Introduction
The rapidly increasing incidence of various age-related chronic and degenerative diseases, such as sarcopenia [1], in the global elderly population, is a major threat to human healthcare. Thus, a mechanistic understanding of aging and developing intervention measures to combat age-related health complications are of utmost importance in these aged society. The most effective and reproducible aging process intervention to date is dietary restriction (DR), which has shown promising results in increasing longevity and providing a healthy lifestyle in all model organisms tested to date [2]. Moreover, DR is known to be consistent in positively affecting various health-related quality of life attributes in humans [3]. Thus, research into the molecular mechanisms of action of DR is important and relevant.
It has been reported that DR may activate the immune response in animal models, including Caenorhabditis elegans [4] and Drosophila [5]. Furthermore, DR promotes production or maintenance of naïve T cells in mice and in primates [6, 7]. In humans, mild DR (14% DR) also improved thymopoiesis in thymus, which is important for immune function [8]. However, depending on the circumstance, DR may also increase severity of viral and parasitic infections [9, 10]. Thus, the immune system is tightly connected with DR. Immunity is also associated with aging, and impaired immunity is a key hallmark of aging in many animal species [11]. A recent study showed that, in C. elegans, innate immune signaling component is necessary for DR-induced longevity and health improvement [4]. Furthermore, DR enhances the lifespan of C. elegans by maintaining the level of p38-ATF-7 (a transcription factor downstream of p38) innate immune response at the level of basal activation [12], suggesting that the immune system plays an important role in the extension of lifespan by DR. However, the detailed mechanisms and effective downstream factors of immunity that mediate the beneficial effects of DR on health span remain unknown.
Caenorhabditis elegans is an invaluable animal model for studying aging [13] or age-associated diseases such as immunosenescence [14]. Several studies with C. elegans have unraveled various intricate molecular pathways regulating longevity that have been proven to be conserved in higher organisms [13]. A feeding-defective mutant strain, eat-2, which mimics DR [15], has been widely used in research to understand the underlying molecular mechanisms of DR-induced lifespan extension. Further, the innate immune system of C. elegans is evolutionarily conserved in the animal kingdom Thus, C. elegans is used in basic research on the mechanisms of resistance to pathogens [16].
F-box proteins are well-conserved proteins from yeast to humans. They have F-box protein motif which functions as a site for protein-protein interaction. The function of the F-box protein motif has been extensively studied in relation to ubiquitin-mediated proteolysis [17]. The function of F-box proteins in relation to the regulation of aging process or behavior response to pathogen has been reported in C. elegans [18, 19]. In this study, we aimed to identify the novel innate immune signaling component that mediates the DR effect, and we found that the F-box protein, FBXC-58, a regulatory component of the innate immune response, is an important downstream effector of DR for extending longevity and improving physical health conditions in C. elegans.
Results
Screen to uncover the innate immune signal mediating the DR effect
To uncover the innate immune signal that mediates the DR effect, we hypothesized that candidate genes would be upregulated in response to both DR and pathogen infection. Based on this assumption, we analyzed previously reported DEG data for DR [12] and the pathogen [Pseudomonas aeruginosa (PA14)]-infected condition [20]. As shown in Figure 1A, we found that 1,085 and 890 genes were upregulated by DR and PA14 infection, respectively. Among these genes, we investigated those (107 genes) that were simultaneously upregulated under both conditions (Figure 1A, Supplementary Table 1) and found that F-box genes (7.5%) fit this criterion (Supplementary Table 1). Among the F-box genes, we found that fbxc-58 was most highly expressed (~4-fold) under DR conditions (induced by eat-2 mutation) (Figure 1B). Furthermore, fbxc-58 expression also increased 2.2-fold under low density of food (LDF) feeding conditions [4] compared with that under ad libitum (AL) conditions (Figure 1C), implying that fbxc-58 was upregulated in response to DR.
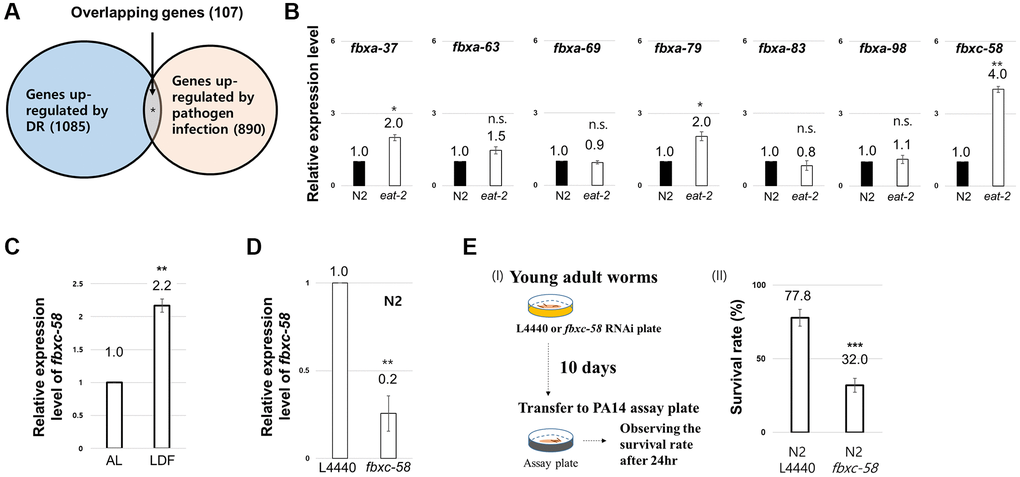
Next, we tested whether fbxc-58 exhibits resistance to pathogen. Note that fbxc-58 RNAi significantly downregulated fbxc-58 expression in C. elegans (Figure 1D). By treating PA14 to fbxc-58 RNAi or L4440 RNAi worms, as shown in Figure 1E, we found that the death rate of fbxc-58 RNAi worms was significantly higher than that of L4440 RNAi worms (P < 0.001). This data implying that fbxc-58 is a pathogen defense-related gene. Thus, fbxc-58 satisfies the condition to be regarded as an immune signaling component whose expression increases in response to DR. Therefore, we further investigated the role of fbxc-58 as a mediator of DR effects.
fbxc-58 mediates DR-induced lifespan extension in C. elegans
We confirmed that the eat-2 mutation, which mimic DR, considerably (30.2%) extended the median lifespan (ML) of C. elegans [ML of L4440 RNAi N2: 17.87 ± 0.44, ML of L4440 RNAi eat-2: 23.27 ± 0.62; P < 0.0001] (Figure 2 and Table 1). To determine the effect of fbxc-58 on longevity in DR, we treated worms with fbxc-58 RNAi. As shown in Figure 2, we found that the median lifespan of eat-2(ad1116) mutants drastically decreased when fbxc-58 was silenced [ML of L4440 RNAi eat-2: 23.27 ± 0.62, ML of fbxc-58 RNAi eat-2: 18.10 ± 0.39; (P < 0.0001)]. In addition, the median lifespan of fbxc-58 RNAi eat-2(ad1116) mutants was similar to that of the wild-type strain [ML of fbxc-58 RNAi N2: 17.99 ± 0.37, ML of fbxc-58 RNAi eat-2: 18.10 ± 0.39; P = 0.86] (Figure 2 and Table 1). Notably, silencing fbxc-58 did not alter the median lifespan of the wild-type strain [ML of L4440 RNAi N2: 17.87 ± 0.44, ML of fbxc-58 RNAi N2: 17.99 ± 0.37; P = 0.89] (Figure 2 and Table 1). Thus, these data imply that fbxc-58 affects DR, playing a pivotal role in extending longevity.
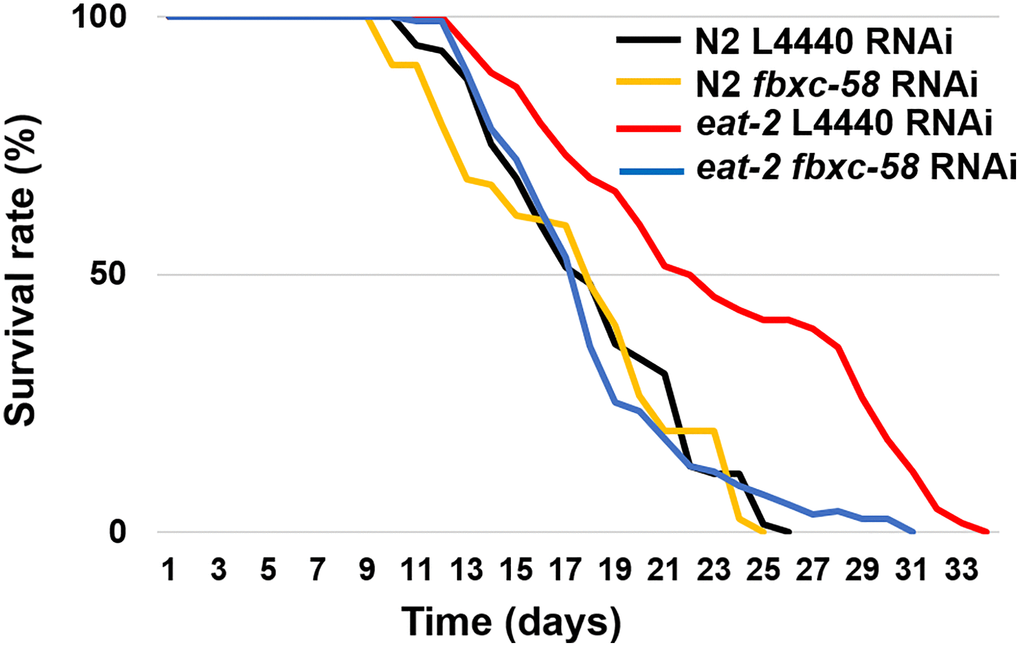
Table 1. Median lifespans of C. elegans cohorts.
| Figure | Strain | RNAi treatment | Mean lifespan (ML) | No. of worms (No. of experiments) |
| Figure 2 | N2 | L4440 RNAi | 17.87 ± 0.44 | 105 (3) |
| N2 | fbxc-58 RNAi | 17.99 ± 0.37 | 131 (3) | |
| eat-2 | L4440 RNAi | 23.27 ± 0.62 | 112 (3) | |
| eat-2 | fbxc-58 RNAi | 18.10 ± 0.39 | 111 (3) | |
| Figure 5C | N2 | L4440 RNAi | 18.06 ± 0.44 | 90 (2) |
| N2 | fbxc-58 RNAi | 17.99 ± 0.39 | 90 (2) | |
| rsks-1 | L4440 RNAi | 21.84 ± 0.42 | 81 (2) | |
| rsks-1 | fbxc-58 RNAi | 17.32 ± 0.37 | 68 (2) |
fbxc-58 delays muscle aging of C. elegans in DR
Aged eat-2(ad1116) mutants showed a 4.9-fold (P < 0.0001) higher body bending rate than that of the aged N2 strain (Figure 3A, Supplementary Figure 1A), although N2 and eat-2(ad1116) mutants showed similar body bending rates in the young adult stage (Supplementary Figure 1B). This data implies that DR alleviates the decline in physical activity of C. elegans during aging. Notably, we found that the improved muscle activity of aged eat-2(ad1116) mutants was significantly diminished by fbxc-58 RNAi (P < 0.05) (Figure 3A, Supplementary Figure 1), implying that fbxc-58 is vital to improving muscle activity through DR in C. elegans. Note that there was no statistical difference in body bending rates by fbxc-58 RNAi in aged N2 strains (Supplementary Figure 2).
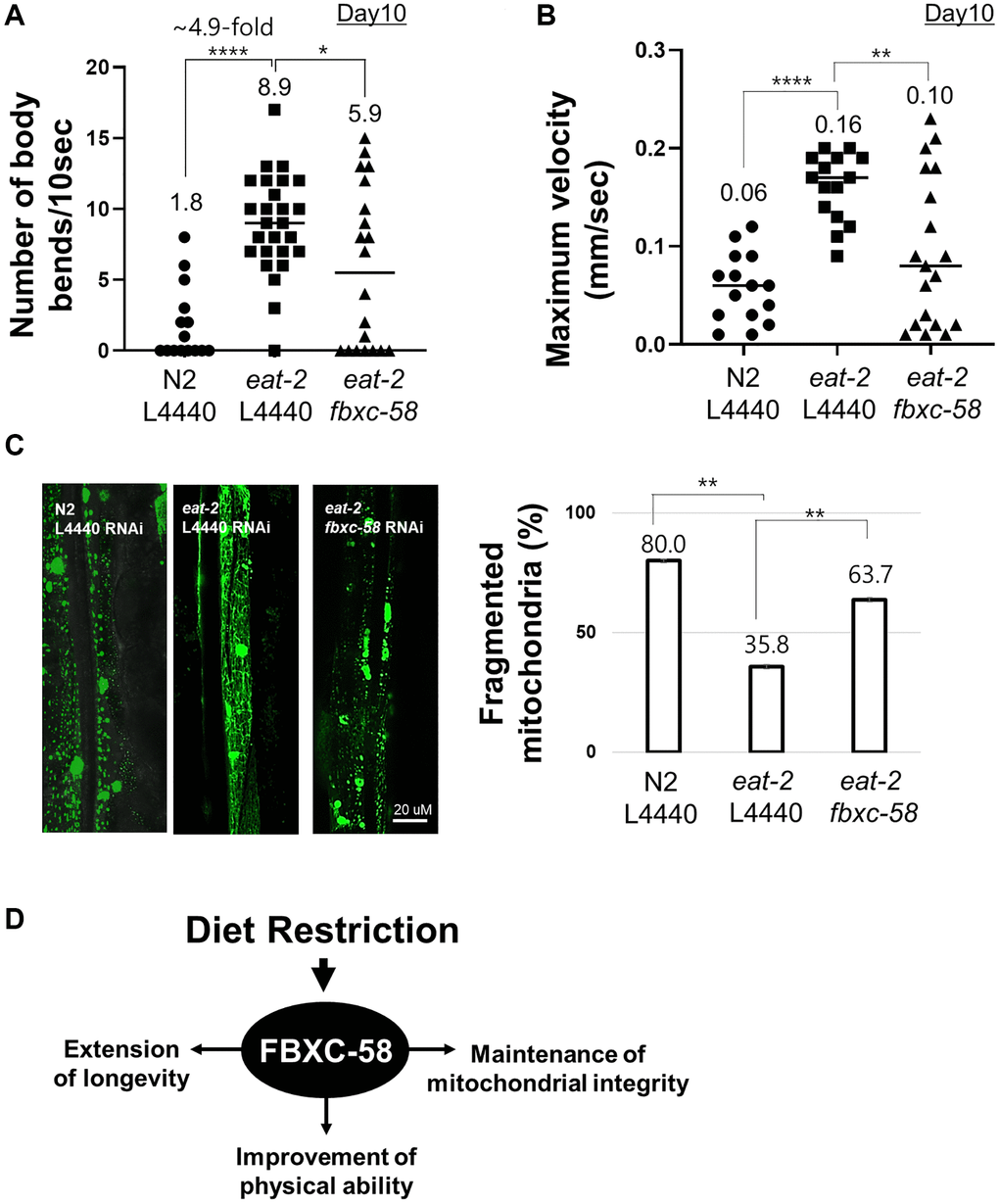
Further, in C. elegans, maximum body movement velocity (MV) is a potent indicator of their health condition and muscle activity [21]. As shown in Figure 3B and Supplementary Figure 3A, the aged eat-2(ad1116) mutant strain exhibited higher MV than that of the aged N2 strain, although MV in the young N2 strain was slightly higher than that of young eat-2(ad1116) mutants (Supplementary Figure 3B). However, the MV of aged eat-2(ad1116) mutants significantly decreased when fbxc-58 was abrogated (P < 0.01) (Figure 3B, Supplementary Figure 3A). This data signifies the essentiality of fbxc-58 in improving the physical health of aged C. elegans through DR. Note that there was no statistical difference in MVs by fbxc-58 RNAi in aged N2 strains (Supplementary Figure 4).
In aging, the disintegration of mitochondrial networks (e.g., mitochondrial fragmentation) in muscle cells is a well-established phenotype of muscle aging in C. elegans [22], and this mitochondrial damage is regarded as one of the leading factors contributing to muscle aging [23]. We found that the proportion of worms with fragmented mitochondria in body wall muscle was significantly lower in L4440 RNAi eat-2(ad1116) mutants (35.8%) than that in the L4440 RNAi N2 strain (80.0%). However, this proportion was increased when fbxc-58 was silenced in eat-2(ad1116) mutants (63.7%) (P < 0.01) (Figure 3C), implying that fbxc-58 plays a pivotal role in maintaining the mitochondrial networks in muscle cells, and it is consistent with the reduced muscle activity in fbxc-58 RNAi eat-2(ad1116) mutants relative to that in L4440 RNAi eat-2(ad1116) mutants (Figure 3A, 3B). Note that fbxc-58 RNAi did not affect the mitochondrial morphology of aged N2 strains (Supplementary Figure 5). Thus, fbxc-58 is necessary for DR-induced longevity extension, muscle mitochondrial protection, and physical activity improvement in aging (Figure 3D).
fbxc-58 is a downstream target of ZIP-2 and PHA-4
To understand the underlying molecular mechanism of fbxc-58 in DR in detail, we examined the correlation between fbxc-58 and zip-2 since ZIP-2 is a well-known immune response effector in C. elegans [24]. Additionally, ZIP-2 is a transcription factor that regulates aging process [25] and the extent of longevity under various DR conditions, including dietary deprivation, diluted peptone, or diluted OP50 without peptone [4]. Note that we confirmed that zip-2 or pha-4 significantly reduced under zip-2 RNAi or pha-4 RNAi conditions, respectively (Figure 4A, 4B). As shown in Figure 4C, we found that fbxc-58 expression was significantly diminished by silencing zip-2 in eat-2(ad1116) mutants compared with that in the L4440 RNAi control. This data implies that fbxc-58 is upregulated by zip-2 under DR conditions. Additionally, we confirmed that C10C5.2 (sequence name of fbxc-58) is among the genes that are downregulated by zip-2 RNAi in previously reported differentially expressed gene (DEG) data [24], indicating that fbxc-58 is a downstream target gene of ZIP-2.
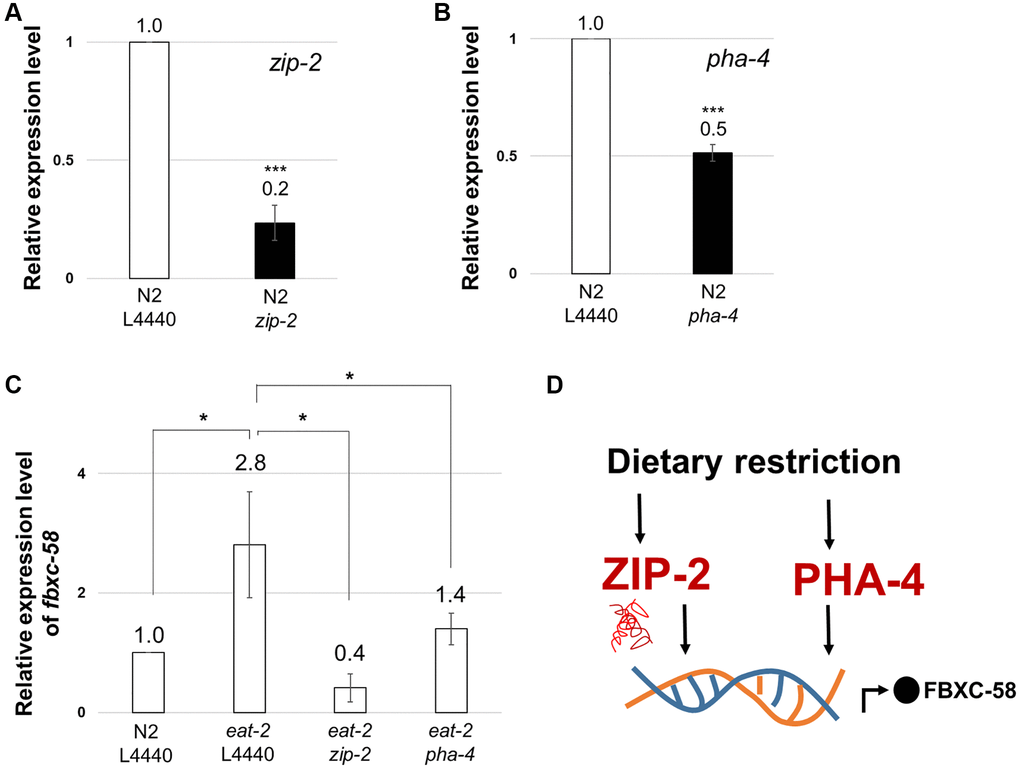
PHA-4 is the forkhead box transcription factor that mediates the lifespan extension induced by eat-2 mutation [26]. We found that fbxc-58 expression was decreased in pha-4 RNAi eat-2(ad1116) mutants compared with that in the L4440 RNAi eat-2(ad1116) mutant strains (Figure 4C). This data implies that fbxc-58 is a downstream target gene of PHA-4. Thus, all of these data corroborate the fact that fbxc-58 is indeed a downstream effector of DR (Figure 4D).
fbxc-58 is a downstream effector of S6 kinase pathway
Mutation in S6 kinase inhibits its translation [27], mediating lifespan extension through DR [28]. zip-2 is necessary for the lifespan extension of the rsks-1 (C. elegans homolog of S6 kinase) mutant strain [4]. Further, PHA-4 is a downstream effector of S6K pathway for longevity in C. elegans [29]. Thus, we tested whether fbxc-58 is necessary for lifespan extension by rsks-1 mutation. We found that fbxc-58 expression was increased in rsks-1(tm1714) mutants compared with that in the N2 strain (Figure 5A). Note that the positive control gene, pha-4 [29], was also increased in rsks-1(tm1714) mutants (Figure 5B). Further, as shown in Figure 5C and Table 1, we observed that rsks-1(tm1714) mutant strains showed elongated ML compared to N2 [ML of L4440 RNAi N2: 18.06 ± 0.44, ML of L4440 RNAi rsks-1: 21.84 ± 0.42; (P < 0.0001)]. In addition, fbxc-58 RNAi significantly reduced the lifespan of rsks-1(tm1714) mutants [ML of L4440 RNAi rsks-1: 21.84 ± 0.42, ML of fbxc-58 RNAi rsks-1: 17.32 ± 0.37; (P < 0.0001)], indicating that fbxc-58 is necessary for extending the longevity of rsks-1(tm1714) mutants, and this implies that fbxc-58-mediated lifespan extension in DR is dependent of S6 kinase signaling pathway (Figure 5D).
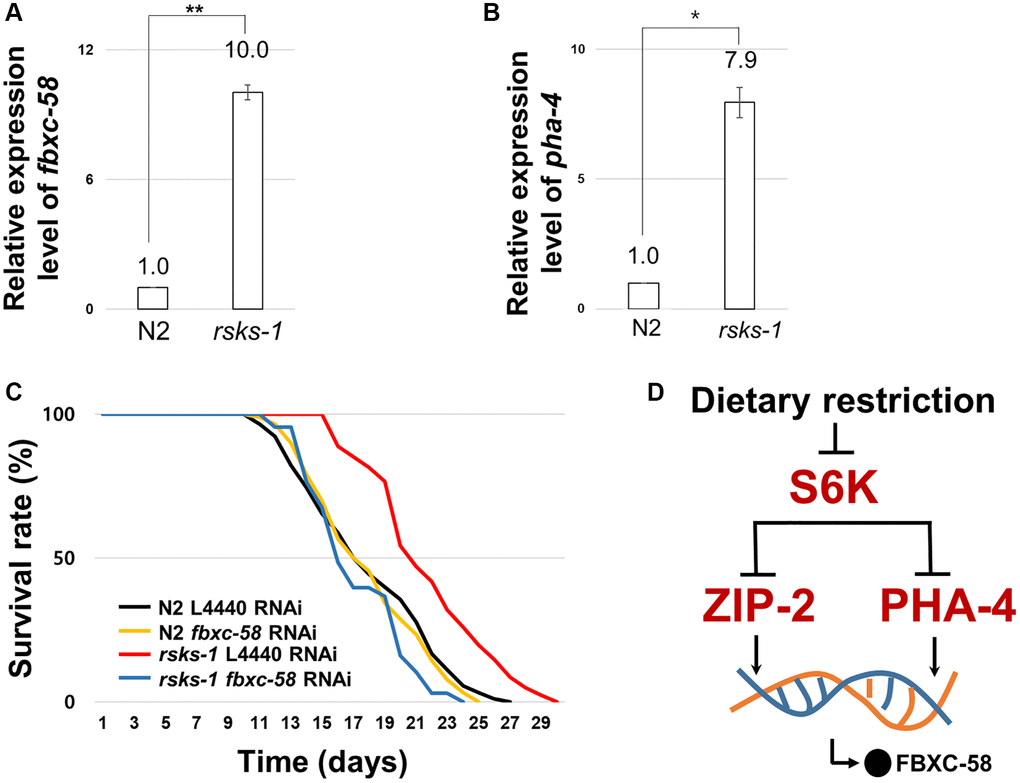
Discussion
DR is a practically effective and reproducible nutritional intervention that extends lifespan in many organisms. However, the underlying mechanisms mediating the effects of DR remain largely unknown. Many studies have shown that DR improves immune function, and immune signaling components are required for DR-induced lifespan extension. These results support the idea that the immune system acts as an important mechanism for DR-induced longevity. Recently, analysis of genes that regulate aging or immune response in animal models, including C. elegans, Drosophila, mice, and even humans, has revealed that aging and immunity are controlled by the same signaling pathways, such as TOR/S6K signaling pathway, pleiotropically [30]. DR-induced longevity is also associated with the modulation of the TOR/S6K signaling pathway [31–33]. Thus, these results suggest that the immune function may be closely associated with aging regulation through DR. In this study, we found that the F-box gene fbxc-58 is a downstream effector of the S6K signaling pathway (Figure 5), and that it regulates both pathogen resistance and aging in C. elegans. Furthermore, fbxc-58 is necessary for the effects of DR on lifespan extension. F-box protein contains conserved F-box domain [19], which acts as a site for protein-protein interaction. F-box protein acts as a modular E3 ubiquitin ligase adaptor protein, and the ubiquitin-dependent mechanisms have been shown to determine lifespan in response to DR [34] or modulate the innate immune response [35]. Therefore, we suggest that gaining insights into the detailed mechanistic aspects of fbxc-58 signaling pathway could elucidate the conserved signaling mechanism that links innate immunity and DR-induced healthy aging in animals.
Further, DR prevents or reduces the burden of age-related diseases or disabilities. Especially, in an aging and sedentary society, sarcopenia, an age-associated muscle disease, is beginning to be recognized as an acute disease condition. Although an effective sarcopenia treatment regime has not yet been identified, nutritional intervention is considered an effective method of preventing sarcopenia [36]. In this study, we found that DR prevents muscle aging via fbxc-58 in C. elegans. fbxc-58 is essential for DR-mediated alleviation of the age-associated decline in muscle activity and protection of mitochondrial network in body wall muscle. Thus, we propose that investigating the molecular mechanism of action of F-box proteins, including fbxc-58, in DR will shed light on means to prevent sarcopenia and offer a potentially practical means of encouraging healthy aging via DR.
Materials and Methods
Strains
The following strains were used. N2 wild-type, eat-2(ad1116), rsks-1(tm1714), PD4251 ccIs4251 [(pSAK2) myo-3p::GFP::LacZ::NLS + (pSAK4) myo-3p::mitochondrial GFP + dpy-20(+)]; dyp-20(e1282), and eat-2(ad1116); PD4251. In this study, all strains were maintained at 20°C.
Quantitative-RT PCR
Total RNA was extracted by using RNeasy Fibrous Tissue Mini Kit (Qiagen, Cat No. 79306). cDNA was generated by using a reverse transcription system (ReverTra Ace™ qPCR RT Master Mix, Toyobo, Cat No. FSQ-201) and was used for quantitative PCR. Quantitative real time PCR was performed with SYBR® Green Realtime PCR Master Mi (Toyobo, Cat No. QPK-201) using A ViiA 7 Real-Time PCR System (ThermoFisher) and analyzed using ΔΔCt methods described in the manufacturer’s manual. Sequences of primers used for quantitative RT-PCR analysis; fbxc-37-Forward: AGTTCGAGATGATCTGCTGC, and fbxc-37-Reverse: TCCAATTGTTAACCACCCATG, fbxc-63-Forward: GATTTTCCAAGCCAAGTCGC, fbxc-63-Reverse: ACTACCACCAGTATAGTCCG, fbxc-69-Forward: GATGAATTATCCGTGCAAACTG, fbxc-69-Reverse: TCACATCACCATTGTAATCCG, fbxc-79-Forward: GATGCTAGGAAGATTGAAGAG, fbxc-79-Reverse: CCATACTGTACTTCAAACGAG, fbxc-83-Forward: GCTCACGTTCACAGAAGAAG, fbxc-83-Reverse: AAAACTGCGTTGTTGGCTTTG, fbxc-98-Forward: GCAAAAGTTAGAGATGTGCTC, fbxc-98-Reverse: CAGTAGAATGCACACGTAATG, fbxc-58-Forward: CAGAAGAGGAGAAACCGAAG, and fbxc-58-Reverse: GCAAGGAGTCTCACTCTTTTC, pha-4-Forward: CTGTTAATCACAGTCAACCTAC, pha-4-Reverse: GTGTTGTTCAGGAAATTCTGG, irg-1-Forward: GCTGAAATTCACTTGTAGTGAG, irg-1-Reverse: GAGACCATAATTTCAATTGCTC, act-3-Forward: AAGTCATCACCGTCGGAAAC, and act-3-Reverse: TTCCTGGGTACATGGTGGTT. All qRT-PCR experiments were performed as three independent experiments.
Low density food (LDF) feeding experiments
To test DR effect by limiting food concentration, the nematode growth medium (NGM) plate without peptone was used for limiting the density of food (OP50 E. coli). After culturing the OP50 strain in Luria-Bertani (LB) broth containing streptomycin (20 mg/mL) at 37°C overnight, the culture mixture was centrifuged, and the pellets were resuspended and washed with M9 buffer containing streptomycin. The concentration of the OP50 on the assay plates was determined using a UV spectrometer, and the optical density (OD) value of bacteria in the LDF condition is 0.36. 200 μL aliquots of LDF (0.36 OD) preparation was spread over the assay plate. Synchronized worms were transferred to NGM (ad libitum, AL) or LDF plates at the young adult stage. After 48 hours, worms were collected and used for RT-qPCR experiments to observe the expression level of fbxc-58 (Figure 1C).
Life span analysis
The number of live animals was scored every 1–2 day until death in solid NGM plate. The death of worm was defined as the failure to respond to gentle prodding on the head or tail with a platinum wire. Life span was assessed at 20°C, and assay was performed without FUDR treatment. Life span was analyzed by Oasis survival analysis software (https://sbi.postech.ac.kr/oasis/) [37].
Qualitative analysis of mitochondrial morphology
The morphological categories of mitochondria are defined as follows. Images showing most of the long interconnected mitochondrial networks were classified as tubular, and images showing most of the short mitochondria or sparse globular mitochondria were classified as fragmented. Mitochondrial morphology was examined in eat-2(ad1116); PD4251 strain that emits fluorescence from mitochondria due to GFP expressed in mitochondria. Worms were immobilized during imaging using 100 mM sodium azide. Imaging was performed using a microscope equipped with an Olympus Fluoview FV3000 confocal laser scanning microscope. FV3000 RS Fluoview software was used to acquire fluorescent z stacks of individual animals (1 μm/slice).
Measurement of worm’s body bend number
The body bend number of worms tested in liquid M9 buffer. The number of changes in the reciprocating motion of bending at the center of the body was counted. The body bend was observed for 10 seconds by an Olympus SZX7 zoom stereo microscope (Olympus Corporation, Japan).
Measurement of worm’s maximum velocity (MV)
The physical assay plate was NGM plate without peptone and with no bacterial lawn. Synchronized single worms were transferred to the physical assay plate and movements recorded immediately. The recording system comprised a stereomicroscope (Olympus SZX7), a CCD camera (Olympus DR74), and imaging software (TUCSEN ISCapture). The locomotion velocity was expressed as mm per second (the distance (mm) between displaced centroids per second). Recorded images were analyzed by ImageJ and wrMTrck (plugin for ImageJ: https://www.phage.dk/plugins). The locomotion velocity data were imported into an Excel spreadsheet, and the peak locomotion velocity in the 30 s period was used as the MV.
RNAi experiments
In this study, commercial C. elegans RNAi feeding libraries generated by the Ahringer laboratory (Geneservice Ltd., Cambridge, UK) was used. For gene expression observation, RNAi treatment was for 72 hours from young adulthood. In addition, each young adult worms were transferred to each RNAi plate and life span (Figure 2, Figure 5C) or physical activities (number of body bends or MV) (Figure 3A, 3B and Supplementary Figures 1–4) or mitochondrial morphology (Figure 3C and Supplementary Figure 5) were examined.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author Contributions
J.H.H. and J.A. conceived and designed the study and wrote the manuscript. J.H.H., F.S.N. and P.G.C. performed the experimental works and J.H.H., J.A., H.D.S., T.H., and C.H.J analyzed the data. J.H.H. and J.A. edited the manuscript.
Acknowledgments
We thank the Caenorhabditis Genetics Center (CGC) for strains.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This work was supported by grants from the Korea Food Research Institute (E0210101).
References
- 1. Kalyani RR, Corriere M, Ferrucci L. Age-related and disease-related muscle loss: the effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol. 2014; 2:819–29. https://doi.org/10.1016/S2213-8587(14)70034-8 [PubMed]
- 2. Fontana L, Partridge L. Promoting health and longevity through diet: from model organisms to humans. Cell. 2015; 161:106–18. https://doi.org/10.1016/j.cell.2015.02.020 [PubMed]
- 3. Martin CK, Bhapkar M, Pittas AG, Pieper CF, Das SK, Williamson DA, Scott T, Redman LM, Stein R, Gilhooly CH, Stewart T, Robinson L, Roberts SB, and Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy (CALERIE) Phase 2 Study Group. Effect of Calorie Restriction on Mood, Quality of Life, Sleep, and Sexual Function in Healthy Nonobese Adults: The CALERIE 2 Randomized Clinical Trial. JAMA Intern Med. 2016; 176:743–52. https://doi.org/10.1001/jamainternmed.2016.1189 [PubMed]
- 4. Hahm JH, Jeong C, Nam HG. Diet restriction-induced healthy aging is mediated through the immune signaling component ZIP-2 in Caenorhabditis elegans. Aging Cell. 2019; 18:e12982. https://doi.org/10.1111/acel.12982 [PubMed]
- 5. Lee JE, Rayyan M, Liao A, Edery I, Pletcher SD. Acute Dietary Restriction Acts via TOR, PP2A, and Myc Signaling to Boost Innate Immunity in Drosophila. Cell Rep. 2017; 20:479–90. https://doi.org/10.1016/j.celrep.2017.06.052 [PubMed]
- 6. Nikolich-Žugich J. Aging of the T cell compartment in mice and humans: from no naive expectations to foggy memories. J Immunol. 2014; 193:2622–9. https://doi.org/10.4049/jimmunol.1401174 [PubMed]
- 7. Messaoudi I, Warner J, Fischer M, Park B, Hill B, Mattison J, Lane MA, Roth GS, Ingram DK, Picker LJ, Douek DC, Mori M, Nikolich-Zugich J. Delay of T cell senescence by caloric restriction in aged long-lived nonhuman primates. Proc Natl Acad Sci U S A. 2006; 103:19448–53. https://doi.org/10.1073/pnas.0606661103 [PubMed]
- 8. Spadaro O, Youm Y, Shchukina I, Ryu S, Sidorov S, Ravussin A, Nguyen K, Aladyeva E, Predeus AN, Smith SR, Ravussin E, Galban C, Artyomov MN, Dixit VD. Caloric restriction in humans reveals immunometabolic regulators of health span. Science. 2022; 375:671–7. https://doi.org/10.1126/science.abg7292 [PubMed]
- 9. Kristan DM. Chronic calorie restriction increases susceptibility of laboratory mice (Mus musculus) to a primary intestinal parasite infection. Aging Cell. 2007; 6:817–25. https://doi.org/10.1111/j.1474-9726.2007.00345.x [PubMed]
- 10. Gardner EM. Caloric restriction decreases survival of aged mice in response to primary influenza infection. J Gerontol A Biol Sci Med Sci. 2005; 60:688–94. https://doi.org/10.1093/gerona/60.6.688 [PubMed]
- 11. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153:1194–217. https://doi.org/10.1016/j.cell.2013.05.039 [PubMed]
- 12. Wu Z, Isik M, Moroz N, Steinbaugh MJ, Zhang P, Blackwell TK. Dietary Restriction Extends Lifespan through Metabolic Regulation of Innate Immunity. Cell Metab. 2019; 29:1192–205.e8. https://doi.org/10.1016/j.cmet.2019.02.013 [PubMed]
- 13. Kenyon CJ. The genetics of ageing. Nature. 2010; 464:504–12. https://doi.org/10.1038/nature08980 [PubMed]
- 14. Youngman MJ, Rogers ZN, Kim DH. A decline in p38 MAPK signaling underlies immunosenescence in Caenorhabditis elegans. PLoS Genet. 2011; 7:e1002082. https://doi.org/10.1371/journal.pgen.1002082 [PubMed]
- 15. Lakowski B, Hekimi S. The genetics of caloric restriction in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 1998; 95:13091–6. https://doi.org/10.1073/pnas.95.22.13091 [PubMed]
- 16. Kim DH, Ewbank JJ. Signaling in the innate immune response. WormBook. 2018; 2018:1–35. https://doi.org/10.1895/wormbook.1.83.2 [PubMed]
- 17. Craig KL, Tyers M. The F-box: a new motif for ubiquitin dependent proteolysis in cell cycle regulation and signal transduction. Prog Biophys Mol Biol. 1999; 72:299–328. https://doi.org/10.1016/s0079-6107(99)00010-3 [PubMed]
- 18. Wu CW, Wang Y, Choe KP. F-Box Protein XREP-4 Is a New Regulator of the Oxidative Stress Response in Caenorhabditis elegans. Genetics. 2017; 206:859–71. https://doi.org/10.1534/genetics.117.200592 [PubMed]
- 19. Saito R, Shinkai Y, Doi M. Intestinal F-box protein regulates quick avoidance behavior of Caenorhabditis elegans to the pathogenic bacterium Pseudomonas aeruginosa. Genes Cells. 2019; 24:192–201. https://doi.org/10.1111/gtc.12666 [PubMed]
- 20. Fletcher M, Tillman EJ, Butty VL, Levine SS, Kim DH. Global transcriptional regulation of innate immunity by ATF-7 in C. elegans. PLoS Genet. 2019; 15:e1007830. https://doi.org/10.1371/journal.pgen.1007830 [PubMed]
- 21. Hahm JH, Kim S, DiLoreto R, Shi C, Lee SJ, Murphy CT, Nam HG. C. elegans maximum velocity correlates with healthspan and is maintained in worms with an insulin receptor mutation. Nat Commun. 2015; 6:8919. https://doi.org/10.1038/ncomms9919 [PubMed]
- 22. Regmi SG, Rolland SG, Conradt B. Age-dependent changes in mitochondrial morphology and volume are not predictors of lifespan. Aging (Albany NY). 2014; 6:118–30. https://doi.org/10.18632/aging.100639 [PubMed]
- 23. Johnson ML, Robinson MM, Nair KS. Skeletal muscle aging and the mitochondrion. Trends Endocrinol Metab. 2013; 24:247–56. https://doi.org/10.1016/j.tem.2012.12.003 [PubMed]
- 24. Estes KA, Dunbar TL, Powell JR, Ausubel FM, Troemel ER. bZIP transcription factor zip-2 mediates an early response to Pseudomonas aeruginosa infection in Caenorhabditis elegans. Proc Natl Acad Sci U S A. 2010; 107:2153–8. https://doi.org/10.1073/pnas.0914643107 [PubMed]
- 25. Hahm JH, Jeong C, Lee W, Koo HJ, Kim S, Hwang D, Nam HG. A cellular surveillance and defense system that delays aging phenotypes in C. elegans. Aging (Albany NY). 2020; 12:8202–20. https://doi.org/10.18632/aging.103134 [PubMed]
- 26. Panowski SH, Wolff S, Aguilaniu H, Durieux J, Dillin A. PHA-4/Foxa mediates diet-restriction-induced longevity of C. elegans. Nature. 2007; 447:550–5. https://doi.org/10.1038/nature05837 [PubMed]
- 27. Dufner A, Thomas G. Ribosomal S6 kinase signaling and the control of translation. Exp Cell Res. 1999; 253:100–9. https://doi.org/10.1006/excr.1999.4683 [PubMed]
- 28. Zid BM, Rogers AN, Katewa SD, Vargas MA, Kolipinski MC, Lu TA, Benzer S, Kapahi P. 4E-BP extends lifespan upon dietary restriction by enhancing mitochondrial activity in Drosophila. Cell. 2009; 139:149–60. https://doi.org/10.1016/j.cell.2009.07.034 [PubMed]
- 29. Sheaffer KL, Updike DL, Mango SE. The Target of Rapamycin pathway antagonizes pha-4/FoxA to control development and aging. Curr Biol. 2008; 18:1355–64. https://doi.org/10.1016/j.cub.2008.07.097 [PubMed]
- 30. Fabian DK, Fuentealba M, Dönertaş HM, Partridge L, Thornton JM. Functional conservation in genes and pathways linking ageing and immunity. Immun Ageing. 2021; 18:23. https://doi.org/10.1186/s12979-021-00232-1 [PubMed]
- 31. Vellai T, Takacs-Vellai K, Zhang Y, Kovacs AL, Orosz L, Müller F. Genetics: influence of TOR kinase on lifespan in C. elegans. Nature. 2003; 426:620. https://doi.org/10.1038/426620a [PubMed]
- 32. Jia K, Chen D, Riddle DL. The TOR pathway interacts with the insulin signaling pathway to regulate C. elegans larval development, metabolism and life span. Development. 2004; 131:3897–906. https://doi.org/10.1242/dev.01255 [PubMed]
- 33. Selman C, Tullet JM, Wieser D, Irvine E, Lingard SJ, Choudhury AI, Claret M, Al-Qassab H, Carmignac D, Ramadani F, Woods A, Robinson IC, Schuster E, et al. Ribosomal protein S6 kinase 1 signaling regulates mammalian life span. Science. 2009; 326:140–4. https://doi.org/10.1126/science.1177221 [PubMed]
- 34. Carrano AC, Liu Z, Dillin A, Hunter T. A conserved ubiquitination pathway determines longevity in response to diet restriction. Nature. 2009; 460:396–9. https://doi.org/10.1038/nature08130 [PubMed]
- 35. Escobar-Henriques M, Langer T. Dynamic survey of mitochondria by ubiquitin. EMBO Rep. 2014; 15:231–43. https://doi.org/10.1002/embr.201338225 [PubMed]
- 36. Colman RJ, Beasley TM, Allison DB, Weindruch R. Attenuation of sarcopenia by dietary restriction in rhesus monkeys. J Gerontol A Biol Sci Med Sci. 2008; 63:556–9. https://doi.org/10.1093/gerona/63.6.556 [PubMed]
- 37. Yang JS, Nam HJ, Seo M, Han SK, Choi Y, Nam HG, Lee SJ, Kim S. OASIS: online application for the survival analysis of lifespan assays performed in aging research. PLoS One. 2011; 6:e23525. https://doi.org/10.1371/journal.pone.0023525 [PubMed]


