Introduction
Aging and wrong lifestyle choices, including inadequate dietary patterns, increase the risk of developing several diseases such as obesity and its-related chronic degenerative diseases. Interestingly, the aging program can be accelerated by obesity [1]. It is thus likely that obesity reduces life- and health span and plays a predominant role in the onset of age-related diseases [2]. In fact, the prevalence of obesity is globally increasing in populations and has become a burden for healthcare systems. Several studies suggest that dietary restriction (DR) regimens (e.g. intermittent fasting, calorie restriction, low calorie diet) reverse obesity and improve health in human by promoting the same molecular and metabolic adaptations that have been shown in animal models of longevity. In particular, DR in humans ameliorates several metabolic and hormonal factors that are implicated in the pathogenesis of an array of age-associated chronic metabolic diseases [3,4].
At present it is difficult to evaluate the effectiveness of DR on lifespan in humans, so that several works proposed predictive non-invasive biomarkers to evaluate the geroprotective role of DR. However, a miscellaneous of biomarkers is investigated in human intervention studies limiting the statistical robustness of the data. Whether a “biomarker-based” approach could be suitable for evaluating the effectiveness of DR still remains a matter of debate.
Precision medicine is a medical model that proposes the customization of healthcare, with the identification of predictors that can help to find the effectiveness of health-promoting dietary interventions. Biomarkers represent potentially predictive tools for precision medicine but, although affordable 'omics'-based technology has enabled faster identification of putative biomarkers [5], their validation is still hindered by low statistical power as well as limited reproducibility of results.
Herein, through meta-analysis we have evaluated the effect size of DR regimens on adipose mass and well-recognized biomarkers of healthy aging. Overall findings provide the geroprotective footprint of DR in humans and highlight a useful platform to validate or monitor the efficiency of dietary treatments to preserve and improve health span and longevity.
Results
Effects of DR on total and visceral adipose mass
DR regimens are effective in slowing aging, and maintaining healthy status in animals [6,7]. Adipose mass quickly and dynamically responds to nutrient/energy fluctuation and its remodelling seems to mediate the beneficial effects of DR [7]. In this section we evaluated the effects of DR on adipose mass (Fig. 1). Interestingly, all studies showed clear evidence on the efficacy of DR in reducing total adipose mass in human (SDM -0.913; 95% CI -0.994, -0.832; p<0.000). Interestingly, we detected higher effectiveness of DR in healthy than unhealthy subjects (SDM -1.843; 95% CI -2.144, -1.542 p<0.000 and SDM -0.813; 95% CI -0.897, -0.728 p<0.000, respectively). Our data reveal that DR was also effective in reducing visceral fat mass (SDM -0.944; 95% CI -1.187, -0.700; p<0.000) (Fig. 1) and identify adipose mass measurement as a feasible approach to evaluate the efficacy of diets low in calories.
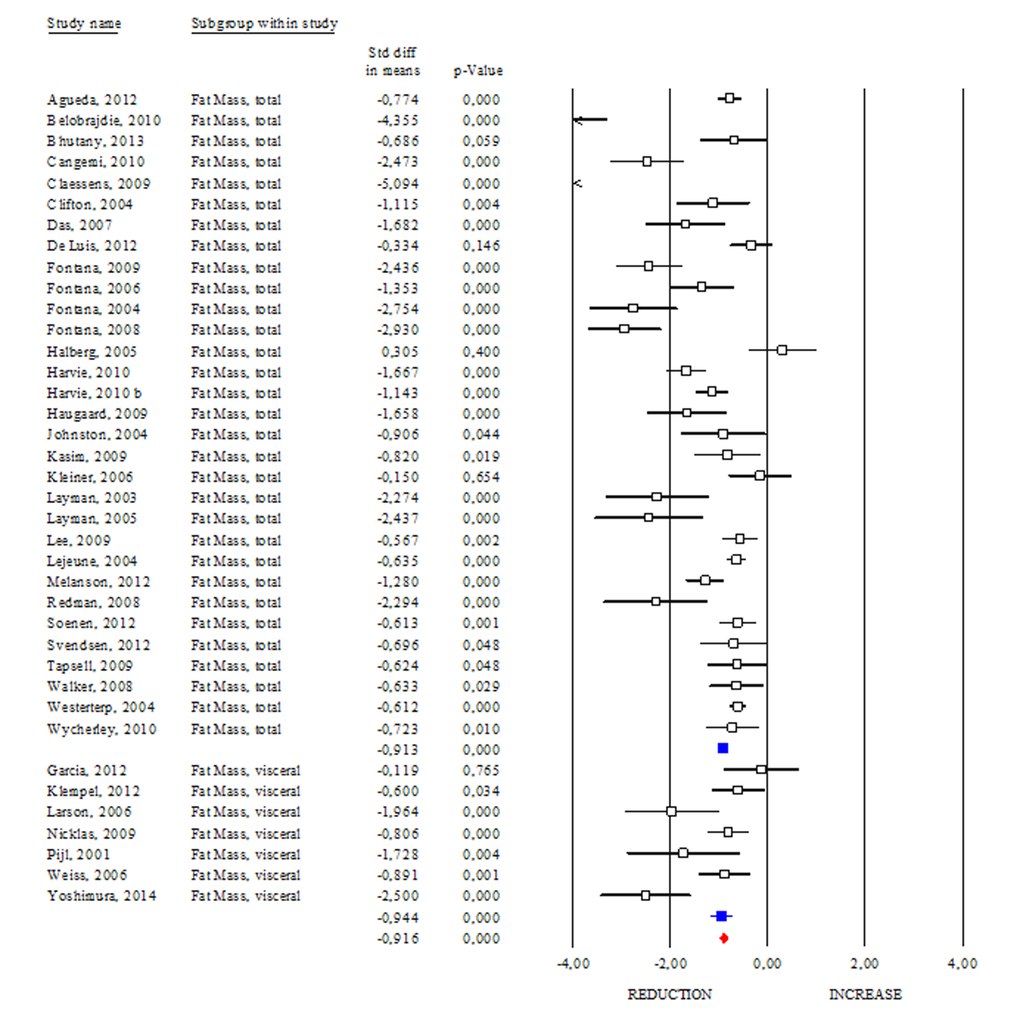
Figure 1. Changes of total and visceral adipose mass after DR. Studies were stratified according to the design of the study. A positive standardized difference in mean (SDM) indicates an increase, whereas a negative SDM indicates the decrease of fat mass (total or visceral). The empty black square indicates the results of each study, whereas empty blu square shows the summary results of each subgroup data. The red diamond resumes overall results of the included studies in the forest plot.
Effects of DR on adipokines and DHEA
Among adipokines, adiponectin has an anti-inflammatory function and correlates with healthy metabolic profile. Reduction of adiponectin production is often revealed in obese and diabetic subjects [8]. These evidences highlight adiponectin as a good candidate to monitor healthy status in human. However, conflicting results emerge from circulating adiponectin levels in centenarians [9,10]. Herein we determined changes of adiponectin levels occurring after DR. As shown in Fig. 2, DR increased adiponectin levels in human (SDM 0.427; 95% CI 0.243, 0.612; p<0.000) independently of healthy status (healthy group: SDM 0.947; 95% CI 0.395, 1.499 p<0.001 and unhealthy group: SDM 0.370; 95% CI 0.155, 0.585 p<0.001). The “satiety hormone” leptin controls dietary behaviour and has been strongly associated with adipose mass. Indeed, reduced leptin levels are associated with diminished visceral adipose mass. However, unclear are evidences about its levels in healthy centenarians [9,10]. Our data reveal that leptin levels were significantly reduced in DR group (SDM -1.383; 95% CI -1.511, -1.255; P<0.000) (Fig. 3).
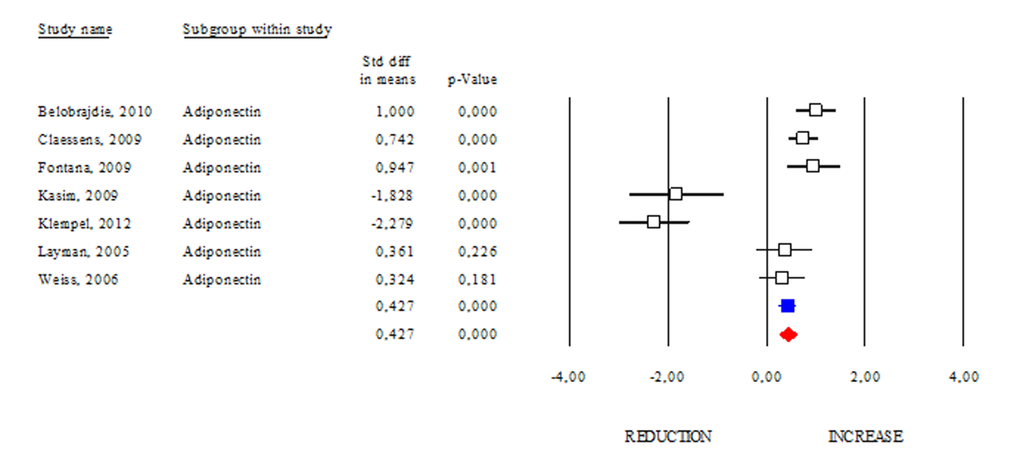
Figure 2. DR effects on circulating adiponectin. Studies were stratified according to the design of the study. A positive standardized difference in mean (SDM) indicates an increase, whereas a negative SDM indicates the decrease of circulating adiponectin. The empty black square indicates the results of each study, whereas empty blu square shows the summary results of each subgroup data. The red diamond resumes overall results of the included studies in the forest plot.
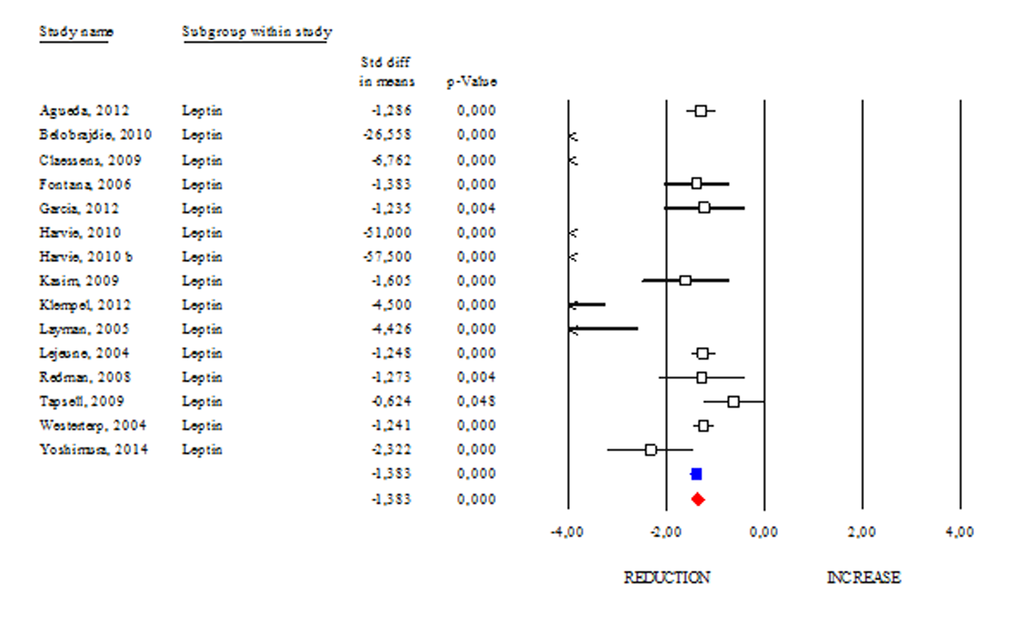
Figure 3. DR effects on circulating leptin. Studies were stratified according to the design of the study. A positive standardized difference in mean (SDM) indicates an increase, whereas a negative SDM indicates the decrease of circulating leptin. The empty black square indicates the results of each study. The red diamond resumes overall results of the included studies in the forest plot.
The hormonal profile of aging includes a marked decrease in the adrenal hormone dehydroepiandrosterone (DHEA) [11]. DHEA is taken up by adipose tissue and seems to reduce its mass protecting against obesity [12]. Epidemiologic data in the elderly cohort of long-living Okinawans (over 65) show relatively high plasma DHEA levels at older ages than the aged-matched counterpart [13]. However, as disclosed in Suppl. Fig. 1, DHEA levels were unchanged after DR (SDM 0.149; 95% CI -0.342, 0.641 p 0.551). Overall findings suggest a tight relationship between changes in circulating adipokines and reduction of adipose mass occurring after DR. Differently, DHEA modulation seems to be independent of calorie intake.
Effects of DR on insulin, IGF-1, HOMA Index and IGBPs
Insulin and insulin growth factors 1 (IGF-1) signalling is an evolutionary conserved pathway linking nutrient levels to fat mass and lifespan. Generally, reduced level of insulin and IGF-1 is associated with increased longevity from yeasts to mammals [14]. Differently, levels of insulin and IGF-1 are commonly higher in subjects affected by age-related diseases or obesity than lean healthy subjects [15]. In our work, we reported clear evidence about DR effects on insulin and IGF-1 levels in human (Fig. 4). In particular, we observed a significant reduction in insulin both in healthy (SDM -1.019; 95% CI -1.362, -0.675 p<0.000) and unhealthy subjects (SDM -0.811; 95% CI -0.893, -0.730 p<0.000). The same trend was detected by analysing the IGF-1 levels (SDM -0.546; 95% CI -0.750, -0.342 p<0.000). Overall data analyses (SDM -0.779; 95% CI -0.851, -0.706 p<0.000) confirm decreased insulin/IGF-1 levels as downstream effect of DR in human.
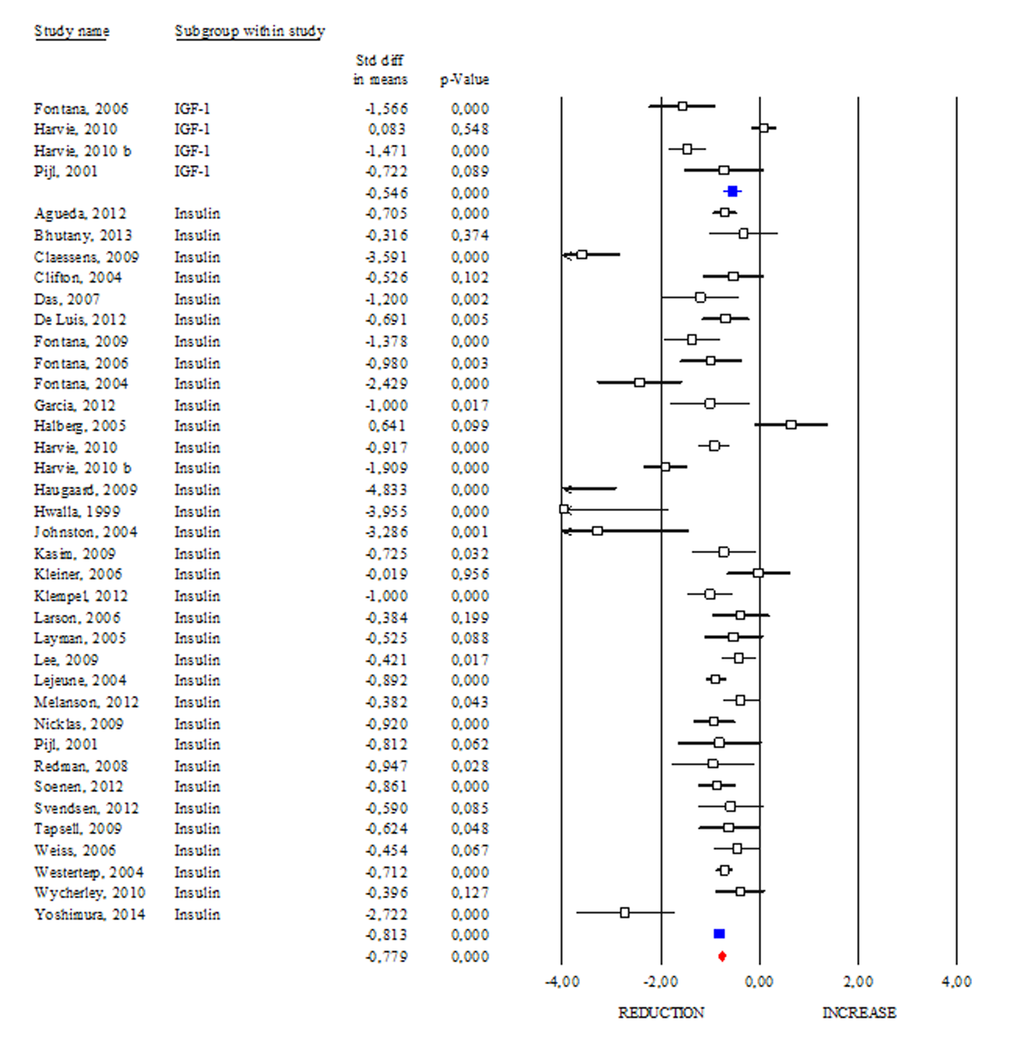
Figure 4. Changes of circulating insulin and insulin growth factor-1 (IGF-1) after DR. Studies were stratified according to the design of the study. A positive standardized difference in mean (SDM) indicates an increase, whereas a negative SDM indicates the decrease of circulating IGF-1 or insulin. The empty black square indicates the results of each study, whereas empty blu square shows the summary results of each subgroup data. The red diamond resumes overall results of the included studies in the forest plot.
The Homeostasis Model Assessment (HOMA) Index is currently a biochemical tool to estimate insulin sensitivity by matching fasting glycaemia and insulinemia [16]. A study carried out on centenarians indicates that they seem to be protected from hyperinsulinaemia, and their insulin resistance is as low, if not lower, than that of healthy younger adults [17]. The correlation between HOMA Index with obesity or aging suggests its prognostic capacity to evaluate the efficacy of health promoting strategies. Accordingly, we reported a significant reduction in the HOMA Index occurring after DR (SDM -0.837; 95% CI -0.990, -0.750 p<0.000) (Fig. 5) and this effect was stronger if dietary treatment was longer than 3 months (data not shown).
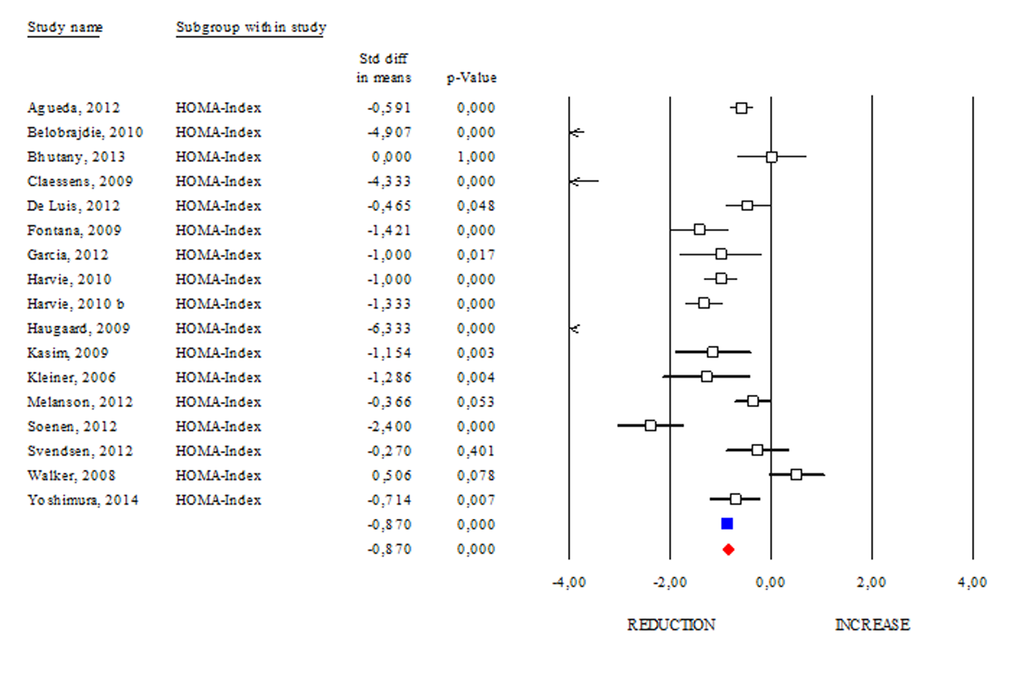
Figure 5. Changes of HOMA Index after DR. Studies were stratified according to the design of the study. A positive standardized difference in mean (SDM) indicates an increase, whereas a negative SDM indicates the decrease of HOMA Index. The empty black square indicates the results of each study. The red diamond resumes overall results of the included studies in the forest plot.
The IGF-binding protein 2 (IGFBP2) is known as a carrier protein for IGF-1 limiting its biological action [18]. However, there are several characterized IGFBPs, which seem to improve metabolic status independently of IGFs binding [19]. Interestingly, some papers reported that DR regimens increase circulating levels of IGFBPs [20]. In our work, we analysed the changes in the levels of the best-known IGFBPs after DR. As shown in Fig. 6, DR similarly modulated IGFBP-1 and IGFBP-2 levels (SDM 1.527; 95% CI 1.248, 1.806 p<0.000 and SDM 1.687; 95% CI 1.387, 1.986 p<0.000, respectively). Differently, DR was ineffective in increasing IGFBP-3 levels (SDM -0.045; 95% CI -0.517, 0.427 p=0.853). These results suggest that IGFBP-1 and -2 are more sensitive to DR than IGFBP-3.
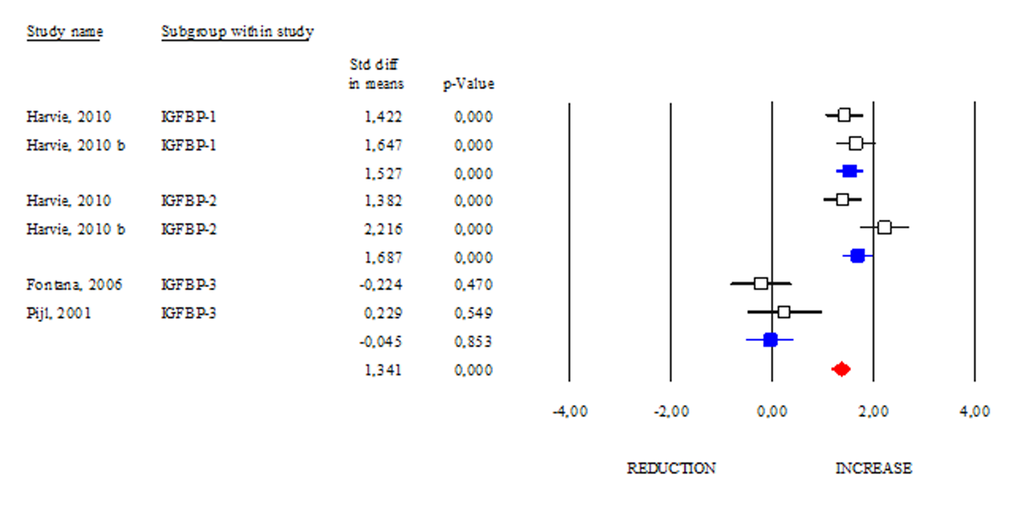
Figure 6. Changes of circulating IGFB-1, IGFBP-2 and IGFBP-3 after DR. Studies were stratified according to the design of the study. A standardized difference in mean (SDM) indicates an increase, whereas a negative SDM indicates the decrease of IGFB-1, IGFBP-2 or IGFBP-3. The empty black square indicates the results of each study, whereas empty blu square shows the summary results of each subgroup data. The red diamond resumes overall results of the included studies in the forest plot.
Effects of DR on inflammatory markers
One of the common features of aging and obesity is the presence of a chronic sterile low-grade inflammatory status, which contributes to the onset of several metabolic perturbations [21]. In our work we evaluated the changes in circulating inflammatory markers observed after DR (Fig. 7). Interestingly, among the evaluated inflammatory markers, only CRP and IL-6 displayed a significant reduction after DR (SDM -0.715; 95% CI -0.862, -0.568 p<0.000 and SDM -0.316; 95% CI -0.515, -0.118 p<0.002, respectively). Although IL-1 and TNF-α are cytokines routinely assayed to monitor systemic inflammation, our data revealed that their level remained unchanged after DR (SDM 0.041; 95% CI -0.181, 0.263 p=0.719 and SDM -0.079; 95% CI -0.264, 0.106 p=0.402, respectively). Overall data regarding CRP, IL-6, IL-1 and TNF-α levels revealed anti-inflammatory effect of DR in human (SDM -0.351; 95% CI -0.442, -0.260 p<0.000) (Fig. 7).
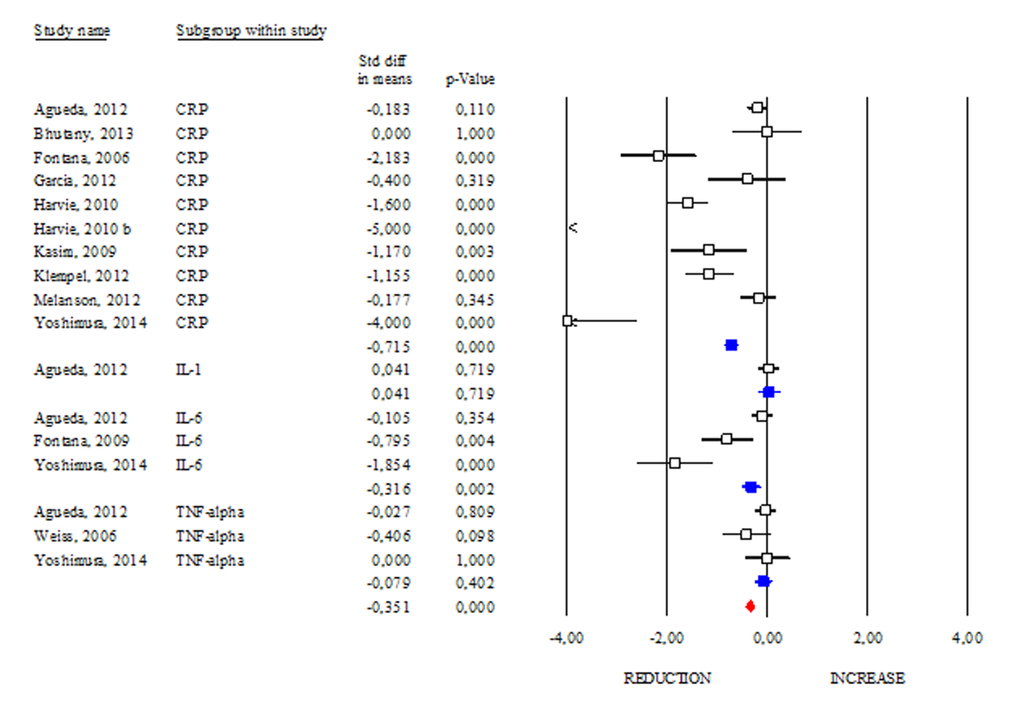
Figure 7. Changes of inflammatory markers after DR. Studies were stratified according to the design of the study. A positive standardized difference in mean (SDM) indicates an increase, whereas a negative SDM indicates the decrease of CRP, IL-1, IL-6 or TNF-alpha. The empty black square indicates the results of each study, whereas empty blu square shows the summary results of each subgroup data. The red diamond resumes overall results of the included studies in the forest plot.
Discussion
Aging is commonly defined as a physiological decline of biological functions in the body. Aging strongly remodels adipose depots by reducing subcutaneous adipose in favour of visceral depots enlargement [22]. Aging and visceral adipose tissue expansion act in synergy in inducing a chronic low grade of inflammatory status, which triggers a systemic metabolic decline in human [21,23]. DR is a promising and feasible strategy that ameliorates body metabolic and inflammatory profile increasing lifespan through evolutionary-conserved mechanisms [4,22,24,25]. Herein we included all studies evaluating the impact of DR on several healthy-associated markers in human including adipose mass. Increased visceral adiposity leads to chronic inflammation, which is often associated with a number of comorbidities (e.g. hyperinsulinemia, hypertension, insulin resistance, glucose intolerance) and reduced life expectancy [26,27]. Through this meta-analysis approach, we confirmed the capacity of DR to reduce total and visceral adipose mass and, interestingly, we observed a more effective visceral adipose mass reduction after DR regimens (-20% in DR: SDM -1.081; 95% CI -1.242, -0.921 p<0.000) (-30/40% in DR: SDM -0.893; 95% CI -1.050, -0.737 p<0.000 and >-40% in DR: SDM -0.678; 95% CI -0.800, -0.555 p<0.000). These findings suggest that to obtain a more effective adipose mass loss, 20% in calorie reduction could be an elective strategy. Central or visceral adiposity perturbs systemic inflammation in animal models and human and relatively to this, the healthy effects of DR could be mediated by visceral adiposity reduction. Indeed, DR significantly diminished the markers of inflammation, highlighting the central role of DR-mediated adipose tissue remodelling in improving inflammatory profile in human. Furthermore, DR also increased adiponectin/leptin ratio, which is commonly associated with ameliorated insulin sensitivity in human. In line with this effect, we demonstrated that DR was successful in reducing insulin, IGF-1 and HOMA index.
The insulin growth factor binding proteins (IGFBPs) are a family of proteins that bind to insulin-like growth factors limiting their biological actions [28]. IGFBP-2 is the most abundant among circulating IGFBPs and its anti-diabetic role as well as direct ability to limit adipogenesis has been demonstrated [29,30]. Actually, high serum levels of IGFBP-2 appear to protect against obesity and type 2 diabetes [30]. IGFBP-1 showed an inverse relation with insulin and BMI in human [31]. Differently, unclear are the evidences about the link between IGFBP-3 and adipose mass. In accordance with the data described above, we observed a strong responsiveness in circulating levels of IGFBP-1 and -2 occurring after DR. However some limitations emerge from this meta-analysis. In particular, statistical analyses on IL-1 and IGFBPs were carried out only evaluating the results obtained from few studies [32-35]. Moreover, it was not possible to evaluate the efficiency of DR in gender or time of treatment subgroups because it was difficult to collect a good number of subjects.
In conclusion, by a meta-analysis approach we have provided evidences about DR efficiency on key hallmarks of aging (Fig. 8) and built a useful platform to evaluate the responses of human to dietary regimens low in calories (Fig. 9).

Figure 8. Geroprotective footprint of dietary restriction.
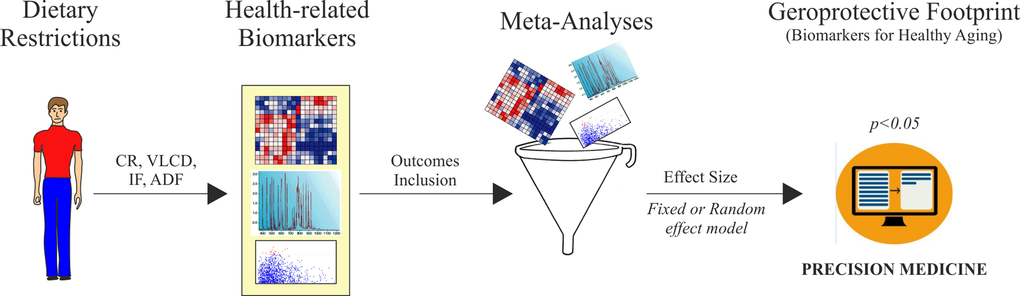
Figure 9. Algorithm development for biomarkers validation of dietary restriction in human. CR: calorie restriction; VLCD: very low calorie diet; IF: intermittent fasting; ADF: alternate-day-fasting.
Materials and Methods
Search strategy and included studies
In our work we analysed human intervention studies and evaluated the impact of DR regimens on adipose mass and some biomarkers of healthy aging (Geromarkers). The Geromarkers included in our meta-analysis were described in Table 1. Two investigators, E.G. and D.L.B., independently carried out study selection and included both studies with an experimental design (EXP) and quasi-experimental design (Q-EXP). EXP studies were randomized with a control group and a parallel or crossover design; whereas Q-EXP included observational studies (pre- and post-intervention or pre- and post-data), non-randomized or uncontrolled studies [36]. Q-EXP studies were pooled together with EXP studies only after assessing whether they were in agreement with EXP studies [37]. Candidate studies were searched in PubMed (finalized February 30, 2016) using the terms ‘calorie or caloric or dietary restriction’, ‘fasting or intermittent fasting or alternate day fasting and ‘adipose tissue or fat mass or fat tissue’’. Inclusion criteria were as follows: human intervention studies with long-term study design (> 3 months); healthy and unhealthy (e.g. dyslipidaemia, obesity, metabolic syndrome) subjects; numerically analysable information about results, study duration and calories reduced in the study. Studies were excluded when: only abstracts were available; duration time of the study was lesser than 3 months; data presentation was incomplete; information about the DR was incomplete. When necessary, efforts were made to contact investigators for clarification or additional data. This research strategy produced a total of 201 studies. Furthermore, a manual research of references from clinical studies and reviews identified 42 additional studies, for a total of 243 studies to be evaluated, 9 of which are reviews [38-46]. A first screening allowed discarding 147 articles whose titles or abstracts were evidently irrelevant to our aim. Of the remaining 96 studies, 53 were rejected whenever: they presented incomplete data; DR was coupled with physical exercise; there were no reported data on adipose mass; they only presented data on weight and fat mass without other parameters (Fig. 10). Therefore, from 243 initial candidates, the 43 studies available for a formal meta-analysis had the following characteristics: they were written in English; they had a period of intervention of at least two weeks; they were carried out exclusively on human subjects. Among the considered studies, 12 were on females [32,34,35,47-55], 4 on males [56-59], and the rest mixed [60-62,33,63-85]. Moreover, 30 studies were intervention studies evaluating the efficacy of calorie restriction [33-35,47-49,51-53,56,58-60,63-67,69,71-73,75,76,79-82,84,85]; 4 were intervention studies evaluating the efficacy of intermittent fasting [50,57,61,70]; 9 were intervention studies evaluating the efficacy of low or very low calorie diets [32,54,55,62,68,74,77,78,83]. The selected studies included human groups with different BMI. In particular, 10 were studies on obese [34,35,48,50,53,54,58,61,64,68], 16 on overweight [51,52,57,59,62,63,65,69,71-73,77-79,82,85], 12 on both obese and overweight [32,47,49,55,60,62,74,75,80,81,83,84], 5 on both normal weight and overweight [33,66,67,70,76]. Finally, the studies were on healthy subjects, with the exception of few articles in which subjects were affected by the following pathologies: chronic osteoarthritis [64]; metabolic syndrome [59]; hyperinsulinemia [58,72], polycystic ovary syndrome [49], type 2 diabetes [84]. Hence, the meta-analysis was based on 43 studies and analysed a total of 2094 subjects. Before analyses, all studies were stratified for gender, healthy status, time of treatment and percentage of calorie reduction and the main characteristics of the included studies were reported in Table 2. Calorie restriction, intermittent or alternate-day-fasting and low calorie diet interventions were overall grouped in dietary restriction (DR) category.
Table 1. Selected biomarkers and number of the studies included in meta-analysis.
| Biomarkers | n. of the studies |
| Fat Mass (total and visceral) | 38 |
| Adipokines (adiponectin and leptin) | 22 |
| IGFBPs (IGFBP-1, -2, -3) | 6 |
| IGF-1 | 4 |
| HOMA-Index | 17 |
| Insulin | 34 |
| Inflammation (TNFa, IL-1, IL-6, CRP) | 17 |
| DHEA | 5 |
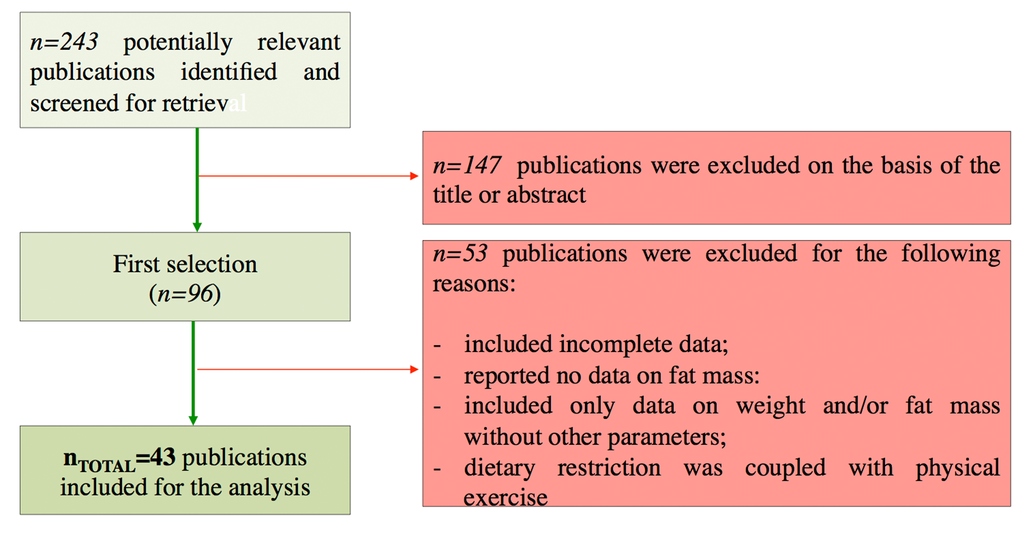
Figure 10. Flow chart of the study identification and selection.
Table 2. Characteristics of the included studies for the meta-analyses.
| Study Design | Gender Stratification | Healthy Status Stratification | Time of Treatment | ||||||
| Unrandomized | Randomized or Controlled | Randomized and Controlled | Cross-Sectional | Yes | No | Yes | No | Brief (<3 months) | Long-Term (>3 months) |
| 4 | 14 | 23 | 2 | 16 | 27 | 36 | 2 | 26 | 16 |
Data analysis
Relevant data of the 43 studies available were entered for formal meta-analytic evaluation into the Comprehensive Meta-Analysis software (Biostat) [86]. Data analysis was performed as previously described [87]. In particular, for the results showed as post-data only, we selected mean, standard deviation and sample size in each group, or difference in means, sample size and p value between groups. When results were reported as pre- and post-data, we used mean, standard deviation, sample size in each group and correlation between baseline and end-point intervention period, or mean change, standard deviation difference, sample size in each group, correlation between baseline and end-point intervention period. For observational studies considering only one group (pre–post-intervention data), we used mean difference, standard deviation of difference and sample size. In all studies, we assumed the correlation between baseline and end-point study period to be 0.5 to produce the most conservative estimate [37,88], To enable a joint comparison, the standardized difference in mean (SDM) was calculated for each outcome. In our analysis, positive SDM indicates increased effect size of calorie restriction on outcome considered. The effect sizes of the included studies were pooled both under a ‘fixed effects model’ or ‘random effect model’. Under fixed effects model we assumed that the true effect is the same in all studies. By contrast, under the random-effects model we allowed that the true effect may vary from one study to the next [37]. Fixed or random effect model was selected following evaluation of heterogeneity between studies based on the I2 test for heterogeneity. When I2 values were low, we selected a fixed effects model, whereas random effects model was selected for I2 values higher than 75%.
Conflicts of Interest
The authors declare no conflict of interests.
Funding
This work was supported by “Uncovering Excellence” – University of Rome Tor Vergata and Ministero della Salute (GR-2011-02348047).
References
- 1. Horvath S, Erhart W, Brosch M, Ammerpohl O, von Schönfels W, Ahrens M, Heits N, Bell JT, Tsai PC, Spector TD, Deloukas P, Siebert R, Sipos B, et al. Obesity accelerates epigenetic aging of human liver. Proc Natl Acad Sci USA. 2014; 111:15538–43. https://doi.org/10.1073/pnas.1412759111 [PubMed]
- 2. Tchkonia T, Morbeck DE, Von Zglinicki T, Van Deursen J, Lustgarten J, Scrable H, Khosla S, Jensen MD, Kirkland JL. Fat tissue, aging, and cellular senescence. Aging Cell. 2010; 9:667–84. https://doi.org/10.1111/j.1474-9726.2010.00608.x [PubMed]
- 3. Al-Regaiey KA. The effects of calorie restriction on aging: a brief review. Eur Rev Med Pharmacol Sci. 2016; 20:2468–73. [PubMed]
- 4. Most J, Tosti V, Redman LM, Fontana L. Calorie restriction in humans: an update. Ageing Res Rev. 2016S1568-1637(16)30183-0. [PubMed]
- 5. Pearson ER. Personalized medicine in diabetes: the role of ‘omics’ and biomarkers. Diabet Med. 2016; 33:712–17. https://doi.org/10.1111/dme.13075 [PubMed]
- 6. Wiesenborn DS, Menon V, Zhi X, Do A, Gesing A, Wang Z, Bartke A, Altomare DA, Masternak MM. The effect of calorie restriction on insulin signaling in skeletal muscle and adipose tissue of Ames dwarf mice. Aging (Albany NY). 2014; 6:900–12. https://doi.org/10.18632/aging.100700 [PubMed]
- 7. Lettieri Barbato D, Tatulli G, Aquilano K, Ciriolo MR. Erratum: mitochondrial Hormesis links nutrient restriction to improved metabolism in fat cell. Aging (Albany NY). 2016; 8:1571. https://doi.org/10.18632/aging.101003 [PubMed]
- 8. Ghoshal K, Bhattacharyya M. Adiponectin: probe of the molecular paradigm associating diabetes and obesity. World J Diabetes. 2015; 6:151–66. https://doi.org/10.4239/wjd.v6.i1.151 [PubMed]
- 9. Pareja-Galeano H, Santos-Lozano A, Sanchis-Gomar F, Fiuza-Luces C, Garatachea N, Gálvez BG, Lucia A, Emanuele E. Circulating leptin and adiponectin concentrations in healthy exceptional longevity. Mech Ageing Dev. 2016S0047-6374(16)30019-7. [PubMed]
- 10. Meazza C, Vitale G, Pagani S, Castaldi D, Ogliari G, Mari D, Laarej K, Tinelli C, Bozzola M. Common adipokine features of neonates and centenarians. J Pediatr Endocrinol Metab. 2011; 24:953–57. https://doi.org/10.1515/JPEM.2011.373 [PubMed]
- 11. Samaras N, Samaras D, Frangos E, Forster A, Philippe J. A review of age-related dehydroepiandrosterone decline and its association with well-known geriatric syndromes: is treatment beneficial? Rejuvenation Res. 2013; 16:285–94. https://doi.org/10.1089/rej.2013.1425 [PubMed]
- 12. Karbowska J, Kochan Z. Effects of DHEA on metabolic and endocrine functions of adipose tissue. Horm Mol Biol Clin Investig. 2013; 14:65–74. https://doi.org/10.1515/hmbci-2013-0009 [PubMed]
- 13. Willcox BJ, Willcox DC, Todoriki H, Fujiyoshi A, Yano K, He Q, Curb JD, Suzuki M. Caloric restriction, the traditional Okinawan diet, and healthy aging: the diet of the world’s longest-lived people and its potential impact on morbidity and life span. Ann N Y Acad Sci. 2007; 1114:434–55. https://doi.org/10.1196/annals.1396.037 [PubMed]
- 14. Parrella E, Longo VD. Insulin/IGF-I and related signaling pathways regulate aging in nondividing cells: from yeast to the mammalian brain. Sci World J. 2010; 10:161–77. https://doi.org/10.1100/tsw.2010.8 [PubMed]
- 15. Balasubramanian P, Longo VD. Growth factors, aging and age-related diseases. Growth Horm IGF Res. 2016; 28:66–68. https://doi.org/10.1016/j.ghir.2016.01.001 [PubMed]
- 16. Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004; 27:1487–95. https://doi.org/10.2337/diacare.27.6.1487 [PubMed]
- 17. Curtis R, Geesaman BJ, DiStefano PS. Ageing and metabolism: drug discovery opportunities. Nat Rev Drug Discov. 2005; 4:569–80. https://doi.org/10.1038/nrd1777 [PubMed]
- 18. Kang HS, Kim MY, Kim SJ, Lee JH, Kim YD, Seo YK, Bae JH, Oh GT, Song DK, Ahn YH, Im SS. Regulation of IGFBP-2 expression during fasting. Biochem J. 2015; 467:453–60. https://doi.org/10.1042/BJ20141248 [PubMed]
- 19. Oh Y. IGF-independent regulation of breast cancer growth by IGF binding proteins. Breast Cancer Res Treat. 1998; 47:283–93. https://doi.org/10.1023/A:1005911319432 [PubMed]
- 20. Levine ME, Suarez JA, Brandhorst S, Balasubramanian P, Cheng CW, Madia F, Fontana L, Mirisola MG, Guevara-Aguirre J, Wan J, Passarino G, Kennedy BK, Wei M, et al. Low protein intake is associated with a major reduction in IGF-1, cancer, and overall mortality in the 65 and younger but not older population. Cell Metab. 2014; 19:407–17. https://doi.org/10.1016/j.cmet.2014.02.006 [PubMed]
- 21. Goldberg EL, Dixit VD. Drivers of age-related inflammation and strategies for healthspan extension. Immunol Rev. 2015; 265:63–74. https://doi.org/10.1111/imr.12295 [PubMed]
- 22. Lettieri Barbato D, Aquilano K. Feast and famine: adipose tissue adaptations for healthy aging. Ageing Res Rev. 2016; 28:85–93. https://doi.org/10.1016/j.arr.2016.05.007 [PubMed]
- 23. Minamino T, Orimo M, Shimizu I, Kunieda T, Yokoyama M, Ito T, Nojima A, Nabetani A, Oike Y, Matsubara H, Ishikawa F, Komuro I. A crucial role for adipose tissue p53 in the regulation of insulin resistance. Nat Med. 2009; 15:1082–87. https://doi.org/10.1038/nm.2014 [PubMed]
- 24. Lettieri Barbato D, Aquilano K, Ciriolo MR. FoxO1 at the nexus between fat catabolism and longevity pathways. Biochim Biophys Acta. 2014; 1841:1555–60. https://doi.org/10.1016/j.bbalip.2014.08.004 [PubMed]
- 25. Emran S, Yang M, He X, Zandveld J, Piper MD. Target of rapamycin signalling mediates the lifespan-extending effects of dietary restriction by essential amino acid alteration. Aging (Albany NY). 2014; 6:390–98. https://doi.org/10.18632/aging.100665 [PubMed]
- 26. Fontana L, Hu FB. Optimal body weight for health and longevity: bridging basic, clinical, and population research. Aging Cell. 2014; 13:391–400. https://doi.org/10.1111/acel.12207 [PubMed]
- 27. Guilherme A, Virbasius JV, Puri V, Czech MP. Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat Rev Mol Cell Biol. 2008; 9:367–77. https://doi.org/10.1038/nrm2391 [PubMed]
- 28. Clemmons DR. Insulin-like growth factor binding proteins and their role in controlling IGF actions. Cytokine Growth Factor Rev. 1997; 8:45–62. https://doi.org/10.1016/S1359-6101(96)00053-6 [PubMed]
- 29. Hedbacker K, Birsoy K, Wysocki RW, Asilmaz E, Ahima RS, Farooqi IS, Friedman JM. Antidiabetic effects of IGFBP2, a leptin-regulated gene. Cell Metab. 2010; 11:11–22. https://doi.org/10.1016/j.cmet.2009.11.007 [PubMed]
- 30. Wheatcroft SB, Kearney MT, Shah AM, Ezzat VA, Miell JR, Modo M, Williams SC, Cawthorn WP, Medina-Gomez G, Vidal-Puig A, Sethi JK, Crossey PA. IGF-binding protein-2 protects against the development of obesity and insulin resistance. Diabetes. 2007; 56:285–94. https://doi.org/10.2337/db06-0436 [PubMed]
- 31. Janssen JA, Stolk RP, Pols HA, Grobbee DE, Lamberts SW. Serum total IGF-I, free IGF-I, and IGFB-1 levels in an elderly population: relation to cardiovascular risk factors and disease. Arterioscler Thromb Vasc Biol. 1998; 18:277–82. https://doi.org/10.1161/01.ATV.18.2.277 [PubMed]
- 32. Harvie MN, Pegington M, Mattson MP, Frystyk J, Dillon B, Evans G, Cuzick J, Jebb SA, Martin B, Cutler RG, Son TG, Maudsley S, Carlson OD, et al. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: a randomized trial in young overweight women. Int J Obes. 2011; 35:714–27. https://doi.org/10.1038/ijo.2010.171 [PubMed]
- 33. Fontana L, Klein S, Holloszy JO. Long-term low-protein, low-calorie diet and endurance exercise modulate metabolic factors associated with cancer risk. Am J Clin Nutr. 2006; 84:1456–62. [PubMed]
- 34. Pijl H, Langendonk JG, Burggraaf J, Frölich M, Cohen AF, Veldhuis JD, Meinders AE. Altered neuroregulation of GH secretion in viscerally obese premenopausal women. J Clin Endocrinol Metab. 2001; 86:5509–15. https://doi.org/10.1210/jcem.86.11.8061 [PubMed]
- 35. Agueda M, Lasa A, Simon E, Ares R, Larrarte E, Labayen I. Association of circulating visfatin concentrations with insulin resistance and low-grade inflammation after dietary energy restriction in Spanish obese non-diabetic women: role of body composition changes. Nutr Metab Cardiovasc Dis. 2012; 22:208–14. https://doi.org/10.1016/j.numecd.2010.06.010 [PubMed]
- 36. Harris AD, Bradham DD, Baumgarten M, Zuckerman IH, Fink JC, Perencevich EN. The use and interpretation of quasi-experimental studies in infectious diseases. Clin Infect Dis. 2004; 38:1586–91. https://doi.org/10.1086/420936 [PubMed]
- 37. Borenstein M. 2009. Introduction to meta-analysis. Chichester, West Sussex, U.K.; Hoboken: John Wiley & Sons.
- 38. Fontana L, Klein S. Aging, adiposity, and calorie restriction. JAMA. 2007; 297:986–94. https://doi.org/10.1001/jama.297.9.986 [PubMed]
- 39. Holloszy JO, Fontana L. Caloric restriction in humans. Exp Gerontol. 2007; 42:709–12. https://doi.org/10.1016/j.exger.2007.03.009 [PubMed]
- 40. Johansson K, Neovius M, Hemmingsson E. Effects of anti-obesity drugs, diet, and exercise on weight-loss maintenance after a very-low-calorie diet or low-calorie diet: a systematic review and meta-analysis of randomized controlled trials. Am J Clin Nutr. 2014; 99:14–23. https://doi.org/10.3945/ajcn.113.070052 [PubMed]
- 41. Longo VD, Fontana L. Calorie restriction and cancer prevention: metabolic and molecular mechanisms. Trends Pharmacol Sci. 2010; 31:89–98. https://doi.org/10.1016/j.tips.2009.11.004 [PubMed]
- 42. Omodei D, Fontana L. Calorie restriction and prevention of age-associated chronic disease. FEBS Lett. 2011; 585:1537–42. https://doi.org/10.1016/j.febslet.2011.03.015 [PubMed]
- 43. Sohal RS, Weindruch R. Oxidative stress, caloric restriction, and aging. Science. 1996; 273:59–63. https://doi.org/10.1126/science.273.5271.59 [PubMed]
- 44. Varady KA, Hellerstein MK. Alternate-day fasting and chronic disease prevention: a review of human and animal trials. Am J Clin Nutr. 2007; 86:7–13. [PubMed]
- 45. Varady KA. Intermittent versus daily calorie restriction: which diet regimen is more effective for weight loss? Obes Rev. 2011; 12:e593–601. https://doi.org/10.1111/j.1467-789X.2011.00873.x [PubMed]
- 46. Wycherley TP, Moran LJ, Clifton PM, Noakes M, Brinkworth GD. Effects of energy-restricted high-protein, low-fat compared with standard-protein, low-fat diets: a meta-analysis of randomized controlled trials. Am J Clin Nutr. 2012; 96:1281–98. https://doi.org/10.3945/ajcn.112.044321 [PubMed]
- 47. Clifton PM, Noakes M, Keogh JB. Very low-fat (12%) and high monounsaturated fat (35%) diets do not differentially affect abdominal fat loss in overweight, nondiabetic women. J Nutr. 2004; 134:1741–45. [PubMed]
- 48. García-Unciti M, Izquierdo M, Idoate F, Gorostiaga E, Grijalba A, Ortega-Delgado F, Martínez-Labari C, Moreno-Navarrete JM, Forga L, Fernández-Real JM, Ibáñez J. Weight-loss diet alone or combined with progressive resistance training induces changes in association between the cardiometabolic risk profile and abdominal fat depots. Ann Nutr Metab. 2012; 61:296–304. https://doi.org/10.1159/000342467 [PubMed]
- 49. Kasim-Karakas SE, Almario RU, Cunningham W. Effects of protein versus simple sugar intake on weight loss in polycystic ovary syndrome (according to the National Institutes of Health criteria). Fertil Steril. 2009; 92:262–70. https://doi.org/10.1016/j.fertnstert.2008.05.065 [PubMed]
- 50. Klempel MC, Kroeger CM, Bhutani S, Trepanowski JF, Varady KA. Intermittent fasting combined with calorie restriction is effective for weight loss and cardio-protection in obese women. Nutr J. 2012; 11:98. https://doi.org/10.1186/1475-2891-11-98 [PubMed]
- 51. Layman DK, Boileau RA, Erickson DJ, Painter JE, Shiue H, Sather C, Christou DD. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J Nutr. 2003; 133:411–17. [PubMed]
- 52. Layman DK, Evans E, Baum JI, Seyler J, Erickson DJ, Boileau RA. Dietary protein and exercise have additive effects on body composition during weight loss in adult women. J Nutr. 2005; 135:1903–10. [PubMed]
- 53. Nicklas BJ, Wang X, You T, Lyles MF, Demons J, Easter L, Berry MJ, Lenchik L, Carr JJ. Effect of exercise intensity on abdominal fat loss during calorie restriction in overweight and obese postmenopausal women: a randomized, controlled trial. Am J Clin Nutr. 2009; 89:1043–52. https://doi.org/10.3945/ajcn.2008.26938 [PubMed]
- 54. Nørrelund H, Børglum J, Jørgensen JO, Richelsen B, Møller N, Nair KS, Christiansen JS. Effects of growth hormone administration on protein dynamics and substrate metabolism during 4 weeks of dietary restriction in obese women. Clin Endocrinol (Oxf). 2000; 52:305–12. https://doi.org/10.1046/j.1365-2265.2000.00937.x [PubMed]
- 55. Svendsen PF, Jensen FK, Holst JJ, Haugaard SB, Nilas L, Madsbad S. The effect of a very low calorie diet on insulin sensitivity, beta cell function, insulin clearance, incretin hormone secretion, androgen levels and body composition in obese young women. Scand J Clin Lab Invest. 2012; 72:410–19. https://doi.org/10.3109/00365513.2012.691542 [PubMed]
- 56. Cangemi R, Friedmann AJ, Holloszy JO, Fontana L. Long-term effects of calorie restriction on serum sex-hormone concentrations in men. Aging Cell. 2010; 9:236–42. https://doi.org/10.1111/j.1474-9726.2010.00553.x [PubMed]
- 57. Halberg N, Henriksen M, Söderhamn N, Stallknecht B, Ploug T, Schjerling P, Dela F. Effect of intermittent fasting and refeeding on insulin action in healthy men. J Appl Physiol (1985). 2005; 99:2128–36. https://doi.org/10.1152/japplphysiol.00683.2005 [PubMed]
- 58. Baba NH, Sawaya S, Torbay N, Habbal Z, Azar S, Hashim SA. High protein vs high carbohydrate hypoenergetic diet for the treatment of obese hyperinsulinemic subjects. Int J Obes Relat Metab Disord. 1999; 23:1202–06. https://doi.org/10.1038/sj.ijo.0801064 [PubMed]
- 59. Lee K, Lee J, Bae WK, Choi JK, Kim HJ, Cho B. Efficacy of low-calorie, partial meal replacement diet plans on weight and abdominal fat in obese subjects with metabolic syndrome: a double-blind, randomised controlled trial of two diet plans - one high in protein and one nutritionally balanced. Int J Clin Pract. 2009; 63:195–201. https://doi.org/10.1111/j.1742-1241.2008.01965.x [PubMed]
- 60. Belobrajdic DP, Frystyk J, Jeyaratnaganthan N, Espelund U, Flyvbjerg A, Clifton PM, Noakes M. Moderate energy restriction-induced weight loss affects circulating IGF levels independent of dietary composition. Eur J Endocrinol. 2010; 162:1075–82. https://doi.org/10.1530/EJE-10-0062 [PubMed]
- 61. Bhutani S, Klempel MC, Kroeger CM, Trepanowski JF, Varady KA. Alternate day fasting and endurance exercise combine to reduce body weight and favorably alter plasma lipids in obese humans. Obesity (Silver Spring). 2013; 21:1370–79. https://doi.org/10.1002/oby.20353 [PubMed]
- 62. Claessens M, van Baak MA, Monsheimer S, Saris WH. The effect of a low-fat, high-protein or high-carbohydrate ad libitum diet on weight loss maintenance and metabolic risk factors. Int J Obes. 2009; 33:296–304. https://doi.org/10.1038/ijo.2008.278 [PubMed]
- 63. Das SK, Gilhooly CH, Golden JK, Pittas AG, Fuss PJ, Cheatham RA, Tyler S, Tsay M, McCrory MA, Lichtenstein AH, Dallal GE, Dutta C, Bhapkar MV, et al. Long-term effects of 2 energy-restricted diets differing in glycemic load on dietary adherence, body composition, and metabolism in CALERIE: a 1-y randomized controlled trial. Am J Clin Nutr. 2007; 85:1023–30. [PubMed]
- 64. de Luis DA, Izaola O, García Alonso M, Aller R, Cabezas G, de la Fuente B. Effect of a commercial hypocaloric diet in weight loss and post surgical morbidities in obese patients with chronic arthropathy, a randomized clinical trial. Eur Rev Med Pharmacol Sci. 2012; 16:1814–20. [PubMed]
- 65. Fontana L, Klein S, Holloszy JO. Effects of long-term calorie restriction and endurance exercise on glucose tolerance, insulin action, and adipokine production. Age (Dordr). 2010; 32:97–108. https://doi.org/10.1007/s11357-009-9118-z [PubMed]
- 66. Fontana L, Meyer TE, Klein S, Holloszy JO. Long-term calorie restriction is highly effective in reducing the risk for atherosclerosis in humans. Proc Natl Acad Sci USA. 2004; 101:6659–63. https://doi.org/10.1073/pnas.0308291101 [PubMed]
- 67. Fontana L, Weiss EP, Villareal DT, Klein S, Holloszy JO. Long-term effects of calorie or protein restriction on serum IGF-1 and IGFBP-3 concentration in humans. Aging Cell. 2008; 7:681–87. https://doi.org/10.1111/j.1474-9726.2008.00417.x [PubMed]
- 68. Haugaard SB, Vaag A, Mu H, Madsbad S. Skeletal muscle structural lipids improve during weight-maintenance after a very low calorie dietary intervention. Lipids Health Dis. 2009; 8:34. https://doi.org/10.1186/1476-511X-8-34 [PubMed]
- 69. Heilbronn LK, de Jonge L, Frisard MI, DeLany JP, Larson-Meyer DE, Rood J, Nguyen T, Martin CK, Volaufova J, Most MM, Greenway FL, Smith SR, Deutsch WA, et al, and Pennington CALERIE Team. Effect of 6-month calorie restriction on biomarkers of longevity, metabolic adaptation, and oxidative stress in overweight individuals: a randomized controlled trial. JAMA. 2006; 295:1539–48. https://doi.org/10.1001/jama.295.13.1539 [PubMed]
- 70. Heilbronn LK, Smith SR, Martin CK, Anton SD, Ravussin E. Alternate-day fasting in nonobese subjects: effects on body weight, body composition, and energy metabolism. Am J Clin Nutr. 2005; 81:69–73. [PubMed]
- 71. Johnston CS, Tjonn SL, Swan PD. High-protein, low-fat diets are effective for weight loss and favorably alter biomarkers in healthy adults. J Nutr. 2004; 134:586–91. [PubMed]
- 72. Kleiner RE, Hutchins AM, Johnston CS, Swan PD. Effects of an 8-week high-protein or high-carbohydrate diet in adults with hyperinsulinemia. MedGenMed. 2006; 8:39. [PubMed]
- 73. Larson-Meyer DE, Heilbronn LK, Redman LM, Newcomer BR, Frisard MI, Anton S, Smith SR, Alfonso A, Ravussin E. Effect of calorie restriction with or without exercise on insulin sensitivity, beta-cell function, fat cell size, and ectopic lipid in overweight subjects. Diabetes Care. 2006; 29:1337–44. https://doi.org/10.2337/dc05-2565 [PubMed]
- 74. Lejeune MP, Kovacs EM, Westerterp-Plantenga MS. Additional protein intake limits weight regain after weight loss in humans. Br J Nutr. 2005; 93:281–89. https://doi.org/10.1079/BJN20041305 [PubMed]
- 75. Melanson KJ, Summers A, Nguyen V, Brosnahan J, Lowndes J, Angelopoulos TJ, Rippe JM. Body composition, dietary composition, and components of metabolic syndrome in overweight and obese adults after a 12-week trial on dietary treatments focused on portion control, energy density, or glycemic index. Nutr J. 2012; 11:57. https://doi.org/10.1186/1475-2891-11-57 [PubMed]
- 76. Meyer TE, Kovács SJ, Ehsani AA, Klein S, Holloszy JO, Fontana L. Long-term caloric restriction ameliorates the decline in diastolic function in humans. J Am Coll Cardiol. 2006; 47:398–402. https://doi.org/10.1016/j.jacc.2005.08.069 [PubMed]
- 77. Redman LM, Rood J, Anton SD, Champagne C, Smith SR, Ravussin E, and Pennington Comprehensive Assessment of Long-Term Effects of Reducing Intake of Energy (CALERIE) Research Team. Calorie restriction and bone health in young, overweight individuals. Arch Intern Med. 2008; 168:1859–66. https://doi.org/10.1001/archinte.168.17.1859 [PubMed]
- 78. Redman LM, Veldhuis JD, Rood J, Smith SR, Williamson D, Ravussin E, Pennington CT, and Pennington CALERIE Team. The effect of caloric restriction interventions on growth hormone secretion in nonobese men and women. Aging Cell. 2010; 9:32–39. https://doi.org/10.1111/j.1474-9726.2009.00530.x [PubMed]
- 79. Soenen S, Bonomi AG, Lemmens SG, Scholte J, Thijssen MA, van Berkum F, Westerterp-Plantenga MS. Relatively high-protein or ‘low-carb’ energy-restricted diets for body weight loss and body weight maintenance? Physiol Behav. 2012; 107:374–80. https://doi.org/10.1016/j.physbeh.2012.08.004 [PubMed]
- 80. Tapsell L, Batterham M, Huang XF, Tan SY, Teuss G, Charlton K, Oshea J, Warensjö E. Short term effects of energy restriction and dietary fat sub-type on weight loss and disease risk factors. Nutr Metab Cardiovasc Dis. 2010; 20:317–25. https://doi.org/10.1016/j.numecd.2009.04.007 [PubMed]
- 81. Lasker DA, Evans EM, Layman DK. Moderate carbohydrate, moderate protein weight loss diet reduces cardiovascular disease risk compared to high carbohydrate, low protein diet in obese adults: A randomized clinical trial. Nutr Metab (Lond). 2008; 5:30. https://doi.org/10.1186/1743-7075-5-30 [PubMed]
- 82. Weiss EP, Racette SB, Villareal DT, Fontana L, Steger-May K, Schechtman KB, Klein S, Holloszy JO, and Washington University School of Medicine CALERIE Group. Improvements in glucose tolerance and insulin action induced by increasing energy expenditure or decreasing energy intake: a randomized controlled trial. Am J Clin Nutr. 2006; 84:1033–42. [PubMed]
- 83. Westerterp-Plantenga MS, Lejeune MP, Nijs I, van Ooijen M, Kovacs EM. High protein intake sustains weight maintenance after body weight loss in humans. Int J Obes Relat Metab Disord. 2004; 28:57–64. https://doi.org/10.1038/sj.ijo.0802461 [PubMed]
- 84. Wycherley TP, Noakes M, Clifton PM, Cleanthous X, Keogh JB, Brinkworth GD. A high-protein diet with resistance exercise training improves weight loss and body composition in overweight and obese patients with type 2 diabetes. Diabetes Care. 2010; 33:969–76. https://doi.org/10.2337/dc09-1974 [PubMed]
- 85. Yoshimura E, Kumahara H, Tobina T, Matsuda T, Ayabe M, Kiyonaga A, Anzai K, Higaki Y, Tanaka H. Lifestyle intervention involving calorie restriction with or without aerobic exercise training improves liver fat in adults with visceral adiposity. J Obes. 2014; 2014:197216. https://doi.org/10.1155/2014/197216 [PubMed]
- 86. Sterne JA, Gavaghan D, Egger M. Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. J Clin Epidemiol. 2000; 53:1119–29. https://doi.org/10.1016/S0895-4356(00)00242-0 [PubMed]
- 87. Lettieri-Barbato D, Tomei F, Sancini A, Morabito G, Serafini M. Effect of plant foods and beverages on plasma non-enzymatic antioxidant capacity in human subjects: a meta-analysis. Br J Nutr. 2013; 109:1544–56. https://doi.org/10.1017/S0007114513000263 [PubMed]
- 88. Higgins JP, Green S, and Cochrane Collaboration. 2008. Cochrane handbook for systematic reviews of interventions. Chichester, West Sussex; Hoboken NJ: Wiley-Blackwell.



