Introduction
Dopamine signaling is associated with cognitive performance and physical function during aging [1]. As a neurotransmitter, dopamine is widely present in animals, plants and microorganisms, and it can regulate a variety of physiological functions including learning and memory, motor behavior and reward decision-making behavior of the body [2]. Studies have shown that dopamine plays an important role in Parkinson’s disease (PD) [3]. Since the motor symptoms of PD are caused by the death of dopamine-releasing nerve cells from the substantia nigra pars compacta (SNc), patients with PD cannot secrete dopamine. On the other hand, the reduction or disappearance of the substantia nigra in the brain affects the transport of dopamine in the striatum, which regulates brain movements [4], and accelerates the aging process in elderly individuals. Increasing dopamine secretion either through transgenic or pharmacological strategies can slow aging and age-related diseases [5–7], and is also able to increase the learning ability and task performance in some older adults to younger adult levels [8, 9]. Dopamine content is generally age-dependent [10], and the older is the age, the faster is the clearance of dopamine [11]. This age-related decline in the dopamine system is prevalent in various animal models such as Drosophila, Caenorhabditis elegans (C. elegans), mice, rats, and monkeys, indicating that degeneration of the dopamine system function may have a conservative molecular mechanism [12]. However, very few studies have identified dopamine contents as a key to regulating aging, and investigating the relationship and the mechanism between dopamine and aging is interesting.
Acein, a nonapeptide of sequence H-Pro-Pro-Thr-Thr-Thr-Lys-Phe-Ala-Ala-OH stimulates dopamine release in the brain [13] by binding with high affinity to the striatal bound-membrane angiotensin converting enzyme (ACE) of rodents [13, 14]. Further structure-activity relationship studies revealed that the C-terminal pentapeptide of Acein H-Thr-Lys-Phe-Ala-Ala-OH is the smallest fragment exhibiting full biological activity [14, 15].
In this study we investigated the effect of Acein on lifespan, using the classic C. elegans aging organism model, in addition we identified the gene related to aging process and its relation with dopamine release.
Results
Acein regulates aging by promoting dopamine release
The effect of Acein on C. elegans longevity was investigated at different concentration. Acein showed the best antiaging activity at a 10 nM concentration, extending the mean lifespan by 25.66% and the median lifespan by 20.00% (Figure 1A, 1B). Forty-eight hours after administration of 10 nM Acein, dopamine secretion in C. elegans was increased by a factor of approximately 2 (Figure 1C). In contrast, 10 nM Acein did not affect the growth and development of L1-stage larvae and adults of C. elegans (Figure 1E, 1F). However, 10 nM Acein significantly increased the number of broods and improved the reproductive ability of C. elegans (Figure 1G). Acein was also able to improve the fitness of C. elegans, including body flexion, head swing and pharyngeal pumping frequency (Figure 1H–1J).
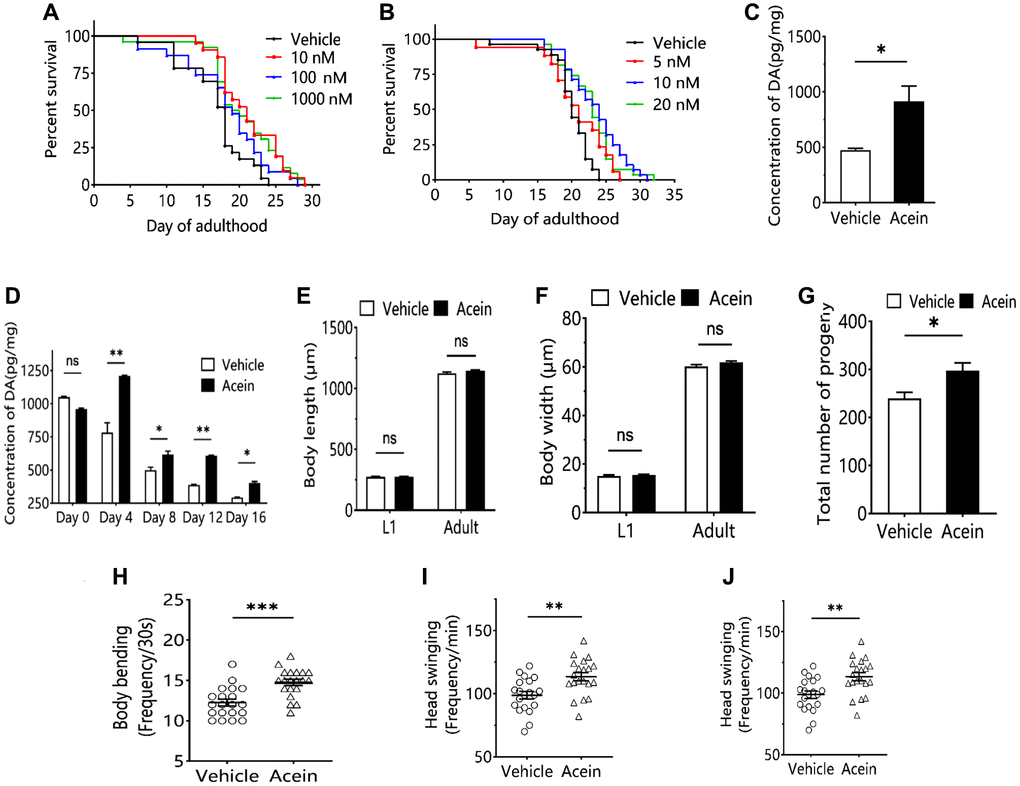
Figure 1. Acein regulates aging by influencing dopamine level. (A) Survival curve of 10 nM, 100 nM and 1000 nM Acein groups. (B) Survival curve of 5 nM, 10 nM and 20 nM Acein groups. (C) Dopamine content in C.elegans after 48 h of 10 nM Acein treatment. (D) Change of dopamine level in C. elegans with age after treatment with 10 nM Acein. (E) C.elegans body length after treatment with 10 nM Acein. (F) C. elegans body width after treatment with 10 nM Acein. (G) C.elegans progeny number after treatment with 10 nM Acein. (H) Body bending detection of C.elegans after treatment with 10 nM Acein. (I) Head swing detection of C.elegans after treatment with 10 nM Acein. (J) Pharyngeal pumping detection of C.elegans. Compared with the vehicle group. nsp > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001.
Transcriptome sequencing revealed clec-126, a key gene for aging regulation in C. elegans by Acein
Transcriptome sequencing of C. elegans tissue samples showed that 98 genes were up-regulated and 26 genes were down-regulated in the 10 nM Acein-treated group (Figure 2A). Further screening was then performed based on HR fold difference and significance level P values (FC ≥ 2 or FC ≤ 0.5 (absolute logFC ≥ 1), P value ≤ 0.05) and good reproducibility between parallel groups. A total of 13 up-regulated genes and 6 down-regulated genes were identified (Figure 2B). qPCR verification showed that Acein could significantly upregulate T02D1.7 and nspf-3 and downregulate the expression of col-41, clec-126 and Y34F4.3. The most significant effect was observed for T02D1.7 on the upside and for clec-126 on the downside (Figure 2C, 2D). Genetic information screening methods indicated that T02D1.7 corresponded to an uncharacterized protein, while the C-type lectin domain protein encoded by clec-126 has carbohydrate binding activity (Supplementary Tables 1, 2).
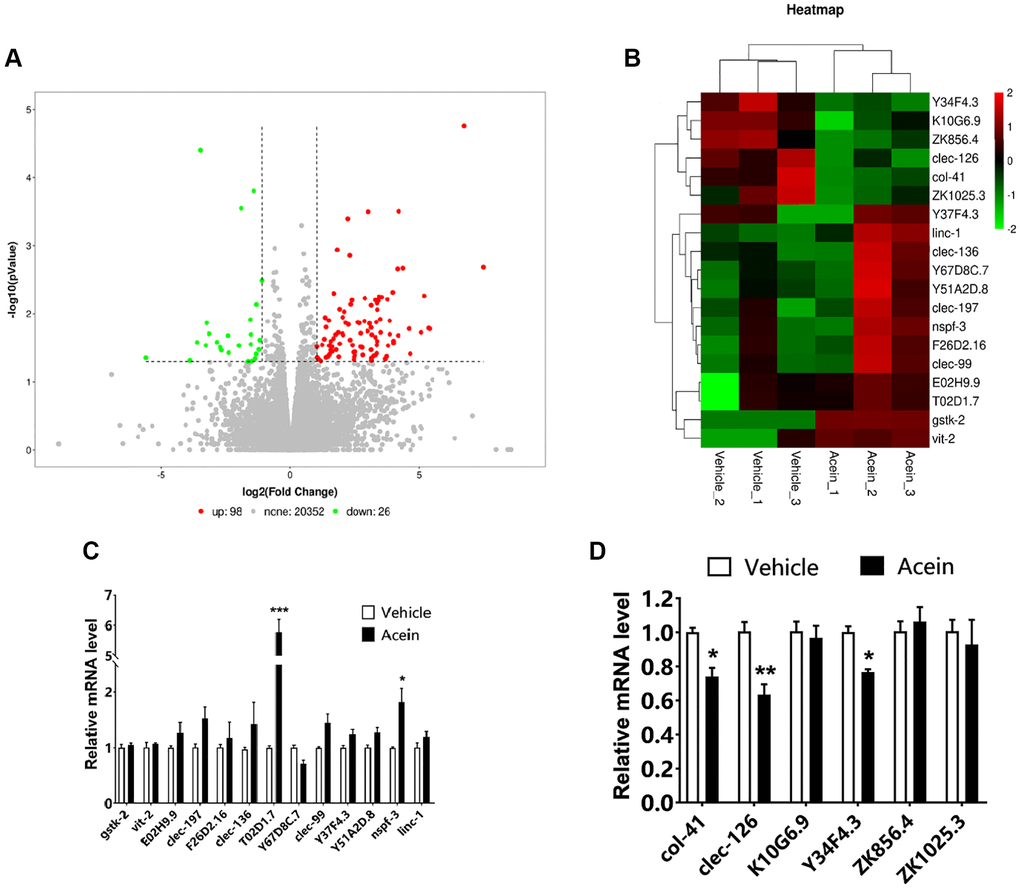
Figure 2. Transcriptome sequencing revealed clec-126, a key gene for the anti-aging activity exerted by Acein. (A) Differential gene scatter diagram, a total of 98 genes were up-regulated (red) and 26 genes were down-regulated (green). (B) Selected differential gene heat map, the screening criteria were FC ≥ 2 or FC ≤ 0.5, P value ≤ 0.05, and the reproducibility of the parallel group was good. (C) qPCR validation results of 13 up-regulated genes. (D) qPCR validation results of 6 down-regulated genes. Compared with the vehicle group, *p < 0.05, **p < 0.01, ***p < 0.001.
Silencing clec-126 can increase dopamine-induced Acein and prolong the lifespan of elderly C. elegans
Low clec-126 expression in clec-126-RNAi C. elegans was verified by qPCR (Figure 3A). Lifespan assays showed that the lifespan of clec-126-RNAi C. elegans was significantly higher, with a maximum increase of 31.19% in mean lifespan and 41.18% in median lifespan (Figure 3B). Subsequently, we investigated whether silencing clec-126 in C. elegans affected dopamine secretion levels. The results showed that silencing clec-126-RNAi could delay the decrease in dopamine content and increase dopamine levels in elderly C. elegans particularly at day 4 (Figure 3C, 3D). Because dopamine may affect the exercise capacity of C. elegans, the frequency of body bending, head swing and pharyngeal pumping was examined. The results showed that clec-126-RNAi did not affect the frequency of body bending, head swing or pharyngeal pumping in C. elegans (Figure 3E–3G). Stress capacity can reflect the body’s resistance to external stress, [16, 17] which is closely related to a healthy lifespan. The results showed that clec-126-RNAi could not significantly affect the survival of C. elegans under heat stress (Figure 3H). With age increasing, the motility of the body gradually decreased, which greatly affected the healthy lifespan and reduced the quality of life of the elderly population, so the head swing and pharyngeal pumping of clec-126-RNAi C. elegans at different ages were measured. The results showed that clec-126-RNAi could significantly slow the decline in head swing ability and pharyngeal pump ability of C. elegans (Figure 3I, 3J).
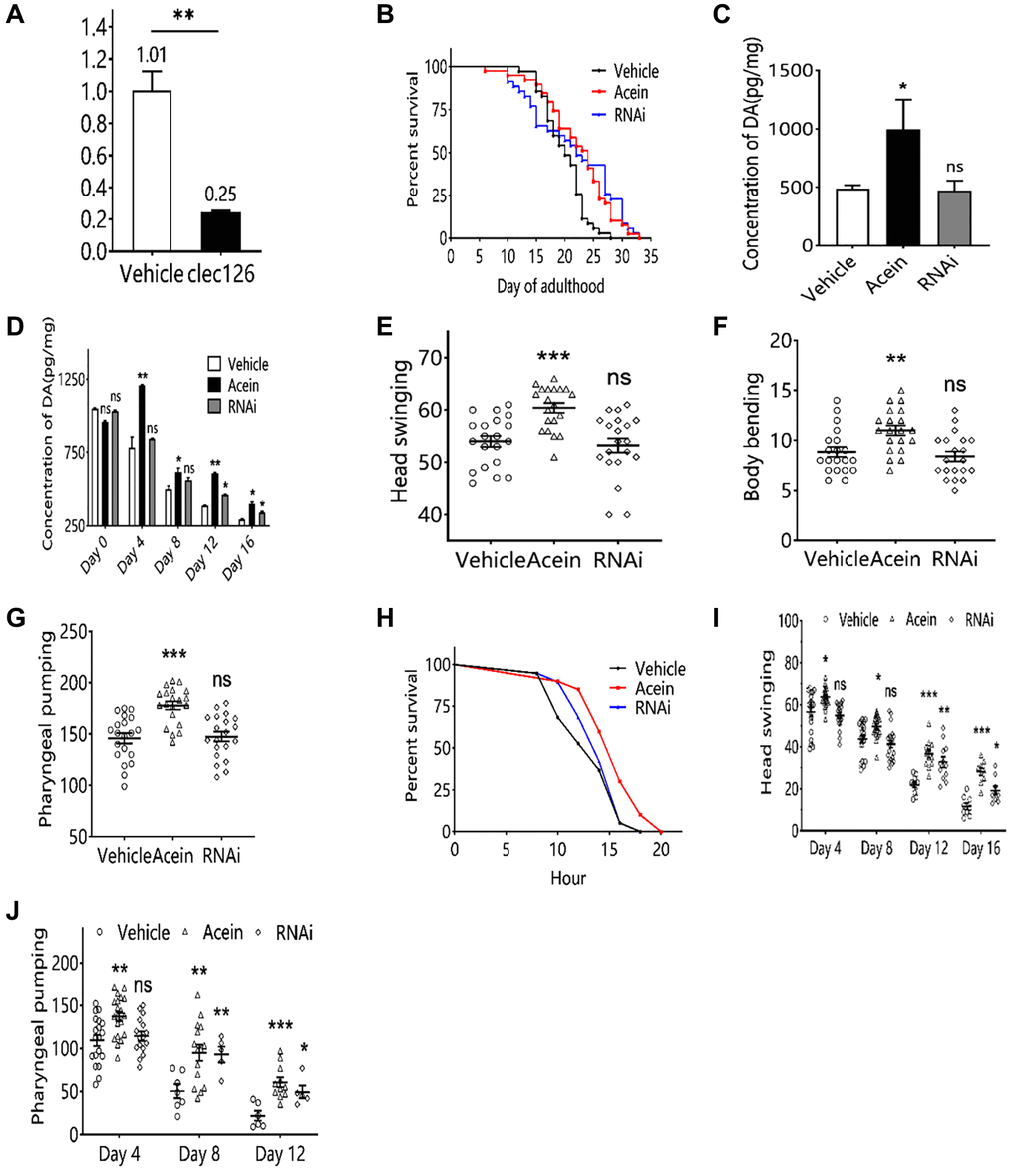
Figure 3. Aging-associated phenotypic assay results in C. elegans with clec-126-RNAi. (A) qPCR validation results of clec-126-RNAi C. elegans. (B) Lifespan experiments for clec-126-RNAi C. elegans. (C) Dopamine changes in C. elegans at 48 h. (D) Changes in dopamine levels in C. elegans with age. (E) Head swing in clec-126-RNAi C. elegans. (F) Body bending in clec-126-RNAi C. elegans. (G) Detection results of pharyngeal pumping in clec-126-RNAi C. elegans. (H) Survival curve on Day 8 in clec-126-RNAi C. elegans group in a thermostressed environment at 35°C. (I) Head swing frequency in clec-126-RNAi C. elegans of different age groups. (J) Pharyngeal pumping frequency in clec-126-RNAi C. elegans of different age groups. Compared with the vehicle group, nsp > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001.
Overexpression of clec-126 in C. elegans reduces dopamine in the elderly and shortens their lifespan
Successful overexpression of clec-126 in C. elegans was verified by qPCR technology (Figure 4A). The results showed that overexpression of clec-126 significantly shortened the lifespan of C. elegans, with a maximum mean reduction of 23.96% and a median reduction of 33.33% (Figure 4B). With age, the dopamine content of C. elegans gradually decreased. Clec-126 overexpression did not affect dopamine levels (p > 0.05) (Figure 4C) but accelerated its breakdown in aged C. elegans and significantly reduced its content in middle-aged C. elegans (p < 0.05) (Figure 4D), thereby accelerating aging. Motor behavior results showed that overexpression of clec-126 in C. elegans had no significant effect on the head swing, body flexion and pharyngeal pumping frequency, suggesting that overexpression of clec-126 did not affect the exercise capacity of C. elegans (Figure 4E–4G). However, overexpression of clec-126 in aged C. elegans significantly accelerated the decline in head swing and pharyngeal pumping ability (Figure 4I, 4J). On the other hand, overexpression of clec-126 had no significant effect on the survival time under heat stress but shortened the overall survival time of C. elegans at 35°C (Figure 4H).
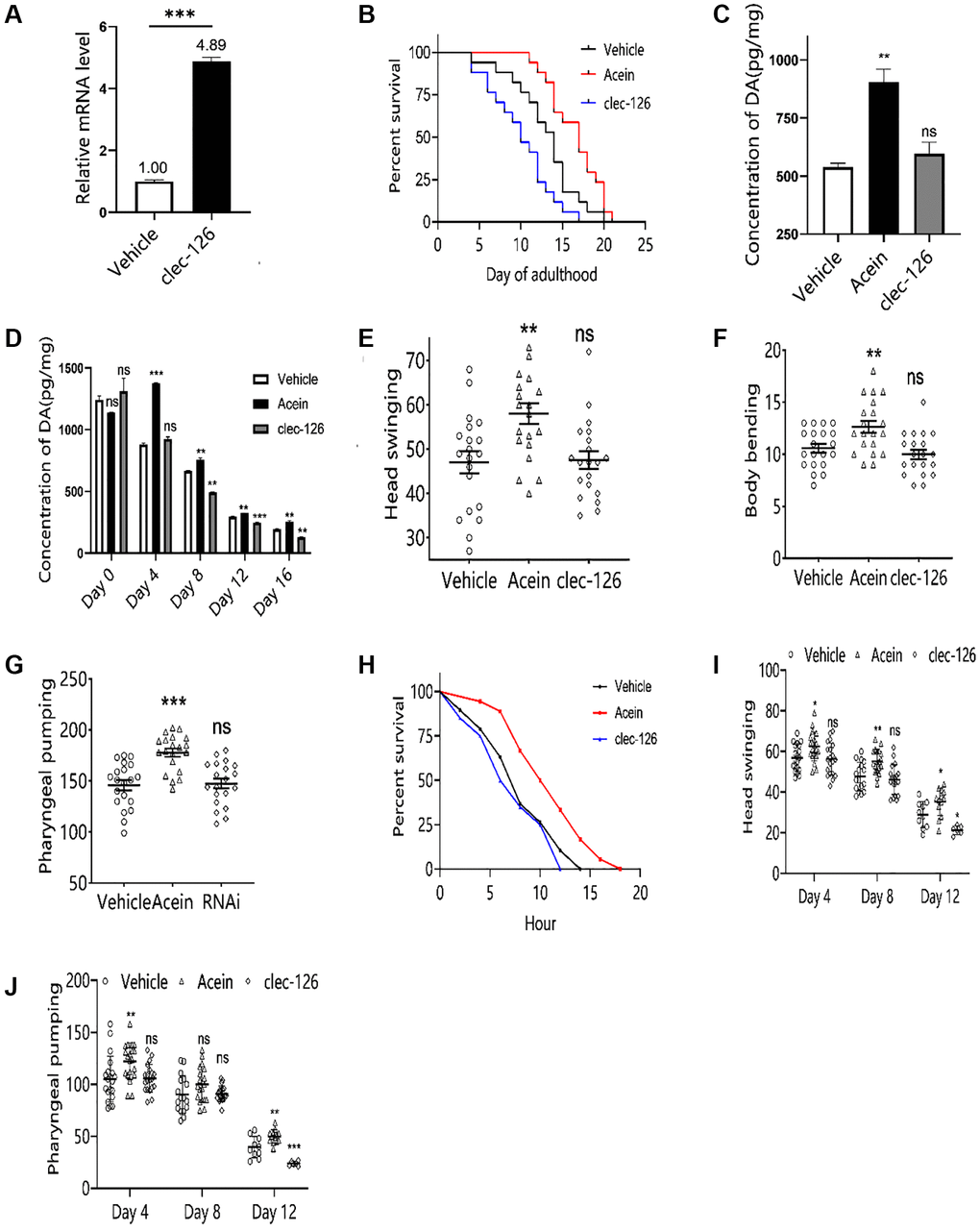
Figure 4. Aging-associated phenotypic assay results in C. elegans overexpressing clec-126. (A) qPCR validation results of C. elegans overexpressing clec-126. (B) Lifespan experiments of C. elegans overexpressing clec-126. (C) Dopamine changes in C. elegans at 48 h. (D) Changes in dopamine levels in C. elegans with age. (E) Head swing in C. elegans overexpressing clec-126. (F) Body bending in C. elegans overexpressing clec-126. (G) Pharyngeal pumping in C. elegans overexpressing clec-126. (H) Survival curve of Day 8 C. elegans overexpressing clec-126 in a thermostressed environment at 35°C. (I) Head swing in C. elegans overexpressing clec-126 of different age groups. (J) Detection results of pharyngeal pumping frequency in C. elegans overexpressing clec-126 of different age groups. Compared with the vehicle group. nsp > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001.
Interaction between clec-126 and other differentially expressed genes
qPCR showed that overexpression of clec-126 resulted in downregulation of T02D1.7 and nspf-3 and upregulation of col-41 and Y34F4.3 in C. elegans (Figure 5A). The analysis showed that clec-126 was negatively correlated with T02D1.7 (r = −0.2812, P = −0.5893) and nspf-3 (r = −0.3859, P = −0.4499) and positively correlated with col-41 (r = 0.9468, P = 0.042, significant) and Y34F4.3 (r = 0.4589, P = 0.3600) (Figure 5B–5E). These observations were consistent with the qPCR results. After successful RNA interference with col-41 in C. elegans, clec-126 did not change significantly (p = 0.1336) (Figure 5F), indicating that col-41 is a downstream gene of clec-126 (Figure 5G).
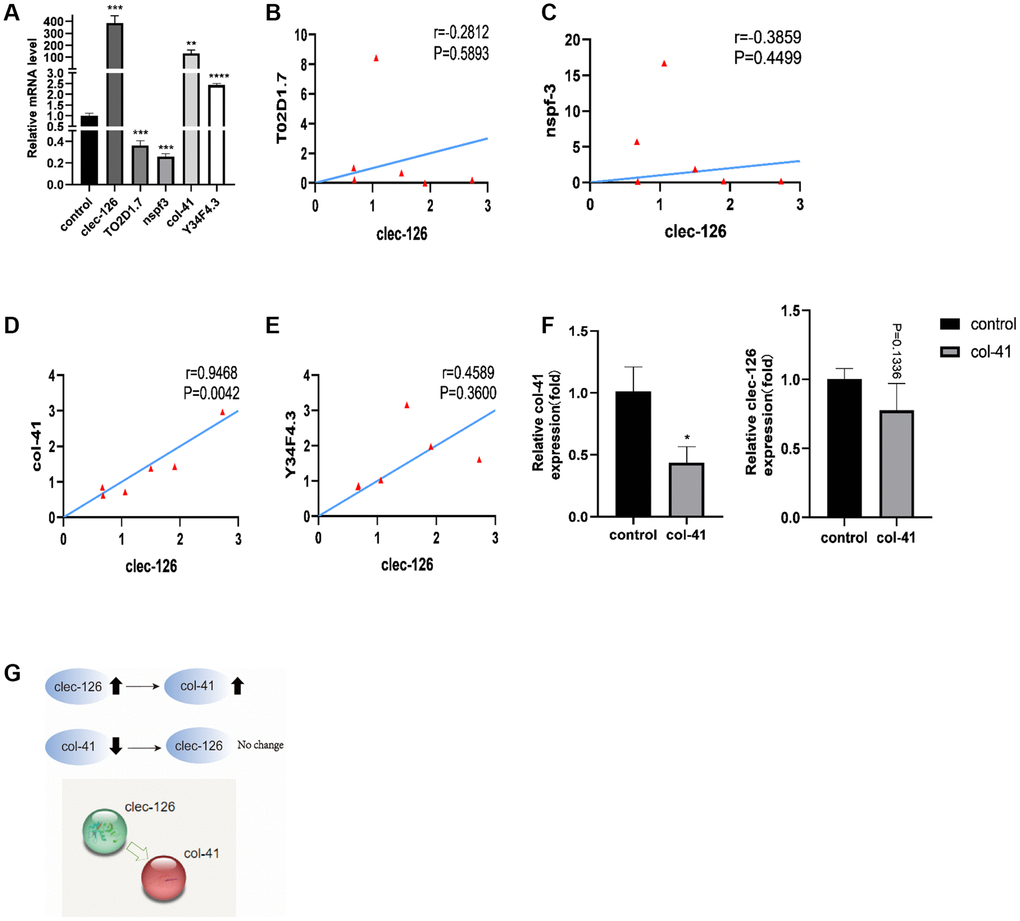
Figure 5. Interaction of clec-126 with four differentially expressed genes, T02D1.7, nspf-3, col-41 and Y34F4.3. (A) Differential expression of T02D1.7, nspf-3, col-41 and Y34F4.3 in overexpressing clec-126 C. elegans. (B–E) Correlation analysis between clec-126 and T02D1.7, nspf-3, col-41 and Y34F4.3. (F) Changes in clec-126 after RNA interference with col-41. (G) Mechanism of action of clec-126 with col-41. Compared with the control group. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
Discussion
An increasing number of studies have shown that changes in the dopamine content of organisms vary with age [18]. The accumulation of dopamine can induce neuronal death and convert harmful substances into stable compounds in the brains of PD patients, thereby ameliorating neurodegenerative diseases and delaying the aging process [19, 20]. In recent studies of the aging process in wild-type C57BL/6 mice, it has been reported that these mice exhibited age-dependent dyskinesia, hindlimb abnormalities, and a decrease in the number of dopaminergic neurons in elderly subjects [21]. Thus, the loss of dopaminergic neurons associated with aging is one of the pathophysiological bases of aging.
Since Acein has been shown to be able to stimulate dopamine secretion in the brain [13], we decided to study its influence on aging. We selected the model organism C. elegans, which is classically used in aging research [22]. The dopamine content in C. elegans gradually decreases with the age. Acein was able to delay the decrease in dopamine levels and increase dopamine content in middle-aged and elderly C. elegans (Figure 1D). Dopamine may affect the growth and development, fertility [23] and the fitness [24] of C. elegans. Therefore, the number of body length, body width, number of broods, and frequency of body bending, head swing, and pharyngeal pumping were examined. We showed that Acein was able to improve the body length and width, number of broods, body flexion frequency, head swing and pharyngeal pumping of C. elegans, with a clear positive effect on aging and lifespan. Along with the study of antiaging activity, we examined the relationships with clec-126, a key gene regulating aging in C. elegans, identified by transcriptome sequencing. The clec-126 gene encodes the C-type lectin domain protein [25] which as a negatively regulated gene, might be linked to body immunity [26]. To date and to our knowledge, no study has reported its role in aging. Our results suggest that clec-126 expression gradually increases during aging in C. elegans in correlation with a decrease in dopamine content, thereby accelerating aging. In contrast, low clec-126 expression correlates with increased dopamine content in C. elegans, thereby delaying aging. This suggests that clec-126 overexpression may be an aging-related factor rather than a longevity-related factor. We showed that clec-126 regulation did not affect dopamine changes in young C. elegans (Figures 3D, 4D), likely due to their well-established function of the dopaminergic system [27] in which neurons connecting the striatum can release dopamine normally [28], and probably plays a role in regulating various physiological functions of the central nervous system [29]. In aged C. elegans, Acein was able to increase dopamine levels and induce downregulation of clec-126, having a positive effect on aging and lifespan. Aging induced by high expression of clec-126 in C. elegans is slowed down by treatment with Acein. Our results suggest that dopamine concentration is responsible for regulating clec-126 expression. In humans, the lectin encoded by the orthologous gene clec is able to bind to glycans, which are receptors of virus-infected host cells, mediate microbial infections and negatively regulate immune function [30]. Clec-126 as a “negative gene” affected the healthy lifespan of C. elegans and therefore might be involved in the immune response of C. elegans. We also demonstrated for the first time that col-41 was a downstream gene of clec-126 and that Acein promoted col-41 expression after downregulating clec-126.
In conclusion, we have shown that Acein increases brood number and the fitness, ameliorates age-related functional decline and delays the aging process of C. elegans by regulating clec-126 (Figure 6) to achieve the goal of prolonging healthy life. We have identified that Acein, most likely via its ability to stimulate dopamine secretion, can regulate the expression of clec-126. However, further studies are needed to determine the antiaging mechanism of Acein and whether clec-126 can serve as a target for age-related pathologies in organisms.
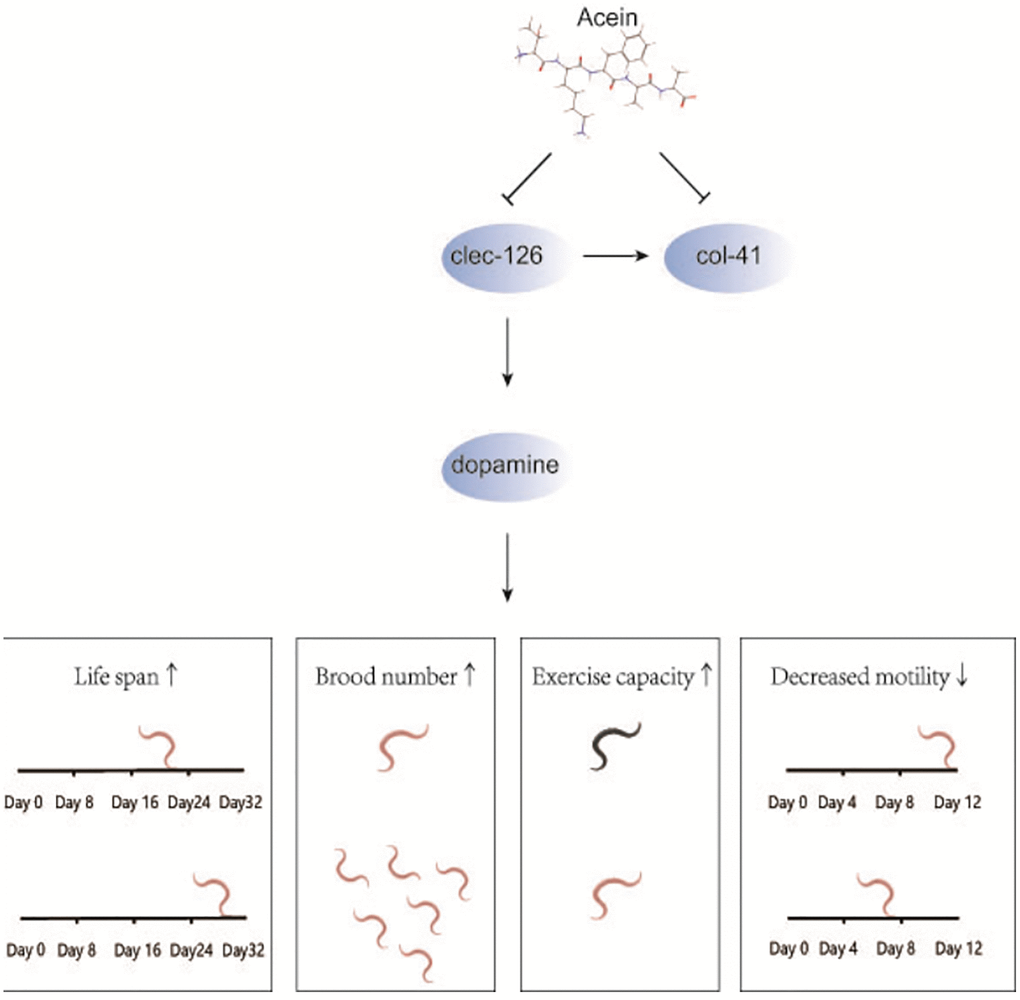
Figure 6. Mechanistic diagram of the antiaging effect of Acein in the clec-126 overexpressing C. elegans model. Acein inhibits the expression of clec-126 to increase dopamine secretion to prolong C. elegans lifespan, increase C. elegans brood number and motility, reduce motility decline, and delay C. elegans senescence. In addition, col-41 was first found to be downstream of clec-126, and clec-126 promotes col-41 expression.
Materials and Methods
Synthesis and characterization of Acein
Acein was synthesized by the Fmoc protection strategy in SPPS with Wang resin. Fmoc-Pro-OH, Fmoc-Thr(OtBu)-OH, Fmoc-Lys(Boc)-OH, Fmoc-Phe-OH and Fmoc-Ala-OH were used (2.2 eq) and couplings were performed using HBTU (2-(1H-benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (HBTU) (2.2 eq) as coupling reagent in the presence of NMM (N-methylmorpholine) (4.4 eq) in a mixture of DMF-DCM (1:1) for 2 h. Each coupling was repeated twice. After coupling, the solvents were removed, and the resin was washed with DMF (3 times), methanol (2 times) and then DCM (2 times) for about 30 s each washing. Removal of the Fmoc protecting group was performed with a mixture of piperidine in DMF (20/80) for 10 min. Deprotection step was repeated twice, then washings were performed as described above. After completion of the couplings, the resin was treated with 5 ml/100 mg resin of the mixture trifuoroacetic acid/triisopropylsilane/water (TFA/TIPS/H2O) (95/2.5/2.5) for 3 h. The resin was filtered, rinsed twice with acetonitrile/water (1:1). The filtrate was collected and concentrated in vacuo. The residue was triturated in ethyl ether and centrifuged. The ethyl ether was removed. This was repeated three times. The residue was dissolved in a mixture of acetonitrile/water (1:1), freeze-dried in liquid nitrogen and lyophilized to yield the peptides as white powders. The crude peptide was purified by semi-preparative HPLC. The eluate with absorption value greater than 100 mV in the main peak was collected. The purity and the molecular weight of Acein were detected by analytical HPLC and LC-MS, respectively. The crude peptide purity of Acein was 82.4%, and the purity after purification was 97.5%. The physical properties of Acein are reported in Supplementary Table 3 and Supplementary Figure 1.
Lifespan assay
Lifespan tests were performed at 20°C on C. elegans growth medium (NGM) plates inoculated with E. coli OP50. C. elegans were synchronized to the L1 stage, and when the larvae developed into the L3/L4 stage, 100 μL of a 5-FUDR solution (15.60 μg/mL) was instilled to inhibit oviposition and reproduction. The first day of lifespan tests was when the C. elegans developed into adults. Once daily, they were administered 10 nM Acein. The C. elegans status was observed daily until the death of all the nematodes. The daily number of deaths was recorded. The standard of judgment of death was when C. elegans were unresponsive to bright light or tapping on the plate, had no pharyngeal muscle movement at high magnification, and remained unresponsive to tapping of the head with a C. elegans picking needle.
Growth and development test
In parallel, L4 stage larvae were collected with a C. elegans picking needle and transferred to a new NGM plate inoculated with OP50. Adult C. elegans were transferred to a new NGM plate after each egg laying until the end of the oviposition period. The number of larvae from all plates was photographed and the number of broods from ten C. elegans was recorded repeatedly in each group.
Exercise capacity assay
Twenty adult C. elegans from each group were randomly selected and placed in new NGM medium. They were allowed to recover for 1 h. Then, an appropriate amount of M9 buffer was added to record both the number of times the C. elegans head swings from side to side and the number of body flexions for 30 s under an inverted microscope. The movement of the pharyngeal pump was amplified under an inverted microscope and more than ten C. elegans were videotaped for 30 s. Then, PotPlayer was used to slow down the video recording by 0.3 times to record the number of C. elegans pharyngeal pumps.
Determination of changes in dopamine levels
A panspecies dopamine assay kit (Sango Biotech, China) was used to measure total dopamine content in approximately 500 C. elegans. Then, a BCA protein concentration test kit (Beyotime, China) was used to measure the total protein content. The relative level of dopamine secretion in C. elegans was expressed as the ratio of total dopamine content to total protein content, which was significantly comparable between different groups [31, 32].
Quantitative real-time PCR
Sequencing of the transcriptome with Unique Molecular Identifiers (UMI) was performed on C. elegans samples. Differentially expressed genes were selected, ranked according to FC size. Genes with low reproducibility between parallel groups were excluded and genes with low expression were selected and then validated. Total RNA from cells was extracted by TRIzol reagent (Sangon Biotech, China) and transcribed into cDNA by a reverse transcription kit (Yisheng, China). cDNA was amplified by SYBR Green PCR Master Mix (Yisheng, China), qRT-PCR (ABI, USA) was performed using QuantStudio3, and GAPDH was used as an endogenous normalization control. The 2−ΔΔCt method was used. The primers are shown in Table 1.
Table 1. Primer sequence information table.
| Target gene | Forward primer | Reverse primer |
| pmp-3 | ATCGTCTTCACTCGGATTGCCTTG | TCCTCTTCCTGCTCATCTCGTTCC |
| gstk-2 | CAACGTATTCTTGTCGCTTCAC | TCACTCATCATCACCAACTCATC |
| vit-2 | TTCACCGCTCATACCTTCTCAAGA | CTCATCAGATTGCTCCTCGTCTCT |
| T02D1.7 | CATCTTCATCTCTTCCGTTCAAG | TTCTGGACTCATGTTGGATTCAC |
| E02H9.9 | AGATTGGAGAGACTGATTGGATGGT | GTCGTGGATGATGAGCATGTAGATT |
| clec-99 | CACATGCTGCCACTCAACAATG | CCTCTAACAGTCCTCGCCACAT |
| F26D2.16 | GTCTCATGTCATTCCGCCATTCG | CCATCCTTCCGCATCCTTTGTATG |
| nspf-3 | TCGCCACTGATGAATATGACC | TATGACGAATCTCCTCCTTTGC |
| clec-197 | TCAAACAGTGAAGCATCTGGAAAGG | TCCACCTCCGTATCCTCCATAGT |
| Y51A2D.8 | GCCTTCAACTTTGCCGTAATC | GCTCTTCTACAATCGTCCTCAAT |
| Y67D8C.7 | TGGATGTTGAGGAAGAAGATATTGG | CCACCACCACCGTACTTGT |
| clec-136 | GGTTCATCAAGCTGACAATGGT | TTCATCGTCTGTATAAGCCTGC |
| linc-1 | ATGTTCCGTCGTGTCTTCGT | CAACTGGAGCCTTGATGGTAAC |
| Y37F4.3 | TTATCAAGAACACCTGCTCCAAGTT | GCTCTCTGAATGGACTCAAGAATGT |
| ZK1025.3 | CAACTTCTCACAATGGTCACACT | TCCACTAACTGCGTATCGGTAG |
| col-41 | TCACAGCCACCAGTCAGAG | GATGGATAGCTTGGAGATGGATAG |
| clec-126 | GCAGTTCTAGCCAGAGTTCCAGT | CCAGGCAACCTCCTCCTTGTT |
| ZK856.4 | CAGACGGAAGAAGCAAGATGAAGT | TCGGAAACTGGGAACTCATCGT |
| K10G6.9 | TGTGTTGTCTACTGGTATCTGGAAC | GCTTCCTCTGCTCATTGATGTACT |
| Y34F4.3 | GCTGGTGCAAGTATCCAATGT | CGAGTGACAATCCGAACGAAT |
RNAi clec-126 in C. elegans
The HT115 strain expressing the dsRNA of clec-126 was constructed and inoculated in NGM culture dishes containing IPTG. Under the induction of IPTG, strain HT115 produced dsRNA clec-126 and C. elegans was able to obtain clec-126 C. elegans RNAi from the edible strain HT115.
Clec-126 overexpression in C. elegans
Clec-126 cDNA expression was driven by the dat-1 promoter. The pPD95.77 vector was digested with HindIII (AAGCTT) and Age I (ACCGGT) restriction enzymes to release the MCS sequence to obtain the linear vector. The fragments of the dat-1 promoter and the clec-126 sequence were amplified, and then Gibson assembly was carried out. The reporter plasmid pCFJ104 (5 ng/ml) and the target plasmid of the Pdat-1::clec-126::GFP::unc-54 3′UTR (20 ng/ml) were formulated into a mixture and then injected into wild-type N2 C. elegans. The primers used for PCR validation of clec-126 were F: TATTGGACAGATGGCTCG; R: TGTCTTGTAGTTCCCGTCA.
Stress capacity assay
When cultured L1 stage larvae of C. elegans reached L3/L4 stages, 15.60 μg/mL of 5-FUDR was added to inhibit egg laying and reproduction. After reaching adulthood, the C. elegans were treated daily with 10 nM Acein. On the 4th and 8th days, they were removed and placed in a 35°C constant temperature incubator for stress experiments. The life and death of C. elegans were recorded every 2 h until the death of the last C. elegans. The determination of death was the same as that in the lifespan experiment, which is described above.
Motor decline assay
After daily treatment with 10 nM Acein, C. elegans were taken on the 4th, 8th and 12th days to detect their capacity for motor behavior including head swing and frequency of pharyngeal pump movements. The detection method was the same as that described for the determination of the movement capacity of C. elegans.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 8. Data are presented as the mean ± standard error of the mean (SEM). A Shapiro-Wilk test was used to determine the normal distribution of the data. Independent sample t tests were used for comparisons between two groups and one-way analysis of variance was used for comparisons between different groups. Survival curves were compared and P values were calculated using log-rank analysis (Mantel-Cox). A value of p < 0.05 indicates a statistically significant difference.
Author Contributions
Jiaqi Wang, Dong Wang and Pr. Hanmei Xu designed the study; Jiaqi Wang, Dong Wang performed the experiments and analyzed the data; Setrerrahmane Sarra, Pr. Hanmei Xu and Pr. Jean Martinez wrote the paper.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This work was supported by the Chinese Industry-University-Research Cooperation Project.
References
- 1. Miller HA, Huang S, Dean ES, Schaller ML, Tuckowski AM, Munneke AS, Beydoun S, Pletcher SD, Leiser SF. Serotonin and dopamine modulate aging in response to food odor and availability. Nat Commun. 2022; 13:3271. https://doi.org/10.1038/s41467-022-30869-5 [PubMed]
- 2. Dobryakova E, Genova HM, DeLuca J, Wylie GR. The dopamine imbalance hypothesis of fatigue in multiple sclerosis and other neurological disorders. Front Neurol. 2015; 6:52. https://doi.org/10.3389/fneur.2015.00052 [PubMed]
- 3. Wright JW, Kawas LH, Harding JW. A Role for the Brain RAS in Alzheimer's and Parkinson's Diseases. Front Endocrinol (Lausanne). 2013; 4:158. https://doi.org/10.3389/fendo.2013.00158 [PubMed]
- 4. Zucca FA, Segura-Aguilar J, Ferrari E, Muñoz P, Paris I, Sulzer D, Sarna T, Casella L, Zecca L. Interactions of iron, dopamine and neuromelanin pathways in brain aging and Parkinson's disease. Prog Neurobiol. 2017; 155:96–119. https://doi.org/10.1016/j.pneurobio.2015.09.012 [PubMed]
- 5. Ohrui T, Tomita N, Sato-Nakagawa T, Matsui T, Maruyama M, Niwa K, Arai H, Sasaki H. Effects of brain-penetrating ACE inhibitors on Alzheimer disease progression. Neurology. 2004; 63:1324–5. https://doi.org/10.1212/01.wnl.0000140705.23869.e9 [PubMed]
- 6. Kehoe PG, Wilcock GK. Is inhibition of the renin-angiotensin system a new treatment option for Alzheimer's disease? Lancet Neurol. 2007; 6:373–8. https://doi.org/10.1016/S1474-4422(07)70077-7 [PubMed]
- 7. Yamada K, Horita T, Takayama M, Takahashi S, Takaba K, Nagata Y, Suzuki N, Kanda T. Effect of a centrally active angiotensin converting enzyme inhibitor, perindopril, on cognitive performance in chronic cerebral hypo-perfusion rats. Brain Res. 2011; 1421:110–20. https://doi.org/10.1016/j.brainres.2011.09.016 [PubMed]
- 8. Chowdhury R, Guitart-Masip M, Lambert C, Dayan P, Huys Q, Düzel E, Dolan RJ. Dopamine restores reward prediction errors in old age. Nat Neurosci. 2013; 16:648–53. https://doi.org/10.1038/nn.3364 [PubMed]
- 9. Shohamy D, Wimmer GE. Dopamine and the cost of aging. Nat Neurosci. 2013; 16:519–21. https://doi.org/10.1038/nn.3385 [PubMed]
- 10. Berry AS, Shah VD, Baker SL, Vogel JW, O'Neil JP, Janabi M, Schwimmer HD, Marks SM, Jagust WJ. Aging Affects Dopaminergic Neural Mechanisms of Cognitive Flexibility. J Neurosci. 2016; 36:12559–69. https://doi.org/10.1523/JNEUROSCI.0626-16.2016 [PubMed]
- 11. Hof PR, Mobbs CV. Handbook of the neuroscience of aging. Academic press. 2010.
- 12. Di Liegro CM, Schiera G, Proia P, Di Liegro I. Physical Activity and Brain Health. Genes (Basel). 2019; 10:720. https://doi.org/10.3390/genes10090720 [PubMed]
- 13. Neasta J, Valmalle C, Coyne AC, Carnazzi E, Subra G, Galleyrand JC, Gagne D, M'Kadmi C, Bernad N, Bergé G, Cantel S, Marin P, Marie J, et al. The novel nonapeptide acein targets angiotensin converting enzyme in the brain and induces dopamine release. Br J Pharmacol. 2016; 173:1314–28. https://doi.org/10.1111/bph.13424 [PubMed]
- 14. Martinez J. Joseph Rudinger memorial lecture: Unexpected functions of angiotensin converting enzyme, beyond its enzymatic activity. J Pept Sci. 2017; 23:741–8. https://doi.org/10.1002/psc.3022 [PubMed]
- 15. Lucas-Valmalle C, Subra G, Verdié P, Kemel ML, Daugé V, Puget K, Martinez J. The C-terminal pentapeptide Acein analogue (JMV3315) stimulates dopamine release in the brain. Aust J Chem. 2023; 76:448–54. https://doi.org/10.1071/CH22232
- 16. Wang H, Liu J, Li T, Liu RH. Blueberry extract promotes longevity and stress tolerance via DAF-16 in Caenorhabditis elegans. Food Funct. 2018; 9:5273–82. https://doi.org/10.1039/c8fo01680a [PubMed]
- 17. Gomes MJ, Martinez PF, Pagan LU, Damatto RL, Cezar MDM, Lima ARR, Okoshi K, Okoshi MP. Skeletal muscle aging: influence of oxidative stress and physical exercise. Oncotarget. 2017; 8:20428–40. https://doi.org/10.18632/oncotarget.14670 [PubMed]
- 18. Chen G, Ahn EH, Kang SS, Xia Y, Liu X, Zhang Z, Ye K. UNC5C Receptor Proteolytic Cleavage by Active AEP Promotes Dopaminergic Neuronal Degeneration in Parkinson's Disease. Adv Sci (Weinh). 2022; 9:e2103396. https://doi.org/10.1002/advs.202103396 [PubMed]
- 19. Kalra J, Prakash A, Kumar P, Majeed AB. Cerebroprotective effects of RAS inhibitors: Beyond their cardio-renal actions. J Renin Angiotensin Aldosterone Syst. 2015; 16:459–68. https://doi.org/10.1177/1470320315583582 [PubMed]
- 20. Qu M, Lin Q, Huang L, Fu Y, Wang L, He S, Fu Y, Yang S, Zhang Z, Zhang L, Sun X. Dopamine-loaded blood exosomes targeted to brain for better treatment of Parkinson's disease. J Control Release. 2018; 287:156–66. https://doi.org/10.1016/j.jconrel.2018.08.035 [PubMed]
- 21. Noda S, Sato S, Fukuda T, Tada N, Hattori N. Aging-related motor function and dopaminergic neuronal loss in C57BL/6 mice. Mol Brain. 2020; 13:46. https://doi.org/10.1186/s13041-020-00585-6 [PubMed]
- 22. Kaletta T, Hengartner MO. Finding function in novel targets: C. elegans as a model organism. Nat Rev Drug Discov. 2006; 5:387–98. https://doi.org/10.1038/nrd2031 [PubMed]
- 23. Nagashima T, Oami E, Kutsuna N, Ishiura S, Suo S. Dopamine regulates body size in Caenorhabditis elegans. Dev Biol. 2016; 412:128–38. https://doi.org/10.1016/j.ydbio.2016.02.021 [PubMed]
- 24. Vidal-Gadea AG, Pierce-Shimomura JT. Conserved role of dopamine in the modulation of behavior. Commun Integr Biol. 2012; 5:440–7. https://doi.org/10.4161/cib.20978 [PubMed]
- 25. Takeuchi T, Sennari R, Sugiura K, Tateno H, Hirabayashi J, Kasai K. A C-type lectin of Caenorhabditis elegans: its sugar-binding property revealed by glycoconjugate microarray analysis. Biochem Biophys Res Commun. 2008; 377:303–6. https://doi.org/10.1016/j.bbrc.2008.10.001 [PubMed]
- 26. C. elegans Sequencing Consortium. Genome sequence of the nematode C. elegans: a platform for investigating biology. Science. 1998; 282:2012–8. https://doi.org/10.1126/science.282.5396.2012 [PubMed]
- 27. Bastioli G, Arnold JC, Mancini M, Mar AC, Gamallo-Lana B, Saadipour K, Chao MV, Rice ME. Voluntary Exercise Boosts Striatal Dopamine Release: Evidence for the Necessary and Sufficient Role of BDNF. J Neurosci. 2022; 42:4725–36. https://doi.org/10.1523/JNEUROSCI.2273-21.2022 [PubMed]
- 28. Liu C, Kaeser PS. Mechanisms and regulation of dopamine release. Curr Opin Neurobiol. 2019; 57:46–53. https://doi.org/10.1016/j.conb.2019.01.001 [PubMed]
- 29. Li N, Jasanoff A. Local and global consequences of reward-evoked striatal dopamine release. Nature. 2020; 580:239–44. https://doi.org/10.1038/s41586-020-2158-3 [PubMed]
- 30. Scur M, Parsons BD, Dey S, Makrigiannis AP. The diverse roles of C-type lectin-like receptors in immunity. Front Immunol. 2023; 14:1126043. https://doi.org/10.3389/fimmu.2023.1126043 [PubMed]
- 31. Katner SN, Neal-Beliveau BS, Engleman EA. Embryonic Methamphetamine Exposure Inhibits Methamphetamine Cue Conditioning and Reduces Dopamine Concentrations in Adult N2 Caenorhabditis elegans. Dev Neurosci. 2016; 38:139–49. https://doi.org/10.1159/000445761 [PubMed]
- 32. Salim C, Rajini PS. Glucose-rich diet aggravates monocrotophos-induced dopaminergic neuronal dysfunction in Caenorhabditis elegans. J Appl Toxicol. 2017; 37:772–80. https://doi.org/10.1002/jat.3426 [PubMed]



