Introduction
Prostate cancer (PCa) is the second prevalent malignancy and the fifth leading cause of cancer death among men worldwide, with more than 1.4 million new cases of PCa and 375, 000 deaths each year [1, 2]. Since most PCa patients are diagnosed with localized disease, the survival rate can be closed to 100% through radical prostatectomy and radiation therapy [3–5]. Unfortunately, nearly 10% of PCa patients in underdeveloped regions will develop metastatic disease, with a 5-year survival rate dropping to around 30% [6]. With rapid aging process, PCa is becoming a major urinary malignancy affecting men’s health.
Prostate-specific antigen (PSA) is the most widely recognized prostate-specific biomarker for PCa [7]. However, its sensitivity and specificity for diagnosing PCa are limited [8]. Besides PCa, elevated levels of total PSA (tPSA) can also occur due to benign prostate conditions such as chronic prostatitis, benign prostatic hyperplasia, urinary tract infections, prostate manipulation, and ejaculation. Due to these limitations, it is challenging to establish a universally applicable threshold for diagnosing PCa. There is an urgent need in clinical practice for biomarkers that can accurately diagnose PCa. Prostate biopsy remains the gold standard for confirming PCa [9]. Patients with tPSA >10 ng/mL require PCa biopsy, but among men who undergo biopsy due to elevated tPSA levels, only about 25–30% are found to have cancer, while the majority experience false-positive results and undergo unnecessary biopsies [10, 11]. There is still no definitive marker to determine whether PCa patients with tPSA levels between 4–10 ng/mL (PSA gray zone) require biopsy [12]. Therefore, there is a clinical need for biomarkers that can accurately guide PCa biopsy decisions.
Due to PCa being an androgen-dependent cancer, androgen deprivation therapy has become an essential treatment option for locally advanced and metastatic PCa, as well as the basis for a variety of novel combination therapy regimens, and is often required throughout the patient's subsequent treatment [6, 13]. In addition, anti-androgen therapy drugs (AAT) such as enzalutamide (ENZ), bicalutamide (BIC) and apalutamide (ARN), competitively blocking androgen binding to androgen receptors (AR) on prostate cells, are also widely used in advanced PCa [14, 15]. In the initial stage of AAT treatment, most tumors can be significantly inhibited, showing ideal efficacy and decreased PSA [16]. But eventually, as the disease progresses, PCa will recur as castration-resistant prostate cancer (CRPC) [17, 18]. Therefore, identifying remarkable biomarkers related to AAT resistance is advantageous to treatment of PCa and helpful to find novel and viable targets.
Long non-coding RNAs (lncRNAs) are RNA transcripts more than 200 nucleotides in length, with no or limited protein-coding potential and limited evolutionary conservation, but particularly significant on various biological functions in cancer cell, including epigenetic regulation, DNA damage repair and cell-cycle regulation [19–22]. Numerous studies have shown that expression of lncRNAs undergoes dysregulation in PCa, which leads to recurrence, progression and metastasis [23, 24]. lncRNA-PCAT1 is positively linked to CRPC progression by perturbing the PHLPP/FKBP51/IKKα complex and activating AKT and NF-κB signaling [24]. High m6A level of lncRNA-NEAT1-1 is related to promote bone metastasis of PCa, and overexpression of NEAT1-1 actually induces PCa metastasis to lung and bone [25]. Therefore, lncRNAs are also frequently serving as effective biomarkers for assessing PCa prognosis. However, the effect of AAT resistance mediated alteration of lncRNAs on PCa prognosis and diagnosis remain poorly understood, which is worth further study and discovery.
Here, in order to investigate the function of AAT resistance-related lncRNAs (ARR-LncRs) and establish a precise AAT resistance-related risk score model for diagnosing PCa (ARR-RSM), we utilized PCa transcriptional data and clinical data from TCGA database. PSA screening data and serum qPCR data from Sichuan and Chongqing were used to compare the diagnostic performance between ARR-RSM and PSA. Our results will provide reliable evidence for the diagnosis of PCa and will offer personalized AAT treatment options for PCa patients.
Materials and Methods
Clinical blood and tissue sample
From December 2020 to March 2023, 36 PCa adjacent-normal tissues and tumor tissues from patients who underwent tumor excision in the First Affiliated Hospital of Chongqing Medical University were collected, and immediately stored in liquid nitrogen until RNA extraction. From December 2020 to December 2022, during the implementation of the project on PCa screening and intervention in Sichuan and Chongqing, we collected blood samples and PSA screening results from 570 patients in the First Affiliated Hospital of Chongqing Medical University.
PCa screening subjects
The screening subjects were from a total of 47 hospitals in Chongqing and Sichuan. The population standard for PCa screening includes: (1) males in good physical condition with a life expectancy of more than 10 years; (2) males over 50 years of age; males over 45 years of age with a family history of PCa; (3) high-risk individuals including males over 50 years of age; males over 45 years of age with a family history of PCa; males over 40 years of age with a baseline PSA >1 μg/L.
Cell culture and treatment
Normal human prostate epithelial cells (RWPE-1) and PCa cells (LNCaP) were purchased from Shanghai Zhong Qiao Xin Zhou Biotechnology Co., Ltd., (Shanghai, China). LNCaP cells were cultured in RPMI-1640 basal medium (Gibco, USA) supplemented with 10% fetal bovine serum (Procell, Wuhan, China), 100 U/ml penicillin (Beyotime, Beijing, China) and 0.1 mg/ml streptomycin (Beyotime, Beijing, China) and incubated in a humidified cell incubator with 5% CO2 at 37°C. LNCaP cells were treated with ENZ, BIC, and ARN solution (MCE, Shanghai, China) for 24 h to simulate drug therapy, and then collected for further experiments. RWPE-1 cells were cultured in special medium (ZQ-1303) and purchased from Shanghai Zhong Qiao Xin Zhou Biotechnology Co., Ltd. (China).
Cell transfection
SiR-AL354989.1 and siR-AC007405.2 were transfected in LNCaP to silence the expressions of AL354989.1 and AC007405.2 (Tsingke, Beijing, China). The sequences used were: siR-AL354989.1-1 (sense: 5′-GGAAGCUUAUGCAAUAGGA(dT)(dT)-3′, antisense: 5′-UCCUAUUGCAUAAGCUUCC(dT)(dT)-3′); siR-AL354989.1-2 (sense: 5′-GAGGAGGACUGCUAGUCAA(dT)(dT)-3′, antisense: 5′-UUGACUAGCAGUCCUCCUC (dT)(dT)-3′); siR-AC007405.2-1 (sense: 5′-CAGAUCUGUGACCCUGCGA(dT)(dT)-3′, antisense: 5′-UCGCAGGGUCACAGAUCUG(dT)(dT)-3′); siR-AC007405.2-2 (sense: 5′-GGAUUGUUGUAACCGCAAA(dT)(dT)-3′, antisense: 5′-UUUGCGGUUACAACAAUCC(dT)(dT)-3′). For transient transfection of LNCaP, 1 × 105 cells were cultured in T25 flasks. According to the manufacturer’s instruction, LNCaP cells were transfected with 10 μL siRNAs (20 μM) and 5 μL Lipofectamine 3000 (Invitrogen, USA) for 6 h in non-FBS RPMI-1640 basal medium, and then cells were cultured in completed RPMI-1640 medium for 48 h. RT-qPCR was used to verify the silencing efficiencies of siR-AL354989.1 and siR-AC007405.2.
RNA isolation and RT-qPCR
The TRIzol reagent (Abclonal, Wuhan, China) was used to extract total RNA from cells and clinical tissues according to the manufacturer’s instruction. Total RNA was extracted from whole blood using the Whole Blood RNA Isolation Kit (Simgen, Hangzhou, China). 1 μg RNA was reverse transcribed using the PrimeScript RT-qPCR kit (Abclonal, China). RT-qPCR was performed using the SYBR(R) Prime-Script RT-qPCR kit (Abclonal, Wuhan, China) on an ABI 7500 real-time PCR system (Applied Biosystems, USA). The 2−ΔΔCt method was used to calculate the Ct values. The lncRNA values were normalized to the expression levels of β-actin. The primer sequences of lncRNAs and β-actin are shown in Table 1.
Table 1. List of primer sequence used in this study.
| β-actin | Forward | CCTTCCTGGGCATGGAGTC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reverse | TGATCTTCATTGTGCTGGGTG | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AC007405.2 | Forward | CTCCTCTACCTAGCCCCCTG | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reverse | GGGGAAAAGCACTGAGGCAA | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AL354989.1 | Forward | GGCCGAAGACCATAAGTGCT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reverse | AGCTTCCTGAGAGGCTCCTT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: F primer: forward primer; R primer: reverse primer. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Colony formation assay
LNCaP cells under different treatments were seeded at 1000 cells per well in a 6-well plate and incubated at 37°C with 5% CO2 for 10 days until visible colonies formed. After incubation, the cultured cells were washed three times with PBS and fixed with a solution of 4% paraformaldehyde and stained with 0.1% crystal violet for 20 minutes. The number of colonies was counted using ImageJ software after capturing image.
Cell counting kit-8 (CCK-8)
CCK-8 was used for the cells’ proliferation assay by following the manufacturer’s protocol. 2 × 103 LNCaP cells per well were placed in a 96-well plate with 200 μL completed RPMI-1640 medium and cultured in an incubator at 5% CO2 and 37°C. The drug sensitivity test of LNCaP cells was also detected by CCK-8. After treating with enzalutamide, bicalutamide and apalutamide for 48 h, 2 × 103 treated LNCaP cells were cultured in a 96-well plate and cultured in an incubator at 5% CO2 and 37°C. CCK-8 reagent 10 μL (Dojindo, Kumamoto, Japan) was added to each well at 0 h, 6 h, 24 h, 48 h, 72 h and incubated at 37°C for 1.5 h. The absorbance was measured at 450 nm by microplate reader.
PCa transcriptome data of TCGA preprocessing
Transcriptome RNA sequencing and clinical data of 492 PCa tumor tissues and 51 non-tumor samples were downloaded from TCGA data portal (https://portal.gdc.cancer.gov/). PCa patients with an overall survival (OS) ≤30 days were excluded from the analysis to eliminate the influence of unpredictable factors such as hemorrhage and infection on OS. The original data of PCa patients were collected for subsequent analysis. The transcriptome RNA sequencing results and clinical data of PCa patients were integrated into a matrix file using a merge script written in the Perl programming language (http://www.perl.org/).
Bioinformatics analysis
We evaluated the survival rate of patients in the ARR-RSM using the survival package. Receiver operating characteristic (ROC) curves were generated, and the area under the curve (AUC) was calculated using the survival ROC package to assess the accuracy of the ARR-RSM. Multivariate and univariate Cox regression analysis was conducted to confirm the independent prognostic factors for PCa patients. To predict the survival rate of PCa patients, we utilized the nomogram provided by the rms package. Kyoto Encyclopedia of Genes and Genomes (KEGG) in gene set enrichment analysis (GSEA) was employed to identify the underlying pathways associated with ARR-RSM.
Statistical analysis
Statistical analysis was performed using SPSS version 21.0 (SPSS, USA) and GraphPad Prism 8.0 (GraphPad Software Inc., USA). The data are presented as means ± SD. The correlation between the expression levels of sARR-LncRs and clinical features was evaluated using Fisher’s exact probability method. Student’s t-test, ANOVA, and post-hoc tests were employed for comparing differences between two or more groups. P < 0.05 was considered statistically significant.
Availability of data and materials
Authors can provide all data sets analyzed during the study on reasonable requirements.
Results
Establishing the ARR-RSM by sARR-LncRs
We obtained transcriptome RNA-sequencing data and clinical data of PCa patients from TCGA database. After that, we utilized the GSE211781 of GEO Database to screen out 985 AAT resistance-related genes (Figure 1). We found that there were 996 lncRNAs associated with AAT resistance by Pearson correlation analysis (|r| > 0.7 and P < 0.05). In parallel, to investigate the relationship between ARR-LncRs and the prognosis of PCa patients, univariate Cox regression analysis was performed and the detailed results were illustrated in the forest map (Figure 2), in which AL354989.1, AL391427.1, SLC25A25-AS1, KRTAP5-AS1 and AC007405.2 were regarded as a remarkable deleterious factor. Next, we established the ARR-RSM by employing multivariate Cox regression analysis. PCa patients were divided into the high-risk and low-risk groups based on the median risk score (Figure 3A). The mortality of PCa patients was positively correlated with the increase of risk score (Figure 3B). In the high-risk group, the expression of AL354989.1 and AC007405.2 were remarkably increased (Figure 3C). Moreover, being revealed by Kaplan-Meier survival curve, PCa patients in the high-risk group showed poorer prognoses (Figure 4A). The high expressions of AL354989.1 and AC007405.2 were significantly related with PCa patients’ poor prognosis (Figure 4B, 4C).
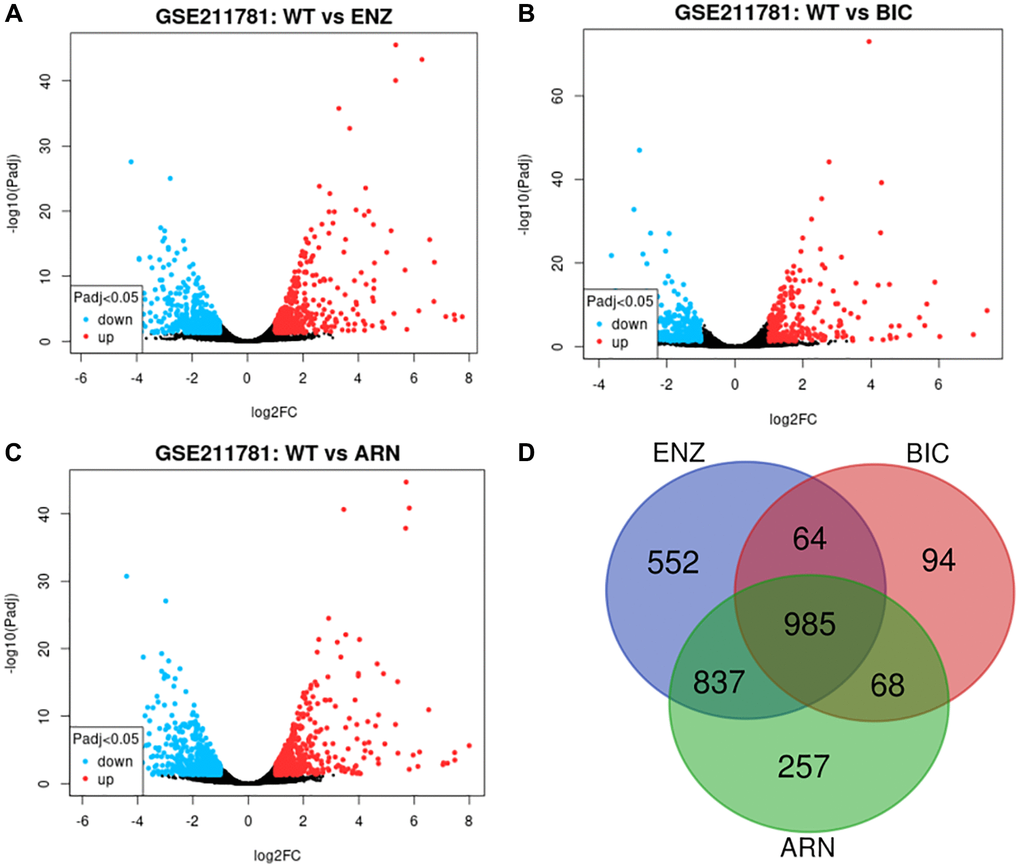
Figure 1. AAT resistance-related gene selected from GSE211781 dataset. The differentially expressed genes between ENZ resistance (A), BIC resistance (B), ARN resistance (C) and WT LNCap cell, log2|FC|≥1, P < 0.05. Venn diagram showed the intersection genes of these differentially expressed genes (D).
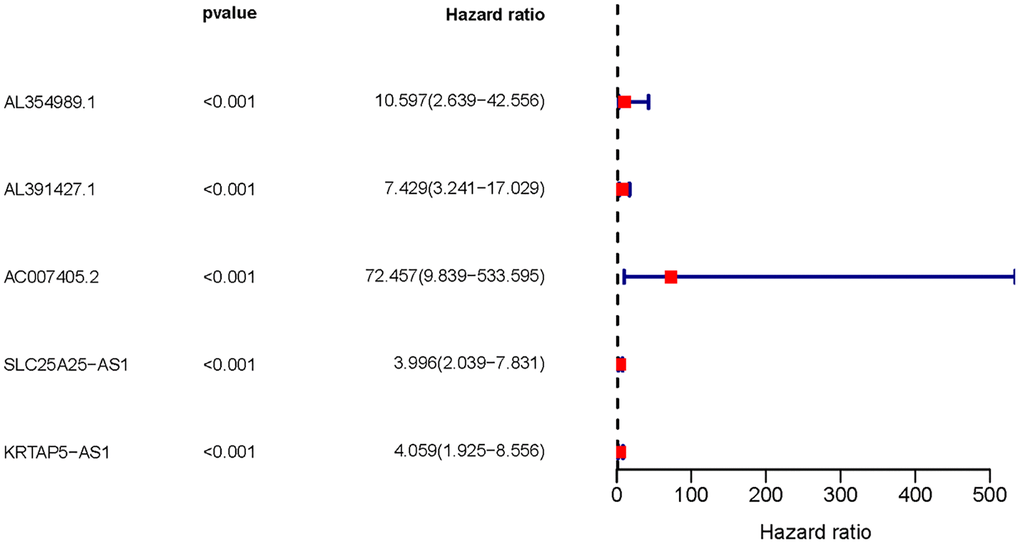
Figure 2. The forest plot of sARR-LncRs. The forest plot showed the hazard ratios of AL354989.1, AL391427.1, AC007405.2, SLC25A25-AS1, and KRTAP5-AS1.
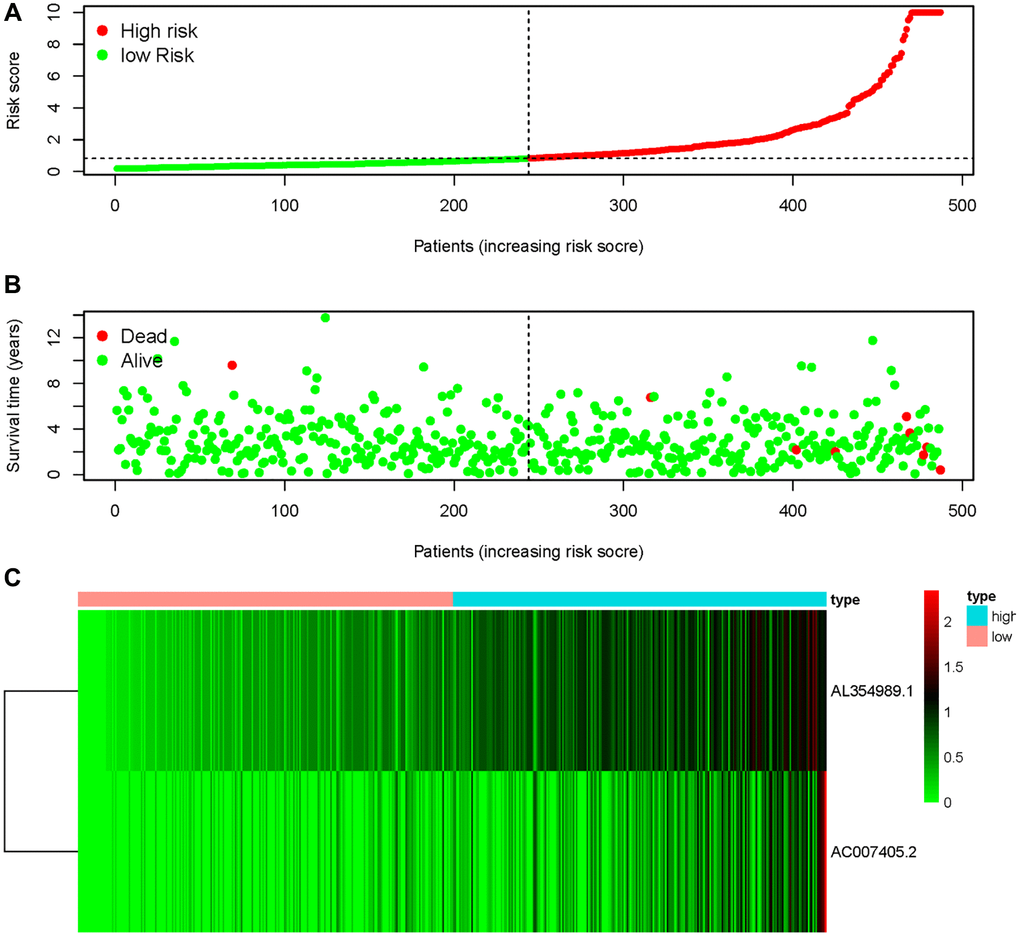
Figure 3. AAT resistance-related risk score model. (A) The risk score of ARR-RSM and the high-risk group were located on the right by the median risk score. (B) The survival status of PCa patients with different risk score. (C) The heatmap of expression of AL354989.1 and AC007405.2 in the ARR-RSM.
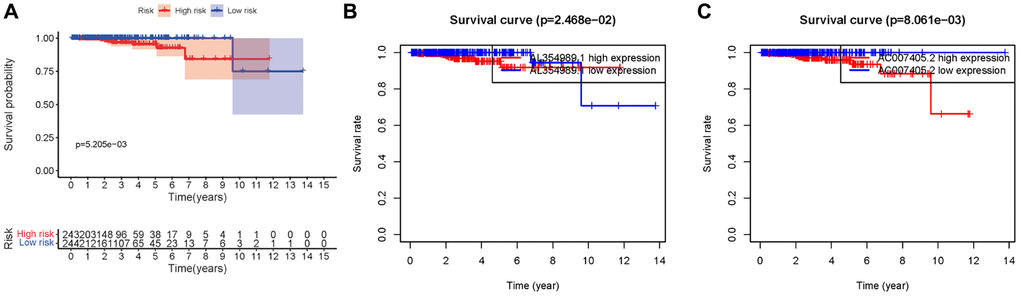
Figure 4. Kaplan-Meier survival curve of ARR-RSM. (A) Kaplan-Meier survival curve of the high-risk group and low-risk group in ARR-RSM. (B, C) The survival curves of sARR-LncRs (AL354989.1 and AC007405.2).
Clinical correlation of sARR-LncRs and potential mechanism of ARR-RSM
Given that sARR-LncRs may suggest a poor prognosis, we used ggpubr package to investigate the correlation between ARR-RSM, sARR-LncRs, and clinicopathologic features of PCa patients, especially T-stage and N-stage. The high-risk score correlated with the advanced T-stage and N-stage (Figure 5A, 5C). The expressions of AC007405.2 were significantly high in advanced T-stage and N-stage, compared to early T-stage and N-stage (Figure 5B, 5D). Univariate and multivariate analysis was performed to explore the potential independent risk factor of PCa patients, revealing that risk score was the only independent risk factor of PCa patients (Table 2). Moreover, we focused on the accuracy of ARR-RSM. The AUCs for ROC curves of ARR-RSM and clinical characters were figured out, which showed that the AUCs of 1-, 3- and 5-year risk scores were 0.984, 0.964 and 0.942, respectively (Figure 6A–6C). Altogether these data suggested that the most accurate and valuable independent risk factor of PCa patients was the ARR-RSM. Following that, ranging from 0 to 100, the points of ARR-RSM were normalized. Drawing the ARR-LncRs expressions line between the total points axis and each prognosis axis, the 1-year, 3-year and 5-year survival probabilities were calculated (Figure 7A). These data therefore indicated that, for predicting the prognoses of PCa patients, ARR-RSM was able to be employed as a risk factor. To further investigate the potential mechanism of ARR-RSM, we then performed the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis of GSEA. As exhibited in Figure 7B, JAK-STAT signaling pathway was the most closely related one to the high-risk group. These results encourage us to further verify the function of sARR-LncRs by basic experiment and examine the association between ARR-RSM and PCa diagnosis and prognosis in clinical samples.
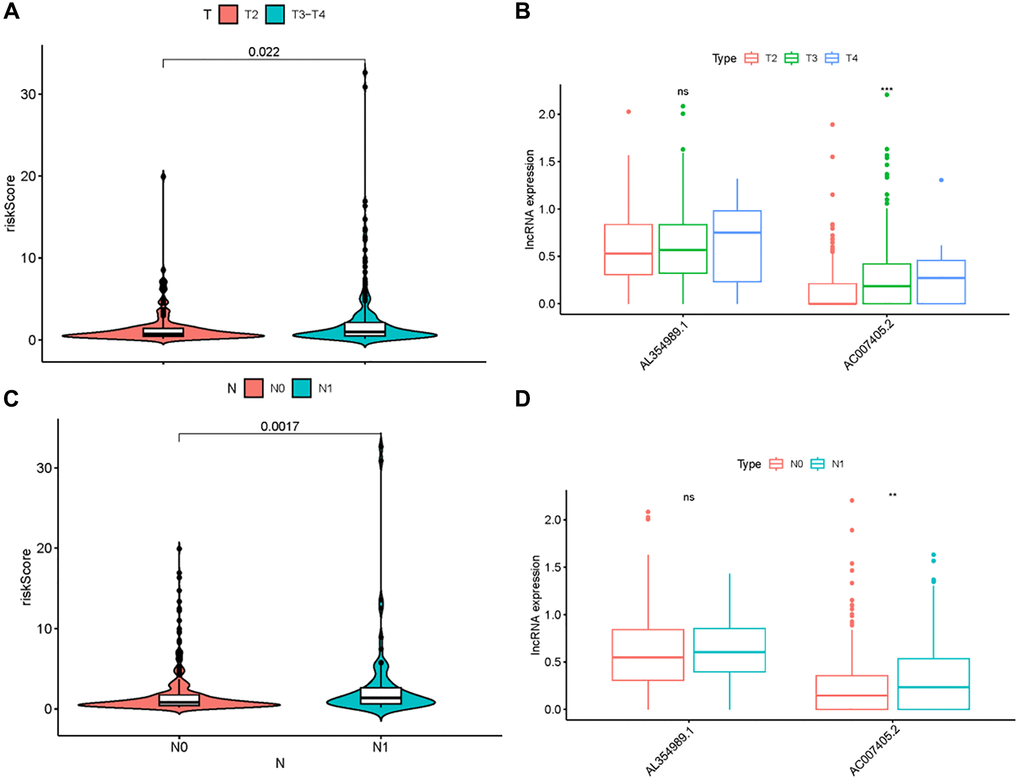
Figure 5. Clinical correlation analysis of ARR-RSM, AL354989.1 and AC007405.2. The relations between the expression levels of risk score, AL354989.1 and AC007405.2 with T-stage (A, B) and N-stage (C, D).
Table 2. Univariate and multivariate Cox analysis of PCa patients.
| Variables | Univariate analysis | Multivariate analysis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR | HR 95% low | HR 95% high | P-value | HR | HR 95% low | HR 95% high | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.060496 | 0.953979 | 1.178907 | 0.276770 | 1.070722 | 0.960997 | 1.192975 | 0.215432 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T-stage | 1.961986 | 0.453625 | 8.485833 | 0.367054 | 1.180598 | 0.227758 | 6.119700 | 0.843240 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N-stage | 3.436471 | 0.761458 | 15.50883 | 0.108378 | 3.965234 | 0.753114 | 20.87741 | 0.104074 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Risk score | 1.030819 | 0.997738 | 1.064998 | 0.038166 | 1.035487 | 1.000663 | 1.071524 | 0.045719 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviation: HR: Hazard Ratio. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
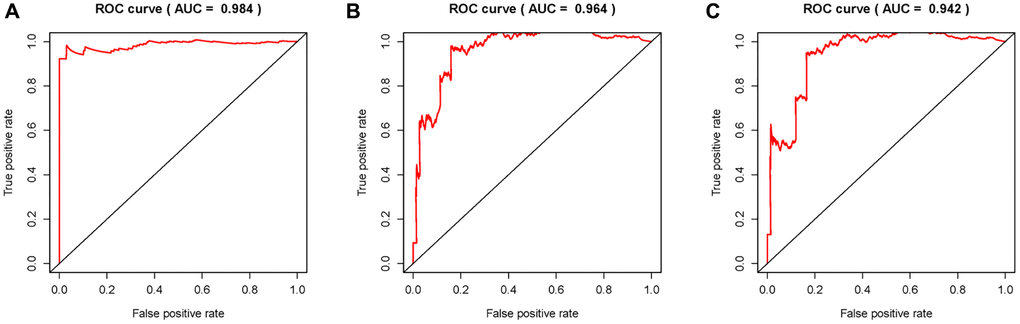
Figure 6. Receiver operating characteristic (ROC) curves. Area under curves (AUCs) of 1-, 3- and 5-year ARR-RSM and clinical features. The 1-, 3- and 5-year AUCs’ values of ARR-RSM were 0.984 (A), 0.964 (B) and 0.942 (C) respectively.
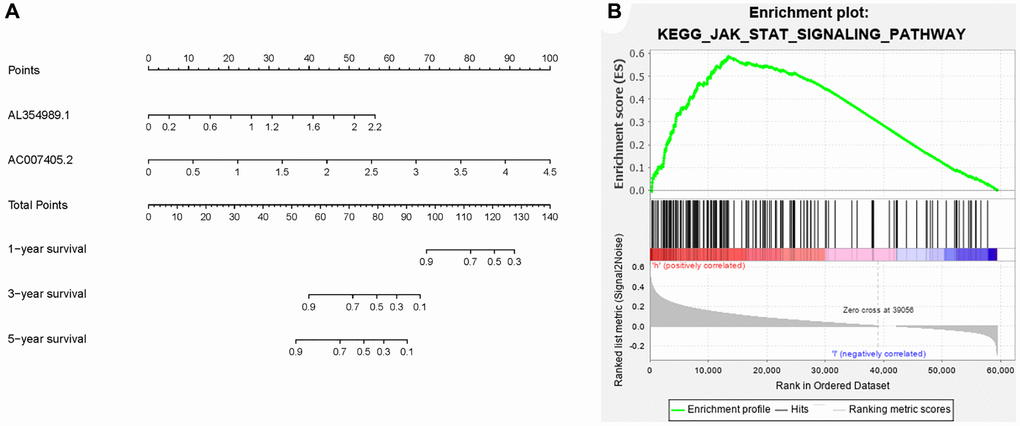
Figure 7. Clinical application of ARR-RSM and underlying pathway. (A) The nomogram of ARR-RSM could predict 1-, 3- and 5-year survival probabilities of PCa patients by detecting the expressions of sARR-LncRs. (B) JAK-STAT pathway was the most enriched pathway in ARR-RSM by KEGG analysis.
PCa screening data
A total of 16,746 men in Chongqing and Sichuan underwent PSA screening. The overall median age was 64.0 years (ranging from 45 to 89 years), with an average age of 63.63 years. The median total PSA level was 2.00 ng/ml (ranging from 0.01 to 102.00 ng/ml), with an average PSA level of 2.45 ng/ml. Among them, there were 1,005 cases (6%) with abnormal PSA levels. The median PSA level for those with abnormal PSA levels was 6.10 ng/ml (ranging from 4.0 to 102 ng/ml), with an average level of 10.55 ng/ml (Supplementary Figure 1A). Among the 570 men tested at the First Affiliated Hospital of Chongqing Medical University, 52 cases (9.12%) had abnormal PSA levels (Supplementary Figure 1B). Among the men with abnormal PSA levels at the First Affiliated Hospital of Chongqing Medical University, 42 cases agreed to undergo prostate biopsy, resulting in a compliance rate of 80.77% (42/52). Among them, 11 cases were diagnosed with PCa, resulting in a positivity rate of 26.19% (11/42) (Supplementary Figure 1C).
AC007405.2 and AL354989.1 obtain remarkable clinical significance and relevance
In order to investigate the expression levels of AC007405.2 and AL354989.1 in PCa, we respectively detected AC007405.2 and AL354989.1 in LNCaP cells and then compared them with normal prostate cells RWPE-1. Expectedly, the expression levels of AC007405.2 and AL354989.1 in LNCaP were significantly higher than RWPE-1 (Figure 8A–8B). The same results were confirmed by comparing 36 paired PCa tissues with adjacent normal tissues. The expressions of AC007405.2 and AL354989.1 in PCa tissues were remarkable higher than that in adjacent normal tissues (Figure 8C, 8D). We performed RT-qPCR to measure the expression levels of AC007405.2 and AL354989.1 in the blood samples of patients with tPSA >4 ng/ml. Based on the prostate biopsy results of patients (Supplementary Table 3) we generated ROC curves using the risk score. As shown in Figure 8E, 8F, the accuracy of diagnosing PCa using the risk score (0.877) is significantly higher than using tPSA (0.815) These findings indicate that by measuring the expression levels of AC007405.2 and AL354989.1 in patients’ blood and calculating the risk score, it is possible to more accurately predict whether a patient has PCa. This provides a novel and feasible diagnostic and predictive indicator for precise PCa diagnosis in clinical settings.
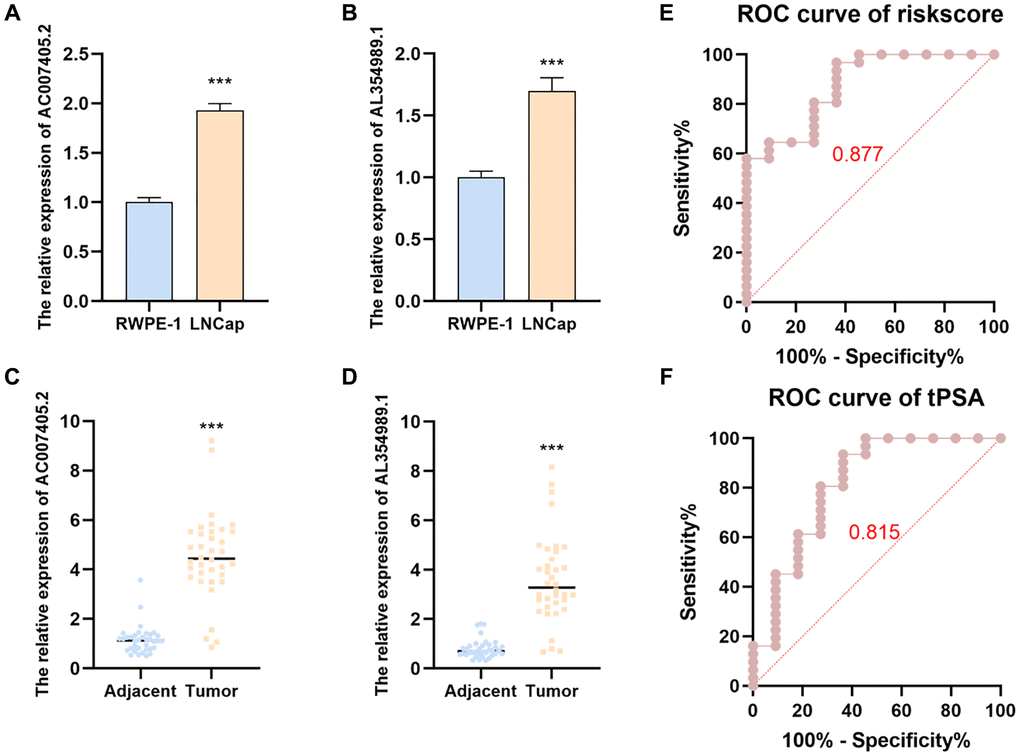
Figure 8. The expression levels and predicted function of AL354989.1 and AC007405.2 in PCa cell lines and clinical samples. AL354989.1 and AC007405.2 expressed higher in LNCaP (A, B) cells and PCa tissues (C, D) than that in prostate normal tissue and RWPE-1 cells, respectively. ROC curves were generated to predict the occurrence of PCa in patients based on the expression levels of AL354989.1 and AC007405.2 in blood samples (E), as well as tPSA values (F).
AC007405.2 and AL354989.1 prominently facilitated the proliferation and AAT resistance of PCa cells
We first performed siRNAs to knockdown the expression of AC007405.2 (Figure 9A) and AL354989.1 (Figure 9B) and investigated the roles of AC007405.2 and AL354989.1 in proliferation of PCa. The results revealed that both siR-AC007405.2 (Figure 9C–9E) and siR-AL354989.1 (Figure 9F–9H) significantly inhibited the proliferation capacity of LNCaP cells. In order to investigate the role of AC007405.2 and AL354989.1 in the process of AAT resistance, we treated LNCaP cells with ENZ, ARN and BIC, respectively. We then detected the expression levels of AC007405.2 and AL354989.1 by RT-qPCR and results showed that AC007405.2 and AL354989.1 were increased by AAT treatment (Figure 10A, 10B). Following that, we silenced AC007405.2 and AL354989.1 by siRNAs and treated with different concentrations of ENZ, ARN, and BIC separately. The results showed that silencing AC007405.2 and AL354989.1 significantly decreased the relative cell activity in the treatments of ENZ (Figure 10C, 10D), ARN (Figure 10E, 10F), and BIC (Figure 10G, 10H). Herein, these results will provide predictions for the clinical efficacy of AAT treatment in PCa and offer novel potential targets for targeted therapy in PCa.
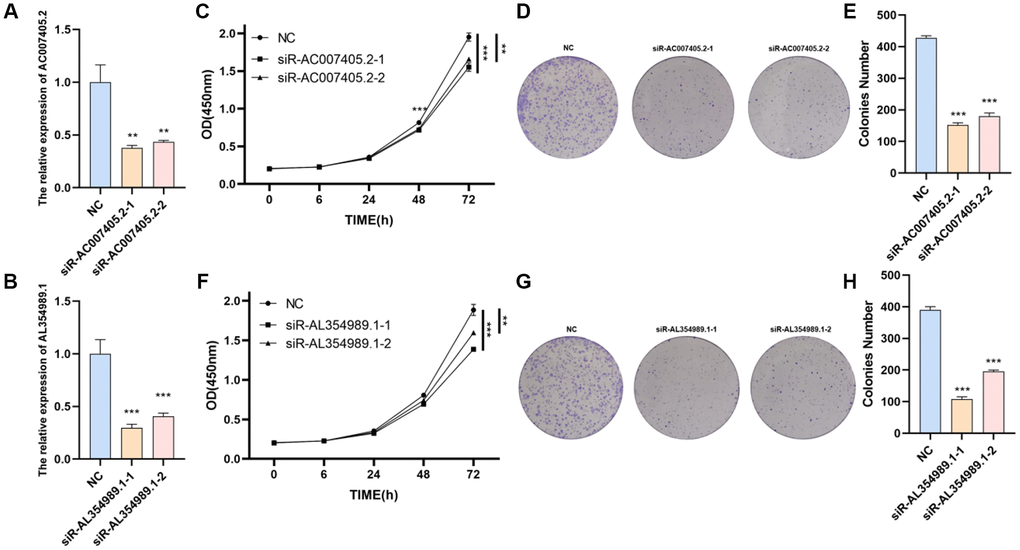
Figure 9. The effects of AL354989.1 and AC007405.2 on proliferation in LNCaP cells. The silencing efficiency of siR-AC007405.2 (A) and siR-AL354989.1 (B). The CCK-8 and clone formation assays showed that silencing of AC007405.2 (C–E) and AL354989.1 (F–H) could decrease the proliferation ability of LNCaP cells.
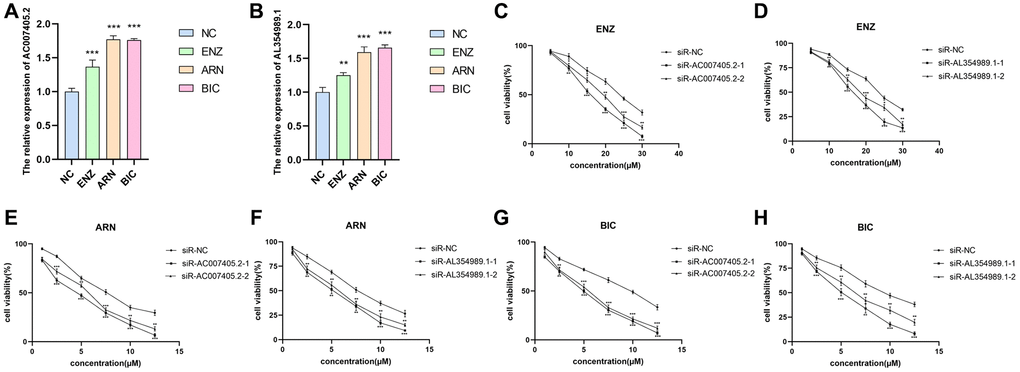
Figure 10. The effects of AL354989.1 and AC007405.2 on AAT resistance in LNCaP cells. The expression levels of AC007405.2 (A) and AL354989.1 (B) were increased with the treatment of ENZ, ARN and BIC, respectively. The drug sensitivity curves of siR-AC007405.2 and siR-AL354989.1 in LNCaP cells under ENZ (C, D), ARN (E, F) and BIC (G, H) treatment, separately.
Discussion
PCa is a prevalent male cancer globally and is increasingly recognized as a significant health concern for middle-aged and elderly men [26]. Timely screening, early diagnosis, and targeted treatment for high-risk individuals are crucial strategies for enhancing the survival rate of PCa [10]. Since the 1980s, PSA combined with digital rectal examination (DRE) has been widely used as an early screening method for PCa, which has significantly improved the early diagnosis rate of PCa. Nevertheless, many other factors, such as prostatitis, benign prostatic hyperplasia, and cycling can cause elevated PSA levels, leading to a high false-positive rate. Furthermore, there exists a gray zone, tPSA levels between 4–10 ng/mL, for prostate biopsy. These false-positive PSA results can lead to patients undergoing unnecessary invasive prostate biopsies [27]. AAT plays a mainstay role in prostate anti-AR therapy. Most patients develop resistance to anti-androgen therapy during long-term treatment, leading to the malignant progression of PCa [28]. Therefore, it is imperative to identify non-invasive molecular biomarkers that can accurately diagnose PCa and evaluate the prognosis of PCa patients, while also providing potential molecular targets for enhancing the therapeutic effect of AAT in PCa.
In response to the current bottleneck in PCa diagnosis and treatment, an increasing number of researchers are beginning to explore molecular biomarkers for diagnosis and treatment. Kuci Emruli et al. conducted a study in which they measured 157 analytes in 363 serum samples from healthy individuals, patients with non-metastatic PCa, and patients with metastatic PCa. They used a recombinant antibody microarray technique. The researchers identified a signature consisting of 69 proteins that could differentiate metastatic PCa patients from healthy controls [29]. The predictive model based on ARG developed by the Hu Dai-Xing team is a reliable prognostic and predictive tool for overall survival and disease-free survival in PCa patients [30]. However, there is still limited research on the predictive role of lncRNAs in the prognosis of PCa patients. In our study, we utilized multivariate Cox regression analysis to construct an ARR-RSM based on the expression levels of AC007405.2 and AL354989.1. This model accurately predicts the prognosis of PCa patients, with ROC values of 0.984, 0.964, and 0.942 for 1, 3, and 5 years, respectively. Its accuracy is significantly higher than risk models constructed by other researchers. Furthermore, we found a significant association between our ARR-RSM and clinical T-stage and N-stage. High riskscore is often associated with poorer prognosis, higher T-stage, and N-stage. Based on our clinical screening of PSA and blood expression levels of AC007405.2 and AL354989.1, we found that ARR-RSM is a more accurate diagnostic biomarker for PCa. RT-qPCR results revealed that AC007405.2 and AL354989.1 are highly expressed in PCa cells (LNCaP) and clinical PCa tissues.
Previously, multiple high-quality studies have reported the pivotal regulatory role of lncRNAs in PCa. Dysregulation of lncRNA OIP5-AS1 expression could mediate PCa cell growth and cadmium-induced ferroptosis. Mechanistically, OIP5-AS1 served as an endogenous sponge of miR-128-3p to regulate the expression of SLC7A11, a surrogate marker of ferroptosis [31]. LncRNA MEG3 was a down-regulated lncRNA in PCa tissues and cells and could inhibit the expression of miR-9-5p, whereas miR-9-5p down-regulated QKI-5 expression. Overexpressed MEG3 and QKI-5 could decrease the abilities of proliferation, migration and invasion in PCa cells effectively and increased the apoptosis rate [32]. LncRNA DSCAM-AS1 was transcriptionally activated by super-enhancers driven by FOXA1 and exhibited lineage-specific expression pattern. DSCAM-AS1 could promote cancer progression by interacting with YBX1 and regulating expression of FOXA1 and ERα [33]. Yuliang Wang et al. indicated that FRGPI is not only a promising and robust prognostic biomarker but also a potential indicator of immunotherapy outcomes in patients with PCa after radical prostatectomy [34]. At present, researchers have focused on the relationship between lncRNA and AAT resistance. Dong et al. found that the LncRNA SNHG4 promotes PCa cell resistance to ENZ through let-7a/RREB1 positive feedback loop and ceRNA network [35]. Chen et al. found that LncRNA ERVH48-1 activates Wnt/β-catenin pathway through sponging miR-4784, which promotes drug resistance and proliferation of PCa [36]. However, the mechanisms of lncRNAs-mediated AAT resistance remain poorly understood. In our study, we discovered the biological roles of AC007405.2 and AL354989.1 in AAT resistance. SiR-AC007405.2 and siR-AL354989.1 can inhibit the proliferation ability of LNCaP. The qPCR analysis revealed a significant upregulation of AC007405.2 and AL354989.1 in LNCaP cells treated with ENZ, ARN, and BIC. SiR-AC007405.2 and siR-AL354989.1 notably enhanced the apoptotic effects induced by ENZ, ARN, and BIC in LNCaP cells. Recently, it has been reported that the increased JAK/STAT and FGFR signal transduction drive AAT resistance in PCa [37]. Similarly, the results of our KEGG enrichment analysis also suggest that ARR-LncRs is mainly enriched in the JAK/STAT signal pathway, which not only reveals the mechanism of ARR-LncRs, but also further confirms the important role of ARR-LncRs in AAT resistance. In summary, AC007405.2 and AL354989.1 could serve as potential molecular therapeutic targets and enhancers of AAT treatment efficacy in the future.
These findings highlight the role of ARR-RSM in the prognostic assessment of PCa patients and the relationships between ARR-LncRs (AC007405.2 and AL354989.1) and clinical features. By establishing ARR-RSM, it can replace PSA as a new screening indicator for early PCa diagnosis, reducing the mortality and incidence rates of PCa. In addition, we can evaluate the potential risk of AAT treatment resistance in patients before treatment and develop personalized treatment plans based on the predictive results, thereby achieving more accurate medical care. In summary, ARR-RSM is an indispensable tool in clinical practice that can help physicians make more accurate diagnostic and treatment decisions, ultimately improving the quality of life for patients. The use of ARR-RSM to predict the presence of PCa was limited to a single-center cohort. In future studies, we will expand our model to multiple centers to achieve the generalizability of ARR-RSM. Regarding the molecular mechanisms underlying the promotion of PCa proliferation and AAT resistance by AC007405.2 and AL354989.1, we have only conducted KEGG pathway analysis, which identified the JAK-STAT pathway. However, further validation is needed through relevant in vitro and in vivo experiments.
Conclusions
In this study, we have uncovered the clinical significance of AC007405.2 and AL354989.1 in predicting the prognosis and diagnosing patients with PCa. Additionally, we have validated their association with various clinical features. Furthermore, we validated the high expressions of AC007405.2 and AL354989.1 in PCa tissues and cell lines, and demonstrated their ability to promote PCa cell proliferation and resistance to AAT treatment. These findings offer potential targets for the treatment of PCa and provide a novel diagnostic and predictive indicator for accurate PCa diagnosis.
Author Contributions
Yuanzhong Deng, Chunlin Zhang and Haitao Yu wrote the paper, verified the clinical significance of ARR-LncRs, and designed the program. Yang Li and Guo Chen were responsible for data analysis. Wei Shi, Xiang Peng, and Zhenwei Feng performed parts of validation experiments. Xuesong Bai and Guo Chen were responsible for clinical samples collection. Nian Liu and Xin Gou funded the program.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement and Consent
Informed written consents were obtained from all patients and their personal information was kept confidential. This study was performed in accordance with the Declaration of Helsinki and approved by the Medical Ethics Committee of the First Affiliated Hospital of Chongqing Medical University (IRB:2020-754).
Funding
This research was funded by Chongqing Medical Scientific Research Project (Joint Project of Chongqing Health Commission and Science and Technology Bureau), Grant Number 2020ZLXM004; Science and Technology Commission of Yuzhong District of Chongqing, Grant Number 20190104.
References
- 1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 71:209–49. https://doi.org/10.3322/caac.21660 [PubMed]
- 2. Ferlay J, Colombet M, Soerjomataram I, Dyba T, Randi G, Bettio M, Gavin A, Visser O, Bray F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur J Cancer. 2018; 103:356–87. https://doi.org/10.1016/j.ejca.2018.07.005 [PubMed]
- 3. Bach C, Pisipati S, Daneshwar D, Wright M, Rowe E, Gillatt D, Persad R, Koupparis A. The status of surgery in the management of high-risk prostate cancer. Nat Rev Urol. 2014; 11:342–51. https://doi.org/10.1038/nrurol.2014.100 [PubMed]
- 4. Costello AJ. Considering the role of radical prostatectomy in 21st century prostate cancer care. Nat Rev Urol. 2020; 17:177–88. https://doi.org/10.1038/s41585-020-0287-y [PubMed]
- 5. Tilki D, Chen MH, Wu J, Huland H, Graefen M, D'Amico AV. Adjuvant Versus Early Salvage Radiation Therapy After Radical Prostatectomy for pN1 Prostate Cancer and the Risk of Death. J Clin Oncol. 2022; 40:2186–92. https://doi.org/10.1200/JCO.21.02800 [PubMed]
- 6. do Pazo C, Webster RM. The prostate cancer drug market. Nat Rev Drug Discov. 2021; 20:663–4. https://doi.org/10.1038/d41573-021-00111-w [PubMed]
- 7. Van Poppel H, Albreht T, Basu P, Hogenhout R, Collen S, Roobol M. Serum PSA-based early detection of prostate cancer in Europe and globally: past, present and future. Nat Rev Urol. 2022; 19:562–72. https://doi.org/10.1038/s41585-022-00638-6 [PubMed]
- 8. Ilic D, Djulbegovic M, Jung JH, Hwang EC, Zhou Q, Cleves A, Agoritsas T, Dahm P. Prostate cancer screening with prostate-specific antigen (PSA) test: a systematic review and meta-analysis. BMJ. 2018; 362:k3519. https://doi.org/10.1136/bmj.k3519 [PubMed]
- 9. Lenfant L, Barret E, Rouprêt M, Rozet F, Ploussard G, Mozer P, and Cancerology Committee of Association Française d'Urologie (CCAFU). Transperineal Prostate Biopsy Is the New Black: What Are the Next Targets? Eur Urol. 2022; 82:3–5. https://doi.org/10.1016/j.eururo.2022.01.046 [PubMed]
- 10. Mottet N, van den Bergh RCN, Briers E, Van den Broeck T, Cumberbatch MG, De Santis M, Fanti S, Fossati N, Gandaglia G, Gillessen S, Grivas N, Grummet J, Henry AM, et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2021; 79:243–62. https://doi.org/10.1016/j.eururo.2020.09.042 [PubMed]
- 11. Eklund M, Jäderling F, Discacciati A, Bergman M, Annerstedt M, Aly M, Glaessgen A, Carlsson S, Grönberg H, Nordström T, and STHLM3 consortium. MRI-Targeted or Standard Biopsy in Prostate Cancer Screening. N Engl J Med. 2021; 385:908–20. https://doi.org/10.1056/NEJMoa2100852 [PubMed]
- 12. Wang B, Zhang S, Meng J, Min L, Luo J, Zhu Z, Bao H, Zang R, Deng S, Zhang F, Ma L, Wang S. Evaporation-Induced rGO Coatings for Highly Sensitive and Non-Invasive Diagnosis of Prostate Cancer in the PSA Gray Zone. Adv Mater. 2021; 33:e2103999. https://doi.org/10.1002/adma.202103999 [PubMed]
- 13. Lowrance W, Dreicer R, Jarrard DF, Scarpato KR, Kim SK, Kirkby E, Buckley DI, Griffin JC, Cookson MS. Updates to Advanced Prostate Cancer: AUA/SUO Guideline (2023). J Urol. 2023; 209:1082–90. https://doi.org/10.1097/JU.0000000000003452 [PubMed]
- 14. Tran C, Ouk S, Clegg NJ, Chen Y, Watson PA, Arora V, Wongvipat J, Smith-Jones PM, Yoo D, Kwon A, Wasielewska T, Welsbie D, Chen CD, et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science. 2009; 324:787–90. https://doi.org/10.1126/science.1168175 [PubMed]
- 15. Dess RT, Sun Y, Jackson WC, Jairath NK, Kishan AU, Wallington DG, Mahal BA, Stish BJ, Zumsteg ZS, Den RB, Hall WA, Gharzai LA, Jaworski EM, et al. Association of Presalvage Radiotherapy PSA Levels After Prostatectomy With Outcomes of Long-term Antiandrogen Therapy in Men With Prostate Cancer. JAMA Oncol. 2020; 6:735–43. https://doi.org/10.1001/jamaoncol.2020.0109 [PubMed]
- 16. Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, Mason M, Matveev V, Wiegel T, Zattoni F, Mottet N, and European Association of Urology. EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur Urol. 2014; 65:467–79. https://doi.org/10.1016/j.eururo.2013.11.002 [PubMed]
- 17. Cheng Q, Butler W, Zhou Y, Zhang H, Tang L, Perkinson K, Chen X, Jiang XS, McCall SJ, Inman BA, Huang J. Pre-existing Castration-resistant Prostate Cancer-like Cells in Primary Prostate Cancer Promote Resistance to Hormonal Therapy. Eur Urol. 2022; 81:446–55. https://doi.org/10.1016/j.eururo.2021.12.039 [PubMed]
- 18. Mukherji D, Omlin A, Pezaro C, Shamseddine A, de Bono J. Metastatic castration-resistant prostate cancer (CRPC): preclinical and clinical evidence for the sequential use of novel therapeutics. Cancer Metastasis Rev. 2014; 33:555–66. https://doi.org/10.1007/s10555-013-9473-1 [PubMed]
- 19. Lin W, Zhou Q, Wang CQ, Zhu L, Bi C, Zhang S, Wang X, Jin H. LncRNAs regulate metabolism in cancer. Int J Biol Sci. 2020; 16:1194–206. https://doi.org/10.7150/ijbs.40769 [PubMed]
- 20. Herman AB, Tsitsipatis D, Gorospe M. Integrated lncRNA function upon genomic and epigenomic regulation. Mol Cell. 2022; 82:2252–66. https://doi.org/10.1016/j.molcel.2022.05.027 [PubMed]
- 21. Zhu H, Chen K, Chen Y, Liu J, Zhang X, Zhou Y, Liu Q, Wang B, Chen T, Cao X. RNA-binding protein ZCCHC4 promotes human cancer chemoresistance by disrupting DNA-damage-induced apoptosis. Signal Transduct Target Ther. 2022; 7:240. https://doi.org/10.1038/s41392-022-01033-8 [PubMed]
- 22. Wang Z, Yang B, Zhang M, Guo W, Wu Z, Wang Y, Jia L, Li S, Xie W, Yang D, and Cancer Genome Atlas Research Network. lncRNA Epigenetic Landscape Analysis Identifies EPIC1 as an Oncogenic lncRNA that Interacts with MYC and Promotes Cell-Cycle Progression in Cancer. Cancer Cell. 2018; 33:706–20.e9. https://doi.org/10.1016/j.ccell.2018.03.006 [PubMed]
- 23. Mirzaei S, Paskeh MDA, Okina E, Gholami MH, Hushmandi K, Hashemi M, Kalu A, Zarrabi A, Nabavi N, Rabiee N, Sharifi E, Karimi-Maleh H, Ashrafizadeh M, et al. Molecular Landscape of LncRNAs in Prostate Cancer: A focus on pathways and therapeutic targets for intervention. J Exp Clin Cancer Res. 2022; 41:214. https://doi.org/10.1186/s13046-022-02406-1 [PubMed]
- 24. Shang Z, Yu J, Sun L, Tian J, Zhu S, Zhang B, Dong Q, Jiang N, Flores-Morales A, Chang C, Niu Y. LncRNA PCAT1 activates AKT and NF-κB signaling in castration-resistant prostate cancer by regulating the PHLPP/FKBP51/IKKα complex. Nucleic Acids Res. 2019; 47:4211–25. https://doi.org/10.1093/nar/gkz108 [PubMed]
- 25. Wen S, Wei Y, Zen C, Xiong W, Niu Y, Zhao Y. Long non-coding RNA NEAT1 promotes bone metastasis of prostate cancer through N6-methyladenosine. Mol Cancer. 2020; 19:171. https://doi.org/10.1186/s12943-020-01293-4 [PubMed]
- 26. Koga Y, Song H, Chalmers ZR, Newberg J, Kim E, Carrot-Zhang J, Piou D, Polak P, Abdulkadir SA, Ziv E, Meyerson M, Frampton GM, Campbell JD, Huang FW. Genomic Profiling of Prostate Cancers from Men with African and European Ancestry. Clin Cancer Res. 2020; 26:4651–60. https://doi.org/10.1158/1078-0432.CCR-19-4112 [PubMed]
- 27. Chen Y, Fan Y, Yang Y, Jin J, Zhou L, He Z, Zhao Z, He Q, Wang X, Yu W, Wu S. Are prostate biopsies necessary for all patients 75years and older? J Geriatr Oncol. 2018; 9:124–9. https://doi.org/10.1016/j.jgo.2017.09.001 [PubMed]
- 28. Kang J, La Manna F, Bonollo F, Sampson N, Alberts IL, Mingels C, Afshar-Oromieh A, Thalmann GN, Karkampouna S. Tumor microenvironment mechanisms and bone metastatic disease progression of prostate cancer. Cancer Lett. 2022; 530:156–69. https://doi.org/10.1016/j.canlet.2022.01.015 [PubMed]
- 29. Kuci Emruli V, Liljedahl L, Axelsson U, Richter C, Theorin L, Bjartell A, Lilja H, Donovan J, Neal D, Hamdy FC, Borrebaeck CAK. Identification of a serum biomarker signature associated with metastatic prostate cancer. Proteomics Clin Appl. 2021; 15:e2000025. https://doi.org/10.1002/prca.202000025 [PubMed]
- 30. Hu D, Jiang L, Luo S, Zhao X, Hu H, Zhao G, Tang W. Development of an autophagy-related gene expression signature for prognosis prediction in prostate cancer patients. J Transl Med. 2020; 18:160. https://doi.org/10.1186/s12967-020-02323-x [PubMed]
- 31. Zhang Y, Guo S, Wang S, Li X, Hou D, Li H, Wang L, Xu Y, Ma B, Wang H, Jiang X. LncRNA OIP5-AS1 inhibits ferroptosis in prostate cancer with long-term cadmium exposure through miR-128-3p/SLC7A11 signaling. Ecotoxicol Environ Saf. 2021; 220:112376. https://doi.org/10.1016/j.ecoenv.2021.112376 [PubMed]
- 32. Wu M, Huang Y, Chen T, Wang W, Yang S, Ye Z, Xi X. LncRNA MEG3 inhibits the progression of prostate cancer by modulating miR-9-5p/QKI-5 axis. J Cell Mol Med. 2019; 23:29–38. https://doi.org/10.1111/jcmm.13658 [PubMed]
- 33. Zhang Y, Huang YX, Wang DL, Yang B, Yan HY, Lin LH, Li Y, Chen J, Xie LM, Huang YS, Liao JY, Hu KS, He JH, et al. LncRNA DSCAM-AS1 interacts with YBX1 to promote cancer progression by forming a positive feedback loop that activates FOXA1 transcription network. Theranostics. 2020; 10:10823–37. https://doi.org/10.7150/thno.47830 [PubMed]
- 34. Wang Y, Fan J, Chen T, Xu L, Liu P, Xiao L, Wu T, Zhou Q, Zheng Q, Liu C, Chan FL, Wu D. A novel ferroptosis-related gene prognostic index for prognosis and response to immunotherapy in patients with prostate cancer. Front Endocrinol (Lausanne). 2022; 13:975623. https://doi.org/10.3389/fendo.2022.975623 [PubMed]
- 35. Dong Q, Qiu H, Piao C, Li Z, Cui X. LncRNA SNHG4 promotes prostate cancer cell survival and resistance to enzalutamide through a let-7a/RREB1 positive feedback loop and a ceRNA network. J Exp Clin Cancer Res. 2023; 42:209. https://doi.org/10.1186/s13046-023-02774-2 [PubMed]
- 36. Chen B, Xu K, Zhang Y, Xu P, Li C, Liu J, Xu Y. LncRNA ERVH48-1 Contributes to the Drug Resistance of Prostate Cancer and Proliferation through Sponging of miR-4784 to the Activation of the Wnt/β-Catenin Pathway. Cancers (Basel). 2023; 15:1902. https://doi.org/10.3390/cancers15061902 [PubMed]
- 37. Chan JM, Zaidi S, Love JR, Zhao JL, Setty M, Wadosky KM, Gopalan A, Choo ZN, Persad S, Choi J, LaClair J, Lawrence KE, Chaudhary O, et al. Lineage plasticity in prostate cancer depends on JAK/STAT inflammatory signaling. Science. 2022; 377:1180–91. https://doi.org/10.1126/science.abn0478 [PubMed]



