Introduction
Multiple myeloma (MM) is an incurable malignant plasma cell disease of blood system, which is characterized by malignant expansion of monoclonal plasma cells in bone marrow or extramedullary position [1]. In recent years, the incidence of MM has increased, and the onset age is mostly over 65 years old. MM develops from an asymptomatic precancerous stage, first with MGUS, then with SMM, and finally with active multiple myeloma [2, 3]. Abnormal plasma cells of MM patients secrete monoclonal immunoglobulin, which leads to end organ damage and characteristic clinical manifestations such as hypercalcemia, renal failure, anemia and osteolytic bone destruction [4].
With the in-depth exploration of the pathogenesis and drug development of MM, many therapies, such as proteasome inhibitors, immunomodulatory drugs, antibody-drug conjugates (ADC) and chimeric antigen receptor (CAR) T-cell therapy, have been applied to MM, and achieved certain curative effects [5]. These drugs alone or in combination can improve the survival rate of newly diagnosed or relapsed MM patients from 25% in the mid-1970s to 55% [6]. The recent follow-up results of a phase III clinical trial for relapsed/refractory (RR) MM patients showed that patients treated with caffezomib, dyramuzumab and dexamethasone (KdD) had a higher realization rate of minimal residual disease-negative (MRD-) than patients treated with caffezomib and dexamethasone (Kd) (28% vs 9%), and the median PFS and OS of KdD group were significantly higher than those of Kd group [7]. Another real-world study included 200 RR MM patients who were treated with elotuzumab, pomalidomide and dexamethasone (EloPD). The overall response rate was 55.4%. Although the median PFS and OS were lower than those in the clinical trial of ELOQUENT-3, it still improved the survival rate of patients with RR MM [8]. The prognosis of MM has been greatly improved, but drug resistance and recurrence have seriously affected the economy and prognosis of patients. Some preclinical research to develop new drugs based on the target of MM may be a promising method. ISB 1342 is a CD38 × CD3 T-cell engager, which can effectively kill MM cell lines with low sensitivity to dartumumab in vitro, which may improve the survival time of patients with dartumumab resistance [9]. At present, MM can’t be cured, and patients still have many recurrences after remission, and the remission time after each recurrence is gradually shortened. With the development of cancer molecular biology and genomics, people’s understanding of tumor molecular phenotype is deepening. The strategy of tumor treatment has gradually changed into the era of precise targeted therapy for genomic changes. Therefore, finding new biomarkers is beneficial to insight into the pathogenesis and improve the prognosis of MM.
RAB22A is a member of RAS oncogene family, and its expression is increased in many cancers such as breast cancer [10], colorectal cancer [11], osteosarcoma [12] and liver cancer [13], and it is involved in the proliferation, migration and immune regulation of malignant tumors. As a small GTPase, RAB22A belongs to RAB5 subfamily, mainly located in early endosomes, Golgi bodies and late endosomes [14]. RAB proteins are involved in the regulation of vesicle transport, early endosome and exosome formation and endosome recovery. In one study, RAB22A mediated the migration and invasion of breast cancer cells by promoting exosome secretion, which can be regulated by targeting RAB22A through tumor suppressor gene miR-19b [15]. Interestingly, another study found that RAB22A-mediated autophagosome containing STING fused with early endosomes, resulting in a new organelle, Rafeesome, which promoted the secretion of vesicles packaging STING into tumor microenvironment, thus mediating anti-tumor immunity [16]. RAB22A can mediate EMT, promote tumor cell migration and lead to tumor progression. MCF2L-AS1 promoted the EMT of colorectal cancer by targeting the miR-105-5p/RAB22A axis, thus making the malignant progression of colorectal cancer [17]. DNMT1 regulated the DNA methylation in the promoter region of miR-211, decreased the expression level of miR-211 in melanoma cells and increased the expression level of its downstream target gene RAB22A, thus promoting the EMT process [18]. In addition, RAB22A, as an immunomodulatory factor, plays an immune role in many tumors. RAB22A controlled MHC-I intrinsic distribution, recycling and trafficking to dendritic cell (DC) phage, which played an extremely important role in the antigen cross-presentation reaction of DC [19]. In another study, RAB22A was significantly up-regulated in hepatocellular carcinoma, which was significantly related to the infiltration of various immune cells and the gene expression of immune checkpoints, and may lead to poor prognosis of hepatocellular carcinoma by regulating immune function [13]. However, RAB22A is rarely studied in hematological tumors, and there is no related research in MM at present. Therefore, it urges us to explore the effects of RAB22A in MM on extracellular vesicle secretion, EMT and immune regulation.
In this study, we analyzed the difference of the expression level of RAB22A between normal people and MM patients, between newly diagnosed and relapsed MM patients by including MM samples in GEO databases. We also explored the related biological functions of high and low RAB22A expression groups through EMT gene set and exosome gene set. In addition, we analyzed the correlation between RAB22A and immune cell infiltration and immune checkpoint genes. Finally, we made a preliminary verification in the clinical samples of our center. Our study explored the effect of RAB22A expression in MM on EMT, exosome secretion and immune regulation, which has guiding significance for the prognosis prediction and drug target development of MM.
Materials and Methods
Source of data
We obtained 78 samples from GSE5900 dataset in GEO database, including 22 normal people, 44 MGUS patients and 12 SMM patients. We also extracted the data of 414 newly diagnosed MM patients from GSE4581 dataset for the analysis of RAB22A expression level and biological function. In addition, we extracted the MSCs expression data of 12 newly diagnosed and 9 relapsed MM patients from GSE146649 dataset, and analyzed the expression levels of RAB22A and exosome-related genes. We use “sva” package to correct batch effect in these data sets.
Analysis of differentially expressed genes (DEGs) of RAB22A
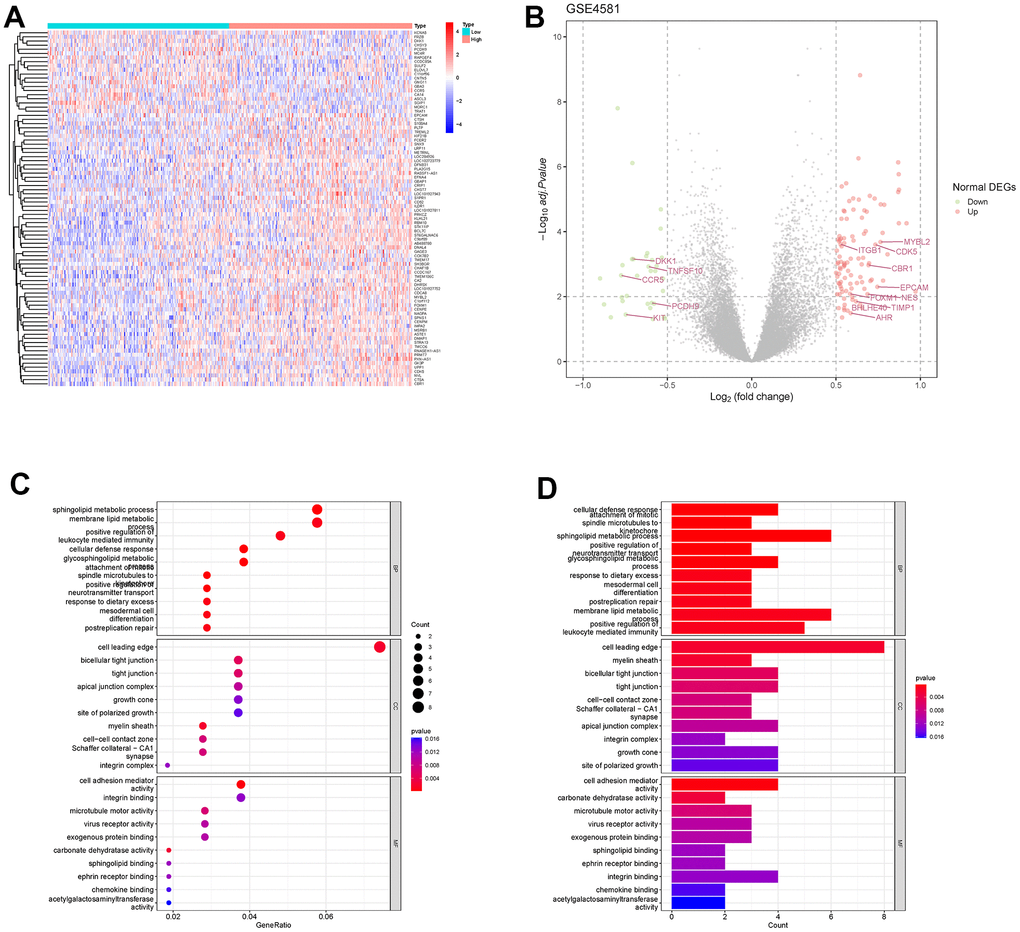
We extracted the RAB22A expression level of 414 newly diagnosed MM patients with GSE4581 by using the “limma” package, and divided the samples into high RAB22A expression group and low RAB22A expression group according to the median value. The heatmap and volcano plot showed the DEGs in the high and low RAB22A expression groups. Gene Ontology (GO) enrichment analysis and Kyoto encyclopedia of genes and genomes (KEGG) enrichment analysis were carried out to further explore the potential biological functions.
Analysis of immune cell infiltration
We used the “CIBERSORT” package for immune cell infiltration analysis. The degree of immune cell infiltration in high and low RAB22A expression groups was analyzed, and the “ggplot2” and “ggpubr” packages were used to draw borplot and barplot. In addition, we further drew scatterplots between the expression level of RAB22A and the infiltration level of immune cells.
Calculation of EMT and exosome scores
We downloaded the “IOBR” package with “calculate_sig_score” function and used “PCA” and “ssGSEA” algorithm to calculate EMT scores and exosomes scores of different RAB22A groups.
Clinical samples and cell lines
We collected the clinical information and bone marrow samples of 71 normal controls and 30 newly diagnosed MM patients in the hematology department of the Affiliated Hospital of Guizhou Medical University. All participants obtained informed consent, and the research was approved by the ethics committee of the Affiliated Hospital of Guizhou Medical University. Human MM cell lines RPMI 8226 and U266 were provided by Hematopoietic Stem Cell Transplantation Center Laboratory in Guizhou province. All human cell lines were tested for mycoplasma contamination and verified by short tandem repeats. Ficoll gradient centrifugation was used to separate MSCs, and the 2-4 generation cells were selected for further experiments. Cell lines were cultured in RPMI-1640 (Gibco, USA) medium with 10% fetal bovine serum (FBS), penicillin (100 units/mL) and streptomycin (100 mg/mL) at 37° C and 5% CO2 with saturating humidity. MSCs were cultured in complete medium of bone marrow mesenchymal stromal cells (BM-MSCs) (OriCell, China), and 48 hours before exosomes were extracted, they were changed to medium containing exosome-depleted FBS (OriCell, China).
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from cells by using Trizol reagent (Invitrogen, USA) according to the manufacturer’s instruction. SYBR Green PCR Master Mix (MCE, China) and PRISM 7500 real-time PCR detection system (ABI, USA) were used for real-time PCR detection. The primers used in this study are as follows: for RAB22A, forward: 5′-TACCAAAGAGGCAAAGCATGT GCG-3′, reverse: 5′-AGACACCATGCAATCACCAACAGC-3′; for β-actin, forward: 5′-GAGACCTTCAACACCCCAGC-3′, reverse:5′-ATGTCACGCACGATTTCCC-3′. The relative expression of mRNA was normalized to β-actin and calculated by 2-ΔCT.
Western blotting analysis
Western blotting was used to detect the expression of RAB22A in normal people and MM patients, and normalized it to β-actin. BCA protein assay kit (Pierce, Hercules, USA) was used to determine the protein concentration. The protein samples were loaded on 10% SDS-PAGE gel and transferred to PVDF membranes. After being sealed with 5% skim milk, the membrane was incubated overnight at 4° C with the following antibodies: anti-RAB22A (1:500, Affinity, China) and anti β-actin (1:3000, Proteintech Group, China). Protein bands were observed using ECL kit (7 Sea Biotechnology, China).
Extraction and identification of exosomes
According to the manufacturer’s instructions, the supernatant of MSCs cultured in medium containing exosome-depleted FBS was collected. Exosome concentration solution (MCE, China) was added to MSC supernatant, and crude exosomes were obtained after centrifugation. Purified exosomes were obtained by centrifugation with exosome purification filter (MCE, China). Western blotting was used to identify exosome surface markers CD63 (Immunoway, USA) and TSG101 (Immunoway, USA).
Immunofluorescence
The exosomes resuspended in 200ul phosphate buffer saline were incubated with PKH26 (Solarbio, China) in the dark for 5 minutes. MM cell lines were inoculated into 24-well plates, and the labeled MSCs-derived exosomes were added. After 24 hours, the cells were collected and fixed with formaldehyde, then 4’,6-diamidino-2-phenylindole (DAPI) was added for nuclear staining. After that, the cells were observed under a fluorescence microscope.
Cell activity detection
Cells were inoculated into a 96-well plate, and exosomes filtered with a 0.22um filter were added. Cell Counting Kit-8 (CCK-8) was added at different time points and incubated at 37° C for 2 hours. The absorbance at 450nm was measured by spectrophotometer.
Statistical analysis
R software package (version 4.1.3) was used for statistical analysis and visualization of results. Shapiro-Wilk normality test was used to test the normality of variables. Unpaired student’s t-test was used to evaluate the statistical significance of the data that conforms to the normal distribution. Using 1: 1 properness score matching (PSM) to eliminate the bias between the control group and the newly diagnosed MM group. P<0.05 indicated statistical significance.
Data availability statement
All data supporting the results of this study can be obtained from the corresponding authors according to reasonable requirements. The datasets presented in this study can be found in online repositories. The names of the repository can be found below: https://portal.gdc.cancer.gov.
Results
The expression of RAB22A increased in MM
Firstly, we explored the expression level of RAB22A in normal people, MGUS, SMM and MM patients, and found that the expression level of RAB22A in SMM and MM patients was significantly higher than that in normal people and MGUS patients (Figure 1A). In order to further clarify the prognostic role of RAB22A in MM, we analyzed the expression level of RAB22A in newly diagnosed and relapsed MM patients. The results showed that the expression level of RAB22A in relapsed MM patients was significantly higher than that in newly diagnosed MM patients (Figure 1B). These results showed the prognostic value of RAB22A for MM patients, which urged us to further explore the biological function of RAB22A.
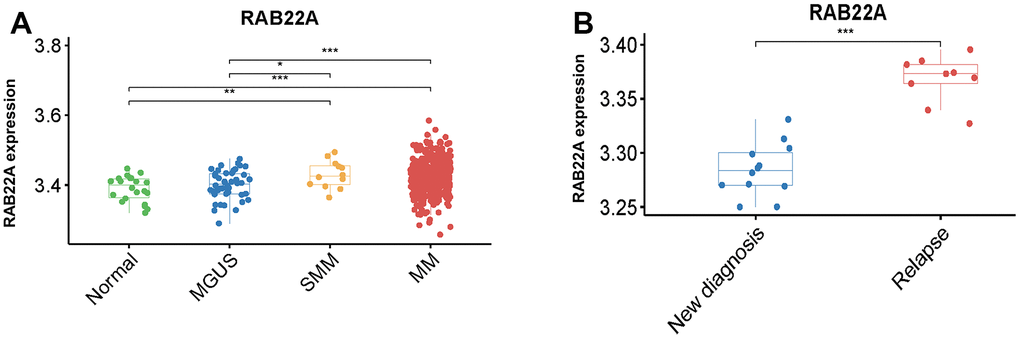
Figure 1. Differential analysis of expression level of RAB22A. (A) Differential expression analysis of RAB22A in normal people, MGUS, SMM and MM patients. (B) Differential expression of RAB22A in MSC of newly diagnosed and relapsed MM patients.
Correlation between RAB22A and EMT
We obtained the DEGs in the high and low RAB22A expression groups, and intersected them with the EMT gene set to obtain 15 EMT genes related to RAB22A. We showed the EMT genes expression levels (Figure 3A, 3B) among different RAB22A expression groups by using heatmap and boxplot. The results showed that the expression levels of MYBL2, ITGB1, CDK5, CBR1, EPCAM, S100A4, FOXM1, TIMP1, BHLHE40 and AHR in high RAB22A expression group were higher than those in low RAB22A expression group, while the expression levels of DKK1, TNFSF10, CCR5, PCDH9 and KIT in high RAB22A expression group were lower. In addition, we drew scatterplots of the correlation between RAB22A and EMT genes (Figure 3D–3R). Finally, we calculated the EMT scores of different RAB22A groups through the “IOBR” package, and the results showed that the EMT scores of high RAB22A expression group were significantly higher than those of low RAB22A expression group (Figure 3C).
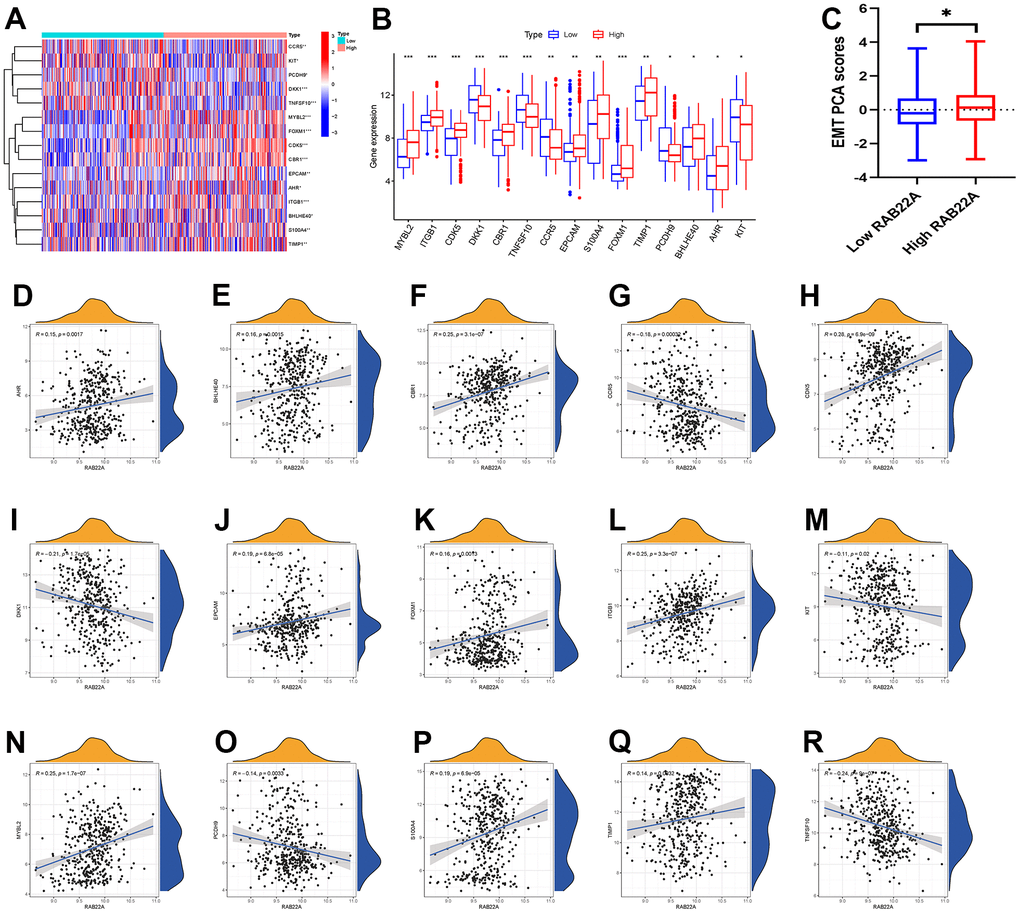
Figure 3. Correlation analysis between RAB22A and EMT. (A, B) EMT-related genes with significantly different expression levels between high and low RAB22A groups. (C) EMT scores of high and low RAB22A groups were calculated. (D–R) Scatterplots and correlation of RAB22A and EMT-related genes.
Correlation between RAB22A expression and exosome scores
Exosomes contain microRNAs (miRNAs), mRNAs, proteins, lipids and so on, which are used for information transmission between cells. MSCs are an important part of bone marrow microenvironment, and BM-MSCs can mediate drug resistance of many hematological neoplasms [24]. BM-MSCs can influence tumor cells through various information interaction ways, and induce tumor cells to escape the killing of chemotherapy drugs. Previous studies have found that exosomes secreted by BM-MSCs in tumor microenvironment affect the progression of leukemia by transporting microRNA-7-5p into acute myeloid leukemia (AML) cells [25]. Therefore, the exosomes secretion of MSCs may be an important factor leading to the progress of hematological tumors, which urges us to pay attention to the genes regulating exosomes secretion.
We extracted the expression data of MSCs from 12 newly diagnosed MM patients and 9 relapsed MM patients in GSE146649 dataset, and calculated the exosome scores with “ssGSEA” algorithm. The results showed that the exosome scores of MSC in relapsed patients was significantly higher than that in newly diagnosed patients (Figure 4A). Then, we further analyzed the expression levels of exosome-related genes in newly diagnosed and relapsed patients. The results showed that the expression levels of CFL1, ACTB, CCT4, RDX, TSG101, FLOT1, CTTN, and STAT3 in relapsed patients were significantly higher than those in newly diagnosed patients (Figure 4B, 4C). In order to further clarify the effect of RAB22A on exosomes secretion, we extracted the expression data of RAB22A from newly diagnosed and relapsed patients, and divided the samples into high and low RAB22A expression groups according to the median value. The results showed that the exosome scores of high RAB22A expression group was significantly higher than that of low RAB22A expression group (Figure 5A). Heatmap and boxplot showed that the expression levels of exosome-related genes in high RAB22A expression group and low RAB22A expression group. The results showed that the expression levels of CFL1, ACTB, CCT4, RDX, TSG101, CD81, FLOT1, PIKFYVE, CTTN and STAT3 in high RAB22A expression group were significantly higher than those in low RAB22A expression group (Figure 5B, 5C). Finally, we showed the correlation between the expression level of RAB22A and exosome genes by scatterplots (Figure 5D–5N).
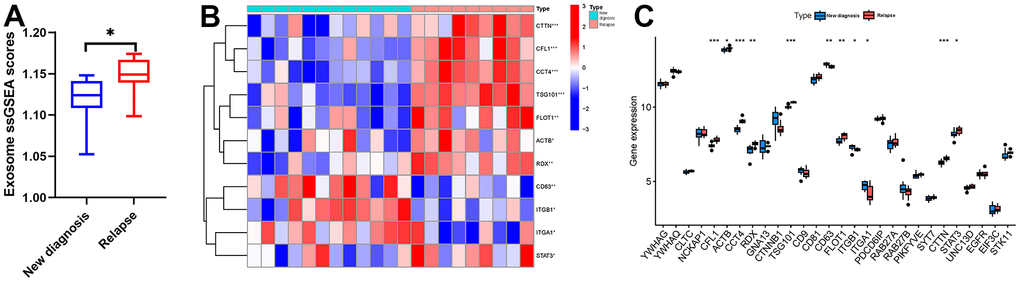
Figure 4. Relationship between MM disease state and exosomes secretion. (A) Exosome scores of MSCs in newly diagnosed and recurrent patients. (B, C) Heatmap and boxplot showed the expression level of exosome-related genes in MSCs of newly diagnosed and relapsed MM patients.
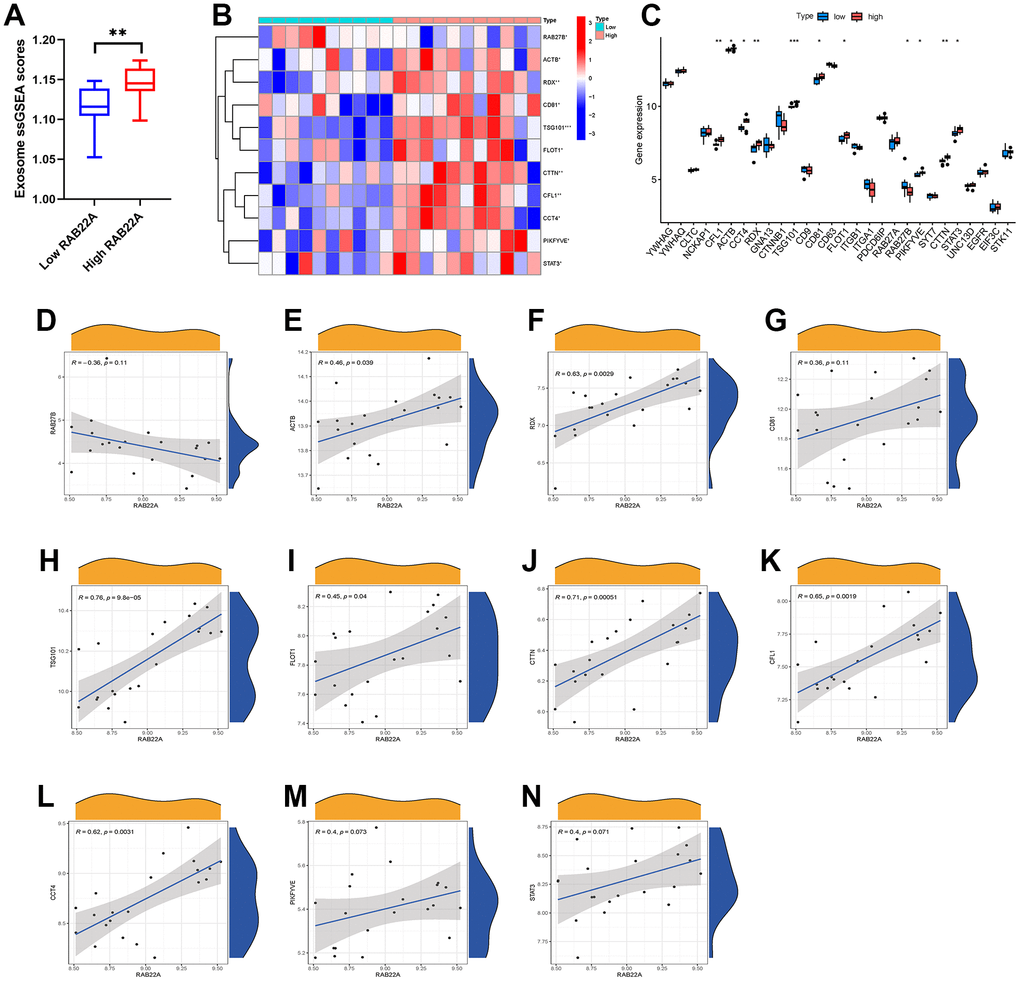
Figure 5. Relationship between RAB22A and exosomes secretion. (A) Exosome scores in high and low RAB22A groups. (B, C) Expression level of exosome-related genes in high and low RAB22A groups. (D–N) Correlation and scatterplots between RAB22A and exosome-related genes.
The correlation between RAB22A and immune cell infiltration and immune checkpoint genes
We explored the infiltration levels of immune cells in different RAB22A expression groups in the samples of GSE4581 data set. The infiltration levels of CD56 dim natural killer cell, gamma delta T cell, myeloid-derived suppressor cell (MDSC), plasmacytoid dendritic cell, type 1 T helper cell and type 2 T helper cell in the high RAB22A expression group were significantly higher than those in the low RAB22A expression group (Figure 6A, 6B). In order to further clarify the correlation between RAB22A and immune cell infiltration, we drew scatterplots. The results showed that monocyte, NK cells resting, eosinophils, T cells regulatory (Treg) and T cells CD4 memory activated were positively correlated with RAB22A. While plasma cells and T cells gamma delta were negatively correlated with RAB22A (Figure 6C–6J).
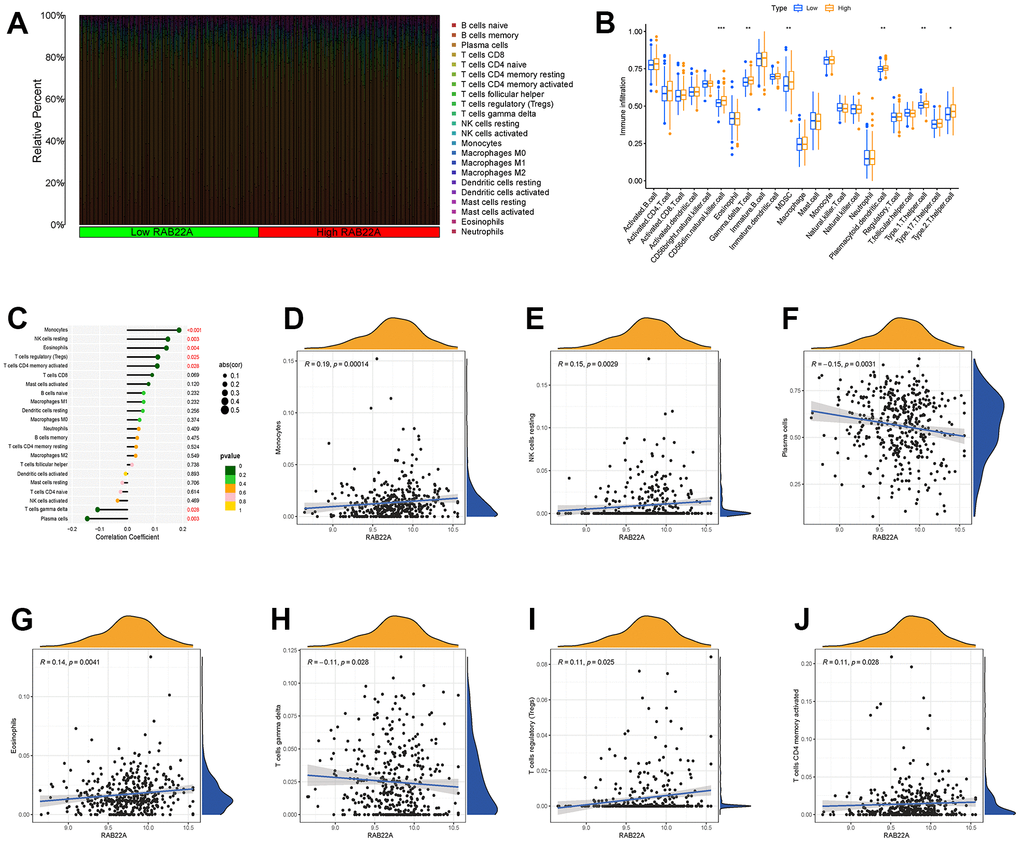
Figure 6. Relationship between RAB22A and immune cell infiltration. (A, B) Infiltration level of immune cells in high and low RAB22A groups. (C) Correlation between RAB22A and infiltration level of immune cells. (D–J) Scatterplots of RAB22A and immune cell infiltration.
In addition, we selected 25 immune checkpoint genes and analyzed the expression levels of these genes in high and low RAB22A expression groups. The results showed that the expression levels of BTNL3, BTNL9, C10orf54, CD209, CD274, CD47, CD80, CTLA4, LAG3, PVR, TDO2, TNFRSF14 and TNFSF14 in the high RAB22A group were lower than those in the low RAB22A group, while CD276, HLA-B, HLA-DMA, HLA-DRA, SIRPA and TNFRSF4 were higher in the high RAB22A group (Figure 7A, 7B).
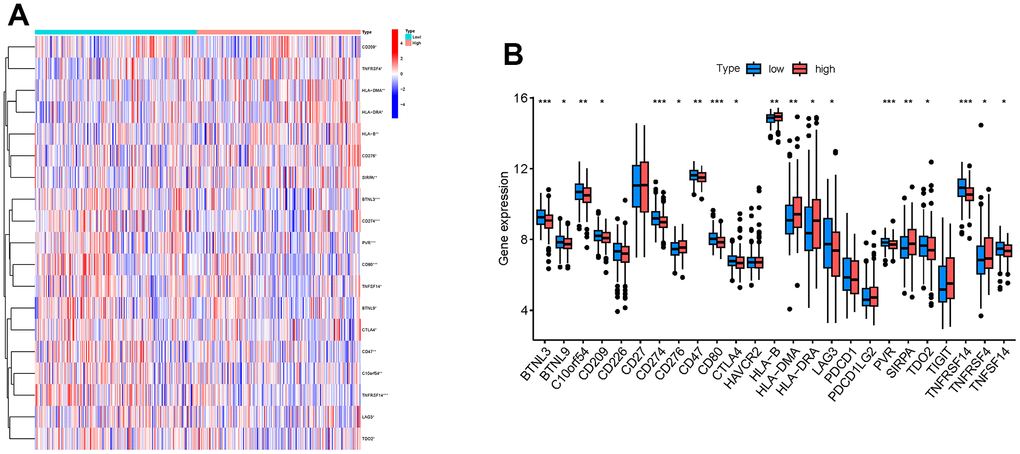
Figure 7. Relationship between RAB22A and immune checkpoint genes. (A) Heatmap showed the gene expression level of immune checkpoint genes in high and low RAB22A groups. (B) Boxplot showed the gene expression level of immune checkpoint genes in high and low RAB22A groups.
RAB22A was associated with m6A RNA methylation regulators
M6A RNA methylation is the most common internal modification in mammalian mRNA, which plays a key biological role by regulating important cellular processes, especially in cancer, where the modification disorder of m6A methylation is often observed. We analyzed the expression levels of m6A-related genes in different RAB22A expression groups. The results showed that the expression levels of METTL14, VIRMA, RBM15 and FMR1 in high RAB22A expression group were significantly higher than those in low RAB22A expression group (Figure 8A, 8B), and the scatterplots showed that these genes were positively correlated with RAB22A (Figure 8C–8G). In addition, we constructed a nomogram (Figure 8H) based on the m6A methylation genes significantly related to RAB22A, which was used to predict the risk value of overexpression of RAB22A. The calibration curve showed that the nomogram had good predictive ability (Figure 8I).
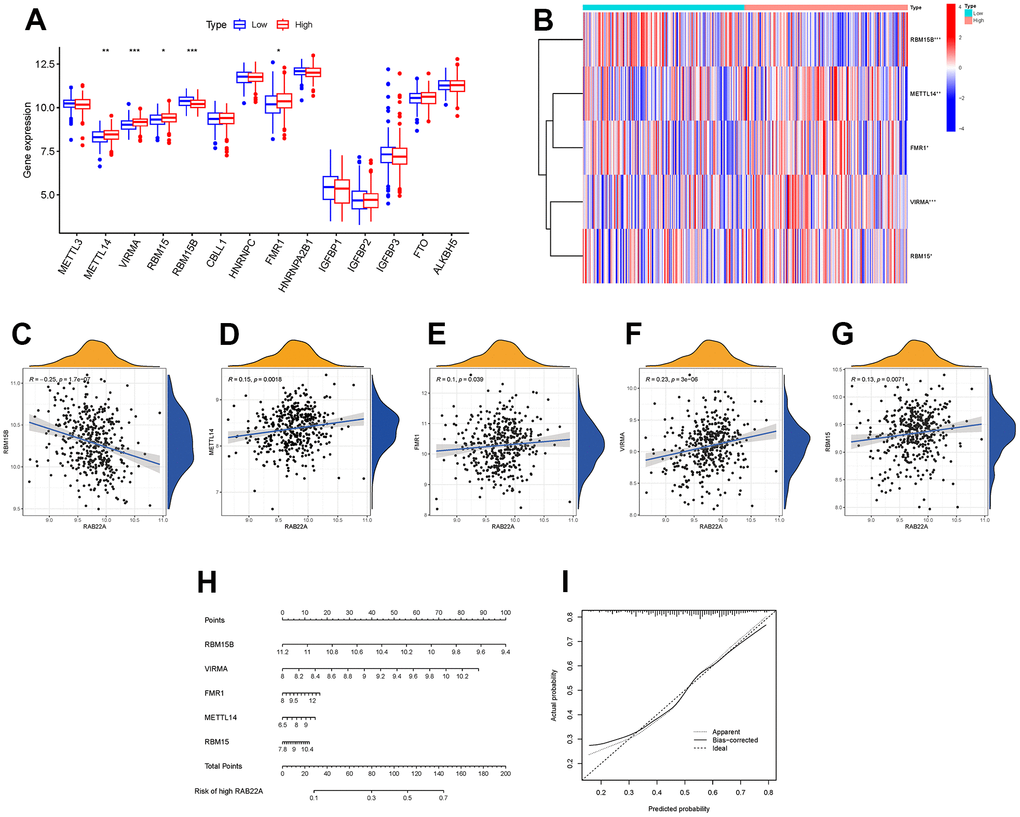
Figure 8. Relationship between RAB22A and m6A methylation. (A, B) Expression level of m6A methylation-related genes in different RAB22A groups. (C–G) Correlation and scatterplots between RAB22A and m6A methylation-related genes. (H, I) Nomogram and calibration curve of the risk of expressing RAB22A.
Drug sensitivity testing
We screened sensitive drugs according to the difference of half maximum inhibition concentration (IC50) between high and low RAB22A expression groups. After consulting the literature, we focused on the drugs that have been used or have prospects in MM. It was worth noting that the IC50 values of doxorubicin, bcl-2 inhibitor (Navitoclax) and cisplatin in low RAB22A expression group were significantly lower than those in high RAB22A expression group (Figure 9A–9C).
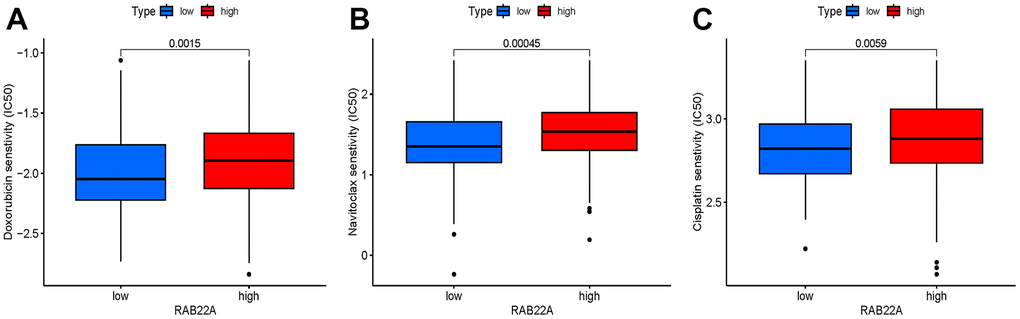
Figure 9. (A–C) Drugs with significantly different IC50 values between high and low RAB22A groups.
The effect of RAB22A on exosome secretion and cell proliferation
We collected the clinical information of 71 normal controls and 30 newly diagnosed MM patients. After 1: 1 propensity score matching (PSM) was performed to eliminate the bias caused by sex and age, 25 normal controls and 25 newly diagnosed MM patients were included for follow-up analysis (Supplementary Table 4). We used western blot and qRT-PCR to verify the difference in the expression level of RAB22A. The results showed that the expression level of RAB22A in MM was significantly higher than that in normal people (Figure 10A, 10B, 10E), and the expression level in MM-BMSC was significantly higher than that in Normal-BMSC (N-BMSC) (Figure 10C, 10D, 10F). Then, we down-regulated RAB22A expression in U266, RPMI 8226 cell lines and MM-MSCs by lentivirus transfection. After down-regulating RAB22A in MM cell line, the cell proliferation was significantly decreased (Figure 10G). We extracted the exosomes of MSCs from the control group and the si-RAB22A group, identified them by exosome markers (Figure 11A), and co-cultured the exosomes from MSCs with MM cell lines. Immunofluorescence staining results showed that the secretion of exosomes decreased after inhibiting the expression of RAB22A in MSCs (Figure 11C). The results of CCK-8 assay showed that compared with the exosomes of MSCs that down-regulated RAB22A, the exosomes of MSCs in control group significantly promoted the proliferation of MM cells (Figure 11B).
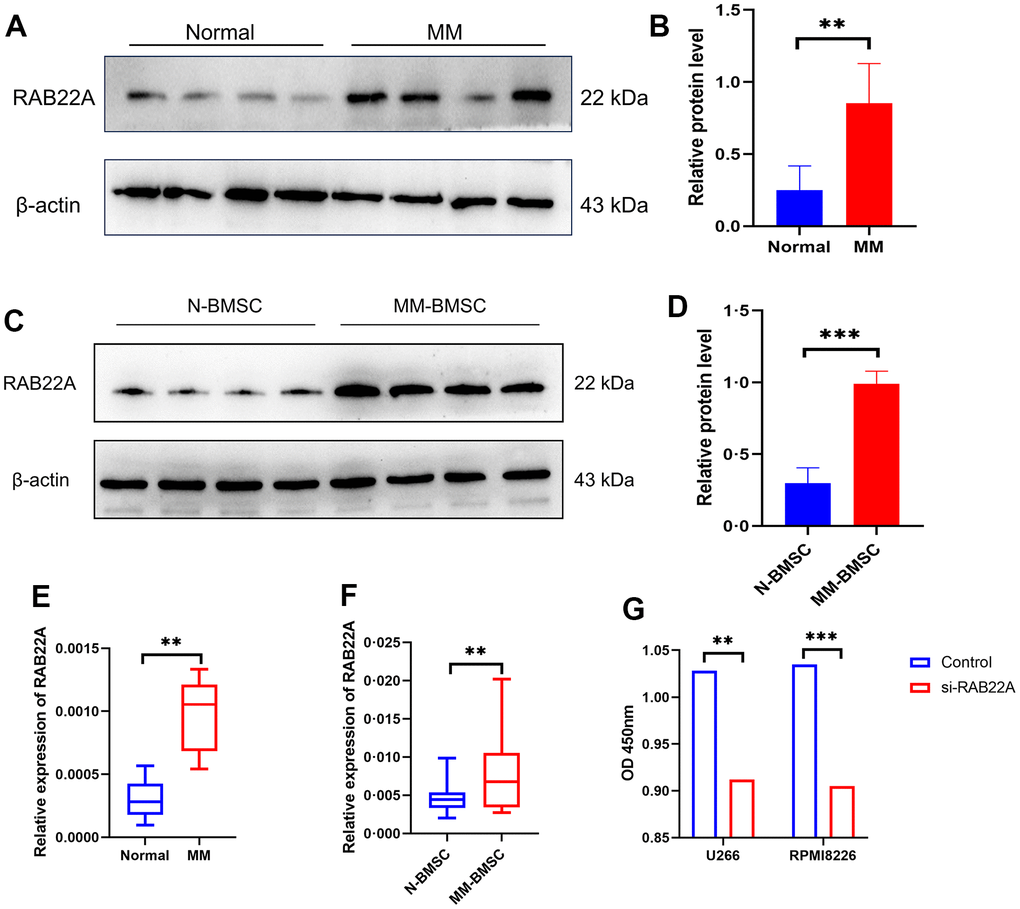
Figure 10. To verify the expression level of RAB22A in clinical samples and cell lines. (A–D) Western blotting results showed that the expression level of RAB22A protein in MM patients was significantly higher than that in normal people. (E, F) qRT-PCR results showed that the expression level of RAB22 gene in MM patients was significantly higher than that in normal people. (G) CCK-8 assay showed that the OD value decreased after down-regulating the expression of RAB22A in MM cell lines.
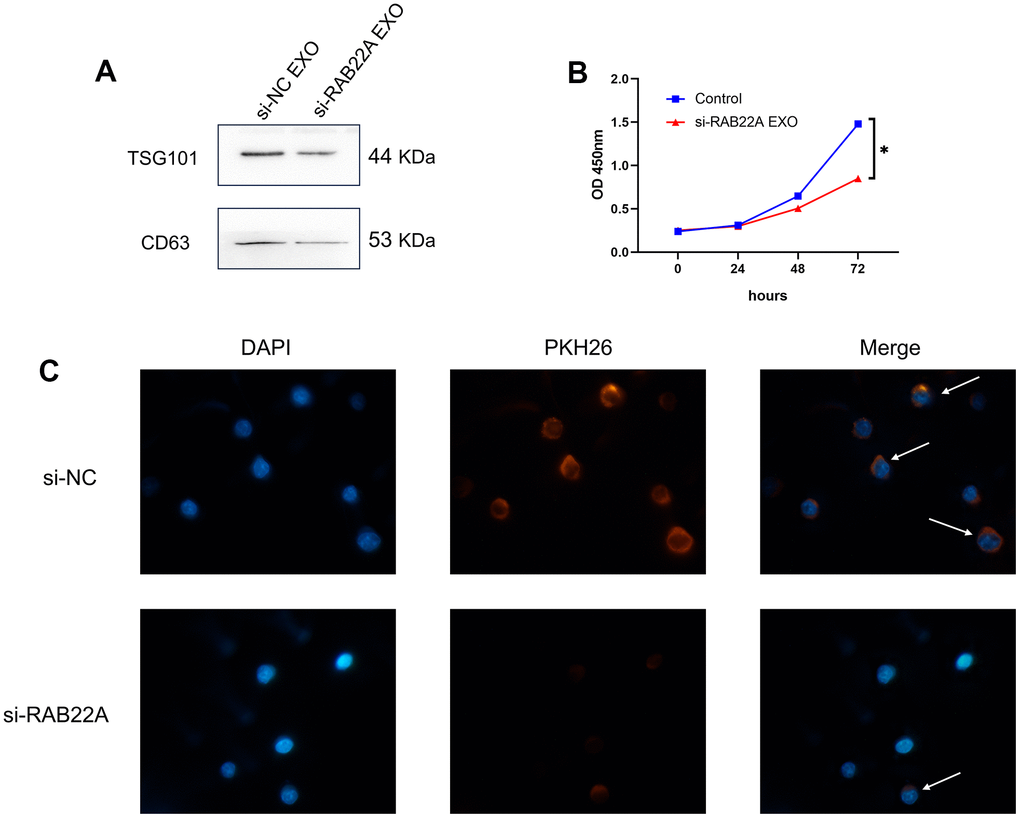
Figure 11. Effects of regulating RAB22A on exosomes secretion and cell proliferation. (A) Verification of exosomes by western blotting. (B) The exosomes of MSCs in control group and down-regulated RAB22A group were co-cultured with MM cell line, and the OD values of MM cells at different time points were detected by CCK-8 method. (C) The exosomes of control group and down-regulated RAB22A group were collected and stained with PKH26. The stained exosomes were co-cultured with MM cells, and the endocytosis of exosomes was observed under fluorescence microscope.
Discussion
Multiple myeloma is a hematological tumor caused by malignant clonal proliferation of bone marrow plasma cells, which is largely caused by the imbalance of oncogenes and abnormal immune system [26]. MM developed from precancerous disease MGUS, and gradually developed into SMM and MM at the rate of 1% per year [27]. RAB22A belongs to the RAS oncogene family, and its abnormal overexpression is involved in the occurrence and progress of many cancers [15, 17]. The significance and mechanism of RAB22A expression in MM are not clear. This study found that RAB22A was significantly overexpressed in SMM and MM, which may lead to the disease progression of MM by regulating exosome secretion, EMT and immune function.
Although the initiation of MM is mainly related to the abnormal activation of key genes and pathways, the support of bone marrow microenvironment is also an important factor leading to the malignant progress of plasma cells. The seed and soil theory in cancer also proves the importance of the interaction between tumor cells and microenvironment for tumor progression and drug resistance. Abnormal bone marrow microenvironment provides protection for tumor cells, which makes drug-resistant tumor cells lurk in it, forming minimal residual disease, and then becomes the source of tumor recurrence and drug resistance. MSCs are one of the important contributing factors in bone marrow microenvironment, and they are a kind of progenitor cells with self-renewal ability and multi-lineage differentiation potential, which can be differentiated into adipocytes, endothelial cells, osteoblasts and fibroblasts [28]. Although MM cells interact with many bone marrow cells, it has been reported that BM-MSCs are the extremely key components to promote the growth, proliferation and drug resistance of MM cells [29]. Cell-to-cell communication between MSCs and MM is necessary for the progress of MM. The invasion of MM cells into bone matrix changed the expression profile of miRNAs in MSCs, which further affected the osteogenic differentiation direction of MSCs, so that osteoclasts were activated and osteoblasts were inhibited, resulting in osteolytic lesions [30–32]. Exosomes are a subtype of membrane-bound extracellular vesicles released by various cells, with a diameter of about 30-150nm. They are rich in mRNAs, miRNAs, proteins and lipids, and can be used for substance exchange and signal transmission between cells [33]. The number of exosomes secreted by tumor cells is much higher than that of normal cells, and the amount of exosomes is related to the malignant degree of tumors. Notably, the gene data carried by RNA directly reflects the protein expression and biological activity of the target cell [34]. The content of miRNAs in exosomes secreted by MM-MSCs is different from that secreted by normal MSC, which leads to the proliferation of MM cells and promotes the disease progression [35]. Another study found that compared with the exosomes of normal MSCs, the expression of miR-483-5p in the exosomes of MM-MSCs was significantly increased, which promoted the proliferation of MM cells and reduced apoptosis by acting on the downstream target TIMP2 [36]. Previous studies have proved that exosome-mediated intercellular communication promotes the malignant progress of MM, but the factors that regulate the exosome secretion of MSCs are not clear. In this study, we co-cultured the exosomes of MM-MSCs from the control group and si-RAB22A group with MM cells. The results of CCK-8 assay showed that the exosomes of MM-MSCs from si-RAB22A group reduced the proliferation of MM. Therefore, it is very important to further explore the genes that affect the secretion of exosomes for the study of drug resistance mechanism and the development of drug targets.
RAB22A is highly expressed in colorectal cancer, hepatocellular carcinoma and breast cancer, and participates in extracellular vesicle secretion, EMT and immune regulation. RAB22A transports the contained substance to the right place by participating in the formation of early endosomes and controlling the formation, movement and fusion of vesicles [37]. In one study [16], RAB22A promoted the formation of extracellular vesicles containing STING, so that STING was transported to the tumor microenvironment. According to literature review, there is no research on the effect of RAB22A on the exosome secretion of MSCs in MM at present, which urges us to pay attention to the effect of RAB22A on the exosome secretion and intercellular communication. In this study, we analyzed the difference of gene expression profile of MSCs between newly diagnosed and relapsed MM patients, and found that the expression level of RAB22A in relapsed patients was significantly higher than that in newly diagnosed patients, and it was related to exosome-related genes and exosome scores. This may suggest that the overexpression of RAB22A promotes the secretion of exosomes in MSCs, which led to the recurrence of MM. Therefore, we further regulated the expression level of RAB22A in MSCs. The results showed that the secretion of exosomes decreased after inhibiting the expression of RAB22A of MM-MSCs, and compared with the exosomes derived from MM-MSCs in the control group, the proliferation of MM cells was significantly inhibited.
EMT is one of the characteristics of solid tumor metastasis, and EMT phenotype also exists in many hematological tumors. A study found that S100A4 was associated with the low survival rate of pancreatic cancer, and TGFβ1 promoted EMT and increased invasiveness by up-regulating S100A4 expression level [38]. In addition, CXCR4 was highly expressed in MM patients with extramedullary invasion, which promoted cell spread and metastasis by inducing EMT phenotype [39]. Another study found that the expression of NSD2 was significantly higher in MM cells with t (4,14) chromosome translocation, and the expression of EMT-related genes was increased by up-regulating transcription factor TWIST1, thus promoting the migration and spread of MM cells [40]. Previous studies have found that RAB22A participates in EMT process in melanoma and colorectal cancer, leading to malignant development of tumor [17, 18]. We analyzed the EMT-related genes that were significantly differentially expressed between the high and low RAB22A expression groups, and calculated the EMT scores of the two groups by “PCA” algorithm. Compared with the low RAB22A expression group, the expression levels of EMT-related genes MYBL2, ITGB1, CDK5, CBR1, EPCAM, S100A4, FOXM1, TIMP1, BHLHE40 and AHR in the high RAB22A expression group were significantly higher, and they had higher EMT scores.
The tumor microenvironment is mainly composed of tumor cells, immune cells, stromal cells and non-cellular components. The components in the tumor microenvironment interact with tumor cells, affecting the occurrence, curative effect and prognosis of tumors. MDSCs are a group of heterogeneous immature myeloid cells with strong immunosuppressive function. The increase of MDSCs leads to poor efficacy of immunotherapy, poor prognosis and disease progression. The expression of MDSCs is increased in hematological tumors, which promotes immune tolerance by inhibiting T cell proliferation. In AML [41], MUC1 induced an increase in the expression of c-myc in extracellular vesicles, which acted on the cyclin of MDSCs, thus promoting the proliferation of MDSCs. In MM, MDSCs promoted the proliferation of MM cells, and played an immunosuppressive role by inhibiting T cell function and Treg cells amplification [42]. Treg cells are a subset of T cells with significant immunosuppression, characterized by the expression of Foxp3, CD25 and CD4, which are related to the disease progression and poor prognosis of multiple myeloma [43, 44], lung cancer [45], hepatocellular carcinoma [46] and breast cancer [47, 48]. Treg cells inhibits the immune response of the body, leading to immune escape of tumor cells [49, 50]. Therefore, Treg cells are considered as the key target of cancer immunotherapy [51]. In our study, the expression level of RAB22A was positively correlated with Treg cells infiltration level, and the infiltration degree of MDSCs in high RAB22A expression group was higher than that in low RAB22A expression group. These results suggested that RAB22A played an important role in regulating immune cell infiltration of MM.
Although this study explored the expression level and biological function of RAB22A in MM through bioinformatics analysis and preliminary verification of clinical samples, there are still some shortcomings. First of all, the transcriptome data of this study came from public databases, which needs to be further verified in clinical samples in the future. Secondly, this study lacked follow-up data and specific treatment information, such as treatment response, survival time, radiotherapy and chemotherapy information, which were very important for the construction of prognosis model. We will further follow up and analyze the prognosis in the clinical samples of our center. Finally, this study did not further analyze the complete transcriptome of exosomes secreted by MSCs. By analyzing the difference of substances transported by exosomes, we will further explore the downstream effector molecules of RAB22A, which is expected to develop new targets in MM. This is what we will continue to explore in the future.
In a word, we confirmed that RAB22A was highly expressed in MM and SMM. RAB22A is related to EMT, exosome secretion, immune cell infiltration and m6A modification. We verified in vitro that RAB22A regulated the proliferation of MM by affecting the secretion of exosomes. RAB22A may be a potential therapeutic target to improve the prognosis of MM.
Author Contributions
BJ.F. participated in the conception, design and manuscript writing of the project. L.W. did the statistical analysis and some experiments. JS. W. designed and revised the manuscript. All authors contributed to the article and approved the final version.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Statement and Consent
The research design was approved by the Institutional Review Committee of the Affiliated Hospital of Guizhou Medical University (approval number is NO.2304534). All methods were carried out in accordance with relevant guidelines and regulations. All participants in the study provided informed consent.
Funding
Financial support was obtained from the National Natural Science Foundation of China (NSFC, No. 82170168 and No. 81960032).
References
- 1. Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011; 364:1046–60. https://doi.org/10.1056/NEJMra1011442 [PubMed]
- 2. Weiss BM, Abadie J, Verma P, Howard RS, Kuehl WM. A monoclonal gammopathy precedes multiple myeloma in most patients. Blood. 2009; 113:5418–22. https://doi.org/10.1182/blood-2008-12-195008 [PubMed]
- 3. Landgren O, Kyle RA, Pfeiffer RM, Katzmann JA, Caporaso NE, Hayes RB, Dispenzieri A, Kumar S, Clark RJ, Baris D, Hoover R, Rajkumar SV. Monoclonal gammopathy of undetermined significance (MGUS) consistently precedes multiple myeloma: a prospective study. Blood. 2009; 113:5412–7. https://doi.org/10.1182/blood-2008-12-194241 [PubMed]
- 4. Rajkumar SV. Multiple myeloma: 2022 update on diagnosis, risk stratification, and management. Am J Hematol. 2022; 97:1086–107. https://doi.org/10.1002/ajh.26590 [PubMed]
- 5. Zhao JH, Xu QL, Ma S, Li CY, Zhang HC, Zhao LJ, Zhang ZY. Recent advance of small-molecule drugs for clinical treatment of multiple myeloma. Eur J Med Chem. 2023; 257:115492. https://doi.org/10.1016/j.ejmech.2023.115492 [PubMed]
- 6. Hemminki K, Försti A, Hansson M. Incidence, mortality and survival in multiple myeloma compared to other hematopoietic neoplasms in Sweden up to year 2016. Sci Rep. 2021; 11:17272. https://doi.org/10.1038/s41598-021-96804-8 [PubMed]
- 7. Usmani SZ, Quach H, Mateos MV, Landgren O, Leleu X, Siegel D, Weisel K, Shu X, Li C, Dimopoulos M. Final analysis of carfilzomib, dexamethasone, and daratumumab vs carfilzomib and dexamethasone in the CANDOR study. Blood Adv. 2023; 7:3739–48. https://doi.org/10.1182/bloodadvances.2023010026 [PubMed]
- 8. Gentile M, Vigna E, Palmieri S, Galli M, Derudas D, Mina R, Della Pepa R, Zambello R, Martino EA, Bruzzese A, Mangiacavalli S, Zamagni E, Califano C, et al. Elotuzumab plus pomalidomide and dexamethasone in relapsed/refractory multiple myeloma: a multicenter, retrospective, real-world experience with 200 cases outside of controlled clinical trials. Haematologica. 2024; 109:245–55. https://doi.org/10.3324/haematol.2023.283251 [PubMed]
- 9. Pouleau B, Estoppey C, Suere P, Nallet E, Laurendon A, Monney T, Pais Ferreira D, Drake A, Carretero-Iglesia L, Macoin J, Berret J, Pihlgren M, Doucey MA, et al. Preclinical characterization of ISB 1342, a CD38 × CD3 T-cell engager for relapsed/refractory multiple myeloma. Blood. 2023; 142:260–73. https://doi.org/10.1182/blood.2022019451 [PubMed]
- 10. He M, Shen L, Jiang C, Gao G, Wang K, Jiao Y, Sun L, Cui Y, Ke Z, Yang Z. Rab22a is a novel prognostic marker for cell progression in breast cancer. Int J Mol Med. 2020; 45:1037–46. https://doi.org/10.3892/ijmm.2020.4486 [PubMed]
- 11. Yin Y, Zhang B, Wang W, Fei B, Quan C, Zhang J, Song M, Bian Z, Wang Q, Ni S, Hu Y, Mao Y, Zhou L, et al. miR-204-5p inhibits proliferation and invasion and enhances chemotherapeutic sensitivity of colorectal cancer cells by downregulating RAB22A. Clin Cancer Res. 2014; 20:6187–99. https://doi.org/10.1158/1078-0432.CCR-14-1030 [PubMed]
- 12. Liao D, Zhong L, Yin J, Zeng C, Wang X, Huang X, Chen J, Zhang H, Zhang R, Guan XY, Shuai X, Sui J, Gao S, et al. Chromosomal translocation-derived aberrant Rab22a drives metastasis of osteosarcoma. Nat Cell Biol. 2020; 22:868–81. https://doi.org/10.1038/s41556-020-0522-z [PubMed]
- 13. Wen F, Meng F, Li X, Li Q, Liu J, Zhang R, Zhao Y, Zhang Y, Wang X, Ju S, Cui Y, Lu Z. Characterization of prognostic value and immunological roles of RAB22A in hepatocellular carcinoma. Front Immunol. 2023; 14:1086342. https://doi.org/10.3389/fimmu.2023.1086342 [PubMed]
- 14. Stenmark H. Rab GTPases as coordinators of vesicle traffic. Nat Rev Mol Cell Biol. 2009; 10:513–25. https://doi.org/10.1038/nrm2728 [PubMed]
- 15. Sun L, He M, Xu N, Xu DH, Ben-David Y, Yang ZY, Li YJ. Regulation of RAB22A by mir-193b inhibits breast cancer growth and metastasis mediated by exosomes. Int J Oncol. 2018; 53:2705–14. https://doi.org/10.3892/ijo.2018.4571 [PubMed]
- 16. Gao Y, Zheng X, Chang B, Lin Y, Huang X, Wang W, Ding S, Zhan W, Wang S, Xiao B, Huo L, Yu Y, Chen Y, et al. Intercellular transfer of activated STING triggered by RAB22A-mediated non-canonical autophagy promotes antitumor immunity. Cell Res. 2022; 32:1086–104. https://doi.org/10.1038/s41422-022-00731-w [PubMed]
- 17. Kong W, Li H, Xie L, Cui G, Gu W, Zhang H, Ma W, Zhou Y. LncRNA MCF2L-AS1 aggravates the malignant development of colorectal cancer via targeting miR-105-5p/RAB22A axis. BMC Cancer. 2021; 21:1069. https://doi.org/10.1186/s12885-021-08668-w [PubMed]
- 18. Yu H, Yang W. MiR-211 is epigenetically regulated by DNMT1 mediated methylation and inhibits EMT of melanoma cells by targeting RAB22A. Biochem Biophys Res Commun. 2016; 476:400–5. https://doi.org/10.1016/j.bbrc.2016.05.133 [PubMed]
- 19. Cebrian I, Croce C, Guerrero NA, Blanchard N, Mayorga LS. Rab22a controls MHC-I intracellular trafficking and antigen cross-presentation by dendritic cells. EMBO Rep. 2016; 17:1753–65. https://doi.org/10.15252/embr.201642358 [PubMed]
- 20. Cao B, Dai W, Ma S, Wang Q, Lan M, Luo H, Chen T, Yang X, Zhu G, Li Q, Lang J. An EV-Associated Gene Signature Correlates with Hypoxic Microenvironment and Predicts Recurrence in Lung Adenocarcinoma. Mol Ther Nucleic Acids. 2019; 17:879–90. https://doi.org/10.1016/j.omtn.2019.07.021 [PubMed]
- 21. McAndrews KM, Kalluri R. Mechanisms associated with biogenesis of exosomes in cancer. Mol Cancer. 2019; 18:52. https://doi.org/10.1186/s12943-019-0963-9 [PubMed]
- 22. Wu J, Zeng D, Zhi S, Ye Z, Qiu W, Huang N, Sun L, Wang C, Wu Z, Bin J, Liao Y, Shi M, Liao W. Single-cell analysis of a tumor-derived exosome signature correlates with prognosis and immunotherapy response. J Transl Med. 2021; 19:381. https://doi.org/10.1186/s12967-021-03053-4 [PubMed]
- 23. Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020; 367:eaau6977. https://doi.org/10.1126/science.aau6977 [PubMed]
- 24. Borella G, Da Ros A, Borile G, Porcù E, Tregnago C, Benetton M, Marchetti A, Bisio V, Montini B, Michielotto B, Cani A, Leszl A, Campodoni E, et al. Targeting the plasticity of mesenchymal stromal cells to reroute the course of acute myeloid leukemia. Blood. 2021; 138:557–70. https://doi.org/10.1182/blood.2020009845 [PubMed]
- 25. Jiang D, Wu X, Sun X, Tan W, Dai X, Xie Y, Du A, Zhao Q. Bone mesenchymal stem cell-derived exosomal microRNA-7-5p inhibits progression of acute myeloid leukemia by targeting OSBPL11. J Nanobiotechnology. 2022; 20:29. https://doi.org/10.1186/s12951-021-01206-7 [PubMed]
- 26. Caraccio C, Krishna S, Phillips DJ, Schürch CM. Bispecific Antibodies for Multiple Myeloma: A Review of Targets, Drugs, Clinical Trials, and Future Directions. Front Immunol. 2020; 11:501. https://doi.org/10.3389/fimmu.2020.00501 [PubMed]
- 27. Kyle RA, Durie BGM, Rajkumar SV, Landgren O, Blade J, Merlini G, Kröger N, Einsele H, Vesole DH, Dimopoulos M, San Miguel J, Avet-Loiseau H, Hajek R, et al, and International Myeloma Working Group. Monoclonal gammopathy of undetermined significance (MGUS) and smoldering (asymptomatic) multiple myeloma: IMWG consensus perspectives risk factors for progression and guidelines for monitoring and management. Leukemia. 2010; 24:1121–7. https://doi.org/10.1038/leu.2010.60 [PubMed]
- 28. Xia C, Wang T, Cheng H, Dong Y, Weng Q, Sun G, Zhou P, Wang K, Liu X, Geng Y, Ma S, Hao S, Xu L, et al. Mesenchymal stem cells suppress leukemia via macrophage-mediated functional restoration of bone marrow microenvironment. Leukemia. 2020; 34:2375–83. https://doi.org/10.1038/s41375-020-0775-3 [PubMed]
- 29. Xu S, De Veirman K, De Becker A, Vanderkerken K, Van Riet I. Mesenchymal stem cells in multiple myeloma: a therapeutical tool or target? Leukemia. 2018; 32:1500–14. https://doi.org/10.1038/s41375-018-0061-9 [PubMed]
- 30. Terpos E, Ntanasis-Stathopoulos I, Gavriatopoulou M, Dimopoulos MA. Pathogenesis of bone disease in multiple myeloma: from bench to bedside. Blood Cancer J. 2018; 8:7. https://doi.org/10.1038/s41408-017-0037-4 [PubMed]
- 31. Berenstein R, Nogai A, Waechter M, Blau O, Kuehnel A, Schmidt-Hieber M, Kunitz A, Pezzutto A, Dörken B, Blau IW. Multiple myeloma cells modify VEGF/IL-6 levels and osteogenic potential of bone marrow stromal cells via Notch/miR-223. Mol Carcinog. 2016; 55:1927–39. https://doi.org/10.1002/mc.22440 [PubMed]
- 32. Berenstein R, Blau O, Nogai A, Waechter M, Slonova E, Schmidt-Hieber M, Kunitz A, Pezzutto A, Doerken B, Blau IW. Multiple myeloma cells alter the senescence phenotype of bone marrow mesenchymal stromal cells under participation of the DLK1-DIO3 genomic region. BMC Cancer. 2015; 15:68. https://doi.org/10.1186/s12885-015-1078-3 [PubMed]
- 33. Paskeh MDA, Entezari M, Mirzaei S, Zabolian A, Saleki H, Naghdi MJ, Sabet S, Khoshbakht MA, Hashemi M, Hushmandi K, Sethi G, Zarrabi A, Kumar AP, et al. Emerging role of exosomes in cancer progression and tumor microenvironment remodeling. J Hematol Oncol. 2022; 15:83. https://doi.org/10.1186/s13045-022-01305-4 [PubMed]
- 34. Xiao Q, Lin C, Peng M, Ren J, Jing Y, Lei L, Tao Y, Huang J, Yang J, Sun M, Wu J, Yang Z, Yang Z, Zhang L. Circulating plasma exosomal long non-coding RNAs LINC00265, LINC00467, UCA1, and SNHG1 as biomarkers for diagnosis and treatment monitoring of acute myeloid leukemia. Front Oncol. 2022; 12:1033143. https://doi.org/10.3389/fonc.2022.1033143 [PubMed]
- 35. Roccaro AM, Sacco A, Maiso P, Azab AK, Tai YT, Reagan M, Azab F, Flores LM, Campigotto F, Weller E, Anderson KC, Scadden DT, Ghobrial IM. BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. J Clin Invest. 2013; 123:1542–55. https://doi.org/10.1172/JCI66517 [PubMed]
- 36. Gu J, Wang M, Wang X, Li J, Liu H, Lin Z, Yang X, Zhang X, Liu H. Exosomal miR-483-5p in Bone Marrow Mesenchymal Stem Cells Promotes Malignant Progression of Multiple Myeloma by Targeting TIMP2. Front Cell Dev Biol. 2022; 10:862524. https://doi.org/10.3389/fcell.2022.862524 [PubMed]
- 37. Magadán JG, Barbieri MA, Mesa R, Stahl PD, Mayorga LS. Rab22a regulates the sorting of transferrin to recycling endosomes. Mol Cell Biol. 2006; 26:2595–614. https://doi.org/10.1128/MCB.26.7.2595-2614.2006 [PubMed]
- 38. Low RRJ, Fung KY, Gao H, Preaudet A, Dagley LF, Yousef J, Lee B, Emery-Corbin SJ, Nguyen PM, Larsen RH, Kershaw NJ, Burgess AW, Gibbs P, et al. S100 family proteins are linked to organoid morphology and EMT in pancreatic cancer. Cell Death Differ. 2023; 30:1155–65. https://doi.org/10.1038/s41418-023-01126-z [PubMed]
- 39. Roccaro AM, Mishima Y, Sacco A, Moschetta M, Tai YT, Shi J, Zhang Y, Reagan MR, Huynh D, Kawano Y, Sahin I, Chiarini M, Manier S, et al. CXCR4 Regulates Extra-Medullary Myeloma through Epithelial-Mesenchymal-Transition-like Transcriptional Activation. Cell Rep. 2015; 12:622–35. https://doi.org/10.1016/j.celrep.2015.06.059 [PubMed]
- 40. Cheong CM, Mrozik KM, Hewett DR, Bell E, Panagopoulos V, Noll JE, Licht JD, Gronthos S, Zannettino ACW, Vandyke K. Twist-1 is upregulated by NSD2 and contributes to tumour dissemination and an epithelial-mesenchymal transition-like gene expression signature in t(4;14)-positive multiple myeloma. Cancer Lett. 2020; 475:99–108. https://doi.org/10.1016/j.canlet.2020.01.040 [PubMed]
- 41. Pyzer AR, Stroopinsky D, Rajabi H, Washington A, Tagde A, Coll M, Fung J, Bryant MP, Cole L, Palmer K, Somaiya P, Karp Leaf R, Nahas M, et al. MUC1-mediated induction of myeloid-derived suppressor cells in patients with acute myeloid leukemia. Blood. 2017; 129:1791–801. https://doi.org/10.1182/blood-2016-07-730614 [PubMed]
- 42. Malek E, de Lima M, Letterio JJ, Kim BG, Finke JH, Driscoll JJ, Giralt SA. Myeloid-derived suppressor cells: The green light for myeloma immune escape. Blood Rev. 2016; 30:341–8. https://doi.org/10.1016/j.blre.2016.04.002 [PubMed]
- 43. Dahlhoff J, Manz H, Steinfatt T, Delgado-Tascon J, Seebacher E, Schneider T, Wilnit A, Mokhtari Z, Tabares P, Böckle D, Rasche L, Martin Kortüm K, Lutz MB, et al. Transient regulatory T-cell targeting triggers immune control of multiple myeloma and prevents disease progression. Leukemia. 2022; 36:790–800. https://doi.org/10.1038/s41375-021-01422-y [PubMed]
- 44. Kawano Y, Zavidij O, Park J, Moschetta M, Kokubun K, Mouhieddine TH, Manier S, Mishima Y, Murakami N, Bustoros M, Pistofidis RS, Reidy M, Shen YJ, et al. Blocking IFNAR1 inhibits multiple myeloma-driven Treg expansion and immunosuppression. J Clin Invest. 2018; 128:2487–99. https://doi.org/10.1172/JCI88169 [PubMed]
- 45. Exposito F, Redrado M, Houry M, Hastings K, Molero-Abraham M, Lozano T, Solorzano JL, Sanz-Ortega J, Adradas V, Amat R, Redin E, Leon S, Legarra N, et al. PTEN Loss Confers Resistance to Anti-PD-1 Therapy in Non-Small Cell Lung Cancer by Increasing Tumor Infiltration of Regulatory T Cells. Cancer Res. 2023; 83:2513–26. https://doi.org/10.1158/0008-5472.CAN-22-3023 [PubMed]
- 46. Zhu AX, Abbas AR, de Galarreta MR, Guan Y, Lu S, Koeppen H, Zhang W, Hsu CH, He AR, Ryoo BY, Yau T, Kaseb AO, Burgoyne AM, et al. Molecular correlates of clinical response and resistance to atezolizumab in combination with bevacizumab in advanced hepatocellular carcinoma. Nat Med. 2022; 28:1599–611. https://doi.org/10.1038/s41591-022-01868-2 [PubMed]
- 47. Lei PJ, Pereira ER, Andersson P, Amoozgar Z, Van Wijnbergen JW, O’Melia MJ, Zhou H, Chatterjee S, Ho WW, Posada JM, Kumar AS, Morita S, Menzel L, et al. Cancer cell plasticity and MHC-II-mediated immune tolerance promote breast cancer metastasis to lymph nodes. J Exp Med. 2023; 220:e20221847. https://doi.org/10.1084/jem.20221847 [PubMed]
- 48. Fattori S, Roux H, Connen E, Robert L, Gorvel L, Le Roy A, Houacine J, Foussat A, Chretien AS, Olive D. Therapeutic Targeting of Tumor-Infiltrating Regulatory T Cells in Breast Cancer. Cancer Res. 2022; 82:3868–79. https://doi.org/10.1158/0008-5472.CAN-22-0654 [PubMed]
- 49. Nishikawa H, Koyama S. Mechanisms of regulatory T cell infiltration in tumors: implications for innovative immune precision therapies. J Immunother Cancer. 2021; 9:e002591. https://doi.org/10.1136/jitc-2021-002591 [PubMed]
- 50. Tanaka A, Sakaguchi S. Regulatory T cells in cancer immunotherapy. Cell Res. 2017; 27:109–18. https://doi.org/10.1038/cr.2016.151 [PubMed]
- 51. van Gulijk M, van Krimpen A, Schetters S, Eterman M, van Elsas M, Mankor J, Klaase L, de Bruijn M, van Nimwegen M, van Tienhoven T, van Ijcken W, Boon L, van der Schoot J, et al. PD-L1 checkpoint blockade promotes regulatory T cell activity that underlies therapy resistance. Sci Immunol. 2023; 8:eabn6173. https://doi.org/10.1126/sciimmunol.abn6173 [PubMed]