Introduction
Down Syndrome (DS) represents human most frequent aneuploidy, characterized by an extra complete or segment of chromosome 21 (trisomy 21), eventually triggering the dysregulation of several factors [1]. Besides the well-established neurodevelopmental defects and neuronal dysfunction, it has been recently reported how DS individuals also display a plethora of conditions related to unbalanced reactive oxygen species (ROS) production [2]. Such phenotype is dependent on chromosome 21 triplication, triggering the upregulation of proteins involved in redox homeostasis as superoxide dismutase 1 (SOD1), the transcription factor BTB and CNC homology 1 (BACH1), the Protein C-ets-2 (ETS2), carbonyl reductase (CBR), S100 calcium-binding protein B (S100B) [3]. Furthermore, it has been reported that the major regulator of antioxidant response elements (ARE) nuclear factor (erythroid-derived 2)-like 2 (NRF2) is transcriptionally activated in DS individuals [4]. This effect has been reported to be related to the hyperphosphorylation of mitogen-activated protein kinases (MAPKs) which in turn mediates NRF2 phosphorylation thus, promoting its dissociation from Kelch-like ECH-Associating protein 1 (Keap1) but preventing its nuclear translocation [5]. In addition, the high oxidative environment might be responsible for several of the outcomes characterizing DS, including accelerated aging, eventually triggering several of the mental disorders characterizing DS individuals. To this regard, recent studies performed in Ts2Cje DS mouse model [5–7], showed an increased risk of developing a type of dementia mimicking the clinical and pathological course of Alzheimer’s disease (AD) already at the age of 40s [8]. These results are strictly related to the triplication of genes associated with AD such as amyloid precursor protein (APP), β-secretase 2 (BACE2), and S100 calcium binding protein B (S100B) [9], which along with the oxidative unbalance characterizing DS, promote the accumulation of amyloid beta-peptide (Aβ) [3]. Furthermore, the chronic condition of oxidative stress accounts for several comorbidities, including cardiovascular diseases, and certain types of cancer characterizing DS individuals [10, 11]. In this context, liver may also represent one of the organs involved in the complex metabolic impairment of DS, even though liver function in DS patients is poorly described, and the molecular basis involved in the pathophysiological processes remain elusive. To this regard, a vascular portohepatic anomaly in DS individuals has been characterized to impair the direct communication between the right portal vein and the inferior vena cava [12], potentially linking congenital vascular malformation and hepatic vascular shunt [13]. Furthermore, DS individuals show a significant enhanced production of hepatitis B antigen which might be responsible for the occurrence of the autoimmune hepatitis, a chronic and progressive inflammation of the liver from an unknown cause [14]. In addition to this, the alternated levels of several amino acids, along with several metabolites involved in the methylation cycle have been detected in blood samples derived from DS individuals, pointing out metabolic dysfunctions as one of the drivers of DS liver pathologies [15]. Consistently, DS individuals, since childhood, are typically obese and also presenting dyslipidemia and hyperinsulinemia [16]. Previous studies showed that DS patients exhibit a marked trend in developing overweight and obesity, mostly as a result of a lower resting metabolic rate, a higher consumption of energy-rich foods, and a poor physical activity. As a result, these subjects display a higher risk in developing type 2 diabetes, dyslipidemia, hyperinsulinemia, hypertension, and cardiovascular diseases [16]. However, the recent improvement of surgical and early therapeutic intervention in DS morbidities, increased patients’ life expectancy. On the other hand, there exists an enhanced risk of chronic noncommunicable disease as non-alcoholic fatty liver disease (NAFLD), a pathology characterized by the accumulation of fat in the liver, a condition strictly associated to the enhanced insulin resistance reported in DS individuals [15–17]. Furthermore, DS liver is characterized by significative changes in liver morphology showing sinusoidal dilatation, central-vein sclerosis, and portal fibrosis [18]. Finally, other key factors to take into due account are several single nucleotide polymorphisms (SNPs), such as patatin like phospholipase domain containing 3 (PNPLA3), Transmembrane 6 superfamily member 2 (TM6SF2), and Klotho eventually promoting hepatic fat accumulation [19]. Given the correlation standing between DS and NAFLD occurrence, further research is needed to fully understand the pathophysiological mechanisms underlying such association.
Materials and Methods
Mouse colony
Ts2Cje (Rb(12.Ts171665Dn)2Cje) mice are an established DS murine model displaying a triple copy of a Robertsonian fusion chromosome where the distal end of Chr16 and Chr12 are located. Parental generations were purchased from Jackson Laboratories (Bar Harbour, ME, USA). The mouse colony was raised by a crossbreed of Ts2Cje trisomic females with euploid (B6EiC3SnF1/J) F1 hybrid males (Eu). The parental generations were purchased from Jackson Laboratories (Bar Harbour, ME, USA). These breeding pairs produce litters containing both trisomic (Ts2Cje) and euploid (Eu) offspring. Pups were genotyped to determine trisomy by standard PCR, using Reinoldth’s method [20, 21]. Mice were housed in clear Plexiglas cages (20 × 22 × 20 cm) under standard laboratory conditions with a temperature of 22 ± 2°C and 70% humidity, a 12-h light/dark cycle, and free access to food and water, as previously described [21]. All the experiments were performed in strict compliance with the Italian National Laws (DL 116/92), and the European Communities Council Directives (86/609/EEC). The experimental protocol was approved by the Italian Ministry of Health (#1183/2016-PR). All efforts were made to minimize the number of animals used in the study and their suffering. For this reason, our cohort was composed by 49 mice subdivided as following: 3 months (6 Eu; 6 TS2Cje); 6 months (5 Eu; 6 TS2Cje); 9 months (6 Eu; 6 TS2Cje) and 12 months (6 Eu; 6 TS2Cje). Immediately after isolation, samples were put into liquid nitrogen and then stored at −80°C until utilization.
GSH evaluation
GSH levels were assayed on tissues homogenized performing a spectrophotometric assay based on the reaction of thiol groups with 2,2-dithio-bis-nitrobenzoic acid at a wavelength of 412 nm (εM = 13,600 M−1 cm−1, where εM is a wavelength-dependent molar absorptivity coefficient) [22]. Measurements were performed quantified using Synergy H1 (Biotek, Milan, Italy) in quadruplicate per each sample.
LOOH measurement
Lipid peroxide assay was performed as already reported [22]. Briefly, the reaction is based on the oxidation of Fe2+ to Fe3+ in the presence of xylenol orange at 560 nm. Measurements were performed quantified using Synergy H1 (Biotek, Milan, Italy) in quadruplicate per each sample.
RNA extraction and cDNA preparation
Tissue sections were resuspended in 500 mL of PRImeZOL Reagent (#AN1100, Canvax Biotech, Andalusia, Spain). RNA extraction was then performed as previously described [23] and resuspended in RNase-free water. RNA was quantified using Synergy H1 (Biotek, Milan, Italy). 2 μg of RNA from each sample were retro-transcribed using High-Capacity cDNA Reverse Transcription Kit (#4368814; Applied Biosystems; Waltham, MA, USA) according to manufacturer instructions.
Real time PCR
Gene expression analysis was performed as previously described [24]. As probe, PowerUP SYBR Green Master Mix (#A25742; Applied Biosystem, Waltham, MA, USA) was used. Primers are listed in Table 1.
Table 1. Real time PCR primers’ list.
| Gene | Forward 5′ --> 3′ | Reverse 5′ --> 3′ | Accession number |
| Hmox1 | TGACACCTGAGGTCAAGCAC | CAGCTCCTCAAACAGCTCAATG | NM_010442.2 |
| Il1β | TGCCACCTTTTGACAGTGATG | CGTCACACACCAGCAGGTTA | NM_008361.4 |
| Il10 | GTAGAAGTGATGCCCCAGGC | GACACCTTGGTCTTGGAGCTTATT | NM_010548.2 |
| Pparα | TGCCTTCCCTGTGAACTGAC | CACAGAGCGCTAAGCTGTGA | NM_001113418.1 |
| Pparγ | GGTCAGTCATGGAACAGCCA | TTCTGGGAGAGGTCTGCAC | NM_001411509.1 |
| Fatp5 | TGTAACGTCCCTGAGCAACC | TAAGCCCACATTGCCCTCTG | NM_009512.2 |
| Cpt2 | GAATGACAGCCAGTTCAGGAAG | GCATGCAGCTCCTTCCCAAT | NM_009949.2 |
| Col1a1 | CCCTGGTCCCTCTGGAAATG | GGACCTTTGCCCCCTTCTTT | NM_007742.4 |
| Gapdh | AACCCTTAAGAGGGATGCTGC | TCTACGGGACGAGGAAACAC | NM_001289726.2 |
Western blot analysis
Tissue sections were homogenized for protein extraction as already described [25]. Briefly, a small section of liver was resuspended in 1 mL of phosphate buffer solution (PBS) and then mechanically homogenized by Dounce homogenizer. The suspension was centrifuged 15 minutes at 13000 Rpm and supernatant was collected for further analysis. Therefore, we performed western blot analysis as described in [26]. Rabbit anti heat shock protein 90 (HSP90) (ab59459), heat shock protein 60 (HSP60) (ab46798), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (ab8245) were purchased from Abcam (Cambridge, UK). Rabbit anti CLPP (PA5-52722) was purchased from Thermo Fisher Scientific (Waltham, MA, USA). Secondary antibody anti Rabbit-HRP (ab6721) was purchased from Abcam (Cambridge, UK).
Histopathological analysis
Liver sections were formalin-fixed, paraffin-embedded and treated for histological examination using a standard method [27]. Two pathologists (R.C. and G.B.) separately evaluated all histological slides, blinded to sample identity. The following histological features were assessed on 5 micron-thick sections stained with hematoxylin and eosin and Masson’s trichrome, as previously described [28]: fibrosis, inflammation, steatosis and hepatocellular ballooning. Fibrosis was graded on a 0–3 scale: 0, absence of fibrosis; 1, portal spaces expanded by fibrosis with or without fibrous septa; 2, portal areas expanded by fibrosis with formation of fibrous bridges; 3, numerous fibrous bridges with formation of nodules. The following score was used to grade inflammation: 0, lack of inflammation; 1, periportal inflammation; 2, mild/moderate portal inflammation; 3, marked portal inflammation. Hepatocellular ballooning was scored on a 0–2 scale: 0, absent; 1, mild; 2, moderate/severe. The following 0–3 score was used to quantify steatosis: 0, absent; 1, mild; 2, moderate; 3, diffuse. For each case, the final histological score was performed by summing the scores of fibrosis, inflammation, steatosis, and ballooning.
Statistics
Data are shown as mean ± standard deviation (SD). Statistical analysis was performed by using Prism 8.0.2. software (GraphPad Software, San Diego, CA, USA). Significant differences were assessed using a one-way ANOVA or the student test, when needed. A value of p < 0.05 was considered statistically significant and symbols used to indicate statistical differences are described in figure legends.
Results
DS liver displays an enhanced exposure to oxidative stress
DS individuals display an impaired ROS scavenging system, which might be also hampering liver homeostasis [2]. To investigate this outcome also in our mice model, we therefore assayed the accumulation of glutathione (GSH) and lipid peroxide (LOOH) in wild type (Eu) and DS Ts2Cje liver homogenates derived from 3-, 6-, 9-, and 12-months old mice (Figure 1). Our cohort was composed by 49 mice: 3 months (6 Eu; 6 TS2Cje); 6 months (5 Eu; 6 TS2Cje); 9 months (6 Eu; 6 TS2Cje) and 12 months (6 Eu; 6 TS2Cje). Ts2Cje, but not Eu, were characterized by a deficit of GSH at 12-months. Any significant change was evident at any stage before the 12-months (Figure 1A). LOOH levels, on the other hand, were increased in 12-months old Ts2Cje liver homogenates compared to the Eu counterpart (Figure 1B). Similarly to GSH, also LOOH levels were not significatively changed at any of the time points assayed before (Figure 1B). Given this evidence, we next sought to investigate heme oxygenase 1 (HMOX1) expression in 3-, 6-, 9-, and 12-months old mice models by qPCR (Figure 1C). Our results show that there is a trend in increasing HMOX1 expression at 3-, 6-, and 9-months, but it turns to be significatively downregulated at 12-months in Ts2Cje livers compared to Eu counterpart (Figure 1C), overall highlighting an important increase in oxidative stress in DS mice model liver, and an impairment of the antioxidant defenses.
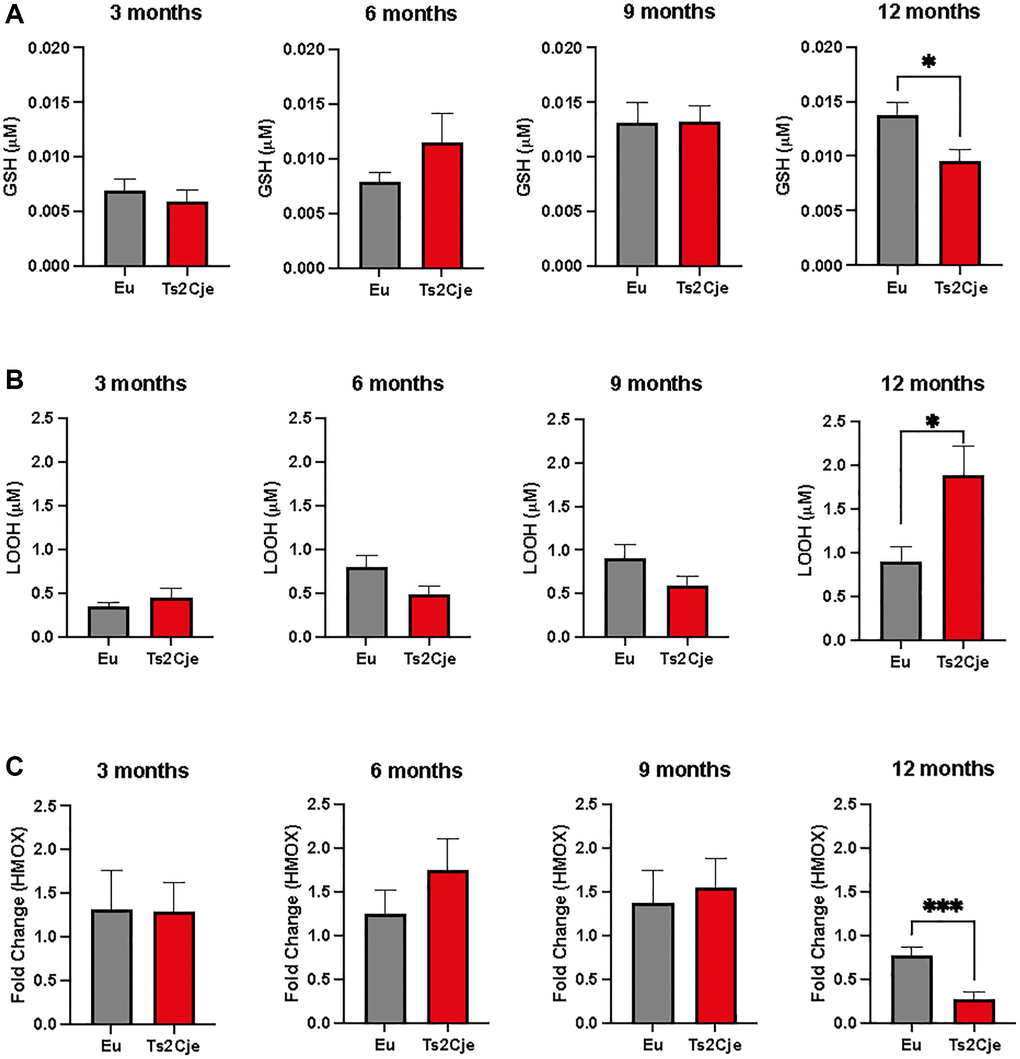
Figure 1. DS mice are characterized by an increased oxidative stress. (A) GSH levels are decreased in Ts2Cje 12-months old mice liver. Representative scheme for GSH quantitation in Eu and Ts2Cje livers obtained from 3-, 6-, 9-, and 12-months old mice. (B) LOOH levels are increased in Ts2Cje 12-months old mice liver. Histograms representative of LOOH spectrophotometric evaluation on Eu and TS2Cje livers obtained from 3-, 6-, 9-, and 12-months old mice. (C) HMOX1 expression is downregulated in Ts2Cje 12-months old livers. Representative histogram of the real-time PCR against HMOX1 on the liver extract obtained from 3-, 6-, 9-, and 12-months old Eu and Ts2Cje mice. GAPDH was used as housekeeping gene. Histograms are representative of four different experiments (*P ≤ 0.05; ***P ≤ 0.001).
DS mice livers show an increase of inflammatory markers
An increase in oxidative stress as the one we reported in Ts2Cje livers, might be correlated with an increase in the overall inflammatory status [29]. The latter is strictly linked to the accumulation of heat shock proteins (Hsps) [30]. For this reason, we sought to investigate the accumulation of Hsp90 and Hsp60 in our model. Western blot analysis unveiled a marked decrease of both Hsp90 and Hsp60 in Ts2Cje liver protein extract compared to Eu counterpart (Figure 2A–2C; Supplementary Figure 1).
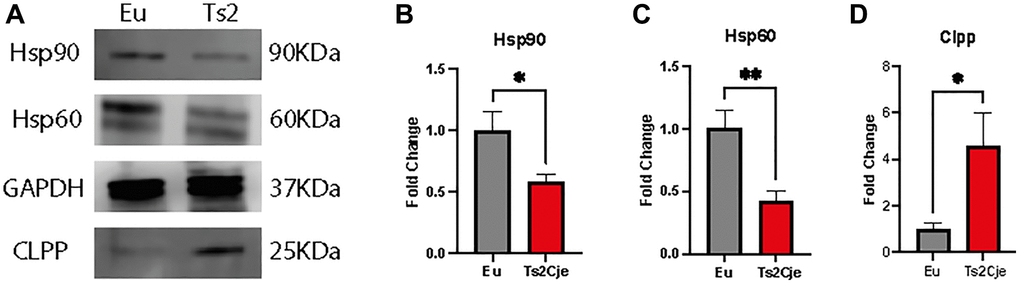
Figure 2. DS mice show a marked increase in liver inflammation. Western blot analysis on 12-months old Eu and Ts2Cje liver extracts against Hsp90, Hsp60, and Clpp (A). Ts2Cje mice showed a decreased Hsp90 (B), Hsp60 (C) levels, while Clpp accumulation is increased (D). GAPDH was used as housekeeping protein. Histograms are representative of four different experiments (*P ≤ 0.05; **P ≤ 0.01).
The increased inflammatory status has been reported to also affect the mitochondrial dynamics [31]. In this context, the Caseinolytic Mitochondrial Matrix Peptidase Proteolytic Subunit (Clpp) has been described as one of the major regulators of mitochondrial quality control system [32]. Interestingly, western blot analysis highlighted a marked increase in Clpp protein level in Ts2Cje liver homogenates compared to Eu counterpart (Figure 2A, 2D; Supplementary Figure 1). Overall, these data describe a scenario in which the increase in oxidative stress triggers an inflammatory status activating Hsp machinery and possibly impairing mitochondrial quality system.
DS liver increases lipid metabolism
The decreased level of Clpp has been described to be one of the NASH hallmarks, a pathology strictly associated to liver lipid accumulation [32]. To assay lipid accumulation in our model we assayed the expression of two members of peroxisome proliferator-activated receptors (PPARs) family. Interestingly, the expression of both PPARα and PPARγ in Ts2Cje liver were markedly decreased compared to the Eu counterpart (Figure 3A, 3B). Furthermore, qPCR analysis also described a marked increase in fatty acid transport protein-5 (FATP5) and Carnitine palmitoyl transferase 2 (CTP2) in Ts2Cje, but not in Eu livers (Figure 3C, 3D). Taken together these data we unveil an increase in fatty acids metabolism in DS mice livers.
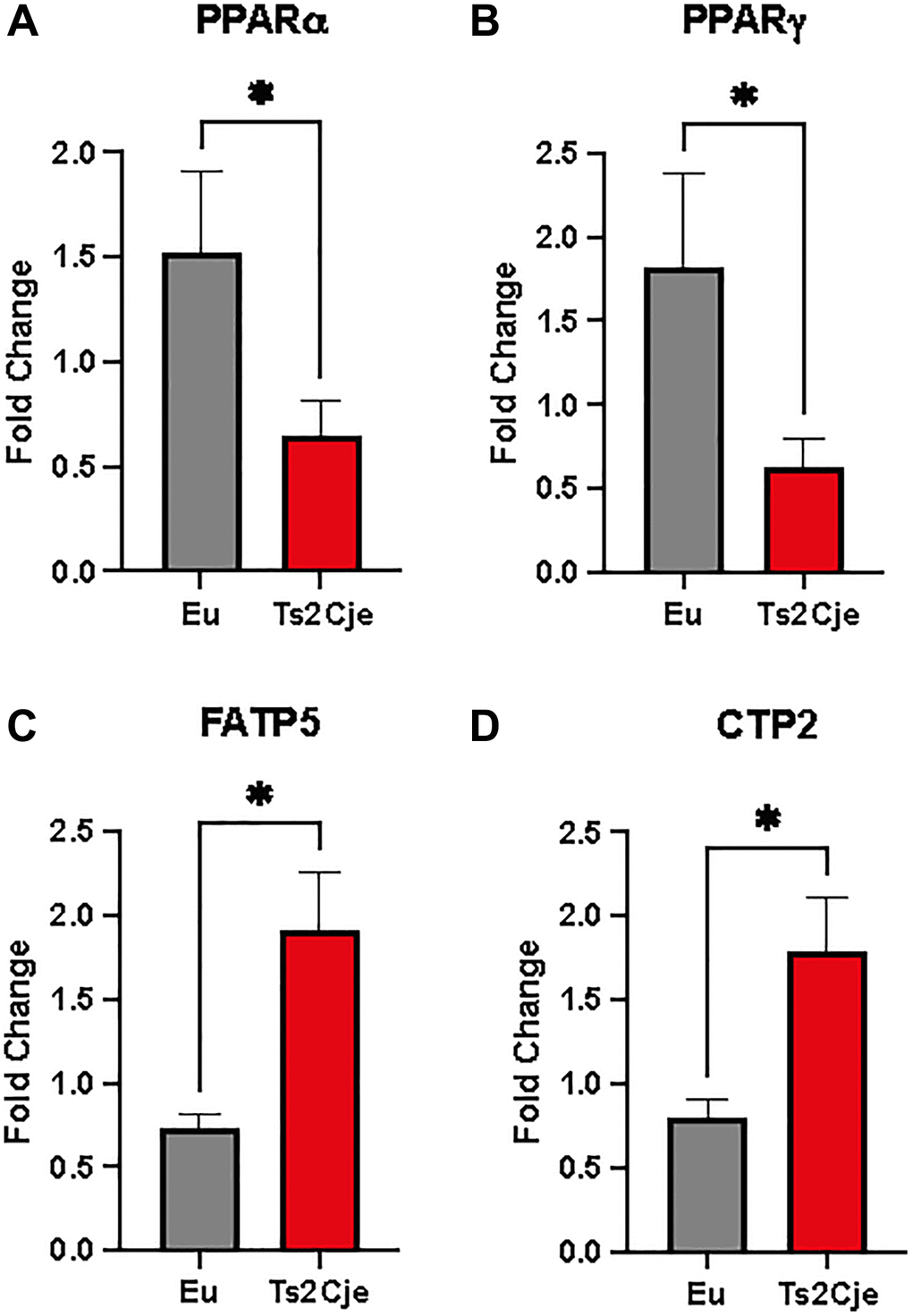
Figure 3. DS mice have a marked increase in lipid metabolism. PPARα, PPARγ, FATP5, and CTP2 are differently regulated in Ts2Cje livers. Representative histogram of the real-time PCR against PPARa (A), PPARg (B), FATP5 (C), and CTP2 (D) on the liver extract obtained from 12-months old Eu and Ts2Cje mice. GAPDH was used as housekeeping gene. Histograms are representative of four different experiments (*P ≤ 0.05).
DS livers display an increased fibrosis
The accumulation of lipids within the hepatic tissue might drive the shift from NASH toward cirrhosis [33]. This process is characterized by the accumulation of collagen. To test this outcome, we performed a qPCR testing Collagen Type I Alpha 1 Chain (Col1a1) expression, which was significantly increased in Ts2Cje livers compared to the Eu counterparts (Figure 4A).
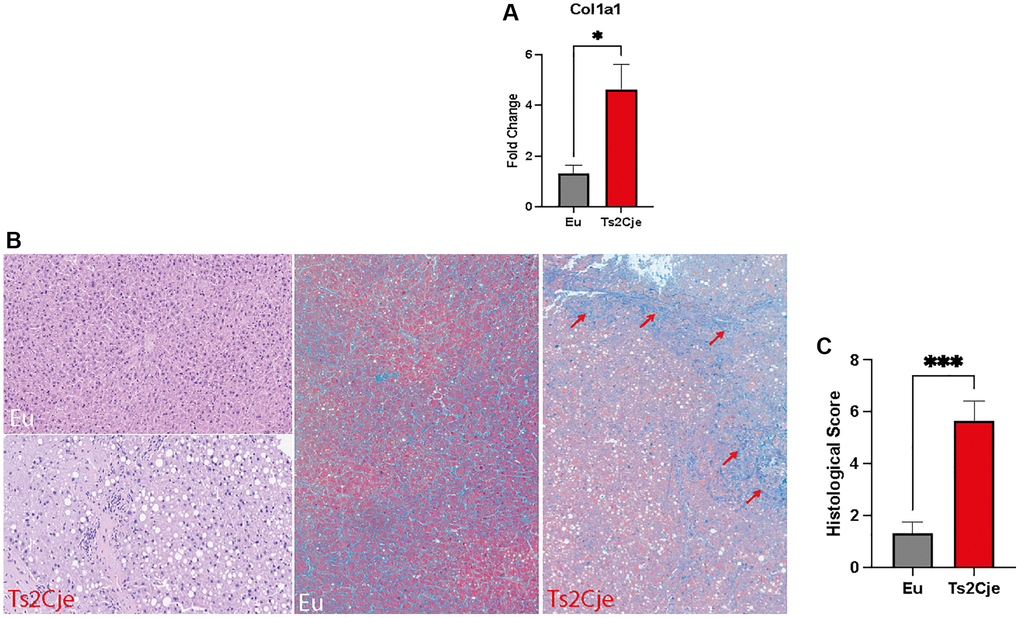
Figure 4. DS mice show a marked increase in fibrotic markers. (A) Col1a1 expression level is increased in Ts2Cje livers. Representative histogram of the real-time PCR against Col1a1 on the liver extract obtained from 12-months old Eu and Ts2Cje mice. GAPDH was used as housekeeping gene. (B) Ts2Cje liver section shows an increased fibrotic level. Representative histological images of the rat livers from control and Ts2Cje group. (Top left panel) Mouse liver from control group showing absence of steatosis (score 0) and mild periportal inflammation (score 1) (hematoxylin and eosin; original magnification 100x). (Bottom left panel) Mouse liver from Ts2Cje group exhibiting moderate steatosis (score 2) and moderate portal inflammation (score 2) (hematoxylin and eosin; original magnification 150x). (Mid panel) Mice liver from control group showing absence of fibrosis (score 0) (Masson’s trichrome; original magnification 50x). (Right panel) Mice liver from Ts2Cje group exhibiting diffuse fibrosis (score 3) with fibrous bridging (arrows) (Masson’s trichrome; original magnification 50x). (C) Histological score quantified as in B. Histograms are representative of four different experiments (*P ≤ 0.05; ***P ≤ 0.001).
Furthermore, we also performed a histological analysis on Ts2Cje and Eu liver sections, evaluating the fibrosis percentage (Figure 4B, 4C). Histologically, Eu group showed lack of fibrosis and hepatocellular ballooning, along with low levels of steatosis and inflammation compared to Ts2Cje counterpart (Figure 4B, 4C). Conversely, the Ts2Cje group fibrosis formation with, at least focal, fibrous septa/bridging and a more widespread inflammation and steatosis than controls (Figure 4B, 4C). Taken together, these data report an increased fibrotic rate in DS liver compared to healthy controls.
Discussion
Trisomy of chromosome 21 is the genetic signature of DS, impairing the cognitive and intellectual abilities of individuals with this condition. Previous reports showed that DS individuals are characterized by changes also on their metabolic profile [15] eventually affecting the homeostasis of several organs, including liver. The aim of the present study was to study the liver of a DS mouse model, which was previously generated and characterized by Barone and Perlugi’s lab [6, 7, 21, 34]. Interestingly, we found that 12-months old mice liver were characterized by a decreased GSH concentration, a well-known antioxidant molecule [35]. The decreased level of antioxidant defenses prompted us to investigate the concentration of LOOH in this context. Consistently, we also showed a concomitant increased oxidative environment in DS 12-months old mice liver as the result of the possible dysregulation of different factors occurring in redox homeostasis, as recently reviewed by our group [3]. Interestingly, we also reported a significant decrease in HMOX1 expression in 12-months old livers which is consistent with the increased expression of transcription factor Bach1, a master transcription repressor having HMOX1 as one of the main targets [36, 37], reported in the brain of DS subject, and further demonstrated in Ts2Cje mice model by Perluigi’s et al. [34, 36]. In particular, HMOX1 protein plays a role in the complex pathophysiological cascade involved in insulin resistance mechanisms, oxidative stress, metabolic syndrome and cardiovascular diseases [37–39]. The rewire of the oxidative balance in DS livers might also have a crucial role in rewiring the inflammatory response of this organ. Interestingly, we report here a repression of IL-1β transcription, along with an increase of IL-10 expression. As recently reported, an enhancement of IL-10 expression inhibits IL-1β production [40]. This result might be related to the increased susceptibility displayed by DS individuals in the development of several infections, given the lack of an inflammatory response [41]. To further assess the inflammatory response in our model, we also evaluated the protein accumulation of Hsp90, which was decreased in DS-livers. Noteworthy, no significative change in Hsp90 accumulation within DS brains has been reported [42] thus suggesting that such pathway may be specifically involved in the liver of DS. In addition, we also reported a decrease in the protein levels of the mitochondrial shock protein Hsp60. This data is consistent with a previous report describing a deficit in Hsp60 on skin fibroblasts derived from DS individuals [43]. In particular, Hsp60 is the major mitochondrial HSP, in charge for preventing protein aggregation following ROS unbalancing [44]. Therefore, we sought to assess the protein accumulation of one of the major regulators of mitochondrial fitness, Clpp, which we found to be upregulated in our DS model. Interestingly, it has been reported that under high fat diet, Clpp downregulation correlates with an increased protection against obesity and hepatic steatosis, also preventing insulin resistance [45, 46]. Furthermore, Clpp is also part of the machinery preventing hepatocytes senescence [45]. Therefore, we here hypothesize that the upregulation of this protein might be linked to the increased insulin resistance extensively described in DS individuals [15]. Furthermore, it might also be part of a compensatory effect preventing cellular senescence and liver cirrhosis, typical of DS individuals. To further investigate such possibility, we also assessed the expression level of PPARα and PPARγ, two ligand-activated transcription factors part of the nuclear hormone receptor superfamily, in turn in charge for regulating adipogenesis and insulin resistance [47]. Interestingly, the downregulation of PPARα and PPARγ, such as the one we highlighted in our model, could be related to the accumulation of lipid droplets within hepatocytes, also contributing to the insulin resistance reported in DS individuals [48, 49]. The accumulation of lipid droplets in DS liver is further supported by our data showing an increased expression of FATP5 and CTP2 in 12-months old liver compared to the wild-type counterpart. The former is responsible for the uptake of long-chain fatty acids [50]. Consistently, its expression has been reported to be inversely correlated to NAFLD progression [51]. The increase in fatty acids uptake might also be correlated to increased CTP2 expression, in turn in charge for initiating fatty acids oxidation eventually promoting their clearance [52]. In this context, fatty acids accumulation might work as a driver for the onset of liver fibrosis. Consistently, we reported an enhanced expression of Col1a1 in 12-month-old DS mice liver. Since an increased Col1a1 is usually associated with an enhanced fibrotic rate in hepatocellular carcinoma [53], we also decided to assess fibrosis of wild-type and DS livers by histopathology. Interestingly, our results corroborated a scenario in which DS livers display higher fibrosis and hepatocellular ballooning, together with increased steatosis and inflammation. Overall, these data unveil a significant increased risk for DS individuals to develop liver pathologies. Interestingly, liver failure has been reported to DS newborns showing transient abnormal myelopoiesis, a pathology characterized by transient appearance of blast cells and eventually also affecting liver homeostasis [54]. Furthermore, a diffuse lobular fibrosis around proliferating ductular elements and residual hepatocytes, as the one characterizing our DS models, was reported in DS newborns individuals presenting a severe liver disease [55]. These effects are further enhanced by the higher obesity rate reported in DS individuals, in turn promoting NAFLD onset by accumulation of hepatic fatty acids [56].
Overall, our work unveils a scenario in which DS liver is characterized by an impaired ROS scavenging system resulting in a significant impairment of redox homeostasis and leading to an impaired response to inflammatory stimuli. Finally, such results correlated with the increased fibrosis of DS animals, along with the accumulation of fatty lipids. In conclusion, our results put the basis for the use of antioxidants supplementation in DS patients to prevent liver-associated pathologies.
Supplementary Materials
Author Contributions
Conceptualization, M.P., G.L.V., E.B., and I.A.B.; Methodology, S.G., J.F., G.B., E.T.; Software, S.G., R.C., G.B.; Validation, S.G., J.F., E.T.; Formal analysis, S.G., A.T., I.A.B.; Investigation, M.P., G.L.V., E.B., I.A.B.; Resources, G.L.V., A.M.A.; Data curation, R.C., A.D.; Writing—original draft preparation, S.G., R.C., G.B., A.T.; Writing—review and editing, S.G., R.C., A.M.A., M.P., G.L.V., E.B., I.A.B.; Visualization, A.D., E.T.; Supervision, M.P., G.L.V., E.B., I.A.B.; Project administration, A.M.A., G.L.V., I.A.B.; Funding acquisition, A.M.A., G.L.V. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
All the experiments were performed in strict compliance with the Italian National Laws (DL 116/92), and the European Communities Council Directives (86/609/EEC). The experimental protocol was approved by the Italian Ministry of Health (#1183/2016-PR).
Funding
This researcher was funded by the researchers supporting project number (RSP2024R261) King Saud University, Riyadh, Saudi Arabia. This work was supported by Piano di Incentivi per la Ricerca di Ateneo 2020–2022, Linea di Intervento 2, “IMYTRA” to G.L.V.
References
- 1. Antonarakis SE, Skotko BG, Rafii MS, Strydom A, Pape SE, Bianchi DW, Sherman SL, Reeves RH. Down syndrome. Nat Rev Dis Primers. 2020; 6:9. https://doi.org/10.1038/s41572-019-0143-7 [PubMed]
- 2. Campos C, Casado Á. Oxidative stress, thyroid dysfunction & Down syndrome. Indian J Med Res. 2015; 142:113–9. https://doi.org/10.4103/0971-5916.164218 [PubMed]
- 3. Perluigi M, Butterfield DA. Oxidative Stress and Down Syndrome: A Route toward Alzheimer-Like Dementia. Curr Gerontol Geriatr Res. 2012; 2012:724904. https://doi.org/10.1155/2012/724904 [PubMed]
- 4. Giallongo S, Rehakova D, Raffaele M, Lo Re O, Koutna I, Vinciguerra M. Redox and Epigenetics in Human Pluripotent Stem Cells Differentiation. Antioxid Redox Signal. 2021; 34:335–49. https://doi.org/10.1089/ars.2019.7983 [PubMed]
- 5. Perluigi M, Tramutola A, Pagnotta S, Barone E, Butterfield DA. The BACH1/Nrf2 Axis in Brain in Down Syndrome and Transition to Alzheimer Disease-Like Neuropathology and Dementia. Antioxidants (Basel). 2020; 9:779. https://doi.org/10.3390/antiox9090779 [PubMed]
- 6. Zuliani I, Lanzillotta C, Tramutola A, Francioso A, Pagnotta S, Barone E, Perluigi M, Di Domenico F. The Dysregulation of OGT/OGA Cycle Mediates Tau and APP Neuropathology in Down Syndrome. Neurotherapeutics. 2021; 18:340–63. https://doi.org/10.1007/s13311-020-00978-4 [PubMed]
- 7. Lanzillotta C, Zuliani I, Tramutola A, Barone E, Blarzino C, Folgiero V, Caforio M, Valentini D, Villani A, Locatelli F, Butterfield DA, Head E, Perluigi M, et al. Chronic PERK induction promotes Alzheimer-like neuropathology in Down syndrome: Insights for therapeutic intervention. Prog Neurobiol. 2021; 196:101892. https://doi.org/10.1016/j.pneurobio.2020.101892 [PubMed]
- 8. Glasson EJ, Sullivan SG, Hussain R, Petterson BA, Montgomery PD, Bittles AH. The changing survival profile of people with Down's syndrome: implications for genetic counselling. Clin Genet. 2002; 62:390–3. https://doi.org/10.1034/j.1399-0004.2002.620506.x [PubMed]
- 9. Giallongo S, Longhitano L, Denaro S, D'Aprile S, Torrisi F, La Spina E, Giallongo C, Mannino G, Lo Furno D, Zappalà A, Giuffrida R, Parenti R, Li Volti G, et al. The Role of Epigenetics in Neuroinflammatory-Driven Diseases. Int J Mol Sci. 2022; 23:15218. https://doi.org/10.3390/ijms232315218 [PubMed]
- 10. Dimopoulos K, Constantine A, Clift P, Condliffe R, Moledina S, Jansen K, Inuzuka R, Veldtman GR, Cua CL, Tay ELW, Opotowsky AR, Giannakoulas G, Alonso-Gonzalez R, et al, and for Down Syndrome International (DSi). Cardiovascular Complications of Down Syndrome: Scoping Review and Expert Consensus. Circulation. 2023; 147:425–41. https://doi.org/10.1161/CIRCULATIONAHA.122.059706 [PubMed]
- 11. Rabin KR, Whitlock JA. Malignancy in children with trisomy 21. Oncologist. 2009; 14:164–73. https://doi.org/10.1634/theoncologist.2008-0217 [PubMed]
- 12. Kieran MW, Vekemans M, Robb LJ, Sinsky A, Outerbridge EW, Der Kaloustian VM. Portohepatic shunt in a Down syndrome patient with an interchange trisomy 47,XY,-2,+der(2),+der(21)t(2;21)(p13;q22.1)mat. Am J Med Genet. 1992; 44:288–92. https://doi.org/10.1002/ajmg.1320440305 [PubMed]
- 13. Burdall OC, Grammatikopoulos T, Sellars M, Hadzic N, Davenport M. Congenital Vascular Malformations of the Liver: An Association With Trisomy 21. J Pediatr Gastroenterol Nutr. 2016; 63:e141–6. https://doi.org/10.1097/MPG.0000000000001405 [PubMed]
- 14. Kaushik SP, Kaye G, Clarke AC. Autoimmune hepatobiliary disease in trisomy 21. J Clin Gastroenterol. 2000; 30:330–2. https://doi.org/10.1097/00004836-200004000-00029 [PubMed]
- 15. Dierssen M, Fructuoso M, Martínez de Lagrán M, Perluigi M, Barone E. Down Syndrome Is a Metabolic Disease: Altered Insulin Signaling Mediates Peripheral and Brain Dysfunctions. Front Neurosci. 2020; 14:670. https://doi.org/10.3389/fnins.2020.00670 [PubMed]
- 16. Valentini D, Alisi A, di Camillo C, Sartorelli MR, Crudele A, Bartuli A, Nobili V, Villani A. Nonalcoholic Fatty Liver Disease in Italian Children with Down Syndrome: Prevalence and Correlation with Obesity-Related Features. J Pediatr. 2017; 189:92–7.e1. https://doi.org/10.1016/j.jpeds.2017.05.077 [PubMed]
- 17. Valentini D, Mosca A, Di Camillo C, Crudele A, Sartorelli MR, Scoppola V, Tarani L, Villani A, Raponi M, Novelli A, Alisi A. PNPLA3 gene polymorphism is associated with liver steatosis in children with Down syndrome. Nutr Metab Cardiovasc Dis. 2020; 30:1564–72. https://doi.org/10.1016/j.numecd.2020.05.012 [PubMed]
- 18. Seeff LB, Levitsky J, Tillman PW, Perou ML, Zimmerman HJ. Histopathology of the liver in Down's syndrome. Am J Dig Dis. 1967; 12:1102–13. https://doi.org/10.1007/BF02233876 [PubMed]
- 19. Crudele A, Dato S, Re OL, Maugeri A, Sanna P, Giallongo S, Oben J, Panera N, De Rango F, Mosca A, Rose G, Passarino G, Alisi A, Vinciguerra M. Pediatric Non-Alcoholic Fatty Liver Disease Is Affected by Genetic Variants Involved in Lifespan/Healthspan. J Pediatr Gastroenterol Nutr. 2021; 73:161–8. https://doi.org/10.1097/MPG.0000000000003123 [PubMed]
- 20. Reinholdt LG, Ding Y, Gilbert GJ, Czechanski A, Solzak JP, Roper RJ, Johnson MT, Donahue LR, Lutz C, Davisson MT. Molecular characterization of the translocation breakpoints in the Down syndrome mouse model Ts65Dn. Mamm Genome. 2011; 22:685–91. https://doi.org/10.1007/s00335-011-9357-z [PubMed]
- 21. Tramutola A, Lanzillotta S, Aceto G, Pagnotta S, Ruffolo G, Cifelli P, Marini F, Ripoli C, Palma E, Grassi C, Di Domenico F, Perluigi M, Barone E. Intranasal Administration of KYCCSRK Peptide Rescues Brain Insulin Signaling Activation and Reduces Alzheimer's Disease-like Neuropathology in a Mouse Model for Down Syndrome. Antioxidants (Basel). 2023; 12:111. https://doi.org/10.3390/antiox12010111 [PubMed]
- 22. Nibali L, Rizzo M, Li Volti G, D'Aiuto F, Giglio RV, Barbagallo I, Pelekos G, Donos N. Lipid subclasses profiles and oxidative stress in aggressive periodontitis before and after treatment. J Periodontal Res. 2015; 50:890–6. https://doi.org/10.1111/jre.12283 [PubMed]
- 23. Longhitano L, Giallongo S, Orlando L, Broggi G, Longo A, Russo A, Caltabiano R, Giallongo C, Barbagallo I, Di Rosa M, Giuffrida R, Parenti R, Li Volti G, et al. Lactate Rewrites the Metabolic Reprogramming of Uveal Melanoma Cells and Induces Quiescence Phenotype. Int J Mol Sci. 2022; 24:24. https://doi.org/10.3390/ijms24010024 [PubMed]
- 24. Longhitano L, Broggi G, Giallongo S, Failla M, Puzzo L, Avitabile T, Tibullo D, Distefano A, Pittalà V, Reibaldi M, Zanghì GN, Longo A, Russo A, et al. Heme Oxygenase-1 Overexpression Promotes Uveal Melanoma Progression and Is Associated with Poor Clinical Outcomes. Antioxidants (Basel). 2022; 11:1997. https://doi.org/10.3390/antiox11101997 [PubMed]
- 25. Herron S, Delpech JC, Madore C, Ikezu T. Using mechanical homogenization to isolate microglia from mouse brain tissue to preserve transcriptomic integrity. STAR Protoc. 2022; 3:101670. https://doi.org/10.1016/j.xpro.2022.101670 [PubMed]
- 26. Cambria D, Longhitano L, La Spina E, Giallongo S, Orlando L, Giuffrida R, Tibullo D, Fontana P, Barbagallo I, Nicoletti VG, Volti GL, Fabro VD, Coda ARD, et al. IGFBP-6 Alters Mesenchymal Stromal Cell Phenotype Driving Dasatinib Resistance in Chronic Myeloid Leukemia. Life (Basel). 2023; 13:259. https://doi.org/10.3390/life13020259 [PubMed]
- 27. Broggi G, Lo Giudice A, Di Mauro M, Asmundo MG, Pricoco E, Piombino E, Caltabiano R, Morgia G, Russo GI. SRSF-1 and microvessel density immunohistochemical analysis by semi-automated tissue microarray in prostate cancer patients with diabetes (DIAMOND study). Prostate. 2021; 81:882–92. https://doi.org/10.1002/pros.24185 [PubMed]
- 28. Mangano K, Cavalli E, Mammana S, Basile MS, Caltabiano R, Pesce A, Puleo S, Atanasov AG, Magro G, Nicoletti F, Fagone P. Involvement of the Nrf2/HO-1/CO axis and therapeutic intervention with the CO-releasing molecule CORM-A1, in a murine model of autoimmune hepatitis. J Cell Physiol. 2018; 233:4156–65. https://doi.org/10.1002/jcp.26223 [PubMed]
- 29. Sharifi-Rad M, Anil Kumar NV, Zucca P, Varoni EM, Dini L, Panzarini E, Rajkovic J, Tsouh Fokou PV, Azzini E, Peluso I, Prakash Mishra A, Nigam M, El Rayess Y, et al. Lifestyle, Oxidative Stress, and Antioxidants: Back and Forth in the Pathophysiology of Chronic Diseases. Front Physiol. 2020; 11:694. https://doi.org/10.3389/fphys.2020.00694 [PubMed]
- 30. Moseley PL. Heat shock proteins and the inflammatory response. Ann N Y Acad Sci. 1998; 856:206–13. https://doi.org/10.1111/j.1749-6632.1998.tb08327.x [PubMed]
- 31. Missiroli S, Genovese I, Perrone M, Vezzani B, Vitto VAM, Giorgi C. The Role of Mitochondria in Inflammation: From Cancer to Neurodegenerative Disorders. J Clin Med. 2020; 9:740. https://doi.org/10.3390/jcm9030740 [PubMed]
- 32. Choi SE, Hwang Y, Lee SJ, Jung H, Shin TH, Son Y, Park S, Han SJ, Kim HJ, Lee KW, Lee G, Kemper JK, Song HK, Kang Y. Mitochondrial protease ClpP supplementation ameliorates diet-induced NASH in mice. J Hepatol. 2022; 77:735–47. https://doi.org/10.1016/j.jhep.2022.03.034 [PubMed]
- 33. Pei K, Gui T, Kan D, Feng H, Jin Y, Yang Y, Zhang Q, Du Z, Gai Z, Wu J, Li Y. An Overview of Lipid Metabolism and Nonalcoholic Fatty Liver Disease. Biomed Res Int. 2020; 2020:4020249. https://doi.org/10.1155/2020/4020249 [PubMed]
- 34. Pagnotta S, Tramutola A, Barone E, Di Domenico F, Pittalà V, Salerno L, Folgiero V, Caforio M, Locatelli F, Petrini S, Butterfield DA, Perluigi M. CAPE and its synthetic derivative VP961 restore BACH1/NRF2 axis in Down Syndrome. Free Radic Biol Med. 2022; 183:1–13. https://doi.org/10.1016/j.freeradbiomed.2022.03.006 [PubMed]
- 35. Averill-Bates DA. The antioxidant glutathione. Vitam Horm. 2023; 121:109–41. https://doi.org/10.1016/bs.vh.2022.09.002 [PubMed]
- 36. Di Domenico F, Pupo G, Mancuso C, Barone E, Paolini F, Arena A, Blarzino C, Schmitt FA, Head E, Butterfield DA, Perluigi M. Bach1 overexpression in Down syndrome correlates with the alteration of the HO-1/BVR-a system: insights for transition to Alzheimer's disease. J Alzheimers Dis. 2015; 44:1107–20. https://doi.org/10.3233/JAD-141254 [PubMed]
- 37. Barbagallo I, Marrazzo G, Frigiola A, Zappala A, Li Volti G. Role of carbon monoxide in vascular diseases. Curr Pharm Biotechnol. 2012; 13:787–96. https://doi.org/10.2174/138920112800399086 [PubMed]
- 38. Barbagallo I, Galvano F, Frigiola A, Cappello F, Riccioni G, Murabito P, D'Orazio N, Torella M, Gazzolo D, Li Volti G. Potential therapeutic effects of natural heme oxygenase-1 inducers in cardiovascular diseases. Antioxid Redox Signal. 2013; 18:507–21. https://doi.org/10.1089/ars.2011.4360 [PubMed]
- 39. Barbagallo I, Nicolosi A, Calabrese G, David S, Cimino S, Madonia M, Cappello F. The role of the heme oxygenase system in the metabolic syndrome. Curr Pharm Des. 2014; 20:4970–4. https://doi.org/10.2174/1381612819666131206103824 [PubMed]
- 40. Sun Y, Ma J, Li D, Li P, Zhou X, Li Y, He Z, Qin L, Liang L, Luo X. Interleukin-10 inhibits interleukin-1β production and inflammasome activation of microglia in epileptic seizures. J Neuroinflammation. 2019; 16:66. https://doi.org/10.1186/s12974-019-1452-1 [PubMed]
- 41. Broers CJ, Gemke RJ, Morré SA, Weijerman ME, van Furth AM. Increased production of interleukin-10 in children with Down syndrome upon ex vivo stimulation with Streptococcus pneumoniae. Pediatr Res. 2014; 75:109–13. https://doi.org/10.1038/pr.2013.173 [PubMed]
- 42. Yoo BC, Vlkolinsky R, Engidawork E, Cairns N, Fountoulakis M, Lubec G. Differential expression of molecular chaperones in brain of patients with Down syndrome. Electrophoresis. 2001; 22:1233–41. https://doi.org/10.1002/1522-2683()22:6%3c1233::AID-ELPS1233%3e3.0.CO;2-M [PubMed]
- 43. Bozner P, Wilson GL, Druzhyna NM, Bryant-Thomas TK, LeDoux SP, Wilson GL, Pappolla MA. Deficiency of chaperonin 60 in Down's syndrome. J Alzheimers Dis. 2002; 4:479–86. https://doi.org/10.3233/jad-2002-4604 [PubMed]
- 44. Martin J, Horwich AL, Hartl FU. Prevention of protein denaturation under heat stress by the chaperonin Hsp60. Science. 1992; 258:995–8. https://doi.org/10.1126/science.1359644 [PubMed]
- 45. Dabravolski SA, Bezsonov EE, Orekhov AN. The role of mitochondria dysfunction and hepatic senescence in NAFLD development and progression. Biomed Pharmacother. 2021; 142:112041. https://doi.org/10.1016/j.biopha.2021.112041 [PubMed]
- 46. Bhaskaran S, Pharaoh G, Ranjit R, Murphy A, Matsuzaki S, Nair BC, Forbes B, Gispert S, Auburger G, Humphries KM, Kinter M, Griffin TM, Deepa SS. Loss of mitochondrial protease ClpP protects mice from diet-induced obesity and insulin resistance. EMBO Rep. 2018; 19:e45009. https://doi.org/10.15252/embr.201745009 [PubMed]
- 47. Gervois P, Torra IP, Fruchart JC, Staels B. Regulation of lipid and lipoprotein metabolism by PPAR activators. Clin Chem Lab Med. 2000; 38:3–11. https://doi.org/10.1515/CCLM.2000.002 [PubMed]
- 48. Wang M, Yan Y, Zhang Z, Yao X, Duan X, Jiang Z, An J, Zheng P, Han Y, Wu H, Wang Z, Glauben R, Qin Z. Programmed PPAR-α downregulation induces inflammaging by suppressing fatty acid catabolism in monocytes. iScience. 2021; 24:102766. https://doi.org/10.1016/j.isci.2021.102766 [PubMed]
- 49. Leonardini A, Laviola L, Perrini S, Natalicchio A, Giorgino F. Cross-Talk between PPARgamma and Insulin Signaling and Modulation of Insulin Sensitivity. PPAR Res. 2009; 2009:818945. https://doi.org/10.1155/2009/818945 [PubMed]
- 50. Doege H, Baillie RA, Ortegon AM, Tsang B, Wu Q, Punreddy S, Hirsch D, Watson N, Gimeno RE, Stahl A. Targeted deletion of FATP5 reveals multiple functions in liver metabolism: alterations in hepatic lipid homeostasis. Gastroenterology. 2006; 130:1245–58. https://doi.org/10.1053/j.gastro.2006.02.006 [PubMed]
- 51. Enooku K, Tsutsumi T, Kondo M, Fujiwara N, Sasako T, Shibahara J, Kado A, Okushin K, Fujinaga H, Nakagomi R, Minami T, Sato M, Uchino K, et al. Hepatic FATP5 expression is associated with histological progression and loss of hepatic fat in NAFLD patients. J Gastroenterol. 2020; 55:227–43. https://doi.org/10.1007/s00535-019-01633-2 [PubMed]
- 52. Parimbelli M, Pezzotti E, Negro M, Calanni L, Allemano S, Bernardi M, Berardinelli A, D'Antona G. Nutrition and Exercise in a Case of Carnitine Palmitoyl-Transferase II Deficiency. Front Physiol. 2021; 12:637406. https://doi.org/10.3389/fphys.2021.637406 [PubMed]
- 53. Ma HP, Chang HL, Bamodu OA, Yadav VK, Huang TY, Wu ATH, Yeh CT, Tsai SH, Lee WH. Collagen 1A1 (COL1A1) Is a Reliable Biomarker and Putative Therapeutic Target for Hepatocellular Carcinogenesis and Metastasis. Cancers (Basel). 2019; 11:786. https://doi.org/10.3390/cancers11060786 [PubMed]
- 54. Park MJ, Sotomatsu M, Ohki K, Arai K, Maruyama K, Kobayashi T, Nishi A, Sameshima K, Takagi T, Hayashi Y. Liver disease is frequently observed in Down syndrome patients with transient abnormal myelopoiesis. Int J Hematol. 2014; 99:154–61. https://doi.org/10.1007/s12185-013-1487-5 [PubMed]
- 55. Ruchelli ED, Uri A, Dimmick JE, Bove KE, Huff DS, Duncan LM, Jennings JB, Witzleben CL. Severe perinatal liver disease and Down syndrome: an apparent relationship. Hum Pathol. 1991; 22:1274–80. https://doi.org/10.1016/0046-8177(91)90111-2 [PubMed]
- 56. De Matteo A, Vajro P. Down Syndrome and Pediatric Nonalcoholic Fatty Liver Disease: A Causal or Casual Relationship? J Pediatr. 2017; 189:11–3. https://doi.org/10.1016/j.jpeds.2017.07.011 [PubMed]



