Introduction
Pancreatic ductal adenocarcinoma (PADA) accounts for approximately ninety percent of pancreatic cancers, and represents a lethal cancer, with an estimated 232000 newly diagnosed cases, 22700 cancer-associated mortality annually, and a less than 5% five-year survival rate [1, 2]. Current standard treatment for PADA is a combination therapy using chemotherapy, molecular targeted therapy, immunotherapy and surgical operation. Unfortunately, despite advancement in therapeutic strategies, a median survival time for PADA patients is only 23 months. On the other hand, we should pay attention to this phenomenon that approximate 27% of patients with resected PDAD can survive for five years. One underlying rationale for this phenomenon is heterogeneity of tumor phenotypes of pancreatic cancer itself. Another potential explanation might be that some patients' response to some treatment regiment, while the others fail to response to the therapy strategies. Therefore, it seems essential to distinguish between patients who would most benefit from the current therapy and those who would be not fit for the treatment [3, 4]. To this end, the present study aimed to define an indicator of distinct survival outcomes.
The circadian clock endows humans the ability to perceive temporal changes of the external environment through a series of physiological activities [5]. The circadian rhythm is a sophisticated biological process, and supervises a series of cyclic biological functions via regulating a diversity of molecular activity and signal transduction processes [6]. In the case that circadian clock is disrupted, the hazard of conditions such as metabolic disorders, cardiovascular disease and cancer will increase [1]. Literatures have revealed a link between circadian rhythm disturbance and human cancer [7]. Although the negative effect of circadian clock disruption has gradually begun to be known [8–10], it remains underappreciated to exploit it for making more effective chronotherapy strategies. Besides, the role of circadian clock in PADA is elusive; therefore, we sought to interrogate the specific association between circadian clock and the pathogenesis of PADA.
In addition, PADA is an exceptionally heterogeneous carcinoma [11], with different phenotype in distinct cases [4, 12]. In despite of immune therapy has been applied for the treatment for cancers [13, 14], the detailed rationale of the role of immunotherapy in PADA is largely to be explored. Emerging data revealed that it is the crosstalk between cancer cells and intro-tumor immune infiltrates that play a pivotal role in the tumorigenesis and progression of cancer [15, 16]. Cytotoxic immune cells such as CD8+ T cell, as well as immune transduction processes are impaired in the intra-tumor microenvironment [7]. Nevertheless, the precise and exact processes of them are still required to be interrogated.
Facing the challenges in the process of treating PADA that how we could precisely distinguish between the low- and high- risk cohorts and how the immune infiltrates crosstalk to cancer cells, we aim to develop an indicator of distinct prognostic outcomes, and investigate its association with immune function and therapeutic efficacy, hoping to incorporate the pharmacological modulation of circadian clock into future therapeutic strategies.
Materials and Methods
Acquisition of data
Gene expression information of PADA patients was acquired from the Cancer Genome Atlas (TCGA) database and Gene Expression Omnibus (GEO), including GSE62452 [17] and GSE28735 [18]. TCGA contains 172 PADA patients with a follow-up time of more than one month, among including of 171 primary tumors and one metastatic tumor. These samples were divided into the discovery set (n = 120) and validation set (n = 52). GSE62452 contains 69 PADA and adjacent non-tumor samples, among which 65 PADA samples had a follow-up time of more than one month. These samples were snap-frozen and sequenced using Affymetrix Human Gene 1.0 ST Array (GPL6244). GSE28735 contains 45 PAD patients, among which 42 PADA patients had a follow-up time of more than one month. These samples were also snap-frozen and sequenced using Affymetrix Human Gene 1.0 ST Array (GPL6244). Gene expression information was got through Cancer Cell Line Encyclopedia (CCLE) database. IC50 values of pancreatic cancer cell lines was got from Genomics of Drug Sensitivity in Cancer (GDSC). In the present study, gene expression profiling from TCGA was subjected to TPM normalization, and all arrays were subjected to RMA normalized in GSE62452 and GSE28735. A total of 104 circadian clock-associated genes that were retrieved in MSigDB database [19]. Basic information for gene expression data was shown in the Table 1.
Table 1. Basic information for gene expression data.
| Database | Normalization method | Samples with OS | Survival time type | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Training set | TPM | 120 | OS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Validation set | TPM | 52 | OS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GSE62452 | RMA | 65 | OS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GSE28735 | RMA | 42 | OS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TPM, transcripts per million; RMA, robust multi-array analysis; OS, overall survival. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Differential expression analysis
To obtain the possible central genes, we executed differential expression analysis with the package edgeR in R. Raw counts were initially utilized to filter out those tremendously low-expressed genes, and then subjected to TMM normalization. Afterwards, processed data were characterized using negative binomial distribution and resulting differential expression calculation. Thresholds used here were P < 0.05 and |logFC| > 1. P < 0.05 is generally used as the significant relevance in the statistics, and |logFC| > 1 is regarded as an apparent fold change between the tumor and control groups in the bioinformatics.
Characterization of intro-tumor immune cell types
Intra-tumor cell types were quantified by six different computational algorithms XCELL, TIMER, QUANTISEQ, MCPCOUNTER, EPIC, and CIBERSORT [20, 21]. The robustness of these approaches has been checked by laboratory assays. These estimation algorithms are basically based on two concepts, i.e., deconvolution and non-overlapping sets of gene lists which can represent for different immune infiltrates. Parameters used here were all in default Data used were TPM values.
Quantitative analysis of circadian clock
Quantification of circadian clock was analyzed via single-sample gene set enrichment analysis [22]. Single-sample gene set enrichment analysis represents a computational strategy that can quantify a specific gene set for individual samples. Here, we used this mathematic strategy to estimate the levels of circadian clock for the comparison between the cancer group and the control group.
Interrogation of physiological and pathological processes involved in interested genes
Physiological and pathological processes that were associated with interested gene lists were interrogated with R package “clusterProfiler” [23]. Physiological and pathological processes were mainly retrieved from two canonical databases: the gene ontology and Kyoto Encyclopedia of Genes and Genomes pathways.
Statistics
Statistics were completed with R language. t test was used for continuous data. Pearson’s correlation analysis was utilized to analyze the association between two continuous variables. P value less than 0.05 was considered statistically relevant.
Data availability
All data in this article are available from the public databases, and are freely available to any scientist wishing to use them for noncommercial purposes, without breaching participant confidentiality. Further information is available from the corresponding author on reasonable request.
Results
Signaling transduction processes associated with the predictive signature
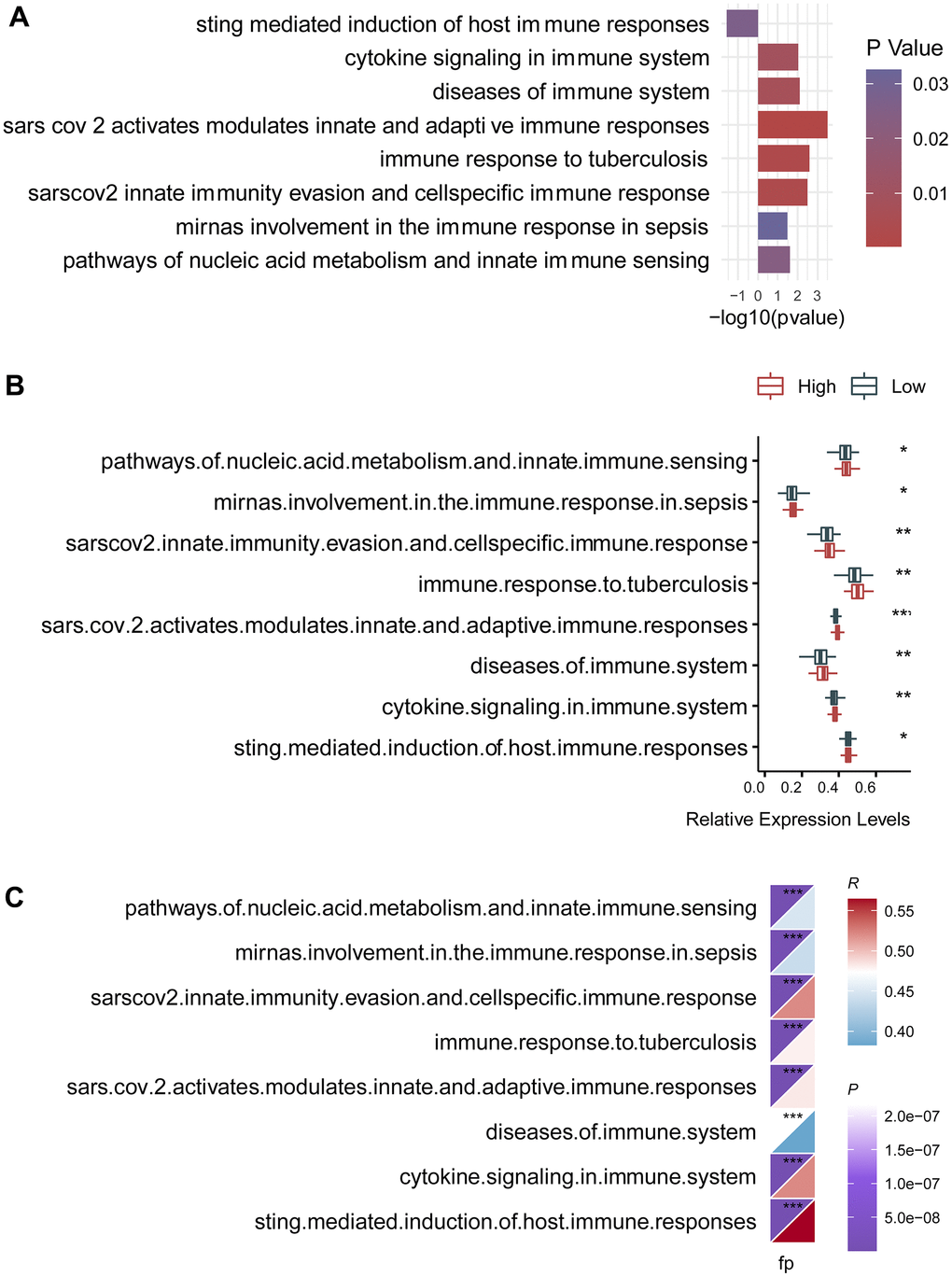
To explore physiological and pathological processes that were associated with the predictive signature, we executed functional enrichment analysis for genes that were mostly associated with the predictive signature. Top ten gene ontology terms were mainly mitotic nuclear division, would healing, nuclear division, and organelle fission (Figure 3A). Top ten signaling transduction processes were mainly focal adhesion, ECM-receptor interaction, cell cycle, proteoglycans in cancer, hippo signaling pathway, p53 signaling pathway, and apoptosis (Figure 3B). Subsequently, crosstalk communication between these physiological and pathological processes were interrogated, and the results showed that chromosome segregation was associated with organelle fission, mitotic sister chromatid segregation and extracellular structure organization (Figure 3C). Besides, these physiological and pathological processes can be clustered into three clusters (Figure 3D). Multiple common genes were observed in these biological processes and signaling pathways (Figure 3E, 3F), furtherly implying crosstalk networks among these pathways.
Association of the gene signature with response to molecular targeted therapy in pancreatic ductal adenocarcinoma
As the circadian clock-related prognostic signature showed its performance in predicting survival and exhibited an effect on immune response, we naturally proposed a hypothesis whether the gene signature could reflect the response to molecular targeted therapy in cancer. To clarify this problem, we investigated drug response of pancreatic cancer cells to a variety of agents using data from GDSC and CCLE database. As expected, the circadian clock-related signature manifested an association with drug response. IC50 of BMS-536924, Erlotinib, Foretinib, Linsitinib, and Sabutoclax exhibited a distinct effect on the low- and the high-risk cohorts (Figure 6A). Quantitative analysis showed that BMS-536924, Foretinib, Linsitinib, and Sabutoclax were more sensitive in the low-risk cancer cells, whereas Erlotinib was more effective in the high-risk cancer cells (Figure 6B). The specific information on these agents was described in Table 2.
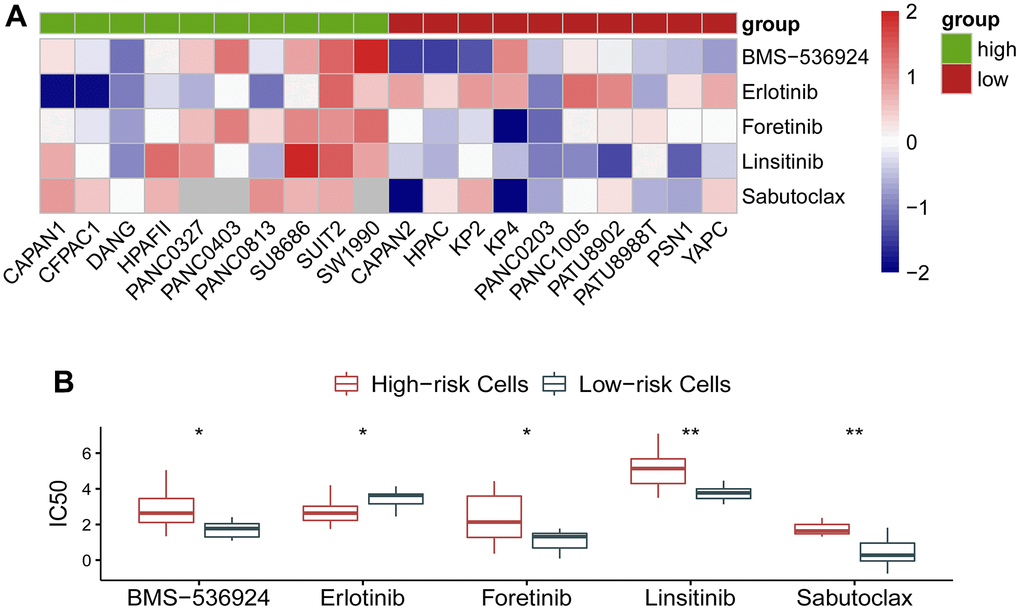
Table 2. Basic information on agents.
| Drug | Targeted molecules | Targeted pathways |
| BMS-536924 | IGF1R, IR | IGF1R signaling |
| Erlotinib | EGFR | EGFR signaling |
| Foretinib | MET, KDR, TIE2, VEGFR3/FLT4, PDGFR, EGFR | RTK signaling |
| Linsitinib | IGF1R | IGF1R signaling |
| Sabutoclax | BCL2, BCL-XL, BFL1, MCL1 | Apoptosis regulation |
Discussion
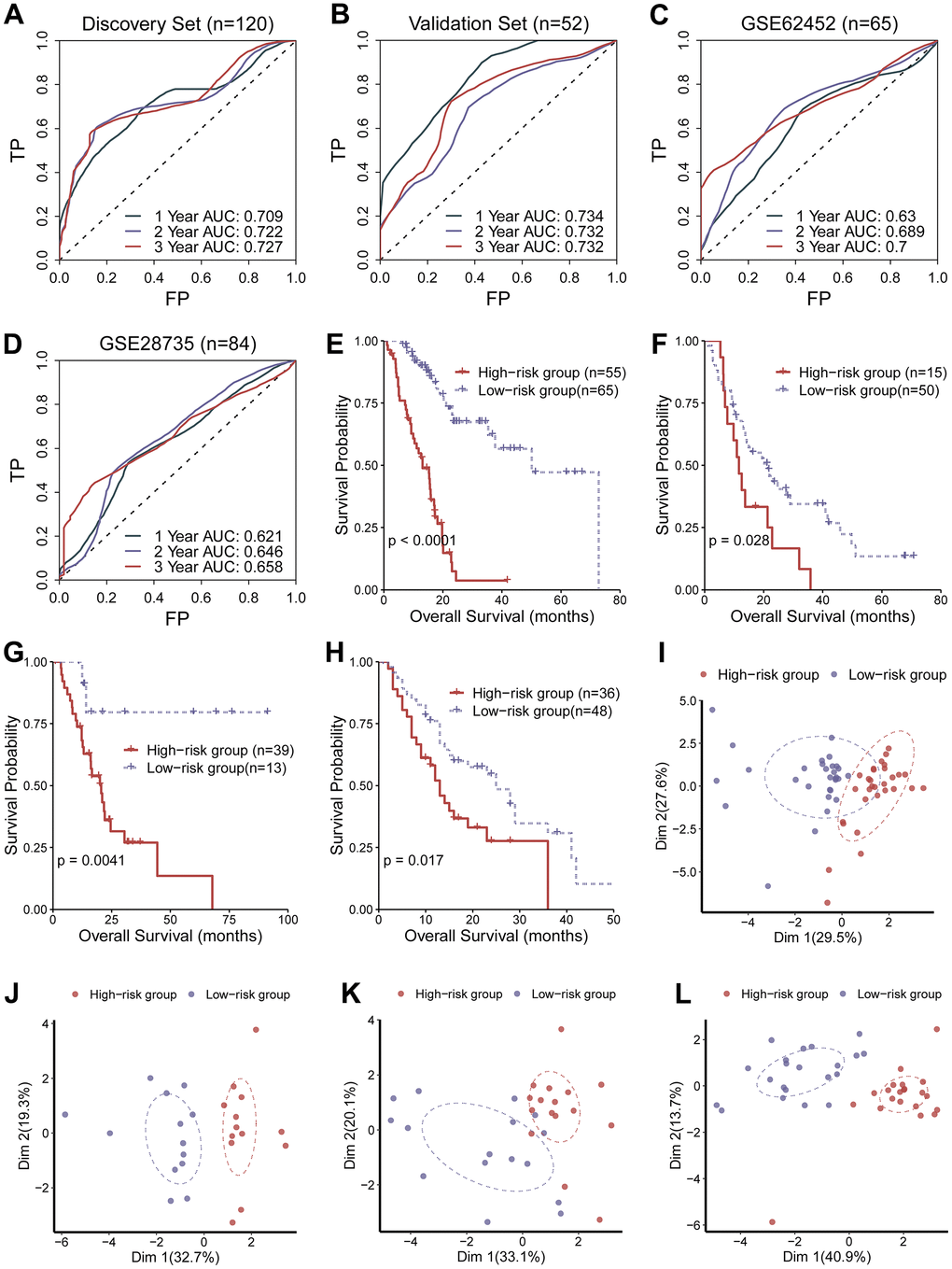
We developed a circadian clock-related prognostic signature that could distinguish pancreatic ductal adenocarcinoma (PADA) patients with unwanted prognostic outcomes from those with desirable prognostic outcomes and be reflective of response to molecular targeted therapy. Remarkably, the ability of the current signature to predict survival outcomes was independently verified in two external data sets, stratifying PADA patients into the low- and the high-risk groups, outperforming current pathological staging system, implying that this circadian clock-related gene signature could serve as a prognostic tool for PADA patients.
Pancreatic cancer represents a fatal condition with a poor prognosis. Surgical removal is still the standard treatment for patients with a resectable lesion [24]. Nevertheless, the option for operation must be chosen cautiously due to a relatively low postoperative mortality and a relatively high incidence of postoperative complications [25–27]. Therefore, it is necessary to identify PADA patients who are suitable for surgery or not. Here, we established this circadian clock-associated signature could be exploited as such an indicator for risk prediction, which would hopefully aid in the formulation of treatment strategies.
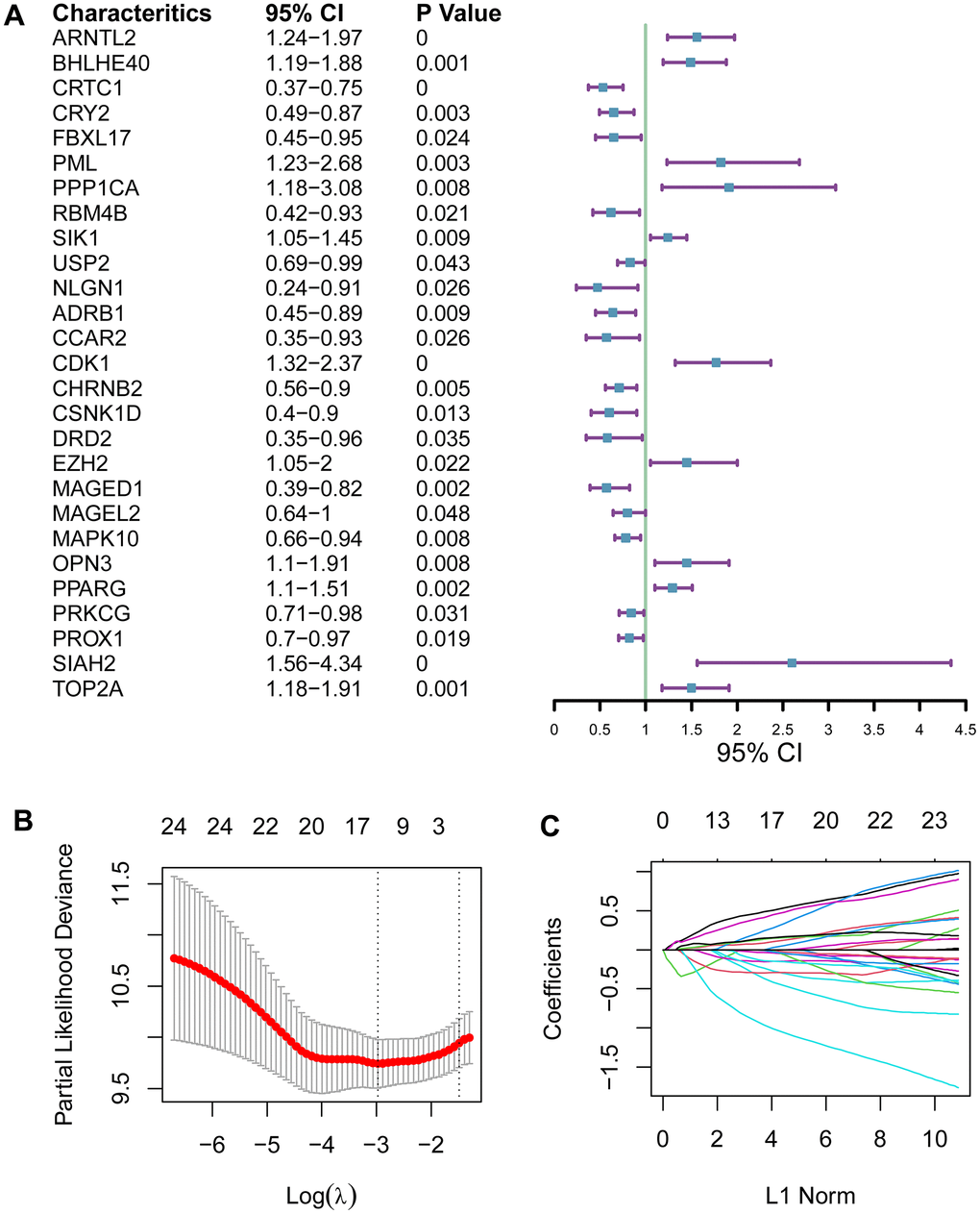
Of the 13 genes identified to constitute the prognostic gene signature, part of them do not have an obvious function in cancer. ARNTL2, BHLHE40 are reported to promote pancreatic ductal adenocarcinoma progression [28, 29]. Interestingly, BHLHE40 can drive pro-tumor neutrophils with hyperactivated glycolysis in pancreatic ductal adenocarcinoma. FBXL17, FBXL8, CDK1, CSNK1D, and PSPC1 have not been well studied in pancreatic ductal cancer. However, several of them have been implicated in pancreatic cancer, with a relatively high expression and a poor survival [30–33]. These suggest that the 13 genes in this prognostic signature are worth further investigation for their potential as novel therapeutic targets.
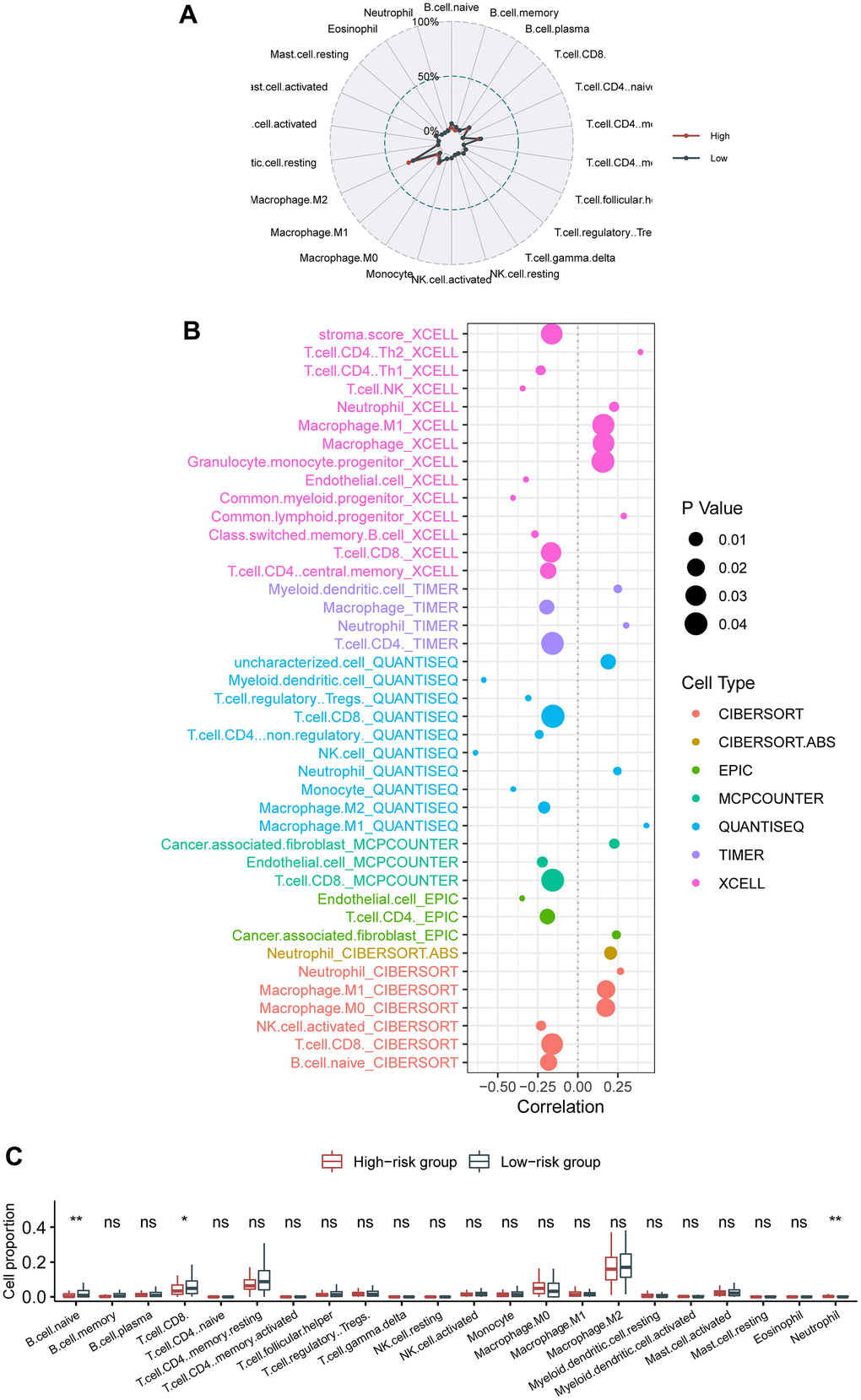
As above mentioned, neutrophils are associated with the proliferation and metastasis of cancer in pancreatic cancer [34, 35]. Consistently, we identified that neutrophils were also elevated in the high-risk group than in the low-risk group. Besides, neutrophil extracellular traps could mediate resistance to checkpoint blockade in pancreatic cancer [36], and targeting neutrophils could prevent pancreatic cancer metastasis [37] and improve effects of immune checkpoint blockade [38].
Besides, our analyses demonstrated that BMS-536924, Foretinib, Linsitinib, and Sabutoclax were more sensitive in the low-risk group, whereas Erlotinib was more effective in the high-risk group. BMS-536924 is reported to has anti-neoplastic activity and can sensitize cancer cells to molecular target drugs [39, 40]. However, its role in pancreatic cancer is required to be investigated. Likewise, Foretinib showed an anti-tumor function [41], while its relationship with drug sensitivity remains elusive. Combination therapy based on Sabutoclax and minocycline was more effective in vivo and vitro experiments [42].
We have to admitted that a prominent limitation in the present research is a lack of in vivo and in vitro assays to further support the findings of this study or interrogate the mechanisms underlying the roles of the signature and pivotal genes in pancreatic cancer. Further laboratory experiments are required to solve this limitation.
Collectively, we built a novel circadian clock-related signature, which can stratify patients of distinct prognostic outcomes and be reflective of therapeutic effects of molecular targeted therapy. These findings could incorporate the pharmacological modulation of circadian clock into future therapeutic strategies.
Supplementary Materials
Author Contributions
J. Y. and L. G. are responsible for designing the study and writing the manuscript, G. S., H. L. and S. G. are responsible for collecting the data and statistics.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This research received no financial support from any funding agency for the research, authorship, and/or publication of this article.
References
- 1. Liu J, Tan Z, Yang S, Song X, Li W. A circadian rhythm-related gene signature for predicting relapse risk and immunotherapeutic effect in prostate adenocarcinoma. Aging (Albany NY). 2022; 14:7170–85. https://doi.org/10.18632/aging.204288 [PubMed]
- 2. Stratford JK, Bentrem DJ, Anderson JM, Fan C, Volmar KA, Marron JS, Routh ED, Caskey LS, Samuel JC, Der CJ, Thorne LB, Calvo BF, Kim HJ, et al. A six-gene signature predicts survival of patients with localized pancreatic ductal adenocarcinoma. PLoS Med. 2010; 7:e1000307. https://doi.org/10.1371/journal.pmed.1000307 [PubMed]
- 3. Wu Z, Huang X, Cai M, Huang P, Guan Z. Novel necroptosis-related gene signature for predicting the prognosis of pancreatic adenocarcinoma. Aging (Albany NY). 2022; 14:869–91. https://doi.org/10.18632/aging.203846 [PubMed]
- 4. Zhang D, Cui F, Peng L, Wang M, Yang X, Xia C, Li K, Yin H, Zhang Y, Yu Q, Jin Z, Huang H. Establishing and validating an ADCP-related prognostic signature in pancreatic ductal adenocarcinoma. Aging (Albany NY). 2022; 14:6299–315. https://doi.org/10.18632/aging.204221 [PubMed]
- 5. Ruan W, Yuan X, Eltzschig HK. Circadian rhythm as a therapeutic target. Nat Rev Drug Discov. 2021; 20:287–307. https://doi.org/10.1038/s41573-020-00109-w [PubMed]
- 6. Smit AE, Schilperoort M, Winter EM. Restoring rhythm to prevent age-related fractures. Aging (Albany NY). 2022; 14:5617–9. https://doi.org/10.18632/aging.204192 [PubMed]
- 7. Oyama Y, Onishi H, Koga S, Murahashi M, Ichimiya S, Nakayama K, Fujimura A, Kawamoto M, Imaizumi A, Umebayashi M, Ohuchida K, Morisaki T, Nakamura M. Patched 1-interacting Peptide Represses Fibrosis in Pancreatic Cancer to Augment the Effectiveness of Immunotherapy. J Immunother. 2020; 43:121–33. https://doi.org/10.1097/CJI.0000000000000305 [PubMed]
- 8. Masri S, Sassone-Corsi P. The emerging link between cancer, metabolism, and circadian rhythms. Nat Med. 2018; 24:1795–803. https://doi.org/10.1038/s41591-018-0271-8 [PubMed]
- 9. Sancar A, Van Gelder RN. Clocks, cancer, and chronochemotherapy. Science. 2021; 371:eabb0738. https://doi.org/10.1126/science.abb0738 [PubMed]
- 10. Bishehsari F, Voigt RM, Keshavarzian A. Circadian rhythms and the gut microbiota: from the metabolic syndrome to cancer. Nat Rev Endocrinol. 2020; 16:731–9. https://doi.org/10.1038/s41574-020-00427-4 [PubMed]
- 11. Sharma NS, Gupta VK, Garrido VT, Hadad R, Durden BC, Kesh K, Giri B, Ferrantella A, Dudeja V, Saluja A, Banerjee S. Targeting tumor-intrinsic hexosamine biosynthesis sensitizes pancreatic cancer to anti-PD1 therapy. J Clin Invest. 2020; 130:451–65. https://doi.org/10.1172/JCI127515 [PubMed]
- 12. Jia L, Zhang Y, Pu F, Yang C, Yang S, Yu J, Xu Z, Yang H, Zhou Y, Zhu S. Pseudogene AK4P1 promotes pancreatic ductal adenocarcinoma progression through relieving miR-375-mediated YAP1 degradation. Aging (Albany NY). 2022; 14:1983–2003. https://doi.org/10.18632/aging.203921 [PubMed]
- 13. Azad A, Yin Lim S, D’Costa Z, Jones K, Diana A, Sansom OJ, Kruger P, Liu S, McKenna WG, Dushek O, Muschel RJ, Fokas E. PD-L1 blockade enhances response of pancreatic ductal adenocarcinoma to radiotherapy. EMBO Mol Med. 2017; 9:167–80. https://doi.org/10.15252/emmm.201606674 [PubMed]
- 14. Hopkins AC, Yarchoan M, Durham JN, Yusko EC, Rytlewski JA, Robins HS, Laheru DA, Le DT, Lutz ER, Jaffee EM. T cell receptor repertoire features associated with survival in immunotherapy-treated pancreatic ductal adenocarcinoma. JCI Insight. 2018; 3:e122092. https://doi.org/10.1172/jci.insight.122092 [PubMed]
- 15. Padoan A, Plebani M, Basso D. Inflammation and Pancreatic Cancer: Focus on Metabolism, Cytokines, and Immunity. Int J Mol Sci. 2019; 20:676. https://doi.org/10.3390/ijms20030676 [PubMed]
- 16. Lutz ER, Wu AA, Bigelow E, Sharma R, Mo G, Soares K, Solt S, Dorman A, Wamwea A, Yager A, Laheru D, Wolfgang CL, Wang J, et al. Immunotherapy converts nonimmunogenic pancreatic tumors into immunogenic foci of immune regulation. Cancer Immunol Res. 2014; 2:616–31. https://doi.org/10.1158/2326-6066.CIR-14-0027 [PubMed]
- 17. Yang S, He P, Wang J, Schetter A, Tang W, Funamizu N, Yanaga K, Uwagawa T, Satoskar AR, Gaedcke J, Bernhardt M, Ghadimi BM, Gaida MM, et al. A Novel MIF Signaling Pathway Drives the Malignant Character of Pancreatic Cancer by Targeting NR3C2. Cancer Res. 2016; 76:3838–50. https://doi.org/10.1158/0008-5472.CAN-15-2841 [PubMed]
- 18. Zhang G, He P, Tan H, Budhu A, Gaedcke J, Ghadimi BM, Ried T, Yfantis HG, Lee DH, Maitra A, Hanna N, Alexander HR, Hussain SP. Integration of metabolomics and transcriptomics revealed a fatty acid network exerting growth inhibitory effects in human pancreatic cancer. Clin Cancer Res. 2013; 19:4983–93. https://doi.org/10.1158/1078-0432.CCR-13-0209 [PubMed]
- 19. Liberzon A, Birger C, Thorvaldsdóttir H, Ghandi M, Mesirov JP, Tamayo P. The Molecular Signatures Database (MSigDB) hallmark gene set collection. Cell Syst. 2015; 1:417–25. https://doi.org/10.1016/j.cels.2015.12.004 [PubMed]
- 20. Newman AM, Liu CL, Green MR, Gentles AJ, Feng W, Xu Y, Hoang CD, Diehn M, Alizadeh AA. Robust enumeration of cell subsets from tissue expression profiles. Nat Methods. 2015; 12:453–7. https://doi.org/10.1038/nmeth.3337 [PubMed]
- 21. Li T, Fu J, Zeng Z, Cohen D, Li J, Chen Q, Li B, Liu XS. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020; 48:W509–14. https://doi.org/10.1093/nar/gkaa407 [PubMed]
- 22. Hänzelmann S, Castelo R, Guinney J. GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinformatics. 2013; 14:7. https://doi.org/10.1186/1471-2105-14-7 [PubMed]
- 23. Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012; 16:284–7. https://doi.org/10.1089/omi.2011.0118 [PubMed]
- 24. Yeo CJ, Cameron JL, Sohn TA, Lillemoe KD, Pitt HA, Talamini MA, Hruban RH, Ord SE, Sauter PK, Coleman J, Zahurak ML, Grochow LB, Abrams RA. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: pathology, complications, and outcomes. Ann Surg. 1997; 226:248–57. https://doi.org/10.1097/00000658-199709000-00004 [PubMed]
- 25. Eppsteiner RW, Csikesz NG, McPhee JT, Tseng JF, Shah SA. Surgeon volume impacts hospital mortality for pancreatic resection. Ann Surg. 2009; 249:635–40. https://doi.org/10.1097/SLA.0b013e31819ed958 [PubMed]
- 26. Yermilov I, Bentrem D, Sekeris E, Jain S, Maggard MA, Ko CY, Tomlinson JS. Readmissions following pancreaticoduodenectomy for pancreas cancer: a population-based appraisal. Ann Surg Oncol. 2009; 16:554–61. https://doi.org/10.1245/s10434-008-0178-6 [PubMed]
- 27. DeOliveira ML, Winter JM, Schafer M, Cunningham SC, Cameron JL, Yeo CJ, Clavien PA. Assessment of complications after pancreatic surgery: A novel grading system applied to 633 patients undergoing pancreaticoduodenectomy. Ann Surg. 2006; 244:931–7. https://doi.org/10.1097/01.sla.0000246856.03918.9a [PubMed]
- 28. Wang Z, Liu T, Xue W, Fang Y, Chen X, Xu L, Zhang L, Guan K, Pan J, Zheng L, Qin G, Wang T. ARNTL2 promotes pancreatic ductal adenocarcinoma progression through TGF/BETA pathway and is regulated by miR-26a-5p. Cell Death Dis. 2020; 11:692. https://doi.org/10.1038/s41419-020-02839-6 [PubMed]
- 29. Wang L, Liu Y, Dai Y, Tang X, Yin T, Wang C, Wang T, Dong L, Shi M, Qin J, Xue M, Cao Y, Liu J, et al. Single-cell RNA-seq analysis reveals BHLHE40-driven pro-tumour neutrophils with hyperactivated glycolysis in pancreatic tumour microenvironment. Gut. 2022. [Epub ahead of print]. https://doi.org/10.1136/gutjnl-2021-326070 [PubMed]
- 30. Lin J, Zhai S, Zou S, Xu Z, Zhang J, Jiang L, Deng X, Chen H, Peng C, Zhang J, Shen B. Positive feedback between lncRNA FLVCR1-AS1 and KLF10 may inhibit pancreatic cancer progression via the PTEN/AKT pathway. J Exp Clin Cancer Res. 2021; 40:316. https://doi.org/10.1186/s13046-021-02097-0 [PubMed]
- 31. Zheng H, Zheng WJ, Wang ZG, Tao YP, Huang ZP, Yang L, Ouyang L, Duan ZQ, Zhang YN, Chen BN, Xiang DM, Jin G, Fang L, et al. Decreased Expression of Programmed Death Ligand-L1 by Seven in Absentia Homolog 2 in Cholangiocarcinoma Enhances T-Cell-Mediated Antitumor Activity. Front Immunol. 2022; 13:845193. https://doi.org/10.3389/fimmu.2022.845193 [PubMed]
- 32. Tanaka T, Li TS, Urata Y, Goto S, Ono Y, Kawakatsu M, Matsushima H, Hirabaru M, Adachi T, Kitasato A, Takatsuki M, Kuroki T, Eguchi S. Increased expression of PHD3 represses the HIF-1 signaling pathway and contributes to poor neovascularization in pancreatic ductal adenocarcinoma. J Gastroenterol. 2015; 50:975–83. https://doi.org/10.1007/s00535-014-1030-3 [PubMed]
- 33. Tian X, Zheng J, Mou W, Lu G, Chen S, Du J, Zheng Y, Chen S, Shen B, Li J, Wang N. Development and validation of a hypoxia-stemness-based prognostic signature in pancreatic adenocarcinoma. Front Pharmacol. 2022; 13:939542. https://doi.org/10.3389/fphar.2022.939542 [PubMed]
- 34. Deng J, Kang Y, Cheng CC, Li X, Dai B, Katz MH, Men T, Kim MP, Koay EA, Huang H, Brekken RA, Fleming JB. DDR1-induced neutrophil extracellular traps drive pancreatic cancer metastasis. JCI Insight. 2021; 6:e146133. https://doi.org/10.1172/jci.insight.146133 [PubMed]
- 35. Schoeps B, Eckfeld C, Prokopchuk O, Böttcher J, Häußler D, Steiger K, Demir IE, Knolle P, Soehnlein O, Jenne DE, Hermann CD, Krüger A. TIMP1 Triggers Neutrophil Extracellular Trap Formation in Pancreatic Cancer. Cancer Res. 2021; 81:3568–79. https://doi.org/10.1158/0008-5472.CAN-20-4125 [PubMed]
- 36. Zhang Y, Chandra V, Riquelme Sanchez E, Dutta P, Quesada PR, Rakoski A, Zoltan M, Arora N, Baydogan S, Horne W, Burks J, Xu H, Hussain P, et al. Interleukin-17-induced neutrophil extracellular traps mediate resistance to checkpoint blockade in pancreatic cancer. J Exp Med. 2020; 217:e20190354. https://doi.org/10.1084/jem.20190354 [PubMed]
- 37. Kajioka H, Kagawa S, Ito A, Yoshimoto M, Sakamoto S, Kikuchi S, Kuroda S, Yoshida R, Umeda Y, Noma K, Tazawa H, Fujiwara T. Targeting neutrophil extracellular traps with thrombomodulin prevents pancreatic cancer metastasis. Cancer Lett. 2021; 497:1–13. https://doi.org/10.1016/j.canlet.2020.10.015 [PubMed]
- 38. Nielsen SR, Strøbech JE, Horton ER, Jackstadt R, Laitala A, Bravo MC, Maltese G, Jensen AR, Reuten R, Rafaeva M, Karim SA, Hwang CI, Arnes L, et al. Suppression of tumor-associated neutrophils by lorlatinib attenuates pancreatic cancer growth and improves treatment with immune checkpoint blockade. Nat Commun. 2021; 12:3414. https://doi.org/10.1038/s41467-021-23731-7 [PubMed]
- 39. Beauchamp MC, Knafo A, Yasmeen A, Carboni JM, Gottardis MM, Pollak MN, Gotlieb WH. BMS-536924 sensitizes human epithelial ovarian cancer cells to the PARP inhibitor, 3-aminobenzamide. Gynecol Oncol. 2009; 115:193–8. https://doi.org/10.1016/j.ygyno.2009.07.009 [PubMed]
- 40. Huang F, Greer A, Hurlburt W, Han X, Hafezi R, Wittenberg GM, Reeves K, Chen J, Robinson D, Li A, Lee FY, Gottardis MM, Clark E, et al. The mechanisms of differential sensitivity to an insulin-like growth factor-1 receptor inhibitor (BMS-536924) and rationale for combining with EGFR/HER2 inhibitors. Cancer Res. 2009; 69:161–70. https://doi.org/10.1158/0008-5472.CAN-08-0835 [PubMed]
- 41. Gortany NK, Panahi G, Ghafari H, Shekari M, Ghazi-Khansari M. Foretinib induces G2/M cell cycle arrest, apoptosis, and invasion in human glioblastoma cells through c-MET inhibition. Cancer Chemother Pharmacol. 2021; 87:827–42. https://doi.org/10.1007/s00280-021-04242-0 [PubMed]
- 42. Quinn BA, Dash R, Sarkar S, Azab B, Bhoopathi P, Das SK, Emdad L, Wei J, Pellecchia M, Sarkar D, Fisher PB. Pancreatic Cancer Combination Therapy Using a BH3 Mimetic and a Synthetic Tetracycline. Cancer Res. 2015; 75:2305–15. https://doi.org/10.1158/0008-5472.CAN-14-3013 [PubMed]