Introduction
Skin aging is a genetically-programmed physiological process, dependent on both environment and genetic background [1, 2]. Two combined aging processes co-exist: extrinsic and intrinsic [3, 4]. Extrinsic aging results from exposure to several external factors such as environmental pollution or ultraviolet (UV) radiation among others [5–9], the “exposome” defining all the external exposures to which an individual is subjected from conception to death [8]. Concerning environmental factors, UV contributes to extrinsic skin aging, especially premature aging, up to 80% [9], with several characteristics, including wrinkles and elasticity loss, that are linked to the alteration of extracellular matrix (ECM) components [10, 11]. In contrast, intrinsic skin aging concerns genetic factors, is linked to senescence, [12] and changes in the endocrine environment that reflect the processes of degradation of the whole organism [13, 14].
Dermal fibroblasts dispersed within the dermis are the main actors of sustained ECM renewal by producing collagen and elastin in particular [15, 16]. In case of a cutaneous lesion, fibroblasts secrete matrix metalloproteinases (MMP) that are endopeptidases able to degrade the damaged matrix, including collagenases such as MMP-1 or stromelysins like MMP3, and that are regulated by the specific endogenous tissue inhibitors of metalloproteinases (TIMPs). Fibroblasts will then synthesize a new remodeling matrix and release cytokines to attract macrophages to encompass and degrade debris. They secrete growth factors to stimulate keratinocyte proliferation and migration, leading to complete wound healing [17, 18]. This is the case of young healthy skin, which exhibits as major morphological features, a thick connective tissue maintained by an effective ECM renewal, a well-stratified epidermis, and an effective wound healing process [19, 20]. In contrast, “aged” skin should be sheerer, with a flat epidermis and a loss of ECM leading to a thinner dermis [21, 22]. The ECM morphological changes of the dermis have been shown to be largely responsible for the loss of elasticity and wrinkles as major hallmarks of aging skin [23]. Indeed, dermal fibroblasts undergo physiological changes with age [18, 24], including a permanent secretion of a secretory pole of factors denoted ‘skin aging–associated secreted proteins’ (SAASP) [11], in contrast to young ones which only transiently secrete such factors, for instance during wound healing [25]. Moreover, the decrease in the number of dermal fibroblasts [19] associated with the alteration of the ECM production and the decrease of their migratory capacity with aging [26], as shown recently in a study of comprehensive proteomic database of soluble proteins and exosomal cargo ‘senescence-associated secretory phenotype’ (SASP) factors originating from multiple senescence inducers and cell types including fibroblasts [27] might be involved in the perturbation of wound healing processes observed in the elderly population [3]. The SASP consists of a myriad of cytokines, including chemokines (CXCLs) and growth factors, as well as proteases that all together initiate inflammation, wound healing, and growth responses in nearby cells.
This study aimed to improve knowledge about age-associated molecular alterations in primary cultured fibroblasts from donors older than 55 years (“old fibroblasts”) compared with fibroblasts from donors of less than 35 years (“young fibroblasts”), either in quiescence condition (absence of serum) or under treatment with transforming growth factor-β1 (TGF-β1) well-known as a major factor for stimulation of wound healing and ECM production by fibroblasts [27]. The secretome and proteome from both aged and young fibroblasts were analyzed by High Pressure Liquid Chromatography/Mass Spectrometry (HPLC/MS). Our comparative analyses of the secretome from young and old fibroblasts revealed a significantly altered expression of less than 75 ones in either quiescence or TGF-β1-stimulated condition (fold change>2, p-value<0.05). The proteome comparison depicted a significant decrease in fibroblast protein secretion with age and conversely an enhancement of more than 60% of protein cytoplasmic accumulation. This enables us to define the specific role in skin aging of some differentially-expressed proteins, especially those linked with the actin cytoskeleton dynamics. The functions of these proteins were also more deeply analyzed by collagen contraction or cell migration using siRNA transfections.
Materials and Methods
Fibroblast culture
Dermal fibroblasts were obtained from healthy sun-protected skin of either young (<35 years-old) or aged (>55 years-old) healthy women undergoing breast reduction mammoplasty in our Department of Plastic Surgery, Saint-Louis Hospital. Dermal fibroblasts were cultured as previously described [18] in Roswell Park Memorial Institute 1640 (RPMI) supplemented with 10% foetal bovine serum (FBS), 1% penicillin/streptomycin, 2 mM L-Glutamine, 25 μg/mL amphotericin B and 5 μg/mL of plasmocin (complete RPMI medium) and were used at the 3rd passage to avoid any exhaustion-replicative senescence [11, 28].
Sample preparation for secretome and proteomic assay
Fibroblasts were either stimulated or not stimulated with TGF-β1 for 24h. Then supernatants were centrifuged (400g, 8 min, 4°C) to remove pellets, before addition of protease inhibitors (Sigma-Aldrich, USA; P8340). Adherent fibroblasts were washed, scraped with a spatula, and centrifuged (400g, 8 min, 4°C). Pellets were lysed with 80 μL RIPA buffer (Rockland, USA; MB-030-0050) including protease inhibitors (Sigma-Aldrich; P8340). Protein measurement was performed by Bradford assay (Bio-Rad, USA; #500-0006) and cell lysates were kept at -80°C until they underwent direct digestion by trypsin before analysis at Jacques Monod Institute (JMI, Paris, France).
Sample preparation prior to LC-MS/MS analysis
50 μg of proteins were precipitated with acetone overnight at -20°C. Samples were centrifuged at 11,000 rpm and 4°C, the pellets were resuspended with 100 μL of NH4HCO3 25mM, then heated at 95°C for 10 minutes. The samples were digested overnight at 37°C with 0.4μg of trypsin (Promega, Madison, WI, USA) in a 25 mM NH4HCO3 buffer per sample. Then peptides were desalted using ZipTip μ-C18 Pipette Tips (Pierce Biotechnology, Rockford, IL, USA).
LC-MS/MS acquisition
MS grade Acetonitrile (ACN), MS grade H2O and MS grade formic acid (FA) were obtained from Thermo Chemical (Waltham, MA, USA). Samples were analyzed by a Q-Exactive Plus coupled to a Nano-LC Proxeon 1000 (Thermo Fisher Scientific, Waltham, MA, USA).
Peptides were loaded with an online preconcentration method and separated by chromatography using a Pepmap-RSLC C18 column (0.75 x 500 mm, 2 μm, 100 Å) from Thermo Fisher Scientific, equilibrated at 50°C and operating at a flow rate of 300 nl/min. Peptides were eluted by a gradient of solvent A (H2O, 0.1 % FA) and solvent B (ACN, 0.1% FA), the column was first equilibrated for 5 min with 95 % of A, then B was raised to 35% in 98 min. Finally, the column was washed with 80% B for 20 min and re-equilibrated at 95% A giving a total run time of 120 min. Peptide masses were analyzed in the Orbitrap cell in full ion scan mode, at a resolution of 70,000, a mass range of m/z 375-1500 and an AGC target of 3×106. MS/MS were performed in the top 20 mode. Peptides were selected for fragmentation by Higher-energy C-trap Dissociation (HCD) with a Normalized Collisional Energy of 27% and a dynamic exclusion of 60 seconds. Fragment masses were measured in the Orbitrap, at a resolution of 17,500, with an AGC target of 2×105, an isolation window of 1.4 Da and a mass range of m/z 200-2000. Monocharged peptides and unassigned charge states were excluded from the MS/MS acquisition. The maximum ion accumulation times were set to 50 ms for MS and 45 ms for MS/MS acquisitions respectively.
Data analysis
Label-free quantification was done on Progenesis QI for Proteomics (Waters, Milford, MA, USA). Between runs, alignment of peptide features and normalization based on all peptide ions were both performed by the Progenesis software. An MGF peak file exported from Progenesis was processed on Proteome Discoverer 2.1 with the mascot node (Mascot version 2.5.1). MS/MS spectra were searched against the Swissprot protein database release 2017_04 with the Homo sapiens taxonomy and a maximum of 2 missed cleavages. Precursor and fragment mass tolerances were set to 10 ppm and 0.02 Da respectively. The following post-translational modifications were included as variables: Oxidation (M), acetyl (N-term), phosphor (STY). Spectra were filtered using a 1% FDR with the percolator node. Multivariate statistics on protein measurements were performed using the Progenesis software and protein abundance was inferred using the top 3 method. A two-group comparison parametric t-test was used to determine differential proteins between the following groups: quiescent young versus quiescent old and TGFβ-stimulated young versus TGFβ-stimulated old cells. Proteins expressed in significant different ways were determined with fold>2, p-value<0.05. A p-value better than 0.05 was used to filter differential significant candidates.
The expression-based heat maps were done using Heatmapper website, following these characteristics: scale type (select direction to scale values) was “row” type, clustering methods (group the data by similar expression levels) was “average Linkage”, and the distance measurement method was “Eucledian”. The schematic molecular pathways were done using the “PathVisio” Software®, after GOrilla and Panthere website analysis.
Molecular biology
RNA extraction, reverse transcription, and quantitative PCR were performed as previously described [17] and primer sequences were purchased from Eurogentec (Seraing, Belgium).
Western blot (WB)
Fibroblasts were lysed in Tris-Triton lysis buffer supplemented with protease inhibitor cocktail (Sigma-Aldrich; P8340) and centrifuged (20800g, 15 min, 4°C) before protein concentration determination by Bradford assay. Migration of 50 μg of denatured proteins was performed in 10% Bolt Bis-Tris Plus polyacrylamide gel containing sodium dodecyl sulfate, followed by transfer to a nitrocellulose membrane using horizontal gel transfer device iBlot™ 2. Membranes were blocked with TBS1X-5% nonfat milk-0.1% Tween for 1h and incubated overnight at 4°C with specific primary antibodies (Abcam, UK): CORO1C (ab15719, 1/500); CFL1 (ab11062, 1/10000); ARP2/3 (ab49671, 1/2200); FLNB (ab97457, 1/500); ACTC1 (66125-lg, 1/5000). Secondary antibodies (anti-mouse, Bio-Rad 70-65k, 1/2000 or anti-rabbit, ECL-NA1934VS, 1/1000) coupled to horseradish peroxidase were incubated for 1h. Signal was detected using an enhanced chemiluminescence detection kit (Bio-Rad; #170-5061) and ImageQuant chemiluminescent. Blot quantification was performed using ImageJ software.
| Gene name | Sequence | |
| Forward | Reverse | |
| β2-microglobulin (β2M) | 5’-TGCTGTCTCCATGTTTGATGTATCT-3’ | 5’-TCTCTGCTCCCCACCTCTAAGT-3’ |
| Coronin 1C (CORO1C) | 5’-AGGAGCAAGACCCATGAGAG-3’ | 5’-TTGGTTCCTGCATATTTTTCG-3’ |
| Cofilin 1 (CFL1) | 5’-AGCCTGCTGGAACCATCTT-3’ | 5’-ACATCTATGGCCGATGTGG-3’ |
| Filamin B (FLNB) | 5’-AACAGCCCCTTCACTGTCAT-3’ | 5’- CATTTACCGGTGCCTCCTC-3’ |
| Actin Alpha Cardiac Muscle 1 (ACTC1) | 5’- AACCTGGTATTGCTGATCGT-3’ | 5’- GCTCAGGGGGAGCAATAATC-3’ |
Immunofluorescence
Immunostaining was performed on fibroblasts seeded on culture chamber slides. Briefly, fibroblasts were seeded at 3500 cells/well on chamber slide, deprived in 0.5% FBS-containing RPMI medium overnight and then activated with 5ng/ml of TGF-β for 24 hours. Fibroblasts were fixed with 4% paraformaldehyde, then permeabilized with Triton X100-0.1%. Staining was performed by overnight incubation with primary antibodies: CORO1C (ab15719, 1/200); CFL1 (SAB2702206, 1/200); FLNB (ab97457, 1/100); ACTC1 (66125-lg, 1/50), followed by an incubation with AF594 secondary antibodies (anti-mouse, A-11001, 1/2000 or anti-goat, A-11058, 1/1000). Imaging was assessed using an Axiovert 200M-fluo microscope and quantification was processed on ImageJ analysis software.
Wound closure by collective migration
The IncuCyte®S3 Live-Cell Analysis System was used for quantitative label-free collective migration measurements over a 5 day-long lasting culture. Fibroblasts were seeded at 10000 cells per well into 96-well plate to obtain a whole confluency after 24h of culture in complete medium. For siRNA transfection, fibroblasts were transfected with Dharmafect-siRNA mixtures for 5h before cell scratching. For inhibiting the part of cell proliferation during fibroblast migration, mitomycin C (10 μg/ml) was added for 2h before cell scratching. Plates were scratched with a semi-automatized scratch system, creating reproductive regular wounds in each well. Cells were washed to remove cellular debris and complete RPMI medium was added. Plates were placed into the IncuCyte ZOOM® system for scanning every hour until the scratch wound was completely closed. Captured images were analysed using the integrated software by calculating the cell confluence in the wound.
Collagen gel (lattice) contraction assay
Fibroblasts (100000 cells/mL) were added to a solution containing Type I Collagen (Corning™), 0.006N acetic acid, 0.005N NaOH and incubated in a 6-well plate for 1 h at 37°C to induce gelation. Two lattices were made for each condition. Dermal collagen lattices were detached allowing floatation. OPTIMEM 1% FBS was added at 2X concentration and pictures were taken daily for one week. The lattice area was measured twice by ImageJ.
Transfection assay
Fibroblast siRNA transfection was carried out using Lipofectamine 2000 Transfection Reagent (Life Technologies, USA) according to the manufacturer’s protocol. Briefly, Lipofectamine was diluted in OPTIMEM medium (1/25) as well as the siRNA stock solution (1/50) and incubated for 5 min at RT. Lipofectamine mix was added to the siRNA mix and incubated for 20 min at RT. The resultant mix was then diluted with OPTIMEM to reach a final concentration of 100 nM of siRNA. Cells were transfected with 100 nM of siRNA targeting p16 (Thermo Fisher Scientific) for 24h before RNA and protein extraction. As control siRNA (SiControl), an irrelevant siRNA sequence pool (Thermo Fisher Scientific) was used.
Statistical analysis
Results were expressed as mean ± SEM from independent experiments. Data were symmetrically distributed in a normal distribution. One Way ANOVA Multiple comparisons or Wilcoxon Rank-Sum test were performed using Prism® (GraphPad 9.1.1) analysis. Differences were considered as significant at *: p<0.05; **: p<0.01; ***: p<0.001.
Results
Secretome study reveals a decrease in protein secretion with age
Senescent cells—and more importantly SASP, the factors they secrete—are widely accepted as drivers of skin aging and multiple age-related cutaneous pathologies. In the present analysis of fibroblast secretome, a total of 945 different proteins between young and old cells was detected in quiescence condition with a significant differential expression of only 16 proteins according to the chosen criteria of selection: fold change>2, p-value<0.05. Under TGF-β1 addition, 944 different proteins between young and old fibroblasts were identified by HPLC with a significant differential expression of 11 proteins (fold change>2, p-value<0.05). The secretome expression-based heat map (Figure 1A) showed that the differentially-secreted proteins were highly expressed in young fibroblasts compared to those from old ones in either quiescent (13/16) or TGF-β1-stimulated (8/13) conditions. The role and function of each identified protein was deeply investigated by a global literature review leading to a general secretory pattern associated with skin aging and correlated with cellular pathways (Figure 1B). We underlined that 19% of the proteins differentially-expressed in quiescence condition between young and old fibroblasts are associated with either actin structure, ECM organization, or enzymatic function, whereas other biological processes including proliferation, inflammation, metabolism, or senescence represent less than 8% each. TGF-β1-treatment induced alterations mainly related to enzymatic function (19%), proliferation/senescence (18%), or actin and ECM organization for 9% each (Figure 1B). A schematic representation of proteins linked to a specific pathway was obtained for quiescence and activation conditions (Figure 1C, 1D, respectively). Noteworthy, the organization of actin and ECM structures appeared to be the major categories linked to the differentially-expressed proteins in both secretome and proteome.
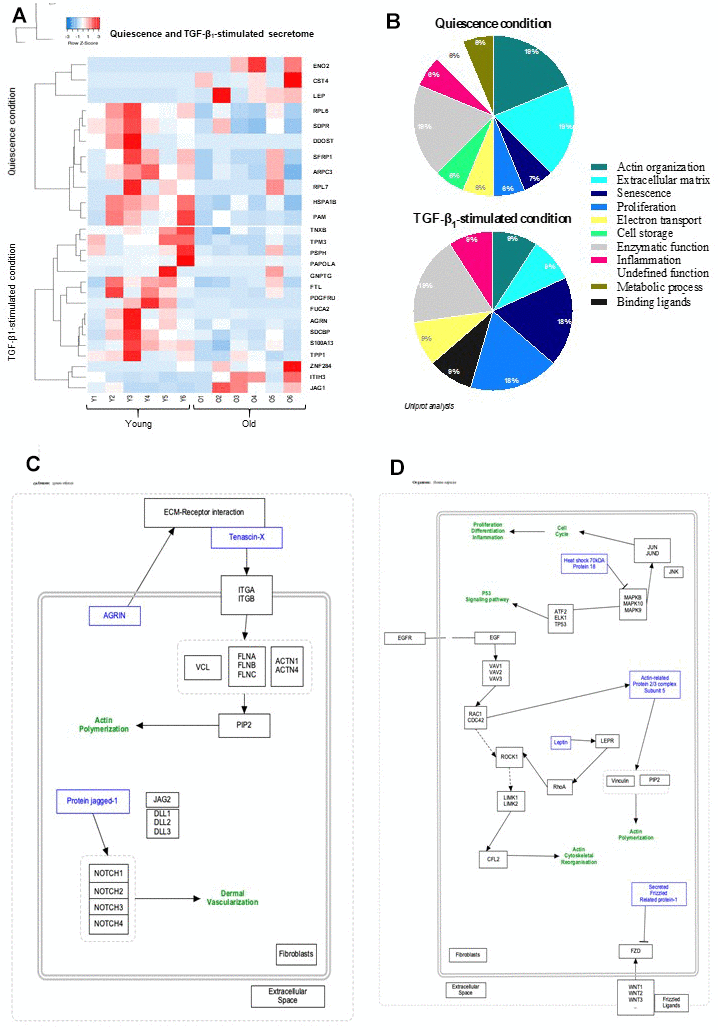
Figure 1. Quiescence and TGF-β1-stimulated SECRETOME heat-map and schematic representations of molecular pathways. (A) Heat-maps of differentially-expressed proteins in young and old fibroblasts (n=6) (HeatMapper). (B) Analysis of cellular processes linked to differentially-expressed proteins (GOrilla and Panthere). (C) Representation of signaling pathways and links between differently expressed proteins in quiescence and (D) TGF-β1-treated condition (PathVisio).
Proteome study revealed a gain in protein production with age
Proteomic study identified a total number of 4169 proteins between quiescent young and old fibroblasts in quiescence condition from which only 63 proteins were statistically differentially-expressed (fold change>2, p-value<0.05). Under TGF-β1 treatment, the young versus old fibroblast comparison of proteomes revealed a difference of 4169 proteins from which only 73 were retained as differentially-expressed according to the criteria of fold change>2 and p-value<0.05. As revealed by the proteome expression-based heat map (Figure 2A) most of these proteins were detected in old fibroblasts rather than in young ones, either in quiescent (34/63) or TGF-β1-treatment (56/73) conditions. Similar to the secretome comparison, actin organization/cytoskeleton function represents 27% of the 63 differentially-expressed proteins in old fibroblasts (Figure 2B) and the same fields were detected under TGF-β1-treated condition: actin with a 18% rate and ECM organization for 8%, although a 23% group of proteins with undefined functions were detected in old fibroblasts (Figure 2B). The schematic pathway representations (Figure 2C, 2D) reported that most of the proteins were linked to actin-cytoskeleton regulation, inflammation (due to the TGF-β1 treatment), and ECM maintenance.
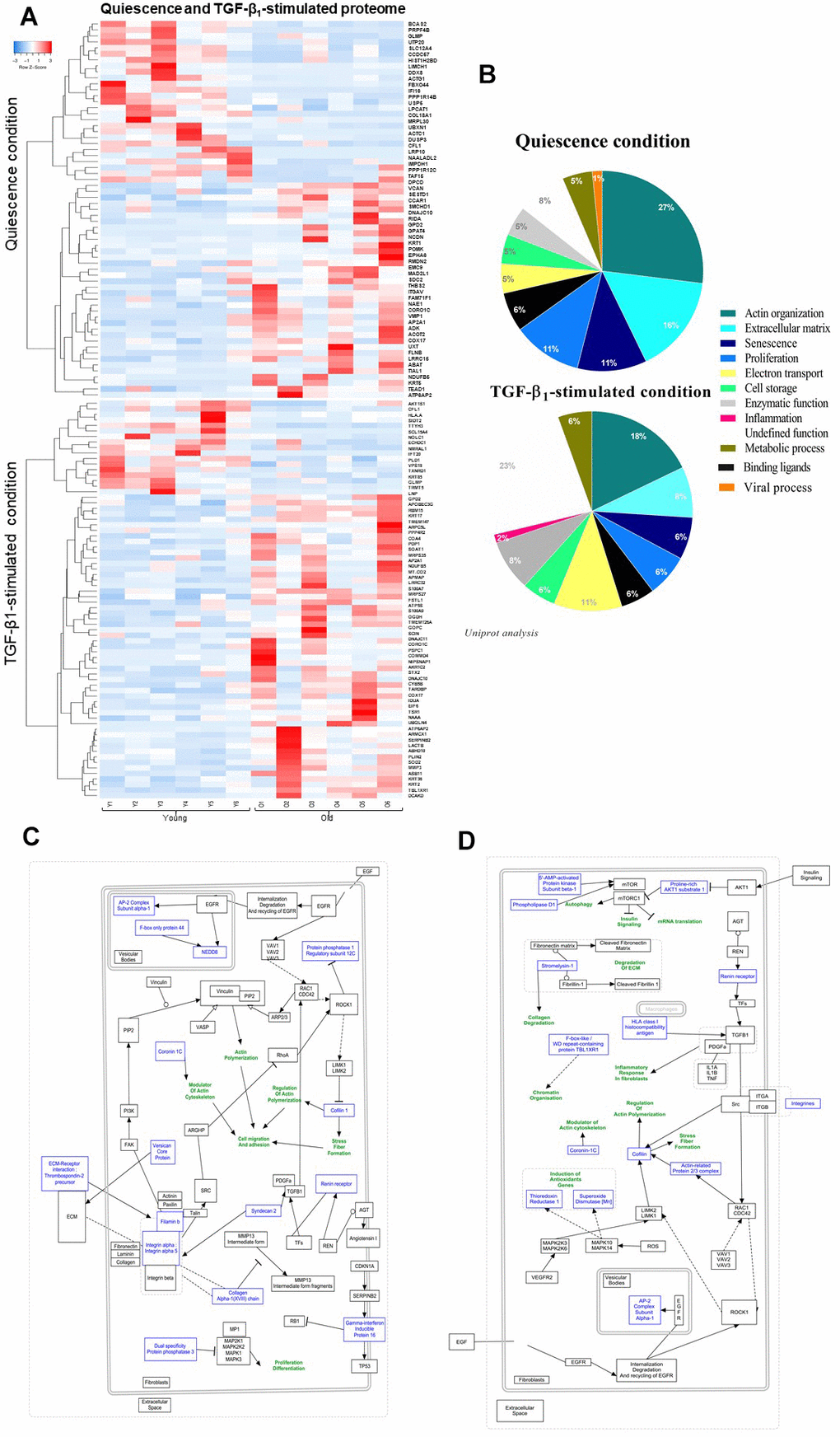
Figure 2. Quiescence and TGF-β1-stimulated PROTEOME heat-map and schematic representations of molecular pathways. (A) Heat-maps of differentially-expressed proteins in young and old fibroblasts (n=6) (HeatMapper). (B) Analysis of cellular processes linked to differentially-expressed proteins (GOrilla and Panthere). (C) Representation of signaling pathways and links between differentially-expressed proteins in quiescence and (D) TGF-β1-treated condition (PathVisio).
Discussion
To identify relevant molecules involved in the main role of human dermal fibroblasts during the process of wound healing and skin aging, a comparative analysis of the secretome and proteome of 12 cultures of dermal fibroblasts, freshly-isolated from young and mature skin, was carried out by HPLC/MS in either quiescence or TGF-β1-treated condition, in the absence of senescence by specific inducers, as described in other previously reported aging model systems [11, 27]. It must be noted that the analyses were assessed in the absence of serum in the culture medium, 24h before and during cell stimulation in order to avoid any serum protein contamination for these secretomic and proteomic assays.
Secretome comparison between young and old fibroblasts showed that more than 70% of the differentially-secreted proteins were significantly downregulated in old fibroblasts, and the main proportion of these secreted proteins was linked to the actin cytoskeleton in both quiescence and TGF-β1-activated condition, suggesting that aging brings a decrease in fibroblast communication and migration within the environment. Other differentially-secreted proteins belong to several biological processes for less than 8% each, including inflammation, electron transport, cell storage, metabolism or senescence. This is in accordance with previous data about aging, reporting that cells become senescent and exert numerous effects through their secretory products known as SAASP [11, 39], and that this could be involved in wound healing alteration during skin aging [40]. Our data are in accordance with the observation obtained from the “SASP Atlas” published by Basisty et al. (2020) as a comprehensive proteomic database of soluble proteins originating from multiple senescence inducers and cell types, showing that the largest pathway associated with all inducers related to tissue and cell structure, mainly include extracellular matrix organization, actin cytoskeleton, integrin interactions, and peptidase regulation.
In the proteome analysis contrary to the secretome one, more than half of the differentially-expressed proteins were upregulated in old cells, but again, almost 25% of altered proteins exhibited a major link to the actin organization/cytoskeleton in either quiescence or activated conditions. As actin organization in fibroblasts has been shown to be substantial during aging [41], our results suggest that actin-related proteins should favor actin-cytoskeleton binding in aged fibroblasts, leading to their rigidification and loss of intracytoplasmic structural organization with consequent morphological changes and consequent alteration in cell migration and wound healing resolution. This might induce a loss of active contractile actin-stress fibers, resulting in nuclear deformation and physical disruption of the cells that could limit their contractibility potential and cause further contraction of the wound merges by fibroblasts.
This might also disturb the production or degradation of the ECM components which will also limit wound healing, as suggested by [42]. This will alter the structure of the dermis composition in links with ECM modification by fibroblasts, as previously reported [43] and supported by our present functional results. Indeed, the second group of upregulated proteins (around 10%) in old fibroblasts was linked to ECM organization.
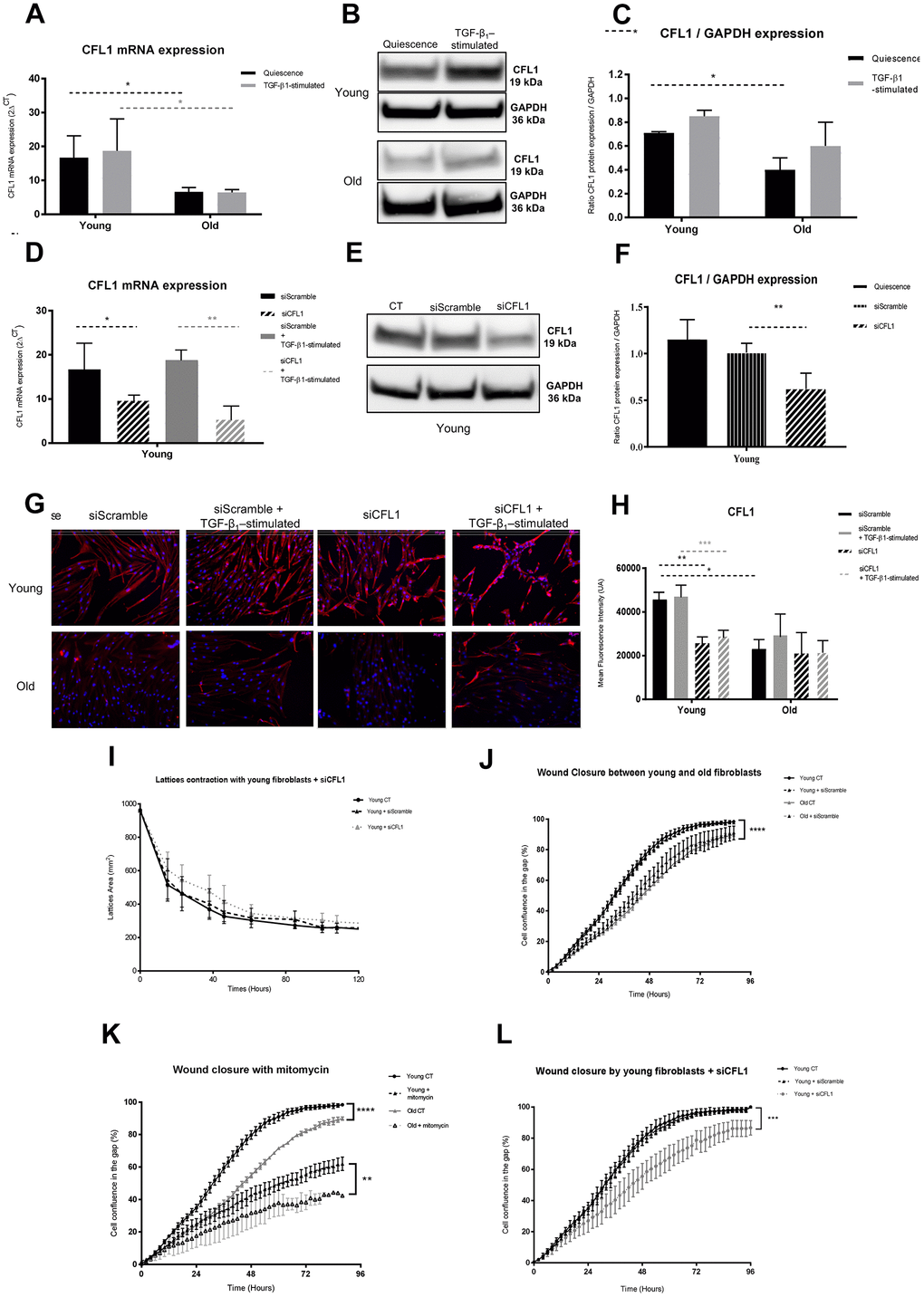
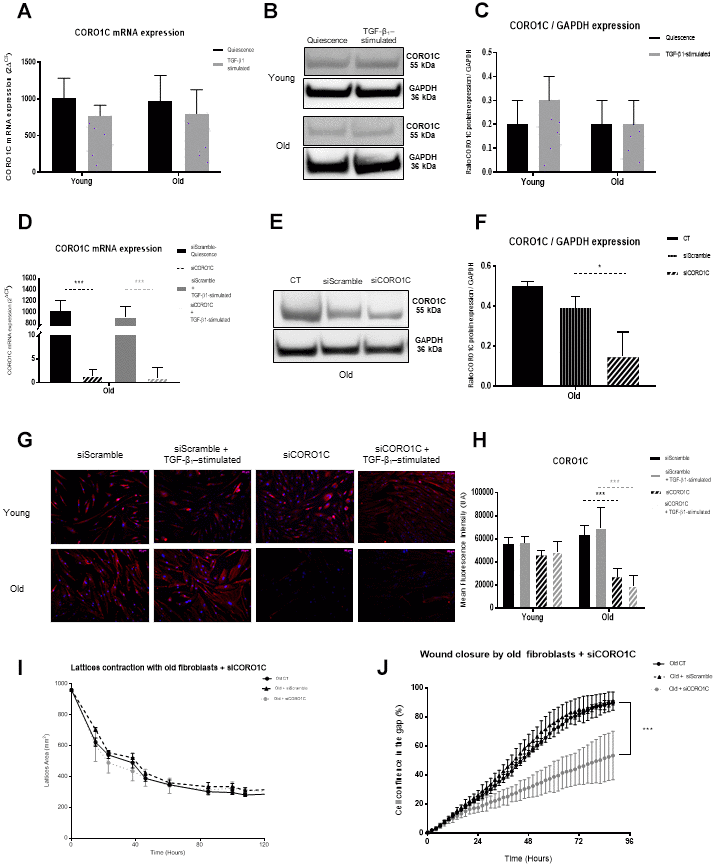
As previously reported [18, 26], we confirmed the significantly lower migratory capacities of old fibroblasts compared to young ones. Beside this fibroblast migration deficiency with age, it has been shown that their numbers decreased [19] and the collagen fibers are more or less abundant and often disorganized and fragmented [44], leading to a defective dermal structure in the elderly. Mechanosensation designs the cell responses to any ECM changes, allowing mechanotransduction through biochemical signals thanks to actin cytoskeleton dynamics [45]. With aging, these mechanical features were shown to be impaired [16]. In particular, the ECM disorganization was reported to favor an alteration in the migration of fibroblasts to the wound site [46]. The modification of ECM components we depicted in old fibroblasts could increase such defect of skin repair. This altered dynamics with aging could be related to our present data showing that the significant alteration of actin-associated proteins, such as CFL1, CORO1C and ARP2/3 in old fibroblasts, did impair cell migration and wound healing.
Accordingly, CFL1, which is a key regulator in actin filament assembly/disassembly (ADF/cofilin), acts upon actin levels and relative binding-proteins – a low cofilin/actin ratio would favor a permanent filament turn-over, whereas a high ratio facilitates CFL1 binding to F-actin in order to stabilize it in saturate severing fragments [47]. CFL1 also works together with the ARP2/3 complex to induce Rac/Rho pathway activity. Moreover, CLF1 loss has been shown to favor an uncontrolled accumulation of contractile actin stress fibers, with a consequent increase in intracellular actin-myosin tension which might promote nuclear deformation and physical disruption as suggested by [48]. We observed a decrease in CFL1 mRNA and protein expression in old fibroblasts and it was indeed associated with a less-effective wound closure by collective fibroblast migration.
CORO1C, which belongs to a family of highly conserved actin-binding proteins that bind to filamentous actin/F-actin, acts as a key regulator of actin assembly/dynamics by working with ARP2/3 complex as well as with Rac1 [49]. CORO1C is located in the lamellipodia, evoking a potent role in motility and migratory ability. Indeed, redistribution of Rac1 from the back of the cell to the front was shown to enable migration, revealing the major involvement of cytoskeleton structure in migrating cells [50]. The overexpression of CORO1C in old fibroblasts seems to down-regulate their motility, although its depletion did not allow it to reach the level of migration of young fibroblasts, suggesting that CORO1C alone was not sufficient to affect motility.
ARP2/3 was another actin-related protein that was down-regulated in old fibroblasts. Identified as a profilin-binding protein, ARP2/3 is a key regulator in the cellular cytoskeleton. It has been shown to be involved in the regulation of actin polymerization although it could not activate this polymerization alone [51]. It can be active with an activating nucleation enhancer factor (NPF) on the formation of branched-actin networks [52], forming the F-actin bypass networks [53]. As with CORO1C, ARP2/3 localization in lamellipodia enables fibroblast migration [54]. Our data demonstrated the involvement of this complex in collective migration since ARP2/3 downregulation by siRNA induced a decrease in migratory capacity of young fibroblasts (data not shown), just as CFL1 did. There is a potent link between the three actin-related molecules that might complementary act together, in common with Rac1, in the regulation of the actin cytoskeleton and the cytoskeleton itself, as illustrated in Figure 4K, to schematize their complementary function in the dynamic actin-polymerization process that could be deregulated with aging.
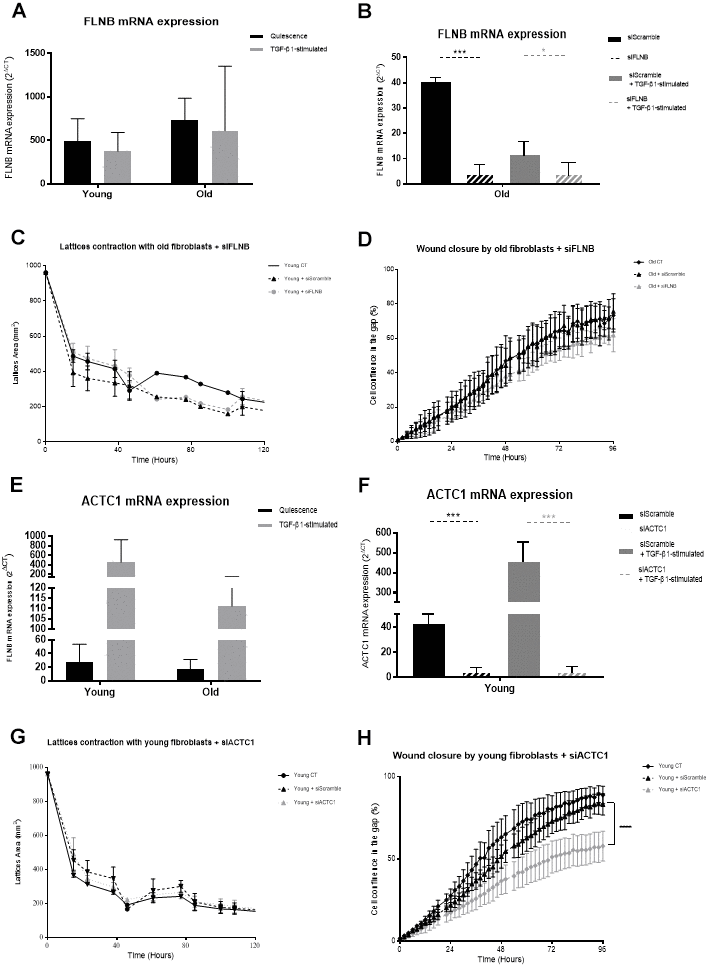
FLNB is another actin-related protein with a key role in the migratory turnover process by connecting filaments of the actin cytoskeleton to the plasma membrane, allowing cells to change shape and to move [55]. Targeting FLNB in fibroblasts was shown to increase matrix metallopeptidase-9 (MMP9) and VEGF secretion which led to induction in tumor growth [56]. Integrin activation has been shown to be involved in the FLNB role in cellular migration [30, 57] in accordance with our data on the decrease of migratory capacity of old fibroblasts after transfection of siRNA targeting FLNB. However, we did not observe a significant alteration in integrin and focal adhesion expression in proteome data or by immunofluorescence analysis, nor an increase in VEGF production by transfected fibroblasts (data not shown).
As actins play a key role in the migratory capacity through their involvement in motility, protusion cell cycle, traction and tail retraction [58, 59], the very low expression of one of the six actin isoforms, ACTC1, in old fibroblasts might be in relation with their altered migratory capacities since ACTC1 depletion in young cells did reduce their migration, suggesting a major involvement of this protein in the wound closure process through fibroblast functions.
Further, the dynamic features, including motility and mechanical forces exhibited by young and old fibroblasts, will be analyzed using Traction Force Microscopy (TFM) and single cell migration. A combined downregulation of the three actin-related proteins we studied would confirm their close association to cytoskeleton behavior and cell motility/migration to further modulate their involvement in aging. Yet they do not seem to be major actors in the merge of traction and contraction of the wounds.
Altogether, our data showed that regulation of fibroblast cytoskeleton through actin-related proteins, combined with ECM organization, is a key process to control skin aging. Noteworthy, our comparative data from the proteomes described several unknown proteins or with undefined functions. These proteins should be further studied in order to investigate more deeply the ongoing aging process and its regulation.
Conclusions
This study revealed a significant decrease in fibroblast protein secretion with age and conversely an enhancement of more than 60% of cytoplasmic protein accumulation. Proteins associated with actin and ECM organization revealed to be the two main fields of proteins modified during aging. In depth analysis of actin-related proteins showed evidence of involvement of proteins like CFL1, CORO1C, ARP2/3-complex, FLNB and ACTC1, in cytoskeleton organization and fibroblast migration capacities, offering new targets to slow characteristic features of skin aging.
Supplementary Materials
Author Contributions
Boismal F. carried out the experiments and their analysis and participated in writing the article. Peltier S. participated in some experiments and analysis. Ly ka so S. carried out the fibroblast sample preparation and their collection for proteomic and secretomic assay. Chevreux G. performed HPLC/MS assay. Blondel L. performed a part of transfection experiments. Setterblad N. provided technical advice for using video-microscopy platform. Serror K., Boccara D., Mimoun M. provided all the surgery skin samples. Guere C., Beauchef G., Dorr MM. and Vie K. as employed by Clarins Laboratories Pontoise France, provided the financial support for this project. Benssussan A., and Michel L. discussed the project. Michel L. conceptualized and supervised the whole project, including writing, review, and editing the paper.
Acknowledgments
We thank the team of the institute’s microscopy and cytometry platform of Saint-Louis Institute (IRSL) conducted by Setterblad N., for their availability in our work. We thank Jean-Michel Camadro, scientific coordinator, Véronique Legros, research engineer, Thibault Leger, research engineer, and Laurent Lignières, research engineer, all from the team of the Proteomique Platform of Institut Jacques Monod, Paris.
Conflicts of Interest
The authors have the following interests. This work was supported by Clarins Laboratories Pontoise France. Christelle Guere, Gallic Beauchef, Marie Dorr and Katell Vie are employed by Clarins Laboratories Pontoise France. There are no patents, products in development or marketed products to declare.
Ethical Statement and Consent
This study was approved by our institution’s ethical committee at Saint-Louis Hospital. Skin tissues were collected in compliance with ethics recommendations. The protocol was assessed according to the Jardé Law with non-opposition signature of the volunteers for research use of surgical residual skin. The written informed consent of non-opposition was given by each woman.
Funding
No funding was provided for this study.
References
- 1. Harman D. The aging process. Proc Natl Acad Sci USA. 1981; 78:7124–8. https://doi.org/10.1073/pnas.78.11.7124 [PubMed]
- 2. Kirkwood TBL, Austad SN. Why do we age? Nature. 2000; 408:233–8. https://doi.org/10.1038/35041682 [PubMed]
- 3. Farage MA, Miller KW, Elsner P, Maibach HI. Intrinsic and extrinsic factors in skin ageing: a review. Int J Cosmet Sci. 2008; 30:87–95. https://doi.org/10.1111/j.1468-2494.2007.00415.x [PubMed]
- 4. Yaar M. Clinical and Histological Features of Intrinsic versus Extrinsic Skin Aging. In: Gilchrest BA, Krutmann J, (eds). Skin Aging. Berlin, Heidelberg: Springer Berlin Heidelberg; 2006:9–21. https://doi.org/10.1007/3-540-32953-6_2
- 5. Boyd AS, Stasko T, King LE
Jr , Cameron GS, Pearse AD, Gaskell SA. Cigarette smoking-associated elastotic changes in the skin. J Am Acad Dermatol. 1999; 41:23–6. https://doi.org/10.1016/s0190-9622(99)70400-7 [PubMed] - 6. Demierre MF, Brooks D, Koh HK, Geller AC. Public knowledge, awareness, and perceptions of the association between skin aging and smoking. J Am Acad Dermatol. 1999; 41:27–30. https://doi.org/10.1016/s0190-9622(99)70401-9 [PubMed]
- 7. Fenske NA, Lober CW. Structural and functional changes of normal aging skin. J Am Acad Dermatol. 1986; 15:571–85. https://doi.org/10.1016/s0190-9622(86)70208-9 [PubMed]
- 8. Krutmann J, Bouloc A, Sore G, Bernard BA, Passeron T. The skin aging exposome. J Dermatol Sci. 2017; 85:152–61. https://doi.org/10.1016/j.jdermsci.2016.09.015 [PubMed]
- 9. Lavker RM, Gerberick GF, Veres D, Irwin CJ, Kaidbey KH. Cumulative effects from repeated exposures to suberythemal doses of UVB and UVA in human skin. J Am Acad Dermatol. 1995; 32:53–62. https://doi.org/10.1016/0190-9622(95)90184-1 [PubMed]
- 10. Brennan M, Bhatti H, Nerusu KC, Bhagavathula N, Kang S, Fisher GJ, Varani J, Voorhees JJ. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem Photobiol. 2003; 78:43–8. https://doi.org/10.1562/0031-8655(2003)078<0043:mmitmc>2.0.co;2 [PubMed]
- 11. Waldera Lupa DM, Kalfalah F, Safferling K, Boukamp P, Poschmann G, Volpi E, Götz-Rösch C, Bernerd F, Haag L, Huebenthal U, Fritsche E, Boege F, Grabe N, et al. Characterization of Skin Aging-Associated Secreted Proteins (SAASP) Produced by Dermal Fibroblasts Isolated from Intrinsically Aged Human Skin. J Invest Dermatol. 2015; 135:1954–68. https://doi.org/10.1038/jid.2015.120 [PubMed]
- 12. Narzt MS, Pils V, Kremslehner C, Nagelreiter IM, Schosserer M, Bessonova E, Bayer A, Reifschneider R, Terlecki-Zaniewicz L, Waidhofer-Söllner P, Mildner M, Tschachler E, Cavinato M, et al. Epilipidomics of Senescent Dermal Fibroblasts Identify Lysophosphatidylcholines as Pleiotropic Senescence-Associated Secretory Phenotype (SASP) Factors. J Invest Dermatol. 2021; 141:993–1006.e15. https://doi.org/10.1016/j.jid.2020.11.020 [PubMed]
- 13. Makrantonaki E, Zouboulis CC, and German National Genome Research Network 2. The skin as a mirror of the aging process in the human organism--state of the art and results of the aging research in the German National Genome Research Network 2 (NGFN-2). Exp Gerontol. 2007; 42:879–86. https://doi.org/10.1016/j.exger.2007.07.002 [PubMed]
- 14. Makrantonaki E, Zouboulis CC. William J. Cunliffe Scientific Awards. Characteristics and pathomechanisms of endogenously aged skin. Dermatology. 2007; 214:352–60. https://doi.org/10.1159/000100890 [PubMed]
- 15. Braverman IM, Fonferko E. Studies in cutaneous aging: I. The elastic fiber network. J Invest Dermatol. 1982; 78:434–43. https://doi.org/10.1111/1523-1747.ep12507866 [PubMed]
- 16. Phillip JM, Aifuwa I, Walston J, Wirtz D. The Mechanobiology of Aging. Annu Rev Biomed Eng. 2015; 17:113–41. https://doi.org/10.1146/annurev-bioeng-071114-040829 [PubMed]
- 17. Brun C, Demeaux A, Guaddachi F, Jean-Louis F, Oddos T, Bagot M, Bensussan A, Jauliac S, Michel L. T-plastin expression downstream to the calcineurin/NFAT pathway is involved in keratinocyte migration. PLoS One. 2014; 9:e104700. https://doi.org/10.1371/journal.pone.0104700 [PubMed]
- 18. Brun C, Jean-Louis F, Oddos T, Bagot M, Bensussan A, Michel L. Phenotypic and functional changes in dermal primary fibroblasts isolated from intrinsically aged human skin. Exp Dermatol. 2016; 25:113–9. https://doi.org/10.1111/exd.12874 [PubMed]
- 19. Gosain A, DiPietro LA. Aging and wound healing. World J Surg. 2004; 28:321–6. https://doi.org/10.1007/s00268-003-7397-6 [PubMed]
- 20. Zouboulis CC, Makrantonaki E. Clinical aspects and molecular diagnostics of skin aging. Clin Dermatol. 2011; 29:3–14. https://doi.org/10.1016/j.clindermatol.2010.07.001 [PubMed]
- 21. Haydont V, Neiveyans V, Zucchi H, Fortunel NO, Asselineau D. Genome-wide profiling of adult human papillary and reticular fibroblasts identifies ACAN, Col XI α1, and PSG1 as general biomarkers of dermis ageing, and KANK4 as an exemplary effector of papillary fibroblast ageing, related to contractility. Mech Ageing Dev. 2019; 177:157–81. https://doi.org/10.1016/j.mad.2018.06.003 [PubMed]
- 22. Talwar HS, Griffiths CEM, Fisher GJ, Hamilton TA, Voorhees JJ. Reduced type I and type III procollagens in photodamaged adult human skin. J Invest Dermatol. 1995; 105:285–90. https://doi.org/10.1111/1523-1747.ep12318471 [PubMed]
- 23. Makrantonaki E, Zouboulis CC. Skin alterations and diseases in advanced age. Drug Discov Today Dis Mech. 2008; 5:e153–62. https://doi.org/10.1016/j.ddmec.2008.05.008
- 24. Tigges J, Krutmann J, Fritsche E, Haendeler J, Schaal H, Fischer JW, Kalfalah F, Reinke H, Reifenberger G, Stühler K, Ventura N, Gundermann S, Boukamp P, Boege F. The hallmarks of fibroblast ageing. Mech Ageing Dev. 2014; 138:26–44. https://doi.org/10.1016/j.mad.2014.03.004 [PubMed]
- 25. Pieraggi MT, Julian M, Bouissou H. Fibroblast changes in cutaneous ageing. Virchows Arch A Pathol Anat Histopathol. 1984; 402:275–87. https://doi.org/10.1007/BF00695081 [PubMed]
- 26. Reed MJ, Ferara NS, Vernon RBB. Impaired migration, integrin function, and actin cytoskeletal organization in dermal fibroblasts from a subset of aged human donors. Mech Ageing Dev. 2001; 122:1203–20. https://doi.org/10.1016/s0047-6374(01)00260-3 [PubMed]
- 27. Basisty N, Kale A, Jeon OH, Kuehnemann C, Payne T, Rao C, Holtz A, Shah S, Sharma V, Ferrucci L, Campisi J, Schilling B. A proteomic atlas of senescence-associated secretomes for aging biomarker development. PLoS Biol. 2020; 18:e3000599. https://doi.org/10.1371/journal.pbio.3000599 [PubMed]
- 28. Makrantonaki E, Brink TC, Zampeli V, Elewa RM, Mlody B, Hossini AM, Hermes B, Krause U, Knolle J, Abdallah M, Adjaye J, Zouboulis CC. Identification of biomarkers of human skin ageing in both genders. Wnt signalling - a label of skin ageing? PLoS One. 2012; 7:e50393. https://doi.org/10.1371/journal.pone.0050393 [PubMed]
- 29. van der Flier A, Sonnenberg A. Structural and functional aspects of filamins. Biochim Biophys Acta. 2001; 1538:99–117. https://doi.org/10.1016/s0167-4889(01)00072-6 [PubMed]
- 30. Nakamura F, Stossel TP, Hartwig JH. The filamins: organizers of cell structure and function. Cell Adh Migr. 2011; 5:160–9. https://doi.org/10.4161/cam.5.2.14401 [PubMed]
- 31. Kanellos G, Zhou J, Patel H, Ridgway RA, Huels D, Gurniak CB, Sandilands E, Carragher NO, Sansom OJ, Witke W, Brunton VG, Frame MC. ADF and Cofilin1 Control Actin Stress Fibers, Nuclear Integrity, and Cell Survival. Cell Rep. 2015; 13:1949–64. https://doi.org/10.1016/j.celrep.2015.10.056 [PubMed]
- 32. Rotty JD, Wu C, Bear JE. New insights into the regulation and cellular functions of the ARP2/3 complex. Nat Rev Mol Cell Biol. 2013; 14:7–12. https://doi.org/10.1038/nrm3492 [PubMed]
- 33. Behrens J, Solga R, Ziemann A, Rastetter RH, Berwanger C, Herrmann H, Noegel AA, Clemen CS. Coronin 1C-free primary mouse fibroblasts exhibit robust rearrangements in the orientation of actin filaments, microtubules and intermediate filaments. Eur J Cell Biol. 2016; 95:239–51. https://doi.org/10.1016/j.ejcb.2016.04.004 [PubMed]
- 34. Baldassarre M, Razinia Z, Burande CF, Lamsoul I, Lutz PG, Calderwood DA. Filamins regulate cell spreading and initiation of cell migration. PLoS One. 2009; 4:e7830. https://doi.org/10.1371/journal.pone.0007830 [PubMed]
- 35. Hu J, Lu J, Lian G, Ferland RJ, Dettenhofer M, Sheen VL. Formin 1 and filamin B physically interact to coordinate chondrocyte proliferation and differentiation in the growth plate. Hum Mol Genet. 2014; 23:4663–73. https://doi.org/10.1093/hmg/ddu186 [PubMed]
- 36. Müller M, Diensthuber RP, Chizhov I, Claus P, Heissler SM, Preller M, Taft MH, Manstein DJ. Distinct functional interactions between actin isoforms and nonsarcomeric myosins. PLoS One. 2013; 8:e70636. https://doi.org/10.1371/journal.pone.0070636 [PubMed]
- 37. Tschumperlin DJ. Fibroblasts and the ground they walk on. Physiology (Bethesda). 2013; 28:380–90. https://doi.org/10.1152/physiol.00024.2013 [PubMed]
- 38. Olson TM, Doan TP, Kishimoto NY, Whitby FG, Ackerman MJ, Fananapazir L. Inherited and de novo mutations in the cardiac actin gene cause hypertrophic cardiomyopathy. J Mol Cell Cardiol. 2000; 32:1687–94. https://doi.org/10.1006/jmcc.2000.1204 [PubMed]
- 39. Ghosh K, Capell BC. The Senescence-Associated Secretory Phenotype: Critical Effector in Skin Cancer and Aging. J Invest Dermatol. 2016; 136:2133–9. https://doi.org/10.1016/j.jid.2016.06.621 [PubMed]
- 40. Demaria M, Desprez PY, Campisi J, Velarde MC. Cell Autonomous and Non-Autonomous Effects of Senescent Cells in the Skin. J Invest Dermatol. 2015; 135:1722–6. https://doi.org/10.1038/jid.2015.108 [PubMed]
- 41. Gourlay CW, Ayscough KR. A role for actin in aging and apoptosis. Biochem Soc Trans. 2005; 33:1260–4. https://doi.org/10.1042/BST0331260 [PubMed]
- 42. Cole MA, Quan T, Voorhees JJ, Fisher GJ. Extracellular matrix regulation of fibroblast function: redefining our perspective on skin aging. J Cell Commun Signal. 2018; 12:35–43. https://doi.org/10.1007/s12079-018-0459-1 [PubMed]
- 43. Haydont V, Neiveyans V, Fortunel NO, Asselineau D. Transcriptome profiling of human papillary and reticular fibroblasts from adult interfollicular dermis pinpoints the ‘tissue skeleton’ gene network as a component of skin chrono-ageing. Mech Ageing Dev. 2019; 179:60–77. https://doi.org/10.1016/j.mad.2019.01.003 [PubMed]
- 44. Shin JW, Kwon SH, Choi JY, Na JI, Huh CH, Choi HR, Park KC. Molecular Mechanisms of Dermal Aging and Antiaging Approaches. Int J Mol Sci. 2019; 20:2126. https://doi.org/10.3390/ijms20092126 [PubMed]
- 45. Wang HB, Dembo M, Hanks SK, Wang Y. Focal adhesion kinase is involved in mechanosensing during fibroblast migration. Proc Natl Acad Sci USA. 2001; 98:11295–300. https://doi.org/10.1073/pnas.201201198 [PubMed]
- 46. Qin Z, Fisher GJ, Voorhees JJ, Quan T. Actin cytoskeleton assembly regulates collagen production via TGF-β type II receptor in human skin fibroblasts. J Cell Mol Med. 2018; 22:4085–96. https://doi.org/10.1111/jcmm.13685 [PubMed]
- 47. Bamburg JR, Bernstein BW. Roles of ADF/cofilin in actin polymerization and beyond. F1000 Biol Rep. 2010; 2:62. https://doi.org/10.3410/B2-62 [PubMed]
- 48. Hensbergen P, Alewijnse A, Kempenaar J, van der Schors RC, Balog CIA, Deelder A, Beumer G, Ponec M, Tensen CP. Proteomic profiling identifies an UV-induced activation of cofilin-1 and destrin in human epidermis. J Invest Dermatol. 2005; 124:818–24. https://doi.org/10.1111/j.0022-202X.2005.23597.x [PubMed]
- 49. Chan KT, Roadcap DW, Holoweckyj N, Bear JE. Coronin 1C harbours a second actin-binding site that confers co-operative binding to F-actin. Biochem J. 2012; 444:89–96. https://doi.org/10.1042/BJ20120209 [PubMed]
- 50. Tilley FC, Williamson RC, Race PR, Rendall TC, Bass MD. Integration of the Rac1- and actin-binding properties of Coronin-1C. Small GTPases. 2015; 6:36–42. https://doi.org/10.4161/21541248.2014.992259 [PubMed]
- 51. Machesky LM, Gould KL. The Arp2/3 complex: a multifunctional actin organizer. Curr Opin Cell Biol. 1999; 11:117–21. https://doi.org/10.1016/s0955-0674(99)80014-3 [PubMed]
- 52. Goley ED, Rodenbusch SE, Martin AC, Welch MD. Critical conformational changes in the Arp2/3 complex are induced by nucleotide and nucleation promoting factor. Mol Cell. 2004; 16:269–79. https://doi.org/10.1016/j.molcel.2004.09.018 [PubMed]
- 53. Goley ED, Welch MD. The ARP2/3 complex: an actin nucleator comes of age. Nat Rev Mol Cell Biol. 2006; 7:713–26. https://doi.org/10.1038/nrm2026 [PubMed]
- 54. Wu C, Asokan SB, Berginski ME, Haynes EM, Sharpless NE, Griffith JD, Gomez SM, Bear JE. Arp2/3 is critical for lamellipodia and response to extracellular matrix cues but is dispensable for chemotaxis. Cell. 2012; 148:973–87. https://doi.org/10.1016/j.cell.2011.12.034 [PubMed]
- 55. Zhou X, Tian F, Sandzén J, Cao R, Flaberg E, Szekely L, Cao Y, Ohlsson C, Bergo MO, Borén J, Akyürek LM. Filamin B deficiency in mice results in skeletal malformations and impaired microvascular development. Proc Natl Acad Sci USA. 2007; 104:3919–24. https://doi.org/10.1073/pnas.0608360104 [PubMed]
- 56. Bandaru S, Zhou AX, Rouhi P, Zhang Y, Bergo MO, Cao Y, Akyürek LM. Targeting filamin B induces tumor growth and metastasis via enhanced activity of matrix metalloproteinase-9 and secretion of VEGF-A. Oncogenesis. 2014; 3:e119. https://doi.org/10.1038/oncsis.2014.33 [PubMed]
- 57. MacPherson M, Fagerholm SC. Filamin and filamin-binding proteins in integrin-regulation and adhesion. Focus on: “FilaminA is required for vimentin-mediated cell adhesion and spreading”. Am J Physiol Cell Physiol. 2010; 298:C206–8. https://doi.org/10.1152/ajpcell.00505.2009 [PubMed]
- 58. Heck T, Vargas DA, Smeets B, Ramon H, Van Liedekerke P, Van Oosterwyck H. The role of actin protrusion dynamics in cell migration through a degradable viscoelastic extracellular matrix: Insights from a computational model. PLoS Comput Biol. 2020; 16:e1007250. https://doi.org/10.1371/journal.pcbi.1007250 [PubMed]
- 59. Kole TP, Tseng Y, Jiang I, Katz JL, Wirtz D. Intracellular mechanics of migrating fibroblasts. Mol Biol Cell. 2005; 16:328–38. https://doi.org/10.1091/mbc.e04-06-0485 [PubMed]