Introduction
Electrocardiogram (ECG) recordings are widely used in clinical routine. Quantitative ECG measures thereby reflect cardiac excitation (RR-interval), atrial (PR-interval) and ventricular (QRS-duration) conduction, and cardiac repolarization (heart rate corrected QTc). All mentioned ECG measures are age-dependent [1]. Some individuals present with altered ECG measures, for example with early-onset PR prolongation, before reaching an expected calendar age. It is unclear if such premature changes reflect underlying subclinical pathologies, or if these individuals are affected by a premature biological age, which does not correspond with their calendar age.
Telomere length has gained scientific interest as a marker of biological aging [2]. Telomeres are tandem repeats of six nucleotides (TTAGGG) located at the end of each chromosome. Intact telomeres prevent spontaneous DNA damage and preserve genomic integrity. However, telomeres shorten during each mitotic cell cycle. As cellular lifetime progresses, telomere shortening leads to apoptosis [3, 4]. Men have shorter telomeres on average compared to women [5]. Furthermore, telomere shortening has been linked to numerous common conditions including obesity, smoking, hypertension, elevated plasma cholesterol levels, cancer, and cardiovascular diseases [6–13].
Here we used data from the large and well-characterized, community-based KORA Study to systematically test associations between ECG measures and telomere length. We hypothesized that shorter telomeres reflect advanced biological age and are associated with age-dependent changes in ECG measures.
Materials and Methods
Study population
The community-based KORA (Cooperative Health Research in the Region of Augsburg) Study has been conducted since 1984 in the population living in and around Augsburg, Germany [14]. From 1999 to 2001, 4,261 individuals age 25-74 years of German nationality, randomly selected through the registration office, were enrolled into the KORA S4 survey. The KORA F4 survey was performed as a seven-year follow-up of KORA S4, conducted between 2006 and 2007. Overall, 3,080 individuals (79.6% of KORA S4) agreed to participate. Details have been reported elsewhere [15]. All participants provided written informed consent to participation in the study. The investigations were carried out in accordance with the Declaration of Helsinki. All study methods were approved by the ethics committee of the Bavarian Chamber of Physicians, Munich (S4: EC No. 99186 and for genetic epidemiological questions 05004, F4: EC No. 06068).
Clinical covariates and electrocardiogram recording
All participants received an assessment of demographic, anthropometric, and clinical characteristics through a standardized personal interview, a physical examination, and a self-administrated questionnaire. All participants further received a twelve-lead Electrocardiogram (Hörmann Bioset 9000) recorded under standardized conditions after 10 min rest in supine position. For the present study, ECGs were filtered for ECG-quality and analyzed using the automated Hannover ECG System (HES) as reported before [16, 17]. We excluded participants presenting with extreme PR values (≤80ms or ≥320ms), second or third degree atrioventricular block, atrial fibrillation, a history of myocardial infarction, Wolff-Parkinson-White syndrome, or in the presence of a pacemaker or implantable cardioverter defibrillator. We further excluded currently pregnant women (Figure 1). For the presented analyses we tested the four commonly used ECG parameters, RR-interval, PR-interval, QRS duration, and QTc corrected using Bazett’s formula.
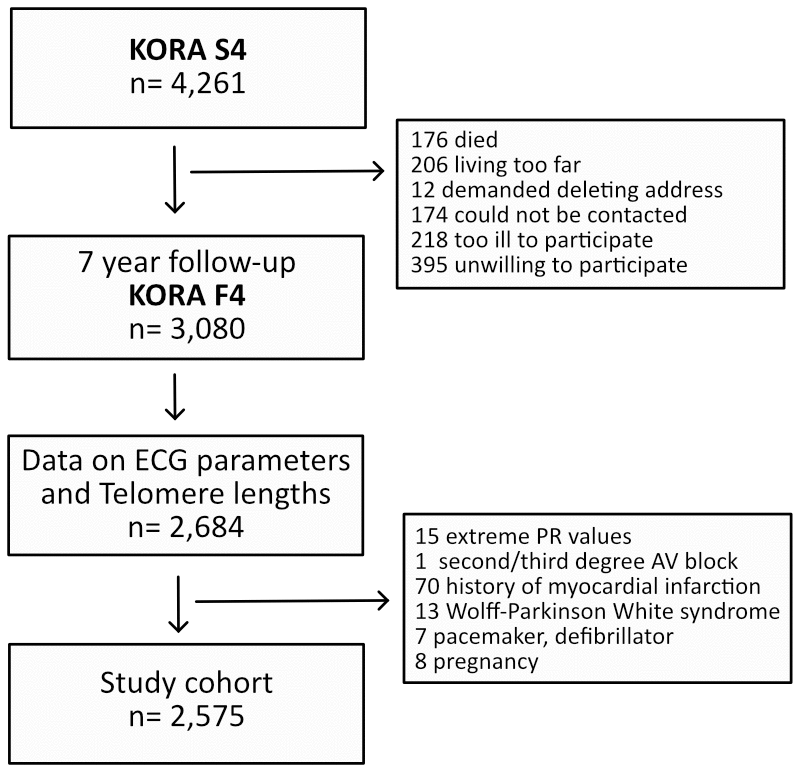
Figure 1. Illustration of the cohort composition.
Measurement of telomere length
Participants provided biosamples for laboratory assessment including genetic analyses upon enrollment into the community-based KORA study and at the same visit as all other measures and characteristics. In all those with available peripheral blood biospecimens, we determined telomere length as previously described in detail [18]. In brief, DNA was extracted from peripheral blood leucocytes and telomere length was determined using a quantitative PCR-based technique. By expressing telomere length as the T/S ratio of the telomere repeat copy number (T) to the single copy gene 36B4 (S), we standardized results irrespective of PCR cycles. For quality control, a standard DNA from a K562 cell line was used to assess variation across PCR plates, and duplicates were used to assess intra-sample variability. The coefficient of variation of telomere length was 3.1%.
Statistical analysis
Discrete data are presented as absolute and relative frequencies. Continuous variables are shown as mean ± standard deviation. Telomere length is used as a continuous variable and is considered normally distributed. Telomere length is compared by sex using the Welch two sample t-test. An unadjusted linear regression is fitted for telomere length and age. We then fitted multivariable-adjusted linear regression models to associate the outcome of ECG measures with the predictor telomere length. We accounted for age, sex, height, and body-mass-index. We present sex-stratified results. All statistical analyses were performed using Rstudio (Version 1.2.1335, Boston, MA, USA). Significance was assumed for a two-sided p <0.05.
Results
The study flow is visualized in Figure 1. Of 3,080 participants enrolled in the KORA F4 study, 2,684 had complete availability of ECG and telomere length data. Of these, 109 individuals fulfilled any exclusion criteria. Hence, 2,575 individuals were included into the final analysis. Baseline characteristics of the study cohort and the distribution of their ECG measures are listed in Table 1. The cohort’s mean age was 54.9±12.9 years and 53.4% were females.
Table 1. Cohort characteristics.
| n=2,575 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Demographics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age; years | 54.9±12.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male sex; n (%) | 1,200 (46.6%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECG measures | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RR-interval; ms | 944±144 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR-interval; ms | 167±23 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QRS-duration; ms | 92±9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc-interval; ms | 425±20 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Telomere assessment | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Relative telomere length | 1.86±0.33 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Telomere length expressed relatively using a T/S ratio of measured telomere length (T) divided by the copy number of the single copy gene 36B4 (S). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The mean telomere length, expressed as the T/S ratio relative to the single copy gene 36B4, was 1.86±0.33. As previously reported, men had significantly shorter telomeres compared to women (1.81 vs. 1.90, p<0.001). Also, telomeres were significantly shorter in older individuals. The age-dependent relation is depicted in Figure 2. Per year of age, the decrease was -0.0099 relative T/S units (standard error 0.0005; p<0.001). This age-dependent relation was likewise observed when stratified by sex (data not presented).
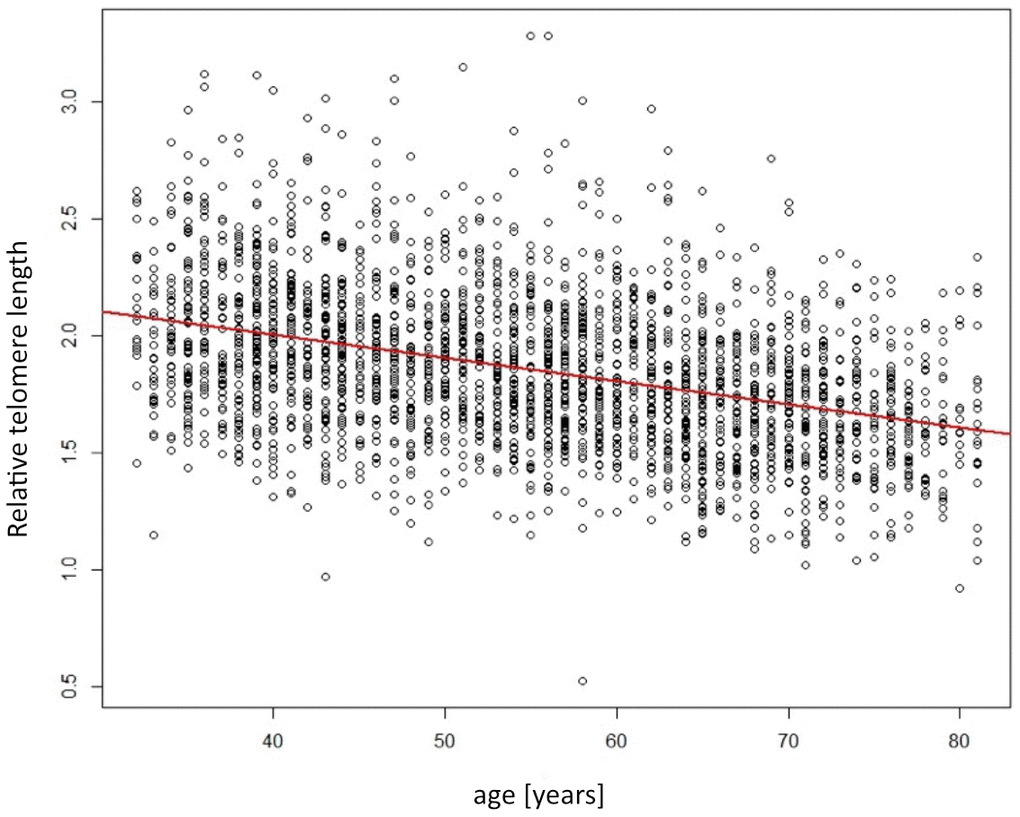
Figure 2. Scatterplot showing the relation of telomere length (y-axis) depending on age (x-axis). Red line indicates the linear regression line.
The associations between ECG measures and telomere length are summarized in Table 2. By unadjusted regression, we found a significant relation between telomere length and RR-interval (p=0.023), PR-interval (p<0.001), and QTc (p<0.001). Regarding the directionality of effects, a shorter telomere length associated with a shorter RR-interval, i.e., a higher heart rate, but inversely correlated with a longer PR-interval and a longer QTc, respectively. However, after accounting for age and other covariates, no significant association between ECG measures and telomere length remained. Also, sex-stratified, adjusted analyses revealed no significant associations (Table 2).
Table 2. Regression results.
| Model 1 | Model 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Beta (SE) | p | Beta (SE) | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RR-interval | 3.1x10-5 (4.2x10-5) | 0.46 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Men | 3.7x10-5 (5.8x10-5) | 0.52 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Women | 2.0x10-5 (6.2x10-5) | 0.75 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR-interval | 1.5x10-4 (2.7x10-4) | 0.58 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Men | -1.2x10-4 (3.7x10-4) | 0.74 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Women | 4.6x10-4 (3.8x10-4) | 0.23 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QRS-duration | 7.3x10-4 (7.2x10-4) | 0.31 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Men | 6.0x10-4 (1.0x10-3) | 0.56 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Women | 7.8x10-4 (1.0x10-3) | 0.43 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| QTc-interval | -8.6x10-3 (9.9x10-3) | 0.38 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Men | -6.2x10-3 (1.4x10-2) | 0.66 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Women | -9.6x10-3 (1.4x10-2) | 0.49 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The table lists the beta coefficients (Beta) and standard errors (SE) of linear regression models for the ECG measures as outcomes and telomere length as predictor. Unadjusted models are described in the manuscript text only. Model 1 is adjusted for age, sex, height, and body-mass-index. Model 2 presents sex-stratified results adjusted for age, height, and body-mass-index. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Discussion
In the large, community-based KORA study, we associated commonly determined ECG measures with peripheral blood leukocyte telomere length as a marker of biological aging. Despite a known age-dependence of the ECG measures, we found no effect of telomere length beyond the influence of calendar age.
The ECG measures in our study reflect the full cardiac cycle from excitation (RR) via atrial (PR) and ventricular (QRS) conduction to ventricular repolarization (QTc). All measures thereby can be influenced by external factors. Such external conditions exemplarily include heart failure and myocardial infarction, which may result in QRS-prolongation [19]. Various drugs can prolong both the PR-interval and QTc [20, 21]. Importantly, also advancing age is an external factor influencing these ECG measures [1]. Consequently, elderly individuals often require a pacemaker due to a symptomatic slowing of heart rate or a symptomatic atrio-ventricular conduction block [22].
Since aging per se is a non-avertible process, the question arises if a biological age differing from the person’s calendar age is a marker that can be approximated clinically. Telomere length is a well-established marker of biological age [2]. For various clinically relevant cardiovascular conditions, an association with telomere length beyond the effect of calendar age has been shown [2, 11].
With the results of our presented analysis, we confirm the prior notion that predominantly RR-interval, PR-interval, and QTc are indeed age-related [23–26]. All three measures show a highly significant association with telomere length as a marker of age in general. However, after accounting for the effect of calendar age, no significant association with telomere length remained. We therefore conclude that biological age, as measured by telomere length, beyond the effect of calendar age is not a relevant contributor to changes in the investigated ECG measures in a community-based cohort.
Whereas telomere length has been studied for the relation to various clinical conditions and markers [6–10], only limited data exist regarding their role on ECG measures. The available prior data are conflicting. A small study in 273 Australian patients with or without diabetes mellitus described a weak correlation with QRS-duration depending on telomere length status [27]. Another study in 222 Japanese patients presenting with several predominantly chronic neurologic conditions investigated ECG measures in relation to semi-quantitatively measured telomere length using Southern blot analysis. The authors report an association with ECG measures including PR-interval, QRS-duration, and QTc [28]. Importantly, both studies did not adjust for age. To the best of our knowledge, the only prior study that investigated ECG measures in relation to telomere length and did account for calendar age is a small investigation in 139 healthy Chinese patients. Telomere length was semi-quantitatively determined using restriction fragment length analysis and the authors did not find a correlation with ECG measures after age adjustment [29]. Given the limitations of the existing data, we present a most systematic analysis of ECG measures representing the full cardiac cycle. Further, we study a well characterized cohort that represents the German general population and is not restricted to a specific underlying disease phenotype. Most importantly, our analyzed cohort comprises 2,575 individuals. It is hence almost ten-fold larger than prior reports. This cohort size warrants sufficient statistical power to also detect weak association signals. We are thus confident that indeed no relevant effect of telomere length on ECG measures is missed due to a lack of power.
Yet, some considerations are required when interpreting our findings: The KORA study enrolled participants of European descent. Since telomere length shares a heritable component [30], our results may not be fully generalizable. Also, we studied a community-based sample where possible pre-existing diseases and conditions have a low prevalence. It may thus be that our findings cannot be extrapolated to large cohorts of patients presenting with a specific underlying condition. We assessed telomere length in peripheral blood leucocytes, where results do not necessarily reflect fully the telomere length in cardiac tissue. Yet, prior data suggest a high correlation across tissues [31]. Furthermore, we were not able to investigate different leukocyte subtypes, which may be characterized by differential telomere length results that may hence have influenced the association with ECG measures. Most importantly, prolongation of the studied ECG measures can itself predispose to clinical conditions. Exemplarily, PR-interval prolongation is an established risk factor for atrial fibrillation (AF) [32]. Even though we did not find a relevant association between ECG measures and telomere length, it remains unresolved if telomere length is relevant for the pathophysiology of selected age-dependent conditions. At least for AF, a relation with telomere length has been suggested, but the results remain conflicting [33–35].
In conclusion, ECG measures are clearly age-dependent. However, in a large, well-characterized, and sufficiently powered cohort we were not able to substantiate the hypothesis that telomere length as a marker of biological age is a relevant contributor to this age-dependent prolongation of ECG measures. It remains to be resolved if telomere length is involved in the pathophysiology of cardiac diseases like AF that are correlated with a prolongation of ECG measures.
Author Contributions
Conception and design of the study: ASF, RF, MW, CG, AP, MM-N, SK, MFS. Data acquisition and analysis: RF, MW, CG, AP, MM-N, MFS. Drafting a significant portion of the manuscript or figures: ASF, MFS.
Acknowledgments
Data collection in the KORA study is conducted in cooperation with the University Hospital of Augsburg.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
Dr. von Falkenhausen was supported by the German Research Foundation (413635475) through the Clinician scientist program in vascular medicine (PRIME). The KORA study was initiated and financed by the Helmholtz Zentrum München – German Research Center for Environmental Health, which is funded by the German Federal Ministry of Education and Research (BMBF) and by the State of Bavaria. Furthermore, KORA research was supported within the Munich Center of Health Sciences (MC-Health), Ludwig-Maximilians-Universität, as part of LMUinnovativ.
References
- 1. Vicent L, Martínez-Sellés M. Electrocardiogeriatrics: ECG in advanced age. J Electrocardiol. 2017; 50:698–700. https://doi.org/10.1016/j.jelectrocard.2017.06.003 [PubMed]
- 2. Rizvi S, Raza ST, Mahdi F. Telomere length variations in aging and age-related diseases. Curr Aging Sci. 2014; 7:161–7. https://doi.org/10.2174/1874609808666150122153151 [PubMed]
- 3. Olovnikov AM. A theory of marginotomy. The incomplete copying of template margin in enzymic synthesis of polynucleotides and biological significance of the phenomenon. J Theor Biol. 1973; 41:181–90. https://doi.org/10.1016/0022-5193(73)90198-7 [PubMed]
- 4. Makarov VL, Hirose Y, Langmore JP. Long G tails at both ends of human chromosomes suggest a C strand degradation mechanism for telomere shortening. Cell. 1997; 88:657–66. https://doi.org/10.1016/s0092-8674(00)81908-x [PubMed]
- 5. Möller P, Mayer S, Mattfeldt T, Müller K, Wiegand P, Brüderlein S. Sex-related differences in length and erosion dynamics of human telomeres favor females. Aging (Albany NY). 2009; 1:733–9. https://doi.org/10.18632/aging.100068 [PubMed]
- 6. Nordfjäll K, Eliasson M, Stegmayr B, Melander O, Nilsson P, Roos G. Telomere length is associated with obesity parameters but with a gender difference. Obesity (Silver Spring). 2008; 16:2682–9. https://doi.org/10.1038/oby.2008.413 [PubMed]
- 7. Lee M, Martin H, Firpo MA, Demerath EW. Inverse association between adiposity and telomere length: The Fels Longitudinal Study. Am J Hum Biol. 2011; 23:100–6. https://doi.org/10.1002/ajhb.21109 [PubMed]
- 8. Valdes AM, Andrew T, Gardner JP, Kimura M, Oelsner E, Cherkas LF, Aviv A, Spector TD. Obesity, cigarette smoking, and telomere length in women. Lancet. 2005; 366:662–4. https://doi.org/10.1016/S0140-6736(05)66630-5 [PubMed]
- 9. Lung FW, Ku CS, Kao WT. Telomere length may be associated with hypertension. J Hum Hypertens. 2008; 22:230–2. https://doi.org/10.1038/sj.jhh.1002314 [PubMed]
- 10. Strandberg TE, Saijonmaa O, Fyhrquist F, Tilvis RS, Strandberg AY, Miettinen TA, Pitkälä KH, Salomaa V. Telomere length in old age and cholesterol across the life course. J Am Geriatr Soc. 2011; 59:1979–81. https://doi.org/10.1111/j.1532-5415.2011.03610_13.x [PubMed]
- 11. Fuster JJ, Andrés V. Telomere biology and cardiovascular disease. Circ Res. 2006; 99:1167–80. https://doi.org/10.1161/01.RES.0000251281.00845.18 [PubMed]
- 12. Riegert-Johnson DL, Boardman LA, Crook JE, Thomas CS, Johnson RA, Roberts ME. Shorter peripheral blood telomeres are a potential biomarker for patients with advanced colorectal adenomas. Int J Biol Markers. 2012; 27:e375–80. https://doi.org/10.5301/JBM.2012.9347 [PubMed]
- 13. Xie H, Wu X, Wang S, Chang D, Pollock RE, Lev D, Gu J. Long telomeres in peripheral blood leukocytes are associated with an increased risk of soft tissue sarcoma. Cancer. 2013; 119:1885–91. https://doi.org/10.1002/cncr.27984 [PubMed]
- 14. Holle R, Happich M, Löwel H, Wichmann HE, and MONICA/KORA Study Group. KORA--a research platform for population based health research. Gesundheitswesen. 2005 (Suppl 1); 67:S19–25. https://doi.org/10.1055/s-2005-858235 [PubMed]
- 15. Laxy M, Knoll G, Schunk M, Meisinger C, Huth C, Holle R. Quality of Diabetes Care in Germany Improved from 2000 to 2007 to 2014, but Improvements Diminished since 2007. Evidence from the Population-Based KORA Studies. PLoS One. 2016; 11:e0164704. https://doi.org/10.1371/journal.pone.0164704 [PubMed]
- 16. Zywietz C, Borovsky D, Götsch G, Joseph G. Methodology of ECG interpretation in the Hannover program. Methods Inf Med. 1990; 29:375–85. https://doi.org/10.1055/s-0038-1634800 [PubMed]
- 17. Sinner MF, Pfeufer A, Perz S, Schulze-Bahr E, Mönnig G, Eckardt L, Beckmann BM, Wichmann HE, Breithardt G, Steinbeck G, Fabritz L, Kääb S, Kirchhof P. Spontaneous Brugada electrocardiogram patterns are rare in the German general population: results from the KORA study. Europace. 2009; 11:1338–44. https://doi.org/10.1093/europace/eup205 [PubMed]
- 18. Codd V, Nelson CP, Albrecht E, Mangino M, Deelen J, Buxton JL, Hottenga JJ, Fischer K, Esko T, Surakka I, Broer L, Nyholt DR, Mateo Leach I, et al, and CARDIoGRAM consortium. Identification of seven loci affecting mean telomere length and their association with disease. Nat Genet. 2013; 45:422–7. https://doi.org/10.1038/ng.2528 [PubMed]
- 19. Cupa J, Strebel I, Badertscher P, Abächerli R, Twerenbold R, Schumacher L, Boeddinghaus J, Nestelberger T, Maechler P, Kozhuharov N, Giménez MR, Wildi K, du Fay de Lavallaz J, et al. Diagnostic and prognostic value of QRS duration and QTc interval in patients with suspected myocardial infarction. Cardiol J. 2018; 25:601–10. https://doi.org/10.5603/CJ.a2018.0033 [PubMed]
- 20. Stöllberger C, Huber JO, Finsterer J. Antipsychotic drugs and QT prolongation. Int Clin Psychopharmacol. 2005; 20:243–51. https://doi.org/10.1097/01.yic.0000166405.49473.70 [PubMed]
- 21. Nikolaidou T, Pellicori P, Zhang J, Kazmi S, Goode KM, Cleland JG, Clark AL. Prevalence, predictors, and prognostic implications of PR interval prolongation in patients with heart failure. Clin Res Cardiol. 2018; 107:108–19. https://doi.org/10.1007/s00392-017-1162-6 [PubMed]
- 22. Bush DE, Finucane TE. Permanent cardiac pacemakers in the elderly. J Am Geriatr Soc. 1994; 42:326–34. https://doi.org/10.1111/j.1532-5415.1994.tb01760.x [PubMed]
- 23. Bay M, Vollenweider P, Marques-Vidal P, Bocchi F, Pruvot E, Schläpfer J. Clinical determinants of the PR interval duration in Swiss middle-aged adults: The CoLaus/PsyCoLaus study. Clin Cardiol. 2020; 43:614–21. https://doi.org/10.1002/clc.23356 [PubMed]
- 24. Chubb H, Ceresnak SR, Motonaga KS, Dubin AM. A proposed method for the calculation of age-dependent QRS duration z-scores. J Electrocardiol. 2020; 58:132–4. https://doi.org/10.1016/j.jelectrocard.2019.12.004 [PubMed]
- 25. Niiranen TJ, Enserro DM, Larson MG, Vasan RS. Multisystem Trajectories Over the Adult Life Course and Relations to Cardiovascular Disease and Death. J Gerontol A Biol Sci Med Sci. 2019; 74:1778–85. https://doi.org/10.1093/gerona/gly249 [PubMed]
- 26. Rautaharju PM, Mason JW, Akiyama T. New age- and sex-specific criteria for QT prolongation based on rate correction formulas that minimize bias at the upper normal limits. Int J Cardiol. 2014; 174:535–40. https://doi.org/10.1016/j.ijcard.2014.04.133 [PubMed]
- 27. Zhou Y, Jelinek H, Hambly BD, McLachlan CS. Electrocardiogram QRS duration and associations with telomere length: A cross-sectional analysis in Australian rural diabetic and non-diabetic population. J Electrocardiol. 2017; 50:450–6. https://doi.org/10.1016/j.jelectrocard.2017.02.010 [PubMed]
- 28. Maeda T, Horiuchi T, Makino N. Epigenetic status of subtelomere of peripheral leukocytes corresponds to cardiographic parameters with a sex association. Geriatr Gerontol Int. 2018; 18:1415–9. https://doi.org/10.1111/ggi.13472 [PubMed]
- 29. Zhang WG, Zhu SY, Zhao DL, Jiang SM, Li J, Li ZX, Fu B, Zhang M, Li DG, Bai XJ, Cai GY, Sun XF, Chen XM. The correlation between peripheral leukocyte telomere length and indicators of cardiovascular aging. Heart Lung Circ. 2014; 23:883–90. https://doi.org/10.1016/j.hlc.2013.12.016 [PubMed]
- 30. Honig LS, Kang MS, Cheng R, Eckfeldt JH, Thyagarajan B, Leiendecker-Foster C, Province MA, Sanders JL, Perls T, Christensen K, Lee JH, Mayeux R, Schupf N. Heritability of telomere length in a study of long-lived families. Neurobiol Aging. 2015; 36:2785–90. https://doi.org/10.1016/j.neurobiolaging.2015.06.017 [PubMed]
- 31. Demanelis K, Jasmine F, Chen LS, Chernoff M, Tong L, Delgado D, Zhang C, Shinkle J, Sabarinathan M, Lin H, Ramirez E, Oliva M, Kim-Hellmuth S, et al, and GTEx Consortium. Determinants of telomere length across human tissues. Science. 2020; 369:eaaz6876. https://doi.org/10.1126/science.aaz6876 [PubMed]
- 32. Hayashi H, Miyamoto A, Kawaguchi T, Naiki N, Xue JQ, Matsumoto T, Murakami Y, Horie M. P-pulmonale and the development of atrial fibrillation. Circ J. 2014; 78:329–37. https://doi.org/10.1253/circj.cj-13-0654 [PubMed]
- 33. Zhang N, Fan C, Gong M, Liang X, Zhang W, Li G, Tse G, Liu T. Leucocyte telomere length and paroxysmal atrial fibrillation: A prospective cohort study and systematic review with meta-analysis. J Clin Lab Anal. 2018; 32:e22599. https://doi.org/10.1002/jcla.22599 [PubMed]
- 34. Siland JE, Geelhoed B, van Gelder IC, van der Harst P, Rienstra M. Telomere length and incident atrial fibrillation - data of the PREVEND cohort. PLoS One. 2017; 12:e0171545. https://doi.org/10.1371/journal.pone.0171545 [PubMed]
- 35. Wang S, Gao Y, Zhao L, Hu R, Yang X, Liu Y. Shortened leukocyte telomere length as a potential biomarker for predicting the progression of atrial fibrillation from paroxysm to persistence in the short-term. Medicine (Baltimore). 2021; 100:e26020. https://doi.org/10.1097/MD.0000000000026020 [PubMed]



