Introduction
Organ damage and septic shock are two major contributing factors to the high mortality associated with sepsis, a syndrome characterized by a disproportionate host immune response to infectious injury [1–3]. The sepsis-related mortality rate is exacerbated by septic shock, which causes circulatory failure, leading to organ hypoperfusion and ultimately organ failure. Adequate organ perfusion is largely dependent on normal diastolic blood pressure, which is influenced by cardiac output and thickness and elasticity of the aortic wall and peripheral vessels. Although aortic dysfunction is known to aggravate sepsis progression, the mechanisms responsible for aortic injury during sepsis remain insufficiently characterized [4].
Recent studies have uncovered approximately 100 different chemical modifications of RNAs that potentially affect, without altering their specific sequences, their folding and structure, stability, and function. The N6-methyladenosine (m6A) modification is the most abundant internal modification in mRNAs and occurs also in most non-coding RNA species, including long non-coding RNAs (lncRNAs). Accumulating evidence indicates a relevant role for m6A methylation in several gene expression steps, affecting transcript stability, export, splicing, and translation [5, 6]. The m6A modification is reversible and depends on the activity of methylases, demethylases, and adapter proteins termed m6A “writers”, “readers”, and “erasers” that mediate respectively the methylation, functional properties, and demethylation of the target RNAs [7, 8].
Changes in the m6A profile of various RNAs were shown to modulate many physiological and biological processes and to contribute to the pathogenesis of cardiovascular diseases [9, 10]. However, there is scarce information about m6A alterations in mRNAs and lncRNAs during sepsis, particularly in vascular tissues [11]. Therefore, we performed genome-wide screening of m6A modifications in lncRNAs and mRNAs from aortic tissues of septic rats and inferred, through bioinformatics analyses, the potential implications of the observed changes. Our findings may help identify new therapeutic targets to reduce the morbidity and mortality associated with septic syndromes.
Results
Sepsis decreases global mRNA m6A levels in the rat aorta
To evaluate potential changes in the m6A profile of aortic RNA species following sepsis, an intraperitoneal injection of lipopolysaccharide (LPS) was applied to rats to establish a sepsis model. Twenty-four h after LPS injection, mean arterial pressure (MAP) decreased by 30% relative to baseline. There was no significant change in MAP in rats injected with saline (control). The aortic tissues were carefully removed from rats after 24 hours of LPS/saline injection. LC-MS-based mRNA modification analysis was next used to detect global m6A levels in aortic mRNA. The results showed that the abundance of m6A sites was significantly decreased in the LPS group compared with the control group (Figure 1).
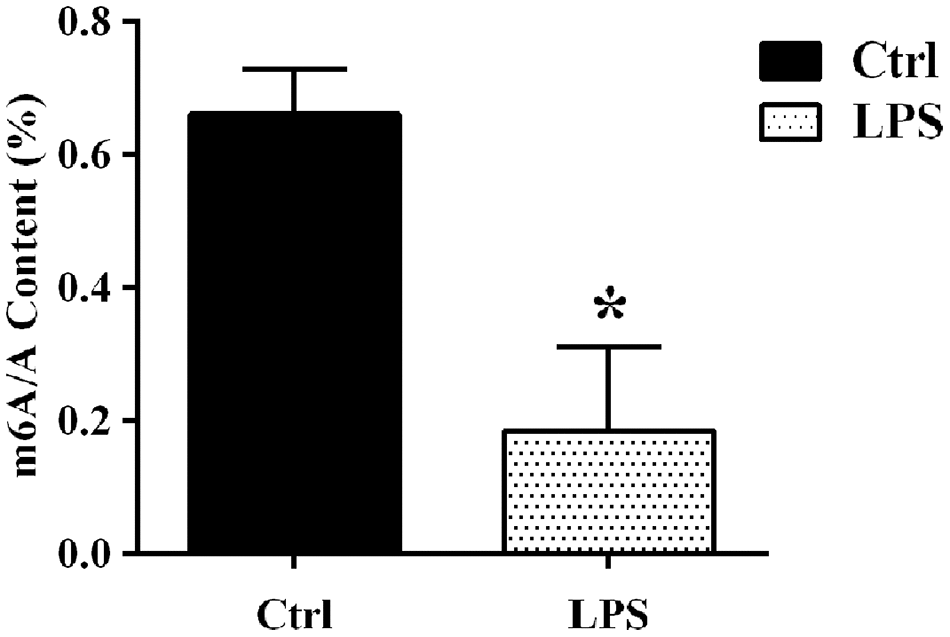
Figure 1. LC-MS-based analysis of sepsis-induced alterations in m6A levels in aortic mRNAs. Ctrl, control.
Microarray-based analysis of differentially m6A-modified mRNAs and lncRNAs
After antibody-based m6A labeling and immunoprecipitation of total aortic RNA, microarray analysis revealed that a total of 263 transcripts (187 mRNAs and 76 lncRNAs) in the LPS group had significantly altered (fold change > 1.5; P < 0.05) m6A abundance compared with the control group. Among these differentially m6A-modified transcripts, 84.8% (156 mRNAs and 67 lncRNAs) showed downregulated m6A levels, whereas the remaining 15.2% (31 mRNAs and 9 lncRNAs) showed instead upregulated m6A levels. Based on m6A fold-change ranking, information on 19 lncRNAs (the 9 hypermethylated and the top 10 hypomethylated ones) and 20 mRNAs (top 10 hypo- and hypermethylated mRNA transcripts) is provided in Supplementary Tables 1, 2. The mRNAs and lncRNAs with significantly altered m6A profiles were then lined up for cluster analysis according to the similarity of their m6A methylation levels and the closeness of their relationship. Variations in m6A patterns between the two groups are depicted in Figure 2 using volcano plots.
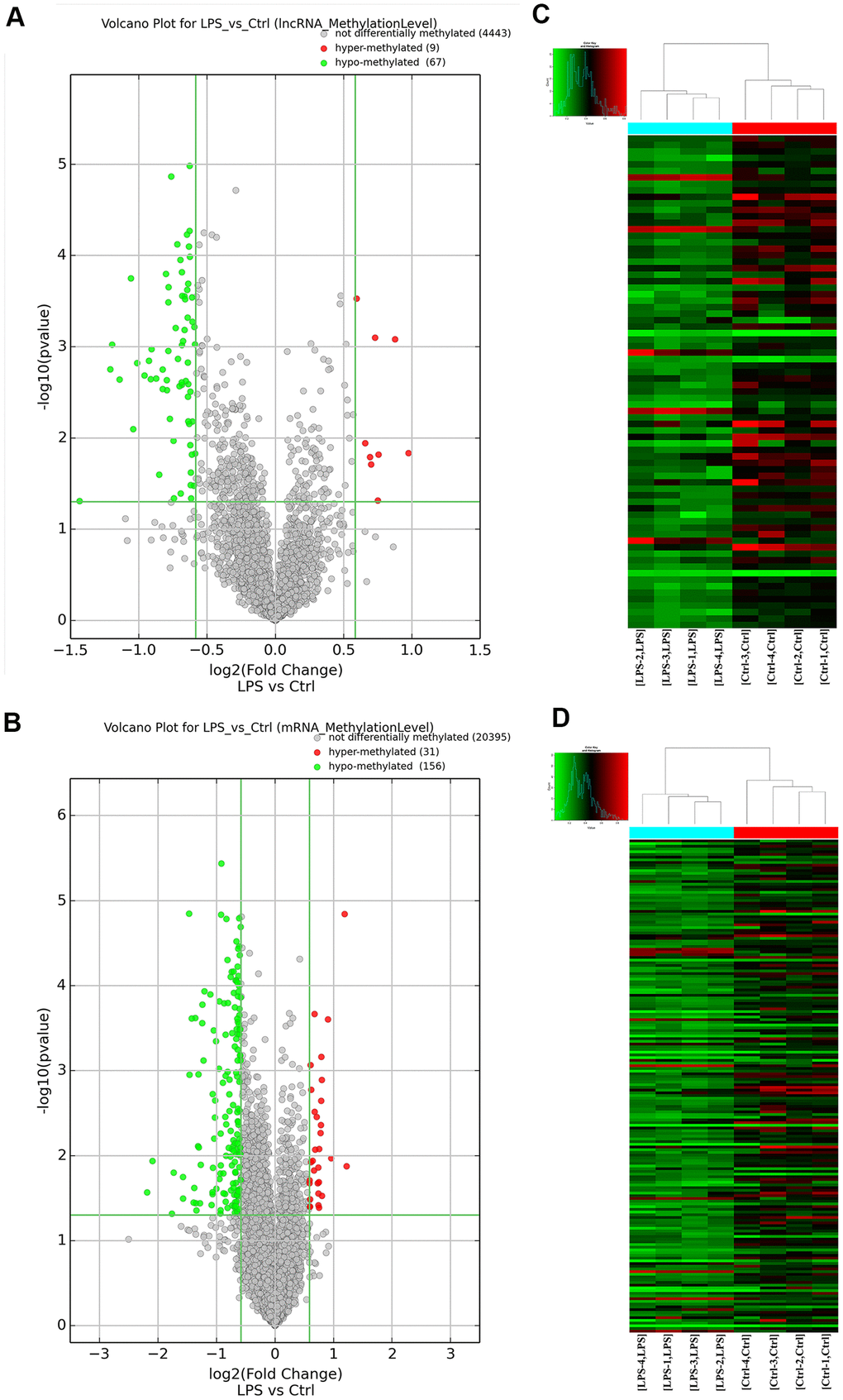
Figure 2. Overview of the m6A methylation map in aortic tissues. (A) Scatter plots showing differentially methylated lncRNAs. (B) Scatter plots showing differentially methylated mRNAs. (C) Hierarchical clustering analysis of lncRNAs with significantly altered m6A levels. (D) Hierarchical clustering analysis of mRNAs with significantly altered m6A levels. Ctrl, control.
GO and KEGG analysis of differentially methylated mRNAs
We performed Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses of the 31 m6A hypermethylated and the 156 m6A hypomethylated mRNAs obtained by microarray. Among the enriched GO terms for the 31 m6A hypermethylated mRNAs, ‘defense response’ in BP, ‘extracellular space’ in CC, and ‘calmodulin-dependent protein kinase activity’ in MF had the highest enrichment scores (Figure 3A, Supplementary Table 3). In turn, the most enriched GO terms for the 156 m6A hypomethylated mRNAs included ‘interspecies interaction between organisms’ in BP, ‘extracellular space’ in CC, and ‘organic anion transmembrane transporter activity’ in MF (Figure 3B, Supplementary Table 4). Meanwhile, KEGG analysis of the 31 m6A hypermethylated mRNAs revealed significant enrichment in ‘complement and coagulation cascades’, ‘inflammatory mediator regulation of transient receptor potential (TRP) channels’, and ‘neuroactive ligand-receptor interaction’ pathways (Figure 3C). Among the 156 m6A hypomethylated mRNAs, nine pathways were enriched, with top scores retrieved for ‘chemokine signaling pathway’ and ‘cytokine-cytokine receptor interaction’ (Figure 3D, Supplementary Table 5).
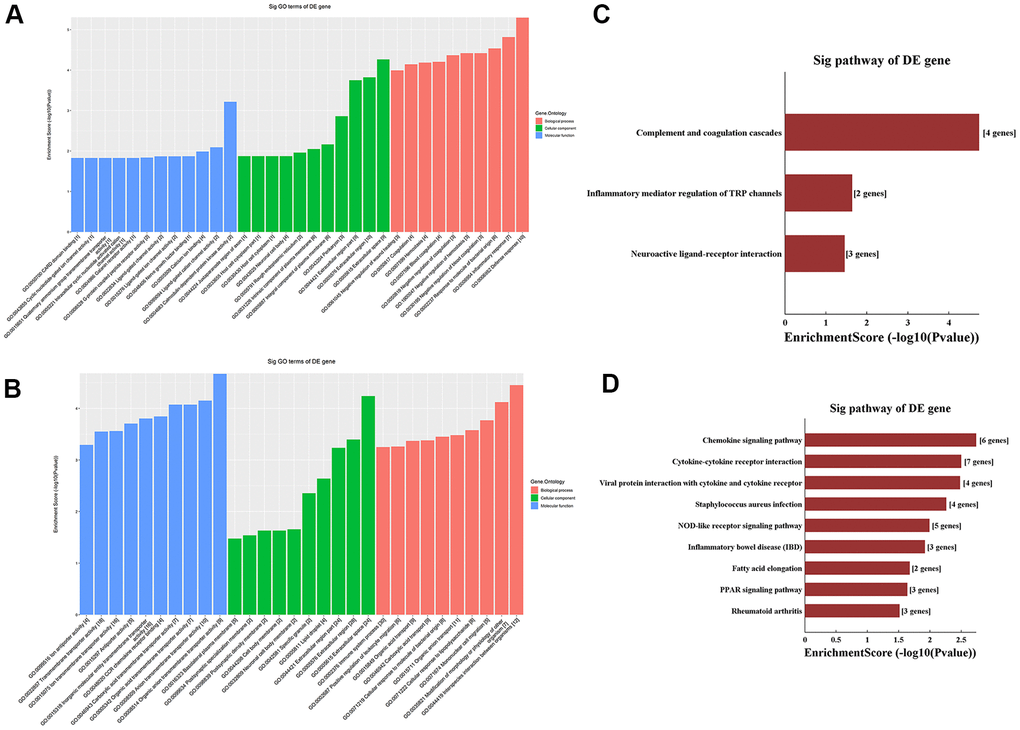
Figure 3. Functional enrichment analysis of differentially methylated mRNAs. (A) Top ten GO terms for hypermethylated mRNAs. (B) Top ten GO terms for hypomethylated mRNAs. (C) Top ten KEGG pathways for hypermethylated mRNAs. (D) Top ten KEGG pathways for hypomethylated mRNAs.
M6A single-base site qPCR with MazF treatment
Based on information in the SRAMP database, we performed motif analysis of m6A ACA sequences in the differentially m6A methylated lncRNAs and mRNAs detected by microarray and verified the presence of m6A-modified sites by qRT-PCR using the MazF enzyme treatment method. Five lncRNAs and five mRNAs with high-confidence m6A-modified sites were thus selected for validation using m6A single-base site qPCR (Table 1). Consistent with microarray data, this analysis confirmed significant downregulation of m6A in the lncRNA XR_343955. In contrast, no significant alterations in m6A levels were detected for the other four candidate lncRNAs. Among the five mRNAs considered, significant m6A downregulation was detected for both ENSRNOT00000010760 (consistent with microarray data) and ENSRNOT00000078131 (contrary to microarray results), while no significant alterations were observed in the other three mRNAs (Figure 4).
Table 1. Targeted lncRNAs and mRNAs from microarray predicted by SRAMP.
| GeneSymbol | Transcript ID | Transcript type | RNA length | Position | Regulation | Fold change | P-value |
| LOC103693543 | XR_595034 | lncRNA | 1931 | 1359 | hyper | 1.835972714 | 0.000829838 |
| LOC103690224 | XR_593937 | lncRNA | 1386 | 914 | hypo | 0.436711298 | 0.000951609 |
| LOC102554997 | XR_343955 | lncRNA | 1787 | 1482 | hypo | 0.60799908 | 7.56021E-05 |
| Leprel2 | XR_353597 | lncRNA | 2222 | 1781 | hypo | 0.630825555 | 0.000655905 |
| LOC103693720 | XR_595701 | lncRNA | 1332 | 1021 | hypo | 0.639273802 | 0.000238771 |
| Tnfrsf26 | ENSRNOT00000066943 | protein_coding | 2294 | 416 | hyper | 2.335986528 | 0.013354003 |
| Fibcd1 | ENSRNOT00000012927 | protein_coding | 4057 | 2519 | hyper | 1.587756196 | 0.014968993 |
| Kng1 | ENSRNOT00000078131 | protein_coding | 1905 | 1377 | hyper | 1.532870761 | 0.011963925 |
| Colgalt2 | ENSRNOT00000030109 | protein_coding | 1875 | 1730 | hypo | 0.411387391 | 0.03636713 |
| Mettl7b | ENSRNOT00000010760 | protein_coding | 1264 | 654 | hypo | 0.415309496 | 0.012844316 |
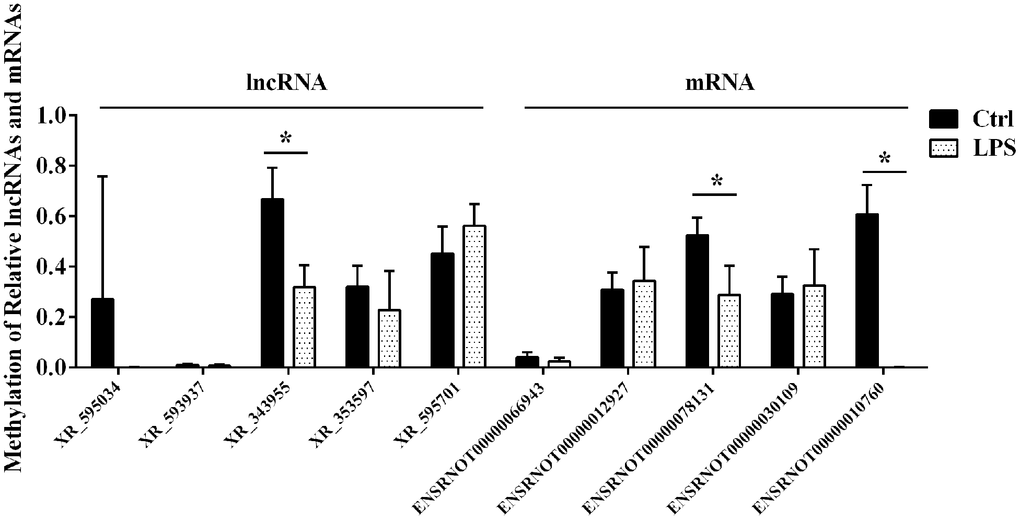
Figure 4. Confirmatory analysis of microarray results. M6A single-base site qPCR was used to validate the top five differentially methylated aortic lncRNAs and mRNAs identified through microarray in the LPS and Ctrl groups. Ctrl, control.
ceRNA network construction and functional enrichment analysis of target mRNAs
Based on m6A single-base site qPCR results, we constructed a competing endogenous RNA (ceRNA) network to identify putative miRNAs and mRNAs regulated by lncRNA-XR_343955. By confining the number of miRNA-IDs to 1000, 59 miRNA binding sites and 118 targeted mRNAs were predicted (Figure 5A). To assess the potential biological functions of lncRNA XR_343955, the 118 mRNAs thus retrieved were analyzed with GO and KEGG. For this mRNA set, the GO terms with the highest enrichment were ‘cell surface receptor signaling pathway’ in BP, ‘membrane part’ in CC, and ‘immunoglobulin receptor activity’ in MF (Figure 5B). In turn, KEGG analysis of the 118 mRNAs indicated cumulative enrichment in 27 pathways, of which the top 10 are shown in Figure 5C.
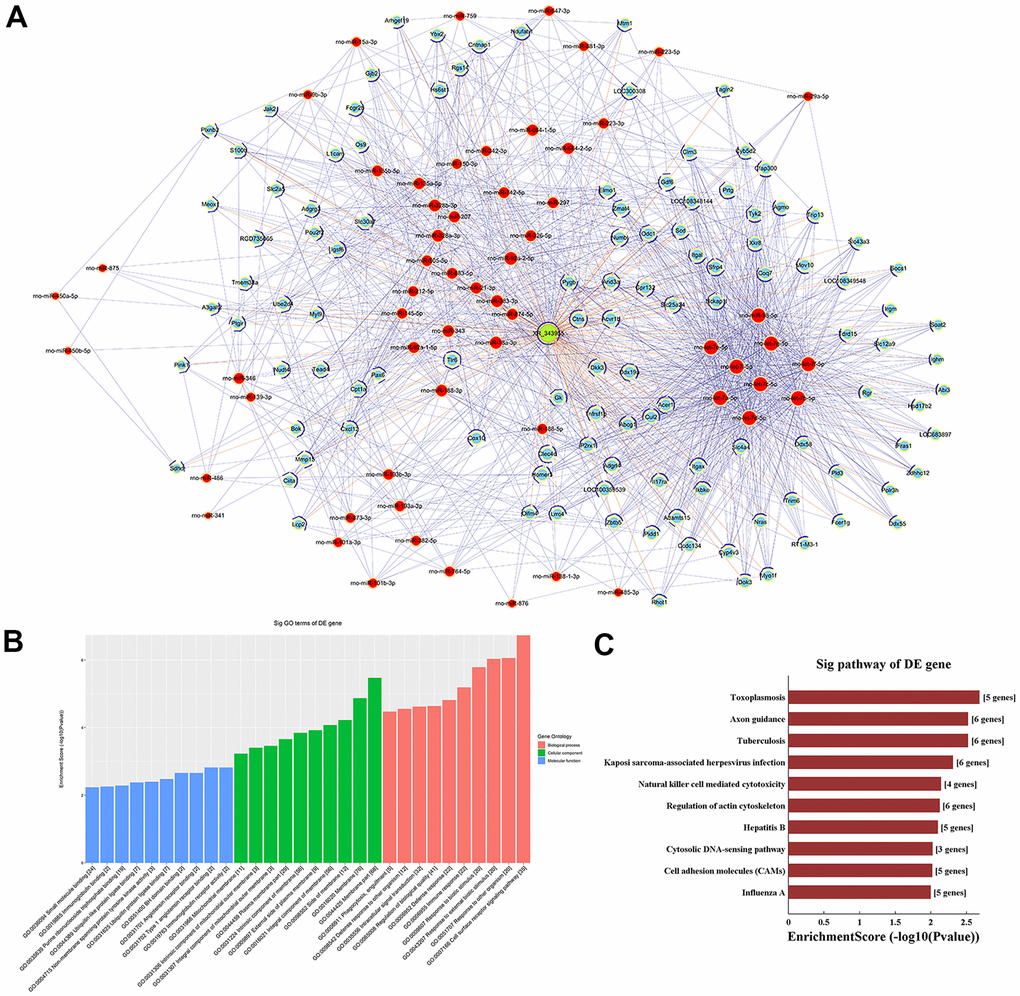
Figure 5. LncRNA-XR_343955-based ceRNA network. (A) XR_343955-associated ceRNA network. Red circles represent miRNAs, blue circles represent mRNAs, and green circles represent lncRNAs. (B) Histogram representation of GO functional classification of predicted mRNAs. (C) Histogram representation of KEGG pathway enrichment for predicted mRNAs.
Expression analysis of m6A effector proteins
We next used qRT-PCR to detect mRNA levels of key proteins regulating the m6A modification, namely m6A writers (METTL3, METTL14, and WTAP), readers (YTHDF 1 and YTHDF 3), and an eraser (FTO), in aortic samples from control and LPS-treated rats. Compared to control, in the LPS group the expression of both METTL3 and WTAP was significantly downregulated, while that of YTHDF 1, YTHDF 3, METTL14, and FTO remained essentially unchanged (Figure 6). These data suggest that the predominant m6A demethylation pattern observed for aortic RNA during sepsis is due, at least in part, to downregulation of the METTL3 methylase and the adapter protein WTAP.
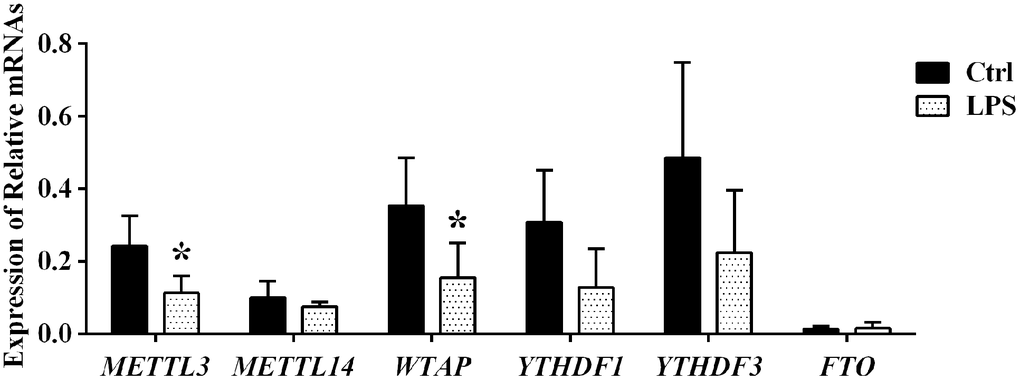
Figure 6. Expression analysis of m6A effector proteins by qPCR.
Discussion
The m6A modification of eukaryotic RNA has a wide-ranging effect on RNA homeostasis [9]. Therefore, alterations in RNA m6A methylation status can lead to cell dysfunction and disease [12]. Recent studies have revealed that the m6A modification not only has a strong and intricate relationship with cardiovascular disease but may regulate also the inflammatory response arising during different physiopathological conditions [13, 14]. Although the clinical significance of m6A profiling in sepsis patients has been recently suggested [11], there is a clear need for studies addressing the specific m6A alterations occurring in tissues and organs affected by sepsis. More extensive data exist for the role of the m6A modification in cardiovascular disease. Using clinical human samples, primary cardiomyocyte cultures, and preclinical pig and mouse models, Prabhu et al. found that increased levels of m6A during ischemia/hypoxia, resulting from downregulated FTO expression, are associated with impaired cardiomyocyte contractile function [13]. Consistently, Song et al. showed increased representation of m6A sites in mouse heart mRNAs following ischemia/reperfusion. Interestingly, this phenomenon was associated with increased expression of METTL3, a component of the m6A writer complex, and autophagy activation [15]. The impact of abnormal m6A expression patterns on inflammation was highlighted by Zhang et al., who reported that the m6A reader protein YT521-B homology domain family 2 (YTHDF2) activates the LPS-induced inflammatory response in preosteoblast MC3T3-E1 cells by regulating the MAPK signaling pathway [16]. In turn, Feng et al. reported that knockdown of METTL3 inhibited inflammation by allowing the expression of an alternatively spliced isoform of MyD88 in human dental pulp cells [17].
To explore potential changes in the m6A methylation profiles of aortic RNA species during sepsis-induced vascular injury, we established a sepsis model by intraperitoneally injecting LPS into Wistar rats. We found that both global levels of m6A and the expression of m6A writer complex proteins were significantly decreased in aortic tissue of LPS-treated rats. These results suggest that sepsis-induced changes in the m6A profile of aortic RNA species may be related to the vascular injury associated with septic syndromes.
GO and KEGG analysis of the minor fraction of mRNAs with upregulated m6A levels revealed their enrichment in coagulation processes. Although coagulation and inflammation represent basic host responses against infection, lack of resolution of these processes may cause damage to host cells and tissues. In sepsis, increased coagulation activity and decreased fibrinolysis caused by inflammation lead to fibrin deposition in the microcirculation. This in turn causes disseminated intravascular coagulation (DIC), ultimately leading to organ dysfunction [18, 19]. In patients with sepsis complicated by severe coagulopathy and/or DIC, organ dysfunction and mortality are significantly increased [20]. Over the past few decades, diverse anticoagulants such as serine protease inhibitors, recombinant human activated protein C, and tissue factor pathway inhibitor have been used as adjunctive therapies for patients with sepsis. However, two meta-analyses, conducted in 2003 and 2016, showed that anticoagulation was not beneficial in reducing mortality and was in turn associated with increased bleeding complications [21, 22]. Aiding the search for safer and more effective anti-DIC therapies, our results suggest that global or mRNA-specific therapeutic modulation of m6A methylation dynamics may be useful to regulate hemostasis and prevent or attenuate sepsis-induced DIC and organ failure.
GO analysis of aortic mRNAs with upregulated m6A levels demonstrated that several transcripts, many of which encode cation channels, were enriched in ion channel activity in the BP category. In turn, GO analysis of mRNAs with downregulated m6A expression demonstrated that numerous transcripts, many of those encoding proteins with organic anion transmembrane transporter activity, were associated with ion transmembrane transporter activity in the MF category.
The cations related to vascular function are primarily calcium (Ca2+) ions. Intracellular Ca2+ plays an important role in the modulation of vascular smooth muscle cell (VSMC) elasticity by affecting contraction and cell signaling [23–26]. In vascular endothelial cells (VECs), Ca2+ levels also influence the production of nitric oxide, a key regulator of vasoconstriction and diastolic function [27, 28]. The large number of cation channel mRNAs with increased m6A abundance detected in aortic tissue during sepsis may be partly responsible for the abnormal intracellular Ca2+ dynamics associated with septic vascular injury. Our KEGG analysis of mRNAs with upregulated m6A modification revealed in turn significant enrichment in the pathway related to inflammatory mediator regulation of TRP channels. This finding is consistent with the results of the GO analysis, since several members of the TRP protein family are Ca2+-selective channels. TRP channels are highly sensitive to various physical and chemical stimuli, and inflammation can lead to an influx of a large amount of Ca2+ into cells by activating TRP channels. In primary human osteoarthritis fibroblast-like synoviocytes, LPS stimulation leads to increased expression of TRP ankyrin 1 (TRPA1), enhanced TRPA1-mediated Ca2+ influx, and synthesis of pro-inflammatory factors [29]. In acute lung injury, TRP vanilloid 4 (TRPV4)-dependent Ca2+ influx contributes to LPS-induced macrophage activation, a process associated with the calcineurin-NFATc3 pathway [30]. These studies suggest that during inflammation the intracellular Ca2+ concentration can be affected via TRP channel activity.
Based on our high-throughput sequencing results, we speculate that increased m6A levels in mRNAs coding for cation channels, particularly TRP channels, contribute to dysregulated Ca2+ dynamics in sepsis and impaired VEC and VSMC function.
Our results showed decreased m6A levels in several aortic mRNAs related to organic anion transmembrane transporter activity. The anions involved in the regulation of vascular function are primarily chloride ions. Although the volume of healthy cells remains relatively stable, cell volume changes often occur during physiological and pathophysiological processes such as proliferation, migration, differentiation, and apoptosis [31]. When the cells swell, Cl- together with Na+ and water flow out through the "Cl- channels", "Ca2+-activated Cl- channels", or "transporters", and a swollen cell can return to normal size. During sepsis, inflammatory factors cause the swelling and dysfunction of VSMCs and VECs, and changes in the Cl- channels affect water drainage and vascular function [32, 33]. Our analysis of microarray data shows m6A levels of solute carrier family (SLC) gene were significantly decreased in aortic tissue during sepsis. The phylogenetically ancient SLC26 gene family encodes multifunctional anion exchangers and anion channels transporting a broad range of substrates, including Cl-, HCO3-, sulfate, oxalate, I−, and formate. It has been reported that SLC 26 member 7 (SLC26A7) was identified as a chloride–bicarbonate anion exchanger and/or as a Cl- channel in the kidney and stomach [34, 35], whose gene mutations cause congenital deafness and dyshormonogenic goiter [36]. Alterations in Cl- channels can affect intracellular water content, vascular tone and arterial blood pressure. These findings may be related to the impairment of vascular function in sepsis. The m6A modification provides a new research direction for vascular function protection.
Our results showed also significantly altered m6A levels in 76 aortic lncRNAs after LPS-induced sepsis. Although most studies on the m6A modification have focused on its regulatory role in mRNA function, recent evidence suggests that the m6A methylation regulates also the synthesis and function of lncRNAs [37]. Consistent with our microarray results, single-base site qPCR confirmed significant downregulation of m6A sites in lncRNA XR_343955 in the aorta of LPS-treated rats. GO and KEGG analyses of 118 predicted mRNAs detected via the ceRNA network for XR_343955 revealed significant enrichment of these transcripts in pathways involving cell adhesion molecules (CAMs). CAMs such as ICAM-1, E-selectin, and VCAM-1 play key roles in the vascular inflammation process by mediating the adhesion of circulating leukocytes to the vascular endothelium before extravasation into the vascular wall [38–41]. During sepsis, secretions of pro-inflammatory HMGB1 by endothelial cells can upregulate the expression of CAMs to promote the inflammatory response by recruiting leukocytes [42, 43]. Therefore, inhibiting the expression of CAMs in VECs is considered a promising treatment for vascular inflammatory diseases. Our results suggest that XR_343955 may regulate CAMs through a ceRNA mechanism, and such capacity may be in turn influenced by sepsis-related changes in XR_343955’s m6A profile.
In mammalian cells, dynamic and reversible m6A modification is governed by the activity of m6A methyltransferases and adapter proteins (i.e. m6A writers: METTL3, METTL14, and WTAP) and m6A demethylases (i.e. m6A erasers: FTO and ALKBH5), which install and remove, respectively, m6A marks in target RNAs. Another regulatory layer is further established by specific RNA-binding proteins (i.e. readers: YTHDF1/3) which bind to the m6A motif to affect RNA function. Using qRT-PCR, we detected that the expression of METTL3 and WTAP in the aorta was significantly downregulated during sepsis, while that of YTHDF 1, YTHDF 3, METTL14, and FTO did not change significantly. Downregulation of the m6A writers METTL3 and WTAP in aortic tissue during sepsis is thus consistent with both LC-MS data, which suggested a decrease in global m6A demethylase activity for the mRNA pool, and with microarray results, which showed decreased m6A levels in 84.8% of the mRNA/lncRNA transcripts with significantly altered m6A status. Dysregulated expression of m6A effectors has shown to contribute to cancer pathogenesis. For example, upregulated METTL3 expression was shown to promote gastric cancer and hepatocellular carcinoma progression by promoting, respectively, epithelial to mesenchymal transition and posttranscriptional silencing of SOCS2 [44, 45]. Therefore, it is plausible that downregulation of METTL3 and WTAP may modulate aortic damage during sepsis.
The m6A modification is an important modification of RNA, which has received extensive attention. However, it is difficult to quickly and efficiently study the relationship between m6A modification of RNA and diseases by relying on traditional biological experiments. Microarray analysis is a powerful tool that can reveal the differential expression profile of m6A-related RNAs underlying specific phenotypic differences. In addition, bioinformatics analysis methods and computational models could be effective ways to identify potential RNAs functions and RNA–disease associations. This might greatly decrease the time and cost of biological experiments. In our study, microarray analysis was performed, followed by bioinformatics analysis using a computer model. This can help researchers quickly and efficiently identify m6A-related RNAs associated with damage of aorta tissues during sepsis. However, our study has some potential limitations. First, differentially expressed m6A-related RNAs are mainly detected in the aorta tissues. Therefore, it is impossible to distinguish whether these changes occurs in vascular smooth muscle cells or vascular endothelial cells. Second, the sample size of microarray analysis was relatively small. Last, there were variations in differentially m6A methylated lncRNAs and mRNAs detected by microarray and m6A single-base site qPCR, which may be due to the methodological differences.
In conclusion, we detected differential abundance of m6A bases in numerous rat aortic mRNAs and lncRNAs, as well as altered expression of m6A writer proteins, following LPS-induced sepsis. GO and KEGG analyses indicated that the differentially m6A-modified mRNAs were mainly related to ‘complement and coagulation cascades’, ‘TRP channels’, and ‘organic anion transmembrane transporter activity’. In turn, ceRNA network analysis suggested the involvement of lncRNA-XR_343955 in the inflammatory response through regulation of CAM-related pathways. These findings suggest that therapeutic modulation of the cellular m6A machinery may be valuable to treat coagulation defects, attenuate inflammatory responses, and preserve vascular integrity in the setting of sepsis.
Materials and Methods
Animals
Eight-week-old male Wistar rats (250–350 g) were purchased from Charles River Laboratories (Beijing, China). Experimental procedures involving the use of animals complied with both ARRIVE guidelines (Consort Group, 2010) as well as with relevant national laws on animal protection, and the protocol was approved by the Ethics Committee on Animal Research at Peking University Health Science Center (Ethics No. LA2020343). Following adaptation to standard laboratory conditions for one week, the experimental rats were randomly allocated to one of two groups: the LPS-induced sepsis group (n = 4) received an intraperitoneal injection of 10 mg/kg LPS (Escherichia coli 055:B5; Sigma-Aldrich, USA; 5 mg of LPS dissolved in 1 mL of 0.9% saline); the control group (n = 4) was intraperitoneally injected with 0.9% saline (2 ml/kg). MAP was noninvasively measured 24 h after the LPS/saline injection. The aortic tissue (n = 4/group) were carefully removed from the anaesthetized rats, immediately frozen in liquid nitrogen and stored at -80° C until analysis.
RNA extraction and quality control
Total RNA from aortic tissues (n = 4 per group) was isolated and assessed as previously described [46]. Briefly, total RNA was isolated from the aortic tissues using TRIzol Reagent according to the manufacturer’s instruction (Invitrogen, USA). The quantity and purity of the total RNA samples were measured by a NanoDrop ND-1000 (ThermoFisher, USA).
LC-MS/MS-Based mRNA m6A modification detection
The mRNA was isolated and purified from total RNA using the NEBNext Poly(A) mRNA Magnetic Isolation Module (NEB, USA) and a Qubit RNA HS Assay kit (Thermo Fisher, USA). The mRNA was then hydrolyzed into single dephosphorylated nucleosides with an enzyme mix. The pretreated nucleoside solution was deproteinized using a Sartorius 10,000-Da MWCO spin filter. LC-MS/MS analysis was performed on an Agilent 6460 QQQ mass spectrometer with an Agilent 1260 HPLC system in multi-reaction monitoring (MRM) detection mode (n = 4/ group).
Detection of m6A-modified mRNAs and lncRNAs by microarray hybridization
Sample preparation and microarray hybridization procedures were based on Arraystar’s standard kit assays and protocols (Arraystar, USA). In this study, up to 27770 mRNAs and 10582 lncRNAs could be detected in a single array using the probes contained in Arraystar’s Rat mRNA and lncRNA Epitranscriptomic Array (m6A). In brief, purified total RNA from aortic tissue of LPS-treated and control rats was immunoprecipitated with polyclonal anti-m6A antibody (Cat 202003Synaptic Systems, USA). The m6A-tagged RNAs were eluted from the immunoprecipitated (IP) magnetic beads and the unmodified RNAs were eluted from the supernatant (Sup). The IP and Sup RNA fractions were then labeled with Cy5 and Cy3, respectively, as cRNAs in separate reactions using the Arraystar Super RNA Labeling Kit. The labeled cRNAs were then combined and hybridized onto an Arraystar Rat mRNA and lncRNA Epitranscriptomic Microarray (4x44K, Arraystar, USA) and scanned in two-color channels with an Agilent Scanner G2505C (Agilent, USA).
Microarray data analysis
Agilent Feature Extraction software (version 11.0.1.1) was used to analyze the acquired array images. The raw intensities of the IP (immunoprecipitation, Cy5-labeled) and Sup (supernatant, Cy3-labeled) RNA fractions were normalized using the average of the log2-scaled spike-in RNA intensities. Following spike-in normalization, the probe signals that displayed present (P) or marginal (M) QC flags in at least 4 out of 8 samples were retained as “All Targets Values” in an Excel sheet for further “m6A methylation level” analyses. The “m6A methylation level” was calculated as follows:
Differentially m6A-methylated RNAs between two comparison groups were identified by filtering by fold change (≥ 1.5) and statistical significance (P < 0.05) thresholds (n = 4 for each group).
M6A single-base site quantitative real-time PCR
The methylated lncRNAs and mRNAs were quantified by m6A single-base site qPCR with the MazF treatment method according to KangChen’s standard protocols (KangChen Biotech., China). In brief, the MazF treatment mixture was dispensed into a 10 μl volume with 1 μg of total RNA from each aortic tissue sample (n = 4 per group) and 20 U mRNA interferase-MazF (Takara, Japan) at 37° C for 30 min. One microgram of nondigested total RNA was reserved. The digested mRNA and the nondigested total RNA samples were subjected to reverse transcription using SuperScript™ III Reverse Transcriptase (Invitrogen, USA) for qPCR with a QuantStudio5 Real-time PCR System (Applied Biosystems, USA). Target lncRNAs and mRNAs were analyzed by SRAMP (http://www.cuilab.cn/sramp) to identify ACA motifs and m6A modification sites [47, 48]. The primers were designed using Primer 5.0 (Supplementary Table 6). Relative expression levels were calculated using the 2-ΔΔCt method, and the test genes were calibrated with MazF- as follows:
The experiments were carried out three times in independent determinations.
Competing endogenous RNA network construction and functional enrichment analysis
Candidate lncRNAs verified by m6A single-base site qPCR were analyzed for ceRNA network construction using a previously described protocol [49]. Differentially m6A-methylated mRNAs as well as target mRNAs predicted by the ceRNA network were classified into GO terms based on the GO database (http://www.geneontology.org). The KEGG (http://www.genome.jp/kegg) database was also interrogated to determine the biochemical pathways enriched by these mRNAs. Hierarchical clustering was performed using R software.
Analysis of m6A methylation regulators
To verify the expression of m6A writer, eraser, and reader proteins, qPCR experiments were performed as previously described [50]. We selected 6 representative proteins involved in m6A modification and binding, for which primers sequences are listed in Supplementary Table 7. The experiments were carried out three times in independent determinations.
Statistical analysis
For qRT-PCR, microarray, and m6A single-base site qPCR data, differences in transcript expression and methylation levels between the LPS and control groups were evaluated using unpaired, two-sided t-test. Fisher’s exact test was applied to evaluate the significance of the GO terms and KEGG pathway identifiers for mRNAs with differential methylation levels as well as for mRNAs predicted by the ceRNA network. The analysis was performed using the limma package on R software, with the recommended cut off of P < 0.05.
Data availability statement
The data that support the findings of this study are openly available in the GenBank database under accession number GSE158943 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE158943).
Supplementary Materials
Author Contributions
HZX and ZJS conceived and planned the experiments; ZJS and YCH performed the experiment and acquired the data; MWN, YNW, and RLX analyzed and interpreted the data; ZJS drafted the manuscript. HZX revised the manuscript. All authors provided critical feedback and help in shaping the research, analysis, and manuscript. All authors have read and approved the final submitted manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This article was supported by the Beijing Natural Science Foundation-Haidian Original Innovation Collaborative Fund (No. L192006).
References
- 1. Zarjou A, Black LM, McCullough KR, Hull TD, Esman SK, Boddu R, Varambally S, Chandrashekar DS, Feng W, Arosio P, Poli M, Balla J, Bolisetty S. Ferritin Light Chain Confers Protection Against Sepsis-Induced Inflammation and Organ Injury. Front Immunol. 2019; 10:131. https://doi.org/10.3389/fimmu.2019.00131 [PubMed]
- 2. Pavon A, Binquet C, Kara F, Martinet O, Ganster F, Navellou JC, Castelain V, Barraud D, Cousson J, Louis G, Perez P, Kuteifan K, Noirot A, et al, and EPIdemiology of Septic Shock (EPISS) Study Group. Profile of the risk of death after septic shock in the present era: an epidemiologic study. Crit Care Med. 2013; 41:2600–09. https://doi.org/10.1097/CCM.0b013e31829a6e89 [PubMed]
- 3. Stevenson EK, Rubenstein AR, Radin GT, Wiener RS, Walkey AJ. Two decades of mortality trends among patients with severe sepsis: a comparative meta-analysis*. Crit Care Med. 2014; 42:625–31. https://doi.org/10.1097/CCM.0000000000000026 [PubMed]
- 4. Kazune S, Grabovskis A, Cescon C, Strike E, Vanags I. Association between increased arterial stiffness and clinical outcomes in patients with early sepsis: a prospective observational cohort study. Intensive Care Med Exp. 2019; 7:26. https://doi.org/10.1186/s40635-019-0252-3 [PubMed]
- 5. Fazi F, Fatica A. Interplay Between N6-Methyladenosine (m6A) and Non-coding RNAs in Cell Development and Cancer. Front Cell Dev Biol. 2019; 7:116. https://doi.org/10.3389/fcell.2019.00116 [PubMed]
- 6. Yue Y, Liu J, He C. RNA N6-methyladenosine methylation in post-transcriptional gene expression regulation. Genes Dev. 2015; 29:1343–55. https://doi.org/10.1101/gad.262766.115 [PubMed]
- 7. Geula S, Moshitch-Moshkovitz S, Dominissini D, Mansour AA, Kol N, Salmon-Divon M, Hershkovitz V, Peer E, Mor N, Manor YS, Ben-Haim MS, Eyal E, Yunger S, et al. Stem cells. m6A mRNA methylation facilitates resolution of naïve pluripotency toward differentiation. Science. 2015; 347:1002–06. https://doi.org/10.1126/science.1261417 [PubMed]
- 8. Lin S, Choe J, Du P, Triboulet R, Gregory RI. The m(6)A Methyltransferase METTL3 Promotes Translation in Human Cancer Cells. Mol Cell. 2016; 62:335–45. https://doi.org/10.1016/j.molcel.2016.03.021 [PubMed]
- 9. Maity A, Das B. N6-methyladenosine modification in mRNA: machinery, function and implications for health and diseases. FEBS J. 2016; 283:1607–30. https://doi.org/10.1111/febs.13614 [PubMed]
- 10. Qin Y, Li L, Luo E, Hou J, Yan G, Wang D, Qiao Y, Tang C. Role of m6A RNA methylation in cardiovascular disease (Review). Int J Mol Med. 2020; 46:1958–72. https://doi.org/10.3892/ijmm.2020.4746 [PubMed]
- 11. Zhang S, Liu F, Wu Z, Xie J, Yang Y, Qiu H. Contribution of m6A subtype classification on heterogeneity of sepsis. Ann Transl Med. 2020; 8:306. https://doi.org/10.21037/atm.2020.03.07 [PubMed]
- 12. Chen Y, Peng C, Chen J, Chen D, Yang B, He B, Hu W, Zhang Y, Liu H, Dai L, Xie H, Zhou L, Wu J, Zheng S. WTAP facilitates progression of hepatocellular carcinoma via m6A-HuR-dependent epigenetic silencing of ETS1. Mol Cancer. 2019; 18:127. https://doi.org/10.1186/s12943-019-1053-8 [PubMed]
- 13. Mathiyalagan P, Adamiak M, Mayourian J, Sassi Y, Liang Y, Agarwal N, Jha D, Zhang S, Kohlbrenner E, Chepurko E, Chen J, Trivieri MG, Singh R, et al. FTO-Dependent N6-Methyladenosine Regulates Cardiac Function During Remodeling and Repair. Circulation. 2019; 139:518–32. https://doi.org/10.1161/CIRCULATIONAHA.118.033794 [PubMed]
- 14. Luo J, Xu T, Sun K. N6-Methyladenosine RNA Modification in Inflammation: Roles, Mechanisms, and Applications. Front Cell Dev Biol. 2021; 9:670711. https://doi.org/10.3389/fcell.2021.670711 [PubMed]
- 15. Song H, Feng X, Zhang H, Luo Y, Huang J, Lin M, Jin J, Ding X, Wu S, Huang H, Yu T, Zhang M, Hong H, et al. METTL3 and ALKBH5 oppositely regulate m6A modification of TFEB mRNA, which dictates the fate of hypoxia/reoxygenation-treated cardiomyocytes. Autophagy. 2019; 15:1419–37. https://doi.org/10.1080/15548627.2019.1586246 [PubMed]
- 16. Zhang Y, Gu X, Li D, Cai L, Xu Q. METTL3 Regulates Osteoblast Differentiation and Inflammatory Response via Smad Signaling and MAPK Signaling. Int J Mol Sci. 2019; 21:199. https://doi.org/10.3390/ijms21010199 [PubMed]
- 17. Feng Z, Li Q, Meng R, Yi B, Xu Q. METTL3 regulates alternative splicing of MyD88 upon the lipopolysaccharide-induced inflammatory response in human dental pulp cells. J Cell Mol Med. 2018; 22:2558–68. https://doi.org/10.1111/jcmm.13491 [PubMed]
- 18. Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013; 369:840–51. https://doi.org/10.1056/NEJMra1208623 [PubMed]
- 19. Iba T, Levi M, Levy JH. Sepsis-Induced Coagulopathy and Disseminated Intravascular Coagulation. Semin Thromb Hemost. 2020; 46:89–95. https://doi.org/10.1055/s-0039-1694995 [PubMed]
- 20. Gando S, Saitoh D, Ogura H, Fujishima S, Mayumi T, Araki T, Ikeda H, Kotani J, Kushimoto S, Miki Y, Shiraishi S, Suzuki K, Suzuki Y, et al, and Japanese Association for Acute Medicine Sepsis Registry Study Group. A multicenter, prospective validation study of the Japanese Association for Acute Medicine disseminated intravascular coagulation scoring system in patients with severe sepsis. Crit Care. 2013; 17:R111. https://doi.org/10.1186/cc12783 [PubMed]
- 21. Freeman BD, Zehnbauer BA, Buchman TG. A meta-analysis of controlled trials of anticoagulant therapies in patients with sepsis. Shock. 2003; 20:5–9. https://doi.org/10.1097/01.shk.0000068327.26733.10 [PubMed]
- 22. Umemura Y, Yamakawa K, Ogura H, Yuhara H, Fujimi S. Efficacy and safety of anticoagulant therapy in three specific populations with sepsis: a meta-analysis of randomized controlled trials. J Thromb Haemost. 2016; 14:518–30. https://doi.org/10.1111/jth.13230 [PubMed]
- 23. Zhu Y, Qu J, He L, Zhang F, Zhou Z, Yang S, Zhou Y. Calcium in Vascular Smooth Muscle Cell Elasticity and Adhesion: Novel Insights Into the Mechanism of Action. Front Physiol. 2019; 10:852. https://doi.org/10.3389/fphys.2019.00852 [PubMed]
- 24. Wu X, Davis GE, Meininger GA, Wilson E, Davis MJ. Regulation of the L-type calcium channel by alpha 5beta 1 integrin requires signaling between focal adhesion proteins. J Biol Chem. 2001; 276:30285–92. https://doi.org/10.1074/jbc.M102436200 [PubMed]
- 25. Wu X, Mogford JE, Platts SH, Davis GE, Meininger GA, Davis MJ. Modulation of calcium current in arteriolar smooth muscle by alphav beta3 and alpha5 beta1 integrin ligands. J Cell Biol. 1998; 143:241–52. https://doi.org/10.1083/jcb.143.1.241 [PubMed]
- 26. Ratz PH, Berg KM, Urban NH, Miner AS. Regulation of smooth muscle calcium sensitivity: KCl as a calcium-sensitizing stimulus. Am J Physiol Cell Physiol. 2005; 288:C769–83. https://doi.org/10.1152/ajpcell.00529.2004 [PubMed]
- 27. Loot AE, Pierson I, Syzonenko T, Elgheznawy A, Randriamboavonjy V, Zivković A, Stark H, Fleming I. Ca2+-sensing receptor cleavage by calpain partially accounts for altered vascular reactivity in mice fed a high-fat diet. J Cardiovasc Pharmacol. 2013; 61:528–35. https://doi.org/10.1097/FJC.0b013e31828d0fa3 [PubMed]
- 28. Ziegelstein RC, Xiong Y, He C, Hu Q. Expression of a functional extracellular calcium-sensing receptor in human aortic endothelial cells. Biochem Biophys Res Commun. 2006; 342:153–63. https://doi.org/10.1016/j.bbrc.2006.01.135 [PubMed]
- 29. Yin S, Wang P, Xing R, Zhao L, Li X, Zhang L, Xiao Y. Transient Receptor Potential Ankyrin 1 (TRPA1) Mediates Lipopolysaccharide (LPS)-Induced Inflammatory Responses in Primary Human Osteoarthritic Fibroblast-Like Synoviocytes. Inflammation. 2018; 41:700–09. https://doi.org/10.1007/s10753-017-0724-0 [PubMed]
- 30. Li M, Fang XZ, Zheng YF, Xie YB, Ma XD, Liu XT, Xia Y, Shao DH. Transient receptor potential vanilloid 4 is a critical mediator in LPS mediated inflammation by mediating calcineurin/NFATc3 signaling. Biochem Biophys Res Commun. 2019; 513:1005–12. https://doi.org/10.1016/j.bbrc.2019.04.020 [PubMed]
- 31. Hoffmann EK, Lambert IH, Pedersen SF. Physiology of cell volume regulation in vertebrates. Physiol Rev. 2009; 89:193–277. https://doi.org/10.1152/physrev.00037.2007 [PubMed]
- 32. Hübner CA, Schroeder BC, Ehmke H. Regulation of vascular tone and arterial blood pressure: role of chloride transport in vascular smooth muscle. Pflugers Arch. 2015; 467:605–14. https://doi.org/10.1007/s00424-014-1684-y [PubMed]
- 33. Chipperfield AR, Harper AA. Chloride in smooth muscle. Prog Biophys Mol Biol. 2000; 74:175–221. https://doi.org/10.1016/s0079-6107(00)00024-9 [PubMed]
- 34. Kim KX, Sanneman JD, Kim HM, Harbidge DG, Xu J, Soleimani M, Wangemann P, Marcus DC. Slc26a7 chloride channel activity and localization in mouse Reissner’s membrane epithelium. PLoS One. 2014; 9:e97191. https://doi.org/10.1371/journal.pone.0097191 [PubMed]
- 35. Tanimura Y, Kiriya M, Kawashima A, Mori H, Luo Y, Kondo T, Suzuki K. Regulation of solute carrier family 26 member 7 (Slc26a7) by thyroid stimulating hormone in thyrocytes. Endocr J. 2021; 68:691–99. https://doi.org/10.1507/endocrj.EJ20-0502 [PubMed]
- 36. Cangul H, Liao XH, Schoenmakers E, Kero J, Barone S, Srichomkwun P, Iwayama H, Serra EG, Saglam H, Eren E, Tarim O, Nicholas AK, Zvetkova I, et al, and UK10K Consortium. Homozygous loss-of-function mutations in SLC26A7 cause goitrous congenital hypothyroidism. JCI Insight. 2018; 3:e99631. https://doi.org/10.1172/jci.insight.99631 [PubMed]
- 37. Dominissini D, Moshitch-Moshkovitz S, Schwartz S, Salmon-Divon M, Ungar L, Osenberg S, Cesarkas K, Jacob-Hirsch J, Amariglio N, Kupiec M, Sorek R, Rechavi G. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature. 2012; 485:201–06. https://doi.org/10.1038/nature11112 [PubMed]
- 38. Frenette PS, Wagner DD. Adhesion molecules--Part II: Blood vessels and blood cells. N Engl J Med. 1996; 335:43–45. https://doi.org/10.1056/NEJM199607043350108 [PubMed]
- 39. Frenette PS, Wagner DD. Adhesion molecules--Part 1. N Engl J Med. 1996; 334:1526–29. https://doi.org/10.1056/NEJM199606063342308 [PubMed]
- 40. Frijns CJ, Kappelle LJ. Inflammatory cell adhesion molecules in ischemic cerebrovascular disease. Stroke. 2002; 33:2115–22. https://doi.org/10.1161/01.str.0000021902.33129.69 [PubMed]
- 41. Ulbrich H, Eriksson EE, Lindbom L. Leukocyte and endothelial cell adhesion molecules as targets for therapeutic interventions in inflammatory disease. Trends Pharmacol Sci. 2003; 24:640–47. https://doi.org/10.1016/j.tips.2003.10.004 [PubMed]
- 42. Jung B, Kang H, Lee W, Noh HJ, Kim YS, Han MS, Baek MC, Kim J, Bae JS. Anti-septic effects of dabrafenib on HMGB1-mediated inflammatory responses. BMB Rep. 2016; 49:214–19. https://doi.org/10.5483/bmbrep.2016.49.4.220 [PubMed]
- 43. Hsueh PT, Lin HH, Wang HH, Liu CL, Ni WF, Liu JK, Chang HH, Sun DS, Chen YS, Chen YL. Immune imbalance of global gene expression, and cytokine, chemokine and selectin levels in the brains of offspring with social deficits via maternal immune activation. Genes Brain Behav. 2018; 17:e12479. https://doi.org/10.1111/gbb.12479 [PubMed]
- 44. Yue B, Song C, Yang L, Cui R, Cheng X, Zhang Z, Zhao G. METTL3-mediated N6-methyladenosine modification is critical for epithelial-mesenchymal transition and metastasis of gastric cancer. Mol Cancer. 2019; 18:142. https://doi.org/10.1186/s12943-019-1065-4 [PubMed]
- 45. Chen M, Wei L, Law CT, Tsang FH, Shen J, Cheng CL, Tsang LH, Ho DW, Chiu DK, Lee JM, Wong CC, Ng IO, Wong CM. RNA N6-methyladenosine methyltransferase-like 3 promotes liver cancer progression through YTHDF2-dependent posttranscriptional silencing of SOCS2. Hepatology. 2018; 67:2254–70. https://doi.org/10.1002/hep.29683 [PubMed]
- 46. Yang Y, Shen F, Huang W, Qin S, Huang JT, Sergi C, Yuan BF, Liu SM. Glucose Is Involved in the Dynamic Regulation of m6A in Patients With Type 2 Diabetes. J Clin Endocrinol Metab. 2019; 104:665–73. https://doi.org/10.1210/jc.2018-00619 [PubMed]
- 47. Zhou Y, Zeng P, Li YH, Zhang Z, Cui Q. SRAMP: prediction of mammalian N6-methyladenosine (m6A) sites based on sequence-derived features. Nucleic Acids Res. 2016; 44:e91. https://doi.org/10.1093/nar/gkw104 [PubMed]
- 48. Imanishi M, Tsuji S, Suda A, Futaki S. Detection of N6-methyladenosine based on the methyl-sensitivity of MazF RNA endonuclease. Chem Commun (Camb). 2017; 53:12930–33. https://doi.org/10.1039/c7cc07699a [PubMed]
- 49. Yang Y, Xu W, Xiang RL, Tian X, Xu KF. Expression profiles and potential functions of long noncoding RNAs and mRNAs in autoimmune pulmonary alveolar proteinosis patients. Aging (Albany NY). 2021; 13:10535–54. https://doi.org/10.18632/aging.202818 [PubMed]
- 50. Chokkalla AK, Mehta SL, Kim T, Chelluboina B, Kim J, Vemuganti R. Transient Focal Ischemia Significantly Alters the m6A Epitranscriptomic Tagging of RNAs in the Brain. Stroke. 2019; 50:2912–21. https://doi.org/10.1161/STROKEAHA.119.026433 [PubMed]



