Introduction
The male abnormal gene family 21 (mab21), was first identified in C. elegans [1]. Since its identification, mab21L1 gene was found to control development of both eye and brain [2–9]. Moreover, it also has a role in regulating axis and dorsal-ventral patterning as well as heart and liver development [3, 4, 10]. Mutations of the human mab21L1 gene cause numerous ocular diseases including ocular coloboma, microcornea and cataract, neural defects, skeletal dysplasia and intellectual disability [7, 9, 11]. However, the functional mechanism of the mab21L1 gene remains largely unknown [8, 12].
Apoptosis is one of the major causes for ocular diseases [13–16]. In lens system, we initially demonstrated that induced lens epithelial cell apoptosis appears to be a common cellular mechanism mediating stress-induced, non-congenital cataractogenesis [17–19]. Subsequently, a number of laboratories have confirmed that lens epithelial cell apoptosis is indeed actively involved in lens pathogenesis as demonstrated from in vivo animal model studies [20–25]. Moreover, transgenic studies with overexpression of various exogenous genes or disruption of several endogenous genes caused apoptosis followed by cataractogenesis or small eye during lens development [26–33].
In lens epithelial cells, apoptosis is mainly mediated by p53 and its downstream targets [26, 30, 33–49]. Bcl-2 family proteins play a crucial role in mediating endogenous apoptotic pathway [50–54], and among which Bak is an important pro-apoptotic protein [55–59] and Mcl-1 is a major anti-apoptotic regulator [60–63].
αB-crystallin is initially known as major lens structural protein that plays an essential role in maintaining the transparency of the ocular lens [64]. Later, αB-crystallin is found to be a member of the small heat shock protein family [65] and act as a molecular chaperone [66–73] and also displays autokinase activity [74, 75]. As a strong antiapoptotic regulator, αB-crystallin is initially shown to protect cells from osmotic [76], thermal [77] and oxidative insult [78]. Later, αB-crystallin is found to prevent induced apoptosis by various factors including staurosporine [54, 79–81], TNF [79, 81], UVA irradiation [81, 82], okadaic acid [38] and hydrogen peroxide [51]. Regarding the antiapoptotic mechanism, previous studies from our laboratory and others have demonstrated that αB-crystallin can directly interact with multiple targets including members of the caspase and Bcl-2 families as well as the tumor suppressor p53 to suppress apoptosis [38, 54, 82–89].
In the present study, we present evidence to show that the mab21L1 gene has a role in promoting survival of lens epithelial cells. Mechanistically, overexpression of Mab21L1 upregulates expression of αB-crystallin. Moreover, we demonstrate that αB-crystallin prevents p53 phosphorylation at multiple sites through abrogating activation of the ATR and CHK1 kinases. As a result of suppressing p53 activity by αB-crystallin, Mab21L1 downregulates expression of Bak but upregulates Mcl-1 during okadaic acid treatment. It is well established that p53 plays an important role in regulating aging [90–92]. By regulating p53 activity, Mab21L1 may be involved in control of aging. Taken together, our results demonstrate that Mab21L1 promotes survival of lens epithelial cells through upregulation of αB-crystallin to suppress ATR/CHK1/p53 pathway.
Results
Establishment of the stable lens epithelial cells expressing vector and Mab21L1
To explore the function of Mab21L1, we have overexpressed the human Mab21L1 cDNA in mouse lens epithelial cells αTN4-1 using the vector p3X-FLAG-CMV-10-Mab21L1 [with cDNA inserted in EcoRI (5’) and Kpn I (3’)] (Supplementary Figure 1). As shown in Figure 1A, 1B, αTN4-1 cells have very low level of endogenous Mab21L1, and overexpression of human Mab21L1 lead to over 2-fold upregulation of Mab21L1. Thus, we established two stable lines: vector-transfected cells, p3X-FLAG-CMV-10-αTN4-1 (Vector-αTN4-1 in short), and Mab21L1-transfected cells, p3X-FLAG-CMV-10-Mab21L1-αTN4-1 (Mab21L1-αTN4-1 in short). The clone 1 from both vector- and Mab21L1-transfected cells were used in most related experimental studies described below.
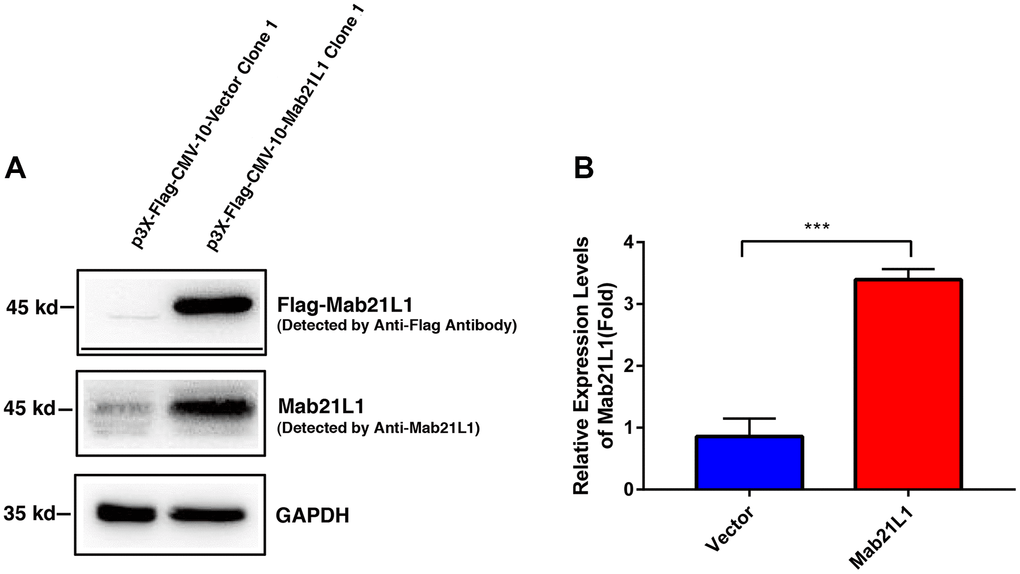
Figure 1. Establishment of vector and MAB21L1 overexpression clones with mouse lens epithelial cells, αTN4-1 line. The plasmids of P3X-Flag-CMV-10-Vector and P3X-Flag-CMV-10-MAB21L1 were transfected into αTN4-1 cells, respectively. After transfection, the P3X-Flag-CMV-10-Vector-αTN4-1 (Vector-αTN4-1 in short) cells and P3X-Flag-CMV-10-MAB21L1-αTN4-1 (MAB21L1-αTN4-1 in short) cells were screened with 600 μg/ml G418 for 4 weeks, and individual clones were obtained. Clone 1 of vector-αTN4-1 and MAB21L1-αTN4-1 cells were confirmed by Western blot analysis (A). (B) Quantitative results of the MAB21L1 protein expression levels in A by Image J software. N=3. *** p<0.001.
Mab21L1 promotes survival of lens epithelial cells treated with okadaic acid
Next, we tested if Mab21L1 expression cells display any property in resisting stresses upon exposure to phosphatase inhibitor, okadaic acid [38]. Both vector- and Mab21L1-transfected cells were treated with 100 nM okadaic acid for 0 to 24 hours. As shown in Figure 2, cells expressing Mab21L1 were much resistant against 100 nM okadaic acid than vector-transfected cells. After 24-hour treatment by 100 nM OA, over 40% vector-transfected cells underwent apoptosis. In contrast, less than 20% cells were induced to undergo apoptosis (Figure 2A, 2C). The apoptotic nature was confirmed with live/dead assay in which the live cells were stained in green color and the dead cells were stained in red (Figure 2B). Thus, Mab21L1 can promote survival of the transfected lens epithelial cells. Mab21L1 also displayed clear protection against UVA irradiation-induced apoptosis (data not shown).
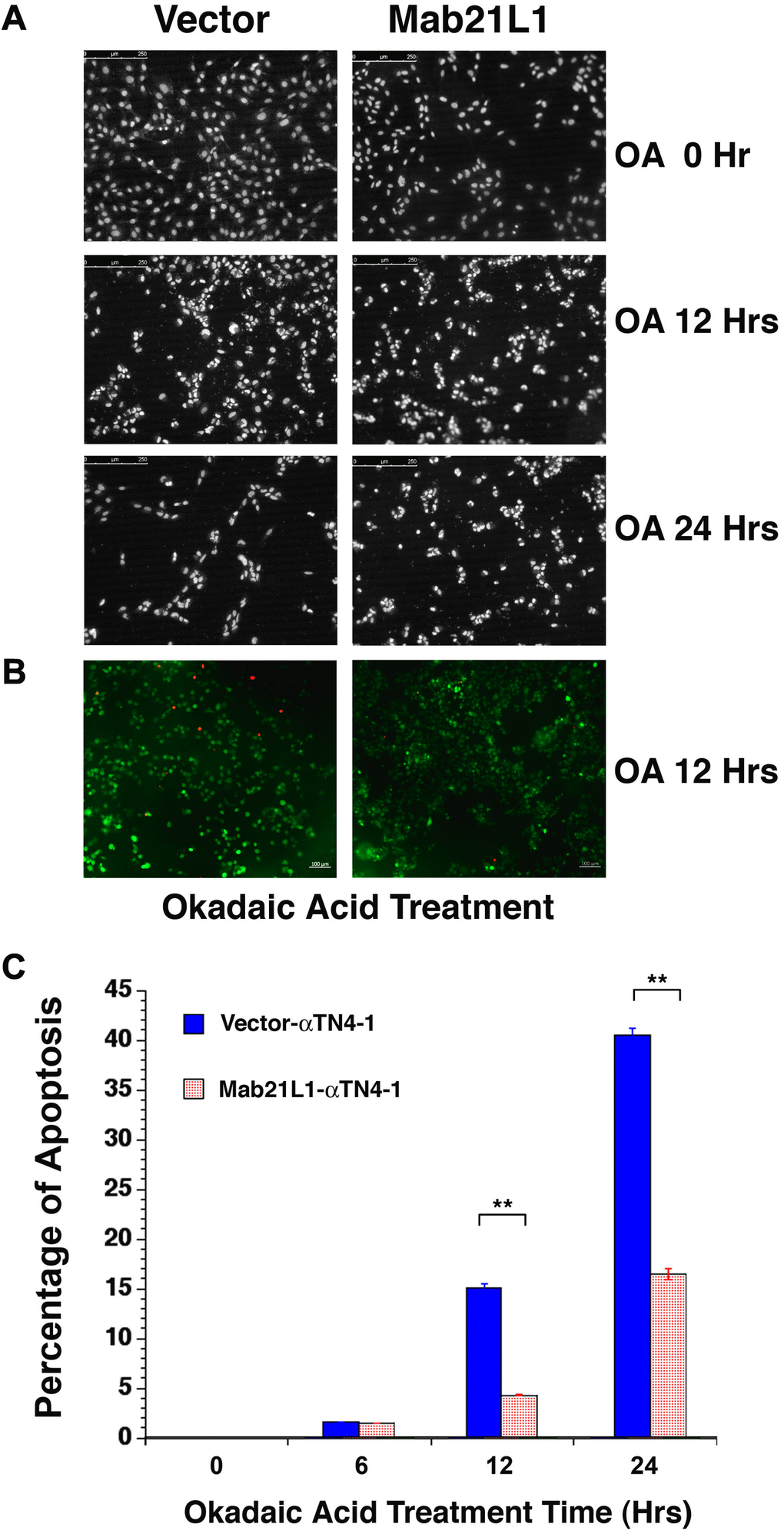
Figure 2. Analysis of okadaic acid (OA)-induced apoptosis of vector-αTN4-1 (vector) and MAB21L1-αTN4-1 (Mab21L1) cells. (A) Hoechst staining of OA-treated vector-αTN4-1 and MAB21L1-αTN4-1 cells for 0 to 24 hours. The apoptotic cells displayed fragmented or condensed nuclei, or were detached from the culture dishes, so that empty space appeared. Both vector-αTN4-1 and MAB21L1-αTN4-1 cells were grown to the density as shown in row one of Figure 2A (approximately 90% confluence, 0 Hr treatment), then subjected to 100 nM okadaic acid (OA) treatment with for 12 and 24 hrs. After OA treatment, the cells were processed for Hoechst staining as described (Mao et al., 2001). (B) Image of live cells (green color) and apoptotic cells (read color) of the vector-αTN4-1 and MAB21L1-αTN4-1 cells after 12 Hrs treatment by OA. (C) Quantitative results of apoptosis rate using live/dead assay as described (Wang et al., 2021). OA treatment for 12 hrs and 24 hrs induced about 15% and 41% apoptosis in vector-transfected cells, respectively. In contrast, only about 5% and 16% apoptosis were detected in MAB21L1-transfected cells after 12h and 24h-treatment with 100 nM OA. Note that MAB21L1 displayed the anti-apoptotic ability in αTN4-1 cells. Scale bar, 250 μm. N=3. ** p<0.01.
Mab21L1 upregulates αB-crystallin during OA-induced apoptosis
To explore how Mab21L1 could promote survival of lens epithelial cells against stress conditions including the okadaic acid and UVA irradiation, we examined expression of αB-crystallin during treatment by okadaic acid. As shown in Figure 3, cells expressing Mab21L1 displayed enhanced expression of αB-crystallin. Moreover, during okadaic acid treatment, in Mab21L1 expression cells, OA-induced downregulation of αB-crystallin was much slower than that in vector-transfected cells. Thus, Mab21L1 expression upregulates αB-crystallin, a small heat shock protein having strong antiapoptotic ability [38, 51, 54, 82–89].
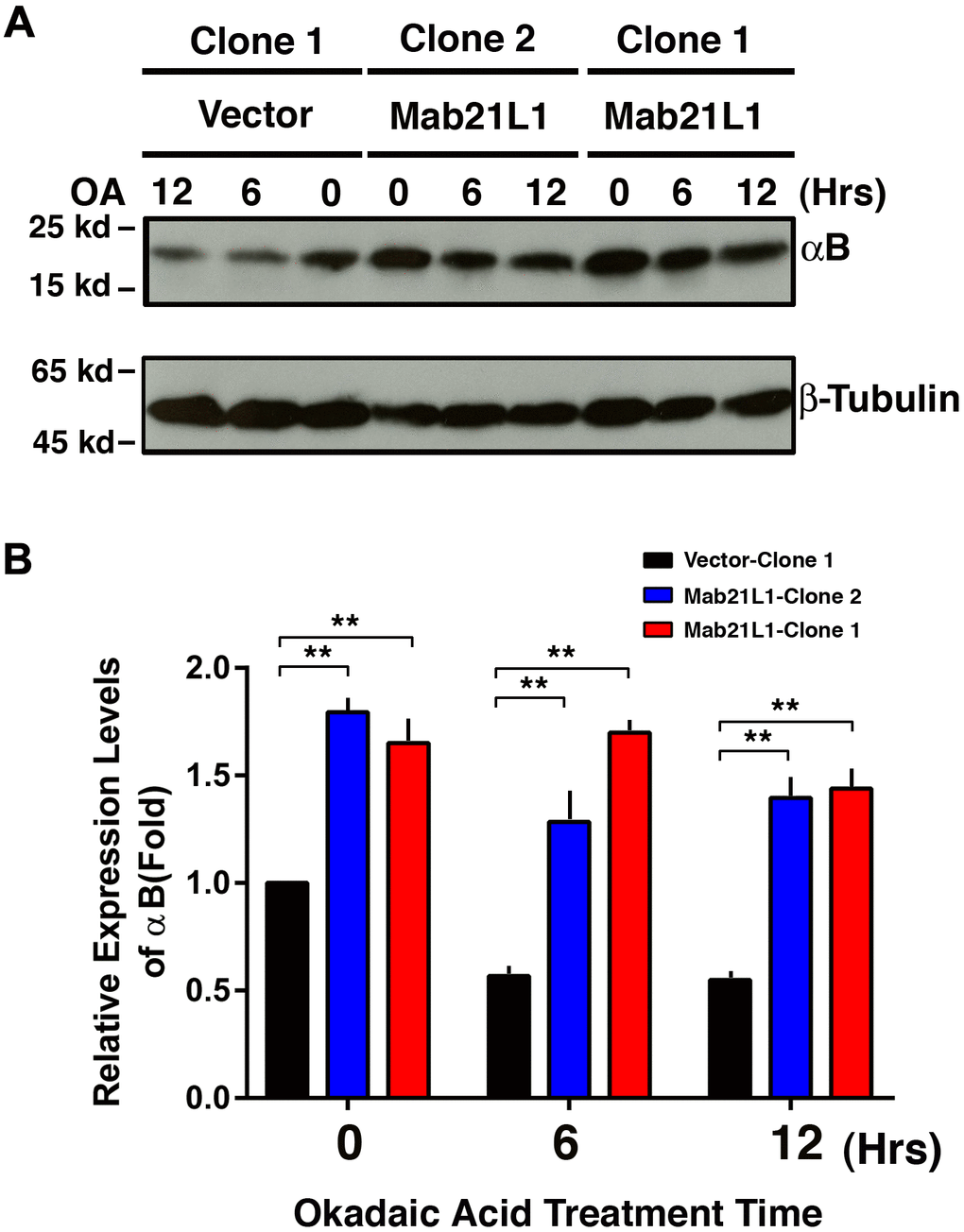
Figure 3. Upregulation of αB-crystallin in Mab21L1-αTN4-1 cells and its attenuated degradation during okadaic acid (OA)-induced apoptosis of the Mab21L1-αTN4-1 cells. Both vector-αTN4-1 and MAB21L1-αTN4-1 cells were grown to about 90% confluence and then subjected to 100 nM OA treatment for 12 and 24 hrs. Thereafter, the cells were harvested for extraction of total proteins which were used for analysis of αB-crystallin expression by Western blot analysis (A). Quantitative results of the αB-crystallin protein expression levels were analyzed by Image J software (B). Note that in the MAB21L1-αTN4-1 cell clones, the αB-crystallin protein expression level was much higher than that in the vector-αTN4-1 cell clone in the absence of 100 nM OA treatment. During OA treatment for 12 and 24 hrs, the degradation of αB-crystallin protein was much slower in MAB21L1-αTN4-1 cell clones than that in vector-αTN4-1 clone. N=3. ** p<0.01.
αB-crystallin prevents UVA-induced phosphorylation of p53 at S-20 and S-37
We and others have previously shown that αB-crystallin can prevent apoptosis through regulation of multiple targets [38, 51, 54, 82–86]. To further explore the possible mechanisms how αB-crystallin prevents apoptosis, we examined the effects of αB-crystallin on p53 phosphorylation at S-20 and S-37. As shown in Figure 4, in vector-transfected cells (pEGFP-HLE), UVA irradiation induces significant hyperphosphorylation of p53 at S-20 and S-37. However, in αB-crystallin expression cells (pEGFP-HαB-HLE), UVA irradiation-induced hyperphosphorylation of p53 was much reduced. Thus, αB-crystallin can prevent UVA-induced hyperphosphorylation of p53 at S-20 and S-37.
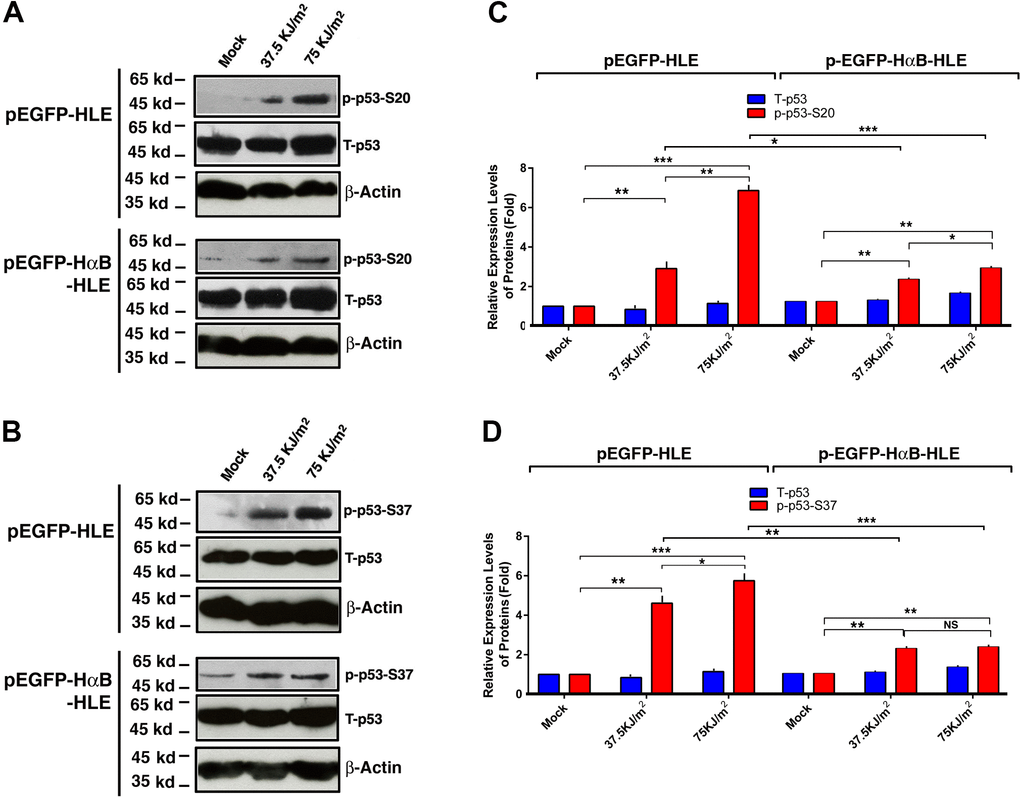
Figure 4. αB-crystallin inhibited p53 hyperphosphorylation under UVA irradiation. Both pEGFP-HLE and pEGFP-HαB-HLE cells (Mao et al., 2004) were grown to 90% confluence, then subjected to mock irradiation, 37.5 KJ/m2 and 75 KJ/m2 UVA irradiation, respectively. The irradiated cells were harvested for extraction of total proteins which were used for analysis of total p53 (T-p53) (A, B), phosphorylated p53 at S-20 (p-p53-S20) (A) and phosphorylated p53 at S-37 (p-p53-S37) (B) by Western blot analysis. Quantitative results of the T-p53, p-p53-S20 (C) and p-p53-S37 (D) levels were analyzed by Image J software. Note that UVA-induced much stronger p53 activity (phosphorylation at S-20 and S-37) in pEGFP-HLE cells than that in pEGFP-HαB-HLE cells. N=3. NS, not significant; *p<0.05, ** p<0.01, *** p<0.001.
αB-crystallin prevents UVA-induced activation of check kinase 1 (CHK1)
To explore how αB-crystallin prevents UVA-induced hyperphosphorylation of p53 at S-20 and S-37, we examined if αB-crystallin has any effect on the upstream kinase, check kinase 1 (CHK1). Previous studies have shown that CHK1 can phosphorylate p53 at these sites [93, 94]. As shown in Figure 5, UVA irradiation induced activation of CHK1 (as reflected by the enhanced phosphorylation at S-345) in vector-transfected cells. In αB-crystallin expression cells, however, activation of CHK1 was largely abrogated. Thus, αB-crystallin is capable of suppressing UVA irradiation-induced activation of CHK1.
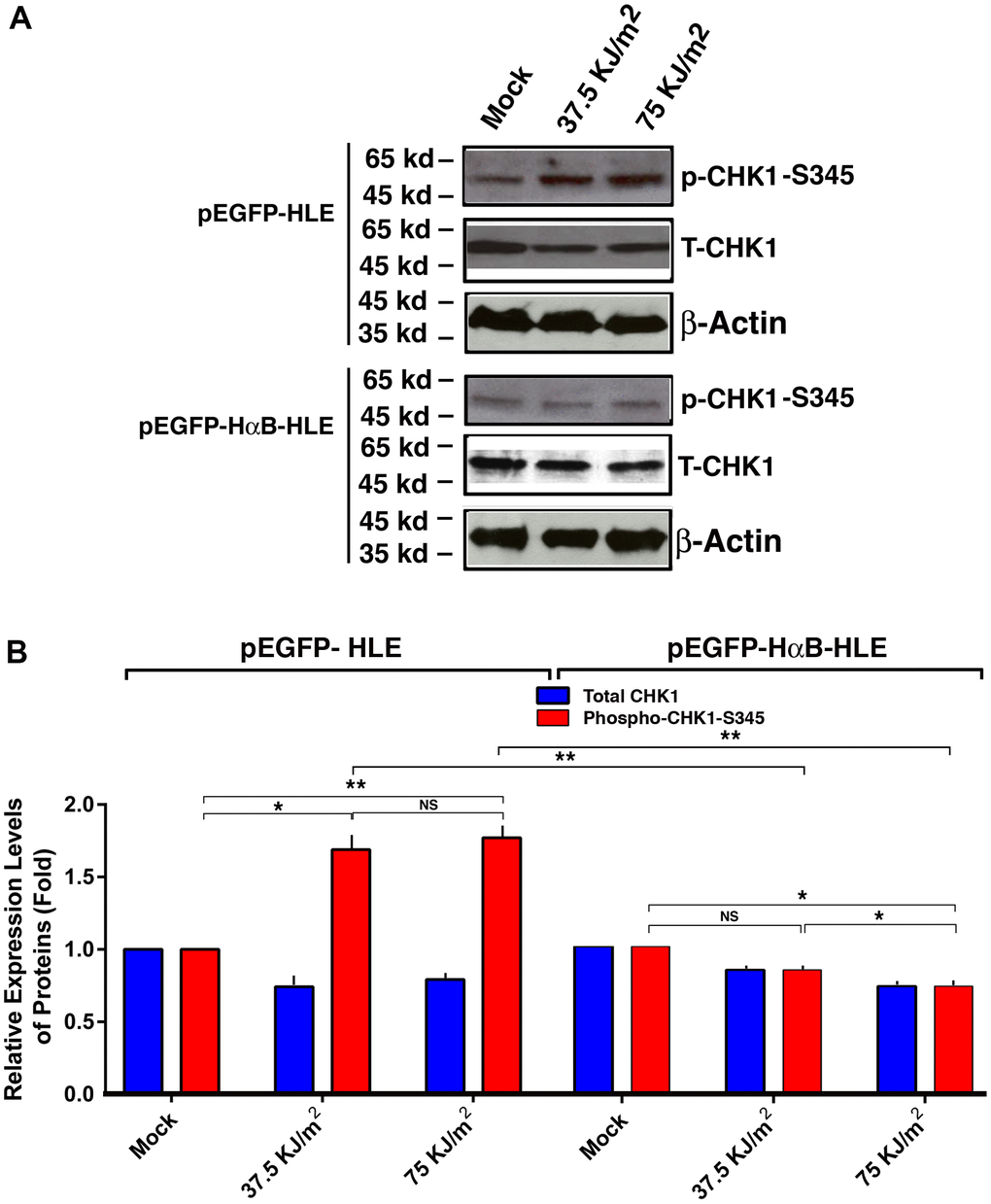
Figure 5. αB-crystallin inhibited CHK1 activation under the UVA irradiation. Both pEGFP-HLE and pEGFP-HαB-HLE cells (Mao et al., 2004) were grown to 90% confluence, then subjected to mock irradiation, 37.5 KJ/m2 and 75 KJ/m2 UVA irradiation, respectively. The irradiated cells were harvested for extraction of total proteins which were used for analysis of total CHK1 (T-CHK1, 54 kd), phosphorylated CHK1 at S-345 and β-Actin (loading control) levels (A) by Western blot analysis. Quantitative results of the T-CHK1 and p-CHK1-S345 against β-actin (loading control) levels in pEGFP-HLE cells and pEGFP-HαB-HLE cells (B) were analyzed by Image J software. Note that UVA induced significant upregulation of CHK1 activity (as reflected by phosphorylation at S345) in pEGFP-HLE cells. In contrast, in pEGFP-HαB-HLE cells, both total CHK1 and phosphorylated CHK1 at S345 were downregulated under UVA irradiation. N=3. NS, not significant. *p<0.05, ** p<0.01.
αB-crystallin prevents UVA-induced activation of ATR kinase
Since CHK1 is activated by the upstream kinase, ATR [95–98], we next examined if αB-crystallin has any effect on the upstream ATR kinase. As shown in Figure 6, UVA irradiation induced activation of ATR kinase (also reflected by the enhanced phosphorylation at S-428 in vector-transfected cells). In αB-crystallin expression cells, however, activation of ATR was totally abrogated. Thus, αB-crystallin is also capable of suppressing UVA irradiation-induced activation of ATR kinase.
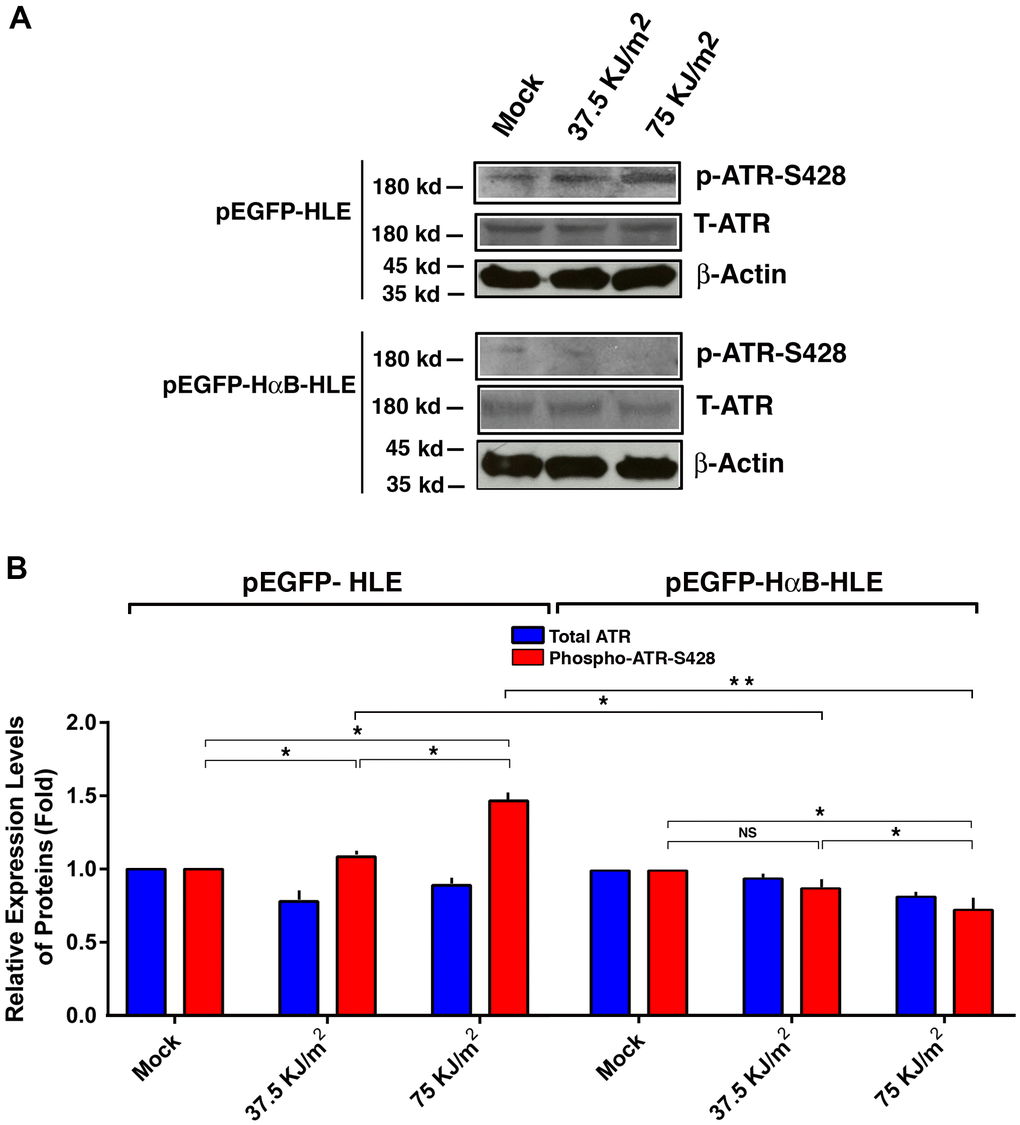
Figure 6. αB-crystallin inhibited ATR activation under the UVA irradiation. Both pEGFP-HLE and pEGFP-HαB-HLE cells (Mao et al., 2004) were grown to 90% confluence, then subjected to mock irradiation, 37.5 KJ/m2 and 75 KJ/m2 UVA irradiation, respectively. The irradiated cells were harvested for extraction of total proteins which were used for analysis of total ATR (T-ATR, 300 kd), phosphorylated ATR at S-428 and β-Actin (loading control) by Western blot analysis (A). Quantitative results of the T-ATR, and p-ATR-S428 levels against loading control in pEGFP-HLE cells and pEGFP-HαB-HLE cells (B) were analyzed by Image J software. Note that UVA induced significant upregulation of ATR activity (as reflected by phosphorylation at S428) in pEGFP-HLE cells. In contrast, in pEGFP-HαB-HLE cells, both total ATR and phosphorylated ATR at S428 were downregulated under UVA irradiation. N=3. NS, not significant. *p<0.05.
Mab21L1 downregulates Bak during OA-induced apoptosis and silence of Bak attenuates OA-induced apoptosis
Since Mab21L1 may prevent p53 hyperphosphorylation through upregulation of αB-crystallin, we next tested if Mab21L1 have any effect on the downstream targets of p53 during okadaic acid-induced apoptosis. As shown in Figure 7A, 7B, in Mab21L1 expression cells, Bak expression was relatively higher than that in vector transfected cells. However, during okadaic acid treatment, we observed that Bak was upregulated in vector-transfected cells. However, in Mab21L1 expression cells, okadaic acid-induced upregulation of Bak was abrogated. Next, we silenced Bak expression in mouse lens epithelial cells, αTN4-1 using CRISPR/Cas9 technology [49] (Figure 7C). As shown in Figure 7D, silence of Bak expression significantly attenuated okadaic acid-induced apoptosis. Thus, Mab21L1 can downregulate Bak level to suppress okadaic acid-induced apoptosis.
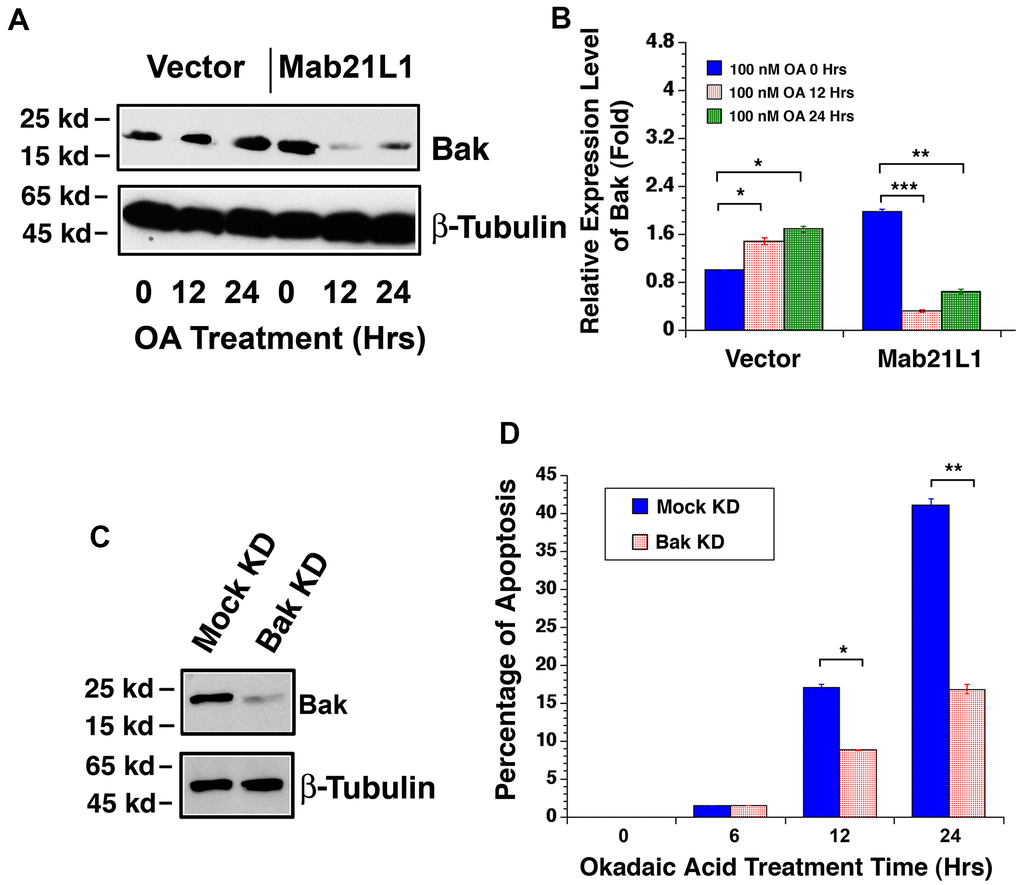
Figure 7. Dynamic change of Bak levels in vector-αTN4-1 and MAB21L1-αTN4-1 cells in the absence and presence of 100 OA treatment and effect of Bak level on OA-induced apoptosis of αTN4-1 cells. (A, B). Dynamic change of Bak levels in vector-αTN4-1 and MAB21L1-αTN4-1 cells in the absence and presence of 100 OA treatment. Both vector-αTN4-1 and MAB21L1-αTN4-1 cells were grown to about 90% confluence and then subjected to 100 nM OA treatment for 12 and 24 hrs. Thereafter, the cells were harvested for extraction of total proteins which were used for analysis of Bak expression by Western blot analysis (A). Quantitative results of the Bak protein expression levels were analyzed by Image J software (B). Note that in the MAB21L1-αTN4-1 cells, the Bak protein expression level was much higher than that in the vector-αTN4-1 cell clone in the absence of 100 nM OA treatment. During OA treatment for 12 and 24 hrs, however, Bak protein was upregulated in vector-αTN4-1 cells. In contrast, in MAB21L1-αTN4-1 cells, Bak protein was significantly degraded. N=3. *p<0.05, ** p<0.01, *** p<0.001. (C, D) Effect of Bak level on OA-induced apoptosis of αTN4-1 cells. Vector-αTN4-1 cells were used as Bak knockdown with CRSPR/Cas9 technology (see Materials and Methods). Both mock and Bak KD clones were verified with Western blot analysis (C). The two types of cells were then subjected to 100 nM OA treatment for 12 and 24 hrs, and the apoptosis rate was determined with live/dead assays (D). N=3. *p<0.05, ** p<0.01.
Mab21L1 promotes Mcl-1 expression during OA-induced apoptosis and overexpression of Mcl-1 suppresses OA-induced apoptosis
To further understand how Mab21L1 regulates survival of lens epithelial cells, we examined the dynamic change of another Bcl-2 family member, the mcl-1 gene encoding an important anti-apoptotic regulator [60–63]. A previous study reported that Mcl-1 expression is related to p53 activity in a reverse relationship [99]. As shown in Figure 8A, 8B, in Mab21L1 expression cells, Mcl-1 expression was slightly lower than that in vector transfected cells. However, during okadaic acid treatment, we observed that Mcl-1 was downregulated in vector-transfected cells. However, in Mab21L1 expression cells, okadaic acid induced upregulation of Mcl-1. Next, we overexpressed Mcl-1 in mouse lens epithelial cells αTN4-1 using the vector, pCI-Mcl-1 (Figure 8C). As shown in Figure 8D, overexpression of Mcl-1 significantly suppressed OA-induced apoptosis. Thus, Mab21L1 can upregulate Mcl-1 level to attenuate OA-induced apoptosis.
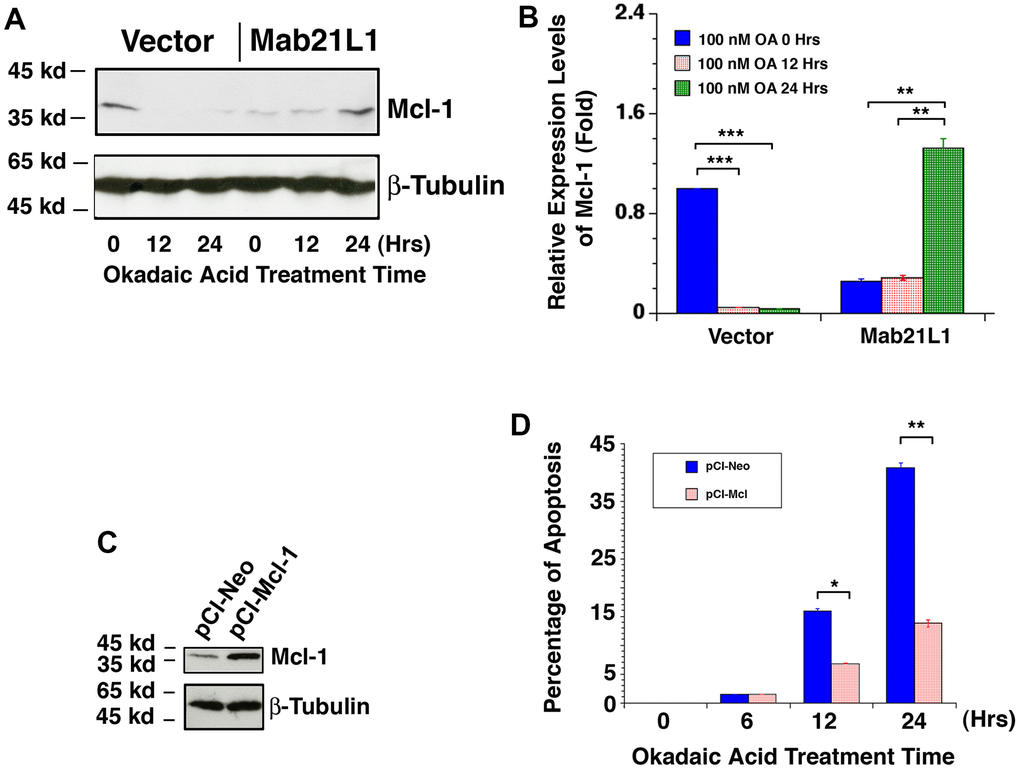
Figure 8. Dynamic change of Mcl-1 levels in vector-αTN4-1 and MAB21L1-αTN4-1 cells in the absence and presence of 100 OA treatment and effect of Mcl-1 level on OA-induced apoptosis of αTN4-1 cells. (A, B) Dynamic change of Mcl-1 levels in vector-αTN4-1 and MAB21L1-αTN4-1 cells in the absence and presence of 100 OA treatment. Both vector-αTN4-1 and MAB21L1-αTN4-1 cells were grown to about 90% confluence and then subjected to 100 nM OA treatment for 12 and 24 hrs. Thereafter, the cells were harvested for extraction of total proteins which were used for analysis of Mcl-1 expression by Western blot analysis (A). Quantitative results of the Mcl-1 protein expression levels were analyzed by Image J software (B). Note that in the MAB21L1-αTN4-1 cells, the Mcl-1 protein expression level was much lower than that in the vector-αTN4-1 cell clone in the absence of 100 nM OA treatment. During OA treatment for 12 and 24 hrs, however, Mcl-1 protein was down-regulated to background level in vector-αTN4-1 cells. In contrast, in MAB21L1-αTN4-1 cells, Mcl-1 protein was significantly upregulated after 24 h treatment by OA. N=3. ** p<0.01, *** p<0.001. (C, D) Effect of Mcl-1 level on OA-induced apoptosis of αTN4-1 cells. Both pCI-αTN4-1 and pCI-Mcl-1-αTN4-1 cell clones were verified with Western blot analysis (C). The two types of pCI-αTN4-1 and pCI-Mcl-1-αTN4-1 cells were then subjected to 100 nM OA treatment for 12 and 24 hrs, and the apoptosis rate was determined with live/dead assays (D). N=3. *p<0.05, ** p<0.01.
Discussion
In the present study, we have demonstrated the followings: 1) Overexpression of human mab21L1 gene cDNA in mouse lens epithelial cells, αTN4-1 significantly promotes survival of lens epithelial cells under treatment by okadaic acid; 2) Overexpression of human mab21L1 gene cDNA in αTN4-1 cells upregulates expression of αB-crystallin gene; 3) αB-crystallin prevents UVA-induced hyperphosphorylation of the tumor suppressor p53 at S-20 and S-37; 4) αB-crystallin inhibits UVA-induced activation of the check kinase 1 (CHK1) and its upstream kinase ATR; 5) Overexpression of human mab21L1 gene cDNA in αTN4-1 cells downregulates the pro-apoptotic factor Bak but upregulates expression of the pro-survival regulator Mcl−1 during okadaic acid treatment. Together, our results demonstrate that MAB21L1 promotes survival of lens epithelial cells through upregulation of αB-crystallin to suppress ATR/CHK1/p53 pathway. (Figure 9).
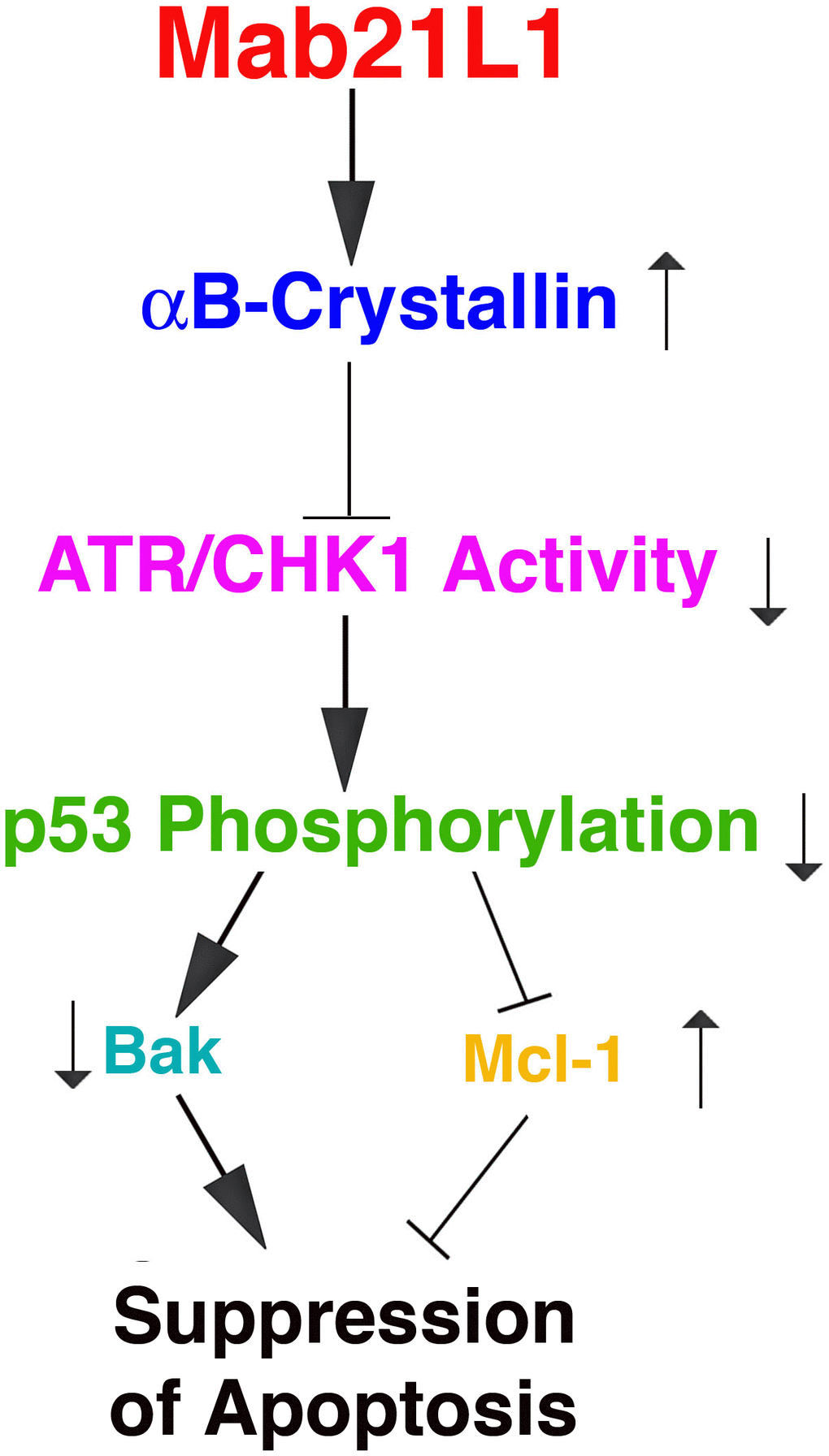
Figure 9. Diagram to show the mechanism mediating Mab21L1 promotion of survival of lens epithelial cells. Mab21L1 promotes survival of lens epithelial cells through upregulation of αB-crystallin to suppress the ATR/CHK1/p53 pathway and thus downregulate Bak expression but enhance Mcl-1 expression. As a result, MAB21L1 can suppress stress (okadaic acid or UVA)-induced apoptosis.
Mab21L1 positively regulates αB-crystallin to promote survival of lens epithelial cells
The mab21 gene was first identified as a cell fate determination gene that regulates sensory ray morphogenesis in male C. elegans [1]. Later it was found that Mab21L1 is a lens lineage-specific transcription factor [100]. It has an important role in regulating lens development [5, 7–9, 11]. Mutations in human Mab21L1 gene causes aberrations in lens ectoderm morphogenesis and lead to congenital cerebellar, ocular, craniofacial and genital (COFG) syndrome [101, 102]. The ocular abnormalities include microphthalmia, coloboma and cataracts [7, 9]. Similar to human mab21L1 gene mutations, Mab21L1-deficient mice display severe cell-autonomous defects in lens placode invagination due to impaired cell proliferation and survival [5] and other deficiency [6, 101]. Using mouse lens epithelial cells, αTN4-1 and the stable lines expressing P3X-Flag-CMV-10 vector, or human mab21L1 gene cDNA, here we demonstrated that human mab21L1 gene cDNA-expression lens epithelial cells are much resistant against stress condition of okadaic acid treatment than the vector-transfected cells (Figure 2). Mechanistically, we demonstrated that Mab21L1 is capable of upregulating αB-crystallin, a strong anti-apoptotic regulator (See below discussion for more details).
How might Mab21L1 up-regulates αB-crystallin? In a recent study using single cell sequencing strategy, Yamada et al. (2021) [8] demonstrated that in Mab21L1 knockout mice, 61 genes were upregulated and 131 genes were downregulated in comparison with the wild type mice. Among these down-regulated genes are two transcription factors: Maf and Sox2 [8]. Since previous studies have shown that both Maf and Sox2 can positively regulates expression of αB-crystallin [100, 103, 104], it is plausible that Mab21L1 may upregulate αB-crystallin through Maf and Sox-2. Alternatively, Mab21L1 may directly bind to the promoter of αB-crystallin gene to control its expression. Whether this is the case is currently under investigation.
αB-crystallin represses ATR/CHK1/p53 pathway to inhibit apoptosis
As an anti-apoptotic regulator, αB-crystallin can prevent cells from induced-apoptosis in several mechanisms based on the studies from us and several other laboratories [38, 51, 54, 82–89]. First, it can suppress the maturation of the members of the caspase family [38, 51, 83–84]. Second, it can also interact with members of the Bcl-2 family including Bax and Bcl-Xs to prevent their translocation into mitochondria [54]. Third, αB-crystallin was found to negatively regulate the Ras/Raf/MEK/ERK signaling pathway to prevent UVA-induced apoptosis [40, 82].
Previous studies have shown that αB-crystallin can also bind to the tumor suppressor, p53 [105]. However, how does αB-crystallin modulate p53 activity remains unknown. Here, using stable lines of human lens epithelial cells HLE expressing pEGFP-vector, or human αB-crystallin gene cDNA, we demonstrated that UVA induced hyperphosphorylation of p53 at S-20 and S-37 in pEGFP-HLE cells (Figure 4A–4D). This UVA-induced p53 hyperphosphorylation is significantly reduced in αB-crystallin expression cells, pEGFP-HαB-HLE (Figure 4A–4D). At present, seventeen phosphorylation sites have been identified [92]. Whether the remaining 15 phosphorylation sites besides S-20 and S-37 can be induced by UVA and whether αB-crystallin also has strong impact on the phosphorylation of the remaining phosphorylation sites remains to be investigated. Nevertheless, our study demonstrates that αB-crystallin can modulate p53 phosphorylation status at S-20 and S-37 to inhibit its activity.
It has been well established that p53 can be phosphorylated by check kinase 1 (CHK1) at S-20 and S-37 [93, 94]. Our results also showed that while in vector-transfected cells (pEGFP-HLE), UVA irradiation increased CHK1 activity, in αB-crystallin expression cells (pEGFP-HαB-HLE), however, both CHK1 protein level and activity were down-regulated under UVA irradiation (Figure 5). Since CHK1 is activated by ATR kinase [95–98], we also examined the differential activation of ATR in vector- and HαB-transfected cells. Our results demonstrated that while in vector-transfected cells, pEGFP-HLE, UVA induced upregulation of ATR activity from 20% to 50% under irradiation with 37.5KJ/m2 to 75 KJ/m2, respectively; in αB-crystallin expression pEGFP-HαB-HLE cells, in contrast, both ATR protein level and activity were slightly down-regulated under UVA irradiation of both doses (Figure 6). Together, out results demonstrate that αB-crystallin can repress ATR/CHK1/p53 pathway to inhibit p53-mediated apoptosis.
αB-crystallin suppression of ATR/CHK1/p53 contributes to Mab21L1 promotion of survival of lens epithelial cells
In the present study, we also observed that in Mab21L1 expression cells, expression of Bak is relatively higher than that in vector-transfected cells (Figure 7), and Mcl-1 expression, on the other hand, is relatively lower than that in vector-transfected cells (Figure 8). However, Mab21L1-induced upregulation of αB-crystallin can override the Bak upregulation and Mcl-1 downregulation to promote survival of lens epithelial cells in the absence of stress conditions.
Under okadaic acid treatment, however, we observed that Bak expression was upregulated and Mcl-1 expression was down-regulated in vector-transfected cells. In contrast, in Mab21L1 expression cells, Bak expression was significantly down-regulated and Mcl-1 was significantly upregulated (Figures 7A, 7B, 8A, 8B). Moreover, when Bak was knocked down or Mcl-1 was overexpressed, okadaic acid-induced apoptosis of αTN4-1 cells was significant inhibited (>50%, Figures 7C, 7D, 8C, 8D). This OA-induced Bak downregulation and Mcl-1 upregulation in Mab21L1 expression cells is closely related with its promotion of αB-crystallin expression. Under the same okadaic acid treatment condition, OA induced significant downregulation of αB-crystallin in vector-transfected cells (Figure 3). In Mab21L1 expression cells, however, OA-induced much less downregulation of αB-crystallin (Figure 3). Thus, Mab21L1-promoted upregulation of αB-crystallin through suppression of ATR/CHK1/p53 pathway, can promote survival of lens epithelial cells (Figure 9).
Materials and Methods
Materials
Various molecular biology reagents were purchased from Invitrogen Life Technologies (Gaithersburg, MD, USA). All the oligos were purchased from Sangon Biotech Co., Ltd. (Shanghai, China). Protein size markers were purchased from GenStar Co., Ltd. (Beijing, China). Various antibodies were obtained from Cell Signaling Technology (Boston, MA, USA); Abcam Inc. (Cambridge, MA, USA); Santa Cruz Biotechnology, Inc. (Dallas, TX, USA); Sigma-Aldrich (St. Louis, MO, USA); Proteintech Co., Ltd. (Wuhan, China); Ray Biotech Co., Ltd. (Beijing, China).
Cell culture, plasmid construction and establishment of gene overexpression or knockout stable cell lines
Mouse lens epithelial cell line αTN4-1 and human lens epithelial cells (HLE) were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal bovine serum (FBS; Atlanta Biologicals) and 1% penicillin/streptomycin in 5% CO2 at 37° C as described before [42, 49, 54, 59, 106, 107].
The CRISPR/Cas9-based gene KO vector pSp Cas9(BB)-2A-Puro (PX459) is a gift from Dr. Mengqing Xiang in Zhongshan Ophthalmic Center of Sun Yat-sen University. The sgRNA sequences used for Bak gene KO were: 5’-CAAGTTGTCCATCTCGGGGTTGG-3’ (target 1) and 5’ TCTTCACCAAGATCGCCTCCAGG 3’ (target 2) as described before [49]. The sgRNA was inserted into PX459 using the BbsI restriction sites. For expression vector, the construction of full-length cDNAs of Mab21L1 or Mcl-1 were cloned by PCR. The Mab21L1 cDNA was inserted into the expression vector, p3xFlag-CMV-10-Mab21L1, at the EcoRI and KpnI sites. The Mcl-1 cDNA was subcloned into the pCI-Neo Vector, at the EcoR1 and Sal I sites.
For establishment of stable cell lines, αTN4-1 cells were transfected with the above plasmids using Lipofectamine 3000 (Life Technologies) according to the manufacturer’s instructions. Forty-eight hours after transfection, cells stably expressing the plasmids were selected by 1.0 μg/mL puromycin for PX459, and 600 μg/ml G418 for p3×FLAG-CMV-10 and pCI-Neo. About 4 weeks, individual clones for the stable cell lines were established and confirmed by western blot analysis and DNA sequencing [49, 106, 107].
pEGFP-HLE and pEGFP-HαB-HLE were established as described before [54, 106]. These cells were cultured in DMEM supplemented with 10% fetal bovine serum (FBS; Atlanta Biologicals), 600 μg/ml G418 and 1% penicillin/streptomycin in 5% CO2 at 37° C.
UVA irradiation
The UVA irradiation was conducted with similar facility described previously [82], which produces an energy level of approximately 1 mW cm−2 S−1 with the wavelength ranging from 320 to 400 nm. The amount of UVA light reaching the lens is between 0.1 and 1 mW cm−2 [108]. The total energy received by the lens epithelial cells was 37.5 kJ m−2 (3.75 J cm−2), or 75 kJ m−2 (7.5 J cm−2). Briefly, the irradiation was conducted with an uncovered 100 mm culture dish containing 100% confluence HLE cell with 10 ml DMEM plus 10% FBS. We conducted UVA irradiation in the presence of culture medium to avoid the cellular responses to nutritional shock (for example, withdrawal of growth factors in serum). Under the present irradiation condition, UVA may elicit production of hydrogen peroxide according to a previous study [108]. A dose of 75 kJ m−2 corresponds to the amount of UVA which our naked skin receives in approximately 1.5 hours of exposure to sunshine in a typical summer day (90° C) in August at noon time.
Protein extraction and Western blot analysis
Total proteins were extracted by RIPA buffer (1% NP-40, 1% sodium deoxycholate, 0.1% SDS, 50 mM Tris-HCl PH8.0, 150 mM NaCl) supplemented with the Protease Inhibitor Cocktail, then cell lysates were sonicated and centrifuged at 13000×rpm for 10 min at 4° C as described [106, 107]. The supernatants were transferred to new tubes. Fifty micrograms of total proteins in each sample was separated by 10% or 12 % SDS-polyacrylamide gel and transferred to PVDF membranes. The protein blots were blocked with 5% nonfat milk in TBST (10 mM Tris HCl, pH8.0, 150 mM NaCl, 0.05% Tween-20) and further incubated with primary antibodies overnight at 4° C. Primary antibodies used in western blot were shown in Supplementary Table 1. The horseradish peroxidase-conjugated secondary antibodies (CST; 7077 and 7074) were then applied for one hour at room temperature. Immunoreactivity was detected with a chemilluminescence detection kit (ECL Ultra; New Cell and Molecular Biotech Co., Ltd.) and the blots were visualized using a Tanon chemiluminescence system (China).
Apoptosis assays
Cell apoptosis was determined by Hoechst staining [38, 51] and Live/Dead Viability/Cytotoxicity [49]. Cells were seeded into 60 mm petri dish (for Hoechst staining) or into 6-well plates about 90% confluence, and then treated with 100 nM Okadaic acid for different time as indicated in the figures to induce cell apoptosis. The apoptotic nature of the treated cells was further verified by Hoechst staining as previously described [51]. The Live/Dead Viability/Cytotoxicity Kit (L3224; Thermo Fisher) was used to distinguish live and dead cells [49]. Live cells are characterized by the presence of ubiquitous intracellular esterase activity, and revealed by calcein AM. The polyanionic dye calcein is well retained within live cells, producing an intense uniform green fluorescence in live cells. EthD-1 enters cells with damaged membranes and displays red fluorescence upon binding to nucleic acids, thereby producing a bright red color in dead cells and EthD-1 is excluded by the membrane of live cells. The images were captured with a Zeiss microscope.
Statistics
All results shown are reported as the mean ± standard deviation (SD). Significance was calculated using the unpaired two-tailed t test. Differences were considered statistically significant at P < 0.05.
Data availability statement
All data are available upon reasonable request.
Author Contributions
YX, ZXH and DW-CL designed research; YX, JWX, QG, YYB, ZXH, LW, XHH performed the experiments; YX, JWX, QG, YYB, ZXH and DW-CL analyzed the data; DW-CL wrote the paper.
Acknowledgments
We thank Dr. Mengqing Xiang for the CRISPR/Cas9-based gene KO vector pSp Cas9(BB)-2A-Puro (PX459). This study was supported in part by the Grants from the National Natural Science Foundation of China (nos. 81770910 and 8216067), the Basic Research Project (ZK[2021]-427) from the Guizhou Provincial Science and Technology Projects. We thank Dr. Paul Russel for the αTN4-1 mouse lens epithelial cell line. We thank all members of David Li’s Laboratory in the State Key Laboratory of Ophthalmology in Zhongshan Ophthalmic Center of Sun Yat-sen University.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This study was supported in part by the grants from the National Natural Science Foundation of China (nos. 81770910 and 8216067), the Basic Research Project (ZK[2021]-427) from the Guizhou Provincial Science and Technology Projects.
Editorial Note
This corresponding author has a verified history of publications using a personal email address for correspondence.
References
- 1. Baird SE, Fitch DH, Kassem IA, Emmons SW. Pattern formation in the nematode epidermis: determination of the arrangement of peripheral sense organs in the C. elegans male tail. Development. 1991; 113:515–26. https://doi.org/10.1242/dev.113.2.515 [PubMed]
- 2. Margolis RL, Stine OC, McInnis MG, Ranen NG, Rubinsztein DC, Leggo J, Brando LV, Kidwai AS, Loev SJ, Breschel TS, Callahan C, Simpson SG, DePaulo JR, et al. cDNA cloning of a human homologue of the Caenorhabditis elegans cell fate-determining gene mab-21: expression, chromosomal localization and analysis of a highly polymorphic (CAG)n trinucleotide repeat. Hum Mol Genet. 1996; 5:607–16. https://doi.org/10.1093/hmg/5.5.607 [PubMed]
- 3. Mariani M, Corradi A, Baldessari D, Malgaretti N, Pozzoli O, Fesce R, Martinez S, Boncinelli E, Consalez GG. Mab21, the mouse homolog of a C. elegans cell-fate specification gene, participates in cerebellar, midbrain and eye development. Mech Dev. 1998; 79:131–5. https://doi.org/10.1016/s0925-4773(98)00180-4 [PubMed]
- 4. Mariani M, Baldessari D, Francisconi S, Viggiano L, Rocchi M, Zappavigna V, Malgaretti N, Consalez GG. Two murine and human homologs of mab-21, a cell fate determination gene involved in Caenorhabditis elegans neural development. Hum Mol Genet. 1999; 8:2397–406. https://doi.org/10.1093/hmg/8.13.2397 [PubMed]
- 5. Yamada R, Mizutani-Koseki Y, Hasegawa T, Osumi N, Koseki H, Takahashi N. Cell-autonomous involvement of Mab21l1 is essential for lens placode development. Development. 2003; 130:1759–70. https://doi.org/10.1242/dev.00399 [PubMed]
- 6. Nguyen D, Yamada R, Yoshimitsu N, Oguri A, Kojima T, Takahashi N. Involvement of the Mab21l1 gene in calvarial osteogenesis. Differentiation. 2017; 98:70–8. https://doi.org/10.1016/j.diff.2017.11.001 [PubMed]
- 7. Seese SE, Deml B, Muheisen S, Sorokina E, Semina EV. Genetic disruption of zebrafish mab21l1 reveals a conserved role in eye development and affected pathways. Dev Dyn. 2021; 250:1056–73. https://doi.org/10.1002/dvdy.312 [PubMed]
- 8. Yamada R, Oguri A, Fujiki K, Shirahige K, Hirate Y, Kanai-Azuma M, Takezoe H, Akimoto Y, Takahashi N, Kanai Y. MAB21L1 modulates gene expression and DNA metabolic processes in the lens placode. Dis Model Mech. 2021; 14:dmm049251. https://doi.org/10.1242/dmm.049251 [PubMed]
- 9. Seese SE, Reis LM, Deml B, Griffith C, Reich A, Jamieson RV, Semina EV. Identification of missense MAB21L1 variants in microphthalmia and aniridia. Hum Mutat. 2021; 42:877–90. https://doi.org/10.1002/humu.24218 [PubMed]
- 10. Sridharan J, Haremaki T, Jin Y, Teegala S, Weinstein DC. Xmab21l3 mediates dorsoventral patterning in Xenopus laevis. Mech Dev. 2012; 129:136–46. https://doi.org/10.1016/j.mod.2012.05.002 [PubMed]
- 11. Wong RL, Chow KL. Depletion of Mab21l1 and Mab21l2 messages in mouse embryo arrests axial turning, and impairs notochord and neural tube differentiation. Teratology. 2002; 65:70–7. https://doi.org/10.1002/tera.10018 [PubMed]
- 12. Huang ZX, Xiang JW, Zhou L, Nie Q, Wang L, Chen ZG, Hu XH, Xiao Y, Qing WJ, Liu YF, Sun Q, Tang XC, Liu FY, et al. The Male Abnormal Gene Family 21 (Mab21) Members Regulate Eye Development. Curr Mol Med. 2016; 16:660–7. https://doi.org/10.2174/1566524016666160824150729 [PubMed]
- 13. Lolley RN. The rd gene defect triggers programmed rod cell death. The Proctor Lecture. Invest Ophthalmol Vis Sci. 1994; 35:4182–91. [PubMed]
- 14. Papermaster DS, Windle J. Death at an early age. Apoptosis in inherited retinal degenerations. Invest Ophthalmol Vis Sci. 1995; 36:977–83. [PubMed]
- 15. Li DWC. The lens epithelium, apoptosis and cataract formation. In Eye Lens Epithelium: Damaging Mechanisms and Lens Transparency. (Glaesser, Hockwin and Vrensen, eds). Chapter 8, pp81–108. Deutsche Akademic der Naturforscher Leopoldina (German Academy of Natural Sciences). Halle. 1997.
- 16. Yan Q, Liu JP, Li DW. Apoptosis in lens development and pathology. Differentiation. 2006; 74:195–211. https://doi.org/10.1111/j.1432-0436.2006.00068.x [PubMed]
- 17. Li WC, Kuszak JR, Dunn K, Wang RR, Ma W, Wang GM, Spector A, Leib M, Cotliar AM, Weiss M. Lens epithelial cell apoptosis appears to be a common cellular basis for non-congenital cataract development in humans and animals. J Cell Biol. 1995; 130:169–81. https://doi.org/10.1083/jcb.130.1.169 [PubMed]
- 18. Li WC, Spector A. Lens epithelial cell apoptosis is an early event in the development of UVB-induced cataract. Free Radic Biol Med. 1996; 20:301–11. https://doi.org/10.1016/0891-5849(96)02050-3 [PubMed]
- 19. Li WC, Kuszak JR, Wang GM, Wu ZQ, Spector A. Calcimycin-induced lens epithelial cell apoptosis contributes to cataract formation. Exp Eye Res. 1995; 61:91–8. https://doi.org/10.1016/s0014-4835(95)80062-x [PubMed]
- 20. Michael R, Vrensen GF, van Marle J, Gan L, Söderberg PG. Apoptosis in the rat lens after in vivo threshold dose ultraviolet irradiation. Invest Ophthalmol Vis Sci. 1998; 39:2681–7. [PubMed]
- 21. Pandya U, Saini MK, Jin GF, Awasthi S, Godley BF, Awasthi YC. Dietary curcumin prevents ocular toxicity of naphthalene in rats. Toxicol Lett. 2000; 115:195–204. https://doi.org/10.1016/s0378-4274(00)00191-0 [PubMed]
- 22. Tamada Y, Fukiage C, Nakamura Y, Azuma M, Kim YH, Shearer TR. Evidence for apoptosis in the selenite rat model of cataract. Biochem Biophys Res Commun. 2000; 275:300–6. https://doi.org/10.1006/bbrc.2000.3298 [PubMed]
- 23. Yoshizawa K, Oishi Y, Nambu H, Yamamoto D, Yang J, Senzaki H, Miki H, Tsubura A. Cataractogenesis in neonatal Sprague-Dawley rats by N-methyl-N-nitrosourea. Toxicol Pathol. 2000; 28:555–64. https://doi.org/10.1177/019262330002800408 [PubMed]
- 24. Ye J, Yao K, Lu D, Wu R, Jiang H. Low power density microwave radiation induced early changes in rabbit lens epithelial cells. Chin Med J (Engl). 2001; 114:1290–4. [PubMed]
- 25. Takamura Y, Kubo E, Tsuzuki S, Akagi Y. Apoptotic cell death in the lens epithelium of rat sugar cataract. Exp Eye Res. 2003; 77:51–7. https://doi.org/10.1016/s0014-4835(03)00083-6 [PubMed]
- 26. Morgenbesser SD, Williams BO, Jacks T, DePinho RA. p53-dependent apoptosis produced by Rb-deficiency in the developing mouse lens. Nature. 1994; 371:72–4. https://doi.org/10.1038/371072a0 [PubMed]
- 27. Yoshida H, Kong YY, Yoshida R, Elia AJ, Hakem A, Hakem R, Penninger JM, Mak TW. Apaf1 is required for mitochondrial pathways of apoptosis and brain development. Cell. 1998; 94:739–50. https://doi.org/10.1016/s0092-8674(00)81733-x [PubMed]
- 28. Gómez Lahoz E, Liegeois NJ, Zhang P, Engelman JA, Horner J, Silverman A, Burde R, Roussel MF, Sherr CJ, Elledge SJ, DePinho RA. Cyclin D- and E-dependent kinases and the p57(KIP2) inhibitor: cooperative interactions in vivo. Mol Cell Biol. 1999; 19:353–63. https://doi.org/10.1128/MCB.19.1.353 [PubMed]
- 29. Blixt A, Mahlapuu M, Aitola M, Pelto-Huikko M, Enerbäck S, Carlsson P. A forkhead gene, FoxE3, is essential for lens epithelial proliferation and closure of the lens vesicle. Genes Dev. 2000; 14:245–54. https://doi.org/10.1101/gad.14.2.245 [PubMed]
- 30. Hettmann T, Barton K, Leiden JM. Microphthalmia due to p53-mediated apoptosis of anterior lens epithelial cells in mice lacking the CREB-2 transcription factor. Dev Biol. 2000; 222:110–23. https://doi.org/10.1006/dbio.2000.9699 [PubMed]
- 31. Liu Y, Zacksenhaus E. E2F1 mediates ectopic proliferation and stage-specific p53-dependent apoptosis but not aberrant differentiation in the ocular lens of Rb deficient fetuses. Oncogene. 2000; 19:6065–73. https://doi.org/10.1038/sj.onc.1203996 [PubMed]
- 32. de Iongh RU, Lovicu FJ, Overbeek PA, Schneider MD, Joya J, Hardeman ED, McAvoy JW. Requirement for TGFbeta receptor signaling during terminal lens fiber differentiation. Development. 2001; 128:3995–4010. https://doi.org/10.1242/dev.128.20.3995 [PubMed]
- 33. Wang WL, Li Q, Xu J, Cvekl A. Lens fiber cell differentiation and denucleation are disrupted through expression of the N-terminal nuclear receptor box of NCOA6 and result in p53-dependent and p53-independent apoptosis. Mol Biol Cell. 2010; 21:2453–68. https://doi.org/10.1091/mbc.e09-12-1031 [PubMed]
- 34. Nakamura T, Pichel JG, Williams-Simons L, Westphal H. An apoptotic defect in lens differentiation caused by human p53 is rescued by a mutant allele. Proc Natl Acad Sci USA. 1995; 92:6142–6. https://doi.org/10.1073/pnas.92.13.6142 [PubMed]
- 35. Pan H, Griep AE. Temporally distinct patterns of p53-dependent and p53-independent apoptosis during mouse lens development. Genes Dev. 1995; 9:2157–69. https://doi.org/10.1101/gad.9.17.2157 [PubMed]
- 36. Li DW, Fass U, Huizar I, Spector A. Okadaic acid-induced lens epithelial cell apoptosis requires inhibition of phosphatase-1 and is associated with induction of gene expression including p53 and bax. Eur J Biochem. 1998; 257:351–61. https://doi.org/10.1046/j.1432-1327.1998.2570351.x [PubMed]
- 37. Pomerantz J, Schreiber-Agus N, Liégeois NJ, Silverman A, Alland L, Chin L, Potes J, Chen K, Orlow I, Lee HW, Cordon-Cardo C, DePinho RA. The Ink4a tumor suppressor gene product, p19Arf, interacts with MDM2 and neutralizes MDM2’s inhibition of p53. Cell. 1998; 92:713–23. https://doi.org/10.1016/s0092-8674(00)81400-2 [PubMed]
- 38. Li DW, Xiang H, Mao YW, Wang J, Fass U, Zhang XY, Xu C. Caspase-3 is actively involved in okadaic acid-induced lens epithelial cell apoptosis. Exp Cell Res. 2001; 266:279–91. https://doi.org/10.1006/excr.2001.5223 [PubMed]
- 39. Yao K, Wang K, Xu W, Sun Z, Shentu X, Qiu P. Caspase-3 and its inhibitor Ac-DEVD-CHO in rat lens epithelial cell apoptosis induced by hydrogen in vitro. Chin Med J (Engl). 2003; 116:1034–8. [PubMed]
- 40. Li DW, Liu JP, Mao YW, Xiang H, Wang J, Ma WY, Dong Z, Pike HM, Brown RE, Reed JC. Calcium-activated RAF/MEK/ERK signaling pathway mediates p53-dependent apoptosis and is abrogated by alpha B-crystallin through inhibition of RAS activation. Mol Biol Cell. 2005; 16:4437–53. https://doi.org/10.1091/mbc.e05-01-0010 [PubMed]
- 41. Cang Y, Zhang J, Nicholas SA, Bastien J, Li B, Zhou P, Goff SP. Deletion of DDB1 in mouse brain and lens leads to p53-dependent elimination of proliferating cells. Cell. 2006; 127:929–40. https://doi.org/10.1016/j.cell.2006.09.045 [PubMed]
- 42. Li DW, Liu JP, Schmid PC, Schlosser R, Feng H, Liu WB, Yan Q, Gong L, Sun SM, Deng M, Liu Y. Protein serine/threonine phosphatase-1 dephosphorylates p53 at Ser-15 and Ser-37 to modulate its transcriptional and apoptotic activities. Oncogene. 2006; 25:3006–22. https://doi.org/10.1038/sj.onc.1209334 [PubMed]
- 43. Ayala M, Strid H, Jacobsson U, Söderberg PG. p53 expression and apoptosis in the lens after ultraviolet radiation exposure. Invest Ophthalmol Vis Sci. 2007; 48:4187–91. https://doi.org/10.1167/iovs.06-0660 [PubMed]
- 44. Hu WF, Gong L, Cao Z, Ma H, Ji W, Deng M, Liu M, Hu XH, Chen P, Yan Q, Chen HG, Liu J, Sun S, et al. αA- and αB-crystallins interact with caspase-3 and Bax to guard mouse lens development. Curr Mol Med. 2012; 12:177–87. https://doi.org/10.2174/156652412798889036 [PubMed]
- 45. Liu FY, Tang XC, Deng M, Chen P, Ji W, Zhang X, Gong L, Woodward Z, Liu J, Zhang L, Sun S, Liu JP, Wu K, et al. The tumor suppressor p53 regulates c-Maf and Prox-1 to control lens differentiation. Curr Mol Med. 2012; 12:917–28. https://doi.org/10.2174/156652412802480835 [PubMed]
- 46. Gao M, Huang Y, Wang L, Huang M, Liu F, Liao S, Yu S, Lu Z, Han S, Hu X, Qu Z, Liu X, Assefa Yimer T, et al. HSF4 regulates lens fiber cell differentiation by activating p53 and its downstream regulators. Cell Death Dis. 2017; 8:e3082. https://doi.org/10.1038/cddis.2017.478 [PubMed]
- 47. Tang X, Chen Z, Deng M, Wang L, Nie Q, Xiang JW, Xiao Y, Yang L, Liu Y, Li DW. The Sumoylation Modulated Tumor Suppressor p53 Regulates Cell Cycle Checking Genes to Mediate Lens Differentiation. Curr Mol Med. 2018; 18:556–65. https://doi.org/10.2174/1566524019666190111154450 [PubMed]
- 48. Nie Q, Chen H, Zou M, Wang L, Hou M, Xiang JW, Luo Z, Gong XD, Fu JL, Wang Y, Zheng SY, Xiao Y, Gan YW, et al. The E3 Ligase PIAS1 Regulates p53 Sumoylation to Control Stress-Induced Apoptosis of Lens Epithelial Cells Through the Proapoptotic Regulator Bax. Front Cell Dev Biol. 2021; 9:660494. https://doi.org/10.3389/fcell.2021.660494 [PubMed]
- 49. Wang L, Zhang L, Gong XD, Fu JL, Gan YW, Hou M, Nie Q, Xiang JW, Xiao Y, Wang Y, Zheng SY, Yang L, Chen H, et al. PP-1β and PP-2Aα modulate cAMP response element-binding protein (CREB) functions in aging control and stress response through de-regulation of αB-crystallin gene and p300-p53 signaling axis. Aging Cell. 2021; 20:e13458. https://doi.org/10.1111/acel.13458 [PubMed]
- 50. Gross A, McDonnell JM, Korsmeyer SJ. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 1999; 13:1899–911. https://doi.org/10.1101/gad.13.15.1899 [PubMed]
- 51. Mao YW, Xiang H, Wang J, Korsmeyer S, Reddan J, Li DW. Human bcl-2 gene attenuates the ability of rabbit lens epithelial cells against H2O2-induced apoptosis through down-regulation of the alpha B-crystallin gene. J Biol Chem. 2001; 276:43435–45. https://doi.org/10.1074/jbc.M102195200 [PubMed]
- 52. Cory S, Adams JM. The Bcl2 family: regulators of the cellular life-or-death switch. Nat Rev Cancer. 2002; 2:647–56. https://doi.org/10.1038/nrc883 [PubMed]
- 53. Czabotar PE, Lessene G, Strasser A, Adams JM. Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol. 2014; 15:49–63. https://doi.org/10.1038/nrm3722 [PubMed]
- 54. Mao YW, Liu JP, Xiang H, Li DW. Human alphaA- and alphaB-crystallins bind to Bax and Bcl-X(S) to sequester their translocation during staurosporine-induced apoptosis. Cell Death Differ. 2004; 11:512–26. https://doi.org/10.1038/sj.cdd.4401384 [PubMed]
- 55. Chittenden T, Harrington EA, O’Connor R, Flemington C, Lutz RJ, Evan GI, Guild BC. Induction of apoptosis by the Bcl-2 homologue Bak. Nature. 1995; 374:733–6. https://doi.org/10.1038/374733a0 [PubMed]
- 56. Farrow SN, White JH, Martinou I, Raven T, Pun KT, Grinham CJ, Martinou JC, Brown R. Cloning of a bcl-2 homologue by interaction with adenovirus E1B 19K. Nature. 1995; 374:731–3. https://doi.org/10.1038/374731a0 [PubMed]
- 57. Kiefer MC, Brauer MJ, Powers VC, Wu JJ, Umansky SR, Tomei LD, Barr PJ. Modulation of apoptosis by the widely distributed Bcl-2 homologue Bak. Nature. 1995; 374:736–9. https://doi.org/10.1038/374736a0 [PubMed]
- 58. Singh G, Guibao CD, Seetharaman J, Aggarwal A, Grace CR, McNamara DE, Vaithiyalingam S, Waddell MB, Moldoveanu T. Structural basis of BAK activation in mitochondrial apoptosis initiation. Nat Commun. 2022; 13:250. https://doi.org/10.1038/s41467-021-27851-y [PubMed]
- 59. Deng M, Chen P, Liu F, Fu S, Tang H, Fu Y, Xiong Z, Hui S, Ji W, Zhang X, Zhang L, Gong L, Hu X, et al. The p53-Bak apoptotic signaling axis plays an essential role in regulating differentiation of the ocular lens. Curr Mol Med. 2012; 12:901–16. https://doi.org/10.2174/156652412802480899 [PubMed]
- 60. Reynolds JE, Yang T, Qian L, Jenkinson JD, Zhou P, Eastman A, Craig RW. Mcl-1, a member of the Bcl-2 family, delays apoptosis induced by c-Myc overexpression in Chinese hamster ovary cells. Cancer Res. 1994; 54:6348–52. [PubMed]
- 61. Zhou P, Qian L, Kozopas KM, Craig RW. Mcl-1, a Bcl-2 family member, delays the death of hematopoietic cells under a variety of apoptosis-inducing conditions. Blood. 1997; 89:630–43. https://doi.org/10.1182/blood.V89.2.630 [PubMed]
- 62. Rinkenberger JL, Horning S, Klocke B, Roth K, Korsmeyer SJ. Mcl-1 deficiency results in peri-implantation embryonic lethality. Genes Dev. 2000; 14:23–7. https://doi.org/10.1101/gad.14.1.23 [PubMed]
- 63. Cleveland AH, Romero-Morales A, Azcona LA, Herrero M, Nikolova VD, Moy S, Elroy-Stein O, Gama V, Gershon TR. Oligodendrocytes depend on MCL-1 to prevent spontaneous apoptosis and white matter degeneration. Cell Death Dis. 2021; 12:1133. https://doi.org/10.1038/s41419-021-04422-z [PubMed]
- 64. Bloemendal H. Lens proteins. CRC Crit Rev Biochem. 1982; 12:1–38. https://doi.org/10.3109/10409238209105849 [PubMed]
- 65. Ingolia TD, Craig EA. Four small Drosophila heat shock proteins are related to each other and to mammalian alpha-crystallin. Proc Natl Acad Sci USA. 1982; 79:2360–4. https://doi.org/10.1073/pnas.79.7.2360 [PubMed]
- 66. Horwitz J. Alpha-crystallin can function as a molecular chaperone. Proc Natl Acad Sci USA. 1992; 89:10449–53. https://doi.org/10.1073/pnas.89.21.10449 [PubMed]
- 67. Rao PV, Horwitz J, Zigler JS
Jr . Alpha-crystallin, a molecular chaperone, forms a stable complex with carbonic anhydrase upon heat denaturation. Biochem Biophys Res Commun. 1993; 190:786–93. https://doi.org/10.1006/bbrc.1993.1118 [PubMed] - 68. Boyle D, Takemoto L. Characterization of the alpha-gamma and alpha-beta complex: evidence for an in vivo functional role of alpha-crystallin as a molecular chaperone. Exp Eye Res. 1994; 58:9–15. https://doi.org/10.1006/exer.1994.1190 [PubMed]
- 69. Wang K, Spector A. The chaperone activity of bovine alpha crystallin. Interaction with other lens crystallins in native and denatured states. J Biol Chem. 1994; 269:13601–8. https://doi.org/10.1016/S0021-9258(17)36872-2 [PubMed]
- 70. Sun TX, Das BK, Liang JJ. Conformational and functional differences between recombinant human lens alphaA- and alphaB-crystallin. J Biol Chem. 1997; 272:6220–5. https://doi.org/10.1074/jbc.272.10.6220 [PubMed]
- 71. Nicholl ID, Quinlan RA. Chaperone activity of alpha-crystallins modulates intermediate filament assembly. EMBO J. 1994; 13:945–53. https://doi.org/10.1002/j.1460-2075.1994.tb06339.x [PubMed]
- 72. Clark JI, Huang QL. Modulation of the chaperone-like activity of bovine alpha-crystallin. Proc Natl Acad Sci USA. 1996; 93:15185–9. https://doi.org/10.1073/pnas.93.26.15185 [PubMed]
- 73. Reddy GB, Reddy PY, Suryanarayana P. alphaA- and alphaB-crystallins protect glucose-6-phosphate dehydrogenase against UVB irradiation-induced inactivation. Biochem Biophys Res Commun. 2001; 282:712–6. https://doi.org/10.1006/bbrc.2001.4642 [PubMed]
- 74. Kantorow M, Piatigorsky J. Alpha-crystallin/small heat shock protein has autokinase activity. Proc Natl Acad Sci USA. 1994; 91:3112–6. https://doi.org/10.1073/pnas.91.8.3112 [PubMed]
- 75. Kantorow M, Horwitz J, van Boekel MA, de Jong WW, Piatigorsky J. Conversion from oligomers to tetramers enhances autophosphorylation by lens alpha A-crystallin. Specificity between alpha A- and alpha B-crystallin subunits. J Biol Chem. 1995; 270:17215–20. https://doi.org/10.1074/jbc.270.29.17215 [PubMed]
- 76. Dasgupta S, Hohman TC, Carper D. Hypertonic stress induces alpha B-crystallin expression. Exp Eye Res. 1992; 54:461–70. https://doi.org/10.1016/0014-4835(92)90058-z [PubMed]
- 77. Aoyama A, Fröhli E, Schäfer R, Klemenz R. Alpha B-crystallin expression in mouse NIH 3T3 fibroblasts: glucocorticoid responsiveness and involvement in thermal protection. Mol Cell Biol. 1993; 13:1824–35. https://doi.org/10.1128/mcb.13.3.1824-1835.1993 [PubMed]
- 78. Mehlen P, Preville X, Chareyron P, Briolay J, Klemenz R, Arrigo AP. Constitutive expression of human hsp27, Drosophila hsp27, or human alpha B-crystallin confers resistance to TNF- and oxidative stress-induced cytotoxicity in stably transfected murine L929 fibroblasts. J Immunol. 1995; 154:363–74. [PubMed]
- 79. Mehlen P, Kretz-Remy C, Préville X, Arrigo AP. Human hsp27, Drosophila hsp27 and human alphaB-crystallin expression-mediated increase in glutathione is essential for the protective activity of these proteins against TNFalpha-induced cell death. EMBO J. 1996; 15:2695–706. https://doi.org/10.1002/j.1460-2075.1996.tb00630.x [PubMed]
- 80. Mehlen P, Schulze-Osthoff K, Arrigo AP. Small stress proteins as novel regulators of apoptosis. Heat shock protein 27 blocks Fas/APO-1- and staurosporine-induced cell death. J Biol Chem. 1996; 271:16510–4. https://doi.org/10.1074/jbc.271.28.16510 [PubMed]
- 81. Andley UP, Song Z, Wawrousek EF, Fleming TP, Bassnett S. Differential protective activity of alpha A- and alphaB-crystallin in lens epithelial cells. J Biol Chem. 2000; 275:36823–31. https://doi.org/10.1074/jbc.M004233200 [PubMed]
- 82. Liu JP, Schlosser R, Ma WY, Dong Z, Feng H, Liu L, Huang XQ, Liu Y, Li DW. Human alphaA- and alphaB-crystallins prevent UVA-induced apoptosis through regulation of PKCalpha, RAF/MEK/ERK and AKT signaling pathways. Exp Eye Res. 2004; 79:393–403. https://doi.org/10.1016/j.exer.2004.06.015 [PubMed]
- 83. Kamradt MC, Chen F, Cryns VL. The small heat shock protein alpha B-crystallin negatively regulates cytochrome c- and caspase-8-dependent activation of caspase-3 by inhibiting its autoproteolytic maturation. J Biol Chem. 2001; 276:16059–63. https://doi.org/10.1074/jbc.C100107200 [PubMed]
- 84. Kamradt MC, Chen F, Sam S, Cryns VL. The small heat shock protein alpha B-crystallin negatively regulates apoptosis during myogenic differentiation by inhibiting caspase-3 activation. J Biol Chem. 2002; 277:38731–6. https://doi.org/10.1074/jbc.M201770200 [PubMed]
- 85. Morozov V, Wawrousek EF. Caspase-dependent secondary lens fiber cell disintegration in alphaA-/alphaB-crystallin double-knockout mice. Development. 2006; 133:813–21. https://doi.org/10.1242/dev.02262 [PubMed]
- 86. Ousman SS, Tomooka BH, van Noort JM, Wawrousek EF, O’Connor KC, Hafler DA, Sobel RA, Robinson WH, Steinman L. Protective and therapeutic role for alphaB-crystallin in autoimmune demyelination. Nature. 2007; 448:474–9. https://doi.org/10.1038/nature05935 [PubMed]
- 87. Aggeli IK, Beis I, Gaitanaki C. Oxidative stress and calpain inhibition induce alpha B-crystallin phosphorylation via p38-MAPK and calcium signalling pathways in H9c2 cells. Cell Signal. 2008; 20:1292–302. https://doi.org/10.1016/j.cellsig.2008.02.019 [PubMed]
- 88. Dimberg A, Rylova S, Dieterich LC, Olsson AK, Schiller P, Wikner C, Bohman S, Botling J, Lukinius A, Wawrousek EF, Claesson-Welsh L. alphaB-crystallin promotes tumor angiogenesis by increasing vascular survival during tube morphogenesis. Blood. 2008; 111:2015–23. https://doi.org/10.1182/blood-2007-04-087841 [PubMed]
- 89. Stegh AH, Kesari S, Mahoney JE, Jenq HT, Forloney KL, Protopopov A, Louis DN, Chin L, DePinho RA. Bcl2L12-mediated inhibition of effector caspase-3 and caspase-7 via distinct mechanisms in glioblastoma. Proc Natl Acad Sci USA. 2008; 105:10703–8. https://doi.org/10.1073/pnas.0712034105 [PubMed]
- 90. Vigneron A, Vousden KH. p53, ROS and senescence in the control of aging. Aging (Albany NY). 2010; 2:471–4. https://doi.org/10.18632/aging.100189 [PubMed]
- 91. Timofeev O, Koch L, Niederau C, Tscherne A, Schneikert J, Klimovich M, Elmshäuser S, Zeitlinger M, Mernberger M, Nist A, Osterburg C, Dötsch V, Hrabé de Angelis M, Stiewe T, and German Mouse Clinic Consortium. Phosphorylation Control of p53 DNA-Binding Cooperativity Balances Tumorigenesis and Aging. Cancer Res. 2020; 80:5231–44. https://doi.org/10.1158/0008-5472.CAN-20-2002 [PubMed]
- 92. Roake CM, Artandi SE. Control of Cellular Aging, Tissue Function, and Cancer by p53 Downstream of Telomeres. Cold Spring Harb Perspect Med. 2017; 7:a026088. https://doi.org/10.1101/cshperspect.a026088 [PubMed]
- 93. Shieh SY, Ahn J, Tamai K, Taya Y, Prives C. The human homologs of checkpoint kinases Chk1 and Cds1 (Chk2) phosphorylate p53 at multiple DNA damage-inducible sites. Genes Dev. 2000; 14:289–300. https://doi.org/10.1101/gad.14.3.289 [PubMed]
- 94. Das KC, Dashnamoorthy R. Hyperoxia activates the ATR-Chk1 pathway and phosphorylates p53 at multiple sites. Am J Physiol Lung Cell Mol Physiol. 2004; 286:L87–97. https://doi.org/10.1152/ajplung.00203.2002 [PubMed]
- 95. Guo Z, Kumagai A, Wang SX, Dunphy WG. Requirement for Atr in phosphorylation of Chk1 and cell cycle regulation in response to DNA replication blocks and UV-damaged DNA in Xenopus egg extracts. Genes Dev. 2000; 14:2745–56. https://doi.org/10.1101/gad.842500 [PubMed]
- 96. Liu Q, Guntuku S, Cui XS, Matsuoka S, Cortez D, Tamai K, Luo G, Carattini-Rivera S, DeMayo F, Bradley A, Donehower LA, Elledge SJ. Chk1 is an essential kinase that is regulated by Atr and required for the G(2)/M DNA damage checkpoint. Genes Dev. 2000; 14:1448–59. https://doi.org/10.1101/gad.14.12.1448 [PubMed]
- 97. Zhao H, Piwnica-Worms H. ATR-mediated checkpoint pathways regulate phosphorylation and activation of human Chk1. Mol Cell Biol. 2001; 21:4129–39. https://doi.org/10.1128/MCB.21.13.4129-4139.2001 [PubMed]
- 98. Wang X, Khadpe J, Hu B, Iliakis G, Wang Y. An overactivated ATR/CHK1 pathway is responsible for the prolonged G2 accumulation in irradiated AT cells. J Biol Chem. 2003; 278:30869–74. https://doi.org/10.1074/jbc.M301876200 [PubMed]
- 99. Bellosillo B, Villamor N, Colomer D, Pons G, Montserrat E, Gil J. In vitro evaluation of fludarabine in combination with cyclophosphamide and/or mitoxantrone in B-cell chronic lymphocytic leukemia. Blood. 1999; 94:2836–43. https://doi.org/10.1182/blood.V94.8.2836.420k35_2836_2843 [PubMed]
- 100. Cvekl A, Duncan MK. Genetic and epigenetic mechanisms of gene regulation during lens development. Prog Retin Eye Res. 2007; 26:555–97. https://doi.org/10.1016/j.preteyeres.2007.07.002 [PubMed]
- 101. Bruel AL, Masurel-Paulet A, Rivière JB, Duffourd Y, Lehalle D, Bensignor C, Huet F, Borgnon J, Roucher F, Kuentz P, Deleuze JF, Thauvin-Robinet C, Faivre L, Thevenon J. Autosomal recessive truncating MAB21L1 mutation associated with a syndromic scrotal agenesis. Clin Genet. 2017; 91:333–8. https://doi.org/10.1111/cge.12794 [PubMed]
- 102. Rad A, Altunoglu U, Miller R, Maroofian R, James KN, Çağlayan AO, Najafi M, Stanley V, Boustany RM, Yeşil G, Sahebzamani A, Ercan-Sencicek G, Saeidi K, et al. MAB21L1 loss of function causes a syndromic neurodevelopmental disorder with distinctive cerebellar, ocular, craniofacial and genital features (COFG syndrome). J Med Genet. 2019; 56:332–9. https://doi.org/10.1136/jmedgenet-2018-105623 [PubMed]
- 103. Cvekl A, Ashery-Padan R. The cellular and molecular mechanisms of vertebrate lens development. Development. 2014; 141:4432–47. https://doi.org/10.1242/dev.107953 [PubMed]
- 104. Cvekl A, Zhang X. Signaling and Gene Regulatory Networks in Mammalian Lens Development. Trends Genet. 2017; 33:677–702. https://doi.org/10.1016/j.tig.2017.08.001 [PubMed]
- 105. Liu S, Li J, Tao Y, Xiao X. Small heat shock protein alphaB-crystallin binds to p53 to sequester its translocation to mitochondria during hydrogen peroxide-induced apoptosis. Biochem Biophys Res Commun. 2007; 354:109–14. https://doi.org/10.1016/j.bbrc.2006.12.152 [PubMed]
- 106. Wang L, Nie Q, Gao M, Yang L, Xiang JW, Xiao Y, Liu FY, Gong XD, Fu JL, Wang Y, Nguyen QD, Liu Y, Liu M, Li DW. The transcription factor CREB acts as an important regulator mediating oxidative stress-induced apoptosis by suppressing αB-crystallin expression. Aging (Albany NY). 2020; 12:13594–617. https://doi.org/10.18632/aging.103474 [PubMed]
- 107. Xiao L, Gong LL, Yuan D, Deng M, Zeng XM, Chen LL, Zhang L, Yan Q, Liu JP, Hu XH, Sun SM, Liu J, Ma HL, et al. Protein phosphatase-1 regulates Akt1 signal transduction pathway to control gene expression, cell survival and differentiation. Cell Death Differ. 2010; 17:1448–62. https://doi.org/10.1038/cdd.2010.16 [PubMed]
- 108. Mahns A, Melchheier I, Suschek CV, Sies H, Klotz LO. Irradiation of cells with ultraviolet-A (320-400 nm) in the presence of cell culture medium elicits biological effects due to extracellular generation of hydrogen peroxide. Free Radic Res. 2003; 37:391–7. https://doi.org/10.1080/1071576031000064702 [PubMed]



