Introduction
Tumor development is a complex process, involving numerous factors such as altered gene expression, gene mutations, changes in protein structure, and changes in the tumor immune microenvironment [1–3]. Cancer poses a significant danger to human life and health and has a significant impact on socio-economic progress. Many tumors are also frequently encountered in clinical work. Breast cancer, head and neck squamous cell carcinoma, sarcoma, and skin cutaneous melanoma are all common tumors in plastic and aesthetic (burn) surgery [4–7]. As of now, the etiology and mechanisms of tumors have not been fully understood [8]. Therefore, the identification of those genes that were highly expressed in various tumor tissues and the assessment of their effects on the survival of tumor patients is important for discovering new prognostic markers for tumors as well as providing clinicians with new therapeutic ideas [9–12]. In recent years, many researchers have focused on mitochondria and have found structural or functional alterations in mitochondria in patients with certain diseases. This suggests that there may be some connection between the structure and function of mitochondria and the development of disease [13–15]. It has been found that in some common diseases including neurodegenerative diseases and cancer, pathological alterations in the cellular environment can cause changes in the function or activity of many proteins associated with mitochondrial fusion or cleavage. These changes will directly affect mitochondrial fusion or cleavage, resulting in altered mitochondrial dynamics [16].
The OPA1 gene is located at 3q28 and is over 100 kb in length with 31 exons [17]. Previous studies have revealed the presence of an OPA1 protein encoded by the OPA1 gene in mitochondria, which is necessary for angiogenesis. On the other hand, angiogenesis is necessary for tumor growth and metastasis [18]. In addition, mitochondria are key organelles required to maintain normal endocrine function, providing energy for hormone production and transport [19]. The endocrine level is related to the occurrence, development, and prognosis of various tumors. Some researchers used endocrine disruptors to affect the function of the endocrine system and interfere with hormone action, which could increase the risk of tumors including reproductive damage, metabolic disorder, breast cancer, and prostate cancer [20]. In animal models, endocrine disruptors have carcinogenic effects on endocrine response tissues such as the breast, prostate, testis, ovary, and thyroid [21–23]. Therefore, it is important to study the factors affecting mitochondrial function and its influence on the endocrine level to find out the occurrence and development of some cancers and find new prevention and treatment measures.
Mitochondria are highly dynamic cellular organelles, exhibiting diverse morphologies such as small spheres, short or long tubules, and interconnected structures [24]. These shapes are regulated by the processes of fusion and fission. Additionally, mitochondria display high mobility in numerous cells, traveling through the cytosol via the cytoskeletal transport system. Consequently, mitochondrial dynamics play a pivotal role in maintaining mitochondrial quality [25]. Some researchers found that mitochondrial fusion was significantly increased in the tissues of patients with liver cancer, mitochondrial fusion was also inhibited in liver cancer cell lines after knockdown of OPA1, and the growth of liver cancer cells cultured in vitro and tumor formation in mice were inhibited [26]. In LUAD, the mitochondrial fusion process caused by OPA1 is activated, enhancing mitochondrial metabolism to promote tumor growth and inhibit apoptosis. OPA1 overexpression can also increase glycolytic activity to enable cancer cells in LUAD patients to achieve immune escape [27]. In addition, researchers found that upregulation of OPA1 expression was associated with poor prognosis in breast cancer, and the research team reduced proliferation, migration, and invasion of breast cancer cells in vitro and in vivo by inhibiting OPA1 expression. Further research showed that OPA1 silencing inhibited breast cancer cell growth and invasion by up-regulating the expression levels of the 148/152 miRNA family while not reducing mitochondrial respiration [28]. In pancreatic ductal adenocarcinoma (PDAC), the presence of cancer stem cells (CSCs) was associated with high invasiveness of PDAC, while OPA1 overexpression was found in CSCs and has a regulatory effect on tumorsphere formation [29]. In summary, we make a conjecture based on previous studies. The expression level of OPA1 may play a very important role in the occurrence and development of pan-cancer.
Therefore, we will explore the level of OPA1 gene expression in pan-cancer, its mutation types, and the relationship of these alterations with patient prognosis and immune microenvironment, as well as the molecular mechanisms that may be involved. This will provide a theoretical basis for further investigation of the possibility of using OPA1 as a new prognostic marker for tumors.
Results
Expression levels of OPA1 in tumors and paraneoplastic tissues
The workflow of our study is shown in Figure 1. We used the TIMER 2.0 database to compare OPA1 expression levels in different tumors and normal tissues in the TCGA database, and the results were displayed in Figure 2A. The results showed that the expression levels of OPA1 in CESC, CHOL, ESCA, GBM, HNSC, KIRC, KIRP, LIHC, LUAD, LUSC, and STAD were significantly higher than that of the corresponding normal tissues (Figure 2A).
Figure 1. The flow chart of the whole study.
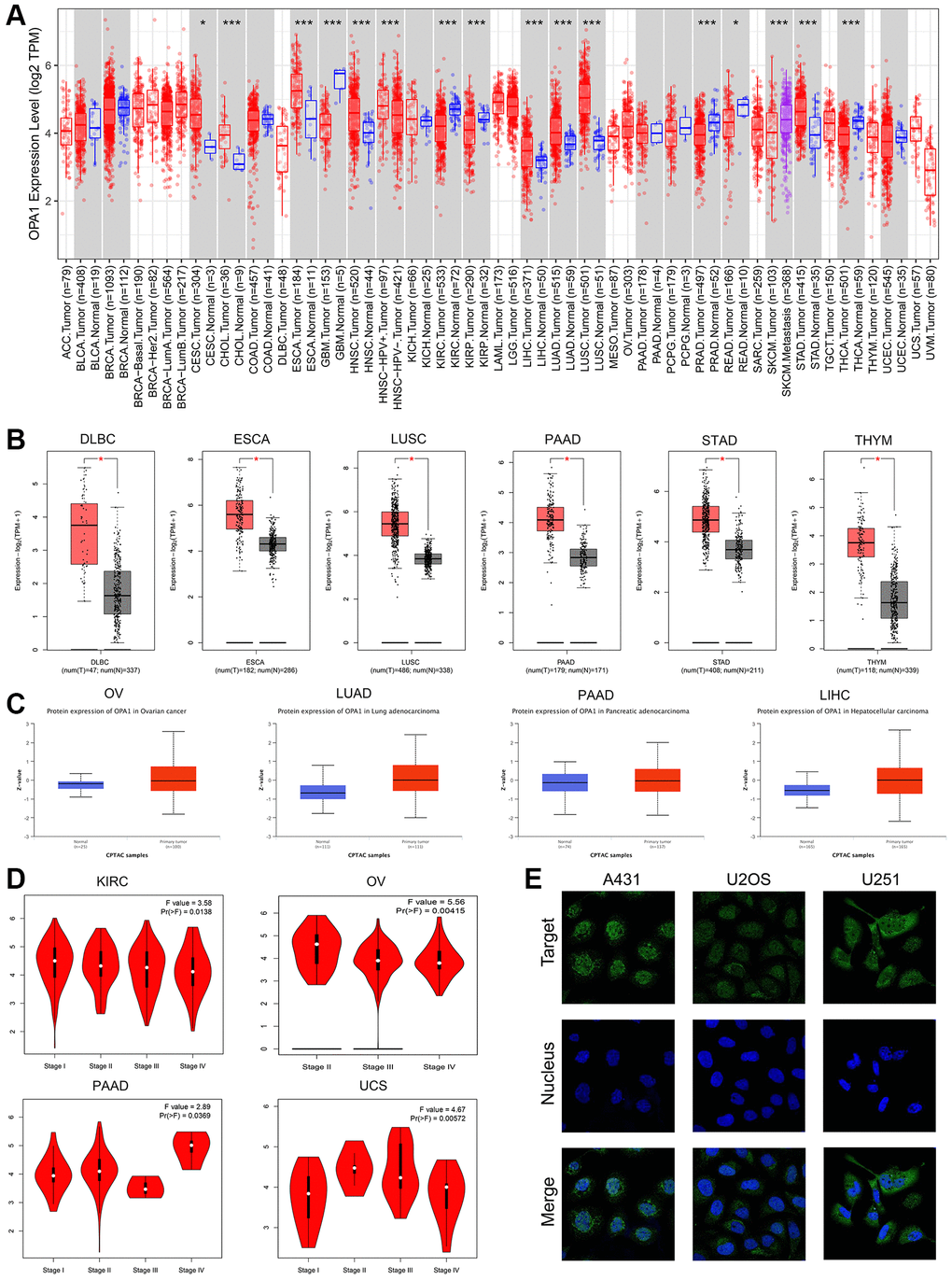
Figure 2. The expression levels of OPA1 in different tumors and normal samples. (A) The expression levels of the OPA1 gene in different tumors and paratumoral tissues were analyzed by TIMER2. *p < 0.05, **p < 0.01, ***p < 0.001. (B) The expression levels of the OPA1 gene in DLBC, ESCA, LUSC, PAAD, STAD, THYM, and the normal samples, were analyzed by GEPIA. *p < 0.05. (C) The protein expression of OPA1 in OV, LUAD, PAAD, LIHC, and the normal samples was analyzed by UALCAN. (D) The expression level of OPA1 in the different stages of KIRC, OV, PAAD, and UCS, was analyzed by GEPIA. (E) The immunofluorescence in osteosarcoma cell lines (A431, U2OS, and U251), was analyzed by HPA.
Using the GEPIA database, we performed a joint analysis of OPA1 expression data in various tumors from the TCGA and GTEx databases and found that OPA1 was expressed at higher levels in DLBC, ESCA, LUSC, PAAD, STAD, and THYM than in control normal tissues, with significance (Figure 2B). By comparing OPA1 expression at the protein level in different tumors and normal tissues using the UALCAN database, we found that OPA1 protein expression levels were significantly elevated in OV, LUAD, PAAD, and LIHC compared to normal tissues (Figure 2C).
The results from the GEPIA2 database also demonstrated that OPA1 expression levels were increased at different stages of KIRC, OV, PAAD, and UCS. (Figure 2D).
In addition, we used the HPA database to observe the subcellular localization of OPA1, and we observed by immunofluorescence imaging that the OPA1 protein was localized both in the nucleus and on the nuclear membrane in A431, U2OS, and U251 cell lines. (Figure 2E).
Prognostic value of OPA1
Based on the median expression level of OPA1 in tumor cells of tumor patients, we divided the patients into high and low-expression groups. Then use the Kaplan-Meier plotter to analyze and visualize the survival of tumor patients with different OPA1 expression levels at different periods. As shown in Figure 3A, patients with tumors including BRCA, HNSC, LUAD, PAAD, THCA, and UCEC, in the OPA1 high expression group, had significantly lower overall survival rates than the patients with these tumors in the OPA1 low expression group. However, the overall survival of CESC, KIRC, LUCS and READ patients in the high OPA1 expression group was higher than that of the low expression group. For patients with BLCA, PAAD, PCPG, and THCA, relapse-free survival was also significantly lower in the OPA1 high-expression group than in the low-expression group. However, patients with LIHC with high OPA1 expression levels had significantly higher relapse-free survival rates than those with LIHC with low OPA1 expression levels (Figure 3B).
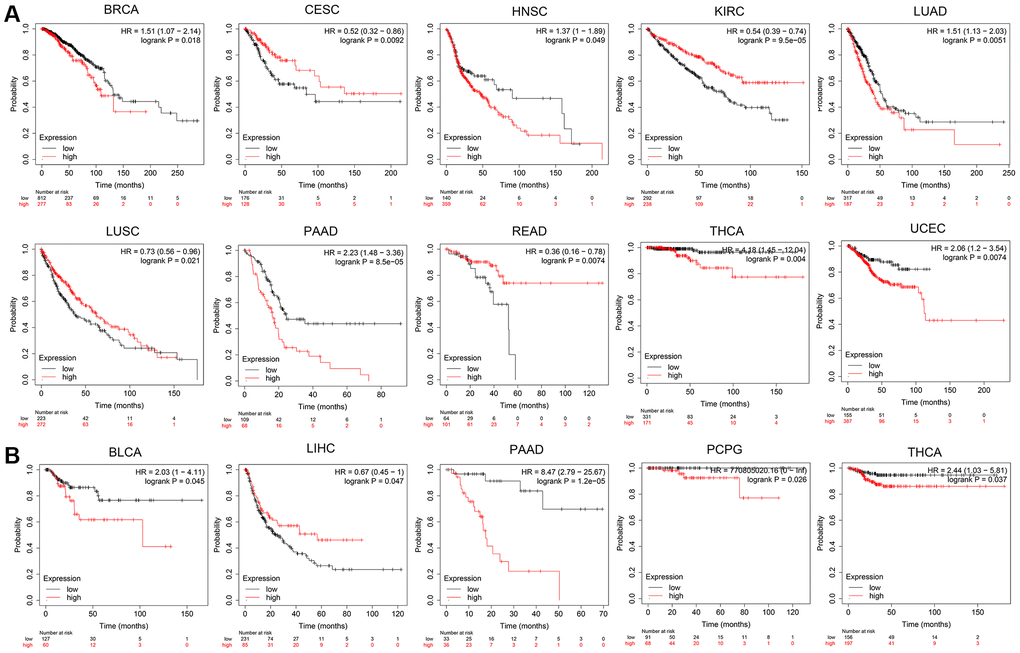
Figure 3. The prognosis comparison of tumor patients in the OPA1 high expression group and the low expression group, analyzed by Kaplan-Meier plotter. (A) Overall survival and (B) relapse-free survival.
The results of the analysis through the GEPIA database were similar to those of Kaplan-Meier plotter. ACC, BLCA, and PAAD patients with low OPA1 expression levels had higher disease-free survival than patients with high OPA1 expression levels. However, CHOL patients with high OPA1 expression levels had higher disease-free survival than those with low OPA1 expression levels (Figure 4A). The results shown in Figure 4B indicated that the overall survival of BRCA, LUAD, MESO, and PAAD patients with high OPA1 expression levels was lower than low-expression group patients. However, CHOL and KIRC patients in the high OPA1 group had higher overall survival than the low OPA1 group (Figure 4B).
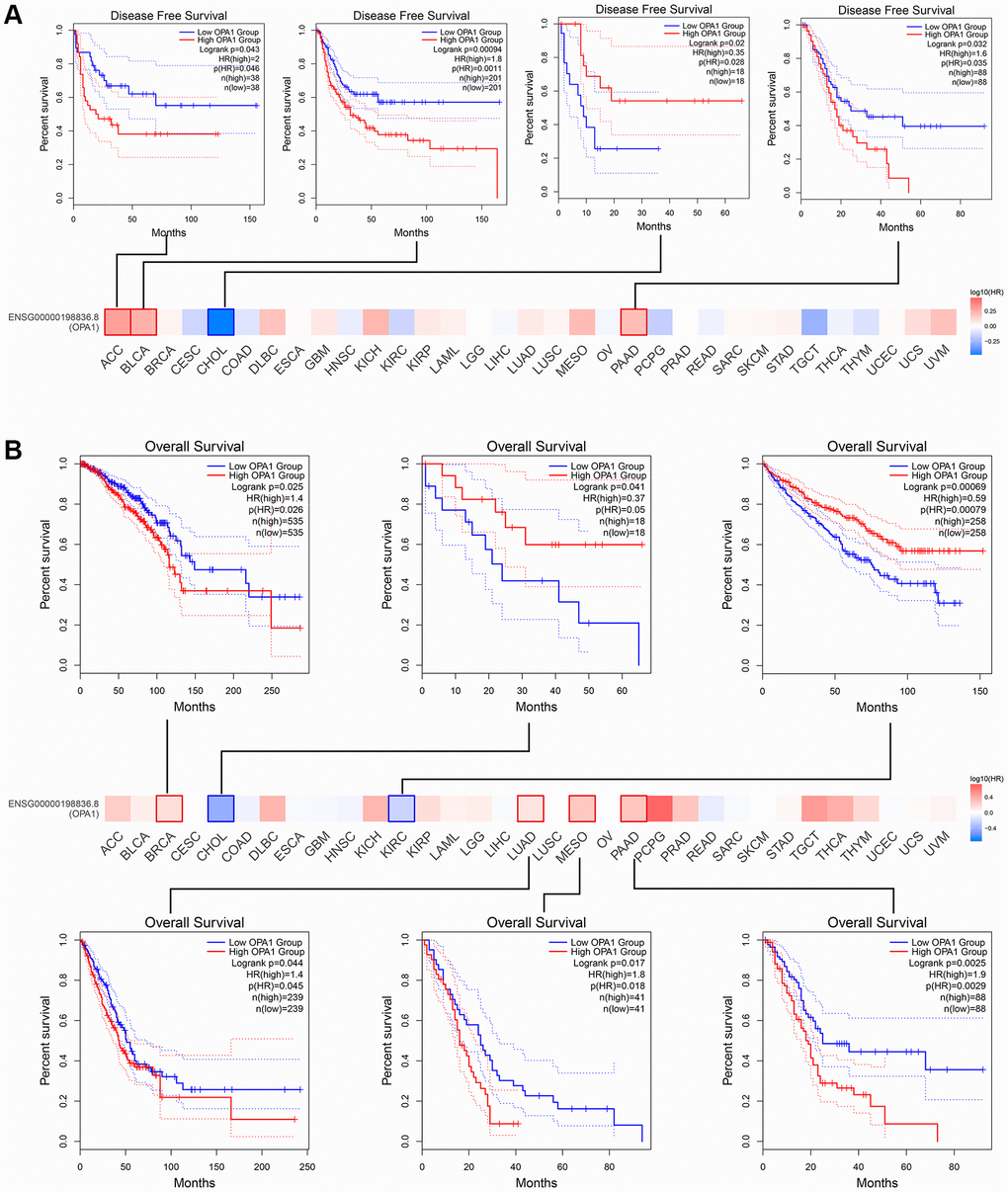
Figure 4. The prognosis comparison of tumor patients in the OPA1 high expression group and the low expression group, analyzed by GEPIA. (A) Disease-free survival and (B) overall survival.
OPA1 gene mutation site analysis
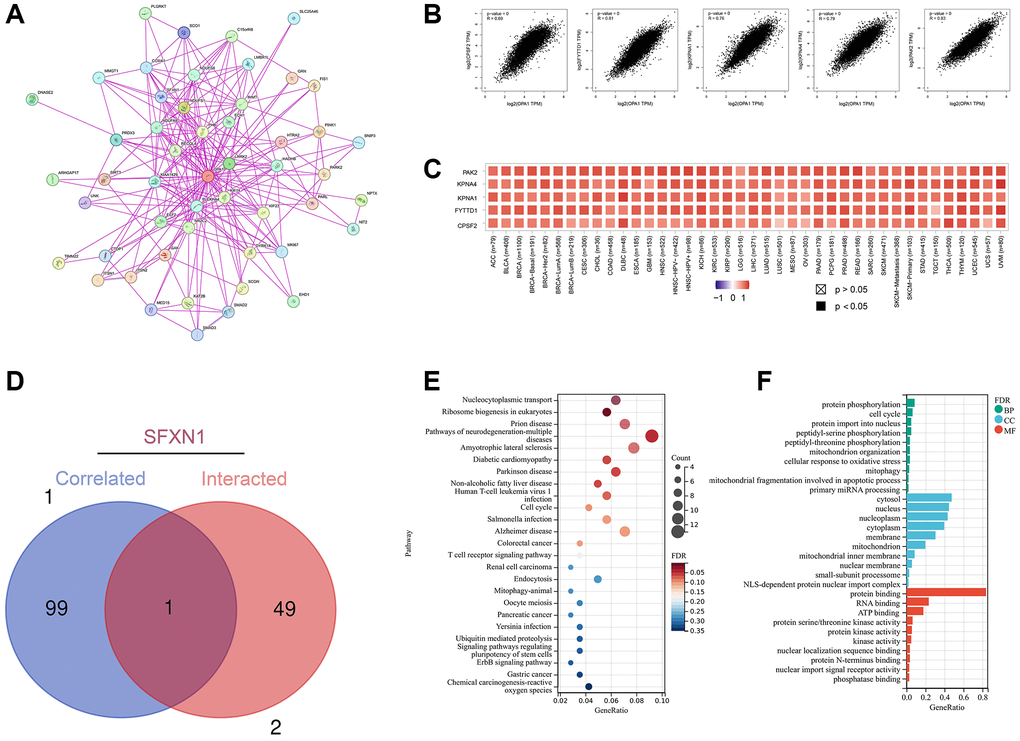
We analyzed the genetic alteration status of OPA1 in different tumor samples from the TCGA cohort. As shown in Figure 5A, the highest frequency of OPA1 alterations (29%) was found in patients with Lung Squamous cell carcinoma, where “amplification” was the predominant type. In addition, the amplification rate of OPA1 in patients with ESCA, OV, CESC, and HNSC all exceeded 10%. Notably, the type of alterations in the OPA1 gene in colorectal adenocarcinoma cases were all mutations (~2% frequency).
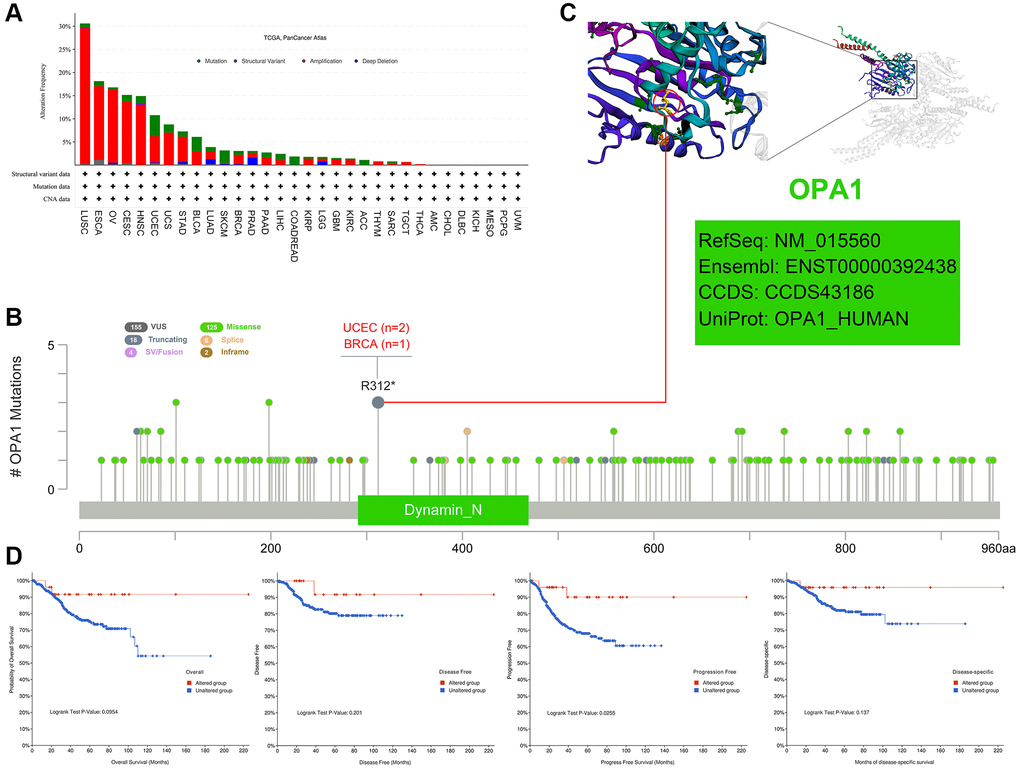
Figure 5. Mutations of OPA1 in individual tumors in the TCGA database, analyzed by cBioPortal. (A) The mutation types of OPA1 in individual tumors. (B) Mutation sites of OPA1 in UCEC and BRCA. (C) 3D structure diagram of OPA1 mutation site. (D) Relationship between OPA1 mutations and survival in UCEC patients.
Figure 5B showed the type, locus, type of cases, and number of mutations in the OPA1 gene. These mutations of unclear significance can neither be classified as favorable nor deleterious mutations and were referred to as VUS mutations. The most common of these were missense mutations. In two UCEC and one BRCA patient, both detected alterations in the R312* site of the dynamin-N protein, which subsequently led to OPA1 protein truncation. Figure 5C showed the R312* site in the 3D structure of the OPA1 protein. We also analyzed the impact of OPA1 mutations on the prognosis of UCEC patients. The results showed that UCEC patients with OPA1 mutations had a significantly higher PFS than UCEC patients without OPA1 mutations (P < 0.05) (Figure 5D).
Relationship between OPA1 expression level and tumor immune microenvironment
Tumor filter cells are an important component of the tumor immune microenvironment and are closely associated with cancer development, progression, and metastasis [30, 31]. Here, we used the TIMER tool to analyze relevant data from the TCGA database to explore the statistical relationship between OPA1 expression levels and the level of immune cell infiltration in pan-cancer. After processing the data using the EPIC, MCPCOUNTER, and TIDE algorithms, we found a statistically positive correlation between OPA1 expression levels in BRCA, ESCA, HNSC, LIHC, LUAD, MESO, OV, PAAD and THYM and the estimated infiltration values of cancer-associated fibroblasts (Figure 6A, 6B).
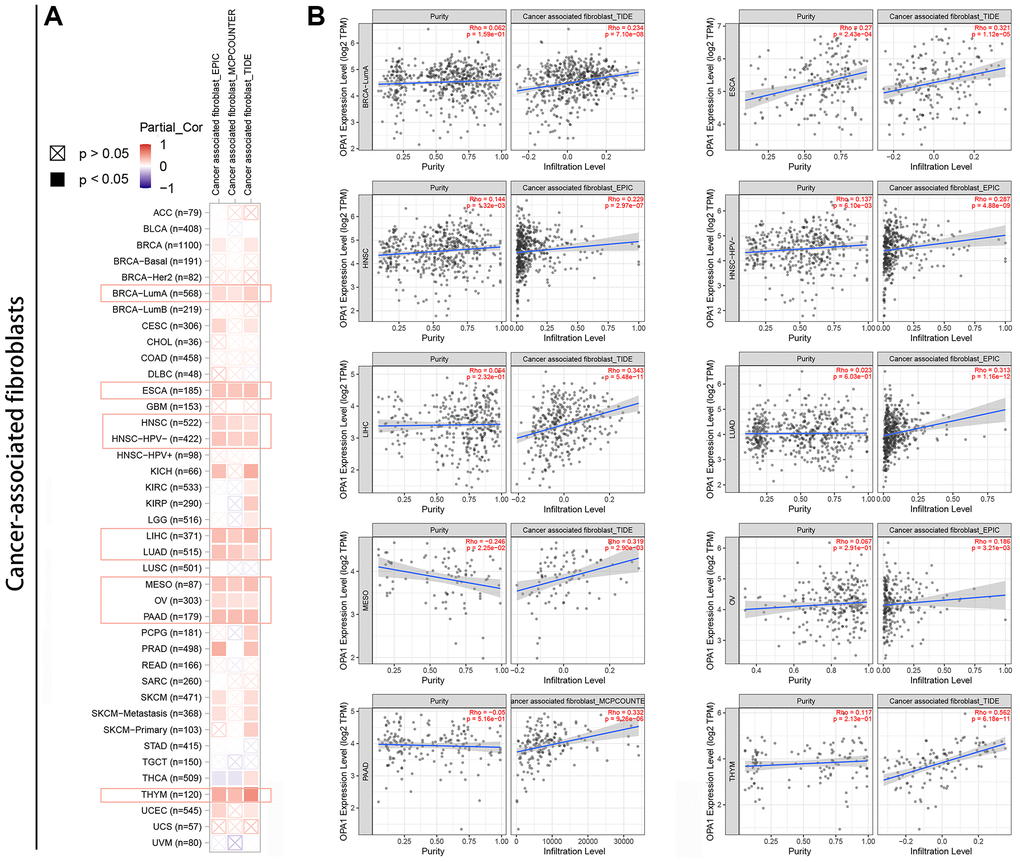
Figure 6. Relationship between OPA1 expression and cancer-associated fibroblast infiltration in pan-cancers. (A) Heat map and (B) scatter plot.
Discussion
Our study indicates a significant presence or mutation of OPA1 in various tumors, demonstrating strong correlations with protein phosphorylation, patient prognosis, immune cell infiltration, and tumor mutational load. OPA1 was found to play important roles in angiogenesis in the physiological state and tumors [18]. Angiogenesis is the formation of new blood vessel branches from old blood vessels, a process that is essential for wound healing and tumor growth [32]. Angiogenesis requires energy, which in human cells are provided by mitochondria, so some researchers have hypothesized that angiogenesis stimulates mitochondrial respiration [33]. However, new research suggests that mitochondrial functions were not limited to energy conversion, but were also involved in complex physiological processes such as cell differentiation, apoptosis, and immune signaling [34].
The function of mitochondria is closely related to their morphology [34]. Under normal physiological conditions, mitochondria perform their corresponding physiological functions by altering the balance of fusion and division [35]. Mitochondrial fusion proteins (MFN) 1 and 2 were located on the outer mitochondrial membrane (OMM) and were responsible for outer mitochondrial membrane fusion, and the optic nerve atrophy protein OPA1 is located on the inner mitochondrial membrane (IMM) and is responsible for inner mitochondrial membrane fusion, and all three together regulate mitochondrial fusion. The cristae formed by the inner membrane invagination of mitochondria were essential for cellular metabolism, and OPA1 plays a crucial role in maintaining the mitochondrial cristae structure [36]. OPA1 may play a regulatory role during apoptosis by participating in cristae junction formation and maintenance [37]. During angiogenesis, OPA1 can coordinate angiogenesis by affecting the activity of the NF-κB signaling pathway and the expression of angiogenic genes [18]. These studies suggest that OPA1 plays important roles in both cell cycle regulation and tumor-related biological processes such as angiogenesis. However, our literature search revealed that there is no article on the pan-cancer analysis of OPA1, so a comprehensive analysis of OPA1 in tumors is necessary. To this end, we analyzed OPA1 expression, genetic alterations, patient survival prognosis, and immune infiltration in a total of 33 different tumor species based on an online database.
Previous studies have shown that mitochondria play a key role in tumor development, including providing energy for tumor cell growth, controlling redox, calcium homeostasis, transcriptional regulation, and controlling cell death. Metabolically, mitochondrial metabolites such as fumaric acid, succinic acid, and 2-hydroxyglutarate (2-HG) drive tumorigenesis, and mitochondria provide energy for anabolic processes in tumor cells [38]. Mitochondria also promote the transformation of normal cells into malignant cells through three main mechanisms: (1) Mitochondrial reactive oxygen species (ROS) promote the accumulation of potential carcinogenic DNA defects. (2) ROS plays an important role in activating potential carcinogenic signaling pathways [39]. (3) Functional defects in mitochondrial permeability transition are necessary for the survival of newly formed malignant tumor precursors [40, 41].
We found that OPA1 was highly expressed in most tumors but had different prognostic effects in patients with different tumors. Patients with ACC, BRCA, HNSC, LUAD, MESO, PAAD, THCA, UCEC, BLCA, and PCPG in the OPA1 high expression group had a worse prognosis, while OPA1 high expression in CHOL, CESC, KIRC, READ, and LIHC patients had the opposite prognostic impact. Taking BRCA as an example, previous researchers have demonstrated through in vivo and in vitro experiments that knockdown of OPA1 expression can reduce proliferation migration and invasion of breast cancer cells, and OPA1 silencing can increase miRNA levels of the 148/152 family that inhibit tumor growth and invasion. Therefore, OPA1 has been listed as a drug target for TNBC inhibition [28]. However, there is no conclusive evidence to explain why upregulation of OPA1 expression is associated with better prognosis in CHOL, CESC, KIRC, READ, and LIHC patients. But combining our research with previous studies, we make the following inference: The reasons may include the following three aspects: First, each cancer has its unique mutation spectrum, OPA1 and a variety of interacting genes play a biological function together, and in the OPA1 interacting genes, the members that affect the development of different tumors are different. Secondly, the high expression of OPA1 promotes mitochondrial function, which may also affect endocrine levels and different tissues have different responses to changes in hormone levels, such as the breast, prostate, testis, ovary, and other tissues are more sensitive to changes in hormone levels, and are more likely to cause cancer due to endocrine interference [21–23]. Thirdly, whether the high expression of OPA1 plays a crucial role in the development of the above tumors also needs to be further verified by experiments. This is because the elevated expression of OPA1 may simply be due to the alteration of normal tissues during the anti-tumor process. Fourth, the statistical difference may be due to insufficient sample size. Taking LIHC as an example, some researchers found that OPA1 downregulation can enhance the sensitivity of hepatocellular carcinoma to chemotherapy drug sorafenib, which indicates that low expression of OPA1 is associated with a better prognosis of hepatocellular carcinoma. Contrary to our results, we believe that this difference may be caused by insufficient sample size [42]. Therefore, although we have demonstrated that high expression of OPA1 is associated with poor prognosis in a variety of tumors, specific analysis is needed for specific tumors.
In the present study, we investigated the mutation of the OPA1 gene in tumors and its relationship with the prognosis of tumor patients. Mutations in OPA1 were significantly associated with high survival rates in CESC. Through gene interaction analysis, we found that, OPA1 expression level was positively correlated with CPSF2 (R = 0.69), FYTTD1 (R = 0.81), KPNA1 (R = 0.76), KPNA4 (R = 0.76), and PAK2 (R = 0.83) genes. Among them, PTC patients with high expression of CPSF2 had a higher risk of recurrence [43], high expression of KPNA-1 was associated with tamoxifen resistance and metastasis in breast cancer patients [44], KPNA-4 can be used as a biomarker for diagnosis and prognosis of hepatocellular carcinoma [45], and can also promote the metastasis of prostate cancer [46], and PAK2 can promote the migration and proliferation of salivary adenoid cystic carcinoma [47]. As for SFXN1, it is a mitochondrial serine transporter required for single-carbon metabolism, which plays an important role in the development of LUAD and can be used as an independent prognostic marker and therapeutic target for LUAD [48, 49]. There is currently no literature reporting a direct interaction between SFXN1 and OPA1. However, phosphorylation modifications, represented by serine residues, play a significant role in mitochondrial dynamics [50]. This could potentially serve as a mechanism for the interaction between SFXN1 and OPA1. Therefore, OPA1 interacts with a variety of genes that affect tumor metastasis and prognosis. OPA1 and these genes may work together in some signaling pathways and play roles in the occurrence and development of tumors.
In addition, we performed an enrichment analysis of OPA1 and its related and interacting genes, and the results showed that these genes play important roles in related signaling pathways such as nucleoplasmic transport and ribosome biosynthesis, and may influence tumor pathogenesis by affecting protein binding, RNA binding, and ATP binding.
The immune microenvironment is also an important influencing factor in tumorigenesis and development. Previous studies have shown that cancer-associated fibroblasts (CAF) are inhibitory intermediates in the tumor microenvironment and are associated with poor prognosis. They can polarize the responsive immune population, including macrophages, by secreting immune regulatory factors. Our prognostic analysis results have shown that the expression level of OPA1 is significantly correlated with the prognosis of cancer patients, so we speculate that OPA1 may affect the prognosis of patients by affecting the infiltration level of cancer-associated fibroblasts. As we suspected, the results showed a significant correlation between high OPA1 expression in tumors and high infiltration levels of cancer-associated fibroblasts.
Conclusion
In our study, we analyzed the role of OPA1 in the development of pancytopenia. Our findings suggest that OPA1 is highly expressed or mutated in a variety of tumors and is strongly associated with protein phosphorylation, patient prognosis, immune cell infiltration, and tumor mutational load. These findings help us to better understand the role of OPA1 in tumorigenesis development and potentially use it as a prognostic marker in some tumors.
Methods
OPA1 gene expression analysis
We used the TIMER 2.0 database to analyze OPA1 expression levels in various tumors and normal tissues adjacent to tumors [51]. Then, OPA1 expression level data from TCGA and GTEx databases were jointly analyzed using the GEPIA tool to obtain OPA1 expression differences in cancer and normal tissues [52]. To explore the expression of OPA1 at the protein level, we used the UALCAN database to mine the TCGA database for OPA1 protein expression data [53]. Then, we analyzed OPA1 gene expression level changes in various tumors at different stages through the GEPIA database. Taking the GEPIA database as an example, we can enter “OPA1” in the search box of GEPIA to search, then click the “DIY Expression” module, select “Boxplot” or “Stage plot”, and then click the tumor name to be analyzed in the “Datasets Selection” column to obtain the expression level of OPA1 in various tumors and tumor stages.
Survival analysis
To explore the association of OPA1 with the prognosis of cancer, we used the Kaplan-Meier plotter and the GEPIA databases to analyze the relationship between OPA1 expression levels and the OS and DFS of tumor patients and listed tumor types with significant statistical differences [54]. We entered “OPA1” in the search box of GEPIA to search, then clicked on the “Survival” module, selected “overall survival” or “RFS”, and then clicked on the tumor name to be analyzed in the “Datasets Selection” column to obtain the relationship between OPA1 and various tumor prognosis. The Kaplan-Meier plotter database operates similarly.
Analysis of OPA1 gene mutations
The cBioPortal database was used to analyze mutations in OPA1 in tumor tissue [55]. First, we found the ‘Quick selection’ section, then clicked on ‘TCGA Pan-Cancer Atlas Study’, and then typed ‘OPA1’ in the search field to query OPA1 gene alterations. In the ‘Cancer Type Summary’ module, there is the TCGA database of OPA1 alteration frequency, mutation type, and CNC in all tumors. By clicking on the “Mutation” module, we queried the information of the OPA1 mutation site displayed as a 3D structure map. In the “Comparison” module, we compared the changes in OS, DFS, PFS, and DSS of cancer patients with and without OPA1 gene alterations in the TCGA database, and the results were represented as Kaplan-Meier plots and indicated with log-rank p-values on the plots.
OPA1 subcellular localization
The Human Protein Atlas (HPA) database contains a wealth of human proteomics, transcriptomics, and systems biology data [56]. It contains a variety of protein expressions in various tumor tissues and normal tissues. Our study used the HPA database to analyze the localization of OPA1 proteins in three tumor cell lines: A431, U2OS, and U251.
Correlation analysis of OPA1 with tumor-associated fibroblast infiltration
To investigate how OPA1 expression in tumor cells affects the level of immune cell infiltration in the tumor microenvironment, we used the “Immunogene” module of the TIMER 2.0 tool to perform an analysis of OPA1 expression and infiltration of cancer-associated fibroblasts in the TCGA database. The immune cells we selected were cancer-related fibroblasts. We used three algorithms, TIDE, MCPCOUNTER, and EPIC, to assess the level of cancer-associated fibroblast and plotted these data as heat and scatter plots.
Tools and websites
cBioPortal: https://www.cBioPortal.org/; DAVID: https://david.ncifcrf.gov/; GEPIA: http://gepia.cancer-pku.cn/; GEPIA2: http://gepia2.cancer-pku.cn/#index; HPA: https://www.proteinatlas.org/; Venn: http://bioinformatics.psb.ugent.be/webtools/Venn/; Kaplan-Meier plotter: http://kmplot.com/analysis/; STRING: https://cn.string-db.org/; TIMER 2.0: http://timer.cistrome.org/; UALCAN: http://ualcan.path.uab.edu/index.html.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Author Contributions
ZW, NX, HZ, FC, and GW conceived and designed the study, and also critically revised the manuscript. ZW, NX, and HZ conducted the analysis and drafted the manuscript. ZW, NX, GL, WY, CZ, GW and DL contributed to the revision of the manuscript. All authors have read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
This study was approved by the Second Xiangya Hospital of Central South University Committee for Clinical Research.
Funding
This work was supported by Hunan Provincial Innovation Foundation for Postgraduate (CX20230116), the Fundamental Research Funds for the Central Universities of Central South University (2023ZZTS0026) and the National Key R&D Program of China: R&D and Demonstration Application of Key Technologies of Cloud-Integrated Visual-Audio-Touch Multi-Mode Interactive Feedback High Simulation Intelligent Virtual Surgery (2022YFF1202600). The funding sources and sponsor had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
References
- 1. Liao CP, Booker RC, Brosseau JP, Chen Z, Mo J, Tchegnon E, Wang Y, Clapp DW, Le LQ. Contributions of inflammation and tumor microenvironment to neurofibroma tumorigenesis. J Clin Invest. 2018; 128:2848–61. https://doi.org/10.1172/JCI99424 [PubMed]
- 2. Zhang K, Zhou J, Wu T, Tian Q, Liu T, Wang W, Zhong H, Chen Z, Xiao X, Wu G. Combined analysis of expression, prognosis and immune infiltration of GINS family genes in human sarcoma. Aging (Albany NY). 2022; 14:5895–907. https://doi.org/10.18632/aging.204191 [PubMed]
- 3. Cui X, Zhang X, Liu M, Zhao C, Zhang N, Ren Y, Su C, Zhang W, Sun X, He J, Gao X, Yang J. A pan-cancer analysis of the oncogenic role of staphylococcal nuclease domain-containing protein 1 (SND1) in human tumors. Genomics. 2020; 112:3958–67. https://doi.org/10.1016/j.ygeno.2020.06.044 [PubMed]
- 4. Petit JY, Lehmann A. [Role of plastic surgery in the treatment of breast cancer]. Bull Acad Natl Med. 1996; 180:317–28. [PubMed]
- 5. Maruo T, Zenda S, Shinozaki T, Tomioka T, Okano W, Sakuraba M, Tahara M, Hayashi R. Comparison of salvage surgery for recurrent or residual head and neck squamous cell carcinoma. Jpn J Clin Oncol. 2020; 50:288–95. https://doi.org/10.1093/jjco/hyz176 [PubMed]
- 6. Stiles ZE, Lohman RF, Mann GN. Plastic Surgery Reconstruction of Sarcoma Resection Defects: Form and Function. Surg Clin North Am. 2022; 102:583–99. https://doi.org/10.1016/j.suc.2022.04.008 [PubMed]
- 7. Dzwierzynski WW. Melanoma Risk Factors and Prevention. Clin Plast Surg. 2021; 48:543–50. https://doi.org/10.1016/j.cps.2021.05.001 [PubMed]
- 8. Myers JA, Miller JS. Exploring the NK cell platform for cancer immunotherapy. Nat Rev Clin Oncol. 2021; 18:85–100. https://doi.org/10.1038/s41571-020-0426-7 [PubMed]
- 9. Jiang G, Chen J, Li Y, Zhou J, Wang W, Wu G, Zhang Y. Association of SMC4 with prognosis and immune infiltration of sarcoma. Aging (Albany NY). 2023; 15:567–82. https://doi.org/10.18632/aging.204503 [PubMed]
- 10. Wu G, Zhou J, Zhu X, Tang X, Liu J, Zhou Q, Chen Z, Liu T, Wang W, Xiao X, Wu T. Integrative analysis of expression, prognostic significance and immune infiltration of RFC family genes in human sarcoma. Aging (Albany NY). 2022; 14:3705–19. https://doi.org/10.18632/aging.204039 [PubMed]
- 11. Wu G, Tian Q, Liu J, Zhou Q, Zou D, Chen Z, Wu T, Wang W, Xia H, Zhou J. Comprehensive analysis of expression and prognosis for LMNB family genes in human sarcoma. Medicine (Baltimore). 2022; 101:e28933. https://doi.org/10.1097/MD.0000000000028933 [PubMed]
- 12. Zhou J, Wang M, Zhou Z, Wang W, Duan J, Wu G. Expression and Prognostic Value of MCM Family Genes in Osteosarcoma. Front Mol Biosci. 2021; 8:668402. https://doi.org/10.3389/fmolb.2021.668402 [PubMed]
- 13. Annesley SJ, Fisher PR. Mitochondria in Health and Disease. Cells. 2019; 8:680. https://doi.org/10.3390/cells8070680 [PubMed]
- 14. Beckley MA, Shrestha S, Singh KK, Portman MA. The role of mitochondria in the pathogenesis of Kawasaki disease. Front Immunol. 2022; 13:1017401. https://doi.org/10.3389/fimmu.2022.1017401 [PubMed]
- 15. Vrettou S, Wirth B. S-Glutathionylation and S-Nitrosylation in Mitochondria: Focus on Homeostasis and Neurodegenerative Diseases. Int J Mol Sci. 2022; 23:15849. https://doi.org/10.3390/ijms232415849 [PubMed]
- 16. Lenaers G, Reynier P, Elachouri G, Soukkarieh C, Olichon A, Belenguer P, Baricault L, Ducommun B, Hamel C, Delettre C. OPA1 functions in mitochondria and dysfunctions in optic nerve. Int J Biochem Cell Biol. 2009; 41:1866–74. https://doi.org/10.1016/j.biocel.2009.04.013 [PubMed]
- 17. Herkenne S, Ek O, Zamberlan M, Pellattiero A, Chergova M, Chivite I, Novotná E, Rigoni G, Fonseca TB, Samardzic D, Agnellini A, Bean C, Di Benedetto G, et al. Developmental and Tumor Angiogenesis Requires the Mitochondria-Shaping Protein Opa1. Cell Metab. 2020; 31:987–1003.e8. https://doi.org/10.1016/j.cmet.2020.04.007 [PubMed]
- 18. Chow J, Rahman J, Achermann JC, Dattani MT, Rahman S. Mitochondrial disease and endocrine dysfunction. Nat Rev Endocrinol. 2017; 13:92–104. https://doi.org/10.1038/nrendo.2016.151 [PubMed]
- 19. La Merrill MA, Vandenberg LN, Smith MT, Goodson W, Browne P, Patisaul HB, Guyton KZ, Kortenkamp A, Cogliano VJ, Woodruff TJ, Rieswijk L, Sone H, Korach KS, et al. Consensus on the key characteristics of endocrine-disrupting chemicals as a basis for hazard identification. Nat Rev Endocrinol. 2020; 16:45–57. https://doi.org/10.1038/s41574-019-0273-8 [PubMed]
- 20. Jenkins S, Rowell C, Wang J, Lamartiniere CA. Prenatal TCDD exposure predisposes for mammary cancer in rats. Reprod Toxicol. 2007; 23:391–6. https://doi.org/10.1016/j.reprotox.2006.10.004 [PubMed]
- 21. Soto AM, Sonnenschein C. Environmental causes of cancer: endocrine disruptors as carcinogens. Nat Rev Endocrinol. 2010; 6:363–70. https://doi.org/10.1038/nrendo.2010.87 [PubMed]
- 22. Buha A, Matovic V, Antonijevic B, Bulat Z, Curcic M, Renieri EA, Tsatsakis AM, Schweitzer A, Wallace D. Overview of Cadmium Thyroid Disrupting Effects and Mechanisms. Int J Mol Sci. 2018; 19:1501. https://doi.org/10.3390/ijms19051501 [PubMed]
- 23. Giacomello M, Pyakurel A, Glytsou C, Scorrano L. The cell biology of mitochondrial membrane dynamics. Nat Rev Mol Cell Biol. 2020; 21:204–24. https://doi.org/10.1038/s41580-020-0210-7 [PubMed]
- 24. Chan DC. Mitochondrial Dynamics and Its Involvement in Disease. Annu Rev Pathol. 2020; 15:235–59. https://doi.org/10.1146/annurev-pathmechdis-012419-032711 [PubMed]
- 25. Li M, Wang L, Wang Y, Zhang S, Zhou G, Lieshout R, Ma B, Liu J, Qu C, Verstegen MMA, Sprengers D, Kwekkeboom J, van der Laan LJW, et al. Mitochondrial Fusion Via OPA1 and MFN1 Supports Liver Tumor Cell Metabolism and Growth. Cells. 2020; 9:121. https://doi.org/10.3390/cells9010121 [PubMed]
- 26. Wang Y, Li Y, Jiang X, Gu Y, Zheng H, Wang X, Zhang H, Wu J, Cheng Y. OPA1 supports mitochondrial dynamics and immune evasion to CD8+ T cell in lung adenocarcinoma. PeerJ. 2022; 10:e14543. https://doi.org/10.7717/peerj.14543 [PubMed]
- 27. Zamberlan M, Boeckx A, Muller F, Vinelli F, Ek O, Vianello C, Coart E, Shibata K, Christian A, Grespi F, Giacomello M, Struman I, Scorrano L, Herkenne S. Inhibition of the mitochondrial protein Opa1 curtails breast cancer growth. J Exp Clin Cancer Res. 2022; 41:95. https://doi.org/10.1186/s13046-022-02304-6 [PubMed]
- 28. Carmona-Carmona CA, Dalla Pozza E, Ambrosini G, Cisterna B, Palmieri M, Decimo I, Cuezva JM, Bottani E, Dando I. Mitochondrial Elongation and OPA1 Play Crucial Roles during the Stemness Acquisition Process in Pancreatic Ductal Adenocarcinoma. Cancers (Basel). 2022; 14:3432. https://doi.org/10.3390/cancers14143432 [PubMed]
- 29. Fridman WH, Galon J, Dieu-Nosjean MC, Cremer I, Fisson S, Damotte D, Pagès F, Tartour E, Sautès-Fridman C. Immune infiltration in human cancer: prognostic significance and disease control. Curr Top Microbiol Immunol. 2011; 344:1–24. https://doi.org/10.1007/82_2010_46 [PubMed]
- 30. Steven A, Seliger B. The Role of Immune Escape and Immune Cell Infiltration in Breast Cancer. Breast Care (Basel). 2018; 13:16–21. https://doi.org/10.1159/000486585 [PubMed]
- 31. Carmeliet P. Angiogenesis in life, disease and medicine. Nature. 2005; 438:932–6. https://doi.org/10.1038/nature04478 [PubMed]
- 32. Guo D, Wang Q, Li C, Wang Y, Chen X. VEGF stimulated the angiogenesis by promoting the mitochondrial functions. Oncotarget. 2017; 8:77020–7. https://doi.org/10.18632/oncotarget.20331 [PubMed]
- 33. Pernas L, Scorrano L. Mito-Morphosis: Mitochondrial Fusion, Fission, and Cristae Remodeling as Key Mediators of Cellular Function. Annu Rev Physiol. 2016; 78:505–31. https://doi.org/10.1146/annurev-physiol-021115-105011 [PubMed]
- 34. Murata D, Arai K, Iijima M, Sesaki H. Mitochondrial division, fusion and degradation. J Biochem. 2020; 167:233–41. https://doi.org/10.1093/jb/mvz106 [PubMed]
- 35. Patten DA, Wong J, Khacho M, Soubannier V, Mailloux RJ, Pilon-Larose K, MacLaurin JG, Park DS, McBride HM, Trinkle-Mulcahy L, Harper ME, Germain M, Slack RS. OPA1-dependent cristae modulation is essential for cellular adaptation to metabolic demand. EMBO J. 2014; 33:2676–91. https://doi.org/10.15252/embj.201488349 [PubMed]
- 36. Cipolat S, Rudka T, Hartmann D, Costa V, Serneels L, Craessaerts K, Metzger K, Frezza C, Annaert W, D'Adamio L, Derks C, Dejaegere T, Pellegrini L, et al. Mitochondrial rhomboid PARL regulates cytochrome c release during apoptosis via OPA1-dependent cristae remodeling. Cell. 2006; 126:163–75. https://doi.org/10.1016/j.cell.2006.06.021 [PubMed]
- 37. Dang L, White DW, Gross S, Bennett BD, Bittinger MA, Driggers EM, Fantin VR, Jang HG, Jin S, Keenan MC, Marks KM, Prins RM, Ward PS, et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature. 2009; 462:739–44. https://doi.org/10.1038/nature08617 [PubMed]
- 38. Sabharwal SS, Schumacker PT. Mitochondrial ROS in cancer: initiators, amplifiers or an Achilles' heel? Nat Rev Cancer. 2014; 14:709–21. https://doi.org/10.1038/nrc3803 [PubMed]
- 39. Czabotar PE, Lessene G, Strasser A, Adams JM. Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol. 2014; 15:49–63. https://doi.org/10.1038/nrm3722 [PubMed]
- 40. Izzo V, Bravo-San Pedro JM, Sica V, Kroemer G, Galluzzi L. Mitochondrial Permeability Transition: New Findings and Persisting Uncertainties. Trends Cell Biol. 2016; 26:655–67. https://doi.org/10.1016/j.tcb.2016.04.006 [PubMed]
- 41. Zhao X, Tian C, Puszyk WM, Ogunwobi OO, Cao M, Wang T, Cabrera R, Nelson DR, Liu C. OPA1 downregulation is involved in sorafenib-induced apoptosis in hepatocellular carcinoma. Lab Invest. 2013; 93:8–19. https://doi.org/10.1038/labinvest.2012.144 [PubMed]
- 42. Ivković I, Limani Z, Jakovčević A, Gajović S, Seiwerth S, Đanić Hadžibegović A, Prgomet D. Prognostic Significance of BRAF V600E Mutation and CPSF2 Protein Expression in Papillary Thyroid Cancer. Biomedicines. 2022; 11:53. https://doi.org/10.3390/biomedicines11010053 [PubMed]
- 43. Tsoi H, Man EP, Leung MH, Mok KC, Chau KM, Wong LS, Chan WL, Chan SY, Luk MY, Cheng CN, Khoo US. KPNA1 regulates nuclear import of NCOR2 splice variant BQ323636.1 to confer tamoxifen resistance in breast cancer. Clin Transl Med. 2021; 11:e554. https://doi.org/10.1002/ctm2.554 [PubMed]
- 44. Xu M, Liang H, Li K, Zhu S, Yao Z, Xu R, Lin N. Value of KPNA4 as a diagnostic and prognostic biomarker for hepatocellular carcinoma. Aging (Albany NY). 2021; 13:5263–83. https://doi.org/10.18632/aging.202447 [PubMed]
- 45. Yang J, Lu C, Wei J, Guo Y, Liu W, Luo L, Fisch G, Li X. Inhibition of KPNA4 attenuates prostate cancer metastasis. Oncogene. 2017; 36:2868–78. https://doi.org/10.1038/onc.2016.440 [PubMed]
- 46. Deng WW, Wu L, Bu LL, Liu JF, Li YC, Ma SR, Yu GT, Mao L, Zhang WF, Sun ZJ. PAK2 promotes migration and proliferation of salivary gland adenoid cystic carcinoma. Am J Transl Res. 2016; 8:3387–97. [PubMed]
- 47. Kory N, Wyant GA, Prakash G, Uit de Bos J, Bottanelli F, Pacold ME, Chan SH, Lewis CA, Wang T, Keys HR, Guo YE, Sabatini DM. SFXN1 is a mitochondrial serine transporter required for one-carbon metabolism. Science. 2018; 362:eaat9528. https://doi.org/10.1126/science.aat9528 [PubMed]
- 48. Chen L, Kang Y, Jiang Y, You J, Huang C, Xu X, Chen F. Overexpression of SFXN1 indicates poor prognosis and promotes tumor progression in lung adenocarcinoma. Pathol Res Pract. 2022; 237:154031. https://doi.org/10.1016/j.prp.2022.154031 [PubMed]
- 49. Shannon N, Gravelle R, Cunniff B. Mitochondrial trafficking and redox/phosphorylation signaling supporting cell migration phenotypes. Front Mol Biosci. 2022; 9:925755. https://doi.org/10.3389/fmolb.2022.925755 [PubMed]
- 50. Li T, Fu J, Zeng Z, Cohen D, Li J, Chen Q, Li B, Liu XS. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020; 48:W509–14. https://doi.org/10.1093/nar/gkaa407 [PubMed]
- 51. Cai Y, He Q, Liu W, Liang Q, Peng B, Li J, Zhang W, Kang F, Hong Q, Yan Y, Peng J, Xu Z, Bai N. Comprehensive analysis of the potential cuproptosis-related biomarker LIAS that regulates prognosis and immunotherapy of pan-cancers. Front Oncol. 2022; 12:952129. https://doi.org/10.3389/fonc.2022.952129 [PubMed]
- 52. Chandrashekar DS, Karthikeyan SK, Korla PK, Patel H, Shovon AR, Athar M, Netto GJ, Qin ZS, Kumar S, Manne U, Creighton CJ, Varambally S. UALCAN: An update to the integrated cancer data analysis platform. Neoplasia. 2022; 25:18–27. https://doi.org/10.1016/j.neo.2022.01.001 [PubMed]
- 53. Chen S, Wei Y, Liu H, Gong Y, Zhou Y, Yang H, Tang L. Analysis of Collagen type X alpha 1 (COL10A1) expression and prognostic significance in gastric cancer based on bioinformatics. Bioengineered. 2021; 12:127–37. https://doi.org/10.1080/21655979.2020.1864912 [PubMed]
- 54. Wu P, Heins ZJ, Muller JT, Katsnelson L, de Bruijn I, Abeshouse AA, Schultz N, Fenyö D, Gao J. Integration and Analysis of CPTAC Proteomics Data in the Context of Cancer Genomics in the cBioPortal. Mol Cell Proteomics. 2019; 18:1893–8. https://doi.org/10.1074/mcp.TIR119.001673 [PubMed]
- 55. Zhou C, Chen Z, Xiao B, Xiang C, Li A, Zhao Z, Li H. Comprehensive analysis of GINS subunits prognostic value and ceRNA network in sarcoma. Front Cell Dev Biol. 2022; 10:951363. https://doi.org/10.3389/fcell.2022.951363 [PubMed]
- 56. Szklarczyk D, Gable AL, Nastou KC, Lyon D, Kirsch R, Pyysalo S, Doncheva NT, Legeay M, Fang T, Bork P, Jensen LJ, von Mering C. The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021; 49:D605–12. https://doi.org/10.1093/nar/gkaa1074 [PubMed]
- 57. Bardou P, Mariette J, Escudié F, Djemiel C, Klopp C. jvenn: an interactive Venn diagram viewer. BMC Bioinformatics. 2014; 15:293. https://doi.org/10.1186/1471-2105-15-293 [PubMed]
- 58. Chen L, Lu D, Sun K, Xu Y, Hu P, Li X, Xu F. Identification of biomarkers associated with diagnosis and prognosis of colorectal cancer patients based on integrated bioinformatics analysis. Gene. 2019; 692:119–25. https://doi.org/10.1016/j.gene.2019.01.001 [PubMed]