Introduction
Glioma is the most common malignant brain tumor in adults [1]. It is generally difficult to treat due to extensive proliferation, invasion, angiogenesis, immunosuppression, and resistance to conventional treatments [2, 3]. Compared with low-grade glioma (LGG, grade II and III), glioblastoma (GBM, WHO grade IV) is more lethal [4]. Even with large surgical resection followed by combined radiotherapy and temozolomide chemotherapy, the median survival of glioblastoma is only 16 months [5].
In solid tumors, a variety of non-cancer cells, including various immune cells, inflammatory cells, vascular cells, fibrotic cells, and even adipocytes, together with cancer cells constitute the tumor microenvironment (TME) [6]. The main tumor-infiltrating immune cells in the tumor microenvironment of glioma are tumor-associated macrophages (TAMs), including blood-circulating monocytes and tissue resident microglia, accounting for about 30-50% of the infiltrating immune cells in GBM [7]. TAMs promote the progression and metastasis of cancer in a variety of ways, such as releasing cancer-promoting growth factors and cytokines, enhancing tumor invasion, inhibiting immune cell function, and stimulating angiogenesis [8–10]. Similar findings have been noticed in gliomas, and TAMs have also been found to mediate drug resistance in glioma immunotherapy [11]. Exploring the regulatory mechanism of TAMs and determining the prognostic value of TAMs related signature will be promising for improvement of the treatment of gliomas.
In this study, we analyzed the tumor-associated macrophages in the glioma by EPIC, MCPCOUNTER and XCELL methods, and found that compared with LGG, TAMs were increased in GBM in the TCGA dataset. The differentially expressed genes (DEGs) between LGG and GBM were selected for weighted correlation network analysis (WGCNA) to identify candidate modules and hub genes that might regulate TAM in glioma. Subsequently, the hub genes were analyzed by univariate cox regression and Kaplan-Meier. The hub genes significantly related to overall survival (OS) were extracted to perform the least absolute shrinkage and selection operator (LASSO) multivariate cox regression algorithm. Finally, nine positive prognostic genes (MYL12A, MSN, S100A4, CHI3L1, PLAUR, EMP3, CASP4, TIMP1 and CCDC109B) were screened out and used to construct a high efficacy prediction model. Moreover, through immune landscape analysis, we found that the risk score was significantly related to tumor microenvironment. In conclusion, we revealed the relationship between TAM and malignancy of glioma, demonstrated the value of TAM related signature in predicting the prognosis of glioma, and provided potential targeted therapy for glioma.
Materials and Methods
Datasets and samples
The expression data and clinical data of merged GBMLGG dataset from The Cancer Genome Atlas (TCGA) were downloaded from University of California Santa Cruz (UCSC) Xena browser (https://xenabrowser.net/datapages/) [12]. The samples with missing data on survival and WHO grade were excluded in this study, and 674 glioma patients we finally obtained in the TCGA dataset. The RNA-seq data and clinical data of mRNAseq-693 and mRNAseq-325 cohorts were downloaded from the Chinese Glioma Genome Atlas (CGGA) data portal (http://www.cgga.org.cn/) [13] Finally, 656 and 309 glioma patients were enrolled in this study, respectively. In addition, three LGG and three GBM samples were collected from patients undergoing surgical treatment from November 2019 to December 2020 in Zhengzhou Central Hospital Affiliated to Zhengzhou University. The clinical diagnosis was confirmed by immunohistochemical staining in the pathology department This study was approved by the institutional review board of Zhengzhou Central Hospital Affiliated to Zhengzhou University, and informed consents were obtained from all patients.
Immune microenvironment analysis
The abundance of tumor-infiltrating macrophage cells in glioma were evaluated by using EPIC, MCPCOUNTER and XCELL algorithms on the TIMER2 platform (http://timer.cistrome.org/) [14]. The ESTIMATE scores, Immune scores and Stromal scores of gliomas were calculated using the R package “estimate”.
Identification of DEGs and GO enrichment analysis
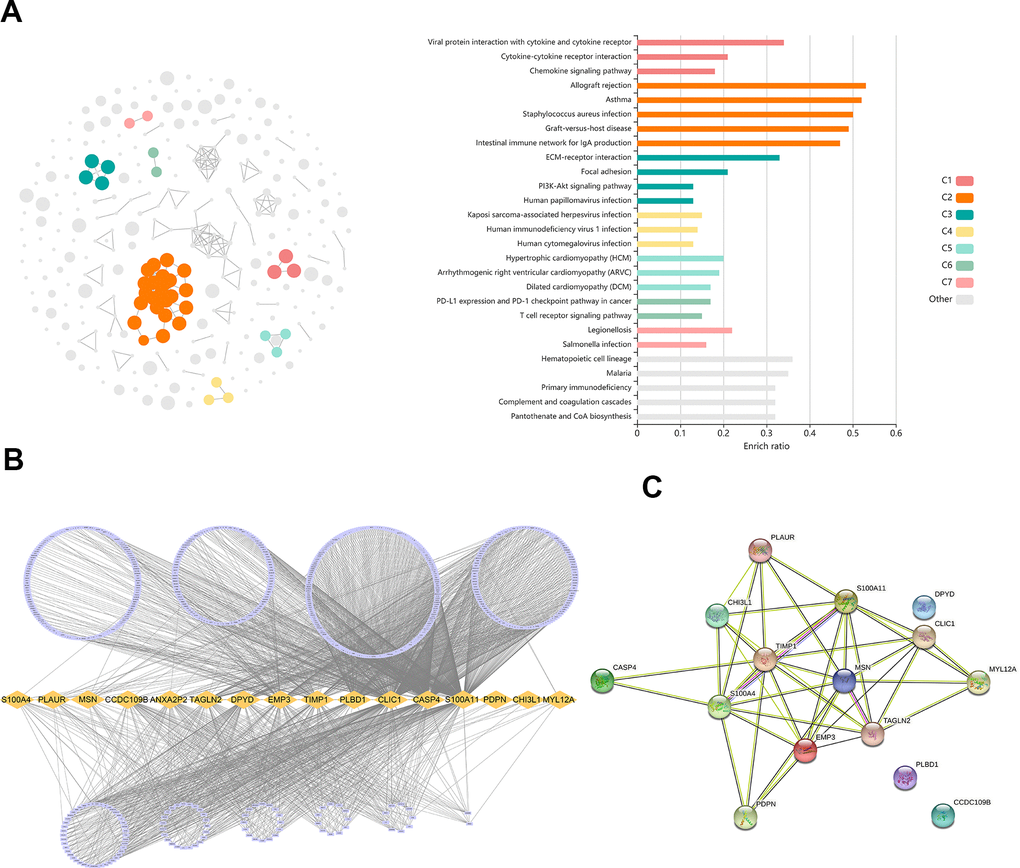
The differently expressed genes (DEGs) (adjusted p-value < 0.05 and |log2FC| ≥ 1) were identified in the TCGA dataset by using the R package“ limma” [15]. Gene enrichment analysis was conducted using the KOBAS-i (http://bioinfo.org/kobas) [16].
Construction of the risk score model
DEGs and clinical traits were incorporated to perform Weighted correlation network analysis (WGCNA) using R package “WGCNA”, the tumor-infiltrating macrophage related hub genes were identified. Univariate Cox regression analysis was performed to identify the hub genes which significantly related to overall survival (OS). After that, the least absolute shrinkage and selection operator (LASSO) multivariate Cox regression algorithm was performed using the R package “glmnet”. to reduce the number of predictors and screen for significant predictors [17, 18]. Finally, the positive genes were screened out, and the coefficients in the risk score signature were constructed based on the most suitable penalty parameter λ. The risk score formula was as follow:
where Coefi is the coefficient, and Expi is the normalized expression of each signature gene.
Immunohistochemical staining
The tissue sections were incubated with anti-CHI3L1 (abcam, ab255297, 1:250), anti-MSN (abcam, ab151542, 1:250) or anti-TIMP1 (abcam, ab211926, 1:250) primary antibody overnight at 4° C. After washing three times, the sections were incubated with horseradish peroxidase-conjugated goat anti-rabbit IgG secondary antibody for 20 minutes, followed by staining with diaminobenzidine. Finally, the sections were counterstained with hematoxylin.
Statistical analysis
One-way ANOVA, Wilcoxon test and t test were used to analyze the significance of differences in gene expression levels and macrophage infiltration levels. Univariate, multivariate, LASSO cox regression and Kaplan-Meier analyses were performed to screen positive prognostic genes and evaluate the risk signature using the R packages “glmnet” and “survival”. Roc curve was drawn using the R package “survivalROC.” All statistical analyses were performed using GraphPad Prism 8, R software and SPSS, and a p value of less than 0.05 was considered statistically significant.
Ethics statement
The studies involving human participants were reviewed and approved by the medical ethics committee of the Zhengzhou Central Hospital Affiliated to Zhengzhou University. Informed consents were obtained from all individual participants included in the study.
Results
Analysis of tumor-associated macrophages (TAMs) in glioma
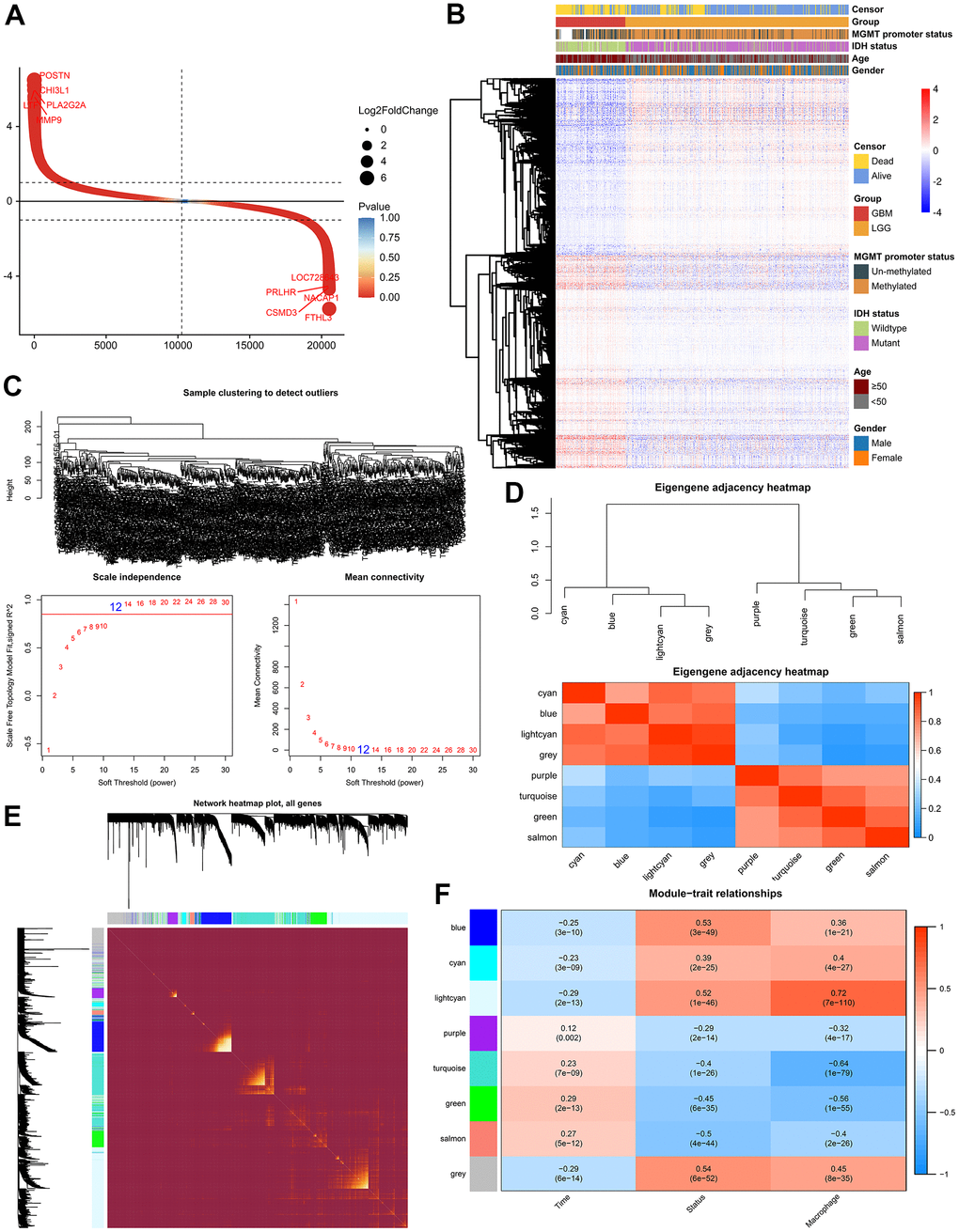
With the progress of glioma, tumor cells secrete a large number of chemokines to recruit immune cells. The increase in the proportion of macrophages in gliomas is related to the degree of malignancy [19]. Previous study had assessed the accuracy of 7 tools at estimating different immune cells from bulk RNA-seq data by developing a systematic approach for benchmarking such computational methods. Finally, the EPIC, MCPCOUNTER and XCELL methods were recommend to predict the level of tumor infiltrating macrophages [20]. Therefore, we analyzed the RNA-seq data from glioma patients in the CGGA mRNAseq-325, CGGA mRNAseq-693 and TCGA merged GBMLGG cohorts by these three methods to characterize the TAM in gliomas (Figure 1 and Supplementary Figure 1). The results showed that compared with LGG, the level of macrophages in GBM was significantly increased (Figure 1A, 1B and Supplementary Figure 1). In addition, the level of macrophages was significantly correlated with overall survival of gliomas (Figure 1C). In different WHO grade, age, MGMT status and IDH mutation status group, TAMs also showed significant differences. However, there was no difference in gender group (Supplementary Figure 2).
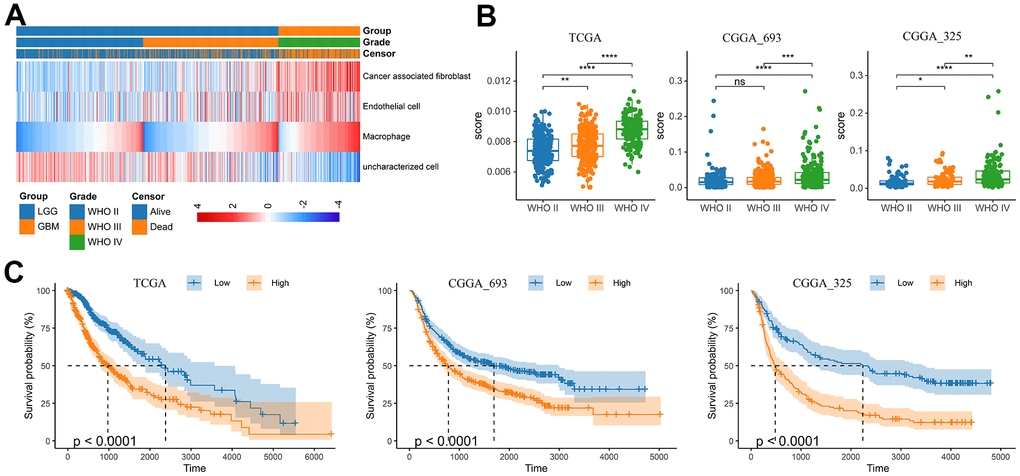
Figure 1. Analysis of tumor infiltrating macrophages (TAM) in glioma. (A) Heatmap was drawn to depict the TAM in glioma from the TCGA merged GBMLGG cohort. (B) TAMs were increased in GBM in the TCGA merged GBMLGG, CGGA mRNAseq-693 and CGGA mRNAseq-325 cohorts, respectively. (C) Kaplan-Meier overall survival curves displayed that increased TAM was related to the poor prognosis and lower survival rate of glioma. *, P< 0.05; **, P< 0.01; ***, P< 0.001, ****, P< 0.0001.
Construction of the risk score signature
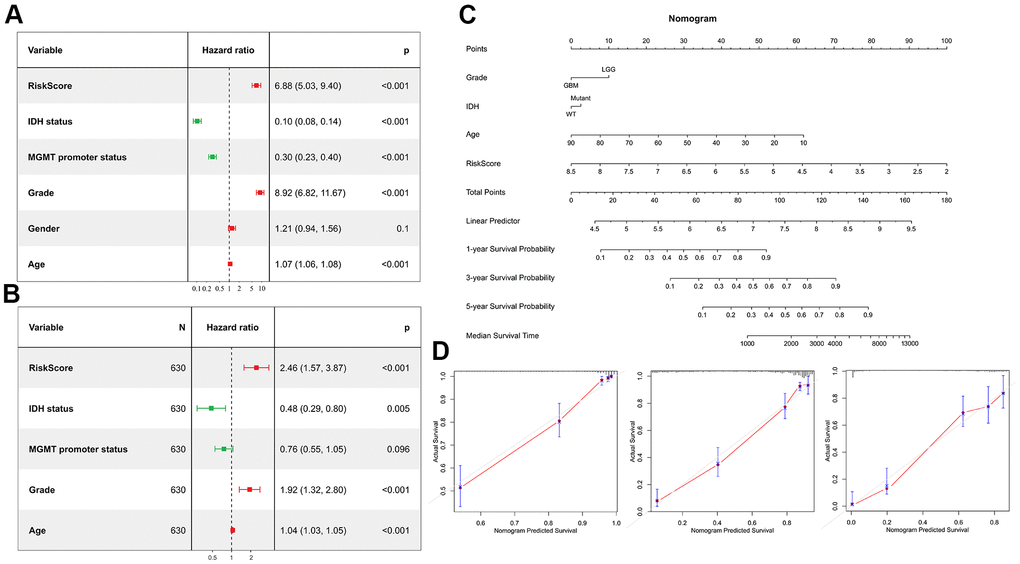
As shown by the Univariate Cox regression analysis, all the 16 hub genes were significantly associated with prognosis in the TCGA dataset (Figure 4A). Subsequently, we performed the least absolute shrinkage and selection operator (LASSO) Cox regression algorithm to analyze the 16 hub genes in the TCGA dataset. Nine prognostic-related genes (MYL12A, MSN, S100A4, CHI3L1, PLAUR, EMP3, CASP4, TIMP1 and CCDC109B) were screened out based on the minimum criteria in the TCGA dataset (Figure 4B, 4C). The results of Kaplan-Meier analyses displayed that the expression of the nine prognostic-related genes was significantly correlated with the overall survival of glioma patients in the TCGA dataset (Figure 4D). Therefore, these nine genes were finally selected to construct the risk score signature (Figure 5A, 5D, 5G). Kaplan-Meier survival analyses showed that high risk score was related to the poor prognosis and lower survival rate of glioma both in the CGGA mRNAseq-325, CGGA mRNAseq-693 and TCGA merged GBMLGG cohorts (Figure 5B, 5E, 5H). In order to assess the sensitivity and specificity of risk score in predicting the 1-, 3- and 5-year survival of glioma patients, we conducted ROC curve analyses in three cohorts and found that the predictive accuracy of the risk score was very high (Figure 5C, 5F, 5I).
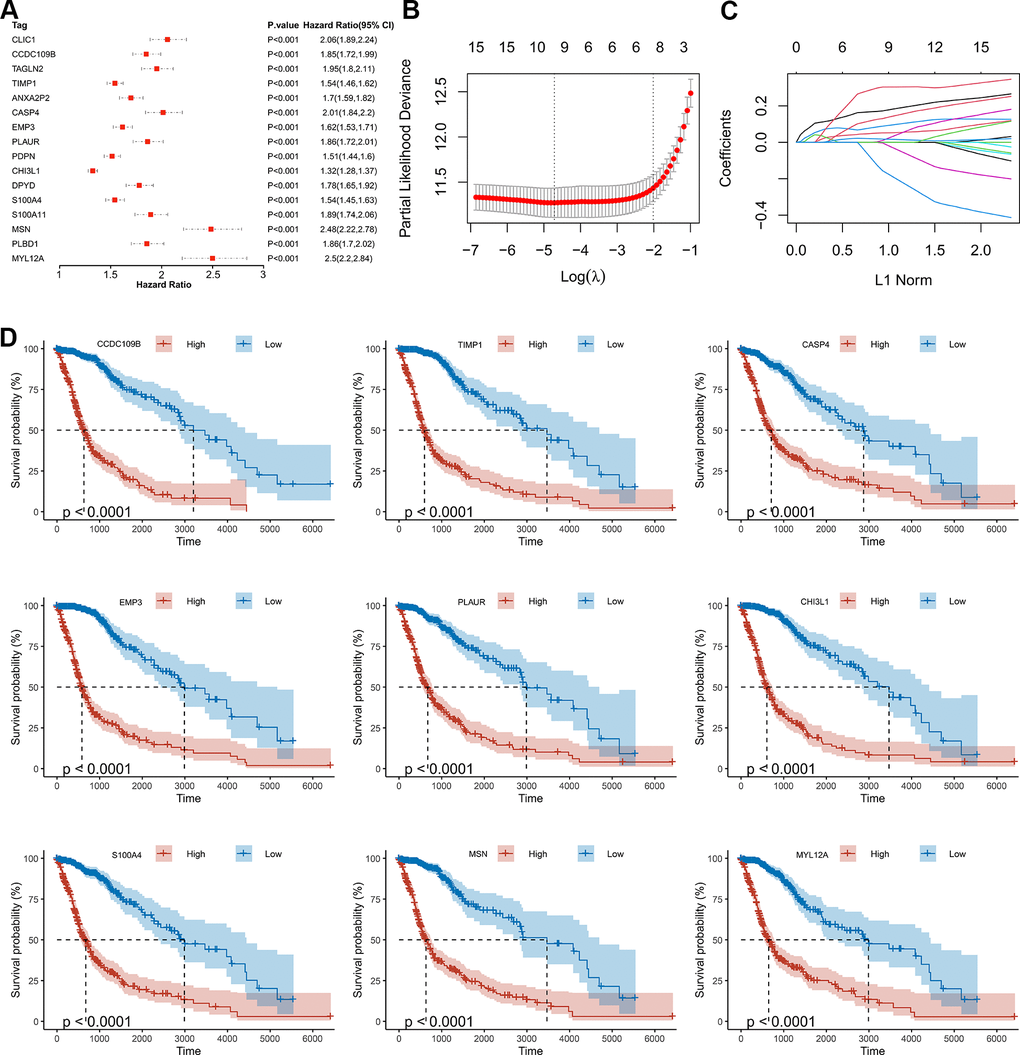
Figure 4. Screening of the prognostic genes in the TCGA dataset. (A) Univariate Cox regression analysis of 16 hub genes in the TCGA dataset. (B) Partial likelihood deviance of different numbers of variables revealed by the LASSO regression model. (C) LASSO coefficient profiles of the selected hub genes. (D) Kaplan-Meier curves displayed highly expressed genes screened out by LASSO were significantly related to poor prognosis in the TCGA dataset.
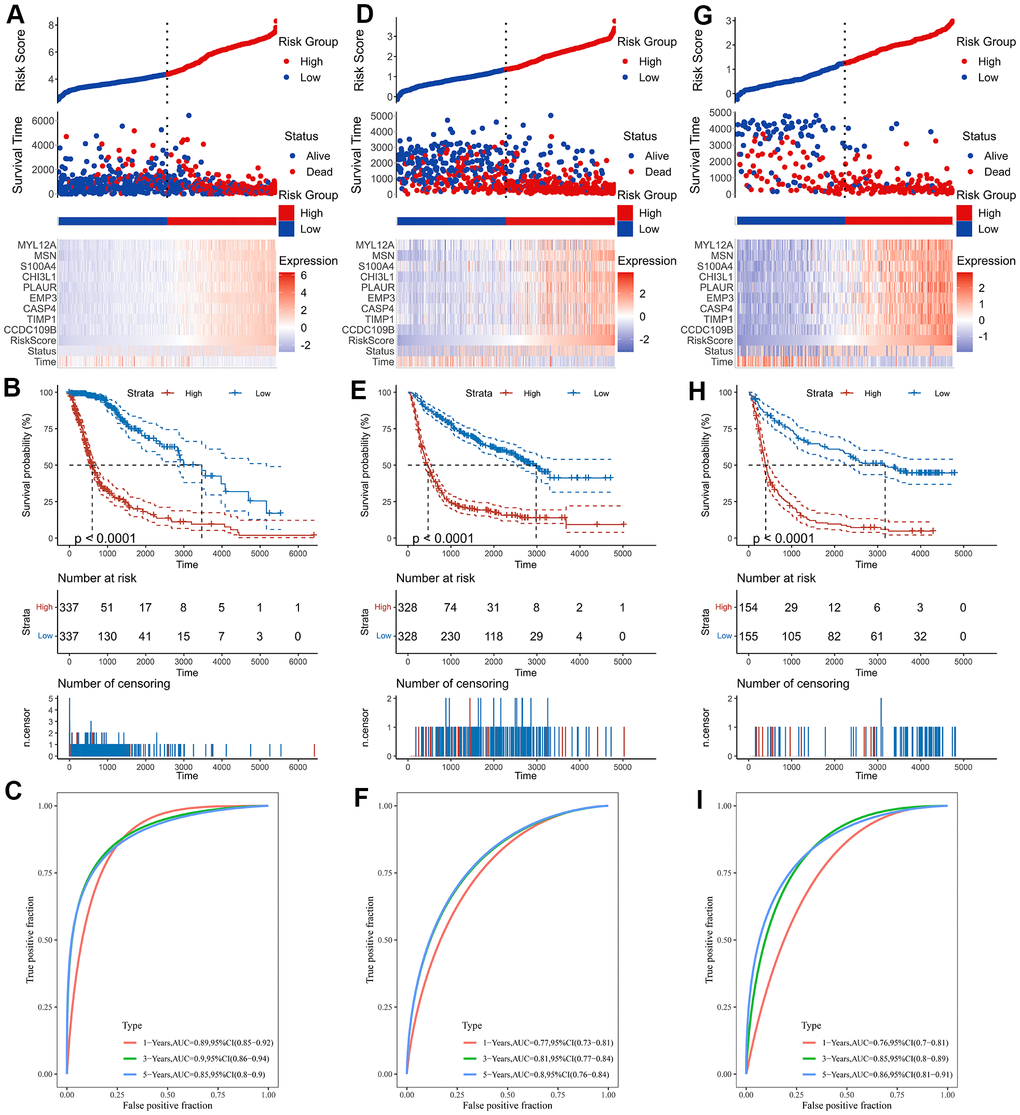
Figure 5. Construction of the risk score signature. (A) Risk signature in the TCGA cohort. (B) Kaplan-Meier analysis according to risk score in the TCGA cohort. (C) The ROC curves depicting the sensitivity and specificity of risk score in predicting the 1-, 3- and 5-year survival in the TCGA cohort. (D) Risk signature in the CGGA mRNAseq-693 cohort. (E) Kaplan-Meier analysis according to risk score in the CGGA mRNAseq-693 cohort. (F) The ROC curves depicting the sensitivity and specificity of risk score in predicting the 1-, 3- and 5-year survival in the CGGA mRNAseq-693 cohort. (G) Risk signature in the CGGA mRNAseq-325 cohort. (H) Kaplan-Meier analysis according to risk score in the CGGA mRNAseq-325 cohort. (I) The ROC curves depicting the sensitivity and specificity of risk score in predicting the 1-, 3- and 5-year survival in the CGGA mRNAseq-325 cohort.
The relationship between risk score and TAM
To better understand the relationship between risk score and TAM, we analyzed the distribution of the survival status, age, WHO grade, risk score and TAM of glioma patients (Figure 6A). The risk scores of gliomas in GBM were significantly higher than the corresponding LGG subtype (Figure 6B). In addition, the infiltration level of macrophages was increased in the high-risk group (Figure 6C). The risk score also was closely related to age, IDH mutation status and MGMT status (Supplementary Figure 3). In addition to the risk score, all nine genes that constructed the risk signature were significantly associated with the TAM in glioma (Figure 6D).
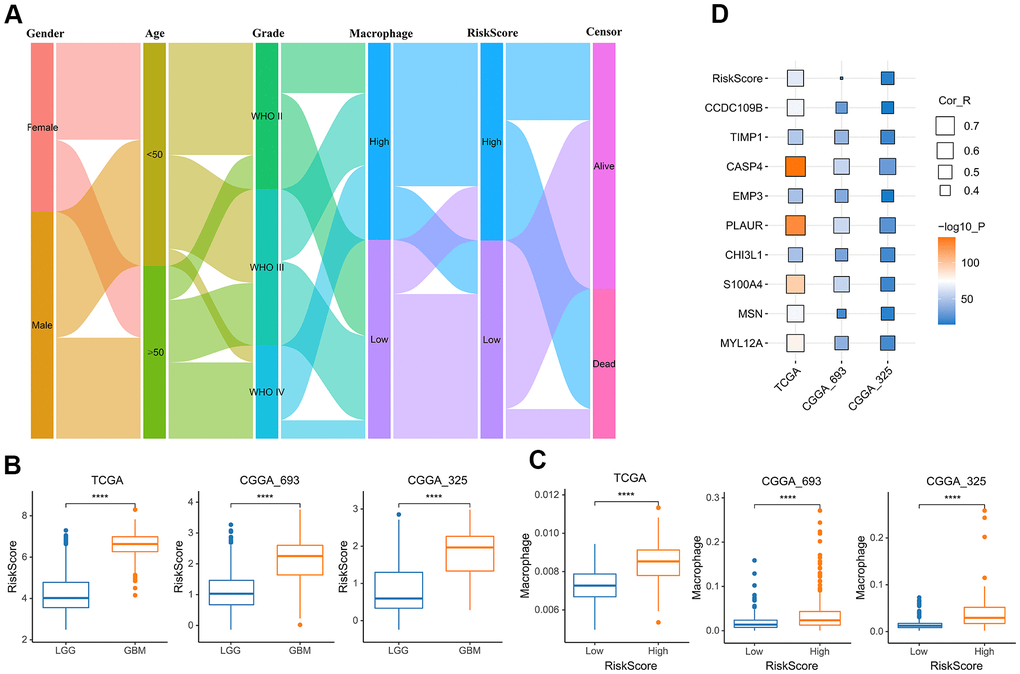
Figure 6. The relationship between risk score and TAM. (A) Sankey Diagram displayed the distribution of the survival status, age, WHO grade, risk score and TAM of glioma patients in the merged GBMLGG cohort. (B) Boxplot showed the risk scores of GBM were higher than those of LGG in the cohort of TCGA merged GBMLGG, CGGA mRNAseq-693 and CGGA mRNAseq-325, respectively. (C) Boxplot showed the TAM of high-risk group was higher than that of low-risk group in the cohort of TCGA merged GBMLGG, CGGA mRNAseq-693 and CGGA mRNAseq-325, respectively. (D) Correlation analysis showed that the risk score and nine prognostic genes were significantly related to TAM in the cohort of TCGA merged GBMLGG, CGGA mRNAseq-693 and CGGA mRNAseq-325, respectively. *, P< 0.05; **, P< 0.01; ***, P< 0.001, ****, P< 0.0001.
Immune microenvironment analysis of glioma
After that, we investigated whether the risk score was associated with tumor immune microenvironment in the TCGA dataset (Figure 7). The results of correlation analysis showed that risk scores were positively correlated with immune scores, stromal scores and ESTIMATE scores both in the CGGA mRNAseq-325, CGGA mRNAseq-693 and TCGA merged GBMLGG cohorts (Figure 7A–7C). And the immune scores, stromal scores and ESTIMATE scores of the high-risk group were higher than those of the low-risk group (Figure 7D–7F).
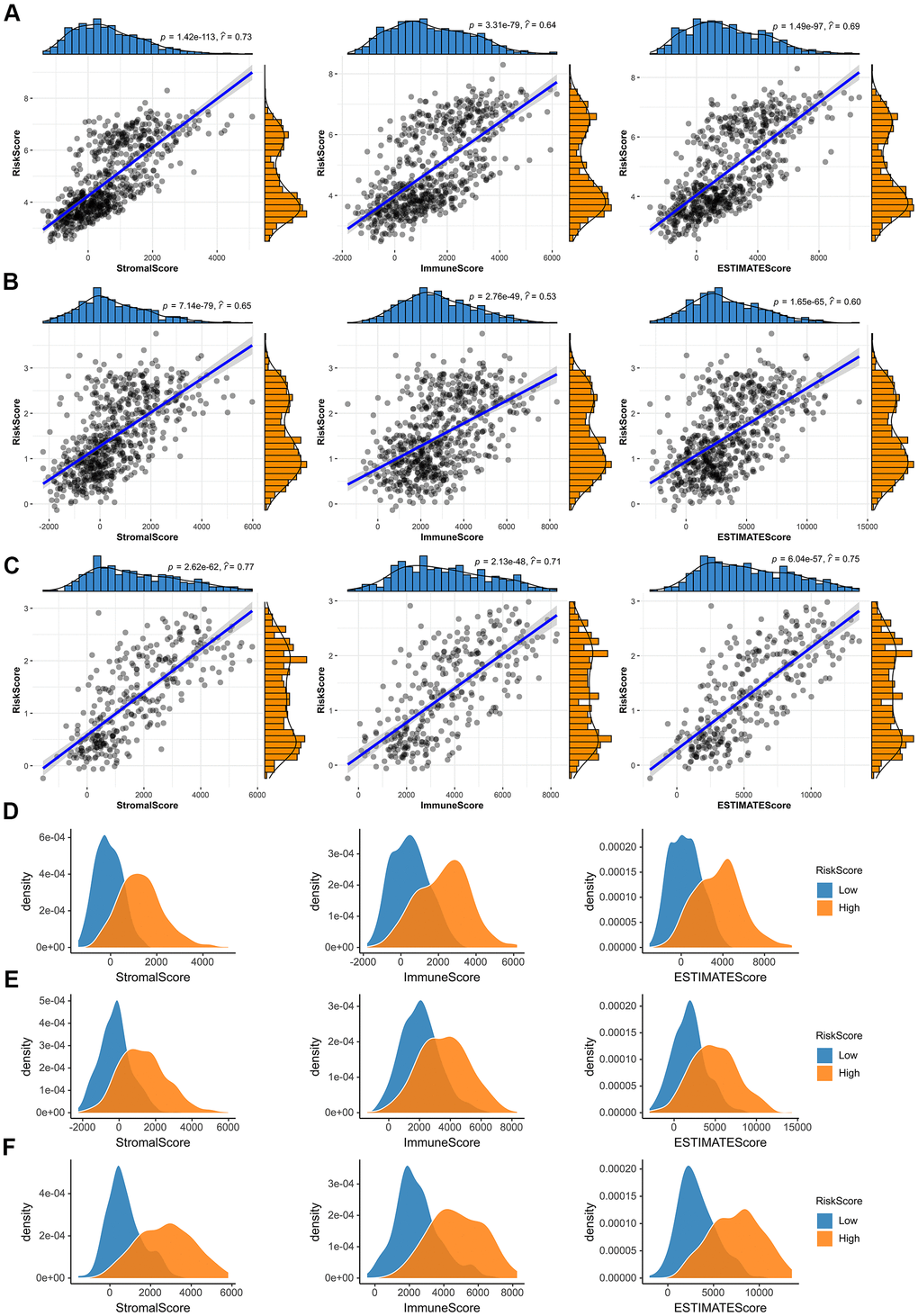
Figure 7. Immune microenvironment analysis of glioma. (A–C) In the TCGA merged GBMLGG, CGGA mRNAseq-693 and CGGA mRNAseq-325 cohorts, the risk score was significantly correlated with the immune scores, stromal scores and ESTIMATE scores, respectively. (D–F) Compared with low-risk group, the immune scores, stromal scores and ESTIMATE scores of high-risk group were higher in the TCGA merged GBMLGG, CGGA mRNAseq-693 and CGGA mRNAseq-325 cohorts.
Validation the expression of the prognostic genes
Immunohistochemical staining were performed to validate the expression of the genes that constructed the risk signature in glioma. Similar to the expression pattern of CGGA mRNAseq-325, CGGA mRNAseq-693 and TCGA merged GBMLGG cohorts, CHI3L1, MSN and TIMP1 were expressed higher in high-grade gliomas than in low-grade gliomas (Figure 9).
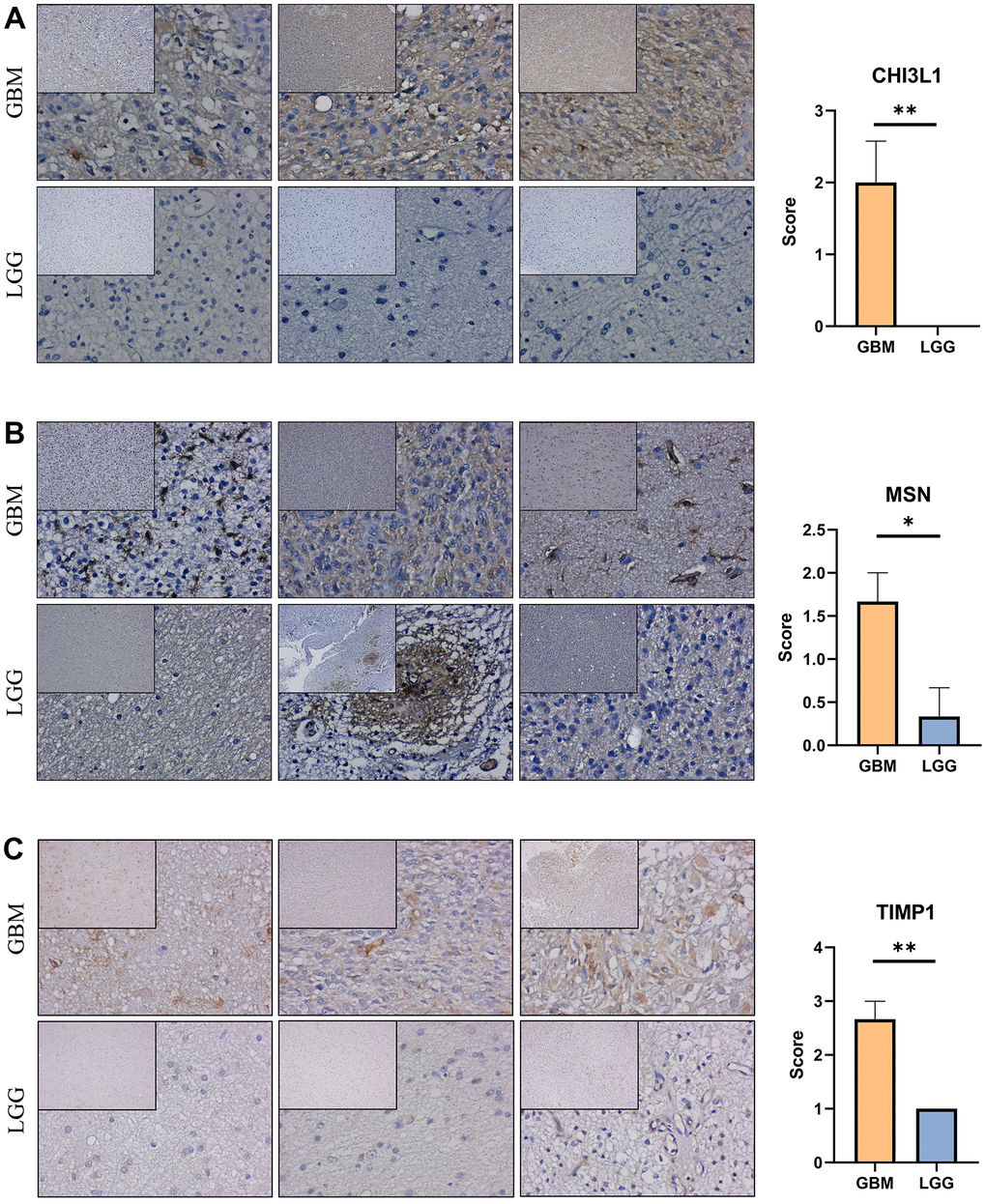
Figure 9. Expression verification of the prognostic genes. (A–C) Immunohistochemical staining analysis of the protein levels of CHI3L1, MSN and TIMP1 between the low-grade and high-grade gliomas. *, P< 0.05; **, P< 0.01; ***, P< 0.001.
TIMP1 affects migration and proliferation of glioma cells
To fully determine the effect of TIMP1 on glioma cells, we knocked down TIMP1 in LN229 cells by transfecting specific siRNA (Figure 10A, 10B). Knockdown TIMP1 significantly inhibited the migration of LN229 cells (Figure 10C). In addition, EdU assay showed that TIMP1 could significantly affect the proliferation of LN229 cells (Figure 10D).
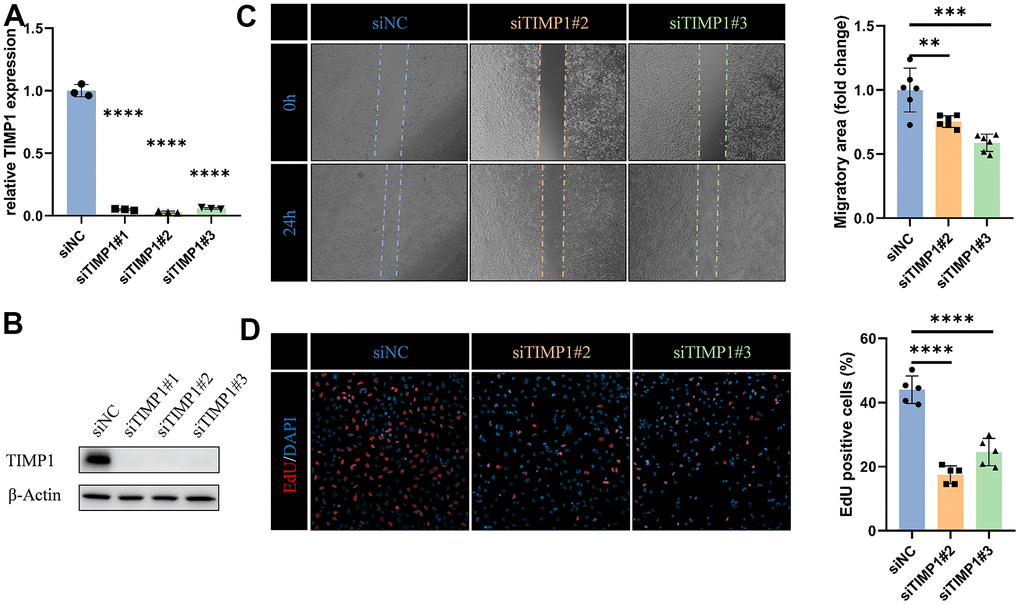
Figure 10. TIMP1 affects migration and proliferation of glioma cells. (A) qRT-PCR and (B) western blot analysis of TIMP1 knockdown efficiency in LN229 cells. (C) Representative images and statistical analysis of cell migration assay in control and TIMP1-deficient endothelial cells at the indicated times. (D) Representative images and statistical analysis of EdU assay in control and TIMP1-deficient LN229 cells. *, P< 0.05; **, P< 0.01; ***, P< 0.001; **** P< 0.0001.
Discussion
Glioma is considered to be one of the most devastating tumors in adults [21]. Currently, maximal safe surgical resection followed by radiotherapy with concurrent temozolomide chemotherapy is still the standard treatment of GBM [22]. However, the median survival of GBM is poor, not exceeding 16 months [5]. In the present study, we built a survival nomogram prediction model, incorporating grade, age, IDH status, and risk score into the model to improve the prediction accuracy of the model.
Inflammation in the tumor microenvironment is an important manifestation of malignant tumors. Chronic inflammation has been proved to be closely related with carcinogenesis. The inflammation-enriched tumor microenvironment has been shown to be responsible for the progression of developing tumors into highly malignant neoplasms, including GBM. Macrophages, as the main inflammatory cells in the tumor microenvironment, regulate the activity of signaling pathways together with glioma cells, and ultimately promote cancer progression, tumor cell migration and invasion, and immunosuppression [23]. Here, we revealed the relationship between TAM and malignancy of glioma, demonstrated the value of TAM related signature in predicting the prognosis of glioma.
Based on superior computational methods and multiple cohorts, we screened core genes and constructed prediction models. The results provided evidence for TAMs-related signature as a valuable prognostic biomarker in glioma. Nine positive prognostic genes (MYL12A, MSN, S100A4, CHI3L1, PLAUR, EMP3, CASP4, TIMP1 and CCDC109B) were included in the prediction model. CHI3L1 is an abundant glycopolymer, which is synthesized and secreted by macrophages and various cells. CHI3L1 has been shown to be associated in multiple cancers [24]. Lately, Chen et al. showed that CHI3L1 promoted macrophage-mediated immune suppression by forming complexes with galectin 3 or galectin 3-binding protein [25]. As a member of the ERM family, MSN localized to filopodia and other membranous protrusions that connects the actin-based cytoskeleton to plasma membranes [26]. In glioblastoma, MSN could increase CD44 expression driven by the Wnt/β-catenin signaling pathway [27]. MYL12A was proved to be involved in DNA damage repair and p53-driven apoptosis [28]. TIMP1 is member of natural inhibitors of the matrix metalloproteinases, which has the effect of controlling the polarization of Natural Killer cells induced by the tumor-related cytokine TGFβ [29]. CCDC109B was involved in the activation of cell death pathway. Xu et al. showed that it plays an important role in mediating the migration and invasion of glioma cells induced by HIF1α [29].
As mentioned above, in previous studies, the role of some prognostic genes in glioma or other cancers have been reported. But we uncovered their potential role on tumor-infiltrating immune cells, which together with cancer cells constitute the tumor microenvironment. Due to the limitations of the research on the signaling pathways involved in target prognostic genes and their realistic role in tumor-associated macrophage infiltration, further researches are needed to explore the molecular mechanism.
Conclusions
Overall, we revealed the relationship between TAM and malignancy of glioma, demonstrated the value of TAM related signature in predicting the prognosis of glioma, and provided potential targeted therapy for glioma.
Supplementary Materials
Author Contributions
Lin-jian Wang: Conceptualization, Methodology, Funding acquisition; Yimeng Xue: Data curation, Writing - Original Draft; Yongli Lou: Writing - Review and Editing.
Conflicts of Interest
All authors state that they have no conflicts of interest.
Funding
This work was supported by the National Natural Science Foundation of China (82101401).
References
- 1. Thakkar JP, Dolecek TA, Horbinski C, Ostrom QT, Lightner DD, Barnholtz-Sloan JS, Villano JL. Epidemiologic and molecular prognostic review of glioblastoma. Cancer Epidemiol Biomarkers Prev. 2014; 23:1985–96. https://doi.org/10.1158/1055-9965.EPI-14-0275 [PubMed]
- 2. Lefranc F, Le Rhun E, Kiss R, Weller M. Glioblastoma quo vadis: Will migration and invasiveness reemerge as therapeutic targets? Cancer Treat Rev. 2018; 68:145–54. https://doi.org/10.1016/j.ctrv.2018.06.017 [PubMed]
- 3. Locarno CV, Simonelli M, Carenza C, Capucetti A, Stanzani E, Lorenzi E, Persico P, Della Bella S, Passoni L, Mavilio D, Bonecchi R, Locati M, Savino B. Role of myeloid cells in the immunosuppressive microenvironment in gliomas. Immunobiology. 2020; 225:151853. https://doi.org/10.1016/j.imbio.2019.10.002 [PubMed]
- 4. Ostrom QT, Cioffi G, Gittleman H, Patil N, Waite K, Kruchko C, Barnholtz-Sloan JS. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2012-2016. Neuro Oncol. 2019; 21:v1–100. https://doi.org/10.1093/neuonc/noz150 [PubMed]
- 5. Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC, Ludwin SK, Allgeier A, Fisher B, Belanger K, Hau P, Brandes AA, Gijtenbeek J, et al, and European Organisation for Research and Treatment of Cancer Brain Tumour and Radiation Oncology Groups, and National Cancer Institute of Canada Clinical Trials Group. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009; 10:459–66. https://doi.org/10.1016/S1470-2045(09)70025-7 [PubMed]
- 6. Wu J, Chen Z, Wickström SL, Gao J, He X, Jing X, Wu J, Du Q, Yang M, Chen Y, Zhang D, Yin X, Guo Z, et al. Interleukin-33 is a Novel Immunosuppressor that Protects Cancer Cells from TIL Killing by a Macrophage-Mediated Shedding Mechanism. Adv Sci (Weinh). 2021; 8:e2101029. https://doi.org/10.1002/advs.202101029 [PubMed]
- 7. Watters JJ, Schartner JM, Badie B. Microglia function in brain tumors. J Neurosci Res. 2005; 81:447–55. https://doi.org/10.1002/jnr.20485 [PubMed]
- 8. Sun X, He X, Zhang Y, Hosaka K, Andersson P, Wu J, Wu J, Jing X, Du Q, Hui X, Ding B, Guo Z, Hong A, et al. Inflammatory cell-derived CXCL3 promotes pancreatic cancer metastasis through a novel myofibroblast-hijacked cancer escape mechanism. Gut. 2022; 71:129–47. https://doi.org/10.1136/gutjnl-2020-322744 [PubMed]
- 9. Colegio OR, Chu NQ, Szabo AL, Chu T, Rhebergen AM, Jairam V, Cyrus N, Brokowski CE, Eisenbarth SC, Phillips GM, Cline GW, Phillips AJ, Medzhitov R. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature. 2014; 513:559–63. https://doi.org/10.1038/nature13490 [PubMed]
- 10. Wynn TA, Chawla A, Pollard JW. Macrophage biology in development, homeostasis and disease. Nature. 2013; 496:445–55. https://doi.org/10.1038/nature12034 [PubMed]
- 11. Zheng Y, Bao J, Zhao Q, Zhou T, Sun X. A Spatio-Temporal Model of Macrophage-Mediated Drug Resistance in Glioma Immunotherapy. Mol Cancer Ther. 2018; 17:814–24. https://doi.org/10.1158/1535-7163.MCT-17-0634 [PubMed]
- 12. Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, Antipin Y, Reva B, Goldberg AP, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012; 2:401–4. https://doi.org/10.1158/2159-8290.CD-12-0095 [PubMed]
- 13. Zhao Z, Meng F, Wang W, Wang Z, Zhang C, Jiang T. Comprehensive RNA-seq transcriptomic profiling in the malignant progression of gliomas. Sci Data. 2017; 4:170024. https://doi.org/10.1038/sdata.2017.24 [PubMed]
- 14. Li T, Fu J, Zeng Z, Cohen D, Li J, Chen Q, Li B, Liu XS. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020; 48:W509–14. https://doi.org/10.1093/nar/gkaa407 [PubMed]
- 15. Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015; 43:e47. https://doi.org/10.1093/nar/gkv007 [PubMed]
- 16. Bu D, Luo H, Huo P, Wang Z, Zhang S, He Z, Wu Y, Zhao L, Liu J, Guo J, Fang S, Cao W, Yi L, et al. KOBAS-i: intelligent prioritization and exploratory visualization of biological functions for gene enrichment analysis. Nucleic Acids Res. 2021; 49:W317–25. https://doi.org/10.1093/nar/gkab447 [PubMed]
- 17. Sun H, Wang S. Penalized logistic regression for high-dimensional DNA methylation data with case-control studies. Bioinformatics. 2012; 28:1368–75. https://doi.org/10.1093/bioinformatics/bts145 [PubMed]
- 18. Pak K, Oh SO, Goh TS, Heo HJ, Han ME, Jeong DC, Lee CS, Sun H, Kang J, Choi S, Lee S, Kwon EJ, Kang JW, Kim YH. A User-Friendly, Web-Based Integrative Tool (ESurv) for Survival Analysis: Development and Validation Study. J Med Internet Res. 2020; 22:e16084. https://doi.org/10.2196/16084 [PubMed]
- 19. Yi L, Xiao H, Xu M, Ye X, Hu J, Li F, Li M, Luo C, Yu S, Bian X, Feng H. Glioma-initiating cells: a predominant role in microglia/macrophages tropism to glioma. J Neuroimmunol. 2011; 232:75–82. https://doi.org/10.1016/j.jneuroim.2010.10.011 [PubMed]
- 20. Sturm G, Finotello F, Petitprez F, Zhang JD, Baumbach J, Fridman WH, List M, Aneichyk T. Comprehensive evaluation of transcriptome-based cell-type quantification methods for immuno-oncology. Bioinformatics. 2019; 35:i436–45. https://doi.org/10.1093/bioinformatics/btz363 [PubMed]
- 21. Ostrom QT, Gittleman H, Liao P, Vecchione-Koval T, Wolinsky Y, Kruchko C, Barnholtz-Sloan JS. CBTRUS Statistical Report: Primary brain and other central nervous system tumors diagnosed in the United States in 2010-2014. Neuro Oncol. 2017; 19:v1–88. https://doi.org/10.1093/neuonc/nox158 [PubMed]
- 22. Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, et al, and European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups, and National Cancer Institute of Canada Clinical Trials Group. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005; 352:987–96. https://doi.org/10.1056/NEJMoa043330 [PubMed]
- 23. Basheer AS, Abas F, Othman I, Naidu R. Role of Inflammatory Mediators, Macrophages, and Neutrophils in Glioma Maintenance and Progression: Mechanistic Understanding and Potential Therapeutic Applications. Cancers (Basel). 2021; 13:4226. https://doi.org/10.3390/cancers13164226 [PubMed]
- 24. Zhao T, Su Z, Li Y, Zhang X, You Q. Chitinase-3 like-protein-1 function and its role in diseases. Signal Transduct Target Ther. 2020; 5:201. https://doi.org/10.1038/s41392-020-00303-7 [PubMed]
- 25. Chen A, Jiang Y, Li Z, Wu L, Santiago U, Zou H, Cai C, Sharma V, Guan Y, McCarl LH, Ma J, Wu YL, Michel J, et al. Chitinase-3-like 1 protein complexes modulate macrophage-mediated immune suppression in glioblastoma. J Clin Invest. 2021; 131:e147552. https://doi.org/10.1172/JCI147552 [PubMed]
- 26. Derouiche A, Geiger KD. Perspectives for Ezrin and Radixin in Astrocytes: Kinases, Functions and Pathology. Int J Mol Sci. 2019; 20:3776. https://doi.org/10.3390/ijms20153776 [PubMed]
- 27. Zhu X, Morales FC, Agarwal NK, Dogruluk T, Gagea M, Georgescu MM. Moesin is a glioma progression marker that induces proliferation and Wnt/β-catenin pathway activation via interaction with CD44. Cancer Res. 2013; 73:1142–55. https://doi.org/10.1158/0008-5472.CAN-12-1040 [PubMed]
- 28. Park I, Han C, Jin S, Lee B, Choi H, Kwon JT, Kim D, Kim J, Lifirsu E, Park WJ, Park ZY, Kim DH, Cho C. Myosin regulatory light chains are required to maintain the stability of myosin II and cellular integrity. Biochem J. 2011; 434:171–80. https://doi.org/10.1042/BJ20101473 [PubMed]
- 29. Albini A, Gallazzi M, Palano MT, Carlini V, Ricotta R, Bruno A, Stetler-Stevenson WG, Noonan DM. TIMP1 and TIMP2 Downregulate TGFβ Induced Decidual-like Phenotype in Natural Killer Cells. Cancers (Basel). 2021; 13:4955. https://doi.org/10.3390/cancers13194955 [PubMed]