Introduction
Acute myeloid leukemia (AML) is a highly heterogeneous disease characterized by dysplasia and dysdifferentiation of primitive and immature myeloid cells in the bone marrow and peripheral blood cells. Moreover, it represents the most common acute leukemia in adults [1–3]. Risk stratification based on cell morphology and cytogenetics-guided conventional therapies in combination with newly developed molecular targeted regimens and hematopoietic stem cell transplantation (HSCT) have greatly improved the prognosis of AML patients, the current survival rate of AML patients remains poor [4–7]. There is an urgent need for the identification of new biomarkers and the molecular mechanisms involved in leukemogenesis and its progression in order to better define the prognostic subgroups and guide rational disease management of AML.
The poly-ADP-ribose polymerase (PARP) family of genes encompasses 17 members that encode ADP-ribosyl transferases enzymes, which are responsible for the post-translational DNA-damage-dependent modification of nuclear proteins that contribute toward a variety of DNA-repair processes [8–11]. The role of poly (ADP-ribose) polymerases (PARP) in malignancy is well established in BRCA1/2 mutant tumors that are known to be deficient in homologous recombination mechanisms, which leads to their unique susceptibility to PARP1/2 inhibition treatment [12, 13]. In spite of their therapeutic promise in breast and ovarian cancer, the clinical application of PARP inhibitors (PARPis) as an effective treatment has not been widely translated to different cancers, partly because mutations affecting DDR-associated genes are not common in other malignancies, including AML [9, 12–15]. Thus, the dependence on PARPs in other malignancies is not well understood. Intriguingly, there was a recent study by Esposito et al. that demonstrated for the first time a potential utility of PARPi-induced lethality for leukemias driven by AML1-ETO and PML-RARa [15]. Moreover, another study by Molenaar et al. showed that IDH1/2 mutations sensitized AML to PARP inhibition [14], potentiating the possibility of targeting PARPs in AML therapy.
However, the transcriptional expression features and clinical significance of diverse PARP proteins in acute myeloid leukemia (AML) have not been fully established. In this study, we analyzed the association between PARP genes expression and clinical prognostic significance from TCGA and GEO databases to uncover the potential dependence on diverse PARPs in AML and the molecular functions of PARP proteins in AML progression, which may further guide the clinical treatments by targeting PARPs in AML.
Methods
Patients’ data
The RNA expression data, clinical and laboratory parameters data, gene mutation data, and survival data of 173 newly diagnosed AML patients in the TCGA dataset were downloaded from the cBioPortal dataset. The expression differences in PARP family members between AML patients and normal patients were analyzed based on three datasets, i.e., TCGA versus GTEx (UCSC Xena project) [16], GSE15061 [17], and Bloodspot [18].
Gene differential expression analysis
The gene expression data of AML patients and normal patients downloaded from the GSE15061 dataset [17] was normalized before the differential expression analysis of AML patients versus normal patients was performed using an unpaired t test. The gene expression data of the TCGA and GTEx datasets downloaded from the UCSC Xena project were already computed by a standard pipeline; therefore, differential expression analysis was conducted using an unpaired t test [16]. Bloodspot included gene expression data, whereby human normal hematopoiesis cells were from GSE42519 [19] and human AML cells were from GSE13159 [20].
Bioinformatics analysis
PARP10 co-expression analysis was conducted using cBioPortal based on 173 RNA-seq datasets from TCGA AML patients [21]. KEGG pathway analysis of the significant co-expressed genes was conducted using Enrichr [22]. Gene set enrichment analysis (GSEA) of the PARP10 high expression versus low expression groups was conducted using GESA software (Broad) [23].
Cell culture, plasmids, virus package, and cell proliferation assay
Lenti-X 293T cells (Clontech, USA) were cultured in Dulbecco’s Modified Eagle Medium (DMEM, Gibco, USA) with 1 mM sodium pyruvate, 4500 mg/L glucose, and 10% fetal bovine serum (FBS). Cas9-expressing MOLM13 (MOLM13-Cas9) cells were cultured in Roswell Park Memorial Institute (RPMI, USA) 1640 (Thermo Fisher Scientific, USA) with 10% FBS. Cells used in this study were cultured with a medium containing 1/100 Penicillin–Streptomycin (Gibco).
LentiCas9-blast (#52962) and lentiGuide-RFP-blasticidin (#167930) were purchased from Addgene (USA). Two sgRNAs [24] were synthesized, annealed, and inserted into a lentiGuide-RFP-blasticidin plasmid. Lentiviral particles containing sgRNAs were produced by transient transfection of Lenti-X 293T cells using the lipofection method. MOLM13-Cas9 cells were incubated with lentiGuide-RFP-blasticidin lentiviral supernatants for 48 hours, and then, subjected to selection with blasticidin (30 ug/mL) for 3 days. A flow cytometry-based growth competition assay was performed by using lentivirus-infected cells without blasticidin selection. Pooled cells were subjected to flow cytometry to detect the percentage of RFP-positive cells at the indicated days, and the percentage of RFP-positive cells was normalized to day 0. The cell proliferation assay was performed by plating 0.1 × 106 cells per well in a 12 well-plate in triplicate, and monitoring the cell density every two days.
Statistical analyses
Median expression of PARP10 was used as threshold to define PARP10 high or low group in the datasets. The relationship between PARP10 expression and the clinical and laboratory parameters was analyzed using the Chi-square test. Moreover, the Kaplan-Meier method and log-rank test were used to generate survival curves and to analyze the survival differences between the different groups. Univariate and multivariate analyses were performed using Cox proportional hazards model. All of the statistical analyses were performed using SPSS 24.0 software (SPSS Inc., Chicago, IL, USA), R software, and Prism GraphPad. All tests were two-sided and statistical significance was P < 0.05.
Results
Differentially expressed PARPs in AML versus normal bone marrow cells
To explore the transcriptional expression profiles of PARP genes in AML, we first analyzed the mRNA levels of the TCGA versus GTEX databases. The results showed that PARP1, PARP2, PARP3, PARP4, PARP5b, PARP6, PARP8, PARP9, PARP10, PARP11, PARP12, PARP13, PARP14, PARP15, and PARP16 were differentially expressed in AML patients compared to normal bone marrow cells (P < 0.05, Figure 1 and Supplementary Figure 1). Next, we conducted a validation analysis of the GEO database (GSE15061) and found that PARP3, PARP6, PARP10, and PARP11 genes exhibited a similar differential expression pattern, whereby these genes showed significantly higher expressions in AML patients compared to normal bone marrow cells, in both databases (Figure 1 and Supplementary Figure 2). The other validation was performed in the Bloodspot dataset, where only PARP6 and PARP10 exhibited significantly higher expressions in AML compared to equivalent normal cells (Figure 1 and Supplementary Figure 3). Therefore, PARP6 and PARP10 genes might play potential roles in AML progression.
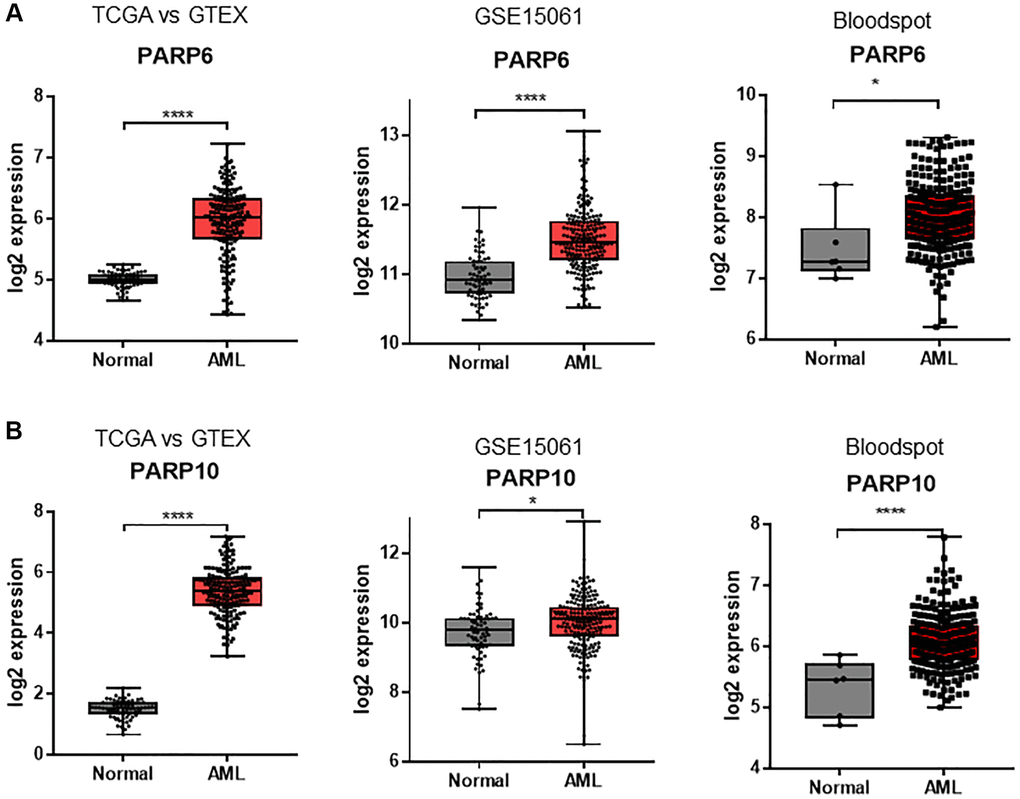
Figure 1. Expression differences of PARPs between AML samples and normal controls in TCGA vs. GTEX, GSE15061, and Bloodspot datasets. (A) PARP6 and (B) PARP10. An unpaired t test was used to estimate the significant differences in expression. *P < 0.05; ****P < 0.001.
High PARP10 expression was associated with poorer survival in AML
To better understand the significance of these two genes (PARP6 and PARP10)—in the prognosis of AML patients, we generated Kaplan-Meier survival curves for the overall survival (OS) and event-free survival (EFS) of patients with high and low expression of these genes (cutoff: median expression). Results showed that patients with higher expressions of PARP10 were significantly associated with poorer OS and EFS compared to patients with lower expressions of PARP10 (OS: median: 0.88 vs. 2.19 years; P = 0.001; EFS: median: 0.65 vs. 1.12 years; P = 0.041), while the expression of PARP6 showed no influence on the prognosis of AML patients (Figure 2 and Supplementary Figure 4). Therefore, we identified PAPR10 as the only gene among the PARP family that was highly expressed and associated with poorer outcomes in AML.
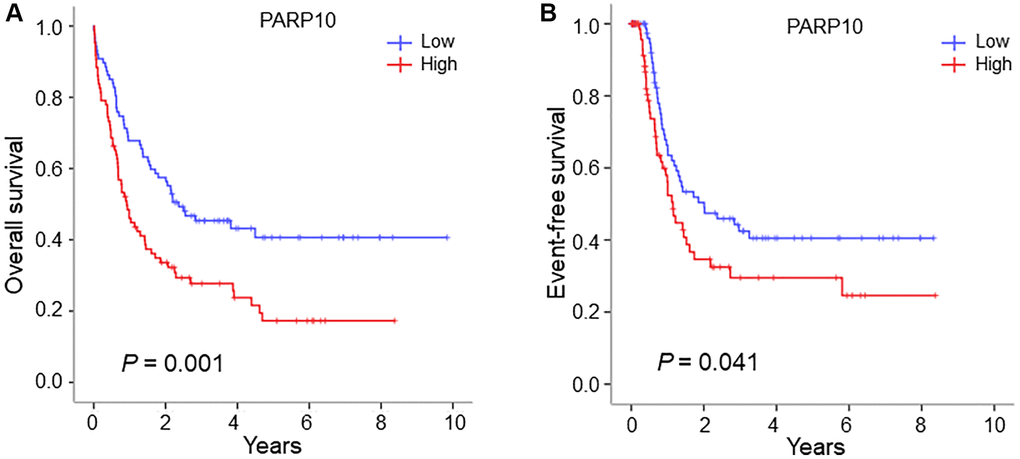
Figure 2. Survival analysis of AML patients with high expression versus low expression in the PARP10 group. (A) Overall survival difference of AML patients with high PARP10 expression versus low PARP10 expression. (B) Event-free survival difference of AML patients with high PARP10 expression versus low PARP10 expression. Log-rank test was used to generate the survival curves and analyze the survival difference between the high- and low-expression groups.
Association of PARP10 expression with clinical characteristics from the TCGA database
We further analyzed the association between PARP10 expression and the clinical characteristics in AML patients taken from the TCGA database (Table 1). We found that the patients present in the high expression PARP10 subgroup were older (≥60 years of age, P = 0.012), possessed more frequent TP53 mutations (P = 0.024), and had shorter overall (P = 0.001) and event-free survivals (P = 0.01). Moreover, the patients who possessed the mutant TP53 showed relatively higher expressions of PARP10 compared to patients carrying the wildtype TP53 (P < 0.05, Figure 3A). PARP10 expression was also significantly associated with FAB subtypes (P = 0.013), ELN risk classification (P = 0.001), and cytogenetic risk classification (P < 0.001). According to ELN risk classification, patients with poorer risks exhibited the highest expression of PARP10, while patients with good risk exhibited the lowest expression of PARP10 (P = 0.001, Figure 3B); cytogenetic risk classification showed a similar pattern (P = 0.006, Figure 3C). No significant differences in sex, white blood cells, bone marrow blasts, peripheral blood blasts, and allo-SCT status were observed in the PARP10 high expression versus low expression groups (P > 0.05) (Table 1). Overall, these data indicated that the high expression of PARP10 was a feature of higher-risk AML, which is more frequently seen in older AML patients, TP53 mutant patients, and those with high-risk karyotypes, suggesting its clinical significance in predicting poorer clinical outcomes of AML patients.
Table 1. Clinical characteristics of AML patients in PARP10 low and high groups from TCGA dataset.
| Clinical parameters | PARP10 low | PARP10 high | P |
| Sex, male/female | 41/46 | 51/35 | 0.109 |
| Age, years (range) | 56 (18–82) | 62 (22-88) | 0.012 |
| WBC, ×109/L (range) | 25.9 (0.4–297.4) | 14.0 (0.6-223.8) | 0.656 |
| BM, % (range) | 72.0 (30–100) | 72.5 (32-99) | 0.69 |
| PB, % (range) | 44.0 (0–98) | 35.5 (0-97) | 0.548 |
| Gene mutations | |||
| NPM1, wildtype/mutant | 62/25 | 63/23 | 0.770 |
| FLT3, wildtype/mutant | 60/27 | 64/22 | 0.426 |
| CEBPA, wildtype/mutant | 82/5 | 78/8 | 0.375 |
| IDH1/IDH2, wildtype/mutant | 72/15 | 69/17 | 0.669 |
| TP53, wildtype/mutant | 84/3 | 75/11 | 0.024 |
| NRAS/KRAS, wildtype/mutant | 78/9 | 76/10 | 0.787 |
| CRC | |||
| Favorable | 26 | 6 | 0 |
| Intermediate | 44 | 48 | |
| Poor | 17 | 29 | |
| ELN risk stratification | |||
| Favorable | 26 | 6 | 0.001 |
| Intermediate | 47 | 54 | |
| Poor | 14 | 23 | |
| FAB | |||
| M0 | 7 | 9 | 0.013 |
| M1 | 16 | 28 | |
| M2 | 19 | 19 | |
| M3 | 14 | 2 | |
| M4 | 22 | 12 | |
| M5 | 6 | 12 | |
| M6 | 1 | 1 | |
| M7 | 1 | 2 | |
| Allo-SCT, NO/Yes | 45/42 | 55/31 | 0.103 |
| OS, years | 2.19 (0–9.84) | 0.88 (0.01–8.38) | 0.001 |
| EFS, years | 1.12 (0–8.33) | 0.65 (0.01–8.38) | 0.01 |
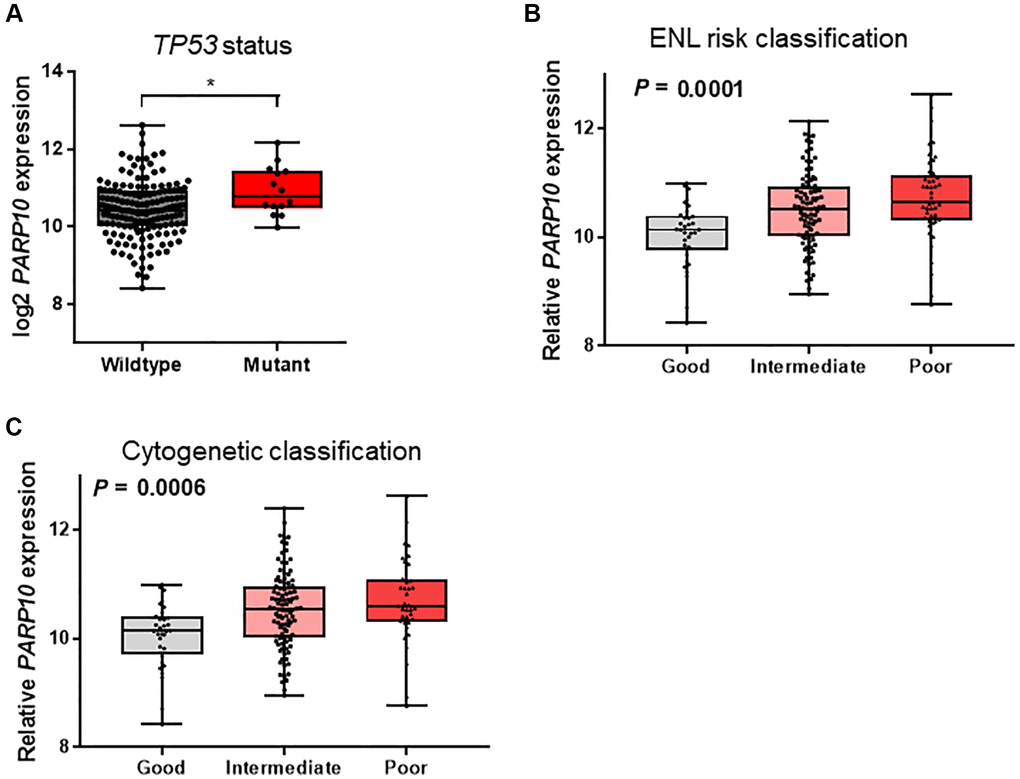
Figure 3. Association of PARP10 expression with TP53 mutation status and risk classifications in AML patients. (A) PARP10 expression difference between AML patients with wildtype and mutant TP53. Statistical significance was estimated using an unpaired t test. (B) PARP10 expression differences among patients with good-risk, intermediate-risk, and poor-risk ENL classifications. (C) PARP10 expression difference among patients with good-risk, intermediate-risk, and poor-risk cytogenetic classifications. Statistical significance was estimated using the Kruskal-Wallis test.
Univariate and multivariate Cox regression analyses identified PARP10 as an independent factor of AML prognosis
To evaluate the prognostic value of PARP10 expression in the presence of other clinical and molecular factors, we included the following dichotomous variables in univariate and multivariate Cox regression analyses: PARP10 expression levels (high vs. low), age (<60 vs. >60), sex (male vs. female), WBC count (<30 vs. ≥30 × 109/L), eight common gene mutations (NPM1, FLT3, CEBPA, IDH1/IDH2, TP53, and NRAS/KRAS; wildtype vs. mutant), and transplant status (no vs. yes) (Table 2).
Univariate Cox regression analysis showed that higher PARP10 expression had an adverse effect on both OS (HR = 1.840, 95% CI 1.007–2.171; P = 0.001) and EFS (HR = 1.567, 95% CI 1.015–2.420; P = 0.043). Lower WBC (<30 × 109/L) and being younger in age (<60 years old) were associated with favorable EFS (HR = 0.485, 95% CI 0.314–0.749; P = 0.001) and OS (HR = 0.319, 95% CI 0.219–0.466; P < 0.001), respectively. Wildtype FLT3 and TP53 were also associated with a more favorable EFS (HR = 0.585 95% CI 0.368–0.930; P = 0.023) and OS (HR = 0.373 95% CI 0.205–0.679; P < 0.001), respectively. Furthermore, not having received transplant contributed to an inferior OS (HR = 1.925, 95% CI 1.314–2.821; P = 0.001), while the other clinical and molecular factors had no effect on either OS or EFS (P > 0.05) (Tables 2 and 3).
Table 2. Cox regression analysis of multivariable for overall survival in AML patients.
| Variables | OS | |||
| Univariate analysis | Multivariate analysis | |||
| Hazard ratio (95% CI) | P value | Hazard ratio (95% CI) | P value | |
| WBC | 0.824 (0.569–1.195) | 0.308 | ||
| Age | 0.319 (0.219–0.466) | 0.000 | 0.420 (0.278–0.634) | 0.000 |
| Sex | 1.056 (0.731–1.526) | 0.77 | ||
| PARP10 | 1.840 (1.267–2.673) | 0.001 | 1.478 (1.007–2.171) | 0.046 |
| NPM1 | 0.87 (0.58–1.30) | 0.486 | ||
| FLT3 | 0.755 (0.504–1.129) | 0.171 | ||
| CEBPA | 1.077 (0.545–2.129) | 0.931 | ||
| IDH1/IDH2 | 1.188 (0.733–1.927) | 0.484 | ||
| TP53 | 0.244 (0.136–0.437) | 0.000 | 0.373 (0.205–0.679) | 0.001 |
| NRAS/KRAS | 0.945 (0.53–1.683) | 0.846 | ||
| Transplant | 1.925 (1.314–2.821) | 0.001 | 1.455 (0.972–2.179) | 0.069 |
Table 3. Cox regression analysis of multivariable for event-free survival in AML patients.
| Variables | EFS | |||
| Univariate analysis | Multivariate analysis | |||
| Hazard ratio (95% CI) | P value | Hazard ratio (95% CI) | P value | |
| WBC | 0.485 (0.314–0.749) | 0.001 | 0.477 (0.301–0.755) | 0.002 |
| Age | 0.71 (0.452–1.115) | 0.137 | ||
| Sex | ||||
| PARP10 | 1.567 (1.015–2.420) | 0.043 | 1.678 (1.069–2.634) | 0.024 |
| NPM1 | 0.753 (0.47–1.206) | 0.238 | ||
| FLT3 | 0.585 (0.368–0.930) | 0.023 | 0.597 (0.369–0.966) | 0.036 |
| CEBPA | 0.705 (0.339–1.466) | 0.349 | ||
| IDH1/IDH2 | 1.181(0.674–2.071) | 0.561 | ||
| TP53 | 0.422 (0.169–1.055) | 0.065 | 0.328 (0.124–0.867) | 0.025 |
| NRAS/KRAS | 0.779 (0.402–1.511) | 0.460 | ||
| Transplant | 0.647 (0.414–1.01) | 0.055 | 0.734 (0.467–1.154) | 0.180 |
Multivariate Cox regression analysis indicated that high PAPR10 expression and TP53 status were independent risk factors for both OS and EFS, even in the presence of the other covariates (P < 0.05, Tables 2 and 3). Age group was also an independent risk factor for OS (P < 0.001) after adjusting for the PARP10 group, TP53, and transplant status. The WBC group and FLT3 status were independent risk factors for EFS after adjusting for the PARP10 group, TP53, and transplant status (P < 0.05). Therefore, our Cox regression analyses identified high PARP10 expression as an independent risk factor for both OS and EFS in AML patients.
Allo-SCT improved the prognosis of AML patients with high PARP10 expression but not patients with low PARP10 expression
As allo-SCT has a profound positive influence on the prognosis of AML [25, 26], we further analyzed the influence of transplant status on OS and EFS in PARP10 high and low AML patients (Figure 4). The results showed that among patients with a high expression of PARP10, allo-SCT significantly increased their OS compared to the group without transplants (P < 0.001), while there was no difference in EFS between these two groups (P = 0.898) (Figure 4A and 4B). Among patients with a low expression of PARP10, allo-SCT did not increase their OS compared to the group without transplants (P = 0.273), whereas allo-SCT significantly decreased the EFS compared to the group without transplants (P = 0.007) (Figure 4C and 4D). These results suggest that allo-SCT therapy can benefit patients with a higher expression of PARP10 while having an adverse effect on those with a lower expression of PARP10. Thus, our results suggest that allo-SCT therapy is highly recommended for patients with higher expressions of PARP10, while it is inappropriate for patients with low expressions of PARP10.
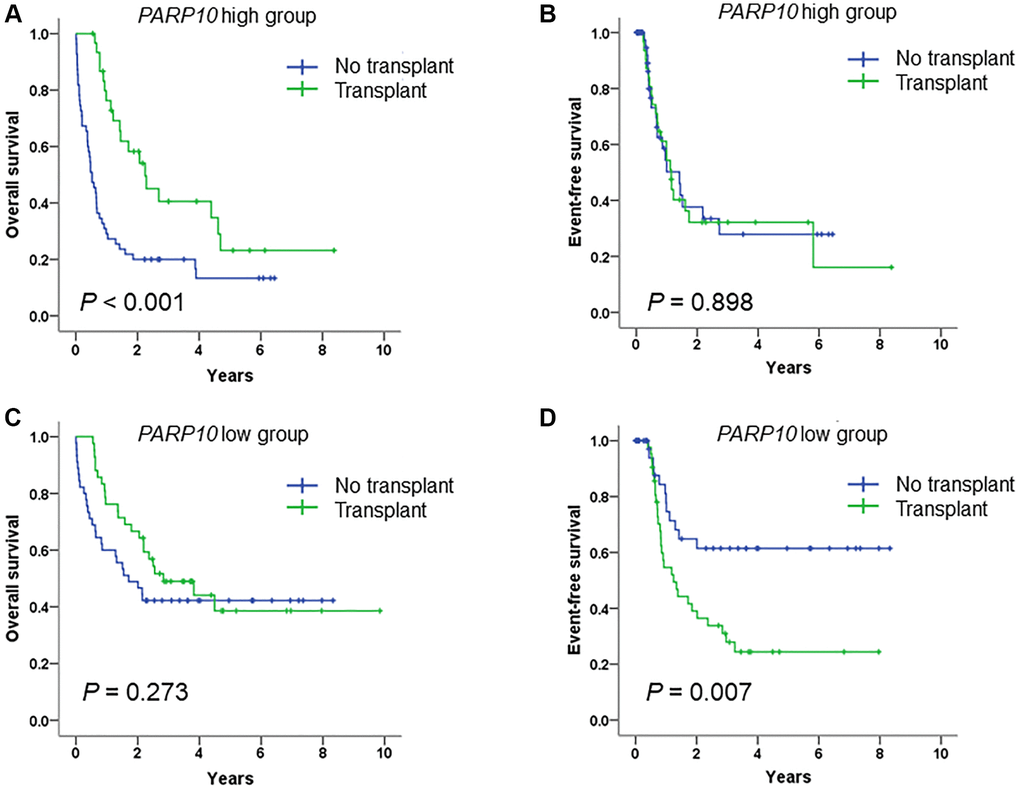
Figure 4. Survival analysis of AML patients who received intensive chemotherapy or intensive chemotherapy followed by allo-SCT, according to PARP10 expression. Overall survival rate (A) and event-free survival rate (B) of AML patients who received intensive chemotherapy versus intensive chemotherapy followed by allo-SCT in the PARP10 high group. Overall survival rate (C) and event-free survival rate (D) of AML patients who received intensive chemotherapy versus intensive chemotherapy followed by allo-SCT in the PARP10 low group.
Potential molecular mechanism mediated by PARP10 in AML
To further explore the mechanism of PARP10 in AML, we performed co-expression network analysis using RNA sequencing data from the 173 AML patients taken from the cBioPortal database [21] and found 558 positively co-expressed genes (r > 0.5; P < 0.05) and 94 negatively co-expressed genes (r < −0.5; P < 0.05) (Supplementary Table 1). KEGG pathway analysis revealed that the 558 positively co-expressed genes were significantly enriched in the oncogenic pathways (e.g., MAPK signaling pathway, AMPK signaling pathway, NF-kappa B signaling pathway, mTOR signaling pathway, and pancreatic cancer), and intriguingly, the chronic myeloid leukemia and acute myeloid leukemia pathways were also among the top enriched pathways (adjusted P < 0.05), indicating that PARP10 is highly relevant to AML, potentially through these functional pathways (Figure 5A and Supplementary Table 2). However, KEGG pathway analysis of the 94 negatively co-expressed genes (r < −0.5; P < 0.05) did not illustrate any significantly enriched pathways (adjusted P > 0.05) (Supplementary Table 3), although this could be due to the limited number of genes.
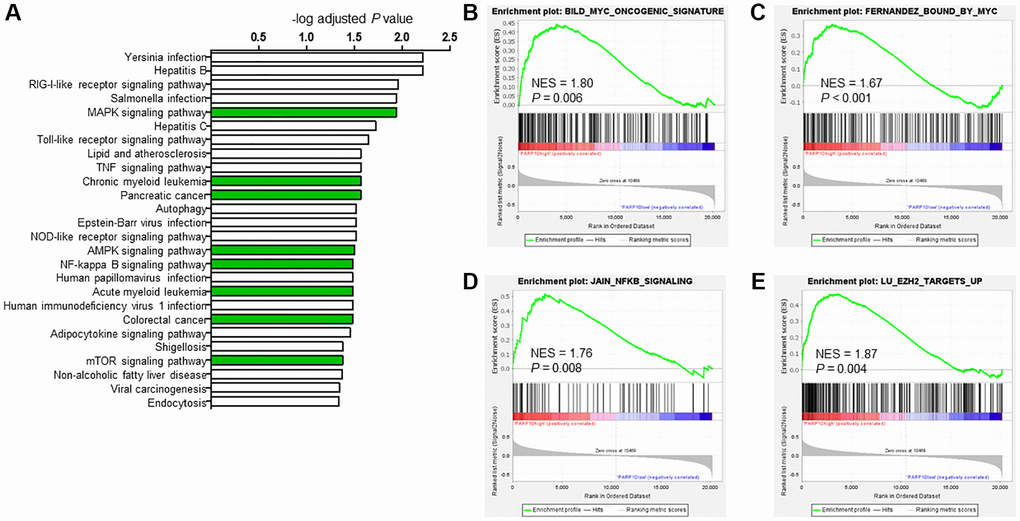
Figure 5. Potential biological functions of PARP10 in AML. (A) KEGG analysis of PARP10 positively co-expressed genes (r > 0.5, P < 0.05). Significantly enriched pathways (adjust P < 0.05) were plotted. And (B–E) GSEA analysis of AML patients based on PARP10 expression. NES: normalized enrichment score.
We next performed gene set enrichment analysis (GSEA) to further explore the involved biological pathways and cofactors of PARP10 in AML. Median expression of PARP10 was used as threshold to define PARP10 high or low group in GSE15061 dataset. For the PARP10 high expression group, the gene sets were significantly enriched in the MYC oncogenic signature (NES = 1.8; P = 0.006), MYC binding (NES = 1.67; P = 0.000), NFKB signaling (NES = 1.76; P = 0.008), and EZH2 targets (NES = 1.87; P = 0.004) (Figure 5B–5E). STRING protein–protein interaction network analysis also demonstrated interaction of PARP10 and MYC (Supplementary Figure 5). As MYC, NFKB, and EZH2 are key transcriptional factors and epigenetic regulators in tumorigenesis [27–29], our data suggest that PARP10 could be involved in transcription and epigenomic regulation in AML.
PARP10 knockout impaired AML cell proliferation in vitro
To further gain insight into the function of PARP10 in AML proliferation, we performed PARP10 knockout in the MOLM13 cell line using CRISPR/Cas-mediated gene editing. Genotyping of the genomic DNA regions that encompass targeting sites by two guide RNAs [24] successfully validated the random insertions and/or deletions introduced by CRISPR/Cas9-mediated PARP10 editing in MOLM13 cells (Figure 6A and Supplementary Table 4). In growth competition assays, which tracked the percentage of sgPARP10–RFP cells in MOLM13-Cas9 cells relative to the non-transduced, the RFP negative cells at days 0, 1, 3, 5, and 7 indicated that the expression of sgPARP10 reduced the growth of MOLM13-Cas9 cells compared to the sgEV control cells (Figure 6B). We also established proliferation curves for the control and PARP10 knockout cells and found PARP10 knockout, by guide RNA1, impaired the proliferation of MOLM13 cells, although it’s not significant at day 8 (P = 0.059), whereas PARP10 knockout by guide RNA2 significantly impaired the proliferation of MOLM13 cells (P < 0.05) (Figure 6C and 6D). Thus, our bioinformatic analysis and functional experiments highlighted the important role of PARP10 in AML cell proliferation, indicating that targeting PARP10 might provide a novel strategy for treating AML.
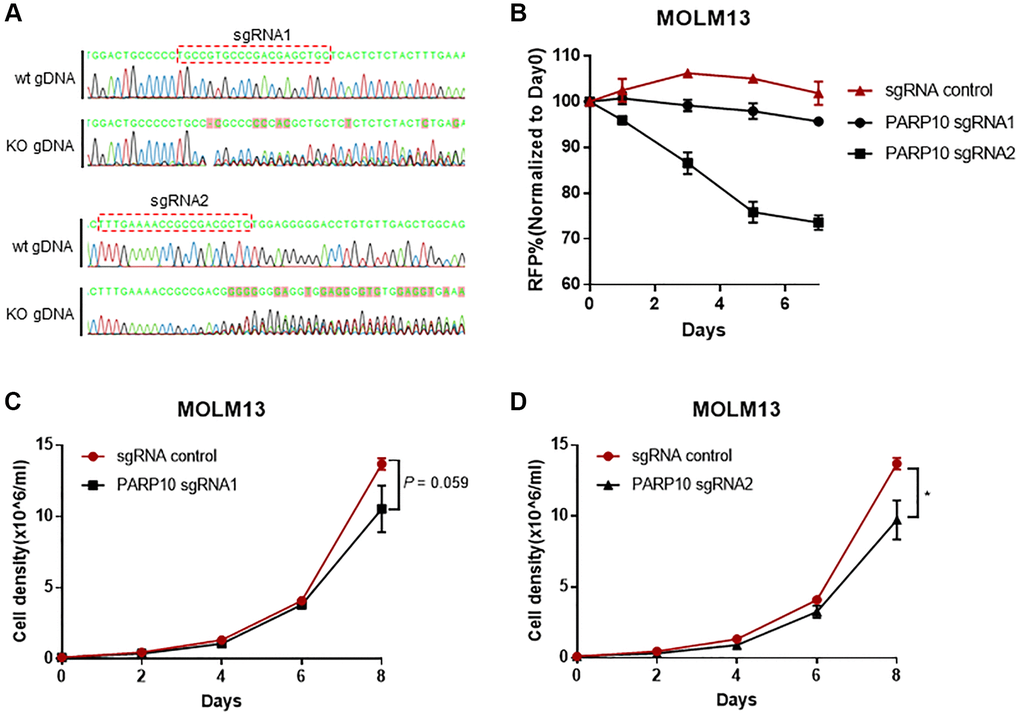
Figure 6. Loss of PARP10 impaired proliferation of AML cells. (A) Sanger sequencing showing random insertions and/or deletions introduced by CRISPR/Cas9-mediated PARP10 editing in MOLM13 cells. (B) Flow cytometry-based RFP competition assay showing PARP10 knockout by two sgRNAs impaired cell proliferation in MOLM13 cells. Proliferation curves of PARP10 knockout cells introduced by sgRNA1 (C) and sgRNA2 (D) versus control MOLM13 cells, the error bars represent the SD from triplicates. The asterisk indicates significant statistical differences between the PARP10 knockout and control cells.
Discussion
The prognosis of AML is poor due to its highly heterogeneous blast; thus, novel diagnostic and prognosis biomarkers are needed to better define the disease and improve stratified therapy [1, 3, 6]. The PARP family of proteins is composed of 17 members, which are responsible for adenosine diphosphate (ADP) ribosylation in the cells, while the PARP family has emerged as important regulatory factors in both DNA and cancer biology [8, 30]. PARP1/2 inhibitors were first approved for the treatment of breast and ovarian cancers [8, 13], and subsequently, many in vitro and in vivo studies have been conducted to investigate their efficacy in other tumor types, although without any satisfactory progress, which is partially due to mutations that affect the DDR-associated genes, which are not common in other malignancies, including AML [9, 12, 13, 15]. There is the possibility that AML survival is independent of PARP1/2, whereas the other PARP family proteins are essential in AML cells; thus, targeting the other PARP family proteins may provide promising cytotoxic activities against leukemia in the clinic. In this study, we comprehensively evaluated the differential expression pattern of PARP family genes across AML patients and healthy donors and analyzed the association between PARP10 expression and clinical parameters as well as AML prognosis.
Through the integrated analyses of three large datasets encompassing gene expression data on AML samples and healthy controls (i.e., TCGA versus GTEX, GSE15061, and Bloodspot) [16–18], we found that the expressions of PARP6 and PARP10 were simultaneously higher in AML cells than in normal cells, while the differential expression patterns of the other genes showed discrepancies across the three datasets, which indicated that targeting PARP6 and PARP10 might offer the more plausible option than targeting PARP1/2 in precision medicine. To better understand the correlation between gene expression and the prognosis of AML, we performed survival analysis using the TCGA LAML dataset and found patients with higher expressions of PARP10 correlated with inferior OS and EFS compared to patients with lower expressions of PARP10, whereas PARP6 expression had no influence on the AML prognosis. Future in vitro and in vivo studies are warranted to dissect the role of PARP10 on leukemia initiation and disease progression.
Survival analysis of the differentially expressed genes (i.e., PARP6 and PARP10) suggested that PARP10 was the only gene whose high expression was associated with inferior clinical outcomes. The Chi-square test of the PARP10 expression group (i.e., high vs. low) and clinical characteristics demonstrated that the high expression of PARP10 was associated with older age, more frequent TP53 mutations, and higher risk classifications, all of which exhibited features of high AML risk and were consistent with the result of predicting inferior clinical outcomes. Intriguingly, multivariate Cox regression analysis identified PARP10 as an independent factor in AML prognosis for both OS and EFS after adjusting for age (<60 vs. >60), WBC count (≥30 vs. <30 × 109/L), FLT3 and TP53 statuses (wildtype vs. mutant), and transplant status (yes vs. no). All these results demonstrate that PARP10 could be used as a potential biomarker, which might contribute to the precise prognosis and stratification of AML. More importantly, we found patients with higher expressions of PARP10 would benefit from allo-SCT therapy, while patients with lower expressions of PARP10 should receive allo-SCT due to the detection of an inferior EFS after this therapy according to our study. Therefore, it is important for clinical physicians or researchers to perform rt-qPCR or high-throughput RNA sequencing to quantify the expression value of PARP10 in clinical management, based on which AML patients could be stratified into different risk group to receive comparable therapies. Furthermore, multicenter studies are warranted to validate these findings and to better stratify future AML therapies, and after that it should be considered that whether PARP10 expression could be included into the novel ELN 2022 AML prognostic recommendations.
Previous studies demonstrated that PARP10 functions as an oncogene in Hela cells [24], while acting as a tumor suppressor in hepatocellular carcinoma cells [31], indicating that it has a dual role in carcinogenesis, in a context-dependent manner. Herein, we analyzed the PARP10 co-expression network in AML for the first time, and our results suggest that PARP10 may work together with other signaling pathways to exert an oncogenic effect in AML, while it also provided hints regarding its underlying molecular mechanism in oncogenesis. Moreover, GSEA analysis indicated that high PARP10 expression was involved in MYC, NFKB, and EZH2 target gene sets in AML patients, providing a potential and interesting direction for further exploration of its biological functions. Intriguingly, PARP10 knockout significantly impaired the proliferation of AML cells in vitro, potentiating that targeting PARP10 might provide a novel strategy for the treatment of AML. Clinical trials evaluating PARP10 inhibition alone or in combination with other drugs in the treatment of AML are warranted to conduct in the future.
However, there are some limitations of the current study. The major limitation of public databases is the batch effect derived from high-throughput sequencing of large number of human samples. Bioinformatics researchers have been dedicated to developing novel algorithms to adjust batch effect, which is always not satisfactory enough when applied to human samples with such great heterogeneity. Another limitation of public databases is that the survival data is taken from clinical cohort decades ago, which might not be able to represent the current status as treatment for AML has been refined and long-term prognosis in patients with AML has gradually improved in recent decades. Thus, direct experiment in the lab (e.g., rt-qPCR) to compare the differential expression of PARP10 in AML and normal cells, and association analysis of PARP10 expression with clinical prognosis in the current treatment cohort are warranted to perform in the future.
In conclusion, our study revealed that PARP10 was aberrantly expressed in AML patients, and its high expression was associated with high-risk factors and poor prognosis and could be an independent poor survival factor in AML patients. Importantly, our results suggested that patients with high expression of PARP10 would benefit from allo-SCT therapy, while those with low expression of PARP10 should not receive allo-SCT due to an inferior EFS after allo-SCT use.
Author Contributions
Dandan Hu and Ling Wang initiated and designed this study. Ling Wang and Chuang Jiang collected and analyzed the data. Chuang Jiang performed the experiments. Ling Wang and Chuang Jiang wrote the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
This study was approved by the Institutional Review Committee of Guangzhou Women and Children's Medical Center affiliated to Guangzhou Medical University, and experiments were conducted based on the approved guidelines.
Funding
This work was supported by Science and Technology Key Project for People’s Livelihood of Guangzhou, China (No. 202206010060).
References
- 1. Shallis RM, Wang R, Davidoff A, Ma X, Zeidan AM. Epidemiology of acute myeloid leukemia: Recent progress and enduring challenges. Blood Rev. 2019; 36:70–87. https://doi.org/10.1016/j.blre.2019.04.005 [PubMed]
- 2. Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T, Dombret H, Ebert BL, Fenaux P, Larson RA, Levine RL, Lo-Coco F, Naoe T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017; 129:424–47. https://doi.org/10.1182/blood-2016-08-733196 [PubMed]
- 3. Döhner H, Weisdorf DJ, Bloomfield CD. Acute Myeloid Leukemia. N Engl J Med. 2015; 373:1136–52. https://doi.org/10.1056/NEJMra1406184 [PubMed]
- 4. Jongen-Lavrencic M, Grob T, Hanekamp D, Kavelaars FG, Al Hinai A, Zeilemaker A, Erpelinck-Verschueren CAJ, Gradowska PL, Meijer R, Cloos J, Biemond BJ, Graux C, van Marwijk Kooy M, et al. Molecular Minimal Residual Disease in Acute Myeloid Leukemia. N Engl J Med. 2018; 378:1189–99. https://doi.org/10.1056/NEJMoa1716863 [PubMed]
- 5. Narayanan D, Weinberg OK. How I investigate acute myeloid leukemia. Int J Lab Hematol. 2020; 42:3–15. https://doi.org/10.1111/ijlh.13135 [PubMed]
- 6. Kayser S, Levis MJ. Advances in targeted therapy for acute myeloid leukaemia. Br J Haematol. 2018; 180:484–500. https://doi.org/10.1111/bjh.15032 [PubMed]
- 7. Taussig DC, Pearce DJ, Simpson C, Rohatiner AZ, Lister TA, Kelly G, Luongo JL, Danet-Desnoyers GA, Bonnet D. Hematopoietic stem cells express multiple myeloid markers: implications for the origin and targeted therapy of acute myeloid leukemia. Blood. 2005; 106:4086–92. https://doi.org/10.1182/blood-2005-03-1072 [PubMed]
- 8. Jagtap P, Szabó C. Poly(ADP-ribose) polymerase and the therapeutic effects of its inhibitors. Nat Rev Drug Discov. 2005; 4:421–40. https://doi.org/10.1038/nrd1718 [PubMed]
- 9. Curtin NJ, Szabo C. Poly(ADP-ribose) polymerase inhibition: past, present and future. Nat Rev Drug Discov. 2020; 19:711–36. https://doi.org/10.1038/s41573-020-0076-6 [PubMed]
- 10. Herceg Z, Wang ZQ. Functions of poly(ADP-ribose) polymerase (PARP) in DNA repair, genomic integrity and cell death. Mutat Res. 2001; 477:97–110. https://doi.org/10.1016/s0027-5107(01)00111-7 [PubMed]
- 11. Javle M, Curtin NJ. The role of PARP in DNA repair and its therapeutic exploitation. Br J Cancer. 2011; 105:1114–22. https://doi.org/10.1038/bjc.2011.382 [PubMed]
- 12. Dziadkowiec KN, Gąsiorowska E, Nowak-Markwitz E, Jankowska A. PARP inhibitors: review of mechanisms of action and BRCA1/2 mutation targeting. Prz Menopauzalny. 2016; 15:215–9. https://doi.org/10.5114/pm.2016.65667 [PubMed]
- 13. Lee JM, Ledermann JA, Kohn EC. PARP Inhibitors for BRCA1/2 mutation-associated and BRCA-like malignancies. Ann Oncol. 2014; 25:32–40. https://doi.org/10.1093/annonc/mdt384 [PubMed]
- 14. Molenaar RJ, Radivoyevitch T, Nagata Y, Khurshed M, Przychodzen B, Makishima H, Xu M, Bleeker FE, Wilmink JW, Carraway HE, Mukherjee S, Sekeres MA, van Noorden CJF, Maciejewski JP. IDH1/2 Mutations Sensitize Acute Myeloid Leukemia to PARP Inhibition and This Is Reversed by IDH1/2-Mutant Inhibitors. Clin Cancer Res. 2018; 24:1705–15. https://doi.org/10.1158/1078-0432.CCR-17-2796 [PubMed]
- 15. Esposito MT, Zhao L, Fung TK, Rane JK, Wilson A, Martin N, Gil J, Leung AY, Ashworth A, So CW. Synthetic lethal targeting of oncogenic transcription factors in acute leukemia by PARP inhibitors. Nat Med. 2015; 21:1481–90. https://doi.org/10.1038/nm.3993 [PubMed]
- 16. Goldman MJ, Craft B, Hastie M, Repečka K, McDade F, Kamath A, Banerjee A, Luo Y, Rogers D, Brooks AN, Zhu J, Haussler D. Visualizing and interpreting cancer genomics data via the Xena platform. Nat Biotechnol. 2020; 38:675–8. https://doi.org/10.1038/s41587-020-0546-8 [PubMed]
- 17. Mills KI, Kohlmann A, Williams PM, Wieczorek L, Liu WM, Li R, Wei W, Bowen DT, Loeffler H, Hernandez JM, Hofmann WK, Haferlach T. Microarray-based classifiers and prognosis models identify subgroups with distinct clinical outcomes and high risk of AML transformation of myelodysplastic syndrome. Blood. 2009; 114:1063–72. https://doi.org/10.1182/blood-2008-10-187203 [PubMed]
- 18. Bagger FO, Sasivarevic D, Sohi SH, Laursen LG, Pundhir S, Sønderby CK, Winther O, Rapin N, Porse BT. BloodSpot: a database of gene expression profiles and transcriptional programs for healthy and malignant haematopoiesis. Nucleic Acids Res. 2016; 44:D917–24. https://doi.org/10.1093/nar/gkv1101 [PubMed]
- 19. Rapin N, Bagger FO, Jendholm J, Mora-Jensen H, Krogh A, Kohlmann A, Thiede C, Borregaard N, Bullinger L, Winther O, Theilgaard-Mönch K, Porse BT. Comparing cancer vs normal gene expression profiles identifies new disease entities and common transcriptional programs in AML patients. Blood. 2014; 123:894–904. https://doi.org/10.1182/blood-2013-02-485771 [PubMed]
- 20. Haferlach T, Kohlmann A, Wieczorek L, Basso G, Kronnie GT, Béné MC, De Vos J, Hernández JM, Hofmann WK, Mills KI, Gilkes A, Chiaretti S, Shurtleff SA, et al. Clinical utility of microarray-based gene expression profiling in the diagnosis and subclassification of leukemia: report from the International Microarray Innovations in Leukemia Study Group. J Clin Oncol. 2010; 28:2529–37. https://doi.org/10.1200/JCO.2009.23.4732 [PubMed]
- 21. Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, Antipin Y, Reva B, Goldberg AP, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012; 2:401–4. https://doi.org/10.1158/2159-8290.CD-12-0095 [PubMed]
- 22. Kuleshov MV, Jones MR, Rouillard AD, Fernandez NF, Duan Q, Wang Z, Koplev S, Jenkins SL, Jagodnik KM, Lachmann A, McDermott MG, Monteiro CD, Gundersen GW, Ma'ayan A. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016; 44:W90–7. https://doi.org/10.1093/nar/gkw377 [PubMed]
- 23. Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A. 2005; 102:15545–50. https://doi.org/10.1073/pnas.0506580102 [PubMed]
- 24. Schleicher EM, Galvan AM, Imamura-Kawasawa Y, Moldovan GL, Nicolae CM. PARP10 promotes cellular proliferation and tumorigenesis by alleviating replication stress. Nucleic Acids Res. 2018; 46:8908–16. https://doi.org/10.1093/nar/gky658 [PubMed]
- 25. Cornelissen JJ, Blaise D. Hematopoietic stem cell transplantation for patients with AML in first complete remission. Blood. 2016; 127:62–70. https://doi.org/10.1182/blood-2015-07-604546 [PubMed]
- 26. Koreth J, Schlenk R, Kopecky KJ, Honda S, Sierra J, Djulbegovic BJ, Wadleigh M, DeAngelo DJ, Stone RM, Sakamaki H, Appelbaum FR, Döhner H, Antin JH, et al. Allogeneic stem cell transplantation for acute myeloid leukemia in first complete remission: systematic review and meta-analysis of prospective clinical trials. JAMA. 2009; 301:2349–61. https://doi.org/10.1001/jama.2009.813 [PubMed]
- 27. Dang CV. MYC on the path to cancer. Cell. 2012; 149:22–35. https://doi.org/10.1016/j.cell.2012.03.003 [PubMed]
- 28. Dolcet X, Llobet D, Pallares J, Matias-Guiu X. NF-kB in development and progression of human cancer. Virchows Arch. 2005; 446:475–82. https://doi.org/10.1007/s00428-005-1264-9 [PubMed]
- 29. Kim KH, Roberts CW. Targeting EZH2 in cancer. Nat Med. 2016; 22:128–34. https://doi.org/10.1038/nm.4036 [PubMed]
- 30. Riffell JL, Lord CJ, Ashworth A. Tankyrase-targeted therapeutics: expanding opportunities in the PARP family. Nat Rev Drug Discov. 2012; 11:923–36. https://doi.org/10.1038/nrd3868 [PubMed]
- 31. Zhao Y, Hu X, Wei L, Song D, Wang J, You L, Saiyin H, Li Z, Yu W, Yu L, Ding J, Wu J. PARP10 suppresses tumor metastasis through regulation of Aurora A activity. Oncogene. 2018; 37:2921–35. https://doi.org/10.1038/s41388-018-0168-5 [PubMed]



