Introduction
Breast cancer stands as the most prevalent malignant tumor in women worldwide. According to the latest statistics, it is projected that in 2023 there will be 297,790 new cases accounting for 31% of female cancers and 43,170 deaths accounting for 15% of female deaths [1, 2]. All these data underscore an urgent need to establish new and effective strategies for the diagnosis and treatment of breast cancer. Due to the significant heterogeneity of breast cancer, there is a growing interest in the clinical realm towards personalized and precise treatment approaches [3, 4]. The pursuit of identifying novel prognostic biomarkers and promising therapeutic targets is recognized as a crucial avenue to address this imperative.
The tumor microenvironment (TME) is an intricate ecosystem composed of malignant cells, a variety of infiltrating immune cells (lymphocytes and myeloid cells), and stromal cells intertwined with noncellular components [5]. Cancer-associated fibroblasts (CAFs) play a pivotal role in the progression of tumors [6]. As a major component of the stroma, CAFs enhance cancer cell proliferation, immune rejection, and treatment resistance by secreting growth factors and inflammatory ligands [7]. For example, CAFs could secrete vascular endothelial growth factor (VEGF) to regulate the tumor vascular network [8], and interleukin 6 (IL-6) to foster the differentiation of myeloid-derived suppressor cells (MDSCs) and suppress cytotoxic T cells [9]. Furthermore, the secretion of CXCL12 by CAFs upregulates the anti-apoptotic proteins Bcl-2 and Survivin, potentially contributing to therapy resistance in breast cancer [10]. CAFs can also shape the extra cellular matrix by secreting collagen, fibrinolytic proteins, hyaluronic acid, and laminin, forming a barrier to drugs or therapeutic immune cell penetration. This impediment prevents deep penetration of drugs and immune cells into tumor tissues, thereby diminishing the efficacy of tumor therapy. The modulation of CAFs or overcoming their barrier effect represents a novel approach in tumor therapy [11, 12]. Nevertheless, there is still a deficiency of breast cancer prognostic models based on CAF-specific gene markers to date.
Phenomenal advances in single-cell RNA sequencing (scRNA-seq) technologies have allowed us to deeply analyze the complex cellular composition inside tumors. Here, we conducted a reanalysis a publicly available breast cancer single-cell RNA-seq cohort containing 26 samples [13]. The specific gene markers of CAFs in breast cancer were interrogated by Wilcoxon rank-sum test algorithm at single-cell resolution. Additionally, combining multiple deconvolution algorithms and Weighted Gene Co-expression Network Analysis (WGCNA), we also identified a group of CAFs marker genes. The overlapped compartment of above markers was selected as the final CAF-associated marker genes. Next, we performed univariate Cox regression and least absolute shrinkage and selection operator (LASSO) regression analyses in The Cancer Genome Atlas (TCGA) breast cancer cohort to build a CAF-related prognostic model. Based on the median risk score, the breast cancer samples were classified into high and low-risk groups. Reasonably, patients in the high-risk group showed markedly inferior outcomes. In addition, the potential predictive power of the prognostic model was validated in several separate validation sets. More importantly, pathway activity, mutational profile, and immune status exhibited dramatic differences between high- and low-risk groups. High-risk patients have disturbed cell cycle progression and demonstrate epithelial mesenchymal transition. In addition, high-risk patients were characterized by more M2 macrophage infiltration and less CD8+ T cell. Finally, we focused on the response to chemotherapeutic agents of patients in different risk groups and interrogated the sensitivities of several anti-cancer drugs among diverse risk group patients. To summarize, we combined scRNA-seq and bulk RNA-seq to construct a robust CAF-related prognostic model, which can be used to guide the prognosis and treatment of breast cancer patients.
Materials and Methods
Single-cell transcriptome analysis
In our study, we initially obtained scRNA-seq data from 26 breast cancer samples from the GEO database (GSE176078) [13]. We performed unsupervised clustering of the individual cells using the read count matrix as input, employing the Seurat package (v4.1.1) in R (v4.1.3) [14]. Stringent quality control criteria were implemented, primarily focusing on the number of detected genes and the proportion of mitochondrial gene counts per cell.
Initially, we filtered out cells with fewer than 200 detected genes and cells with more than 15% mitochondrial gene counts. We used the Harmony algorithm to integrate the multi-sample data and correct batch effects [15]. Subsequently, dimension reduction clustering and differential expression analysis were performed following the Seurat-guided tutorial. Principal component analysis (PCA) and uniform manifold approximation and projection (UMAP) dimension reduction were carried out using the top 20 principal components. Cell cluster annotations were based on canonical gene markers.
Weighted gene co-expression network analysis
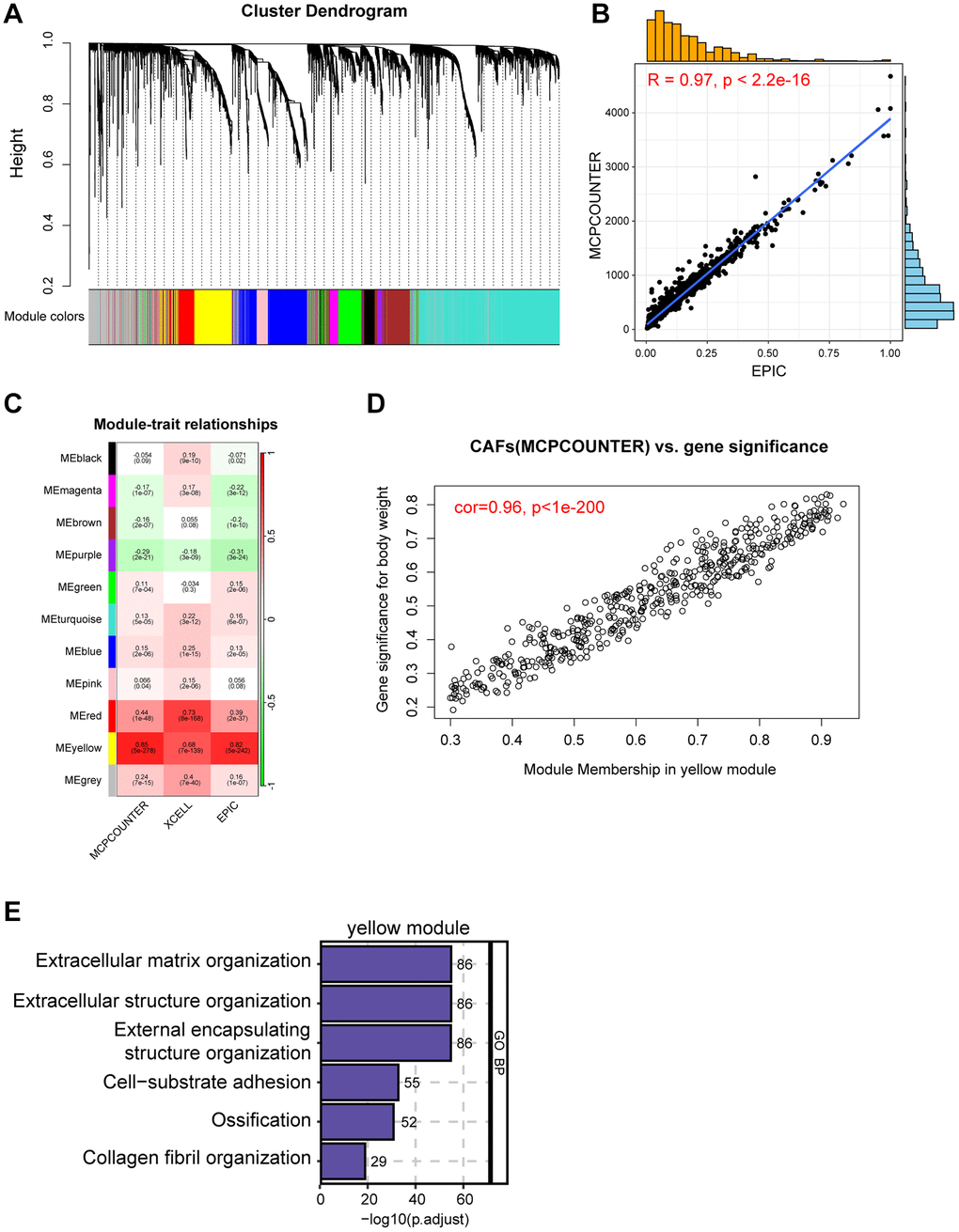
We used the WGCNA package to obtain genes most related to CAFs content [16]. Samples were clustered to ascertain the overall relevance of all samples in the dataset, and outliers were excluded. The soft thresholding power β was chosen based on the lowest power for which the scale-free topology fit index reached a high value. The minimum gene number/module was set to 50 and, finally, 11 modules were generated. Next, we undertook correlation analyses between modules and traits to find the most relevant modules for CAFs content.
Collection of public datasets
RNA-sequencing expression matrix and clinical information of breast cancer samples and para cancerous tissues from The Cancer Genome Atlas (TCGA) database were downloaded from UCSC Xena (https://xena.ucsc.edu/). Three additional independent datasets (GSE20685, GSE37751 and GSE58812) were obtained from the GEO database (https://www.ncbi.nlm.nih.gov/geo/) [17–19]. We downloaded somatic mutation data from Genomic Data Commons (GDC, https://portal.gdc.cancer.gov/). Somatic mutation data sorted in the form of Mutation Annotation Format (MAF) were analyzed and then used to calculate Tumor mutation burden (TMB) using the R package maftools [20].
Biological functional analysis between high/low-risk group patients
The DESeq2 R package was used to perform differentially expressed genes (DEGs) analysis. DEGs were determined with a cutoff of an adjust p-value of less than 0.05 and |Log2 fold change| greater than 1 [22]. The clusterProfiler R package was used to perform gene set enrichment analysis (GSEA) [23]. With the use of Fisher’s exact test, those with false discovery rate FDR-corrected p-values of less than 0.05 were regarded as marked indicators. Single sample gene set enrichment analysis was performed via the R package GSVA [24]. Gene signatures of recurrent cancer cell states were collected from the previous study. The ITH score was calculated using the DEPTH R package [25].
Tumor immune microenvironment in breast cancer patients
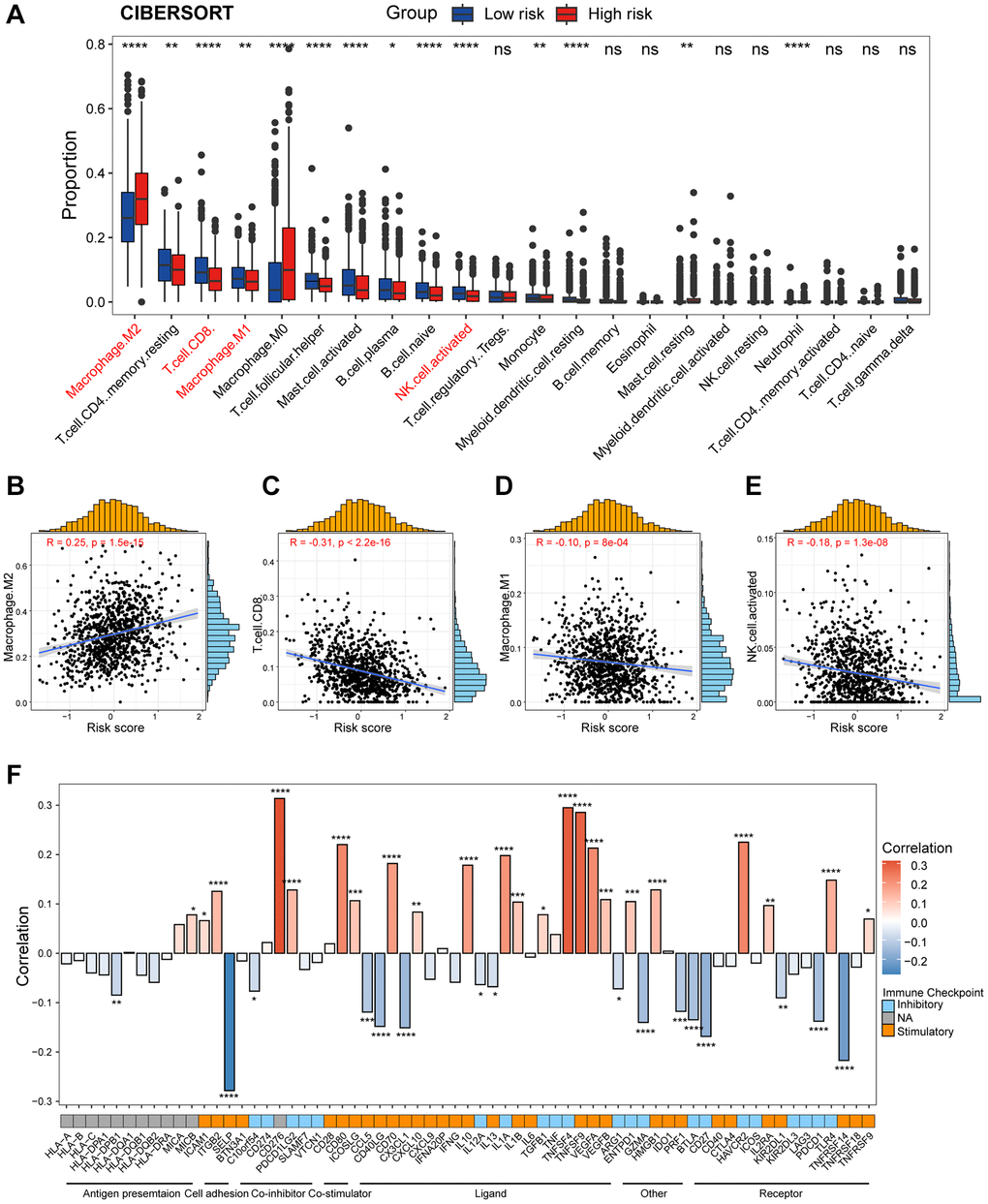
To study the infiltration of immune cells, we used TIMER2.0, an efficient algorithm for predicting immune cell infiltration of bulk tumor gene expression data (http://timer.cistrome.org/). In addition, we collected series of tumor immunomodulators from the literatures and calculated the correlation of risk score with them.
Predicting drug responses and immunotherapy sensitivity
We used the R package oncoPredict to assess the predictive ability of risk score chemotherapeutic agents by calculating patients IC50 for various common chemotherapeutic agents. The Wilcoxon rank test was then used to compare the difference in IC50 between the high/low-risk groups.
Univariate and multivariable Cox regression
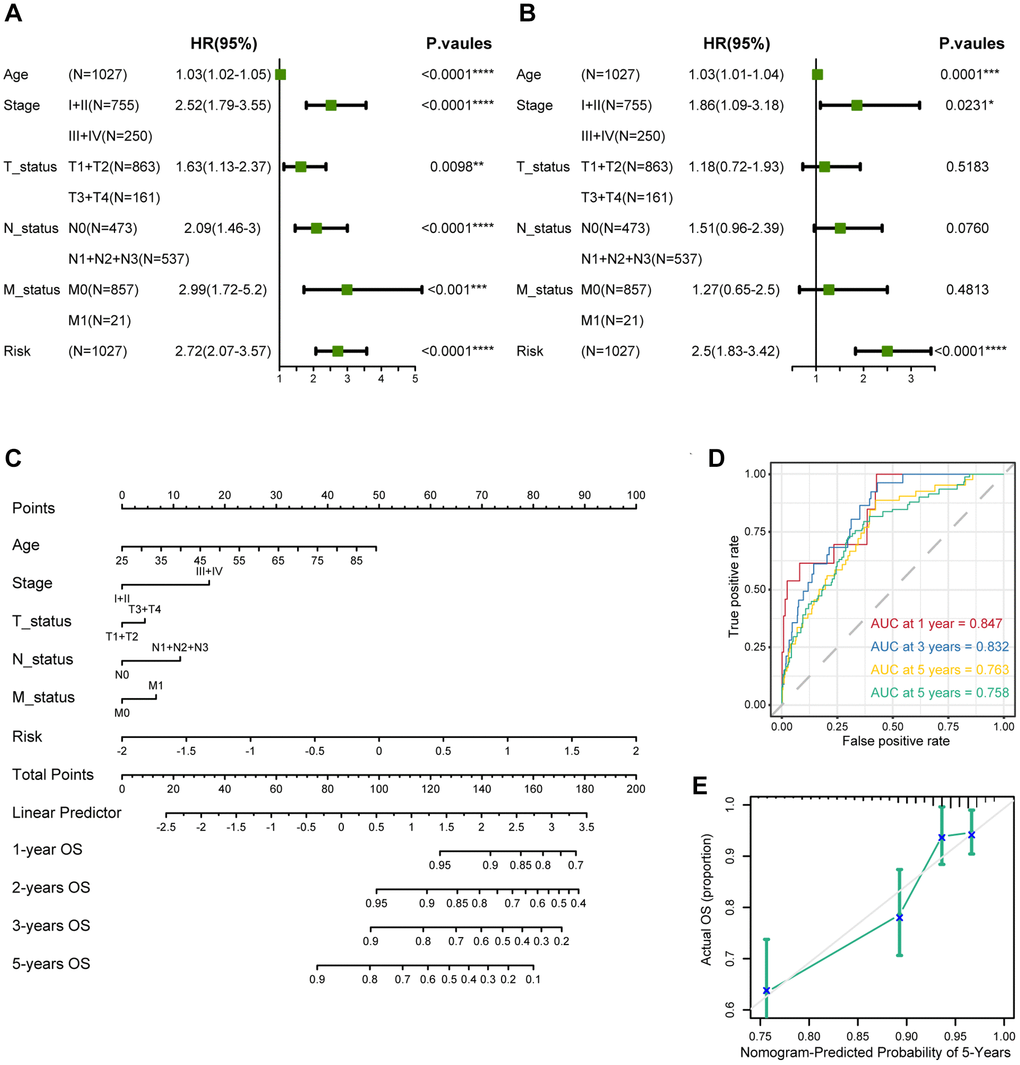
We performed univariate Cox regression on breast cancer patients with gene expression and overall survival. Multivariate Cox regression was used to evaluate independent risk factors in the same cohort. Genes and factors with a false discovery rate (FDR) <0.05 were considered statistically associated with patient survival. The results of univariate and multivariate Cox regression were acquired and visualized by using the R package forestplot.
Establishment of the nomogram
This study used the Cox regression model along with the R package rms to build an OS prediction nomogram that set 1-, 2-, 3-, and 5-year OS as the endpoints.
Statistical analysis
All statistical analyses were performed using R version 4.1.3 (https://www.r-project.org/) and its adequate packages. Statistical significance was set at p ≤ 0.05.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Results
Interrogating the cellular constitution of breast cancer at single-cell resolution
In order to meticulously investigate the cellular constitution of breast cancer at single-cell resolution and identify cell markers of CAFs, we re-analyzed the scRNA-seq data of tumors from 26 breast cancer samples. Firstly, we integrated these data and corrected the potential batch effects through Harmony algorithm. Following rigorous quality control and data filtering, data of 29733 genes within 85408 cells was obtained. Three major compartments in the TME of breast cancer, including the immune subset, the stromal subset, and the epithelial subset were identified (Figure 1A). UMAP visualization showed that scRNA-seq data from different samples were integrated and mixed uniformly (Figure 1B). Using canonical lineage markers, we annotated each cell subpopulation among three main cellular subsets as epithelial cells, T cells, B cells, plasma cells, myeloid cells, endothelial cells, pericytes, cycling cells, and CAFs (Figure 1C, Supplementary Table 1). For instance, the immune subset consisted of T cells which were identified by expressions of CD2, CD3D and CD3E, B cells with high MS4A1, CD79A, and CD79B expressions, plasma cells with high IGHG1, IGKC, and JCHAIN expressions, and myeloid cells which were identified by significant expressions of LYZ, C1QA and C1QB (Figure 1C). Additionally, we found several stromal subpopulations including endothelial cells, pericytes, and CAFs. We annotated the endothelial cells due to the unique expression of ACKR1, PLVAP and PECAM1, as well as pericytes with expressions of RGS5, ACTA2, TAGLN (Figure 1C, 1D). Moreover, with a special focus on the CAFs, we observed CAFs showed significant expressions of DCN, COL1A1 and LUM, which were believed to be uniquely expressed proteins on the surface of CAFs and played a role in the development and function of CAFs. As expected, the Gene Ontology (GO) enrichment analysis exhibited that CAFs gene markers were enriched in pathways including extracellular matrix organization, extracellular structure organization and collagen fibril organization (Figure 1E). Collectively, we interrogated the cellular constitution of breast cancer in detail at single-cell resolution and identified cellular marker genes of each subpopulation, especially for CAFs.
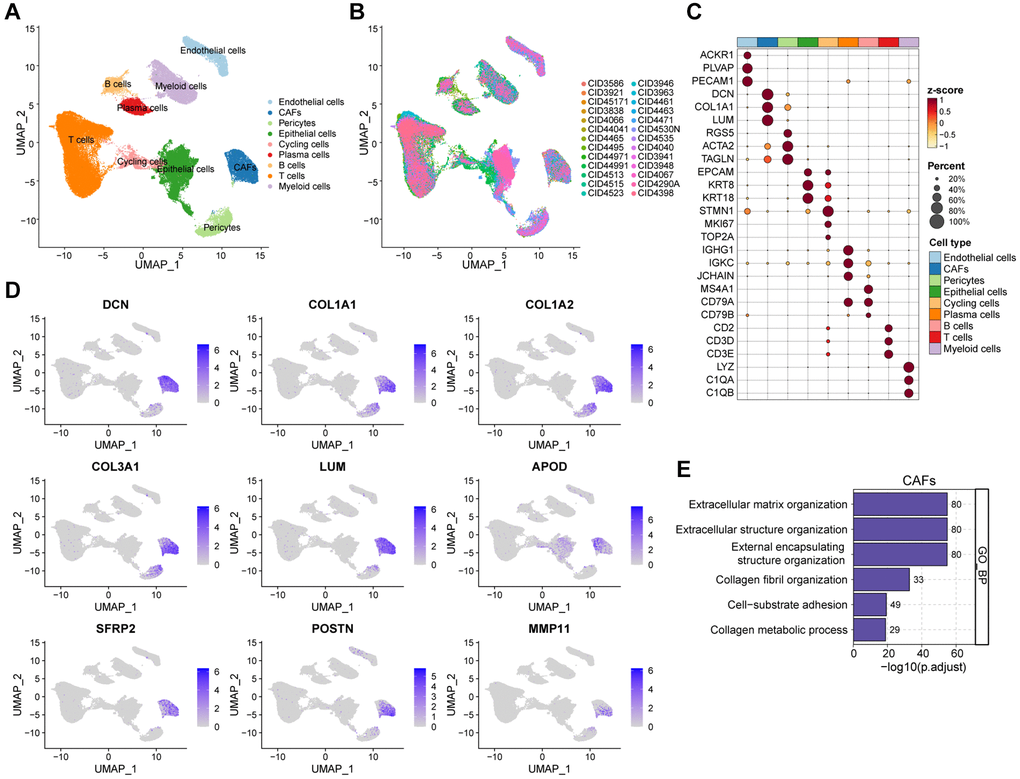
Figure 1. Interrogating the cellular constitution of breast cancer at single-cell resolution. (A, B) UMAP plot showing the major cell subpopulations in breast cancer. (C) Bubble heatmap showing expression levels of selected signature genes in breast cancer. Dot size indicates fraction of expressing cells, colored based on normalized expression levels. (D) Feature plots to further identify various CAFs, based on the expression levels of marker genes. (E) GO enrichment of CAFs signature genes.
Analyses of clinicopathological characteristics based on the CAF-associated prognostic signature
Besides the distinct survival outcomes observed between high/low-risk populations, notable variances were also observed in their clinicopathologic features. An incremental rise in the risk score was noted with advancing clinical stages, implying a potential correlation between the risk score and the progression of tumors (Figure 4A). Subsequently, we investigated the prognostic role of risk score in patients within tumor stage I-IV. Patients with lower risk score exhibited significantly better prognoses in stages II and III, while outcomes in stages I and IV did not show a similar improvement (Supplementary Figure 3A–3D). This phenomenon might be attributed to the limited number of patients with stage I and IV breast cancer and the growing emphasis on early breast cancer prevention. Among the different PAM50 molecular subtypes, the risk score exhibited a notable elevation in the LumB subtype and minimal in the normal-like subtype, which accounts for only a small fraction of breast cancers (Figure 4B). Similarly, we investigated the prognostic implications of risk score in patients within different PAM50 subtypes. Analysis revealed that patients with elevated risk score experienced poorer prognose in the Lum A/B and Basal-like subtypes, but not significantly in the Her2 and normal-like subtypes (Supplementary Figure 3E–3I). Furthermore, elderly patients had higher risk score, which may indicate that it played a role in the aging process (Figure 4C). Intra-tumor heterogeneity (ITH) is one of the crucial ingredients contributing to the failure of cancer therapies and patient mortality, higher ITH levels are associated with the development of therapeutic resistance [26]. Conspicuously, patients in high-risk group displayed elevated levels of ITH (Figure 4D). Risk score was positively correlated with ITH score, implying that high-risk patients were more resistant to single treatment (Figure 4E). Summarily, above results showed us high/low-risk breast cancer patients presented with unique clinicopathological profiles.
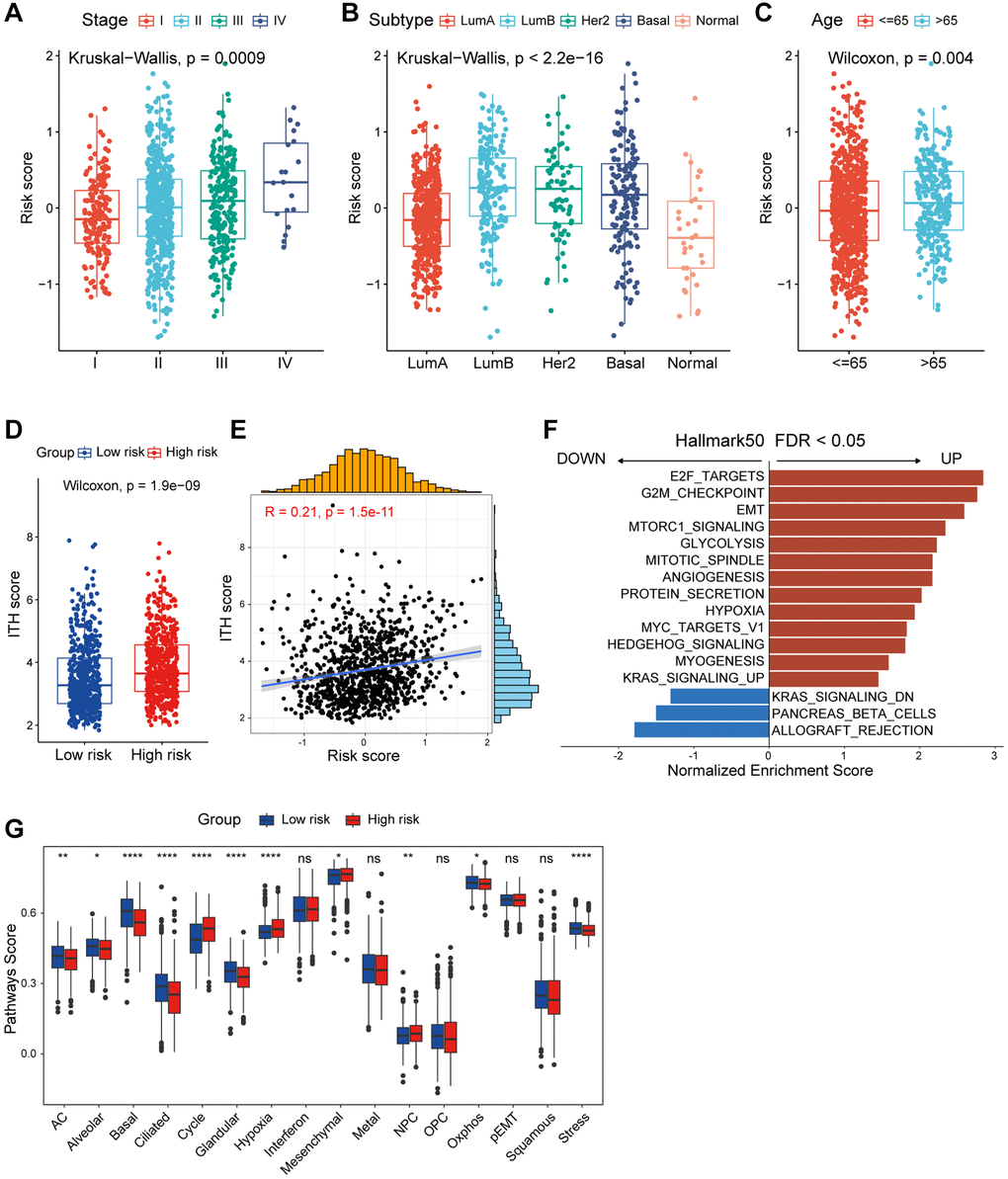
Figure 4. Functional and genomic features of CAF-related risk score-based classification. (A) Levels of risk score in different tumor stages of breast cancer. (B) Levels of risk score in different molecular subtypes of breast cancer. (C) Levels of risk score in different age groups of breast cancer. (D) Boxplot showing the levels of ITH in high/low-risk groups. Paired two-sided Wilcoxon test. (E) Scatter plot showing the correlation between the risk score and ITH score. (F) Bar plot showing different pathways enriched in high/low-risk groups of breast cancer calculated by GSEA. (G) Boxplots showing the signature score of 16 cancer cell states in high/low-risk groups of breast cancer scored by GSVA. Paired two-sided Wilcoxon test. The asterisks represent the statistical P-value (*p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001; nsp > 0.05).
Discussion
Breast cancer, the gravest neoplastic affliction affecting women worldwide, is a multifaceted and exceedingly heterogeneous ailment [32]. Tumor heterogeneity refers to the variance in patients’ response to identical treatments [33]. The tumor microenvironment (TME) encompasses non-malignant cells and constituents within the tumor, as well as the molecules they generate and emit. It is widely recognized that the direct and indirect interactions between tumor cells and the TME are pivotal in the genesis, progression, metastasis, and therapy of the malignancy. CAFs occupy a central role in the TME, exercising indispensable roles in extracellular matrix remodeling, maintenance of stem cell characteristics, angiogenesis, regulation of tumor metabolism, immune response modulation, and facilitation of cancer cell proliferation, migration, invasion, and resistance to therapeutic interventions through manifold mechanisms. Literature reports outline that CAFs are capable of establishing interactive associations with other cells in TME, thereby establishing an immunosuppressive loop that further enhances immune inhibition in the TME. For instance, in triple-negative breast cancer (TNBC), CAFs lend support to immunosuppression by recruiting monocytes to the tumor via the CXCL12-CXCR4 axis, thereby inducing their transformation into tumor-promoting lipid-associated macrophages [34]. Furthermore, CAFs have been scientifically demonstrated to impede CD8+ T cell infiltration and bestow resistance against immune checkpoint blockades (ICBs) therapies [35]. The firmly established capability of CAFs to promote tumorigenesis designates them as promising targets for immunotherapeutic interventions [36]. Nonetheless, precise biomarkers specifically linked with CAFs in breast cancer remain ambiguous, presenting significant challenges in clinical management. We are steadfast in our resolve to explore novel markers associated with CAFs in breast cancer and contribute towards improving clinical treatment strategies.
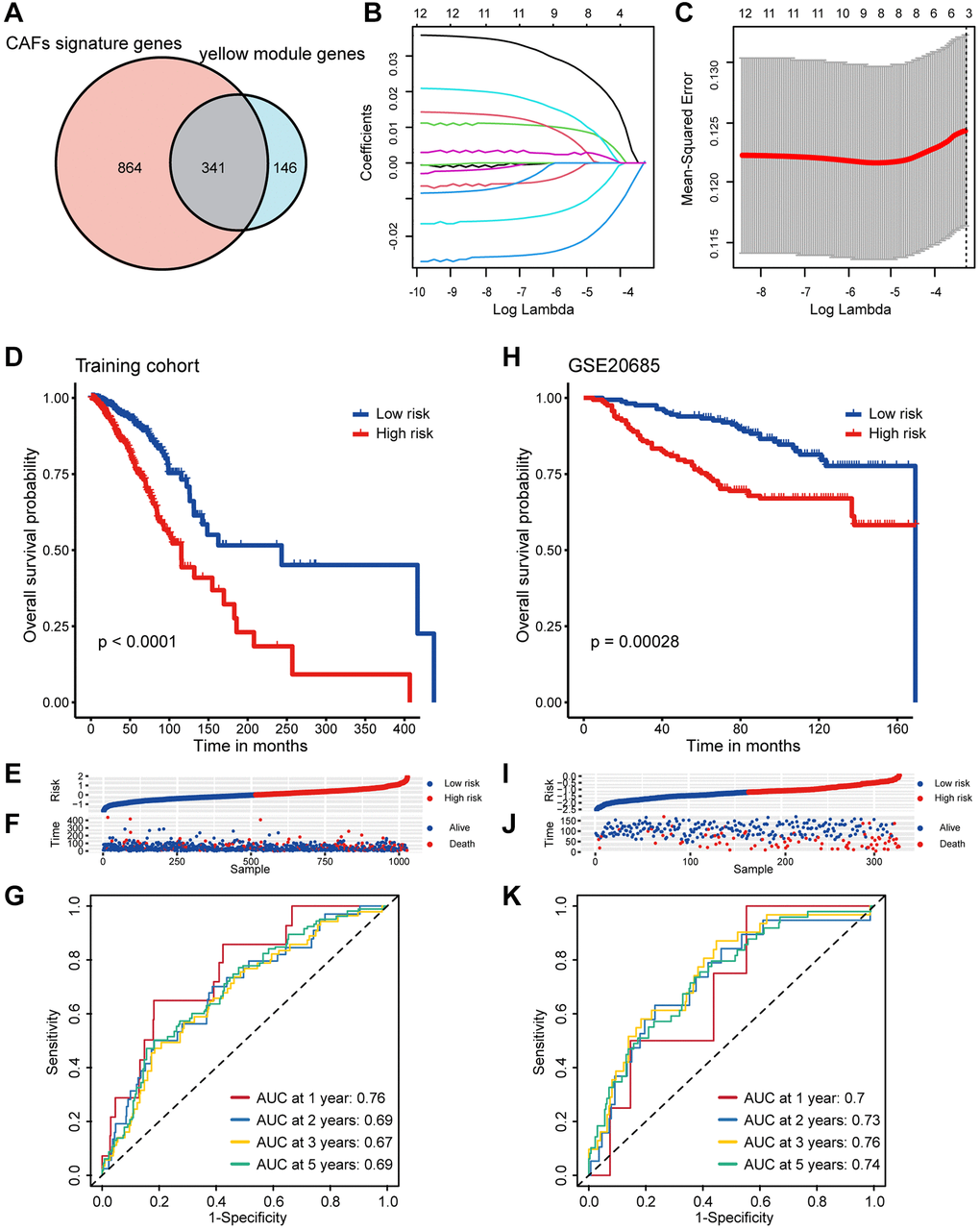
Bulk RNA-seq examines variations between tissue samples at the genomic, transcriptomic, and epigenomic levels. In contrast, single-cell RNA-seq is a sequencing analysis technique performed at the individual cell level, elucidating differences within tissue samples [37]. The combination of these two methodologies offers a comprehensive, multi-omics investigation into the heterogeneity of tumor tissue. The integration of scRNA-seq and bulk RNA-seq carries significant potential in revealing profound biological insights and represents the prevailing trend and future trajectory of histological analysis. In this study, we re-analyzed single cell sequencing data with 26 samples breast cancer and distinctly identified fibroblast subpopulations, defining 1,205 CAFs markers. To explore biological markers of CAFs in bulk RNA-seq, we performed WGCNA. To ensure the robustness of the marker genes, we employed three bioinformatics methodologies to quantify the infiltration of CAFs in breast cancer tissue. Module correlation analysis revealed a robust association between the yellow module and all three methods, thereby establishing the yellow module as a marker for CAFs in bulk RNA-seq. The marker genes derived from scRNA-seq were intersected with the CAFs module genes in bulk RNA-seq, resulting in the identification of 341 genes pertaining to CAFs. Subsequently, univariate Cox and LASSO regression analyses were carried out to screen for eight candidate genes with prognostic relevance, comprising five risk genes (CERCAM, EMP1, SDC1, PRKG1, XG) and three protective genes (TNN, WLS, PDLIM4). While some of these eight candidate genes have already been investigated for their noteworthy roles in tumors, including breast cancer, others await more comprehensive examination. For instance, EMP1 has been recognized as a biomarker for gefitinib resistance in lung cancer and contributes to prednisolone resistance in patients with acute lymphoblastic leukemia [38]. SDC1 functions as a cell surface proteoglycan to attach the cytoskeleton to the mesenchymal matrix and regulates exosome biogenesis. Furthermore, SDC1 can promote the progression of triple-negative breast cancer by activating the c-src/FAK signaling pathway [39]. We consider SDC1 to be a focal point for our future research endeavors. Grounded on these eight prognostically relevant genes, we developed a CAF-related prognostic model and calculated a CAF-related risk score for each breast cancer patient. Based on the median risk score, patients were categorized into high/low-risk groups, with individuals in the high-risk category exhibiting a notably poorer prognosis. Furthermore, to validate the stability of this prognostic model, we subjected it to verification using three independent validation sets. Intriguingly, the risk score exhibited a significant correlation with clinical stage, PAM50 staging, and age of the patients. Moreover, patients with higher risk score also displayed increased ITH, cancer gene mutations, and activation of pro-cancer molecular pathways. Importantly, patients with lower risk score demonstrated significantly higher infiltration of immune cells, including CD8+ T cells, M1 macrophages, and NK cells, whereas high-risk patients showed elevated M2 macrophage infiltrates. Additionally, we delved into the TIME at the single-cell level and noted increased infiltration of myeloid cells, specifically macrophages and monocytes, in high-risk patients, alongside decreased infiltration of DCs. Consequently, the elimination of M2 macrophages or their progenitors may serve as a promising therapeutic strategy for patients in high-risk populations. We further screened breast cancer patients in the high/low-risk groups for a variety of drugs and identified Palbociclib, Oxaliplatin, and Camptothecin as especially suitable options for individuals in the low-risk category. Most notably, we developed an effective prognostic marker based on CAF-related genes and presented a novel and accurate classification system, alongside therapeutic strategies, for breast cancer patients.
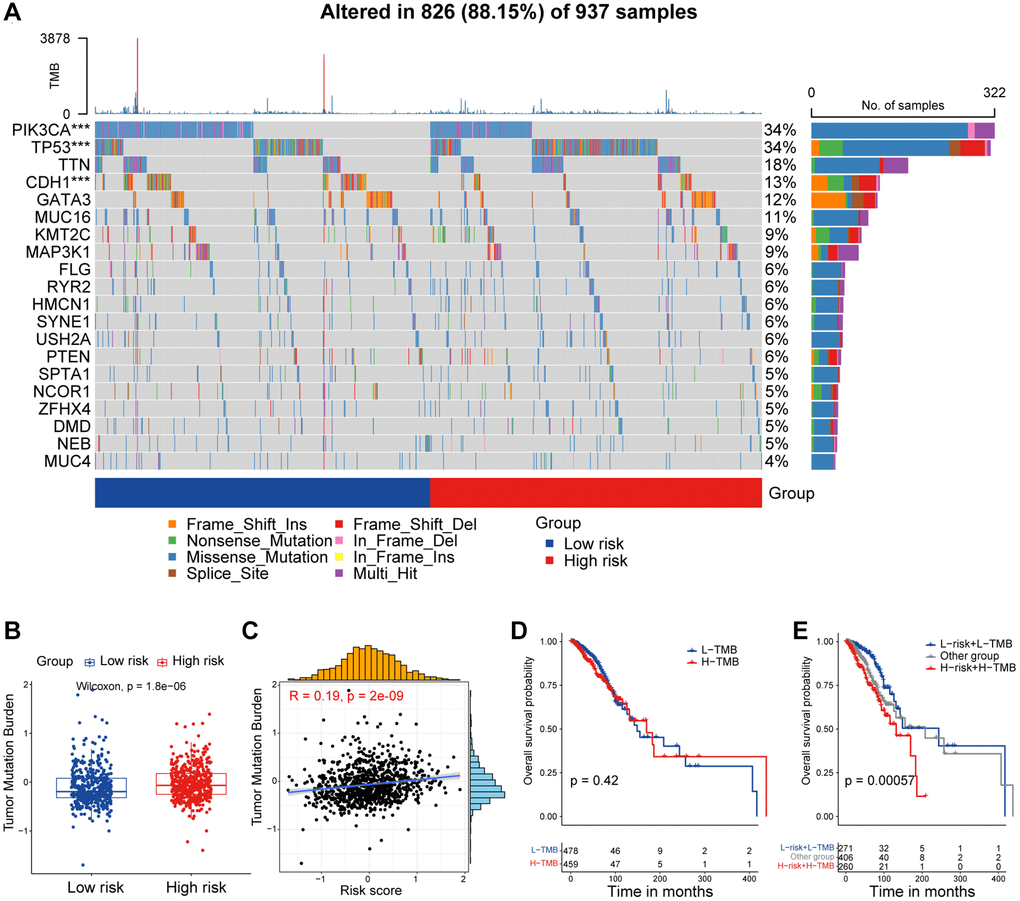
Currently, the management of breast cancer predominantly still rely on clinical staging and pathologic staging [40]. In this study, we observed a progressive increase in the risk score corresponding to the clinical stage, with patients in stages III and IV displaying significantly higher risk score compared to patients in stages I and II. Furthermore, disparities in risk score across patients classified under different PAM50 subtypes, with higher score observed in LumB and Her2 patients, and the lowest score in normal-like patients. Interestingly, we found that risk score tended to be higher in breast cancer patients aged 65 and above, indicating a potential association with the aging process. Given the inherent heterogeneity of tumors, especially in breast cancer, it becomes crucial to investigate the underlying mechanisms and transcriptional differences that contribute to divergent survival outcomes between high and low-risk patient groups. ITH represents a source of genetic variation that can promote cancer progression and the development of drug resistance, consequently culminating in treatment failure. Higher levels of ITH indicate an increased propensity of acquiring drug resistance. Our findings revealed a markedly higher ITH score among high-risk patients, with a positive correlation between risk score and ITH score. The increased heterogeneity and resistance to treatment may contribute to the unfavorable prognosis observed in high-risk patients. Through GSEA enrichment analysis, we identified enrichment of E2F targets, G2M checkpoint, and MITOTIC spindle pathways in the high-risk group, pathways typically associated with cell cycle regulation [41]. This implies a disruption in the regulation of cell cycle progression within high-risk group. Additionally, we observed elevated score for EMT, glycolysis, angiogenesis, and hypoxia in the high-risk group, which aligns with well-established knowledge that CAFs can secrete VEGF to regulate tumor vascular networks, induce EMT in epithelial cells, and contribute to metabolic reprogramming in tumor cells [42]. Our findings further corroborate these associations. The observed heterogeneity in cancer cells can largely be attributed to the redeployment of modules typically expressed in different cellular and developmental contexts. In our investigation, we scrutinized unique attributes of two risk categories by elucidating recently defined cancer cell states that represent fundamental units of tumor transcriptional variability. Notably, the high-risk group exhibited higher characterization score for cycle and hypoxia, while displaying lower score for astrocyte (AC)-like, alveolar, basic, ciliated, and glandular states. TMB not only reflects the extent of genetic alterations within tumor but also provides insight into its immunogenicity. TMB has been identified as a predictive marker for ICB response in melanoma, reflecting its association with immunogenicity in this context [43]. Our study demonstrated elevated TMB in the high-risk group, positively correlating with the risk score. Furthermore, the combined assessment of TMB and CAF-related risk score exhibited strong prognostic performance in predicting patient survival.
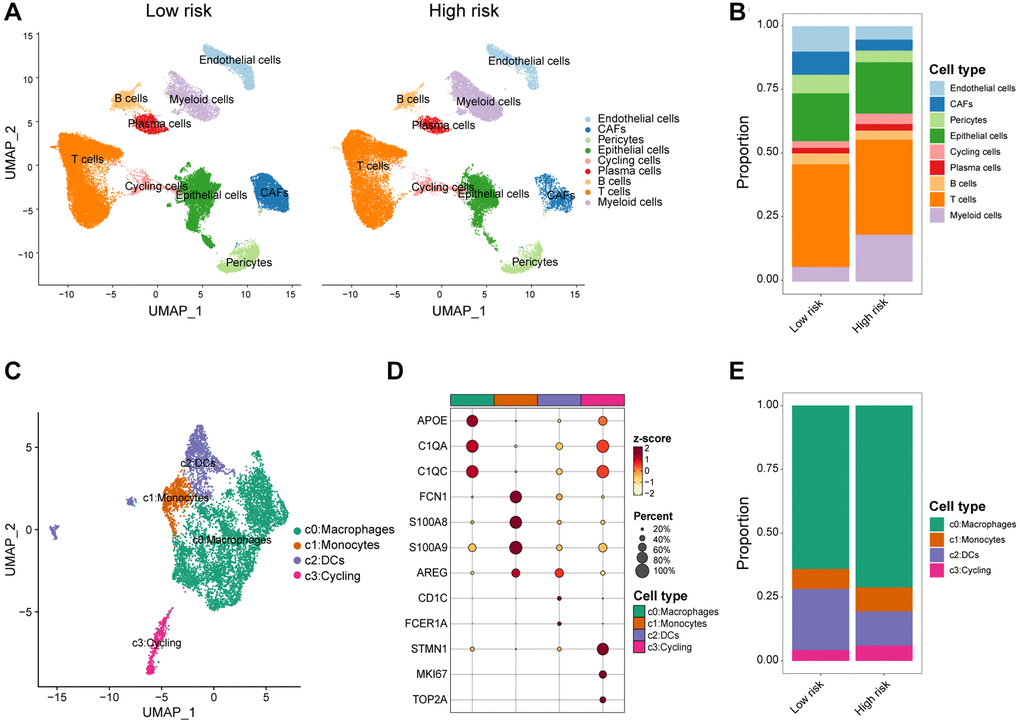
While previous research has emphasized the role of transcriptional and epigenetic variation within tumor cells in tumor development, recent attention has transitioned towards the TIME, which encompasses tumor cells, stromal cells, and infiltrating immune cells. Distinct immune microenvironments can influence the response of tumor cells to both the host’s immune system and external treatments, playing a crucial role in tumor immune evasion and drug resistance [44]. Macrophages typically function in phagocytosis and removal of cellular debris, whereas under specific circumstances they polarize into classically activated M1 macrophages and alternatively activated M2 macrophages [45, 46]. Polarization of M1-polarized macrophages can be stimulated by Th1 cytokines such as interferon-gamma (IFN-γ), tumor necrosis factor-α (TNF-α), and bacterial lipopolysaccharide (LPS), whereas M2-polarized macrophages can be triggered by interleukin 4 (IL-4), interleukin 13 (IL-13), and transforming growth factor-β (TGF-β) [47, 48]. Classically activated M1 macrophages have potent anti-infective, anti-tumor, and removal of apoptotic cells and necrotic tissues, whereas selectively activated M2 macrophages are mainly involved in their healing, angiogenesis, and immunosuppression [49]. Within the TIME, macrophages can be induced by CAFs to differentiate predominantly into M2 macrophages, which are known to promote tumor growth and metastasis. Furthermore, CAFs possess the ability to attract infiltration and aggregation of M2 macrophages through the secretion of various growth factors and cytokines. Our findings revealed significantly elevated levels of M2 macrophage infiltration, accompanied by decreased levels of M1 macrophages, in patients classified as high-risk. Furthermore, both CD8+ T cells and activated NK cells, which are critical subsets of the immune system with anti-tumor effects, displayed reduced infiltration levels in the high-risk group. Additionally, our analysis at the single-cell level demonstrated that the high-risk population exhibited higher levels of macrophage infiltration and lower levels of DCs. Consequently, the elimination of M2 macrophages or their progenitors presents itself as a potential therapeutic strategy for individuals within high-risk populations.
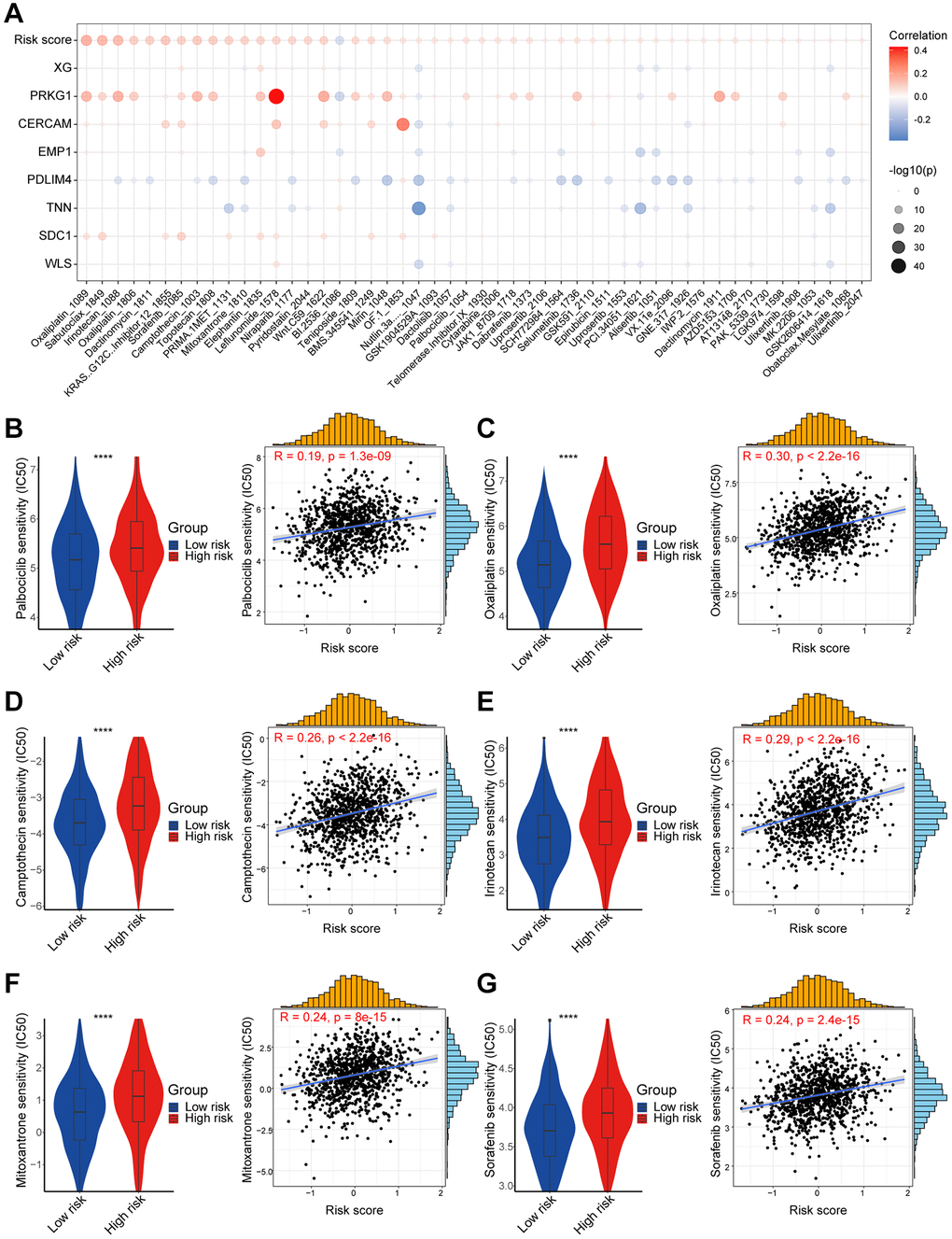
After exploring the differences between the transcriptional, genetic, and immune microenvironments between high/low-risk risk groups, we worked to screen several anticancer medications for different breast cancer patients. The IC50 of drugs such as Palbociclib, Oxaliplatin, and Camptothecin was significantly lower in the low-risk group. Refreshingly, in the I-SPY2 data, patients with non-responding Her2-positive metastatic breast cancer treated with patuximab had lower risk score. It has been shown that PDPN-positive CAF can promote resistance to trastuzumab in Her2-positive breast cancer by secreting the immunosuppressive factors indoleamine 2,3-dioxygenase 1 (IDO1) as well as tryptophan 2,3-dioxygenase 2 (TDO2) [50]. This could potentially provide a framework for guiding the treatment of breast cancer patients. The nomogram enables integration of our model with standard clinical variables of the patient, including age, pathological staging, etc., thereby offering clinical guidance for personalized treatment. However, the exact function of the above treatments remains to be further confirmed in future prospective studies.
While our prognostic model for breast cancer, based on cancer-associated fibroblasts, demonstrated commendable performance in both the training and validation cohorts, it is imperative to acknowledge certain constraints. Primarily, it is essential to substantiate the expression and prognostic significance of marker genes associated with candidate CAFs at the protein level through further examination. Moreover, despite screening these prognostic genes based on CAFs markers, it should be noted that they are not exclusively expressed by CAFs. Lastly, we must be mindful of the potential presence of inherent bias resulting from the retrospective recruitment of breast cancer patients.
Author Contributions
FY and ZG contributed to the conception and design of this study. ZF and YH collected and analyzed the data. ZF drafted the original manuscript. ZG and ZF polished and revised the manuscript. This manuscript has been read and approved by all authors.
Conflicts of Interest
The authors declare that they have no competing interests.
Funding
No funding was used for this paper.
References
- 1. Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023; 73:17–48. https://doi.org/10.3322/caac.21763 [PubMed]
- 2. Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022; 72:7–33. https://doi.org/10.3322/caac.21708 [PubMed]
- 3. Yeo SK, Guan JL. Breast Cancer: Multiple Subtypes within a Tumor? Trends Cancer. 2017; 3:753–60. https://doi.org/10.1016/j.trecan.2017.09.001 [PubMed]
- 4. Marusyk A, Almendro V, Polyak K. Intra-tumour heterogeneity: a looking glass for cancer? Nat Rev Cancer. 2012; 12:323–34. https://doi.org/10.1038/nrc3261 [PubMed]
- 5. Danenberg E, Bardwell H, Zanotelli VRT, Provenzano E, Chin SF, Rueda OM, Green A, Rakha E, Aparicio S, Ellis IO, Bodenmiller B, Caldas C, Ali HR. Breast tumor microenvironment structures are associated with genomic features and clinical outcome. Nat Genet. 2022; 54:660–9. https://doi.org/10.1038/s41588-022-01041-y [PubMed]
- 6. Hu D, Li Z, Zheng B, Lin X, Pan Y, Gong P, Zhuo W, Hu Y, Chen C, Chen L, Zhou J, Wang L. Cancer-associated fibroblasts in breast cancer: Challenges and opportunities. Cancer Commun (Lond). 2022; 42:401–34. https://doi.org/10.1002/cac2.12291 [PubMed]
- 7. Biffi G, Tuveson DA. Diversity and Biology of Cancer-Associated Fibroblasts. Physiol Rev. 2021; 101:147–76. https://doi.org/10.1152/physrev.00048.2019 [PubMed]
- 8. De Palma M, Biziato D, Petrova TV. Microenvironmental regulation of tumour angiogenesis. Nat Rev Cancer. 2017; 17:457–74. https://doi.org/10.1038/nrc.2017.51 [PubMed]
- 9. Xiang H, Ramil CP, Hai J, Zhang C, Wang H, Watkins AA, Afshar R, Georgiev P, Sze MA, Song XS, Curran PJ, Cheng M, Miller JR, et al. Cancer-Associated Fibroblasts Promote Immunosuppression by Inducing ROS-Generating Monocytic MDSCs in Lung Squamous Cell Carcinoma. Cancer Immunol Res. 2020; 8:436–50. https://doi.org/10.1158/2326-6066.CIR-19-0507 [PubMed]
- 10. Gao ZJ, Fang Z, Yuan JP, Sun SR, Li B. Integrative multi-omics analyses unravel the immunological implication and prognostic significance of CXCL12 in breast cancer. Front Immunol. 2023; 14:1188351. https://doi.org/10.3389/fimmu.2023.1188351 [PubMed]
- 11. Chen X, Song E. Turning foes to friends: targeting cancer-associated fibroblasts. Nat Rev Drug Discov. 2019; 18:99–115. https://doi.org/10.1038/s41573-018-0004-1 [PubMed]
- 12. Jenkins L, Jungwirth U, Avgustinova A, Iravani M, Mills A, Haider S, Harper J, Isacke CM. Cancer-Associated Fibroblasts Suppress CD8+ T-cell Infiltration and Confer Resistance to Immune-Checkpoint Blockade. Cancer Res. 2022; 82:2904–17. https://doi.org/10.1158/0008-5472.CAN-21-4141 [PubMed]
- 13. Wu SZ, Al-Eryani G, Roden DL, Junankar S, Harvey K, Andersson A, Thennavan A, Wang C, Torpy JR, Bartonicek N, Wang T, Larsson L, Kaczorowski D, et al. A single-cell and spatially resolved atlas of human breast cancers. Nat Genet. 2021; 53:1334–47. https://doi.org/10.1038/s41588-021-00911-1 [PubMed]
- 14. Hao Y, Hao S, Andersen-Nissen E, Mauck WM
3rd , Zheng S, Butler A, Lee MJ, Wilk AJ, Darby C, Zager M, Hoffman P, Stoeckius M, Papalexi E, et al. Integrated analysis of multimodal single-cell data. Cell. 2021; 184:3573–87.e29. https://doi.org/10.1016/j.cell.2021.04.048 [PubMed] - 15. Korsunsky I, Millard N, Fan J, Slowikowski K, Zhang F, Wei K, Baglaenko Y, Brenner M, Loh PR, Raychaudhuri S. Fast, sensitive and accurate integration of single-cell data with Harmony. Nat Methods. 2019; 16:1289–96. https://doi.org/10.1038/s41592-019-0619-0 [PubMed]
- 16. Langfelder P, Horvath S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008; 9:559. https://doi.org/10.1186/1471-2105-9-559 [PubMed]
- 17. Kao KJ, Chang KM, Hsu HC, Huang AT. Correlation of microarray-based breast cancer molecular subtypes and clinical outcomes: implications for treatment optimization. BMC Cancer. 2011; 11:143. https://doi.org/10.1186/1471-2407-11-143 [PubMed]
- 18. Terunuma A, Putluri N, Mishra P, Mathé EA, Dorsey TH, Yi M, Wallace TA, Issaq HJ, Zhou M, Killian JK, Stevenson HS, Karoly ED, Chan K, et al. MYC-driven accumulation of 2-hydroxyglutarate is associated with breast cancer prognosis. J Clin Invest. 2014; 124:398–412. https://doi.org/10.1172/JCI71180 [PubMed]
- 19. Jézéquel P, Loussouarn D, Guérin-Charbonnel C, Campion L, Vanier A, Gouraud W, Lasla H, Guette C, Valo I, Verrièle V, Campone M. Gene-expression molecular subtyping of triple-negative breast cancer tumours: importance of immune response. Breast Cancer Res. 2015; 17:43. https://doi.org/10.1186/s13058-015-0550-y [PubMed]
- 20. Mayakonda A, Lin DC, Assenov Y, Plass C, Koeffler HP. Maftools: efficient and comprehensive analysis of somatic variants in cancer. Genome Res. 2018; 28:1747–56. https://doi.org/10.1101/gr.239244.118 [PubMed]
- 21. Beyene KM, El Ghouch A. Time-dependent ROC curve estimation for interval-censored data. Biom J. 2022; 64:1056–74. https://doi.org/10.1002/bimj.202000382 [PubMed]
- 22. Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014; 15:550. https://doi.org/10.1186/s13059-014-0550-8 [PubMed]
- 23. Wu T, Hu E, Xu S, Chen M, Guo P, Dai Z, Feng T, Zhou L, Tang W, Zhan L, Fu X, Liu S, Bo X, Yu G. clusterProfiler 4.0: A universal enrichment tool for interpreting omics data. Innovation (Camb). 2021; 2:100141. https://doi.org/10.1016/j.xinn.2021.100141 [PubMed]
- 24. Hänzelmann S, Castelo R, Guinney J. GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinformatics. 2013; 14:7. https://doi.org/10.1186/1471-2105-14-7 [PubMed]
- 25. Song D, Wang X. DEPTH2: an mRNA-based algorithm to evaluate intratumor heterogeneity without reference to normal controls. J Transl Med. 2022; 20:150. https://doi.org/10.1186/s12967-022-03355-1 [PubMed]
- 26. Dentro SC, Leshchiner I, Haase K, Tarabichi M, Wintersinger J, Deshwar AG, Yu K, Rubanova Y, Macintyre G, Demeulemeester J, Vázquez-García I, Kleinheinz K, Livitz DG, et al, and PCAWG Evolution and Heterogeneity Working Group and the PCAWG Consortium. Characterizing genetic intra-tumor heterogeneity across 2,658 human cancer genomes. Cell. 2021; 184:2239–54.e39. https://doi.org/10.1016/j.cell.2021.03.009 [PubMed]
- 27. Barkley D, Moncada R, Pour M, Liberman DA, Dryg I, Werba G, Wang W, Baron M, Rao A, Xia B, França GS, Weil A, Delair DF, et al. Cancer cell states recur across tumor types and form specific interactions with the tumor microenvironment. Nat Genet. 2022; 54:1192–201. https://doi.org/10.1038/s41588-022-01141-9 [PubMed]
- 28. Alexandrov LB, Nik-Zainal S, Wedge DC, Aparicio SA, Behjati S, Biankin AV, Bignell GR, Bolli N, Borg A, Børresen-Dale AL, Boyault S, Burkhardt B, Butler AP, et al, and Australian Pancreatic Cancer Genome Initiative, and ICGC Breast Cancer Consortium, and ICGC MMML-Seq Consortium, and ICGC PedBrain. Signatures of mutational processes in human cancer. Nature. 2013; 500:415–21. https://doi.org/10.1038/nature12477 [PubMed]
- 29. Barroso-Sousa R, Jain E, Cohen O, Kim D, Buendia-Buendia J, Winer E, Lin N, Tolaney SM, Wagle N. Prevalence and mutational determinants of high tumor mutation burden in breast cancer. Ann Oncol. 2020; 31:387–94. https://doi.org/10.1016/j.annonc.2019.11.010 [PubMed]
- 30. Jardim DL, Goodman A, de Melo Gagliato D, Kurzrock R. The Challenges of Tumor Mutational Burden as an Immunotherapy Biomarker. Cancer Cell. 2021; 39:154–73. https://doi.org/10.1016/j.ccell.2020.10.001 [PubMed]
- 31. Wolf DM, Yau C, Wulfkuhle J, Brown-Swigart L, Gallagher RI, Lee PRE, Zhu Z, Magbanua MJ, Sayaman R, O'Grady N, Basu A, Delson A, Coppé JP, et al, and I-SPY2 Investigators. Redefining breast cancer subtypes to guide treatment prioritization and maximize response: Predictive biomarkers across 10 cancer therapies. Cancer Cell. 2022; 40:609–23.e6. https://doi.org/10.1016/j.ccell.2022.05.005 [PubMed]
- 32. Giaquinto AN, Sung H, Miller KD, Kramer JL, Newman LA, Minihan A, Jemal A, Siegel RL. Breast Cancer Statistics, 2022. CA Cancer J Clin. 2022; 72:524–41. https://doi.org/10.3322/caac.21754 [PubMed]
- 33. Polyak K. Heterogeneity in breast cancer. J Clin Invest. 2011; 121:3786–8. https://doi.org/10.1172/JCI60534 [PubMed]
- 34. Timperi E, Gueguen P, Molgora M, Magagna I, Kieffer Y, Lopez-Lastra S, Sirven P, Baudrin LG, Baulande S, Nicolas A, Champenois G, Meseure D, Vincent-Salomon A, et al. Lipid-Associated Macrophages Are Induced by Cancer-Associated Fibroblasts and Mediate Immune Suppression in Breast Cancer. Cancer Res. 2022; 82:3291–306. https://doi.org/10.1158/0008-5472.CAN-22-1427 [PubMed]
- 35. Wu SZ, Roden DL, Wang C, Holliday H, Harvey K, Cazet AS, Murphy KJ, Pereira B, Al-Eryani G, Bartonicek N, Hou R, Torpy JR, Junankar S, et al. Stromal cell diversity associated with immune evasion in human triple-negative breast cancer. EMBO J. 2020; 39:e104063. https://doi.org/10.15252/embj.2019104063 [PubMed]
- 36. Lappano R, Rigiracciolo DC, Belfiore A, Maggiolini M, De Francesco EM. Cancer associated fibroblasts: role in breast cancer and potential as therapeutic targets. Expert Opin Ther Targets. 2020; 24:559–72. https://doi.org/10.1080/14728222.2020.1751819 [PubMed]
- 37. Liu SQ, Gao ZJ, Wu J, Zheng HM, Li B, Sun S, Meng XY, Wu Q. Single-cell and spatially resolved analysis uncovers cell heterogeneity of breast cancer. J Hematol Oncol. 2022; 15:19. https://doi.org/10.1186/s13045-022-01236-0 [PubMed]
- 38. Wang YW, Cheng HL, Ding YR, Chou LH, Chow NH. EMP1, EMP 2, and EMP3 as novel therapeutic targets in human cancer. Biochim Biophys Acta Rev Cancer. 2017; 1868:199–211. https://doi.org/10.1016/j.bbcan.2017.04.004 [PubMed]
- 39. Luo P, Gong Y, Weng J, Wen F, Luo J, Hu C, Xiao J, Shu J. CircKIF4A combines EIF4A3 to stabilize SDC1 expression to activate c-src/FAK and promotes TNBC progression. Cell Signal. 2023; 108:110690. https://doi.org/10.1016/j.cellsig.2023.110690 [PubMed]
- 40. McDonald ES, Clark AS, Tchou J, Zhang P, Freedman GM. Clinical Diagnosis and Management of Breast Cancer. J Nucl Med. 2016 (Suppl 1); 57:9S–16. https://doi.org/10.2967/jnumed.115.157834 [PubMed]
- 41. Fang Z, Gao ZJ, Yu X, Sun SR, Yao F. Identification of a centrosome-related prognostic signature for breast cancer. Front Oncol. 2023; 13:1138049. https://doi.org/10.3389/fonc.2023.1138049 [PubMed]
- 42. Sahai E, Astsaturov I, Cukierman E, DeNardo DG, Egeblad M, Evans RM, Fearon D, Greten FR, Hingorani SR, Hunter T, Hynes RO, Jain RK, Janowitz T, et al. A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer. 2020; 20:174–86. https://doi.org/10.1038/s41568-019-0238-1 [PubMed]
- 43. Snyder A, Makarov V, Merghoub T, Yuan J, Zaretsky JM, Desrichard A, Walsh LA, Postow MA, Wong P, Ho TS, Hollmann TJ, Bruggeman C, Kannan K, et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N Engl J Med. 2014; 371:2189–99. https://doi.org/10.1056/NEJMoa1406498 [PubMed]
- 44. Fu T, Dai LJ, Wu SY, Xiao Y, Ma D, Jiang YZ, Shao ZM. Spatial architecture of the immune microenvironment orchestrates tumor immunity and therapeutic response. J Hematol Oncol. 2021; 14:98. https://doi.org/10.1186/s13045-021-01103-4 [PubMed]
- 45. Chen S, Saeed AFU, Liu Q, Jiang Q, Xu H, Xiao GG, Rao L, Duo Y. Macrophages in immunoregulation and therapeutics. Signal Transduct Target Ther. 2023; 8:207. https://doi.org/10.1038/s41392-023-01452-1 [PubMed]
- 46. Yunna C, Mengru H, Lei W, Weidong C. Macrophage M1/M2 polarization. Eur J Pharmacol. 2020; 877:173090. https://doi.org/10.1016/j.ejphar.2020.173090 [PubMed]
- 47. Weng YS, Tseng HY, Chen YA, Shen PC, Al Haq AT, Chen LM, Tung YC, Hsu HL. MCT-1/miR-34a/IL-6/IL-6R signaling axis promotes EMT progression, cancer stemness and M2 macrophage polarization in triple-negative breast cancer. Mol Cancer. 2019; 18:42. https://doi.org/10.1186/s12943-019-0988-0 [PubMed]
- 48. Liu Z, Gao Z, Li B, Li J, Ou Y, Yu X, Zhang Z, Liu S, Fu X, Jin H, Wu J, Sun S, Sun S, Wu Q. Lipid-associated macrophages in the tumor-adipose microenvironment facilitate breast cancer progression. Oncoimmunology. 2022; 11:2085432. https://doi.org/10.1080/2162402X.2022.2085432 [PubMed]
- 49. DeNardo DG, Ruffell B. Macrophages as regulators of tumour immunity and immunotherapy. Nat Rev Immunol. 2019; 19:369–82. https://doi.org/10.1038/s41577-019-0127-6 [PubMed]
- 50. Du R, Zhang X, Lu X, Ma X, Guo X, Shi C, Ren X, Ma X, He Y, Gao Y, Liu Y. PDPN positive CAFs contribute to HER2 positive breast cancer resistance to trastuzumab by inhibiting antibody-dependent NK cell-mediated cytotoxicity. Drug Resist Updat. 2023; 68:100947. https://doi.org/10.1016/j.drup.2023.100947 [PubMed]