Introduction
Estrogens are a class of sex hormones produced primarily by the ovaries and placenta, including estradiol, estrone and estriol. Estrogen plays an important role in slowing down aging. Estrogen has the ability to maintain bone mineral density, maintain the elasticity of blood vessels, preserve skin elasticity and moisture content, and protect cognitive functions of the brain [1–4]. For this reason, estrogen replacement therapy (ERT) is often used to improve various symptoms experienced by postmenopausal women [5]. However, this therapy is not without side effects. Excess estrogen causes overexpression of estrogen receptors (ERα and ERβ), which damages tissues and leads to autoimmune diseases and tumors [6]. Increased cases of breast and ovarian cancer, systemic lupus erythematosus, and multiple sclerosis suggest that the dangers of too much estrogen are worsening [7–9]. This study found that estrogen will induce inflammatory lesions in the aorta of female mice, similar to Takayasu arteritis (TAK).
Vascular smooth muscle cells (VSMCs) are differentiated cells located in the middle layer of arteries that possess contractile function and express marker proteins (e.g., ACTA2, MYH11, and CNN1) [10]. Despite a high degree of differentiation, VSMCs maintain a high degree of plasticity. Under harmful microenvironmental stimuli, VSMCs are activated from a contractile phenotype to a dedifferentiated mesenchymal cell state, in which they further differentiate into fibroblasts, osteoblasts, adipocytes, macrophages, and other characteristic cells [11, 12]. In addition, the plasticity of VSMCs is also closely related to the proinflammatory molecular environment, and this phenotypic transformation ability also induces secondary inflammatory responses [13, 14]. In vascular diseases, such as atherosclerosis [15], vascular calcification [16, 17], diabetes [18], and aortic aneurysms [19], VSMCs exhibit a hypodifferentiated state and dedifferentiate to a macrophage-like phenotype. Macrophage-like VSMCs obtain partial properties of immune cells, incorporating chemotaxis, phagocytosis, and proinflammatory factor release, consequently leading to a chronic inflammatory state in the vessel wall [14, 20]. Thus, increased plasticity of VSMCs is associated with the progression of vascular disease [21].
As research has progressed, emerging factors have been considered to influence the phenotypic transformation of VSMCs. Stimuli that trigger VSMC phenotypic regulation include injury, mechanical force, intracellular microenvironment, extracellular microenvironment, abnormal substance accumulation, and intercellular interactions [22–25]. Given that the prevalence of cardiovascular disease is much lower in premenopausal women than in men or postmenopausal women, estrogen is often considered beneficial to blood vessels [26]. Estrogen also has a role in vasodilating blood vessels, protecting vascular endothelial cells, and repairing damaged blood vessels [27, 28]. Estradiol (E2) is the most predominant and biologically active of estrogens. However, the effects of estrogen on blood vessels are contradictory and controversial [29, 30]. In postmenopausal women, Susana et al. found that, although short-term estrogen intervention exerts an anti-inflammatory effect, long-term use shifts to a proinflammatory state, whereas raloxifene treatment has no proinflammatory effect [31]. In rats with spinal cord injury, Samantaray et al. found that low doses of estrogen reduce inflammation, attenuate cell death, and promote angiogenesis [32]. In addition, estrogen or estrogen receptor (ER) signaling is highly expressed in breast cancer [33], endometriosis [34], and allergic respiratory inflammation [35], and it is positively correlated with systemic inflammation. Zhang et al. exposed rat VSMCs to gradient concentrations of E2 (10−9 to 10−5 M) and found a concentration-dependent promotion of VSMCs growth in the range of physiological levels, while they reported that high concentrations of E2 have a growth inhibitory effect [36].
Krüppel-like factor 4 (KLF4) is a member of the zinc-finger transcription factor family required for mammalian embryonic development and various diseases. KLF4 acts as a key initiator responsible for the dedifferentiation of VSMCs from a contractile phenotype to mesenchymal-like phenotype [37, 38]. Von Hippel-Lindau (VHL) is a ubiquitin protein ligase that regulates the stability of KLF4 protein. Binding of estrogen to ERα results in ubiquitin-dependent downregulation of VHL, which in turn leads to accumulation of KLF4 [39, 40]. Furthermore, if a binding element for hypoxia-inducible factor-1 alpha (HIF-1α) is found in the upstream regulatory region of the KLF4 locus, then VHL also regulates KLF4 by degrading HIF-1α [41, 42].
Therefore, the aim of the present study was to investigate the effect of estrogen on the phenotypic transformation of VSMCs and its role and potential mechanisms in inflammatory changes in large arteries.
Results
High estrogen induces Takayasu arteritis-like symptoms in female mice
Three months after exogenous intraperitoneal administration of E2 to female mice, we performed X-ray angiography on the mice. X-ray angiography confirmed significant stenotic segments in the suprarenal and subrenal abdominal aorta as well as bead-like changes in the carotid artery, changes in the superior mesenteric artery (including stenosis of secondary arteries), and atrophy of the kidney in the E2 group (Figure 1A). Blood pressure testing of mice at the end of model establishment revealed that the mean arterial pressure in the caudal artery of mice in the E2 group was attenuated, mainly due to a decrease in systolic blood pressure, and the pulse rate in the caudal artery was also significantly attenuated (Figure 1B–1E).
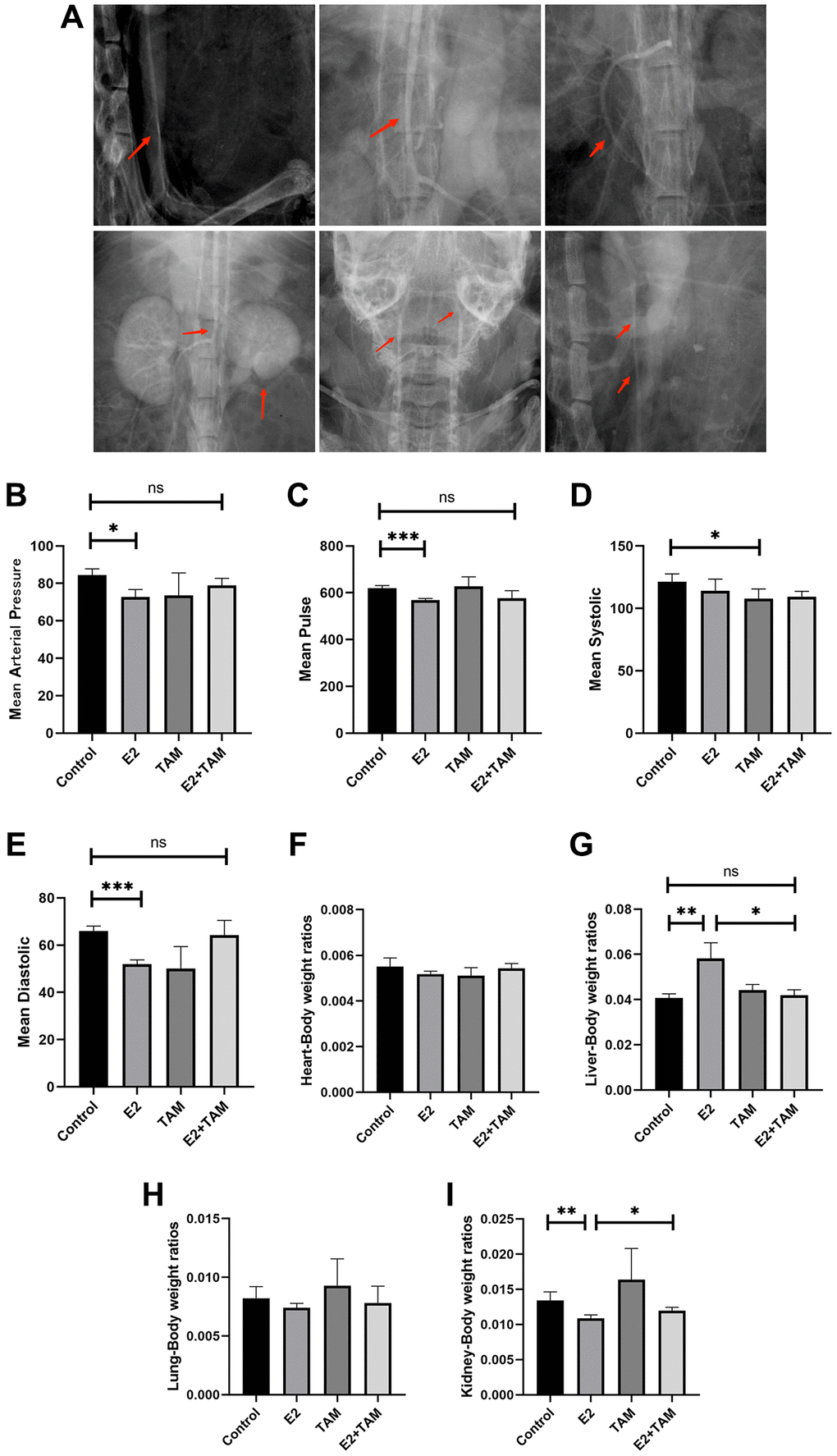
Figure 1. Estrogen-induced inflammatory changes in the aortas of mice. (A) After three months of continuous intraperitoneal injection of E2 in mice, x-ray angiography was performed to observe the condition of the aorta, branches and organs. (B–E) Blood pressure and pulse rate in the tail artery of mice were monitored using a noninvasive sphygmomanometer (n = 6). (F–I) After sacrificing the mice, the hearts, livers, lungs, and kidneys were collected, and the organ/body wet-weight ratios were calculated (n = 6). *P < 0.05, **P < 0.01, ***P < 0.001, indicating statistically significant data between groups; Abbreviation: ns indicates no statistical significance.
Arteritis not only affects localized organs, but may also affect the overall metabolic and nutritional status of the animal. This can lead to weight loss, which is a sign of systemic inflammation or systemic stress. As a result of these changes, changes in organ/body weight ratios may indicate the impact of arteritis on overall health. Measurement and calculation of the organ/body weight wet weight ratios of model mice demonstrated that E2 did not significantly change the mass of the heart and lungs. In addition, E2 significantly increased the liver/body weight wet weight ratio but decreased the kidney/body weight wet weight ratio, which may have been related to the narrowing of the renal artery triggered by E2, resulting in atrophy due to insufficient blood supply to the kidney (Figure 1F–1I).
Pathological staining of mouse abdominal aorta with HE, Masson’s trichrome, and Picro-Sirius Red revealed thickened arterial wall, disorganized smooth muscle layer structure, VSMCs proliferation, loosely arranged collagen fibers, aggregation of proliferating VSMCs into clusters and bundles, and visible hyaline degeneration of cytoplasm in the E2 group (Figure 2A). The detection of mouse serum by ELISA confirmed that serum estrogen, CRP, and IL-6 levels were significantly elevated in the E2 group (Figure 2B–2D). In addition, Ficonlin-1, a potential specific marker for TAK [43, 44], was also highly expressed in the E2 group (Figure 2E).
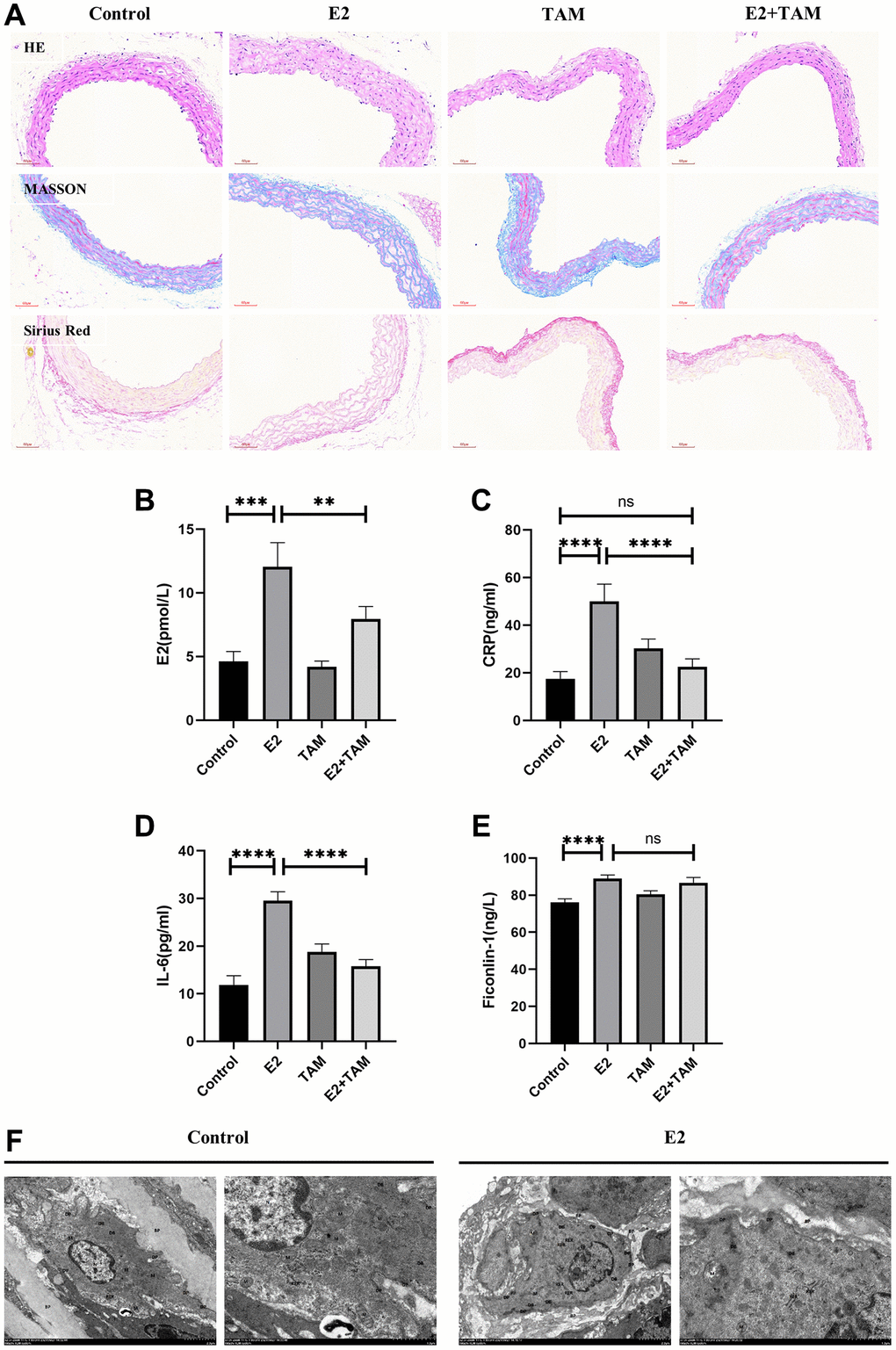
Figure 2. Detection of pathological changes in the aorta and serum inflammatory markers in mice. (A) The abdominal aorta was examined by pathology using HE, Masson’s trichome, and Picro-Sirius Red staining. (B–E) Peripheral blood of mice was taken and serum was centrifuged for analysis. E2, CRP, IL-6 and Ficonlin-1 levels were measured by ELISA. (F) The abdominal aorta of the model group was observed by TEM, which indicated slight swelling of organelles. The nucleus (N) was irregularly shaped and shrunken. Mitochondria (M) were abundant. The RER showed no obvious dilation and a large number of ribosomes attached to the surface. DPs and DBs were abundant and evenly distributed. Magnification 2500× and 7000×. **P < 0.01, ***P < 0.001, ****P < 0.0001, indicating statistically significant data between groups; Abbreviation: ns indicates no statistical significance.
According to the 2022 American College of Rheumatology/EULAR Classification Criteria for TAK [45]. In summary, C57 female mice intravenously injected with E2 for 3 months showed the following manifestations: (1) abnormal blood pressure and pulse rate in the caudal artery, including decreased blood pressure and reduced pulse rate; (2) inflammatory response as indicated by increased serum levels of CRP and IL-6; (3) elevated expression of Ficonlin-1, a potential serum marker of TAK.; (4) pathological manifestations, including thickening of the wall of the abdominal aorta and hyperplastic degeneration of the smooth muscle layer; (5) organ changes, including atrophy of the kidney; and 6) characteristic imaging manifestations as demonstrated by X-ray angiography, which revealed stenosis in the secondary and higher arterial branches of the abdominal aorta, carotid artery, and superior mesenteric artery. These pathologic manifestations are very similar to TAK.
Estrogen induces conversion of VSMCs to a macrophage-like phenotype in mice
TEM analysis of the abdominal aorta of E2 group mice (Figure 2F) showed that VSMCs exhibited a synthetic cell state with abundant mitochondria (M). The rough endoplasmic reticulum (RER) did not show significant expansion, and a large number of ribosomal attachments were visible on the surface. Moreover, dense plaques (DPs) and dense bodies (DBs) were abundant, and VSMCs organelles were mildly swollen. In addition, the cytoplasm was reduced, and the nucleus was shrunk. The TEM results demonstrated that VSMCs showed an active synthetic state with increased cellular protein synthesis and secretion, which differed from the stable contractile phenotype (Figure 2F).
In the E2 group, double staining of the abdominal aorta with ACTA2 and CD68 revealed expression of the CD68 macrophage marker on VSMCs, which was not found in the normal group (Figure 3A). Meanwhile, aortic F4/80 fluorescence intensity was elevated in the E2 group (Supplementary Figure 1A). Western blot analysis of whole mouse aortas revealed increased expression of KLF4, a key protein for VSMC phenotypic transformation, in the E2 group (Figure 3B, 3C and Supplementary Figure 2). In addition, the expression of contractile phenotype-related proteins, ACTA2 was reduced, while the expression of macrophage marker proteins, CD68 was increased (Figure 3B, 3C).
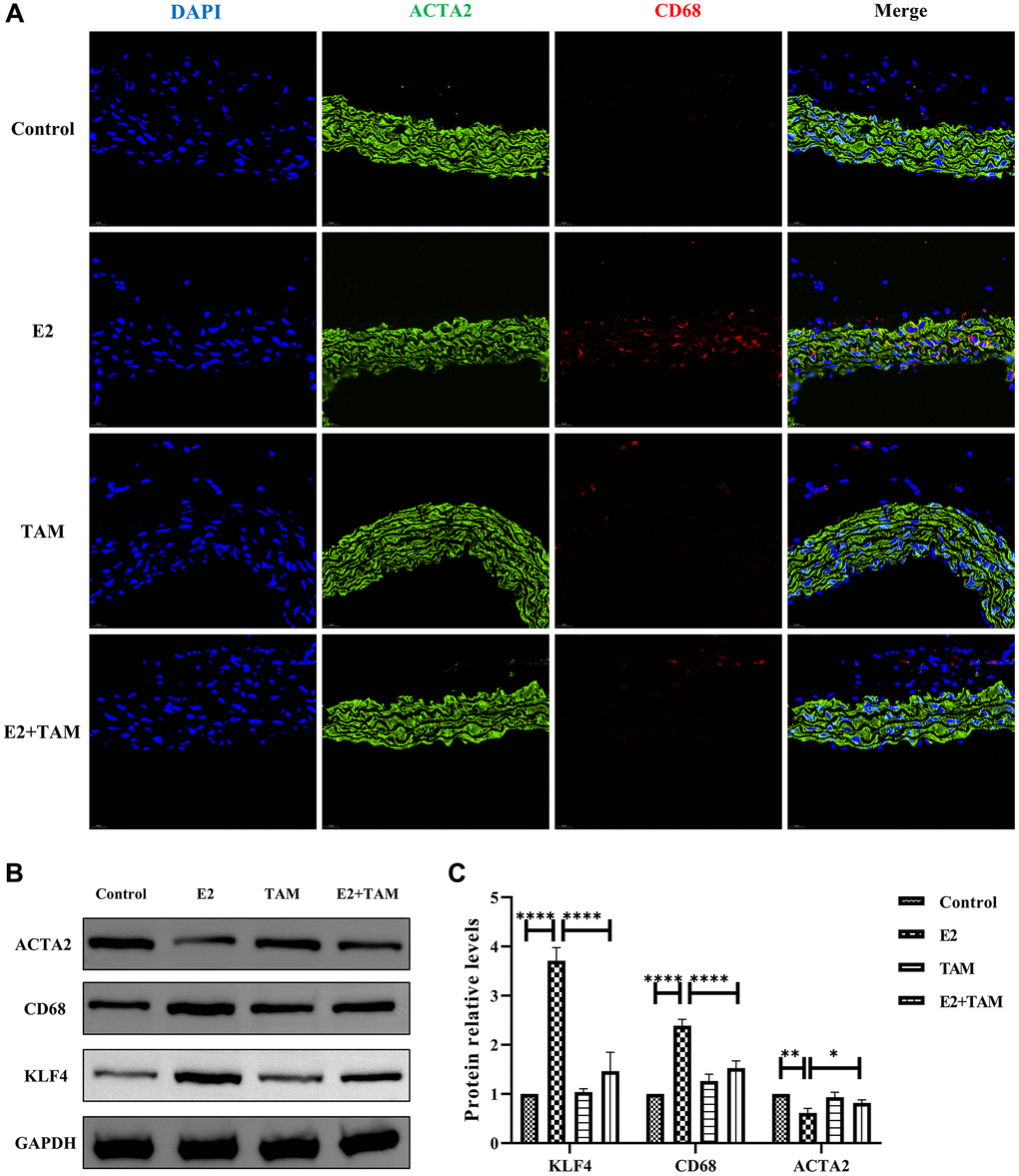
Figure 3. E2 induces transformation of VSMCs into a macrophage-like phenotype in mice and TAM has protective effect on blood vessels. (A) The paraffin sections of abdominal aorta were stained with ACTA2 (green) and CD68 (red). Magnification 630×. (B, C) The whole aortic tissue was lysed, and western blot analysis was performed to analyze the protein levels of KLF4, CD68 and ACTA2 (n = 4). *P < 0.05, **P < 0.01, ****P < 0.0001, indicating statistically significant data between groups.
This suggests that administration of exogenous estrogen to female mice induces the conversion of vascular smooth muscle cells to a macrophage-like phenotype.
Inhibition of estrogen with TAM improves the manifestation of aortic inflammation in mice in vivo
TAM antagonizes the effect of E2 by binding to ESR1. In the mouse model of polyarteritis, TAM ameliorated the E2-induced attenuation of mean arterial pressure and slowing of pulse rate in the tail artery of mice (Figure 1B–1E). Regarding the organ/body weight wet weight ratio, TAM treatment alone, which had no significant effect on organ weight in mice, attenuated the E2-induced weight gain of the liver and did not show E2-induced kidney atrophy (Figure 1F–1I). In addition, TAM reduced the high estrogen, CRP, and IL-6 levels induced by E2, and it also attenuated the inflammatory response. However, there was no significant change in Ficonlin-1 levels in the E2+TAM group compared to the E2 group (Figure 2B–2E).
Pathological staining of the abdominal aorta of mice with HE, Masson’s trichome, and Picro-Sirius Red was performed, which demonstrated that the abdominal aorta of mice in the E2+TAM group was similar to the normal group without the pathological manifestations associated with the E2 group (Figure 2A). Compared to the E2 group, the protein expression of ACTA2 (contractile phenotype) was increased in the E2+TAM group, while the protein expression of CD68 (macrophage marker) was decreased in the E2+TAM group (Figure 3B, 3C). In addition, the expression of KLF4 was lower in the E2+TAM group compared to the E2 group (Figure 3B, 3C). And in immunofluorescence staining, TAM decreased the expression of CD68 (Figure 3A and Supplementary Figure 1B).
Therefore, these findings indicated that TAM ameliorates the pathological manifestations associated with TAK in mice by inhibiting the conversion of VSMCs to a macrophage-like phenotype.
Estrogen-induced conversion of MOVAS cells to a macrophage-like phenotype
Estrogen affected the phenotype of VSMCs in a concentration- and time-dependent manner. The present study confirmed that low concentrations of estrogen maintained the contractile phenotype of VSMCs, while higher concentrations of estrogen induced significant changes in the phenotype of MOVAS cells. After treatment of MOVAS cells with 0.1 nM, 1 nM, 10 nM, 100 nM, 1 μM, and 10 μM E2 for 4 days, and the mRNA expression of ACTA2 and CD68 was detected. The results indicated that 10 μM E2 induced the phenotypic transformation of MOVAS cells (Figure 4A).
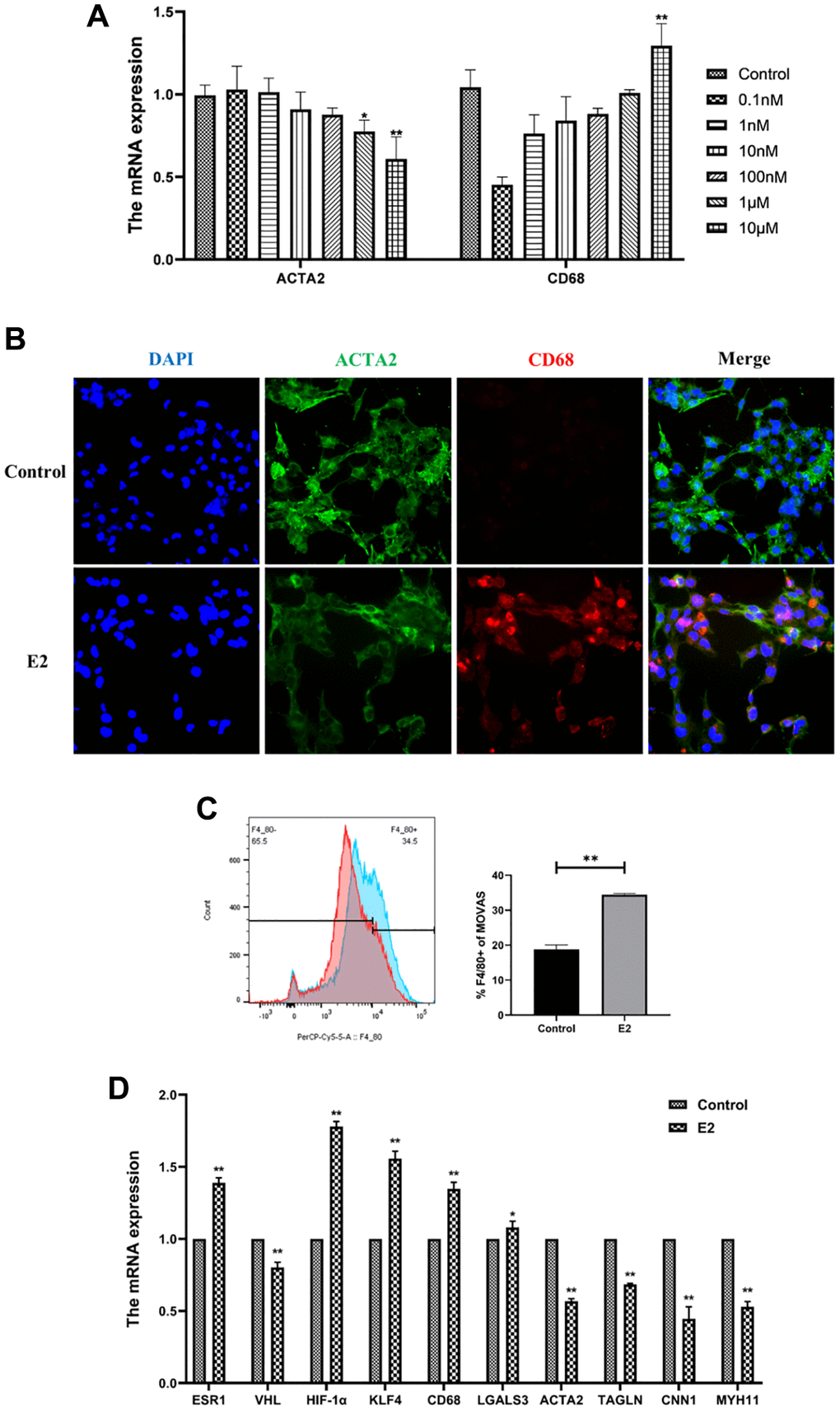
Figure 4. E2 induces transformation of MOVAS cells into a macrophage-like phenotype. (A) MOVAS cells were treated with E2 (0.1 nM, 1 nM, 10 nM, 100 nM, 1 μM, and 10 μM) for 4 days. The contractile phenotype marker, ACTA2, and the macrophage marker, CD68, were detected by qRT-PCR. (B–D) MOVAS cells were treated with 10 μM E2 for 4 days (n = 4). (B) ACTA2 and CD68 levels in MOVAS cells were detected by immunofluorescence analysis. (C) F4/80 antibody expression on the surface of MOVAS cells was detected by FCM analysis (n = 3). (D) Contraction phenotype-related markers (ACTA2, TAGLN, CNN1, and MYH11) and macrophage-like phenotype-related markers (CD68 and LGALS3) in MOVAS cells were evaluated by qRT-PCR (n = 4). *P < 0.05, **P < 0.01, indicating statistically significant data between groups.
Treatment of MOVAS cells with 10 μM E2 for 4 days significantly upregulated CD68 expression in MOVAS cells according to immunofluorescence analysis (Figure 4B). FCM analysis demonstrated that estrogen induced a significant increase in F4/80+ MOVAS cells (Figure 4C). In addition, qRT-PCR indicated that the mRNA expression levels of smooth muscle contraction-related genes, namely, ACTA2, TAGLN, MYH11, and CNN1, were decreased, while the mRNA expression levels of macrophage-related genes, namely, CD68 and LGALS3, were increased (Figure 4D).
This suggests that E2 can induce MOVAS cells to express macrophage markers in an in vitro cellular assay.
High estrogen induces phenotypic trans-differentiation of vascular smooth muscle cells by inhibiting VHL/HIF1a/KLF4 axis
ERα is a novel proteasomal degradation target of the VHL E3 ligase. In the present study, high levels of estrogen increased the expression levels of ERα and increased the degradation rate of VHL. Tamoxifen (TAM), an estradiol competitive antagonist, bound to ESR1 and inhibited the degradation of VHL (Figure 5A). Thus, these results indicated that TAM inhibits the conversion of MOVAS cells to a macrophage-like phenotype and decreases the F4/80+ cell percentage (Figure 6A, 6B). In addition, TAM decreases HIF-1α expression in mouse aortic sections (Supplementary Figure 3).
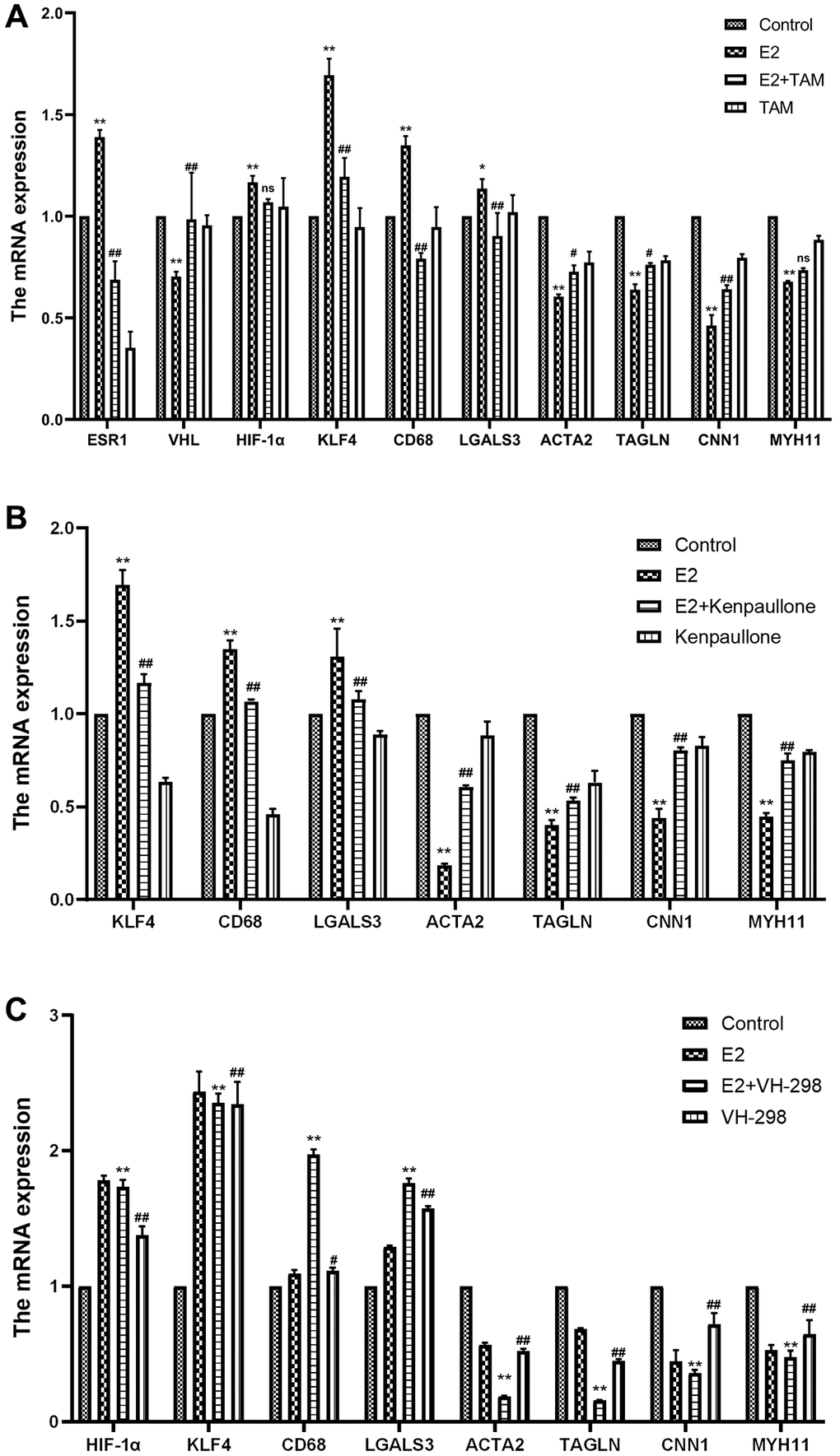
Figure 5. Effect of high-dose estrogen on the expression of MOVAS cell markers. (A) MOVAS cells were treated with 10 μM E2 with or without TAM. After 4 days, the contraction phenotypic markers and macrophage-like phenotypic markers of MOVAS cells were detected by qRT-PCR as well as the mRNA expression of related cellular pathways (n = 4). (B) MOVAS cells were treated with 10 μM E2 with or without kenpaullone. After 4 days, the mRNA expression levels of related genes were detected by qRT-PCR (n = 4). (C) MOVAS cells were treated with 10 μM E2 with or without VH-298 for 4 days, and the mRNA expression levels of related genes were detected by qRT-PCR (n = 4). *VS control group, *P < 0.05, **P < 0.01, indicating statistically significant data between groups; #VS. E2 group, #P < 0.05, ##P < 0.01, indicating statistically significant data between groups; ns indicates no statistical significance.
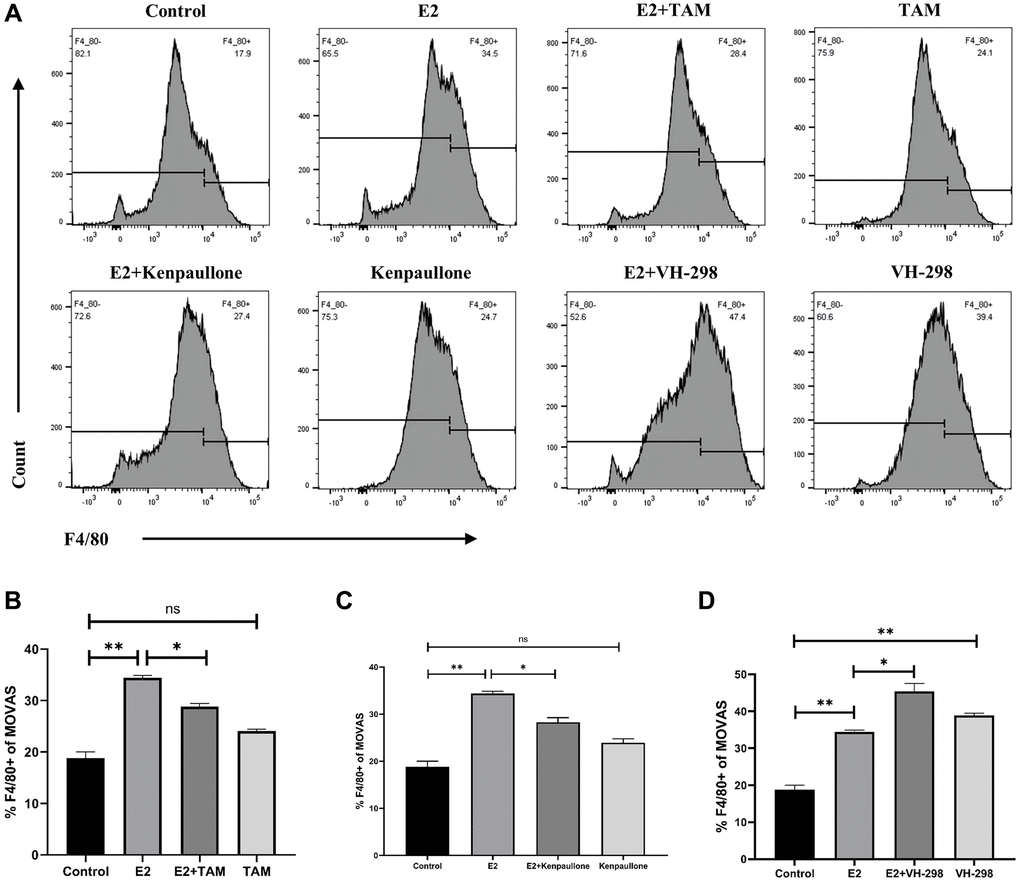
Figure 6. E2 induces phenotypic transformation by regulating the VHL/HIF-1α/KLF4 axis. (A–D) MOVAS cells were treated with 10 μM E2 for 4 days with or without the following inhibitors: Tamoxifen (1 μM), kenpaullone (3 μM), and VH-298 (10 μM). The expression of the F4/80 macrophage marker on MOVAS cells was detected by FCM analysis (n = 3). *P < 0.05, **P < 0.01, indicating statistically significant data between groups; ns indicates no statistical significance.
Kenpaullone is a small molecule inhibitor of KLF4. Treatment with kenpaullone significantly reduced the mRNA expression of E2-induced macrophage-related genes (Figure 5B) and significantly reduced the number of F4/80+ cells in MOVAS cells as detected by FCM analysis (Figure 6A, 6C).
VH-298 is a highly efficient inhibitor of the VHL/HIF-α interaction, and it induces the production of HIF-1α by blocking the VHL-triggered hypoxic response, which in turn causes the accumulation of KLF4 and initiates the phenotypic transformation of MOVAS cells. Treatment of MOVAS cells with VH-298 alone decreased the expression of genes associated with the contractile phenotype but increased the expression of genes associated with the macrophage-like phenotype (Figure 5C). This promotion of phenotypic switching was enhanced by cotreatment with E2. FCM analysis demonstrated that E2+VH-298 treatment increased the percentage of F4/80+ MOVAS cells (Figure 6A, 6D).
These results suggested that E2 induces the conversion of MOVAS cells to a macrophage-like phenotype by regulating the VHL/HIF-1α/KLF4 axis.
Discussion
KLF4 plays a key role in VSMC phenotypic transformation. For the first time, the present study demonstrated that estrogen activated KLF4 by regulating VHL/HIF-1α, which in turn promoted phenotypic transformation of MOVAS cells. Estrogen treatment initiated the transformation of VSMCs to a macrophage-like phenotype by downregulating VHL and reducing the degradation of HIF-1α, thereby inducing the accumulation of the KLF4 transcription factor (Figure 7).
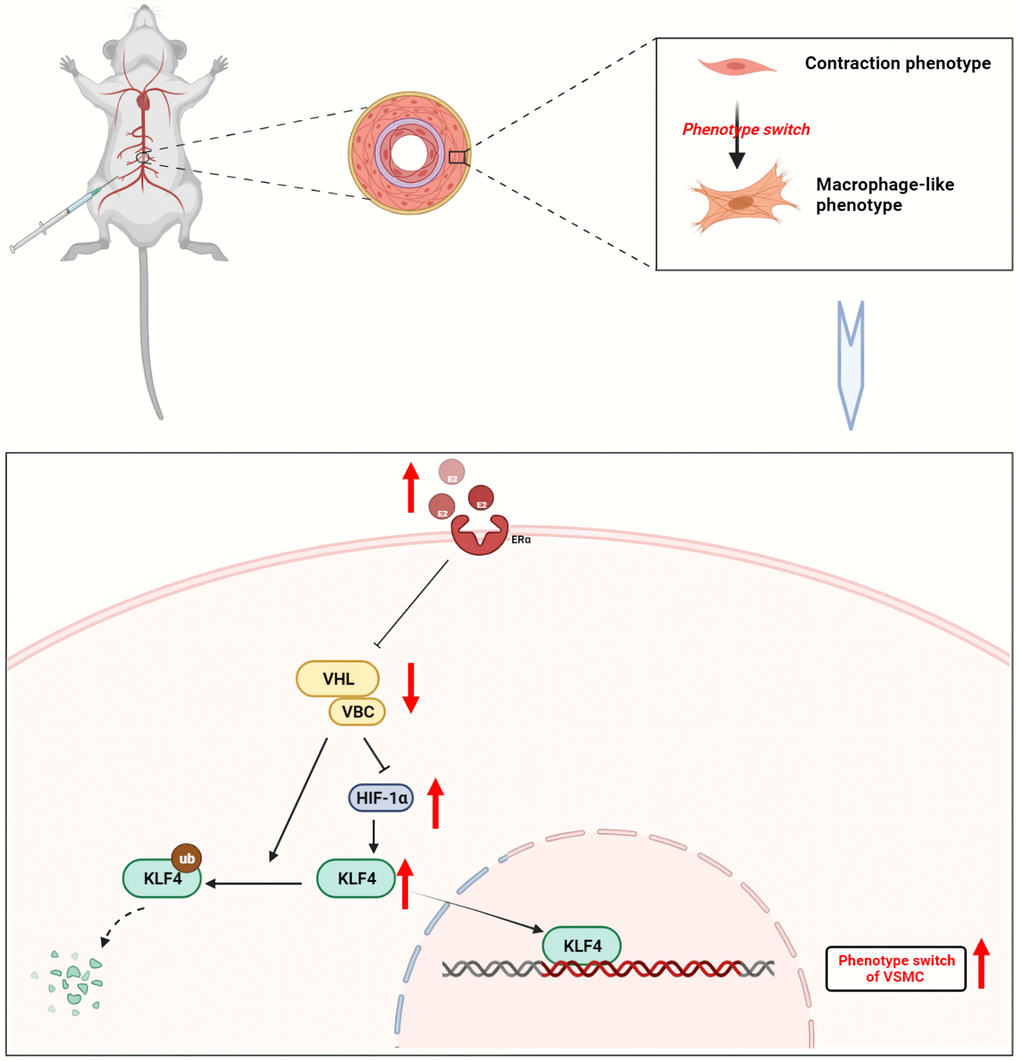
Figure 7. Mechanism of estrogen-induced conversion of VSMCs to a macrophage-like phenotype leading to inflammation in mouse aorta. Upon binding to its receptor, estrogen inhibits the binding of VHL/VBC. VHL induces both ubiquitination degradation of KLF4 and acts to hydrolyze HIF-1α. When VHL expression is reduced, KLF4 expression rises, which in turn enters the nucleus and participates in the phenotype of vascular smooth muscle cells. Created with BioRender.
Large vessel vasculitis mainly includes TAK and giant cell arteritis (GCA), in which the histological lesions show an inflammatory infiltrate mainly located in the tunica media and tunica adventitia of the aorta [46]. VSMCs are major components of the media, and their phenotypic transformation influences the development of vascular inflammatory lesions [47]. TAK is a rare vasculitis that occurs primarily in young women and is characterized by a chronic idiopathic inflammation involving the aorta and its associated branches [48]. Because TAK presents with non-specific symptoms in the acute early stage (headache, fever, and muscle pain), the diagnosis of TAK is often delayed until the onset of substantial arterial injury [49]. TAK initially triggers thickening, fibrosis, and thrombosis of the vessel wall, which may progress to stenosis, occlusion, dilatation, or aneurysm of the affected artery, and it may even be life-threatening [50]. The etiology of TAK is unclear, but there is evidence that pathogenic T cells and macrophages contribute to the chronic inflammatory lesion in the presence of genetic susceptibility, environmental triggers, microorganisms, pathogens, and their antigenic components [51–53]. Given that the proportion of female patients is 5–12 times higher than that of male patients, estrogen is considered an independent risk factor for the pathogenesis of TAK [54]. Most patients with TAK have been diagnosed with hyperestrogenemia. Hyperestrogen exerts a degenerative, atrophic, and profibrotic effect on VSMCs, and the thrombogenic properties of estrogen may also contribute to the development of TAK [55]. In addition, supplementation of exogenous estrogen to female rats induces a disease model similar to human TAK, suggesting that estrogen induces aortic inflammation [56]. Because research on the correlation between estrogen and large vessel inflammation is lacking, the present study investigated the mechanism of estrogen-induced inflammatory changes in large arteries of mice.
By referring to various diagnostic criteria for multiple aortitis, such as the 1990 American College of Rheumatology (ACR) criteria [57], the 2010 European EULAR/PRES/PRINTO criteria [58], and the 2015 Chinese diagnostic criteria for aortitis [59], the present animal model was considered as a mouse model of multiple aortitis. In particular, angiography revealed stenosis of the abdominal aorta, superior mesenteric artery, and carotid artery in mice accompanied by atrophy of the kidneys, supporting successful establishment of the TAK model in mice. For the first time, the present study demonstrated that high doses of estrogen induce VSMCs to convert to a macrophage-like phenotype in this model.
We have previously reported that the transformation of VSMCs to a macrophage-like phenotype occurs in a mouse model of abdominal aortic aneurysm and is associated with chronic inflammation of blood vessels [19, 60]. In contrast, vascular inflammatory diseases are often associated with high estrogen levels and are epidemiologically more prevalent in women of childbearing age [61]. For example, in systemic lupus erythematosus (SLE), estrogen exacerbates the inflammatory response by targeting key immune pathways, including type 1 interferon (IFN) responses, differentiation of CD4+ T helper cells, and survival of self-reactive B cells [62]. Regarding the development of TAK, estrogen plays a pivotal role as high estrogen levels and sustained action of estrogen are involved in the mechanism of TAK injury by inducing the conversion of VSMCs to a macrophage-like phenotype.
KLF4 is a zinc-finger transcription factor that plays a key role in cell proliferation and differentiation in physiological and pathological states, and KLF4 is thought to be a switch for VSMC phenotypic transformation [63]. Transcription of SMC contractile phenotype marker genes is controlled by common cis-acting elements, including multiple CC(A/T)6GG(CArG) elements and a transforming growth factor-β control element (TCE) [63]. KLF4, a transacting factor of TCE, is overexpressed and promotes phenotypic transformation by binding to the TCE in response to the stimulus of induced VSMC phenotypic transformation [64]. In addition, epigenetic regulation is also an important factor determining the differentiation status of VSMCs, especially in the field of histone acetylation and methylation [65]. Interestingly, when a VSMC initiates transformation, the exact type of cell into which it transforms is uncertain and may depend on the stimulus. The present study found that high doses of estrogen induce VSMCs to express macrophage markers, but we did not explore whether this translates into other cellular phenotypes.
During estrogen-induced phenotypic transformation of VSMCs, estrogen leads to VHL degradation by binding to ERα, and the regulation of VHL on KLF4 is not dependent on HIF-1α. VHL directly targets KLF4 for ubiquitination degradation and also acts as a substrate specific splice for the VCB-Cul2 E3 ubiquitin ligase complex. The complex targets HIF-1α for protein hydrolysis, and HIF-1α has two binding elements at the upstream regulatory region motif of KLF4, which can be targeted to induce upregulation of KLF4 [66, 67]. Sivritas et al. treated rat aortic VSMCs with estrogen and found a rapid increase in KLF4 expression and a 50% reduction in cell proliferation after estrogen binding to Erα [68]. These results suggest that estrogen may produce an inhibitory effect on VSMCs proliferation and induce differentiation, which is contrary to previous knowledge.
Although estrogen is generally considered to have a protective effect on blood vessels, adverse effects of estrogen on blood vessels have been reported in recent years. First, estrogen has age-related inflammatory effects on blood vessels. E2 is cardioprotective in young females and postmenopausal women, but vasotoxic effects of E2 have been observed in older females and postmenopausal women [69], which may be related to age-dependent regulation of ERs as older women have reduced expression of ERα and increased expression of ERβ and the novel GPR30 ER [31, 70]. Second, estrogen has differential effects on large vessels and microvessels, which is attributed to different non-genomic mechanisms. In general, estrogen exerts its early anti-inflammatory effects on large vessels probably through inhibition of NF-κB expression, while it exerts its pro-inflammatory effects on microvessels by increasing adhesion molecules (ICAM-1 and VCAM-1) [70]. These results are contradictory to the present study but may be relevant to the pathogenesis of acute and chronic inflammation. Estrogen increases vascular NO production by activating eNOS and iNOS, and the resulting changes in NO concentration determine the effect on inflammation [71]. Thus, estrogen may have an early acute anti-inflammatory effect on the vasculature, which then migrates into a chronic proinflammatory process. Third, there are different ER-mediated signaling pathways. Estrogen has two main receptors, namely, ERα and ERβ. It has been reported that long-term exposure to estrogen upregulates proinflammatory ERα expression in endothelial cells (ECs) (e.g., fetal sheep placental internal arteries) but downregulates anti-inflammatory ERβ expression in human endothelium [72, 73]. In addition, the anti-inflammatory mechanism mediated by the novel GPR30 ER is not fully understood [28]. Fourth, high levels of estrogen have adverse effects on blood vessels. Small doses of estrogen reduce the persistent inflammatory response and promote angiogenesis after chronic spinal cord injury in rats, whereas high doses of estrogen do not have this effect [32]. In addition, natural estrogens have anti-inflammatory properties, while synthetic estrogens have proinflammatory and carcinogenic effects [74]. Barreno et al. first reported inflammatory changes and atherosclerosis in the blood vessels of the testes and epididymis in men taking long-term exogenous estrogens [75]. Therefore, the effects of estrogen levels, as well as the duration of exposure, on the vasculature still need further investigation.
As phytoestrogens have been identified as a natural, plant-based alternative to synthetic sources of estrogen. Over the past 10–15 years, there has been increasing interest in the possibility of estrogen supplementation through diet and supplements in order to improve estrogen levels in postmenopausal women [76, 77]. A growing body of evidence suggests that the consumption of these plant-derived compounds can prevent the onset or attenuate the progression of pathological conditions such as aging, decline in mental acuity, dysmetabolism including atherosclerosis and diabetes, stroke, menopausal symptoms, and osteoporosis [78]. In terms of nutrition, the nutritional effects of estrogen on the uterus and the antioxidant effects of phytoestrogens have also received attention from researchers [79, 80]. It is worth noting that, based on the results of this experiment, there are still potential risks during estrogen application.
ERT, a treatment by supplementing or replacing estrogen in the body, has been used for a long time as a mainstream therapy for disease prevention in postmenopausal women, for the prevention of menopausal osteoporosis, enhancement of immune function, and improvement of symptoms of endocrine disorders in menopausal patients. Estrogen replacement therapy has the following advantages: (1) Relief of menopausal symptoms: ERT can help relieve menopausal symptoms such as insomnia, hot flashes, and vasodilatory instability (i.e., night sweats). Hot flashes may be due to disruption of the thermoregulatory center caused by a decrease in estrogen levels, leading to increased vasodilation and sweating [81]. (2) Improvement of metabolic syndrome: ERT is beneficial for blood pressure, glycemic control, low density lipoprotein, cholesterol, triglycerides and lipoprotein a [82]. (3) Protect bone density: Estrogen has a protective effect on bones, slowing the loss of bone density and strength and reducing the risk of pathologic fracture breaks. (4) Improve cognition: cognitive decline and dementia syndromes such as Alzheimer's disease can be prevented in postmenopausal women through the use of ERT [83]. (5) Reducing the risk of cardiovascular disease: estrogen promotes blood vessel formation and protects the heart from ischemic damage; estrogen prevents endothelial damage, platelet aggregation, and plaque formation, which ultimately reduces the risk of atherosclerosis; estrogen has demonstrated cardiovascular benefits in several research trials, but the mode of administration and the age of initiation are still topics of discussion [84]. In addition, estrogen replacement therapy has the following disadvantages: (1) Increased risk of breast cancer: long-term use of ERT may increase the risk of breast cancer. (2) Increased risk of endometrial cancer: long-term use of ERT may increase the risk of endometrial cancer in women who have not yet had their uterus removed. 3) Cardiovascular Disease Risk: Excessive ERT may increase the risk of stroke and venous thromboembolism, especially in older women with pre-existing cardiovascular disease or high-risk factors [85]. In conclusion, estrogen replacement therapy has advantages in relieving menopausal symptoms and protecting bone density, but its role in cardiovascular disease is unclear.
Conclusion
In summary, the present study demonstrated that estrogen induces MOVAS cells to transform into a macrophage-like phenotype in a dose- and time-dependent manner. In addition, high dose and prolonged estrogen treatment induces inflammatory lesions in large arteries in female mice similar to human TAK. Moreover, estrogen binding to ERα targets VSMC phenotypic transformation through the VHL/HIF-1α/KLF4 axis. These data suggested that estrogen may play a key role in the development of TAK by regulating the phenotype transformation of VSMCs to a macrophage-like phenotype, providing new insights into the mechanisms of phenotypic transformation in vascular disease.
Materials and Methods
Animal treatment
Female C57B6/J mice (6–8 weeks old and weighing 20 ± 2 g) were purchased from the Shanxi Medical University Experimental Animal Center (Taiyuan, China) and maintained in a specific pathogen-free environment at our facility (SYXK 2021-0001). All animals were fed standard food and had free access to water. All animal experiments were conducted in a humane manner and according to the Institutional Animal Care Instructions. The mice were randomly divided into the following four groups: control, no treatment; E2, 400 μg/kg estrogen; TAM, 10 mg/kg Tamoxifen; E2+TAM, combined treatment of 400 μg/kg estrogen and 10 mg/kg Tamoxifen. Stock solutions were prepared by dissolving drugs in 100% ethanol, which were then dissolved in sterile saline for intraperitoneal injections, which were given once a day for three months. The above drugs were purchased from MedChemExpress (Monmouth Junction, NJ, USA).
Cell culture and treatment
The mouse vascular aortic smooth muscle cell line, MOVAS, was purchased from KeyGen Biotech (Nanjing, China). Cells were cultured in high glucose Dulbecco’s Modified Eagle’s Medium (Gibco, Grand Island, NY, USA) supplemented with 10% FBS (CellMax, Beijing, China) and 1% penicillin/streptomycin (Gibco, Grand Island, NY, USA) at 37°C in a humidified 5% CO2 atmosphere. Cells were seeded in 6-well plates at a density of 1.0 × 104 cells/cm2.
To induce the transformation of MOVAS cells into a macrophage-like phenotype, cells were exposed to estrogen with or without the addition of the following drugs: Tamoxifen, which blocks estrogen action; kenpaullone, a small molecule inhibitor of KLF4; and VH-298, a highly potent inhibitor of the VHL/HIF-α interaction. After the treatment at the required concentration and time, the culture medium was discarded, and cells were collected for further analyses. The above drugs were purchased from MedChemExpress (Monmouth Junction, NJ, USA).
Measurement of blood pressure and pulse
The blood pressure of the tail artery and the heart rate were measured by the tail-cuff method in a quiet environment after completion of the injections. The tail was preheated to approximately 38°C and fixed in a comfortable position. The compression cuff was placed at the root of the tail, inflated, and pressurized. When the pressure reached 270 mm Hg (1 mm Hg = 0.133 kPa), the pressure reduced automatically and slowly, and the blood pressure and heart rate were recorded by the BP-2000 Blood Pressure Analysis System (Visitech systems, Apex, NC, USA), which was connected to a pressure transducer and an amplifier. The blood pressure and heart rates were measured three times a day for five days for all mice, and the means were calculated and used as the blood pressure and heart rate. The appetite, activity, body temperature, body weight (BW), and other reactions of the animals were observed daily until the end of the experiment.
Serum analysis
ELISAs were used to analyze mouse serum using the following kits according to the manufacturer’s instructions: E2 (Ruixin Biotech, RXJ203008M), CRP (Ruixin Biotech, RX203183M), IL-6 (Ruixin Biotech, RX203049M), and FCN1 (Meimian, MM-46191M2).
X-ray angiography
After anesthetization with pentobarbital sodium, the heart was immediately exposed at the left margin of the sternal incision, and the left ventricle was punctured with a 1.0 mL syringe with a needle at the apex of the heart. Then, 0.5 mL of iodoxanol was quickly injected, and the mouse was immediately placed into a small animal X-ray imaging system for vascular imaging. The imaging parameters were 38 kV, 6 s, and 0.2 mA. Vascular stenosis and renal morphology were observed after image collection.
Histopathological examination
Abdominal aortic tissues were fixed in 10% formalin for 24 h at 4°C and then embedded in paraffin blocks, which were cut into sections (5 μm). The sections were stained with hematoxylin and eosin (HE), Masson’s Trichrome reagent, and Picro-Sirius Red reagent according to standard routine protocols.
Immunofluorescent staining
Cryosections were fixed for 10 min in ice-cold methanol, washed with phosphate buffered saline (PBS), and blocked with 3% BSA. The sections were then incubated with the following primary antibodies: rabbit anti-ACTA2 (Servicebio, GB111364, 1:500) and mouse anti-CD68 (Santa Cruz Biotechnology, sc-20060, 1:200). After washing, the sections were incubated with the following secondary antibodies: FITC-conjugated goat anti-rabbit (Servicebio, GB22303, 1:300) and Cy3-conjugated donkey anti-mouse (Servicebio, GB21301, 1:300). The sections were then counterstained with DAPI (Boster Biotech, AR1176) to visualize nuclei and imaged using a Leica TCSSP8 DMI8 LASX microscope with Leica LASX software.
Quantitative real-time PCR
Total RNA of MOVAS cells was extracted using the M5 Universal RNA Mini Kit Tissue/Cell RNA Rapid Extraction Kit (Mei5 Biotechnology Co., Ltd., Beijing, China). The concentration of total RNA was determined with a SpectraMax QuickDrop (Molecular Devices, Shanghai, China) according to the manufacturer’s protocol. cDNA was synthesized using the PrimeScript™ RT Master Mix (Perfect Real Time) (Takara Bio Inc., Beijing, China) and a C1000 Touch Thermal Cycler (Bio-Rad, Hercules, CA, USA) according to manufacturer’s protocols. qRT-PCR was performed using 2X M5 HiPer Realtime PCR Super mix with Low Rox (Mei5 Biotechnology Co., Ltd., Beijing, China) and a QuantStudio 6 Flex Real-Time PCR System (Thermo Fisher Scientific Inc., Waltham, MA, USA). The cycle threshold (Ct) values were normalized to the ACTB gene as the loading control. Relative gene expression levels were calculated using the 2−ΔΔCt method. Each experiment was repeated in triplicate. The primer sequences (Sangon Biotech Co., Ltd., Shanghai, China) used in this study are listed in Table 1.
Table 1. Primers designed for quantitative amplification.
| Genes | Forward Primer (5′ to 3′) | Reverse Primer (5′ to 3′) |
| Actb | GTGCTATGTTGCTCTAGACTTCG | ATGCCACAGGATTCCATACC |
| Esr1 | CCTGGCTGGAGATTCTGATGATTGG | TCCACCATGCCTTCCACACATTTAC |
| Vhl | TGATGGACTTCTGGTTAACCAA | TTTCAGGGTATACACTGGCAAT |
| Hif-1α | GAATGAAGTGCACCCTAACAAG | GAGGAATGGGTTCACAAATCAG |
| Klf4 | AGTTTGTGCTGAAGGCGTCTCTG | TGGGCTTCCTTTGCTAACACTGATG |
| Cd68 | CCTCTTGCTGCCTCTCATCATTGG | GGCTGGTAGGTTGATTGTCGTCTG |
| Lgals3 | TATGACCTGCCCTTGCCTGGAG | CTGTTTGCGTTGGGTTTCACTGTG |
| Acta2 | CGTGGCTATTCCTTCGTGACTACTG | CGTCAGGCAGTTCGTAGCTCTTC |
| Tagln | ACTCTAATGGCTTTGGGCAGTTTGG | CCTCTTATGCTCCTGGGCTTTCTTC |
| Cnn1 | CAAAGTCAATGTGGGAGTCAAG | CAGTTTGGGATCATAGAGGTGA |
| Myh11 | AAGCAGCTAAAGGACAAAACAG | ATGTCACATTGTCATTTAGCGG |
Western blot
Mouse vascular tissue was placed in RIPA lysis solution containing phosphatase and protease inhibitors and thoroughly ground with a tissue crusher. Protein was quantified with a BCA Protein Assay Kit (Boster Biotech, Wuhan, China) according to the manufacturer’s instruction. Proteins were separated by electrophoresis using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) for 1–1.5 h at 130 V. Subsequently, proteins were transferred to polyvinylidene fluoride membranes (Merck Millipore, Burlington, MA, USA) for 1 h at 320 mA. Membranes were blocked with 5% nonfat dry milk/TBST for 1.5 h at room temperature and then incubated with the following primary antibodies overnight at 4°C: GAPDH (Servicebio, GB15004, 1:2000), KLF4 (Santa Cruz Biotechnology, sc-393462, 1:800), ACTA2 (Santa Cruz Biotechnology, sc-53142, 1:800), SM22α (Proteintech, 10493-1-AP, 1:2000), CD68 (Santa Cruz Biotechnology, sc-20060, 1:800), and EMR1 (Proteintech, 27044-1-AP, 1:1000). After washing, the membranes were incubated with the appropriate HRP-conjugated secondary antibody (Boster Biotech, BA1056, 1:8000) for 1 h at room temperature. Protein signals were visualized using PierceTM ECL Western Blotting Substrate (Boster Biotech, Wuhan, China) and a ChemiDoc System (BioRad, Hercules, CA, USA). Densitometric analyses were performed using Image Lab software (BioRad).
Flow cytometry (FCM) analysis
The expression of the F4/80 macrophage marker in MOVAS cells was detected by FCM. Briefly, MOVAS cells were resuspended in 100 μL of PBS and 2.5 μL of antibody followed by incubation for 0.5 h. Cells were then centrifuged, washed three times with PBS, resuspended in 100 μL of PBS, and analyzed by FCM.
Transmission electron microscope (TEM) analysis
Tissue samples were prefixed with 3% glutaraldehyde, postfixed in 1% osmium tetroxide, dehydrated in an acetone series, infiltrated in Epox 812, and embedded. Semi-thin sections were stained with methylene blue, and ultra-thin sections were cut with a diamond knife and stained with uranyl acetate and lead citrate. Sections were examined with a JEM-1400-FLASH TEM.
Statistical analyses
All statistical analyses were performed using GraphPad Prism 8.0 (GraphPad Software, La Jolla, CA, USA), and the results are presented as the mean ± SEM. Testing for normality was performed by the Kolmogorov–Smirnov test with Dallal–Wilkinson–Lilliefors correction. When comparing two groups, nonparametric Mann-Whitney U-test or unpaired Student’s t-test was used according to the distribution. When comparing three or more groups, ANOVA test was performed with subsequent Dunn’s test and adjustment for multiple comparisons. A two-sided P < 0.05 was considered statistically significant.
Data availability statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding authors.
Supplementary Materials
Author Contributions
RZ and HW contributed equally to this research. RZ and HW conceived the study and carried out the data analysis, interpretation, and manuscript writing. XC and KF helped to collect and analyze the data. TG, XQ and SG helped to perform the experiments. GZ and HD conceived the study and revised the manuscript. All authors contributed to the manuscript and approved its final version.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
All animal experiments were conducted in compliance with National Institutes of Health guidelines and were approved by the Ethics Committee of the second Hospital of Shanxi Medical University (Approval No. DW2022020).
Funding
The research was backed by four projects, including the Shanxi Leading Talent Team Building Program (Grant No. 202204051002010), the Shanxi Provincial Science and Technology Department Centralized Guided Local Projects (Grant No. YDZJSX2021C026), the National Natural Science Foundation of China (Grant No. 81870354) and the Youth Fund of the Second Hospital of Shanxi Medical University (Grant No. 202002-5).
References
- 1. Management of osteoporosis in postmenopausal women: the 2021 position statement of The North American Menopause Society. Menopause. 2021; 28:973–97. https://doi.org/10.1097/GME.0000000000001831 [PubMed]
- 2. Zouboulis CC, Blume-Peytavi U, Kosmadaki M, Roó E, Vexiau-Robert D, Kerob D, Goldstein SR. Skin, hair and beyond: the impact of menopause. Climacteric. 2022; 25:434–42. https://doi.org/10.1080/13697137.2022.2050206 [PubMed]
- 3. Knowlton AA, Lee AR. Estrogen and the cardiovascular system. Pharmacol Ther. 2012; 135:54–70. https://doi.org/10.1016/j.pharmthera.2012.03.007 [PubMed]
- 4. Brann DW, Lu Y, Wang J, Zhang Q, Thakkar R, Sareddy GR, Pratap UP, Tekmal RR, Vadlamudi RK. Brain-derived estrogen and neural function. Neurosci Biobehav Rev. 2022; 132:793–817. https://doi.org/10.1016/j.neubiorev.2021.11.014 [PubMed]
- 5. Vigneswaran K, Hamoda H. Hormone replacement therapy - Current recommendations. Best Pract Res Clin Obstet Gynaecol. 2022; 81:8–21. https://doi.org/10.1016/j.bpobgyn.2021.12.001 [PubMed]
- 6. Patel S, Homaei A, Raju AB, Meher BR. Estrogen: The necessary evil for human health, and ways to tame it. Biomed Pharmacother. 2018; 102:403–11. https://doi.org/10.1016/j.biopha.2018.03.078 [PubMed]
- 7. Blakemore J, Naftolin F. Aromatase: Contributions to Physiology and Disease in Women and Men. Physiology (Bethesda). 2016; 31:258–69. https://doi.org/10.1152/physiol.00054.2015 [PubMed]
- 8. Pan M, Zhou J, Pan X, Wang J, Qi Q, Wang L. Drugs for the treatment of postmenopausal symptoms: Hormonal and non-hormonal therapy. Life Sci. 2023; 312:121255. https://doi.org/10.1016/j.lfs.2022.121255 [PubMed]
- 9. Farkas S, Szabó A, Hegyi AE, Török B, Fazekas CL, Ernszt D, Kovács T, Zelena D. Estradiol and Estrogen-like Alternative Therapies in Use: The Importance of the Selective and Non-Classical Actions. Biomedicines. 2022; 10:861. https://doi.org/10.3390/biomedicines10040861 [PubMed]
- 10. Owens GK. Regulation of differentiation of vascular smooth muscle cells. Physiol Rev. 1995; 75:487–517. https://doi.org/10.1152/physrev.1995.75.3.487 [PubMed]
- 11. Yap C, Mieremet A, de Vries CJM, Micha D, de Waard V. Six Shades of Vascular Smooth Muscle Cells Illuminated by KLF4 (Krüppel-Like Factor 4). Arterioscler Thromb Vasc Biol. 2021; 41:2693–707. https://doi.org/10.1161/ATVBAHA.121.316600 [PubMed]
- 12. Allahverdian S, Chaabane C, Boukais K, Francis GA, Bochaton-Piallat ML. Smooth muscle cell fate and plasticity in atherosclerosis. Cardiovasc Res. 2018; 114:540–50. https://doi.org/10.1093/cvr/cvy022 [PubMed]
- 13. Castro PR, Barbosa AS, Pereira JM, Ranfley H, Felipetto M, Gonçalves CAX, Paiva IR, Berg BB, Barcelos LS. Cellular and Molecular Heterogeneity Associated with Vessel Formation Processes. Biomed Res Int. 2018; 2018:6740408. https://doi.org/10.1155/2018/6740408 [PubMed]
- 14. Sorokin V, Vickneson K, Kofidis T, Woo CC, Lin XY, Foo R, Shanahan CM. Role of Vascular Smooth Muscle Cell Plasticity and Interactions in Vessel Wall Inflammation. Front Immunol. 2020; 11:599415. https://doi.org/10.3389/fimmu.2020.599415 [PubMed]
- 15. Feil S, Fehrenbacher B, Lukowski R, Essmann F, Schulze-Osthoff K, Schaller M, Feil R. Transdifferentiation of vascular smooth muscle cells to macrophage-like cells during atherogenesis. Circ Res. 2014; 115:662–7. https://doi.org/10.1161/CIRCRESAHA.115.304634 [PubMed]
- 16. Kendrick J, Chonchol M. The role of phosphorus in the development and progression of vascular calcification. Am J Kidney Dis. 2011; 58:826–34. https://doi.org/10.1053/j.ajkd.2011.07.020 [PubMed]
- 17. Liang X, He W, Zhang H, Luo D, Zhang Z, Liu A, Wang J, Huang H. Inflammatory Cells Accelerated Carotid Artery Calcification via MMP9: Evidences From Single-Cell Analysis. Front Cardiovasc Med. 2021; 8:766613. https://doi.org/10.3389/fcvm.2021.766613 [PubMed]
- 18. Lazaro I, Lopez-Sanz L, Bernal S, Oguiza A, Recio C, Melgar A, Jimenez-Castilla L, Egido J, Madrigal-Matute J, Gomez-Guerrero C. Nrf2 Activation Provides Atheroprotection in Diabetic Mice Through Concerted Upregulation of Antioxidant, Anti-inflammatory, and Autophagy Mechanisms. Front Pharmacol. 2018; 9:819. https://doi.org/10.3389/fphar.2018.00819 [PubMed]
- 19. Cao G, Lu Z, Gu R, Xuan X, Zhang R, Hu J, Dong H. Deciphering the Intercellular Communication Between Immune Cells and Altered Vascular Smooth Muscle Cell Phenotypes in Aortic Aneurysm From Single-Cell Transcriptome Data. Front Cardiovasc Med. 2022; 9:936287. https://doi.org/10.3389/fcvm.2022.936287 [PubMed]
- 20. Bennett MR, Sinha S, Owens GK. Vascular Smooth Muscle Cells in Atherosclerosis. Circ Res. 2016; 118:692–702. https://doi.org/10.1161/CIRCRESAHA.115.306361 [PubMed]
- 21. Frismantiene A, Philippova M, Erne P, Resink TJ. Smooth muscle cell-driven vascular diseases and molecular mechanisms of VSMC plasticity. Cell Signal. 2018; 52:48–64. https://doi.org/10.1016/j.cellsig.2018.08.019 [PubMed]
- 22. Shi J, Yang Y, Cheng A, Xu G, He F. Metabolism of vascular smooth muscle cells in vascular diseases. Am J Physiol Heart Circ Physiol. 2020; 319:H613–31. https://doi.org/10.1152/ajpheart.00220.2020 [PubMed]
- 23. Sazonova OV, Isenberg BC, Herrmann J, Lee KL, Purwada A, Valentine AD, Buczek-Thomas JA, Wong JY, Nugent MA. Extracellular matrix presentation modulates vascular smooth muscle cell mechanotransduction. Matrix Biol. 2015; 41:36–43. https://doi.org/10.1016/j.matbio.2014.11.001 [PubMed]
- 24. Lacolley P, Regnault V, Nicoletti A, Li Z, Michel JB. The vascular smooth muscle cell in arterial pathology: a cell that can take on multiple roles. Cardiovasc Res. 2012; 95:194–204. https://doi.org/10.1093/cvr/cvs135 [PubMed]
- 25. Tóth A, Balogh E, Jeney V. Regulation of Vascular Calcification by Reactive Oxygen Species. Antioxidants (Basel). 2020; 9:963. https://doi.org/10.3390/antiox9100963 [PubMed]
- 26. Khalil RA. Estrogen, vascular estrogen receptor and hormone therapy in postmenopausal vascular disease. Biochem Pharmacol. 2013; 86:1627–42. https://doi.org/10.1016/j.bcp.2013.09.024 [PubMed]
- 27. Barnabas O, Wang H, Gao XM. Role of estrogen in angiogenesis in cardiovascular diseases. J Geriatr Cardiol. 2013; 10:377–82. https://doi.org/10.3969/j.issn.1671-5411.2013.04.008 [PubMed]
- 28. Aryan L, Younessi D, Zargari M, Banerjee S, Agopian J, Rahman S, Borna R, Ruffenach G, Umar S, Eghbali M. The Role of Estrogen Receptors in Cardiovascular Disease. Int J Mol Sci. 2020; 21:4314. https://doi.org/10.3390/ijms21124314 [PubMed]
- 29. Iorga A, Cunningham CM, Moazeni S, Ruffenach G, Umar S, Eghbali M. The protective role of estrogen and estrogen receptors in cardiovascular disease and the controversial use of estrogen therapy. Biol Sex Differ. 2017; 8:33. https://doi.org/10.1186/s13293-017-0152-8 [PubMed]
- 30. Dehaini H, Fardoun M, Abou-Saleh H, El-Yazbi A, Eid AA, Eid AH. Estrogen in vascular smooth muscle cells: A friend or a foe? Vascul Pharmacol. 2018; 111:15–21. https://doi.org/10.1016/j.vph.2018.09.001 [PubMed]
- 31. Novella S, Heras M, Hermenegildo C, Dantas AP. Effects of estrogen on vascular inflammation: a matter of timing. Arterioscler Thromb Vasc Biol. 2012; 32:2035–42. https://doi.org/10.1161/ATVBAHA.112.250308 [PubMed]
- 32. Samantaray S, Das A, Matzelle DC, Yu SP, Wei L, Varma A, Ray SK, Banik NL. Administration of low dose estrogen attenuates persistent inflammation, promotes angiogenesis, and improves locomotor function following chronic spinal cord injury in rats. J Neurochem. 2016; 137:604–17. https://doi.org/10.1111/jnc.13610 [PubMed]
- 33. Maharjan CK, Mo J, Wang L, Kim MC, Wang S, Borcherding N, Vikas P, Zhang W. Natural and Synthetic Estrogens in Chronic Inflammation and Breast Cancer. Cancers (Basel). 2021; 14:206. https://doi.org/10.3390/cancers14010206 [PubMed]
- 34. Zhao Y, Gong P, Chen Y, Nwachukwu JC, Srinivasan S, Ko C, Bagchi MK, Taylor RN, Korach KS, Nettles KW, Katzenellenbogen JA, Katzenellenbogen BS. Dual suppression of estrogenic and inflammatory activities for targeting of endometriosis. Sci Transl Med. 2015; 7:271ra9. https://doi.org/10.1126/scitranslmed.3010626 [PubMed]
- 35. Cephus JY, Gandhi VD, Shah R, Brooke Davis J, Fuseini H, Yung JA, Zhang J, Kita H, Polosukhin VV, Zhou W, Newcomb DC. Estrogen receptor-α signaling increases allergen-induced IL-33 release and airway inflammation. Allergy. 2021; 76:255–68. https://doi.org/10.1111/all.14491 [PubMed]
- 36. Zhang L, Zhu C, Zhang X, Wan Y, Song J. Dual effects of estrogen on vascular smooth muscle cells: receptor-mediated proliferative vs. metabolite-induced pro-senescent actions. Steroids. 2011; 76:309–16. https://doi.org/10.1016/j.steroids.2010.12.002 [PubMed]
- 37. Shankman LS, Gomez D, Cherepanova OA, Salmon M, Alencar GF, Haskins RM, Swiatlowska P, Newman AA, Greene ES, Straub AC, Isakson B, Randolph GJ, Owens GK. KLF4-dependent phenotypic modulation of smooth muscle cells has a key role in atherosclerotic plaque pathogenesis. Nat Med. 2015; 21:628–37. https://doi.org/10.1038/nm.3866 [PubMed]
- 38. Majesky MW, Horita H, Ostriker A, Lu S, Regan JN, Bagchi A, Dong XR, Poczobutt J, Nemenoff RA, Weiser-Evans MC. Differentiated Smooth Muscle Cells Generate a Subpopulation of Resident Vascular Progenitor Cells in the Adventitia Regulated by Klf4. Circ Res. 2017; 120:296–311. https://doi.org/10.1161/CIRCRESAHA.116.309322 [PubMed]
- 39. Hu D, Zhou Z, Davidson NE, Huang Y, Wan Y. Novel insight into KLF4 proteolytic regulation in estrogen receptor signaling and breast carcinogenesis. J Biol Chem. 2012; 287:13584–97. https://doi.org/10.1074/jbc.M112.343566 [PubMed]
- 40. Zhou Z, Song X, Chi JJ, Gius DR, Huang Y, Cristofanilli M, Wan Y. Regulation of KLF4 by posttranslational modification circuitry in endocrine resistance. Cell Signal. 2020; 70:109574. https://doi.org/10.1016/j.cellsig.2020.109574 [PubMed]
- 41. Frost J, Galdeano C, Soares P, Gadd MS, Grzes KM, Ellis L, Epemolu O, Shimamura S, Bantscheff M, Grandi P, Read KD, Cantrell DA, Rocha S, Ciulli A. Potent and selective chemical probe of hypoxic signalling downstream of HIF-α hydroxylation via VHL inhibition. Nat Commun. 2016; 7:13312. https://doi.org/10.1038/ncomms13312 [PubMed]
- 42. Shulman M, Shi R, Zhang Q. Von Hippel-Lindau tumor suppressor pathways & corresponding therapeutics in kidney cancer. J Genet Genomics. 2021; 48:552–9. https://doi.org/10.1016/j.jgg.2021.05.016 [PubMed]
- 43. Okuzaki D, Kobayashi S, Sakurai MA, Torigata K, Okamoto A, Matsumoto T, Daida H, Ito A, Nojima H. Ficolin 1 Expression is Elevated in the Peripheral Blood Mononuclear Cells of Takayasu’s Vasculitis Patients. J Mol Biomark Diagn. 2012; 3:1000125. https://doi.org/10.4172/2155-9929.1000125
- 44. Huang R, He Y, Sun B, Liu B. Bioinformatic Analysis Identifies Three Potentially Key Differentially Expressed Genes in Peripheral Blood Mononuclear Cells of Patients with Takayasu's Arteritis. Cell J. 2018; 19:647–53. https://doi.org/10.22074/cellj.2018.4991 [PubMed]
- 45. Grayson PC, Ponte C, Suppiah R, Robson JC, Gribbons KB, Judge A, Craven A, Khalid S, Hutchings A, Danda D, Luqmani RA, Watts RA, Merkel PA, and DCVAS Study Group. 2022 American College of Rheumatology/EULAR Classification Criteria for Takayasu Arteritis. Arthritis Rheumatol. 2022; 74:1872–80. https://doi.org/10.1002/art.42324 [PubMed]
- 46. Le Joncour A, Desbois AC, Leroyer AS, Tellier E, Régnier P, Maciejewski-Duval A, Comarmond C, Barete S, Arock M, Bruneval P, Launay JM, Fouret P, Blank U, et al. Mast cells drive pathologic vascular lesions in Takayasu arteritis. J Allergy Clin Immunol. 2022; 149:292–301.e3. https://doi.org/10.1016/j.jaci.2021.05.003 [PubMed]
- 47. Ashraf JV, Al Haj Zen A. Role of Vascular Smooth Muscle Cell Phenotype Switching in Arteriogenesis. Int J Mol Sci. 2021; 22:10585. https://doi.org/10.3390/ijms221910585 [PubMed]
- 48. de Souza AW, de Carvalho JF. Diagnostic and classification criteria of Takayasu arteritis. J Autoimmun. 2014; 48-49:79–83. https://doi.org/10.1016/j.jaut.2014.01.012 [PubMed]
- 49. Mason JC. Takayasu arteritis--advances in diagnosis and management. Nat Rev Rheumatol. 2010; 6:406–15. https://doi.org/10.1038/nrrheum.2010.82 [PubMed]
- 50. Russo RAG, Katsicas MM. Takayasu Arteritis. Front Pediatr. 2018; 6:265. https://doi.org/10.3389/fped.2018.00265 [PubMed]
- 51. Mirault T, Guillet H, Messas E. Immune response in Takayasu arteritis. Presse Med. 2017; 46:e189–96. https://doi.org/10.1016/j.lpm.2017.07.002 [PubMed]
- 52. Espinoza JL, Ai S, Matsumura I. New Insights on the Pathogenesis of Takayasu Arteritis: Revisiting the Microbial Theory. Pathogens. 2018; 7:73. https://doi.org/10.3390/pathogens7030073 [PubMed]
- 53. Zaldivar Villon MLF, de la Rocha JAL, Espinoza LR. Takayasu Arteritis: Recent Developments. Curr Rheumatol Rep. 2019; 21:45. https://doi.org/10.1007/s11926-019-0848-3 [PubMed]
- 54. Seyahi E. Takayasu arteritis: an update. Curr Opin Rheumatol. 2017; 29:51–6. https://doi.org/10.1097/BOR.0000000000000343 [PubMed]
- 55. Shimamoto T. Injury and repair in arterial tissue: contraction and blebbing of endothelial cells in atherogenesis and thrombogenesis and abnormal secretion of sex hormone in Takayasu's disease. Angiology. 1974; 25:682–717. https://doi.org/10.1177/000331977402501008 [PubMed]
- 56. Hong Z, Chen F, Fu W, Xu X. [Effect of estrogen on hemodynamics and vascular remodeling with Takayasu arteritis]. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2001; 18:22–5. [PubMed]
- 57. Arend WP, Michel BA, Bloch DA, Hunder GG, Calabrese LH, Edworthy SM, Fauci AS, Leavitt RY, Lie JT, Lightfoot RW
Jr . The American College of Rheumatology 1990 criteria for the classification of Takayasu arteritis. Arthritis Rheum. 1990; 33:1129–34. https://doi.org/10.1002/art.1780330811 [PubMed] - 58. Ozen S, Pistorio A, Iusan SM, Bakkaloglu A, Herlin T, Brik R, Buoncompagni A, Lazar C, Bilge I, Uziel Y, Rigante D, Cantarini L, Hilario MO, et al, and Paediatric Rheumatology International Trials Organisation (PRINTO). EULAR/PRINTO/PRES criteria for Henoch-Schönlein purpura, childhood polyarteritis nodosa, childhood Wegener granulomatosis and childhood Takayasu arteritis: Ankara 2008. Part II: Final classification criteria. Ann Rheum Dis. 2010; 69:798–806. https://doi.org/10.1136/ard.2009.116657 [PubMed]
- 59. Kong X, Ma L, Wu L, Chen H, Ma L, Sun Y, Wu W, Ji Z, Zhang Z, Yang C, Ye S, Chen S, Dai S, et al. Evaluation of clinical measurements and development of new diagnostic criteria for Takayasu arteritis in a Chinese population. Clin Exp Rheumatol. 2015; 33:S-48–55. [PubMed]
- 60. Cao GM, Xuan XZ, Dong HL. Low expression of integrin signaling pathway genes is associated with abdominal aortic aneurysm: a bioinformatic analysis by WGCNA. Eur Rev Med Pharmacol Sci. 2022; 26:2847–60. https://doi.org/10.26355/eurrev_202204_28615 [PubMed]
- 61. Sabbatini AR, Kararigas G. Estrogen-related mechanisms in sex differences of hypertension and target organ damage. Biol Sex Differ. 2020; 11:31. https://doi.org/10.1186/s13293-020-00306-7 [PubMed]
- 62. Hughes GC, Choubey D. Modulation of autoimmune rheumatic diseases by oestrogen and progesterone. Nat Rev Rheumatol. 2014; 10:740–51. https://doi.org/10.1038/nrrheum.2014.144 [PubMed]
- 63. Yoshida T, Hayashi M. Role of Krüppel-like factor 4 and its binding proteins in vascular disease. J Atheroscler Thromb. 2014; 21:402–13. https://doi.org/10.5551/jat.23044 [PubMed]
- 64. Xie C, Ritchie RP, Huang H, Zhang J, Chen YE. Smooth muscle cell differentiation in vitro: models and underlying molecular mechanisms. Arterioscler Thromb Vasc Biol. 2011; 31:1485–94. https://doi.org/10.1161/ATVBAHA.110.221101 [PubMed]
- 65. Spin JM, Maegdefessel L, Tsao PS. Vascular smooth muscle cell phenotypic plasticity: focus on chromatin remodelling. Cardiovasc Res. 2012; 95:147–55. https://doi.org/10.1093/cvr/cvs098 [PubMed]
- 66. Wang P, Zhao L, Gong S, Xiong S, Wang J, Zou D, Pan J, Deng Y, Yan Q, Wu N, Liao B. HIF1α/HIF2α-Sox2/Klf4 promotes the malignant progression of glioblastoma via the EGFR-PI3K/AKT signalling pathway with positive feedback under hypoxia. Cell Death Dis. 2021; 12:312. https://doi.org/10.1038/s41419-021-03598-8 [PubMed]
- 67. Shan F, Huang Z, Xiong R, Huang QY, Li J. HIF1α-induced upregulation of KLF4 promotes migration of human vascular smooth muscle cells under hypoxia. J Cell Physiol. 2020; 235:141–50. https://doi.org/10.1002/jcp.28953 [PubMed]
- 68. Sivritas D, Becher MU, Ebrahimian T, Arfa O, Rapp S, Bohner A, Mueller CF, Umemura T, Wassmann S, Nickenig G, Wassmann K. Antiproliferative effect of estrogen in vascular smooth muscle cells is mediated by Kruppel-like factor-4 and manganese superoxide dismutase. Basic Res Cardiol. 2011; 106:563–75. https://doi.org/10.1007/s00395-011-0174-z [PubMed]
- 69. Novella S, Pérez-Cremades D, Mompeón A, Hermenegildo C. Mechanisms underlying the influence of oestrogen on cardiovascular physiology in women. J Physiol. 2019; 597:4873–86. https://doi.org/10.1113/JP278063 [PubMed]
- 70. Bowling MR, Xing D, Kapadia A, Chen YF, Szalai AJ, Oparil S, Hage FG. Estrogen effects on vascular inflammation are age dependent: role of estrogen receptors. Arterioscler Thromb Vasc Biol. 2014; 34:1477–85. https://doi.org/10.1161/ATVBAHA.114.303629 [PubMed]
- 71. Chambliss KL, Shaul PW. Estrogen modulation of endothelial nitric oxide synthase. Endocr Rev. 2002; 23:665–86. https://doi.org/10.1210/er.2001-0045 [PubMed]
- 72. Ihionkhan CE, Chambliss KL, Gibson LL, Hahner LD, Mendelsohn ME, Shaul PW. Estrogen causes dynamic alterations in endothelial estrogen receptor expression. Circ Res. 2002; 91:814–20. https://doi.org/10.1161/01.res.0000038304.62046.4c [PubMed]
- 73. Tschugguel W, Dietrich W, Zhegu Z, Stonek F, Kolbus A, Huber JC. Differential regulation of proteasome-dependent estrogen receptor alpha and beta turnover in cultured human uterine artery endothelial cells. J Clin Endocrinol Metab. 2003; 88:2281–7. https://doi.org/10.1210/jc.2002-021165 [PubMed]
- 74. Felty Q, Yoo C, Kennedy A. Gene expression profile of endothelial cells exposed to estrogenic environmental compounds: implications to pulmonary vascular lesions. Life Sci. 2010; 86:919–27. https://doi.org/10.1016/j.lfs.2010.04.007 [PubMed]
- 75. Peña Barreno C, Gonzalez-Peramato P, Nistal M. Vascular and inflammatory effects of estrogen and anti-androgen therapy in the testis and epididymis of male to female transgender adults. Reprod Toxicol. 2020; 95:37–44. https://doi.org/10.1016/j.reprotox.2020.05.001 [PubMed]
- 76. Romani AMP. The controversy on the beneficial effect of phytoestrogens in diabetic treatment in postmenopausal women. Biochem Pharmacol. 2021; 190:114619. https://doi.org/10.1016/j.bcp.2021.114619 [PubMed]
- 77. Sirotkin AV, Harrath AH. Phytoestrogens and their effects. Eur J Pharmacol. 2014; 741:230–6. https://doi.org/10.1016/j.ejphar.2014.07.057 [PubMed]
- 78. Branca F, Lorenzetti S. Health effects of phytoestrogens. Forum Nutr. 2005; 100–11. https://doi.org/10.1159/000083773 [PubMed]
- 79. Lephart ED. Phytoestrogens (Resveratrol and Equol) for Estrogen-Deficient Skin-Controversies/Misinformation versus Anti-Aging In Vitro and Clinical Evidence via Nutraceutical-Cosmetics. Int J Mol Sci. 2021; 22:11218. https://doi.org/10.3390/ijms222011218 [PubMed]
- 80. Zhang M, van Ravenzwaay B, Rietjens IMC. Development of a Generic Physiologically Based Kinetic Model to Predict In Vivo Uterotrophic Responses Induced by Estrogenic Chemicals in Rats Based on In Vitro Bioassays. Toxicol Sci. 2020; 173:19–31. https://doi.org/10.1093/toxsci/kfz216 [PubMed]
- 81. Prokai-Tatrai K, Prokai L. The impact of 17β-estradiol on the estrogen-deficient female brain: from mechanisms to therapy with hot flushes as target symptoms. Front Endocrinol (Lausanne). 2024; 14:1310432. https://doi.org/10.3389/fendo.2023.1310432 [PubMed]
- 82. Kim JE, Choi J, Park J, Lee JK, Shin A, Park SM, Kang D, Choi JY. Associations of postmenopausal hormone therapy with metabolic syndrome among diabetic and non-diabetic women. Maturitas. 2019; 121:76–82. https://doi.org/10.1016/j.maturitas.2018.12.012 [PubMed]
- 83. Wong GRM, Lee EJA, Liaw QY, Rajaram H. The role of oestrogen therapy in reducing risk of Alzheimer's disease: systematic review. BJPsych Open. 2023; 9:e194. https://doi.org/10.1192/bjo.2023.579 [PubMed]
- 84. Javed A, Ravi PC, Bilal Delvi S, Faraz Hussain I, Acosta G AJ, Iqbal W, Krishnamaneni V, Alasaadi S, Pradhan S, Vashisht R, Modi S. The Relationship Between Myocardial Infarction and Estrogen Use: A Literature Review. Cureus. 2023; 15:e46134. https://doi.org/10.7759/cureus.46134 [PubMed]
- 85. Mohammed K, Abu Dabrh AM, Benkhadra K, Al Nofal A, Carranza Leon BG, Prokop LJ, Montori VM, Faubion SS, Murad MH. Oral vs Transdermal Estrogen Therapy and Vascular Events: A Systematic Review and Meta-Analysis. J Clin Endocrinol Metab. 2015; 100:4012–20. https://doi.org/10.1210/jc.2015-2237 [PubMed]



