Introduction
Breast cancer (BC) is a prevalent and predominantly female tumor disease that can be classified into different subtypes based on molecular features, histopathological characteristics, and clinical outcomes. Based on the expression levels of hormone receptors (estrogen receptor, progesterone receptor) and HER2/neu (human epidermal growth factor receptor 2), breast cancer can be classified into three subtypes, including hormone receptor-positive (ER+ or PR+)/HER2-negative (HER2-) subtype, hormone receptor-negative (ER- and PR-)/HER2-positive (HER2+) subtype, and hormone receptor-negative/HER2-negative (triple-negative) subtype. Breast cancer has a high incidence and mortality rate [1] with approximately 1.6 million new cases and a mortality rate of around 6.4% globally each year [2, 3] with approximately 1.6 million new cases and a mortality rate of around 6.4% globally each year [4]. Despite significant advances in breast cancer treatment in recent years, the mortality rate remains high, and many patients are diagnosed and treated late due to the lack of effective methods for early diagnosis and screening [4]. The pathogenesis of breast cancer is complex and multifactorial, involving genetic and environmental factors. Many genes, such as the tumor suppressor genes BRCA1 and BRCA2, have been found to be closely associated with the occurrence and development of breast cancer [5]. Approximately 75% of breast cancer patients have mutations or low expression of BRCA1 or BRCA2 [5]. Therefore, exploring the genes and proteins that are abnormally expressed during the occurrence and development of breast cancer, studying the molecular characteristics involved in breast cancer, and identifying new prognostic biomarkers are of great significance for better understanding of its pathological mechanism, developing new effective diagnostic and therapeutic methods, and improving clinical outcomes.
Ferroptosis is a novel form of programmed cell death characterized by the imbalance of redox status and iron metabolism in cells. Due to the high energy demand of tumor cells, they exhibit high levels of oxidative reactions and obvious iron addiction compared to normal cells, with high levels of iron death markers, such as reactive oxygen species (ROS) and ferrous ions (Fe2+) [6]. Studies have shown that inducing ferroptosis of breast cancer cells is an important approach to improve or treat breast cancer [7, 8]. Many clinical drugs such as metformin [7], simvastatin [9], lapatinib [10], etc., can improve the occurrence, development, and prognosis of breast cancer by inducing ferroptosis. Thus, exploring ferroptosis-related genes closely associated with breast cancer is of great significance for studying its pathogenesis, diagnosis, treatment, and prognosis assessment.
In this study, we applied bioinformatics and machine learning methods to screen ferroptosis-related genes closely associated with breast cancer and verified their functions through in vitro experiments. This study will further enrich our understanding of the role of ferroptosis in the occurrence, development, and prognosis of breast cancer and provide a basis for the molecular mechanism study, diagnosis, treatment, and prognosis assessment of breast cancer.
Materials and Methods
Data download and preprocessing
Transcriptome data and clinical information related to breast cancer were obtained from The Cancer Genome Atlas (TCGA) database (https://portal.gdc.cancer.gov). A total of 1204 transcriptome data samples were included, consisting of 1091 breast cancer tumor tissue samples and 113 adjacent normal tissue samples. The clinical information contained 1097 breast cancer patients. After excluding breast cancer patients without transcriptome data and breast cancer patients with overall survival time less than 10 days, clinical data of 1060 breast cancer patients were selected for prognostic analysis. Table 1 shows the statistical description of the clinical data of 1060 breast cancer patients. The transcriptome data were processed as follows: the Ensemble ID in the transcriptome dataset was converted to gene names based on the gene annotation information in the TCGA database, and a new expression matrix was generated by extracting the protein-coding genes in the dataset. The expression matrix was then normalized and standardized. A total of 259 ferroptosis-related genes were obtained from the FerrDB database (http://www.zhounan.org/ferrdb/).
Table 1. Clinical characteristics of the 1060 breast cancer patients from TCGA database.
| Characteristics | Alive (N = 913) | Dead (N = 147) | Total (N = 1060) |
| Gender | |||
| Female | 902 (85.09%) | 146 (13.77%) | 1048 (98.87%) |
| Male | 11 (1.04%) | 1 (0.09%) | 12 (1.13%) |
| Histological type | |||
| Infiltrating Carcinoma NOS | 1 (0.10%) | 0 (0%) | 1 (0.10%) |
| Infiltrating Ductal Carcinoma | 653 (66.09%) | 104 (10.53%) | 757 (76.62%) |
| Infiltrating Lobular Carcinoma | 177 (17.91%) | 22 (2.23%) | 199 (20.14%) |
| Medullary Carcinoma | 3 (0.30%) | 2 (0.20%) | 5 (0.51%) |
| Metaplastic Carcinoma | 7 (0.71%) | 1 (0.10%) | 8 (0.81%) |
| Mucinous Carcinoma | 14 (1.42%) | 3 (0.30%) | 17 (1.72%) |
| Not Available | 1 (0.10%) | 0 (0%) | 1 (0.10%) |
| Pathologic stage | |||
| Stage I | 165 (15.73%) | 16 (1.53%) | 181 (17.25%) |
| Stage II | 537 (51.19%) | 63 (6.01%) | 600 (57.20%) |
| Stage III | 195 (18.59%) | 43 (4.10%) | 238 (22.69%) |
| Stage IV | 4 (0.38%) | 15 (1.43%) | 19 (1.81%) |
| Stage X | 5 (0.48%) | 6 (0.57%) | 11 (1.05%) |
| Pathologic T | |||
| T1 | 245 (23.11%) | 33 (3.11%) | 278 (26.23%) |
| T2 | 538 (50.75%) | 75 (7.08%) | 613 (57.83%) |
| T3 | 106 (10.00%) | 24 (2.26%) | 130 (12.26%) |
| T4 | 22 (2.08%) | 14 (1.32%) | 36 (3.40%) |
| TX | 2 (0.19%) | 1 (0.09%) | 3 (0.28%) |
| Pathologic N | |||
| N0 | 455 (42.92%) | 43 (4.06%) | 498 (46.98%) |
| N1 | 295 (27.83%) | 59 (5.57%) | 354 (33.40%) |
| N2 | 97 (9.15%) | 21 (1.98%) | 118 (11.13%) |
| N3 | 58 (5.47%) | 15 (1.42%) | 73 (6.89%) |
| NX | 8 (0.75%) | 9 (0.85%) | 17 (1.60%) |
| Pathologic M | |||
| M0 | 760 (72.11%) | 117 (11.10%) | 877 (83.21%) |
| M1 | 4 (0.38%) | 17 (1.61%) | 21 (1.99%) |
| MX | 143 (13.57%) | 13 (1.23%) | 156 (14.80%) |
| Age | |||
| Mean ± SD | 57.76 ± 12.56 | 60.47 ± 14.43 | 58.13 ± 12.86 |
| Median (min-max) | 58 (26.00, 89.00) | 62.00 (26.00, 89.00) | 58.00 (26.00, 89.00) |
| Overall survival time | |||
| Mean ± SD | 1210.05 ± 1158.61 | 1617.74 ± 1309.93 | 1266.59 ± 1188.48 |
| Median (min-max) | 791.00 (010.00, 8605.00) | 1174.00 (30.00, 7455.00) | 876.50 (10.00, 8605.00) |
Materials and reagents
The human breast cancer cell line MCF-7 was purchased from the Cell Bank of the Chinese Academy of Sciences. DMEM medium and fetal bovine serum were purchased from Gibco (Grand Island, NY, USA); Op-timen medium was purchased from Procell Life Science and Technology (Wuhan, China); Lipofectamine 2000 transfection reagent was purchased from Invitrogen (Thermo Fisher Scientific, Carlsbad, CA, USA). Small interfering RNA targeting SLC2A1 (siRNA-SLC2A1), pcDNA-TXNIP overexpression plasmid (TXNIP-OE), and pcDNA-ATF3 overexpression plasmid (ATF3-OE) were designed and synthesized by Sangon Biotech (Shanghai, China); SLC2A1, TXNIP, and ATF3 primers were synthesized by Sangon Biotech (Shanghai, China). Primary antibodies against SLC2A1 (#4393), TXNIP (#13113), and ATF3 (#12691) were purchased from Abcam (Cambridge, UK), and anti-actin was purchased from Proteintech Group (Wuhan, China). HRP-conjugated secondary antibody was purchased from Elabscience Biotechnology (Wuhan, China). BCA protein assay kit was purchased from Elabscience Biotechnology (Wuhan, China). TRIzol reagent, PrimeScript RT reagent Kit, and SYBR Green Master Mix were purchased from Thermo Fisher Scientific (Wilmington, NC, USA). Reactive oxygen species (ROS) and Fe2+ detection assay kits were purchased from Yeasen Biotechnology (Shanghai, China), glutathione (GSH) detection assay kits were purchased from Beijing Solarbio Science and Technology (Beijing, China), malondialdehyde (MDA) assay kits were purchased from Nanjing Jiancheng Biological Engineering Research Institute (Nanjing, China). CCK-8 assay kit was purchased from New Cell and Molecular Biotech (Suzhou, China). GIEMS staining solution was purchased from Gibco (Grand Island, NY, USA).
Identification of differentially expressed genes associated with ferroptosis
Differential expression analysis of transcriptome data was performed using the R package “limma”. A fold change threshold of 2.5 and a P < 0.05 were set to identify differentially expressed genes (DEGs) between tumor (BC group, n = 1091) and adjacent tissues (NC group, n = 113). To further investigate the relationship between DEGs and ferroptosis, the identified DEGs were compared with ferroptosis-related genes downloaded from the FerrDB database (http://www.zhounan.org/ferrdb). By overlapping the two gene sets, a list of ferroptosis-related DEGs was identified, which may play critical roles in breast cancer.
Functional enrichment analysis
To further investigate the biological functions and pathways associated with ferroptosis, the differentially expressed genes (DEGs) related to ferroptosis were input into the DAVID database (https://david.ncifcrf.gov/) for functional enrichment analysis. Gene Ontology (GO) annotation and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis were performed to identify GO terms and KEGG pathways that were significantly enriched (P < 0.05). The results of the functional enrichment analysis were visualized using R software. The identified enriched GO terms and KEGG pathways could provide insights into the molecular mechanism of ferroptosis in breast cancer.
Cell culture and transfection
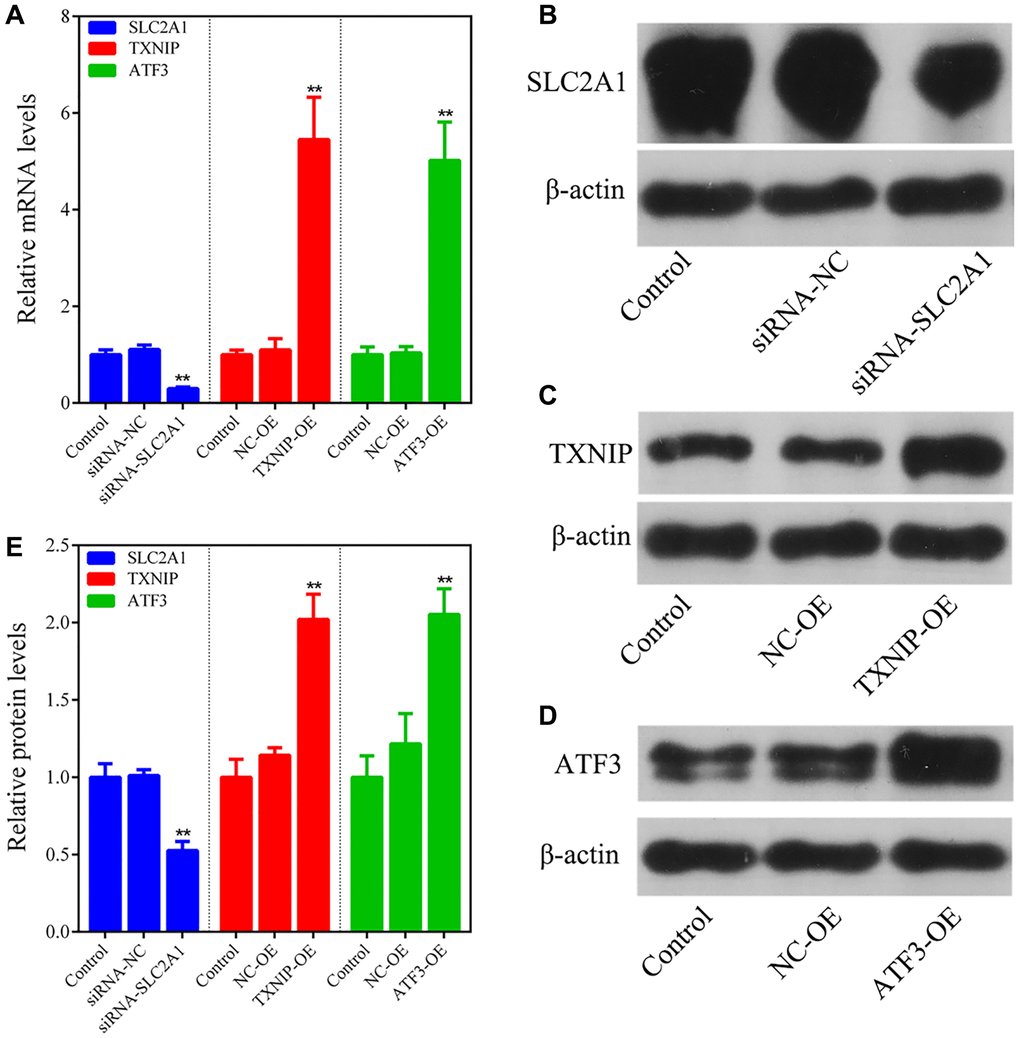
The breast cancer cell line MCF-7 was cultured in high-glucose DMEM supplemented with 10% FBS and 1% penicillin-streptomycin at 37°C and 5% CO2 in a cell culture incubator. Cells in logarithmic growth phase were digested with trypsin and seeded into 6-well plates at an appropriate density. After the cells grew to approximately 60% confluency, Lipofectamine 2000 transfection reagent was used to transfect siRNA-SLC2A1, TXNIP overexpression plasmid, and ATF3 overexpression plasmid mixed with Opti-MEM medium into MCF-7 cells. Empty plasmids (siRNA-NC and NC-OE) were also transfected into MCF-7 cells as negative controls. After incubating for 6 h in the cell culture incubator, the medium was replaced, and the transfection efficiency was detected by q-PCR and Western blotting 24 h later. The cells were divided into four groups according to the treatment: normal control group (Control), SLC2A1 knockdown group (siRNA-SLC2A1), TXNIP overexpression group (TXNIP-OE), and ATF3 overexpression group (ATF3-OE). The cells in each group were used for subsequent experiments 24 h after transfection.
Quantitative reverse transcription-PCR (q-PCR)
First, total RNA was extracted from the transfected cells and normal cells using the TRIzol reagent according to the manufacturer’s instructions. The RNA concentration and purity were determined using a NanoDrop spectrophotometer (Applied Thermo Fisher Scientific, Wilmington, NC, USA). Then, cDNA was synthesized from the extracted RNA using the PrimeScript RT reagent Kit according to the manufacturer’s instructions. Finally, q-PCR was performed using the SYBR Green PCR Master Mix on the ABI 7500 Real-Time PCR System (Applied Biosystems, Wilmington, USA). The primers utilized in q-PCR are detailed in Table 2.
Table 2. Primer sequences for q-PCR assay.
| Gene | Forward primer (5′–3′) | Reverse primer (5′–3′) | Product size (bp) |
| SLC2A1 | 5′-GGCTTCTCCAACTGGACCTC-3′ | 5′-CCGGAAGCGATCTCATCGAA-3′ | 176 |
| TXNIP | 5′-ATGCCACCCAAGCATTCCTTA-3′ | 5′-AGGAAGCTCAAAGCCGAACT-3′ | 153 |
| ATF3 | 5′-GGAGTGCCTGCAGAAAGAGT-3′ | 5′-CCATTCTGAGCCCGGACAAT-3′ | 147 |
| β-actin (ACTB) | 5′-ACACAGTGCTGTCTGGTGG-3′ | 5′-CAGAGTACTTGCGCTCAGGA-3′ | 129 |
Western blot
To validate the expression of ferroptosis-related genes in breast cancer cell line MCF-7, Western blot analysis was performed. Briefly, after transfection with siRNA-SLC2A1, TXNIP overexpression plasmid, and ATF3 overexpression plasmid, respectively, the cells were harvested and lysed using RIPA buffer containing protease inhibitor cocktail. The protein concentration was determined by the BCA protein assay kit. Equal amounts of protein were separated by SDS-PAGE and transferred onto PVDF membranes. After blocking with 5% non-fat milk, the membranes were incubated with primary antibodies against the selected ferroptosis-related genes and β-actin (loading control) at 4°C overnight. After washing with TBST, the membranes were incubated with HRP-conjugated secondary antibodies at room temperature for 1 h. The protein bands were visualized using ECL reagents and analyzed using ImageJ software. The relative expression levels of the ferroptosis-related genes were normalized to β-actin.
Cell proliferation assay
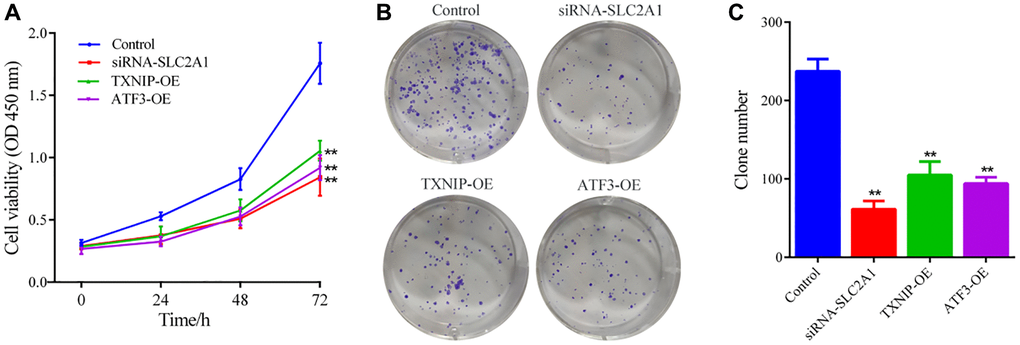
To assess cell proliferation, single-cell suspensions of the Control group and experimental groups (siRNA-SLC2A1, TXNIP-OE, ATF3-OE) were seeded into 96-well plates at a density of approximately 2000 cells per well and incubated at 37°C and 5% CO2 in a cell culture incubator for 24, 48, and 72 h. At each time point, 10 μL of CCK8 working solution was added to each well, and the cells were incubated for 30 min at 37°C. The absorbance (A) was measured at 450 nm wavelength according to the instructions of the CCK8 kit.
Colony formation assay
To evaluate colony formation ability, cells from each group were digested with trypsin and made into single-cell suspensions. Approximately 1000 cells were seeded into each well of a 6-well plate and cultured at 37°C and 5% CO2 in a cell culture incubator for 21 days, with the medium changed every 3 days. After incubation, the cells were stained with GIEMS solution for 15 min and photographed under a microscope.
Detection of cell ferroptosis
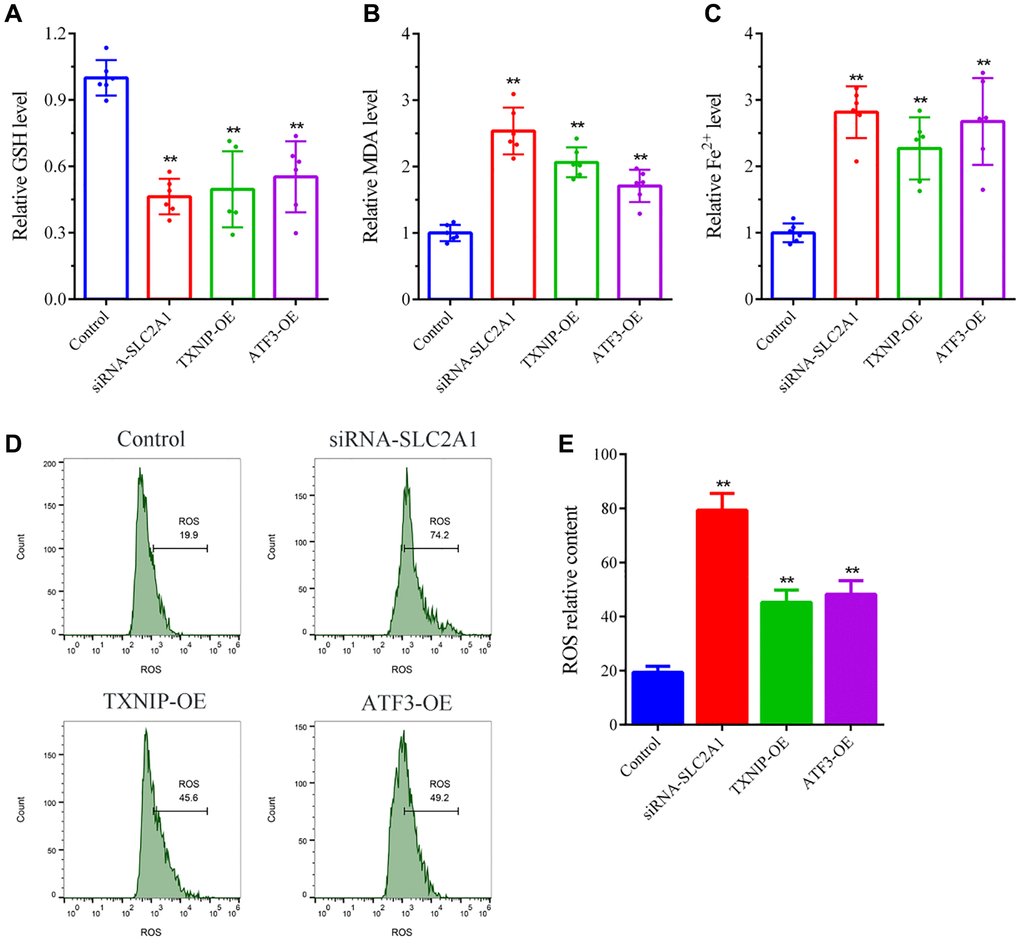
The levels of GSH, MDA, and ferrous iron ions (Fe2+) in cells were measured according to the instructions of the detection kit. First, the transfected cells were cultured for 24 h, and then collected and lysed using RIPA cell lysis buffer containing 1% protease and phosphatase inhibitor. The lysates were centrifuged at 13000 × g for 5 min, and the supernatant was collected. The cell lysates were mixed with the assay reagents, and the absorbance was measured using a microplate reader. The detection wavelengths for GSH, MDA, and Fe2+ were 412 nm, 532 nm, and 593 nm, respectively. Furthermore, the reactive oxygen species (ROS) levels in cells were measured according to the instructions of a ROS detection kit. Transfected cells were cultured for 24 h and collected to prepare a single-cell suspension. A suitable amount of 10 μmol/L DCFH-DA probe solution was added to the cells, and they were incubated at 37°C for 30 min. After washing the cells twice with serum-free cell culture medium, the fluorescence intensity was measured using a flow cytometer with an excitation wavelength of 488 nm and an emission wavelength of 525 nm.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 8.3.0 software. All experimental data were repeated at least three times and presented as mean ± standard deviation (SD). Differences between groups were compared using the independent sample t-test and one-way analysis of variance (ANOVA). A p-value less than 0.05 was considered statistically significant.
Availability of data and materials
The data sets supporting the results of this article are included within the article. All data, models, and code generated or used during the study appear in the submitted article.
Results
Screening of differentially expressed genes associated with ferroptosis
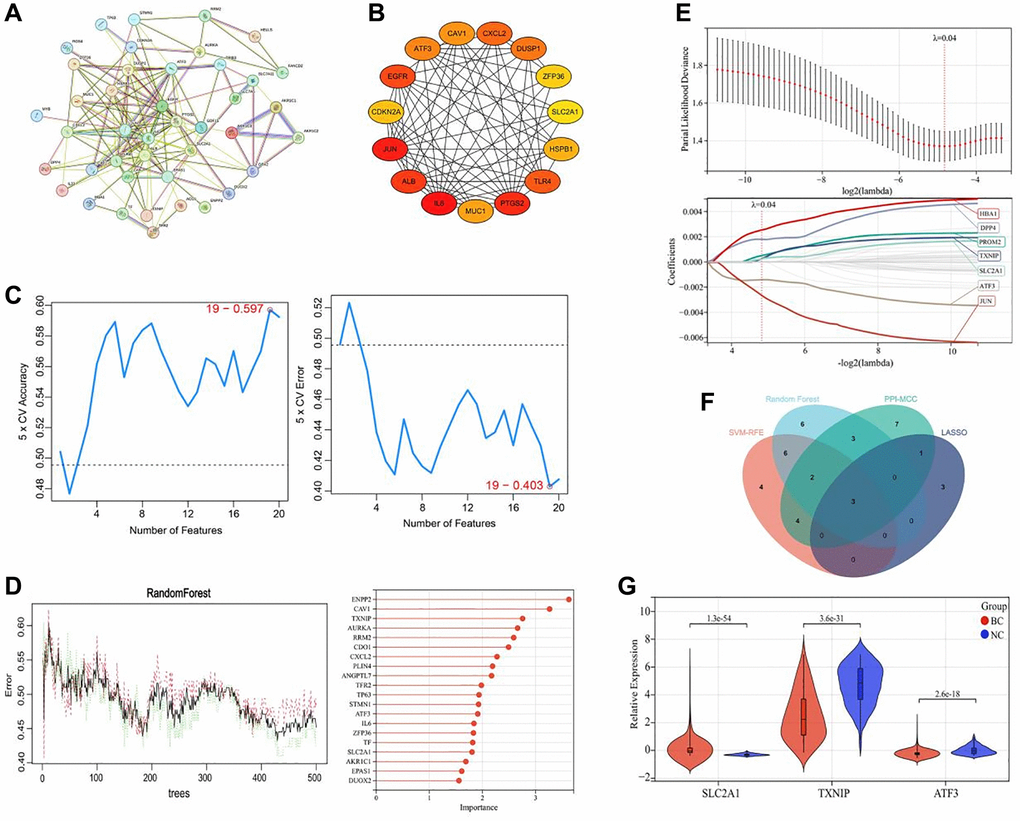
A total of 3031 differentially expressed genes (DEGs) were identified between tumor (BC group) and adjacent tissues (NC group), including 1275 significantly upregulated genes and 1756 significantly downregulated genes (Figure 1A). To further investigate the relationship between DEGs and ferroptosis, the identified DEGs were compared with ferroptosis-related genes downloaded from the Ferroptosis Database (FerrDB). By overlapping the two gene sets, a list of 52 ferroptosis-related DEGs was identified, which may play critical roles in breast cancer (Figure 1B). The expression levels of these ferroptosis-related DEGs are presented in a heatmap (Figure 1C).
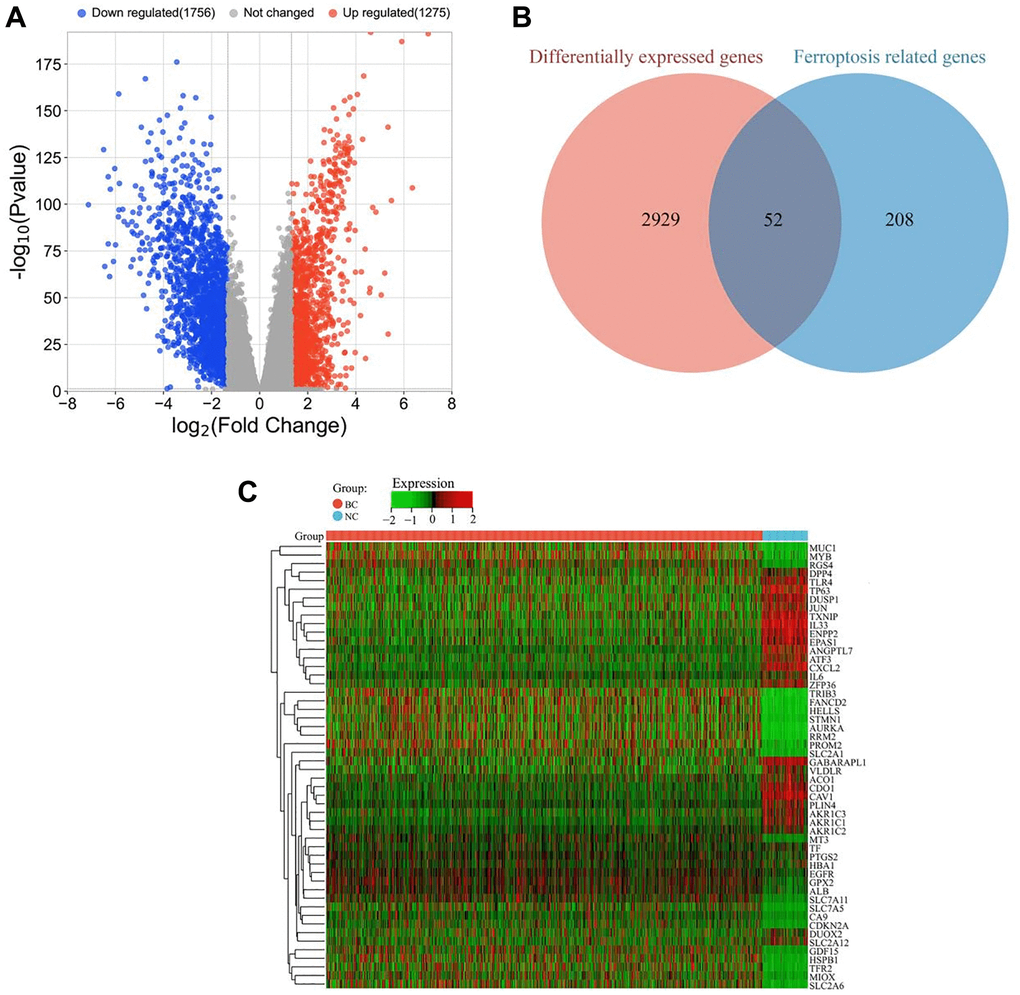
Figure 1. Identification of ferroptosis-related DEGs in breast cancer. (A) Volcano plot of all DEGs between tumor (BC group, n = 1091) and adjacent tissues (NC group, n = 113). Red and blue dots represent significantly upregulated and downregulated genes, respectively. (B) Venn diagram showing the overlap between ferroptosis-related genes and DEGs. (C) Heatmap of differentially expressed ferroptosis-related genes in tumor (BC group, n = 1091) and adjacent tissues (NC group, n = 113). The color scale represents the relative expression level of each gene, with red indicating high expression and green indicating low expression.
Functional enrichment analysis
The GO analysis showed that the ferroptosis-related DEGs were mainly involved in biological process (BPs) such as positive regulation of transcription from RNA polymerase II promoter, negative regulation of transcription from RNA polymerase II promoter, and response to hypoxia, as well as cellular components (CCs) such as cytosol, plasma membrane, nucleus, and extracellular exosome. In addition, they were enriched in molecular functions (MFs) such as identical protein binding, protein kinase binding, ubiquitin protein ligase binding, and protein homodimerization activity. These results suggest that the ferroptosis-related DEGs may play important roles in the regulation of transcription, hypoxia response, and protein-protein interactions (Figure 2A).
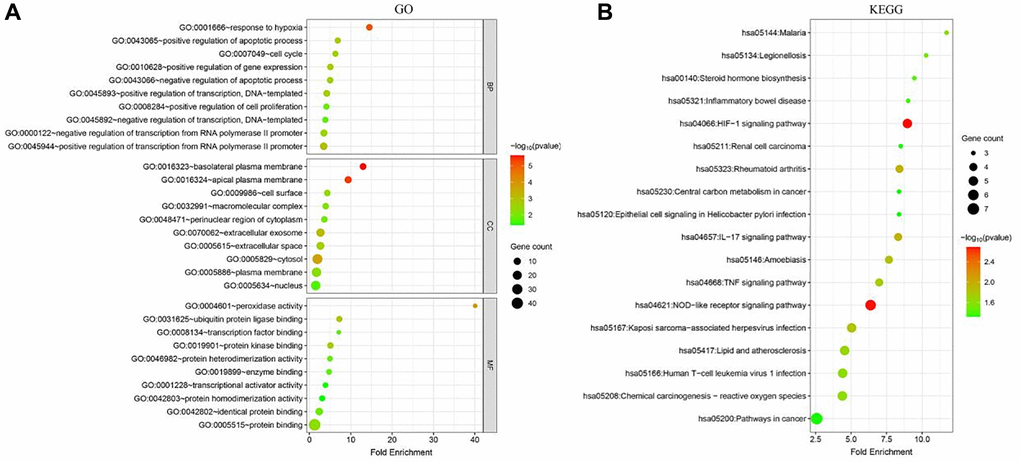
Figure 2. Functional enrichment analysis of ferroptosis-related DEGs in breast cancer. (A) Gene Ontology (GO) analysis of ferroptosis-related DEGs. The top 10 enriched terms in each category (biological process, cellular component, and molecular function) are shown. (B) Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis of ferroptosis-related DEGs. The top 10 enriched pathways are shown.
The KEGG pathway analysis revealed that the ferroptosis-related DEGs were significantly enriched in several cancer-related pathways, including Pathways in cancer, Central carbon metabolism in cancer, HIF-1 signaling pathway, and NOD-like receptor signaling pathway. These pathways are known to be associated with cancer development and progression, suggesting that the ferroptosis-related DEGs may play important roles in breast cancer (Figure 2B).
Construction of the prognostic risk model
We used multivariate Cox regression analysis to construct a risk score model, which was calculated as follows: RiskScore = SLC2A1 × 0.813354160440223 − TXNIP × 0.473133422604816 − ATF3 × 0.21764547803389237. The survival status plot of breast cancer patients based on the prognostic model is shown in Figure 4A. Based on the risk score of each patient, all tumor samples were divided into high-risk and low-risk groups. The results showed that the overall survival rate of the high-risk group was significantly lower than that of the low-risk group (P < 0.001), as shown in Figure 4B. ROC curve analysis validated the good predictive ability of the prognostic model for survival. The AUC values of the prognostic model for predicting the survival of breast cancer patients at 365 days, 1095 days, and 1825 days were 0.79, 0.73, and 0.73, respectively, as shown in Figure 4C.
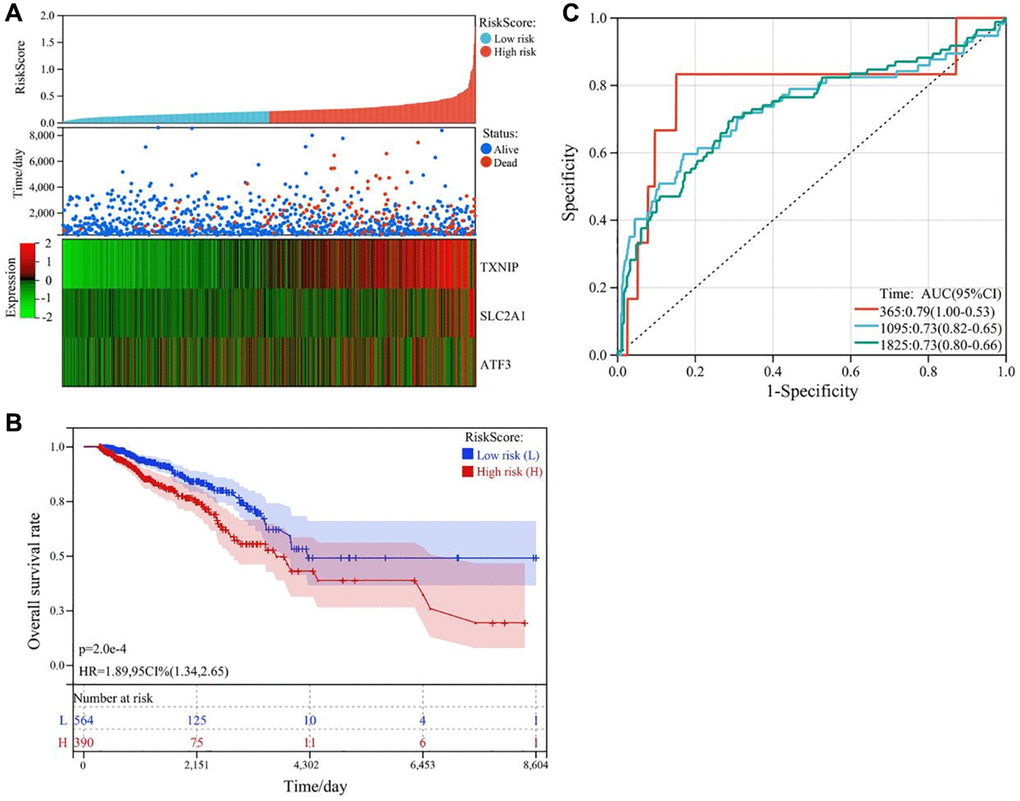
Figure 4. Construction and validation of the ferroptosis-related gene prognostic risk model in breast cancer. (A) Survival status plot of breast cancer patients based on the prognostic model. (B) Kaplan-Meier curve analysis of the overall survival rate of high-risk (n = 530) and low-risk (n = 530) groups. (C) ROC curve analysis of the prognostic model for predicting the survival of breast cancer patients at 365 days, 1095 days, and 1825 days. The AUC values were 0.79, 0.73, and 0.73, respectively.
Discussion
Breast cancer is the most common cancer among women, with a complex and multifactorial pathogenesis that remains unclear. Despite advances in diagnosis and treatment, breast cancer still accounts for a significant proportion of cancer-related deaths. Therefore, there is an urgent need to identify novel approaches for early diagnosis, effective treatment, and accurate prognosis evaluation. Recent studies have highlighted the critical role of ferroptosis in breast cancer development and progression. Many ferroptosis-related genes have been found to be closely related to the growth of breast cells. Cystine/glutamate transporter (SLC7A11), encoded by the SLC7A11 gene, is a multi-channel transmembrane protein that promotes GSH synthesis and reduces ROS to inhibit cell ferroptosis by transporting cystine into cells and exchanging intracellular glutamate [11, 12]. Inhibition of SLC7A11 expression and methylation levels can increase lipid ROS levels and induce ferroptosis in breast cancer cells, thus inhibiting tumor growth [7]. Phospholipid hydroperoxide glutathione peroxidase (GPX4) is a ferroptosis inhibitory protein that converts lipid hydroperoxides into alcohols using reduced GSH to alleviate cell oxidative damage [13]. It was found that GPX4 is highly expressed in breast cancer tissues and its high expression is closely associated with poor prognosis of breast cancer patients [14]. Inhibition of GPX4 expression in breast cells can trigger ferroptosis, a form of iron-dependent cell death, which can suppress tumor cell proliferation and induce cell apoptosis [15].
Integrative analysis of expression profiling data using bioinformatics methods is one of the most effective approaches for identifying disease pathogenesis, biomarkers, and prognostic features, which has the advantages of low cost and high efficiency. Machine learning is an artificial intelligence method that uses statistical algorithms applied to datasets by computers and is widely used for feature information screening in high-throughput data. The combination of bioinformatics methods and machine learning provides a more reliable and effective technique for screening disease-associated genes, which has gradually become a technical hotspot in omics research. In this study, bioinformatics and machine learning were combined for research on the ferroptosis mechanism in breast cancer. We performed differential expression analysis on breast cancer-related datasets from the TCGA database, and identified 52 DEGs associated with ferroptosis in cancer and adjacent tissues. Functional analysis revealed that these genes are involved in multiple signaling pathways closely related to breast cancer progression, such as the HIF-1 and NOD-like receptor signaling pathways. Previous studies have shown that the HIF-1 pathway can influence breast cancer growth by regulating tumor cell metabolism, the Wnt/β-catenin signaling pathway, angiogenesis, and other pathways [16–19]. Activation of the NOD-like receptor pathway can promote estrogen receptor signaling and ROS production, which further inhibits breast cancer cell growth [20]. The NOD-like receptor pathway is also an important pathway for activating the body’s immune signal, promoting the secretion of inflammatory factors such as TNF-α, and further killing breast cancer cells [21]. Additionally, the HIF-1 and NOD-like receptor pathways can regulate cell ferroptosis [22, 23], although their association with ferroptosis in breast cancer cells has not been reported.
We utilized four machine learning algorithms to identify three key ferroptosis-related genes (TXNIP, SLC2A1, ATF3) that are closely associated with the development and prognosis of colon cancer from 52 differentially expressed genes. A prognostic model was constructed using these three genes, and the results showed that high expression of SLC2A1 is associated with poor prognosis in breast cancer patients, while low expression of TXNIP and ATF3 is associated with poor prognosis. The accuracy of this prognostic model was further validated through Kaplan-Meier survival curve analysis and ROC curve analysis. Thioredoxin-interacting protein (TXNIP), also known as thioredoxin-binding protein-2, is a widely expressed protein in various tissues and organs [24]. TXNIP negatively regulates the activity of thioredoxin (TXN) by binding to it, leading to oxidative stress, mitochondrial dysfunction and cell death [25]. As an inducer of ferroptosis, TXNIP causes iron accumulation and accumulation of lipid ROS by inhibiting the activity of GSH, GPX4, and other redox enzymes, ultimately inducing cell death [26]. TXNIP has great potential as a therapeutic target for breast cancer, as studies have shown that it is a good biomarker and prognostic evaluation gene for breast cancer. Park et al. showed that TXNIP expression was reduced in breast cancer patients and that TXNIP downregulation activated estrogen receptor signaling, which in turn enhanced the proliferative activity of breast cancer cell [27]. Downregulation of TXNIP can promote glucose uptake and Warburg effect, while forced overexpression of TXNIP can inhibit these processes [28]. Chen et al. showed that TXNIP can inhibit the proliferation of TNBC resistant cells and promote apoptosis by increasing ROS synthesis and DNA damage caused by doxorubicin, thus reducing TNBC resistance to chemotherapy [29]. In addition, studies have shown that TXNIP expression is downregulated in other tumors such as hepatocellular carcinoma, lung cancer and bladder cancer. TXNIP can inhibit tumor cell growth and metastasis by suppressing metabolic reprogramming and promoting oxidative stress [30]. Activating transcription factor 3 (ATF3) is a stress-induced transcription factor (e.g., by DNA damage, oxidative stress, and cellular injury) that plays an important role in the progression of breast cancer and other tumor diseases. Low expression of ATF3 is closely related to poor prognosis in breast cancer patients [31]. Upregulating ATF3 expression in breast cancer cells can inhibit their growth, migration, and invasion [31]. Research has found that ATF3 regulates transcription by binding to the promoter sequences of multiple oncogenes and tumor suppressor genes, exerting anti-tumor effects [32]. Furthermore, ATF3 can induce ferroptosis by inhibiting the Xc− amino acid antiporter to deplete intracellular GSH and promote the production of lipid peroxides, leading to cell death [33]. ATF3 can also bind to the promoter of ferroptosis-related gene SLC7A11 and induce cell death by suppressing its expression [33]. Solute carrier family 2 member 1 (SLC2A1), also known as glucose transporter 1 (GLUT1), is the earliest discovered glucose transporter. SLC2A1 is highly expressed in breast cancer and other tumor diseases and is closely associated with the progression and metastasis of various cancers [34, 35]. Studies have found that SLC2A1 can resist ferroptosis by promoting glycolysis [36]. Inhibition of SLC2A1 can suppress the glycolysis process, cell proliferation, migration, and metastasis of breast cancer cells [1]. Additionally, inhibition of SLC2A1 can increase autophagic flux to suppress tamoxifen resistance in breast cancer cells [37]. In summary, the three key ferroptosis-related genes (TXNIP, SLC2A1, ATF3) are closely associated with the development and prognosis of breast cancer. However, it is still unclear whether TXNIP, SLC2A1, and ATF3 affect the growth of breast cancer cells by regulating the ferroptosis mechanism.
To validate the reliability of combining bioinformatics and machine learning analysis, we employed in vitro gene silencing and overexpression techniques to observe the effects of SLC2A1 knockdown, TXNIP upregulation, and ATF3 upregulation on the growth of breast cancer cells and ferroptosis-related indicators. The results showed that consistent with the results of bioinformatics and machine learning analysis, SLC2A1 is a breast cancer risk gene, and its knockdown can induce iron death of breast cancer cells and inhibit cancer cell growth. TXNIP and ATF3 are tumor suppressor genes in breast cancer, and their upregulation can trigger the ferroptosis mechanism and suppress the growth of breast cancer cells.
Conclusion
This study identified three key ferroptosis-related genes (TXNIP, SLC2A1, ATF3) associated with breast cancer by integrating bioinformatics analysis and multiple machine learning algorithms, and preliminarily validated their roles through cell experiments. The innovation lies in the effective strategy of combining bioinformatics and machine learning to screen key genes. However, this study has some limitations, including lack of in vivo validation. Future directions include validating the results in animal models and elucidating the molecular mechanisms. In summary, the study further confirms the important role of the ferroptosis mechanism in the occurrence, development, and treatment of breast cancer, providing new insights for the study of the pathogenesis, diagnosis, and prognosis evaluation of breast cancer.
Author Contributions
The authors’ contributions are as follows: Experiments and Writing: SL, she is the first author. Experiments, Investigation, Grammar modification and data analysis: YM-B and BZ. All authors contributed to the article and approved the submitted version.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This study was supported by the Ningxia Key Research and Development Plan project (2021BEG03112) and Ningxia Key Research and Development Plan project (2022BEG03144).
References
- 1. Wu Q, Ba-Alawi W, Deblois G, Cruickshank J, Duan S, Lima-Fernandes E, Haight J, Tonekaboni SAM, Fortier AM, Kuasne H, McKee TD, Mahmoud H, Kushida M, et al. GLUT1 inhibition blocks growth of RB1-positive triple negative breast cancer. Nat Commun. 2020; 11:4205. https://doi.org/10.1038/s41467-020-18020-8 [PubMed]
- 2. Sun YS, Zhao Z, Yang ZN, Xu F, Lu HJ, Zhu ZY, Shi W, Jiang J, Yao PP, Zhu HP. Risk Factors and Preventions of Breast Cancer. Int J Biol Sci. 2017; 13:1387–97. https://doi.org/10.7150/ijbs.21635 [PubMed]
- 3. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, Forman D, Bray F. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015; 136:E359–86. https://doi.org/10.1002/ijc.29210 [PubMed]
- 4. Zuo S, Yu J, Pan H, Lu L. Novel insights on targeting ferroptosis in cancer therapy. Biomark Res. 2020; 8:50. https://doi.org/10.1186/s40364-020-00229-w [PubMed]
- 5. Narod SA, Salmena L. BRCA1 and BRCA2 mutations and breast cancer. Discov Med. 2011; 12:445–53. [PubMed]
- 6. Mou Y, Wang J, Wu J, He D, Zhang C, Duan C, Li B. Ferroptosis, a new form of cell death: opportunities and challenges in cancer. J Hematol Oncol. 2019; 12:34. https://doi.org/10.1186/s13045-019-0720-y [PubMed]
- 7. Yang J, Zhou Y, Xie S, Wang J, Li Z, Chen L, Mao M, Chen C, Huang A, Chen Y, Zhang X, Khan NUH, Wang L, Zhou J. Metformin induces Ferroptosis by inhibiting UFMylation of SLC7A11 in breast cancer. J Exp Clin Cancer Res. 2021; 40:206. https://doi.org/10.1186/s13046-021-02012-7 [PubMed]
- 8. Yang F, Xiao Y, Ding JH, Jin X, Ma D, Li DQ, Shi JX, Huang W, Wang YP, Jiang YZ, Shao ZM. Ferroptosis heterogeneity in triple-negative breast cancer reveals an innovative immunotherapy combination strategy. Cell Metab. 2023; 35:84–100.e8. https://doi.org/10.1016/j.cmet.2022.09.021 [PubMed]
- 9. Yao X, Xie R, Cao Y, Tang J, Men Y, Peng H, Yang W. Simvastatin induced ferroptosis for triple-negative breast cancer therapy. J Nanobiotechnology. 2021; 19:311. https://doi.org/10.1186/s12951-021-01058-1 [PubMed]
- 10. Ma S, Henson ES, Chen Y, Gibson SB. Ferroptosis is induced following siramesine and lapatinib treatment of breast cancer cells. Cell Death Dis. 2016; 7:e2307. https://doi.org/10.1038/cddis.2016.208 [PubMed]
- 11. Yadav P, Sharma P, Sundaram S, Venkatraman G, Bera AK, Karunagaran D. SLC7A11/ xCT is a target of miR-5096 and its restoration partially rescues miR-5096-mediated ferroptosis and anti-tumor effects in human breast cancer cells. Cancer Lett. 2021; 522:211–24. https://doi.org/10.1016/j.canlet.2021.09.033 [PubMed]
- 12. Koppula P, Zhuang L, Gan B. Cystine transporter SLC7A11/xCT in cancer: ferroptosis, nutrient dependency, and cancer therapy. Protein Cell. 2021; 12:599–620. https://doi.org/10.1007/s13238-020-00789-5 [PubMed]
- 13. Yang WS, SriRamaratnam R, Welsch ME, Shimada K, Skouta R, Viswanathan VS, Cheah JH, Clemons PA, Shamji AF, Clish CB, Brown LM, Girotti AW, Cornish VW, et al. Regulation of ferroptotic cancer cell death by GPX4. Cell. 2014; 156:317–31. https://doi.org/10.1016/j.cell.2013.12.010 [PubMed]
- 14. Lee N, Carlisle AE, Peppers A, Park SJ, Doshi MB, Spears ME, Kim D. xCT-Driven Expression of GPX4 Determines Sensitivity of Breast Cancer Cells to Ferroptosis Inducers. Antioxidants (Basel). 2021; 10:317. https://doi.org/10.3390/antiox10020317 [PubMed]
- 15. Li H, Liu W, Zhang X, Wu F, Sun D, Wang Z. Ketamine suppresses proliferation and induces ferroptosis and apoptosis of breast cancer cells by targeting KAT5/GPX4 axis. Biochem Biophys Res Commun. 2021; 585:111–6. https://doi.org/10.1016/j.bbrc.2021.11.029 [PubMed]
- 16. Ferrer CM, Lynch TP, Sodi VL, Falcone JN, Schwab LP, Peacock DL, Vocadlo DJ, Seagroves TN, Reginato MJ. O-GlcNAcylation regulates cancer metabolism and survival stress signaling via regulation of the HIF-1 pathway. Mol Cell. 2014; 54:820–31. https://doi.org/10.1016/j.molcel.2014.04.026 [PubMed]
- 17. Liu X, Xie P, Hao N, Zhang M, Liu Y, Liu P, Semenza GL, He J, Zhang H. HIF-1-regulated expression of calreticulin promotes breast tumorigenesis and progression through Wnt/β-catenin pathway activation. Proc Natl Acad Sci U S A. 2021; 118:e2109144118. https://doi.org/10.1073/pnas.2109144118 [PubMed]
- 18. de Heer EC, Jalving M, Harris AL. HIFs, angiogenesis, and metabolism: elusive enemies in breast cancer. J Clin Invest. 2020; 130:5074–87. https://doi.org/10.1172/JCI137552 [PubMed]
- 19. Jin J, Qiu S, Wang P, Liang X, Huang F, Wu H, Zhang B, Zhang W, Tian X, Xu R, Shi H, Wu X. Cardamonin inhibits breast cancer growth by repressing HIF-1α-dependent metabolic reprogramming. J Exp Clin Cancer Res. 2019; 38:377. https://doi.org/10.1186/s13046-019-1351-4 [PubMed]
- 20. Raut PK, Kim SH, Choi DY, Jeong GS, Park PH. Growth of breast cancer cells by leptin is mediated via activation of the inflammasome: Critical roles of estrogen receptor signaling and reactive oxygen species production. Biochem Pharmacol. 2019; 161:73–88. https://doi.org/10.1016/j.bcp.2019.01.006 [PubMed]
- 21. Cao X, Xu J. Insights into inflammasome and its research advances in cancer. Tumori. 2019; 105:456–64. https://doi.org/10.1177/0300891619868007 [PubMed]
- 22. Feng X, Wang S, Sun Z, Dong H, Yu H, Huang M, Gao X. Ferroptosis Enhanced Diabetic Renal Tubular Injury via HIF-1α/HO-1 Pathway in db/db Mice. Front Endocrinol (Lausanne). 2021; 12:626390. https://doi.org/10.3389/fendo.2021.626390 [PubMed]
- 23. Xie SS, Deng Y, Guo SL, Li JQ, Zhou YC, Liao J, Wu DD, Lan WF. Endothelial cell ferroptosis mediates monocrotaline-induced pulmonary hypertension in rats by modulating NLRP3 inflammasome activation. Sci Rep. 2022; 12:3056. https://doi.org/10.1038/s41598-022-06848-7 [PubMed]
- 24. Pan M, Zhang F, Qu K, Liu C, Zhang J. TXNIP: A Double-Edged Sword in Disease and Therapeutic Outlook. Oxid Med Cell Longev. 2022; 2022:7805115. https://doi.org/10.1155/2022/7805115 [PubMed]
- 25. Alhawiti NM, Al Mahri S, Aziz MA, Malik SS, Mohammad S. TXNIP in Metabolic Regulation: Physiological Role and Therapeutic Outlook. Curr Drug Targets. 2017; 18:1095–103. https://doi.org/10.2174/1389450118666170130145514 [PubMed]
- 26. Singh LP, Yumnamcha T, Devi TS. Mitophagy, Ferritinophagy and Ferroptosis in Retinal Pigment Epithelial Cells Under High Glucose Conditions: Implications for Diabetic Retinopathy and Age-Related Retinal Diseases. JOJ Ophthalmol. 2021; 8:77–85. [PubMed]
- 27. Park JW, Lee SH, Woo GH, Kwon HJ, Kim DY. Downregulation of TXNIP leads to high proliferative activity and estrogen-dependent cell growth in breast cancer. Biochem Biophys Res Commun. 2018; 498:566–72. https://doi.org/10.1016/j.bbrc.2018.03.020 [PubMed]
- 28. Wang Y, Chen S. TXNIP Links Anticipatory Unfolded Protein Response to Estrogen Reprogramming Glucose Metabolism in Breast Cancer Cells. Endocrinology. 2022; 163:bqab212. https://doi.org/10.1210/endocr/bqab212 [PubMed]
- 29. Chen Y, Feng X, Yuan Y, Jiang J, Zhang P, Zhang B. Identification of a novel mechanism for reversal of doxorubicin-induced chemotherapy resistance by TXNIP in triple-negative breast cancer via promoting reactive oxygen-mediated DNA damage. Cell Death Dis. 2022; 13:338. https://doi.org/10.1038/s41419-022-04783-z [PubMed]
- 30. Chen Y, Ning J, Cao W, Wang S, Du T, Jiang J, Feng X, Zhang B. Research Progress of TXNIP as a Tumor Suppressor Gene Participating in the Metabolic Reprogramming and Oxidative Stress of Cancer Cells in Various Cancers. Front Oncol. 2020; 10:568574. https://doi.org/10.3389/fonc.2020.568574 [PubMed]
- 31. Li L, Sun RM, Jiang GQ. ATF3 Demethylation Promotes the Transcription of ARL4C, Which Acts as a Tumor Suppressor in Human Breast Cancer. Onco Targets Ther. 2020; 13:3467–76. https://doi.org/10.2147/OTT.S243632 [PubMed]
- 32. Ku HC, Cheng CF. Master Regulator Activating Transcription Factor 3 (ATF3) in Metabolic Homeostasis and Cancer. Front Endocrinol (Lausanne). 2020; 11:556. https://doi.org/10.3389/fendo.2020.00556 [PubMed]
- 33. Wang L, Liu Y, Du T, Yang H, Lei L, Guo M, Ding HF, Zhang J, Wang H, Chen X, Yan C. ATF3 promotes erastin-induced ferroptosis by suppressing system Xc(). Cell Death Differ. 2020; 27:662–75. https://doi.org/10.1038/s41418-019-0380-z [PubMed]
- 34. Liu XS, Yang JW, Zeng J, Chen XQ, Gao Y, Kui XY, Liu XY, Zhang Y, Zhang YH, Pei ZJ. SLC2A1 is a Diagnostic Biomarker Involved in Immune Infiltration of Colorectal Cancer and Associated With m6A Modification and ceRNA. Front Cell Dev Biol. 2022; 10:853596. https://doi.org/10.3389/fcell.2022.853596 [PubMed]
- 35. Zhang X, Pang X, Zhang Z, Liu Q, Zhang H, Xiang Q, Cui Y. Co-expression and prognosis analyses of GLUT1-4 and RB1 in breast cancer. BMC Cancer. 2021; 21:1026. https://doi.org/10.1186/s12885-021-08763-y [PubMed]
- 36. Song X, Liu J, Kuang F, Chen X, Zeh HJ
3rd , Kang R, Kroemer G, Xie Y, Tang D. PDK4 dictates metabolic resistance to ferroptosis by suppressing pyruvate oxidation and fatty acid synthesis. Cell Rep. 2021; 34:108767. https://doi.org/10.1016/j.celrep.2021.108767 [PubMed] - 37. Sun M, Zhao S, Duan Y, Ma Y, Wang Y, Ji H, Zhang Q. GLUT1 participates in tamoxifen resistance in breast cancer cells through autophagy regulation. Naunyn Schmiedebergs Arch Pharmacol. 2021; 394:205–16. https://doi.org/10.1007/s00210-020-01893-3 [PubMed]