Introduction
Renal cell carcinoma (RCC) represents around 3-5% of all oncological diagnoses worldwide, with higher incidence occurring in Western countries [1]. RCC has several pathological types and the histological classification is important due to different molecular targeted therapy or surgical treatment. Clear cell type accounts for 75% of all RCC cases. It is considered as malignant according to WHO classification and has poor prognosis comparing with other subtypes such as papillary or chromophobe [2, 3]. Although kidney renal clear cell carcinoma (KIRC) is highly aggressive and popular, relatively fewer are known about its etiology. The possible risk factors of KIRC include hereditary diseases or genetic aberration of mTOR pathway proteins [4]. Therefore, it is necessary to consider the newly change in KIRC and to discover novel biomarkers for improving the prognosis of KIRC.
Phosphoglycerate mutase-1 (PGAM1) is a glycolytic enzyme that has been extensively studied in different types of malignancies [5]. Numerous studies have demonstrated that PGAM1 is frequently activated in glycolysis and highly expressed in various types of cancer, including KIRC [6, 7]. In KIRC, PGAM1 has been found to play a crucial role in tumor proliferation, and its high expression is associated with abnormal glycolysis and the formation of KIRC, making it a potential therapeutic target for cancer therapy [8, 9]. In addition to PGAM1, another subfamily of PGAM enzymes, PGAM5, has also been shown to regulate several aspects of cancer cell death, including apoptosis and necrosis, by altering mitochondrial function [10]. The role of PGAM5 in cancer is not as extensively studied as PGAM1, but it is emerging as an important player in cancer cell biology [11]. The dysregulation of PGAM enzymes, particularly PGAM1, plays a significant role in cancer cell metabolism and proliferation, making them attractive targets for cancer therapy. The immune microenvironment has been shown to be crucial for tumorigenesis in KIRC [12]. This study is the first to report the involvement of this metabolic gene in immune cell infiltration in KIRC. Further research is needed to fully understand the mechanisms underlying their involvement in cancer progression and to develop effective therapeutic strategies.
The aim of our study was to explore the theragnostic potential of PGAM1 in KIRC through an integrated analysis. This included analyzing differential gene expression, protein correlation, pathway analysis, and prognostic analysis across various tumor types and stages. We also examined the correlation of PGAM1 expression with immune-infiltrating cells and immunomodulatory factors. Our findings indicate that PGAM1 could serve as a strong prognostic biomarker and is closely associated with immune mechanisms, highlighting its potential as an immunotherapeutic agent for KIRC.
Materials and Methods
Analysis of gene expression differences and prognostic significance in KIRC
In this study on differential expression of KIRC genes and their prognostic significance, various databases and tools were used to analyze gene expression levels in KIRC tumors and adjacent normal tissues, and survival analysis was performed as described in a previous study [13, 14]. Briefly, TNMplot, UALCAN (The University of ALabama at Birmingham CANcer data analysis Portal), and Gene Expression Omnibus (GEO) were accessed to retrieve relevant data. The Human Protein Atlas was also consulted to obtain immunohistochemical (IHC) staining images for further analysis. In conducting survival analysis, the Kaplan-Meier plotter tool was utilized to assess the relationship between clinical stages of KIRC and various factors such as immune cell content and tumor mutational burdens. To determine optimal patient groupings for survival analysis, the tool was configured to use the “Auto select best cut off” feature, which selects the best-performing cutoff value. Overall, the various analyses performed in this study provide important insights into the gene expression and prognostic significance of KIRC, and may inform future research and clinical decision-making in this area.
Single-cell transcriptomic and immune profiling analysis
Single-cell RNA sequencing (scRNA-seq) has emerged as a powerful tool to explore cellular heterogeneity and gene expression at the single-cell level. In this study, scRNA-seq data were obtained from the GSE159115 and GSE121636 datasets of the GEO database. Quality control (QC) was performed using the R package Seurat to ensure the inclusion of high-quality cells and reduce the influence of batch effects. To identify distinct cell subpopulations, uniform manifold approximation and projection (UMAP) clustering was performed using the “BiocManager” and Gene Set Variation Analysis (“GSVA”) packages in R. Cell type annotation was performed by comparing the expression profiles of previously recognized cellular marker genes with those of the identified cell subpopulations using the “SingleR” package in R. The association between gene expression and immune cell infiltration/abundance was explored using the “Gene” module of TIMER, focusing on macrophages, dendritic cells (DC), and CD4+ T, CD8+ T, and B cells. Finally, the association between immune infiltration and overall survival of KIRC patients was estimated using several algorithms, including TIMER, EPIC, MCPCOUNTER, CIBERSORT, CIBERSORT-ABS, QUANTISEQ, and XCELL.
Processing of spatial transcriptomics data using seurat algorithm
We performed analysis based on previously published articles and established spatial transcriptome data of kidney injury model animals [15]. The spatial transcriptomics data were processed using the R package Seurat (v4.1.0) and log normalization was performed to normalize the data. To integrate Seurat objects into a single ST dataset and remove batch effects, we utilized the functions SelectIntegrationFeatures, FindIntegrationAnchors, and IntegrateData. To reduce dimensionality, we performed the function RunPCA, and then used the functions FindNeighbors and FindClusters to cluster similar ST points. Initially, different clusters were divided based on hematoxylin-eosin stained (H&E) sections and annotated by unsupervised cluster analysis. Upon annotating clusters with cell markers, we observed that some clusters highly expressed multiple cell markers. To address this, we employed the ssGSEA algorithm to score common cell types based on the average expression matrix of different clusters, which was confirmed to be more effective in ST [16].
Analysis of human KIRC specimens
Tissue microarray (TMA) slides (CL2) consisting of human renal cancer, metastatic, and normal tissues were procured from SuperBioChips Laboratories (Seoul, Republic of Korea). To perform an immunohistochemistry (IHC), the protocol described in a prior study [17] was followed.
Colocalization analysis of immunofluorescence staining
We employed a method that has been described previously, which is summarized as follows [18]. Immunofluorescence analysis was performed by fixing the tissue with 4% paraformaldehyde at room temperature for 10 minutes, permeabilizing it with 0.2% Triton X-100 for 5 minutes, blocking it with 5% BSA for 30 minutes, and incubating it overnight at 4° C with the specified primary antibodies PGAM1_A4170 and CD163_A22619 purchased from Abclonal (Woburn, MA, USA). After washing with PBST, the samples were incubated with secondary antibodies for 30 minutes at room temperature. The BX61VS® Fully Motorized Fluorescence Microscope (Olympus Corporation, Tokyo, Japan) at ×20 magnification was used to digitize all glass slides with high precision. The whole-slide images were viewed and analyzed with Olympus VS-ASW® software at Li-Tzung Pathology Laboratory (Kaohsiung, Taiwan).
Pharmacogenetic prediction model
We developed a pharmacogenetic prediction model to analyze drug sensitivity based on PGAM1 expression, using the knockdown-screen data repository of the Genomics of Drug Sensitivity in Cancer (GDSC) algorithm in Q-omics v.1.0. Pearson correlation coefficient analysis was performed to assess the correlation between PGAM1 expression levels and drug dose levels [19].
Statistical analysis
The statistical analysis in this study followed the methods described in a previous publication [20]. Pearson’s correlation coefficient was used to evaluate gene expression correlation, while a t-test or Fisher’s exact test was used for the comparison between two groups, and one-way ANOVA was used for comparison within one group, all analyzed using GraphPad Prism software (GraphPad Software, La Jolla, CA, USA). A p-value of less than 0.05 was considered statistically significant.
Results
Differential expression and genetic variation analysis of PGAM1 in KIRC
We first conducted an analysis of the expression levels of PGAM family genes in the male reproductive system and observed that PGAM1 exhibited higher expression in male reproductive organs of fat fruits (Figure 1A). We subsequently explored the association of PGAM1 mutations with common cancer progression genes, such as VHL, PBRM1, and SETD2 (Figure 1B). Subsequently, we conducted an analysis in which we extracted data pertaining to PGAM1 expression from the TCGA database, focusing on patients diagnosed with Kidney Renal Clear Cell Carcinoma (KIRC). This information was presented using a waterfall plot, offering a visual representation of the top 25 genes that exhibited notable alterations in response to PGAM1 expression changes. This analysis delved into the genetic variations that occur at different levels of PGAM1 expression and their connections with genes that are frequently found to be mutated in KIRC. The insights gained from this examination are depicted graphically in Figure 1C, providing a comprehensive view of these associations and their potential implications in the context of KIRC. The mutation landscape identified various types of mutations, including splice site, missense, frameshift, and nonsense mutations, as well as in-frame ins/dels. Moreover, we used protein-protein interactions to evaluate regulatory network maps and inferred underlying mechanisms. Our analysis revealed that PGAM1 was associated with VHL, which was consistent with the results from Figure 1C. Furthermore, we found that the intermediate factor connecting these two genes was HIF1A (Figure 1D). We conducted univariate and multivariate Cox regression analyses to evaluate the association of PGAM1 expression with OS. Our findings indicated that stage, sex, age, and PGAM1 (high vs low) were significantly associated with OS (Figure 1E). Finally, we applied multivariate Cox regression analysis to the same variables and found that the risk score could serve as an independent prognostic factor (P < 0.05) (Figure 1F).
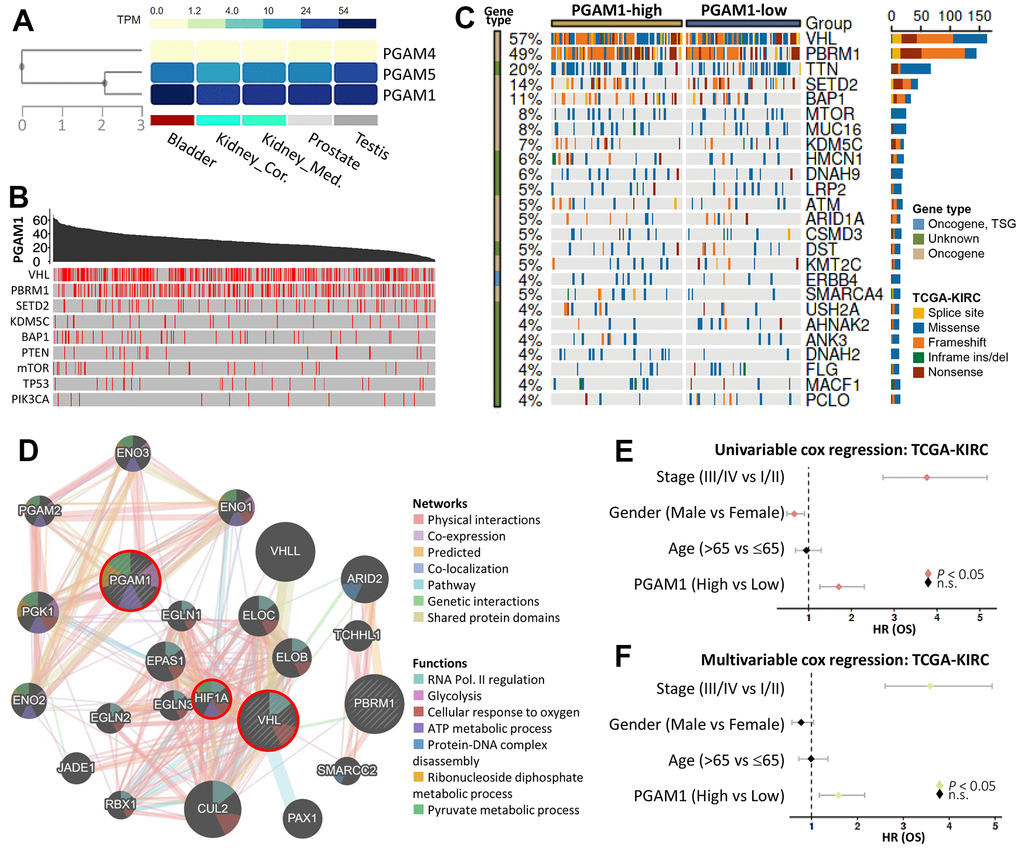
Figure 1. Gene landscape and characteristics of PGAM1 in KIRC. (A) PGAM1 gene expression levels in the male urinary system were examined. (B) The relationship between PGAM1 and nine highly mutated genes in KIRC was investigated, with mutation sites indicated by red lines. (C) The frequency of mutations was compared between PGAM1-high and PGAM1-low groups using Fisher’s exact test. Mutation types, driver mutation types, and groups are shown in the right panel. (D) A PGAM1 interaction network was generated using the Reactome database. (E, F) Univariate and multivariate Cox regression models were used to calculate hazard ratios for PGAM1 at different stages of KIRC.
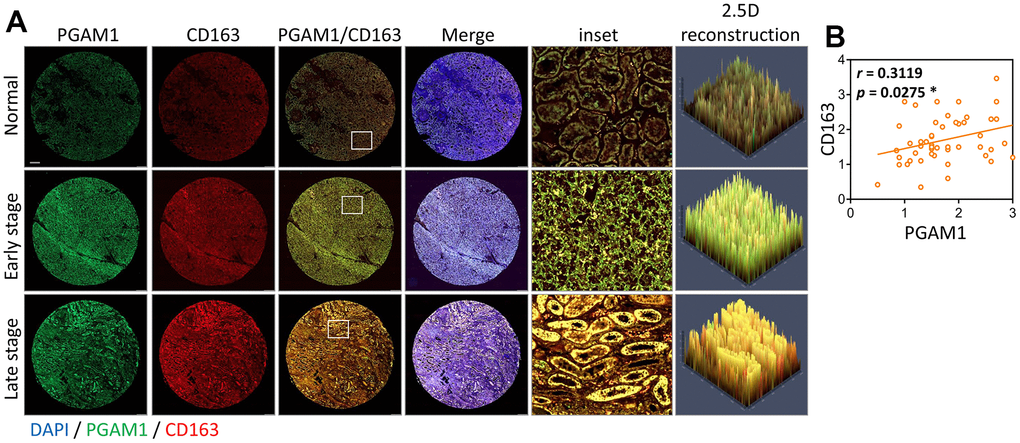
Subsequently, we conducted a comprehensive assessment of various kidney cancers and discovered that the levels of phosphoglycerate mutase-1 (PGAM1) were elevated in both clear cell renal cell carcinoma (KIRC) and papillary renal cell carcinoma (KIRP) but reduced in chromophobe renal cell carcinoma (KICH) (Figure 2A). Upon scrutinizing the expression of PGAM1 across diverse sample types, cancer stages, metastasis stages, and ccRCC subtypes in The Cancer Genome Atlas (TCGA) (Figure 2B–2E), we observed that PGAM1 expression was significantly amplified in the first stage of metastatic KIRC, and both subtypes of ccRCC exhibited an upward trend. Furthermore, analysis of immunohistochemical staining data from the Human Protein Atlas verified that the PGAM1 protein was upregulated in KIRC tumor tissues relative to normal tissues (Figure 2F). Strikingly, Kaplan-Meier analysis revealed that patients with elevated PGAM1 immunohistochemical scores had a shorter overall survival time (Figure 2G), underscoring the potential clinical significance of PGAM1 in ccRCC.
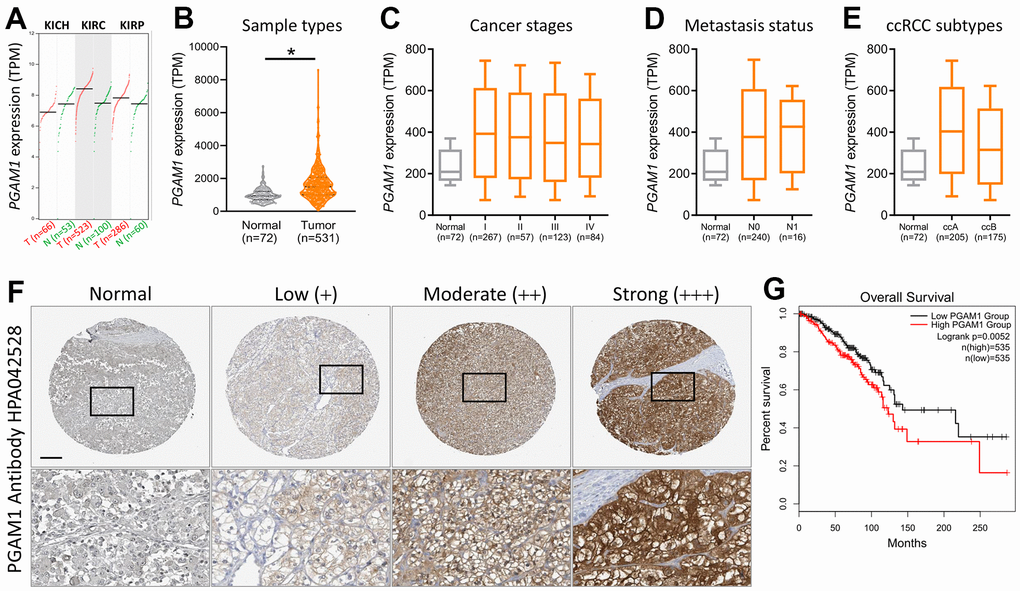
Figure 2. Evaluation of the diagnostic potential of PGAM1 expression in KIRC biopsy specimens. (A) PGAM1 gene expression levels in renal cancer. (B) Comparison of PGAM1 expression between KIRC tumor and non-tumor tissues. Boxplots depicting PGAM1 expression levels across different stages (C), metastasis status (D), and ccRCC subtypes (E) of KIRC. (F) Comparative immunohistochemical analysis of PGAM1 expression in KIRC tissue samples from four different patients based on the Human Protein Atlas. (G) Prognostic significance of PGAM1 mRNA levels for overall survival, as determined using the Kaplan-Meier plotter dataset.
Spatial transcriptomics evaluation of PGAM1 expression in injured kidneys
To assess the changes and distribution of PGAM1 expression in renal injury, we conducted an analysis of previously published data [15] that utilized two models of injured kidneys for spatial transcriptomic sequencing. The intact kidney tissue was sectioned coronally to expose each major physiologic region, cryosectioned and placed on a tissue capture area on a dedicated 10× Genomics Visium slide embedded with oligonucleotide sequences. Marker gene expression was visualized using Seurat, and distinct regions were identified by biomarkers in proximal tubules (Lrp2) and collecting ducts (Aqp2) (Figure 3A). Our results showed that PGAM1 expression was higher in the proximal tubules of renal injury compared to the control group. Furthermore, we also analyzed the gene levels of common kidney injury biomarkers as controls, including lipocalin/lipocalin 2 (Lcn2), kidney injury molecule 1 (Havcr1), insulin-like growth factor binding protein 7 (Igfbp7), all of which have significantly high expression in kidney injury. Therefore, we concluded that the metabolic gene PGAM1 was highly expressed in the damaged kidney, which may be related to the damage of this region and the change of metabolic pathway.
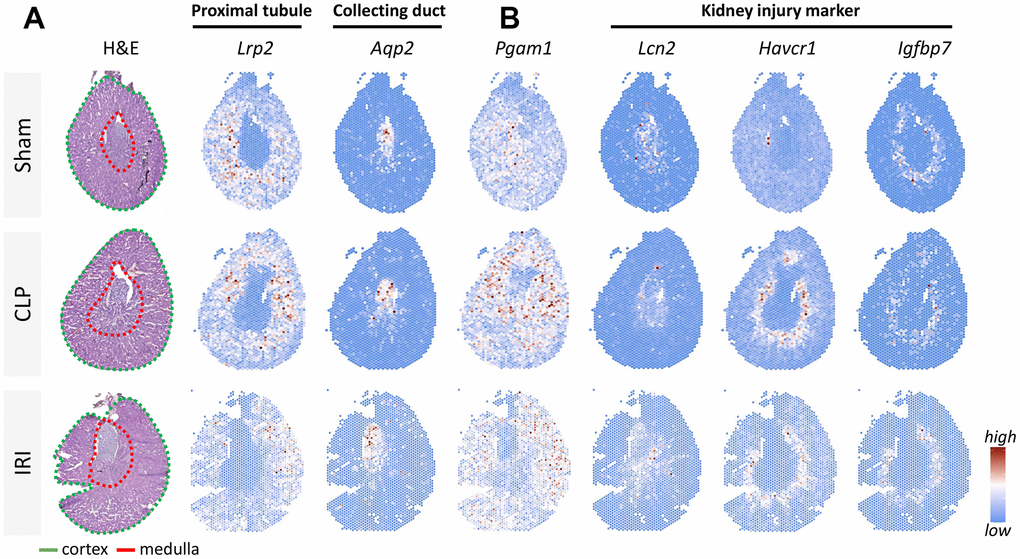
Figure 3. Resolving spatial relationships of cell type and gene expression using spatial transcriptomics in a mouse kidney injury model. (A) H&E-stained sections of 3 mouse models: sham operation, ischemia/reperfusion injury (IRI), and cecal ligation and puncture (CLP), respectively. Different regions of the cortex (Lrp2) and medulla (Aqp2) were labeled using tissue-specific biomarkers. (B) Analysis of Pgam1 and different biomarkers of renal injury (Lcn2, Kim1, Havcr1).
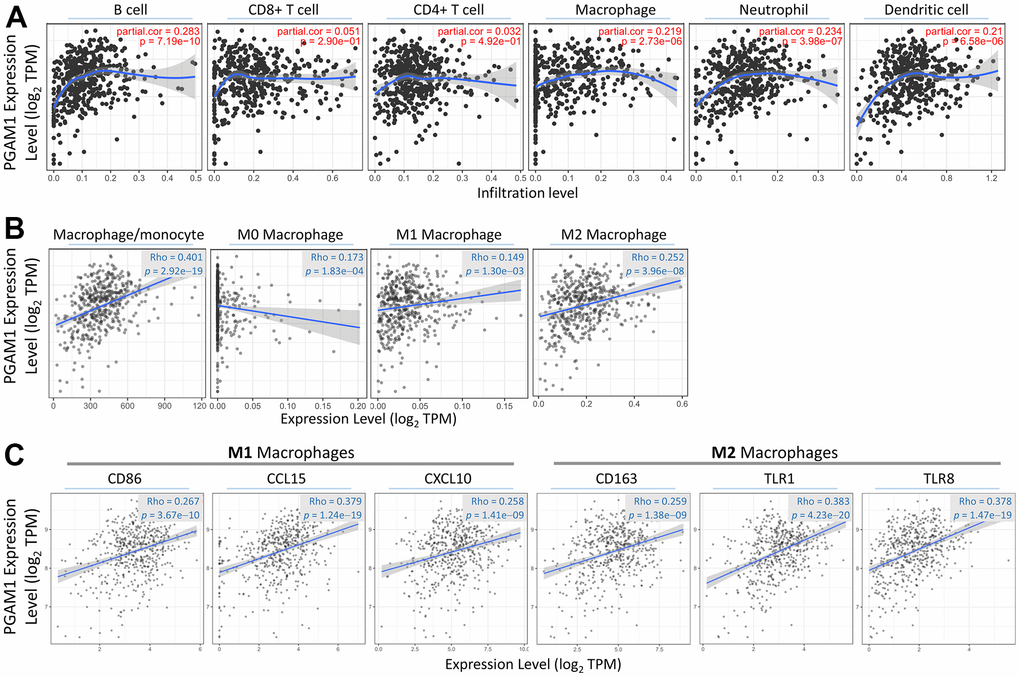
Correlation analysis between PGAM1 and infiltrating immune cells
In light of the role of the tumor immune microenvironment (TIME) in promoting kidney damage and recruiting immune cells, we further investigated the relationship between metabolism and the immune response in tumors. Our analysis showed that overexpression of PGAM1 resulted in Branch amino acid degradation, as reflected in the KEGG pathway analysis (Figure 4A). Moreover, the activation of the Immunoglobulin complex was observed in the GO-cellular component analysis (Figure 4B), and the same result was reflected in the GO-molecular function analysis, where the ratio of Immunoglobulin receptor binding and Immune response-mediated biological function was greatly increased (Figure 4C, 4D). Based on these findings, we hypothesize that kidney damage, which requires high energy, not only leads to metabolic conversion and abnormality but also promotes infiltration of immune cells, playing a significant role in the tumor immune microenvironment. The activation of the Immunoglobulin complex and increased Immunoglobulin receptor binding observed in our analysis suggests that immune cells may be recruited to the damaged kidney, contributing to the immune response in tumors.
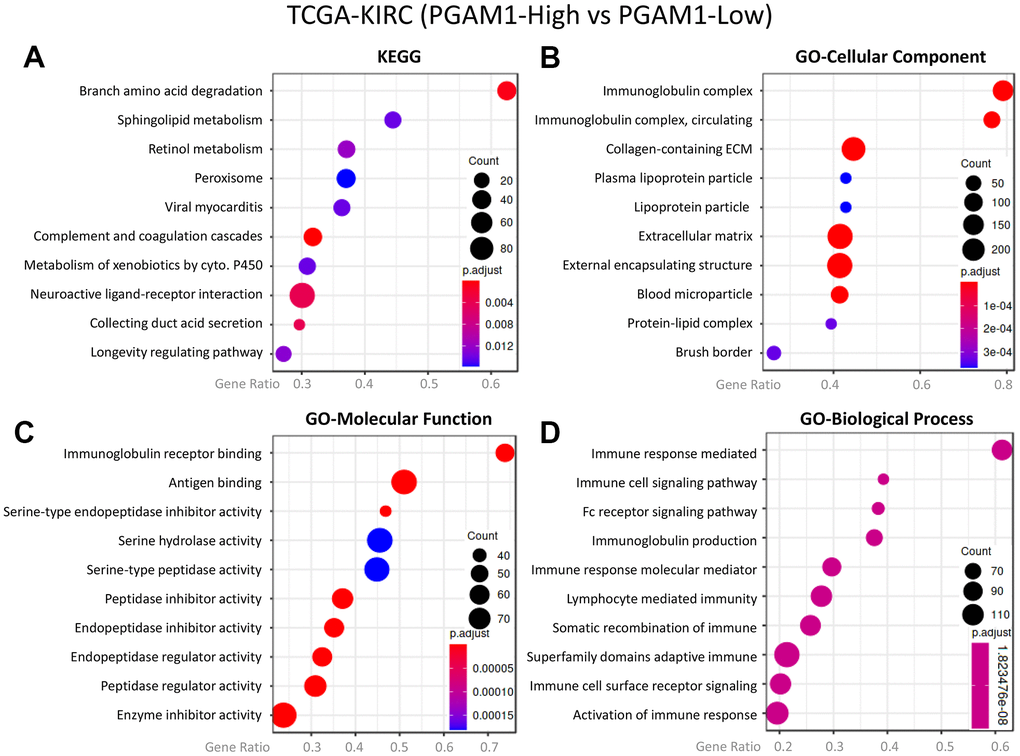
Figure 4. The co-expression genes of PGAM1 in KIRC were subjected to enrichment analysis. The target genes were analyzed using (A) Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway, (B) cellular component, focusing on (C) molecular function and (D) biological process.
Evaluation of PGAM1 transcriptome using single-cell RNA sequencing database analysis
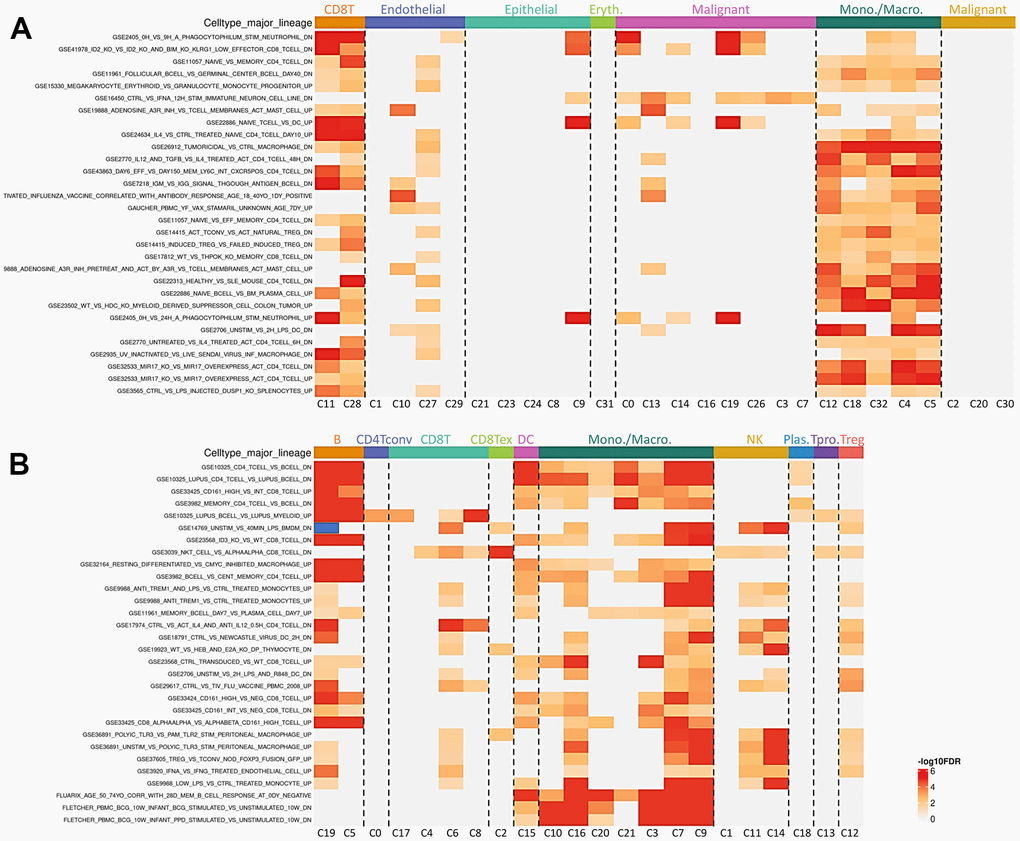
To assess the transcriptome of PGAM1 in renal cancer at the single-cell level and to explore the heterogeneity of different cell types in the renal cancer microenvironment, we conducted an analysis of two publicly available single-cell RNA-sequencing databases (GSE159115 and GSE121636). A total of 8 samples from GSE159115, comprising 8 types of cells and 32 clusters, and 3 samples from GSE121636, comprising 10 cells and 21 clusters, were included in the study. Using UMAP plots, we identified and visualized 8 and 10 major cell populations in GSE159115 and GSE121636, respectively (Figure 5A, 5B). After quality control and batch effect removal, we analyzed 27,669 (GSE159115) and 25,681 (GSE121636) cells, respectively. We determined cell type-specific markers for each cluster based on the top-ranked differentially expressed genes, which were used for cell type classification. Figure 5C, 5D shows the clinical information for each cell population, where differences in the corresponding proportions of each cluster were observed for different clinical characteristics. We further analyzed the expression and distribution of PGAM1 in these two single-cell sequencing databases. As shown in Figure 6A, 6B, we found that PGAM1 expression levels in different cell types of renal cancer were high in the same region as the high glycolysis model. In addition, oxidative phosphorylation in Figure 6B was generally increased, which was also reflected in the Inflammatory response.
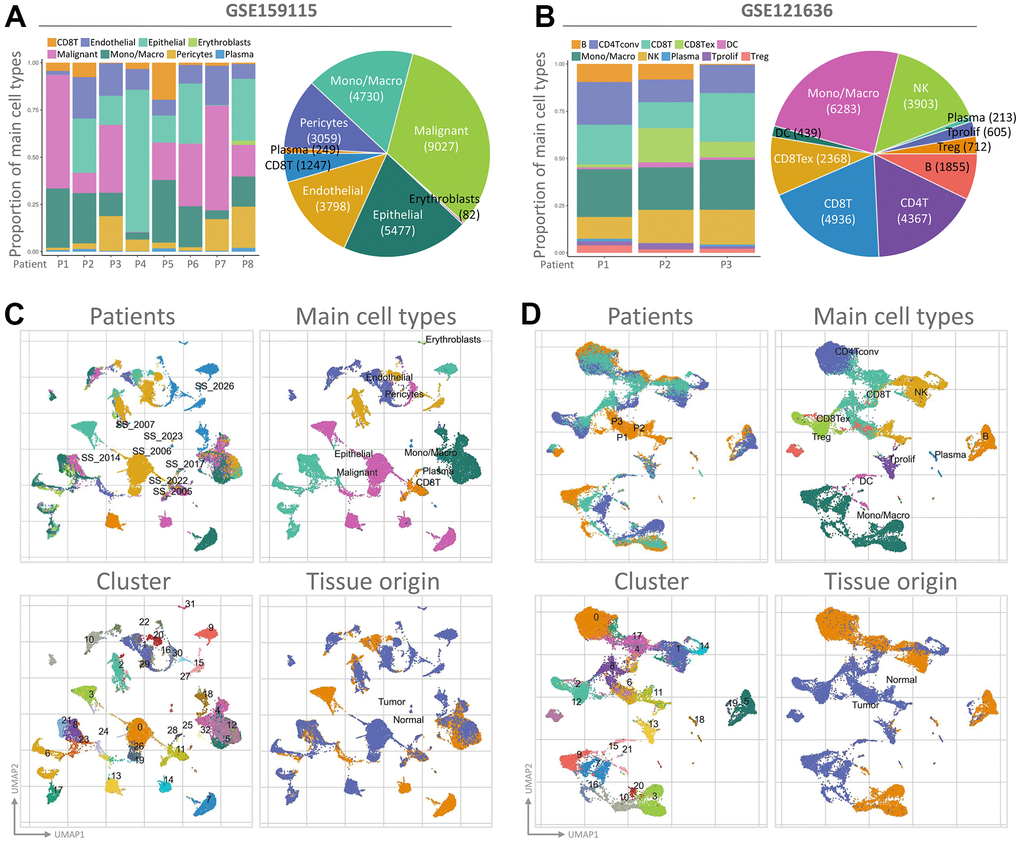
Figure 5. The use of single-cell RNA sequencing analysis has allowed for the identification of immune cell populations. (A, B) The relative proportions of each cell type found within the two datasets, while showcasing the proportion of integrated immune cells present within the databases. UMAP is an abbreviation for the Unified Manifold Approximation and Projection technique used in this study. Visual representations of all KIRC cells from both GEO datasets are depicted in (C, D) through the utilization of Unified Manifold Approximation and Projection (UMAP) and assigned specific colors according to clusters.
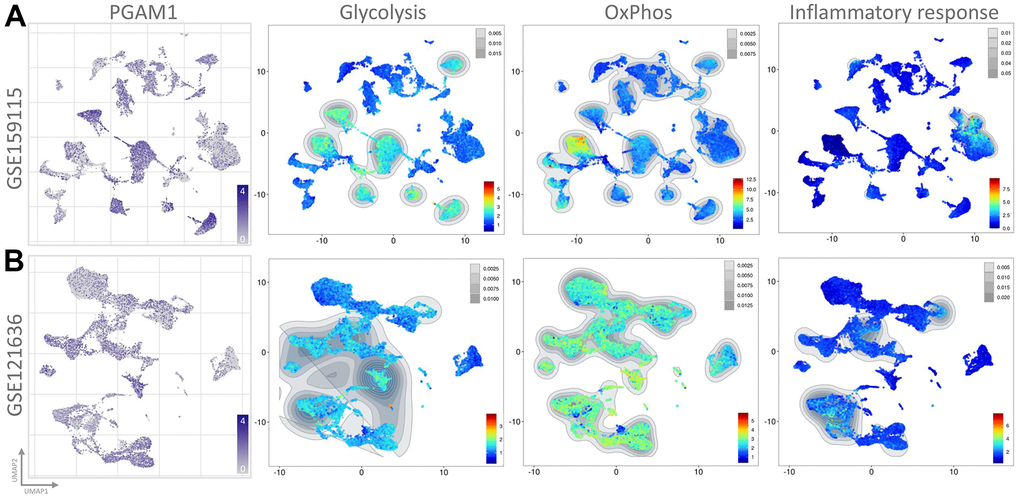
Figure 6. The single-cell transcriptomes of patient-derived cultures treated with PGAM1 were presented. The expression clusters of PGAM1 were visualized using UMAP plots in (A, B), while the UMAP plots of each distinct cluster were analyzed through Gene Set Enrichment Analysis (GSEA).
Pharmacogenetic screening for potential PGAM1 inhibitors in KIRC
In this study, we aimed to identify drugs with potential efficacy against KIRC by analyzing the GDSC repository for drugs that exhibit enhanced potency in the presence of high PGAM1 expression. We performed a cross-correlation analysis to investigate the effects of 473 drugs on shRNA-mediated PGAM1 in various KIRC cells. Our analysis identified four drugs, namely GNE-317, MS-275, AC45971100, and NSC-35468, that displayed altered potency (Figure 10A). Notably, KIRC cell lines with high shPGAM1 efficiency exhibited increased sensitivity to these drugs (Figure 10B–10E). Collectively, these findings suggest that GNE-317, MS-275, AC45971100, and NSC-35468 hold potential as anticancer agents targeting PGAM1 to regulate KIRC growth.
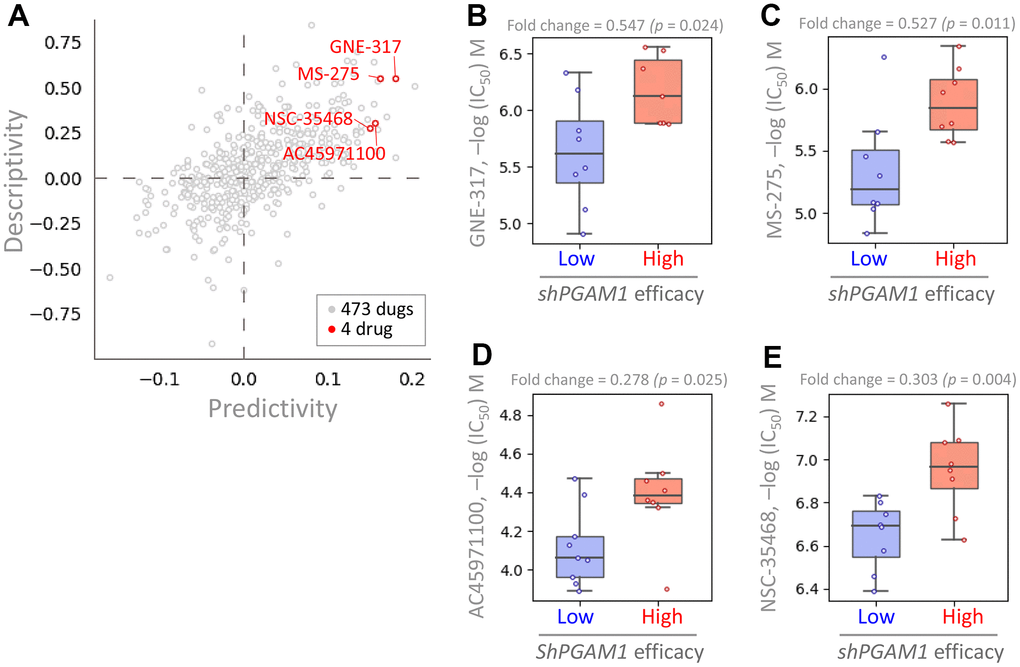
Figure 10. Analysis of drug sensitivity and cytotoxicity in renal cancer cells. (A) The PGAM1 gene was queried in the pharmacogenetics database to identify gene signatures and potential drugs. Predictivity refers to the fold change in efficacy of short hairpin PGAM1 (shPGAM1), which indicates the efficiency of PGAM1 knockdown using shRNA, between cells with high and low response to the target drug. The drug sensitivity of the shPGAM1 gene to various chemical drugs was evaluated in KIRC cell lines. The boxplots show the log of the half maximal inhibitory concentration (IC50) values of GNE-317 (B), MS-275 (C), AC45971100 (D), and NSC-35468 (E).
Discussion
Kidney injury and KIRC are two distinct conditions that can be associated with metabolic and glycolysis changes [21]. Chronic kidney disease (CKD) refers to a progressive loss of kidney function over time [22]. This can be caused by a variety of factors, including high blood pressure, diabetes, and other underlying medical conditions. In many cases, CKD is asymptomatic and may not be diagnosed until it has reached an advanced stage [23]. KIRC, on the other hand, is a type of kidney cancer that arises from the cells lining the small tubes within the kidney. This type of cancer is known for its potential to spread quickly to other parts of the body and can be difficult to treat [24]. Both CKD and KIRC have been associated with changes in metabolism and glycolysis. Metabolism refers to the chemical processes that occur within cells to generate energy, while glycolysis is the breakdown of glucose into energy [25, 26].
In cases of CKD, changes in metabolism and glycolysis may be related to the impaired function of the kidney. Studies have shown that in CKD patients, there is a decrease in the activity of key enzymes involved in glycolysis, such as hexokinase and pyruvate kinase [22]. This leads to a reduced ability of the kidneys to generate energy from glucose and may contribute to the development of insulin resistance and other metabolic abnormalities [27, 28]. Furthermore, CKD is associated with alterations in the metabolism of lipids and proteins, which can lead to the accumulation of toxic byproducts that can damage the kidneys further. The metabolic changes associated with CKD can have significant impacts on the function of the kidney [29].
One of the key metabolic alterations in KIRC is an increase in glycolysis, which is a process by which glucose is broken down to produce energy. This increase in glycolysis is thought to be due to mutations in the von Hippel-Lindau (VHL) tumor suppressor gene, which is commonly found in KIRC. The loss of VHL function leads to the stabilization of hypoxia-inducible factor (HIF), which in turn promotes glycolysis and angiogenesis [30, 31]. In addition to glycolysis, KIRC is also associated with changes in other metabolic pathways. For example, there is evidence to suggest that the tumor cells may undergo a shift towards increased fatty acid oxidation, which can provide an alternative source of energy for the tumor cells. There are also changes in the tricarboxylic acid (TCA) cycle, which is a key metabolic pathway that generates energy in the form of ATP [32, 33]. Understanding the metabolic and glycolysis changes associated with CKD and KIRC is an important area of research that may lead to new diagnostic and treatment approaches for these conditions.
The PGAM1 gene encodes a mutase that facilitates the reversible conversion of 3-phosphoglycerate (3-PGA) to 2-phosphoglycerate (2-PGA) in the glycolytic pathway, and its emerging research focus is on the regulation of cell damage and energy metabolism [34]. However, the clinical relevance, association with TIME, and therapeutic significance of PGAM1 in KIRC patients remain unknown. In this study, we employed a multi-omics approach, experimental studies, and clinical validation to uncover novel roles of PGAM1 in KIRC. Our findings demonstrate that elevated PGAM1 expression is an independent diagnostic biomarker that correlates with advanced clinical status and poor prognosis in KIRC. Moreover, we identified macrophages as key contributors to PGAM1 upregulation in tumor tissues, and analysis of spatial transcriptome and single-cell sequencing showed that PGAM1 expression is associated with immune TIME [35]. Mechanistically, PGAM1 overexpression in KIRC may be attributed to metabolic abnormalities or the high energy requirements of metastatic cancer cells, as PGAM1 and HIF1A exhibit direct regulation [11]. Overall, our study highlights the intricate relationship between metabolism and the immune response in the tumor microenvironment and provides insight into the potential mechanisms underlying kidney damage and the recruitment of immune cells.
In our pharmacogenomic data, we present four potential small-molecule drugs. GNE-317 is a PI3K/mTOR inhibitor, and our Figure 1B indicates an association between PGAM1 mutations and PIK3CA/mTOR alterations. Previous studies have shown that blocking PI3K/mTOR significantly inhibits various cancers, including gastric cancer, colorectal cancer, cervical cancer, and more [36–38]. GNE-275 is an HDAC inhibitor widely used in the treatment of various cancers, including prostate cancer, Diffuse Intrinsic Pontine Gliomas, and liver cancer [39–41]. AC45971100, also known as fluometuron, is an immune modulator that may exert its effects by inhibiting mitochondrial enzyme dihydroorotate dehydrogenase and effectively suppressing the growth of breast cancer cells [42]. NSC35468, or Podophyllotoxin bromide, is less commonly reported but has been suggested in literature to induce mitochondrial inner membrane depolarization and caspase-dependent apoptosis in colorectal cancer [43].
Conclusions
The function of the PGAM1 gene in renal cancer remains unknown. Our study demonstrates that increased PGAM1 expression is linked to poor prognosis in KIRC and may impact the tumor microenvironment and macrophage infiltration. As a result, PGAM1 could be a valuable diagnostic, prognostic, and immune-related therapeutic target for KIRC (Figure 11). Additional research is required to confirm our findings and investigate the immunomodulatory effects and mechanisms of PGAM1 in KIRC. This study highlights the inverse relationship between PGAM1 and immune response and identifies several genes worthy of further study as potential diagnostic biomarkers, therapeutic targets, and treatment options.
Figure 11. The proposed model depicts the potential significance of PGAM1 in various aspects of KIRC, such as diagnosis, prognosis, tumor immune microenvironment, and potential precision treatments.
Author Contributions
C.-Y. W. and J.-H. H. performed experiments and drafted the manuscript. Y.-D. T. and R. C. drew the figures, and discussed the concepts of the manuscript. Y.-L. T. and C.-H. K. provided valuable discussion. C.-J. L. reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This research was funded by the Ministry of Science and Technology (111-2314-B-075B-004-MY3) and Kaohsiung Veterans General Hospital (VGHKS111-147), Kaohsiung Municipal Minsheng Hospital (KMSH-11201) and Yen Tjing Ling Medical Foundation (CI-111-16 and CI-112-10).
References
- 1. Capitanio U, Bensalah K, Bex A, Boorjian SA, Bray F, Coleman J, Gore JL, Sun M, Wood C, Russo P. Epidemiology of Renal Cell Carcinoma. Eur Urol. 2019; 75:74–84. https://doi.org/10.1016/j.eururo.2018.08.036 [PubMed]
- 2. Linehan WM, Vasselli J, Srinivasan R, Walther MM, Merino M, Choyke P, Vocke C, Schmidt L, Isaacs JS, Glenn G, Toro J, Zbar B, Bottaro D, Neckers L. Genetic basis of cancer of the kidney: disease-specific approaches to therapy. Clin Cancer Res. 2004; 10:6282S–9S. https://doi.org/10.1158/1078-0432.CCR-050013 [PubMed]
- 3. Lopez-Beltran A, Scarpelli M, Montironi R, Kirkali Z. 2004 WHO classification of the renal tumors of the adults. Eur Urol. 2006; 49:798–805. https://doi.org/10.1016/j.eururo.2005.11.035 [PubMed]
- 4. Shingarev R, Jaimes EA. Renal cell carcinoma: new insights and challenges for a clinician scientist. Am J Physiol Renal Physiol. 2017; 313:F145–54. https://doi.org/10.1152/ajprenal.00480.2016 [PubMed]
- 5. Li N, Liu X. Phosphoglycerate Mutase 1: Its Glycolytic and Non-Glycolytic Roles in Tumor Malignant Behaviors and Potential Therapeutic Significance. Onco Targets Ther. 2020; 13:1787–95. https://doi.org/10.2147/OTT.S238920 [PubMed]
- 6. Liu L, Wang S, Zhang Q, Ding Y. Identification of potential genes/proteins regulated by Tiam1 in colorectal cancer by microarray analysis and proteome analysis. Cell Biol Int. 2008; 32:1215–22. https://doi.org/10.1016/j.cellbi.2008.07.004 [PubMed]
- 7. Ren F, Wu H, Lei Y, Zhang H, Liu R, Zhao Y, Chen X, Zeng D, Tong A, Chen L, Wei Y, Huang C. Quantitative proteomics identification of phosphoglycerate mutase 1 as a novel therapeutic target in hepatocellular carcinoma. Mol Cancer. 2010; 9:81. https://doi.org/10.1186/1476-4598-9-81 [PubMed]
- 8. Hitosugi T, Zhou L, Elf S, Fan J, Kang HB, Seo JH, Shan C, Dai Q, Zhang L, Xie J, Gu TL, Jin P, Alečković M, et al. Phosphoglycerate mutase 1 coordinates glycolysis and biosynthesis to promote tumor growth. Cancer Cell. 2012; 22:585–600. https://doi.org/10.1016/j.ccr.2012.09.020 [PubMed]
- 9. Li C, Shu F, Lei B, Lv D, Zhang S, Mao X. Expression of PGAM1 in renal clear cell carcinoma and its clinical significance. Int J Clin Exp Pathol. 2015; 8:9410–5. [PubMed]
- 10. Wang Z, Jiang H, Chen S, Du F, Wang X. The mitochondrial phosphatase PGAM5 functions at the convergence point of multiple necrotic death pathways. Cell. 2012; 148:228–43. https://doi.org/10.1016/j.cell.2011.11.030 [PubMed]
- 11. Sheng H, Tang W. Glycolysis Inhibitors for Anticancer Therapy: A Review of Recent Patents. Recent Pat Anticancer Drug Discov. 2016; 11:297–308. https://doi.org/10.2174/1574892811666160415160104 [PubMed]
- 12. Wang Y, Yin C, Geng L, Cai W. Immune Infiltration Landscape in Clear Cell Renal Cell Carcinoma Implications. Front Oncol. 2021; 10:491621. https://doi.org/10.3389/fonc.2020.491621 [PubMed]
- 13. Shen J, Gao H, Li B, Huang Y, Shi Y. The integration of machine learning and multi-omics analysis provides a powerful approach to screen aging-related genes and predict prognosis and immunotherapy efficacy in hepatocellular carcinoma. Aging (Albany NY). 2023; 15:6848–64. https://doi.org/10.18632/aging.204876 [PubMed]
- 14. Cheng M, Fan X, He M, Dai X, Liu X, Hong J, Zhang L, Liao L. Identification of an endoplasmic reticulum stress-related prognostic risk model with excellent prognostic and clinical value in oral squamous cell carcinoma. Aging (Albany NY). 2023; 15. https://doi.org/10.18632/aging.204983 [PubMed]
- 15. Melo Ferreira R, Sabo AR, Winfree S, Collins KS, Janosevic D, Gulbronson CJ, Cheng YH, Casbon L, Barwinska D, Ferkowicz MJ, Xuei X, Zhang C, Dunn KW, et al. Integration of spatial and single-cell transcriptomics localizes epithelial cell-immune cross-talk in kidney injury. JCI Insight. 2021; 6:e147703. https://doi.org/10.1172/jci.insight.147703 [PubMed]
- 16. Peng Z, Ye M, Ding H, Feng Z, Hu K. Spatial transcriptomics atlas reveals the crosstalk between cancer-associated fibroblasts and tumor microenvironment components in colorectal cancer. J Transl Med. 2022; 20:302. https://doi.org/10.1186/s12967-022-03510-8 [PubMed]
- 17. Tzeng YT, Tsui KH, Tseng LM, Hou MF, Chu PY, Sheu JJ, Li CJ. Integrated analysis of pivotal biomarker of LSM1, immune cell infiltration and therapeutic drugs in breast cancer. J Cell Mol Med. 2022; 26:4007–20. https://doi.org/10.1111/jcmm.17436 [PubMed]
- 18. Li CJ, Chang CH, Tsang YL, Fang SH, Chen SN, Chiang AJ. Prognostic significance of ferroptosis pathway gene signature and correlation with macrophage infiltration in cervical squamous cell carcinoma. Int Immunopharmacol. 2022; 112:109273. https://doi.org/10.1016/j.intimp.2022.109273 [PubMed]
- 19. Lee J, Kim Y, Jin S, Yoo H, Jeong S, Jeong E, Yoon S. Q-omics: Smart Software for Assisting Oncology and Cancer Research. Mol Cells. 2021; 44:843–50. https://doi.org/10.14348/molcells.2021.0169 [PubMed]
- 20. Li CJ, Lin HY, Ko CJ, Lai JC, Chu PY. A Novel Biomarker Driving Poor-Prognosis Liver Cancer: Overexpression of the Mitochondrial Calcium Gatekeepers. Biomedicines. 2020; 8:451. https://doi.org/10.3390/biomedicines8110451 [PubMed]
- 21. Wettersten HI, Aboud OA, Lara PN
Jr , Weiss RH. Metabolic reprogramming in clear cell renal cell carcinoma. Nat Rev Nephrol. 2017; 13:410–9. https://doi.org/10.1038/nrneph.2017.59 [PubMed] - 22. Zhu Z, Hu J, Chen Z, Feng J, Yang X, Liang W, Ding G. Transition of acute kidney injury to chronic kidney disease: role of metabolic reprogramming. Metabolism. 2022; 131:155194. https://doi.org/10.1016/j.metabol.2022.155194 [PubMed]
- 23. Chen TK, Knicely DH, Grams ME. Chronic Kidney Disease Diagnosis and Management: A Review. JAMA. 2019; 322:1294–304. https://doi.org/10.1001/jama.2019.14745 [PubMed]
- 24. Bahadoram S, Davoodi M, Hassanzadeh S, Bahadoram M, Barahman M, Mafakher L. Renal cell carcinoma: an overview of the epidemiology, diagnosis, and treatment. G Ital Nefrol. 2022; 39:2022–vol3. [PubMed]
- 25. Semenza GL. Hypoxia-inducible factors: mediators of cancer progression and targets for cancer therapy. Trends Pharmacol Sci. 2012; 33:207–14. https://doi.org/10.1016/j.tips.2012.01.005 [PubMed]
- 26. Qi X, Li Q, Che X, Wang Q, Wu G. The Uniqueness of Clear Cell Renal Cell Carcinoma: Summary of the Process and Abnormality of Glucose Metabolism and Lipid Metabolism in ccRCC. Front Oncol. 2021; 11:727778. https://doi.org/10.3389/fonc.2021.727778 [PubMed]
- 27. Zhao YY. Metabolomics in chronic kidney disease. Clin Chim Acta. 2013; 422:59–69. https://doi.org/10.1016/j.cca.2013.03.033 [PubMed]
- 28. Gronda E, Jessup M, Iacoviello M, Palazzuoli A, Napoli C. Glucose Metabolism in the Kidney: Neurohormonal Activation and Heart Failure Development. J Am Heart Assoc. 2020; 9:e018889. https://doi.org/10.1161/JAHA.120.018889 [PubMed]
- 29. Kalim S, Rhee EP. An overview of renal metabolomics. Kidney Int. 2017; 91:61–9. https://doi.org/10.1016/j.kint.2016.08.021 [PubMed]
- 30. Linehan WM, Ricketts CJ. The Cancer Genome Atlas of renal cell carcinoma: findings and clinical implications. Nat Rev Urol. 2019; 16:539–52. https://doi.org/10.1038/s41585-019-0211-5 [PubMed]
- 31. Tello D, Balsa E, Acosta-Iborra B, Fuertes-Yebra E, Elorza A, Ordóñez Á, Corral-Escariz M, Soro I, López-Bernardo E, Perales-Clemente E, Martínez-Ruiz A, Enríquez JA, Aragonés J, et al. Induction of the mitochondrial NDUFA4L2 protein by HIF-1α decreases oxygen consumption by inhibiting Complex I activity. Cell Metab. 2011; 14:768–79. https://doi.org/10.1016/j.cmet.2011.10.008 [PubMed]
- 32. Sudarshan S, Karam JA, Brugarolas J, Thompson RH, Uzzo R, Rini B, Margulis V, Patard JJ, Escudier B, Linehan WM. Metabolism of kidney cancer: from the lab to clinical practice. Eur Urol. 2013; 63:244–51. https://doi.org/10.1016/j.eururo.2012.09.054 [PubMed]
- 33. Courtney KD, Bezwada D, Mashimo T, Pichumani K, Vemireddy V, Funk AM, Wimberly J, McNeil SS, Kapur P, Lotan Y, Margulis V, Cadeddu JA, Pedrosa I, et al. Isotope Tracing of Human Clear Cell Renal Cell Carcinomas Demonstrates Suppressed Glucose Oxidation In Vivo. Cell Metab. 2018; 28:793–800.e2. https://doi.org/10.1016/j.cmet.2018.07.020 [PubMed]
- 34. Yang GJ, Tao F, Zhong HJ, Yang C, Chen J. Targeting PGAM1 in cancer: An emerging therapeutic opportunity. Eur J Med Chem. 2022; 244:114798. https://doi.org/10.1016/j.ejmech.2022.114798 [PubMed]
- 35. Jiang X, Sun Q, Li H, Li K, Ren X. The role of phosphoglycerate mutase 1 in tumor aerobic glycolysis and its potential therapeutic implications. Int J Cancer. 2014; 135:1991–6. https://doi.org/10.1002/ijc.28637 [PubMed]
- 36. Yang L, Dong Z, Li S, Chen T. ESM1 promotes angiogenesis in colorectal cancer by activating PI3K/Akt/mTOR pathway, thus accelerating tumor progression. Aging (Albany NY). 2023; 15:2920–36. https://doi.org/10.18632/aging.204559 [PubMed]
- 37. Shen X, Wang M, Chen W, Xu Y, Zhou Q, Zhu T, Wang G, Cai S, Han Y, Xu C, Wang W, Meng L, Sun H. Senescence-related genes define prognosis, immune contexture, and pharmacological response in gastric cancer. Aging (Albany NY). 2023; 15:2891–905. https://doi.org/10.18632/aging.204524 [PubMed]
- 38. Chiang AJ, Li CJ, Tsui KH, Chang C, Chang YI, Chen LW, Chang TH, Sheu JJ. UBE2C Drives Human Cervical Cancer Progression and Is Positively Modulated by mTOR. Biomolecules. 2020; 11:37. https://doi.org/10.3390/biom11010037 [PubMed]
- 39. Moi D, Bonanni D, Belluti S, Linciano P, Citarella A, Franchini S, Sorbi C, Imbriano C, Pinzi L, Rastelli G. Discovery of potent pyrrolo-pyrimidine and purine HDAC inhibitors for the treatment of advanced prostate cancer. Eur J Med Chem. 2023; 260:115730. https://doi.org/10.1016/j.ejmech.2023.115730 [PubMed]
- 40. Ehteda A, Khan A, Rajakumar G, Vanniasinghe AS, Gopalakrishnan A, Liu J, Tsoli M, Ziegler DS. Microtubule-targeting combined with HDAC inhibition is a novel therapeutic strategy for Diffuse Intrinsic Pontine Gliomas. Mol Cancer Ther. 2023. [Epub ahead of print]. https://doi.org/10.1158/1535-7163.MCT-23-0179 [PubMed]
- 41. Liu YS, Chang YC, Kuo WW, Chen MC, Wang TF, Chen TS, Lin YM, Li CC, Liao PH, Huang CY. Calreticulin nuclear translocalization alleviates CaM/CaMKII/CREB signaling pathway to enhance chemosensitivity in HDAC inhibitor-resistant hepatocellular carcinoma cells. Aging (Albany NY). 2022; 14:5097–115. https://doi.org/10.18632/aging.204131 [PubMed]
- 42. Zewail M, E Gaafar PM, Ali MM, Abbas H. Lipidic cubic-phase leflunomide nanoparticles (cubosomes) as a potential tool for breast cancer management. Drug Deliv. 2022; 29:1663–74. https://doi.org/10.1080/10717544.2022.2079770 [PubMed]
- 43. Lee SO, Kwak AW, Lee MH, Seo JH, Cho SS, Yoon G, Chae JI, Joo SH, Shim JH. Picropodophyllotoxin Induces G1 Cell Cycle Arrest and Apoptosis in Human Colorectal Cancer Cells via ROS Generation and Activation of p38 MAPK Signaling Pathway. J Microbiol Biotechnol. 2021; 31:1615–23. https://doi.org/10.4014/jmb.2109.09012 [PubMed]