Introduction
Breast cancer is the top malignant tumor that threatens the health of women worldwide with incidence rate of 47.8% and mortality rate of 13.6% [1]. In the past two decades, people have gradually realized that breast cancer is heterogeneous, varying in pathology, genetics, and molecular biology [2, 3]. Breast cancer consists of luminal A, luminal B, human epidermal growth factor receptor 2 (HER2)-enriched, and basal-like molecular subtypes relevant to the clinical practice [4]. Under the guidance of molecular typing, the diagnosis and treatment of breast cancer has gradually entered the era of individualized diagnosis and treatment [5]. The 5-year survival rate and the quality of life of breast cancer patients have been improved [6]. However, some patients will progress to an advanced stage, and advanced breast cancer is still an incurable disease with a median survival of about 24 to 30 months [7, 8].
Early diagnosis has become the key to save the lives of breast cancer patients. Currently, tissue- and serum-based biomarkers are widely used for early-stage tumor screening and to predict disease progression or recurrence [9]. Commonly used biomarkers for breast cancer include estrogen receptor (ER), progesterone receptor (PR), and HER2 [10]. Since the occurrence and progression are heterogeneous, researchers strive to identify novel biomarkers for optimal breast cancer control [11].
Bioinformatic analysis has contributed much to the finding of novel biomarkers, especially in cancer [12]. Kim et al. have suggested SURF4 as an oncogene in cancer [13]. SURF4 is the human homologous gene of the yeast cargo receptor Erv29p with a molecular size of 30 kD [14, 15]. SURF4 contains 7 transmembrane domains and a double-lysine endoplasmic reticulum carboxyl-terminal sequence, which can interact with the ER Golgi Intermediate compartment-53 and p24 protein [16]. However, the role of SURF4 in breast cancer has not been demonstrated yet.
In this study, high expression of SURF4 was first found in breast cancer. Their relationship was further evaluated using bioinformatics analysis. The diagnostic value and independent predictive value in overall survival (OS) and relapse-free survival (RFS) were evaluated. Besides, the nomogram and Gene set enrichment analysis (GSEA) were conducted. The cell and tissue experiments were carried out for final validation.
Results
Patient characteristics
In total, 1104 patients were analyzed, 1090 of which (98.73%) were females. As shown in Supplementary Table 1, 589 (53.45%) patients were less than 60 years old, and 790 (71.56%) were diagnosed of infiltrating ductal carcinoma. As for molecular subtype, 142 (12.86%) patients were basal-like type, 67 (6.07%) patients were HER2 overexpression type, 422 (38.22%) patients were luminal A type, 194 (17.57%) patients were luminal B type, and 24 (2.17%) patients were normal-like type. Most patients (56.7%) were in stage II.
High expression of SURF4 in tumor
Compared with normal tissue, SURF4 is significantly (P < 0.001) highly expressed in tumor (Figure 1A). Besides, SURF4 is significantly (P < 0.001) highly expressed in breast cancer in comparison with paired normal breast tissue (Figure 1B).
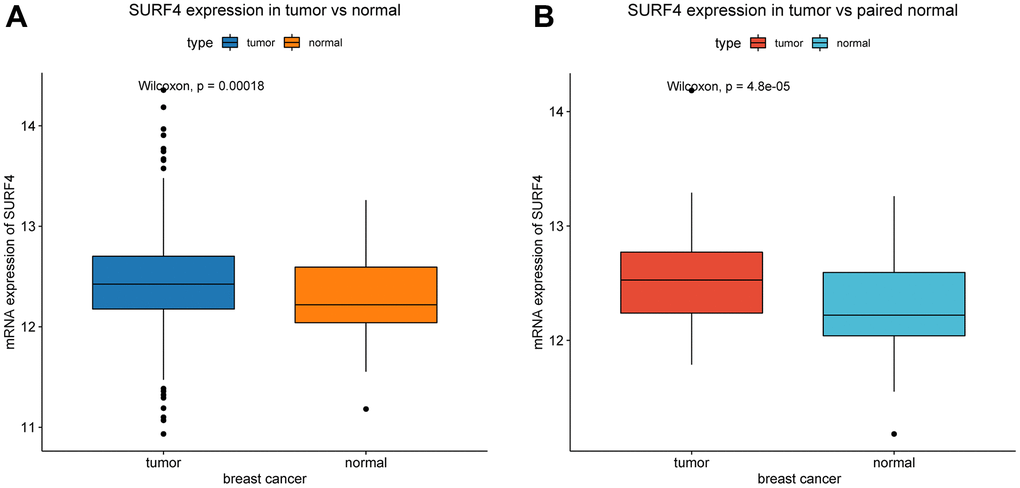
Figure 1. Expression of SURF4 in breast cancer. (A) The expression of SURF4 in tumor and normal tissues. (B) The expression of SURF4 in tumor and paired normal tissues.
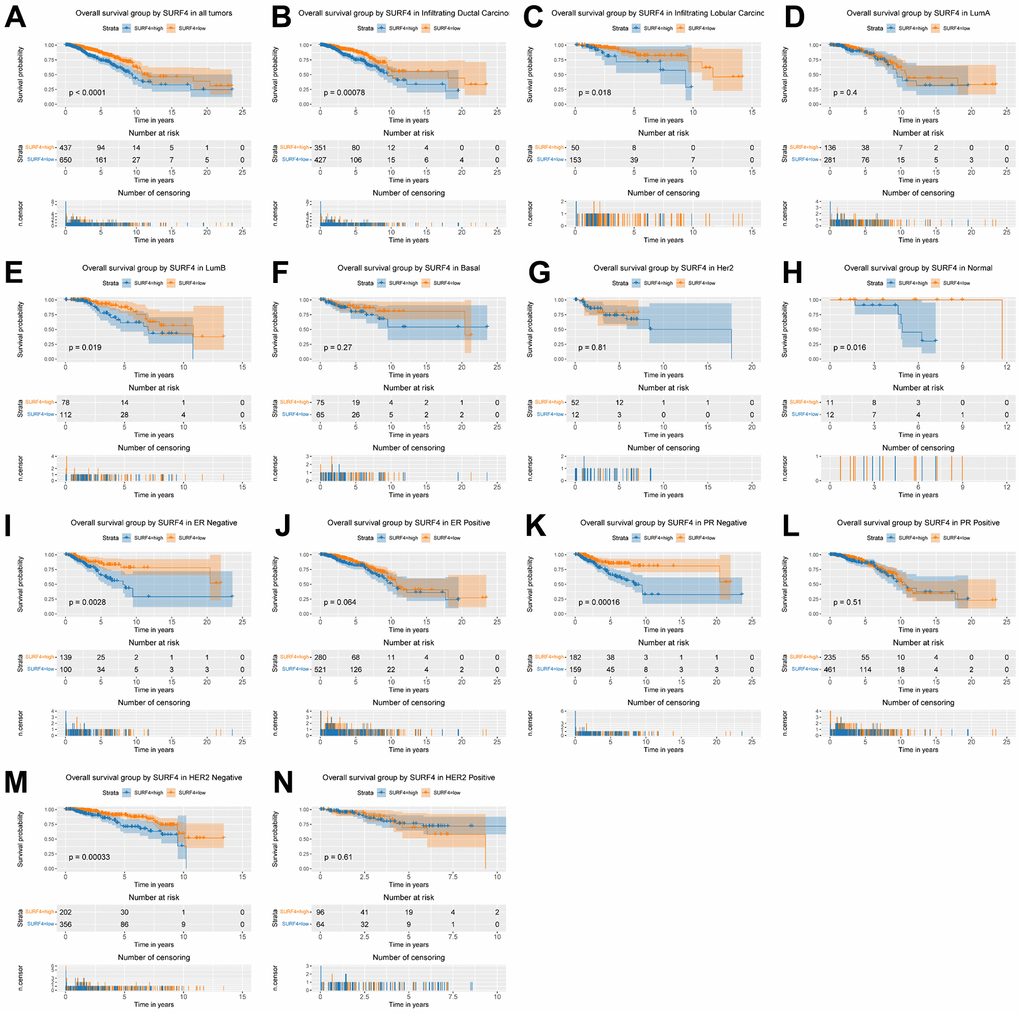
Furthermore, the expression of SURF4 grouped by distinctive characteristics were evaluated. As shown in Figure 2A–2H, the expression of SURF4 was highest in T4 (P = 0.014), infiltrating ductal carcinoma (P < 0.001), ER negative (P < 0.001), PR negative (P < 0.001), and HER2 positive (P < 0.001) patients. Besides, as shown in Figure 3A–3K, high expression of SURF4 was observed in female (P = 0.026), patients without lymph node metastasis (P = 0.037), HER2 overexpression type (P < 0.001), and deceased patients (P = 0.016). Other characteristics showed no significantly statistical differences.
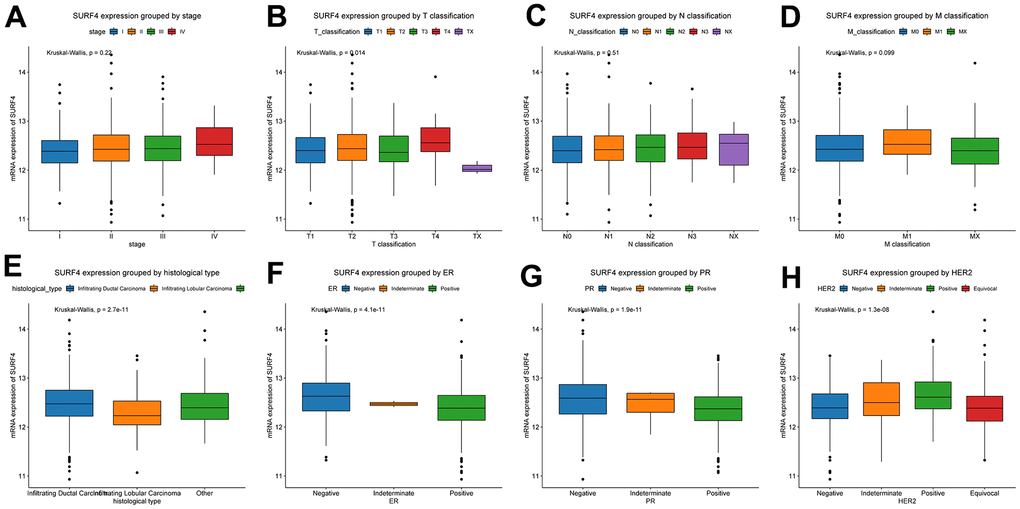
Figure 2. Expression of SURF4 in breast cancer and its relationship with clinicopathological parameters. Expression of SURF4 grouped by (A) stage, (B) T classification, (C) N classification, (D) M classification, (E) histological type, (F) ER, (G) PR, and (H) HER2.
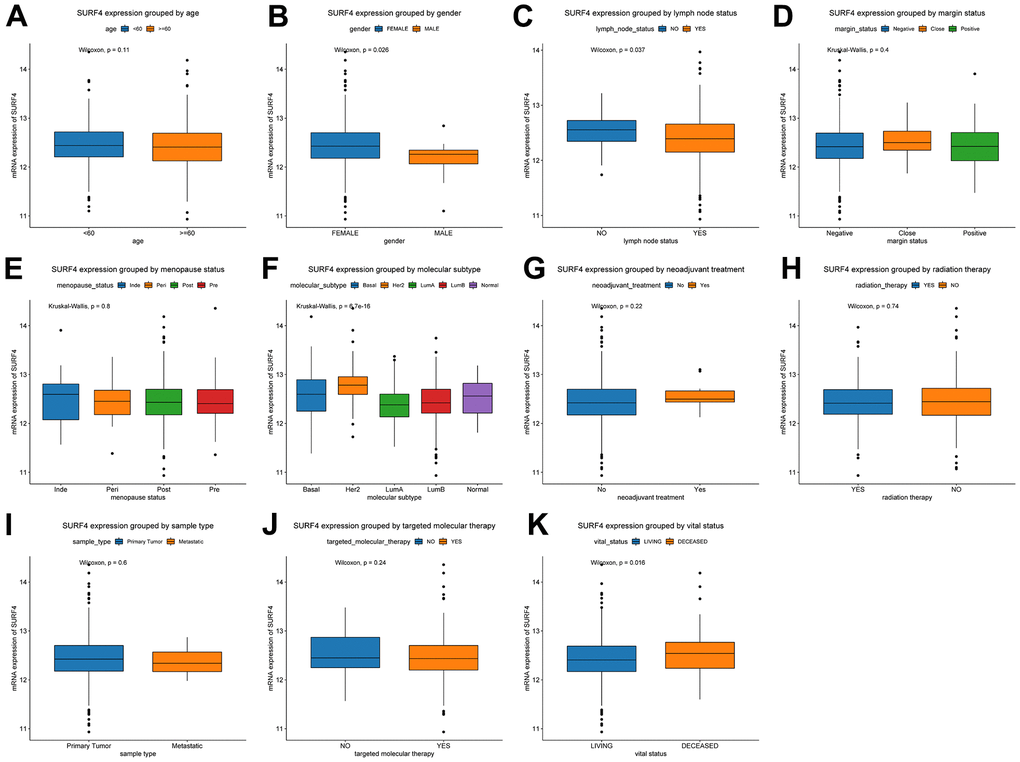
Figure 3. Expression of SURF4 in breast cancer and its relationship with clinicopathological parameters. Expression of SURF4 grouped by (A) age, (B) gender, (C) lymph node status, (D) margin status, (E) menopause status, (F) molecular subtype, (G) neoadjuvant treatment, (H) radiation therapy, (I) sample type, (J) targeted molecular therapy, and (K) vital status.
Diagnostic value of SURF4 expression for breast cancer
The receiver operating characteristic (ROC) curves were plotted to evaluate the diagnostic value of SURF4 expression for breast cancer. As shown in Supplementary Figure 1A, the cut-off value between normal and tumor was 12.241, and the AUC was 0.606. Besides, the AUC for stage I-IV (Supplementary Figure 1B–1E) was 0.581, 0.609, 0.613, and 0.693, respectively.
Predictive value of SURF4 for survival
The nomograms of SURF4 were generated to predicate survival of breast cancer patients. Patients with high SURF4 expression showed shorter OS (Figure 8). Besides, the patients with higher age, equivocal HER2, higher stages, or positive margin status also had shorter OS.
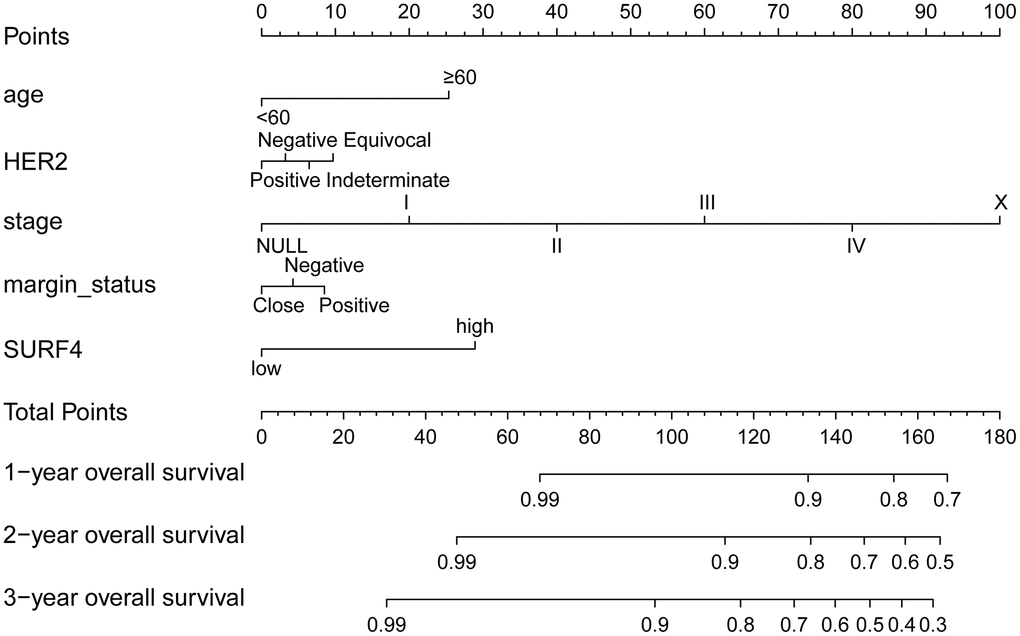
Figure 8. The nomogram about SURF4 and OS. 1-, 3- and 5-year related survival probabilities were obtained by drawing a line straight down to the risk axis.
Expression of SURF4 in TIMER database
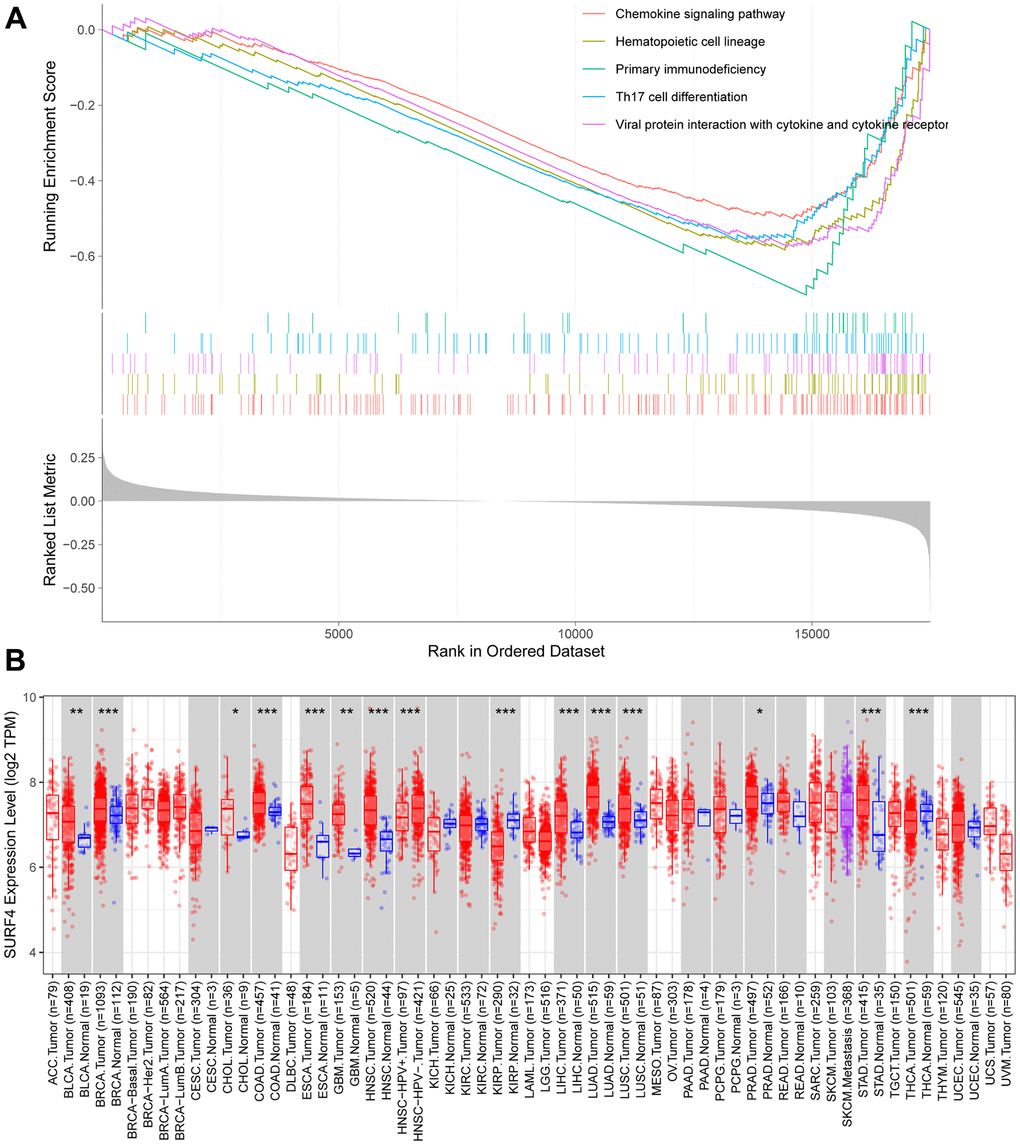
The expressions of SURF4 in different cancers were further studied using TIMER database (Figure 9B). Significant high SURF4 expression was found in bladder urothelial carcinoma, etc.
High SURF4 expression in breast cancer tissue and cell
As shown in Figure 10A, SURF4 expression was significantly higher in tumor than adjacent normal tissue (P < 0.001). As shown in Figure 10B, significant high SURF4 expression (P < 0.01) was observed in MCF7 (Human luminal A type breast cancer cell), BT474 (Human luminal B type breast cancer cell), SKBR3 (Human HER2 overexpression type breast cancer cell), MDAMB231 and 4T1 (Human triple-negative breast cancer cell). Meanwhile, HaCaT (Human immortalized epidermal cell) and MCF10A (Human normal breast cell) showed much lower SURF4 expression. Importantly, 4T1 cell line showed the highest SURF4 expression and used subsequently for in vitro experiment. The high SURF4 expression in breast cancer tissue was further validated by immunohistochemistry (IHC) (Figure 10C).
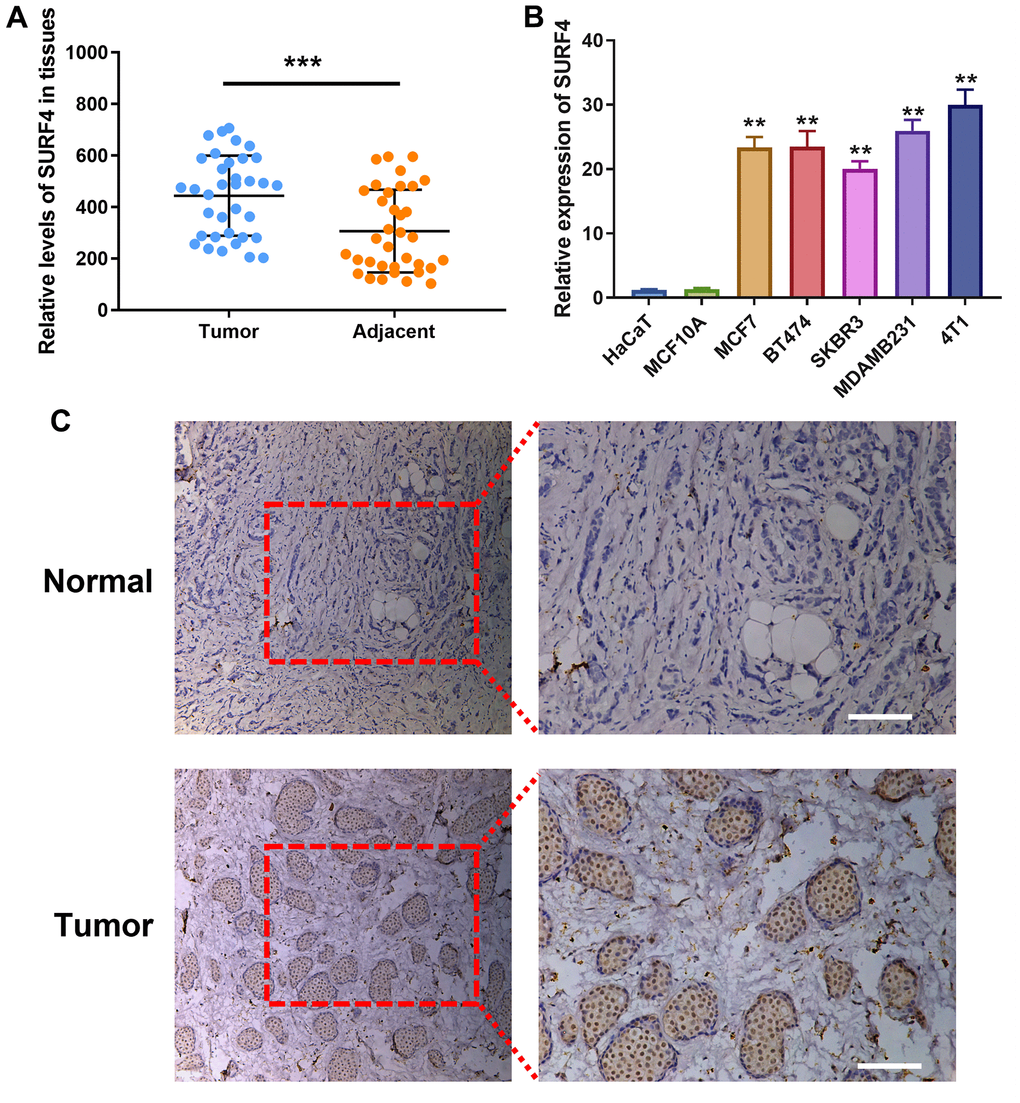
Figure 10. Expression of SURF4 in human breast tissues and cell lines. (A) SURF4 expression in tumor and adjacent normal tissue by qRT-PCR (N = 35). (B) SURF4 expression in HaCaT (Human immortalized epidermal cell), MCF10A (Human normal breast cell), MCF7 (Human luminal A type breast cancer cell), BT474 (Human luminal B type breast cancer cell), SKBR3 (Human HER2 overexpression type breast cancer cell), MDAMB231 and 4T1 (Human triple-negative breast cancer cell) by qRT-PCR in triplicate. (C) SURF4 expression in normal and tumor tissue by IHC. The tissues were from single patient. Scale bar = 100 μm. **P < 0.01; ***P < 0.001.
SURF4 promoted the proliferation and migration of 4T1 cells
The successful over-expression (O-SURF4) and knockdown (si-SURF4) of SURF4 was shown in Figure 11A. As shown in Figure 11B, si-SURF4 group showed decreased cell proliferation (P < 0.01), while O-SURF4 group showed increased cell proliferation (P < 0.01). The results of colony formation (Figure 11C) and living/dead cell staining (Figure 11D, 11E) were consistent. Si-SURF4 group showed fewer colonies (P < 0.05) and more dead cells (P < 0.01), while O-SURF4 group showed more colonies (P < 0.05) and fewer dead cells (P < 0.05). Finally, the migration distance was shorter in si-SURF4 group (P < 0.05) and longer in O-SURF4 group (Figure 11F, 11G, P < 0.01). The results indicated that SURF4 promoted the proliferation and migration of 4T1 cells.
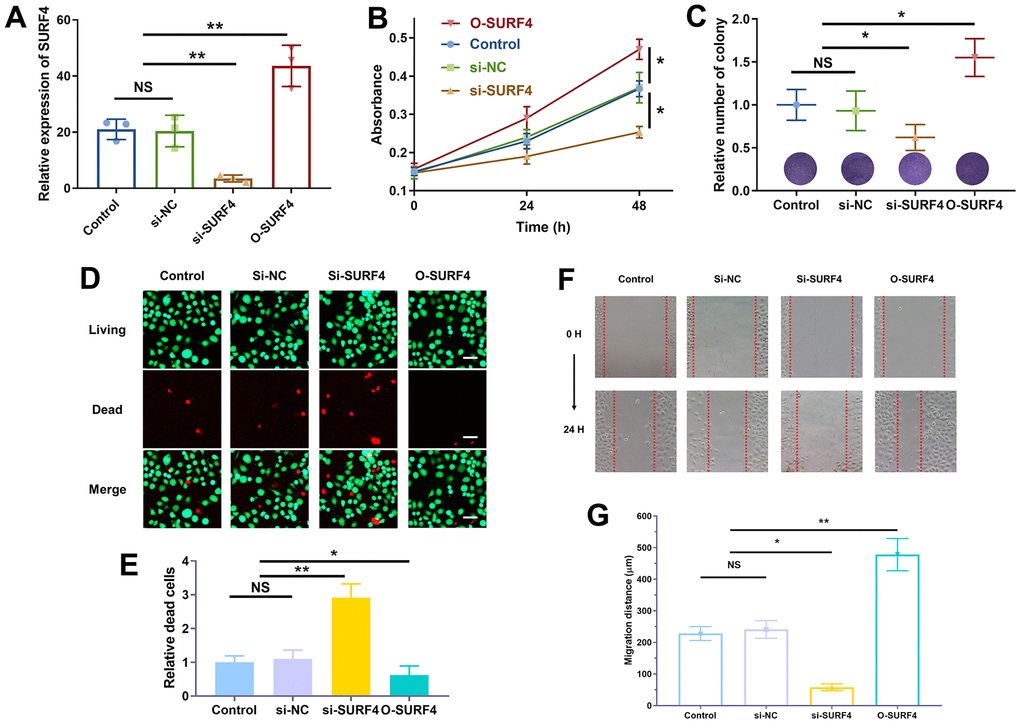
Figure 11. SURF4 promoted cell proliferation and migration of breast cancer cell 4T1. (A) Efficiency of plasmid transfection by qRT-PCR. (B) The cell viability by CCK8 assay. (C) Cell proliferation ability by colony formation assay. (D, E) Cell proliferation ability by living/dead staining assay. Scale bar = 50 μm. (F, G) Cell migration ability by wound healing assay. The experiments were repeated for 3 times. 40 x under light microscopy. NS, no significance; *P < 0.05; **P < 0.01.
Discussion
In 2020, the incidence of breast cancer reached 11.7%, surpassing lung cancer for the first time to become the world’s most commonly diagnosed cancer. Continuous exploration of earlier diagnosis methods has become the most urgent task [17]. Breast cancer is a very heterogeneous disease [18]. It has become widely accepted that breast cancer can be classified according to molecular markers. In 2001, Peru et al. applied complementary DNA microarray technology to detect postoperative specimens, and divided breast cancer into different subtypes with different clinical prognosis, namely luminal A, luminal B, basal-like, HER2 overexpression, and normal-like type [19].
Diverse types of breast cancer have different biological characteristics and treatment methods. Hormone receptor-positive luminal A and luminal B have the highest proportions in breast cancer, and can be treated with endoprostheses with relatively good prognosis [20]. The 5-year selective ER modulator tamoxifen therapy can reduce the recurrence rate by about 50% and the mortality by about 30% [21]. Compared with luminal A type, luminal B type has the following characteristics: lower expression levels of estrogen or estrogen-related genes, low or no expression of PR, higher grade of tumor and higher expression levels of proliferation-related genes, growth factor receptor pathways such as IGF-1R and PI3K/AKT/mTOR are easily activated, etc. [22]. HER2 overexpression type has poor biological behavior and high recurrence rate, but its treatment has made significant progress with the application of targeted drugs such as trastuzumab, pertuzumab, and lapatinib, etc. [5, 23]. The basal-like type lacking hormone receptors and overexpressing HER2 is often considered triple-negative breast cancer, which can only be treated with chemotherapy, and is more common in patients with BRCA1 mutations or African ancestry [24, 25].
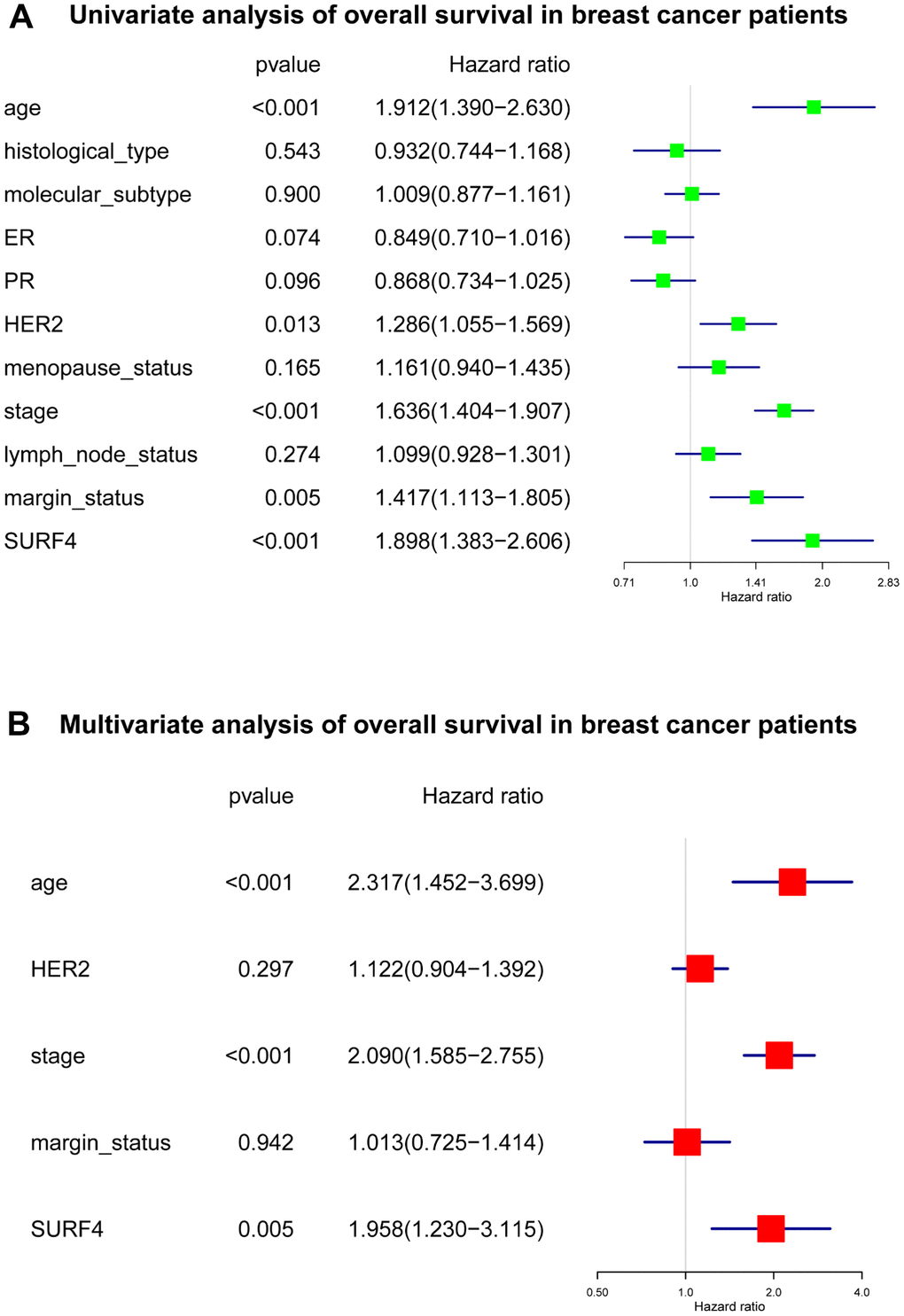
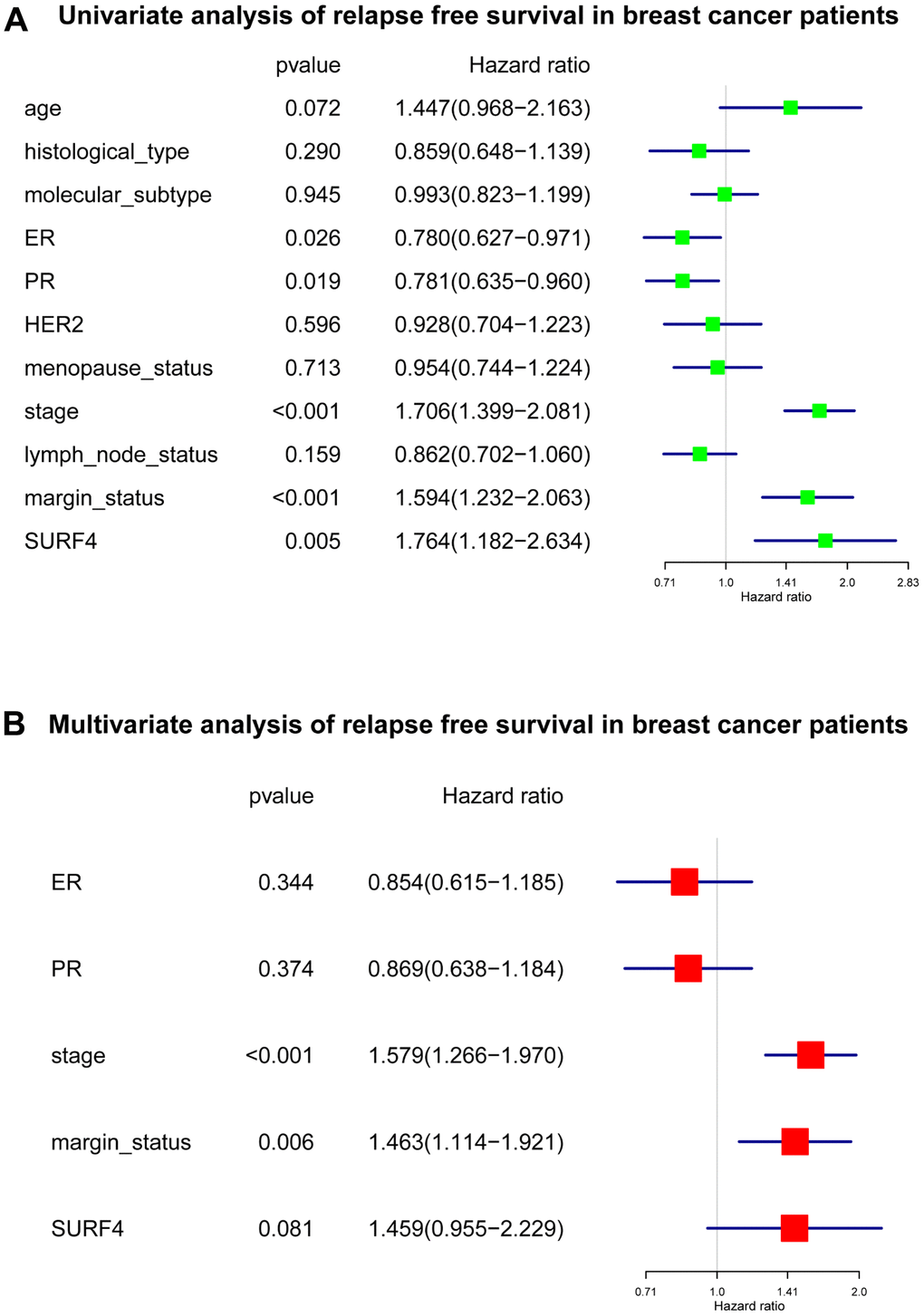
Our results indicated that SURF4 was significantly (P < 0.001) highly expressed in tumor. High expression of SURF4 was observed in T4, infiltrating ductal carcinoma, ER negative, PR negative, HER2 positive, female, patients without lymph node metastasis, HER2 overexpression type, and deceased patients. The diagnostic ability increased with stage increased as the AUC for stage I-IV was 0.581, 0.609, 0.613, and 0.693, respectively. The age, stage, and SURF4 expression exhibited independent prognostic value for OS of breast cancer. Patients with high SURF4 expression, higher age, equivocal HER2, higher stages, or positive margin status had shorter OS. The stage and margin status exhibited independent prognostic value for RFS of breast cancer.
As is known, Ki-67 is a nuclear antigen associated with proliferating cells, and currently a widely used tumor cell proliferation activity marker [26]. Ki-67 expression level is significantly higher in malignant tumors [27]. Ki-67 has been used as one of the proliferation markers in early breast cancer [28]. Relevant studies have shown that changes in the expression level of Ki-67 can be used as a sensitivity indicator to predict the efficacy of neoadjuvant endocrine therapy [29]. Neoadjuvant endocrine therapy trials IMPACT and Z1031 showed a survival benefit after inhibition of Ki-67 expression [30, 31]. Neoadjuvant endocrine therapy has been used as the primary endpoint, but its value in predicting the efficacy of neoadjuvant chemotherapy remains controversial [32].
Limited studies have showed the role of SURF4 in cancer. Yue et al. found SURF4 possessed the ability for maintaining stemness of ovarian cancer, and may serve as a potential target [33]. Kim et al. reported that SURF4 could induce cellular transformation and cell migration in vitro and has oncogenic transformation ability in vivo [13]. Our study reported a novel biomarker SURF4 in breast cancer. High SURF4 expression was confirmed in breast cancer tissue and cells. SURF4 expression was significantly higher in tumor. The high SURF4 expression in breast cancer tissue was further validated by IHC. Besides, significant high SURF4 expression (P < 0.01) was observed in MCF7 (Human luminal A type breast cancer cell), BT474 (Human luminal B type breast cancer cell), SKBR3 (Human HER2 overexpression type breast cancer cell), MDAMB231 and 4T1 (Human triple-negative breast cancer cell). The in vitro experiments suggested SURF4 promoted the proliferation and migration of 4T1 cells. Combing the results of GSEA analysis, SURF4 may influence the development of breast cancer by controlling chemokine signaling pathway, etc. However, the underlying mechanisms need to be studied in the future. Also, the in vivo experiments can improve the evidence power of the findings.
In conclusion, high expression of SURF4 was first found in breast cancer. SURF4 expression exhibited independent prognostic value for OS, and patients with high SURF4 expression had shorter OS. SURF4 promoted the proliferation and migration of 4T1 cells. SURF4 may be a biomarker to play a role in diagnosis and prognosis of breast cancer. Our findings may indicate SURF4 as a novel therapeutic target for treatment of breast cancer.
Materials and Methods
Data processing and comparison
The files of mRNA expression and associated clinical data were acquired from the The Cancer Genome Atlas (TCGA) database [34]. Non-parametric rank sum tests were used to evaluate the SURF4 mRNA expression. Wilcoxon rank sum tests and Kruskal-Wallis tests were used to compare two and multiple subgroups, respectively. Chi-square tests along with Fisher’s exact tests were used to evaluate the characteristics correlated with SURF4 expression.
Diagnostic value evaluation
The pROC program was used for visualization of ROC curves, which were plotted to evaluate the diagnostic value of SURF4 [35]. According to the cut-off value obtained from ROC curves between normal and tumor, the patients were further divided into the low and high SURF4 expression groups. The area under the ROC curves (AUC) was also calculated.
Survival evaluation and nomogram plotting
The Kaplan-Meier curves were used to analyze the OS and RFS by the R survival package [36]. To investigate the independent predictive ability of SURF4 in breast cancer, univariate and multivariate Cox analysis were carried out. According to SURF4 expression, the patients with breast cancer were divided into groups. Based on different SURF4 expression, the age, HER2, stage, margin status, and 1,3,5-year OS were compared.
GSEA analysis and TIMER database mining
To investigate the relation between SURF4 expression and enriched signaling pathways, the TCGA database was first searched, and then GSEA analysis was performed. The TIMER database was used for study of SURF4 expression in different cancers.
Sample collection
Breast cancer and adjacent tissues were obtained from 35 subjects, and placed in liquid nitrogen immediately after resection. The study was approved by the institutional ethical committee and conformed to the Declaration of Helsinki.
Cell culture and plasmid transfection
The cells were cultured in 1640 medium containing 10% fetal bovine serum, and transfected with the si-NC (negative control), si-SURF4 (small-interfering RNAs against SURF4) and O-SURF4(overexpressed SURF4) plasmids. All the plasmids were purchased from Genepharma (Shanghai, China).
Real-time quantitative PCR
Total RNA was extracted using TRIzol (Invitrogen, USA). 1 mL of isolated RNA was used for the reverse transcription. The real-time quantitative PCR (RT-qPCR) procedure was then completed. The experiments were repeated for 3 times. The primers are as follows: SURF4 (Forward: 5′-CCTTTAAGGCTTGGCCTACG-3′; Reverse: 5′- GGGCCAGGTTCCTCATCAAA-3′), and β-actin (Forward: 5′-GGAGCGAGATCCCTCCAAAAT-3′; Reverse: 5′-GGCTGTTGTCATACTTCTCATGG-3′).
Immunohistochemistry staining
The tissues were from single patient. The immunohistochemistry (IHC) staining was performed according to the manual instructions (#13079, Cell Signaling Technology, MA, USA). Anti-SURF4 primary antibody (ab133369, Abcam company, Shanghai, China) and corresponding secondary antibody (rabbit) were used. The fluorescence microscope was used for imaging.
Cell proliferation and migration assay
After a 24-hour culture period with different plasmids, the absorbance at 450 nm was measured after the addition of 10 μL of CCK-8 reagent (CK04, Dojindo company, Beijing, China) [37]. After being scraped to create a 1-mm gap, the cells treated with different plasmids were grown for 48 hour, and images were obtained at 0 and 48 hours to record the migration distance [38]. The experiments were repeated for 3 times.
Statistical analysis
R3.5.1 was used to perform bioinformatics analysis [39]. The Kaplan-Meier curve was used to analyze the survival rate [40]. To investigate the independent predictive capability of SURF4 in breast cancer, univariate and multivariate Cox analyses were used. It was statistically significant at P < 0.05.
Author Contributions
J.Z. and B.X. designed this study and had full access to all of data in the study; J.Z. and F.M. extracted the data; J.H., C.L., and F.G. analyzed and interpreted the data; J.Z. and J.H. performed the experiments; J.Z. and F.M. wrote the paper. All authors reviewed the manuscript.
Conflicts of Interest
The authors have no conflicts of interest.
Ethical Statement and Consent
The study was approved by the Ethics Committee of Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College. The written consent of all subjects or their legal representatives has been obtained.
Funding
This study was funded by the National Key Research and Development Program of China (2021YFF1201300).
Editorial Note
This corresponding author has a verified history of publications using a personal email address for correspondence.
References
- 1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 71:209–49. https://doi.org/10.3322/caac.21660 [PubMed]
- 2. Sørlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS, Thorsen T, Quist H, Matese JC, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA. 2001; 98:10869–74. https://doi.org/10.1073/pnas.191367098 [PubMed]
- 3. Stephens PJ, Tarpey PS, Davies H, Van Loo P, Greenman C, Wedge DC, Nik-Zainal S, Martin S, Varela I, Bignell GR, Yates LR, Papaemmanuil E, Beare D, et al, and Oslo Breast Cancer Consortium (OSBREAC). The landscape of cancer genes and mutational processes in breast cancer. Nature. 2012; 486:400–4. https://doi.org/10.1038/nature11017 [PubMed]
- 4. Harbeck N, Gnant M. Breast cancer. Lancet. 2017; 389:1134–50. https://doi.org/10.1016/S0140-6736(16)31891-8 [PubMed]
- 5. Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012; 490:61–70. https://doi.org/10.1038/nature11412 [PubMed]
- 6. Allemani C, Matsuda T, Di Carlo V, Harewood R, Matz M, Nikšić M, Bonaventure A, Valkov M, Johnson CJ, Estève J, Ogunbiyi OJ, Azevedo E Silva G, Chen WQ, et al, and CONCORD Working Group. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018; 391:1023–75. https://doi.org/10.1016/S0140-6736(17)33326-3 [PubMed]
- 7. Cardoso F, Spence D, Mertz S, Corneliussen-James D, Sabelko K, Gralow J, Cardoso MJ, Peccatori F, Paonessa D, Benares A, Sakurai N, Beishon M, Barker SJ, Mayer M. Global analysis of advanced/metastatic breast cancer: Decade report (2005-2015). Breast. 2018; 39:131–8. https://doi.org/10.1016/j.breast.2018.03.002 [PubMed]
- 8. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 9. Kwong GA, Ghosh S, Gamboa L, Patriotis C, Srivastava S, Bhatia SN. Synthetic biomarkers: a twenty-first century path to early cancer detection. Nat Rev Cancer. 2021; 21:655–68. https://doi.org/10.1038/s41568-021-00389-3 [PubMed]
- 10. Loibl S, Poortmans P, Morrow M, Denkert C, Curigliano G. Breast cancer. Lancet. 2021; 397:1750–69. https://doi.org/10.1016/S0140-6736(20)32381-3 [PubMed]
- 11. Loke SY, Lee ASG. The future of blood-based biomarkers for the early detection of breast cancer. Eur J Cancer. 2018; 92:54–68. https://doi.org/10.1016/j.ejca.2017.12.025 [PubMed]
- 12. Zou J, Wang E. Cancer Biomarker Discovery for Precision Medicine: New Progress. Curr Med Chem. 2019; 26:7655–71. https://doi.org/10.2174/0929867325666180718164712 [PubMed]
- 13. Kim J, Hong CM, Park SM, Shin DH, Kim JY, Kwon SM, Kim JH, Kim CD, Lim DS, Lee D. SURF4 has oncogenic potential in NIH3T3 cells. Biochem Biophys Res Commun. 2018; 502:43–7. https://doi.org/10.1016/j.bbrc.2018.05.116 [PubMed]
- 14. Emmer BT, Lascuna PJ, Tang VT, Kotnik EN, Saunders TL, Khoriaty R, Ginsburg D. Murine Surf4 is essential for early embryonic development. PLoS One. 2020; 15:e0227450. https://doi.org/10.1371/journal.pone.0227450 [PubMed]
- 15. Reeves JE, Fried M. The surf-4 gene encodes a novel 30 kDa integral membrane protein. Mol Membr Biol. 1995; 12:201–8. https://doi.org/10.3109/09687689509027508 [PubMed]
- 16. Mitrovic S, Ben-Tekaya H, Koegler E, Gruenberg J, Hauri HP. The cargo receptors Surf4, endoplasmic reticulum-Golgi intermediate compartment (ERGIC)-53, and p25 are required to maintain the architecture of ERGIC and Golgi. Mol Biol Cell. 2008; 19:1976–90. https://doi.org/10.1091/mbc.e07-10-0989 [PubMed]
- 17. Cardoso F, Kyriakides S, Ohno S, Penault-Llorca F, Poortmans P, Rubio IT, Zackrisson S, Senkus E, and ESMO Guidelines Committee. Electronic address: [email protected]. Early breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up†. Ann Oncol. 2019; 30:1194–220. https://doi.org/10.1093/annonc/mdz173 [PubMed]
- 18. Kravchenko J, Akushevich I, Seewaldt VL, Abernethy AP, Lyerly HK. Breast cancer as heterogeneous disease: contributing factors and carcinogenesis mechanisms. Breast Cancer Res Treat. 2011; 128:483–93. https://doi.org/10.1007/s10549-011-1347-z [PubMed]
- 19. Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, et al. Molecular portraits of human breast tumours. Nature. 2000; 406:747–52. https://doi.org/10.1038/35021093 [PubMed]
- 20. Ignatiadis M, Sotiriou C. Luminal breast cancer: from biology to treatment. Nat Rev Clin Oncol. 2013; 10:494–506. https://doi.org/10.1038/nrclinonc.2013.124 [PubMed]
- 21. Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. Lancet. 2015; 386:1341–52. https://doi.org/10.1016/S0140-6736(15)61074-1 [PubMed]
- 22. Creighton CJ. The molecular profile of luminal B breast cancer. Biologics. 2012; 6:289–97. https://doi.org/10.2147/BTT.S29923 [PubMed]
- 23. LaBoy C, Siziopikou KP, Rosen L, Blanco LZ
Jr , Pincus JL. Clinicopathologic features of unexpectedly HER2 positive breast carcinomas: An institutional experience. Pathol Res Pract. 2021; 222:153441. https://doi.org/10.1016/j.prp.2021.153441 [PubMed] - 24. Foulkes WD, Stefansson IM, Chappuis PO, Bégin LR, Goffin JR, Wong N, Trudel M, Akslen LA. Germline BRCA1 mutations and a basal epithelial phenotype in breast cancer. J Natl Cancer Inst. 2003; 95:1482–5. https://doi.org/10.1093/jnci/djg050 [PubMed]
- 25. Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K, Karaca G, Troester MA, Tse CK, Edmiston S, Deming SL, Geradts J, Cheang MCU, et al. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006; 295:2492–502. https://doi.org/10.1001/jama.295.21.2492 [PubMed]
- 26. Sun X, Kaufman PD. Ki-67: more than a proliferation marker. Chromosoma. 2018; 127:175–86. https://doi.org/10.1007/s00412-018-0659-8 [PubMed]
- 27. Menon SS, Guruvayoorappan C, Sakthivel KM, Rasmi RR. Ki-67 protein as a tumour proliferation marker. Clin Chim Acta. 2019; 491:39–45. https://doi.org/10.1016/j.cca.2019.01.011 [PubMed]
- 28. Urruticoechea A, Smith IE, Dowsett M. Proliferation marker Ki-67 in early breast cancer. J Clin Oncol. 2005; 23:7212–20. https://doi.org/10.1200/JCO.2005.07.501 [PubMed]
- 29. Goncalves R, DeSchryver K, Ma C, Tao Y, Hoog J, Cheang M, Crouch E, Dahiya N, Sanati S, Barnes M, Sarian LOZ, Olson J, Allred DC, Ellis MJ. Development of a Ki-67-based clinical trial assay for neoadjuvant endocrine therapy response monitoring in breast cancer. Breast Cancer Res Treat. 2017; 165:355–64. https://doi.org/10.1007/s10549-017-4329-y [PubMed]
- 30. Dowsett M, Smith IE, Ebbs SR, Dixon JM, Skene A, Griffith C, Boeddinghaus I, Salter J, Detre S, Hills M, Ashley S, Francis S, Walsh G, and IMPACT Trialists. Short-term changes in Ki-67 during neoadjuvant treatment of primary breast cancer with anastrozole or tamoxifen alone or combined correlate with recurrence-free survival. Clin Cancer Res. 2005; 11:951s–8s. [PubMed]
- 31. Ellis MJ, Suman VJ, Hoog J, Lin L, Snider J, Prat A, Parker JS, Luo J, DeSchryver K, Allred DC, Esserman LJ, Unzeitig GW, Margenthaler J, et al. Randomized phase II neoadjuvant comparison between letrozole, anastrozole, and exemestane for postmenopausal women with estrogen receptor-rich stage 2 to 3 breast cancer: clinical and biomarker outcomes and predictive value of the baseline PAM50-based intrinsic subtype--ACOSOG Z1031. J Clin Oncol. 2011; 29:2342–9. https://doi.org/10.1200/JCO.2010.31.6950 [PubMed]
- 32. Dowsett M, Nielsen TO, A’Hern R, Bartlett J, Coombes RC, Cuzick J, Ellis M, Henry NL, Hugh JC, Lively T, McShane L, Paik S, Penault-Llorca F, et al, and International Ki-67 in Breast Cancer Working Group. Assessment of Ki67 in breast cancer: recommendations from the International Ki67 in Breast Cancer working group. J Natl Cancer Inst. 2011; 103:1656–64. https://doi.org/10.1093/jnci/djr393 [PubMed]
- 33. Yue Y, Xia L, Xu S, Wang C, Wang X, Lu W, Xie X. SURF4 maintains stem-like properties via BIRC3 in ovarian cancer cells. J Gynecol Oncol. 2020; 31:e46. https://doi.org/10.3802/jgo.2020.31.e46 [PubMed]
- 34. Cai H, Jiao Y, Li Y, Yang Z, He M, Liu Y. Low CYP24A1 mRNA expression and its role in prognosis of breast cancer. Sci Rep. 2019; 9:13714. https://doi.org/10.1038/s41598-019-50214-z [PubMed]
- 35. Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez JC, Müller M. pROC: an open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinformatics. 2011; 12:77. https://doi.org/10.1186/1471-2105-12-77 [PubMed]
- 36. Liu N, Zhou Y, Lee JJ. IPDfromKM: reconstruct individual patient data from published Kaplan-Meier survival curves. BMC Med Res Methodol. 2021; 21:111. https://doi.org/10.1186/s12874-021-01308-8 [PubMed]
- 37. Cai H, Dai X, Guo X, Zhang L, Cao K, Yan F, Ji B, Liu Y. Ataxia telangiectasia mutated inhibitor-loaded copper sulfide nanoparticles for low-temperature photothermal therapy of hepatocellular carcinoma. Acta Biomater. 2021; 127:276–86. https://doi.org/10.1016/j.actbio.2021.03.051 [PubMed]
- 38. Cai H, Wang R, Guo X, Song M, Yan F, Ji B, Liu Y. Combining Gemcitabine-Loaded Macrophage-like Nanoparticles and Erlotinib for Pancreatic Cancer Therapy. Mol Pharm. 2021; 18:2495–506. https://doi.org/10.1021/acs.molpharmaceut.0c01225 [PubMed]
- 39. Heinemann J. Cluster Analysis of Untargeted Metabolomic Experiments. Methods Mol Biol. 2019; 1859:275–85. https://doi.org/10.1007/978-1-4939-8757-3_16 [PubMed]
- 40. Ranstam J, Cook JA. Kaplan-Meier curve. Br J Surg. 2017; 104:442. https://doi.org/10.1002/bjs.10238 [PubMed]