Introduction
The average global prevalence of dyslipidemia is about 20% [1–3], and it is higher in patients with premature coronary arteriosclerotic diseases (CAD) [4] and chronic kidney disease [5]. Dyslipidemia plays crucial roles in the pathogenesis of hypertension, CAD, stroke, and chronic kidney failure that differ based on etiology, and identification of novel and safe treatments that decrease blood lipid levels, especially the level of low-density lipoprotein cholesterol (LDL-C), would be beneficial [6]. It is well known that lifestyle, environmental, and genetic factors all contribute to dyslipidemia. In the past decade, technological advances in biological sequencing technology have enabled genome-wide association studies (GWASs) that have helped thoroughly characterize risk factors for dyslipidemia.
The tubulin beta class I gene (TUBB, gene ID: 203068) located on chromosome 6 in humans encodes a beta tubulin protein that is an essential component of microtubules. Microtubules perform many cellular functions, including chromosome segregation, maintenance of cell shape, transport and motility, and organelle distribution [7]. As a substrate of peptidylarginine deiminases, TUBB participates in citrullination, which is related to growth, infiltration, and drug resistance in some tumor cells. Thus, mutations in TUBB are involved in complex diseases [8] and carcinoma [9]. The WW domain-containing oxidoreductase gene (WWOX, gene ID: 51741) on human chromosome 16 encodes a member of the short-chain dehydrogenases/reductases protein family that is involved in a variety of important cellular processes, including induction of apoptosis, cell development, and steroid metabolism [10, 11]. Mutations in WWOX are not only associated with multiple types of cancer [11], but also with lipid metabolism [12]. Both TUBB and WWOX are ubiquitously expressed in many tissues, including fat (reads per kilobase per million mapped reads (RPKM) 97.7 and 1.6, respectively), heart (RPKM 67.2 and 0.5) and liver (RPKM 58.1 and 0.9). Because of their functions and tissue distribution, single nucleotide polymorphisms in the TUBB and WWOX genes can have similar effects on lipid profiles. Recently, GWASs revealed that SNPs at locations rs3132584 and rs3130685 in TUBB are associated with LDL-C profile [13], while SNPs at rs2222896 and rs2548861 in WWOX are associated with LDL-C and high-density lipoprotein cholesterol (HDL-C) profiles [14], respectively. However, associations between TUBB and WWOX and lipid traits in the Chinese population have not been examined; we hypothesize that modifications of TUBB and WWOX affect blood lipid levels in this population as well.
The incidence of dyslipidemia in Chinese adults is as high as 34% [15]. Previous studies revealed that several lipid-related genes are associated with different lipid traits in the Maonan and Han populations [16–18]. The Maonan ethnicity is one of 55 recognized minorities in China, and seventy percent (64,500) of Maonan people live in Huanjiang Maonan Autonomous County of Guangxi Zhuang Autonomous Region according to a wire report from Xinhua News Agency (Beijing, May 20th, 2020). The Maonan people maintain a unique lifestyle and wedding culture characterized by a fat-rich diet, recreational use of alcohol and tobacco, and intra-ethnic marriages. As a result, the Maonan population has a singular genetic background and specific environmental risk factors that are particularly informative in genetic studies of dyslipidemia. In this study, we therefore explored associations between TUBB-WWOX SNPs, their haplotypes, and gene-gene (G × G) and gene-environment (G × E) interactions and the prevalence of dyslipidemia in the Maonan population.
Results
Participant characteristics
As shown in Table 1, no statistically significant differences in mean age, gender ratio, height, cigarette smoking, and proportion over 65 years old were observed between the normal and dyslipidemia groups (P > 0.05). However, mean weight, waist circumference, body mass index (BMI), alcohol consumption, systolic blood pressure (SBP), diastolic blood pressure (DBP), pulse pressure (PP), fasting blood-glucose (FBS), hypertension morbidity, and proportion with BMI greater than 24 kg/m2 and with FBS of at least 7.0 mM were higher in the dyslipidemia group than in the normal group (P < 0.01). In addition, total cholesterol (TC), triglyceride (TG), LDL-C, and apolipoprotein (Apo) B levels were also significantly higher in the dyslipidemia group than in the normal group (P < 0.001). In contrast, HDL-C and ApoA1 levels and ApoA1/ApoB ratio were significantly lower in the dyslipidemia group than in the normal group (P < 0.001).
Table 1. General and biochemical characteristics of the participants.
| Parameter | Normal | Dyslipidemia | t (χ2) | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number | 864 | 1129 | – | – | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male/female | 431/433 | 545/584 | 0.509 | 0.476 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, years1 | 55.31±13.08 | 56.04±12.88 | -1.240 | 0.215 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Height, cm | 156.34±8.01 | 156.75±8.32 | -1.123 | 0.262 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight, kg | 54.48±110.29 | 57.06±11.07 | -5.365 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Waistline, cm | 76.06±9.31 | 80.33±9.47 | -8.199 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body mass index (BMI), kg/m2 | 22.21±3.31 | 23.11±3.39 | -5.971 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cigarette smoking, n (%)2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 cigarette/day | 622(71.99) | 818(72.45) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤ 20 cigarettes/day | 192(22.22) | 239(21.17) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| > 20 cigarettes/day | 50(5.79) | 72(6.38) | 0.544 | 0.762 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alcohol consumption, n (%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 g/day | 607(70.25) | 841(74.49) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤ 25 g/day | 50(5.79) | 84(7.44) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| > 25 g/day | 207(23.96) | 204(18.07) | 11.633 | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Systolic blood pressure, mmHg | 127.80±20.45 | 135.34±22.30 | -7.844 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diastolic blood pressure, mmHg | 79.83±12.29 | 84.53±12.91 | -8.276 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pulse pressure, mmHg | 48.91±15.00 | 51.31±16.39 | -3.404 | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fasting blood-glucose (FBS), mM | 6.03±0.94 | 6.32±1.23 | -5.915 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total cholesterol, mM | 4.33±0.50 | 4.89±0.72 | -20.024 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Triglyceride, mM | 1.00(1.07) | 1.56(2.02) | -28.603 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL-C, mM | 1.30±0.18 | 1.15±0.17 | 19.195 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C, mM | 2.90±0.15 | 3.43±0.31 | -49.871 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Apolipoprotein (Apo)A1, g/L | 1.32±0.18 | 1.28±0.16 | 5.931 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoB, g/L | 0.94±0.14 | 1.00±0.10 | -10.755 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1/ApoB | 1.45±0.32 | 1.29±0.24 | 11.697 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension, n (%) | 288(33.33) | 571(43.96) | 59.334 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age > 65 years | 194(22.45) | 255(22.59) | 0.0050 | 0.944 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI > 24 kg/m2 | 255(22.59) | 475(42.07) | 55.200 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FBS ≥ 7.0 mM, n (%) | 164(18.98) | 495(43.84) | 52.194 | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol. 1Normally distributed quantitative data were assessed in a t-test and are described as means ± SD. 2Qualitative data were assessed using the chi-square test. 3Non-normally distributed quantitative data were assessed using the Wilcoxon-Mann-Whitney test and are described as medians (interquartile range). P < 0.05 indicated a statistically significant difference. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Genotype and allele frequencies
Information for four target SNPs (rs3132584, rs3130685, rs2222896, and rs2548861) in the TUBB and WWOX genes among the Chinese Han Beijing (CHB) population in NCBI dbSNP Build 132 is shown in Table 2; genotypic and allelic frequencies of these four SNPs in the Maonan population are presented in Table 3. The frequencies of point mutations at all four SNPs were similar between the CHB and Maonan populations. Mutations from G to T and from C to T were observed at the TUBB rs3132584 and rs3130685 SNPs, respectively, while mutations from A to G and from T to G were observed at the WWOX rs2222896 and rs2548861 SNPs, respectively. However, genotypic and allelic frequencies of the four SNPs differed between the ethnic groups; in particular, the frequency of the rs3132584TT genotype was 5.0% in Maonan population but was 0% in the CHB population. In this study, allelic and genotypic distributions of the four SNPs in both groups were consistent with the Hardy Weinberg Equilibrium (HWE, P > 0.05 for all). Additionally, allele and genotype frequencies of the rs3132584, rs3130685, and rs2222896 SNPs differed between the normal and dyslipidemia groups (P ≤ 0.01 for all).
Table 2. Frequencies of TUBB and WWOX SNPs in the Chinese Han Beijing population [n(%)].
| Gene | Chromosome | SNP | Allele1 | Allele2 | PHWE | Genotype detail of CHB | Source | Phenotype | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TUBB | 6 | rs3132584 | G(0.8171) | *T(0.1829) | 0.4386 | GG(0.6341) | GT(0.3591) | — | 1000 Genome | ↑LDL(G) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 6 | rs3130685 | *C(0.4556) | T(0.5444) | 1.0000 | CC(0.2000) | CT(0.5111) | TT(0.2889) | 1000 Genome | ↓LDL(T) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WWOX | 16 | rs2222896 | *A(0.3659) | G(0.6341) | 0.7518 | AA(0.1220) | AG(0.3902) | GG(0.4878) | 1000 Genome | ↑LDL(G) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 16 | rs2548861 | *T(0.4512) | G(0.5488) | 0.4028 | TT(0.1707) | TG(0.5610) | GG(0.2683) | 1000 Genome | ↓HDL(T) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP, single nucleotide polymorphism; HWE, Hardy-Weinberg equilibrium; CHB, Chinese Han Beijing. Allele1 and the allele2 indicate reference and alternate alleles, respectively; *allele indicates the minor allele. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 3. Genotypic and allelic frequencies of TUBB and WWOX SNPs in normal and dyslipidemia groups [n(%)].
| Gene SNP | Genotype (allele) | Total (n =1993) | Normal (n = 864) | Dyslipidemia (n = 1229) | χ2 | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TUBB | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3132854 | GG | 1223(61) | 467(54) | 756(67) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GT | 676(34) | 337(39) | 339(30) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | 94(5) | 60(7) | 34(3) | 40.978 | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G | 3122(78) | 1271(74) | 1851(82) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T | 864(22) | 457(26) | 407(18) | 40.896 | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHWE | 0.95 | 1.000 | 0.690 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3130685 | CC | 361(18) | 138(16) | 223(20) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT | 980(49) | 415(48) | 565(50) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | 652(33) | 311(36) | 341(30) | 9.282 | 0.010 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T | 2284(57) | 1037(60) | 1247(55) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C | 1702(43) | 691(40) | 1011(45) | 9.163 | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHWE | 0.850 | 1.000 | 0.720 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WWOX | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896 | AA | 395(20) | 259(30) | 136(12) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AG | 961(48) | 444(51) | 517(46) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GG | 637(32) | 161(19) | 476(42) | 167.338 | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G | 2235(56) | 766(44) | 1469(65) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A | 1751(44) | 962(56) | 789(35) | 170.763 | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHWE | 0.340 | 0.240 | 0.840 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2548861 | TT | 287(14) | 118(14) | 169(15) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG | 947(48) | 404(47) | 543(48) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GG | 759(38) | 342(40) | 417(37) | 1.670 | 0.434 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G | 2465(62) | 1088(63) | 1377(61) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T | 1521(38) | 640(37) | 881(39) | 1.626 | 0.202 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHWE | 0.780 | 1.000 | 0.750 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Qualitative data were assessed using the Chi-square test; P < 0.05 indicated a statistically significant difference. HWE, Hardy-Weinberg equilibrium. PHWE > 0.05 indicated a statistically significant difference. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Associations between genotypes and alleles, serum lipid levels, and dyslipidemia
Associations between the four TUBB and WWOX SNPs and serum lipid levels are shown in Figure 1. Serum levels of HDL-C, LDL-C, and ApoA1 differed among the three rs2222896 genotypes in the normal group (P ≤ 0.001 for all); rs2222896G allele carriers had higher LDL-C and lower HDL-C levels than those without rs2222896G. However, there were no differences in serum lipid levels depending on genotype for the other three SNPs. In the dyslipidemia group, some lipid parameters differed significantly depending on genotype for the rs3132584, rs2222896, and rs2548861 SNPs (P < 0.0125 for all). Furthermore, higher TC levels and lower ApoA1/ApoB ratio were associated with rs2222896G and rs2548861T alleles, higher LDL-C and ApoB levels and lower HDL-C levels were associated with the rs3132584G, rs2222896G, and rs2548861T alleles, lower ApoA1 levels were associated with the rs2222896G allele, and lower ApoA1/ApoB ratio was associated with the rs2548861T and rs2222896G alleles.
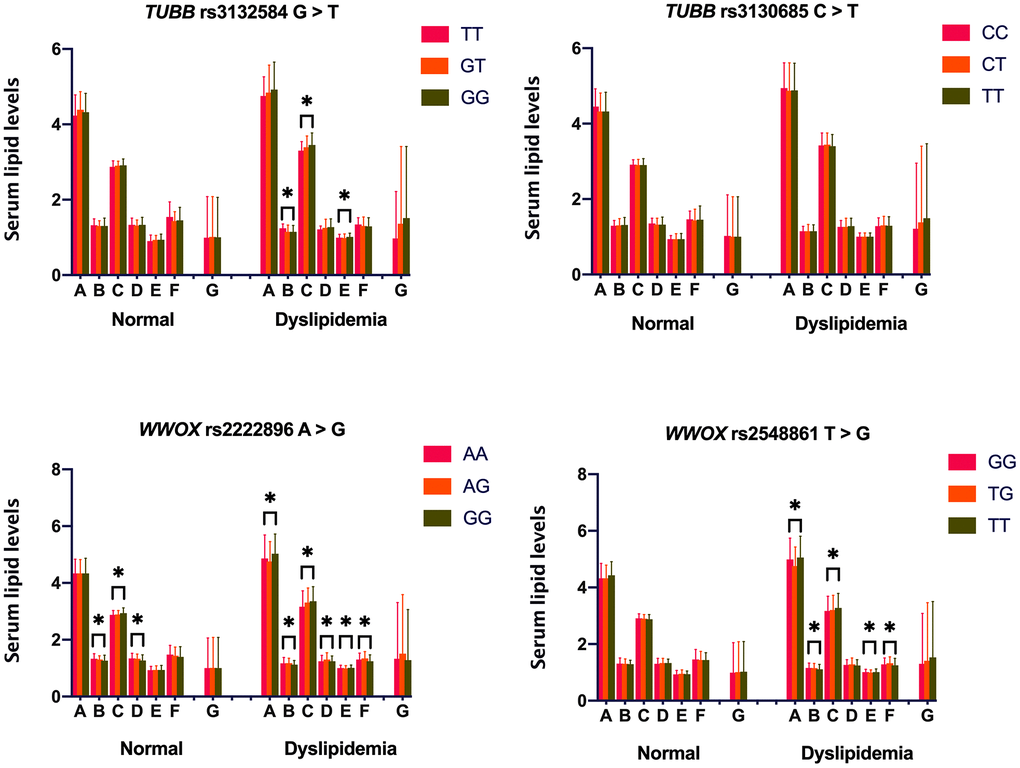
Figure 1. Associations between TUBB-WWOX genotypes and serum lipid levels in normal and dyslipidemia groups. A = Total cholesterol (mM); B = High-density lipoprotein cholesterol (mM); C = Low-density lipoprotein cholesterol (mM); D = Apolipoprotein (Apo) A1 (g/L); E = ApoB (g/L); F = ApoA1/ApoB ratio; and G = Triglyceride (mM). *P-value < 0.006 indicated a statistically significant difference after Bonferroni correction.
Because environmental factors can affect lipid phenotypes, we adjusted for the risk factors of age, sex, drinking, smoking, FBS, and BMI, to clarify relationships between genotypes and lipid phenotypes (Table 4). Overall, in the normal group, serum HDL-C levels were lower in rs2222896G allele carriers than in rs2222896G non-carriers (P ≤ 0.001), LDL-C was higher, but ApoA1 was lower, in subjects with the rs2222896GG genotype compared to those with the rs2222896AA genotype (P < 0.001), ApoB levels were higher in subjects with the rs3132584GG genotype than in those with the rs3132584TT genotype (P = 0.006), and ApoA1/ApoB ratio was lower in subjects with the rs2222896GG and rs3132584GT genotypes than in those with the rs2222896AA and rs3132584TT genotypes, respectively (P = 0.012 and P = 0.007). In the dyslipidemia group, serum TC and ApoB levels were higher, but ApoA1/ApoB ratio was lower, in subjects with the rs2548861GG genotype than in those with the rs2548861TG genotype (P ≤ 0.005 for all), serum HDL-C and ApoA1 levels and ApoA1/ApoB ratio were lower, but LDL-C levels were higher, in subjects with the rs2222896GG genotype than in those with the rs2222896AA genotype, and serum HDL-C levels were lower in subjects with the rs3132584GT, rs3132584GG, and rs2548861TT genotypes than in those with the rs3132584TT and rs2548861GG genotypes (P = 0.001 for all).
Table 4. Meaningful associations between the four SNPs and serum lipid levels in the normal and dyslipidemia groups.
| Lipid | SNP | Genotype | Beta | t | P | 95% CI of Beta | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reference | Alternate | Lower | Upper | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Normal | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL-C | rs2222896 | AA | AG | -0.131 | -3.313 | 0.001 | -0.076 | -0.019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896 | AA | GG | -0.165 | -4.232 | 0.000 | -0.112 | -0.041 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C | rs2222896 | AA | GG | 0.137 | 3.500 | 0.000 | 0.024 | 0.084 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1 | rs2222896 | AA | GG | -0.160 | -4.080 | 0.000 | -0.109 | -0.038 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Apo B | rs3132584 | TT | GG | 0.188 | 2.739 | 0.006 | 0.015 | 0.090 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1/ApoB | rs2222896 | AA | GG | -0.099 | -2.509 | 0.012 | -0.143 | -0.017 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3132584 | TT | GT | -0.188 | -2.708 | 0.007 | -0.210 | -0.034 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dyslipidemia | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC | 2548861 | GG | TG | -0.156 | -4.832 | 0.000 | -0.318 | -0.134 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL-C | rs3132584 | TT | GT | -0.272 | -3.302 | 0.001 | -0.159 | -0.041 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | GG | -0.296 | -3.598 | 0.000 | -0.164 | -0.048 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896 | AA | GG | -0.155 | -3.270 | 0.001 | -0.085 | -0.021 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2548861 | GG | TT | -0.113 | -3.471 | 0.001 | -0.084 | -0.023 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C | rs2222896 | AA | GG | 0.136 | 2.843 | 0.005 | 0.027 | 0.146 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1 | rs2222896 | AA | GG | -0.167 | -3.535 | 0.000 | -0.083 | -0.024 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Apo B | 2548861 | GG | TG | -0.089 | -2.695 | 0.007 | -0.031 | -0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1/ApoB | rs2222896 | AA | GG | -0.120 | -2.549 | 0.011 | -0.102 | -0.013 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2548861 | GG | TG | 0.085 | 2.577 | 0.010 | 0.010 | 0.070 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC, total cholesterol; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; ApoA1, apolipoprotein A1; ApoB, apolipoprotein B; CI, confidence interval. P values were calculated by multivariable linear regression. P < 0.0125 indicated a statistically significant difference after Bonferroni correction and adjusting for genotype, age, gender, drinking, smoking, fasting blood-glucose, and body mass index. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Associations between the four SNPs and dyslipidemia are shown in Table 5. rs3132584, rs3130685, and rs2222896 were associated with dyslipidemia (P < 0.05). Subjects with the rs3132584TT [codominant: adjusted odds ratio (OR) = 0.35, 95% confidence interval (CI) = 0.23–0.54, P < 0.0001] or rs3132584GT+TT (dominant: adjusted OR = 0.58, 95% CI = 0.48–0.70, P < 0.0001) genotypes had a lower risk of dyslipidemia than those with the rs3132584GG genotype. In contrast, subjects with the rs3130685CC (codominant: adjusted OR = 1.47, 95% CI = 1.13–1.92, P = 0.0096) or rs3130685 CT+TT (dominant: adjusted OR = 1.30, 95% CI = 1.08–1.57, P = 0.0064) genotypes had a higher risk of dyslipidemia than those with the rs3130685TT genotype. Finally, subjects with the rs2222896AA (codominant: adjusted OR = 0.18, 95% CI = 0.14–0.23, P < 0.0001) or rs2222896 AG+AA (dominant: adjusted OR = 0.31, 95% CI = 0.26–0.39, P < 0.0001) genotypes had a lower risk of dyslipidemia than those with the rs2222896GG genotype.
Table 5. Associations between genetic models of the four SNPs and dyslipidemia.
| Gene | Model | Genotype | χ2 | P | OR (95% CI) | *P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reference | Alternate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TUBB | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3132584 G > T | Codominant | GG | GT | 24.275 | 0.000 | 0.62(0.51–0.75) | < 0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | 23.917 | 0.000 | 0.35(0.23–0.54) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dominant | GG | GT+TT | 34.413 | 0.000 | 0.58(0.48–0.70) | < 0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recessive | GG+GT | TT | 16.846 | 0.000 | 0.42(0.27–0.64) | < 0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Log-additive | GT | TT | 6.456 | 0.011 | 0.61(0.52–0.71) | < 0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3130685C> T | Codominant | TT | CT | 4.542 | 0.033 | 1.24(1.02–1.52) | 0.0096 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | 8.448 | 0.004 | 1.47(1.13–1.92) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dominant | TT | CT+CC | 7.458 | 0.006 | 1.30(1.08–1.57) | 0.0064 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recessive | TT+CT | CC | 4.714 | 0.030 | 1.29(1.03–1.64) | 0.029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Log-additive | CT | CC | 1.848 | 0.174 | 1.22(1.07–1.38) | 0.0024 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WWOX | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896A>G | Codominant | GG | AG | 71.311 | 0.000 | 0.39(0.32–0.49) | < 0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AA | 164.027 | 0.000 | 0.18(0.14–0.23) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dominant | GG | AG+AA | 124.580 | 0.000 | 0.31(0.26–0.39) | < 0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recessive | GG+AG | AA | 99.024 | 0.000 | 0.32(0.25–0.40) | < 0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Log-additive | AG | AA | 42.060 | 0.000 | 0.42(0.37–0.48) | < 0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2548861T>G | Codominant | GG | TG | 0.985 | 0.321 | 1.10(0.91–1.34) | 0.4300 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | 1.315 | 0.251 | 1.17(0.89–1.55) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dominant | GG | TG+TT | 1.455 | 0.228 | 1.12(0.93–1.34) | 0.2300 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recessive | GG+TG | TT | 0.683 | 0.409 | 1.11(0.86–1.43) | 0.4100 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Log-additive | TG | TT | 0.216 | 0.642 | 1.09(0.96–1.24) | 0.2000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR, odds ratio; CI, confidence interval. P and *P < 0.0125 indicated a statistically significant difference. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Associations between haplotype frequencies, serum lipid levels, and dyslipidemia
Linkage disequilibrium (LD) was observed between the WWOX rs2222896 and rs2548861 SNPs in the normal and dyslipidemia groups (D′ = 0.870 or R2 = 0.350 and D′ = 0.810 or R2 = 0.220, respectively; Figure 2). Frequencies of the common haplotypes [lowest frequency threshold (LFT) > 0.03] of the two WWOX SNPs are shown in Table 6. The frequencies of the rs2222896A-rs2548861T, rs2222896G-rs2548861G, rs2222896A-rs2548861G, and rs2222896G-rs2548861T haplotypes differed between the two groups (P < 0.05 for all). The rs2222896G-rs2548861G and rs2222896G-rs2548861T haplotypes were associated with increased risk of dyslipidemia, whereas the rs2222896A-rs2548861T and rs2222896A-rs2548861G haplotypes were protective factors against dyslipidemia (P < 0.007 for all).
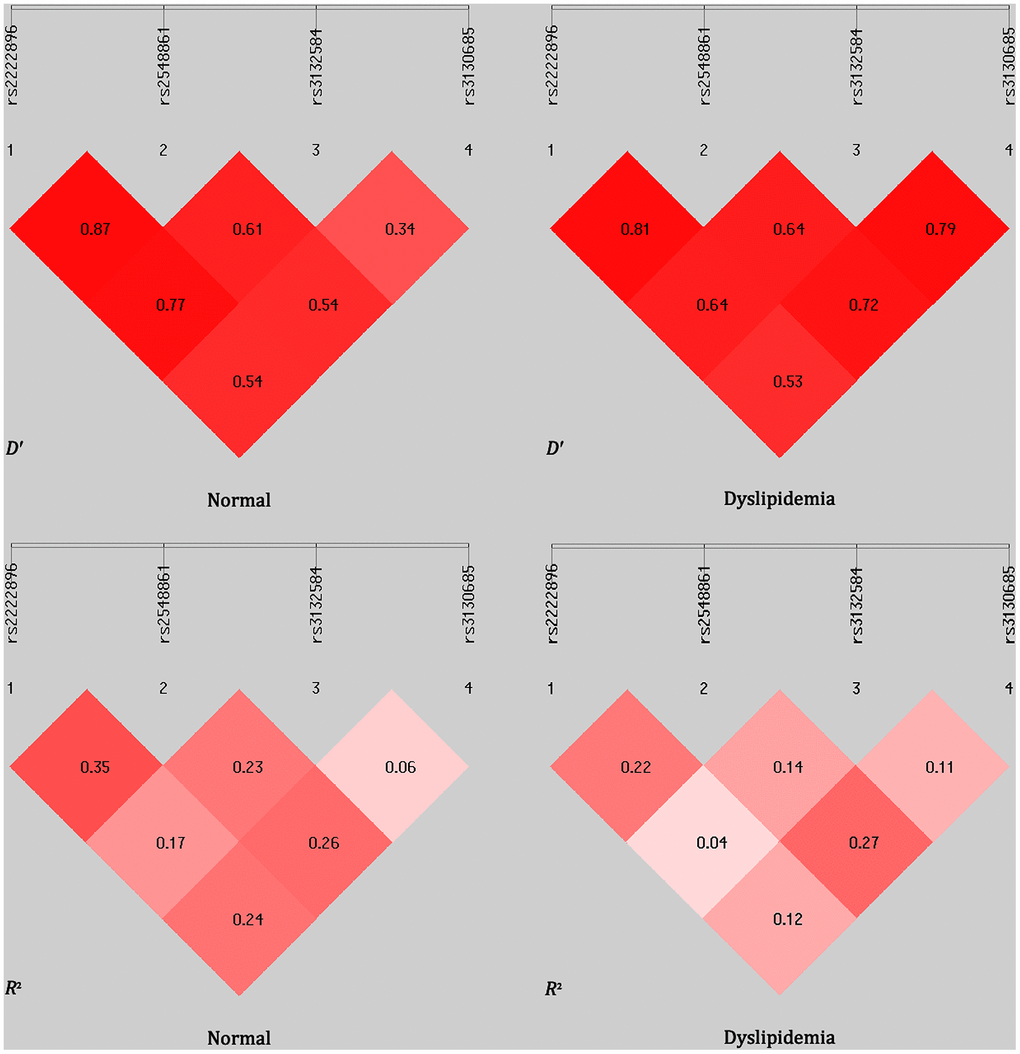
Figure 2. Linkage disequilibrium (LD) analysis for the four TUBB and WWOX SNPs in normal and dyslipidemia groups. LD status is indicated by D′ and R2 values.
Table 6. Haplotype frequencies of the two WWOX SNPs in the normal and dyslipidemia groups [n(frequency)].
| Label | Haplotype | Normal (n=1728) | Dyslipidemia (n=2258) | χ2 | P | OR (95% CI) | *P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1 | rs2222896A–rs2548861T | 604(0.349) | 451(0.199) | 112.869 | 2.30× 10−26 | 0.464(0.420–0.535) | < 0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H2 | rs2222896G–rs2548861G | 730(0.422) | 1039(0.460) | 5.632 | 0.017 | 1.165(1.026–1.322) | 0.003 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H3 | rs2222896A–rs2548861G | 358(0.207) | 338(0.149) | 22.445 | 2.16× 10−6 | 0.673(0.571–0.793) | 0.007 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H4 | rs2222896G–rs2548861T | 36(0.020) | 430(0.190) | 272.729 | 2.88× 10−61 | 11.055(7.818–15.633) | < 0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H, haplotype; OR, odds ratio; CI, confidence interval. P and *P < 0.05 indicated a statistically significant difference. n = total number with that haplotype. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Associations between the four haplotypes and serum lipid levels are shown in Figure 3. rs2222896G-rs2548861G haplotype carriers in the normal group had lower HDL-C levels than those without rs2222896G-rs2548861G haplotype (P = 0.001), rs2222896A-rs2548861G haplotype carriers had higher ApoA1 and HDL-C levels and ApoA1/ApoB ratio, but lower TG levels, than those without rs2222896A-rs2548861G haplotype (P ≤ 0.002), and rs2222896G-rs2548861T haplotype carriers had higher TC levels than those without rs2222896G-rs2548861T haplotypes (P < 0.001). In the dyslipidemia group, rs2222896A-rs2548861T haplotype carriers had lower TC and ApoB levels, but higher HDL-C and ApoA levels and ApoA1/ApoB ratio than those without corresponding haplotype (P < 0.001), rs2222896A-rs2548861G haplotype carriers had lower LDL-C and higher ApoA1 and TG levels than those without corresponding haplotype (P ≤ 0.004), and rs2222896G-rs2548861T haplotype carriers had lower ApoA1 and HDL-C levels and ApoA1/ApoB ratio than those without corresponding haplotype (P < 0.001).
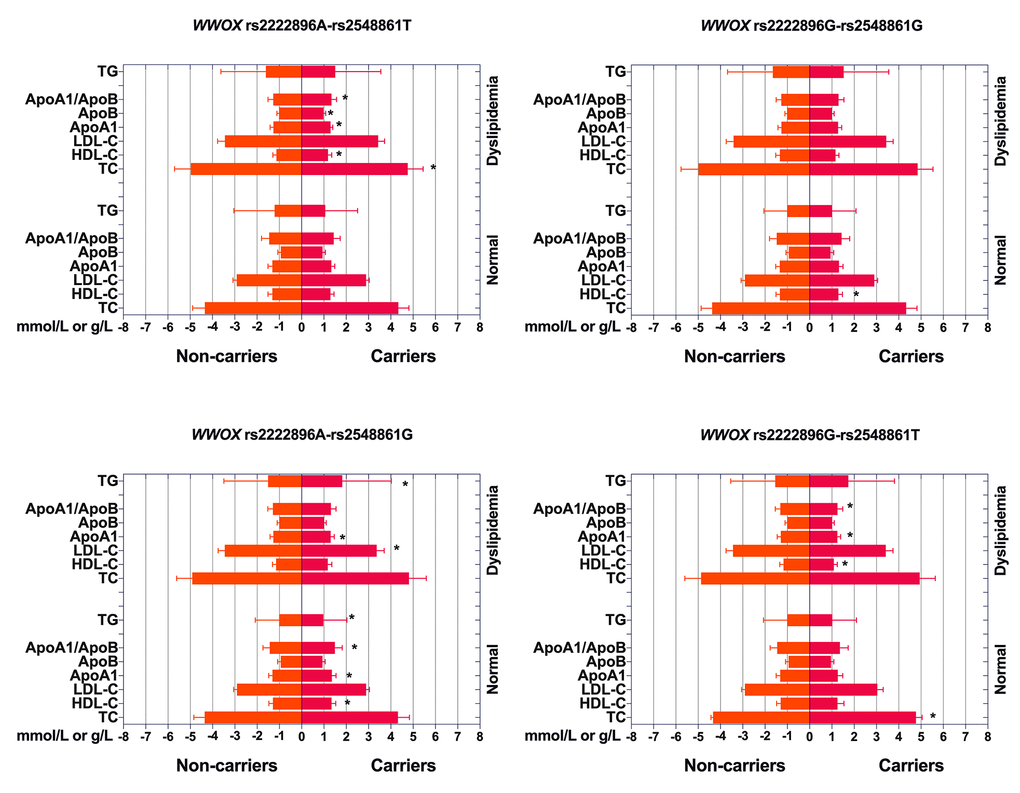
Figure 3. Associations between WWOX haplotypes, serum lipid levels, and dyslipidemia. *P-value < 0.006 indicated a statistically significant difference after Bonferroni correction.
As shown in Table 7, after adjusting for sex, age, BMI, FBS, smoking, and drinking in the normal group, the rs2222896G-rs2548861T haplotype was associated with increased levels of serum TC and LDL-C (P < 0.001), the rs2222896A-rs2548861G haplotype was associated with increased serum HDL-C and ApoA1 levels and ApoA1/ApoB ratio, but decreased serum TG levels (P ≤ 0.006), and the rs2222896G-rs2548861G haplotype was associated with decreased serum HDL-C levels (P < 0.001). In the dyslipidemia group, the rs2222896A-rs2548861T haplotype was associated with decreased serum TC and ApoB levels and increased serum HDL-C and ApoA1 levels and ApoA1/ApoB ratio (P < 0.001), the rs2222896A-rs2548861G haplotype was associated with decreased serum LDL-C levels and increased serum ApoA1 (P ≤ 0.006) and TG levels (P < 0.001), and the rs2222896G-rs2548861T haplotype was associated with decreased serum ApoA1 and HDL-C levels and ApoA1/ApoB ratio (P < 0.001).
Table 7. Association between WWOX haplotypes and serum lipid levels in normal and dyslipidemia groups.
| Lipid | Haplotype | Subjects | Beta | t | P | 95%CI of Beta | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower | Upper | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Normal | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC | rs2222896G–rs2548861T | carriers/non-carriers | 0.128 | 3.765 | 0.000 | 0.212 | 0.673 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG | rs2222896A–rs2548861G | carriers/non-carriers | -0.167 | -4.912 | 0.000 | -0.085 | -0.036 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL-C | rs2222896A–rs2548861G | carriers/non-carriers | 0.151 | 4.414 | 0.000 | 0.034 | 0.089 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896G–rs2548861G | carriers/non-carriers | -0.124 | -3.610 | 0.000 | -0.074 | -0.022 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C | rs2222896G–rs2548861T | carriers/non-carriers | 0.136 | 4.012 | 0.000 | 0.074 | 0.217 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1 | rs2222896A–rs2548861G | carriers/non-carriers | 0.109 | 3.163 | 0.002 | 0.017 | 0.071 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1/ApoB | rs2222896A–rs2548861G | carriers/non-carriers | 0.095 | 2.772 | 0.006 | 0.020 | 0.116 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dyslipidemia | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC | rs2222896A–rs2548861T | carriers/non-carriers | -0.138 | -4.632 | 0.000 | -0.290 | -0.118 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896G–rs2548861G | carriers/non-carriers | -0.083 | -2.796 | 0.005 | -0.233 | -0.041 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG | rs2222896A–rs2548861G | carriers/non-carriers | 0.126 | 4.207 | 0.000 | 0.093 | 0.256 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL-C | rs2222896A–rs2548861T | carriers/non-carriers | 0.136 | 4.530 | 0.000 | 0.026 | 0.067 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896G–rs2548861T | carriers/non-carriers | -0.193 | -6.556 | 0.000 | -0.099 | -0.053 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C | rs2222896A–rs2548861G | carriers/non-carriers | -0.120 | -4.006 | 0.000 | -0.135 | -0.046 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1 | rs2222896A–rs2548861T | carriers/non-carriers | 0.121 | 4.053 | 0.000 | 0.020 | 0.058 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896A–rs2548861G | carriers/non-carriers | 0.083 | 2.773 | 0.006 | 0.009 | 0.054 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896G–rs2548861T | carriers/non-carriers | -0.117 | -3.931 | 0.000 | -0.064 | -0.022 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoB | rs2222896A–rs2548861T | carriers/non-carriers | -0.117 | -3.913 | 0.000 | -0.036 | -0.012 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ApoA1/ApoB | rs2222896A–rs2548861T | carriers/non-carriers | 0.142 | 4.769 | 0.000 | 0.040 | 0.097 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896G–rs2548861T | carriers/non-carriers | -0.106 | -3.570 | 0.000 | -0.091 | -0.026 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TC, total cholesterol; TG, triglyceride; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; ApoA1, apolipoprotein A1; ApoB, apolipoprotein B; CI, confidence interval. P values were calculated by multivariable linear regression. P < 0.0125 indicated a statistically significant difference after Bonferroni correction and adjusting for genotype, age, gender, drinking, smoking, fasting blood-glucose, and body mass index. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Interactions between the four haplotypes and environmental factors on dyslipidemia are shown in Figure 4. Dyslipidemia morbidity was increased in those with the rs2222896G-rs2548861G and rs2222896G-rs2548861T haplotypes compared to those without corresponding haplotypes (OR = 1.44, 95% CI = 1.18−1.76, P < 0.001 and OR = 15.08, 95% CI = 9.24−24.60, P < 0.001, respectively); environmental factors such as BMI > 24 kg/m2 and FBS ≥ 7.0 mM also increased dyslipidemia morbidity. In contrast, dyslipidemia morbidity was decreased in those with the rs2222896A-rs2548861T and rs2222896A-rs2548861G haplotypes compared to those without corresponding haplotypes (OR = 0.47, 95% CI = 0.39−0.56, P < 0.001 and OR = 0.70, 95% CI = 0.56−0.86, P < 0.001, respectively). Interactions between haplotype and environmental factors had different effects on the incidence of dyslipidemia than those observed for each individual locus. Interactions between the rs2222896G-rs2548861G and rs2222896G-rs2548861T haplotypes and smoking, high BMI, or FBS increased the risk of dyslipidemia, as did the interaction between the rs2222896G-rs2548861T haplotype and sex or age (P < 0.05 for all). Furthermore, interactions between the rs2222896A-rs2548861T haplotype and sex, age, BMI, drinking, or smoking, and between the rs2222896G-rs2548861T haplotype and BMI or FBS decreased the effects of the environmental factors on dyslipidemia (P < 0.05 for all).
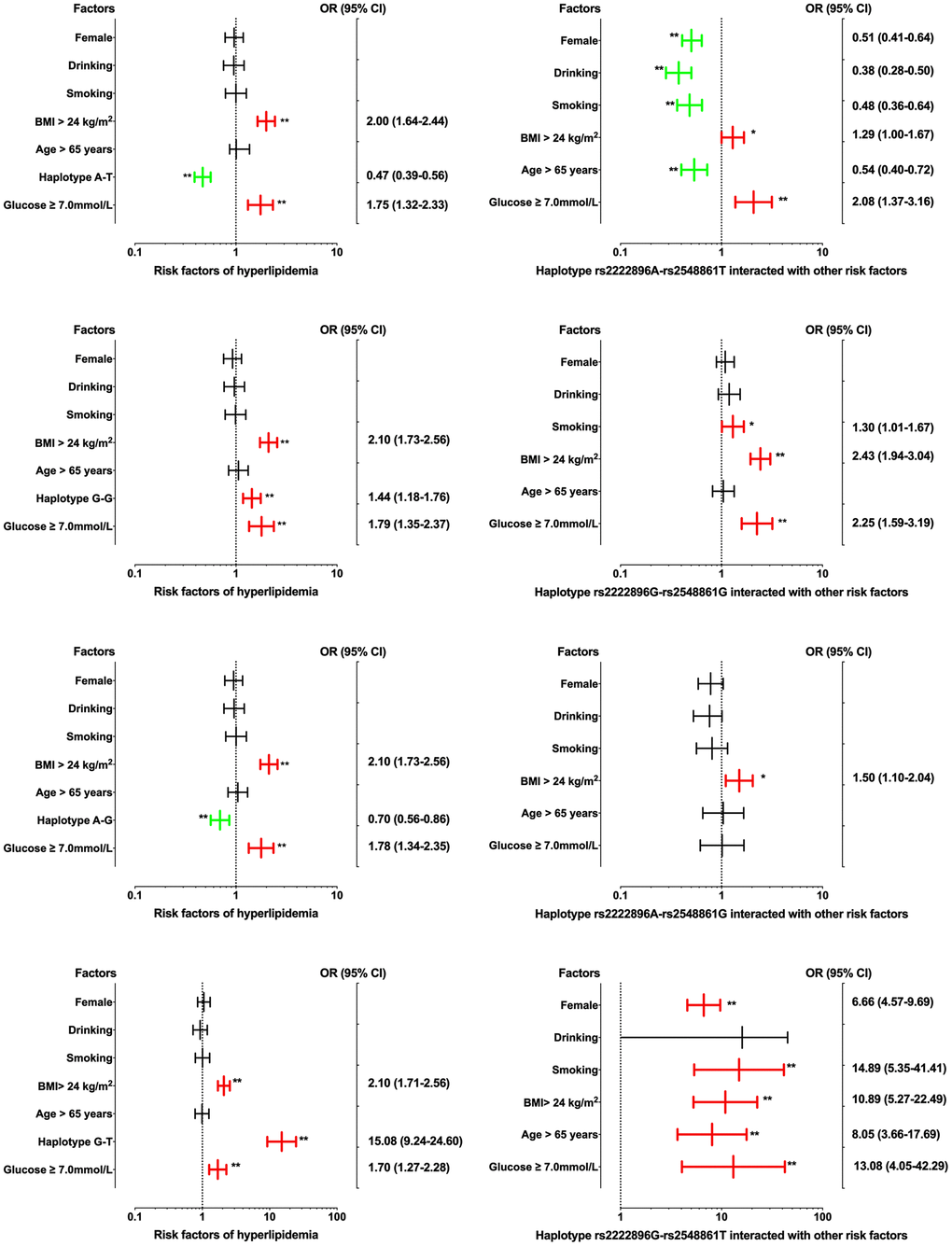
Figure 4. Associations between WWOX haplotypes, environmental factors, and their interactions on dyslipidemia. *P-value < 0.05 and **P-value < 0.001.
Associations between G × G interactions and dyslipidemia
As shown in Table 8, thirteen TUBB-WWOX G × G interactions were observed in the normal and dyslipidemia groups (LEF for gene interactions > 0.03). Frequencies of nine of these G × G interactions, namely G-C-A-T, G-C-A-G, G-C-G-T, T-T-A-G, T-T-G-T, T-T-G-G, T-C-G-G, T-T-A-T, and T-C-G-T, differed significantly between the two groups (P < 0.05). Among them, only the T-T-G-T interaction was associated with increased incidence of dyslipidemia (OR = 1.371, 95% CI = 1.112–1.689, P = 0.003), while the other eight interactions, particularly T-T-G-G, were associated with reduced incidence of dyslipidemia (OR = 0.795, 95% CI = 0.640–0.988, P = 0.038).
Table 8. Frequencies of gene-gene interactions in the normal and dyslipidemia groups [n (frequency)].
| Label | Gene-gene interaction | Normal | Dyslipidemia | χ2 | P | OR (95% CI) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G1 | G2 | G3 | G4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 1 | G | C | A | T | 412 | (0.238) | 260 | (0.115) | 128.782 | 0.000 | 0.328(0.217–0.398) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 2 | G | C | A | G | 489 | (0.283) | 449 | (0.199) | 55.068 | 0.000 | 0.506(0.423–0.606) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 3 | G | C | G | T | 375 | (0.217) | 388 | (0.172) | 16.853 | 0.000 | 0.683(0.569–0.819) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 4 | G | C | G | G | 479 | (0.277) | 612 | (0.271) | 0.300 | 0.584 | 0.951(0.796–1.137) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 5 | G | T | A | T | 357 | (0.207) | 388 | (0.172) | 1.800 | 0.180 | 0.884(0.373–1.059) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 6 | G | T | G | G | 523 | (0.303) | 634 | (0.281) | 3.847 | 0.050 | 0.835(0.697–1.000) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 7 | T | T | A | G | 185 | (0.107) | 196 | (0.087) | 5.180 | 0.023 | 0.771(0.616–0.965) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 8 | T | T | G | T | 185 | (0.107) | 307 | (0.136) | 8.761 | 0.003 | 1.371(1.112–1.689) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 9 | T | T | G | G | 199 | (0.115) | 217 | (0.096) | 4.926 | 0.038 | 0.795(0.640–0.988) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 10 | G | T | A | G | 470 | (0.272) | 576 | (0.255) | 2.241 | 0.134 | 0.873(0.731–1.043) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 11 | T | C | G | G | 257 | (0.149) | 108 | (0.048) | 121.925 | 0.000 | 0.250(0.195–0.320) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 12 | T | T | A | T | 234 | (0.135) | 180 | (0.080) | 36.219 | 0.000 | 0.511(0.410–0.636) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I 13 | T | C | G | T | 211 | (0.122) | 102 | (0.045) | 81.633 | 0.000 | 0.307(0.238–0.397) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G1, TUBB rs3132584 G>T; G2, TUBB rs3130685 C>T; G3, WWOX rs2222896 A>G; G4, WWOX rs2548861 T>G; I, interaction. Gene interaction frequencies < 0.03 in the two groups were excluded. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Models of the effects of different interactions on dyslipidemia
Using generalized multifactor dimensionality reduction (GMDR) analysis and adjusting by covariates, we identified the fourteen best interactive models of SNP-SNP, haplotype-haplotype, G × G, and G × E effects on dyslipidemia (Table 9). The two- to four-locus interactive models for SNP-SNP and SNP-environment were rs2222896-rs3130685, rs2222896-rs3130685-rs2548861, rs2222896-rs3130685-rs2548861-rs3132584, rs2222896-rs3130685-age > 65 years, rs2222896-rs3132584-FBS ≥ 7.0 mM, and rs2222896-rs3132584-drinking (cross-validation (CV) constancy of 10 of 10, balanced accuracy test (Bal. Acc.) ≥ 64.98%, and permutation test P < 0.001 for all). The two- to three-locus interactive models for haplotype-haplotype and haplotype-environment were H1-H3 (interaction of rs2222896A-rs2548861T and rs2222896A-rs2548861G haplotypes), H2-H3-H4 (interaction of rs2222896G-rs2548861G, rs2222896A-rs2548861G, and rs2222896G-rs2548861T haplotypes), H1-H3-BMI > 24 kg/m2, and H1-H3-FBS ≥ 7.0 mM (CV constancy of 10 of 10, Bal. Acc. ≥ 62.05%, and permutation test P < 0.001 for all). Additionally, the two- to three-locus models of G × G and G × E interactions were I1-I2 (G-C-A-T and G-C-A-G), I3-I12 (G-C-G-T and T-T-A-T), I4-I8-I11 (G-C-G-G, T-T-G-T, and T-C-G-G), I4-I5-I10 (G-C-G-G, G-T-A-T, and G-T-A-G), and I1-I11-BMI > 24 kg/m2 (CV constancy of 10 of 10, Bal. Acc. ≥ 56.87%, and permutation test P < 0.001 for all).
Table 9. Best models for different types of interactions.
| Interactive model | Training Bal. Acc. | Testing Bal. Acc. | CV consistency | Sign test P | Permutation test P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP-SNP | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A-B | 0.7189 | 0.7208 | 10/10 | 0.001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A-B-C | 0.8061 | 0.8043 | 10/10 | 0.001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A-B-C-D | 0.8434 | 0.8290 | 10/10 | 0.001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP-environment | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A-B-F | 0.7189 | 0.7208 | 10/10 | 0.001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A-D-H | 0.6492 | 0.6509 | 10/10 | 0.001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A-D-J | 0.6483 | 0.6498 | 10/10 | 0.001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haplotype–haplotype | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1-H3 | 0.6176 | 0.6176 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haplotype-environment | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1-H3-G | 0.6469 | 0.6469 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1-H3-H | 0.6381 | 0.6369 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gene-gene | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I1-I2 | 0.6233 | 0.6233 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I3-I12 | 0.5687 | 0.5687 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I4-I8-I11 | 0.6950 | 0.6950 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I4-I5-I10 | 0.6488 | 0.6488 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gene-environment | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I1-I11-G | 0.6302 | 0.6268 | 10/10 | <0.0001 | <0.0001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| A = rs2222896, B = rs3130685, C = rs2548861, D = rs3132584, E = female, F = age > 65 years, G = body mass index > 24 kg/m2, H = fasting blood-glucose ≥ 7.0 mM, I = smoking, J = drinking; Bal. Acc., balanced accuracy; CV, cross-validation. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Furthermore, associations between the fourteen interactions and dyslipidemia were further verified through logistic regression analysis as shown in Figure 5 and Table 10. The following interactions between genotypes, haplotypes, and factors increased the risk of dyslipidemia: rs3130685TT-rs2548861TG+TT (OR = 3.099, 95% CI = 2.397–4.005, P < 0.001) and rs3130685CT+CC-rs2548861GG (OR = 3.136, 95% CI = 2.489–3.952, P < 0.001) compared to rs3130685TT-rs2548861GG; rs3130685TT-rs2222896AG+GG (OR = 2.532, 95% CI = 1.982–3.263, P < 0.001) and rs3132584GT+GG-rs2222896AG+GG (OR = 2.548, 95% CI = 2.035–3.189, P < 0.001) compared to rs3130685TT-rs2222896AA; rs2222896AG+GG-nondrinking compared to rs2222896AA-nondrinking (OR = 1.792, 95% CI = 1.490–2.155, P < 0.001); H1-H3 compared to H1–H3 non-carriers (OR = 3.315, 95% CI = 2.681–4.098, P < 0.001); I4 non-carriers-I10 carriers (OR = 2.295, 95% CI = 1.783–2.954, P < 0.001) and I4 carriers-I10 non-carriers (OR = 2.877, 95% CI = 2.253–3.675, P < 0.001) compared to I4–I10 non-carriers; and I4 non-carriers-I8 carriers (OR = 5.292, 95% CI = 3.694–7.582, P < 0.001) and I4 carriers-I8 non-carriers (OR = 1.465, 95% CI = 1.219–1.760, P < 0.001) compared to I4–I8 non-carriers. In contrast, interactions between the following genotypes, phenotypes, and factors decreased the risk of dyslipidemia: rs3132584GT+GG-rs2222896AA compared to rs3132584TT-rs2222896AA (OR = 0.441, 95% CI = 0.345–0.563, P < 0.001); rs2222896AA-drinking compared to rs2222896AA-nondrinking (OR = 0.298, 95% CI = 0.196–0.455, P < 0.001); and I1-I1 carriers compared to I1-I11 non-carriers (OR = 0.114, 95% CI = 0.097–0.164, P < 0.001).
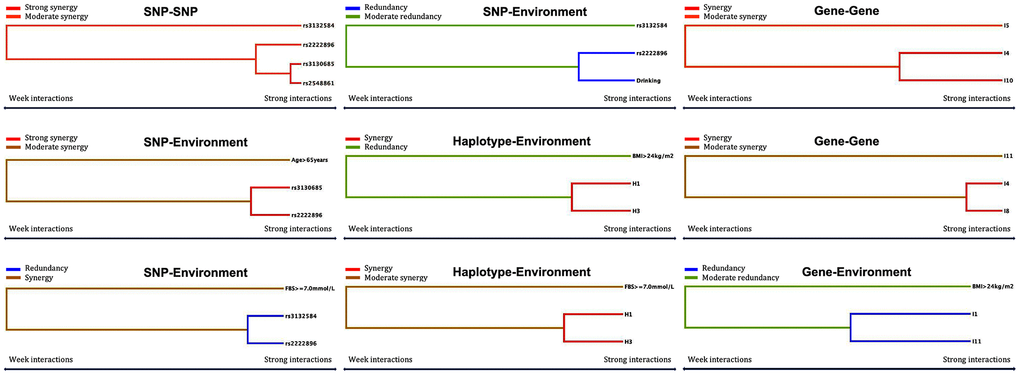
Figure 5. Optimal interactions affecting dyslipidemia shown in a dendrogram. Elements that interact strongly with each other appear close together in the leaves of the tree, while elements that interact weakly appear far from each other.
Table 10. Risk of dyslipidemia with different types of interactions.
| Variable 1 | Variable 2 | OR (95% CI) | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP-SNP interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3130685 | rs2548861 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | GG | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | TG+TT | 3.099(2.397–4.005) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT+CC | GG | 3.136(2.489–3.952) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT+CC | TG+TT | 0.588(0.489–0.706) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3130685 | rs2222896 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | AA | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | AG+GG | 2.532(1.982–3.236) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT+CC | AA | 1.317(0.929–1.867) | 0.123 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT+CC | AG+GG | 1.236(1.027–1.486) | 0.025 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3132584 | rs2222896 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | AA | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | AG+GG | 1.526×109(0.000–NA) | 0.998 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GT+GG | AA | 0.441(0.345–0.563) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GT+GG | AG+GG | 2.548(2.035–3.189) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP-environment interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896 | Drinking | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AA | No | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AA | Yes | 0.298(0.196–0.455) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AG+GG | No | 1.792(1.490–2.155) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AG+GG | Yes | 1.344(1.077–1.678) | 0.009 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haplotype-haplotype interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1 | H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non–carriers | 3.315(2.681–4.098) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Carriers | 0.704(0.563–0.881) | 0.002 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Non–carriers | 0.485(0.403–0.583) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gene-gene interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I1 | I11 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non-carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Carriers | 1.271(0.862–1.875) | 0.226 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Non-carriers | 0.808(0.648–1.006) | 0.057 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 0.114(0.079–0.164) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I4 | I8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non-carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Carriers | 5.292(3.694–7.582) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Non-carriers | 1.465(1.219–1.760) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 0.394(0.294–0.529) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I4 | I10 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non-carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Carriers | 2.295(1.783–2.954) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Non-carriers | 2.877(2.253–3.675) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 0.483(0.399–0.586) | 0.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1, rs2222896A-rs2548861T haplotype; H3, rs2222896A-rs2548861G haplotype; I1, rs3132584G-rs3130685C-rs2222896A-rs2548861T; I4, rs3132584G-rs3130685C-rs2222896G-rs2548861G; I8, rs3132584T-rs3130685T-rs2222896G-rs2548861T; I10, rs3132584G-rs3130685T-rs2222896A-rs2548861G; I11, rs3132584T-rs3130685C-rs2222896G-rs2548861G; OR, odds ratio; CI, confidence interval. Different types of interactions were analyzed by logistic regression. P < 0.006 indicated a statistically significant difference after Bonferroni correction and adjusting for body mass index and fasting blood-glucose. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Risk of hypertension based on different interactive models of dyslipidemia
The effects of traditional and potential genetic risks associated with dyslipidemia on hypertension are shown in Tables 11, 12. In this study, dyslipidemia, female sex, age > 65 years, BMI > 24 kg/m2, and FBS ≥ 7.0 mM were clear risk factors for hypertension (P < 0.05). Moreover, interactions between the rs3132584 and rs2222896 SNPs increased the risk of hypertension 1.523-fold (P < 0.006) after adjustment for BMI, sex, age, and FBS.
Table 11. Risk of hypertension in the study population [n(frequency)].
| Risk factor | Normal (n) | Hypertension (n) | χ2 | P | OR (95% CI) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 1017(0.510) | 976(0.490) | 3.936 | 0.047 | 1.235(1.003–1.522) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 1442(0.724) | 551(0.276) | 0.133 | 0.715 | 0.956(0.751–1.217) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking | 1448(0.727) | 545(0.273) | 1.303 | 0.254 | 1.149(0.905–1.457) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dyslipidemia | 864(0.434) | 1129(0.566) | 42.037 | 0.000 | 1.884(1.556–2.282) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age > 65 years | 1544(0.775) | 449(0.225) | 55.483 | 0.000 | 2.369(1.888–2.927) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI > 24 kg/m2 | 1293(0.649) | 700(0.351) | 25.641 | 0.000 | 1.660(1.364–2.020) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FBS ≥ 7.0 mM | 1725(0.866) | 268(0.134) | 29.408 | 0.000 | 2.139(1.625–2.816) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI, body mass index; FBS, fasting blood-glucose; OR, odds ratio; CI, confidence interval. P < 0.05 indicated a statistically significant difference. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 12. Effects of meaningful interactive models of dyslipidemia on hypertension.
| Variable 1 | Variable 2 | OR (95% CI) | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP-SNP interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3130685 | rs2548861 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | GG | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT+CC | TG+TT | 0.936(0.771–1.137) | 0.506 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3130685 | rs2222896 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | AA | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CT+CC | AG+GG | 0.895(0.739–1.085) | 0.259 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs3132584 | rs2222896 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | AA | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GT+GG | AG+GG | 1.523(1.189–1.950) | 0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SNP-environment interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| rs2222896 | Drinking | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AA | No | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AG+GG | Yes | 1.264(0.997–1.603) | 0.053 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haplotype-haplotype interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1 | H3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non-carriers | 0.917(0.525–1.601) | 0.761 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gene-gene interaction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I1 | I11 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non-carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 0.448(0.202–0.993) | 0.048 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I4 | I8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non-carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 0.819(0.417–1.609) | 0.562 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I4 | I10 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-carriers | Non-carriers | 1 | – | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carriers | Carriers | 0.650(0.411–1.026) | 0.064 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H1, rs2222896A-rs2548861T haplotype; H3, rs2222896A-rs2548861G haplotype; I1, rs3132584G-rs3130685C-rs2222896A-rs2548861T; I4, rs3132584G-rs3130685C-rs2222896Grs2548861G; I8, rs3132584T-rs3130685T-rs2222896G-rs2548861T; I10, rs3132584G-rs3130685T-rs2222896A-rs2548861G; I11, rs3132584T-rs3130685C-rs2222896G-rs2548861G; OR, odds ratio; CI, confidence interval. Different types of interactions were analyzed by logistic regression. P < 0.006 indicated a statistically significant difference after Bonferroni correction and adjusting for body mass index and fasting blood-glucose. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
Dyslipidemia, a multifactorial non-communicable chronic disease, contributes to pathogenesis of arteriosclerotic diseases and is characterized by high TC, TG, and LDL-C and low HDL-C levels [6]. LDL-C and other ApoB-rich lipoproteins accumulate in the arterial wall, playing a key role in atherosclerotic plaque formation and subsequent related cardiovascular events [19]. Studies have shown that the risk of developing carotid plaque increases by 62% for every 1 mM increase in serum LDL-C concentration [20]. Lipid levels in the plasma are influenced by many factors, and genetic variation in lipid-related genes leads to diverse phenotypes in different countries and ethnicities. Previous GWASs demonstrated associations between the TUBB and WWOX genes and LDL-C and HDL-C levels, and differences in the expression of these two genes in different populations contributes to diversity in lipid traits. According to recently updated data from 1000Genomes (http://www.ncbi.nlm.nih.gov/snp/), alternate allele frequencies in American, European, East Asian, and African populations, respectively, are as follows: TUBB rs3132584T, 0.1800, 0.1769, 0.1796, and 0.4803; TUBB rs3130685T, 0.5320, 0.4632, 0.5367, and 0.5479; WWOX rs2222896G, 0.5750, 0.6252, 0.6131, and 0.8313; and WWOX rs2548861G, 0.6341, 0.5656, 0.6282, and 0.2829. In this study, the frequencies of these allelic mutations in the Maonan population differed significantly from their frequencies in the CHB population: frequencies of the rs3132584T, rs3130685T, rs2222896G, and rs2548861G alleles in the Maonan and the CHB populations, respectively, were 0.2168 vs. 0.1829, 0.5730 vs. 0.5444, 0.5607 vs. 0.6341, and 0.6184 vs. 0.5488. Moreover, the frequency of the mutant homozygous TT genotype at rs3132584 was 0.0471 in the Maonan population but 0 in the CHB population, further demonstrating the differences in gene distribution between these two ethnic groups. Furthermore, although genotypic frequencies for rs3132584, rs3130685, and rs2222896 in the Maonan population differed significantly between the normal and dyslipidemia groups, only rs2222896 genotype was correlated with various serum lipid parameters. Normal group subjects with the rs2222896GG genotype had higher LDL-C and lower HDL-C and ApoA1 concentrations than those with other genotypes. This association between rs2222896 and serum LDL-C level was consistent with a previous study [21]. Additionally, Yamada et al. identified two novel loci in TUBB, rs3132584 and rs3130685, that were associated with high LDL-C in an exome-wide association study of early-onset dyslipidemia [13]. Another study of 2911 Japanese subjects revealed that the reference alleles rs3132584A and rs3130685C were associated with high LDL-C (P < 0.0001). In this study, we found that a rs3132584 mutation from G to T was present at a higher frequency. In the dyslipidemia group, rs3132584G was associated with high LDL-C levels; however, we observed little association between rs3130685C and LDL-C concentration. Two previous studies [14, 22] indicated that the rs2548861 SNP in WWOX was associated with HDL-C concentration in the Young Finns Study population (n = 6,728, P = 6.9 × 10-7), but another [23] failed to replicate this result in the Spanish population (n = 801). In our study, an association between rs2548861 and HDL-C concentration was observed in the dyslipidemia group (n = 1129, P = 0.001) but not in the normal group (n = 864, P = 0.597). We also found that HDL-C concentration in the dyslipidemia group might be affected by other non-genetic factors. Thus, we speculated that mutations at rs2548861 were not correlated with HDL-C concentration in these subjects. A correlation analysis between genetic models and disease risk was then conducted to understand the relationship between alleles at these four SNP sites and dyslipidemia. We found that codominant, dominant, recessive, and log-additive models of the rs3132584, rs3130685, and rs2222896 SNPs identified significant differences in dyslipidemia. Using the log-additive models, we identified the direction of the associations between each minor allele frequency (MAF) of these three SNPs and dyslipidemia. The rs3132584T and rs2222896A alleles were potential protective factors, while rs3130685C was a risk factor, for dyslipidemia. Various lipid-related SNPs and the frequencies of different alleles in the TUBB and WWOX genes can therefore lead to different clinical lipid phenotypes in different populations.
Genetic mechanisms are influenced by many types of interactions [24–26], including G × G and G × E interactions, which can have synergistic or contrasting effects on gene expression [27, 28]. Interactions among genes can include SNP-SNP interactions between individual genes, haplotype-haplotype interactions, and the combined effects of mutations in genes on different chromosomes [29, 30]. Detailed information on the four target SNPs was obtained from the 1000 Genome database. It indicated that there was a large gap between rs3132584 and rs3130685 at chromosomal positions 30720650 and 31238429, respectively, that prevented LD between them. In contrast, rs2222896 and rs2548861 at chromosomal positions 78058601 and 78624496, respectively, were about 500 kb from each other, resulting in a weak LD between them. Of the four common haplotypes observed for rs2222896 and rs2548861 alleles, rs2222896A-rs2548861T and rs2222896A-rs2548861G were associated with reduced risk of dyslipidemia while rs2222896G-rs2548861G and rs2222896G-rs2548861T were associated with elevated risk for dyslipidemia. In addition, multi-site mutations in TUBB and WWOX altered the incidence of dyslipidemia in the Maonan population. Thirteen types of multi-site mutation combinations for the two genes were screened using SHEsisPlus online software (http://shesisplus.bio-x.cn/SHEsis.html). Among these 13 combinations, rs3132584T-rs3130685T-rs2222896G-rs2548861T increased the risk of dyslipidemia 1.371-fold; all of the remaining 12 combinations could potentially decrease the risk of dyslipidemia. Although TUBB and WWOX are located on two different chromosomes, interaction models were successfully screened using the GMDR method (https://sourceforge.net/projects/gmdr/), and the effects of G × G and G × E interactions on dyslipidemia were examined further. The fourteen highest-performing models of interactions linked to dyslipidemia were identified; all had a 10 of 10 CV, training and testing balanced accuracy values greater than 0.5, and sign and permutation test P values less than 0.05. Furthermore, an intuitive interactive dendrogram and logistic regression analysis indicated that the rs3130685-rs2548861, rs3130685-rs2222896, H1-H3, I4-I8, and I4-110 interactions exhibited strong synergistic effects increasing the risk of dyslipidemia, while the rs3132584-rs2222896 and I1-I11 interactions decreased the risk of dyslipidemia. This study therefore demonstrated that WWOX haplotype and the combined effects of TUBB × WWOX have larger impacts on serum lipid levels and dyslipidemia than single SNPs in those genes [31]; additional experiments with larger samples sizes are needed to confirm this finding.
Many environmental factors, including aging, sex, unhealthy lifestyle (high-fat diet [32, 33], smoking, drinking), high blood glucose, and weight and obesity, are directly or indirectly involved in the pathogenesis of dyslipidemia. Moreover, these environmental factors can interact with lipid-related genes to alter lipid expression profiles [34, 35]. Several studies demonstrate that the incidence of hyperlipidemia is higher in elderly people (1259/1657, 76%, mean age 69 years) than in young people (203/494, 41%, mean age 29 years); this difference was attributed to age-associated declines in ribosome coverage in the vicinity of start codons and increases near stop codons, alterations in expression of genes associated with lipid metabolism, loss of hepatic LDL receptors, and decreases in sex hormone levels [36, 37]. Furthermore, the risk of dyslipidemia was higher in postmenopausal women and elderly men [38, 39]; food pickling processes can increase concentrations of secondary metabolites in lemons and onions, thus endangering health [40]; and heavy (8 or more drinks per week for women or 15 or more drinks per week for men) rather than low or moderate alcohol consumption was also linked to dangerous increases in lipid levels [41]. The Maonan ethnic group is located mainly in the Huanjiang region, which is known as the “hometown of cattle and grain” due to the prevalence of agriculture and beef cattle industries. Beef, duck, half-cooked chicken, animal offal, and pickled sour pork, snails, and vegetables, are common dishes in this region. In addition, consumption of wine made from rice, corn, sweet potatoes, pumpkins, etc., three times per day with meals and smoking cigarettes made from local dried tobacco leaves is common among Maonan men. Many high-risk environmental factors may therefore affect the Maonan population. We used random stratified sampling in our examination of dyslipidemia risk in this population to eliminate systematic differences in age and gender structure between the two groups. We found no differences in either proportion of subjects > 65 years old or severity of smoking between subgroups, but severity of alcohol consumption, BMI > 24 kg/m2, and FBS ≥ 7.0 mM were significantly higher in the dyslipidemia group than in the normal group, and these factors interacted with the TUBB and WWOX SNPs to affect the prevalence of dyslipidemia. Pairwise multiple regression interaction models and optimal interaction models from GMDR revealed that the rs2222896G-rs2548861G haplotype interacted with smoking, overweight/obesity, or FBS ≥ 7.0 mM, while the rs2222896G-rs2548861T haplotype interacted with old age, female sex, smoking, FBS ≥ 7.0 mM, or overweight status/obesity to exacerbate the risk of dyslipidemia. In addition, age > 65 years increased the synergistic effect of rs2222896-rs3130685, and FBS ≥ 7.0 mM increased the synergistic effects of H1-H3 or rs2222896-rs3132584 on dyslipidemia. In contrast, interactions between the rs2222896A-rs2548861T haplotype and female sex, old age, smoking, and drinking, and between the rs2222896A-rs2548861G haplotype and overweight status/obesity or FBS ≥ 7.0 mM, decreased the risk of dyslipidemia. Finally, overweight status/obesity decreased the protective effects of H1-H3 or I1-I11 interactions against dyslipidemia, and drinking weakened the increased risk of dyslipidemia associated with rs2222896, possibly because most subjects were low-moderate drinkers.
Blood pressure is also modified by multiple genetic and environmental factors and their interactions [42–44], and dyslipidemia is a common independent risk for hypertension [45]. We therefore further explored differences in the incidence of hypertension between the two groups and found that the prevalence of hypertension was associated with gender, age, BMI, FBS, smoking, drinking, and dyslipidemia. Interestingly, we also found that the rs3132584 and rs2222896 SNP interaction that affected risk of dyslipidemia also significantly increased the risk of hypertension; this might be a novel explanation of how dyslipidemia contributes to the pathogenesis of hypertension.
Some important strengths and weaknesses of this study should be considered when interpreting the results. Notable strengths include the following: (1) identification of differences in TUBB and WWOX mutation frequencies between the Maonan ethnic group and the CHB population provides novel data for human genomic studies; (2) detection of TUBB-WWOX interactions and G × E interactions that affected the prevalence of dyslipidemia might provide new therapeutic targets for dyslipidemia; and (3) this study characterized an integrated effect of the TUBB rs3132584 and WWOX rs2222896 SNPs on dyslipidemia that could also predict the risk of hypertension, identifying a possible novel pathogenic mechanism by which dyslipidemia can lead to hypertension. However, the following limitations should also be considered: (1) this study did not include other variable risk factors, such as daily exercise habits and anti-hyperlipidemia interventions; (2) despite the unique diet (high salt and sour, high fat and alcohol) associated with the Maonan population, intake of these foods was not accurately characterized and their effects on dyslipidemia could not be examined; (3) larger sample sizes are needed to verify the results of this study; and (4) biological function studies are needed to further examine the effects of interactions between these two genes on dyslipidemia.
In summary, this study identified associations between TUBB, WWOX, environmental factors, serum lipid levels, and dyslipidemia in the Maonan population. Our findings revealed that, compared to single-locus effects, TUBB-WWOX-environment interactions can result in synergistic or contrasting effects on incidence of dyslipidemia, thereby increasing or reducing the risk of dyslipidemia.
Materials and Methods
Participants
A total of 1993 unrelated subjects were selected from our previous sample library in 2015 through stratified random sampling [46]; all subjects were from three generations of the Maonan ethic group and lived in the Maonan Autonomous County of Huanjiang in Southern China. Subjects were separated into normal lipid and dyslipidemia groups with similar age and gender distributions. The mean age of the 864 normal group subjects was 55.31 ± 13.08 years; 431 (49.88%) were males and 433 (50.12%) were females. The mean age of the 1129 dyslipidemia group subjects was 56.04 ± 12.88 years; 545 (48.27%) were males and 584 (51.73%) were females. All subjects were generally healthy without a history of diabetes, thyroid disease, autoimmune disease, cardiovascular disease, stroke, renal disease, tumor, or mental illness. The participants did not take medicine or eat foods with known lipid-lowering properties. The study was reviewed and approved by the Ethics Committee of the First Affiliated Hospital, Guangxi Medical University (No. Lunshen 2014-KY-Guoji-001; March 7, 2014). Subjects who met inclusion criteria and signed informed consent were enrolled after professional investigators explained the experimental rules.
Laboratory values
Serum lipid levels were measured in peripheral blood (3 mL) collected in the morning after fasting for more than 8 hours using an autoanalyzer (type 7170A; Hitachi Ltd., Tokyo, Japan). Serum TC, TG, HDL-C, and LDL-C levels were determined using commercially available enzymatic assay kits, and serum ApoA1 and ApoB levels were determined by turbidimetric immuno-assay [46–48].
Variable definitions
Dyslipidemia is defined by elevated levels of TC, LDL-C, and TG and decreased HDL-C levels [6]. In this study, normal ranges for serum TC, TG, HDL-C, LDL-C, ApoA1, and ApoB levels and ApoA1/ApoB ratio were 3.10−5.17 mM, 0.56−1.70 mM, 0.90−1.81 mM, 2.70−3.10 mM, 1.00−1.78 g/L, 0.63−1.14 g/L, and 1.00−2.50, respectively. Dyslipidemia was therefore defined by any of the following either alone or in combination: TC > 5.17 mM, TG > 1.7 mM, LDL-C > 3.10 mM, and/or HDL-C < 0.9 mM [49]. Blood pressure was measured after the subject had been sitting for at least 5 minutes. An average SBP ≥ 140 mmHg and/or DBP ≥ 90 mmHg for three measurements over two days was defined as hypertension [50]. BMI was determined by dividing weight (kilograms) by height (meters squared); overweight and obese categories in China [51] are defined as BMI > 24 kg/m2 and BMI ≥ 28 kg/m2, respectively.
SNP selection
The basic principles of gene and SNP selection were as follows: (1) TUBB and WWOX were selected from previous GWASs examining lipid metabolism. (2) SNPs correlated with lipid levels were selected using Haploview (Broad Institute of MIT and Harvard, Cambridge, MA, USA version 4.2). (3) SNP information was obtained from NCBI dbSNP Build 132 (http://www.ncbi.nlm.nih.gov/snp/). (4) Only SNPs with MAF greater than 10% were included. (5) Correlations between the identified TUBB rs3132584 and rs3130685 SNPs and WWOX rs2222896 and rs2548861 SNPs and blood lipids were confirmed in previous studies [13, 14, 22, 23].
DNA quality control and genotyping
We used a phenol-chloroform method to extract DNA from peripheral blood. DNA was stored at -80° C until 10 μL (10 ng/μL) of each sample was sent to the Department for Next-Generation Sequencing, Sangon Biotech Co., Ltd. (Shanghai, China). The absorbance of the DNA sample was measured at 260 nm and 280 nm using a Shimadzu UV-1601 spectrophotometer. An A260 nm/A280 nm ratio of = 1.8 indicates a pure DNA sample. SNP genotyping was performed on HiSeq XTen sequencers (Illumina, San Diego, CA, USA). The forward and reverse primers for amplification of the four SNPs via polymerase chain reaction were as follows: 5′-AGATCGGTGCCAAGGTAAGAAT-3′ and 5′-CAAACCCGAAGAGCCCTTTTACTA-3′ for rs3132584; 5′-TGTCAAATGCCATTCTTTAAACCTCATGT-3′ and 5′-TTGGGACACTTTACTCCTGAATC-3′ for rs3130685; 5′-AGCTAAAGCCTGTAGACCATCAACATA-3′ and 5′-TCCTCTTTAACACAGGTGAAATGCA-3′ for rs2222896; and 5′-CCAAACCCTAAATAATTGTCTGGATGTT-3′ and 5′-TCTGAATTTCTGCCAAGTTAGATG-3′ for rs2548861.
Statistical analyses
SPSS software, version 25 (SPSS Inc., Chicago, IL, USA) was used to perform statistical analyses. Normally distributed data are presented as means ± standard deviation (SD) and differences between groups were identified using Student’s unpaired t-test and one-way analysis of variance (ANOVA). Non-normally distributed data are shown as interquartile ranges and medians and differences between groups were identified using Mann-Whitney nonparametric tests. Differences in qualitative variables, including different ratios and HWE between the two groups, were analyzed using Chi square tests. Associations between genotypes or haplotypes and continuous serum lipid level data were assessed by multivariable linear regression. Differences in serum lipid levels associated with genotypes and haplotypes were considered statistically significant at P < 0.0125, and G × G or G × E interactions were considered significant at P < 0.006 (corresponding to P < 0.05 after Bonferroni correction for four or eight independent tests). OR values and 95% CIs were calculated by multiple logistic regression after adjustment for stratified risk factors including age, sex, tobacco and alcohol consumption, BMI, and FBS. HWE, genotypic and haplotypic frequencies, and D′ and R2 values used to describe LD were calculated using SHEsisPlus online software. GMDR online software was used to screen optimal SNP-SNP, haplotype-haplotype, G × G, and G × E interaction models. All other data visualizations were generated using GraphPad Prism (version 8.0.0).
Author Contributions
C.-X.L. conceived and designed the study, collected data, performed statistical analyses, and drafted the manuscript. R.-X.Y. improved the design of study, organized the epidemiology investigation, collected samples, and further revised the manuscript. Z.-H.S. collected data and performed statistical analyses. P.-F.Z., G.-X.D., Y.-Z.G., and B.-L.W. collected samples. All authors read and approved the final manuscript.
Acknowledgments
The authors thank all participant of the present study, the research staff of Sangon Biotech Co., Ltd. (Shanghai, China), and the editors and reviewers of the Aging-US journal.
Conflicts of Interest
The authors have no financial or other conflicts of interest to report.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81460169).
References
- 1. Bucholz EM, Rodday AM, Kolor K, Khoury MJ, de Ferranti SD. Prevalence and predictors of cholesterol screening, awareness, and statin treatment among US adults with familial hypercholesterolemia or other forms of severe dyslipidemia (1999-2014). Circulation. 2018; 137:2218–30. https://doi.org/10.1161/CIRCULATIONAHA.117.032321 [PubMed]
- 2. Noubiap JJ, Bigna JJ, Nansseu JR, Nyaga UF, Balti EV, Echouffo-Tcheugui JB, Kengne AP. Prevalence of dyslipidaemia among adults in Africa: a systematic review and meta-analysis. Lancet Glob Health. 2018; 6:e998–1007. https://doi.org/10.1016/S2214-109X(18)30275-4 [PubMed]
- 3. Halcox JP, Banegas JR, Roy C, Dallongeville J, De Backer G, Guallar E, Perk J, Hajage D, Henriksson KM, Borghi C. Prevalence and treatment of atherogenic dyslipidemia in the primary prevention of cardiovascular disease in Europe: EURIKA, a cross-sectional observational study. BMC Cardiovasc Disord. 2017; 17:160. https://doi.org/10.1186/s12872-017-0591-5 [PubMed]
- 4. Poorzand H, Tsarouhas K, Hozhabrossadati SA, Khorrampazhouh N, Bondarsahebi Y, Bacopoulou F, Rezaee R, Jafarzadeh Esfehani R, Morovatdar N. Risk factors of premature coronary artery disease in Iran: a systematic review and meta-analysis. Eur J Clin Invest. 2019; 49:e13124. https://doi.org/10.1111/eci.13124 [PubMed]
- 5. Gai Z, Wang T, Visentin M, Kullak-Ublick GA, Fu X, Wang Z. Lipid accumulation and chronic kidney disease. Nutrients. 2019; 11:722. https://doi.org/10.3390/nu11040722 [PubMed]
- 6. Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, Chapman MJ, De Backer GG, Delgado V, Ference BA, Graham IM, Halliday A, Landmesser U, et al, and ESC Scientific Document Group. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020; 41:111–88. https://doi.org/10.1093/eurheartj/ehz455 [PubMed]
- 7. Jiang Z, Cui Y, Wang L, Zhao Y, Yan S, Chang X. Investigating citrullinated proteins in tumour cell lines. World J Surg Oncol. 2013; 11:260. https://doi.org/10.1186/1477-7819-11-260 [PubMed]
- 8. Sferra A, Petrini S, Bellacchio E, Nicita F, Scibelli F, Dentici ML, Alfieri P, Cestra G, Bertini ES, Zanni G. TUBB variants underlying different phenotypes result in altered vesicle trafficking and microtubule dynamics. Int J Mol Sci. 2020; 21:1385. https://doi.org/10.3390/ijms21041385 [PubMed]
- 9. Yele V, Pindiprolu SK, Sana S, Ramamurty DS, Madasi JR, Vadlamani S. Synthesis and preclinical evaluation of indole triazole conjugates as microtubule targeting agents that are effective against MCF-7 breast cancer cell lines. Anticancer Agents Med Chem. 2020. [Epub ahead of print]. https://doi.org/10.2174/1871520620666200925102940 [PubMed]
- 10. Johannsen J, Kortüm F, Rosenberger G, Bokelmann K, Schirmer MA, Denecke J, Santer R. A novel missense variant in the SDR domain of the WWOX gene leads to complete loss of WWOX protein with early-onset epileptic encephalopathy and severe developmental delay. Neurogenetics. 2018; 19:151–56. https://doi.org/10.1007/s10048-018-0549-5 [PubMed]
- 11. Iatan I, Choi HY, Ruel I, Reddy MV, Kil H, Lee J, Odeh MA, Salah Z, Abu-Remaileh M, Weissglas-Volkov D, Nikkola E, Civelek M, Awan Z, et al. The WWOX gene modulates high-density lipoprotein and lipid metabolism. Circ Cardiovasc Genet. 2014; 7:491–504. https://doi.org/10.1161/CIRCGENETICS.113.000248 [PubMed]
- 12. Khawaled S, Nigita G, Distefano R, Oster S, Suh SS, Smith Y, Khalaileh A, Peng Y, Croce CM, Geiger T, Seewaldt VL, Aqeilan RI. Pleiotropic tumor suppressor functions of WWOX antagonize metastasis. Signal Transduct Target Ther. 2020; 5:43. https://doi.org/10.1038/s41392-020-0136-8 [PubMed]
- 13. Yamada Y, Kato K, Oguri M, Horibe H, Fujimaki T, Yasukochi Y, Takeuchi I, Sakuma J. Identification of 12 novel loci that confer susceptibility to early-onset dyslipidemia. Int J Mol Med. 2019; 43:57–82. https://doi.org/10.3892/ijmm.2018.3943 [PubMed]
- 14. Weissglas-Volkov D, Pajukanta P. Genetic causes of high and low serum HDL-cholesterol. J Lipid Res. 2010; 51:2032–57. https://doi.org/10.1194/jlr.R004739 [PubMed]
- 15. Pan L, Yang Z, Wu Y, Yin RX, Liao Y, Wang J, Gao B, Zhang L, and China National Survey of Chronic Kidney Disease Working Group. The prevalence, awareness, treatment and control of dyslipidemia among adults in China. Atherosclerosis. 2016; 248:2–9. https://doi.org/10.1016/j.atherosclerosis.2016.02.006 [PubMed]
- 16. Liu CX, Yin RX, Shi ZH, Deng GX, Zheng PF, Wei BL, Guan YZ. EHBP1 SNPs, their haplotypes, and gene-environment interactive effects on serum lipid levels. ACS Omega. 2020; 5:7158–69. https://doi.org/10.1021/acsomega.9b03522 [PubMed]
- 17. Yang S, Yin RX, Miao L, Zhang QH, Zhou YG, Wu J. Association between the LIPG polymorphisms and serum lipid levels in the Maonan and Han populations. J Gene Med. 2019; 21:e3071. https://doi.org/10.1002/jgm.3071 [PubMed]
- 18. Qiu L, Yin RX, Nie RJ, Hu XJ, Khounphinith E, Zhang FH. The CXCL12 SNPs and their haplotypes are associated with serum lipid traits. Sci Rep. 2019; 9:19524. https://doi.org/10.1038/s41598-019-55725-3 [PubMed]
- 19. Borén J, Chapman MJ, Krauss RM, Packard CJ, Bentzon JF, Binder CJ, Daemen MJ, Demer LL, Hegele RA, Nicholls SJ, Nordestgaard BG, Watts GF, Bruckert E, et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease: pathophysiological, genetic, and therapeutic insights: a consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2020; 41:2313–30. https://doi.org/10.1093/eurheartj/ehz962 [PubMed]
- 20. Pan J, Liu J, Wang H, Li W, Du X, Lin Q, Zhang X, Qi D, Tu J, Ning X, Yang Q, Wang J. Association of carotid atherosclerosis with lipid components in asymptomatic low-income Chinese: a population-based cross-sectional study. Front Neurol. 2020; 11:276. https://doi.org/10.3389/fneur.2020.00276 [PubMed]
- 21. Kathiresan S, Manning AK, Demissie S, D’Agostino RB, Surti A, Guiducci C, Gianniny L, Burtt NP, Melander O, Orho-Melander M, Arnett DK, Peloso GM, Ordovas JM, Cupples LA. A genome-wide association study for blood lipid phenotypes in the Framingham heart study. BMC Med Genet. 2007 (Suppl 1); 8:S17. https://doi.org/10.1186/1471-2350-8-S1-S17 [PubMed]
- 22. Lee JC, Weissglas-Volkov D, Kyttälä M, Dastani Z, Cantor RM, Sobel EM, Plaisier CL, Engert JC, van Greevenbroek MM, Kane JP, Malloy MJ, Pullinger CR, Huertas-Vazquez A, et al. WW-domain-containing oxidoreductase is associated with low plasma HDL-C levels. Am J Hum Genet. 2008; 83:180–92. https://doi.org/10.1016/j.ajhg.2008.07.002 [PubMed]
- 23. Sáez ME, González-Pérez A, Martínez-Larrad MT, Gayán J, Real LM, Serrano-Ríos M, Ruiz A. WWOX gene is associated with HDL cholesterol and triglyceride levels. BMC Med Genet. 2010; 11:148. https://doi.org/10.1186/1471-2350-11-148 [PubMed]
- 24. Rand DM, Mossman JA. Mitonuclear conflict and cooperation govern the integration of genotypes, phenotypes and environments. Philos Trans R Soc Lond B Biol Sci. 2020; 375:20190188. https://doi.org/10.1098/rstb.2019.0188 [PubMed]
- 25. Zan Y, Carlborg Ö. Dynamic genetic architecture of yeast response to environmental perturbation shed light on origin of cryptic genetic variation. PLoS Genet. 2020; 16:e1008801. https://doi.org/10.1371/journal.pgen.1008801 [PubMed]
- 26. Zhang ZH, Zhu YJ, Wang SL, Fan YY, Zhuang JY. Importance of the interaction between heading date genes Hd1 and Ghd7 for controlling yield traits in rice. Int J Mol Sci. 2019; 20:516. https://doi.org/10.3390/ijms20030516 [PubMed]
- 27. Zheng PF, Yin RX, Liu CX, Deng GX, Guan YZ, Wei BL. SYNE1-QK1 SNPs, G × G and G × E interactions on the risk of hyperlipidaemia. J Cell Mol Med. 2020; 24:5772–85. https://doi.org/10.1111/jcmm.15239 [PubMed]
- 28. Barghi N, Hermisson J, Schlötterer C. Polygenic adaptation: a unifying framework to understand positive selection. Nat Rev Genet. 2020; 21:769–81. https://doi.org/10.1038/s41576-020-0250-z [PubMed]
- 29. Miao L, Yin RX, Pan SL, Yang S, Yang DZ, Lin WX. BCL3-PVRL2-TOMM40 SNPs, gene-gene and gene-environment interactions on dyslipidemia. Sci Rep. 2018; 8:6189. https://doi.org/10.1038/s41598-018-24432-w [PubMed]
- 30. Deng GX, Yin RX, Guan YZ, Liu CX, Zheng PF, Wei BL, Wu JZ, Miao L. Association of the NCAN-TM6SF2-CILP2-PBX4-SUGP1-MAU2 SNPs and gene-gene and gene-environment interactions with serum lipid levels. Aging (Albany NY). 2020; 12:11893–913. https://doi.org/10.18632/aging.103361 [PubMed]
- 31. Guo T, Yin RX, Pan L, Yang S, Miao L, Huang F. Integrative variants, haplotypes and diplotypes of the CAPN3 and FRMD5 genes and several environmental exposures associate with serum lipid variables. Sci Rep. 2017; 7:45119. https://doi.org/10.1038/srep45119 [PubMed]
- 32. Zhang Q, Fan XY, Guo WL, Cao YJ, Lin YC, Cheng WJ, Chen LJ, Rao PF, Ni L, Lv XC. The protective mechanisms of macroalgae laminaria japonica consumption against lipid metabolism disorders in high-fat diet-induced hyperlipidemic rats. Food Funct. 2020; 11:3256–70. https://doi.org/10.1039/d0fo00065e [PubMed]
- 33. Hu Q, Zhang H, Gutiérrez Cortés N, Wu D, Wang P, Zhang J, Mattison JA, Smith E, Bettcher LF, Wang M, Lakatta EG, Sheu SS, Wang W. Increased Drp1 acetylation by lipid overload induces cardiomyocyte death and heart dysfunction. Circ Res. 2020; 126:456–70. https://doi.org/10.1161/CIRCRESAHA.119.315252 [PubMed]
- 34. Jha CK, Mir R, Elfaki I, Javid J, Babakr AT, Banu S, Chahal SMS. Evaluation of the Association of Omentin 1 rs2274907 A>T and rs2274908 G>A Gene Polymorphisms with Coronary Artery Disease in Indian Population: A Case Control Study. J Pers Med. 2019; 9:30. https://doi.org/10.3390/jpm9020030 [PubMed]
- 35. Wang XB, Cui NH, Zhang S, Guo SR, Liu ZJ, Ming L. PARP-1 variant Rs1136410 confers protection against coronary artery disease in a Chinese Han population: a two-stage case-control study involving 5643 subjects. Front Physiol. 2017; 8:916. https://doi.org/10.3389/fphys.2017.00916 [PubMed]
- 36. Rosada A, Kassner U, Weidemann F, König M, Buchmann N, Steinhagen-Thiessen E, Spira D. Hyperlipidemias in elderly patients: results from the Berlin Aging Study II (BASEII), a cross-sectional study. Lipids Health Dis. 2020; 19:92. https://doi.org/10.1186/s12944-020-01277-9 [PubMed]
- 37. Anisimova AS, Meerson MB, Gerashchenko MV, Kulakovskiy IV, Dmitriev SE, Gladyshev VN. Multifaceted deregulation of gene expression and protein synthesis with age. Proc Natl Acad Sci USA. 2020; 117:15581–90. https://doi.org/10.1073/pnas.2001788117 [PubMed]
- 38. Cho L, Davis M, Elgendy I, Epps K, Lindley KJ, Mehta PK, Michos ED, Minissian M, Pepine C, Vaccarino V, Volgman AS, and ACC CVD Womens Committee Members. Summary of updated recommendations for primary prevention of cardiovascular disease in women: JACC state-of-the-art review. J Am Coll Cardiol. 2020; 75:2602–18. https://doi.org/10.1016/j.jacc.2020.03.060 [PubMed]
- 39. Barrientos G, Llanos P, Basualto-Alarcón C, Estrada M. Androgen-regulated cardiac metabolism in aging men. Front Endocrinol (Lausanne). 2020; 11:316. https://doi.org/10.3389/fendo.2020.00316 [PubMed]
- 40. Farag MA, Tawfike AF, Donia MS, Ehrlich A, Wessjohann LA. Influence of pickling process on Allium cepa and Citrus limon metabolome as determined via mass spectrometry-based metabolomics. Molecules. 2019; 24:928. https://doi.org/10.3390/molecules24050928 [PubMed]
- 41. Wood AM, Kaptoge S, Butterworth AS, Willeit P, Warnakula S, Bolton T, Paige E, Paul DS, Sweeting M, Burgess S, Bell S, Astle W, Stevens D, et al, and Emerging Risk Factors Collaboration/EPIC-CVD/UK Biobank Alcohol Study Group. Risk thresholds for alcohol consumption: combined analysis of individual-participant data for 599 912 current drinkers in 83 prospective studies. Lancet. 2018; 391:1513–23. https://doi.org/10.1016/S0140-6736(18)30134-X [PubMed]
- 42. Levy D, Larson MG, Benjamin EJ, Newton-Cheh C, Wang TJ, Hwang SJ, Vasan RS, Mitchell GF. Framingham heart study 100K project: genome-wide associations for blood pressure and arterial stiffness. BMC Med Genet. 2007 (Suppl 1); 8:S3. https://doi.org/10.1186/1471-2350-8-S1-S3 [PubMed]
- 43. Yu Z, Coresh J, Qi G, Grams M, Boerwinkle E, Snieder H, Teumer A, Pattaro C, Köttgen A, Chatterjee N, Tin A. A bidirectional mendelian randomization study supports causal effects of kidney function on blood pressure. Kidney Int. 2020; 98:708–16. https://doi.org/10.1016/j.kint.2020.04.044 [PubMed]
- 44. Huang Y, Ollikainen M, Muniandy M, Zhang T, van Dongen J, Hao G, van der Most PJ, Pan Y, Pervjakova N, Sun YV, Hui Q, Lahti J, Fraszczyk E, et al. Identification, heritability, and relation with gene expression of novel DNA methylation loci for blood pressure. Hypertension. 2020; 76:195–205. https://doi.org/10.1161/HYPERTENSIONAHA.120.14973 [PubMed]
- 45. Arnett DK, Graf GA. Metabolomics, lipid pathways, and blood pressure change. Arterioscler Thromb Vasc Biol. 2020; 40:1801–03. https://doi.org/10.1161/ATVBAHA.120.314816 [PubMed]
- 46. Guo T, Yin RX, Lin WX, Wang W, Huang F, Pan SL. Association of the variants and haplotypes in the DOCK7, PCSK9 and GALNT2 genes and the risk of hyperlipidaemia. J Cell Mol Med. 2016; 20:243–65. https://doi.org/10.1111/jcmm.12713 [PubMed]
- 47. Miao L, Yin RX, Pan SL, Yang S, Yang DZ, Lin WX. Weighted gene co-expression network analysis identifies specific modules and hub genes related to hyperlipidemia. Cell Physiol Biochem. 2018; 48:1151–63. https://doi.org/10.1159/000491982 [PubMed]
- 48. Yin RX, Wu DF, Miao L, Aung LH, Cao XL, Yan TT, Long XJ, Liu WY, Zhang L, Li M. Several genetic polymorphisms interact with overweight/obesity to influence serum lipid levels. Cardiovasc Diabetol. 2012; 11:123. https://doi.org/10.1186/1475-2840-11-123 [PubMed]
- 49. Cao XL, Yin RX, Huang F, Wu JZ, Chen WX. Chromosome 9p21 and ABCA1 genetic variants and their interactions on coronary heart disease and ischemic stroke in a Chinese han population. Int J Mol Sci. 2016; 17:586. https://doi.org/10.3390/ijms17040586 [PubMed]
- 50. Gabb GM, Mangoni AA, Anderson CS, Cowley D, Dowden JS, Golledge J, Hankey GJ, Howes FS, Leckie L, Perkovic V, Schlaich M, Zwar NA, Medley TL, Arnolda L. Guideline for the diagnosis and management of hypertension in adults - 2016. Med J Aust. 2016; 205:85–89. https://doi.org/10.5694/mja16.00526 [PubMed]
- 51. Fan Y, Li W, Liu H, Wang L, Zhang S, Li W, Liu H, Leng J, Shen Y, Tuomilehto J, Yu Z, Yang X, Liu M, Hu G. Effects of obesity and a history of gestational diabetes on the risk of postpartum diabetes and hyperglycemia in Chinese women: obesity, GDM and diabetes risk. Diabetes Res Clin Pract. 2019; 156:107828. https://doi.org/10.1016/j.diabres.2019.107828 [PubMed]



