Introduction
Locally recurrent rectal cancer (LRRC) is defined as the recurrence of rectal cancer (READ) only within the pelvis after radical resection [1]. LRRC is associated with symptoms of significant morbidity, such as rectal bleeding, bowel obstruction, chronic pain, fistulas, malodorous tumor discharge, tenesmus, and pelvic sepsis [2, 3]. LRRC generally occurs within 2-3 years after the initial surgery [4]. Non-recurrent rectal cancer (NRRC) is defined as READ that has not recurred within 3 years after radical surgery. Historically, the introduction of total mesenteric excision (TME) has greatly reduced the local recurrence rate from 30% to 10% [3]. Combined with chemoradiotherapy, the local control (LC) rate for clinically localized READ ranges between 92% and 96% [5, 6]. Up to 50% of recurrent READ patients have synchronously diagnosed distant metastases [7, 8]. Meanwhile, nearly half of the patients with recurrence of READ are limited in the pelvis, which is defined as LRRC [8–10].
The causes and mechanism of LRRC have become a focus of current clinical medical research, highlighting the need for prevention of LRRC. Risk factors for LRRC occurrence include anastomotic leak, incomplete resection, intraoperative tumor perforation, high-grade pathology, and lack of adjuvant chemoradiotherapy [11]. LRRC is associated with short-term mortality, high reoperation rate, and additive healthcare costs [12]. Without treatment, the life expectancy of LRRC patients is limited and the quality of life is usually poor. Three major strategies for reducing the recurrence of READ are wider surgical coverage and neoadjuvant radiotherapy and neoadjuvant chemotherapy [13, 14]. However, neoadjuvant chemoradiation may significantly increase morbidity. Since LRRC portends a significantly worse oncologic outcome and quality of life, remarkable advances in surgical techniques and adjuvant chemoradiotherapy are improving the overall survival (OS) of LRRC. Although modern surgical techniques and multimodal therapies have proved to be effective, LRRC shows a significant treatment challenge. Some research showed a 5-year survival rate of 35% to 50% after surgery for LRRC [15, 16]. Chemotherapy alone can prolong OS to12 to 15 months, but it is not curable [8]. About 40% of LRRC patients have an opportunity for surgical treatment, but performing the operation can be a challenging option under such a situation [17]. Reoperation is very difficult and often needs to be combined with organ resection. The incidence of perioperative complications is specifically high, which is rare in domestic medical units. Therefore, how to prevent LRRC is the key to improve the survival and life quality of READ patients.
The long-term outcome of surgical treatment mainly depends on the clear margin resection [16]. We speculated that the molecular mechanism of LRRC may also be related to the occurrence and prognosis of LRRC. The signal pathway related to distant metastasis of READ has been studied in detail as a hot spot. However, the molecular mechanism of LRRC has not been reported yet. Whole-exome sequencing (WES) can reveal mutations within exon coding regions. This study was based on WES to explore mutations associated with LRRC. 5 trios of LRRC and 3 pairs of NRRC samples were sequenced by WES. The data was analyzed systematically by bioinformatics. Gene Expression Profiling Interactive Analysis (GEPIA) and Human Protein Atlas (HPA) were performed to validate the candidate genes. Immunohistochemistry was used to detect the protein expression of candidate genes in LRRC, PRC, and NRRC specimens. Our findings provided a basis for predicting LRRC and developing new therapeutic agents.
Materials and Methods
Patients and specimens
The present study was reviewed and approved by the Ethics Committee of Huashan Hospital affiliated to Fudan University. We collected 21 samples, including 15 tissue samples from five LRRC patients (PRC, adjacent normal rectal tissue, and LRRC) and three NRRC patients (NRRC and adjacent normal rectal tissue) from May 2019 to November 2019 at Huashan Hospital, Fudan University. HE-stained sections from each sample were subjected to an independent pathology review to confirm that the tumor specimens were histologically consistent with LRRC and that the adjacent tissue specimens contained no tumor cells. All participants provided written informed consent for genetic analysis.
Whole-exome sequencing
For every individual, the genomic DNA of cells from one or two regions of the PRC, LRRC, and matched normal rectal tissue samples were sequenced. DNA was extracted from formalin-fixed and paraffin-embedded (FFPE) tissue blocks using MagPure FFPE DNA/RNA LQ Kit (Magen, Guangzhou, China). The DNA was then subjected to an additional quality and quantity evaluation step by utilizing NanoDrop ultra-micro spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, USA) and Qubit 1X dsDNA HS Assay Kit (Thermo Fisher Scientific, Wilmington, DE, USA). Exome-coding DNA was captured with Agilent SSEL XT HS Human All Exon V6 (Agilent Technologies, Santa Clara, CA, USA), and the libraries were sequenced on an Illumina NovaSeq Platform (Illumina, San Diego, CA, USA), which produced 350-bp paired-end reads.
Sequencing data analysis
Raw sequencing data were filtered using the Trim Galore program to remove low quality reads at both ends of the sequencing [18]. The FastQC package (http://www.bioinformatics.babraham.ac.uk/projects/fastqc) was used to assess the quality score distribution of the sequencing reads. Read sequences were mapped to the human reference genome (GRCH37/hg19) using Burrows-Wheeler Aligner (BWA, v.0.7.15) with the default parameters, and duplicates were marked and discarded using Picard (http://broadinstitute.github.io/picard). After alignment by BWA, the reads were subjected to local realignment and recalibration using the Genome Analysis Toolkit (GATK). Variants and genotypes calling were also performed using Genome Analysis Toolkit (GATK, v3.7).
Point substitutions in all tumor samples were counted and the proportion of each point substitution (C> A, C> G, C> T, T> A, T> C, and T> G) was calculated for both PRC and LRRC samples. The results were visualized by GraphPad Prism 8.
ANNOVAR was utilized to annotate all called variants. The SNVs of the adjacent normal rectal tissue in PRC, LRRC, and NRRC samples were removed for further analysis. Mutations were screened before being subjected to pathway enrichment analysis. The minor allele frequency (MAF) of variants was evaluated in the 1000 Genome Project (http://www.ncbi.nlm.nih.gov/variation/tools/1000genomes/), ExAC (http://exac.broadinstitute.org/), variants of MAF<1% and annotated “.” (No annotation information in the database) were retained. Subsequently, the pathogenicity of variants was predicted according to SIFT (http://sift.jcvi.org), Polyphen2 (http://genetics.bwh.harvard.edu/pph2) and FATHMM (http://fathmm.biocompute.org.uk/). Variants predicted by the three tools as “D (damaging)” were retained.
CNVs were identified using open source software called CNVkit (v0.9.7), a tool kit to infer and visualize the copy numbers from targeted DNA sequencing data. Genes of copy number over “3” were selected, the copy number of “2” meant no CNV, the copy number over “2” meant a copy gain in some paired chromosomes. The CNVs of the adjacent normal rectal tissue in PRC, LRRC, and NRRC samples were removed for further analysis.
Enrichment analyses of gene ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG)
The specific genes of PRC, LRRC, and NRRC samples were used to perform GO and KEGG enrichment analysis by DAVID 6.8 (https://david.ncifcrf.gov/). The results were considered as statistically significant if P value< 0.05. The top 15 enriched GO terms were visualized by BMKCloud (http://www.biocloud.net/), a free online platform for data analysis. The top 15 KEGG pathways were visualized by R ggplot2 package. GO functional analysis was divided into three parts: biological process (BP), cellular component (CC), and molecular function (MF).
Mutations of driver genes
To look for potential LRRC drivers, mutations in 155 TCGA rectal adenocarcinoma (https://www.cbioportal.org/) and 339 MSK READ patients were included in the analysis for comparison. Forty-eight genes with a high frequency of occurrence in both cases (TCGA Freq ≥ 10%; MSK Freq ≥ 5%) were selected. The mutation patterns of these genes in PRC and LRRC tissues were then compared.
mRNA and protein expression levels of hub genes
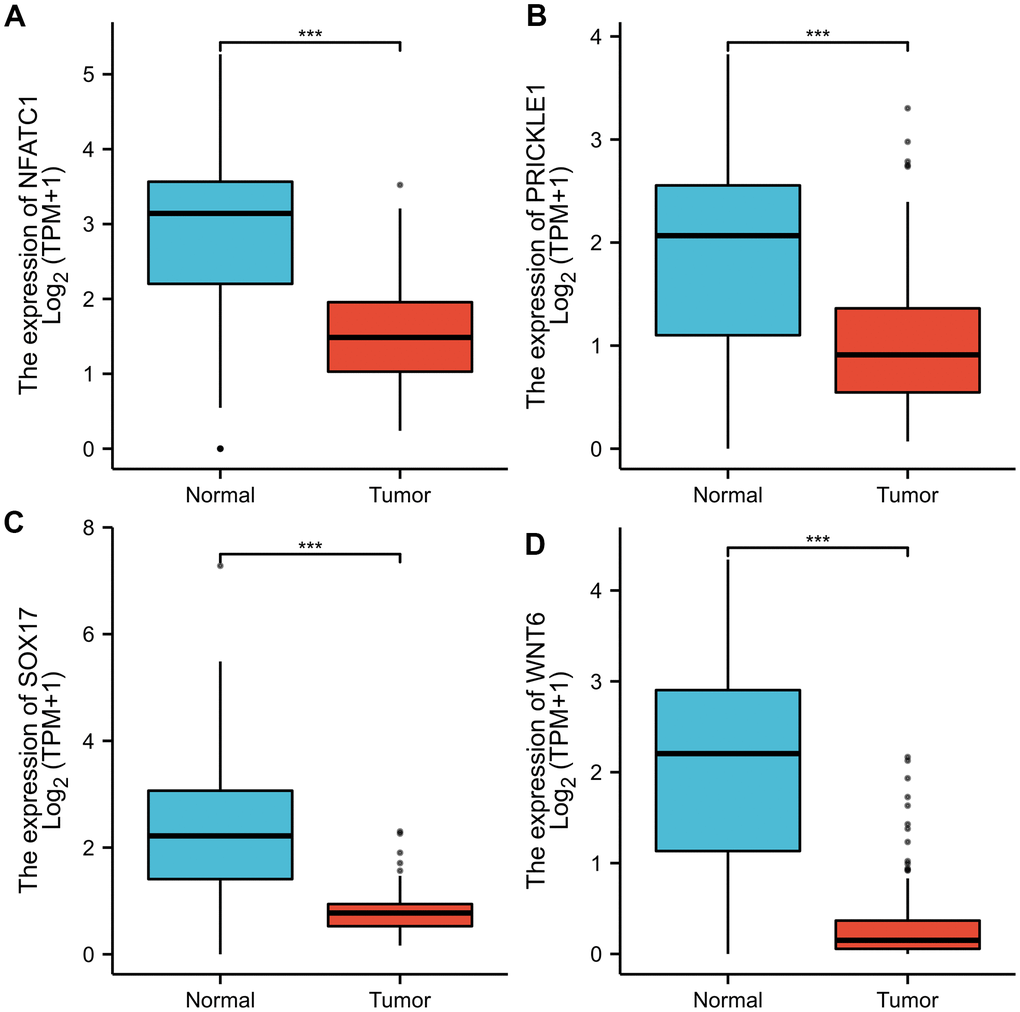
To validate the expression of the key DEGs, the Gene Expression Profiling Interactive Analysis (GEPIA) website (http://gepia2.cancer-pku.cn/#index) was applied to analyze the data of RNA sequencing expression based on thousands of samples from the GTEx project and TCGA [19]. The association between overall survival (OS) and the genes expressed in GC patients was determined using GEPIA. The lower and upper 50% of gene expression were set as the standard for analysis. Log-rank test results with P<0.05 were regarded as statistically significant. Besides, the GEPIA was employed to visualize the mRNA expression of hub genes in tumors and normal samples.
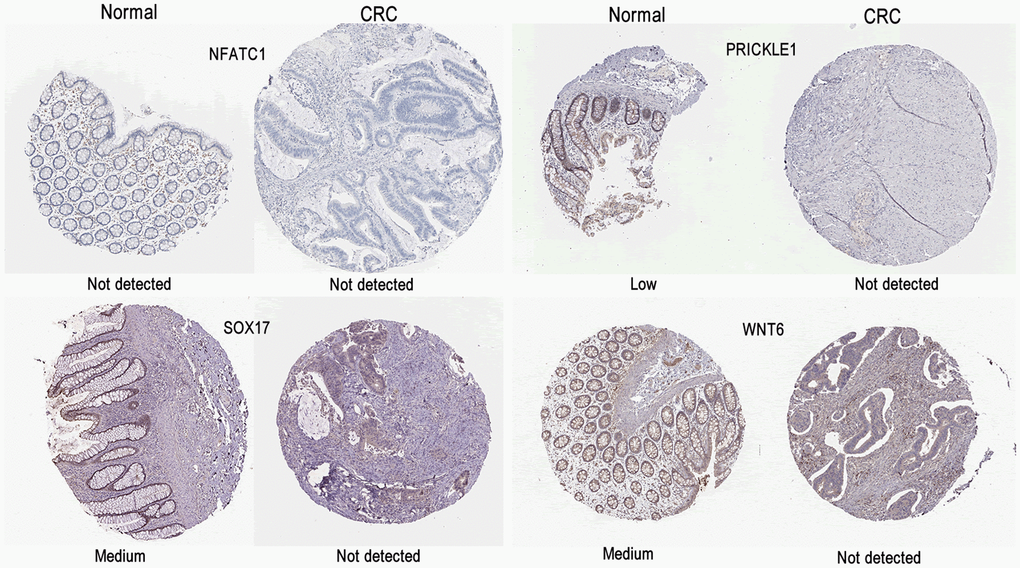
The Human Protein atlas (HPA) database (https://www.proteinatlas.org/) is a free online database that provides abundant transcriptome and proteome data on human normal or pathological tissues through RNA-sequence analysis and immunohistochemical analysis. In the present study, the protein expression and distribution of hub genes were investigated in GC tissues and compared with normal tissues in HPA [18].
Immunohistochemistry
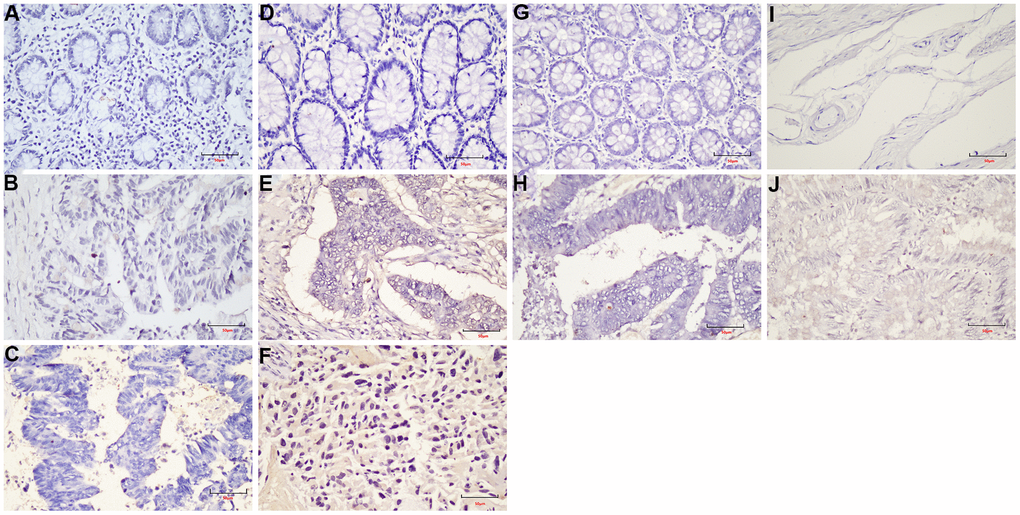
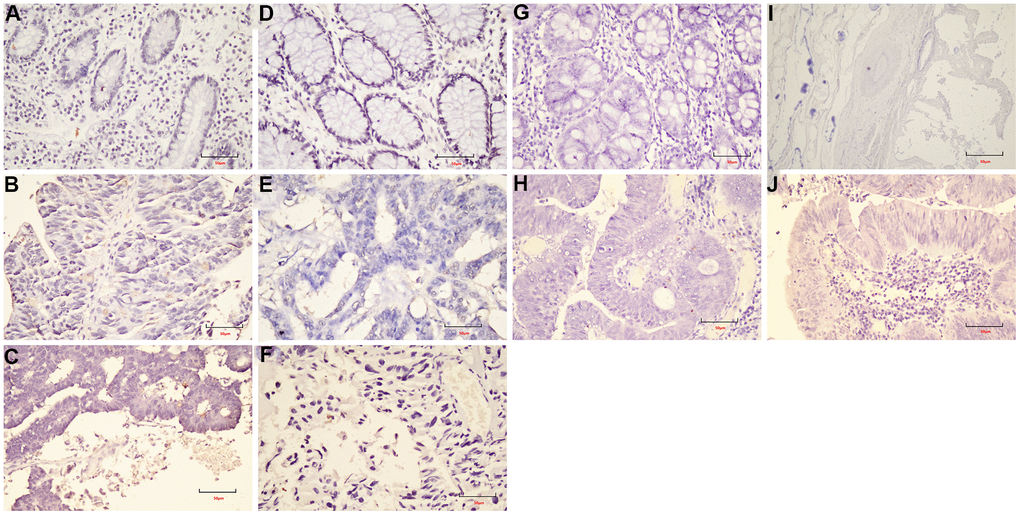
The Formalin-Fixed and Paraffin-Embedded (FFPE) samples were used in this study. 3-mm tumor sections were incubated with commercial rabbit polyclonal antibodies against LEF1 (ab137872, abcam) and cyclin D1 (ab16663, abcam) at 1/100 dilution overnight at 4° C. Then, the sections were conjugated with HRP-Sheep Anti-Rabbit IgG-HRP-Sheep Anti-Mouse IgG antibody (BOSTER BA1056, 1:500 dilution;) at room temperature for 2 h, then covered by 3, 3-diaminobenzidine (DAB) (Vector Laboratories, Burlingame, CA). Elivision plus kit for immunohistochemistry (IHC) (KIT-990, MXB) was used in this study.
Statistical analyses
Fisher’s exact test was used to assess differences in the count data. Top 15 significant GO terms of BP, MF, and CC were listed according to the p-value. Top 15 significant KEGG pathways were listed according to the p-value. P<0.05 was considered statistically significant.
Results
WES data
The clinical characteristics of the eight patients are listed in Supplementary Table 1. Age range is 45-71 years. The gender included 8 males. The tumor size included 4 patients (2cm), 3 patients (3cm), and 1 patient (4cm), and 1 patient (5cm). The pathologic 4 stage I, 3 stage II, and 1 stage III. The sequencing quality of the WES was analyzed and the raw data, Q30 (proportion of mapped reads) and mean depth of each sample are shown. As shown in Supplementary Table 2, all samples had Q30 ratio >90%, good sequencing quality, >90% of mapped reads, and an average sequencing depth > 200 x, which was sufficient to identify mutations.
Mutational signatures of LRRC and PRC tumors
All point substitutions were divided into six groups (C>A, C>G, C>T, T>A, T>C and T>G) according to the direction of the mutation. In total, 97862 and 47292 somatic substitutions were identified in all PRC and LRRC tissues by comparing them with the matched adjacent normal samples. The proportion of each group was different, but the proportion of all six mutant groups did not differ significantly between LRRC and matched PRC tissues (Figure 1).
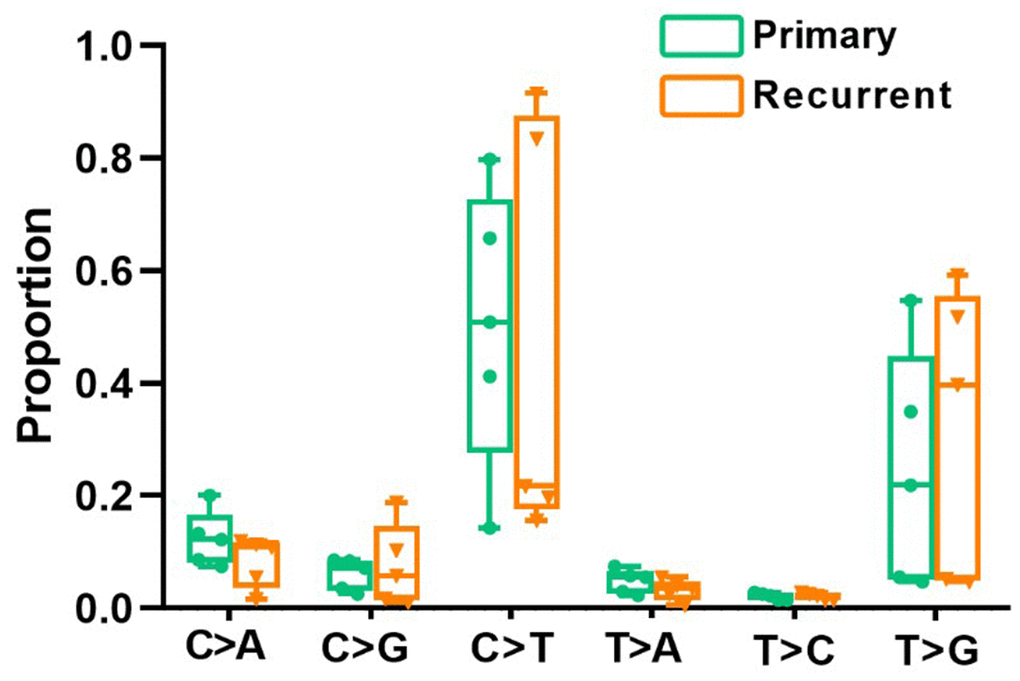
Figure 1. Divergent mutational features in LRRC and PRC samples of LRRC patients. Six mutational subtypes in LRRC and PRC tissues. LRRC tissues are presented in orange, and the PRC tissues are presented in cyan.
The common variations in LRRC and PRC tumors
After removing the control SNVs from PRC and LRRC samples, the focus was on common variations. In PRC, four genes, including MYCN, SCRIB, SNAPC4, and MED15, were identified with eight mutations (Table 1). Notably, the SCRIB mutation (c.T233G) was predicted as “D (deleterious)” by SIFT and Polyphen2 software. In the LRRC, two genes, including SCRIB and RUSC2 (Table 2), were found to have three mutations. Interestingly, LRRC had the same SCRIB mutation as PRC. In addition, two frame shift mutations were found in the RUSC2 gene (c.831_833del, c.3465_3467del).
Table 1. Common variations in PRC samples.
| Gene | Mutation type | Variants | 1000g2015_eas | ExAC eas | SIFT | Polyphen2 |
| MYCN | Frame shift insertion | c.87dupC | - | - | - | - |
| SCRIB | Nonsynonymous | c.233T>G | - | - | D | D |
| SNAPC4 | Nonsynonymous | c.3158T>A | - | - | D | B |
| MED15 | Synonymous | c.1035G>C | - | - | - | - |
| c.1197G>C | - | - | - | - | ||
| c.1170G>C | ||||||
| c.1248G>C | ||||||
| c.1368G>C |
Table 2. Common variations in LRRC samples.
| Gene | Mutation type | Variants | 1000g2015_eas | ExAC eas | SIFT | Polyphen2 |
| SCRIB | Nonsynonymous | c.233T>G | - | - | D | D |
| RUSC2 | Non-frameshift deletion | c.831_833del c.3465_3467del | - | - | - | - |
Wnt signaling pathway is one of the specific pathways in LRRC
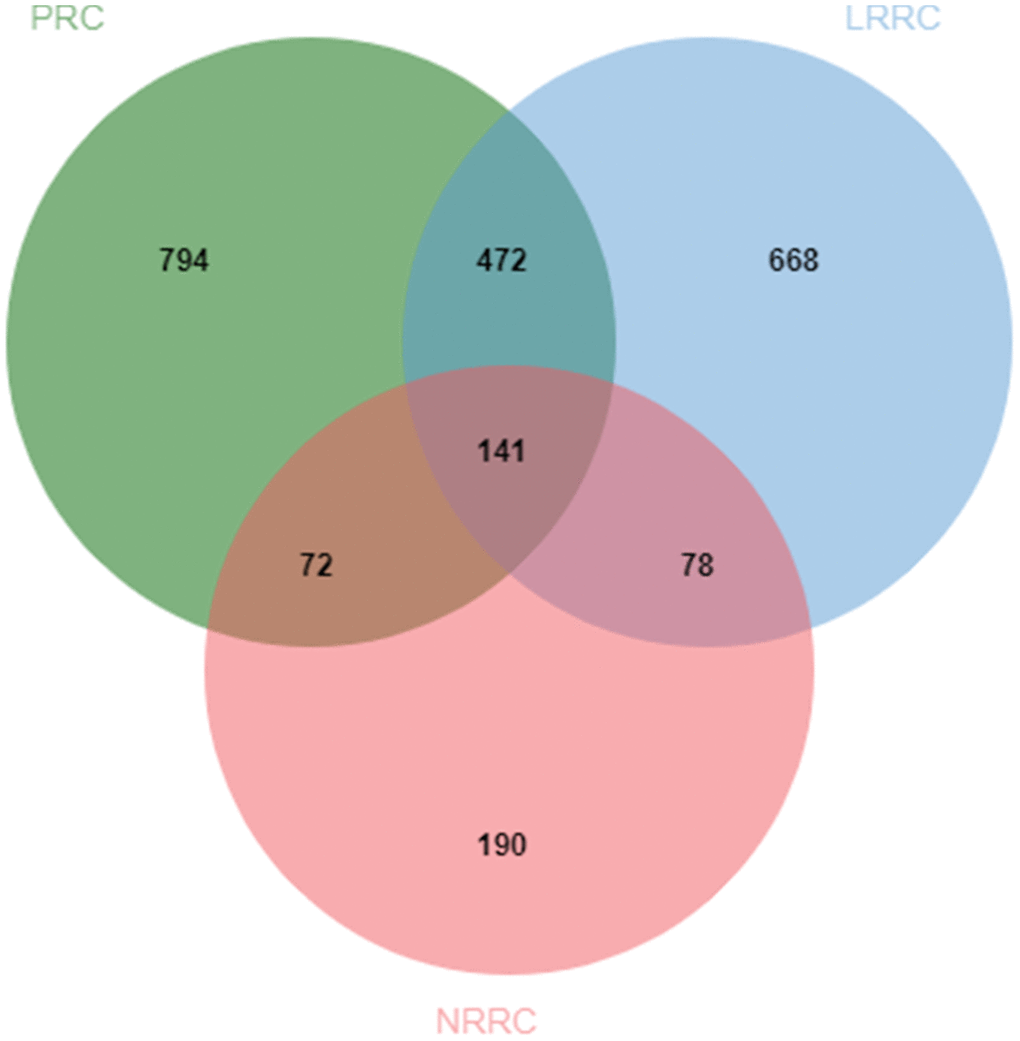
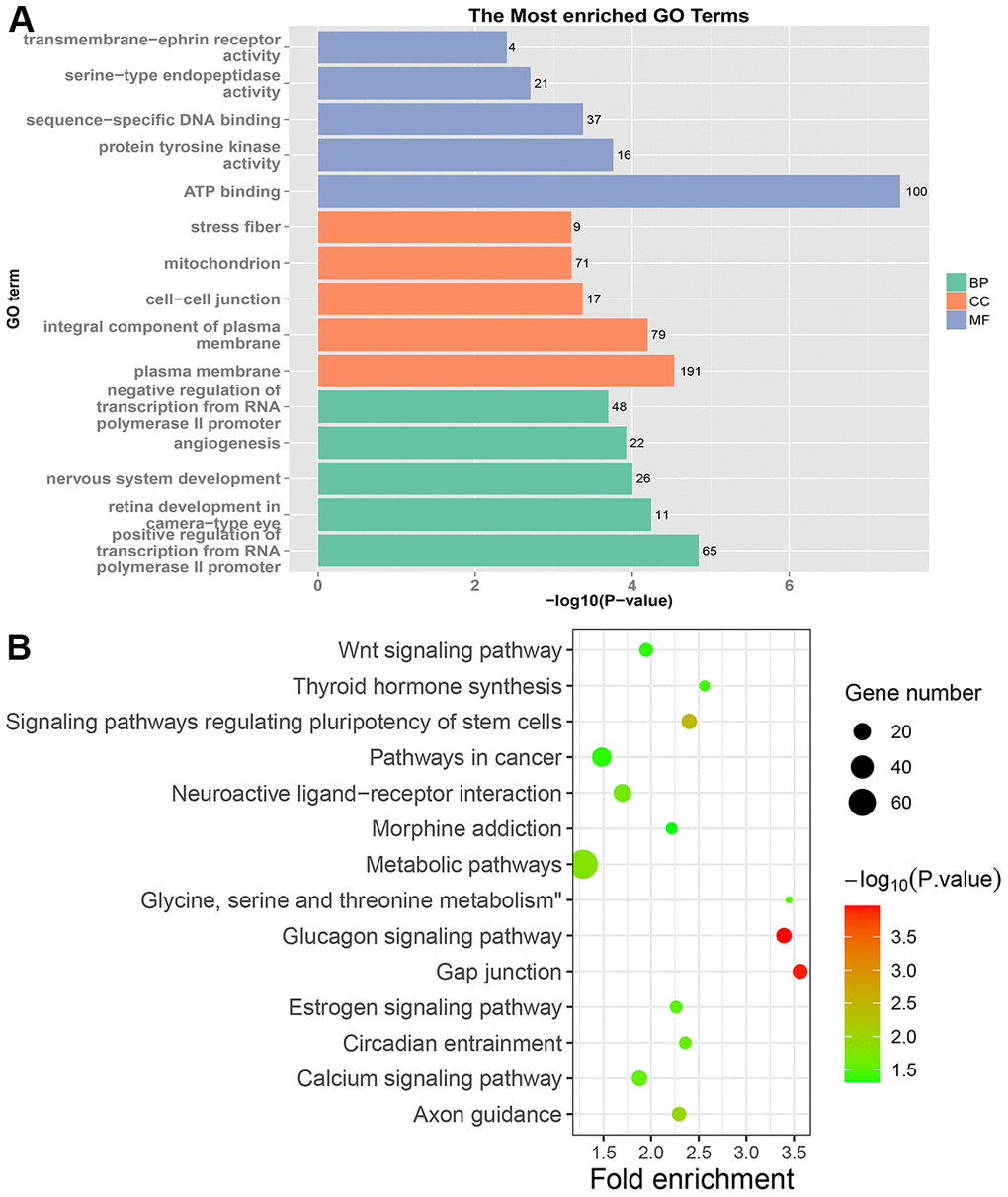
Venn analysis of the enriched KEGG pathway was performed on five sets of samples. As shown in Figure 6, there are 7 specific pathways in LRRC, including Wnt signaling pathway, Gap junction, Glucagon signaling pathway, Axon guidance, Thyroid hormone synthesis, Morphine addiction and Glycine, serine and threonine metabolism.
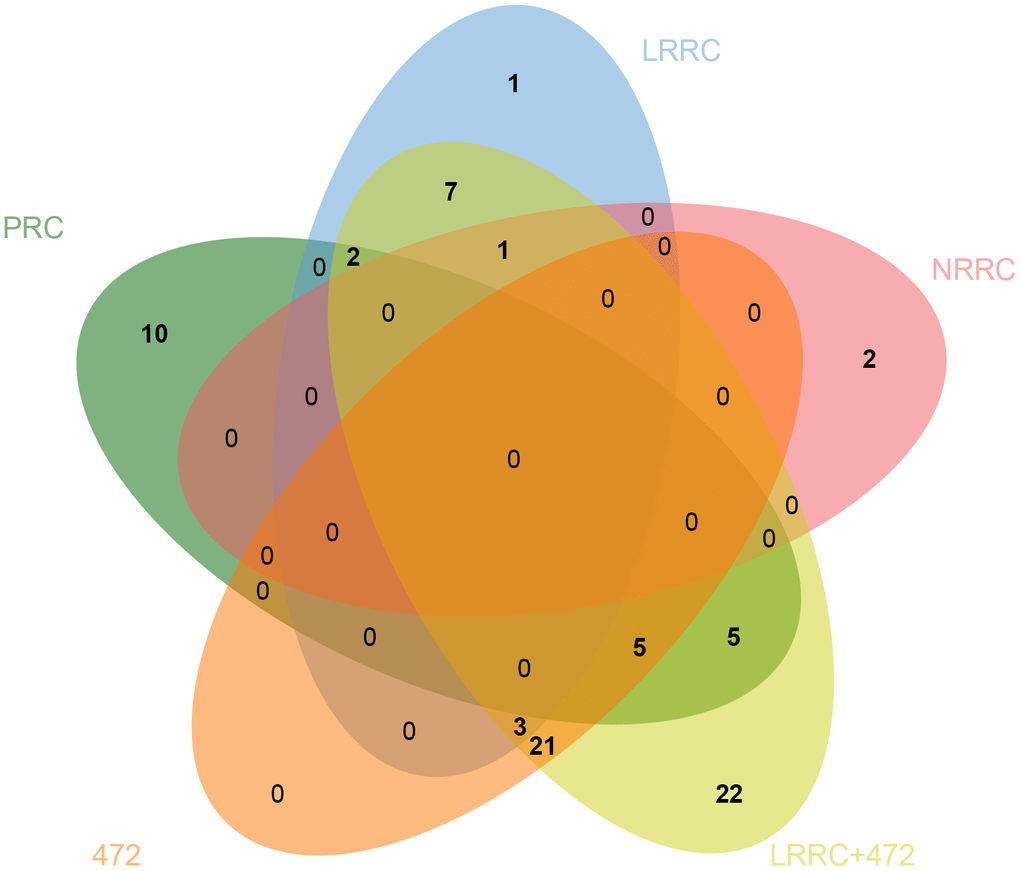
Figure 6. Venn diagram of specific pathways in PRC, LRRC, NRRC, 472, and LRRC+472. 472, 472 common genes; LRRC+472, LRRC+ 472 common genes.
Analysis of driver genes in LRRC
Based on the database, 48 possible driver genes were selected. As shown in Figure 11, Mutations in the genes, including MUC17, TTN, SYNE1, MUC16, FAT4, FLG, CSMD1, FAT3, RYR1, COL6A3, NEB, OBSCN, ZFHX4, TCF7L2, ERN3, NOTCH3, KMT2C, and PTRRT, were detected in both LRRC and PRC samples from five LRRC patients. Mutations in PEG3, NRAS, and BRAF did not occur in the PRC samples of the five patients. In LRRC samples, PEG3 mutations were present in 4 samples, NRAS in 1 sample, and BRAF in 1 sample. The other 27 mutations were present in some of the LRRC or PRC samples.
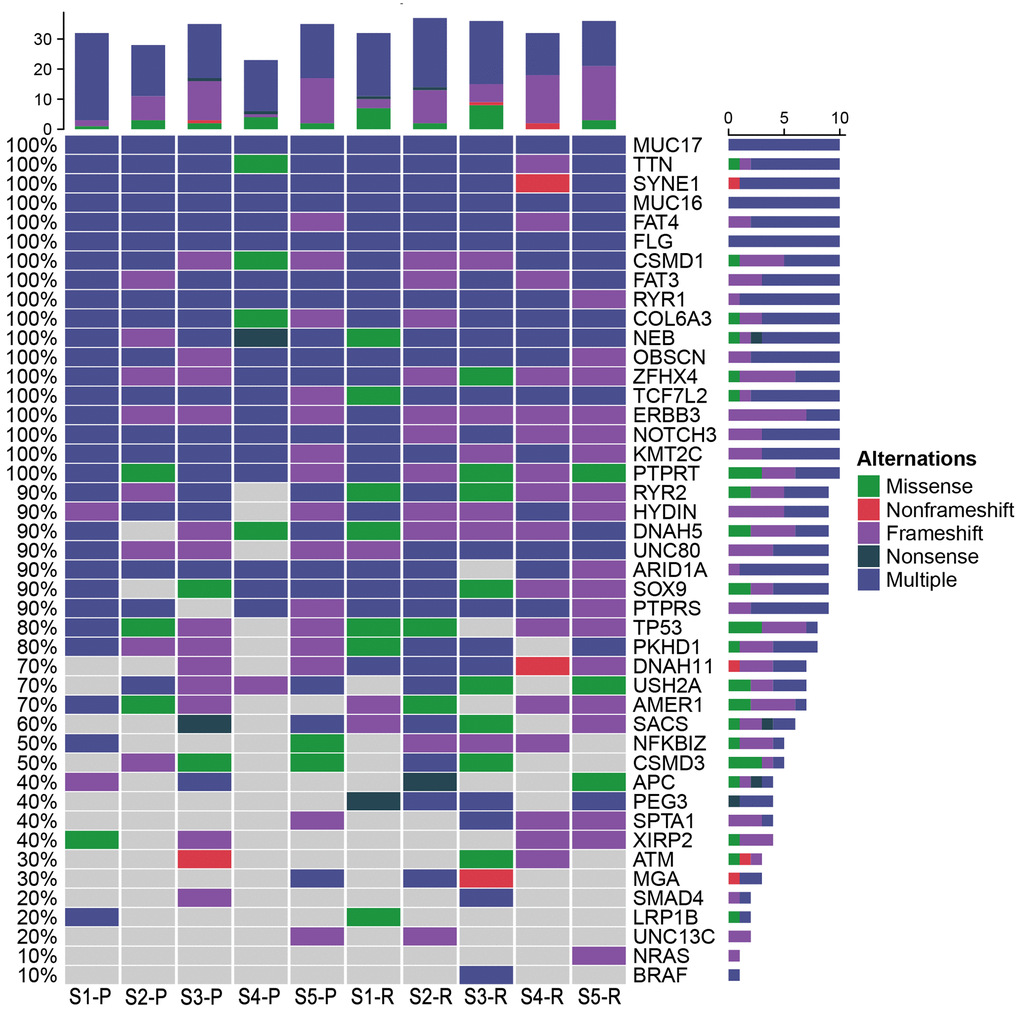
Figure 11. Potential driver genes related to LRRC. Forty-eight genes included (1) genes reported as significantly mutated genes in the previous study in CRC; (2) genes with a higher mutation rate in the cohort (TCGA Freq≥10%; MSK Freq≥5%).
The CNVs in PRC and LRRC
After removing the control CNVs from PRC and LRRC samples, the focus was on specific CNVs. As shown in Table 5, FGFR1 and MYC were found to be genes specific for CNVs in PRC and LRRC, respectively. Besides, FGFR3 CNVS were occurred in both PRC and LRRC. The copy numbers of FGFR3 in PRC and LRRC tumors were 3.542 and 3.548, respectively. Due to the sample size, after removing control CNVs, no CNVs was found in NRRC. There were no significant CNVs related to Wnt pathway.
Table 5. The CNVs of PRC and LRRC samples.
| Sample | Chromosome | Start | End | Gene | Depth | Copy_number |
| P1-primary | Chr4 | 1803536 | 1803776 | FGFR3 | 677.633 | 3.12 |
| P2-primary | Chr4 | 1803536 | 1803776 | FGFR3 | 493.65 | 3.04 |
| P3-primary | Chr4 | 1803536 | 1803776 | FGFR3 | 699.067 | 3.27 |
| P3-primary | Chr11 | 69588723 | 69588963 | FGF4 | 655.746 | 3.24 |
| P3-primary | Chr8 | 38282001 | 38282241 | FGFR1 | 886.567 | 3.79 |
| P4-primary | Chr4 | 1803284 | 1803510 | FGFR3 | 414.624 | 4.15 |
| P5-primary | Chr4 | 1803536 | 1803776 | FGFR3 | 986.671 | 4.13 |
| P5-primary | Chr11 | 69588723 | 69588963 | FGF4 | 580.725 | 3.04 |
| P1-recurrent | Chr4 | 1803284 | 1803510 | FGFR3 | 273.934 | 3.71 |
| P2-recurrent | Chr4 | 1803536 | 1803776 | FGFR3 | 543.558 | 3.37 |
| P3-recurrent | Chr4 | 1803284 | 1803510 | FGFR3 | 200.765 | 3.38 |
| P4-recurrent | Chr4 | 1803536 | 1803776 | FGFR3 | 772.854 | 3.92 |
| P4-recurrent | Chr11 | 69588723 | 69588963 | FGF4 | 494.054 | 3.08 |
| P5-recurrent | Chr4 | 1803536 | 1803776 | FGFR3 | 472.738 | 3.36 |
| P5-recurrent | Chr8 | 128750764 | 128751028 | MYC | 2600.53 | 12.58 |
Discussion
WES has a high sensitivity to common, rare, and low-frequency mutations. It could find the most disease-related mutations in the exon region, and only needs to sequence about 1% of the genome [20]. WES had been widely used in CRC to detect the mutational landscape of CRC populations and provided novel insights into the treatment and prognosis of CRC in the clinic [21].
LRRC refers to the recurrence of READ after radical resection. In addition to negative distal and circumferential margins, radical resection also requires no lateral or distant lymph node metastasis. Attention should also be paid to tumor-free operations, including irrigation before anastomosis. However, the evolution of LRRC at the genome level remains unknown. In the present study, WES was performed on PRC, LRRC, and NRRC samples to detect the mutational characteristics of LRRC and discover the specific genes and related pathways. The results of the study showed that LRRC tissues exhibit different mutation profiles.
Many studies have explored the relationship between mutations in key driver genes and CRC metastasis [22]. The specific genes of PRC were found, including MYCN, SCRIB, SNAPC4, and MED15. MYCN is a member of the MYC family. The amplification of MYCN is related to many tumors, most notably neuroblastoma [23]. A frame shift mutation (c.87dupC) was found in the MYCN gene in 5 PRC samples, which was a clinically unknown mutation. SCRIB is a membrane protein that is involved in the maintenance of the apical basal cell polarity of epithelial tissues. It plays a tumor-suppressive role in the progression of skin and liver cancer [24]. Notably, a pathogenic mutation of SCRIB (c.T233G) was found in both PRC and LRRC samples. SCRIB affects tumor development by negatively modulating the Wnt/β-catenin signaling pathway [25]. It is suggested that SCRIB may affect the occurrence and development of READ through the Wnt/β-catenin signaling pathway. SNAPC4 gene encodes the largest subunit of the small nuclear RNA-activating protein (SNAP) complex, and its role in cancer is unknown. A nonsynonymous mutation was found in the SNAPC4 gene (c.T3158A) was damaged (SIFT). MED15 is part of the multiprotein mediator complex, which plays a cancer-promoting role in urothelial bladder cancer (BCa) and renal cell carcinoma (RCa).
Two specific genes of LRRC samples were found, including SCRIB and RUSC2. Non-frameshift mutations of the RUSC2 (c.831_833del, c.3465_3467del) were found in LRRC samples. RUSC2 interacts with the SHD domain of GIT2 and reduces GIT2 degradation, which regulates lung cancer progression through EGFR signaling [26]. Whether RUSC2 affects the progression of LRRC through EGFR signaling needs further research.
FGFR1 and MYC were the specific genes of CNVs for PRC and LRRC, respectively. Genetic aberrations in FGFRs have been reported in a variety of cancers, including gastric, lung, and breast cancers [27]. FGFR1 amplification was previously shown to be associated with resistance to endocrine therapy, shorter time to distant metastasis, and shorter overall survival in HR+ breast cancer [28]. MYC amplification plays an important role in the progression of CRC [29].
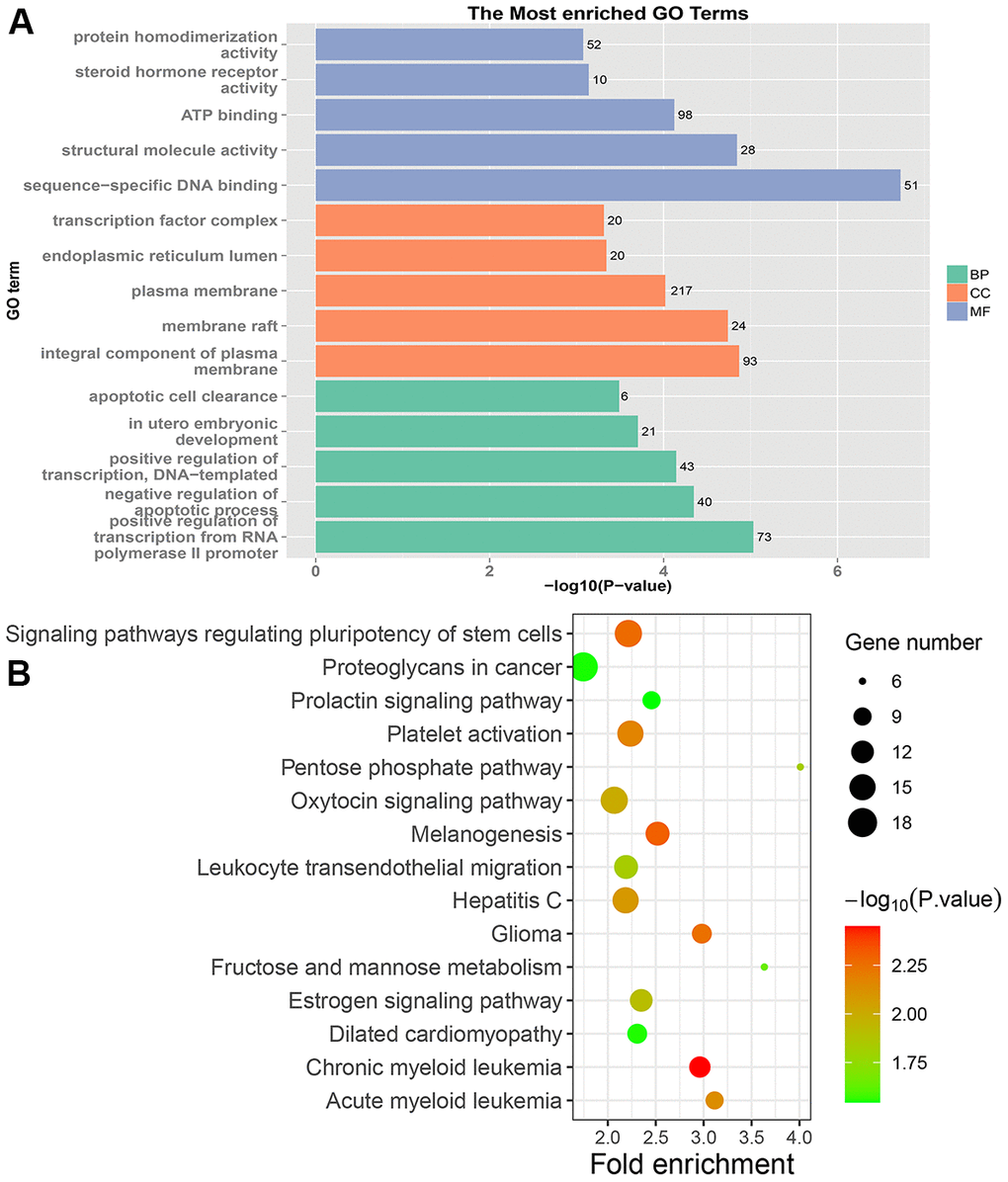
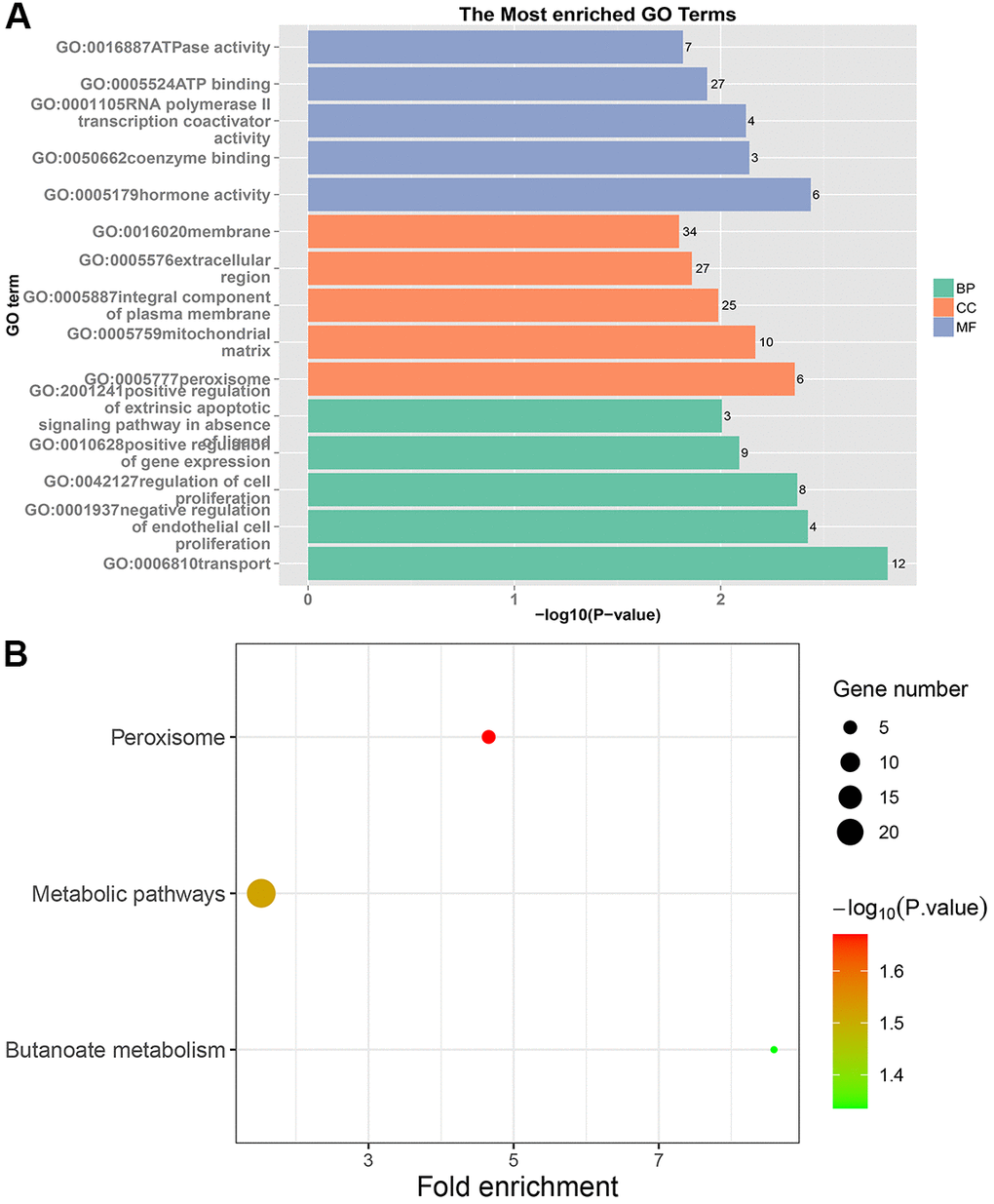
The pathways involved in PRC and LRRC are different. The specific pathways enriched in the PRC were the pentose phosphate pathway, fructose and mannose metabolism, proteoglycans in cancer, ECM-receptor interaction, and protein digestion and absorption. They were involved in the occurrence, development, and metastasis of CRC [30–33]. The pathogenic genes of NRRC were mainly enriched in peroxisome and butanoate metabolism. In CRC tissues, the peroxisome proliferator-activated receptor (PPAR) signaling pathway was down-regulated [34]. Several studies have shown that PPARγ activation promotes cell cycle arrest, apoptosis, and differentiation in many human tumors, and selective synthetic ligands have been shown to act as potential antitumor drugs [35]. It is suggested that the peroxisome limits the LRRC through the PPAR signaling pathway.
In this study, the specific signaling pathways of LRRC were Wnt signaling pathway, gap junction, glucagon signaling pathway, axon guidance, thyroid hormone synthesis, morphine addiction, glycine, serine and threonine metabolism. The present study focused on the Wnt signaling pathway. The PRICKLE1 mutation (c.C875T) and WNT6 mutation (c.G629A) were predicted as “D (deleterious)” by SIFT, Polyphen2 and FATHMM software, which were critical to the occurrence of LRRC (Table 4). The expression of LEF1 and cyclin D1 proteins of Wnt signaling pathway was higher in PRC compared to NRRC. Carbonic anhydrase IV (CA4) inhibits the Wnt signaling pathway by targeting the WTAP-WT1-TBL1 axis and is a novel tumor suppressor in CRC [36]. Agrin (AGRN) may act as an oncogenic indicator of READ through activation of the WNT pathway, which could help in the development of optimal treatments for READ [37]. Activation of Wnt signaling as a mechanism of chemoresistance in recurrent small cell lung cancer (SCLC) [38]. PRC1, a novel Wnt target, functions in a positive feedback loop that reinforces Wnt signaling to promote early Hepatocellular carcinoma (HCC) recurrence [39]. AKIP1 is a novel regulator of Wnt/β-catenin signaling and early relapse of HCC [40]. These findings suggest that the development of small molecule drugs that target the Wnt pathway may be important for LRRC.
Neoplastic transformation is frequently associated with a loss of gap junction intercellular communication and a reduction in the expression of connexins in various tumor types [41]. Furthermore, gap junctions may have distinct functional roles in cell growth and cell invasion, as a gap junction inhibitor decreases the invasion of prostate cancer cells [42]. Studies have found that glucagon signaling pathway may be involved in the progression of CRC [43]. Synthesis and growth of tumor proteins can be stimulated by glucagon in situ [44]. Several lines of evidence indicate that axon guidance genes are involved not only in neural development but also in cancer development. ROBO1 and ROBO2, crucial regulators of axon guidance, are considered potential tumor suppressor genes [45]. Multiple studies have shown a significant connection between hypothyroidism and pancreatic, gastric, and breast cancer [46]. Deregulation of Thyroid Hormones (THs) system in Colorectal Cancer (CRC) suggests that these hormones may play roles in CRC pathogenesis [47]. Morphine promotes tumorigenesis and cetuximab resistance via EGFR signaling activation in human colorectal cancer [48]. Glycolysis and glycine, serine and threonine were activated in CRC, and these alterations may promote cell proliferation [49].
Besides, this study found two KEGG pathways shared by PRC and LRRC, including signaling pathways regulating pluripotency of stem cells and estrogen signaling pathway. Cancer stem cells (CSCs) are involved in the occurrence and recurrence of CRC and have been identified [50]. Estrogen signaling pathway may be involved in the occurrence of CRC, but the specific mechanism is not yet clear [51]. These two pathways may be involved in the progression from PRC to LRRC.
There are certain limitations in the present study. The number of patients used for this study was too small. Although this study can’t yield a definitive conclusion, Wnt signaling pathway may play a critical role in LRRC. Although we used some tractable methods to validate the possible biological significance of Wnt signaling pathway mutations in LRRC, the specific functions and molecular mechanisms of the Wnt signaling pathway in LRRC need to be further explored.
Conclusions
We described the mutation characteristics of LRRC, PRC, and NRRC in the present study. Our results showed that LRRC, PRC, and NRRC had different genomic characteristics and involved different pathways. The signaling pathway of Wnt signaling pathway, Gap junction, Glucagon signaling pathway, Axon guidance, Thyroid hormone synthesis, Morphine addiction, and Serine and threonine metabolism may be related to the occurrence of LRRC. The genes including NFATC1, PRICKLE1, SOX17, and WNT6, related to Wnt signaling pathway, may play a critical role in LRRC. Further translational and clinical research is imperative to investigate new therapeutic strategies for LRRC.
Supplementary Materials
Author Contributions
Conceptualization, Yi Yang, Xiaodong Gu, Zhenyang Li and Jianbin Xiang; Data curation, Yi Yang and Jianbin Xiang; Funding acquisition, Jianbin Xiang; Methodology, Yi Yang, Xiaodong Gu, Zhenyang Li, Chuang Zheng, Zihao Wang, Minwei Zhou, Zongyou Chen, Dongbing Li and Jianbin Xiang; Writing – original draft, Yi Yang, Xiaodong Gu, Zhenyang Li, Mengzhen Li and Dongbing Li and Jianbin Xiang; Writing – review and editing, Yi Yang, Xiaodong Gu, Zhenyang Li, Chuang Zheng, Zihao Wang, Minwei Zhou, Zongyou Chen, Mengzhen Li, Dongbing Li and Jianbin Xiang.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by Pearl River S&T Nova Program of Guangzhou (201806010020 and 201906010020) and International Science and Technology Cooperation Projects of Guangzhou Development Zone (2017GH12).
References
- 1. Beyond TME Collaborative. Consensus statement on the multidisciplinary management of patients with recurrent and primary rectal cancer beyond total mesorectal excision planes. Br J Surg. 2013; 100:1009–14. https://doi.org/10.1002/bjs.9192 [PubMed]
- 2. Harji DP, Sagar PM. Advancing the surgical treatment of locally recurrent rectal cancer. Br J Surg. 2012; 99:1169–71. https://doi.org/10.1002/bjs.8879 [PubMed]
- 3. Mirnezami AH, Sagar PM, Kavanagh D, Witherspoon P, Lee P, Winter D. Clinical algorithms for the surgical management of locally recurrent rectal cancer. Dis Colon Rectum. 2010; 53:1248–57. https://doi.org/10.1007/DCR.0b013e3181e10b0e [PubMed]
- 4. Bhangu A, Ali SM, Cunningham D, Brown G, Tekkis P. Comparison of long-term survival outcome of operative vs nonoperative management of recurrent rectal cancer. Colorectal Dis. 2013; 15:156–63. https://doi.org/10.1111/j.1463-1318.2012.03123.x [PubMed]
- 5. Bouchard P, Efron J. Management of recurrent rectal cancer. Ann Surg Oncol. 2010; 17:1343–56. https://doi.org/10.1245/s10434-009-0861-2 [PubMed]
- 6. van Gijn W, Marijnen CA, Nagtegaal ID, Kranenbarg EM, Putter H, Wiggers T, Rutten HJ, Påhlman L, Glimelius B, van de Velde CJ, and Dutch Colorectal Cancer Group. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol. 2011; 12:575–82. https://doi.org/10.1016/S1470-2045(11)70097-3 [PubMed]
- 7. Bakx R, Visser O, Josso J, Meijer S, Slors JF, van Lanschot JJ. Management of recurrent rectal cancer: a population based study in greater Amsterdam. World J Gastroenterol. 2008; 14:6018–23. https://doi.org/10.3748/wjg.14.6018 [PubMed]
- 8. Palmer G, Martling A, Cedermark B, Holm T. A population-based study on the management and outcome in patients with locally recurrent rectal cancer. Ann Surg Oncol. 2007; 14:447–54. https://doi.org/10.1245/s10434-006-9256-9 [PubMed]
- 9. Pawlik TM, Skibber JM, Rodriguez-Bigas MA. Pelvic exenteration for advanced pelvic malignancies. Ann Surg Oncol. 2006; 13:612–23. https://doi.org/10.1245/ASO.2006.03.082 [PubMed]
- 10. Heriot AG, Tekkis PP, Darzi A, Mackay J. Surgery for local recurrence of rectal cancer. Colorectal Dis. 2006; 8:733–47. https://doi.org/10.1111/j.1463-1318.2006.01018.x [PubMed]
- 11. Cai Y, Li Z, Gu X, Fang Y, Xiang J, Chen Z. Prognostic factors associated with locally recurrent rectal cancer following primary surgery (Review). Oncol Lett. 2014; 7:10–16. https://doi.org/10.3892/ol.2013.1640 [PubMed]
- 12. Ashraf SQ, Burns EM, Jani A, Altman S, Young JD, Cunningham C, Faiz O, Mortensen NJ. The economic impact of anastomotic leakage after anterior resections in English NHS hospitals: are we adequately remunerating them? Colorectal Dis. 2013; 15:e190–98. https://doi.org/10.1111/codi.12125 [PubMed]
- 13. Fujita S, Mizusawa J, Kanemitsu Y, Ito M, Kinugasa Y, Komori K, Ohue M, Ota M, Akazai Y, Shiozawa M, Yamaguchi T, Bandou H, Katsumata K, et al, and Colorectal Cancer Study Group of Japan Clinical Oncology Group. Mesorectal Excision With or Without Lateral Lymph Node Dissection for Clinical Stage II/III Lower Rectal Cancer (JCOG0212): A Multicenter, Randomized Controlled, Noninferiority Trial. Ann Surg. 2017; 266:201–07. https://doi.org/10.1097/SLA.0000000000002212 [PubMed]
- 14. Sebag-Montefiore D, Stephens RJ, Steele R, Monson J, Grieve R, Khanna S, Quirke P, Couture J, de Metz C, Myint AS, Bessell E, Griffiths G, Thompson LC, Parmar M. Preoperative radiotherapy versus selective postoperative chemoradiotherapy in patients with rectal cancer (MRC CR07 and NCIC-CTG C016): a multicentre, randomised trial. Lancet. 2009; 373:811–20. https://doi.org/10.1016/S0140-6736(09)60484-0 [PubMed]
- 15. Harris CA, Solomon MJ, Heriot AG, Sagar PM, Tekkis PP, Dixon L, Pascoe R, Dobbs BR, Frampton CM, Harji DP, Kontovounisios C, Austin KK, Koh CE, et al. The Outcomes and Patterns of Treatment Failure After Surgery for Locally Recurrent Rectal Cancer. Ann Surg. 2016; 264:323–29. https://doi.org/10.1097/SLA.0000000000001524 [PubMed]
- 16. Westberg K, Palmer G, Hjern F, Johansson H, Holm T, Martling A. Management and prognosis of locally recurrent rectal cancer - A national population-based study. Eur J Surg Oncol. 2018; 44:100–07. https://doi.org/10.1016/j.ejso.2017.11.013 [PubMed]
- 17. Tanis PJ, Doeksen A, van Lanschot JJ. Intentionally curative treatment of locally recurrent rectal cancer: a systematic review. Can J Surg. 2013; 56:135–44. https://doi.org/10.1503/cjs.025911 [PubMed]
- 18. Lin C, Zhang Y, Zhang K, Zheng Y, Lu L, Chang H, Yang H, Yang Y, Wan Y, Wang S, Yuan M, Yan Z, Zhang R, et al. Fever Promotes T Lymphocyte Trafficking via a Thermal Sensory Pathway Involving Heat Shock Protein 90 and α4 Integrins. Immunity. 2019; 50:137–51.e6. https://doi.org/10.1016/j.immuni.2018.11.013 [PubMed]
- 19. Wei J, Wang Y, Shi K, Wang Y. Identification of Core Prognosis-Related Candidate Genes in Cervical Cancer via Integrated Bioinformatical Analysis. Biomed Res Int. 2020; 2020:8959210. https://doi.org/10.1155/2020/8959210 [PubMed]
- 20. Veltman JA, Brunner HG. De novo mutations in human genetic disease. Nat Rev Genet. 2012; 13:565–75. https://doi.org/10.1038/nrg3241 [PubMed]
- 21. Liu Z, Yang C, Li X, Luo W, Roy B, Xiong T, Zhang X, Yang H, Wang J, Ye Z, Chen Y, Song J, Ma S, et al. The landscape of somatic mutation in sporadic Chinese colorectal cancer. Oncotarget. 2018; 9:27412–22. https://doi.org/10.18632/oncotarget.25287 [PubMed]
- 22. Huang D, Sun W, Zhou Y, Li P, Chen F, Chen H, Xia D, Xu E, Lai M, Wu Y, Zhang H. Mutations of key driver genes in colorectal cancer progression and metastasis. Cancer Metastasis Rev. 2018; 37:173–87. https://doi.org/10.1007/s10555-017-9726-5 [PubMed]
- 23. Zhao Z, Shelton SD, Oviedo A, Baker AL, Bryant CP, Omidvarnia S, Du L. The PLAGL2/MYCN/miR-506-3p interplay regulates neuroblastoma cell fate and associates with neuroblastoma progression. J Exp Clin Cancer Res. 2020; 39:41. https://doi.org/10.1186/s13046-020-1531-2 [PubMed]
- 24. Kapil S, Sharma BK, Patil M, Elattar S, Yuan J, Hou SX, Kolhe R, Satyanarayana A. The cell polarity protein Scrib functions as a tumor suppressor in liver cancer. Oncotarget. 2017; 8:26515–31. https://doi.org/10.18632/oncotarget.15713 [PubMed]
- 25. Daulat AM, Wagner MS, Walton A, Baudelet E, Audebert S, Camoin L, Borg JP. The Tumor Suppressor SCRIB is a Negative Modulator of the Wnt/β-Catenin Signaling Pathway. Proteomics. 2019; 19:e1800487. https://doi.org/10.1002/pmic.201800487 [PubMed]
- 26. Duan B, Cui J, Sun S, Zheng J, Zhang Y, Ye B, Chen Y, Deng W, Du J, Zhu Y, Chen Y, Gu L. EGF-stimulated activation of Rab35 regulates RUSC2-GIT2 complex formation to stabilize GIT2 during directional lung cancer cell migration. Cancer Lett. 2016; 379:70–83. https://doi.org/10.1016/j.canlet.2016.05.027 [PubMed]
- 27. Katoh M. Fibroblast growth factor receptors as treatment targets in clinical oncology. Nat Rev Clin Oncol. 2019; 16:105–22. https://doi.org/10.1038/s41571-018-0115-y [PubMed]
- 28. Turner N, Pearson A, Sharpe R, Lambros M, Geyer F, Lopez-Garcia MA, Natrajan R, Marchio C, Iorns E, Mackay A, Gillett C, Grigoriadis A, Tutt A, et al. FGFR1 amplification drives endocrine therapy resistance and is a therapeutic target in breast cancer. Cancer Res. 2010; 70:2085–94. https://doi.org/10.1158/0008-5472.CAN-09-3746 [PubMed]
- 29. Ben-David E, Bester AC, Shifman S, Kerem B. Transcriptional dynamics in colorectal carcinogenesis: new insights into the role of c-Myc and miR17 in benign to cancer transformation. Cancer Res. 2014; 74:5532–40. https://doi.org/10.1158/0008-5472.CAN-14-0932 [PubMed]
- 30. Shibuya N, Inoue K, Tanaka G, Akimoto K, Kubota K. Augmented pentose phosphate pathway plays critical roles in colorectal carcinomas. Oncology. 2015; 88:309–19. https://doi.org/10.1159/000369905 [PubMed]
- 31. Brown DG, Rao S, Weir TL, O’Malia J, Bazan M, Brown RJ, Ryan EP. Metabolomics and metabolic pathway networks from human colorectal cancers, adjacent mucosa, and stool. Cancer Metab. 2016; 4:11. https://doi.org/10.1186/s40170-016-0151-y [PubMed]
- 32. Zhou F, Tang D, Xu Y, He H, Wu Y, Lin L, Dong J, Tan W, Dai Y. Identification of microRNAs and their Endonucleolytic Cleavaged target mRNAs in colorectal cancer. BMC Cancer. 2020; 20:242. https://doi.org/10.1186/s12885-020-06717-4 [PubMed]
- 33. Wang J, Yu S, Chen G, Kang M, Jin X, Huang Y, Lin L, Wu D, Wang L, Chen J. A novel prognostic signature of immune-related genes for patients with colorectal cancer. J Cell Mol Med. 2020; 24:8491–504. https://doi.org/10.1111/jcmm.15443 [PubMed]
- 34. Zhang X, Yao J, Shi H, Gao B, Zhang L. LncRNA TINCR/microRNA-107/CD36 regulates cell proliferation and apoptosis in colorectal cancer via PPAR signaling pathway based on bioinformatics analysis. Biol Chem. 2019; 400:663–75. https://doi.org/10.1515/hsz-2018-0236 [PubMed]
- 35. Blanquicett C, Roman J, Hart CM. Thiazolidinediones as anti-cancer agents. Cancer Ther. 2008; 6:25–34. [PubMed]
- 36. Zhang J, Tsoi H, Li X, Wang H, Gao J, Wang K, Go MY, Ng SC, Chan FK, Sung JJ, Yu J. Carbonic anhydrase IV inhibits colon cancer development by inhibiting the Wnt signalling pathway through targeting the WTAP-WT1-TBL1 axis. Gut. 2016; 65:1482–93. https://doi.org/10.1136/gutjnl-2014-308614 [PubMed]
- 37. Wang ZQ, Sun XL, Wang YL, Miao YL. Agrin promotes the proliferation, invasion and migration of rectal cancer cells via the WNT signaling pathway to contribute to rectal cancer progression. J Recept Signal Transduct Res. 2021; 41:363–70. https://doi.org/10.1080/10799893.2020.1811325 [PubMed]
- 38. Wagner AH, Devarakonda S, Skidmore ZL, Krysiak K, Ramu A, Trani L, Kunisaki J, Masood A, Waqar SN, Spies NC, Morgensztern D, Waligorski J, Ponce J, et al. Recurrent WNT pathway alterations are frequent in relapsed small cell lung cancer. Nat Commun. 2018; 9:3787. https://doi.org/10.1038/s41467-018-06162-9 [PubMed]
- 39. Chen J, Rajasekaran M, Xia H, Zhang X, Kong SN, Sekar K, Seshachalam VP, Deivasigamani A, Goh BK, Ooi LL, Hong W, Hui KM. The microtubule-associated protein PRC1 promotes early recurrence of hepatocellular carcinoma in association with the Wnt/β-catenin signalling pathway. Gut. 2016; 65:1522–34. https://doi.org/10.1136/gutjnl-2015-310625 [PubMed]
- 40. Cui Y, Wu X, Lin C, Zhang X, Ye L, Ren L, Chen M, Yang M, Li Y, Li M, Li J, Guan J, Song L. AKIP1 promotes early recurrence of hepatocellular carcinoma through activating the Wnt/β-catenin/CBP signaling pathway. Oncogene. 2019; 38:5516–29. https://doi.org/10.1038/s41388-019-0807-5 [PubMed]
- 41. Mesnil M. Connexins and cancer. Biol Cell. 2002; 94:493–500. https://doi.org/10.1016/s0248-4900(02)00025-4 [PubMed]
- 42. Tate AW, Lung T, Radhakrishnan A, Lim SD, Lin X, Edlund M. Changes in gap junctional connexin isoforms during prostate cancer progression. Prostate. 2006; 66:19–31. https://doi.org/10.1002/pros.20317 [PubMed]
- 43. Wu Z, Liu Z, Ge W, Shou J, You L, Pan H, Han W. Analysis of potential genes and pathways associated with the colorectal normal mucosa-adenoma-carcinoma sequence. Cancer Med. 2018; 7:2555–66. https://doi.org/10.1002/cam4.1484 [PubMed]
- 44. Fiocca R, Rindi G, Capella C, Grimelius L, Polak JM, Schwartz TW, Yanaihara N, Solcia E. Glucagon, glicentin, proglucagon, PYY, PP and proPP-icosapeptide immunoreactivities of rectal carcinoid tumors and related non-tumor cells. Regul Pept. 1987; 17:9–29. https://doi.org/10.1016/0167-0115(87)90029-2 [PubMed]
- 45. Je EM, Gwak M, Oh H, Choi MR, Choi YJ, Lee SH, Yoo NJ. Frameshift mutations of axon guidance genes ROBO1 and ROBO2 in gastric and colorectal cancers with microsatellite instability. Pathology. 2013; 45:645–50. https://doi.org/10.1097/PAT.0000000000000007 [PubMed]
- 46. Brown AR, Simmen RC, Simmen FA. The role of thyroid hormone signaling in the prevention of digestive system cancers. Int J Mol Sci. 2013; 14:16240–57. https://doi.org/10.3390/ijms140816240 [PubMed]
- 47. Zohreh B, Masoumeh V, Fakhraddin N, Omrani GH. Apigenin-mediated Alterations in Viability and Senescence of SW480 Colorectal Cancer Cells Persist in The Presence of L-thyroxine. Anticancer Agents Med Chem. 2019; 19:1535–42. https://doi.org/10.2174/1871520619666190704102708 [PubMed]
- 48. Lu H, Zhang H, Weng ML, Zhang J, Jiang N, Cata JP, Ma D, Chen WK, Miao CH. Morphine promotes tumorigenesis and cetuximab resistance via EGFR signaling activation in human colorectal cancer. J Cell Physiol. 2021; 236:4445–54. https://doi.org/10.1002/jcp.30161 [PubMed]
- 49. Gu J, Xiao Y, Shu D, Liang X, Hu X, Xie Y, Lin D, Li H. Metabolomics Analysis in Serum from Patients with Colorectal Polyp and Colorectal Cancer by 1H-NMR Spectrometry. Dis Markers. 2019; 2019:3491852. https://doi.org/10.1155/2019/3491852 [PubMed]
- 50. Szaryńska M, Olejniczak A, Kobiela J, Spychalski P, Kmieć Z. Therapeutic strategies against cancer stem cells in human colorectal cancer. Oncol Lett. 2017; 14:7653–68. https://doi.org/10.3892/ol.2017.7261 [PubMed]
- 51. Lv J, Li L. Hub Genes and Key Pathway Identification in Colorectal Cancer Based on Bioinformatic Analysis. Biomed Res Int. 2019; 2019:1545680. https://doi.org/10.1155/2019/1545680 [PubMed]