Introduction
With over one million new cases annually, gastric cancer (GC) is the fifth most diagnosed malignancies globally [1]. Moreover, a recent study reported the incidences of GC increased significantly in the younger generation [2]. GC is often associated with unfavorable outcomes, currently it is the third most common cause of cancer-related deaths [1]. As a heterogeneous, complex and multifactorial disease; the inter-patient, intra-patient, and intra-tumoral heterogeneities in GC were crucial barriers in treatment determination [1, 3, 4]. For example, due to the biological differences between tumors from Western and Eastern countries, it is difficult to identify an international accepted standard-of-care therapy [3]. In Asia, surgery plus adjuvant chemotherapy is more frequent choice, while neoadjuvant chemotherapy/radiotherapy is preferred outside of Asia [1, 3, 4]. Now it is generally accepted that the optimal treatment is dependent on the genomic and molecular characteristics of GC. Remarkably, both two well-known proposals, The Cancer Genome Atlas (TCGA) in the US [5] and the Asian Cancer Research Group (ACRG) in Asia [6], established microsatellite instability-high (MSI-H) as a distinct subgroup of GC.
Microsatellites are short and repetitive DNA sequences that distributed randomly through the whole genome. Tumors with MSI prone to a high mutation rate as consequence of a deficient DNA mismatch repair (dMMR) machinery [7]. The high frequencies of gene mutations can induce the presence of neoantigens and a peculiar immunological microenvironment. In fact, MSI/dMMR has emerged as a tumor-agnostic biomarker for immunotherapy since its approval by the US Food and Drug Administration (FDA) in 2017 [7, 8]. However, giving approximately 97% of tumors were microsatellite instability-low/microsatellite instability-stable (MSI-L/MSS) [9], MSI test is not always conducted in real-world practice. Indeed, current guidelines only recommend MSI testing for colorectal and endometrial cancers in Europe [10, 11]. Although the utility of MSI status may help to identify the most effective treatment, the examination of MSI/dMMR during routine diagnostic activity was not recommended in GC partly because there were no reports regarding the prevalence of MSI-H GC worldwide or in various regions. On the other hand, although the association between MSI-H and various clinicopathological factors or the efficacy of treatments have been examined in GC [1, 3, 12], the results were often ambiguous or conflicted due to the biological complexity of GC. Moreover, there are many inconsistent results due to the limited patients enrolled and/or different methods for measuring MSI. Hence, a comprehensive overview of MSI-H GC could have both basic and clinical importance considering no single study has adequate power to draw any solid conclusions.
Here, with accumulating evidence available, we collected 43246 GC patients from 134 studies and carried out a pooled analysis to estimate the overall proportion of patients with MSI-H GC. To evaluate the performances of different MSI testing method, we investigated the prevalence of MSI-H GC examined by polymerase chain reaction (PCR), immunohistochemistry (IHC), and next-generation sequencing (NGS), respectively. Next, we estimated the incidences of five potential epidemiological and risk factors in patients with MSI-H GC and patients with MSI-L/MSS GC, and compared them by calculate the odds ratios (ORs). Similar comparisons were also conducted in nine clinicopathological features and six molecular biomarkers. Moreover, the 5-year survival rates in patients who were treated with conventional strategies were examined. Lastly, we compared the objective response rate (ORR) and overall survival (OS) in MSI-H GC patients and MSI-L/MSS GC patients who were treated with immune checkpoint inhibitors (ICIs). Our panoramic overview on MSI-H gastric cancer may have implications in the personalization of tumor diagnosis, treatment and prognosis.
Materials and Methods
Search strategy and selection criteria
This study was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement [13]. A systematic search of Embase, PubMed and Cochrane databases for articles describing prevalence, risk factors, clinicopathological characteristics, molecular biomarkers, and outcomes of MSI-H GC versus MSI-L/MSS GC from inception to December 2022 was carried out. The keywords used were “microsatellite”, “mismatch repair”, “replication error”, and “gastric cancer”. All investigators preformed the initial search independently, carefully reviewed the title and abstract for relevance, and classified the potential articles as excluded, included and uncertain. For uncertain articles, the full-texts were reviewed for the confirmation of eligibility. Any discrepancy was resolved by discussion.
Both inclusion and exclusion criteria were pre-specified. Studies were eligible if they met the following criteria: (1) original articles, including retrospective and prospective cohort studies, on human gastric cancer; (2) published in the English language; (3) available information regarding the proportions, risk factors, clinicopathological characteristics, molecular biomarkers, or outcomes of MSI-H GC. Exclusion criteria were: (1) other studies on this topic, including pre-clinical papers, review articles, early versions of data later published; (2) studies in the pediatric population; (3) data from unpublished studies. When multiple publication from the same databases occurred, we removed the overlapping data and only included the most recent and/or most complete reporting studies.
Data extraction and analysis
MSI status was determined by PCR, IHC, and NGS. Treatment methods were classified in immunotherapy and conventional therapy. All authors independently extracted study-level information regarding study characteristics (authors, year of publication, country/region of origin, MSI testing method, and number of patients), risk factors (family history of cancer, Epstein-Barr virus [EBV] infection, H. pylori infection, smoking, and drinking), clinicopathological characteristics (age, sex, tumor location, Lauren’s classification, WHO classification, TNM stage), molecular biomarkers (HER2, P53, KRAS, PD-L1, CD8, and tumor mutation burden [TMB]), treatment methods, and clinical outcomes (ORR and OS). Objective response included complete response and partial response determined by tumor assessments from radiological examinations or physical tests. OS was defined as the time period between the date of diagnosis and the date of death by any cause.
The primary outcomes of this study were: (1) the proportion of MSI-H globally; (2) comparison of risk factors, clinicopathological characteristics, and molecular biomarkers between MSI-H GC and MSI-L/MSS GC; and (3) the prognostic and predictive value of MSI-H as a biomarker.
Quality assessment
Joanna Briggs Institute (JBI) Critical Appraisal Tool was applied for quality assessment [14]. The JBI assessment rates the risk of bias of cohorts according to appropriateness of sample frame, adequacy of sample size, sampling method, methods for identification and measurement of relevant conditions, data analysis, statistical analysis, and response rate adequacy. Potential publication bias was assessed by visual inspection of Begg’s funnel plots, in which the log odds ratios (ORs) were plotted against their standard errors [15].
Statistical analysis
Statistical heterogeneity for the pooled estimates was evaluated by the Cochrane’s Q statistic and the Higgins I2 measure [16]. The I2 statistic was calculated to assess the extent of inconsistency contributable to the heterogeneity across different studies. The assumption of homogeneity was considered invalid for I2>25%. When I2 >25%, the effect size was calculated by a random-effects model using the DerSimonian and Laird approach; otherwise, a fixed-model were conducted. The pooled OR and incidences for both prespecified subgroup analysis and post-hoc analyses were calculated using fixed-effects model or random-effects model depending on the heterogeneity of included trials. Analysis of proportions was conducted with a generalized linear mixed model with Clopper-Pearson intervals to estimate the overall proportion and corresponding 95% confidence interval (CI) [17]. Hazard ratio (HR) was applied to compare the survivals in patients treated with ICI-based regimens and chemotherapy. The eligible trials reported their HRs calculated from Cox proportional-hazards models. Two-sided P <0.05 were considered statistically significant. All analysis was conducted by MedCalc 18.2.1 and RStudio 1.3.1093.
Results
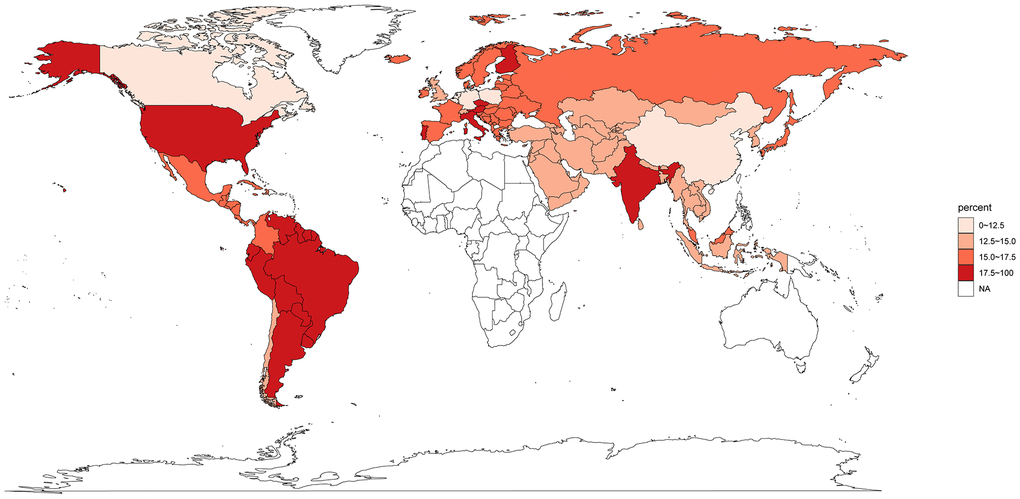
The initial search from PubMed, Embase and Cochrane databases yielded 3,250 related papers. After carefully screening and reviewing, 134 cohorts were eligible for the final analysis. A flow chart showing the selection process is presented in Figure 1. All data used for analysis were obtained from published manuscripts. These studies were conducted in Austria, Belgium, Brazil, Canada, China, Chile, Colombia, Czech, Finland, Germany, Hong Kong, India, Italy, Japan, Korea, Malaysia, Netherlands, Poland, Portugal, Spain, Switzerland, Taiwan, UK, and US. Additionally, five international studies [18–24] involved medical centers from other countries including Argentina, Australia, Denmark, Estonia, Greece, Guatemala, Hungary, Ireland, Israel, Latvia, Lithuania, Mexico, New Zealand, Norway, Peru, Puerto Rico, Romania, Russia, Singapore, South Africa, Thailand, and Turkey. The quality of these eligible studies, according to the JBI assessment rates [14], were generally moderate to good (Supplementary Table 1). A total of 43246 patients were enrolled, 4919 with MSI-H GC and 38327 with MSI-L/MSS disease. The overall proportion of patients with MSI-H GC was 14.5% (95% CI, 13.3%-15.8%), it was highest in the South America (21.8%; 95% CI, 17.1%-26.9%), followed by North America (17.9%; 5.9%-34.7%), Europe (16.8%; 14.1%-19.8%), and Asia (13.7%; 12.4%-15.1%) (Figure 2). The global proportion of MSI-H GC remained relatively stable over time periods (14.9%, 95% CI 13.1%-16.9% for before year 2010 vs. 13.2%, 95% CI 10.7%-15.9% for year 2010 and beyond; p=0.38).
Different methods were applied to evaluate the MSI status in these eligible studies. PCR was applied in 77 cohorts, IHC in 18 studies, the combination of PCR and IHC in 27 trials, and NGS-based testing in 11 studies. Consist with previous studies [25–27], these different assays showed similar diagnostic performance. Of note, although the prevalence in cohorts that used NGS alone or in combination (13.2%; 95% CI, 9.3%-17.6%) was lower compared with those using PCR and/or IHC (14.9%; 13.7%-16.2%), the difference was not significant (p=0.39).
Next, we evaluated the potential epidemiological and risk factors which could increase the incidence of MSI-H GC. As shown in Table 1, MSI-H was independent of familial predisposition (OR, 1.04; 95% CI, 0.64-1.70; p=0.87), H. pylori infection (0.95; 0.59-1.53; p=0.83), smoking (0.55; 0.29-1.05; p=0.07), and alcohol consumption (0.89; 0.25-3.17; p=0.85). However, patients with MSI-H GC were less likely to have EBV infection than were patients with MSI-L/MSS disease (OR, 0.43; 95% CI, 0.21-0.86; p=0.02).
Table 1. Characteristics of MSI-H gastric cancer versus MSI-L/MSS gastric cancer.
| Cohorts, n | Patients, n | Proportion in MSI-H patients (95% CI) | Proportion in MSI-L /MSS patients (95% CI) | Odds ratio (95% CI) | P-value | I 2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Risk factors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Family history of cancer | 13 | 3517 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 13 | 770 | 32.0% (21.3-43.8) | 32.5% (20.1-46.3) | 1.04 (0.64-1.70) | 0.87 | 74 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 13 | 2143 | 68.0% (56.2-78.7) | 67.5% (53.7-79.9) | 0.96 (0.59-1.57) | 0.87 | 74 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Epstein-Barr Virus infection | 18 | 3820 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 18 | 311 | 3.4% (1.3-6.4) | 10.3% (7.4-13.6) | 0.43 (0.21-0.86) | 0.02 | 50 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 18 | 3509 | 96.6% (93.6-98.7) | 89.7% (86.4-92.6) | 2.35 (1.17-4.74) | 0.02 | 50 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H. pylori infection | 12 | 1702 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 12 | 938 | 60.0% (40.3-78.2) | 56.0% (43.1-68.6) | 0.95 (0.59-1.53) | 0.83 | 49 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 12 | 764 | 40.0% (21.8-59.7) | 44.0% (31.4-56.9) | 1.05 (0.65-1.70) | 0.83 | 49 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking status | 3 | 383 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never/Former smoker | 3 | 178 | 58.0% (30.9-82.8) | 43.7% (27.9-60.3) | 1.82 (0.95-3.48) | 0.07 | 31 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current smoker | 3 | 205 | 42.0% (17.2-69.1) | 56.3% (39.7-72.1) | 0.55 (0.29-1.05) | 0.07 | 31 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking status | 3 | 408 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Never/Former drinker | 3 | 216 | 49.7% (14.2-85.3) | 49.9% (8.9-90.9) | 1.13 (0.32-4.05) | 0.85 | 72 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Current drinker | 3 | 192 | 50.3% (14.7-85.8) | 50.1% (9.1-91.1) | 0.89 (0.25-3.17) | 0.85 | 72 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Clinicopathological characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender | 86 | 32366 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 86 | 20867 | 57.2% (54.3-59.7) | 66.8% (64.3-69.2) | 0.67 (0.61-0.75) | <0.001 | 43 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 86 | 11499 | 42.8% (40.3-45.4) | 33.2% (30.8-35.7) | 1.49 (1.34-1.65) | <0.001 | 43 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 28 | 6433 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| <=65 year | 28 | 2754 | 38.2% (28.4-48.5) | 50.1% (42.9-57.3) | 0.55 (0.43-0.71) | <0.001 | 51 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >65 year | 28 | 3679 | 61.8% (51.5-71.7) | 49.9% (42.6-57.1) | 1.80 (1.41-2.32) | <0.001 | 51 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor location | 43 | 13387 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cardia tumor | 43 | 2588 | 12.1% (9.2-15.3) | 19.9% (16.5-23.5) | 0.55 (0.43-0.71) | <0.001 | 42 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Body tumor | 43 | 4037 | 23.1% (20.2-26.0) | 31.8% (29.0-34.7) | 0.63 (0.56-0.71) | <0.001 | 5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antrum tumor | 43 | 6762 | 63.6% (58.7-68.3) | 46.9% (43.0-50.9) | 2.17 (1.85-2.53) | <0.001 | 32 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lauren classification | 62 | 20007 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intestinal | 62 | 9706 | 66.3% (60.7-71.6) | 49.8% (47.0-52.6) | 2.02 (1.74-2.34) | <0.001 | 45 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diffuse | 62 | 7976 | 22.4% (17.5-27.7) | 40.0% (36.9-43.1) | 0.45 (0.39-0.52) | <0.001 | 29 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mixed | 62 | 2325 | 10.2% (7.2-13.7) | 8.5% (6.2-11.2) | 1.14 (0.89-1.47) | 0.29 | 51 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WHO classification | 26 | 8749 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tubular | 26 | 3823 | 53.1% (45.2-61.0) | 43.7% (37.1-50.4) | 1.22 (1.11-1.34) | <0.001 | 50 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Poor differentiation | 26 | 4528 | 42.1% (33.2-51.4) | 46.9% (39.3-54.4) | 0.79 (0.64-0.97) | 0.02 | 40 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Signet ring cell | 26 | 303 | 2.0% (0.8-3.7) | 4.1% (1.6-7.5) | 0.28 (0.18-0.45) | <0.001 | 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mucinous | 26 | 95 | 1.9% (0.7-3.6) | 1.0% (0.4-1.9) | 2.02 (1.22-3.35) | 0.01 | 32 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor Stage | 57 | 18208 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Early stage | 57 | 6063 | 35.2% (29.7-41.0) | 30.7% (25.6-36.0) | 1.26 (1.07-1.48) | 0.01 | 49 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Late stage | 57 | 12145 | 64.8% (59.0-70.3) | 69.3% (64.0-74.4) | 0.80 (0.68-0.94) | 0.01 | 49 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T | 45 | 22152 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1/T2 | 45 | 8478 | 37.9% (31.7-44.3) | 35.3% (29.9-41.0) | 1.07 (0.93-1.25) | 0.35 | 49 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3/T4 | 45 | 13674 | 62.1% (55.7-68.3) | 64.7% (59.0-70.1) | 0.93 (0.80-1.08) | 0.35 | 49 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N | 65 | 26025 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N+ | 65 | 16509 | 56.2% (51.4-60.9) | 64.5% (60.1-68.8) | 0.68 (0.60-0.78) | <0.001 | 42 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N- | 65 | 9516 | 43.8% (39.1-48.6) | 35.5% (31.2-39.9) | 1.46 (1.29-1.66) | <0.001 | 42 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M | 26 | 10031 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M0 | 26 | 8855 | 92.7% (88.1-96.2) | 87.7% (81.4-92.8) | 2.40 (1.88-3.08) | <0.001 | 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M1 | 26 | 1176 | 7.3% (3.8-11.9) | 12.3% (7.2-18.6) | 0.42 (0.33-0.53) | <0.001 | 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Molecular biomarkers | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HER2 expression | 11 | 4860 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HER2+ | 11 | 299 | 2.6% (1.5-4.3) | 8.8% (5.9-12.2) | 0.33 (0.19-0.55) | <0.001 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HER2- | 11 | 4561 | 97.4% (95.8-98.6) | 91.2% (87.8-94.1) | 3.07 (1.82-5.19) | <0.001 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TP53 status | 17 | 2766 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TP53 mutant | 17 | 1180 | 24.4% (16.9-32.8) | 45.6% (34.2-57.3) | 0.39 (0.30-0.50) | <0.001 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TP53 non-mutant | 17 | 1586 | 75.6% (67.2-83.1) | 54.4% (42.7-65.8) | 2.56 (2.00-3.29) | <0.001 | 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KRAS status | 11 | 1934 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KRAS mutant | 11 | 160 | 20.7% (13.6-28.8) | 4.4% (3.2-5.8) | 5.56 (3.76-8.23) | <0.001 | 16 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KRAS non-mutant | 11 | 1774 | 79.3% (71.2-86.4) | 95.6% (94.2-96.8) | 0.18 (0.12-0.27) | <0.001 | 16 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PD-L1 expression* | 17 | 6129 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PD-L1+ | 17 | 2139 | 61.0% (48.7-72.6) | 29.8% (19.9-40.8) | 4.04 (2.94-5.56) | <0.001 | 47 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PD-L1- | 17 | 3990 | 39.0% (27.4-51.3) | 70.2% (59.2-80.1) | 0.25 (0.18-0.34) | <0.001 | 47 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CD8 expression | 6 | 1677 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High expression | 6 | 1029 | 73.3% (59.8-84.8) | 58.6% (35.7-79.6) | 2.34 (1.28-4.27) | 0.006 | 63 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low expression | 6 | 648 | 26.7% (15.2-40.2) | 41.4% (20.4-64.3) | 0.43 (0.23-0.78) | 0.006 | 63 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor mutation burden | 4 | 1253 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >10 mutations/Mb | 4 | 318 | 97.9% (91.5-100.0) | 13.1% (3.6-27.2) | 241.65 (16.52-3535.77) | <0.001 | 78 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| <=10 mutations/Mb | 4 | 935 | 2.1% (0.0-8.5) | 86.9% (72.8-96.4) | 0.004 (0.00-0.06) | <0.001 | 78 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *The threshold for PD-L1 positivity/negativity was that PD-L1 stained cell accounting for 1% of tumor cells, or immune and tumor cells evaluated by IHC. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The bold values mean p<0.05. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The incidences of MSI-H GC were higher in female (OR, 1.49; 95% CI, 1.34-1.65; p<0.001) and older age (>65 years; 1.80; 1.41-2.32; p<0.001). MSI-H tumors were more likely found in the lower gastric body (OR, 2.17; 95% CI, 1.85-2.53; p<0.001), but not in the upper (0.55; 0.43-0.71; p<0.001) and middle body (0.63; 0.56-0.71; p<0.001). Compared with MSI-L/MSS GC, more MSI-H tumors were classified as Lauren intestinal subtype (OR, 2.02; 95% CI, 1.74-2.34; p<0.001), less as diffuse subtype (0.45; 0.39-0.52; p<0.001). According to WHO classification, MSI-H tumors were more likely to identified as tubular subtype (OR, 1.22; 95% CI; 1.11-1.34; p<0.001) and mucinous subtype (2.02; 1.22-3.35; p=0.01), but less as signet ring cell subtype (0.28; 0.18-0.45; p<0.001) and poorly differentiated subtype (0.79; 0.64-0.97; p=0.02). Moreover, MSI-H tumors were more often diagnosed at early disease stages (OR, 1.26; 95% CI, 1.07-1.48; p=0.01).
MSI-H was associated with higher proportion of KRAS mutation (OR, 5.56; 95% CI, 3.76-8.23; p<0.001), PD-L1 positivity (4.04; 2.94-5.56; p<0.001), CD8 overexpression (2.34; 1.28-4.27; p=0.006), and high TMB (TMB>10 mutants/Mb; 241.65; 16.52-3535.77; p<0.001), but lower proportion of HER2+ (0.33; 0.19-0.55; p<0.001) and TP53 mutation (0.39; 0.30-0.50; p<0.001).
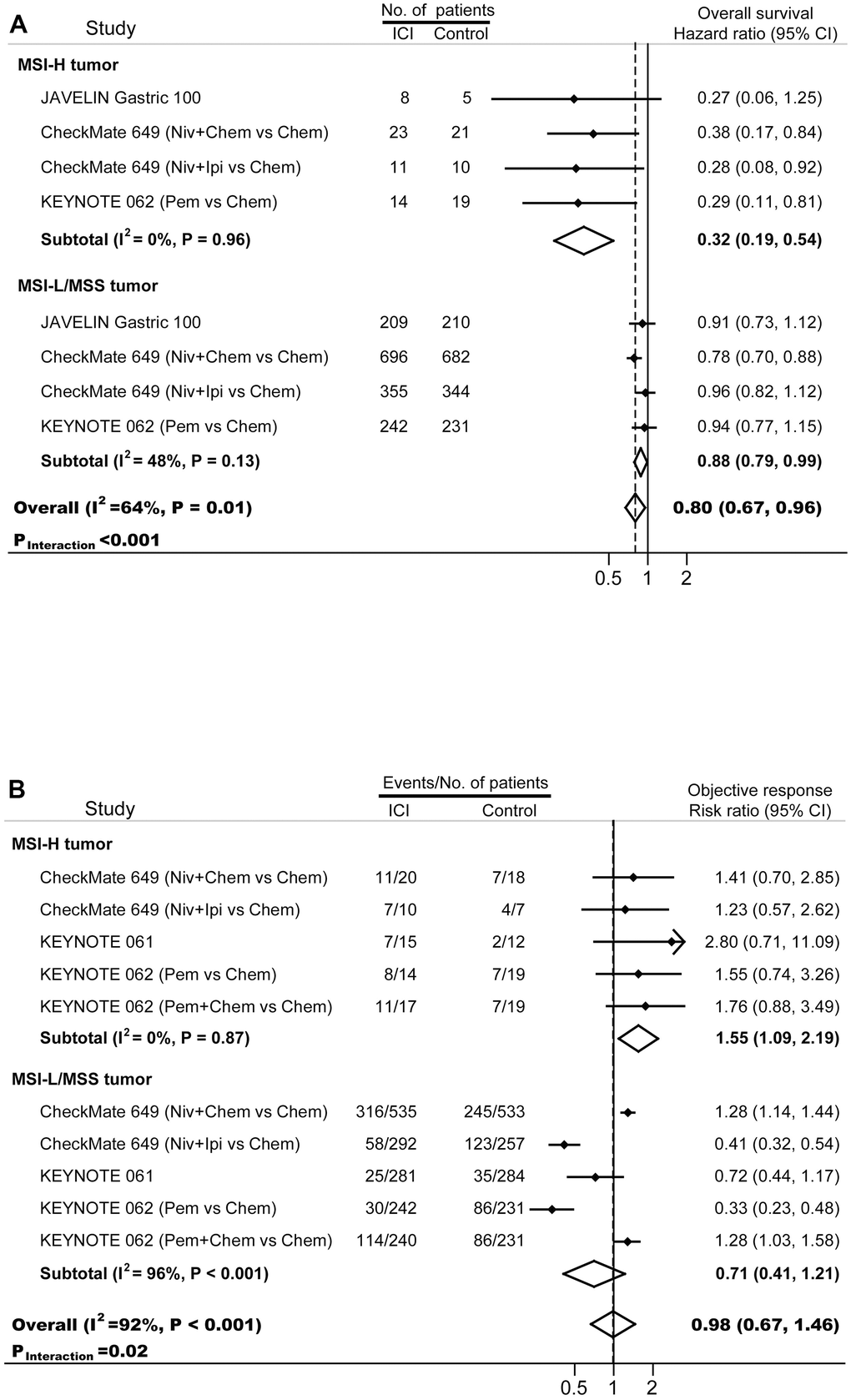
The final important aspect to analyze is the clinical outcomes of patients with MSI-H tumors. In 40 cohorts enrolled 17081 patients treated with conventional strategies, 5-year survival rate in MSI-H patients (70.3%; inter quartile range, 57.5%-77.0%) was significant higher compared with that in MSI-L/MSS patients (43.7%, inter quartile range, 36.8%-56.4%; p<0.001). Four phase III randomized trials, KEYNOTE-061 [18, 20], KEYNOTE-062 [18, 21], JAVELIN Gastric 100 [22], and CheckMate-649 [19, 23], were included to estimate the activity and efficacy of immunotherapy (Figure 3). Compared with standard treatments, immunotherapy decreased the risk of death by 68% (HR, 0.32; 95% CI, 0.19-0.54) in patients with MSI-H GC and by 12% (HR, 0.88; 95% CI, 0.79-0.99) in patients with MSI-L/MSS GC. The survival outcomes were significantly different between these two subgroups (pinteraction<0.001). More patients with MSI-H GC responded to immunotherapy than to chemotherapy (risk ratio, 1.55; 95% CI, 1.09-2.19; p<0.001); whereas similar proportion of MSI-L/MSS GC patients showed responses to immunotherapy and chemotherapy (risk ratio, 0.71; 95% CI, 0.41-1.21). The treatment effect in term of objective response was significantly different between MSI-H GC and MSI-L/MSS GC (pinteraction=0.02).
The Begg’s funnel plots were conducted to evaluate the potential publication bias from every eligible study (Supplementary Figure 1). No significant publication bias was observed.
Discussion
With published data from 134 cohorts with over 40,000 patients, our pooled analysis first demonstrated that about 14.5% of gastric cancer were secondary to MSI-H globally. The highest proportion of MSI-H GC occurred in South America, and the lowest in East Asia. As the prevalence of MSI-H in colorectal tumor, the most frequently studied tumor types, was about 14.2% in the US [28], we recommended MSI testing should be the first-line analysis during standard diagnostical activity. Moreover, we compared the incidences of five epidemiological and risk factors, nine clinicopathological features and six molecular biomarkers in patients with MSI-H GC and patients with MSI-L/MSS GC, confirmed the distinct characteristics of MSI-H GC. Lastly, our data revealed that MSI-H was a predictive biomarker for better survivals in both conventional treatments and immunotherapy. Since MSI-H tumors were often diagnosed at early stage and had favorable outcomes, less aggressive treatment strategies might be considered in clinical practice. Our panoramic review on MSI-H GC may assist in design and/or interpretation of clinical trials, provide references in drug development, and constitute complementary information in drafting the clinical practice guideline.
Gastric cancer was a malignancy strongly associated with the geographical background. It was well-established that the incidences, clinicopathological characteristics, treatment strategies, and outcomes showed great geographic variations [1, 2]. Due to the important role of MSI-H as a biomarker in cancer immunotherapy, the determination of the prevalence of MSI-H GC from different countries/regions appeared to be an essential prerequisite for worldwide clinical development of ICI-based treatment. Here, our study presented the first global estimated of gastric cancer secondary to MSI-H disease. As expected, the prevalence of MSI-H GC also differed across countries. Interestingly, although the highest incidence of GC was observed among Asiatic population and lowest in Europe and Northern America [2], our data revealed that, compared with Western countries, the proportion of MSI-H GC was significantly lower in East Asia. Surprisingly, the frequency of MSI-H GC was highest in Hispanics/Latinos. Considering the limited enrollment of Latinos in clinical trials, the surveillance strategies for MSI status needed to improve in these patient populations. Currently, the exact explanations for this disproportionate distribution of MSI-H GC are unclear. It seemed that the combination of genetic predisposition, dietary habits and other environmental factors played major roles. For example, in an Italian population highly susceptible to GC, MSI-H was believed to cause the genetic alterations in non-invasive neoplasia [29]. Additionally, the role of dietary risk factors in MSI-H GC was evaluated in a population-based study [30]. They discovered that MSI-H GC was associated with a specific diet pattern, frequent consumption of fresh vegetables and fruits can significantly reduce the risk of MSI-H GC, while high consumption of meat paste, red meat, and nitrite increased the risk. Accordingly, the so-called western-style food habit, which was often referred to refined compounds, red meat, and processed meat, might be a potential reason for the upregulated proportion of MSI-H GC among Western countries. Another hypothesis is that GC is more likely diagnosed at a younger age in East Asia [31, 32] and MSI-H is often associated with older age. Age might be an un-neglected factor in explain the different prevalence of MSI-H GC between Asia and Western countries.
Consist with previous reports [12, 33], our study demonstrated that MSI-H GC occurred more often in older patients. The tumors in young patients and old patients showed different clinicopathological and molecular characteristics. GC in old patients were usually located in the lower body, with relatively low metastasis, and were present in about 10% synchronous GC [34]. These features were often observed in MSI-H GC. In contrast, tumors in young patients were located in middle body, with high metastasis, and occurring in 3% synchronous GC [34]. Moreover, epigenetic changes were involved in the development of GC in old patients [35]. Age related gene methylation could increase the chances of development of malignant neoplasms as CpG island methylation played a key role in the in activation of many genes [35]. For example, it was reported that the methylation of hMLH1 and its loss of expression were greatly upregulated in aged patient [36], which could significantly increase the possibility of microsatellite instability.
The Epstein-Barr Virus infects 90% of the population worldwide and can directly cause EBV-associated GC [37]. This specific subtype of GC represented a distinct etiologic entity which was associated with mutations in PIK3A, hypermethylation of CDKN2A, amplification of JAK2, proximal location, male gender, and poorly differentiated histology characteristically with lymphocytic infiltration [38]. Interestingly, none of these features were dominated in MSI-H GC. Indeed, experiments revealed a mutually elite pattern between the presence of EBV positivity and MSI-H that are independent of each other [5, 37], suggesting EBV-associated GC and MSI-H GC involved different molecular pathways during cancer development. Indeed, our analysis here revealed that almost all MSI-H GC were EBV negative (~97%). The EBV positive and MSI-H tumors might belong to the special subset of GC with increased number of lymphocytes [39]. Interestingly, high proportion PD-L1expression was found in both EBV-associated GC [38] and MSI-H GC, and more patients responded to ICI-based immunotherapy compared with other subtypes [3, 37].
Through examining a series of clinicopathological features and molecular biomarkers, our study revealed that a specific genetic profile and distinct clinicopathological characteristics were associated with MSI-H GC. TP53 was the most commonly mutated gene in tumors and associated with poor outcomes in cancers [40]. In the TCGA analysis, although TP53 mutations were often observed in chromosomally unstable tumors, they were rarely found in MSI-H cases [5, 41]. It might because MSH2 and TP53 genes protected the genome integrity by different pathways [42]. Consist with previous findings in colorectal cancer [43], our data here demonstrated that, compared with MSI-L/MSS GC, the proportion of TP53 mutations decreased significantly in MSI-H tumors. It was suggested that only a special restricted pattern of P53 expression was preferentially associated with MSI-H phenotype [44]. Interestingly, TP53 can exert anti-tumor immune activities by increasing antigen presentation, reducing PD-L1 expression [45, 46], and TP53 dysfunction could repress immunogenic activity by decrease the expression levels of almost all immune-related gene pathways [41]. This might indicate that TP53 and its associated genes could be a potential biomarker in cancer immunotherapy. Currently, HER2 was the only biomarker which was routinely examined and widely used for targeted therapy in GC [1, 3]. It is a subtype included in the chromosomal instability (CIN) category according to TCGA classification [5], and MSS/TP53 inactive category based on the ACRG classification [6]. HER2+ GC was more commonly associated with proximal location, metastasis, male gender, advanced tumor stage at diagnosis and poor prognosis [47–49]. However, most MSI-H tumors did not have these features. Indeed, our data showed that less than 3% of all MSI-H GC were HER2 positive. It seemed that HER2 positive and MSI-H tumors demonstrated a mutually negative association. This suggested that HER2 and MSI could modulate the tumor microenvironment and the immunologic response in different pathways [50]. It was reported there was a synergistic effect of HER2-targeted therapy and immunotherapy [51], which might explain the recent accelerated approval of pembrolizumab in combination with trastuzumab plus chemotherapy for patients with HER2+ gastric cancer by FDA [52].
In MSI-H tumors, due to the massive production of abnormal tumor-specific neoantigens which could activate recruitment of lymphocytes, a robust correlation between tumor infiltrating lymphocytes (TILs) and MSI was confirmed [53], and a permissive inflamed microenvironment was established [54]. This strong activation of the immune system was one of the explanations for the favorable prognosis and the low rates of metastasis in MSI GC [55]. As expected, our results showed that MSI-H GC had higher PD-L1 expression, CD8+ TIL, and TMB. Numerous evidences revealed the superior efficacy of immunotherapy-based regimens compared with conventional treatment in MSI-H/dMMR patients, even in trials with unfavorable results in the overall population [18, 21]. In the present meta-analysis, ICI-based regimens significantly improved overall survivals and objective response rates in the subgroup of patients with MSI-H GC. Furthermore, the interaction between the outcomes and MSI status remained significant, suggesting that, even if some patients with MSI-L/MSS GC may benefit from immunotherapy (mainly those with PD-L1 positive and/or high tumor mutation burden), the efficacy and activity of immunotherapy in the MSI-H arm is higher compared with the overall MSI-L/MSS counterpart. In fact, because the prognostic value of PD-L1 expression was controversial [56], it was suggested that the combined assessment of MSI status and PD-L1 expression were more powerful than PD-L1 alone in guiding patients’ stratification for immunotherapy [57].
In the past several years, many studies evaluated the clinical relevance of the MSI status as a positive predictor in GC patients [24, 58]. It had been argued that it was due to the correlation between MSI-H with relatively early TNM stage at diagnosis and Lauren intestinal histotype [12, 59]. Interestingly, MSI-H GC was often associated with longer survivals even in patients with advanced disease since these tumors had a lower prevalence of lymph node metastases and a lower ability to invade serosal layers [60]. Because of the prognostic relevance, MSI status should be considered in the therapeutic decision-making to avoid potential excessive medical treatment. For example, it was known that peri-operative chemotherapy is guideline-endorsed treatment for GC [1, 3]. However, in MAGIC trial [61], patients with MSI-H tumor exhibited unfavorable survivals in the chemotherapy plus surgery arm. In CLASSIC study [62], patients with MSI-H GC experience no benefit from chemotherapy in term of disease-free survival. These results confirmed that lack of survival benefit from peri-operative chemotherapy, and hence transforming the clinical practice of operable MSI-H GC.
Our study has several clinical implications. First of all, although the utility of MSI status in clinical practice may help to identify the most effective treatment, the MSI test in gastric cancer is not always conducted in real-world. For example, the diagnosis of MSI status is required only in patients with colorectal and endometrial cancer in Europe [10, 11]. Giving the relatively high prevalence of MSI-H GC in western countries, the examination of MSI/dMMR should be recommended during routine diagnostic activity in gastric cancer. Second, MSI-H GC is associated with relatively early stage at diagnosis, Lauren intestinal histotype, lower prevalence of metastases, and hence favorable outcomes. Additionally, peri-operative chemotherapy exhibited poorly prognosis in patients with MSI-H GC despite it is endorsed by guidelines in gastric cancer treatment [61]. Accordingly, in clinical practice, less aggressive treatment strategies may be considered for patients with MSI-H GC. Furthermore, since patients with MSI-H GC were regarded as a special immune-sensitive papulation, immunotherapy should be routinely available for those with advanced MSI-H GC. Another potential use of this study is in the economic analysis. With the MSI status testing, different treatment strategy will be carried out to achieve the best clinical benefit. Considering ICIs are among the most expensive agents in the world, the financial consequences are significant for patients, their families, and the whole society.
To our knowledge, the present study provides the most comprehensive analysis of the existing literature regarding the panoramic landscape of MSI-H GC to date. However, our study is not without limitations. First, there are very few studies from South-East Asia, South Asia, and Eastern Mediterranean region, and sample size are relatively small. Accordingly, it is cautious to properly interpret the prevalence of MSI-H GC from these regions and more data are needed. Additionally, we cannot extract any information regarding MSI-H GC from Africa and Oceania region. Second, there are substantial heterogeneities in some comparisons, which might arise from the large number of enrolled patients included in the pooled analysis. We clarify the potential sources of the heterogeneities by performing meta-regression and subgroup analysis where appropriate. Third, we conduct the current study at the trial level, no features at individual levels are investigated. It may reduce the reliability in the association between MSI-H and variables in specific subgroup analysis. Fourth, some included information, such as HER2 and CD8 expression status, are reported from various medical centers by different investigators. These data are potentially associated with subjectivity. Our study is subject to any errors and bias from the original researchers.
Conclusions
In summary, this study conducts a systematic overview of the global burden, risk factors, clinicopathological characteristics, molecular biomarkers, and clinical outcomes of MSI-H gastric cancer. We provide high-level evidence showing that 15% GC patients have MSI-H disease, which is associated with a specific genetic profile and distinct clinicopathological characteristics. Accordingly, MSI/dMMR should be determined as the first-line analysis during GC standard diagnostic activity. Moreover, giving MSI-H tumors are often diagnosed at relatively early stage and have favorable outcomes, less aggressive treatment strategies may be considered in clinical practice. For patients with advanced MSI-H GC, immunotherapy should be routinely available. Further investigations are needed to better understand the significant etiological factors associated with MSI-H GC.
Author Contributions
Zhishan Zhang: conception, methodology, validation, formal analysis, writing-original draft; Jinyuan Huang: methodology, validation, formal analysis, investigation, writing-original draft; Yingying Li: methodology, formal analysis, investigation, validation; Huimeng Yan: methodology, validation, formal analysis; Junxing Xie: methodology, investigation; Jing Wang: investigation; Bin Zhao: conception, writing-review and editing, supervision.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
No funding was provided for this study.
References
- 1. Smyth EC, Nilsson M, Grabsch HI, van Grieken NC, Lordick F. Gastric cancer. Lancet. 2020; 396:635–48. https://doi.org/10.1016/S0140-6736(20)31288-5 [PubMed]
- 2. Arnold M, Park JY, Camargo MC, Lunet N, Forman D, Soerjomataram I. Is gastric cancer becoming a rare disease? A global assessment of predicted incidence trends to 2035. Gut. 2020; 69:823–9. https://doi.org/10.1136/gutjnl-2019-320234 [PubMed]
- 3. Joshi SS, Badgwell BD. Current treatment and recent progress in gastric cancer. CA Cancer J Clin. 2021; 71:264–79. https://doi.org/10.3322/caac.21657 [PubMed]
- 4. Han L, Cui DJ, Huang B, Yang Q, Huang T, Lin GY, Chen SJ. CLDN5 identified as a biomarker for metastasis and immune infiltration in gastric cancer via pan-cancer analysis. Aging (Albany NY). 2023; 15:5032–51. https://doi.org/10.18632/aging.204776 [PubMed]
- 5. Cancer Genome Atlas Research Network. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014; 513:202–9. https://doi.org/10.1038/nature13480 [PubMed]
- 6. Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, Liu J, Yue YG, Wang J, Yu K, Ye XS, Do IG, Liu S, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015; 21:449–56. https://doi.org/10.1038/nm.3850 [PubMed]
- 7. Baretti M, Le DT. DNA mismatch repair in cancer. Pharmacol Ther. 2018; 189:45–62. https://doi.org/10.1016/j.pharmthera.2018.04.004 [PubMed]
- 8. Lu M, Zhao B, Liu M, Wu L, Li Y, Zhai Y, Shen X. Pan-cancer analysis of SETD2 mutation and its association with the efficacy of immunotherapy. NPJ Precis Oncol. 2021; 5:51. https://doi.org/10.1038/s41698-021-00193-0 [PubMed]
- 9. Bonneville R, Krook MA, Kautto EA, Miya J, Wing MR, Chen HZ, Reeser JW, Yu L, Roychowdhury S. Landscape of Microsatellite Instability Across 39 Cancer Types. JCO Precis Oncol. 2017; 2017:PO.17.00073. https://doi.org/10.1200/PO.17.00073 [PubMed]
- 10. Benson AB, Venook AP, Al-Hawary MM, Arain MA, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Farkas L, Garrido-Laguna I, Grem JL, Gunn A, et al. Colon Cancer, Version 2.2021, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2021; 19:329–59. https://doi.org/10.6004/jnccn.2021.0012 [PubMed]
- 11. Stoffel EM, Mangu PB, Gruber SB, Hamilton SR, Kalady MF, Lau MW, Lu KH, Roach N, Limburg PJ, American Society of Clinical Oncology, and European Society of Clinical Oncology. Hereditary colorectal cancer syndromes: American Society of Clinical Oncology Clinical Practice Guideline endorsement of the familial risk-colorectal cancer: European Society for Medical Oncology Clinical Practice Guidelines. J Clin Oncol. 2015; 33:209–17. https://doi.org/10.1200/JCO.2014.58.1322 [PubMed]
- 12. Ratti M, Lampis A, Hahne JC, Passalacqua R, Valeri N. Microsatellite instability in gastric cancer: molecular bases, clinical perspectives, and new treatment approaches. Cell Mol Life Sci. 2018; 75:4151–62. https://doi.org/10.1007/s00018-018-2906-9 [PubMed]
- 13. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J, Moher D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009; 339:b2700. https://doi.org/10.1136/bmj.b2700 [PubMed]
- 14. Munn Z, Moola S, Riitano D, Lisy K. The development of a critical appraisal tool for use in systematic reviews addressing questions of prevalence. Int J Health Policy Manag. 2014; 3:123–8. https://doi.org/10.15171/ijhpm.2014.71 [PubMed]
- 15. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50:1088–101. [PubMed]
- 16. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–60. https://doi.org/10.1136/bmj.327.7414.557 [PubMed]
- 17. Schwarzer G, Chemaitelly H, Abu-Raddad LJ, Rücker G. Seriously misleading results using inverse of Freeman-Tukey double arcsine transformation in meta-analysis of single proportions. Res Synth Methods. 2019; 10:476–83. https://doi.org/10.1002/jrsm.1348 [PubMed]
- 18. Chao J, Fuchs CS, Shitara K, Tabernero J, Muro K, Van Cutsem E, Bang YJ, De Vita F, Landers G, Yen CJ, Chau I, Elme A, Lee J, et al. Assessment of Pembrolizumab Therapy for the Treatment of Microsatellite Instability-High Gastric or Gastroesophageal Junction Cancer Among Patients in the KEYNOTE-059, KEYNOTE-061, and KEYNOTE-062 Clinical Trials. JAMA Oncol. 2021; 7:895–902. https://doi.org/10.1001/jamaoncol.2021.0275 [PubMed]
- 19. Janjigian YY, Shitara K, Moehler M, Garrido M, Salman P, Shen L, Wyrwicz L, Yamaguchi K, Skoczylas T, Campos Bragagnoli A, Liu T, Schenker M, Yanez P, et al. First-line nivolumab plus chemotherapy versus chemotherapy alone for advanced gastric, gastro-oesophageal junction, and oesophageal adenocarcinoma (CheckMate 649): a randomised, open-label, phase 3 trial. Lancet. 2021; 398:27–40. https://doi.org/10.1016/S0140-6736(21)00797-2 [PubMed]
- 20. Shitara K, Özgüroğlu M, Bang YJ, Di Bartolomeo M, Mandalà M, Ryu MH, Fornaro L, Olesiński T, Caglevic C, Chung HC, Muro K, Goekkurt E, Mansoor W, et al, and KEYNOTE-061 investigators. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial. Lancet. 2018; 392:123–33. https://doi.org/10.1016/S0140-6736(18)31257-1 [PubMed]
- 21. Shitara K, Van Cutsem E, Bang YJ, Fuchs C, Wyrwicz L, Lee KW, Kudaba I, Garrido M, Chung HC, Lee J, Castro HR, Mansoor W, Braghiroli MI, et al. Efficacy and Safety of Pembrolizumab or Pembrolizumab Plus Chemotherapy vs Chemotherapy Alone for Patients With First-line, Advanced Gastric Cancer: The KEYNOTE-062 Phase 3 Randomized Clinical Trial. JAMA Oncol. 2020; 6:1571–80. https://doi.org/10.1001/jamaoncol.2020.3370 [PubMed]
- 22. Moehler M, Dvorkin M, Boku N, Özgüroğlu M, Ryu MH, Muntean AS, Lonardi S, Nechaeva M, Bragagnoli AC, Coşkun HS, Cubillo Gracian A, Takano T, Wong R, et al. Phase III Trial of Avelumab Maintenance After First-Line Induction Chemotherapy Versus Continuation of Chemotherapy in Patients With Gastric Cancers: Results From JAVELIN Gastric 100. J Clin Oncol. 2021; 39:966–77. https://doi.org/10.1200/JCO.20.00892 [PubMed]
- 23. Shitara K, Ajani JA, Moehler M, Garrido M, Gallardo C, Shen L, Yamaguchi K, Wyrwicz L, Skoczylas T, Bragagnoli AC, Liu T, Tehfe M, Elimova E, et al. Nivolumab plus chemotherapy or ipilimumab in gastro-oesophageal cancer. Nature. 2022; 603:942–8. https://doi.org/10.1038/s41586-022-04508-4 [PubMed]
- 24. Pietrantonio F, Miceli R, Raimondi A, Kim YW, Kang WK, Langley RE, Choi YY, Kim KM, Nankivell MG, Morano F, Wotherspoon A, Valeri N, Kook MC, et al. Individual Patient Data Meta-Analysis of the Value of Microsatellite Instability As a Biomarker in Gastric Cancer. J Clin Oncol. 2019; 37:3392–400. https://doi.org/10.1200/JCO.19.01124 [PubMed]
- 25. Dedeurwaerdere F, Claes KB, Van Dorpe J, Rottiers I, Van der Meulen J, Breyne J, Swaerts K, Martens G. Comparison of microsatellite instability detection by immunohistochemistry and molecular techniques in colorectal and endometrial cancer. Sci Rep. 2021; 11:12880. https://doi.org/10.1038/s41598-021-91974-x [PubMed]
- 26. Shimozaki K, Hayashi H, Tanishima S, Horie S, Chida A, Tsugaru K, Togasaki K, Kawasaki K, Aimono E, Hirata K, Nishihara H, Kanai T, Hamamoto Y. Concordance analysis of microsatellite instability status between polymerase chain reaction based testing and next generation sequencing for solid tumors. Sci Rep. 2021; 11:20003. https://doi.org/10.1038/s41598-021-99364-z [PubMed]
- 27. Kang SY, Kim DG, Ahn S, Ha SY, Jang KT, Kim KM. Comparative analysis of microsatellite instability by next-generation sequencing, MSI PCR and MMR immunohistochemistry in 1942 solid cancers. Pathol Res Pract. 2022; 233:153874. https://doi.org/10.1016/j.prp.2022.153874 [PubMed]
- 28. Gutierrez C, Ogino S, Meyerhardt JA, Iorgulescu JB. The Prevalence and Prognosis of Microsatellite Instability-High/Mismatch Repair-Deficient Colorectal Adenocarcinomas in the United States. JCO Precis Oncol. 2023; 7:e2200179. https://doi.org/10.1200/PO.22.00179 [PubMed]
- 29. Rugge M, Bersani G, Bertorelle R, Pennelli G, Russo VM, Farinati F, Bartolini D, Cassaro M, Alvisi V. Microsatellite instability and gastric non-invasive neoplasia in a high risk population in Cesena, Italy. J Clin Pathol. 2005; 58:805–10. https://doi.org/10.1136/jcp.2004.025676 [PubMed]
- 30. Palli D, Russo A, Ottini L, Masala G, Saieva C, Amorosi A, Cama A, D’Amico C, Falchetti M, Palmirotta R, Decarli A, Mariani Costantini R, Fraumeni JF
Jr . Red meat, family history, and increased risk of gastric cancer with microsatellite instability. Cancer Res. 2001; 61:5415–9. [PubMed] - 31. Lee KS, Oh DK, Han MA, Lee HY, Jun JK, Choi KS, Park EC. Gastric cancer screening in Korea: report on the national cancer screening program in 2008. Cancer Res Treat. 2011; 43:83–8. https://doi.org/10.4143/crt.2011.43.2.83 [PubMed]
- 32. Zeng H, Chen W, Zheng R, Zhang S, Ji JS, Zou X, Xia C, Sun K, Yang Z, Li H, Wang N, Han R, Liu S, et al. Changing cancer survival in China during 2003-15: a pooled analysis of 17 population-based cancer registries. Lancet Glob Health. 2018; 6:e555–67. https://doi.org/10.1016/S2214-109X(18)30127-X [PubMed]
- 33. Puliga E, Corso S, Pietrantonio F, Giordano S. Microsatellite instability in Gastric Cancer: Between lights and shadows. Cancer Treat Rev. 2021; 95:102175. https://doi.org/10.1016/j.ctrv.2021.102175 [PubMed]
- 34. Arai T, Takubo K. Clinicopathological and molecular characteristics of gastric and colorectal carcinomas in the elderly. Pathol Int. 2007; 57:303–14. https://doi.org/10.1111/j.1440-1827.2007.02101.x [PubMed]
- 35. Shokal U, Sharma PC. Implication of microsatellite instability in human gastric cancers. Indian J Med Res. 2012; 135:599–613. [PubMed]
- 36. Nakajima T, Akiyama Y, Shiraishi J, Arai T, Yanagisawa Y, Ara M, Fukuda Y, Sawabe M, Saitoh K, Kamiyama R, Hirokawa K, Yuasa Y. Age-related hypermethylation of the hMLH1 promoter in gastric cancers. Int J Cancer. 2001; 94:208–11. https://doi.org/10.1002/ijc.1454 [PubMed]
- 37. Pikuła A, Kwietniewska M, Rawicz-Pruszyński K, Ciseł B, Skórzewska M, Gęca K, Franciszkiewicz-Pietrzak K, Kurylcio A, Mielko J, Polkowski WP. The importance of Epstein-Barr virus infection in the systemic treatment of patients with gastric cancer. Semin Oncol. 2020; 47:127–37. https://doi.org/10.1053/j.seminoncol.2020.04.001 [PubMed]
- 38. Tanabe H, Mizukami Y, Takei H, Tamamura N, Omura Y, Kobayashi Y, Murakami Y, Kunogi T, Sasaki T, Takahashi K, Ando K, Ueno N, Kashima S, et al. Clinicopathological characteristics of Epstein-Barr virus and microsatellite instability subtypes of early gastric neoplasms classified by the Japanese and the World Health Organization criteria. J Pathol Clin Res. 2021; 7:397–409. https://doi.org/10.1002/cjp2.209 [PubMed]
- 39. Watanabe H, Enjoji M, Imai T. Gastric carcinoma with lymphoid stroma. Its morphologic characteristics and prognostic correlations. Cancer. 1976; 38:232–43. https://doi.org/10.1002/1097-0142(197607)38:1<232::aid-cncr2820380135>3.0.co;2-4 [PubMed]
- 40. Wang X, Sun Q. TP53 mutations, expression and interaction networks in human cancers. Oncotarget. 2017; 8:624–43. https://doi.org/10.18632/oncotarget.13483 [PubMed]
- 41. Jiang Z, Liu Z, Li M, Chen C, Wang X. Immunogenomics Analysis Reveals that TP53 Mutations Inhibit Tumor Immunity in Gastric Cancer. Transl Oncol. 2018; 11:1171–87. https://doi.org/10.1016/j.tranon.2018.07.012 [PubMed]
- 42. Otozai S, Ishikawa-Fujiwara T, Oda S, Kamei Y, Ryo H, Sato A, Nomura T, Mitani H, Tsujimura T, Inohara H, Todo T. p53-Dependent suppression of genome instability in germ cells. Mutat Res. 2014; 760:24–32. https://doi.org/10.1016/j.mrfmmm.2013.12.004 [PubMed]
- 43. Samowitz WS, Holden JA, Curtin K, Edwards SL, Walker AR, Lin HA, Robertson MA, Nichols MF, Gruenthal KM, Lynch BJ, Leppert MF, Slattery ML. Inverse relationship between microsatellite instability and K-ras and p53 gene alterations in colon cancer. Am J Pathol. 2001; 158:1517–24. https://doi.org/10.1016/S0002-9440(10)64102-8 [PubMed]
- 44. Nyiraneza C, Jouret-Mourin A, Kartheuser A, Camby P, Plomteux O, Detry R, Dahan K, Sempoux C. Distinctive patterns of p53 protein expression and microsatellite instability in human colorectal cancer. Hum Pathol. 2011; 42:1897–910. https://doi.org/10.1016/j.humpath.2010.06.021 [PubMed]
- 45. Cortez MA, Ivan C, Valdecanas D, Wang X, Peltier HJ, Ye Y, Araujo L, Carbone DP, Shilo K, Giri DK, Kelnar K, Martin D, Komaki R, et al. PDL1 Regulation by p53 via miR-34. J Natl Cancer Inst. 2015; 108:djv303. https://doi.org/10.1093/jnci/djv303 [PubMed]
- 46. Wang B, Niu D, Lai L, Ren EC. p53 increases MHC class I expression by upregulating the endoplasmic reticulum aminopeptidase ERAP1. Nat Commun. 2013; 4:2359. https://doi.org/10.1038/ncomms3359 [PubMed]
- 47. Bermúdez A, Arranz-Salas I, Mercado S, López-Villodres JA, González V, Ríus F, Ortega MV, Alba C, Hierro I, Bermúdez D. Her2-Positive and Microsatellite Instability Status in Gastric Cancer-Clinicopathological Implications. Diagnostics (Basel). 2021; 11:944. https://doi.org/10.3390/diagnostics11060944 [PubMed]
- 48. Matsuoka T, Yashiro M. Biomarkers of gastric cancer: Current topics and future perspective. World J Gastroenterol. 2018; 24:2818–32. https://doi.org/10.3748/wjg.v24.i26.2818 [PubMed]
- 49. Carlomagno N, Incollingo P, Tammaro V, Peluso G, Rupealta N, Chiacchio G, Sandoval Sotelo ML, Minieri G, Pisani A, Riccio E, Sabbatini M, Bracale UM, Calogero A, et al. Diagnostic, Predictive, Prognostic, and Therapeutic Molecular Biomarkers in Third Millennium: A Breakthrough in Gastric Cancer. Biomed Res Int. 2017; 2017:7869802. https://doi.org/10.1155/2017/7869802 [PubMed]
- 50. Kahraman S, Yalcin S. Recent Advances in Systemic Treatments for HER-2 Positive Advanced Gastric Cancer. Onco Targets Ther. 2021; 14:4149–62. https://doi.org/10.2147/OTT.S315252 [PubMed]
- 51. Clifton GT, Peoples AG. Immunotherapy as a partner for HER2-directed therapies. Expert Rev Anticancer Ther. 2021; 21:739–46. https://doi.org/10.1080/14737140.2021.1894932 [PubMed]
- 52. Janjigian YY, Kawazoe A, Yañez P, Li N, Lonardi S, Kolesnik O, Barajas O, Bai Y, Shen L, Tang Y, Wyrwicz LS, Xu J, Shitara K, et al. The KEYNOTE-811 trial of dual PD-1 and HER2 blockade in HER2-positive gastric cancer. Nature. 2021; 600:727–30. https://doi.org/10.1038/s41586-021-04161-3 [PubMed]
- 53. Giampieri R, Maccaroni E, Mandolesi A, Del Prete M, Andrikou K, Faloppi L, Bittoni A, Bianconi M, Scarpelli M, Bracci R, Scartozzi M, Cascinu S. Mismatch repair deficiency may affect clinical outcome through immune response activation in metastatic gastric cancer patients receiving first-line chemotherapy. Gastric Cancer. 2017; 20:156–63. https://doi.org/10.1007/s10120-016-0594-4 [PubMed]
- 54. Ma C, Patel K, Singhi AD, Ren B, Zhu B, Shaikh F, Sun W. Programmed Death-Ligand 1 Expression Is Common in Gastric Cancer Associated With Epstein-Barr Virus or Microsatellite Instability. Am J Surg Pathol. 2016; 40:1496–506. https://doi.org/10.1097/PAS.0000000000000698 [PubMed]
- 55. Ganesh K, Stadler ZK, Cercek A, Mendelsohn RB, Shia J, Segal NH, Diaz LA
Jr . Immunotherapy in colorectal cancer: rationale, challenges and potential. Nat Rev Gastroenterol Hepatol. 2019; 16:361–75. https://doi.org/10.1038/s41575-019-0126-x [PubMed] - 56. Shen X, Zhao B. Efficacy of PD-1 or PD-L1 inhibitors and PD-L1 expression status in cancer: meta-analysis. BMJ. 2018; 362:k3529. https://doi.org/10.1136/bmj.k3529 [PubMed]
- 57. Morihiro T, Kuroda S, Kanaya N, Kakiuchi Y, Kubota T, Aoyama K, Tanaka T, Kikuchi S, Nagasaka T, Nishizaki M, Kagawa S, Tazawa H, Fujiwara T. PD-L1 expression combined with microsatellite instability/CD8+ tumor infiltrating lymphocytes as a useful prognostic biomarker in gastric cancer. Sci Rep. 2019; 9:4633. https://doi.org/10.1038/s41598-019-41177-2 [PubMed]
- 58. Di Bartolomeo M, Morano F, Raimondi A, Miceli R, Corallo S, Tamborini E, Perrone F, Antista M, Niger M, Pellegrinelli A, Randon G, Pagani F, Martinetti A, et al, and ITACA-S study group. Prognostic and Predictive Value of Microsatellite Instability, Inflammatory Reaction and PD-L1 in Gastric Cancer Patients Treated with Either Adjuvant 5-FU/LV or Sequential FOLFIRI Followed by Cisplatin and Docetaxel: A Translational Analysis from the ITACA-S Trial. Oncologist. 2020; 25:e460–8. https://doi.org/10.1634/theoncologist.2019-0471 [PubMed]
- 59. Polom K, Marano L, Marrelli D, De Luca R, Roviello G, Savelli V, Tan P, Roviello F. Meta-analysis of microsatellite instability in relation to clinicopathological characteristics and overall survival in gastric cancer. Br J Surg. 2018; 105:159–67. https://doi.org/10.1002/bjs.10663 [PubMed]
- 60. Velho S, Fernandes MS, Leite M, Figueiredo C, Seruca R. Causes and consequences of microsatellite instability in gastric carcinogenesis. World J Gastroenterol. 2014; 20:16433–42. https://doi.org/10.3748/wjg.v20.i44.16433 [PubMed]
- 61. Smyth EC, Wotherspoon A, Peckitt C, Gonzalez D, Hulkki-Wilson S, Eltahir Z, Fassan M, Rugge M, Valeri N, Okines A, Hewish M, Allum W, Stenning S, et al. Mismatch Repair Deficiency, Microsatellite Instability, and Survival: An Exploratory Analysis of the Medical Research Council Adjuvant Gastric Infusional Chemotherapy (MAGIC) Trial. JAMA Oncol. 2017; 3:1197–203. https://doi.org/10.1001/jamaoncol.2016.6762 [PubMed]
- 62. Choi YY, Kim H, Shin SJ, Kim HY, Lee J, Yang HK, Kim WH, Kim YW, Kook MC, Park YK, Kim HH, Lee HS, Lee KH, et al. Microsatellite Instability and Programmed Cell Death-Ligand 1 Expression in Stage II/III Gastric Cancer: Post Hoc Analysis of the CLASSIC Randomized Controlled study. Ann Surg. 2019; 270:309–16. https://doi.org/10.1097/SLA.0000000000002803 [PubMed]



