Introduction
Malignant neoplasms, a major health concern, are leading causes of mortality worldwide, placing a significant strain on healthcare systems [1]. Even with substantial progress in cancer treatments, the prognosis for various cancers remains a critical issue, reflecting gaps in our current therapeutic approaches [2, 3]. Central to this challenge is the tumor microenvironment (TME), including the tumor immune microenvironment (TIME), which is crucial in cancer progression and metastasis [4]. Understanding and targeting the complexities of TME and TIME is essential for developing effective cancer treatments and improving patient outcomes [5].
Cyclin-dependent kinase 16 (CDK16) is a distinct member of the “cyclin-dependent kinase” (CDK) family, playing a crucial role in various cellular processes. These include cell cycle regulation, vesicle trafficking, spindle orientation, skeletal myogenesis, neurite outgrowth, transportation of secretory cargo, spermatogenesis, glucose transport, as well as in the regulation of cell apoptosis, growth, proliferation, metastasis, and autophagy [6]. Located on chromosome Xp11.3 in humans, CDK16 is associated with X-linked congenital diseases [7]. Commonly expressed in mammalian tissues, it often acts as an oncogene [8]. As a PCTAIRE kinase, its activity is modulated by the binding of Cyclin Y or its homolog Cyclin Y-like 1 to the N and C-terminal regions of CDK16 [7, 9]. Crucially, CDK16 has been identified as playing a pivotal role in the progression of various cancers, including lung, prostate, breast, melanoma, and hepatocellular carcinomas [10–14]. Despite its significance, the influence of CDK16 on the tumor microenvironment, particularly its correlation with immune therapies, has been historically overlooked. This study conducts a comprehensive analysis of CDK16’s role in pan-cancer and its function within the TME, thereby highlighting its considerable potential in cancer therapy.
In our study, we examined CDK16’s role in various cancers, focusing on its expression, genetic variations, and prognostic relevance. Using data from diverse databases for 33 cancer types, we applied methods like GSEA to assess CDK16’s impact on tumor progression and its connection to immune pathways. We explored the relationship between CDK16 expression, immune cell infiltration, and immune gene expression. Our results highlight a strong link between increased CDK16 expression and an immunosuppressive tumor microenvironment, implying CDK16’s role in tumor-induced immunosuppression and potential implications for immune therapy. This underscores CDK16’s importance in cancer progression and its potential influence on immune therapy outcomes.
Materials and Methods
Data source
RNAseq data and clinical information data for pan-cancer were obtained from The Cancer Genome Atlas (TCGA) and Genotype-Tissue Expression (GTEx), which were downloaded from UCSC Xena (https://xenabrowser.net/datapages/). Immunocell infiltration data for CDK16 were downloaded from TIMER2 (http://timer.cistrome.org/) and ImmuCellAI (http://bioinfo.life.hust.edu.cn/ImmuCellAI#!/) databases.
GSEA
To assess the link between CDK16 and its associated genes in human cancers, we employed Gene Set Enrichment Analysis (GSEA). For this analysis, we utilized Pearson correlation coefficients and the R package called “clusterProfiler”. Through these comprehensive analyses, we were able to obtain valuable insights into the relationship between CDK16 and the expression patterns of its associated genes in human cancers.
Immune infiltration analysis
The R package “ESTIMATE” was used to assess the TME in pan-cancer. Immune cell infiltration data were obtained from the TIMER2 database and published study (Zeng et al., 2019). CDK16 expression and immune cell infiltration levels were correlated to explore the relationship between CDK16 and immune cell infiltration.
Cell culture
The lung cancer cell line H1299 was obtained from ATCC (Manassas, VA, USA). The cells were cultured in RPMI-1640 medium (Gibco, China) supplemented with 10% fetal bovine serum (Gibco, China). The cultures were maintained at 37°C in a humidified atmosphere containing 5% CO2.
CCK-8 assay
To assess cell viability, the CCK-8 assay was employed. After transfecting the cells with siRNA for 48 hours, they were transferred to 96-well plates at a density of 3000 cells per well and cultured as described above. CCK-8 reagent (Beyotime, Shanghai, China) was added to each well and cultured for two hours. The optical density at 450 nm was measured at 0, 24, 48, and 72 hours using an iD3 microplate reader.
RNA extraction and qRT-PCR
According to the manufacturer’s protocol, total RNA was isolated using TRIzol reagent (Pufei, Shanghai, China). The primers sequences used for qRT-PCR were obtained from Applied Biosystems (Ribo, Guangzhou, China) as below (5′–3′): CDK16 forward: 5′-TTGGGCCGTTGGCTGTTC-3′, reverse: 5′-GTGCTCACGGCGGCTC-3′; GAPDH forward: 5′-CAGTGCCAGCCTCGTCTAT-3′, reverse: 5′-AGGGGCCATCCACAGTCTTC-3′. Relative expression levels were calculated according to the 2−ΔΔCt method. Statistical analyses were performed with GraphPad Prism.
Statistical analysis
We used t-tests to assess the disparity in CDK16 gene expression levels between cancer tissues and normal tissues. Survival impacts were analyzed using the Kaplan-Meier method and log-rank test, reporting hazard ratios, confidence intervals, and p-values for clarity. Correlation analyses were conducted with Spearman’s or Pearson’s tests based on data type. All analyses were performed using R software (version 4.0.2), with a significance threshold set at a p-value of less than 0.05, ensuring the reliability of our findings.
Data availability statement
The original data supporting the conclusions of this paper will be provided by the authors without reservation.
Results
Expression of CDK16 in pan-cancer
In our study, we assessed CDK16 expression in a pan-cancer context using data sourced from The Cancer Genome Atlas (TCGA) and the Genotype-Tissue Expression (GTEx) project. Our findings revealed that CDK16 was notably overexpressed in 23 out of 33 cancer types examined. These included Bladder Urothelial Carcinoma (BLCA), Breast Invasive Carcinoma (BRCA), Cervical Squamous Cell Carcinoma and Endocervical Adenocarcinoma (CESC), Cholangiocarcinoma (CHOL), Colon Adenocarcinoma (COAD), Lymphoid Neoplasm Diffuse Large B-cell Lymphoma (DLBC), Esophageal Carcinoma (ESCA), Glioblastoma Multiforme (GBM), Head and Neck Squamous Cell Carcinoma (HNSC), Kidney Chromophobe (KICH), Lower Grade Glioma (LGG), Liver Hepatocellular Carcinoma (LIHC), Lung Adenocarcinoma (LUAD), Lung Squamous Cell Carcinoma (LUSC), Ovarian Serous Cystadenocarcinoma (OV), Pancreatic Adenocarcinoma (PAAD), Rectum Adenocarcinoma (READ), Sarcoma (SARC), Skin Cutaneous Melanoma (SKCM), Stomach Adenocarcinoma (STAD), Thymoma (THYM), Uterine Corpus Endometrial Carcinoma (UCEC), and Uterine Carcinosarcoma (UCS). Conversely, CDK16 exhibited lower expression levels in five cancer types, specifically Kidney Renal Clear Cell Carcinoma (KIRC), Acute Myeloid Leukemia (LAML), Prostate Adenocarcinoma (PRAD), Testicular Germ Cell Tumor (TGCT), and Thyroid Carcinoma (THCA), as delineated in Figure 1A and Supplementary Table 1. Notably, the highest expression of CDK16 was observed in ovarian tumor tissues (OV), with the lowest in liver hepatocellular carcinoma (LIHC), as shown in Figure 1B. In normal tissues, the highest expression of CDK16 was identified in the testis (Figure 1C). Additionally, a comparative analysis between tumor tissues and adjacent normal tissues revealed that CDK16 was overexpressed in 15 cancer types, specifically BLCA, BRCA, CHOL, COAD, UCEC, ESCA, THCA, HNSC, READ, LUAD, LIHC, PRAD, STAD, LUSC, and KICH, while it showed lower expression in KIRC, as illustrated in Figure 2A–2P. This differential expression pattern of CDK16 highlights its potential as a biomarker for cancer diagnosis and as a therapeutic target, particularly in cancers with significant overexpression.
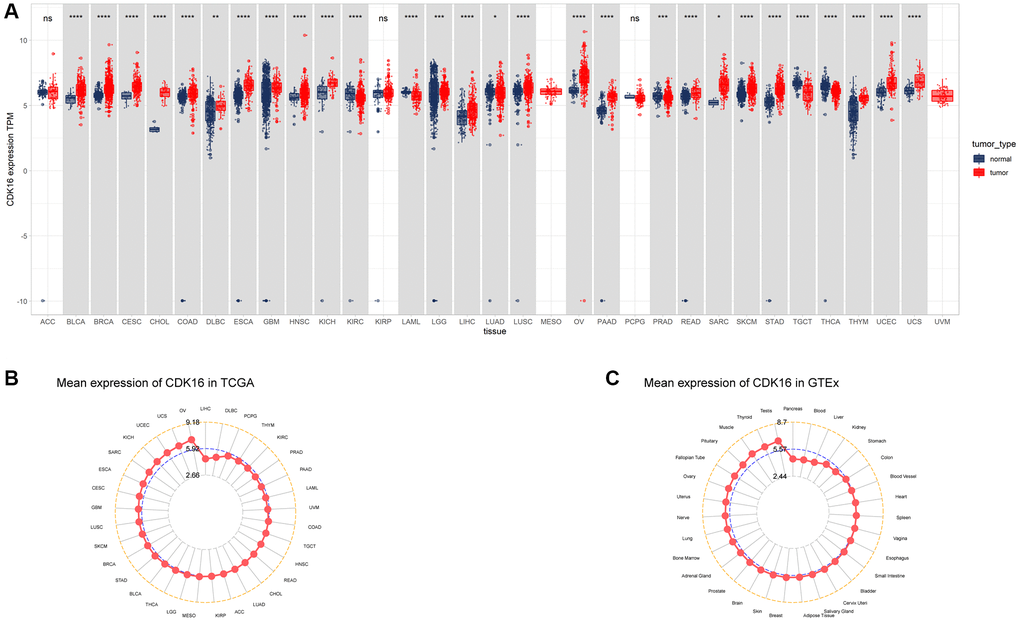
Figure 1. Expression of CDK16 in pan-cancer. (A) Pan-cancer expression of CDK16. (B) CDK16 expression in tumor tissues from TCGA cohort. (C) CDK16 expression in normal tissues from GTEx cohort. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
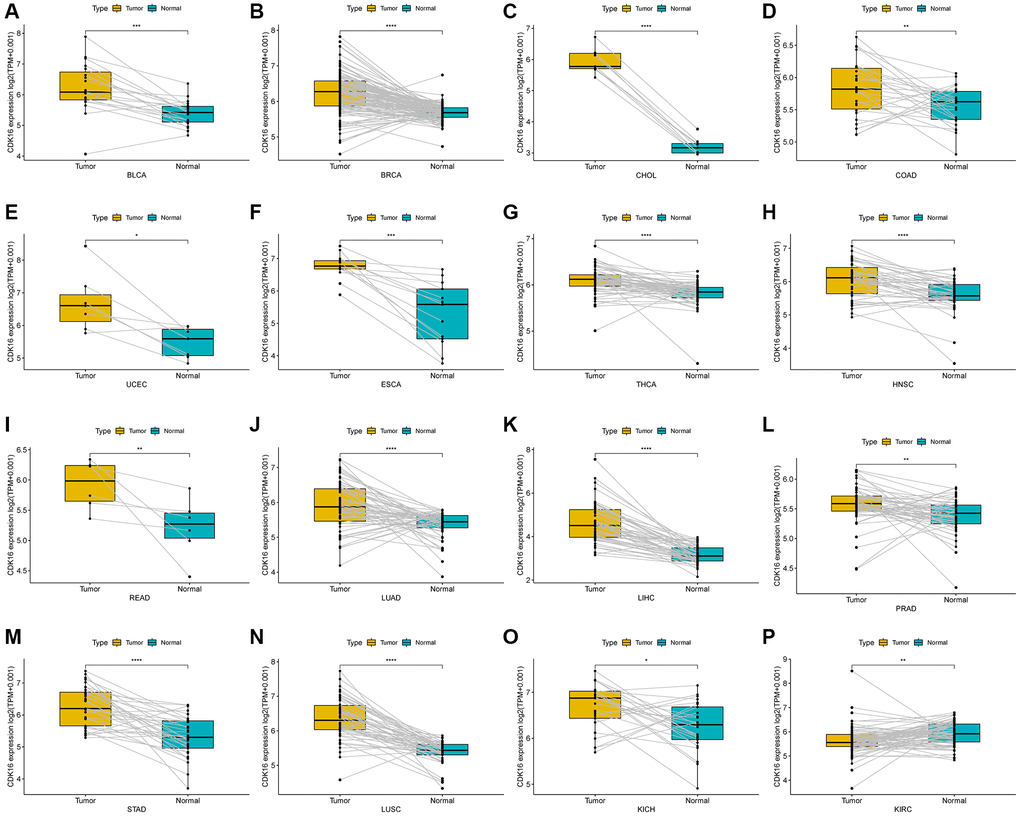
Figure 2. Expression of CDK16 in paired tumor and adjacent normal tissues. (A–P) CDK16 expression in paired tumor and adjacent normal tissues from TCGA in indicated tumor types. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Prognostic role of CDK16
In the univariate Cox regression analysis (UniCox), CDK16 emerged as a significant risk factor for overall survival (OS) in a range of cancers, namely LIHC, LGG, PCPG, KIRC, UCEC, UVM, BRCA, and MESO (Figure 3A). This finding indicates the potential of CDK16 as a prognostic marker across these diverse cancer types. Kaplan-Meier survival analysis further revealed a correlation between high CDK16 expression and poorer OS in ACC, UVM, SARC, UCEC, LIHC, and KICH (Figure 3B–3G). This pattern highlights the importance of monitoring CDK16 levels in these specific cancers for better prognostic assessment. Concerning disease-specific survival (DSS) outcomes, elevated CDK16 expression was associated with increased risk in KIRC, PCPG, LIHC, LGG, UVM, PRAD, BRCA, UCEC, MESO, and ESCA (Figure 4A). This suggests that CDK16 could play a significant role in the disease course of these cancers. The study also examined the impact of CDK16 on disease-free interval (DFI). High CDK16 expression was predictive of a shorter DFI in PRAD, LIHC, SARC, and ACC (Figure 4B), indicating its potential as an early marker for recurrence or progression in these cancers. Lastly, for progression-free interval (PFI), increased CDK16 expression was linked to worse PFI in PRAD, ACC, LIHC, PCPG, UCEC, UVM, ESCA, LGG, SARC, MESO, BRCA, and KIRC (Figure 4C). This broad association underscores the potential of CDK16 as a marker for monitoring cancer progression across various types. Overall, the expanded analysis underscores the significance of CDK16 in a variety of cancers, suggesting its potential utility as a biomarker for prognosis and therapeutic targeting.
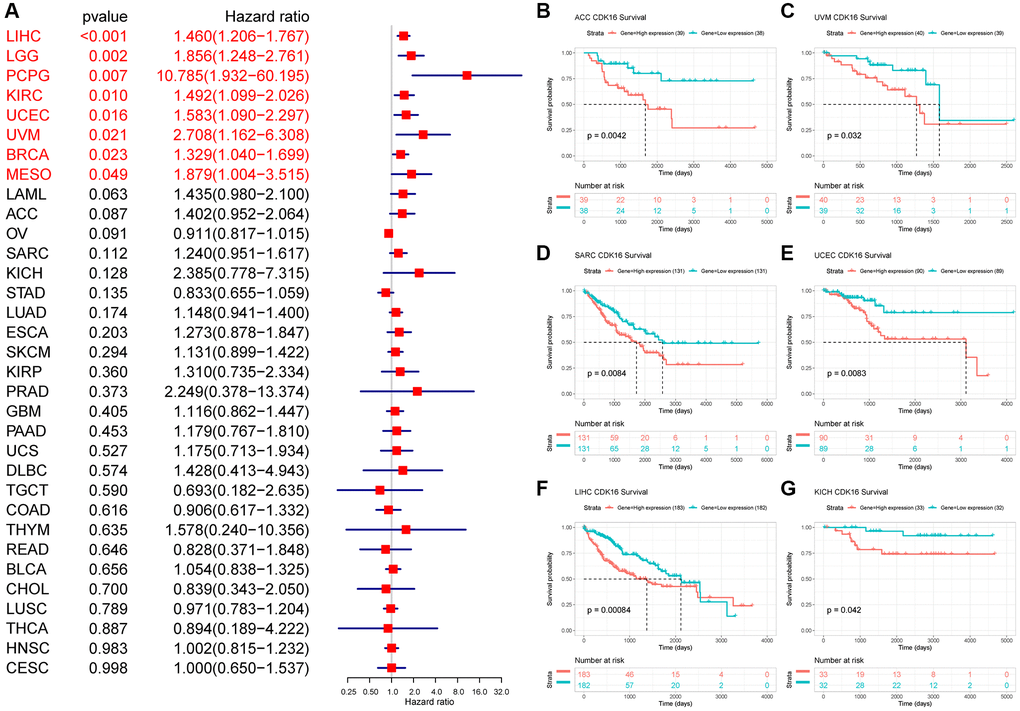
Figure 3. Relationship between CDK16 level and OS. (A) The univariate Cox regression OS analysis of CDK16 in TCGA pan-cancer. Red color represents significant results (p < 0.05). (B–G) Kaplan-Meier curves showing OS in pan-cancer.
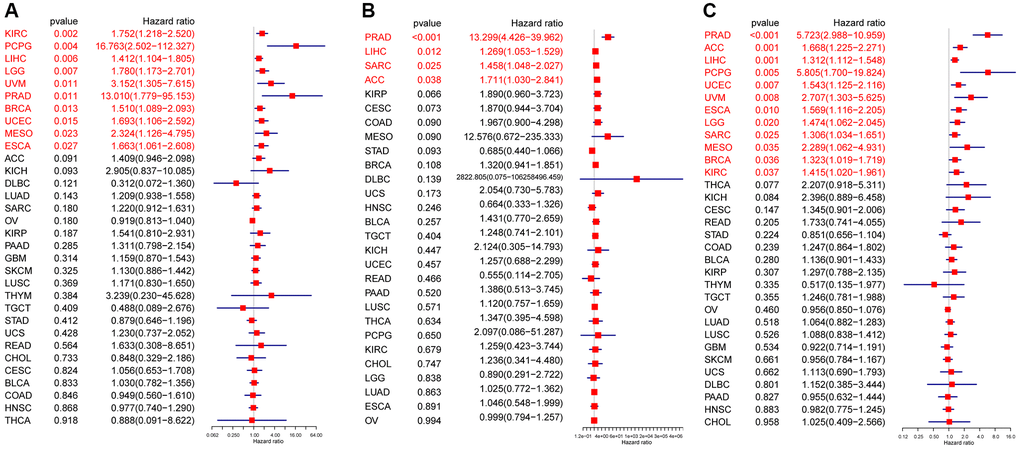
Figure 4. Prognostic value of CDK16. Forest plots showing results of univariate Cox regression analysis for (A) DSS, (B) DFI, and (C) PFI. Red color represents significant results (p < 0.05).
Gene alteration of CDK16 in pan-cancer
The established link between genetic alterations and the development of cancer forms the cornerstone of modern oncological research. In our study, we harnessed the advanced data analysis tools provided by cBioPortal to scrutinize the genetic alterations specific to CDK16 across a range of tumor types. Our analysis revealed that the genetic alterations in CDK16 are predominantly in the forms of ‘amplification’ and ‘mutation’ (Figure 5A). This finding not only contributes to the growing body of knowledge about CDK16 but also suggests its potential impact on the pathophysiology of various cancers. Moving beyond this, our investigation also focused on the relationship between CDK16 expression and copy number alterations (CNA). We observed a consistently positive correlation between these two factors across most tumor types we examined (Figure 5B). This correlation is particularly intriguing, as it hints at a more complex role of CDK16 in the genomic landscape of cancer, potentially influencing tumor behavior and response to therapies.
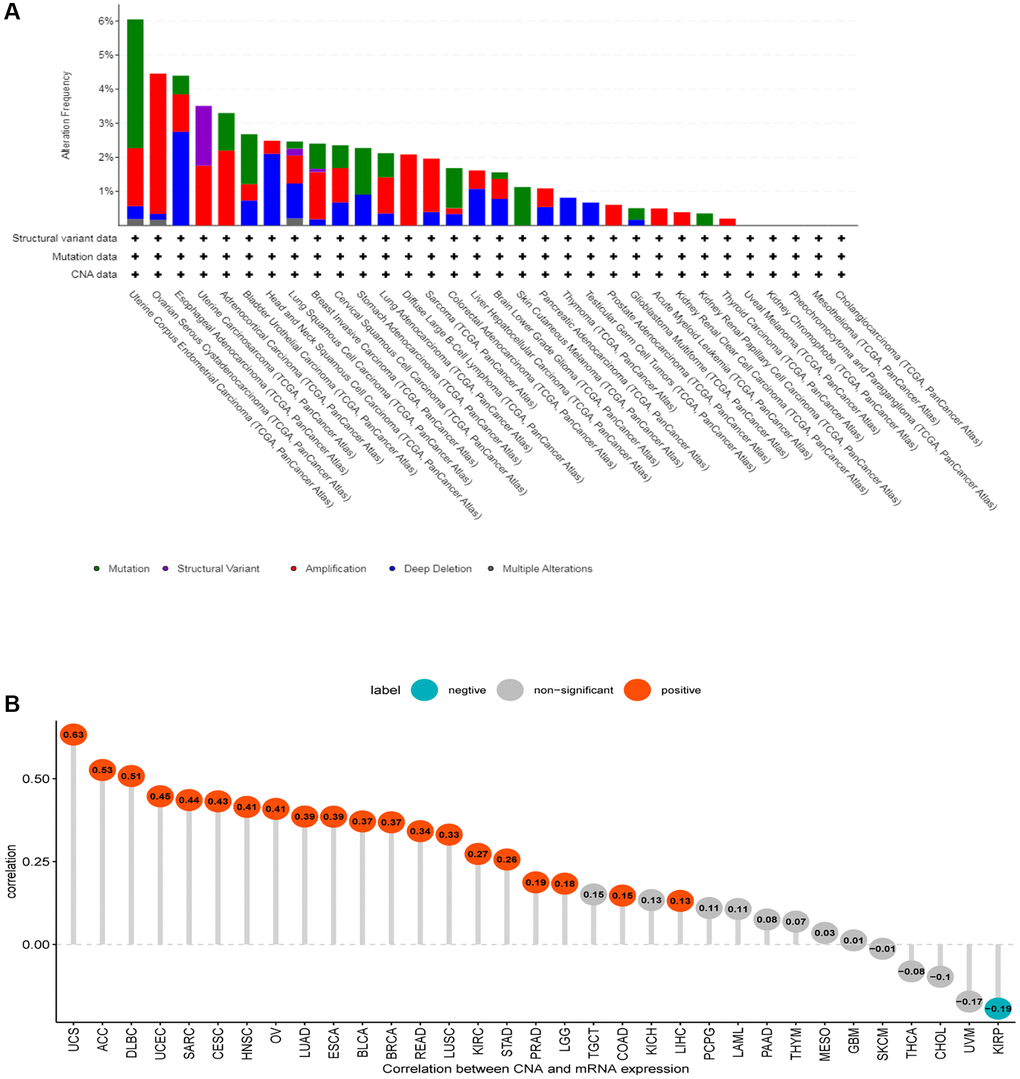
Figure 5. Gene alteration of CDK16. (A) The mutation and CNA status of CDK16 in TCGA-pan-cancer. (B) The correlation between CDK16 expression and CNA.
GSEA analysis of CDK16 in pan-cancer
In an effort to delineate the potential roles of CDK16 in the advancement of tumors, a comprehensive Gene Set Enrichment Analysis (GSEA) was carried out, focusing on the expression levels of CDK16 across a spectrum of 33 distinct cancer types. The results of this analysis are graphically represented in Figure 6A–6F. These findings shed light on the extensive involvement of CDK16 in crucial cell cycle-related pathways. Notably, the pathways include those governing cell cycle regulation and DNA repair mechanisms, which are integral to the proliferation and survival of cancer cells in various types of tumors. The analysis delineates a significant correlation between elevated CDK16 expression and the activation of genes that are pivotal in these pathways. In addition to its prominent role in cell cycle and DNA repair pathways, the study also unveiled that CDK16 is intricately involved in the regulation of immunomodulatory pathways. This encompasses both innate and adaptive immune systems, highlighting a versatile role of CDK16 in modulating the immune response within the tumor microenvironment. The implications of such involvement are twofold: firstly, CDK16’s influence on the immune system may contribute to the immune evasion strategies employed by tumors; secondly, it suggests potential therapeutic targets within these pathways that could be exploited to enhance anti-tumor immunity.
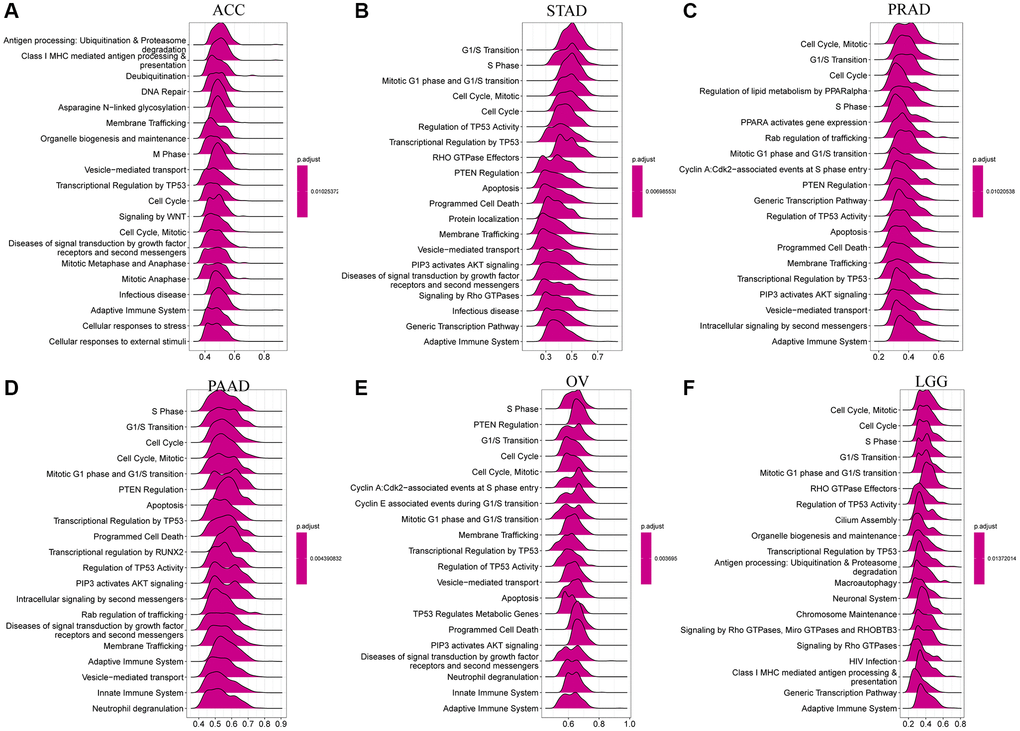
Figure 6. GSEA of CDK16 in TCGA pan-cancer. (A–F) The top 6 significant pathways of GSEA results across the indicated tumor types. Red color represents immune-related pathways.
Relationship between CDK16 and immune cell infiltration
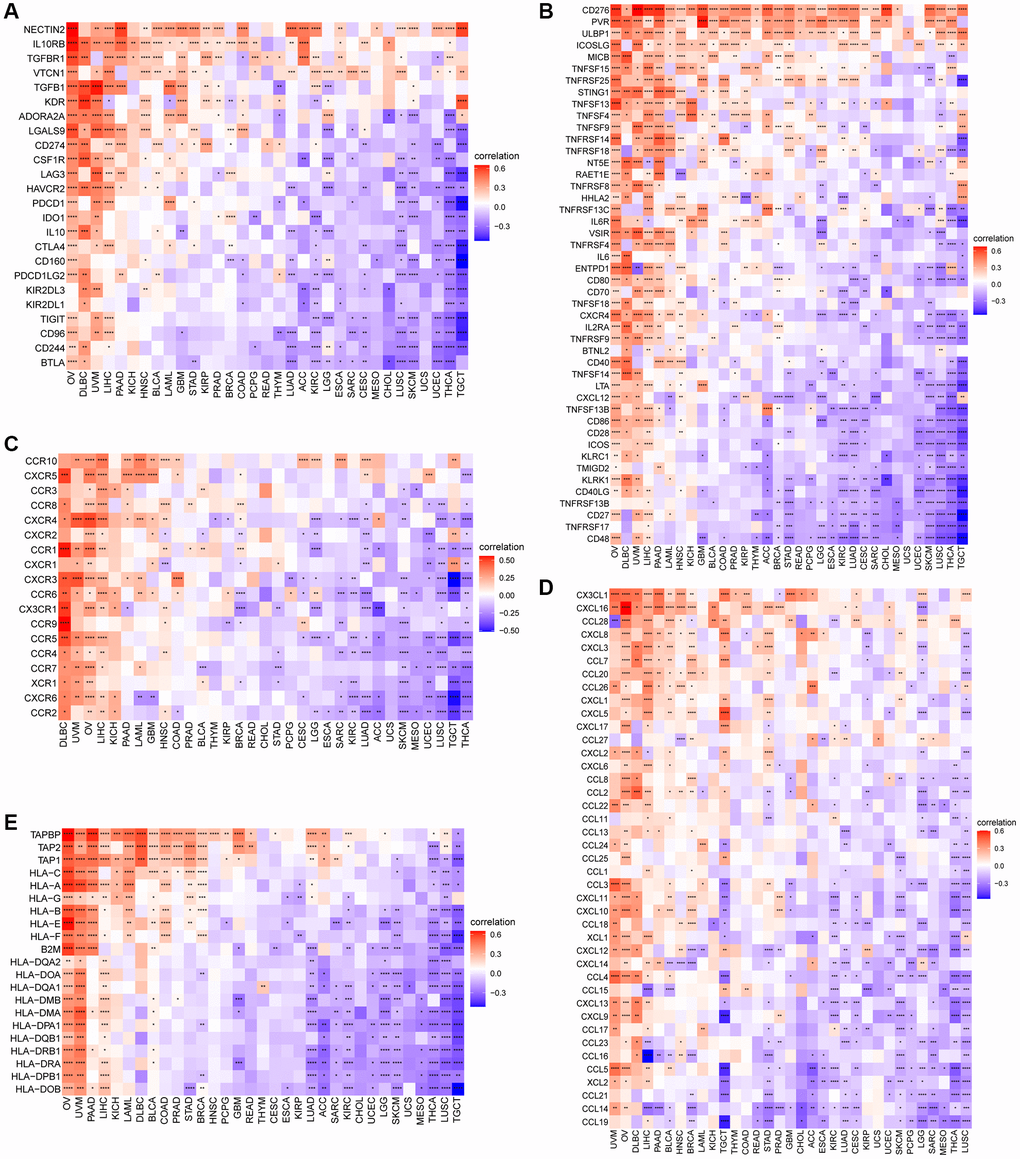
We evaluated the association between CDK16 expression and stromal as well as immune scoring, employing the ‘ESTIMATE’ algorithm for this analysis. As depicted in Figure 6, it was observed that CDK16 expression potentially influences immune scores, stromal scores, and ESTIMATE scores in the majority of tumors (Figure 7A). To validate these observations, we further examined the Tumor Microenvironment (TME)-related pathways influenced by CDK16. This included an analysis of immune-related pathways, stroma-related pathways, and DNA repair-related pathways, drawing on data sources from published research [15]. The results corroborated our initial findings, demonstrating a close association between CDK16 expression and various immune-related pathways. These pathways encompass immune checkpoints, antigen processing mechanisms, and CD8 T cell effector functions across a broad range of cancers (Figure 7B).
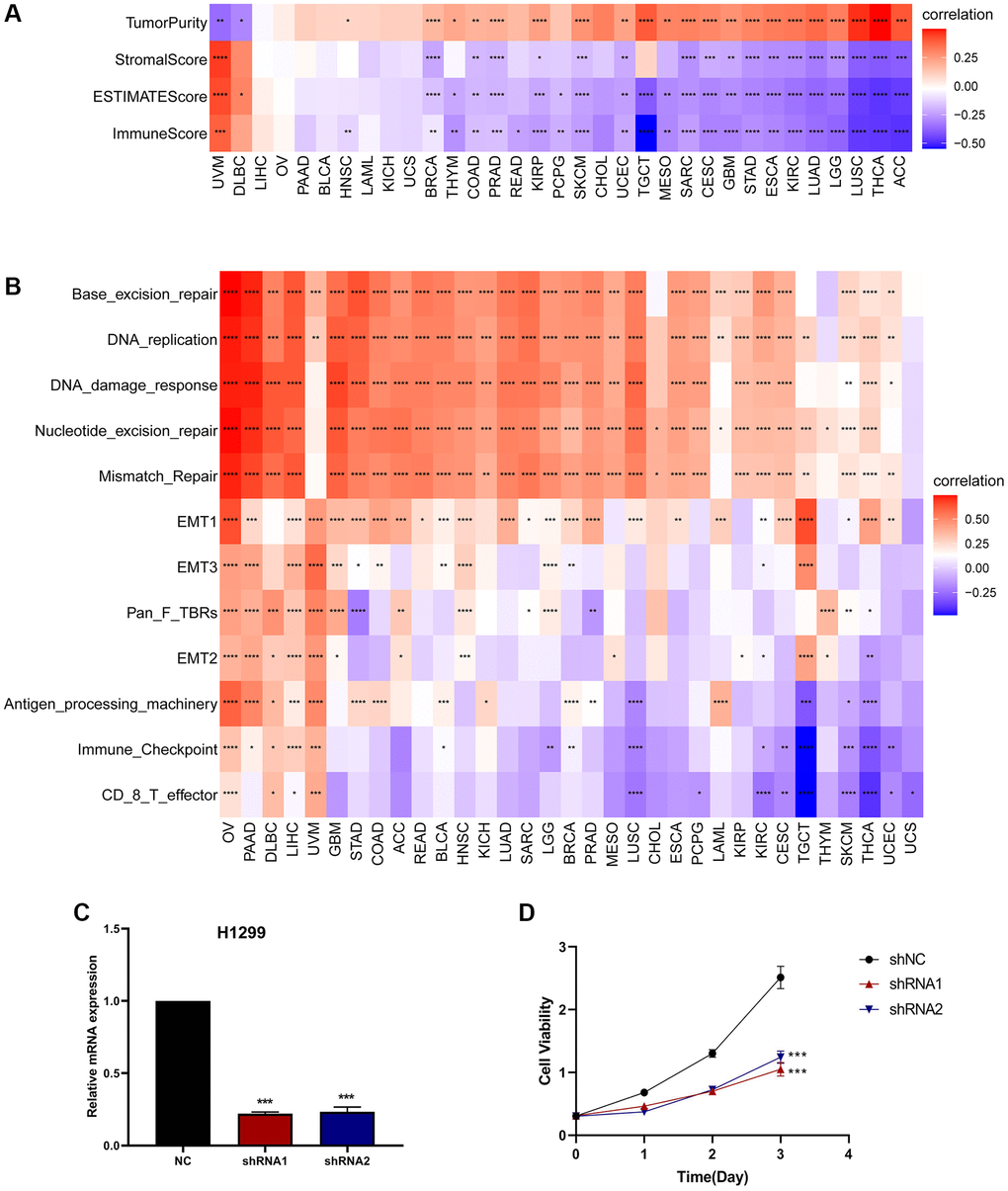
Figure 7. The relationship between CDK16 and the regulation of the tumour microenvironment. (A) Heatmap represents the correlation between CDK16 expression and TME scores in pan-cancer. (B) The relationship between CDK16 and the tumour microenvironment. (C) After H1299 cells were transfected with si-CDK16, the level of CDK16 was evaluated by qRT-PCR. (D) The cell viability of cells was examined by CCK-8 assay. Red represents positive correlation, blue represents negative correlation, and the darker the color, the stronger the correlation. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
In conjunction with our GSEA results, these findings collectively provide robust evidence supporting the expansive pro-cancer effects of CDK16. To further substantiate CDK16’s role in promoting cell proliferation, we successfully downregulated its expression in H1299 lung cancer cells using two specific CDK16 shRNAs (Figure 7C). Following this, we conducted Cell Counting Kit-8 (CCK8) assays on these cells. The results from these assays revealed a significant reduction in the viability of H1299 lung cancer cells following CDK16 knockdown (Figure 7D). This outcome lends strong support to our hypothesis that CDK16 functions as an oncogene. Furthermore, when combined with our GSEA results, it becomes evident that CDK16 exerts a broad pro-oncogenic effect across multiple cancer types. Consequently, we proceeded to delve deeper into the mechanistic action of CDK16 in the Tumor Immune Microenvironment (TIME).
Dysfunctional immune cell infiltration during tumorigenesis and progression can lead to a loss of immune system control over tumor growth and may even promote tumor development, resulting in immune escape. To explore this further, we utilized immune cell infiltration data from the TIMER2 and ImmuCellAI databases. Our investigation into the relationship between CDK16 expression and immune cell infiltration revealed that high CDK16 expression is associated with significantly poorer immune cell infiltration in pan-cancer (Figure 8 and Supplementary Figure 1).

Figure 8. The relationship between CDK16 and the immune cell infiltration. Correlation between CDK16 and different immune cells from TIMER2 database.
Immunotherapy
Immune checkpoint inhibitors (ICIs) have demonstrated potential efficacy in cancer patients exhibiting a high tumor mutation burden (TMB) or microsatellite instability (MSI). Therefore, we investigated the correlation between ICIs and CDK16 expression across multiple cancer types. Figure 6A illustrates a strong positive association between CDK16 expression and TMB in ten cancers, namely malignant MESO, PCPG, LUAD, TGCT, STAD, BLCA, OV, SKCM, BRCA, and THCA (Figure 10A). Similarly, CDK16 expression showed a positive correlation with MSI in eleven cancers, including TCGT, MESO, GBM, SARC, CESC, STAD, LUSC, LUAD, LGG, LIHC, and BRCA (Figure 10B). These findings underscore the potential involvement of CDK16 in immunotherapeutic approaches. Hence, it is reasonable to propose that CDK16 expression may influence the response to ICI treatment and prognosis in patients affected by various cancer types. To test this hypothesis, we collected data from cohorts of patients who underwent immunotherapy and evaluated CDK16 expression across different cancer types. Kaplan-Meier analysis revealed that low CDK16 expression was associated with prolonged overall survival and improved overall response rate in patients treated with nivolumab, a PD-1 inhibitor utilized in cancer immunotherapy (Figure 11A, 11B). These findings emphasize the significant role of CDK16 expression in determining patient response to immunotherapy.
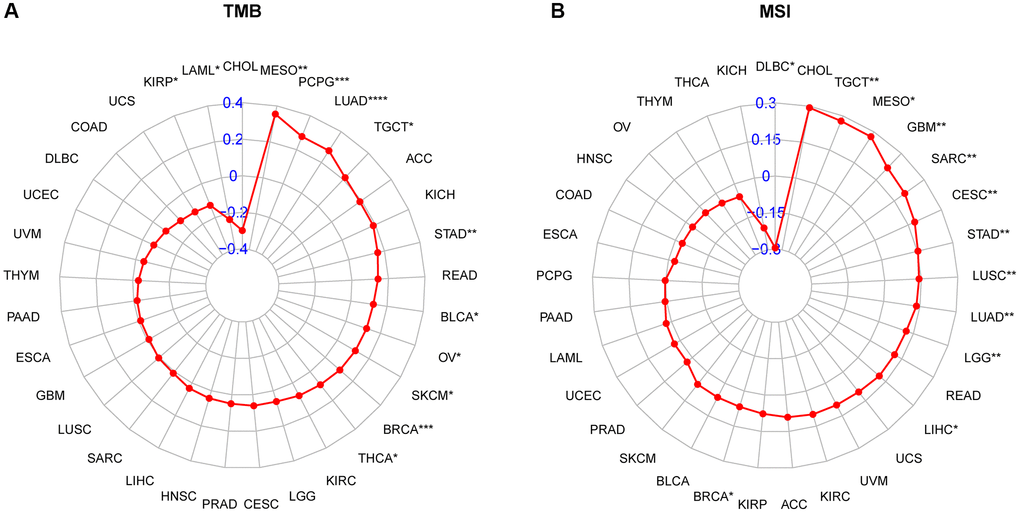
Figure 10. Relationship of CDK16 expression and tumor mutational burden (TMB), microsatellite instability (MSI). (A) Radar map illustrating the relationship between CDK16 expression and TMB. (B) Radar map illustrating the relationship between CDK16 expression and MSI. The red lines represent correlation coefficients, and blue values represent ranges. Spearman correlation test, *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001.
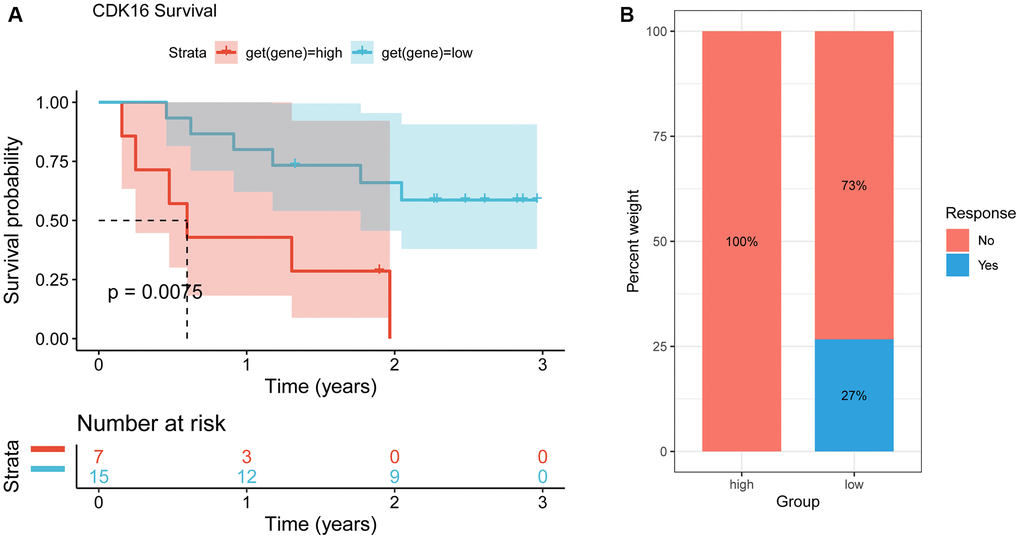
Figure 11. Correlation between CDK16 and immunotherapy. (A) The Kaplan-Meier overall survival analysis of responsive patients in high- and low- CDK16 expression groups (GSE91061). (B) The Kaplan-Meier OS analysis and percentage of responsive patients in high- and low-CDK16 expression groups of GSE91061 cohort.
Discussion
Cancer is a complex and multifaceted disease driven by dysregulation of various cellular pathways, including those involved in cell metabolism, cell death, inflammation, migration, invasion, and immune evasion [16, 17]. The tumor microenvironment (TME) plays a critical role in tumor progression and metastasis, serving as a key mediator of these processes [18, 19]. Additionally, the TME has emerged as a crucial factor in predicting clinical outcomes and determining the response of tumor cells to immunotherapy. Emerging evidence suggests that the immunosuppressive nature of the TME can facilitate tumor growth and dampen the efficacy of immunotherapy in cancer patients [20, 21]. The intricate interplay between tumor cells and the immune system within the TME creates an immunosuppressive state that enables cancer cells to evade immune detection and destruction [22, 23]. Given the significant impact of immunosuppression in cancer, there is an urgent need to identify potential biomarkers that can shed light on the immunological landscape of the TME. These biomarkers could aid in patient stratification, treatment selection, and monitoring of therapeutic response. By uncovering molecular signatures indicative of immunosuppression, researchers and clinicians can develop targeted interventions to reverse immunosuppressive mechanisms and enhance the effectiveness of immunotherapy.
CDK16 plays a crucial role in regulating biological functions rhythmically. However, its role in the initiation and progression of tumors has been largely overlooked [24]. Previous studies have only sparingly assessed the significance of CDK16 in cancer, such as its ability to modulate cancer cell growth and apoptosis through a p27-dependent mechanism [25]. Recognizing the potential importance of CDK16 in cancer, we conducted a comprehensive analysis of CDK16 in a pan-cancer context, investigating its expression, genetic alterations, biological functions, and its relationship with immune regulation and treatment response. Firstly, we observed overexpression of CDK16 in various cancer types, indicating its potential role as a promoter in tumor development. This finding aligns with existing literature supporting the concept of CDK16 as a potential cancer biomarker. Previous research has shown that high CDK16 expression is associated with tumor growth and infiltration, consistent with our findings. Furthermore, our analysis revealed that high CDK16 expression is correlated with poor prognosis, emphasizing the significance of CDK16 as a potential prognostic marker. Elevated CDK16 expression is linked to shorter overall survival and higher recurrence risk, suggesting a critical role for CDK16 in the malignant progression of tumors. This finding is in line with existing literature supporting the importance of CDK16 in assessing the prognosis of cancer patients. The use of CDK16 as a prognostic marker may help guide treatment strategies to improve patient survival. Additionally, we investigated genetic alterations in CDK16. Gene amplification and mutation were identified as the primary types of genetic alterations in CDK16, which may be related to tumor development and treatment response. These genetic alterations may lead to abnormal CDK16 activity, promoting tumor growth. This discovery provides clues for further research into the molecular mechanisms of CDK16 and may serve as a target for novel treatment strategies.
On the other hand, our gene enrichment analysis and immune regulation study revealed the multifaceted role of CDK16. CDK16 is not only involved in crucial pathways such as the cell cycle and DNA repair but also plays a role in regulating immune responses. This suggests that CDK16 may affect immune cell infiltration in the tumor microenvironment, influencing immune responses. This finding is of significant importance for understanding the potential mechanisms of immunotherapy and provides new insights for the development of novel immunotherapeutic strategies. Furthermore, we delved into the relationship between CDK16 and immune therapy, which holds significant importance in the current landscape of cancer immunotherapy. Tumor Mutational Burden (TMB) is widely recognized as a crucial determinant of immune therapy efficacy [26, 27]. Previous research has established that high TMB correlates with improved clinical responses to immune checkpoint inhibitors (ICIs) in patients with melanoma [28], head and neck cancers [29], and urothelial carcinoma [30]. TMB serves as a prognostic and predictive biomarker for human cancer immunotherapy. Microsatellite Instability (MSI) is also a key biomarker for ICI treatment response [31]. Our study has revealed a positive correlation between CDK16 expression levels and TMB as well as MSI, further emphasizing the potential significance of CDK16 in the context of immune therapy. Low CDK16 expression is associated with a more favorable prognosis in immune therapy, suggesting CDK16 as a potential predictive factor for immunotherapy. This finding is poised to provide a solid foundation for the development of personalized immune therapy strategies, aiding in the identification of patients who are likely to benefit from immune therapy.
While this research has provided valuable insights, we acknowledge its limitations. Most of the analyses were conducted using publicly available databases. To validate the research findings, further in vitro and in vivo experiments, as well as large-scale prospective studies, are required. Future research efforts should aim to delve deeper into and explore the findings of this study.
In conclusion, our research results provide a comprehensive understanding of the role of CDK16 in various cancers and lay the foundation for future research and clinical applications. Further research will help uncover the exact mechanisms of CDK16 and develop new treatment strategies to improve cancer patient outcomes. Additionally, CDK16 as a potential target for immunotherapy warrants further investigation to expand the application of immunotherapy in cancer treatment.
Author Contributions
JQ, GW, and MH acquired the data and drafted the manuscript. JQ and ZY performed the experiments, YX, ZY and LD performed statistical analysis and technical support. ZZ and YW revised and finalized the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Statement
Our study is based on open-source data from the TCGA database, so there are no ethical issues and other conflicts of interest. The Ethics Committee of local legislation and institutional has granted exemptions from approval for research related to the use of such public databases.
Funding
No funding was used for this paper.
References
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020; 70:7–30. https://doi.org/10.3322/caac.21590 [PubMed]
- 2. Zhang C, Zhang Z, Zhang G, Zhang Z, Luo Y, Wang F, Wang S, Che Y, Zeng Q, Sun N, He J. Clinical significance and inflammatory landscapes of a novel recurrence-associated immune signature in early-stage lung adenocarcinoma. Cancer Lett. 2020; 479:31–41. https://doi.org/10.1016/j.canlet.2020.03.016 [PubMed]
- 3. Kandoth C, McLellan MD, Vandin F, Ye K, Niu B, Lu C, Xie M, Zhang Q, McMichael JF, Wyczalkowski MA, Leiserson MDM, Miller CA, Welch JS, et al. Mutational landscape and significance across 12 major cancer types. Nature. 2013; 502:333–9. https://doi.org/10.1038/nature12634 [PubMed]
- 4. Garassino MC, Gadgeel S, Esteban E, Felip E, Speranza G, Domine M, Hochmair MJ, Powell S, Cheng SY, Bischoff HG, Peled N, Reck M, Hui R, et al. Patient-reported outcomes following pembrolizumab or placebo plus pemetrexed and platinum in patients with previously untreated, metastatic, non-squamous non-small-cell lung cancer (KEYNOTE-189): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet Oncol. 2020; 21:387–97. https://doi.org/10.1016/S1470-2045(19)30801-0 [PubMed]
- 5. Nishino M, Ramaiya NH, Hatabu H, Hodi FS. Monitoring immune-checkpoint blockade: response evaluation and biomarker development. Nat Rev Clin Oncol. 2017; 14:655–68. https://doi.org/10.1038/nrclinonc.2017.88 [PubMed]
- 6. Wang X, Liu R, Li S, Xia W, Guo H, Yao W, Liang X, Lu Y, Zhang H. The roles, molecular interactions, and therapeutic value of CDK16 in human cancers. Biomed Pharmacother. 2023; 164:114929. https://doi.org/10.1016/j.biopha.2023.114929 [PubMed]
- 7. Mikolcevic P, Sigl R, Rauch V, Hess MW, Pfaller K, Barisic M, Pelliniemi LJ, Boesl M, Geley S. Cyclin-dependent kinase 16/PCTAIRE kinase 1 is activated by cyclin Y and is essential for spermatogenesis. Mol Cell Biol. 2012; 32:868–79. https://doi.org/10.1128/MCB.06261-11 [PubMed]
- 8. Roskoski R
Jr . Cyclin-dependent protein serine/threonine kinase inhibitors as anticancer drugs. Pharmacol Res. 2019; 139:471–88. https://doi.org/10.1016/j.phrs.2018.11.035 [PubMed] - 9. Gu X, Shen H, Bai W, Xiang Z, Li X, Zhang R, Shi F, Li H, Zhu G, Guo S. Endometrial cancer prognosis prediction using correlation models based on CDK family genes. Front Genet. 2022; 13:1021600. https://doi.org/10.3389/fgene.2022.1021600 [PubMed]
- 10. Wang H, Liu H, Min S, Shen Y, Li W, Chen Y, Wang X. CDK16 overexpressed in non-small cell lung cancer and regulates cancer cell growth and apoptosis via a p27-dependent mechanism. Biomed Pharmacother. 2018; 103:399–405. https://doi.org/10.1016/j.biopha.2018.04.080 [PubMed]
- 11. Yanagi T, Krajewska M, Matsuzawa S, Reed JC. PCTAIRE1 phosphorylates p27 and regulates mitosis in cancer cells. Cancer Res. 2014; 74:5795–807. https://doi.org/10.1158/0008-5472.CAN-14-0872 [PubMed]
- 12. Li X, Li J, Xu L, Wei W, Cheng A, Zhang L, Zhang M, Wu G, Cai C. CDK16 promotes the progression and metastasis of triple-negative breast cancer by phosphorylating PRC1. J Exp Clin Cancer Res. 2022; 41:149. https://doi.org/10.1186/s13046-022-02362-w [PubMed]
- 13. Zhang C, Lv H, Zhang F, Ji A. LncRNA HCG18 facilitates melanoma progression by modulating miR-324-5p/CDK16 axis. Am J Transl Res. 2022; 14:1246–57. [PubMed]
- 14. Liu Q, Wang C, Jiang Z, Li S, Li F, Tan HB, Yue SY. circRNA 001306 enhances hepatocellular carcinoma growth by up-regulating CDK16 expression via sponging miR-584-5p. J Cell Mol Med. 2020; 24:14306–15. https://doi.org/10.1111/jcmm.16047 [PubMed]
- 15. Thorsson V, Gibbs DL, Brown SD, Wolf D, Bortone DS, Ou Yang TH, Porta-Pardo E, Gao GF, Plaisier CL, Eddy JA, Ziv E, Culhane AC, Paull EO, et al, and Cancer Genome Atlas Research Network. The Immune Landscape of Cancer. Immunity. 2018; 48:812–30.e14. https://doi.org/10.1016/j.immuni.2018.03.023 [PubMed]
- 16. Samstein RM, Lee CH, Shoushtari AN, Hellmann MD, Shen R, Janjigian YY, Barron DA, Zehir A, Jordan EJ, Omuro A, Kaley TJ, Kendall SM, Motzer RJ, et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat Genet. 2019; 51:202–6. https://doi.org/10.1038/s41588-018-0312-8 [PubMed]
- 17. Wu G, He M, Yin X, Wang W, Zhou J, Ren K, Chen X, Xue Q. The Pan-Cancer Landscape of Crosstalk Between TRP Family and Tumour Microenvironment Relevant to Prognosis and Immunotherapy Response. Front Immunol. 2022; 13:837665. https://doi.org/10.3389/fimmu.2022.837665 [PubMed]
- 18. Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, Merad M, Coussens LM, Gabrilovich DI, Ostrand-Rosenberg S, Hedrick CC, Vonderheide RH, Pittet MJ, Jain RK, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018; 24:541–50. https://doi.org/10.1038/s41591-018-0014-x [PubMed]
- 19. Xie J, Zhang J, Tian W, Zou Y, Tang Y, Zheng S, Wong CW, Deng X, Wu S, Chen J, Mo Y, Xie X. The Pan-Cancer Multi-Omics Landscape of FOXO Family Relevant to Clinical Outcome and Drug Resistance. Int J Mol Sci. 2022; 23:15647. https://doi.org/10.3390/ijms232415647 [PubMed]
- 20. da Silva PHR, Borges BC, Uehara IA, Soldi LR, de Araújo RA, Silva MJB. Chemokines and the extracellular matrix: Set of targets for tumor development and treatment. Cytokine. 2021; 144:155548. https://doi.org/10.1016/j.cyto.2021.155548 [PubMed]
- 21. Pan S, Sun S, Liu B, Hou Y. Pan-cancer Landscape of the RUNX Protein Family Reveals their Potential as Carcinogenic Biomarkers and the Mechanisms Underlying their Action. J Transl Int Med. 2022; 10:156–74. https://doi.org/10.2478/jtim-2022-0013 [PubMed]
- 22. Greten FR, Grivennikov SI. Inflammation and Cancer: Triggers, Mechanisms, and Consequences. Immunity. 2019; 51:27–41. https://doi.org/10.1016/j.immuni.2019.06.025 [PubMed]
- 23. Rundqvist H, Veliça P, Barbieri L, Gameiro PA, Bargiela D, Gojkovic M, Mijwel S, Reitzner SM, Wulliman D, Ahlstedt E, Ule J, Östman A, Johnson RS. Cytotoxic T-cells mediate exercise-induced reductions in tumor growth. Elife. 2020; 9:e59996. https://doi.org/10.7554/eLife.59996 [PubMed]
- 24. Cole AR. PCTK proteins: the forgotten brain kinases? Neurosignals. 2009; 17:288–97. https://doi.org/10.1159/000231895 [PubMed]
- 25. Jia Q, Xie B, Zhao Z, Huang L, Wei G, Ni T. Lung cancer cells expressing a shortened CDK16 3'UTR escape senescence through impaired miR-485-5p targeting. Mol Oncol. 2022; 16:1347–64. https://doi.org/10.1002/1878-0261.13125 [PubMed]
- 26. Xie J, Luo X, Deng X, Tang Y, Tian W, Cheng H, Zhang J, Zou Y, Guo Z, Xie X. Advances in artificial intelligence to predict cancer immunotherapy efficacy. Front Immunol. 2023; 13:1076883. https://doi.org/10.3389/fimmu.2022.1076883 [PubMed]
- 27. Wu G, Dong Y, Hu Q, Ma H, Xu Q, Xu K, Chen H, Yang Z, He M. HGH1 and the immune landscape: a novel prognostic marker for immune-desert tumor microenvironment identification and immunotherapy outcome prediction in human cancers. Cell Cycle. 2023; 22:1969–85. https://doi.org/10.1080/15384101.2023.2260163 [PubMed]
- 28. Li X, Liu R, Su X, Pan Y, Han X, Shao C, Shi Y. Harnessing tumor-associated macrophages as aids for cancer immunotherapy. Mol Cancer. 2019; 18:177. https://doi.org/10.1186/s12943-019-1102-3 [PubMed]
- 29. Snyder A, Makarov V, Merghoub T, Yuan J, Zaretsky JM, Desrichard A, Walsh LA, Postow MA, Wong P, Ho TS, Hollmann TJ, Bruggeman C, Kannan K, et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N Engl J Med. 2014; 371:2189–99. https://doi.org/10.1056/NEJMoa1406498 [PubMed]
- 30. Kowanetz M, Zou W, Shames D, Cummings C, Rizvi N, Spira A, Frampton G, Leveque V, Flynn S, Mocci S, Shankar G, Funke R, Ballinger M, et al. OA20.01 Tumor Mutation Burden (TMB) is Associated with Improved Efficacy of Atezolizumab in 1L and 2L+ NSCLC Patients. Oncol Res Treat. 2017; 40:220. https://doi.org/10.1016/j.jtho.2016.11.343
- 31. Xie J, Zheng Z, Tuo L, Deng X, Tang H, Peng C, Zou Y. Recent advances in exosome-based immunotherapy applied to cancer. Front Immunol. 2023; 14:1296857. https://doi.org/10.3389/fimmu.2023.1296857 [PubMed]