Introduction
Given its elevated aggressiveness and unfavorable prognosis, pancreatic adenocarcinoma (PAAD) represents a substantial threat to human life and well-being. Epidemiological studies show that pancreatic cancer is the fourth most lethal form of cancer, while being the 12th most frequent tumor worldwide [1, 2]. Pancreatic ductal adenocarcinoma (PDAC) comprises the majority of PAAD cases, displaying low responsiveness to radiotherapy, a challenging resection rate, and a notably poor prognosis, with a 5-year survival rate of less than 7% [1, 3]. The progression of PAAD from genetic mutation to abnormal cell proliferation and precancerous lesions, culminating in early carcinoma, spans an approximate duration of 5-20 years. However, a PAAD tumor can rapidly progress from a tiny lump to an advanced stage in as little as 6 to 20 months. Moreover, the asymptomatic progression of pancreatic tumors often leads to the diagnosis of pancreatic cancer at advanced stages in most individuals. Therefore, early identification and prediction of disease progression in PAAD are of significant clinical importance.
Pancreatic cancer therapy and patient outcomes can be greatly improved by a deeper understanding of the pathogenesis and development of PAAD, as well as the identification of novel molecular targets for the disease. Rapid advances in omics technology over the past few decades have greatly aided our understanding of the molecular pathogenesis of PAAD [4, 5]. Akin to numerous malignancies, PAAD intricately engages multifaceted molecular cascades. Presently, researchers have adeptly fashioned a variety of intricate polygenic prognostic risk models leveraging advanced bioinformatics methodologies. Gene signatures obtained from omic data have been developed to forecast PAAD clinical outcomes [6, 7]. A multigene prognostic model serves as a valuable tool for assessing patients’ overall survival duration and risk of recurrence. It enables the identification of high-risk individuals with a poor prognosis, facilitating timely and systematic intervention. Conversely, it allows for the appropriate avoidance of unnecessary treatment burden among low-risk patients. In light of this, unique multigene signatures are required for predicting PAAD outcomes and recurrence.
Throughout the progression of cancer, the microenvironment of PAAD cells is conducive to their survival, proliferation, and distant metastasis [8]. Research has underscored the fundamental importance of the ongoing interaction between cancer cells and stromal cells in the process of cancer development and progression [9]. Cancer-associated fibroblasts (CAFs) are comparatively prevalent stromal cell components that have been observed in a variety of malignancies, including breast cancer, prostate cancer, and hepatocellular carcinoma, and their communications with cancer cells have been shown to be essential for cancer development [10–12]. Noteworthy is the possibility that certain biological traits, including pronounced interstitial fibrosis, are linked to the poor prognosis of pancreatic cancer [13]. Tumor cell proliferation, metastasis, and chemotherapy resistance may all be influenced by CAFs due to the secretion of various growth factors, chemokines, and cytokines, as well as the degradation of extracellular matrix proteins [14, 15]. The tumor-promoting properties of CAFs might be maintained even in the absence of cancer cells [16]. Subsequently, blocking CAF-derived effects or inhibiting CAF-secreted components that promote tumor formation and progression has emerged as a viable method for PAAD treatments.
Previous study has identified five new CAFs subclusters based on the marker genes with distinct signaling patterns and immune status in PAAD using single-cell sequence transcriptomic data [17]. However, there is still a lack of knowledge on how PAAD prognosis and immunotherapy response are connected to the features of CAF on a systemic level. Transcriptomic and single-cell RNA-sequencing (scRNA-seq) data pertaining to PAAD were obtained from the GEO and TCGA databases. We classified CAFs into distinct subclusters and established a CAF-based risk signature for PAAD. To enhance the clinical applicability of CAF characteristics in predicting PAAD, we devised a distinctive nomogram that integrates the CAF-based risk signature alongside clinicopathological variables. Finally, the immunological landscape and response to immunotherapy that underlie the CAF-based signature were further explored, and the clinical significance was established. Our findings could offer fresh perspectives on the pathogenesis of PAAD, paving the way for more individualized therapies and better results for PAAD patients.
Materials and Methods
Data collection and processing
We obtained scRNA-seq data (GSE155698) from the Gene Expression Omnibus (GEO) database, encompassing pancreatic cancer tumor samples from 16 patients alongside 3 adjacent normal pancreas samples. Scanning the scRNA-seq data involved an initial screening of single cells, ensuring each gene was expressed in at least three cells, while maintaining a minimum threshold of 250 expressed genes per cell. To determine the relative abundance of mitochondria and rRNA, the Seurat R package was employed [18]. Each single cell was then configured to express at least 6000 genes and have a UMI greater than 100 for further screening. After everything was filtered out, 50,527 cells were left. Utilizing The Cancer Genome Atlas (TCGA) database, we accessed transcriptome data, single-nucleotide variant (SNV) data, copy number variant (CNV) data, and associated clinical information for PAAD. Additionally, expression matrices from GSE78229 and GSE85916 were acquired from the GEO database for validation purposes.
Clustering of CAFs
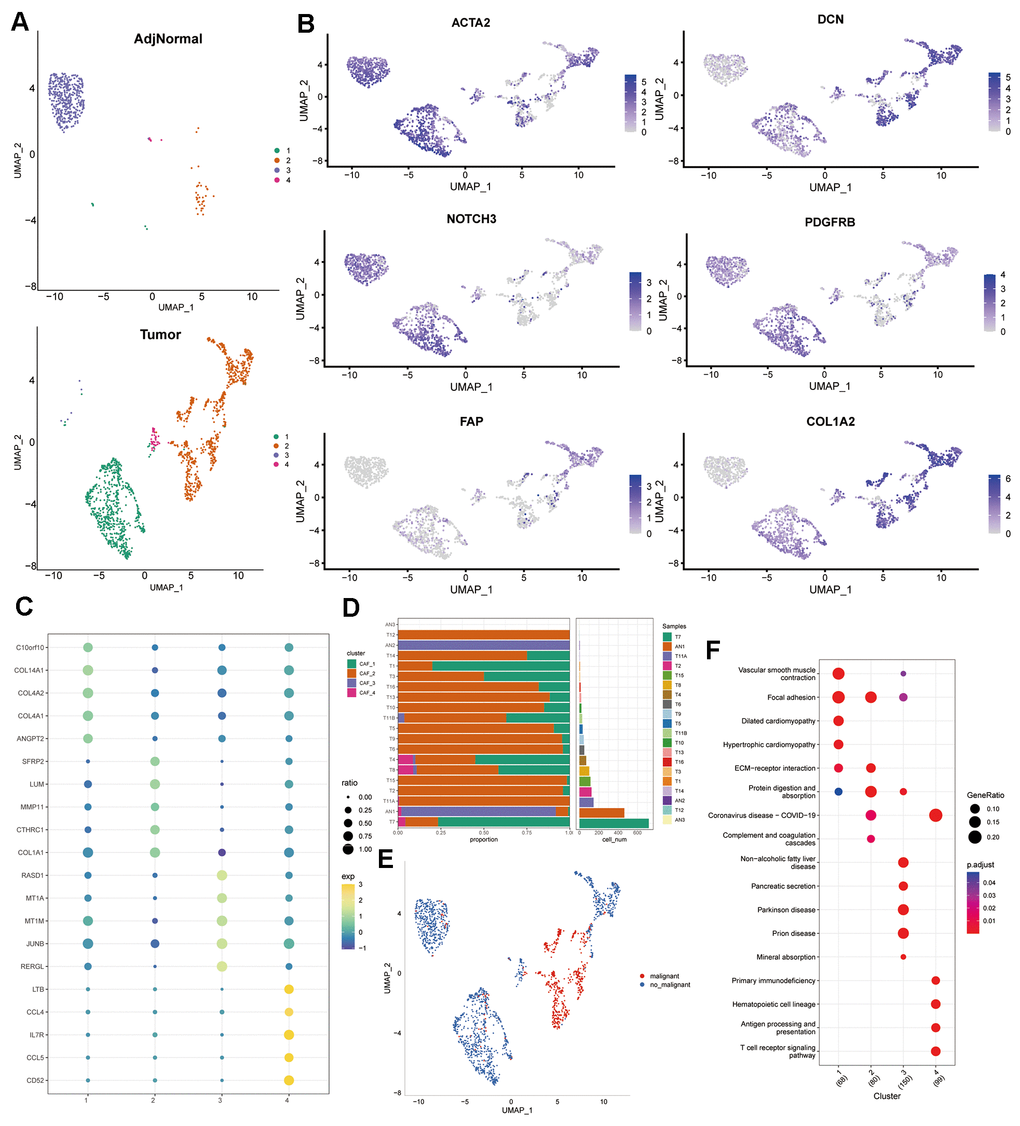
In an effort to better describe the CAF signature, we redeployed the scRNA-seq data of PAAD. After log normalization, cells that had either over 6000 or under 250 genes being actively expressed were filtered out. We addressed batch effects and conducted non-linear dimensional reduction. Using the FindNeighbors and FindClusters functions, individual cells were partitioned into separate subgroups. Subsequently, this subdivision was visualized via t-distributed stochastic neighbor embedding (t-SNE) dimensional reduction. Six genes were used to annotate fibroblasts, including ACTA2, FAP, PDGFRB, NOTCH3, DCN, and COL1A2. Using the same approach, FindNeighbors and FindClusters were used to re-cluster the fibroblasts, which was visualized with the TSNE dimensionality reduction. We utilized the FindAllMarkers function to identify specific marker genes within each CAF cluster. Subsequently, functional enrichments were conducted on these CAF marker genes using the Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis via the clusterProfiler package [19]. The CopyKAT R package was used to distinguish tumor cells from normal cells by analyzing CNV features among CAFs clusters [20].
Identification of prognostic genes
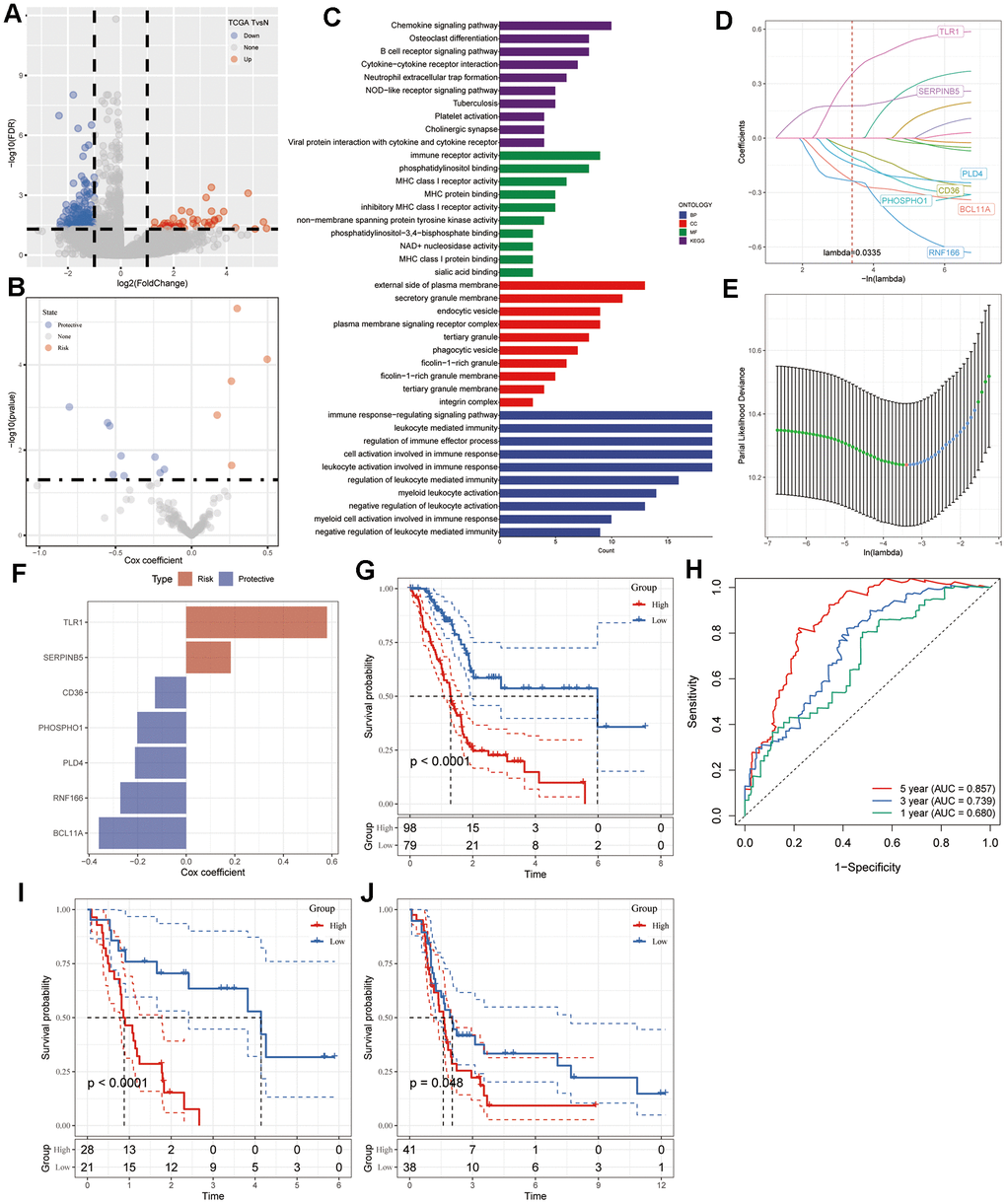
Differentially expressed genes (DEGs) between 177 pancreatic tumor samples and 4 normal tissue samples were first identified in the TCGA-PAAD cohort. We then analyzed the DEGs for CAF clusters to determine which genes are most strongly correlated with CAFs (P < 0.001, cor > 0.4). Further prognosis-associated genes were identified through univariate Cox regression analysis, considering a significance threshold of P < 0.05. To reduce the pool of prognostic genes, initial analysis involved least absolute shrinkage and selection operator (LASSO) Cox regression, yielding seven genes. Subsequently, a multivariate Cox regression employing stepwise regression constructed the risk signature, calculating coefficients via the formula: risk score = Σβi*Expi. Patients were stratified into high- or low-risk groups based on their risk scores. An evaluation of the risk signature’s predictive performance was conducted using receiver operating characteristic (ROC) analysis. Analyses in the validation cohorts were carried out in a similar manner.
Nomogram construction
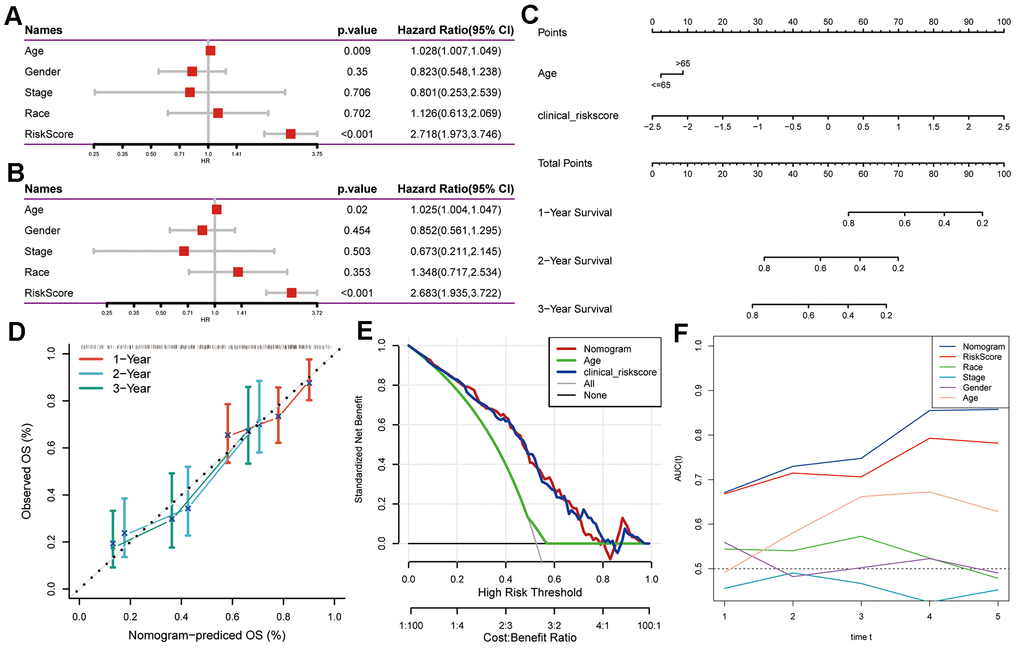
To create a clinically applicable nomogram model, we initiated univariate and multivariate Cox regression analyses incorporating the risk signature and clinicopathological factors such as age, gender, stage, and race. The resulting multivariate Cox model, including variables with P < 0.05, was utilized to devise a prognostic nomogram for PAAD using the rms package [21]. The prediction efficacy of the model was assessed by the calibration curve and the reliability was assessed using decision curve analysis (DCA).
Immune infiltration analysis
The immune and stromal scores of the tumor microenvironment (TME) were calculated using the ESTIMATE algorithm and the relative abundance of infiltrating immune cells in the TME was evaluated by the CIBERSORT and MCPcounter algorithms [22].
Evaluation of responses to immunotherapy
We collected clinical and transcriptomic data of PAAD patients treated with anti-PD-L1 therapies from the IMvigor210 cohort available at http://research-pub.gene.com/IMvigor210CoreBiologies. Furthermore, GSE78220 comprises pre-treatment melanoma samples that underwent anti-PD-1 checkpoint blockade (ICB) immunotherapy. This transcriptomic dataset can be accessed to assess the potential utility of the risk signature in predicting response to ICB therapy [23].
Statistical analysis
R software (version 3.6.3) was used for all statistical testing. The Pearson or Spearman correlation method was used for correlation analysis. The Wilcoxon test was used to examine the differences between the two groups. K-M curves with Log-rank testing were used to compare survival times across groups. Statistical significance was assumed at a P-value of less than 0.05.
Data availability statement
Publicly available datasets were analyzed in this study. The data can be found in GEO and TCGA databases.
Results
Expression of distinct signaling patterns in CAF clusters
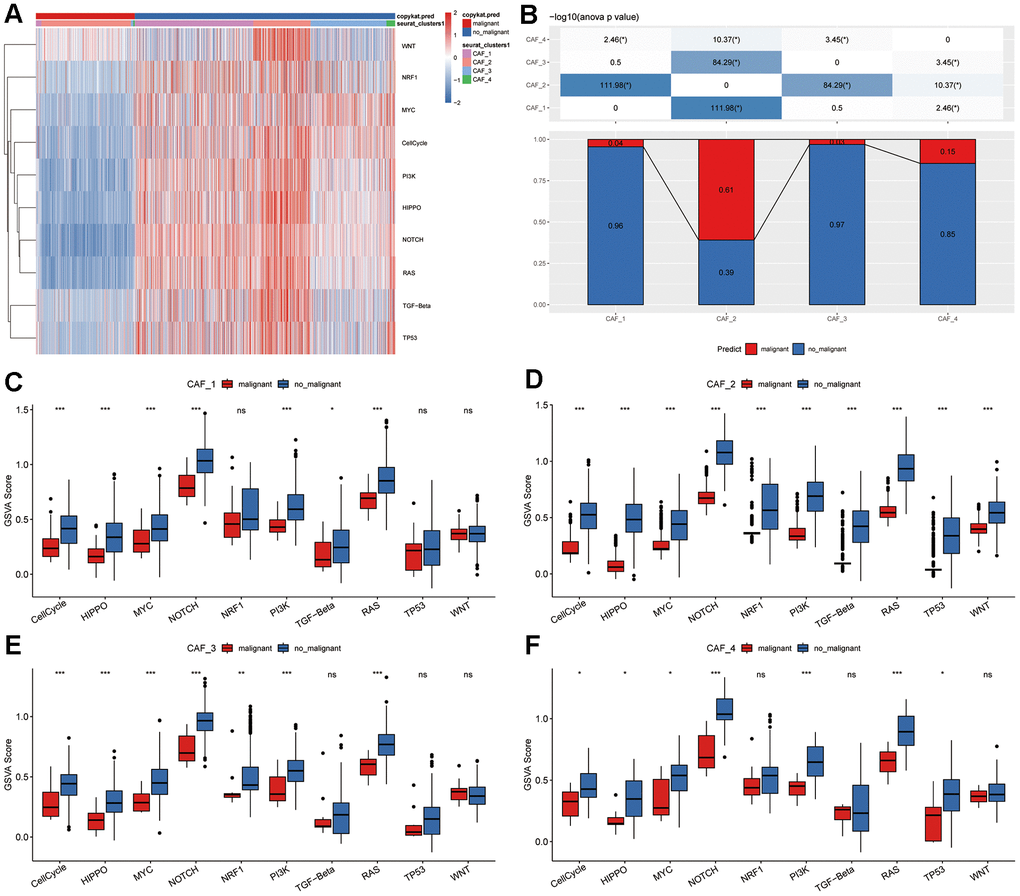
To investigate the potential role of CAF clusters in tumorigenesis, the expression of ten tumor-related pathways were evaluated in the four CAF clusters, including WNT, NRF1, MYC, CellCycle, PI3K, HIPPO, NOTCH, RAS, TGF-Beta, and TP53 signaling pathways. The GSVA scores of these pathways across the four CAF clusters were presented in the heatmap (Figure 2A). CAF_2 cluster exhibited notably higher proportions of malignant cells compared to the other three clusters (Figure 2B). Moreover, the CAF_4 cluster displayed a higher proportion of malignant cells compared to both CAF_1 and CAF_3 clusters. Furthermore, we conducted a comparison of GSVA scores for ten tumor-related pathways between malignant and non-malignant cells within each CAF cluster (Figure 2C–2F). Non-malignant cells had considerably higher GSVA scores for most signaling pathways in all CAF clusters, while minor variations among groups were observed.
Associations between CAF clusters and clinical characteristics of PAAD patients
To investigate potential associations between CAF clusters and clinical characteristics of PAAD patients, the ssGSEA scores for specific marker genes—C10orf10, COL14A1, COL4A2, COL4A1, and ANGPT2 for CAF_1; SFRP2, LUM, MMP11, CTHRC1, and COL1A1 for CAF_2; RASD1, MT1A, MT1M, JUNB, and RERGL for CAF_3; and LTB, CCL4, IL7R, CCL5, and CD52 for CAF_4 (as identified in Figure 1C)—were calculated within each CAF cluster based on the TCGA cohort. Supplementary Figure 2A demonstrated that the CAF_4 cluster displayed notably lower scores in tumor samples in comparison to normal samples, with no statistically significant differences observed in the remaining CAF clusters. Following this, to assess the prognostic significance, the PAAD samples within the TCGA cohort were stratified based on high and low CAF scores. There was no correlation between CAF_1 and CAF_3 clusters and PAAD prognosis, while the CAF_2 and CAF_4 clusters fared better for low-CAF score samples than high-CAF score samples. (Supplementary Figure 2B). Next, we set out to establish the clinical correlations of CAF clusters. When PAAD patients were segmented into two categories based on gender (female vs. male), the disparity of CAF_1 score, but not other CAF clusters, was observed (Supplementary Figure 2C–2F). However, no clinical associations between CAF clusters and age were discovered. Furthermore, CAF_3 and CAF_4 scores were shown to be considerably higher in white persons compared to Asian people, whereas no associations were found between CAF scores and pathological stages.
Mutation and pathway analyses of seven prognostic genes
The genetic mutations (SNV) within the seven genes forming the risk signature were subsequently examined. More samples were found to contain SNV mutations in TLR1, BCL11A, PHOSPHO1, and SERPINB5, however, no SNV mutation was found in CD36, PLD4, or RNF166 (Figure 5A). Pathway enrichment on SNV data showed that TGF-Beta and NRF2 pathways were the most affected pathways in PAAD, while more samples were involved in the RTK-RAS and TP53 pathways (Figure 5B). Within the seven genes comprising the risk signature, only a few samples displayed CNV alterations (Figure 5C). Hence, we explored the connections between these risk genes and diverse molecular indicators in PAAD, aiming to enhance our understanding of how these genes are linked to the disease. The results demonstrated that CD36, PHOSPHO1, PLD4, TLR1, BCL11A, and RNF166 were all significantly negatively correlated with Aneuploidy Score, Fraction Altered, and Number of Segments, whereas SERPINB5 exhibited notably positive correlations with Homologous Recombination Defects and Number of Segments (Figure 5D). Interestingly, the Nonsilent Mutation Rate displayed no correlation with any of these identified risk genes. Additionally, we delved into the potential pathways linked to each risk gene (Figure 5E, 5F). The heatmap revealed significant correlations of these seven genes with a total of 60 pathways, encompassing pathways like adipocytokine signaling pathway, base excision repair, and cell adhesion molecules cams.
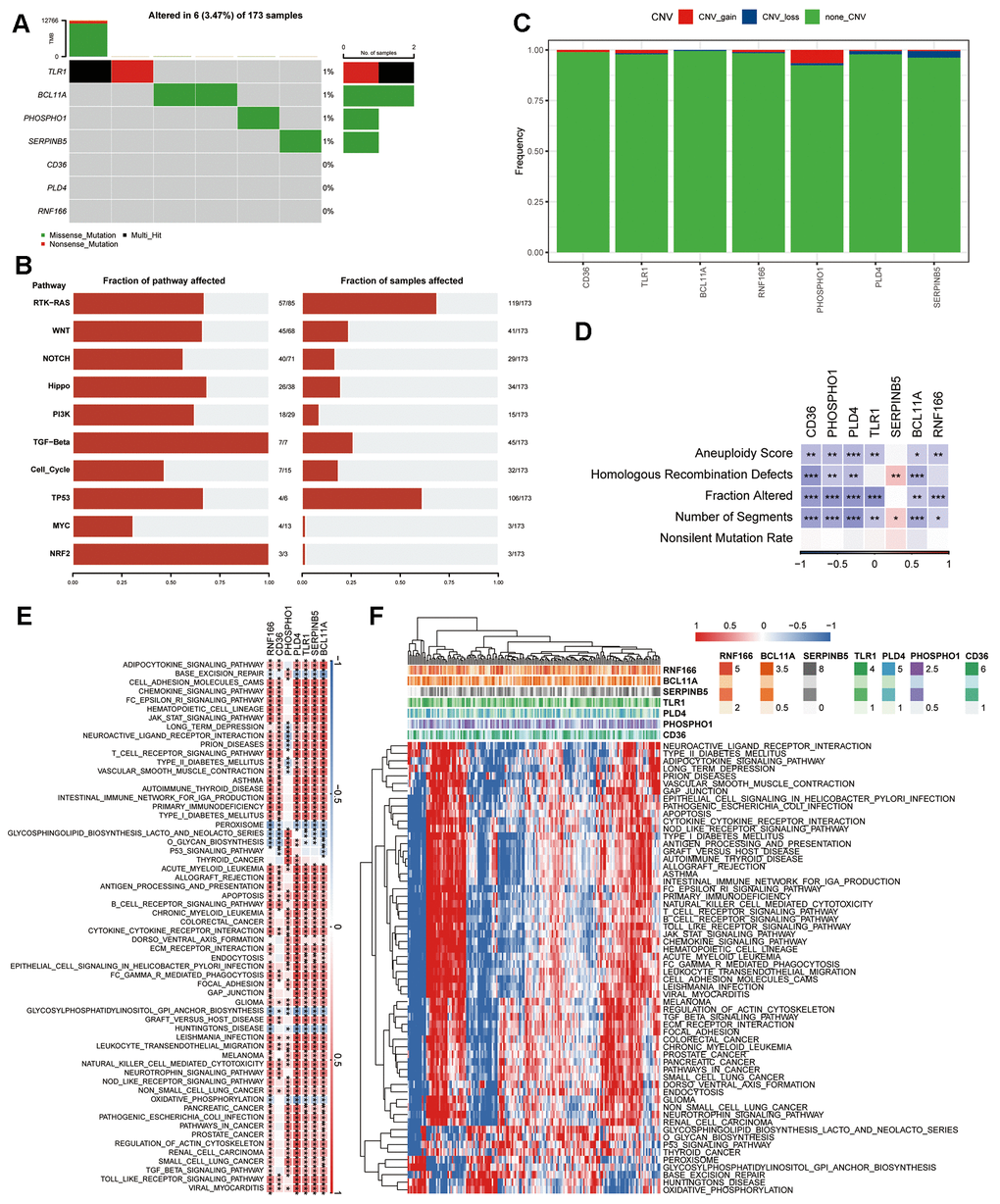
Relationship between immune infiltration and seven prognostic genes
We then investigated the association between the seven prognostic genes and immune infiltration. Initially, we computed the immune score, stromal score, and ESTIMATE score utilizing the ESTIMATE algorithm (Supplementary Figure 3A–3D). Our correlation analysis revealed that CD36, PHOSPHO1, PLD4, TLR1, and RNF166 exhibited positive correlations with stromal score, immune score, and ESTIMATE score. Conversely, BCL11A showed positive correlations solely with immune score and ESTIMATE score. Notably, SERPINB5 did not display significant correlations with any of these three scores. We then compared the immune score across groups that were defined by the median expression level of respective genes. For the genes CD36, PHOSPHO1, PLD4, TLR1, BCL11A, and RNF166, individuals with high expression had significantly higher immune score than those with low expression (Supplementary Figure 3E–3K). However, individuals with low SERPINB5 expression had higher immune score. We utilized the CIBERSORT algorithm to compute the abundance of infiltrating immune cells and their correlations with the seven prognostic genes. The correlation heatmap showed that CD36, PHOSPHO1, PLD4, BCL11A, and RNF166 presented a significantly positive correlation with CD8+ T cells, naïve and memory B cells, whereas negative correlation with CD4+ memory resting T cells. TLR1 was positively correlated with CD4+ memory activated T cells, while negatively correlated with activated NK cells and activated mast cells, similar to SERPINB5 (Supplementary Figure 3L). We further validated our results using the MCPcounter algorithm. Significantly positive correlations were observed between RNF166, TLR1, PLD4, PHOSPHO1, CD36, and infiltrating immune cells, while SERPINB5 was only positively correlated with fibroblasts, which was consistent with previous results (Supplementary Figure 3M).
The prediction of risk signature to the efficacy of immunotherapy
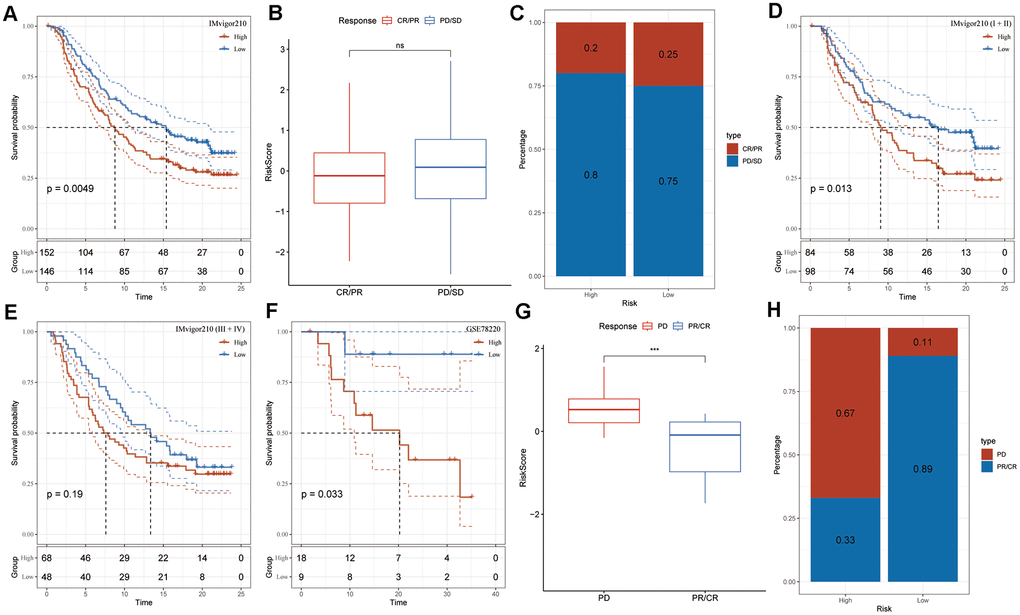
The utilization of T-cell immunotherapy in cancer treatment has shown promise in extending patients’ survival. Consequently, we conducted an analysis on the IMvigor210 and GSE78220 cohorts to evaluate the predictive efficacy of the risk signature for immune-checkpoint therapy. Patients categorized in the IMvigor210 low-risk group exhibited better clinical outcomes and longer overall survival in comparison to those in the high-risk group (Figure 6A, P = 0.0049). However, no significant difference in risk score was observed between patients with complete/partial response (CR/PR) and those with progressive/stable disease (PD/SD), and the proportion of PD/SD patients in the high-risk group did not significantly differ from that in the low-risk group (Figure 6B, 6C). Particularly, substantial differences in survival probability were noted among various risk groups for patients diagnosed at Stages I and II (Figure 6D, P = 0.013), but not for those at Stages III and IV (Figure 6E, P = 0.19). These findings suggested higher sensitivity of the risk score for patients diagnosed at earlier stages. Similarly, patients within the low-risk group in the GSE78220 cohort displayed significantly prolonged overall survival compared to those in the high-risk group (Figure 6F, P = 0.033). Moreover, in contrast to IMvigor210, PD patients exhibited notably higher risk scores than PR/CR patients (Figure 6G), and the proportion of PD patients in the high-risk group was significantly elevated compared to the low-risk group in the GSE78220 cohort (Figure 6H). These outcomes indicate the effective predictive capacity of the risk signature in determining patient responses to immunotherapy.
Discussion
Since CAFs have been proved to be engaged in carcinogenesis by releasing numerous substances into the TME, increasing evidence has shown that this dynamic interaction between tumor cells and stroma cells leads to tumor development [9, 24]. Here, using scRNA-seq data, we systematically characterized and classified CAFs of PAAD to better understand their variety. At the conclusion of the study, we identified four distinct clusters of CAFs, each exhibiting unique characteristics potentially influencing the biological regulation of the TME. Currently, there remains a scarcity of studies focusing on the predictive relevance of CAF-secreted factors or a CAF-associated risk signature in PAAD. Zhao et al. identified five novel subcluster of CAFs based on the marker genes and both CAF-C2 and CAF-C4 subgroups had significantly negative correlation with prognosis [17]. Inconsistently, our study identified four subgroups in PAAD using single-cell sequencing dataset, while both CAF_2 and CAF_4 subgroups showed poor prognosis, which were established by tallying up DEGs from the four groups into a single score. The distinct prognostic significance of CAF clusters might be attributed in part to the notable variation we observed in the TP53 signaling pathway between malignant and non-malignant cells within CAF_2 and CAF_4 subgroups, while such variation wasn’t evident in CAF_1 and CAF_3. Earlier research has indicated the involvement of TP53 mutation in the progression of PAAD tumors [25, 26].
The predictive efficacy of two CAF subgroups led us to develop a CAF-based risk signature consisting of seven genes. CD36, PHOSPHO1, PLD4, BCL11A, and RNF166 were the protective genes, while TLR1 and SERPINB5 were the risk genes. Notably, TLR1, BCL11A, PHOSPHO1, and SERPINB5 were all found to have SNV mutations in our research. Overall patient survival is correlated with the presence of a sense SNV mutation, which alters protein activity or function that leads to PAAD progression [27]. Despite the lack of evidence from independent research, our findings imply a possible role for SNV mutations in these risk genes in PAAD development. Additionally, we discovered a strong association between the seven genes and 60 pathways, some of which have previously been linked to carcinogenesis, including apoptosis [28], cytokine-cytokine receptor interaction [29], leukocyte transendothelial migration [30], and natural killer cell mediated cytotoxicity [31]. Therefore, these findings point us in the right path for future research into how the control of these risk genes manifests in PAAD.
Recent research indicates that tumor growth may be facilitated by CAFs’ interactions with the infiltrating immune cells in the TME [32]. In our investigation, we employed three distinct algorithms to assess the connections between seven prognostic genes and immune infiltration. The ESTIMATE analysis indicated significant positive correlations between four protective genes and one risk gene with stromal score, immune score, and ESTIMATE score. These results suggested a possible interaction between these genes and tumor immune microenvironment in PAAD and, by extension, that these genes could be valuable therapeutic targets for the disease. The tumor immune microenvironment encompasses diverse immune cells within tumor islets, collectively orchestrating the stage for an effective anticancer immune response in the TME. Tumor cells are able to escape immune cell surveillance when CAFs collaborate with these cells to create an immunosuppressive tumor microenvironment [33]. After calculating the immune cell abundance with CIBERSORT algorithm, multiple immune cells were found to correlate with the predictive genes, including naïve and memory B cells, CD8+ and CD4+ naïve T cells, and activated NK cells, which was consistent with the results of MCPcounter analysis. Compared to tumors without tertiary lymphoid structures (TLSs), PDAC tumors have an increased number of memory B cells [34]. Comparing TLSs from the TME to peripheral blood, there was a decrease in the number of IgD+ and IgM+/IgD+ naïve B cells, indicating that class switching occurred in the TME [35]. T cells play a role in tumor development, and therapies derived from T cells, like checkpoint blockade and chimeric antigen receptor T (CAR-T) cell therapy, have exhibited promising outcomes [36]. When a NK cell detects surface markers associated with oncogenic transformation, it may quickly and efficiently eliminate any number of neighboring cells. This unusual ability of NK cells can be used to stimulate both antibody and T cell responses, thus strengthening the case for their use as anticancer agents [37].
The majority of patients, however, exhibit either inherent or acquired resistance to immunotherapies [38]. According to our findings, the risk signature could identify individuals who were more likely to respond favorably to immunotherapies. Recent research has shown that mesothelial cell-derived CAFs can function as antigen-presenting cells by directly ligating and inducing antigen-specific regulatory T cells from naïve CD4+ T cells [39]. Therefore, CAFs might play a role in immune evasion within pancreatic cancer, offering insights into enhancing cancer immunotherapy strategies. Additionally, there was an observed positive correlation of M1 macrophages with the risk genes (TLR1 and SERPINB5), whereas M2 macrophages exhibited a positive correlation with the protective gene (BCL11A), hinting at a potential involvement of the risk genes in macrophage polarization. Studies revealed that LRRC15+ CAFs directly diminish CD8+ T cell activity and limit the response to checkpoint blockade through a TGFβ-dependent pathway [40]. Enhancements in patient survival and the response to immunotherapy could potentially result from the advancement of treatments aimed at re-establishing the balanced fibroblast state, thereby reducing the population of disease-promoting LRRC15+ myofibroblasts. Our results, on the other hand, suggested that a CAF-based signature might forecast a patient’s receptivity to anti-PD-L1 immunotherapy. These findings revealed previously unknown information on CAF’s function in modifying the TME and cancer niche. However, additional research is still needed to fully understand CAF-TIME communication’s function in PAAD.
Our study has a few limitations. Firstly, the development of CAF clustering and the CAF-based risk signature relied on retrospective data from open resources. It’s crucial to validate its effectiveness in other prospective and multi-center PAAD cohorts in the near future. Secondly, our focus was primarily on the potential predictive value of the CAF-based risk signature. Further research is necessary to unravel the mechanisms underlying the signature’s involvement in the progression of PAAD.
Conclusions
In summary, our study provided a comprehensive characterization of PAAD CAF populations, identifying four distinct CAF subgroups exhibiting varied traits. The DEGs among these groups were found to be enriched in pathways like vascular smooth muscle contraction, focal adhesion, ECM-receptor interaction, and protein digestion and absorption. Leveraging two clusters significantly associated with PAAD prognosis, we formulated a CAF-based predictive risk signature composed of seven genes. This signature, when combined with clinicopathological parameters in a nomogram, demonstrated excellent prognostic performance for PAAD patient outcomes. Moreover, our findings suggested that this signature correlated with the immune landscape of PAAD, potentially serving as a tool to predict responses to PD-L1 blockade immunotherapy.
Supplementary Materials
Author Contributions
JM and LH designed, conceptualized, and conducted the whole research. ZC conducted the GEO and TCGA dataset analysis of PAAD. JM contributed to the result interpretation and manuscript preparation. LH revised the manuscript critically, with all authors providing feedback. All authors contributed to the article and approved the final submitted version.
Acknowledgments
This article is based on bioinformatics analysis of existing publicly available data, not yet experimental or funded. We sincerely thank the scientists who shared their data on the public database.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
No funding was provided for this study.
References
- 1. Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J Clin. 2021; 71:7–33. https://doi.org/10.3322/caac.21654 [PubMed]
- 2. Feng RM, Zong YN, Cao SM, Xu RH. Current cancer situation in China: good or bad news from the 2018 Global Cancer Statistics? Cancer Commun (Lond). 2019; 39:22. https://doi.org/10.1186/s40880-019-0368-6 [PubMed]
- 3. Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016; 66:115–32. https://doi.org/10.3322/caac.21338 [PubMed]
- 4. Huang X, Zhang G, Tang T, Liang T. Identification of tumor antigens and immune subtypes of pancreatic adenocarcinoma for mRNA vaccine development. Mol Cancer. 2021; 20:44. https://doi.org/10.1186/s12943-021-01310-0 [PubMed]
- 5. Zhang D, Wu S, Pan S, Wang M, Wang Z, He Z, Zhang G, Cui F, Song Y, Li W, Shi X, Huang H, Xu H. Single-cell sequencing reveals heterogeneity between pancreatic adenosquamous carcinoma and pancreatic ductal adenocarcinoma with prognostic value. Front Immunol. 2022; 13:972298. https://doi.org/10.3389/fimmu.2022.972298 [PubMed]
- 6. Wu M, Li X, Zhang T, Liu Z, Zhao Y. Identification of a Nine-Gene Signature and Establishment of a Prognostic Nomogram Predicting Overall Survival of Pancreatic Cancer. Front Oncol. 2019; 9:996. https://doi.org/10.3389/fonc.2019.00996 [PubMed]
- 7. Chi H, Peng G, Wang R, Yang F, Xie X, Zhang J, Xu K, Gu T, Yang X, Tian G. Cuprotosis Programmed-Cell-Death-Related lncRNA Signature Predicts Prognosis and Immune Landscape in PAAD Patients. Cells. 2022; 11:3436. https://doi.org/10.3390/cells11213436 [PubMed]
- 8. Farrow B, Albo D, Berger DH. The role of the tumor microenvironment in the progression of pancreatic cancer. J Surg Res. 2008; 149:319–28. https://doi.org/10.1016/j.jss.2007.12.757 [PubMed]
- 9. Zhang W, Huang P. Cancer-stromal interactions: role in cell survival, metabolism and drug sensitivity. Cancer Biol Ther. 2011; 11:150–6. https://doi.org/10.4161/cbt.11.2.14623 [PubMed]
- 10. Musa M. Single-cell analysis on stromal fibroblasts in the microenvironment of solid tumours. Adv Med Sci. 2020; 65:163–9. https://doi.org/10.1016/j.advms.2019.12.001 [PubMed]
- 11. Gandellini P, Andriani F, Merlino G, D’Aiuto F, Roz L, Callari M. Complexity in the tumour microenvironment: Cancer associated fibroblast gene expression patterns identify both common and unique features of tumour-stroma crosstalk across cancer types. Semin Cancer Biol. 2015; 35:96–106. https://doi.org/10.1016/j.semcancer.2015.08.008 [PubMed]
- 12. Sukowati CH, Anfuso B, Crocé LS, Tiribelli C. The role of multipotent cancer associated fibroblasts in hepatocarcinogenesis. BMC Cancer. 2015; 15:188. https://doi.org/10.1186/s12885-015-1196-y [PubMed]
- 13. Di Maggio F, El-Shakankery KH. Desmoplasia and Biophysics in Pancreatic Ductal Adenocarcinoma: Can We Learn From Breast Cancer? Pancreas. 2020; 49:313–25. https://doi.org/10.1097/MPA.0000000000001504 [PubMed]
- 14. Terada T, Makimoto K, Terayama N, Suzuki Y, Nakanuma Y. Alpha-smooth muscle actin-positive stromal cells in cholangiocarcinomas, hepatocellular carcinomas and metastatic liver carcinomas. J Hepatol. 1996; 24:706–12. https://doi.org/10.1016/s0168-8278(96)80267-4 [PubMed]
- 15. Song J, Ge Z, Yang X, Luo Q, Wang C, You H, Ge T, Deng Y, Lin H, Cui Y, Chu W, Yao M, Zhang Z, et al. Hepatic stellate cells activated by acidic tumor microenvironment promote the metastasis of hepatocellular carcinoma via osteopontin. Cancer Lett. 2015; 356:713–20. https://doi.org/10.1016/j.canlet.2014.10.021 [PubMed]
- 16. Littlepage LE, Egeblad M, Werb Z. Coevolution of cancer and stromal cellular responses. Cancer Cell. 2005; 7:499–500. https://doi.org/10.1016/j.ccr.2005.05.019 [PubMed]
- 17. Zhao G, Wang C, Jiao J, Zhang W, Yang H. The novel subclusters based on cancer-associated fibroblast for pancreatic adenocarcinoma. Front Oncol. 2022; 12:1045477. https://doi.org/10.3389/fonc.2022.1045477 [PubMed]
- 18. Butler A, Hoffman P, Smibert P, Papalexi E, Satija R. Integrating single-cell transcriptomic data across different conditions, technologies, and species. Nat Biotechnol. 2018; 36:411–20. https://doi.org/10.1038/nbt.4096 [PubMed]
- 19. Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012; 16:284–7. https://doi.org/10.1089/omi.2011.0118 [PubMed]
- 20. Gao R, Bai S, Henderson YC, Lin Y, Schalck A, Yan Y, Kumar T, Hu M, Sei E, Davis A, Wang F, Shaitelman SF, Wang JR, et al. Delineating copy number and clonal substructure in human tumors from single-cell transcriptomes. Nat Biotechnol. 2021; 39:599–608. https://doi.org/10.1038/s41587-020-00795-2 [PubMed]
- 21. Zhang S, Tong YX, Zhang XH, Zhang YJ, Xu XS, Xiao AT, Chao TF, Gong JP. A novel and validated nomogram to predict overall survival for gastric neuroendocrine neoplasms. J Cancer. 2019; 10:5944–54. https://doi.org/10.7150/jca.35785 [PubMed]
- 22. Chen B, Khodadoust MS, Liu CL, Newman AM, Alizadeh AA. Profiling Tumor Infiltrating Immune Cells with CIBERSORT. Methods Mol Biol. 2018; 1711:243–59. https://doi.org/10.1007/978-1-4939-7493-1_12 [PubMed]
- 23. Hugo W, Zaretsky JM, Sun L, Song C, Moreno BH, Hu-Lieskovan S, Berent-Maoz B, Pang J, Chmielowski B, Cherry G, Seja E, Lomeli S, Kong X, et al. Genomic and Transcriptomic Features of Response to Anti-PD-1 Therapy in Metastatic Melanoma. Cell. 2016; 165:35–44. https://doi.org/10.1016/j.cell.2016.02.065 [PubMed]
- 24. Biffi G, Tuveson DA. Diversity and Biology of Cancer-Associated Fibroblasts. Physiol Rev. 2021; 101:147–76. https://doi.org/10.1152/physrev.00048.2019 [PubMed]
- 25. Qi D, Song X, Xue C, Yao W, Shen P, Yu H, Zhang Z. AKT1/FOXP3 axis-mediated expression of CerS6 promotes p53 mutant pancreatic tumorigenesis. Cancer Lett. 2021; 522:105–18. https://doi.org/10.1016/j.canlet.2021.06.024 [PubMed]
- 26. Yue P, Zhu C, Gao Y, Li Y, Wang Q, Zhang K, Gao S, Shi Y, Wu Y, Wang B, Xie J, Meng X. Development of an autophagy-related signature in pancreatic adenocarcinoma. Biomed Pharmacother. 2020; 126:110080. https://doi.org/10.1016/j.biopha.2020.110080 [PubMed]
- 27. Yandım C, Karakülah G. Repeat expression is linked to patient survival and exhibits single nucleotide variation in pancreatic cancer revealing LTR70:r.879A>G. Gene. 2022; 822:146344. https://doi.org/10.1016/j.gene.2022.146344 [PubMed]
- 28. Chen X, Zeh HJ, Kang R, Kroemer G, Tang D. Cell death in pancreatic cancer: from pathogenesis to therapy. Nat Rev Gastroenterol Hepatol. 2021; 18:804–23. https://doi.org/10.1038/s41575-021-00486-6 [PubMed]
- 29. McAndrews KM, Chen Y, Darpolor JK, Zheng X, Yang S, Carstens JL, Li B, Wang H, Miyake T, Correa de Sampaio P, Kirtley ML, Natale M, Wu CC, et al. Identification of Functional Heterogeneity of Carcinoma-Associated Fibroblasts with Distinct IL6-Mediated Therapy Resistance in Pancreatic Cancer. Cancer Discov. 2022; 12:1580–97. https://doi.org/10.1158/2159-8290.CD-20-1484 [PubMed]
- 30. Ondruššek R, Kvokačková B, Kryštofová K, Brychtová S, Souček K, Bouchal J. Prognostic value and multifaceted roles of tetraspanin CD9 in cancer. Front Oncol. 2023; 13:1140738. https://doi.org/10.3389/fonc.2023.1140738 [PubMed]
- 31. Nelson A, Gebremeskel S, Lichty BD, Johnston B. Natural killer T cell immunotherapy combined with IL-15-expressing oncolytic virotherapy and PD-1 blockade mediates pancreatic tumor regression. J Immunother Cancer. 2022; 10:e003923. https://doi.org/10.1136/jitc-2021-003923 [PubMed]
- 32. Barrett RL, Puré E. Cancer-associated fibroblasts and their influence on tumor immunity and immunotherapy. Elife. 2020; 9:e57243. https://doi.org/10.7554/eLife.57243 [PubMed]
- 33. Mao X, Xu J, Wang W, Liang C, Hua J, Liu J, Zhang B, Meng Q, Yu X, Shi S. Crosstalk between cancer-associated fibroblasts and immune cells in the tumor microenvironment: new findings and future perspectives. Mol Cancer. 2021; 20:131. https://doi.org/10.1186/s12943-021-01428-1 [PubMed]
- 34. J Gunderson A, Rajamanickam V, Bui C, Bernard B, Pucilowska J, Ballesteros-Merino C, Schmidt M, McCarty K, Philips M, Piening B, Dubay C, Medler T, Newell P, et al. Germinal center reactions in tertiary lymphoid structures associate with neoantigen burden, humoral immunity and long-term survivorship in pancreatic cancer. Oncoimmunology. 2021; 10:1900635. https://doi.org/10.1080/2162402X.2021.1900635 [PubMed]
- 35. Downs-Canner SM, Meier J, Vincent BG, Serody JS. B Cell Function in the Tumor Microenvironment. Annu Rev Immunol. 2022; 40:169–93. https://doi.org/10.1146/annurev-immunol-101220-015603 [PubMed]
- 36. Kudo M. Immuno-Oncology Therapy for Hepatocellular Carcinoma: Current Status and Ongoing Trials. Liver Cancer. 2019; 8:221–38. https://doi.org/10.1159/000501501 [PubMed]
- 37. Shimasaki N, Jain A, Campana D. NK cells for cancer immunotherapy. Nat Rev Drug Discov. 2020; 19:200–18. https://doi.org/10.1038/s41573-019-0052-1 [PubMed]
- 38. Desbois M, Wang Y. Cancer-associated fibroblasts: Key players in shaping the tumor immune microenvironment. Immunol Rev. 2021; 302:241–58. https://doi.org/10.1111/imr.12982 [PubMed]
- 39. Huang H, Wang Z, Zhang Y, Pradhan RN, Ganguly D, Chandra R, Murimwa G, Wright S, Gu X, Maddipati R, Müller S, Turley SJ, Brekken RA. Mesothelial cell-derived antigen-presenting cancer-associated fibroblasts induce expansion of regulatory T cells in pancreatic cancer. Cancer Cell. 2022; 40:656–73.e7. https://doi.org/10.1016/j.ccell.2022.04.011 [PubMed]
- 40. Krishnamurty AT, Shyer JA, Thai M, Gandham V, Buechler MB, Yang YA, Pradhan RN, Wang AW, Sanchez PL, Qu Y, Breart B, Chalouni C, Dunlap D, et al. LRRC15+ myofibroblasts dictate the stromal setpoint to suppress tumour immunity. Nature. 2022; 611:148–54. https://doi.org/10.1038/s41586-022-05272-1 [PubMed]