Introduction
Acute promyelocytic leukemia (APL), which is caused by a balanced translocation t(15;17)(q22;q12) that results in the fusion transcript PML-RARA, is a rare type of acute myeloid leukemia (AML) that accounts for 10–15% of newly diagnosed cases [1]. It is induced by aberrant promyelocytes in the bone marrow proliferating malignantly and failing to differentiate, preventing normal white blood cell generation [2]. Clinical manifestations of APL mainly include anemia, hemorrhage, infection, hepatosplenomegaly, lymphadenopathy, and bone pain [3, 4]. Among them, bleeding tendency and thrombotic events are the most common causes of mortality in APL patients [5–8]. APL is a kind of leukemia with a dangerous prognosis in the early phase [9]. With all-trans retinoic acid (ATRA) and arsenic trioxide (ATO) development, APL has progressed from a fatal illness to one of the most treatable cancers [10].
In 1973, anthracycline monotherapy was successfully utilized in APL for the first time [11]. In the mid-1980s, ATRA was utilized in patients with APL, and it had a high response rate [12, 13]. ATO is the main effective component of the traditional Chinese medicine arsenic. Chinese scholars first used it in the treatment of APL and achieved a remarkable curative effect. At present, ATO is considered to be one of the most successful medications for the treatment of APL. However, treatment refractoriness and side effects have been noted in the clinical environment. Anthracyclines can cause severe cardiotoxicity, and ATRA syndrome (RAS) is the most serious complication of ATRA induction in the treatment of APL [14]. Moreover, prolonged ATO exposure can easily cause arsenic poisoning, including skin lesions, neurological effects, diabetes and bladder cancer [15]. Furthermore, patients receiving ATO must be hospitalized for intravenous injection, which is inconvenient and not cost-effective. Consequently, an oral drug with comparable efficacy, fewer side effects, and improved patient compliance should be developed in order to offer patients an improved quality of life as well as consolidation and maintenance therapy.
Realgar, a traditional Chinese medicine (TCM), has been applied for more than 1800 years by traditional Chinese doctors [16]. Similar to ATO, realgar is also a kind of mined ore that contains arsenic. Currently, it is widely accepted that the benefit and risk of arsenic are solely dependent on the chemical forms of arsenic rather than the arsenic content alone [17]. Realgar’s monoclinic crystal structure is cradle-type with an octatomic ring spatially and includes approximately 90% As4S4, of which the crystalline form is α-As4S4. Previous studies have shown that realgar is less toxic than ATO [18, 19]. Since 1963, it has been included in the Chinese Pharmacopoeia (ChP). Employed either externally or orally, it is effective in treating a variety of exterior and interior disorders [20, 21]. In recent years, realgar has been shown to have anticancer properties [22–27]. Similar clinical observations also support realgar as a drug for various types of hematological malignancies [28]. In China, realgar has been utilized as a single treatment for APL [29]. Chen et al. revealed that realgar can induce apoptosis and differentiation in APL cells [30]. In a clinical study including 129 APL patients, the results showed that oral realgar treatment can achieve high rates of complete remission, tolerable side effects and long-term disease-free survival [31]. In addition, realgar, which can be administered orally, is preferable to ATO, which can only be administered by injection. Because oral administration is more conducive to consolidating and maintaining therapy, which can improve the compliance and quality of life of APL patients, realgar is a promising candidate worthy of further exploration for the treatment of APL.
Despite the fact that realgar has been used to treat APL in China for many years, its molecular mechanism remains unclear. The purpose of this study was to investigate the effect of realgar on the death of the APL cell line NB4 and to clarify the underlying mechanism. In this study, APL cells were treated with different concentrations of realgar, and cell viability, apoptosis, morphological changes, ATP levels and cell cycle arrest were measured, and we evaluated, for the first time, the expression of Bcl-2, Bax, Cyt-C and AIF at the mRNA and protein levels following realgar treatment. Our study is helpful for understanding the potential mechanism of realgar in the treatment of APL.
Materials and Methods
Realgar and preparation of realgar solutions
Realgar containing arsenic sulfide (As4S4) with a purity of 93.4% was obtained from Hubei Pharmaceutical Co., Ltd. (Wuhan, China). In the current investigation, 3 g of realgar was sieved through a 500 mesh sieve, added to distilled water to a volume of 300 mL, and mixed for 30 minutes using a magnetic stirrer. It was then put into an M-110EH high-pressure microjet granulator (ATSE Nanotechnology Ltd., Suzhou, China) for 10 cycles at 30,000 rpm, followed by 30 minutes of ultrasonic vibration. After grinding, the realgar nanoparticles were spherical and uniform in size, and the average diameter measured by a Malvern Zetasizer Nano-ZS (Malvern Instruments, Britain) was approximately 72.63 nm. The details of realgar nanoparticles Dynamic Light Scattering (DLS) and particle size are shown in Supplementary Figure 1. After degassing using a 0.22 μm micropore screen, the concentration of total arsenic was determined by inductively coupled plasma-atomic emission spectrometry (ICP–AES). Finally, stock solutions of realgar (1000 μg/mL) were prepared in RPMI 1640 medium, and stored airtight and light-shielded at 4°C.
Cells and cell culture
The APL cell line (NB4, obtained from ATCC) was cultured in RPMI-1640 media (Solarbio, Beijing, China) supplemented with 10% fetal bovine serum (HyClone, Logan, UT, USA), 100 μg/mL streptomycin (Sigma, St. Louis, MO, USA) and 100 U/mL penicillin (Sigma, St. Louis, MO, USA). APL cells were cultured at 37°C in an incubator with 5% CO2 and 95% air. Cell numbers were determined by an automated cell counter (Muse; Merck Millipore, Billerica, MA, USA). All experiments were performed on an ultraclean bench.
Cell counting kit-8 (CCK-8) assay
APL cells in the logarithmic growth phase were cultured in 96-well plates (Thermo Fisher Scientific Ltd., China). In brief, 100 μL of the cell solutions (cell density of 8.0 × 104/mL) were added to each well of the 96-well plate. Next, realgar solutions at concentrations of 0, 12.5, 25.0, 50.0 and 75.0 μg/mL (the concentration of arsenic contained corresponds to 0.3, 0.6, 1.2 and 1.5 μg/mL, respectively) were added to each well. Then, the cells were cultivated for 24, 48, and 72 hours in an incubator at 37°C with 5% CO2 and water saturation humidity. Finally, each well’s optical density (OD) at 450 nm was determined using a microplate reader (BioTek Instruments, Winooski, VT, USA). The experiment was carried out three times independently. Furthermore, 50% inhibitory concentration (IC50) was the concentration of realgar required to inhibit the growth of APL cells by 50%.
Cell morphology
NB4 cells were seeded in 6-well plates at a density of 1.2 × 105 cells/well and then exposed to different concentrations (0, 12.5, 25.0, 50.0 and 75.0 μg/mL) of realgar (final volume: 3 mL) for 24 h. At the indicated times, morphological alterations were observed using a light field microscope (IX71, Olympus, Japan).
Apoptosis/necrosis analysis
NB4 cells were plated at a density of 1.2 × 105 cells/well in 6-well plates, and then the cells were exposed to various concentrations (i.e., 0, 12.5, 25.0, 50.0, 75.0 μg/mL) of realgar (final volume: 3 mL) for 24 h. Next, the cells were collected at the specified time, gently resuspended by adding 195 μL Annexin V-FITC Binding Buffer (Beyotime, China), incubated with 5 μL Annexin V-FITC and 10 μL PI staining solution for 20 minutes at room temperature (20−25°C) and protected from light, and placed on ice for detection by flow cytometry (BD FACSVerse, USA). Annexin V-positive and PI-negative cells indicated the early stages of apoptosis, whereas both Annexin V- and PI-positive cells indicated the late stages of apoptosis.
Cell cycle analysis
Cell cycle arrest analysis was carried out by flow cytometry after APL cells were exposed to different concentrations (0, 12.5, 25.0, 50.0 and 75.0 μg/mL) of realgar and stained with the DNA-specific dye ethidium bromide. In short, 1.2 × 105 cells were seeded in a six-well tissue culture plate, and various concentrations (0, 12.5, 25.0, 50.0 and 75.0 μg/mL) of realgar were added and incubated for an additional 24 h. Next, the cells were collected, washed with 1× PBS, and centrifuged. Then, the obtained cells were rewashed in PBS, fixed in 70% ice-cold ethanol and kept at −20°C. Finally, the cells were rewashed with PBS and stained with ethidium bromide, followed by analysis by flow cytometry (BD FACSVerse, USA).
Determination of ATP level
To investigate the effect of realgar on the production of ATP, the ATP content was evaluated using an ATP assay kit (Solarbio, BC0300, China). Briefly, APL cells were cultured with various concentrations of realgar for 24 hours (i.e., 0, 12.5, 25.0, 50.0, 75.0 μg/mL), and then ATP-releasing reagent was added and ultrasonicated on ice for 10 minutes, followed by centrifugation at 12,000 g at 4°C for 10 minutes to collect the supernatants. Finally, a MultiMode Microplate Reader (BioTek Instruments, Winooski, VT, USA) was used to determine the results.
Immunofluorescence analysis
NB4 cells were treated with realgar under optimal conditions (75.0 μg/mL, 72 h) as determined by the CCK-8 assay. Following treatment, the cells were spread onto slides, fixed with 4% paraformaldehyde for 20 minutes at 4°C, and permeabilized in 0.1% Triton for 10 minutes at room temperature. Then, the cells were incubated with 10% goat serum (Beyotime Institute of Biotechnology) for 30 minutes at room temperature before the primary antibody reaction. After that, the slides were treated for 8 hours at 4°C with rabbit anti-human polyclonal antibody against RARa (1:200; Santa Cruz Biotechnology, Inc.), followed by FITC-conjugated goat anti-rabbit IgG (1:200, Zhongshan Goldenbridge Biotechnology Co., Ltd.) at room temperature for 1 h. Next, microscopy was used to observe the cells after counterstaining with DAPI (1 μg/mL, 5 min). A fluorescence microscope (×400; Nikon Corp, Tokyo, Japan) and a confocal laser scanning microscope (×400; Leica Microsystems GmbH, Wetzlar, Germany) were used to observe the cells.
Quantitative reverse transcription PCR (qPCR)
We used qPCR to investigate the expression of Bcl-2, Bax, Cytochrome C (Cyt-C), and apoptosis-inducing factor (AIF) mRNA levels in cells treated with different concentrations of realgar. In brief, APL cells (1.2 × 105/well) were seeded in six-well plates and treated for 24, 48 and 72 h with various concentrations (0, 12.5, 25.0, 50.0, and 75.0 μg/mL) of realgar solution. Then, the cells were collected, and total RNA was extracted by an RNA Tissue/Cell Rapid Extraction Kit (Shandong Sparkjade Science Co., Ltd., China). Total RNA was quantified by a microspectrophotometer, followed by generation of single-stranded cDNA from 600 ng total RNA. Furthermore, qPCR was performed in order to measure the gene level using cDNA synthesized by reverse transcription. Table 1 lists the target-specific primers, and the PCR program was configured as follows: 94°C for 3 min, then 94°C for 10 s, then 40 cycles of 94°C for 5 s, and 60°C for 34 s. After normalization to the relevant endogenous GAPDH control, the fold changes were determined using the 2−ΔΔCt technique.
Table 1. Primer sequences for the target genes determined by quantitative PCR.
| ID | Primer | Sequences (5′ to 3′) |
| AIF | Forward | GTG ATT TGG GCC CCG ATG TT |
| Reverse | GGG GTG CTG GGA GGA ATA GT | |
| Cyt-C | Forward | TCC GGC TGG TAG TAG TTC CG |
| Reverse | TTC CGC CAT GGT GCT GAA TC | |
| Bax | Forward | CGG GTT GTC GCC CTT TTC TA |
| Reverse | GCT CCC GGA GGA AGT CCA AT | |
| Bcl-2 | Forward | TGA GTT CGG TGG GGT CAT GT |
| Reverse | TCA GTC ATC CAC AGG GCG AT | |
| GAPDH | Forward | TGC ACC ACC AAC TGC TTA GC |
| Reverse | GGC ATG GAC TGT GGT CAT GAG |
Western blotting
Total proteins were extracted from the realgar-treated NB4 cells, and the protein concentration was measured using a BCA Test Kit (Beyotime, China). The extracted proteins were denatured with reducing sample buffer at 100°C for 7 min. Then, 40 μg of protein was loaded onto a 10% polyacrylamide gel. Proteins were separated (30 min at 80 V, then 120 V until completed) with Tris-HCl running buffer in an electrophoresis apparatus (BioRad, Hercules, CA, USA). Then, the proteins were transferred (90 min, 100 V) from the acrylamide gel to a polyvinylidene fluoride (PVDF) membrane. Blots were blocked with 5% bovine serum albumin (BSA) for 1 h, followed by incubation with monoclonal anti-Bax (1:1000, ab32503, Abcam, USA), Bcl-2 (1:1000, ab32124, Abcam, USA), AIF (1:1000, ab32516, Abcam, USA), and Cyt-C (1:5000, ab133504, Abcam, USA) antibodies at 4°C overnight. Subsequently, the membranes were washed frequently with Tris-buffered saline Tween (TBS-T; 5 min, 3 times), followed by incubation with goat anti-rabbit IgG antibody (1:20,000) at room temperature for 1 h. After repeated washings with TBS-T, the blots were treated with a chemiluminescent detection kit (Thermo Fisher Scientific, China). The internal reference was represented by an antibody against actin. The quality of the protein was assessed using a Typhoon PhosphorImager (GE Healthcare, Piscataway, NJ, USA).
Statistical analysis
All experiments were performed at least three times independently, and the data are expressed as the mean ± the standard deviation. For the significance test, one-way analyses of variance and post hoc methods depending on Newman–Keuls tests were applied. P < 0.05 was considered to be significant. SPSS 25.0 was used to analyze the data.
Results
Realgar inhibits NB4 cell proliferation
The dose- and time-dependent responses of NB4 cells to realgar are shown in Figure 1. When the concentration of realgar was increased, the survival rate of NB4 cells was reduced in a time-dependent manner. The IC50 values of realgar in NB4 cells were 48.0, 27.0, and 14.0 μg/mL after 24, 48 and 72 h, respectively.
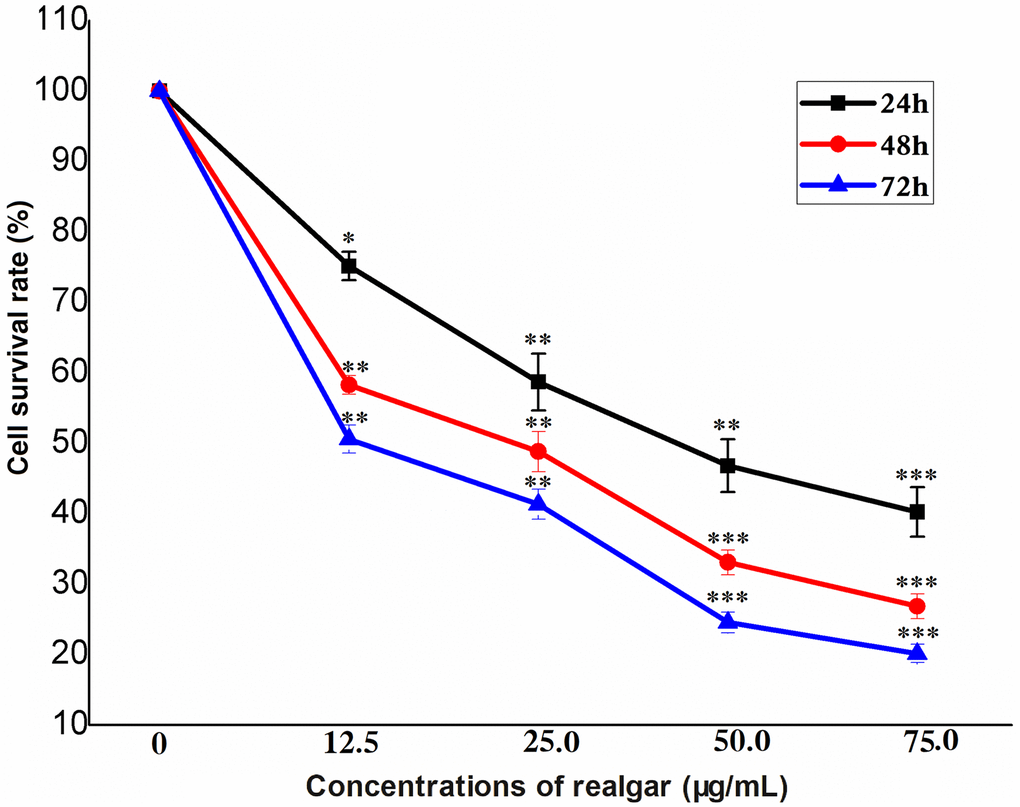
Figure 1. Cell survival after NB4 cells were exposed to different concentrations of realgar. Cell survival was evaluated using a CCK-8 assay after treatment with different concentrations of realgar for 24 h, 48 h or 72 h. N = 3.
Change in cell morphology
As illustrated in (Figure 2A–2E), we found that the number of intact NB4 cells decreased, while the number of damaged cells increased, including cell contraction, nuclear condensation, and debris. The higher the concentration of realgar, the more significant the alteration of cell morphology. According to these results, realgar has a significant cytotoxic impact on NB4 cells.
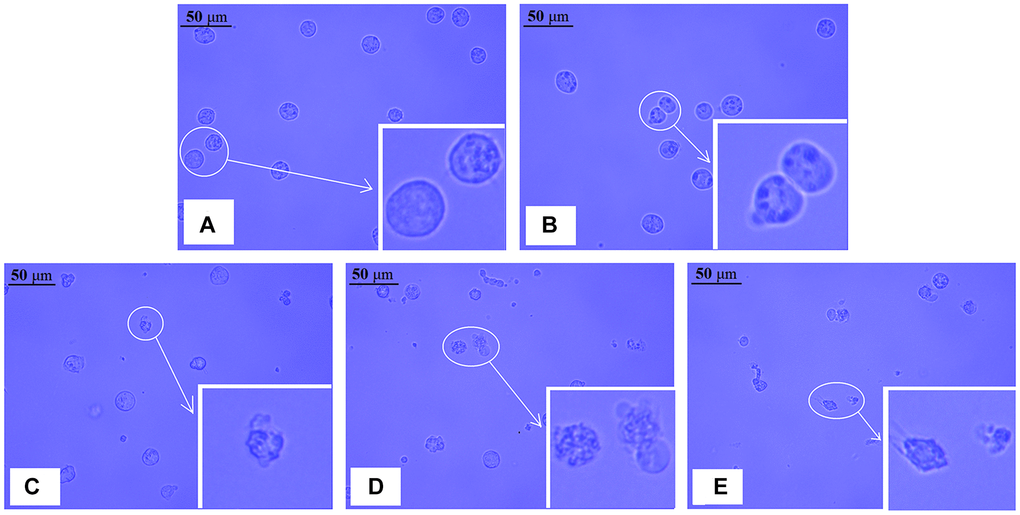
Figure 2. Morphological changes in NB4 cells after exposure to different concentrations of realgar for 24 h. The cells were treated with different concentrations of realgar for 24 h, and typical images were captured by a light field microscope. (A) Control cells; (B) cells exposed to 12.5 μg/ml of realgar; (C) cells exposed to 25.0 μg/ml of realgar; (D) cells exposed to 50.0 μg/ml of realgar; and (E) cells exposed to 75.0 μg/ml of realgar. Inset, magnified cells.
Annexin V-FITC/PI staining analysis
Annexin-V-FITC and PI double staining was used to distinguish between live cells, early apoptotic cells, and late apoptotic/necrotic cells after realgar treatment of NB4 cells (Figure 3A). We observed that the amount of early apoptosis and late apoptosis/necrosis in NB4 cells was noticeably increased in a concentration-dependent manner after being cultured with various concentrations of realgar for 24 hours (Figure 3B and 3C). The results indicate that apoptosis can be easily induced in NB4 cells by realgar treatment.
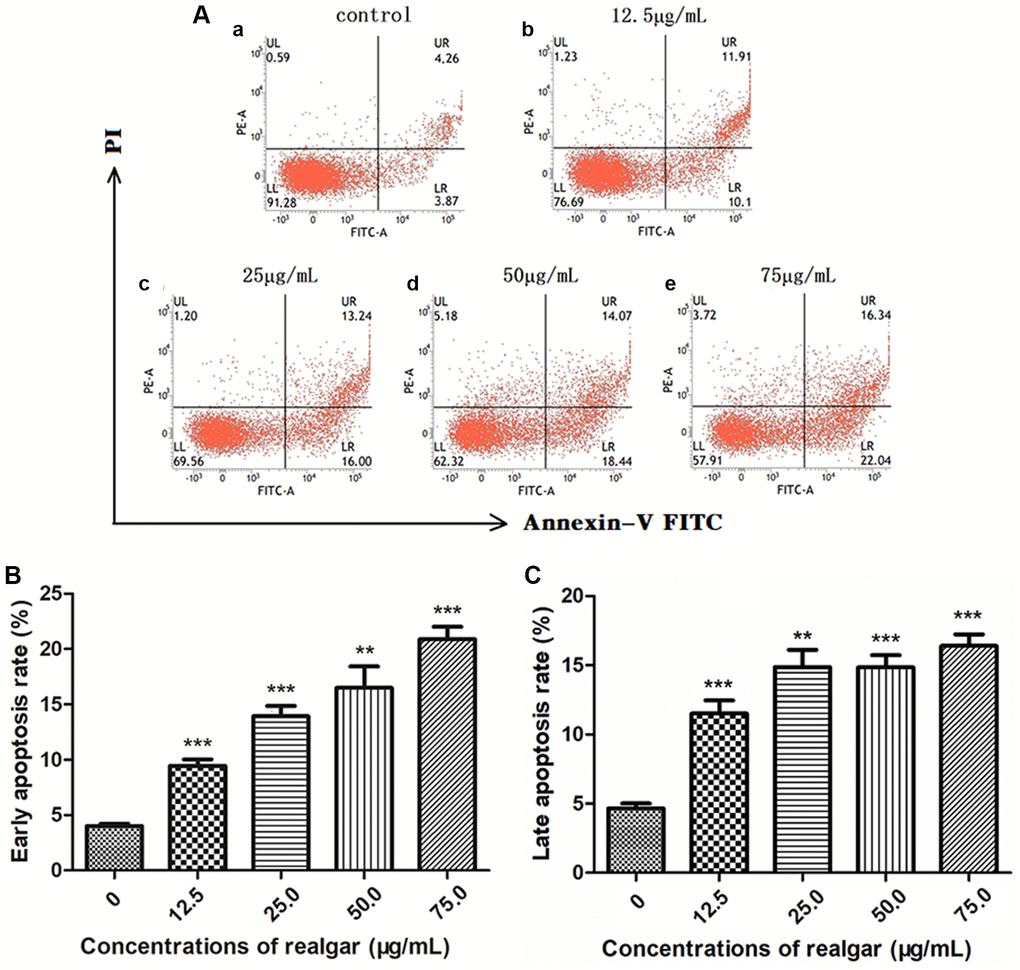
Figure 3. Analysis of apoptosis/necrosis of NB4 cells after exposure to different concentrations of realgar for 24 h. The cells were treated with different concentrations of realgar (0, 12.5, 25.0, 50.0 and 75.0 μg/ml) for 24 h, followed by staining with the Annexin V-FITC/PI apoptosis staining kit. Apoptosis/necrosis analysis was performed by flow cytometry. (A) (a) Untreated cells; (b) cells treated with 12.5 μg/ml of realgar; (c) cells treated with 25.0 μg/ml of realgar; (d) cells treated with 50.0 μg/ml of realgar; and (e) cells treated with 75.0 μg/ml of realgar. In addition, histogram analysis was performed on early apoptosis (B) and late apoptosis (C). Data were obtained from three independent experiments and are presented as the mean ± S.D. N = 3.
Effect of realgar treatment on cell cycle arrest
To verify whether realgar suppressed the proliferation of NB4 cells by triggering cell cycle arrest, we evaluated the change in the cell cycle distribution after NB4 cells were treated with realgar. Figure 4A shows that realgar caused S and G2/M phase accumulation, and correspondingly, G0/G1 phase was decreased in NB4 cells. After exposure to various concentrations of realgar for 24 h, it was discovered that the S phase increased from 3.69% to 7.43%, 8.30%, 12.07%, and 17.84%, and the G2/M phase increased from 4.12% to 6.54%, 8.02%, 11.04%, and 13.86%, and there was a statistically significant difference compared to the control samples (Figure 4B). These results suggested that realgar can induce APL cell cycle arrest to inhibit proliferation in a dose-dependent manner.
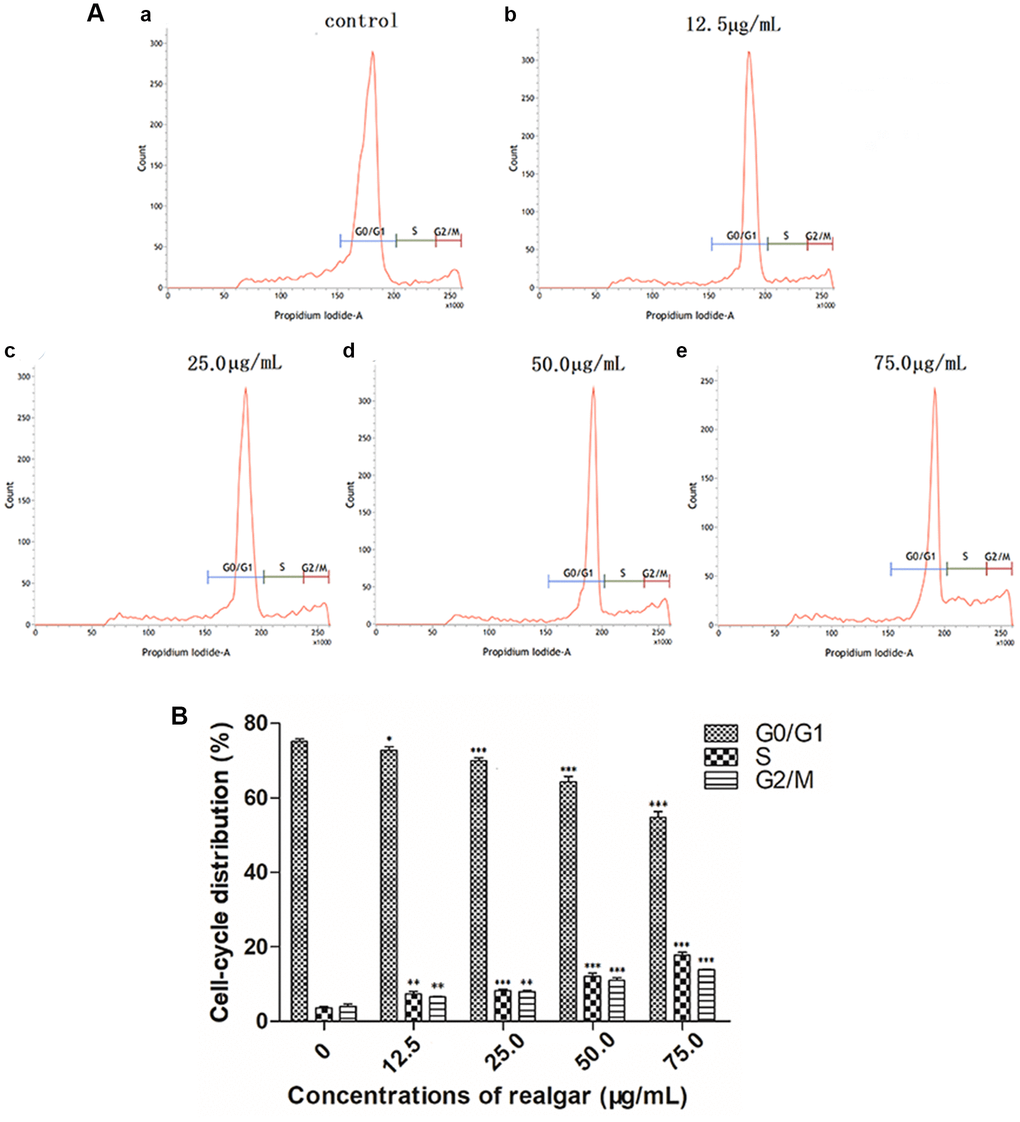
Figure 4. Effect of realgar on APL cell cycle arrest. (A) (a) Control, (b) 12.5 μg/mL, (c) 25.0 μg/mL, (d) 50.0 μg/mL, and (e) 75.0 μg/ml. The statistical data were generated by flow cytometry software and plotted as generated. (B) Histogram analysis of cell cycle phase distributions of NB4 cells after treatments with 0, 12.5, 25.0, 50.0 and 75.0 μg/mL of realgar. Data were obtained from three independent experiments and are presented as the mean ± SD. *P < 0.05, **P < 0.01 and ***P < 0.001.
ATP levels
After 24 hours of treatment with different concentrations of realgar for 24 hours, the ATP levels in NB4 cells declined from 100% to 76.409%, 54.008%, and 48.825% (Figure 5). The results showed that ATP generation by NB4 cells was drastically inhibited following exposure to realgar.
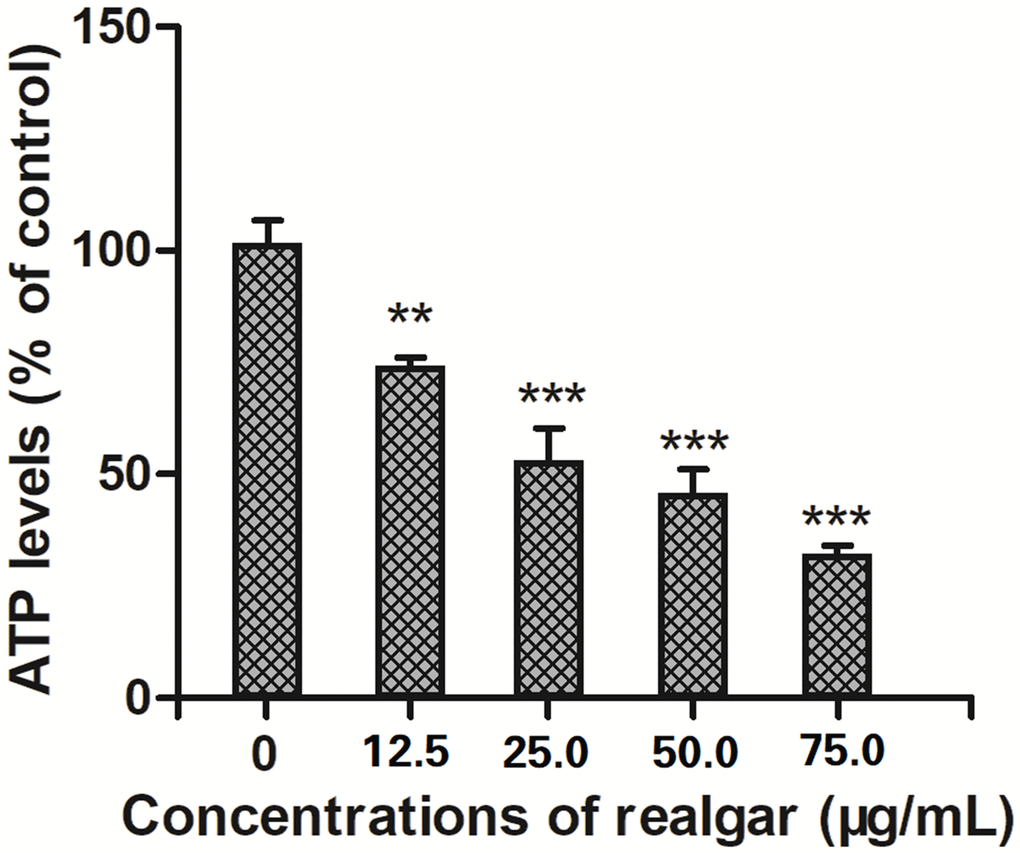
Figure 5. Effect of different concentrations of realgar on ATP generation in APL cells for 24 h. The cells were treated with different concentrations of realgar (0, 12.5, 25.0, 50.0 and 75.0 μg/ml) for 24 h. Then, the ATP levels were measured using a commercial ATP assay kit according to the manufacturer's instructions. Data were obtained from three independent experiments and are presented as the mean ± SD. **P < 0.01 and ***P < 0.001.
Bcl-2, Bax, Cyt-C, and AIF gene expression
In this study, the impact of different concentrations of realgar on the levels of Bcl-2, Bax, Cyt-C, and AIF gene expression in NB4 cells was investigated using qPCR. As shown in (Figure 6A–6D), the expression levels of the Bcl-2, Bax, Cyt-C and AIF genes were differentially regulated after the cells were treated with realgar. We observed that the level of Bcl-2 in NB4 cells decreased in a dose dependent manner to 0.849-, 0.751-, 0.687- and 0.609-fold compared with the control samples after exposure to realgar, whereas Bax increased to 1.307-, 1.564-, 1.733- and 1.838-fold, Cyt-C increased to 1.301-, 1.415-, 1.552- and 1.672-fold, and AIF increased to 1.324-, 1.401-, 1.572- and 1.679-fold. These findings demonstrate that realgar treatment can cause NB4 cell death.
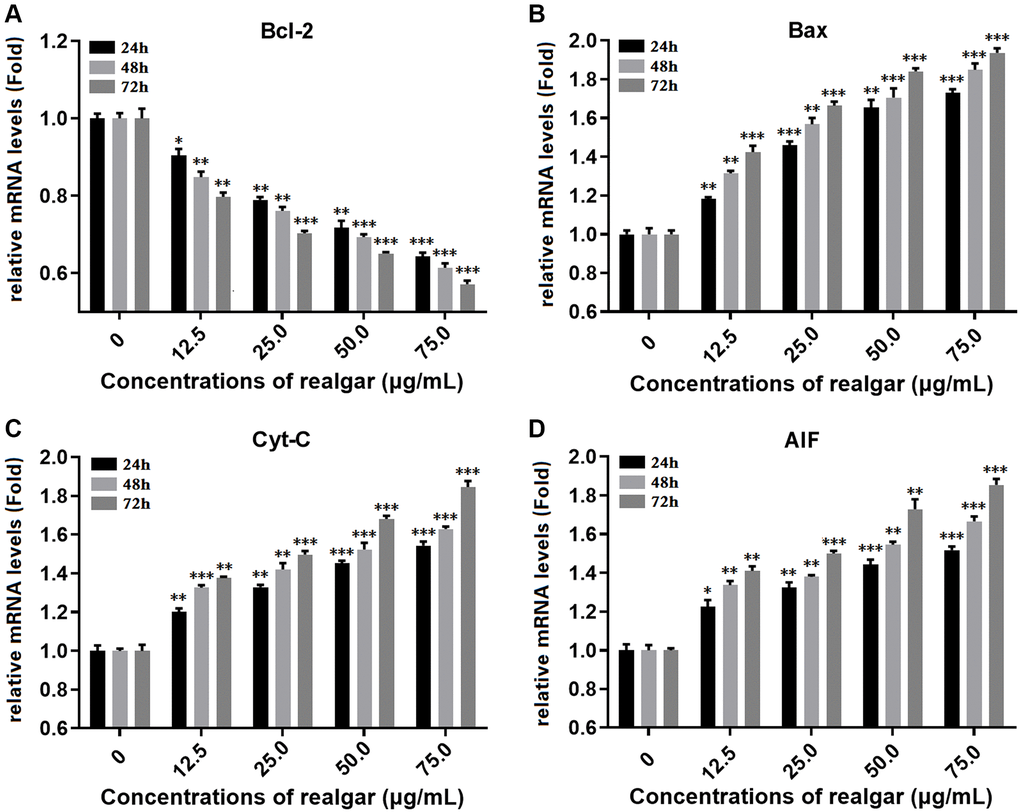
Figure 6. Measurement of the gene expression of Bcl-2, Bax, Cyt-C and AIF. NB4 cells were treated with different concentrations of realgar (0, 12.5, 25.0, 50.0 and 75.0 μg/ml) for 24, 48 and 72 h, and the mRNA expression levels of Bcl-2, Bax, Cyt-C and AIF were compared to those in untreated cells. (A) Bcl-2 mRNA level; (B) Bax mRNA level; (C) Cyt-C mRNA level and (D) AIF mRNA level. The results are expressed as the mean ± S.D. (standard deviation) of three independent experiments. *P < 0.05, **P < 0.01 and ***P < 0.001.
Bcl-2, Bax, Cyt-C, and AIF protein expression
Typically, deregulated cell proliferation and cell apoptosis are intimately coupled. Therefore, we evaluated the effect of realgar on cell apoptosis by evaluating the expression of apoptosis-associated proteins in NB4 cells upon realgar stimulation. Based on the cell proliferation data, the optimal concentration and stimulation time (75 μg/ml, 72 h) were used in this experiment. The results of the immunoblotting analysis showed that realgar treatment clearly downregulated the expression of the antiapoptotic protein Bcl-2 and promoted the expression of the proapoptotic protein Bax. In addition, the levels of Cyt-C and AIF were also elevated (Figure 7A and 7B).
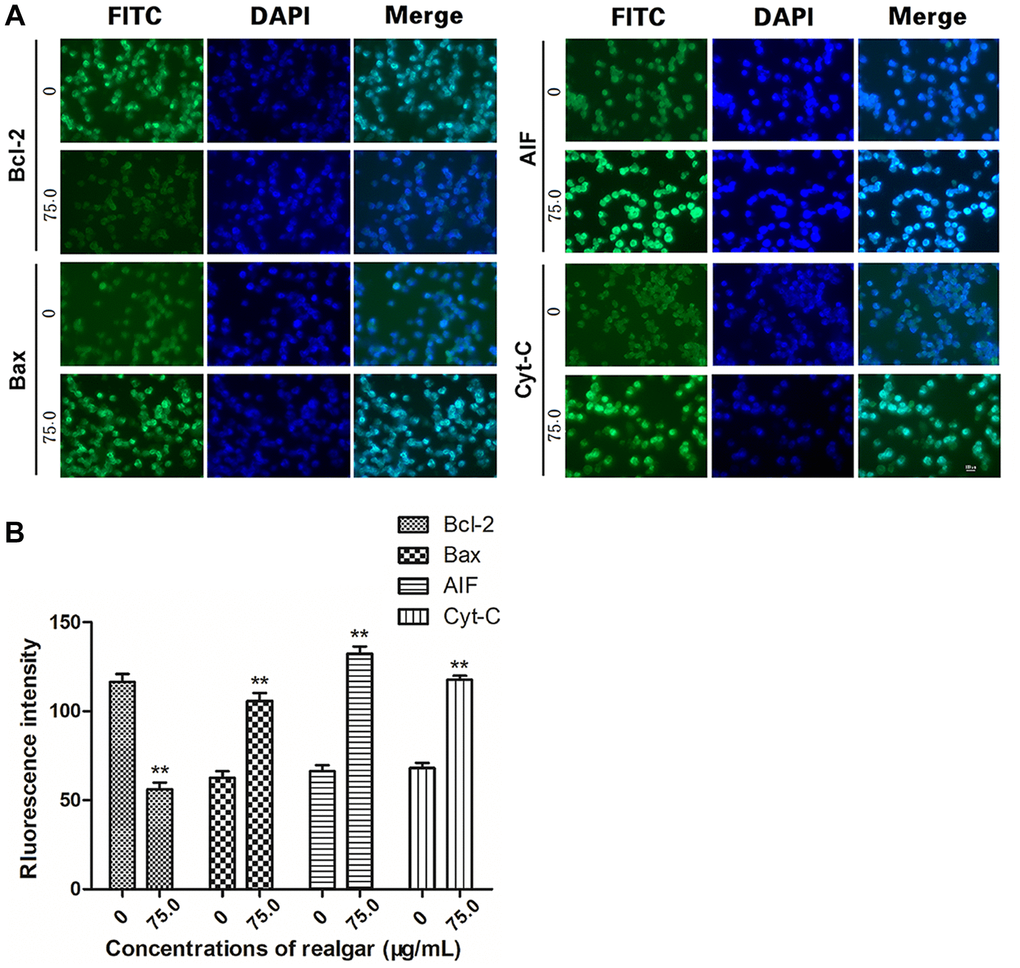
Figure 7. Determination of the target protein levels. NB4 cells were treated with or without realgar (75.0 μg/ml) for 72 h. The levels of Bcl-2, Bax, Cyt-C, and AIF were measured using immunofluorescence. (A) Cell nuclei were stained with 4',6-diamidino-2-phenylindole (DAPI; blue). (B) The fluorescence intensity of each protein is shown. Original magnification, × **P < 0.01.
To further verify this outcome, we performed western blotting analysis (Figure 8A). The results also showed that the level of Bcl-2 was elevated upon realgar stimulation, and in contrast, the expression of Bax, Cyt-C and AIF was decreased (Figure 8A and 8B). This finding indicates that the apoptotic pathway mediated by mitochondria in NB4 cells may be triggered by realgar exposure, implying that realgar-induced apoptosis in NB4 cells could be related to the Bcl-2, Bax, Cyt-C, and AIF signaling pathways.
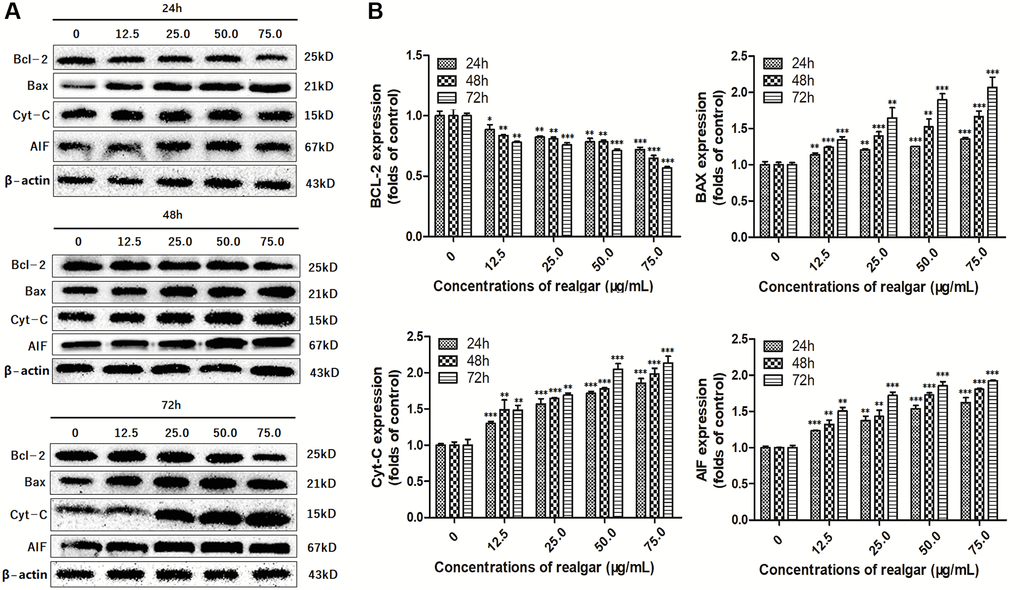
Figure 8. Evaluation of the Bcl-2, Bax, Cyt-C and AIF protein levels. Western blot analysis was performed in order to determine the Bcl-2, Bax, Cyt-C and AIF protein levels in NB4 cells after treatment with different concentrations of realgar (0, 12.5, 25.0, 50.0 and 75.0 μg/ml, respectively) for 24, 48 and 72 h, and three independent experiments were repeated. (A) Bcl-2, Bax, Cyt-C and AIF protein levels; (B) Histogram analysis of the target protein expression levels in NB4 cells. *P < 0.05, **P < 0.01, and ***P < 0.001 vs. relevant control samples.
Discussion
For more than 1500 years, traditional Chinese doctors have employed realgar, a traditional Chinese medicine, to treat hematological malignancies by oral administration [18, 32]. Employed either externally or orally, it has a significant therapeutic effect on carbuncles, lumps, snake bites, furuncles, parasitoses, scrofula and syphilis, among other external and interior ailments [20, 21]. Realgar is less toxic than ATO and can be administered orally. Numerous clinical studies have demonstrated that oral realgar alone or in combination with other drugs has a good therapeutic effect for APL patients [33–35]. Therefore, realgar is regarded as a promising therapeutic candidate and is worthy of further exploitation in the treatment of APL. Nevertheless, the underlying mechanism behind realgar therapy is largely unclear. In this study, we evaluated the effects of realgar on APL using the APL cell line NB4. We demonstrated that realgar has a cytotoxic impact on APL cells, inducing them to die. We hypothesized that the mechanism of realgar-induced APL cell death may be related to mitochondria-mediated apoptosis.
A CCK8 assay was used to analyze the inhibitory impact of realgar on cell proliferation. We observed that by increasing the concentration of realgar cultured with the cells and lengthening the culture period, APL cell viability gradually decreased (Figure 1). Chen et al. found that after treatment with realgar in an MTT assay, ATRA-sensitive NB4 and ATRA-resistant MR2 cells were significantly inhibited in a dose- and time-dependent manner [30]. This result is similar to our findings, indicating that realgar has a cytotoxic effect on tumor cells.
At present, apoptosis is generally thought to be the most common mechanism of cell death [36]. We found that the morphology of APL cells was altered dramatically after treatment with realgar for 24 hours. The greater the concentration of realgar used to treat APL cells, the more severe the morphological alterations (Figure 2). These results are also in agreement with our Annexin V-FITC/PI staining results (Figure 3). AIF is a protein with apoptosis-inducing activity that is located in the membrane space of mitochondria. When cells are stimulated by apoptosis, AIF molecules are released from mitochondria to the cytoplasm, then translocated to the nucleus, and combined with chromosomal DNA to agglutinate around the chromosomal nucleus and break DNA into large fragments of approximately 50 Kb. DNA damage can cause the regular processes of cells to be disrupted, eventually leading to death [37]. As a consequence, we can deduce that realgar may cause APL cells (NB4) to die by breaking DNA chains.
Cell cycle arrest has become one of the most successful cancer therapies after years of study into the cell cycle and its role in cancer development [38]. Flow cytometry analysis was performed in APL cells following treatment with realgar to determine cell cycle arrest caused by realgar. According to this study, realgar arrests the APL cell cycle in the S and G2/M phases in a concentration-dependent manner (Figure 4). Our findings support the hypothesis that realgar can cause cell cycle arrest in APL cells. Since the cell cycle-arrested cells could not enter the mitotic phase, realgar may be a potential therapy for preventing APL cell division. However, Ye et al. explored the effect of realgar on another APL cell line, HL-60, and showed that realgar-induced membrane toxicity may play an important role in the induction of apoptosis in APL cells [39].
Mitochondria are vital to cell survival. They not only produce ROS but also generate ATP through the oxidative phosphorylation system. In pathological situations, the malfunctioning of mitochondria results in ATP exhaustion [40]. We investigated the influence of realgar on the mitochondrial function of APL cells and showed that the amount of ATP produced decreased as the concentration of realgar increased (Figure 5), indicating that realgar can impact mitochondrial activity in APL cells. Based on these observations, we infer that realgar disrupts mitochondrial homeostasis, induces mitochondrial malfunction, and activates the mitochondria-mediated apoptotic signaling pathway, resulting in APL cell death. In contrast to our findings, Hai Y et al. discovered that realgar triggered NB4 cell differentiation by partly degrading PML/RARa via the ubiquitin–proteasome pathway [22].
Apoptosis, also called programmed cell death, is a kind of cell death that is vital for maintaining homeostasis [41]. It has been demonstrated that various proteins are involved in apoptosis. Bcl-2 family proteins, as important regulators of cell apoptosis, are mainly located in mitochondria and regulate cell apoptosis by influencing mitochondrial function [42]. Within the Bcl-2 family, Bcl-2 protein inhibits cell apoptosis by regulating the integrity of mitochondria and endoplasmic reticulum, promoting the release of Cyt-C and the activity of caspases [43]. In addition, Bcl-2 can integrate with other apoptosis-promoting proteins, such as Bax, and inhibit the function of Bax [44]. In the context of cellular stress, the Bcl-2/Bax expression ratio has been suggested to indicate cell survival. Over the last decade, evidence has emerged that Cyt-C plays a vital function in programmed cell death [45, 46]. The release of Cyt-C from mitochondria into the cytoplasm initiates a cascade reaction, activates caspase signaling and regulates cell apoptosis. In addition, AIF exists in the inner mitochondrial membrane and is a factor that can induce caspase-independent apoptosis. In the process of apoptosis, AIF is transferred from mitochondria to the cytoplasm and then enters the nucleus, causing DNA agglutination and fragmentation in the nucleus, resulting in cell death [47].
To determine the underlying mechanism of realgar-induced APL cell death, we evaluated the effect of realgar on mitochondrial function. The expression of Bcl-2, Bax, Cyt-C, and AIF at the mRNA and protein levels was evaluated before and after exposure to various concentrations of realgar using immunofluorescence, western blotting, and qPCR experiments. Our results showed that Bcl-2 was decreased and Bax, Cyt-C, and AIF were increased at the mRNA level in a concentration-dependent manner after APL cells were treated with realgar (Figure 6). In addition, Bcl-2 was decreased, whereas Bax, Cyt-C, and AIF were increased at the protein level in a concentration-dependent manner after APL cells were treated with realgar (Figures 7 and 8). Therefore, our findings demonstrate that realgar induces APL cell death mainly through the abnormal regulation of mitochondrial function and that Bcl-2, Bax, Cyt-C, and AIF play essential roles in realgar-induced APL cell death. Interestingly, Xie et al. found that after realgar treatment, the expression of Caspase-3, Caspase-9 and Cyt-C in NB4 cells increased, thus initiating the caspase-dependent intrinsic apoptosis pathway [48].
Qi et al. studied the differential proteome after realgar was used to interfere with the APL retinoic acid-resistant cell line NB4-R1 [49, 50]. A total of 21 differentially expressed proteins were screened, of which 5 proteins were downregulated and 16 proteins were upregulated. Mass spectrometry showed that the differentially expressed proteins SET (I2PP2A), RPP0, RPP2, PCBP1, ACTB, HMGB1, PHB, RhoGDI2 and elFSA1 were closely related to the proliferation and apoptosis of APL cells. Wang et al. discovered changes in the gene expression profile after realgar acted on NB4 cells and found that the altered expression of PSMC2, PSMD1 and ITGB1 genes in particular may play an important role in the apoptosis and differentiation of NB4 cells [51, 52]. The interaction between the realgar-induced apoptosis effect and genomics and proteomics needs to be further investigated.
As shown in Figure 9, APL cells exposed to realgar reduced ATP production, destroyed the nucleus, and induced APL cell cycle arrest at the S and G2/M phases. In addition, realgar also decreased Bcl-2 expression while increasing Bax, Cyt-C, and AIF expression at the mRNA and protein levels. Our findings indicate that Bcl-2, Bax, Cyt-C, and AIF might play an essential role in realgar-mediated APL cell death.
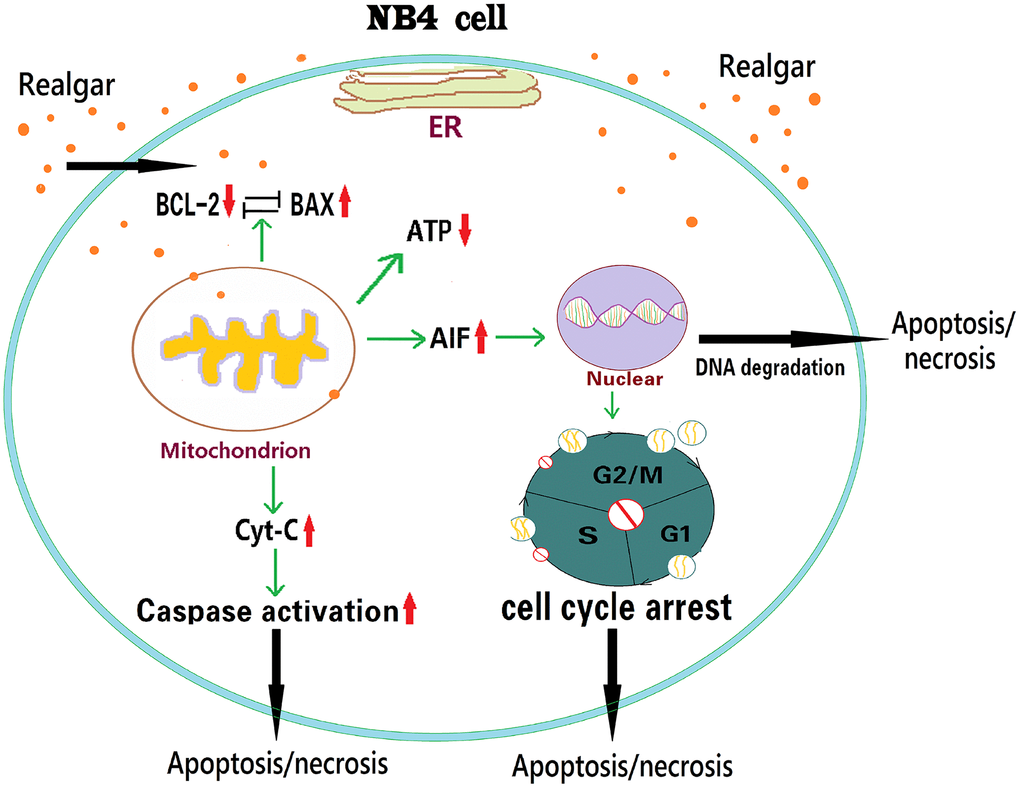
Figure 9. Schematic illustration of how realgar induces APL cell death via the Bcl-2/Bax/Cyt-C/AIF signaling pathway.
Conclusion
Overall, our results demonstrate that realgar can effectively induce APL (NB4) cell death in vitro in a dose- and time-dependent manner. Realgar treatment of APL cells can reduce ATP production, damage the nucleus, and induce APL cell cycle arrest at the S and G2/M phases. In addition, it can also disrupt mitochondrial homeostasis by decreasing Bcl-2 expression and increasing Bax, Cyt-C, and AIF expression at the mRNA and protein levels. Therefore, Bcl-2, Bax, Cyt-C, and AIF may play essential roles in APL cell death. Our findings provide new insight into realgar-induced APL cell death involved in mitochondria-mediated apoptosis, and indicate that realgar may be a potential therapeutic for APL in clinical practice. Animal studies should be performed next to confirm the therapeutic effects of ZnO NPs on MM.
Supplementary Materials
Author Contributions
Zonghong Li: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing–original draft. Ruiming Zhang: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing–original draft. Xuewei Yin: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing-original draft. Nana Li: Software, Supervision, Validation. Siyuan Cui: Data curation, Investigation, Methodology. Teng Wang: Investigation, Methodology, Validation. Xing Tan: Investigation, Methodology, Validation. Mingyue Shen: Investigation, Validation. Yun Guo: Investigation, Visualization. Jinxin Wang: Investigation, Visualization. Dadong Guo: Conceptualization, Resources, Supervision, Writing-review and editing. Ruirong Xu: Conceptualization, Funding acquisition, Project administration, Writing-review and editing. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This study was funded by the National Natural Science Foundation of China (81974547, 81903984 and 81473511), the National Natural Science Foundation of Shandong Province (ZR2019PH018) and the Development Project of Medicine and Health Science Technology of Shandong Province (2013WS0249).
References
- 1. Yilmaz M, Kantarjian H, Ravandi F. Acute promyelocytic leukemia current treatment algorithms. Blood Cancer J. 2021; 11:123. https://doi.org/10.1038/s41408-021-00514-3 [PubMed]
- 2. Borrow J, Goddard AD, Sheer D, Solomon E. Molecular analysis of acute promyelocytic leukemia breakpoint cluster region on chromosome 17. Science. 1990; 249:1577–80. https://doi.org/10.1126/science.2218500 [PubMed]
- 3. Tallman MS, Abutalib SA, Altman JK. The double hazard of thrombophilia and bleeding in acute promyelocytic leukemia. Semin Thromb Hemost. 2007; 33:330–8. https://doi.org/10.1055/s-2007-976168 [PubMed]
- 4. Kayser S, Schlenk RF, Platzbecker U. Management of patients with acute promyelocytic leukemia. Leukemia. 2018; 32:1277–94. https://doi.org/10.1038/s41375-018-0139-4 [PubMed]
- 5. Breccia M, Avvisati G, Latagliata R, Carmosino I, Guarini A, De Propris MS, Gentilini F, Petti MC, Cimino G, Mandelli F, Lo-Coco F. Occurrence of thrombotic events in acute promyelocytic leukemia correlates with consistent immunophenotypic and molecular features. Leukemia. 2007; 21:79–83. https://doi.org/10.1038/sj.leu.2404377 [PubMed]
- 6. Breccia M, Lo Coco F. Thrombo-hemorrhagic deaths in acute promyelocytic leukemia. Thromb Res. 2014 (Suppl 2); 133:S112–6. https://doi.org/10.1016/S0049-3848(14)50019-9 [PubMed]
- 7. Rajpurkar M, Alonzo TA, Wang YC, Gerbing RB, Gamis AS, Feusner JH, Gregory J, Kutny MA. Risk Markers for Significant Bleeding and Thrombosis in Pediatric Acute Promyelocytic Leukemia; Report From the Children's Oncology Group Study AAML0631. J Pediatr Hematol Oncol. 2019; 41:51–5. https://doi.org/10.1097/MPH.0000000000001280 [PubMed]
- 8. Ku GH, White RH, Chew HK, Harvey DJ, Zhou H, Wun T. Venous thromboembolism in patients with acute leukemia: incidence, risk factors, and effect on survival. Blood. 2009; 113:3911–7. https://doi.org/10.1182/blood-2008-08-175745 [PubMed]
- 9. Yanada M, Matsushita T, Asou N, Kishimoto Y, Tsuzuki M, Maeda Y, Horikawa K, Okada M, Ohtake S, Yagasaki F, Matsumoto T, Kimura Y, Shinagawa K, et al. Severe hemorrhagic complications during remission induction therapy for acute promyelocytic leukemia: incidence, risk factors, and influence on outcome. Eur J Haematol. 2007; 78:213–9. https://doi.org/10.1111/j.1600-0609.2006.00803.x [PubMed]
- 10. Kayser S, Schlenk RF, Lebon D, Carre M, Götze KS, Stölzel F, Berceanu A, Schäfer-Eckart K, Peterlin P, Hicheri Y, Rahme R, Raffoux E, Chermat F, et al. Characteristics and outcome of patients with low-/intermediate-risk acute promyelocytic leukemia treated with arsenic trioxide: an international collaborative study. Haematologica. 2021; 106:3100–6. https://doi.org/10.3324/haematol.2021.278722 [PubMed]
- 11. Bernard J, Weil M, Boiron M, Jacquillat C, Flandrin G, Gemon MF. Acute promyelocytic leukemia: results of treatment by daunorubicin. Blood. 1973; 41:489–496. Blood. 2016; 128:1779. https://doi.org/10.1182/blood-2016-08-735084 [PubMed]
- 12. Huang ME, Ye YC, Chen SR, et al. Use of all-trans retinoic acid in the treatment of acute promyelocytic leukemia. Blood. 1988; 72:567–572. Blood. 2016; 128:3017. https://doi.org/10.1182/blood-2016-11-750182 [PubMed]
- 13. Daenen S, Vellenga E, van Dobbenburgh OA, Halie MR. Retinoic acid as antileukemic therapy in a patient with acute promyelocytic leukemia and Aspergillus pneumonia. Blood. 1986; 67:559–61. [PubMed]
- 14. Gallagher RE, Moser BK, Racevskis J, Poiré X, Bloomfield CD, Carroll AJ, Ketterling RP, Roulston D, Schachter-Tokarz E, Zhou DC, Chen IM, Harvey R, Koval G, et al. Treatment-influenced associations of PML-RARα mutations, FLT3 mutations, and additional chromosome abnormalities in relapsed acute promyelocytic leukemia. Blood. 2012; 120:2098–108. https://doi.org/10.1182/blood-2012-01-407601 [PubMed]
- 15. Gurnari C, De Bellis E, Divona M, Ottone T, Lavorgna S, Voso MT. When Poisons Cure: The Case of Arsenic in Acute Promyelocytic Leukemia. Chemotherapy. 2019; 64:238–47. https://doi.org/10.1159/000507805 [PubMed]
- 16. Shi F, Feng N, Omari-Siaw E. Realgar nanoparticle-based microcapsules: preparation and in-vitro/in-vivo characterizations. J Pharm Pharmacol. 2015; 67:35–42. https://doi.org/10.1111/jphp.12314 [PubMed]
- 17. Cohen SM, Arnold LL, Eldan M, Lewis AS, Beck BD. Methylated arsenicals: the implications of metabolism and carcinogenicity studies in rodents to human risk assessment. Crit Rev Toxicol. 2006; 36:99–133. https://doi.org/10.1080/10408440500534230 [PubMed]
- 18. Liu J, Liang SX, Lu YF, Miao JW, Wu Q, Shi JS. Realgar and realgar-containing Liu-Shen-Wan are less acutely toxic than arsenite and arsenate. J Ethnopharmacol. 2011; 134:26–31. https://doi.org/10.1016/j.jep.2010.11.052 [PubMed]
- 19. Hai Y, Song P, Wang X, Zhao L, Xie Q, Li J, Li Y, Li H. Realgar transforming solution as a novel arsenic agent with a lower risk of cardiotoxicity. J Pharmacol Sci. 2019; 140:162–70. https://doi.org/10.1016/j.jphs.2019.06.003 [PubMed]
- 20. Wu J, Shao Y, Liu J, Chen G, Ho PC. The medicinal use of realgar (As4S4) and its recent development as an anticancer agent. J Ethnopharmacol. 2011; 135:595–602. https://doi.org/10.1016/j.jep.2011.03.071 [PubMed]
- 21. Liu J, Lu Y, Wu Q, Goyer RA, Waalkes MP. Mineral arsenicals in traditional medicines: orpiment, realgar, and arsenolite. J Pharmacol Exp Ther. 2008; 326:363–8. https://doi.org/10.1124/jpet.108.139543 [PubMed]
- 22. Hai Y, Wang X, Song P, Li JY, Zhao LH, Xie F, Tan XM, Xie QJ, Yu L, Li Y, Wu ZR, Li HY. Realgar transforming solution-induced differentiation of NB4 cell by the degradation of PML/RARα partially through the ubiquitin-proteasome pathway. Arch Pharm Res. 2019; 42:684–94. https://doi.org/10.1007/s12272-019-01170-9 [PubMed]
- 23. Zhang L, Tian W, Kim S, Ding W, Tong Y, Chen S. Arsenic sulfide, the main component of realgar, a traditional Chinese medicine, induces apoptosis of gastric cancer cells in vitro and in vivo. Drug Des Devel Ther. 2014; 9:79–92. https://doi.org/10.2147/DDDT.S74379 [PubMed]
- 24. Ding W, Zhang L, Kim S, Tian W, Tong Y, Liu J, Ma Y, Chen S. Arsenic sulfide as a potential anti-cancer drug. Mol Med Rep. 2015; 11:968–74. https://doi.org/10.3892/mmr.2014.2838 [PubMed]
- 25. Ding W, Ji T, Xiong W, Li T, Pu D, Liu R. Realgar, a traditional Chinese medicine, induces apoptosis of HPV16-positive cervical cells through a HPV16 E7-related pathway. Drug Des Devel Ther. 2018; 12:3459–69. https://doi.org/10.2147/DDDT.S172525 [PubMed]
- 26. Liu J, Wei LX, Wang Q, Lu YF, Zhang F, Shi JZ, Li C, Cherian MG. A review of cinnabar (HgS) and/or realgar (As4S4)-containing traditional medicines. J Ethnopharmacol. 2018; 210:340–50. https://doi.org/10.1016/j.jep.2017.08.037 [PubMed]
- 27. Wu JZ, Ho PC. Evaluation of the in vitro activity and in vivo bioavailability of realgar nanoparticles prepared by cryo-grinding. Eur J Pharm Sci. 2006; 29:35–44. https://doi.org/10.1016/j.ejps.2006.05.002 [PubMed]
- 28. Wang XB, Gao HY, Hou BL, Huang J, Xi RG, Wu LJ. Nanoparticle realgar powders induce apoptosis in U937 cells through caspase MAPK and mitochondrial pathways. Arch Pharm Res. 2007; 30:653–8. https://doi.org/10.1007/BF02977662 [PubMed]
- 29. Lu DP, Wang Q. Current study of APL treatment in China. Int J Hematol. 2002 (Suppl 1); 76:316–8. https://doi.org/10.1007/BF03165273 [PubMed]
- 30. Chen S, Fang Y, Ma L, Liu S, Li X. Realgar-induced apoptosis and differentiation in all-trans retinoic acid (ATRA)-sensitive NB4 and ATRA-resistant MR2 cells. Int J Oncol. 2012; 40:1089–96. https://doi.org/10.3892/ijo.2011.1276 [PubMed]
- 31. Lu DP, Qiu JY, Jiang B, Wang Q, Liu KY, Liu YR, Chen SS. Tetra-arsenic tetra-sulfide for the treatment of acute promyelocytic leukemia: a pilot report. Blood. 2002; 99:3136–43. https://doi.org/10.1182/blood.v99.9.3136 [PubMed]
- 32. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016; 66:7–30. https://doi.org/10.3322/caac.21332 [PubMed]
- 33. Lou Y, Ma Y, Jin J, Zhu H. Oral Realgar-Indigo Naturalis Formula Plus Retinoic Acid for Acute Promyelocytic Leukemia. Front Oncol. 2021; 10:597601. https://doi.org/10.3389/fonc.2020.597601 [PubMed]
- 34. Lu SY, Wen-Jing L, Lou R, Ma R, Zhu JH, Jiang H. Oral etoposide combined with oral arsenic plus retinoic acid for two cases with newly diagnosed high-risk acute promyelocytic leukemia during COVID19 pandemic. Leuk Res Rep. 2021; 16:100258. https://doi.org/10.1016/j.lrr.2021.100258 [PubMed]
- 35. Zhang L, Yang XM, Chen J, Hu L, Yang F, Zhou Y, Zhao BB, Zhao W, Zhu XF. Population Pharmacokinetics and Safety of Oral Tetra-Arsenic Tetra-Sulfide Formula in Pediatric Acute Promyelocytic Leukemia. Drug Des Devel Ther. 2021; 15:1633–40. https://doi.org/10.2147/DDDT.S305244 [PubMed]
- 36. Bisht G, Rayamajhi S. ZnO Nanoparticles: A Promising Anticancer Agent. Nanobiomedicine (Rij). 2016; 3:9. https://doi.org/10.5772/63437 [PubMed]
- 37. Li Z, Guo D, Yin X, Ding S, Shen M, Zhang R, Wang Y, Xu R. Zinc oxide nanoparticles induce human multiple myeloma cell death via reactive oxygen species and Cyt-C/Apaf-1/Caspase-9/Caspase-3 signaling pathway in vitro. Biomed Pharmacother. 2020; 122:109712. https://doi.org/10.1016/j.biopha.2019.109712 [PubMed]
- 38. Otto T, Sicinski P. Cell cycle proteins as promising targets in cancer therapy. Nat Rev Cancer. 2017; 17:93–115. https://doi.org/10.1038/nrc.2016.138 [PubMed]
- 39. Ye HQ, Gan L, Yang XL, Xu HB. Membrane-associated cytotoxicity induced by realgar in promyelocytic leukemia HL-60 cells. J Ethnopharmacol. 2006; 103:366–71. https://doi.org/10.1016/j.jep.2005.08.014 [PubMed]
- 40. Khalil B, Liévens JC. Mitochondrial quality control in amyotrophic lateral sclerosis: towards a common pathway? Neural Regen Res. 2017; 12:1052–61. https://doi.org/10.4103/1673-5374.211179 [PubMed]
- 41. Taylor RC, Cullen SP, Martin SJ. Apoptosis: controlled demolition at the cellular level. Nat Rev Mol Cell Biol. 2008; 9:231–41. https://doi.org/10.1038/nrm2312 [PubMed]
- 42. Hardwick JM, Soane L. Multiple functions of BCL-2 family proteins. Cold Spring Harb Perspect Biol. 2013; 5:a008722. https://doi.org/10.1101/cshperspect.a008722 [PubMed]
- 43. Um HD. Bcl-2 family proteins as regulators of cancer cell invasion and metastasis: a review focusing on mitochondrial respiration and reactive oxygen species. Oncotarget. 2016; 7:5193–203. https://doi.org/10.18632/oncotarget.6405 [PubMed]
- 44. Martinou JC, Youle RJ. Mitochondria in apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev Cell. 2011; 21:92–101. https://doi.org/10.1016/j.devcel.2011.06.017 [PubMed]
- 45. Czabotar PE, Westphal D, Dewson G, Ma S, Hockings C, Fairlie WD, Lee EF, Yao S, Robin AY, Smith BJ, Huang DC, Kluck RM, Adams JM, Colman PM. Bax crystal structures reveal how BH3 domains activate Bax and nucleate its oligomerization to induce apoptosis. Cell. 2013; 152:519–31. https://doi.org/10.1016/j.cell.2012.12.031 [PubMed]
- 46. Kim EJY, Anko ML, Flensberg C, Majewski IJ, Geng FS, Firas J, Huang DCS, van Delft MF, Heath JK. BAK/BAX-Mediated Apoptosis Is a Myc-Induced Roadblock to Reprogramming. Stem Cell Reports. 2018; 10:331–8. https://doi.org/10.1016/j.stemcr.2017.12.019 [PubMed]
- 47. Hangen E, Féraud O, Lachkar S, Mou H, Doti N, Fimia GM, Lam NV, Zhu C, Godin I, Muller K, Chatzi A, Nuebel E, Ciccosanti F, et al. Interaction between AIF and CHCHD4 Regulates Respiratory Chain Biogenesis. Mol Cell. 2015; 58:1001–14. https://doi.org/10.1016/j.molcel.2015.04.020 [PubMed]
- 48. Xie Q, Yu L, Wang X, Wu Z, Zhi D, Yang J, Guo Z, Wu T, Sun Y, Zhao L, Ding X, Khan A, Li H. A novel realgar-indigo naturalis formula more effectively induces apoptosis in NB4 cells. Pak J Pharm Sci. 2019; 32:957–62. [PubMed]
- 49. Qi J, Zhang M, He PC, Wang HL, Wang XY. [Proteomic study of retinoid acid resistant NB4R1 cells apoptosis induced by realgar]. Zhonghua Xue Ye Xue Za Zhi. 2010; 31:752–7. [PubMed]
- 50. Qi J, Zhang M, He PC. [Establishment of two-dimensional electrophoresis proteomic profiles of retinoid acid resistant human acute promyelocytic leukemia NB4-R1 cells with apoptosis induced by realgar]. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2011; 31:391–6. [PubMed]
- 51. Wang HY, Liu SX. [Investigation on NB4 cell responses to realgar by cDNA microarray]. Zhongguo Zhong Yao Za Zhi. 2002; 27:600–4. [PubMed]
- 52. Wang H, Liu S, Lu X, Zhao X, Chen S, Li X. Gene expression profile changes in NB4 cells induced by realgar. Chin Med J (Engl). 2003; 116:1074–7. [PubMed]



