Introduction
ATPases associated with diverse cellular activities (AAA+ ATPase) are vital enzymes in all organisms, which are involved in various cellular processes, such as DNA replication, protein degradation, membrane fusion, signal transduction, and gene expression [1, 2]. Chemical inhibition on ATPase activity of the AAA+ ATPases impedes cancer progression, including multiple myeloma (MM) [3, 4]. The valosin containing protein (VCP, also known as p97 in mammals) is a member of the AAA+ ATPase family that is a multifunctional protein influencing protein metabolism and intracellular homeostasis to regulate cellular processes, such as cell cycle, DNA replication, virus replication, protein homeostasis, genome stability and repair, mitosis, and transcription [5–11].
It is known that VCP is elevated in many cancers and associated with poor prognosis, such as lymphoma [12], colorectal cancer [13], ovarian cancer [14] and prostate cancer [15]. Phospho-Ser784-VCP is required for DNA repair, checkpoint signaling, and cell survival in response to a broad range of genotoxins and correlates with poor outcomes in chemotherapy-treated breast cancer patients [16]. VCP regulates actin and cell motility via the Rho-ROCK dependent pathway in cancer development [17]. The interaction of VCP and ubiquitin regulatory X (UBX) cofactors mediates the binding of various E3 ligases to direct VCP for certain protein degradation processes [18]. VCP regulates the expression and function of the transcription factor c-Myc, and VCP-dependent degradation of ubiquitylated c-Myc on chromatin is important for promoting its function on gene expression [19]. In addition, VCP has been implicated in a variety of metabolic processes at the gene expression level in a diverse range of cancer cell lines and in patient-derived MM cells. The depletion of Glutamine leads to elevated expression of VCP, whereas VCP inhibition hampers the metabolic processes and intracellular amino acid turnover [20].
Recently, some studies have revealed that VCP is a potential target for cancer therapy including MM, and confirmed the significant biological effects of small molecule inhibitors against VCP [21–23]. Quinazoline-based VCP inhibitors induce G1 cell cycle arrest, decrease cap-dependent translation and mediate programmed cell death in ovarian cancer cells [14]. DBeQ, a selective and reversible inhibitor of VCP ATPase, arrests cell cycle at G1 phase and blocks the degradation of p21 and p27 in breast cancer cells [24]. In addition, DBeQ cooperates with bortezomib to induce MM cell apoptosis [22]. NMS-873 targeting a region in VCP spans D1 and D2 domains of adjacent polymers to activate the unfolded protein response (UPR), interferes with autophagy and induces cancer cell death [25]. Targeting VCP with CB-5083 (a potent inhibitor of VCP’s D2 ATPase domain) leads to an accumulation of poly-ubiquitinated proteins, retention of endoplasmic reticulum-associated degradation (ERAD) substrates, and generation of irresolvable proteotoxic stress, finally causes the activation of apoptotic arm of the UPR [26]. Additionally, CB-5083 promotes tumor cell killing following ionizing radiation both in vitro and in vivo [27]. However, due to the toxicity of CB-5083, two Phase I clinical trials of CB-5083 are discontinued. It is of great urgency to develop novel compounds to meet medical demand. Our team designed and synthesized a series of VCP inhibitors. Among these inhibitors, compound 35 (VCP20) exhibited significant in vitro activities and microsomal stabilities [28].
In the present study, we examined the inhibitory effect of VCP20 on MM models. Inhibition of VCP with VCP20 suppressed MM cell proliferation through inhibiting NF-κB signaling pathway. In addition, VCP20 inhibited osteoclast differentiation, however MM cell-derived exosomes attenuated its inhibitory effect probably due to the high expression of VCP in MM cell-derived exosomes. Furthermore, VCP20 prolonged the survival of MM model mice and improved their bone destruction. Taken together, we disclose that VCP is a potential therapeutic target for MM and VCP20 may be a promising agent for MM treatment.
Results
Increased VCP expression correlates with poor survival in MM patients
Initially, we examined VCP expression in normal plasma (NP, n = 22), monoclonal gammopathy of undetermined significance (MGUS, a pre-MM disease, n = 44) and MM samples (n = 351) from gene expression profiling (GEP) database. The levels of VCP were obviously increased in MM samples compared with NP and MGUS samples (***p < 0.0001) (Figure 1A). Notably, MM patients with high VCP expression suffered from poor clinical outcomes relative to low-VCP-expressing patients in TT2 cohort, which was presented as shorter response duration of overall survival (OS) (n = 351, *p = 0.0456) (Figure 1B). Furthermore, we tested the mean values of VCP levels in eight widely recognized subgroups, and found that elevated VCP was particularly prevalent in proliferation (PR) group, the worst subgroup in MM characterized by high proliferation (***p < 0.0001) (Figure 1C). Here, we assume that VCP may be a high-risk gene to promote MM progression.
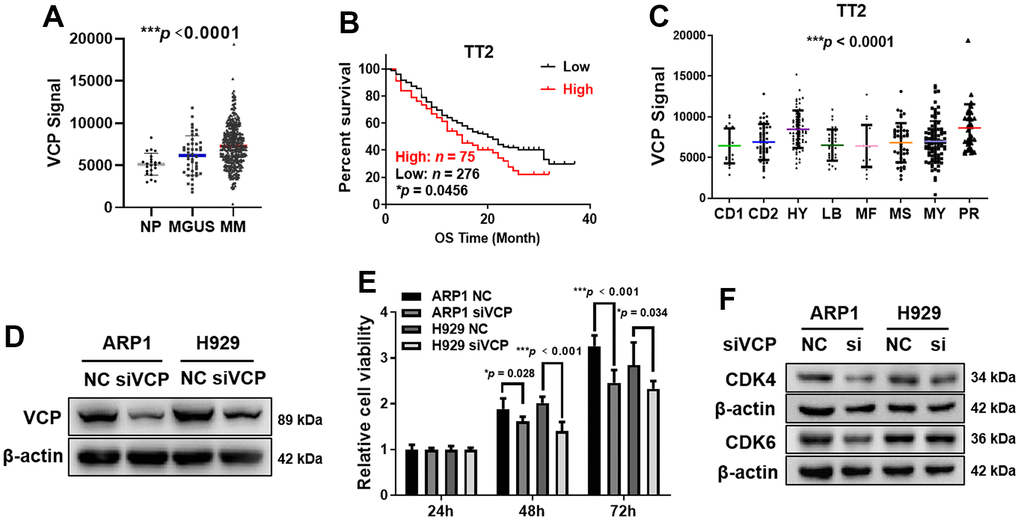
Figure 1. Elevated VCP is associated with poor survival in MM patients and siVCP inhibits MM cell proliferation in vitro. (A) VCP mRNA levels were significantly increased in MM samples (n = 351) compared with NP (n = 22) and MGUS (n = 44) samples. (B) Higher VCP level was associated with a shorter OS in TT2 cohort (n = 351, High: n = 75, Low: n = 276). (C) Box plot representing VCP expression in eight MM subgroups from TT2 cohort. (D) WB analysis of VCP expression in siVCP cells. (E) MTT assay examined the effect of siVCP on MM cell proliferation. (F) WB analysis showed that siVCP inhibited CDK4/6 expression in MM cells. The MM patient survival data were plotted by Kaplan-Meier curve, and the survival of patients with low and high expression of VCP was compared through a log-rank test. Data are presented as the mean ± SD; *p < 0.05; **p < 0.01; ***p < 0.001.
Downregulation of VCP inhibits MM cell viability in vitro
To examine whether VCP is an oncogene in MM, we interfered VCP expression by using small interfering RNA (siRNA) in ARP1 and H929 cells. WB analysis demonstrated successful knockdown of VCP in ARP1 and H929 cells at protein level (Figure 1D and Supplementary Figure 1A). Subsequently, we adopted MTT assay to explore the correlation between VCP and cell proliferation, which showed that MM cell proliferation was significantly decreased in siVCP cells compared with negative control (NC) after 72 h (***p < 0.001, *p = 0.034) (Figure 1E). As key regulators of cellular transition from G1 to S phase [29], CDK4/6 can promote cell cycle dysregulation and regulate cell proliferation in MM cells [30]. We measured CDK4/6 protein expression upon siVCP and found that interfering VCP decreased the expression of CDK4/6 (Figure 1F and Supplementary Figure 1A), suggesting that VCP participated in regulating MM cell cycle via altering CDK4/6 expression. Above results indicate that VCP may act as an oncogene in MM and targeting VCP leads to a decreased cell proliferation in MM.
Compound VCP20 specifically targets VCP to impede MM cell proliferation and promote cell apoptosis in vitro
CB-5083 was the first selective VCP inhibitor with the required pharmacological properties and showed promising preclinical activity, however it presented high toxicity in the clinical trials [26, 28]. It is certainly worthwhile to explore high-effective VCP inhibitors with low-toxicity. Therefore, our team synthesized a series of novel VCP inhibitors, including VCP20 (compound 35) with better VCP inhibitory activities (36.4 nM) (Figure 2A) [28]. Molecular docking of receptor-based 3D-QSAR models for VCP and VCP20 showed that VCP20 exclusively bound into VCP active pocket (Figure 2B), indicating that VCP20 specifically targeted VCP protein.
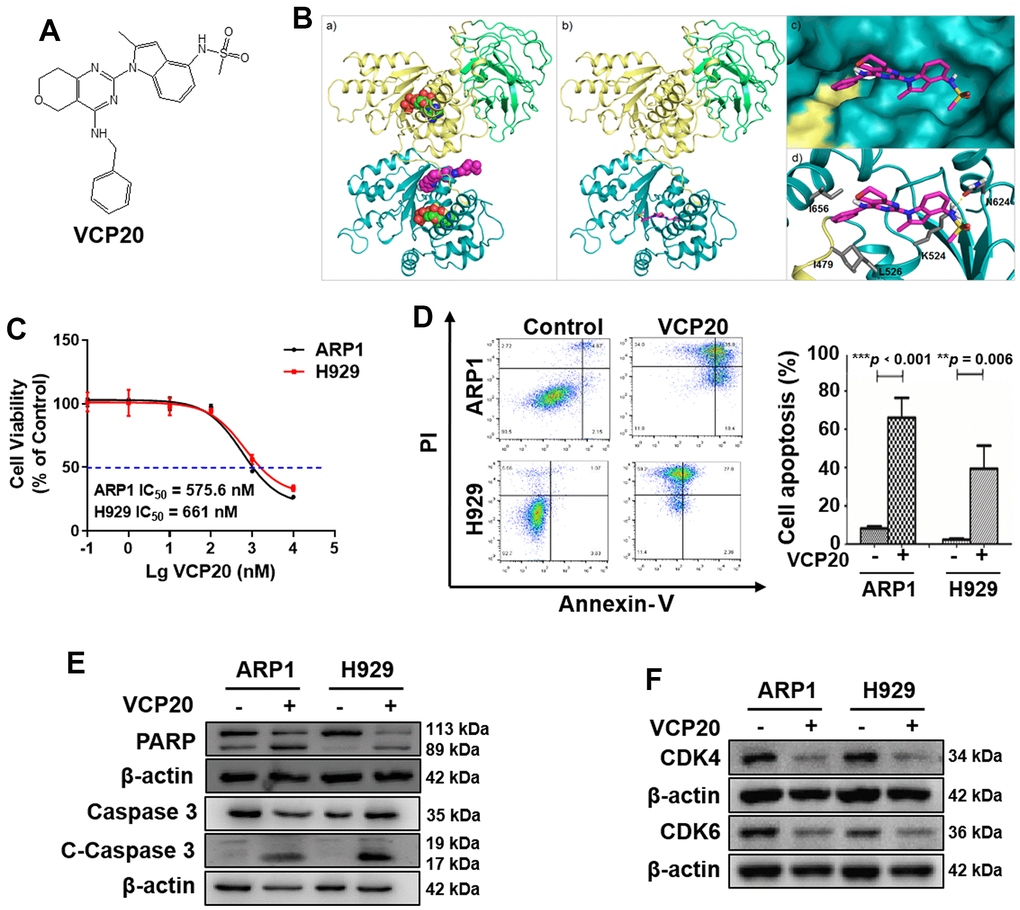
Figure 2. VCP20 inhibits MM cell proliferation and induces cell apoptosis. (A) The structure of VCP20. (B) Predicted binding modes of VCP20 targeting VCP. (C) Inhibition ratio of VCP20 in MM cells. (D) Flow cytometry analysis indicated that VCP20 induced MM cell apoptosis. (E) WB analysis showed that VCP20 increased the expressions of apoptotic protein. (F) WB analysis showed that VCP20 decreased the CDK4/6 expression. The concentration of VCP20 was as follow: ARP1, 575.6 nM; H929, 661 nM. Data are presented as the mean ± SD; **p < 0.01; ***p < 0.001.
We performed MTT assay to evaluate the IC50 of VCP20 on ARP1 and H929 cells. The results showed that VCP20 suppressed MM cell proliferation evidently (ARP1, IC50 = 575.6 nM; H929, IC50 = 661 nM) (Figure 2C). Moreover, we tested the effect of VCP20 on the apoptosis level of MM cells. Flow cytometry assays demonstrated that VCP20 significantly induced MM cell apoptosis (***p < 0.001, **p = 0.006) (Figure 2D). We also detected the expressions of cleaved-PARP and cleaved-Caspase 3, and found that VCP20 induced the elevation of cleaved-PARP and cleaved-Caspase 3 in MM cells (Figure 2E and Supplementary Figure 1B). In addition, VCP20 decreased the expression of CDK4/6 (Figure 2F and Supplementary Figure 1B), which was consistent with the result in siVCP cells. These data indicate that VCP20 inhibits MM cell proliferation and accelerates cell apoptosis.
VCP regulates MM cell proliferation via NF-κB signaling pathway
Since above findings have shed light on the vital role of VCP in MM progression, we followed to investigate the underlying mechanism. Through RNA-seq technology, we identified 164 downregulated genes and 102 upregulated genes between ARP1 siVCP and NC cells, and made the volcano plot of these differentially expressed genes. Volcano plot showed the upregulated (red) and downregulated (blue) genes upon silencing VCP in MM cells. (Figure 3A). KEGG pathway enrichment analysis for these differentially expressed genes demonstrated that the NF-kappa B (NF-κB) signaling was ranked in the top pathway (Figure 3B). It is known that NF-κB transcription factors play key roles in the pathogenesis of MM [31, 32]. We performed WB assay to examine the effect of VCP on the expressions of NF-κB P65 (P65) and phosphorylated-NF-κB P65 (P-P65) in MM cells. The expressions of P65 and P-P65 were observably decreased upon VCP inhibition. Furthermore, the expression of P65 in both of the cytoplasm and nucleus were significantly inhibited by siVCP (Figure 3C and Supplementary Figure 2A). Similar to the above results, the expressions of P65 and P-P65 were also downregulated in ARP1 and H929 cells with VCP20 treatment (Figure 3D and Supplementary Figure 2B). NF-κB activation is associated with the degradation of IκBα [33], and VCP interacts with IκBα to accelerate IκBα degradation and NF-κB activation [34]. Therefore, we detected the effect of VCP20 on IκBα protein expression. It was found that VCP20 enhanced IκBα protein expression remarkably (Figure 3E and Supplementary Figure 2B). Subsequently, we examined the effect of VCP20 on the level of IκBα ubiquitination. Intriguingly, ubiquitination of IκBα was decreased in both ARP1 and H929 cells after treated with VCP20 (Figure 3F). Taken together, these findings unveil that VCP20 inhibits NF-κB activation by decreasing the ubiquitination of IκBα to suppress MM cell proliferation.
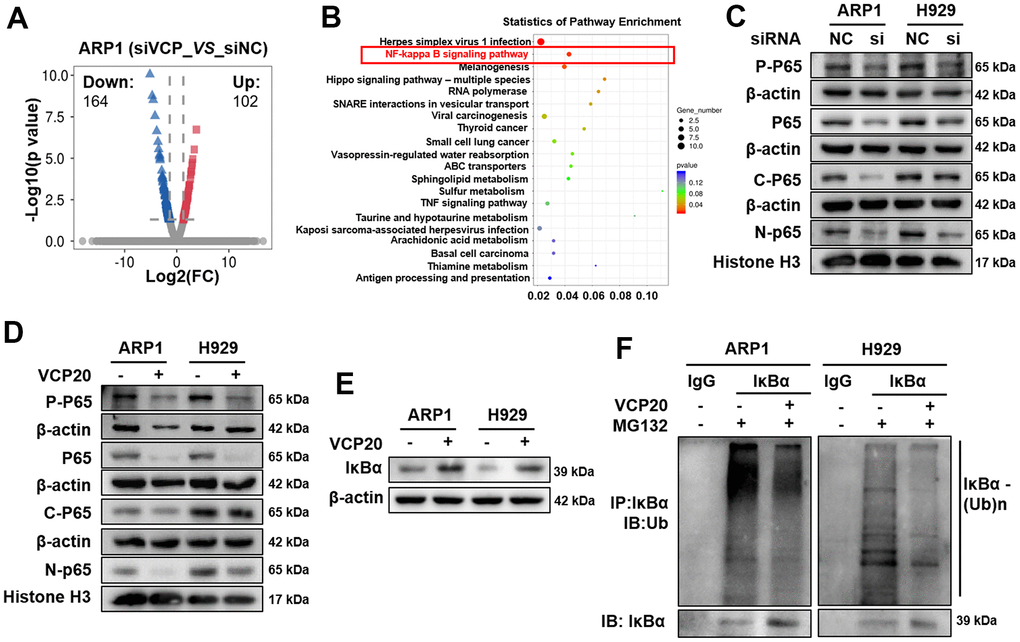
Figure 3. VCP regulates MM cell proliferation via modulating NF-κB signaling pathway. (A) Volcano plots showed the upregulated (red) and downregulated (blue) genes upon silencing VCP in MM cells. (B) KEGG pathway analysis of RNA-seq data indicated that VCP was associated with NF-κB signaling pathway. (C) WB analysis of P-P65 and P65 expressions in siVCP cells compared with NC cells. (D) WB analysis of P-P65 and P65 expressions in the cells treated with VCP20 compared with WT cells. (E) VCP20 increased IκBα expression. (F) The ubiquitination level of IKBα in MM cells with the treatment of VCP20 or not.
VCP20 inhibits osteoclast differentiation via inactivating NF-κB signaling pathway
One of the characteristics of MM is bone disease caused by the imbalance between osteoclast and osteoblast activity [35]. NF-κB is a dimeric transcription factor complex that plays a vital role in osteoclastogenesis [36]. It has been confirmed that VCP is involved in MM progression via NF-κB pathway, hence we checked whether VCP20 would be associated with osteoclastogenesis. TRAP staining showed that VCP20 significantly inhibited the formation of osteoclast cells (*p = 0.0106) (Figure 4A, 4B). MM cell-derived exosomes can promote osteoclast differentiation and aggravate bone disease in MM [37, 38]. As VCP is associated with membrane fusion and considered as an exosome protein marker [39], we questioned whether VCP could exist in the exosomes derived from MM cells and promote osteoclast differentiation through exosomes. It was found that exosomes from ARP1 WT cells could be isolated by gradient ultracentrifugation, which were confirmed by using transmission electron microscopy (Figure 4C). The existence of VCP protein in exosomes derived from ARP1 WT cells was detected by WB method (Figure 4D). Furthermore, MTT assays showed that VCP20 inhibited MM cell proliferation significantly, meanwhile exosomes derived from ARP1 WT cells alleviated the inhibition effect of VCP20 on MM cell proliferation remarkably (***p < 0.001) (Figure 4E). We also detected the effects of exosomes extracted from ARP1 WT cells on osteoclastogenesis. TRAP activity results revealed that the exosomes significantly reversed the inhibition effect of VCP20 on the formation of osteoclast compared with Control cells (*p = 0.035, *p = 0.033, **p = 0.0058) (Figure 4F, 4G). Furthermore, the expressions of NFATC1 (an osteoclast marker [40]), P65 and P-P65 protein in RAW264.7 cells were examined by WB assay. WB results indicated that the expressions of NFATC1 and P-P65 were markedly decreased upon VCP20 treatment, in contrast exosomes derived from ARP1 WT cells partially increased the expressions of NFATC1 and P-P65 (Figure 4H and Supplementary Figure 3). In addition, RT-PCR analysis showed that the relative mRNA levels of osteoclast differentiation-related genes, such as ACP5, CTSK, MMP9 and TM7SF4, were significantly decreased in VCP20 plus RANKL-induced RAW264.7 cells compared with RANKL-induced RAW264.7 cells, in contrast exosomes from ARP1 WT cells partially reversed the inhibitory effect (*p < 0.05, *p < 0.01, ***p < 0.001) (Figure 4I). Collectively, these findings suggest that VCP20 suppresses osteoclastogenesis via inhibiting NF-κB signaling pathway, and exosomes derived from MM cells partially abrogates the inhibition effect of VCP20 on osteoclastogenesis.
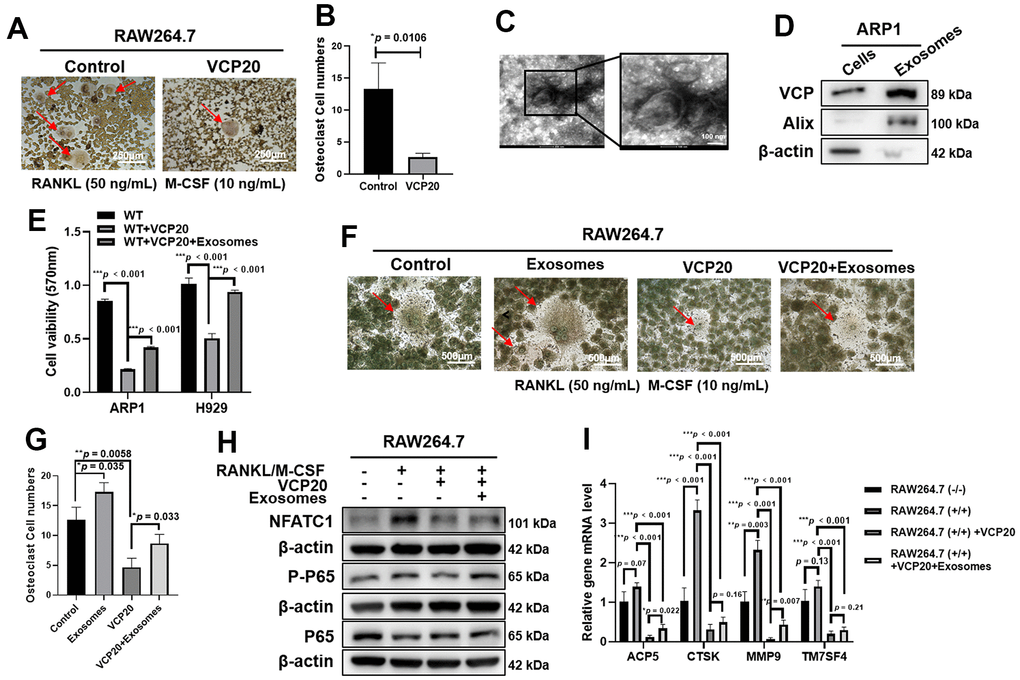
Figure 4. VCP20 inhibits osteoclast differentiation via downregulating NF-κB P65. (A) TRAP staining showed that VCP20 inhibited osteoclast differentiation in RAW 264.7 cells treated with RANKL and M-CSF. Scale bar: 250 μm. (B) Quantitative analysis of multinucleated osteoclasts. (C) The biological characteristics of exosomes were detected by transmission electron microscopy. Scale bar: 100 nm. (D) Detection of VCP, Alix and β-actin in ARP1 WT cells and exosomes from ARP1 WT cells by WB analysis. (E) Exosomes from ARP1 WT cells rescued cell proliferation inhibited by VCP20 in MM cells (1×106 MM cells were treated with 20 μg exosomes or not for 48 h). (F) TRAP staining revealed that exosomes from ARP1 WT cells induced osteoclast differentiation. Scale bar: 250 μm. (G) Quantitative analysis of multinucleated osteoclasts. (H) WB assay confirmed that exosomes from ARP1 WT cells increased the expressions of P-P65 and NFATC1 that were downregulated by VCP20 in RAW264.7 cells. (I) RT-PCR assay showed that VCP20 decreased the levels of osteoclast differentiation related markers, and this effect was partially compensated by exosomes from ARP1 WT cells. VCP20: 100 nM; exosomes: 2 μg/mL each well. Data are presented as the mean ± SD; *p < 0.05; **p < 0.01; ***p < 0.001.
VCP20 retards xenograft tumor formation, improves bone destruction and prolongs the survival of 5TMM3VT model mice in vivo
As to further verify the role of VCP20 in vivo, we established cell-line derived xenograft model by injecting 1×106 ARP1 WT cells subcutaneously to NOD/SCID mice (Control and VCP20; n = 4). After 31 days, the tumors in VCP20 group were much smaller than the tumors in Control group (Figure 5A, 5B). The mean weight of tumors in VCP20 group was significantly lower than that in Control group (*p = 0.015) (Figure 5C). Time course regression analyses of growth rate showed that the average volume of the tumors in VCP20 group were also significantly lower than that in Control group statistically (**p = 0.0013) (Figure 5D). Above data demonstrates that targeting VCP by VCP20 retards the development of MM in vivo, and VCP20 may act as a promising compound for MM treatment.
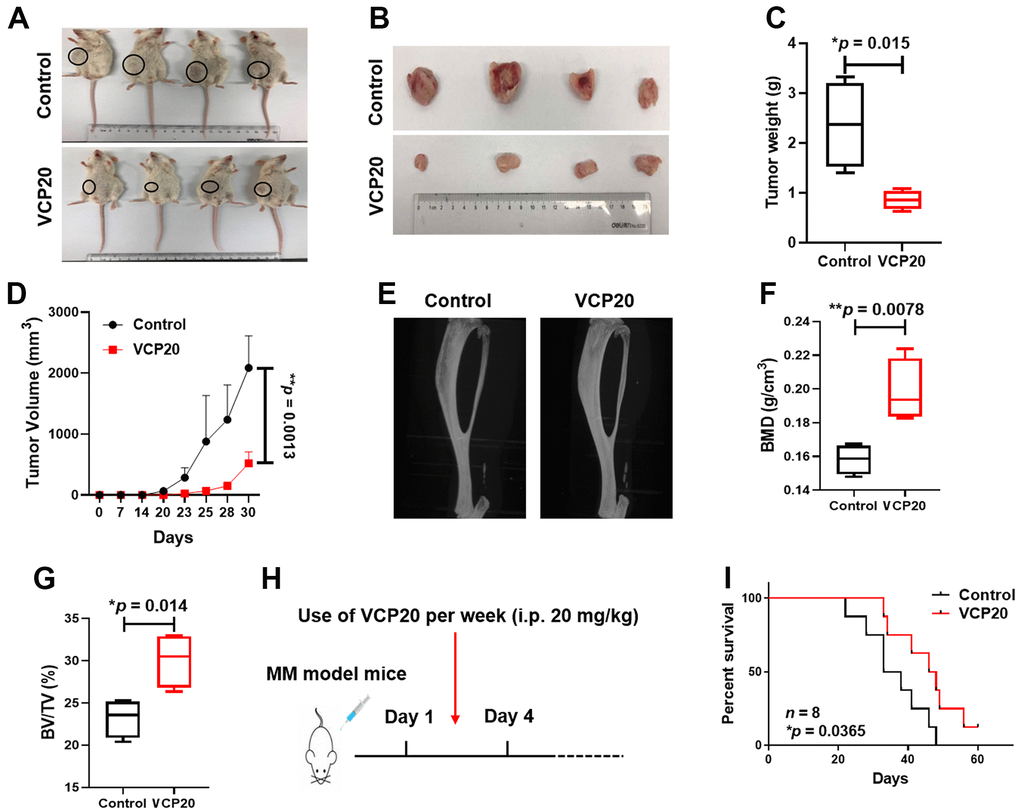
Figure 5. VCP20 impedes MM cell proliferation and prolongs MM mice survival in vivo. (A) Photographic images of xenograft mice at Day 31 (n = 4, VCP20: i.p. 20 mg/kg, twice a week). (B) Images of the harvested xenograft tumors. (C) Mean tumor weight of NOD/SCID mice. (D) Time course of tumor growth. V = 0.52 × larger diameter × (smaller diameter)2. (E) Representative micro-CT images of the bones. (F) BMD of 5TMM3VT mice in Control and VCP20 groups. (G) BV/TV of 5TMM3VT mice in Control and VCP20 groups. (H) VCP20 treatment started from the day after injection (VCP20: i.p. 20 mg/kg, twice a week) and continued until the mice were dead. (I) VCP20 extended the survival period of MM mice (n = 8). Data are presented as the mean ± SD; *p < 0.05; **p < 0.01.
In addition, we employed 5TMM3VT mouse model to evaluate the effect of VCP20 on osteoclastogenesis in vivo. After one month of modeling, the 5TMM3VT model mice presented paralysis of the hind limbs. Micro-CT analysis indicated that the bone destruction was milder in VCP20 mice than Control mice (Figure 5E). The differences between bone mineral density (BMD) and bone volume fraction (BV/TV) in two groups were deeply analyzed. The mean BMD was 0.158 g/cm3 and BV/TV was 23.2% in control mice, by contrast the mean BMD was 0.198 g/cm3 and BV/TV was 30.06% in VCP20 mice (**p = 0.0078, *p = 0.014) (Figure 5F, 5G), suggesting that VCP20 significantly improved bone destruction in MM mice. We further confirmed the therapeutic effect of VCP20 on the survival of MM mice. It was found that VCP20 significantly prolonged the survival of MM mice relative to the untreated mice (*p = 0.0365) (Figure 5H, 5I). All the data above demonstrate that targeting VCP by VCP20 improves bone marrow environment and prolongs the survival of MM mice in vivo.
Discussion
MM is a plasma cell malignancy characterized by complex heterogeneous cytogenetic abnormalities [41]. Although novel targeted drugs have shown great prospects in overcoming conventional drug resistance and improving patient prognosis, MM remains incurable. Therefore, it is an emergency to seek novel targets and related inhibitors to improve MM treatment. To date, elevated VCP expression has been identified in many cancer types and associated with patient outcomes, which is correlated with aggressiveness and drug resistance [18, 42, 43]. VCP promotes cancer cell proliferation or invasion and metastasis in lymphoma [12], colorectal cancer [13], prostate cancer [15], lung cancer [34], breast cancer [16, 44] and pancreatic cancer [45]. In present study, we investigated the function of VCP in MM to confirm if VCP was representing a potential therapeutic target, and identified that a specific VCP inhibitor (VCP20) targeted VCP to inhibit MM cell proliferation and osteoclast differentiation.
We clarified that VCP mRNA level was elevated in MM, which was associated with poor survival of patients with MM. Schweitzer K et al. have demonstrated that VCP promotes Cullin-RING-ubiquitin-ligase/proteasome-dependent degradation of IκBα [46]. Silencing VCP leads to depressed IκBα degradation and NF-κB activation [34]. Our data confirmed that inhibition of VCP significantly inactivated NF-κB signaling pathway to impede MM cell proliferation. It is a promising strategy to develop small-molecule inhibitors of VCP for cancer treatment. The reported inhibitors of VCP include CB-5083 [45], CB-5339 [47], NMS-873 [48], PPA [42], DBeQ [49], OSSL_325096 [21], FQ393 [50] and so on. CB-5083 entered two phase I clinical trials, but the trials were halted for the toxicities caused by off-target effects [51, 52]. VCP20 (compound of 35) is a novel pyrimidine structure VCP inhibitor designed from CB-5083, which shows excellent enzymatic inhibitory activity [28]. In this study, VCP20 was employed to treat MM in vitro and in vivo. VCP20 significantly inhibited MM cell proliferation via inhibiting the ubiquitination degradation of IκBα to inactivate NF-κB signaling pathway.
Like most malignancies, MM cells alter its microenvironment to promote tumor progression that confers resistance to conventional therapy [53]. Paget's disease of bone (PDB) is a common skeletal disorder characterized by abnormal focal bone remodeling [54], in which VCP is a susceptibility gene in PDB pathogenesis [55]. We assumed that VCP might participate in the regulation of bone marrow microenvironment. We examined whether VCP20 could affect osteoclast differentiation. Intriguingly, VCP20 evidently inhibited osteoclast differentiation to improve bone lesion, suggesting that targeting VCP might be an impactful way to ameliorate bone marrow microenvironment. Exosomes secreted by stromal cells and tumor cells are critical regulators of intercellular communication in tumor microenvironment, which are particularly suitable for the transport and intracellular delivery of proteins and nucleic acids [56, 57]. Tumor-derived exosomes remodel the bone marrow environment, inhibit anti-leukemia immunity, mediate drug resistance and interfere with immunotherapies in hematological malignancies [58]. MM cell-derived exosomes promote osteoclast differentiation to support MM cell proliferation [37]. Of note, we identified VCP existing in MM cell-derived exosomes, which indicated that VCP could be released to bone marrow microenvironment to take effect.
Alberto Bosque et al. have reported that targeting VCP may be a new selective strategy to prevent tumoral exosome secretion in cancer treatment [59]. Our further study revealed that MM cell-derived exosomes significantly reversed the inhibitory effect of VCP20 on MM cell proliferation and osteoclast differentiation, indicating that VCP released by exosomes promoted MM progression and VCP20 could alleviate the cell proliferation promotion caused by VCP secretion. Moreover, mechanism study indicated that VCP20 inhibited NF-κB signaling pathway and suppressed NFATC1 expression, leading to restraining osteoclastogenesis. To further extend our research, we employed 5TMM3VT mouse model to validate the therapeutic effect of VCP20 in vivo. We observed that VCP20 significantly prolonged the survival of MM mice and improved the bone destruction. In the present study, we revealed that VCP20 effectively inhibited MM cell proliferation and bone lesion formation through targeting VCP in the bone marrow microenvironment.
In conclusion, we demonstrate for the first time that VCP20 targeting VCP is a promising agent for inhibiting MM cell proliferation and improving bone marrow microenvironment in MM. Figure 6 illustrates the carcinogenesis role of VCP in MM and inhibition of VCP impedes MM progression via NF-κB signaling pathway. Targeting VCP with VCP20 may thus be a novel strategy in the treatment of MM.
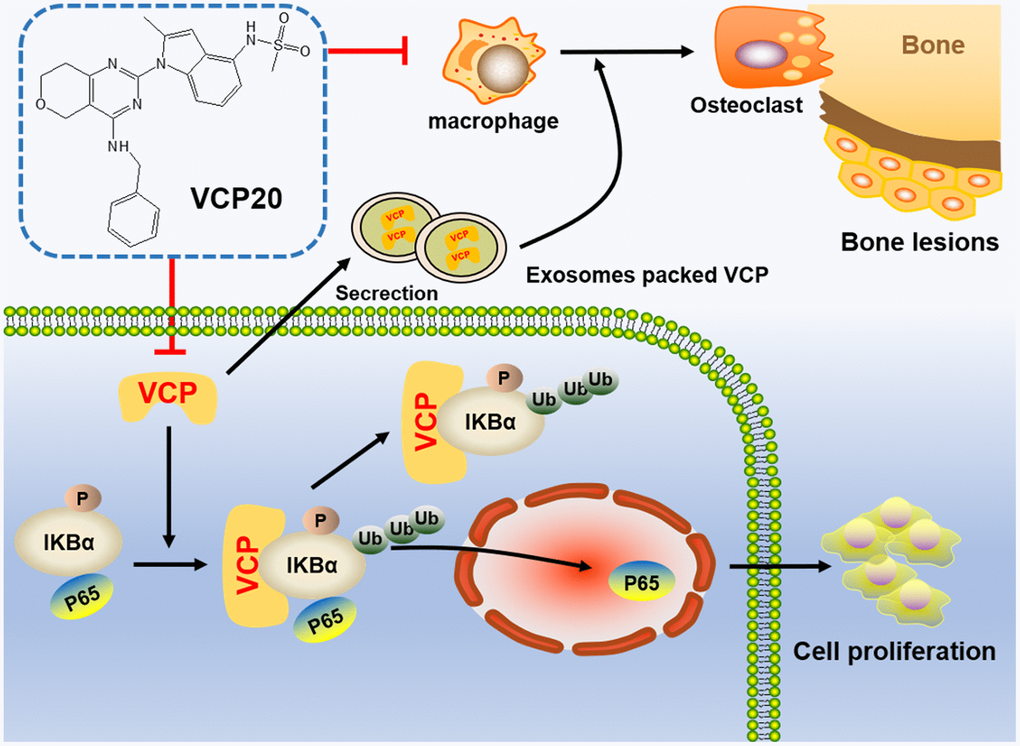
Figure 6. Schematic depiction illustrates that VCP20 targeting VCP is a promising agent for inhibiting cellular proliferation and improving bone marrow microenvironment in MM.
Materials and Methods
Gene expression profiling
The Gene expression profiling (GEP) cohorts were collected from GEO database. Total therapy 2 (TT2) cohort was collected from GSE2658.
Antibodies and reagents
The following antibodies were used, VCP (10736-1-AP, Proteintech, Wuhan, China), P65 (8242S, Cell Signaling Technology, Danvers, MA, USA), P-P65 (3033S, Cell Signaling Technology), PARP (9542S, Cell Signaling Technology), Cleaved Caspase-3 (9661S, Cell Signaling Technology), CDK4 (11026-1-AP, Proteintech), CDK6 (14052-1-AP, Proteintech), Histone H3 (17168-1-AP, Proteintech), CD63 (216130, Abcam, Cambridge, UK), IκBα (9242S, Cell Signaling Technology), Ubiquitin (43124S, Cell Signaling Technology), β-actin (60008-1-Ig, Proteintech). Secondary antibodies included goat anti-Rabbit IgG (H+L) HRP (FMS-Rb01, Fcmacs, Nanjing, China) or mouse (S0002, Affinity, Changzhou, China). MTT was purchased from Solarbio (M8180, Beijing, China). Propidium iodide (PI) was purchased from Beyotime (ST511, Shanghai, China), Annexin-V-FITC was purchased from Biolegend (640906, San Diego, CA, USA). Compound VCP20 was obtained from Professor Yongqiang Zhu (Nanjing Normal University).
Cell lines and cell culture
Human MM cell lines, ARP1 and H929, were cultured in RPMI 1640 medium (Biological Industries, Kibbutz Beit Haemek, Israel) and RAW264.7 were cultured in DMEM medium (Biological Industries) containing 10% fetal bovine serum (Invigentech; Irvine, CA, USA), penicillin and streptomycin solution (NCM, Suzhou, China). All cells were cultured in humidified 5% CO2 incubators at 37° C.
Transient transfection
Small interfering RNA (siRNA) was transfected into MM cells by using electro transmitter (BTX, Holliston, MA, USA). The process was performed as described previously [60]. The specific siRNAs were synthesized by Shanghai Genepharma. The sequences of siRNAs were as following: negative control (NC, sense 5'-UUCUCCGAACGUGUCACGUTT-3' and anti-sense 5'-ACGUGACACGUUCGGAGAATT-3'); VCP (sense 5'- CCAACAGACCCAACAGCAUTT-3' and anti-sense 5'- AUGCUGUUGGGUCUGUUGGTT-3').
Cell proliferation and viability assay
MM cells (5×103) were seeded in each well of 96-well plate. The relative cell viability was calculated as the ratio of absorbance at a certain time relative to the mean value of 24-hour absorbance.
As to detect the effect of VCP20 on MM cell proliferation, cells were treated with different concentrations of VCP20 for 48 h to calculate the IC50 values in MM cells. Cell viability was evaluated by MTT assay according to the manufacturer’s instructions. The absorbance was finally determined at 570 nm using the microplate reader (Thermo Fisher, Manassas, VA, USA). The results from vehicle-treated cells were considered as 100% viability.
Transcriptomic RNA-sequencing (RNA-seq)
ARP1 NC and ARP1 siVCP cells were used for RNA-seq analyses. The procedure of RNA-seq was performed as previously described [61]. All data analysis and processing were performed by Lc-Bio Technologies (Hangzhou) Co., Ltd. (Hangzhou, China). We have uploaded the data of RNA-seq in GEO database (GSE210920, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE210920).
In vivo ubiquitylation assay
MM cells were incubated with 20 μM MG132 for 12 h before collection, and lysed in IP lysis buffer, followed by Co-IP and Western blotting (WB) analyses. Ubiquitylation assay was performed according to the protocol of the Pierce™ Direct Magnetic IP/Co-IP kit (88828, Thermo Fisher, Waltham, MA, USA). Briefly, the cell lysate was subjected to immunoprecipitation with IκBα antibody, and immunoprecipitation was subsequently separated by SDS-PAGE and immunoblotted with a Ubiquitin antibody to detect the ubiquitination level of IκBα. WB was performed as described previously [61].
Molecular docking based alignment
The crystal structure of VCP was retrieved from Protein Data Bank (https://www.rcsb.org/), and was prepared by the Molecular Operating Environment (MOE) software (Chemical Computing Group, Inc.: Montreal, Canada). The binding mode between VCP20 and VCP was manually built and refined by MOE.
Flow cytometry analysis of cell apoptosis
Cell apoptosis was analyzed by flow cytometry as reported in the literatures [62]. Flow cytometry equipped with Guava easyCyte System (Merck Millipore, Darmstadt, Germany) was applied to detect cell apoptosis.
Isolation and application of exosomes
Exosomes were purified from MM cell-derived conditioned medium. The conditioned medium was collected after 48 h and centrifuged at 300×g for 10 min at 4° C, followed by 1,000×g for 10 min and 10,000×g for 30 min at 4° C. The supernatant was ultracentrifuged at 100,000×g for 70 min at 4° C. The exosomes were washed with PBS followed by a second ultracentrifugation at 100,000×g for 70 min at 4° C and then resuspended in PBS.
Tartrate-resistant acid phosphatase (TRAP) activity staining
RAW264.7 cells (1.5×103) were seeded in 24-well plate with recombinant murine sRANKL (315-11, Peprotech, Cranbury, NJ, USA) and M-CSF (315-02, Peprotech) on Day 2. The cells were treated with VCP20 (100 nM) or exosomes (2 μg/mL each well). After 6 days, the cells were stained for TRAP activity.
Real-time PCR
TRIeasy Total RNA Extraction Reagent (19201ES60, Yeasen, Shanghai, China) was used to extract total RNA. Complementary DNA was synthesized by using reverse transcription kit (11123ES10, Yeasen). Real-time quantitative PCR was performed with SYBR Green master Mix (11198ES03, Yeasen). The Sequences of primers were as follows, GAPDH: Forward Sequence CATCACTGCCACCCAGAAGACTG and Reverse Sequence ATGCCAGTGAGCTTCCCGTTCAG; ACP5: Forward Sequence GCGACCATTGTTAGCCACATACG and Reverse Sequence CGTTGATGTCGCACAGAGGGAT; CTSK: Forward Sequence AGCAGAACGGAGGCATTGACTC and Reverse Sequence CCCTCTGCATTTAGCTGCCTTTG; MMP9: Forward Sequence GCTGACTACGATAAGGACGGCA and Reverse Sequence TAGTGGTGCAGGCAGAGTAGGA; TM7SF4: Forward Sequence TTTGCCGCTGTGGACTATCTGC and Reverse Sequence GCAGAATCATGGACGACTCCTTG.
MM xenografts
ARP1 WT cells (2×106) were injected subcutaneously into abdominal of 6~8 weeks old NOD/SCID mice and divided into two groups randomly (Control and VCP20, n = 4 per group). On Day 3 after cell injection, intraperitoneal administration of VCP20 (20 mg/kg) was performed twice weekly in VCP20 group. In all cases, tumor diameter was measured with calipers 2~3 times weekly. When the tumor diameter reached 15 mm, the mice were sacrificed. All animal work was performed in accordance with government-published recommendations for the Care and Use of laboratory animals and guidelines of Institutional Ethics Review Boards of Nanjing University of Chinese Medicine (Ethics Registration No. 201905A003).
5TMM3VT MM mouse model
5TMM3VT cells (1×106) were injected through the tail vein into C57BL/KaLwRij mice from Harlan Laboratories (Indianapolis, IN, USA). The mice were divided into two groups (Control and VCP20). On Day 3 after cell injection, intraperitoneal administration of VCP20 (20 mg/kg) was performed twice weekly in VCP20 group. The mice of tibia model were sacrificed by spinal dislocation. The bone mineral density (BMD) and bone volume faction (bone volume/total volume [BV/TV]) of the tibia were analyzed by micro-computed tomography (micro-CT) (SkyScan 1176, Bruker microCT, Germany) to assess osteolysis. The death time of the mice was recorded to generate the survival curve (n = 8 per group).
Statistical analysis
All values were expressed as means ± SD. Two-tailed Student’s t-test and One-way analysis of variance (ANOVA) (≥ three groups) were used to determine significance between experimental groups. The Kaplan-Meier method was used to evaluate the correlation of VCP expression with MM patient survival. In all cases, significance was defined as p < 0.05.
Supplementary Materials
Author Contributions
YY and CG designed and conceived this project. RW, YC, WH, XL, MJ, XL, ZD and ZW performed the experiments. RW, YC and MK analyzed the data. RW and YC drafted the manuscript. YZ provided VCP inhibitors and analyzed related data. SC offered the clinical data and analyzed related data. YY, CG, YZ and SC edited the manuscript. All authors read and approved the final version of the manuscript.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical Statement
All animal work was performed in accordance with government-published recommendations for the Care and Use of laboratory animals and guidelines of Institutional Ethics Review Boards of Nanjing University of Chinese Medicine (Ethics Registration No. 201905A003).
Funding
This work was supported by National Natural Science Foundation of China (82073885 to YY); Natural Science Foundation of Jiangsu Province (BK20200097 to CG); A Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (Traditional Chinese Medicine).
References
- 1. Zhang S, Mao Y. AAA+ ATPases in Protein Degradation: Structures, Functions and Mechanisms. Biomolecules. 2020; 10:629. https://doi.org/10.3390/biom10040629 [PubMed]
- 2. Zhang G, Li S, Cheng KW, Chou TF. AAA ATPases as therapeutic targets: Structure, functions, and small-molecule inhibitors. Eur J Med Chem. 2021; 219:113446. https://doi.org/10.1016/j.ejmech.2021.113446 [PubMed]
- 3. Assimon VA, Tang Y, Vargas JD, Lee GJ, Wu ZY, Lou K, Yao B, Menon MK, Pios A, Perez KC, Madriaga A, Buchowiecki PK, Rolfe M, et al. CB-6644 Is a Selective Inhibitor of the RUVBL1/2 Complex with Anticancer Activity. ACS Chem Biol. 2019; 14:236–44. https://doi.org/10.1021/acschembio.8b00904 [PubMed]
- 4. Wang Y, Huang J, Li B, Xue H, Tricot G, Hu L, Xu Z, Sun X, Chang S, Gao L, Tao Y, Xu H, Xie Y, et al. A Small-Molecule Inhibitor Targeting TRIP13 Suppresses Multiple Myeloma Progression. Cancer Res. 2020; 80:536–48. https://doi.org/10.1158/0008-5472.CAN-18-3987 [PubMed]
- 5. Vaz B, Halder S, Ramadan K. Role of p97/VCP (Cdc48) in genome stability. Front Genet. 2013; 4:60. https://doi.org/10.3389/fgene.2013.00060 [PubMed]
- 6. Ramanathan HN, Zhang S, Douam F, Mar KB, Chang J, Yang PL, Schoggins JW, Ploss A, Lindenbach BD. A Sensitive Yellow Fever Virus Entry Reporter Identifies Valosin-Containing Protein (VCP/p97) as an Essential Host Factor for Flavivirus Uncoating. mBio. 2020; 11:e00467–20. https://doi.org/10.1128/mBio.00467-20 [PubMed]
- 7. Meyer H, Weihl CC. The VCP/p97 system at a glance: connecting cellular function to disease pathogenesis. J Cell Sci. 2014; 127:3877–83. https://doi.org/10.1242/jcs.093831 [PubMed]
- 8. Arita M, Wakita T, Shimizu H. Valosin-containing protein (VCP/p97) is required for poliovirus replication and is involved in cellular protein secretion pathway in poliovirus infection. J Virol. 2012; 86:5541–53. https://doi.org/10.1128/JVI.00114-12 [PubMed]
- 9. Wang T, Wang B, Huang H, Zhang C, Zhu Y, Pei B, Cheng C, Sun L, Wang J, Jin Q, Zhao Z. Enterovirus 71 protease 2Apro and 3Cpro differentially inhibit the cellular endoplasmic reticulum-associated degradation (ERAD) pathway via distinct mechanisms, and enterovirus 71 hijacks ERAD component p97 to promote its replication. PLoS Pathog. 2017; 13:e1006674. https://doi.org/10.1371/journal.ppat.1006674 [PubMed]
- 10. Yi Z, Fang C, Zou J, Xu J, Song W, Du X, Pan T, Lu H, Yuan Z. Affinity Purification of the Hepatitis C Virus Replicase Identifies Valosin-Containing Protein, a Member of the ATPases Associated with Diverse Cellular Activities Family, as an Active Virus Replication Modulator. J Virol. 2016; 90:9953–66. https://doi.org/10.1128/JVI.01140-16 [PubMed]
- 11. Wu KX, Phuektes P, Kumar P, Goh GY, Moreau D, Chow VT, Bard F, Chu JJ. Human genome-wide RNAi screen reveals host factors required for enterovirus 71 replication. Nat Commun. 2016; 7:13150. https://doi.org/10.1038/ncomms13150 [PubMed]
- 12. Zhu W, Li D, Xiao L. Upregulation of valosin-containing protein (VCP) is associated with poor prognosis and promotes tumor progression of orbital B-cell lymphoma. Onco Targets Ther. 2018; 12:243–53. https://doi.org/10.2147/OTT.S182118 [PubMed]
- 13. Fu Q, Jiang Y, Zhang D, Liu X, Guo J, Zhao J. Valosin-containing protein (VCP) promotes the growth, invasion, and metastasis of colorectal cancer through activation of STAT3 signaling. Mol Cell Biochem. 2016; 418:189–98. https://doi.org/10.1007/s11010-016-2746-6 [PubMed]
- 14. Bastola P, Neums L, Schoenen FJ, Chien J. VCP inhibitors induce endoplasmic reticulum stress, cause cell cycle arrest, trigger caspase-mediated cell death and synergistically kill ovarian cancer cells in combination with Salubrinal. Mol Oncol. 2016; 10:1559–74. https://doi.org/10.1016/j.molonc.2016.09.005 [PubMed]
- 15. Duscharla D, Reddy Kami Reddy K, Dasari C, Bhukya S, Ummanni R. Interleukin-6 induced overexpression of valosin-containing protein (VCP)/p97 is associated with androgen-independent prostate cancer (AIPC) progression. J Cell Physiol. 2018; 233:7148–64. https://doi.org/10.1002/jcp.26639 [PubMed]
- 16. Zhu C, Rogers A, Asleh K, Won J, Gao D, Leung S, Li S, Vij KR, Zhu J, Held JM, You Z, Nielsen TO, Shao J. Phospho-Ser784-VCP Is Required for DNA Damage Response and Is Associated with Poor Prognosis of Chemotherapy-Treated Breast Cancer. Cell Rep. 2020; 31:107745. https://doi.org/10.1016/j.celrep.2020.107745 [PubMed]
- 17. Khong ZJ, Lai SK, Koh CG, Geifman-Shochat S, Li HY. A novel function of AAA-ATPase p97/VCP in the regulation of cell motility. Oncotarget. 2020; 11:74–85. https://doi.org/10.18632/oncotarget.27419 [PubMed]
- 18. Costantini S, Capone F, Polo A, Bagnara P, Budillon A. Valosin-Containing Protein (VCP)/p97: A Prognostic Biomarker and Therapeutic Target in Cancer. Int J Mol Sci. 2021; 22:10177. https://doi.org/10.3390/ijms221810177 [PubMed]
- 19. Heidelberger JB, Voigt A, Borisova ME, Petrosino G, Ruf S, Wagner SA, Beli P. Proteomic profiling of VCP substrates links VCP to K6-linked ubiquitylation and c-Myc function. EMBO Rep. 2018; 19:e44754. https://doi.org/10.15252/embr.201744754 [PubMed]
- 20. Parzych K, Saavedra-García P, Valbuena GN, Al-Sadah HA, Robinson ME, Penfold L, Kuzeva DM, Ruiz-Tellez A, Loaiza S, Holzmann V, Caputo V, Johnson DC, Kaiser MF, et al. The coordinated action of VCP/p97 and GCN2 regulates cancer cell metabolism and proteostasis during nutrient limitation. Oncogene. 2019; 38:3216–31. https://doi.org/10.1038/s41388-018-0651-z [PubMed]
- 21. Nishimura N, Radwan MO, Amano M, Endo S, Fujii E, Hayashi H, Ueno S, Ueno N, Tatetsu H, Hata H, Okamoto Y, Otsuka M, Mitsuya H, et al. Novel p97/VCP inhibitor induces endoplasmic reticulum stress and apoptosis in both bortezomib-sensitive and -resistant multiple myeloma cells. Cancer Sci. 2019; 110:3275–87. https://doi.org/10.1111/cas.14154 [PubMed]
- 22. Auner HW, Moody AM, Ward TH, Kraus M, Milan E, May P, Chaidos A, Driessen C, Cenci S, Dazzi F, Rahemtulla A, Apperley JF, Karadimitris A, Dillon N. Combined inhibition of p97 and the proteasome causes lethal disruption of the secretory apparatus in multiple myeloma cells. PLoS One. 2013; 8:e74415. https://doi.org/10.1371/journal.pone.0074415 [PubMed]
- 23. Le Moigne R, Aftab BT, Djakovic S, Dhimolea E, Valle E, Murnane M, King EM, Soriano F, Menon MK, Wu ZY, Wong ST, Lee GJ, Yao B, et al. The p97 Inhibitor CB-5083 Is a Unique Disrupter of Protein Homeostasis in Models of Multiple Myeloma. Mol Cancer Ther. 2017; 16:2375–86. https://doi.org/10.1158/1535-7163.MCT-17-0233 [PubMed]
- 24. Shi X, Zhu K, Ye Z, Yue J. VCP/p97 targets the nuclear export and degradation of p27Kip1 during G1 to S phase transition. FASEB J. 2020; 34:5193–207. https://doi.org/10.1096/fj.201901506R [PubMed]
- 25. Magnaghi P, D’Alessio R, Valsasina B, Avanzi N, Rizzi S, Asa D, Gasparri F, Cozzi L, Cucchi U, Orrenius C, Polucci P, Ballinari D, Perrera C, et al. Covalent and allosteric inhibitors of the ATPase VCP/p97 induce cancer cell death. Nat Chem Biol. 2013; 9:548–56. https://doi.org/10.1038/nchembio.1313 [PubMed]
- 26. Anderson DJ, Le Moigne R, Djakovic S, Kumar B, Rice J, Wong S, Wang J, Yao B, Valle E, Kiss von Soly S, Madriaga A, Soriano F, Menon MK, et al. Targeting the AAA ATPase p97 as an Approach to Treat Cancer through Disruption of Protein Homeostasis. Cancer Cell. 2015; 28:653–65. https://doi.org/10.1016/j.ccell.2015.10.002 [PubMed]
- 27. Kilgas S, Singh AN, Paillas S, Then CK, Torrecilla I, Nicholson J, Browning L, Vendrell I, Konietzny R, Kessler BM, Kiltie AE, Ramadan K. p97/VCP inhibition causes excessive MRE11-dependent DNA end resection promoting cell killing after ionizing radiation. Cell Rep. 2021; 35:109153. https://doi.org/10.1016/j.celrep.2021.109153 [PubMed]
- 28. Wang X, Bai E, Zhou H, Sha S, Miao H, Qin Y, Liu Z, Wang J, Zhang H, Lei M, Liu J, Hai O, Zhu Y. Discovery of a new class of valosine containing protein (VCP/P97) inhibitors for the treatment of non-small cell lung cancer. Bioorg Med Chem. 2019; 27:533–44. https://doi.org/10.1016/j.bmc.2018.12.036 [PubMed]
- 29. Goel S, Bergholz JS, Zhao JJ. Targeting CDK4 and CDK6 in cancer. Nat Rev Cancer. 2022; 22:356–72. https://doi.org/10.1038/s41568-022-00456-3 [PubMed]
- 30. Ely S, Di Liberto M, Niesvizky R, Baughn LB, Cho HJ, Hatada EN, Knowles DM, Lane J, Chen-Kiang S. Mutually exclusive cyclin-dependent kinase 4/cyclin D1 and cyclin-dependent kinase 6/cyclin D2 pairing inactivates retinoblastoma protein and promotes cell cycle dysregulation in multiple myeloma. Cancer Res. 2005; 65:11345–53. https://doi.org/10.1158/0008-5472.CAN-05-2159 [PubMed]
- 31. Fabre C, Mimura N, Bobb K, Kong SY, Gorgun G, Cirstea D, Hu Y, Minami J, Ohguchi H, Zhang J, Meshulam J, Carrasco RD, Tai YT, et al. Correction: Dual Inhibition of Canonical and Noncanonical NF-κB Pathways Demonstrates Significant Antitumor Activities in Multiple Myeloma. Clin Cancer Res. 2019; 25:2938. https://doi.org/10.1158/1078-0432.CCR-19-0959 [PubMed]
- 32. Gilmore TD. Multiple myeloma: lusting for NF-kappaB. Cancer Cell. 2007; 12:95–7. https://doi.org/10.1016/j.ccr.2007.07.010 [PubMed]
- 33. Ji J, Ding K, Luo T, Zhang X, Chen A, Zhang D, Li G, Thorsen F, Huang B, Li X, Wang J. TRIM22 activates NF-κB signaling in glioblastoma by accelerating the degradation of IκBα. Cell Death Differ. 2021; 28:367–81. https://doi.org/10.1038/s41418-020-00606-w [PubMed]
- 34. Wang Y, Zhang J, Li YJ, Yu NN, Liu WT, Liang JZ, Xu WW, Sun ZH, Li B, He QY. MEST promotes lung cancer invasion and metastasis by interacting with VCP to activate NF-κB signaling. J Exp Clin Cancer Res. 2021; 40:301. https://doi.org/10.1186/s13046-021-02107-1 [PubMed]
- 35. Westhrin M, Kovcic V, Zhang Z, Moen SH, Nedal TMV, Bondt A, Holst S, Misund K, Buene G, Sundan A, Waage A, Slørdahl TS, Wuhrer M, Standal T. Monoclonal immunoglobulins promote bone loss in multiple myeloma. Blood. 2020; 136:2656–66. https://doi.org/10.1182/blood.2020006045 [PubMed]
- 36. de la Rica L, García-Gómez A, Comet NR, Rodríguez-Ubreva J, Ciudad L, Vento-Tormo R, Company C, Álvarez-Errico D, García M, Gómez-Vaquero C, Ballestar E. NF-κB-direct activation of microRNAs with repressive effects on monocyte-specific genes is critical for osteoclast differentiation. Genome Biol. 2015; 16:2. https://doi.org/10.1186/s13059-014-0561-5 [PubMed]
- 37. Zhang Y, Yu X, Sun R, Min J, Tang X, Lin Z, Xie S, Li X, Lu S, Tian Z, Gu C, Teng L, Yang Y. Splicing factor arginine/serine-rich 8 promotes multiple myeloma malignancy and bone lesion through alternative splicing of CACYBP and exosome-based cellular communication. Clin Transl Med. 2022; 12:e684. https://doi.org/10.1002/ctm2.684 [PubMed]
- 38. Zhang L, Lei Q, Wang H, Xu C, Liu T, Kong F, Yang C, Yan G, Sun L, Zhao A, Chen W, Hu Y, Xie H, et al. Tumor-derived extracellular vesicles inhibit osteogenesis and exacerbate myeloma bone disease. Theranostics. 2019; 9:196–209. https://doi.org/10.7150/thno.27550 [PubMed]
- 39. Cordeiro L, Riou C, Uzbekov R, Gérard N. Avian sperm increase in vitro the release of exosomes from SST-enriched organoids. Reproduction. 2021; 161:375–84. https://doi.org/10.1530/REP-20-0421 [PubMed]
- 40. Lorenzo J. The many ways of osteoclast activation. J Clin Invest. 2017; 127:2530–2. https://doi.org/10.1172/JCI94606 [PubMed]
- 41. Hideshima T, Mitsiades C, Tonon G, Richardson PG, Anderson KC. Understanding multiple myeloma pathogenesis in the bone marrow to identify new therapeutic targets. Nat Rev Cancer. 2007; 7:585–98. https://doi.org/10.1038/nrc2189 [PubMed]
- 42. Zhang G, Li S, Wang F, Jones AC, Goldberg AFG, Lin B, Virgil S, Stoltz BM, Deshaies RJ, Chou TF. A covalent p97/VCP ATPase inhibitor can overcome resistance to CB-5083 and NMS-873 in colorectal cancer cells. Eur J Med Chem. 2021; 213:113148. https://doi.org/10.1016/j.ejmech.2020.113148 [PubMed]
- 43. Chapman E, Maksim N, de la Cruz F, La Clair JJ. Inhibitors of the AAA+ chaperone p97. Molecules. 2015; 20:3027–49. https://doi.org/10.3390/molecules20023027 [PubMed]
- 44. Li C, Huang Y, Fan Q, Quan H, Dong Y, Nie M, Wang J, Xie F, Ji J, Zhou L, Zheng Z, Wang L. p97/VCP is highly expressed in the stem-like cells of breast cancer and controls cancer stemness partly through the unfolded protein response. Cell Death Dis. 2021; 12:286. https://doi.org/10.1038/s41419-021-03555-5 [PubMed]
- 45. Wang J, Chen Y, Huang C, Hao Q, Zeng SX, Omari S, Zhang Y, Zhou X, Lu H. Valosin-Containing Protein Stabilizes Mutant p53 to Promote Pancreatic Cancer Growth. Cancer Res. 2021; 81:4041–53. https://doi.org/10.1158/0008-5472.CAN-20-3855 [PubMed]
- 46. Schweitzer K, Pralow A, Naumann M. p97/VCP promotes Cullin-RING-ubiquitin-ligase/proteasome-dependent degradation of IκBα and the preceding liberation of RelA from ubiquitinated IκBα. J Cell Mol Med. 2016; 20:58–70. https://doi.org/10.1111/jcmm.12702 [PubMed]
- 47. Roux B, Vaganay C, Vargas JD, Alexe G, Benaksas C, Pardieu B, Fenouille N, Ellegast JM, Malolepsza E, Ling F, Sodaro G, Ross L, Pikman Y, et al. Targeting acute myeloid leukemia dependency on VCP-mediated DNA repair through a selective second-generation small-molecule inhibitor. Sci Transl Med. 2021; 13:eabg1168. https://doi.org/10.1126/scitranslmed.abg1168 [PubMed]
- 48. Pan M, Yu Y, Ai H, Zheng Q, Xie Y, Liu L, Zhao M. Mechanistic insight into substrate processing and allosteric inhibition of human p97. Nat Struct Mol Biol. 2021; 28:614–25. https://doi.org/10.1038/s41594-021-00617-2 [PubMed]
- 49. Chou TF, Brown SJ, Minond D, Nordin BE, Li K, Jones AC, Chase P, Porubsky PR, Stoltz BM, Schoenen FJ, Patricelli MP, Hodder P, Rosen H, Deshaies RJ. Reversible inhibitor of p97, DBeQ, impairs both ubiquitin-dependent and autophagic protein clearance pathways. Proc Natl Acad Sci USA. 2011; 108:4834–9. https://doi.org/10.1073/pnas.1015312108 [PubMed]
- 50. Feng Q, Zheng J, Zhang J, Zhao M. Synthesis and In Vitro Evaluation of 2-[3-(2-Aminoethyl)-1H-indol-1-yl]-N-benzylquinazolin-4-amine as a Novel p97/VCP Inhibitor Lead Capable of Inducing Apoptosis in Cancer Cells. ACS Omega. 2020; 5:31784–91. https://doi.org/10.1021/acsomega.0c04478 [PubMed]
- 51. Banerjee S, Bartesaghi A, Merk A, Rao P, Bulfer SL, Yan Y, Green N, Mroczkowski B, Neitz RJ, Wipf P, Falconieri V, Deshaies RJ, Milne JL, et al. 2.3 Å resolution cryo-EM structure of human p97 and mechanism of allosteric inhibition. Science. 2016; 351:871–5. https://doi.org/10.1126/science.aad7974 [PubMed]
- 52. Wang F, Li S, Gan T, Stott GM, Flint A, Chou TF. Allosteric p97 Inhibitors Can Overcome Resistance to ATP-Competitive p97 Inhibitors for Potential Anticancer Therapy. ChemMedChem. 2020; 15:685–94. https://doi.org/10.1002/cmdc.201900722 [PubMed]
- 53. Mitsiades CS, Mitsiades NS, Munshi NC, Richardson PG, Anderson KC. The role of the bone microenvironment in the pathophysiology and therapeutic management of multiple myeloma: interplay of growth factors, their receptors and stromal interactions. Eur J Cancer. 2006; 42:1564–73. https://doi.org/10.1016/j.ejca.2005.12.025 [PubMed]
- 54. Lu B, Jiao Y, Wang Y, Dong J, Wei M, Cui B, Sun Y, Wang L, Zhang B, Chen Z, Zhao Y. A FKBP5 mutation is associated with Paget’s disease of bone and enhances osteoclastogenesis. Exp Mol Med. 2017; 49:e336. https://doi.org/10.1038/emm.2017.64 [PubMed]
- 55. Chung PY, Beyens G, de Freitas F, Boonen S, Geusens P, Vanhoenacker F, Verbruggen L, Van Offel J, Goemaere S, Zmierczak HG, Westhovens R, Devogelaer JP, Van Hul W. Indications for a genetic association of a VCP polymorphism with the pathogenesis of sporadic Paget’s disease of bone, but not for TNFSF11 (RANKL) and IL-6 polymorphisms. Mol Genet Metab. 2011; 103:287–92. https://doi.org/10.1016/j.ymgme.2011.03.021 [PubMed]
- 56. Shtam TA, Kovalev RA, Varfolomeeva EY, Makarov EM, Kil YV, Filatov MV. Exosomes are natural carriers of exogenous siRNA to human cells in vitro. Cell Commun Signal. 2013; 11:88. https://doi.org/10.1186/1478-811X-11-88 [PubMed]
- 57. Sun Z, Yang S, Zhou Q, Wang G, Song J, Li Z, Zhang Z, Xu J, Xia K, Chang Y, Liu J, Yuan W. Emerging role of exosome-derived long non-coding RNAs in tumor microenvironment. Mol Cancer. 2018; 17:82. https://doi.org/10.1186/s12943-018-0831-z [PubMed]
- 58. Boyiadzis M, Whiteside TL. The emerging roles of tumor-derived exosomes in hematological malignancies. Leukemia. 2017; 31:1259–68. https://doi.org/10.1038/leu.2017.91 [PubMed]
- 59. Bosque A, Dietz L, Gallego-Lleyda A, Sanclemente M, Iturralde M, Naval J, Alava MA, Martínez-Lostao L, Thierse HJ, Anel A. Comparative proteomics of exosomes secreted by tumoral Jurkat T cells and normal human T cell blasts unravels a potential tumorigenic role for valosin-containing protein. Oncotarget. 2016; 7:29287–305. https://doi.org/10.18632/oncotarget.8678 [PubMed]
- 60. Wei R, Cui X, Min J, Lin Z, Zhou Y, Guo M, An X, Liu H, Janz S, Gu C, Wang H, Yang Y. NAT10 promotes cell proliferation by acetylating CEP170 mRNA to enhance translation efficiency in multiple myeloma. Acta Pharm Sin B. 2022; 12:3313–25. https://doi.org/10.1016/j.apsb.2022.01.015 [PubMed]
- 61. Wei R, Zhu Y, Zhang Y, Zhao W, Yu X, Wang L, Gu C, Gu X, Yang Y. AIMP1 promotes multiple myeloma malignancy through interacting with ANP32A to mediate histone H3 acetylation. Cancer Commun (Lond). 2022; 42:1185–206. https://doi.org/10.1002/cac2.12356 [PubMed]
- 62. Gu C, Wang Y, Zhang L, Qiao L, Sun S, Shao M, Tang X, Ding P, Tang C, Cao Y, Zhou Y, Guo M, Wei R, et al. AHSA1 is a promising therapeutic target for cellular proliferation and proteasome inhibitor resistance in multiple myeloma. J Exp Clin Cancer Res. 2022; 41:11. https://doi.org/10.1186/s13046-021-02220-1 [PubMed]



