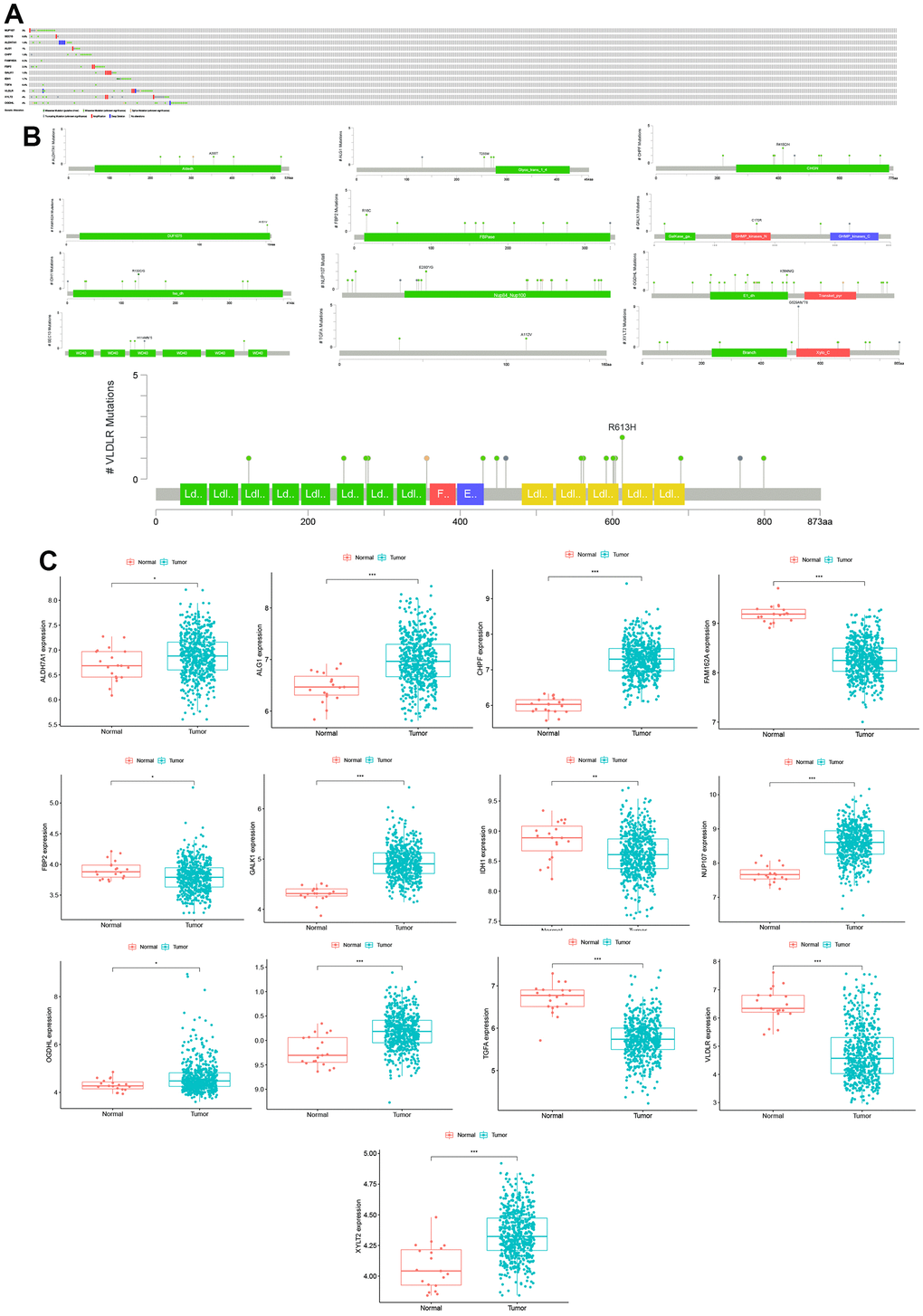
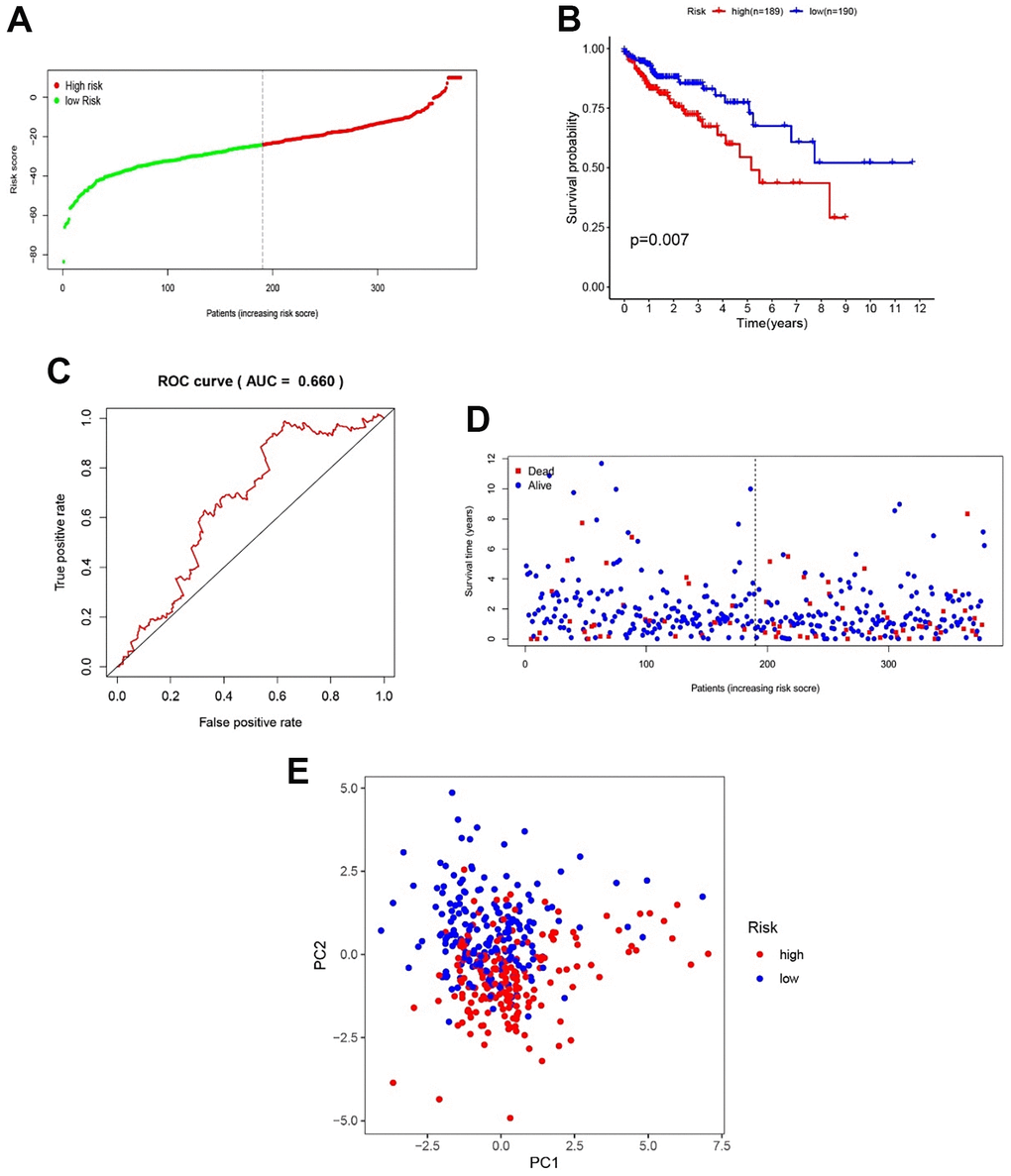
Introduction
Colon cancer (CC) is the third most common gastrointestinal malignant tumor derived from glandular epithelial cells, with a high mortality and increasing incidence [1]. Despite recent improvements in the diagnosis and treatment of CC, including surgery, radiotherapy, chemotherapy, neoadjuvant chemotherapy, and immunotherapy, the 5-year relative survival rate remains poor, and distant metastasis remains the main cause of CC-associated death [2, 3]. Therefore, there is an urgent need to explore potentially useful diagnostic and prognostic biomarkers for CC patients.
Glycometabolism, a vital pathway in living cells, differs significantly between normal cells and tumor cells. The transformation of glucose utilization from oxidative phosphorylation to glycolysis is now regarded as a major feature of cancer. This kind of change in energy metabolism is regulated by genetic changes and tumor microenvironment pressure, causing an increase in the cell proliferation rate and conferring resistance on cells [4]. The activation of glycolysis can affect other phenotypic processes, such as epithelial-to-mesenchymal transition (EMT) [5], which is closely associated with glycolysis. Zhao et al. reported that activated glycolysis promotes pancreatic cancer cell stemness and EMT [6]. Numerous studies have reported that the disturbance of immune regulation is closely associated with cancer, whereas the relationship between tumor immunity and CC glycolysis has not been explored. Hence, understanding the underlying interplay between glycolysis and cancer progression is a critical goal in cancer research that can clarify cancer pathogenesis and facilitate the development of effective therapeutic approaches.
In our study, gene set enrichment analysis (GSEA) of glycolysis-related pathways was conducted using 566 CC and 19 normal control array data from the Gene Expression Omnibus (GEO) to identify differentially expressed glycolysis genes in CC patients. Then, following Cox regression analysis, the patients were classified into low- and high-risk groups according to glycolysis-related expression and risk score. A signature consisting of 13 genes was constructed, which was closely associated with poor overall survival (OS), older age, high tumor grades, high EMT, different immune cell infiltration and targeted sensitive immune drugs. The risk score could serve as an independent pathological factor. Finally, the expression of the 13 signature genes was validated in our patient samples. Overall, our data revealed a strong association between glycolysis metabolism and clinical prognosis in CC.
Results
Clinical characteristics of CC patients
Overall, 566 CC and 19 normal control data stored on the Affymetrix Human Genome U133 Plus 2.0 Array platform were downloaded from a GEO dataset. The clinical characteristics of the samples, including sex, age, survival status, survival time, clinical TNM stage, clinical T stage, clinical N stage, and clinical M stage, are shown in Table 1.
Table 1. Clinical information of CC patients.
| Variables | GSE39582 Patients (566) | TCGA Patients (385) |
| Fustat | ||
| Alive | 370 | 314 |
| Death | 191 | 71 |
| unknown | 5 | |
| Age (year) | ||
| <=65 | 222 | 159 |
| >65 | 344 | 226 |
| Gender | ||
| Male | 256 | 205 |
| Female | 310 | 180 |
| TNM Stage | ||
| I | 33 | 66 |
| II | 264 | 151 |
| III | 205 | 103 |
| IV | 60 | 54 |
| 0 | 4 | 11 |
| T stage | ||
| T1 | 11 | 9 |
| T2 | 45 | 68 |
| T3 | 367 | 263 |
| T4 | 119 | 44 |
| Tis | 3 | 1 |
| T0 | 1 | 0 |
| unknown | 20 | 0 |
| M stage | ||
| M0 | 482 | 286 |
| MI | 61 | 54 |
| MX | 0 | 39 |
| unknown | 23 | 6 |
| N stage | ||
| N0 | 302 | 231 |
| N1 | 134 | 88 |
| N2 | 98 | 66 |
| N3 | 6 | 0 |
| unknown | 26 | 0 |
Differentially expressed glycolysis genes between the CC and normal control samples
Six glycolysis-related gene sets were obtained from the Molecular Signatures Database: REACTOME_GLYCOLYSIS, WP_COMPUTATIONAL_MODEL_OF_AEROBIC_GLYCOLYSIS, WP_GLYCOLYSIS_IN_SENESCENCE, REACTOME_REGULATION_OF_GLYCOLYSIS_BY_FRUCTOSE_2_6_BISPHOSPHATE_METABOLISM, HALLMARK_GLYCOLYSIS, and GO_GLYCOLYTIC_PROCESS. A total of 301 glycolysis-related genes were selected from the six gene sets for additional research, and 217 glycolysis-related genes were found to be significantly and differentially expressed in the CC samples compared with the controls (Figure 1).
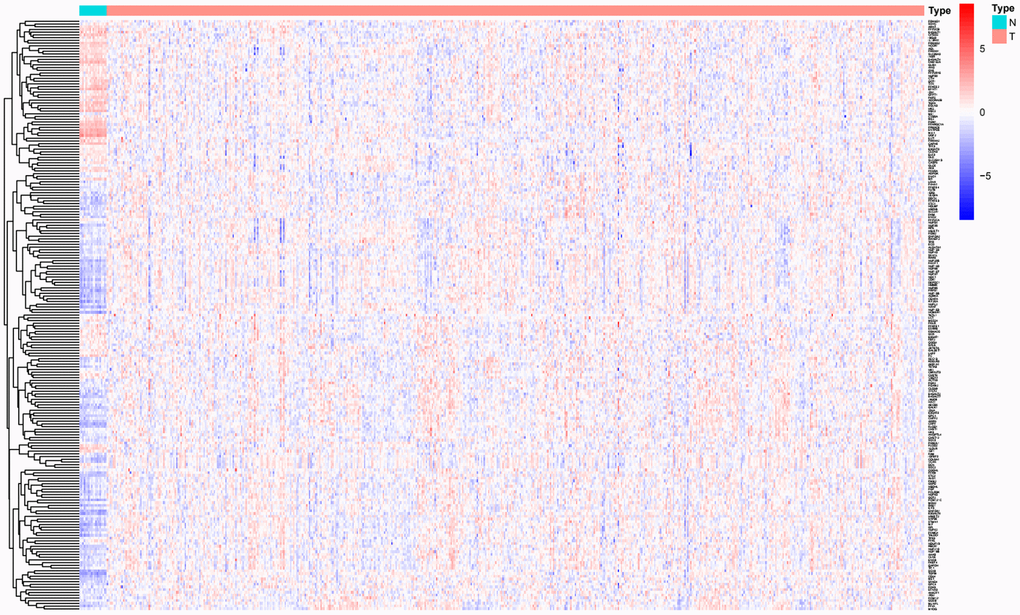
Figure 1. Significantly differentially expressed glycolysis-related genes between CC and normal controls.
Efficacy of the risk score in CC patients
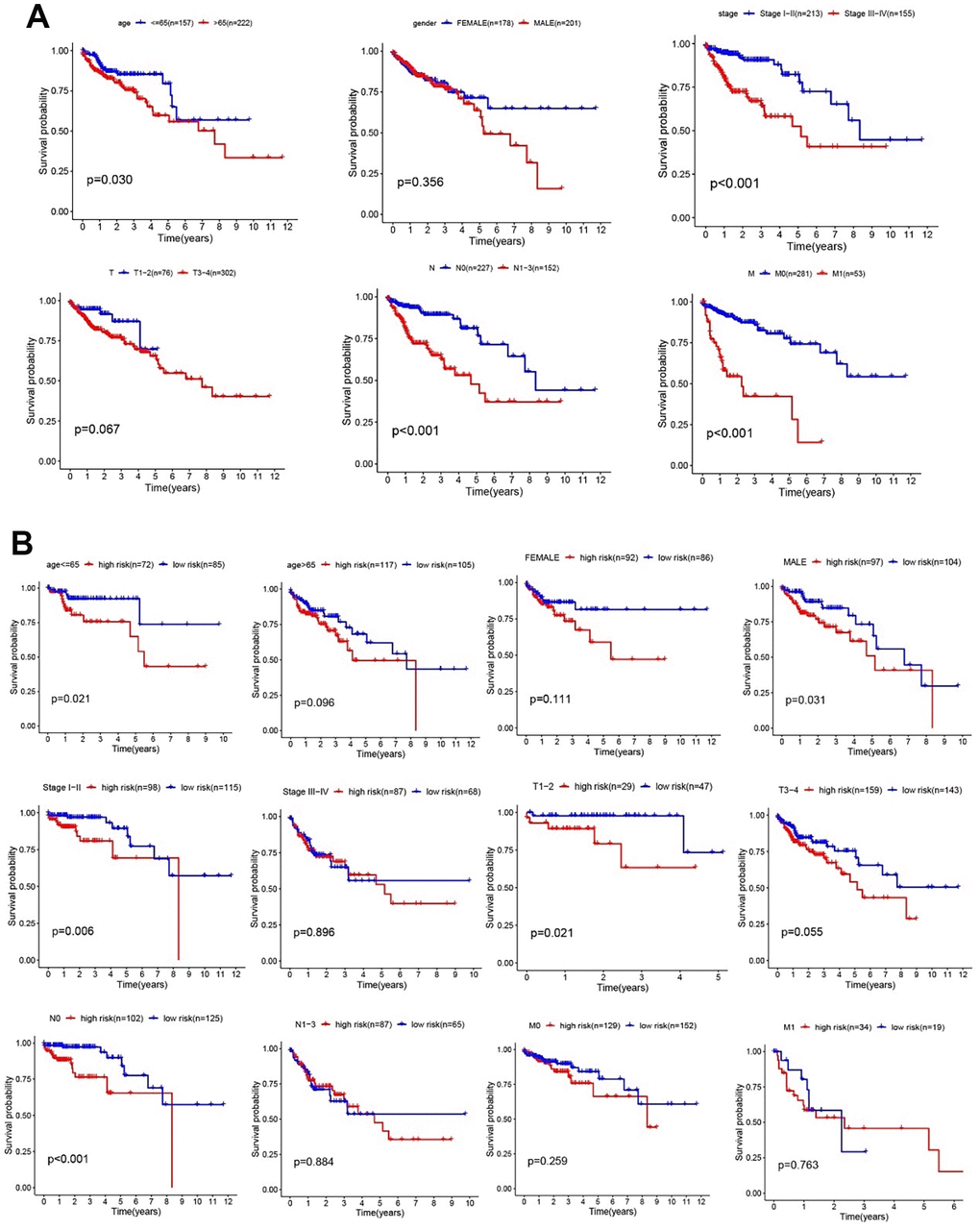
Based on the gene model, 280 CC patients and 281 CC patients were classified into high- and low-risk groups by the median risk score (Figure 3A). KM analysis was carried out between the low- and high-risk groups, and the high-risk group had a significantly poorer prognosis (P < 0.05, Figure 3B). ROC analysis was carried out, and the AUC was calculated to be 0.716, which showed a good prediction effect (Figure 3C). The expression of the 13 genes was calculated; TGFA FBP2, CHPF, and VLDLR were significantly higher in the high-risk group, and NUP107, OGDHL, SEC13, IDH1, GALK1, FAM162A, ALG1, and XYLT2 ALDH7A1 were downregulated (P < 0.05, Figure 3D). The risk plot indicates that the high-risk group was closely related to poor prognosis (Figure 3E). PCA showed that high- and low-risk groups could be significantly distinguished using our model, which reduced the dimension of the expression of multiple genes (Figure 3F). All the analysis results indicated that our gene model had good efficacy based on the risk score.
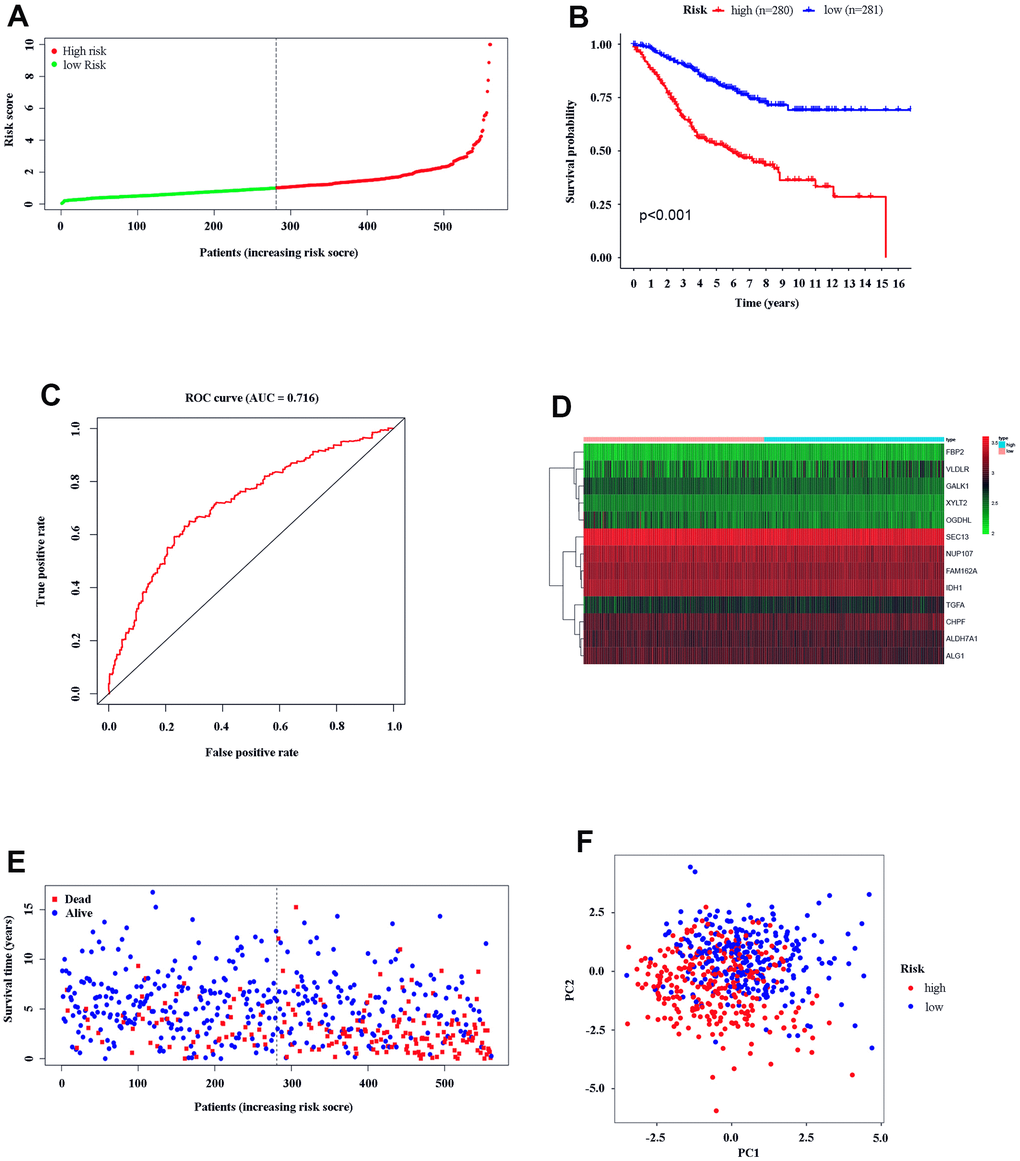
Figure 3. Text efficacy of the gene model in CC patients. (A) CC patient distribution according to the median risk score. (B) Kaplan–Meier analysis between the low- and high-risk groups. (C) ROC analysis of the risk score. (D) Model gene expression in the low- and high-risk groups. (E) The relationship between patient survival status and risk score. (F) PCA between the low- and high-risk groups.
Independent risk factor for risk score in CC patients and its relationship with clinical characteristics of the patients
Univariate and multivariate independent prognostic analyses were carried out to identify independent prognostic factors, including age, sex, TNM stage, and risk score. The results showed that age, TNM stage, and risk score were independent prognostic factors in CC patients, and they were all positively correlated with poor survival prognosis (P < 0.05, Figure 4A, 4B). For a more accurate analysis of the relationship between risk factors and patient OS, a series of Kaplan–Meier curve analyses was carried out. The results revealed that an age of > 65 years, TNM III–IV, T3-4, N1–3 and M1 were positively related to poor OS (P < 0.05, Figure 4C). The relationship between patients’ clinical characteristics and OS in the low- and high-risk groups was determined, and high-risk patients were positively associated with a poor OS when age ≤ 65 years, age>65 years, female sex, male sex, TNM I–II, TNM III–IV, T1–2, T3–4, M0, M1, N0, and N1-3 subgroups (P < 0.05, Figure 4D). These results demonstrate that our glycolysis-related gene model could distinguish clinical features and show the reliability of our glycolysis-related gene model in CC.
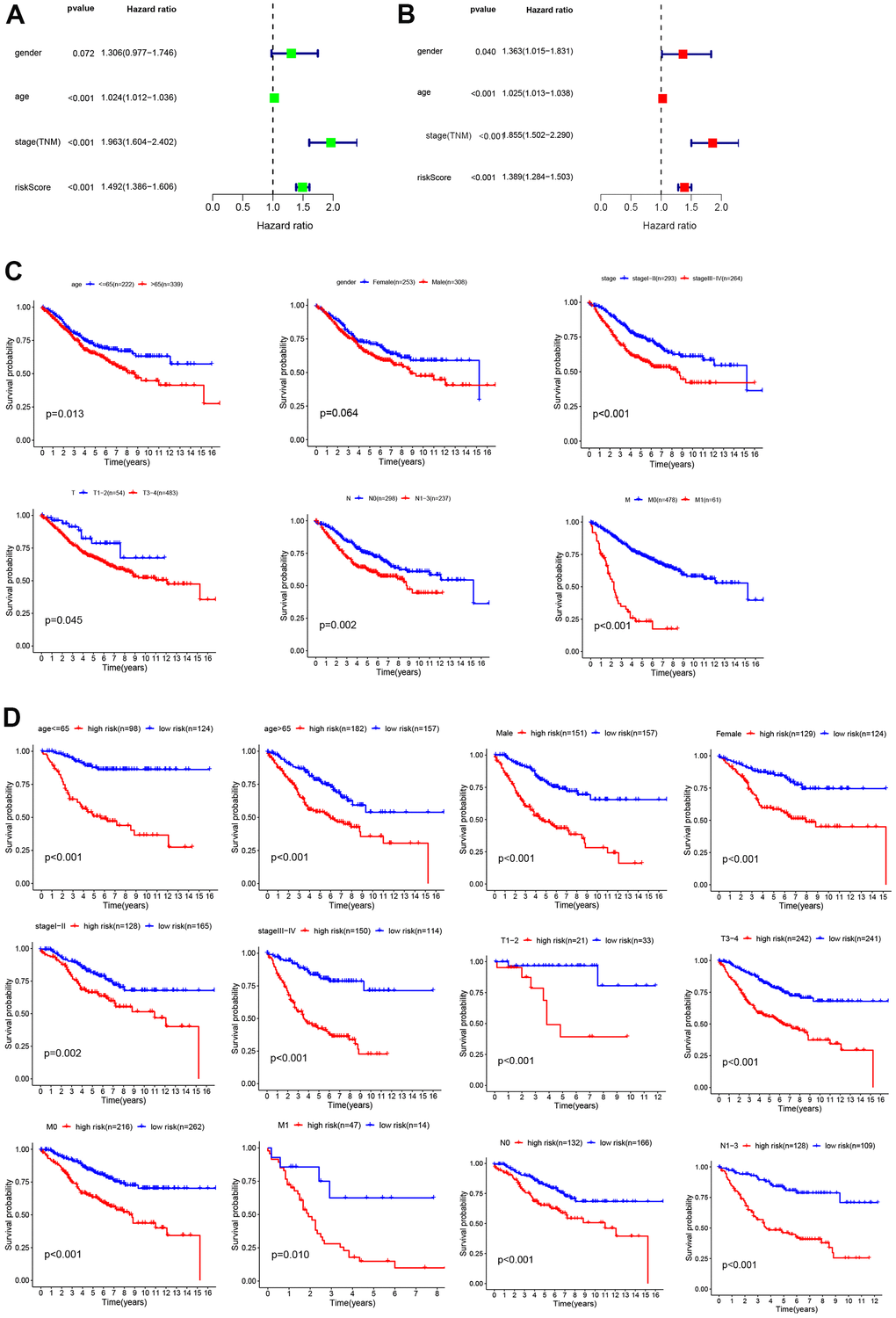
Figure 4. Analysis of risk factors. Univariate (A) and multivariate Cox regression analyses (B) of the relationship between risk core and other clinical features. (C) Kaplan–Meier analysis of clinical features, including age, sex, TNM stage, T stage, N stage, and M stage. (D) Kaplan–Meier analysis of clinical features in low- and high-risk subgroups.
Role of EMT in a high glycolysis tumor environment
Studies have demonstrated that glycolysis can promote EMT in cancers. To explore the role of EMT in CC, we analyzed the expression of EMT biomarkers (SNAI1, SNAI2, TWIST1, TWIST2, and ZEB2) in our study. SNAI1 and TWIST1 showed significantly higher expression in CC tissues (P < 0.05, Figure 5A). Moreover, SNAI1, SNAI2, TWIST1, TWIST2, and ZEB2 expression was upregulated in the high-risk group (P < 0.05, Figure 5B). This suggests that EMT plays a role in the high-glycolytic tumor environment of CC patients. The relationship between glycolysis and EMT in CC requires further research.
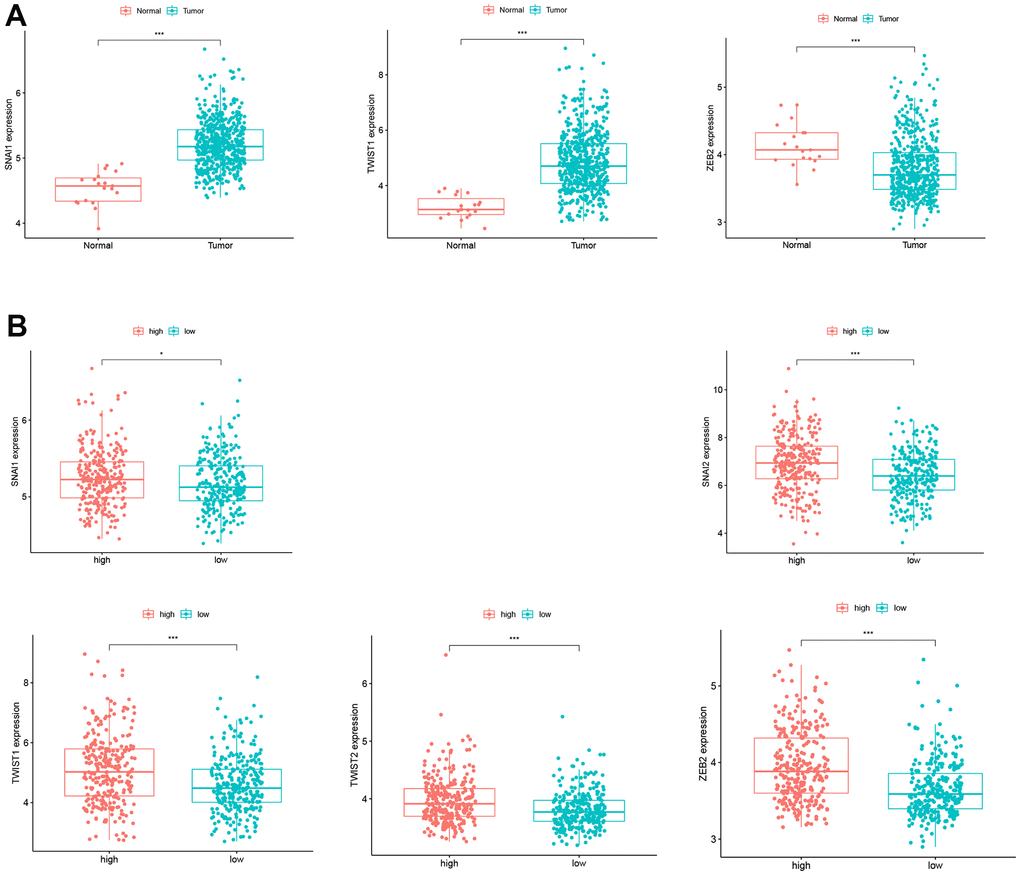
Figure 5. Expression of EMT biomarkers. (A) SNAI1 and TWIST1 expression in CC patients and normal controls. (B) SNAI1, SNAI2, TWIST1, TWIST2, and ZEB2 expression in the low- and high-risk groups.
Tumor immunity microenvironment between the low- and high-risk groups
We investigated the fractions of 22 types of infiltrated immune cells in every single cancer among all 561 CC samples (Figure 6A). The results showed that every sample had its own immune cell infiltration characteristics. The heatmap and vioplot of infiltrated immune cells were performed between the low- and high-risk groups according to the glycolysis-related gene model (Figure 6B, 6C). In low-risk group tissues, the top five most abundant fractions of infiltrating immune cells were M2 macrophages, CD8 T cells, plasma cells, follicular helper T cells, and resting memory CD4 T cells, and this was also true for the high-risk group (Figure 6B, 6C). However, no significant fractions of memory B cells, CD8 T cells, naïve CD T cells, resting CD4 memory T cells, activated CD4 memory T cells, follicular helper T cells, activated NK cells, monocytes, M0 macrophages, resting dendritic cells, activated mast cells, or eosinophils were observed in either the low- or high-risk group. (P > 0.05, Figure 6C). Naïve B cells, M1 macrophages, M2 macrophages, resting mast cells, and neutrophils were significantly higher in the high-risk group than in the low-risk group (p < 0.05, Figure 6C). Plasma cells, Tregs, gamma delta T cells, resting NK cells, and activated dendritic cells were higher in the low-risk group (p < 0.05, Figure 6C).
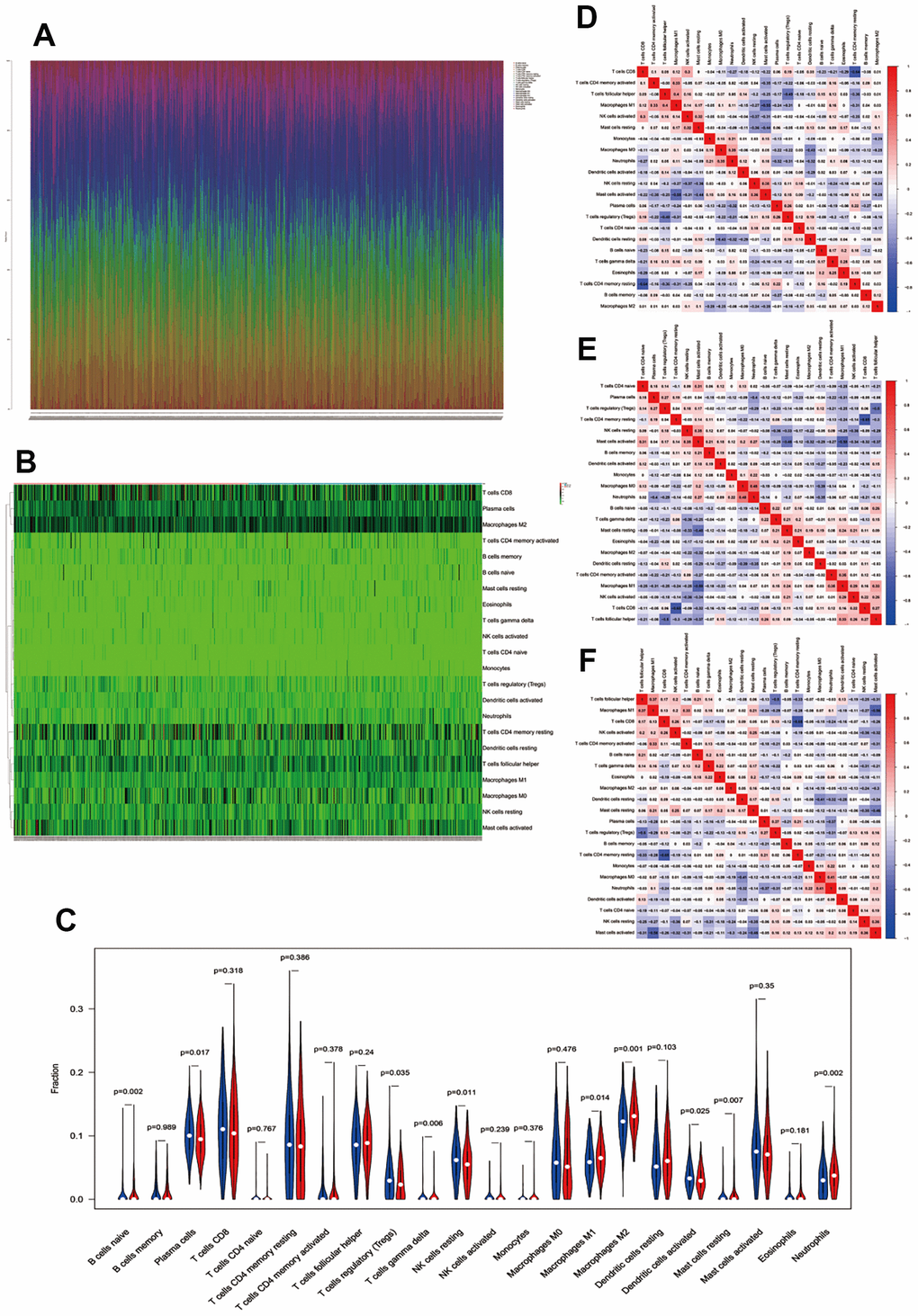
Figure 6. Immune infiltration between the low- and high-risk groups. (A) Fractions of immune cells in every single CC sample. (B, C) Heatmap and vioplot of immune cells between the low- and high-risk groups. Correlation between immune cells in low-risk samples (D), high-risk samples (E), and all colon cancer samples (F).
Finally, the correlation of immune cells in CC tissues in both the low- and high-risk groups was analyzed. In the low-risk group, a moderate to high negative correlation was observed between the T cells CD4 memory resting and T cells CD8, M1 macrophages and activated mast cells, T cells follicular helper and Tregs, activated mast cells and resting mast cells, and resting dendritic cells and M0 macrophages; however, a positive correlation was observed between the T cells follicular helper and M1 macrophages, activated T cells CD4 memory and M1 macrophages, T cells CD8 and activated NK cells, activated NK cells and resting mast cells, resting NK cells and activated mast cells, and M0 macrophages and neutrophils (Figure 6D). In the high-risk group, a moderate to high negative correlation was observed between the resting memory CD4 T cells and CD8 T cells, M1 macrophages and activated mast cells, follicular helper T cells and Tregs, resting mast cells and activated mast cells, and neutrophils and plasm cells. However, a naïve positive correlation was observed between neutrophils and M0 macrophages, resting NK cells and activated mast cells, follicular helper T cells and M1 macrophages, activated M1 macrophages and CD4 memory T cells, and activated mast cells and CD4 T cells (Figure 6E). A negative correlation was observed between resting memory CD4 T cells and CD8 T cells, M1 macrophages and activated mast cells, follicular helper T cells and Tregs, resting mast cells and activated mast cells, and resting dendritic cells and M0 macrophages. However, a positive relationship was observed in all CC patients between M1 macrophages and follicular helper T cells, M1 macrophages and activated memory CD4 T cells, activated mast cells and resting NK cells, CD8 T cells and activated NK cells, and activated NK cells and resting mast cells (Figure 6F). In the low-risk group, high-risk group, and all cancer samples, a negative correlation was observed between resting memory CD4 T cells and CD8 T cells, M1 macrophages and activated mast cells, follicular helper T cells and Tregs, and activated mast cells and resting mast cells; however, a positive correlation was observed between follicular helper T cells and M1 macrophages and activated memory CD4 T cells and M1 macrophages.
Tumor immune sensitive drug prediction
These studies have confirmed that glycolysis is closely related to colon cancer immunity. To further study colon cancer-sensitive tumor immune drugs in the glycolytic microenvironment, the top 400 genes of the absolute value of significantly differential expression multiples between the high- and low-risk groups were screened (P < 0.05, Figure 9A). GO function analysis of the differentially expressed genes suggested that the functions of the differentially expressed genes were enriched in DNA replication, rRNA metabolic process, mitochondrial matrix, mitochondrial inner membrane, catalytic activity and acting on RNA and tubulin binding, which are related to cell metabolism (P < 0.05, Figure 9B). The KEGG results demonstrated that the differentially expressed genes were enriched in metabolism-related pathways such as carbon metabolism, the cell cycle, apoptosis, DNA replication and the proteome (P < 0.05, Figure 9C). We screened tumor immune-related drugs through Dreimt [7], and the top 400 genes were input into the software.
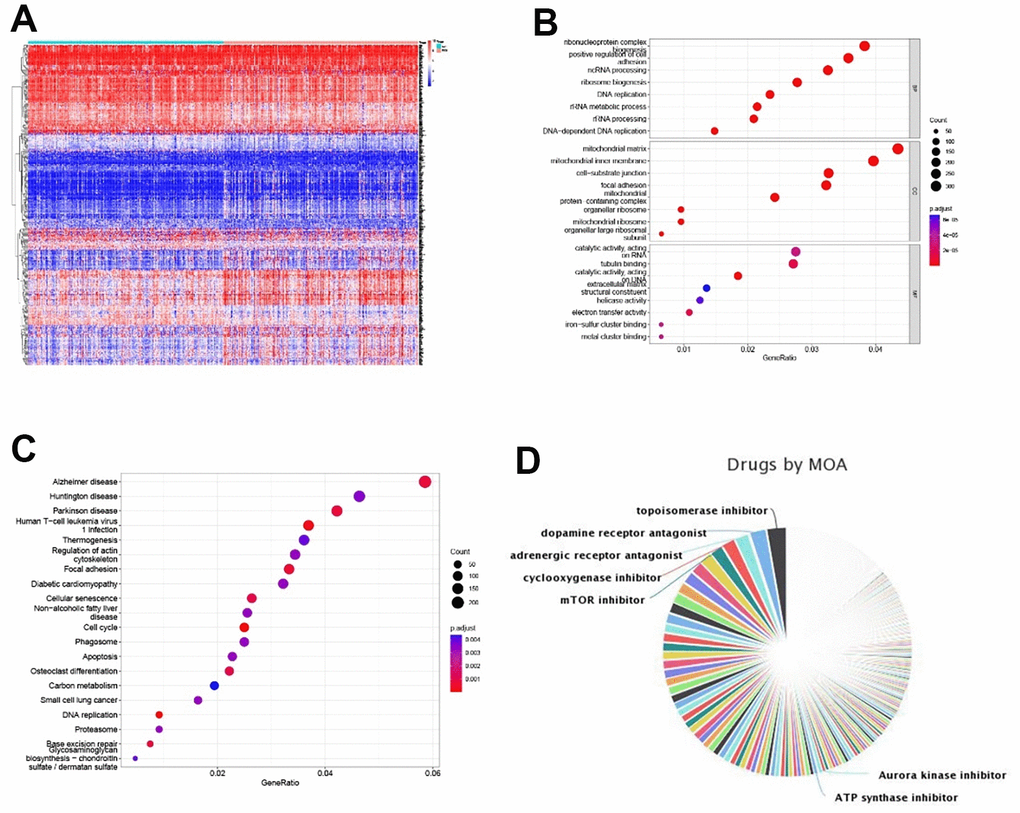
Figure 9. Tumor immune sensitive drug prediction. (A) Top 200 up- and downregulated significantly expressed genes between the high- and low-risk groups. (B, C) GO and KEGG enrichment analyses of differentially expressed genes. (D) Pie chart of tumor immune-sensitive drug types.
The results showed that the top 3 drug types were topoisomerase inhibitors, dopamine receptor antagonists and regenerative receptor antagonists (Figure 9D). The drugs were further screened (tau absolute value > 90, top 15 of drug specificity score > 0.7), and pirarubicin/mitoxantrone (topoisomerase inhibitor), daunorubicin (topoisomerase inhibitor, RNA synthesis inhibitor), BMS-387032/CGP-60474/JNJ-7706621/AT-7519/alvocidib/purvalanol-a/PHA-793887/AZD-5438 (CDK inhibitor), bisindolylmaleimide-ix (PKC inhibitor), dactinomycin RNA (polymerase inhibitor), fludarabine (ribonucleotide reductase inhibitor) and PF-562271 (focal adhesion kinase inhibitor) were filtered out. Our data provide a theoretical basis for tumor immune targeted therapy related to glycolysis in patients with colorectal cancer.
Validation of model gene expression in CC patient samples
To lay the foundation for the next step of our basic research, we validated the expression of all the model genes through western blotting. Our results showed that NUP107, SEC13, ALDH7A1, ALG1, CHPF, GALK1, XYLT2, and OGDHL were highly expressed, whereas FAM162A, FBP2, IDH1, TGFA, and VLDLR were expressed at low levels in CC patients (P < 0.05, Figure 10). These gene expression levels were consistent with the gene expression in GSE39582.
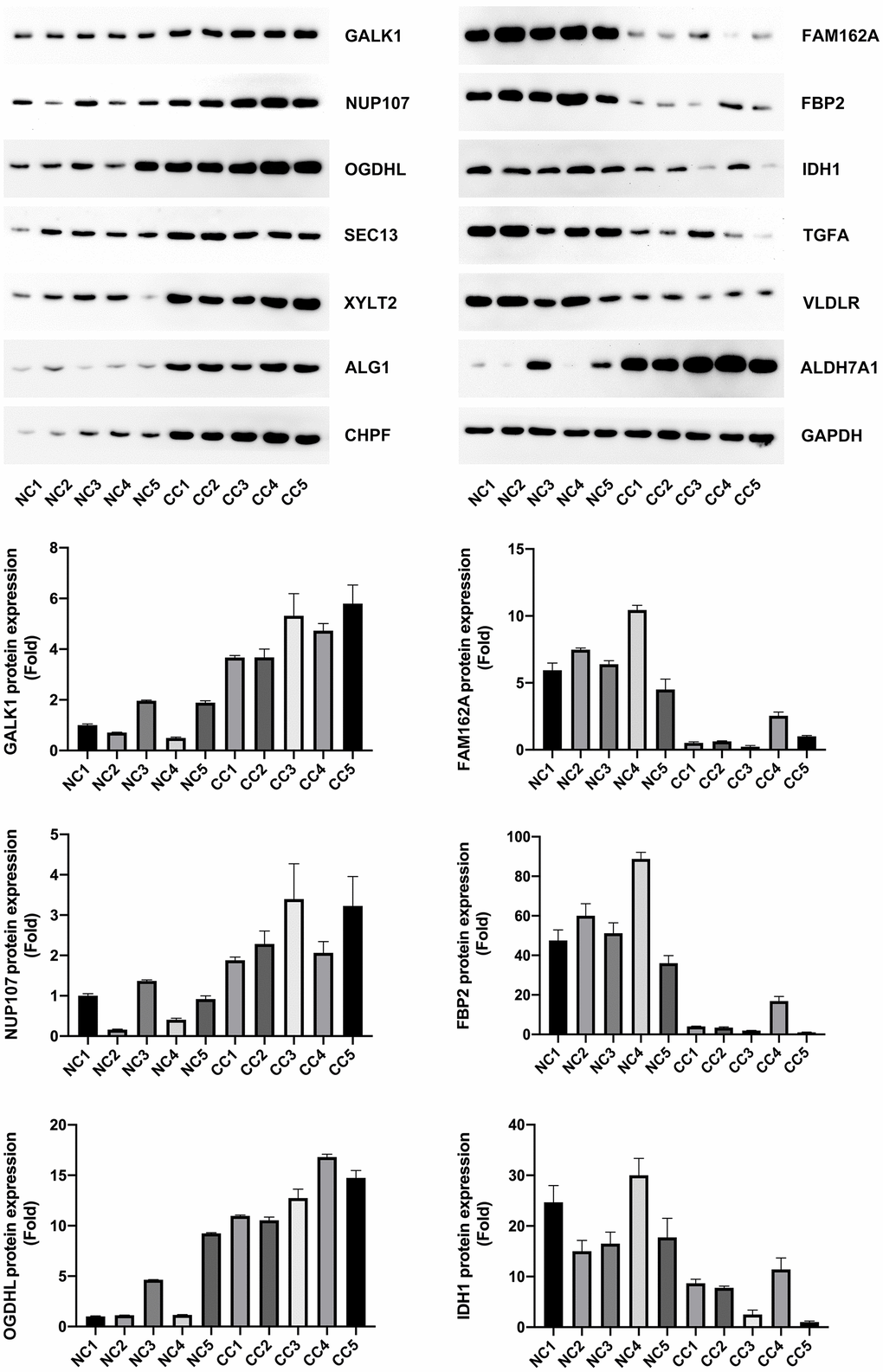
Figure 10. Model gene expression confirmed using western blotting.
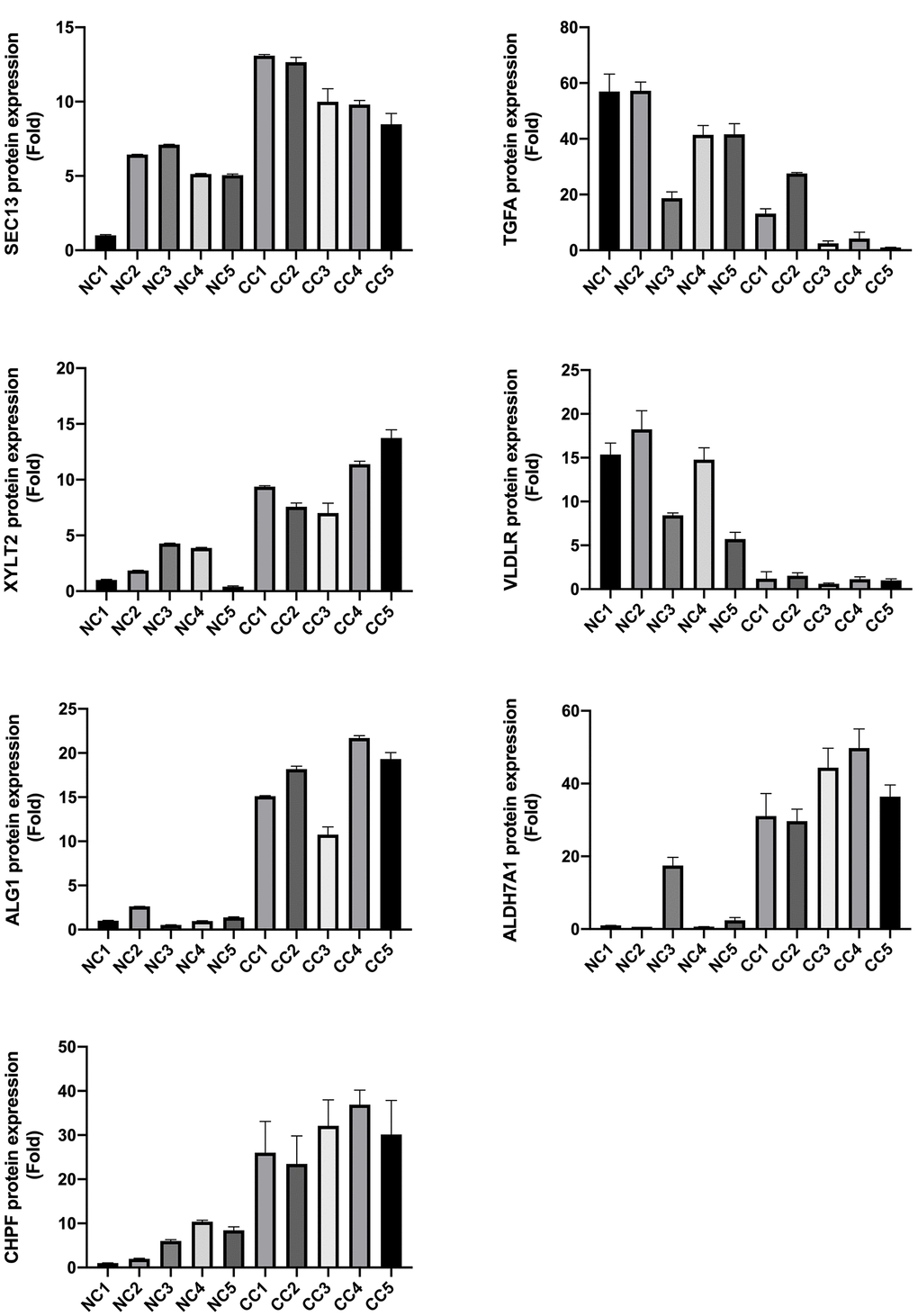
Figure 10. Model gene expression confirmed using western blotting.
Discussion
CC is a common gastrointestinal carcinoma that affects human health and was responsible for 1,148,515 new cases and 576,858 deaths globally in 2020 [8]. In recent years, there have been several advancements in the treatment of CC patients, including surgery, radiation, chemotherapy, adjuvant chemotherapy, and immunotherapy; however, the mortality of CC patients remains high [8, 9]. Thus, it is necessary to discover novel biomarkers as potential therapeutic targets to improve the prognosis of CC. Glycolysis is highly activated in tumor cells, and increasing evidence has revealed that reprogramming of glycolysis metabolism is closely related to the occurrence and development of tumors [10].
In this study, we collected six glycolysis-related gene sets from the GSEA database, from which we selected a total of 301 mRNAs in 566 CC patients for further analysis. Then, 217 differentially expressed glycolysis-related genes were screened for the construction of a prognostic gene model. After Cox regression analyses, a signature of 13 glycolysis-related genes (NUP107, SEC13, ALDH7A1, ALG1, CHPF, FAM162A, FBP2, GALK1, IDH1, TGFA, VLDLR, XYLT2, and OGDHL), all of which were independent risk factors for OS, was established between CC patient and normal tissues. This signature may be considered as a novel potential therapeutic target. Previous studies have reported that NUP37 promotes lung cancer cell proliferation and inhibits apoptosis [11], while the overexpression of TGFA and HPF induces cell proliferation and invasion in lung cancer [11, 12]. The downregulation of ALDH7A1 increases cell migration and invasiveness in hepatocellular and renal clear carcinomas [12]. Furthermore, FBP2 overexpression inhibits sarcoma cell growth [13], VLDLR overexpression inhibits CC cell proliferation and migration [14], OGDHL silencing contributes to the survival of HCC patients via the regulation of glutamine metabolic pathways [15], and IDH1 mutation promotes glioma cell proliferation and migration via EMT [16]. However, the specific roles of SEC13, ALG1, FAM162A, GALK1, and XYLT2 in cancer remain unclear.
The prediction capability of the model was assessed through PCA, Kaplan–Meier curve analysis, and ROC analysis. The clinical variables and risk scores were also analyzed. The PCA results showed that our model specifically divided samples into low- and high-risk groups based on the risk score. Kaplan–Meier curve analysis revealed that a high-risk score was related to poorer patient prognosis. The expression of the signature genes was validated via PCR and western blotting, and the results were consistent with the gene expression in GSE39582. These results suggest that our model might be useful for predicting the prognosis of patients with CC based on the risk score. Our gene expression signature might serve as a workable tool for the classification of CC patients and may provide the basis for personalized treatment.
Among the clinical variables, age > 65 years, TNM III–IV, T3-4, N1-3, M1, and a high-risk score were identified as independent prognostic risk factors. To evaluate the clinical efficacy of the model, the relationships between the risk score and clinical features were analyzed. We found that high-risk scores in the age ≤ 65 years, age> 65 years, female sex, male sex, TNM I-II, TNM III-IV, T1-2, T3-4, M0, M1, N0, and N1-3 subgroups were highly correlated with poor OS in patients. These results established the reliability of the gene model for CC. Although not all clinical features could be distinguished in the TCGA dataset, it may be because TCGA colon cancer data were not as rich as GSE39582, which was the vital reason that we used the GSE39582 dataset to construct our prediction model. Moreover, our glycolysis-related gene signatures could clearly distinguish the tumor stage and provide the basis for potential early individualized treatment.
A study reported that EMT promotes CC cell invasion and migration and is involved in the invasion and metastasis of CC [17]. However, glycolysis is closely related to EMT in cancers [5]. In this study, the risk score was positively associated with the expression of EMT biomarkers (SNAI1, SNAI2, TWIST1, TWIST2, and ZEB2). These results suggest that high glycolysis metabolism is accompanied by changes in other regulatory pathways in the tumor microenvironment.
A previous study reported differences in immune infiltration between CC and normal tissues [18]; however, the role of tumor immunity in colon cancer glycolysis remains unclear. Our data showed that M1 macrophages and plasma cells were more and less infiltrated in the high-risk group, respectively; however, Ge et al. reported that M1 macrophages and plasma cells infiltrated significantly more in CC than in normal tissues [18]. Through immune cell correlation analysis, a moderate to high negative correlation was observed between the M1 macrophages and activated mast cells and the activated mast cells and resting mast cells in the high-risk group, low-risk group, and all cancer samples. Moreover, a positive correlation was observed between follicular helper T cells and M1 macrophages and activated memory CD4 T cells and M1 macrophages, which is consistent with the results of Ge et al. in CC compared to normal tissues. According to the above results, M1 macrophages could be a potential target for therapy and may play an important role in CC and CC glycolysis; however, the specific mechanisms remain unknown and need further research. Then, tumor immune-sensitive drugs were predicted, and pirarubicin/methoxantrone (topoisomerase inhibitor), daunorubicin (topoisomerase inhibitor, RNA synthesis inhibitor), and fludarabine (ribonucleotide reductase inhibitor) were approved for use in related clinical cancer patients. Our data provide a theoretical basis for tumor immune targeted therapy related to glycolysis in patients with colorectal cancer.
In summary, we identified and validated a prognostic model based on 13 glycolysis-related genes, including NUP107, SEC13, ALDH7A1, ALG1, CHPF, FAM162A, FBP2, GALK1, IDH1, TGFA, VLDLR, XYLT2, and OGDHL, for CC. This model could be positively related to poor prognosis in CC, EMT activation, and CC microenvironment immunity. The final related tumor immune-sensitive drugs were filtered. These findings could be used for the prognosis of CC patients and identifying novel biomarkers for CC therapy.
Materials and Methods
Patient and control data
The Affymetrix Human Genome U133 Plus 2.0 array gene expression profile and corresponding clinical information data of 566 CC patients and 19 normal control tissues were downloaded from a GEO dataset (GSE39582). The clinical information included survival time, survival status, sex, age, tumor-node-metastasis (TNM) stage, clinical T stage, clinical N stage, and clinical M stage.
Independent prognostic value of risk score
To assess the prognostic value of the model, we used univariate and multivariate Cox regression analyses to determine the prognostic factors. The factors (age, sex, TNM stage, and risk score) with p values less than 0.05 in univariate and multivariate Cox regression analyses were considered as independent factors.
Clinical application of the gene model
To examine the efficiency of our model in CC, we analyzed the correlation between the gene model and clinical features (age, sex, clinical TNM stage, clinical T stage, clinical N stage, and clinical M stage). All patients were classified into two subgroups based on age (> 65 and ≤ 65 years old), sex (female and male), TNM stage (TNM stage I-II and TNM stage III-IV), clinical T stage (T1-2 and T3-4), clinical M stage (M0 and M1), and clinical N stage (N0 and N1-3). Survival analyses were performed between the two groups.
Immune cell infiltration between low- and high-risk patients
Subsequently, CIBERSORT, including 22 immune cells (seven kinds of T cells, naïve B cells, memory B cells, plasma cells, resting NK cells, activated NK cells, monocytes, M0–M2 macrophages, resting dendritic cells, activated dendritic cells, resting mast cells, activated mast cells, eosinophils, and neutrophils), was used for immune cell infiltration evaluation.
Functional enrichment analyses between low- and high-risk groups
Gene ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses were conducted using the org.Hs.e.g..db R package and clusterProfiler R package. A P value of < 0.05 was identified as statistically significant.
Model validation using western blot analysis
CC patient and control tissues were collected from the Affiliated Kunshan Hospital of Jiangsu University. The patient tissues were preserved in liquid nitrogen and lysed using RIPA lysis buffer (Strong) (MedChemExpress, China). A BCA Protein Quantification Kit (Vazyme, China) was used to measure the protein concentration. Proteins were transferred to nitrocellulose membranes (Millipore, Billerica, MA, USA) after separation through 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis. Then, the membranes were incubated with primary antibodies against the following: NUP107/SEC13/ALDH7A1/ALG1/CHPF/FAM162A/FBP2/GALK1/IDH1/TGFA/VLDLR/XYLT2/OGDHL (Abcam, USA, 1:1000) and GAPDH (Abcam, USA, 1:3000). The membranes were washed and incubated with horseradish peroxidase-labeled goat anti-rabbit IgG (1:5000; Sigma). The signals were visualized using BIO-RADXR.
Statistical analysis
Statistical analysis was performed using SPSS 21.0 software (SPSS, Chicago, IL, USA). Data were presented as the means ± standard deviation (SD). The difference between the groups was determined using ANOVA with repeated measures. To compare the difference between the two groups, the independent sample t test was used. P<0.05 was considered as significant.
Author Contributions
G.L. and J.C. conceived and designed the study. X.W. collected the data. G.L. analyzed the results. G.L. and J.C. drafted the manuscript. All authors participated in the discussion of results and reviewed the manuscript.
Conflicts of Interest
The authors declare that there is no conflicts of interest.
Ethical Statement
The tissue collection was approved by Affiliated Kunshan Hospital of Jiangsu University.
Funding
This work was supported by General Surgery Clinical Basis and Innovation Team (CXTD21-C06).
References
- 1. Harada S, Morlote D. Molecular Pathology of Colorectal Cancer. Adv Anat Pathol. 2020; 27:20–6. https://doi.org/10.1097/PAP.0000000000000247 [PubMed]
- 2. Mumprecht V, Detmar M. Lymphangiogenesis and cancer metastasis. J Cell Mol Med. 2009; 13:1405–16. https://doi.org/10.1111/j.1582-4934.2009.00834.x [PubMed]
- 3. Kaminski MF, Robertson DJ, Senore C, Rex DK. Optimizing the Quality of Colorectal Cancer Screening Worldwide. Gastroenterology. 2020; 158:404–17. https://doi.org/10.1053/j.gastro.2019.11.026 [PubMed]
- 4. Abbaszadeh Z, Çeşmeli S, Biray Avcı Ç. Crucial players in glycolysis: Cancer progress. Gene. 2020; 726:144158. https://doi.org/10.1016/j.gene.2019.144158 [PubMed]
- 5. Georgakopoulos-Soares I, Chartoumpekis DV, Kyriazopoulou V, Zaravinos A. EMT Factors and Metabolic Pathways in Cancer. Front Oncol. 2020; 10:499. https://doi.org/10.3389/fonc.2020.00499 [PubMed]
- 6. Zhao H, Duan Q, Zhang Z, Li H, Wu H, Shen Q, Wang C, Yin T. Up-regulation of glycolysis promotes the stemness and EMT phenotypes in gemcitabine-resistant pancreatic cancer cells. J Cell Mol Med. 2017; 21:2055–67. https://doi.org/10.1111/jcmm.13126 [PubMed]
- 7. Troulé K, López-Fernández H, García-Martín S, Reboiro-Jato M, Carretero-Puche C, Martorell-Marugán J, Martín-Serrano G, Carmona-Sáez P, Glez-Peña D, Al-Shahrour F, Gómez-López G. DREIMT: a drug repositioning database and prioritization tool for immunomodulation. Bioinformatics. 2021; 37:578–9. https://doi.org/10.1093/bioinformatics/btaa727 [PubMed]
- 8. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 71:209–49. https://doi.org/10.3322/caac.21660 [PubMed]
- 9. Labianca R, Beretta GD, Kildani B, Milesi L, Merlin F, Mosconi S, Pessi MA, Prochilo T, Quadri A, Gatta G, de Braud F, Wils J. Colon cancer. Crit Rev Oncol Hematol. 2010; 74:106–33. https://doi.org/10.1016/j.critrevonc.2010.01.010 [PubMed]
- 10. Pavlova NN, Thompson CB. The Emerging Hallmarks of Cancer Metabolism. Cell Metab. 2016; 23:27–47. https://doi.org/10.1016/j.cmet.2015.12.006 [PubMed]
- 11. Huang L, Wang T, Wang F, Hu X, Zhan G, Jin X, Zhang L, Li Y. NUP37 silencing induces inhibition of cell proliferation, G1 phase cell cycle arrest and apoptosis in non-small cell lung cancer cells. Pathol Res Pract. 2020; 216:152836. https://doi.org/10.1016/j.prp.2020.152836 [PubMed]
- 12. Andrejeva D, Kugler JM, Nguyen HT, Malmendal A, Holm ML, Toft BG, Loya AC, Cohen SM. Metabolic control of PPAR activity by aldehyde dehydrogenase regulates invasive cell behavior and predicts survival in hepatocellular and renal clear cell carcinoma. BMC Cancer. 2018; 18:1180. https://doi.org/10.1186/s12885-018-5061-7 [PubMed]
- 13. Huangyang P, Li F, Lee P, Nissim I, Weljie AM, Mancuso A, Li B, Keith B, Yoon SS, Simon MC. Fructose-1,6-Bisphosphatase 2 Inhibits Sarcoma Progression by Restraining Mitochondrial Biogenesis. Cell Metab. 2020; 31:1032. https://doi.org/10.1016/j.cmet.2020.04.009 [PubMed]
- 14. Kim BK, Yoo HI, Lee AR, Choi K, Yoon SK. Decreased expression of VLDLR is inversely correlated with miR-200c in human colorectal cancer. Mol Carcinog. 2017; 56:1620–9. https://doi.org/10.1002/mc.22618 [PubMed]
- 15. Dai W, Xu L, Yu X, Zhang G, Guo H, Liu H, Song G, Weng S, Dong L, Zhu J, Liu T, Guo C, Shen X. OGDHL silencing promotes hepatocellular carcinoma by reprogramming glutamine metabolism. J Hepatol. 2020; 72:909–23. https://doi.org/10.1016/j.jhep.2019.12.015 [PubMed]
- 16. Lu J, Li D, Zeng Y, Wang H, Feng W, Qi S, Yu L. IDH1 mutation promotes proliferation and migration of glioma cells via EMT induction. J BUON. 2019; 24:2461–7. [PubMed]
- 17. Todosi AM, Gavrilescu MM, Aniţei GM, Filip B, Scripcariu V. Colon cancer at the molecular level--usefulness of epithelial-mesenchymal transition analysis. Rev Med Chir Soc Med Nat Iasi. 2012; 116:1106–11. [PubMed]
- 18. Ge P, Wang W, Li L, Zhang G, Gao Z, Tang Z, Dang X, Wu Y. Profiles of immune cell infiltration and immune-related genes in the tumor microenvironment of colorectal cancer. Biomed Pharmacother. 2019; 118:109228. https://doi.org/10.1016/j.biopha.2019.109228 [PubMed]