Introduction
Renal cell carcinoma (RCC) is a common and highly malignant tumor of the urinary system [1]. According to the latest GLOBOCAN estimations, 431,288 people worldwide are newly diagnosed with RCC, accounting for 2.2% of all new cancer cases [2]. RCC is a heterogeneous group of tumors, of which clear cell renal cell carcinoma (ccRCC) accounts for 75–90% [3–5]. The prognosis of early ccRCC is good; however, the 5-year survival rate of patients with metastatic ccRCC is less than 20% [6]. In recent years, targeted therapy and immune checkpoint inhibitors (ICIs) have been shown to be crucial and effective strategies for treating advanced RCC [7, 8]. Therefore, exploring reliable predictors of immunotherapy is critical for delivering precision therapy in ccRCC.
Cuproptosis, a copper-triggered modality of mitochondrial cell death, plays an important role in ccRCC [9]. Protein fatty acylation is a highly conserved post-translational modification of lysine [10]. Ferredoxin 1 (FDX1) is an upstream regulator of protein fatty acylation, and the Dihydrolipoamide S-acetyltransferase (DLAT) acts as a downstream component of FDX1 [10–12]. During this process, copper ions directly bind and contribute to the oligomerization of lipoylated DLAT, thus regulating the tricarboxylic acid (TCA) cycle [12]. Multiple studies have shown that DLAT is associated with the prognosis of liver, gastric, and pancreatic cancer [13–15]. However, its expression level and predictive value for ccRCC remain unclear.
In the present study, we aimed to investigate the role of DLAT in ccRCC. Multiple databases and ccRCC cell lines were used to assess the association between DLAT and clinical performance of ccRCC patients. Here, DLAT methylation, protein transcription, and immunotherapy value were evaluated. Our findings were verified using quantitative real-time PCR (qRT-PCR) and western blot in 75 ccRCC cohorts collected by our institute (Ningbo Urology and Nephrology Hospital [NBUNH]) and three cell lines.
Results
Expression analysis of DLAT
The TCGA RNA-seq analysis showed that mRNA levels of DLAT were lower in cancer tissues compared with the paracancerous tissues in BRCA, COAD, HNSC, KIRC, KIRP, PPAD, READ, and THCA (Figure 1A). Moreover, DLAT mRNA levels were lower in renal cancer tissues than that in paracancerous tissues from the TCGA-KIRC, ICGC (RECA-EU) and GEO (GSE36895) databases (Figures 1B–1D). Consistent with above database, the results were statistically significant in paired comparisons in our NBUNH cohort (P< .05) (Figure 1E).
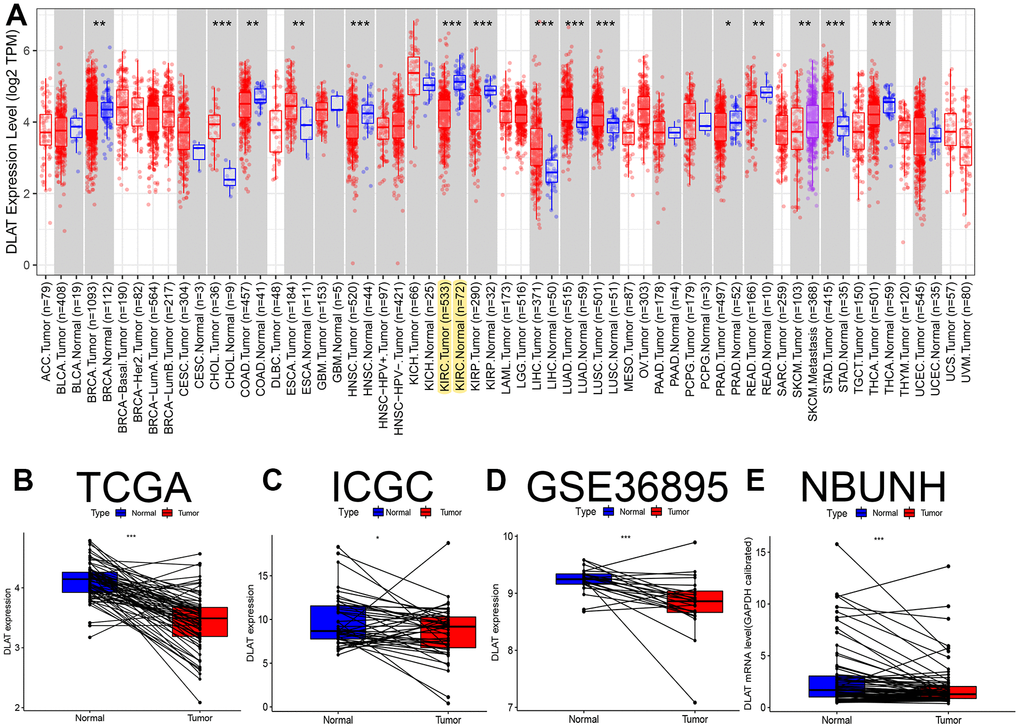
Figure 1. Pan-cancer analysis and expression level verification of DLAT in ccRCC. (A) Expression levels of DLAT in multiple tumors. Yellow background color indicates the expression of DLAT in KIRC. (B) Comparison of paired DLAT expression levels in ccRCC from TCGA. (C) Comparison of paired DLAT expression levels in ccRCC from ICGC (RECA-EU). (D) Comparison of paired DLAT expression levels in ccRCC from GEO (GSE36895). (E) Comparison of paired DLAT expression levels in ccRCC from NBUNH data cohort. *: P< 0.05; **: P< 0.01; ***: P< 0.001.
Analysis of survival and prognosis of DLAT expression
According to the median expression levels of DLAT in the TCGA cohort, patients were divided into high- and low-expression groups. The Kaplan–Meier curve showed that the overall survival (OS) in the low-expression group was lower than that in the high-expression group (P< .01, Figure 2A). In the E-MTAB-1980 cohort, the same results were obtained when the optimal cutoff DLAT expression level was selected (P< .01, Figure 2B). The ROC curve showed that DLAT performed well in distinguishing between benign and malignant tumors. The AUC value in the TCGA database was 0.919 (95% CI=0.888–0.945) (Figure 2C), whereas in the GSE36895 cohort, it was 0.849 (95% CI=0.729–0.949) (Figure 2D).
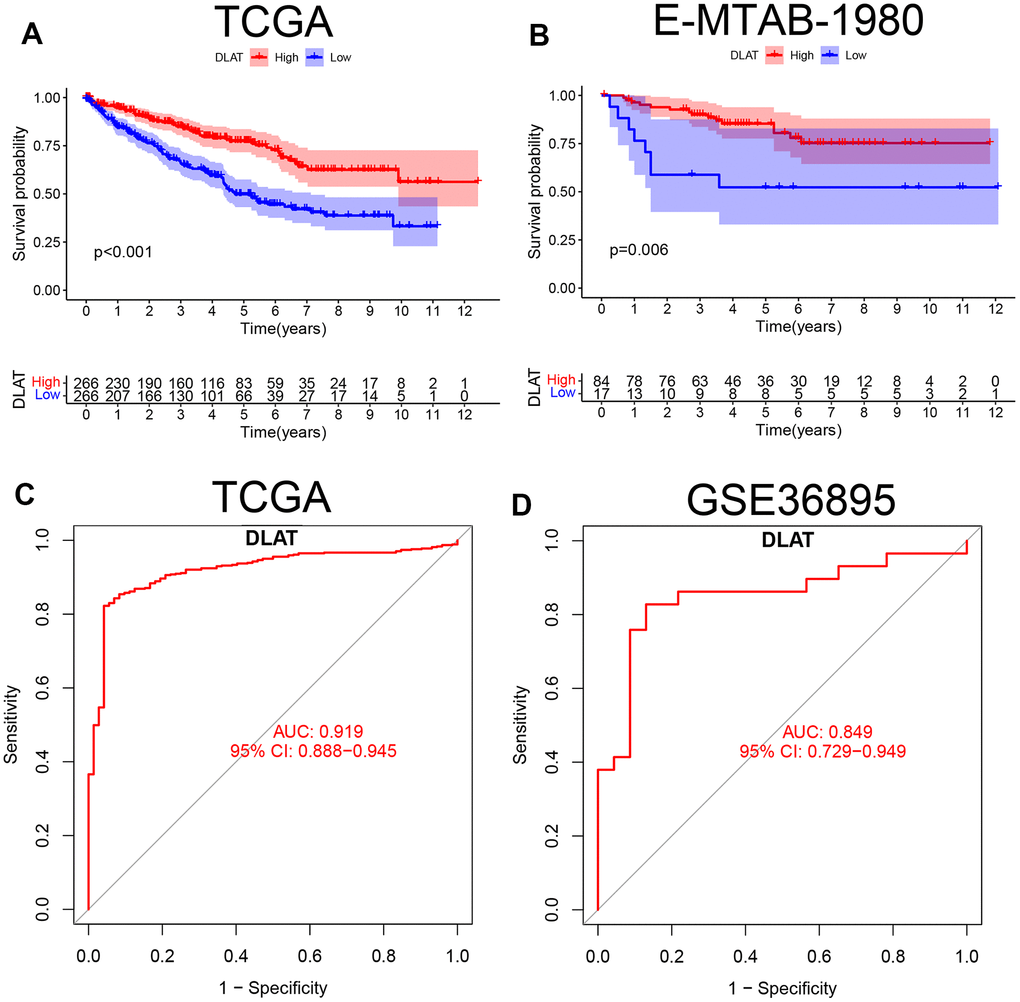
Figure 2. K-M survival curve and ROC of DLAT in ccRCC. (A) Kaplan-Meier survival curve between high- and low-expression of DLAT groups from TCGA. (B) Kaplan-Meier survival curve between high- and low-expression of DLAT groups from E-MTAB-1980. (C) ROC curves and AUC values of DLAT differences in ccRCC tissue and healthy controls from TCGA. (D) ROC curves and AUC values of DLAT differences in ccRCC tissue and healthy controls from GSE36859.
Clinical value of DLAT
Clinical characteristics were compared between the DLAT high- and low-expression groups in the TCGA cohort. The results showed that DLAT expression was lower in grade 3–4 than in grade 1–2 tumors, in stage III–IV than in stage I–II, in T3–4 than in T1–2, and in M1 than in M0 metastatic stage (P< .01, Figure 3A). Univariate and multivariate Cox analyses showed that DLAT expression could be used as an independent prognostic indicator of OS in ccRCC, apart from other clinical factors (Figure 3B, 3C).
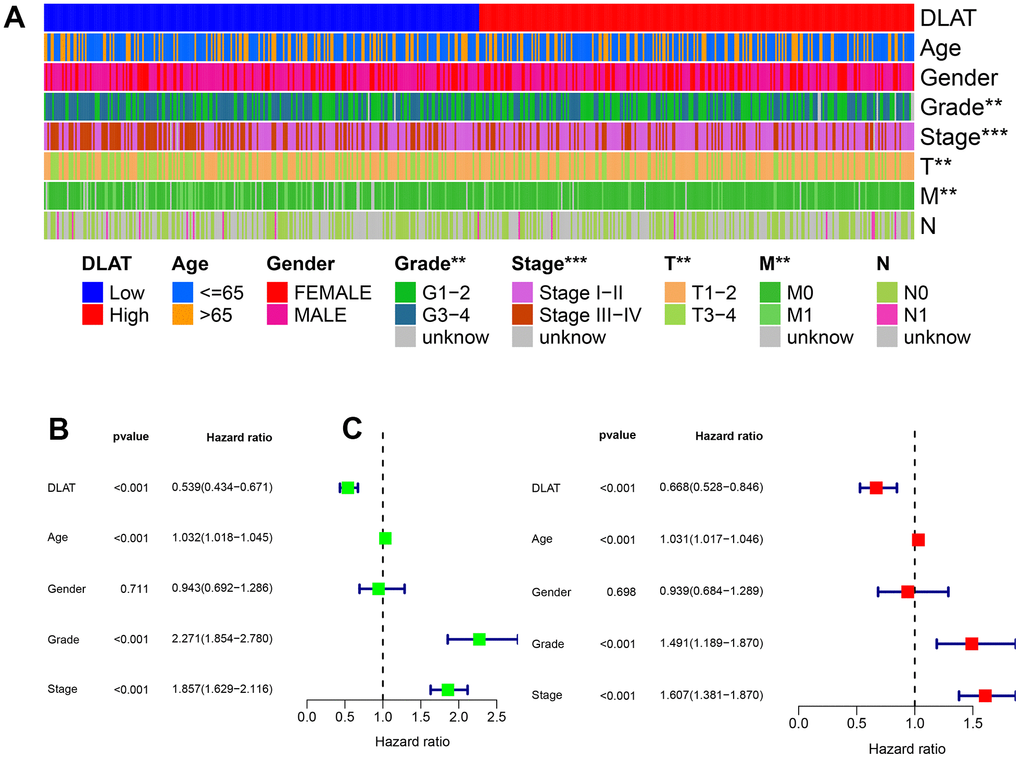
Figure 3. Clinical correlation and independent prognostic analysis. (A) Association between DLAT and traditional clinical features from TCGA. Blue on the left represents low DLAT expression, while red on the right represents high DLAT expression; and each line represents one clinical feature. (B, C) Univariate and multivariate Cox regression analyses confirmed the independent prognosis value of DLAT. *: P< 0.05; **: P< 0.01; ***: P< 0.001.
DLAT correlation and functional analysis
A total of 182 genes were related to DLAT with a correlation coefficient greater than 0.7. These included the copper cell death genes ATP7A, SLC31A1, DLD, and DBT (details are provided in Supplementary Material 1). The co-expression circular plot showed a correlation between 13 genes with the largest absolute values of the correlation coefficient, including DLD (Figure 4A). Biological processes (BP), cellular components (CC), and molecular functions (MF) were analyzed by GO enrichment analysis (Supplementary Material 2 and Figure 4B). KEGG pathway analysis showed that the co-expressed genes were mainly related to valine, leucine, and isoleucine degradation (Figure 4C and Supplementary Material 3).
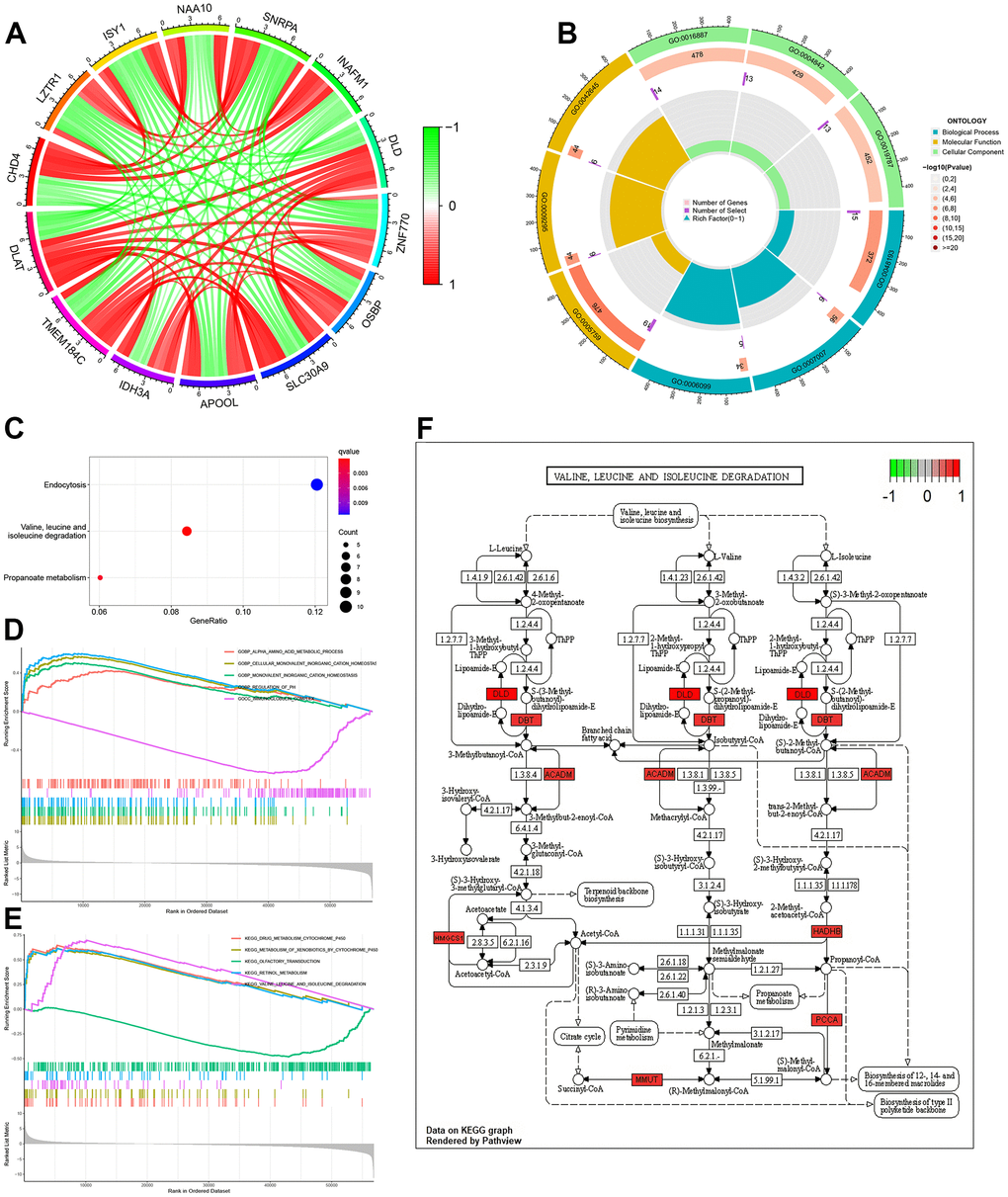
Figure 4. Function, pathway and GSEA analyses of DLAT-related genes. (A) Co-expression cycle graph of the 13 genes closely related to DLAT. The red line represents a positive correlation, while the green line represents a negative correlation. (B) GO circle graph of co-expressed genes. First circle: The top 9 GO terms, with the coordinate scale showing the gene number outside the circle. Second circle: The number of GO terms and Q values in the background gene. Third circle: Number of GO term associated genes. Fourth Circle: Abundance factor values for each GO term associated gene. (C) Bubble map of co-expressed genes from KEGG. (D) Function enrichment analysis between high- and low-expression groups of DLAT by GSEA. (E) Pathways analysis between high- and low-expression groups of DLAT by GSEA. (F) Pathways for valine, leucine and isoleucine degradation. Red represents key genes with high expression, while green represents key genes with low expression. BP: biological process; CC: cell component; MF: molecular function; GSEA: Gene Set Enrichment Analysis.
GSEA was performed using the c5.go.v7.4. symbols.gmt dataset, and the results showed that the main function related to differentiate DLAT expression included alpha amino acid metabolic process, cellular monovalent inorganic cation homeostasis, monovalent inorganic cation homeostasis, regulation of PH and immunoglobulin complex (Supplementary Material 4 and Figure 4D). In addition, the main pathways related to DLAT included drug metabolism-cytochrome P450, metabolism of xenobiotics by cytochrome P450, olfactory transduction, retinol metabolism and valine, leucine, and isoleucine degradation (Figure 4E and Supplementary Material 5). The results of KEGG pathway analysis (Figure 4C) and GSEA (Figure 4E) showed that DLAT was closely related to the degradation pathways of valine, leucine and isoleucine. Further analysis revealed that the DLAT-related genes, including DLD, DBT, ACADM, HMGCS1, HADHB, PCCA and MMUT were functioned in this pathway (Figure 4F).
Validation in vivo and in vitro
In normal human samples in the GTEx database, DLAT was relatively highly expressed in bone, muscle and heart tissues and intermediately expressed in normal kidney tissue (Figure 5A). In the cancer cell lines in the CCLE database, DLAT expression was relatively high in adrenal cancer, malignant rhabdoid tumor, and lung cancer and relatively low in kidney cancer, liposarcoma, and head and neck cancer (Figure 5B). During mRNA transcription, various modifications such as methylation affect transcription results. In this study, we examined the prognostic value of DLAT methylation in KIRC using the MethSurv. The DNA methylation heat map showed the highest DLAT methylation level in cg08065721 and the lowest methylation level in cg11372927 (Figure 5C). The final protein expression level of DLAT was consistent with its mRNA expression level. DLAT expression was high in the tubules of normal kidney tissues and low in those of tumor tissues (Figure 5D). Among the 33 CCLE RCC cell lines, DLAT had the highest expression in the KMRC20 cell line and the lowest expression in the BFTC909 cell line (Figure 5E).
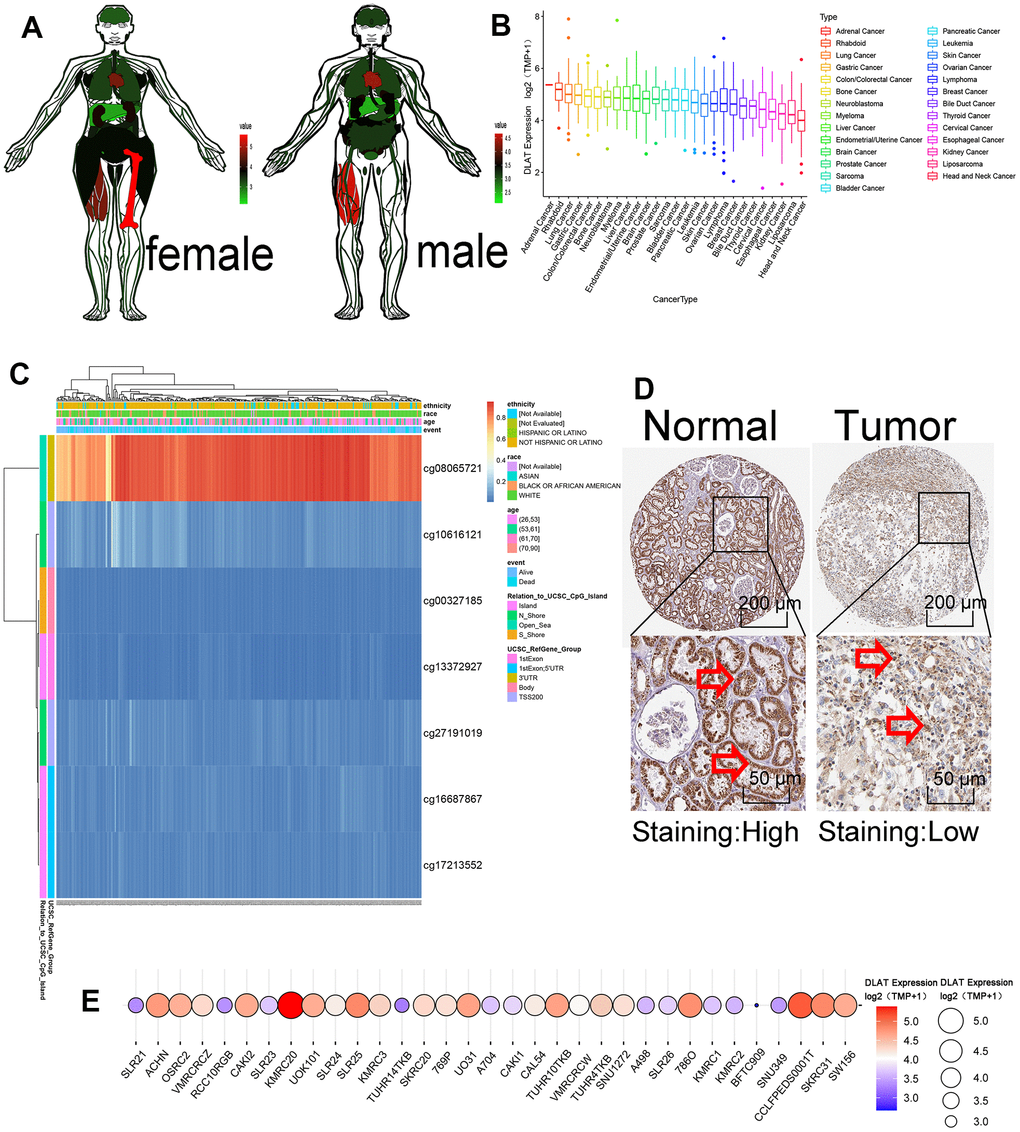
Figure 5. Methylation and protein expressions of DLAT. (A) DLAT expression levels in various human tissues. Red represents high expression and green represents low expression. (B) The expression level of DLAT in each tumor cell line. Decreased expression from left to right. (C) DNA methylation hot diagram of DLAT. Red represents high expression and blue means low expression. (D) DLAT protein level based on Human Protein Atlas. Normal tissues are on the left, while tumor tissues are on the right. The arrow is marked with DLAT protein. (E) DLAT expression at 33 kidney cancer cell lines. Red represents high expression, and blue represents low expression. The size of the circle represents DLAT expression.
In vitro experiments showed that the expression of DLAT in normal renal cells (HK-2) was significantly higher than that in renal cancer cells (OS-RC-2 and 786-O), regardless of the mRNA or protein expression (Figures 6A, 6B).
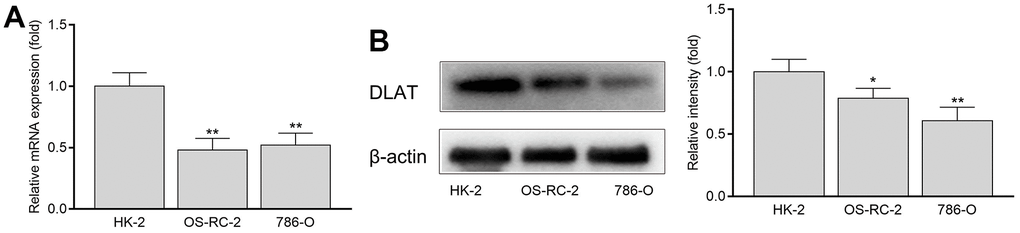
Figure 6. The mRNA and protein expression of DLAT in HK-2, OS-RC-2 and 786-O cells. (A) DLAT mRNA expression. (B) DLAT protein expression.
Immune infiltration and TME analysis
DLAT expression is correlated to tumor immune infiltration and TME. We used the CIBERSORT method to explore 22 types of immune cells, 13 of which were found to be differentially expressed in ccRCC (P< .05, Figure 7A). Further analyses showed that DLAT was correlated with 14 types of immune cells (Figure 7B). Analysis of the TME showed that both ImmuneScore and ESTIMATEScore increased in the low DLAT expression group (P< .001, Figure 7C).
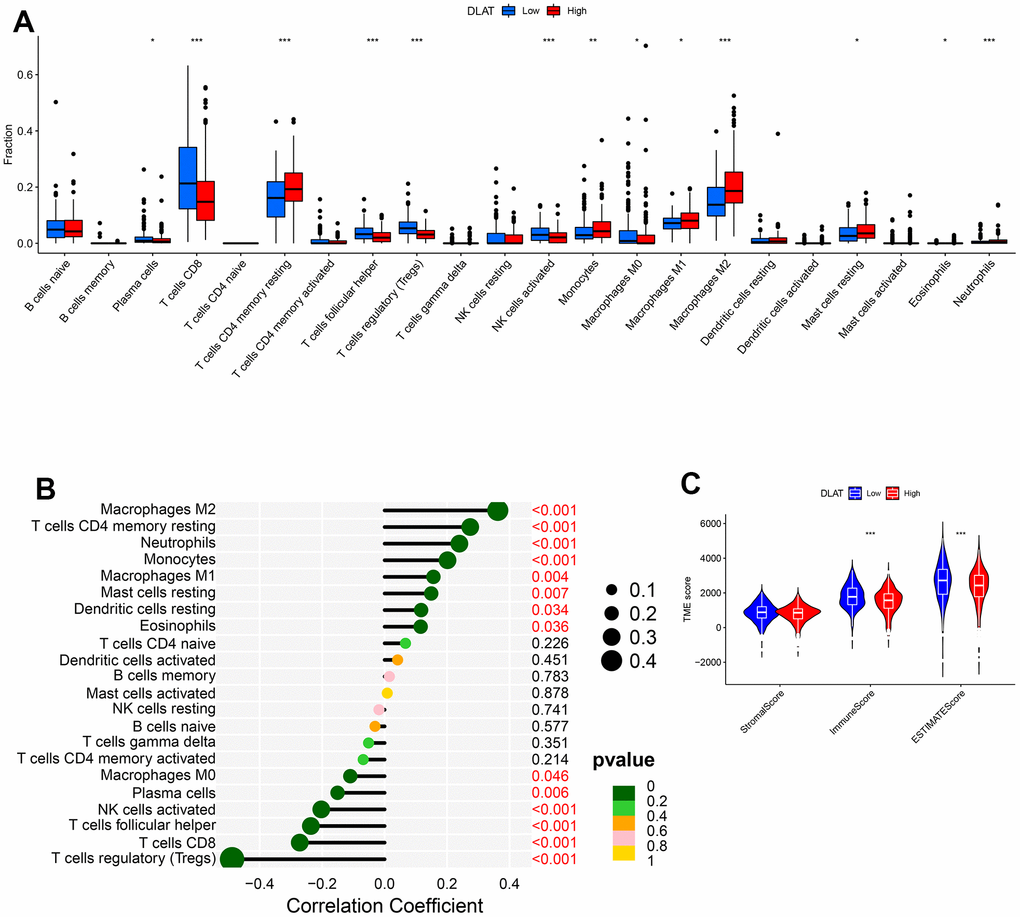
Figure 7. DLAT-mediated immune invasion and micro-environment. (A) Boxplot shows the differences of the 22 types of immune cells in high- and low-expression groups of DLAT. (B) Lollipop plot indicates the correlation between DLAT and 22 types of immune cells. (C) Violin graph shows the relationship between DLAT expression and TME (higher Immune and ESTIMATE Scores were witnessed in low-expression group of DLAT.).
Sensitivity to immunotherapy
Immune checkpoints are reliable molecules for assessing patient response to immunotherapy. Here, we assessed the differences in DLAT expression in relation to common immune checkpoints. The results showed that the expression of LAG-3, TIGIT, CTLA4, and PD-1 was upregulated, whereas that of HER-2, PD-L2, PD-L1, and TIM-3 was downregulated in the DLAT low-expression group (Figure 8A). In addition, TIDE-based simulations of the two main mechanisms of tumor immune evasion provide predictive outcomes for immunotherapy. Patients with DLAT low-expression showed a higher TIDE score and more obvious immune dysfunction and exclusion (Figures 8B–8E).
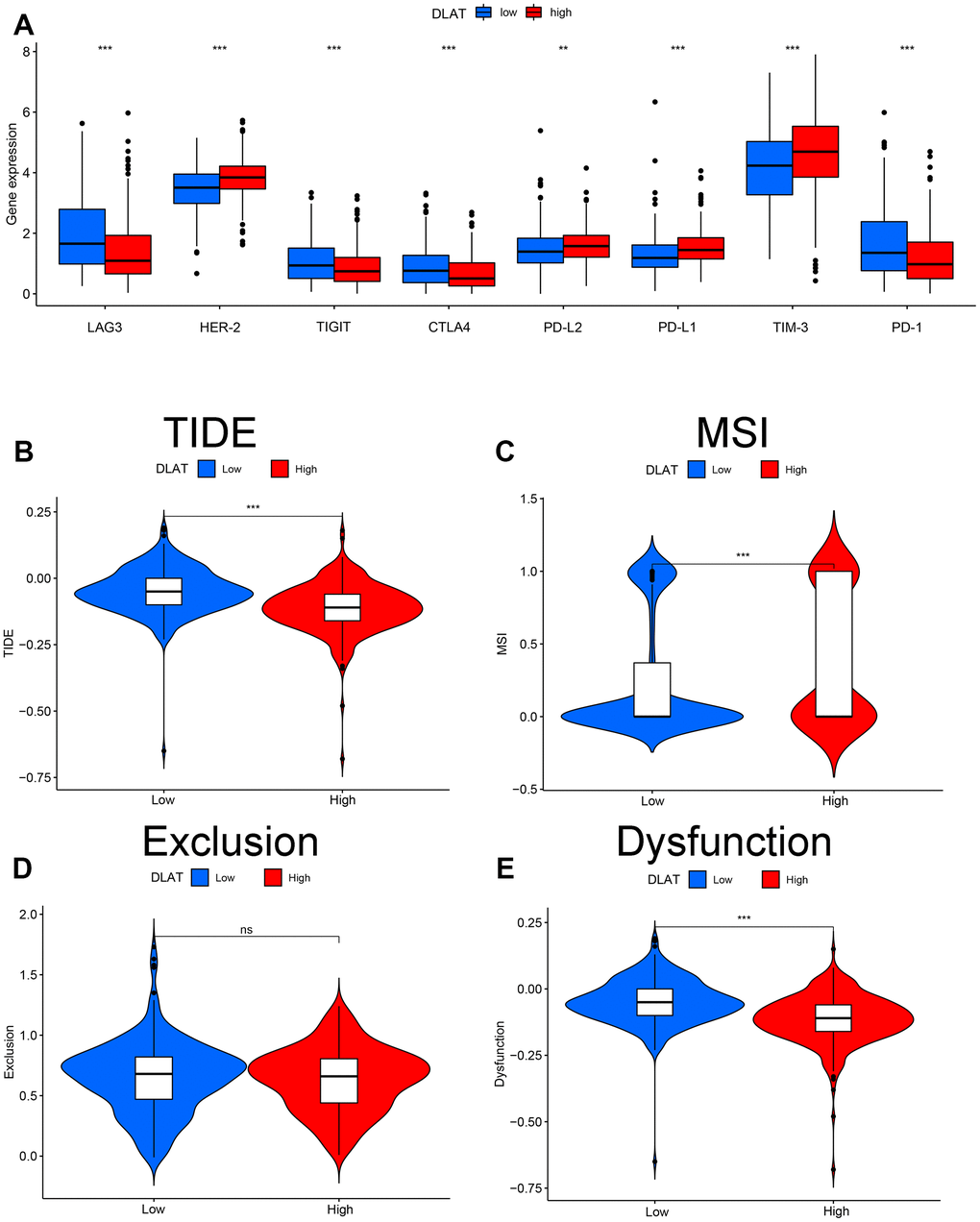
Figure 8. Effect of DLAT on immune regulatory and TIDE in ccRCC. (A) Gene expression of each immune checkpoint in high- and low-expression of DLAT from TCGA. DLAT expression distribution in the TIDE data set: (B) TIDE, (C) MSI, (D) Exclusion, and (E) Dysfunction.
Discussion
In this study, we determined the role of DLAT in ccRCC patients, and discussed its main functions, especially its key role in immunotherapy.
DLAT is a component of the pyruvate dehydrogenase complex and participates in the TCA cycle in the glucose metabolism pathway. Lipid acylation of DLAT, which is induced by FDX1, directly binds to copper ions and contributes to form protein oligomers. Subsequently, it induces protein toxicity stress and promotes copper death, which enables a crucial role of DLAT during the process of cuproptosis. Therefore, DLAT has the potential to be therapeutic targets as well as the tumor prognostic indicators of ccRCC.
Multiple researches suggested that DLAT plays vital roles in various tumors. Through examination of multiple databases (TCGA, ICGC and GEO database), we confirmed that the expression level of DLAT in ccRCC was lower than that in normal tissues. Furthermore, 75 pairs of ccRCC and adjacent tissues in our database (NBUNH), as well as two renal cancer cells (OS-RC-2 and 786-O) and HK-2 normal cells were also verified, and similar results were found in the study of Jiang et al., whose research conducted a qRT-PCR study on 40 matched ccRCC tissues [16]. To further verify the function of DLAT in renal cancer cell lines, researchers constructed a lentivirus overexpressing DLAT vector that effectively inhibited the growth and metastasis of renal cancer cells [16]. Our results, based on the TCGA and ArrayExpress databases, showed that patients with high DLAT expression had better OS, which indicated that DLAT functions as a tumor suppressor (P< .05). According to ROC curve analysis, DLAT expression is a good indicator to distinguish benign from malignant renal tumors, which is consistent with the previous results reported by the study of Jiang et al. [16].
ccRCC has a unique metabolic profile, characterized by elevated levels of lactate, glutamate, pyruvate, glutamine, and creatine and decreased levels of acetate, malate, and amino acids such as valine, alanine, and aspartate [17, 18]. More severe metabolic dysregulation, including changes in valine, leucine, and isoleucine levels, has been observed in stage IV ccRCC [19]. Our results of KEGG and GSEA pathway analysis showed that DLAT was closely associated with the degradation pathway of valine, leucine, and isoleucine. Based on the above observation, we further examined the related genes in valine, leucine, and isoleucine degradation pathway. In this pathway, 7 genes (DLD, DBT, ACADM, HMGCS1, HADHB, PCCA and MMUT) were positively correlated with DLAT gene. Among these 7 genes, DLD and DBT were cuproptosis-related genes. Hence, we consider that the valine, leucine, and isoleucine degradation pathway is closely related to cuproptosis, of which DLAT may promote the progression of this pathway.
Consistent with our research, many studies have identified that high copper levels in tumors can regulate kinase activity, inhibit autophagy, and regulate fat metabolism [20–22]. Zheng reported that disulfiram/copper co-delivery triggered tumor cell autophagy, induced immunogenic cell death, activated tumor-infiltrating macrophages and dendritic cells, and primed T and NK (natural killer) cells, resulting in anti-tumor immunity and tumor regression [23]. We hypothesize that DLAT might promote apoptosis by influencing energy production. A recent research published in Science points out that cuproptosis is an unconventional mechanism of cell death concerning the protein lipoylation in TCA cycle [24]. Moreover, it is feasible to manage intracellular copper levels within a certain range to selectively kill tumor cells [25]. This is promising for novel insights to exploit copper toxicity in tumor therapy regimens.
Immunotherapy for tumors focuses on restoring host anti-tumor immune responses by blocking immune checkpoints [26, 27]. Huang et al. [28] reported that DLAT can promote the metabolism and biogenesis of liver hepatocellular carcinoma cells through MET kinase mediated phosphorylation. Fang et al. reported that DLAT was correlated with progression, chemo-resistance, and immune infiltration in pancreatic adeno-carcinoma [29]. It is reported that inhibitors of CTLA-4, LAG-3, PD-1, PD-L1 and other immune checkpoints have been shown to improve the OS of patients with advanced ccRCC [15, 30, 31]. The present study showed that immune checkpoints of LAG-3, TIGIT, CTLA4, and PD-1 expressions, as well as ImmuneScore and ESTIMATEScore were upregulated in DLAT low-expressed group. In addition, TIDE was applied to model the two major mechanisms of tumor immune evasion to provide predictive outcomes for immunotherapy. Results showed that the patients with low DLAT expression had higher TIDE scores compared with DLAT high-expressed group. Therefore, we consider that patients with low DLAT expression would develop more pronounced immune dysfunction and exclusion, resulting in poorer response to immunotherapy.
This study provides evidences for comprehensively understanding the role of DLAT on ccRCC. Although the robustness of our results was confirmed through multiple validations, such as datasets from multiple external databases, protein expression levels, and basic experiments, this study has some limitations. Specifically, the 76 ccRCC specimens collected were all early-stage tumors and did not cover all clinical statuses; therefore, additional immunotherapy cohorts are needed to validate and optimize the findings.
Conclusions
We demonstrated the abnormal downregulation of DLAT mRNA and protein expression levels in ccRCC tissues. DLAT could serve as a biomarker to predict the clinical efficacy and sensitivity of targeted therapy in patients with ccRCC.
Materials and Methods
Data collection and experiment queuing
RNA sequencing and clinical information of patients with ccRCC were obtained from The Cancer Genome Atlas Kidney Renal Clear Cell Carcinoma (TCGA-KIRC) database (https://portal.gdc.cancer.gov/; normal samples [N]=72; kidney tumor samples [T]=532). The International Cancer Genome Consortium (ICGC) (Renal Cell Cancer [RECA]-EU; http://dcc.icgc.org; N=45; T=91), ArrayExpress (E-MTAB-1980; https://www.ebi.ac.uk/arrayexpress/; T=101), and Gene Expression Omnibus (GEO; GSE36895; https://www.ncbi.nlm.nih.gov/geo/; N=29; T=23) databases were used for validation. The clinical data for each database are presented in Table 1.
Table 1. Summary clinical characteristics of ccRCC patients.
| Characteristics | TCGAN=532 | ICGC N=91 | E-MTAB-1990 N=101 | GSE36895 N=29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age category | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| <65/≥65/NA | 333/199 | 57/34 | 52/49 | 15/13/1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male/ Female | 345/187 | 52/39 | 77/24 | 17/12 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vital status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Alive/ Dead | 357/175 | 61/30 | 78/23 | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Grade | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G1/G2/G3/G4/NA | 14/228/206/76/8 | NA | 13/59/22/5/2 | 2/13/6/8/0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I/ II/ III/ IV/ NA | 266/57/123/83/3 | NA | 66/10/13/12/0 | 5/2/3/6/13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1/ T2/ T3/ T4/ NA | 272/69/180/11 | NA | 68/11/21/1/0 | 14/4/8/3/0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M0/ M1/ MX | 421/79/32 | NA | 89/12/0 | 16/4/9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N0/ N1/N2 / NA | 240/16/0/276 | NA | 94/3/4/0 | 11/3/15/0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NA: Clinical data are unknown. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Renal cancer samples from 75 pairs of ccRCC patients were collected from NBUNH. This study was approved by the Ethics Committee of Ningbo Urology and Nephrology Hospital. Written informed consent to participate in the study was obtained from all participants.
Cell cultures
The normal kidney cell line (HK-2) and renal cancer cell lines (786-O and ACHN) were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). HK-2 cells were cultured in Dulbecco’s modified DMEM medium (HyClone Laboratories, Logan, UT, USA) and OS-RC-2 and 786-O cells were cultured in RMPI-1640 medium (HyClone, Logan, UT, USA). All cells were incubated at 37° C in 5% CO2 after supplementing the culture medium with 10% heat-inactivated fetal bovine serum (HyClone, Auckland, New Zealand), 100 U/mL streptomycin, and 100 mg/mL penicillin (HyClone, Logan, UT, USA).
RNA extraction, reverse transcription, and qRT-PCR
Total RNA was extracted from clinical samples or cells using the E.Z.N.A.® Total RNA Kit (Omega Bio-Tek, Norcross, GA, USA). RNA (1 μg) was reverse-transcribed into cDNA using ABScript II RT Master Mix (ABclonal, Woburn, MA, USA). RT-qPCR was performed on a 7500 real-time PCR system with 2X Universal SYBR Green Fast qPCR Mix (ABclonal, Woburn, MA, USA) according to the manufacturer’s instructions. The primer sequences used were as follows: DLAT, sense: 5'-CCTCCCACAGGTCCTGGAAT-3',’ anti-sense: 5'-GTGCAATAACCCGACGAATGT-3'; GAPDH, sense: 5'-GGAAGCTTGTCATCAATGGAAATC-3’, anti-sense: 5'-TGATGACCCTTTTGGCTCCC-3’. Relative gene expression was normalized to that of GAPDH and the 2− ΔΔCt method was used to calculate the relative expression levels of DLAT.
Western blot analysis
Cell lysates were harvested in radioimmunoprecipitation assay (RIPA) buffer (Solarbio, Beijing, China) containing 1% PMSF protease inhibitor (Solarbio). Total protein concentration was calculated using a BCA Protein Assay Kit (Beyotime, Beijing, China). Total protein (20 μg) samples were loaded and separated using 12% SDS-PAGE, transferred to PVDF membranes, blocked with 5% non-fat dry milk, and incubated overnight with diluted primary antibodies against DLAT (Proteintech, Wuhan, China) or β-actin (Proteintech) at 4° C. The blots were then washed with TBST, incubated with horseradish peroxidase-labeled secondary antibodies (Boster, Wuhan, China), and visualized using an enhanced chemiluminescence reagent.
Comparison of expression levels, Kaplan–Meier survival, and receiver operating characteristic (ROC) analysis
TIMER2.0 (http://timer.cistrome.org) was used to determine DLAT expression levels in various tumor types. Boxplots were used to visualize the expression levels of DLAT in discrete and paired tissues. Survival curves were generated to analyze the different survival outcomes in the high- and low-expression groups. ROC curve was drawn, and the area under the curve (AUC) was calculated to evaluate the specificity and sensitivity of DLAT expression in predicting benign and malignant tumors.
Clinical relevance and independence analysis
The R package Complex Heatmap was used to draw heat maps to compare clinic-pathological parameters in the high- and low-expression groups. Univariate and multivariate Cox regression analyses were performed in R to assess the independence value of DLAT expression in distinguishing ccRCC in addition to traditional clinical characteristics.
Functional and pathway analysis of co-expressed genes
|Pearson’s correlation coefficient|>0.7 and P< .001 were considered as the cutoff criteria for genes correlated to DLAT. The R packages circlize, corrplot, clusterProfiler, org.Hs.eg.db, dplyr, enrichplot, ggplot2, RColorBrewer, ComplexHeatmap, R.utils, and Pathview were used for co-expressed results and Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses. The Molecular Signatures Database (MSigDB) gene sets c5.go.v7.4. symbols.gmt, and c2.cp.kegg.v7.4. symbols.gmt (http://software.broadinstitute.org/gsea/msigdb/index.jsp) were used for Gene Set Enrichment Analysis (GSEA).
mRNA expression, methylation, and protein expression of DLAT
The Genotype-Tissue Expression (GTEx) database (https://gtexportal.org/home/) was used to verify DLAT expression in normal human organs. The Cancer Cell Line Encyclopedia (CCLE) database (https://sites.broadinstitute.org/ccle) was used to verify the expression levels of DLAT in tumor cell lines. The MethSurv tool (https://biit.cs.ut.ee/methsurv/) was used to evaluate the prognostic value of DLAT for different CpG methylation patterns in ccRCC patients. The Human Protein Atlas database (https://www.proteinatlas.org/) was used to determine protein expression levels of DLAT.
Immune infiltration and tumor microenvironment (TME)
The CIBERSORT algorithm was used to evaluate the relationship between DLAT expression and the 22 types of immune cells. The lollipop plot allowed the visualization of the correlation between DLAT expression and immune cell types. The ESTIMATE algorithm was used to evaluate the immune microenvironment (ImmuneScore, StromalScore, ESTIMATEScore and TumorPurity) levels between high and low DLAT expression groups. These operations were performed in R using the packages limma, estimate, e1071, reshape2, vioplot, ggExtra, and ggpubr.
Immune checkpoints and immune response prediction
The relationship between DLAT expression and hotspot immune checkpoints was visualized using box plots. The Tumor Immune Dysfunction and Exclusion (TIDE) database (http://tide.dfci.harvard.edu) was used to predict immunotherapy responses to differential expression levels of DLAT.
Statistical analysis
All our operations and statistical analyses were performed using R software version v4.1.1 (https://www.r-project.org/). Statistically significant differences between two groups were determined using the Student’s t-test, and those among three or more groups were calculated using ANOVA. Pearson’s correlation was used to analyze the correlation between two different genes. A P value < .05 was considered statistically significant. NS, P> .05; *, P< .05; **, P< .01; ***, P< .001.
Data availability statement
The datasets for this study can be found in ICGC at http://dcc.icgc.org, KIRC at https://portal.gdc.cancer.gov/, ArrayExpress at https://www.ebi.ac.uk/arrayexpress/, and GEO at https://www.ncbi.nlm.nih.gov/geo/. The authors confirm that the data supporting the findings of this study are available within the article and its Supplementary Materials.
Consent
The consent was obtained from the patients for the publication of this paper.
Supplementary Materials
Author Contributions
SH and CC contributed to the study conception and design, and wrote the first draft of the manuscript. SH conducted the experiments. KZ and CC performed the statistical analyses. XW and XW organized the database. DC and GW supervised the paper. All authors contributed to manuscript revision and read and approved the submitted version.
Acknowledgments
We are grateful to the people who have created and administered the TCGA, GEO, ICGC, ArrayExpress, CCLE, Human Protein Atlas, and TIDE databases for gene expression and image information for our study and to the developers of the MethSurv tool for providing methylation data. We thank NBUNH for the tissue samples and the basic laboratory of NBUNH for the experimental support.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
Ethical Statement and Consent
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Ethics Committee of Ningbo Urology and Nephrology Hospital. Written informed consent was obtained from the patients involved in this study.
Funding
This research was funded by the Natural Science Foundation of Ningbo (grant number: 2023J233), General Health Foundation of Zhejiang Province (grant number: 2022KY1177), and Science and Technology Project of Yinzhou (grant number: 2022AS033, 2023AS028, 2023AS062 and 2023AS064).
References
- 1. Capitanio U, Bensalah K, Bex A, Boorjian SA, Bray F, Coleman J, Gore JL, Sun M, Wood C, Russo P. Epidemiology of Renal Cell Carcinoma. Eur Urol. 2019; 75:74–84. https://doi.org/10.1016/j.eururo.2018.08.036 [PubMed]
- 2. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 71:209–49. https://doi.org/10.3322/caac.21660 [PubMed]
- 3. Yin L, Li W, Xu A, Shi H, Wang K, Yang H, Wang R, Peng B. SH3BGRL2 inhibits growth and metastasis in clear cell renal cell carcinoma via activating hippo/TEAD1-Twist1 pathway. EBioMedicine. 2020; 51:102596. https://doi.org/10.1016/j.ebiom.2019.12.005 [PubMed]
- 4. Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM, Piñeros M, Znaor A, Bray F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2019; 144:1941–53. https://doi.org/10.1002/ijc.31937 [PubMed]
- 5. Ljungberg B, Albiges L, Abu-Ghanem Y, Bensalah K, Dabestani S, Fernández-Pello S, Giles RH, Hofmann F, Hora M, Kuczyk MA, Kuusk T, Lam TB, Marconi L, et al. European Association of Urology Guidelines on Renal Cell Carcinoma: The 2019 Update. Eur Urol. 2019; 75:799–810. https://doi.org/10.1016/j.eururo.2019.02.011 [PubMed]
- 6. Coppin C, Kollmannsberger C, Le L, Porzsolt F, Wilt TJ. Targeted therapy for advanced renal cell cancer (RCC): a Cochrane systematic review of published randomised trials. BJU Int. 2011; 108:1556–63. https://doi.org/10.1111/j.1464-410X.2011.10629.x [PubMed]
- 7. Hammers HJ, Plimack ER, Infante JR, Rini BI, McDermott DF, Lewis LD, Voss MH, Sharma P, Pal SK, Razak ARA, Kollmannsberger C, Heng DYC, Spratlin J, et al. Safety and Efficacy of Nivolumab in Combination With Ipilimumab in Metastatic Renal Cell Carcinoma: The CheckMate 016 Study. J Clin Oncol. 2017; 35:3851–8. https://doi.org/10.1200/JCO.2016.72.1985 [PubMed]
- 8. Motzer RJ, Escudier B, McDermott DF, George S, Hammers HJ, Srinivas S, Tykodi SS, Sosman JA, Procopio G, Plimack ER, Castellano D, Choueiri TK, Gurney H, et al, and CheckMate 025 Investigators. Nivolumab versus Everolimus in Advanced Renal-Cell Carcinoma. N Engl J Med. 2015; 373:1803–13. https://doi.org/10.1056/NEJMoa1510665 [PubMed]
- 9. Yuan H, Qin X, Wang J, Yang Q, Fan Y, Xu D. The cuproptosis-associated 13 gene signature as a robust predictor for outcome and response to immune- and targeted-therapies in clear cell renal cell carcinoma. Front Immunol. 2022; 13:971142. https://doi.org/10.3389/fimmu.2022.971142 [PubMed]
- 10. Tang D, Chen X, Kroemer G. Cuproptosis: a copper-triggered modality of mitochondrial cell death. Cell Res. 2022; 32:417–8. https://doi.org/10.1038/s41422-022-00653-7 [PubMed]
- 11. Li SR, Bu LL, Cai L. Cuproptosis: lipoylated TCA cycle proteins-mediated novel cell death pathway. Signal Transduct Target Ther. 2022; 7:158. https://doi.org/10.1038/s41392-022-01014-x [PubMed]
- 12. Tsvetkov P, Coy S, Petrova B, Dreishpoon M, Verma A, Abdusamad M, Rossen J, Joesch-Cohen L, Humeidi R, Spangler RD, Eaton JK, Frenkel E, Kocak M, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022; 375:1254–61. https://doi.org/10.1126/science.abf0529 [PubMed]
- 13. Bai WD, Liu JY, Li M, Yang X, Wang YL, Wang GJ, Li SC. A Novel Cuproptosis-Related Signature Identified DLAT as a Prognostic Biomarker for Hepatocellular Carcinoma Patients. World J Oncol. 2022; 13:299–310. https://doi.org/10.14740/wjon1529 [PubMed]
- 14. Huang X, Zhou S, Tóth J, Hajdu A. Cuproptosis-related gene index: A predictor for pancreatic cancer prognosis, immunotherapy efficacy, and chemosensitivity. Front Immunol. 2022; 13:978865. https://doi.org/10.3389/fimmu.2022.978865 [PubMed]
- 15. Liu YF, Zhang ZC, Wang SY, Fu SQ, Cheng XF, Chen R, Sun T. Immune checkpoint inhibitor-based therapy for advanced clear cell renal cell carcinoma: A narrative review. Int Immunopharmacol. 2022; 110:108900. https://doi.org/10.1016/j.intimp.2022.108900 [PubMed]
- 16. Jiang A, Luo P, Chen M, Fang Y, Liu B, Wu Z, Qu L, Wang A, Wang L, Cai C. A new thinking: deciphering the aberrance and clinical implication of copper-death signatures in clear cell renal cell carcinoma. Cell Biosci. 2022; 12:209. https://doi.org/10.1186/s13578-022-00948-7 [PubMed]
- 17. Hakimi AA, Reznik E, Lee CH, Creighton CJ, Brannon AR, Luna A, Aksoy BA, Liu EM, Shen R, Lee W, Chen Y, Stirdivant SM, Russo P, et al. An Integrated Metabolic Atlas of Clear Cell Renal Cell Carcinoma. Cancer Cell. 2016; 29:104–16. https://doi.org/10.1016/j.ccell.2015.12.004 [PubMed]
- 18. Wettersten HI, Hakimi AA, Morin D, Bianchi C, Johnstone ME, Donohoe DR, Trott JF, Aboud OA, Stirdivant S, Neri B, Wolfert R, Stewart B, Perego R, et al. Grade-Dependent Metabolic Reprogramming in Kidney Cancer Revealed by Combined Proteomics and Metabolomics Analysis. Cancer Res. 2015; 75:2541–52. https://doi.org/10.1158/0008-5472.CAN-14-1703 [PubMed]
- 19. Li HJ, Li WX, Dai SX, Guo YC, Zheng JJ, Liu JQ, Wang Q, Chen BW, Li GH, Huang JF. Identification of metabolism-associated genes and pathways involved in different stages of clear cell renal cell carcinoma. Oncol Lett. 2018; 15:2316–22. https://doi.org/10.3892/ol.2017.7567 [PubMed]
- 20. Tang S, Liang C, Hou W, Hu Z, Chen X, Zhao J, Zhang W, Duan Z, Bai L, Zheng S. ATP7B R778L mutant hepatocytes resist copper toxicity by activating autophagy and inhibiting necroptosis. Cell Death Discov. 2023; 9:344. https://doi.org/10.1038/s41420-023-01641-5 [PubMed]
- 21. Wen H, Qu C, Wang Z, Gao H, Liu W, Wang H, Sun H, Gu J, Yang Z, Wang X. Cuproptosis enhances docetaxel chemosensitivity by inhibiting autophagy via the DLAT/mTOR pathway in prostate cancer. FASEB J. 2023; 37:e23145. https://doi.org/10.1096/fj.202300980R [PubMed]
- 22. Zhou C, Yang J, Liu T, Jia R, Yang L, Sun P, Zhao W. Copper metabolism and hepatocellular carcinoma: current insights. Front Oncol. 2023; 13:1186659. https://doi.org/10.3389/fonc.2023.1186659 [PubMed]
- 23. Zheng Z, Zhang J, Jiang J, He Y, Zhang W, Mo X, Kang X, Xu Q, Wang B, Huang Y. Remodeling tumor immune microenvironment (TIME) for glioma therapy using multi-targeting liposomal codelivery. J Immunother Cancer. 2020; 8:e000207. https://doi.org/10.1136/jitc-2019-000207 [PubMed]
- 24. Kahlson MA, Dixon SJ. Copper-induced cell death. Science. 2022; 375:1231–2. https://doi.org/10.1126/science.abo3959 [PubMed]
- 25. Ge EJ, Bush AI, Casini A, Cobine PA, Cross JR, DeNicola GM, Dou QP, Franz KJ, Gohil VM, Gupta S, Kaler SG, Lutsenko S, Mittal V, et al. Connecting copper and cancer: from transition metal signalling to metalloplasia. Nat Rev Cancer. 2022; 22:102–13. https://doi.org/10.1038/s41568-021-00417-2 [PubMed]
- 26. Bedke J, Albiges L, Capitanio U, Giles RH, Hora M, Ljungberg B, Marconi L, Klatte T, Volpe A, Abu-Ghanem Y, Dabestani S, Fernández-Pello S, Hofmann F, et al. The 2022 Updated European Association of Urology Guidelines on the Use of Adjuvant Immune Checkpoint Inhibitor Therapy for Renal Cell Carcinoma. Eur Urol. 2023; 83:10–4. https://doi.org/10.1016/j.eururo.2022.10.010 [PubMed]
- 27. Fitzgerald KN, Duzgol C, Knezevic A, Shapnik N, Kotecha R, Aggen DH, Carlo MI, Shah NJ, Voss MH, Feldman DR, Motzer RJ, Lee CH. Progression-free Survival After Second Line of Therapy for Metastatic Clear Cell Renal Cell Carcinoma in Patients Treated with First-line Immunotherapy Combinations. Eur Urol. 2023; 83:195–9. https://doi.org/10.1016/j.eururo.2022.10.017 [PubMed]
- 28. Huang X, Gan G, Wang X, Xu T, Xie W. The HGF-MET axis coordinates liver cancer metabolism and autophagy for chemotherapeutic resistance. Autophagy. 2019; 15:1258–79. https://doi.org/10.1080/15548627.2019.1580105 [PubMed]
- 29. Fang Z, Wang W, Liu Y, Hua J, Liang C, Liu J, Zhang B, Shi S, Yu X, Meng Q, Xu J. Cuproptosis-Related Gene DLAT as a Novel Biomarker Correlated with Prognosis, Chemoresistance, and Immune Infiltration in Pancreatic Adenocarcinoma: A Preliminary Study Based on Bioinformatics Analysis. Curr Oncol. 2023; 30:2997–3019. https://doi.org/10.3390/curroncol30030228 [PubMed]
- 30. Chen YW, Rini BI, Beckermann KE. Emerging Targets in Clear Cell Renal Cell Carcinoma. Cancers (Basel). 2022; 14:4843. https://doi.org/10.3390/cancers14194843 [PubMed]
- 31. Lee CH, Jung SJ, Seo WI, Chung JI, Lee DS, Jeong DH, Jeon Y, Choi I. Coexpression of lymphocyte-activation gene 3 and programmed death ligand-1 in tumor infiltrating immune cells predicts worse outcome in renal cell carcinoma. Int J Immunopathol Pharmacol. 2022; 36:3946320221125588. https://doi.org/10.1177/03946320221125588 [PubMed]



