Introduction
Ischemic stroke is a common cause of severe disability and death worldwide [1]. To reduce primary injury due to acute ischemic stroke and limit infarct size, timely reperfusion therapy (thrombolysis or thrombectomy) is required [2]. However, post-ischemic reperfusion itself causes damage and dysfunction in a process known as cerebral ischemia-reperfusion (I/R) injury [3, 4]. In this process, reperfusion triggers an inflammatory cascade [5], a key mechanism contributing to secondary neuronal damage and death following the initial ischemic episode [6, 7]. Overzealous up-regulation of endogenous neuroinflammatory processes leads to destruction of hypoxic tissue, induction of apoptosis, and initiation of an inflammatory cascade feed-forward loop that can enlarge the damaged region [8–10].
Microglia, the major central nervous system resident innate immune cells, play an important role in modulating neuroinflammation [11]. Cerebral IR-activated microglia undergo polarization into either “classically activated” M1 or “alternately activated” M2 phenotypes [12, 13]. While M1 microglia produce a large number of pro-inflammatory factors (e.g. IL-6, TNF-α, and IL-1β) [14–16] that impede post-injury neural regeneration and produce poorer longterm neurological outcomes [17], M2 microglia are able to perform efferocytosis and produce numerous protective and trophic factors that promote neurogenesis [18]. Decreasing microglial-mediated neuroinflammatory injury by targeting M1/M2 polarization may therefore have therapeutic potential in ischemic stroke.
Our previous meta-analysis revealed that mesenchymal stem cell (MSC) transplantation after ischemic stroke significantly improves neurological deficits and quality of life [19]. In addition, our previous original research demonstrated that MSCs are able to protect brain tissue, regulate inflammatory responses after traumatic brain injury [20] and stroke [21, 22], and can regulate microglia-mediated inflammatory responses [23]. However, the mechanisms by which MSCs modulate microglia-mediated inflammation remain unclear. For the above reasons, elucidation of these mechanisms would be of significant benefit.
Relative to human bone marrow-derived MSCs (BMSCs), human umbilical cord MSCs (hUMSCs) are more readily obtained, exhibit superior viability, are compatible with therapeutic methods featuring higher levels of patient acceptability and compliance, and are not susceptible to immune-mediated graft rejection [24, 25]. In addition, although hUMSCs are considered more primitive than bone marrow derived MSCs, they do not induce teratomas, but do exhibit immunomodulatory capabilities [26]. Stroke occurs frequently at 45-65 years old, and there is an autologous bone marrow aging problem. In clinical research, it can avoid pain during bone marrow extraction and enhance volunteer compliance. We therefore selected hUMSCs for use in the current research.
Although information regarding MSC therapeutic mechanisms of action is limited and fewer than 1% of transplanted MSCs reach and engraft at target sites (since most become entrapped in the pulmonary capillary bed due to their large size) [27], their therapeutic effect is nonetheless frequently observed [28], currently mainly attributed to MSC-produced paracrine factors. We reasoned that transplanted MSC-derived exosomes (Exos) generated in vivo may be one factor contributing to the distal paracrine therapeutic effects of stem cell therapy.
Small non-coding microRNAs (miRNAs) can act as inhibitors of mRNA transcription and protein translation in most cell types [29]. Cell-derived exosomes contain numerous miRNAs that are able to act both locally and - via entering circulation - at distal sites [30, 31]. Exosomes are also internalized by neighboring or distal cells, thereby modulating the function of these recipient cells [32–34]. A recent study demonstrated that MSC-derived exosome content member miR-146a decreases inflammation and enhances anti-sepsis therapeutic efficacy [35]. Our sequencing data demonstrate that hUMSC-derived exosomes contain large amounts of miR-146a-5p. We thus hypothesized that hUMSC-derived exosomes (hUMSC-Exos), via provision of miR-146a-5p to microglia and consequent regulation of microglial gene expression, decrease microglia-mediated inflammation in the ischemic mouse brain. We injected wild-type and miR-146a-5p knockdown (miR-146a-5p k/d) hUMSC-Exos into ischemic mice to test this hypothesis as well as to explore mechanisms of potential therapeutic activity.
Results
Characterization of hUMSCs and hUMSC-Exos confirms hUMSC phenotype and demonstrates typical exosomal features
In vitro, hUMSCs were expanded to the third and fifth generations (Figure 1A). Flow cytometry was used to characterize hUMSC surface phenotype (Figure 1B). The majority of hUMSCs were positive for expression of CD73 (100%), CD105 (99.96%), and CD90 (100%), and negative for expression of CD45 (0.06%), HLA-DR (0.50%), CD34 (0.06%), CD11b (0.06%), and CD9 (0.46%). Such results are representative of the known hUMSC phenotype, confirming hUMSC identity. After removal of dead cells and debris from the hUMSC-conditioned culture medium, secreted extracellular vesicles were harvested by differential centrifugation. Such vesicles displayed typical exosomal features, including morphology and size (30-150 nm) as detected by electron microscopy (Figure 1C) and NanoSight analysis (Figure 1D), and the presence of Exos markers CD9, Alix, and TSG101 [36] as detected by western blot (Figure 1E). Therefore, they were designated hUMSC-Exos.
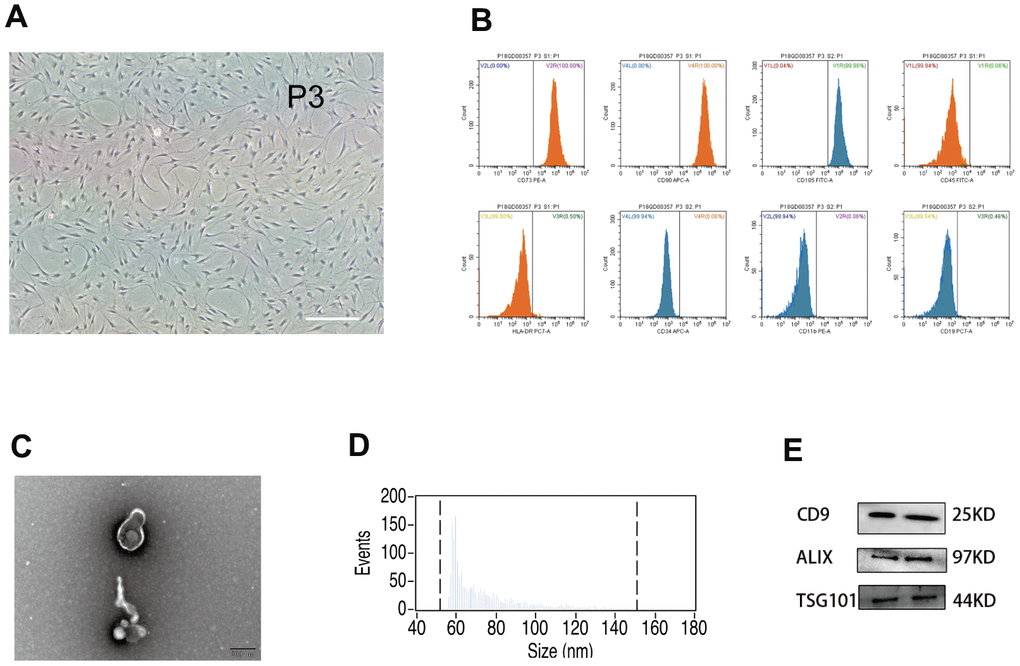
Figure 1. Analysis of human umbilical mesenchymal stem cells (hUMSCs) and hUMSC-derived exosomes (hUMSC-Exos). (A) Representative micrographs of cultured hUMSCs at passage 3 (P3). Scale bar: 200 μm. (B) Flow cytometry analysis of hUMSC CD73, CD105, CD90, CD11b, CD19, CD34, CD45, and HLA-DR expression. (C) Representative electron micrographs of hUMSC-Exos. Scale bar: 200 nm. (D) Exosome particle size and concentration. (E) Western blot analysis of Exos-specific markers CD9, ALIX, and TSG101. Each blot represents three independent experiments of two samples each.
In a murine model, hUMSC-Exos alleviate ischemic stroke injury and inflammation
We examined the in vivo effects of hUMSC-Exos in a murine model of ischemic stroke. Mice were randomly allocated to vehicle-only (phosphate-buffered saline (PBS)) or experimental (hUMSC-Exo in PBS) groups, and interventions were intravenously administered via the tail vein four hours post-reperfusion (Figure 2A). Seventy-two hours post-reperfusion, neurological function scores were examined. Lower Bederson scale and higher grip strength test scores (Figure 2C, P < 0.05) were observed in the experimental group. After experimental animal euthanasia, brain slices were obtained to assess infarct volume via 2,3,5-triphenyltetrazolium chloride (TTC) staining, demonstrating smaller infarct size in the experimental group (Figure 2B, p < 0.05). Additionally, the area of ischemic penumbra was smaller in the experimental group (Figure 2D). Furthermore, hematoxylin and eosin (H&E) staining demonstrated lower levels of tissue edema and cell edema and fewer contracted nuclei in the experimental group (Figure 2E). Immunohistochemical detection of IL-6 and NFκB in the ischemic penumbra demonstrated significantly lower expression of these proteins in the experimental group (Figure 2F, p < 0.05). Finally, infiltration of PKH26 (red)-labeled intravenous Exos into the site of brain injury was demonstrated by fluorescence microscopy (Figure 2G). Taken together, these results suggest that circulating hUMSC-Exos infiltrate the relevant anatomical site and are protective against I/R injury after ischemic stroke, in part by decreasing levels of local neuroinflammation.
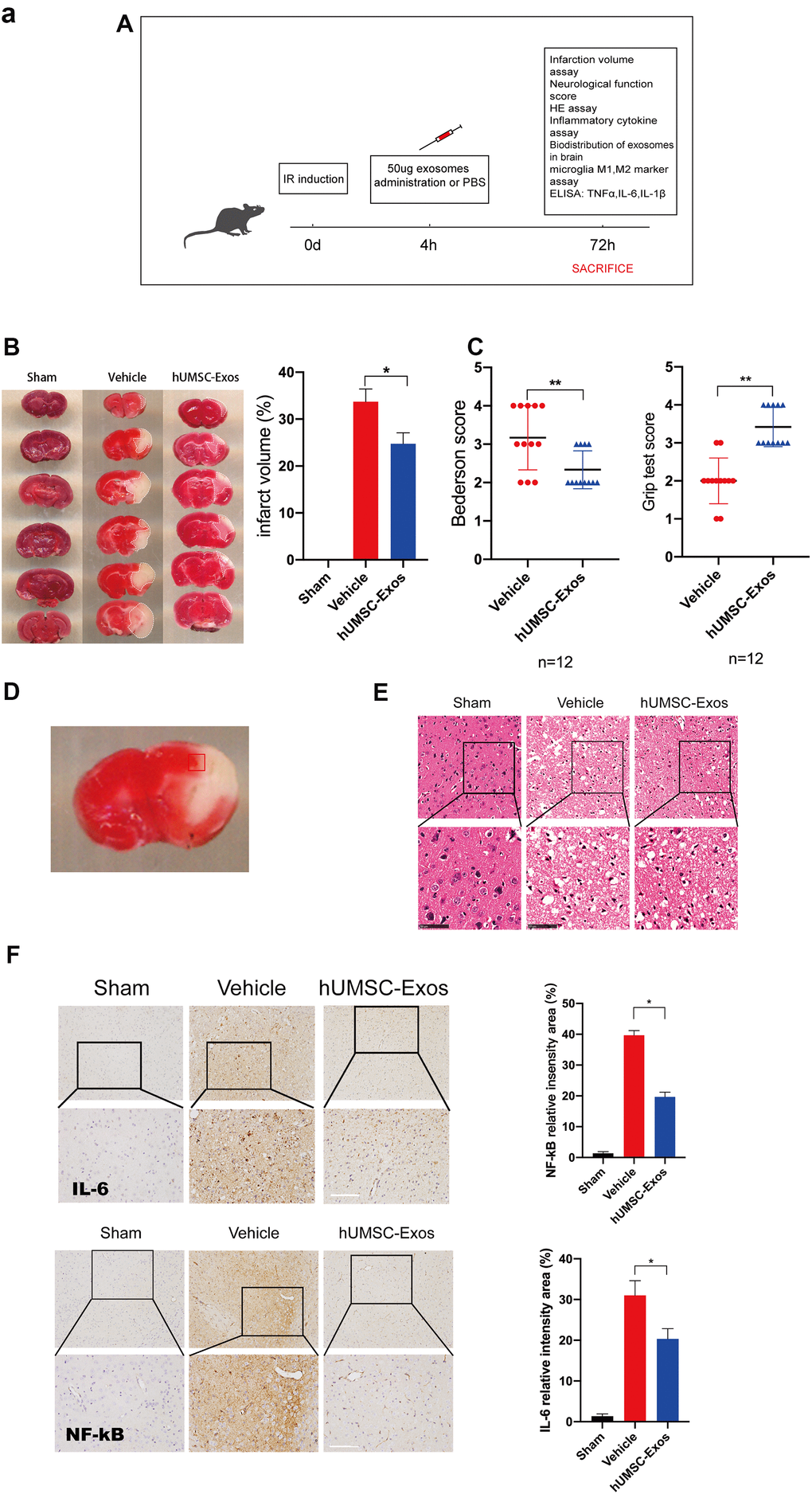
Figure 2. Treatment with hUMSC-Exos attenuates microglia-mediated inflammation and neurological deficits after ischemic stroke. (A) Schematic of the protocol. (B) Representative photomicrographs of TTC-stained tissue from the control, vehicle-only, and experimental groups, with associated infarct size as calculated using ImageJ software. Data are expressed as mean ± SEM (n = 12 per group). Significant differences are indicated (*p < 0.05). (C) Neurological deficit scores in the vehicle-only and experimental groups 72 hours post-reperfusion. Data are expressed as mean ± SEM (n = 12 per group). Significant differences are indicated (*p < 0.05, **p < 0.01). (D) The red box indicates the cerebral ischemic penumbra. (E) H&E staining. Scale bar: 50 μm. (F) Representative photomicrographs of IL-6 and NFκB in the ischemic penumbra 72 hours post-reperfusion, with associated relative intensities as calculated using ImageJ software. Scale bar: 50 μm. Data are expressed as mean ± SEM (n = 6 per group). Significant differences are indicated (*p < 0.05).
(G) Red fluorescence indicates PKH26-labeled exosomes which have accessed the site of cerebral damage. Scale bar: 50 μm. (H) Microglial M1 markers IBA-1 and CD16 in the ischemic penumbra 3 days following ischemic stroke, in the control, vehicle-only, and experimental groups. Scale bar: 50 μm. Associated M1 counts are shown (A, B). (I) Microglial M2 markers IBA-1 and CD206 in the ischemic penumbra 3 days following ischemic stroke, in the control, vehicle-only, and experimental groups. Scale bar: 50 μm. Associated M2 counts - from the same animals in which M1 counts were determined - are shown (C, D). Significant differences are indicated (*p < 0.05). (J) Lower protein levels of pro-inflammatory cytokines IL-6, TNF-α, and IL-1β in the experimental group. Data are expressed as mean ± SEM (experiments were performed in triplicate). Significant differences are indicated (*p < 0.05, **p < 0.01).
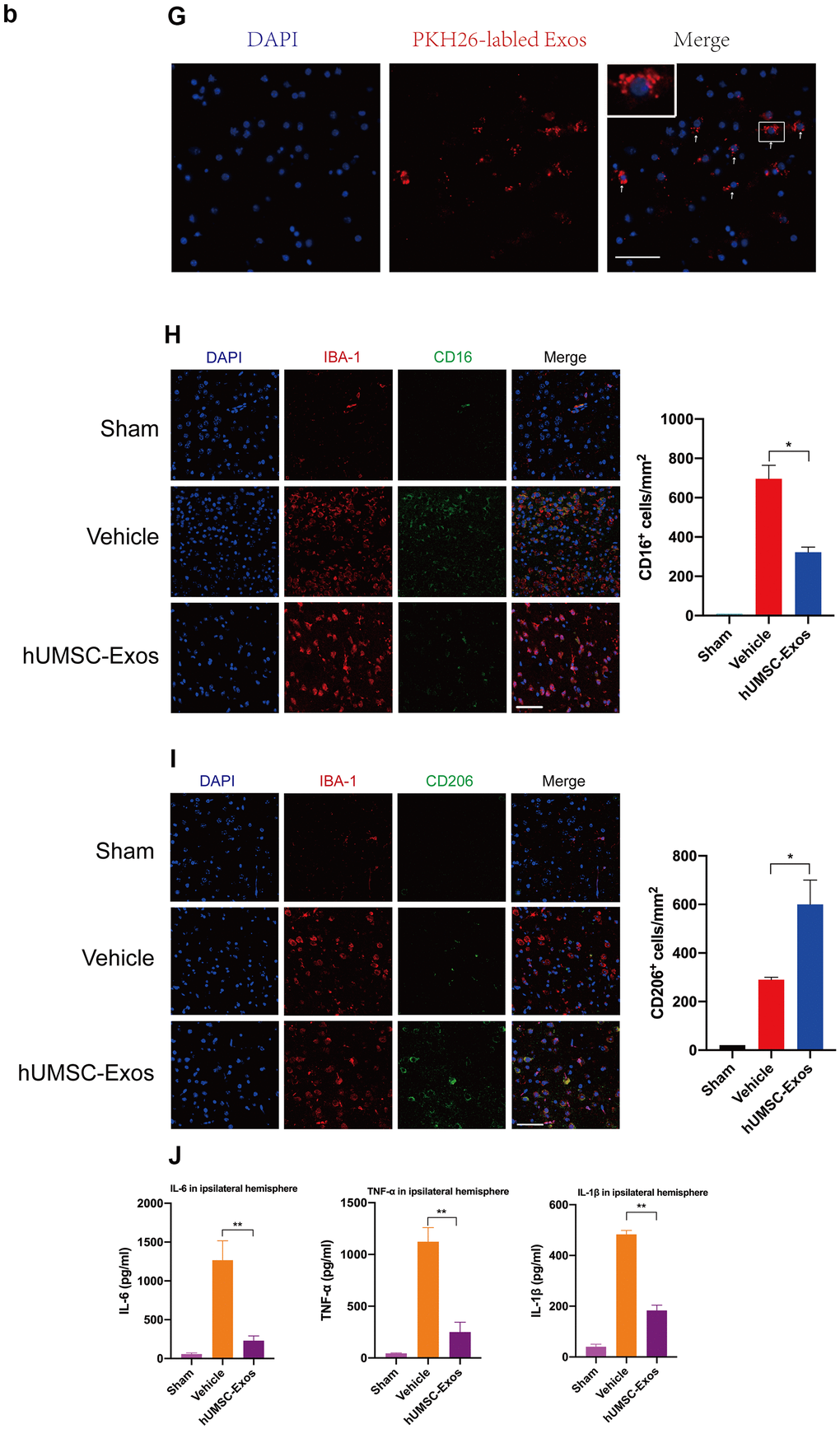
Figure 2. Treatment with hUMSC-Exos attenuates microglia-mediated inflammation and neurological deficits after ischemic stroke. (G) Red fluorescence indicates PKH26-labeled exosomes which have accessed the site of cerebral damage. Scale bar: 50 μm. (H) Microglial M1 markers IBA-1 and CD16 in the ischemic penumbra 3 days following ischemic stroke, in the control, vehicle-only, and experimental groups. Scale bar: 50 μm. Associated M1 counts are shown (A, B). (I) Microglial M2 markers IBA-1 and CD206 in the ischemic penumbra 3 days following ischemic stroke, in the control, vehicle-only, and experimental groups. Scale bar: 50 μm. Associated M2 counts - from the same animals in which M1 counts were determined - are shown (C, D). Significant differences are indicated (*p < 0.05). (J) Lower protein levels of pro-inflammatory cytokines IL-6, TNF-α, and IL-1β in the experimental group. Data are expressed as mean ± SEM (experiments were performed in triplicate). Significant differences are indicated (*p < 0.05, **p < 0.01).
We next examined the effect of hUMSC-Exos on activated microglia in vivo. Treatment with hUMSC-Exos markedly decreased the presence of IBA-1+CD16+ cells at 72 h post-stroke (Figure 2H, p < 0.05) but markedly increased the presence of IBA-1+CD206+ cells at 72h post-stroke (Figure 2I, p < 0.05). Since IBA-1 is a marker of brain microglia [37], CD16 is an M1 marker [38], and CD206 is an M2 marker [39], this suggests that hUMSC-Exos treatment decreased and increased, respectively, the number of M1 and M2 microglia. Expression of pro-inflammatory cytokines IL-6, TNF-α, and IL-1β was also significantly decreased in the experimental group (Figure 2J, p < 0.01). Taken together, these results suggest that hUMSC-Exos may decrease microglia-mediated neuroinflammation after ischemic stroke in mice.
Microglial pro-inflammatory activity is also decreased by hUMSC-Exos in vitro
First, we tested whether red fluorescent dye (PKH26)-labeled hUMSC-Exos are internalized during co-culture with BV2 microglia. After 6 h, microglia had efficiently internalized hUMSC-Exos as indicated by intracellular fluorescence (Figure 3A). To further validate the direct effects of hUMSC-Exo on activated microglia, the latter were cultured in serum-free medium for 6 h in a hypoxic incubator (to mimic oxygen-glucose deprivation (OGD)) prior to culture in conventional medium with or without hUMSC-Exos. After 24 h, IL-6, TNF-α, and IL-1β intracellular transcription and supernatant protein levels were determined by real-time polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA), respectively. Treatment with hUMSC-Exos significantly decreased both IL-6, TNF-α, and IL-1β transcription and protein levels (Figure 3B, 3C, p < 0.01). Results suggest that hUMSC-Exos decrease microglial pro-inflammatory activity in vitro.
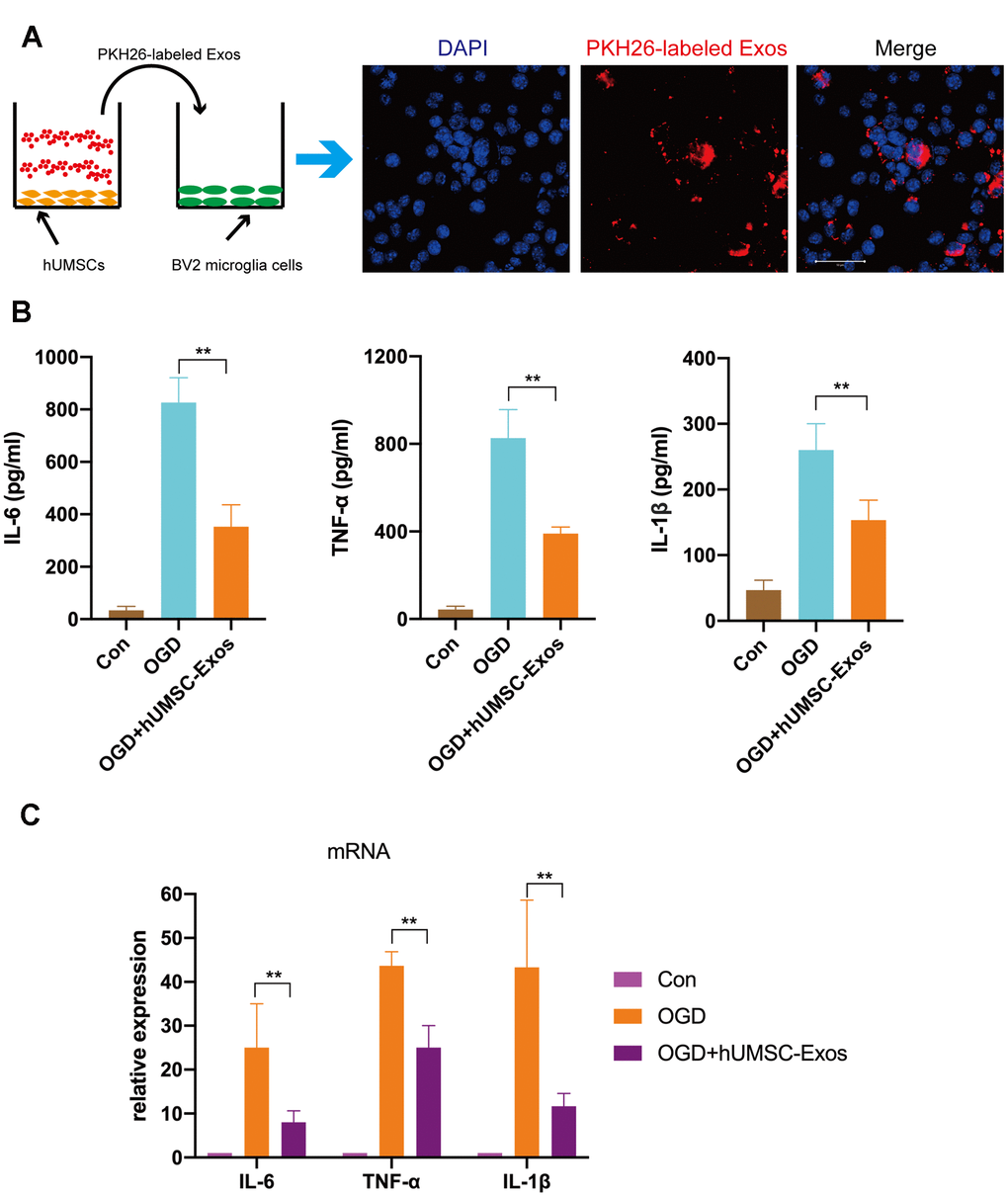
Figure 3. Treatment with hUMSC-Exos reduces microglial pro-inflammatory activity in vitro. (A) Confocal imaging demonstrating uptake of PKH-26-labeled exosomes (red) by BV2 microglia. Scale bar: 50 μm. (B) Lower protein levels of pro-inflammatory cytokines IL-6, TNF-α, and IL-1β in the hUMSC-Exos treatment group. (C) Levels of IL-6, TNF-α, and IL-1β mRNA as detected using qRT-PCR. Data are expressed as mean ± SEM (experiments were performed at least in triplicate). Significant differences are indicated (*p < 0.05, **p < 0.01).
Microglial pro-inflammatory activity is attenuated by hUMSC-Exosomal miRNAs in vitro
In an attempt to elucidate at least one mechanism by which hUMSC-Exos modulate microglial activity, we investigated whether hUMSC-Exosomal miRNAs attenuate microglial pro-inflammatory activity in vitro. To demonstrate that miRNAs are a key functional component of these Exos, we conducted small interfering RNA (siRNA) knockdown of Drosha (an essential polymerase required for miRNA synthesis) in hUMSC (Figure 4A, 4B) in order to generate miRNA-depleted hUMSC-Exos (Figure 4C, 4D). Treatment of activated microglia with wild-type or Drosha-knockdown hUMSC-Exos demonstrated that miRNA depletion significantly weakened the anti-inflammatory effect of hUMSC-Exos (Figure 4E). Results suggest that hUMSC-Exosomal miRNAs contribute to microglial modulation.
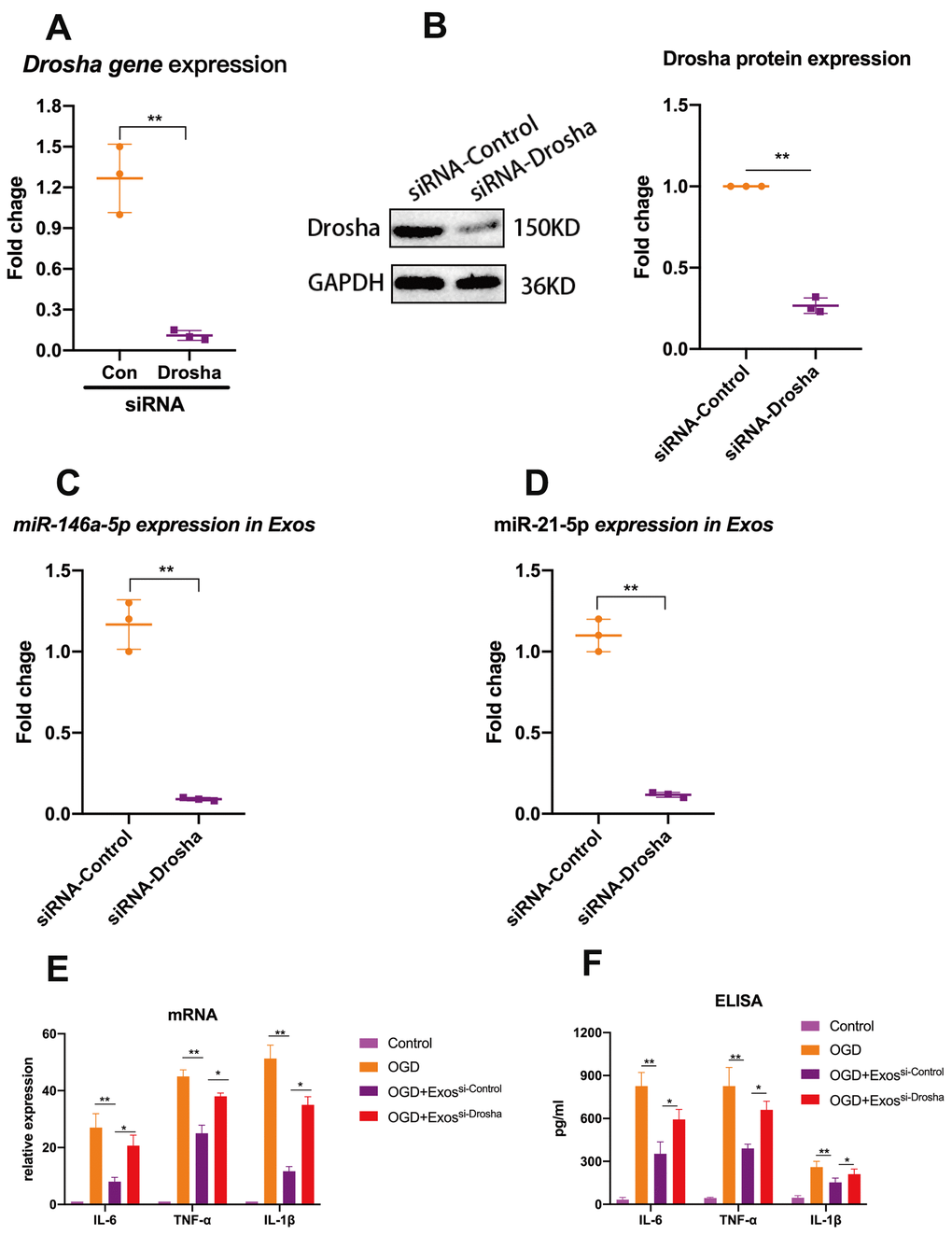
Figure 4. Exosomal miRNAs are implicated in hUMSC-Exos-mediated attenuation of microglial pro-inflammatory activity. (A, B) After 24 hours’ siRNA-Drosha transfection, hUMSC Drosha knockdown efficiency was evaluated by qPCR quantitation of Drosha mRNA and western blot-based quantitation of Drosha protein. Western blots are representative of three independent experimental replicates. (C, D) Exosomal miR-146a-5p and miR-21-5p content was significantly decreased after Drosha knockdown. (E) Protein levels of the pro-inflammatory cytokines IL-6, TNF-α, and IL-1β in hUMSC-Exos were decreased after Drosha knockdown. (F) Detection of IL-6, TNF-α, and IL-1β mRNA levels via qRT-PCR. Data are expressed as mean ± SEM. (A-F) Each experiment is representative of n = 3 per group. Significant differences are indicated (*p < 0.05, **p < 0.01).
Specifically, hUMSC-Exosomal miR-146a-5p attenuates microglial pro-inflammatory activity in vitro through suppression of the IRAK1/TRAF6 signaling pathway
To determine which hUMSC-Exosomal miRNAs may contribute to microglial polarization and attenuation of microglial pro-inflammatory activity, BV2 microglia were exposed to OGD with or without subsequent hUMSC-Exos treatment for 24 hours, and hUMSC-Exosomal small RNA expression analysis and deep sequencing were performed. The following formula was used to calculate corrected miRNA expression: reads per million (RPM) = (number of reads mapping to miRNA/number of clean reads) *106. To the best of our knowledge, this is the first study to report sequencing of hUMSC-Exos-derived miRNAs. The analysis revealed that several hundred miRNA species are present within hUMSC-Exos. Among the top ten miRNAs identified as significantly differentially expressed between hUMSC-Exos treated and untreated microglia (Figure 5A), miR-146a-5p was higher in the treatment group and is known to modulate inflammation [40]. As demonstrated by PCR, treatment with hUMSC-Exos significantly increases BV2 microglial miR-146a-5p content (Figure 5B). Results suggest that hUMSC-Exosomal miR-146a-5p is internalized by microglia (or that hUMSC-Exos induce microglial miR-146a-5p expression) and may contribute to modulating inflammation. The in vitro experimental scheme is pictured (Figure 5C). Western blot analysis demonstrated decreased IL-6, TNF-α, and IL-1β, as well as IRAK1/TRAF6 signaling pathway member IRAK1, TRAF6, and NFκB (p65) protein expression after hUMSC-Exos treatment. Furthermore, expression of IL-6, TNF-α, and IL-1β was higher after treatment with miR-146a-5p knockdown hUMSC-Exos (Figure 5D–5F). Results suggest that hUMSC-Exosomal miR-145-5p contributes to modulating OGD-induced microglial pro-inflammatory activity via suppression of the IRAK1/TRAF6 signaling pathway.
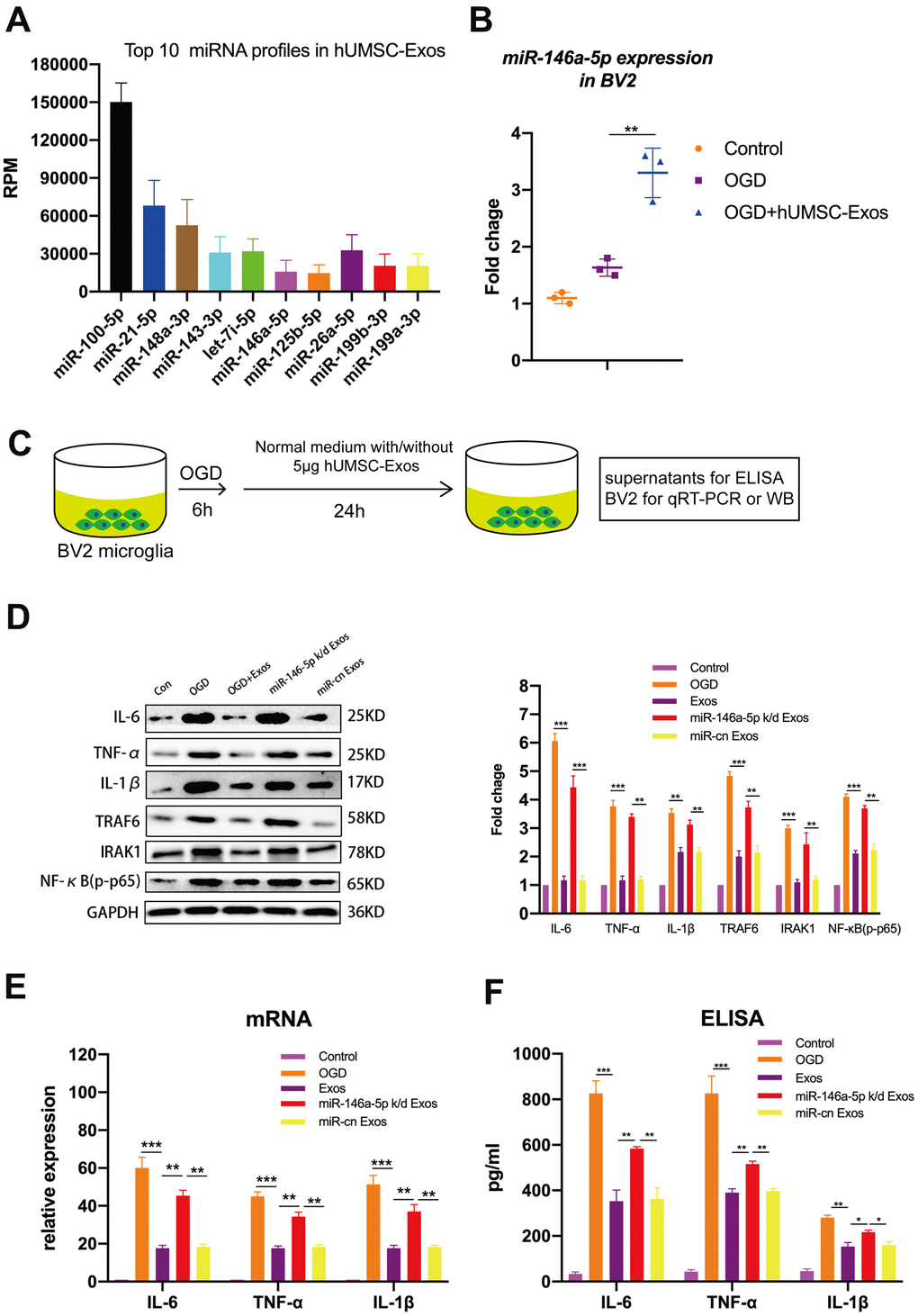
Figure 5. Exosomal miR-146a-5p decreases microglial pro-inflammatory activity by suppressing the IRAK1/TRAF6 signaling pathway in vitro. (A) Expression levels of the top ten hUMSC-Exosomal miRNAs, including MiR-146a-5p. (B) After post-OGD exposure to hUMSC-Exos, BV2 microglia exhibited significantly increased miR-146a-5p content. Data were normalized to levels of U6. (C) In vitro experimental scheme. (D) Expression of pro-inflammatory cytokines IL-6, TNF-α, and IL-1β, as well as signaling pathway IRAK1, TRAF6, and NFκB (p65) in microglia treated with wild-type versus miR-146a-5p knockdown hUMSC-Exos. (E) Determination of IL-6, TNF-α, and IL-1β mRNA levels via qRT-PCR. (F) Determination of supernatant IL-6, TNF-α, and IL-1β protein levels via ELISA. Data are expressed as mean ± SEM. (A–F) Each experiment is representative of n = 3 per group. Significant differences are indicated (*p < 0.05, **p < 0.01, ***p < 0,001).
Microglia-mediated neuroinflammation and neural deficits resulting from ischemic stroke are attenuated by hUMSC-Exosomal miR-146a-5p in mice
To investigate whether hUMSC-Exosomal miR-146a-5p exerts neuroprotective effects in the ischemic mouse brain via IRAK1/TRAF6 signaling pathway modulation, both wild-type and miR-146a-5p knockdown hUMSC-Exos were administered in a murine ischemic stroke model. Relative to treatment with miR-146a-5p knockdown hUMSC-Exos, treatment with wild-type hUMSC-Exos significantly reduced infarct volume three days post-ischemia (Figure 6A) and resulted in lower Bederson scale scores and higher grip strength test scores (Figure 6B, 6C). Results suggest that hUMSC-Exosomal miR-145a-5p may attenuate I/R damage and neural deficits after ischemic stroke in a murine model.
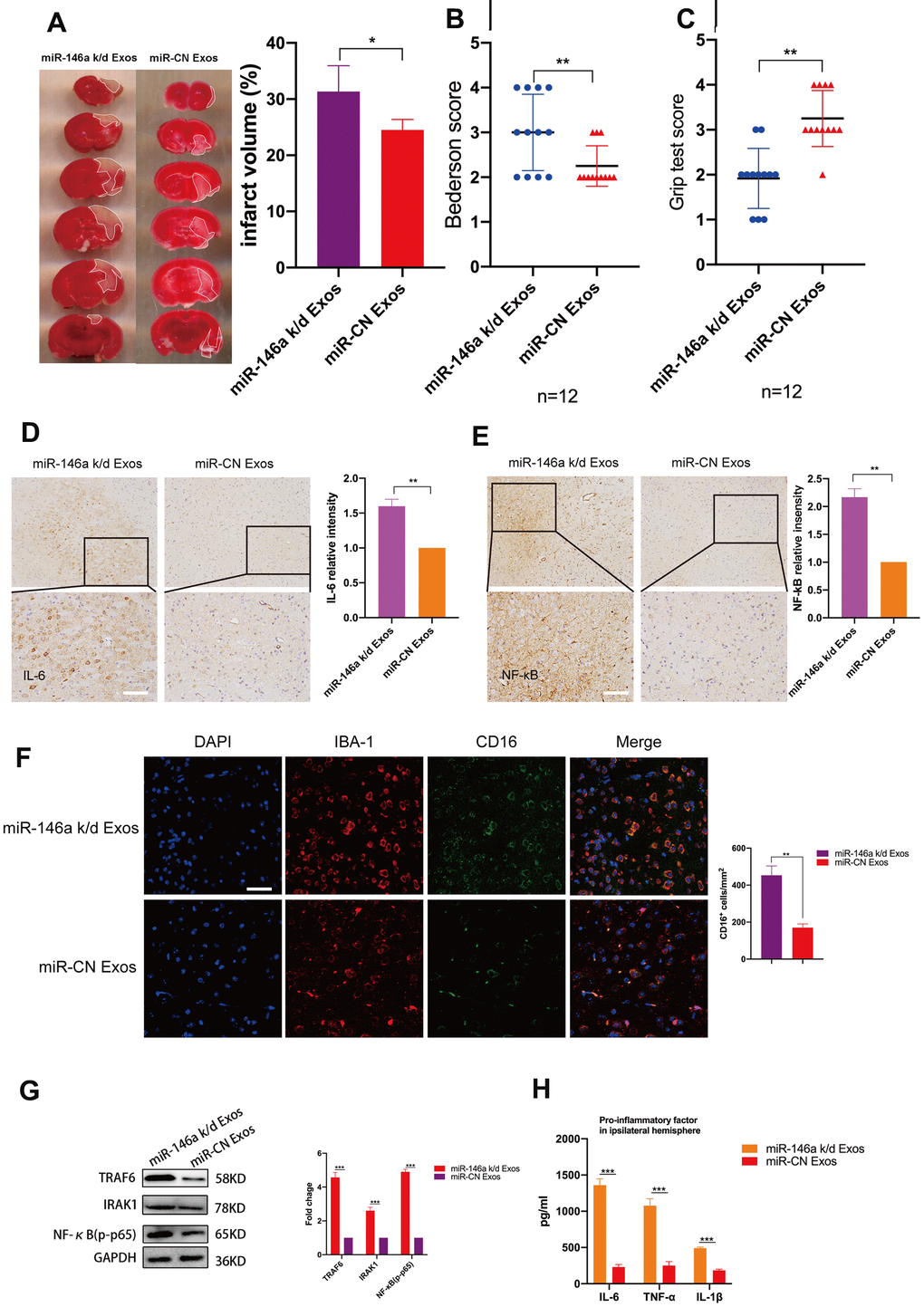
Figure 6. Treatment with hUMSC-Exos decreases neuroinflammation and is neuroprotective by down-regulating IRAK1/TRAF6 signaling pathway activity in vivo. (A) Representative photomicrographs of TTC-stained tissue from wild-type versus miR-146a-5p knockdown hUMSC-Exos groups, with infarct size as calculated using ImageJ software. Data are expressed as mean ± SEM (n = 6 per group). Significant differences are indicated (*p < 0.05). (B, C) Neurological deficit scores in vehicle-only versus experimental groups at 72 hours post-reperfusion. Data are expressed as mean ± SEM (n = 12 per group). Significant differences are indicated (*p < 0.05, **p < 0.01). (D, E) Representative photomicrographs of IL-6 and NFκB in the ischemic penumbra 72 hours post-reperfusion, with associated relative intensities as calculated using ImageJ software. Scale bar: 50 μm. Data are expressed as mean ± SEM (n = 6 per group). Significant differences are indicated (*p < 0.05). (F) Microglial M1 markers IBA-1 and CD16 in the ischemic penumbra 3 days following ischemic stroke. (G) Expression of signaling pathway IRAK1, TRAF6, and NFκB (p65) in the wild-type versus miR-146a-5p knockdown groups. (H) Determination of IL-6, TNF- α, and IL-1β protein levels via ELISA. Data are expressed as mean ± SEM (experiments were performed in triplicate). Significant differences are indicated (*p < 0.05, **p < 0.01, ***p < 0,001).
Immunohistochemistry demonstrated that levels of IL-6 and NFκB in the ischemic penumbra were significantly lower in the group treated with wild-type hUMSC-Exos (Figure 6D, 6E, p < 0.01). Furthermore, immunofluorescent labeling demonstrated that significantly fewer microglia were activated in the group treated with wild-type hUMSC-Exos (Figure 6F, p < 0.01). Western blots demonstrated that expression of IRAK1/TRAF6 signaling pathway proteins IRAK1, TRAF6, and NFκB (p65) was significantly decreased in the group treated with wild-type hUMSC-Exos (Figure 6G, p < 0.001). Finally, levels of IL-6, TNF-α, and IL-1β were significantly reduced in the group treated with wild-type hUMSC-Exos (Figure 6H, p < 0.001). Results suggest that hUMSC-Exosomal miR-146a-5p may attenuate microglia-mediated neuroinflammation after ischemic stroke in a murine model.
Discussion
In vivo, OGD activates microglial-mediated inflammatory response [41]. During the acute period after stroke, microglia secrete pro-inflammatory cytokines IL-6, TNF-α, and IL-1β [38], which can induce secondary cytotoxicity. Studies have proven that decreasing microglia-mediated neuroinflammation is beneficial during stroke recovery [13, 17]. Although MSC transplantation is neuroprotective after both traumatic brain injury and stroke, at least in part via modulating microglia-mediated neuroinflammation, mechanisms of the latter remain incompletely understood. However, since over 99% of transplanted MSCs become entrapped in the pulmonary vasculature without impeding therapeutic effect, mechanisms likely involve distally acting MSC-produced paracrine factors which may hold promise as cell-free therapies. Nearly all cell types secrete exosomes, which are important mediators of cellular communication.
One important group of Exos cargo molecules is miRNAs (short non-coding RNAs that inhibit target gene expression by directly binding their mRNAs) [42]. Prior studies report that Exos play an important role in transmitting miRNAs between cells [43–46], via interstitial fluid and circulation delivering biologically active miRNAs to both neighboring and distant cells [47]. For example, adipose tissue constitutes an important source of circulating exosomal miRNAs that serve as a previously unrecognized form of adipokine to regulate gene expression in distant tissues [48], adipose tissue-resident macrophage-derived exosomal miRNAs modulate insulin sensitivity [43], and exosomal transfer of miR-181b from cardiosphere-derived cells (CDCs) into macrophages reduces PKCδ transcription (a mechanism underlying the post-reperfusion cardioprotective effects of CDCs) [49]. The present study investigated the potential therapeutic role hUMSC-Exos in ameliorating I/R injury.
Results suggest that in a murine model of ischemic stroke, quality-controlled hUMSC-Exos delivered intravenously four hours post-reperfusion are able to traverse the blood-brain barrier to access the site of ischemic injury and are then taken up by local microglia, in which exosome-derived miR-146a-5p inhibits IRAK1/TRAF6 signaling pathway-mediated NFκB activation and consequent M1 polarization and production of potent pro-inflammatory cytokines (instead favoring M2 polarization), ultimately resulting in decreased: I/R-induced tissue edema, cell death, extent of the ischemic infarct and penumbra, and functional motor deficits.
This is consistent with prior studies which have demonstrated that immunofluorescently labeled Exos are detectable both extracellularly and intracellularly at sites of brain injury [50] and are taken up by recipient cells [50, 51]. It has also previously been demonstrated that MSC-derived Exos exert powerful effects in the context of ischemic stroke, for example ameliorating inflammation-induced astrocyte alterations [52]. Regarding mechanisms, pro-neurogenic effects of UMSC-Exos may be partially attributable to histone deacetylase 6 (HDAC6) inhibition by exosomal miR-26a [53]. Furthermore, it is known that without miRNA-184 and -210, MSC-derived extracellular vesicles lose the ability to promote neurogenesis and angiogenesis [54]. Although it is difficult to completely exclude the effects of other exosomal cargo molecule groups on microglia in the present study, miRNAs are considered a key functional element. The miRNA miR-146a-5p is a well-known anti-inflammatory molecule with a key role in inflammatory disorders [55–57]. The receptor proteins IRAK1 and TRAF6 are abundant in the cytoplasm and nucleus of various cell types [58]. They are largely involved in Toll-like receptor (TLR)-initiated pathways, leading to expression of pro-inflammatory mediators [59]. Overexpression of IRAK1 and TRAF6 can activate NFκB [60], a transcription factor which is a key activator of pro-inflammatory gene expression programs [57]. Through binding the 3′UTR of the mRNAs encoding IRAK1 and TRAF6, miR-146a-5p down-modulates inflammatory responses [61, 62].
Apart from recommending follow-up research into the therapeutic potential of cell-free miR-146a-5p in the context of I/R injury and inflammatory disorders, results raise a number of interesting theoretical questions. For example, it is unknown whether hUMSC-Exosomal cargo is also perhaps involved in regulating glial ion transporters (which are involved in the Warburg effect, glial activation, neuroinflammation, and neuronal damage during glioma [63]). Additionally, might other miRNAs impact microglial function after ischemic stroke? Might hUMSC-Exos also impact the function of other local cell types (e.g. neurons, astrocytes, and/or oligodendrocytes) after ischemic stroke? Finally, by which mechanisms might such effects occur?
Conclusions
In conclusion, hUMSC-Exos and or miR-146a-5p represent novel therapeutic options for the improvement of outcomes after ischemic stroke (Figure 7), warranting further investigation.
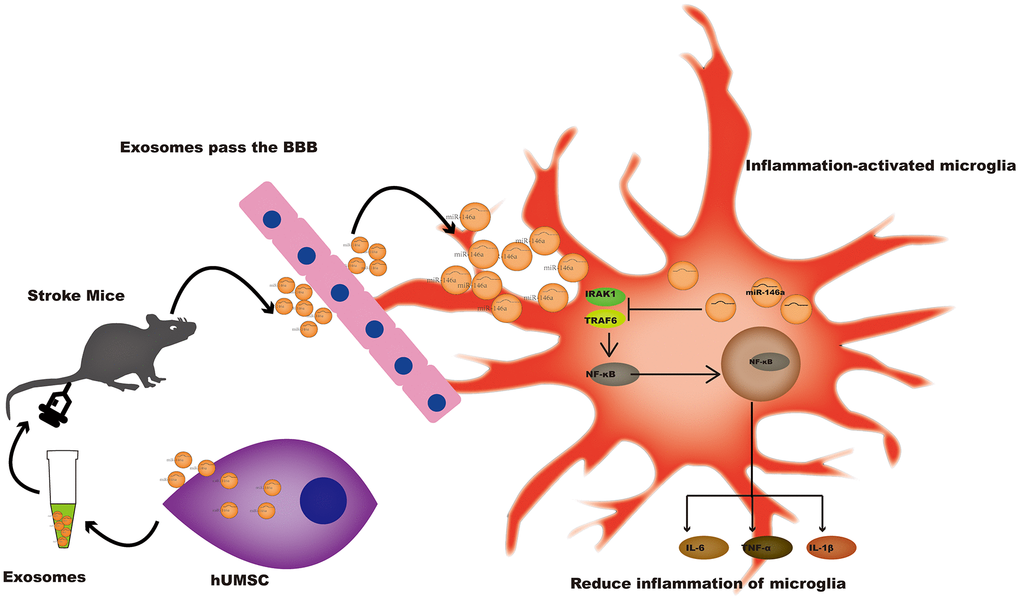
Figure 7. A potential mechanism contributing to the hUMSC-Exos-induced decrease in microglia-mediated neuroinflammation after ischemic stroke. After injection of hUMSC-Exos into the tail vein of the murine ischemic stroke model, they traversed the blood-brain barrier and were internalized by microglia at the site of cerebral injury. Exosomal miR-146a-5p may decrease microglia-mediated neuroinflammation by suppressing the IRAK1/TRAF6 signaling pathway.
Materials and Methods
Animals
Experimental animals (C57BL/6 mice aged 8 weeks, weighing 20-30 g each) were purchased from the Animal Experiment Center of Southern Medical University (Guangzhou, China), fed a standard laboratory diet with ad libitum access to food and water, and maintained at a temperature of 22 ± 1° C and a humidity of 65–70 % in a controlled room with a 12 h light–dark cycle. All experimental procedures were approved by the Southern Medical University Ethics Committee and performed in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Murine model of ischemic stroke
As described previously [64], mice were anesthetized via intraperitoneal injection (35 mg/kg sodium pentobarbital). Transient middle cerebral artery occlusion (MCAO) was produced by advancing a 4-0 nylon monofilament (0.23–0.25 mm) (Yushun Bio Technology Co. Ltd., China) via the left common carotid artery to occlude the middle cerebral artery for 60 minutes, prior to filament withdrawal (reperfusion). Success of blood-flow occlusion and restoration was verified using a laser Doppler flowmeter (Moor LAB, Moor Instruments, Devon, UK). During the MCAO procedure, head temperature was maintained at 36° C. At four hours post-reperfusion, 250 μL PBS with or without 50 μg hUMSC-Exos was injected into the tail vein (experimental and vehicle-only groups). Mice in a third (control) group received no injection.
Evaluation of motor function
At 24 hours post-reperfusion, global neurological and motor function was assessed (Table 1) using a modified Bederson Scale and grip strength evaluation [65] by a researcher blinded to group allocation. For grip strength evaluation, taut string (50 cm) was suspended between two vertical supports at a height of 40 cm. Each mouse was placed midway on the string and rated as shown in Table 1.
Table 1. Neurological function score.
| Score | A modified Bederson score | Score | The grip test |
| 0 | No deficit. | 0 | Falls off. |
| 1 | Forelimb flexion. | 1 | Hangs onto string by one or both forepaws. |
| 2 | As for 1, plus decreased resistance to lateral push. | 2 | As for 1, and attempts to climb onto string. |
| 3 | Unidirectional circling. | 3 | Hangs onto string by one or both forepaws plus one or both hind paws. |
| 4 | Longitudinal spinning or seizure activity. | 4 | Hangs onto string by fore and hind paws plus tail wrapped around string. |
| 5 | No movement. | 5 | Escape (to the supports). |
Evaluation of cerebral infarct volume
At 72 hours post-reperfusion, mice were euthanized. Thereafter, the brain was removed for coronal sectioning. Infarct size was measured using 2% 2,3,5-triphenyltetrazolium chloride (TTC) staining in conjunction with microscopy. Infarct volume was evaluated by a blinded observer using ImageJ software version 1.61 (National Institutes of Health, Bethesda, MD, USA).
H&E staining
Fresh brain tissues were fixed using 4% PFA (pH 7.4), gradually dehydrated, embedded in paraffin, cut into 4-μm-thick sections using a microtome, and stained with H&E to visualize cellular structures by microscopy.
Immunofluorescent staining
Four-micron-thick coronal brain sections were deparaffinized in xylene, rehydrated via an alcohol gradient, and washed with PBS (0.01 M, pH 7.4). Sections were blocked using 5% bovine serum albumin (BSA) for 60 min at room temperature prior to overnight incubation at 4° C with the following primary antibodies: goat anti-IBA-1 (1:200 dilution; ab48004, Abcam), rabbit anti-CD16 (1:200 dilution; ab252908, Abcam), and goat anti-CD206 (1:200 dilution, R&D Systems). Automated image analysis was performed using ImageJ software version 1.61 (National Institutes of Health, Bethesda, MD, USA). Cell numbers were calculated per square millimeter from three random microscopic fields (200 × magnification) on three sections (a total of nine images) (n = 6 animals per group). All counts were performed in a blinded manner.
Cell types and cell culture methods
Human umbilical cords were provided by Guangzhou Saliai Stem Cell Science and Technology Co. Ltd. for hUMSC isolation. Thereafter, hUMSCs were cultured in complete Dulbecco’s modified Eagle’s medium (DMEM)/F12 (Gibco) supplemented with 10% fetal bovine serum (FBS) (Gibco, Australia), and cells between the 3rd and 5th generations were used for subsequent experiments. The BV2 murine microglial cell line was obtained from Xiehe Medical University (Beijing, China). Microglia were cultured in DMEM supplemented with 10% heat-inactivated FBS, 100 μg/mL streptomycin, and 100 U/mL penicillin (HyClone).
Exosome isolation, characterization, and treatment
After hUMSCs were cultured in DMEM/F12 medium supplemented with 10% (FBS), culture supernatants containing Exos were centrifuged at 2,000 x g for 20 min at "4° C, followed by 10, 000 x g for 30 min at 4° C, and the supernatant was passed through a 0.22 μm filter (Millipore), to pellet and exclude contaminating dead cells and debris. Thereafter, the filtrate was subjected to ultracentrifugation at 110,000 g for 70 min at 4° C to pellet Exos. Pellets were washed with PBS, followed by ultracentrifugation at 110,000 × g for 70 min at 4° C to re-pellet Exos, which were then resuspended in PBS. The bicinchoninic acid (BCA) Protein Assay kit (KeyGEN BioTECH) was used to estimate Exos concentration. For transmission electron microscopy (JEM-1200EX, JEOL Ltd.), 5-10 μl of each sample was added to a copper mesh and precipitated for 3 min. Remaining liquid was carefully pipetted from the filter paper edge. Thereafter, filter paper was rinsed with PBS and phosphotungstic acid was used for negative staining prior to drying at room temperature for 2 min and imaging (operating voltage: 80-120 kV).
Determining Exos internalization by microglia via fluorescence microscopy
Exos were labeled using the red fluorescent membrane dye PKH67 (Sigma). Labeled Exos were washed by resuspension in 10 mL PBS, pelleted by ultracentrifugation as described above, and finally resuspended in 100 μl PBS. For cell treatment, a volume of suspension corresponding to 2 μg of Exos was added to 2×105 recipient cells prior to 24 hours of incubation.
In vitro OGD and “reperfusion” model
An anaerobic chamber containing 95% N2 and 5% CO2 was used in conjunction with deoxygenated glucose-free DMEM (Gibco) was used to simulate microglial OGD. After 6 hours, culture medium was replaced with maintenance medium, and cells were moved to a regular incubator to recover for 24 hours.
RT-PCR
Total RNA was extracted from cells and brain tissues using Trizol reagent (Life Technologies) and from Exos using an Exosome RNA Purification Kit (EZB-exo-RN1), both according to the manufacturer’s instructions. Reverse transcription was performed using the PrimeScript RT reagent Kit (RR037A, Takara Bio Inc., Shiga, Japan). Real-time PCR was conducted using SYBR Green PCR Master Mix (Applied TaKaRa, Otsu, Shiga, Japan) in conjunction with an Applied Biosystems 7500 Fast Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). Sequences of all primers are provided (Table 2).
Table 2. Primers used for real-time PCR.
| Genes | Primer sequences |
| IL-6 | FORWARD ACTTCCATCCAGTTGCCTTCTTGG |
| REVERSE TTAAGCCTCCGACTTGTGAAGTGG | |
| TNF-α | FORWARD GCGACGTGGAACTGGCAGAAG |
| REVERSE GCCACAAGCAGGAATGAGAAGAGG | |
| IL-1β | FORWARD ACTTCCATCCAGTTGCCTTCTTGG |
| REVERSE TGCTCATGTCCTCATCCTGGAAGG | |
| DROSHA | FORWARD AAGGCAAGACGCACAGGAATTAGG |
| REVERSE TCTGCCAGCATTGTTGGTCATAGG | |
| Arg-1 | FORWARD CATATCTGCCAAAGACATCGTG |
| REVERSE GACATCAAAGCTCAGGTGAATC | |
| miR-21-5p | FORWARD GCGCGTAGCTTATCAGACTGA |
| REVERSE AGTGCAGGGTCCGAGGTATT | |
| miR-146a-5p | FORWARD CGCGTGAGAACTGAATTCCA |
| REVERSE AGTGCAGGGTCCGAGGTATT | |
| U6 | FORWARD CTCGCTTCGGCAGCACA |
| REVERSE AACGCTTCACGAATTGCGT |
Quantitation of supernatant cytokines by ELISA
Concentrations (pg/mL) of IL-6, TNF-α, and IL-1β in BV2 microglia culture supernatants and damaged cerebral tissue were determined via ELISA (kits from R&D Systems, Minneapolis, MN, USA), performed as per manufacturer instructions. Briefly, standards and samples were added to a 96-well ELISA plate pre-coated with biotinylated anti-IL-6, anti-TNF-α, and anti-IL-1β. Unbound substances were washed away, and enzyme-linked polyclonal antibodies specific for IL-6, TNF-α, and IL-1β were added to the corresponding wells. Plates were incubated for 2 hours, washed four times, and enzyme substrate was added prior to 30 min incubation. Color development was terminated using stop solution and absorbance at 450 nm was determined using a microplate reader. The concentration of each sample was calculated from a standard curve prepared using the cytokine standards. Each experiment was performed in triplicate.
Transfection with siRNA
In order to decrease miRNA synthesis, siRNA-Drosha (Ribobio) was transfected into recipient hUMSC using riboFECTTMCP Reagent (Ribobio). After 24 hours, transfection efficiencies were evaluated via qPCR or western blots.
Western blots
Tissues, cells, and Exos were lysed using RIPA buffer (KeyGEN BioTECH), followed by protein quantitation using a Bradford Protein Assay kit (KeyGEN BioTECH). Briefly, lysates were subjected to SDS–PAGE and transferred onto PVDF membranes (Millipore). Membranes were incubated overnight at 4° C with primary antibodies specific for the following proteins, as required by each experiment: TSG101 (1:1000 dilution; ab125011, Abcam), CD9 (1:1000 dilution; ab92726, Abcam), ALIX (1:1000 dilution; ab117600, Abcam), IRAK1 (1:1000 dilution; #511166, ZEN BIO), TRAF6 (1:1000 dilution; #380803, ZEN BIO), NFκB (p65) (1:1000 dilution; ab76302, Abcam), GAPDH (1:5000 dilution; 60004-1-Ig, Proteintech), IL-6 (1:1000 dilution; ab6672, Abcam), TNF-α (1:1000 dilution; Ab8348, Abcam), IL-1β (1:1000 dilution; 12242S, CST), and Drosha (1:1000 dilution; ab183732, Abcam). Thereafter, membranes were incubated with the relevant horseradish peroxidase (HRP)-conjugated secondary antibody to visualize protein spots using an enhanced chemiluminescence (ECL) kit (Thermo Scientific). Housekeeping protein GAPDH was selected as an internal control. Each experiment was conducted in triplicate.
Microarray analysis
The miRNA content of hUMSC-Exos (n = 3) total RNA was profiled via small RNA deep sequencing analysis (Illumina). Library preparation and miRNA sequencing were performed by Ribobio (Guangzhou, China). Briefly, total RNA samples were fractionated and only small RNAs (18-30 nucleotides in length) were used for library preparation. After amplification by PCR, products were sequenced using the Illumina HiSeq 2500 platform.
Statistical analysis
All data are expressed as mean ± standard error (SE). Differences between two groups were analyzed by Student’s t-test (two-tailed), while differences between multiple groups were analyzed by one-way ANOVA in conjunction with the Bonferroni/Dunn post hoc test. A p-value < 0.05 was considered statistically significant. All statistical analyses were carried out using GraphPad Prism 8 software.
Ethics approval
All experimental procedures were approved by the Ethics Committee of Southern Medical University and performed in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
Author Contributions
ZFZ, XXZ, RZ, and XDJ designed the experiments; YX, ZMF, FL, and JBH performed the experiments; QOU, BKL, and TH performed data collection and analysis; STH, YPT, and ZZL prepared the manuscript; HTS, YQC, and YXZ provided suggestions regarding experimental procedures and manuscript revision. XDJ and RZ revised the manuscript and provided financial support. All authors approved the final manuscript prior to submission.
Acknowledgments
We thank Guangzhou Saliai Stem Cell Science and Technology Co. Ltd. for providing hUMSCs, and Dr. Yuxin Chen from Southern Medical University for his technical assistance.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was supported by grants from the Funds for the National Natural Science Foundation of China (NSFC) projects (No.81874077), Key Natural Science Foundation of Guangdong (No. 2016B030230004), Key Projects of Health Collaborative Innovation of Guangzhou (No. 201803040016), Guangdong Provincial Department of Education Key Project (No. Guangdong Education Letter (2018) 17) to Prof. Xiaodan Jiang, and Youth Programs of NSFC (No.81701200) to Run Zhang.
References
- 1. Feigin VL, Nguyen G, Cercy K, Johnson CO, Alam T, Parmar PG, Abajobir AA, Abate KH, Abd-Allah F, Abejie AN, Abyu GY, Ademi Z, Agarwal G, et al, and GBD 2016 Lifetime Risk of Stroke Collaborators. Global, regional, and country-specific lifetime risks of stroke, 1990 and 2016. N Engl J Med. 2018; 379:2429–37. https://doi.org/10.1056/NEJMoa1804492 [PubMed]
- 2. Yang JL, Yang YR, Chen SD. The potential of drug repurposing combined with reperfusion therapy in cerebral ischemic stroke: a supplementary strategy to endovascular thrombectomy. Life Sci. 2019; 236:116889. https://doi.org/10.1016/j.lfs.2019.116889 [PubMed]
- 3. Wang J, Zhang W, Lv C, Wang Y, Ma B, Zhang H, Fan Z, Li M, Li X. A novel biscoumarin compound ameliorates cerebral ischemia reperfusion-induced mitochondrial oxidative injury via Nrf2/Keap1/ARE signaling. Neuropharmacology. 2020; 167:107918. https://doi.org/10.1016/j.neuropharm.2019.107918 [PubMed]
- 4. Chen H, Guan B, Wang B, Pu H, Bai X, Chen X, Liu J, Li C, Qiu J, Yang D, Liu K, Wang Q, Qi S, Shen J. Glycyrrhizin prevents hemorrhagic transformation and improves neurological outcome in ischemic stroke with delayed thrombolysis through targeting peroxynitrite-mediated HMGB1 signaling. Transl Stroke Res. 2020; 11:967–82. https://doi.org/10.1007/s12975-019-00772-1 [PubMed]
- 5. Xue J, Yu Y, Zhang X, Zhang C, Zhao Y, Liu B, Zhang L, Wang L, Chen R, Gao X, Jiao P, Song G, Jiang XC, Qin S. Sphingomyelin synthase 2 inhibition ameliorates cerebral ischemic reperfusion injury through reducing the recruitment of toll-like receptor 4 to lipid rafts. J Am Heart Assoc. 2019; 8:e012885. https://doi.org/10.1161/JAHA.119.012885 [PubMed]
- 6. Shi YW, Zhang XC, Chen C, Tang M, Wang ZW, Liang XM, Ding F, Wang CP. Schisantherin a attenuates ischemia/reperfusion-induced neuronal injury in rats via regulation of TLR4 and C5aR1 signaling pathways. Brain Behav Immun. 2017; 66:244–56. https://doi.org/10.1016/j.bbi.2017.07.004 [PubMed]
- 7. Maier CM, Hsieh L, Crandall T, Narasimhan P, Chan PH. Evaluating therapeutic targets for reperfusion-related brain hemorrhage. Ann Neurol. 2006; 59:929–38. https://doi.org/10.1002/ana.20850 [PubMed]
- 8. Kalogeris T, Bao Y, Korthuis RJ. Mitochondrial reactive oxygen species: a double edged sword in ischemia/reperfusion vs preconditioning. Redox Biol. 2014; 2:702–14. https://doi.org/10.1016/j.redox.2014.05.006 [PubMed]
- 9. Gan Y, Liu Q, Wu W, Yin JX, Bai XF, Shen R, Wang Y, Chen J, La Cava A, Poursine-Laurent J, Yokoyama W, Shi FD. Ischemic neurons recruit natural killer cells that accelerate brain infarction. Proc Natl Acad Sci USA. 2014; 111:2704–09. https://doi.org/10.1073/pnas.1315943111 [PubMed]
- 10. Shi K, Tian DC, Li ZG, Ducruet AF, Lawton MT, Shi FD. Global brain inflammation in stroke. Lancet Neurol. 2019; 18:1058–66. https://doi.org/10.1016/S1474-4422(19)30078-X [PubMed]
- 11. Heneka MT, Kummer MP, Latz E. Innate immune activation in neurodegenerative disease. Nat Rev Immunol. 2014; 14:463–77. https://doi.org/10.1038/nri3705 [PubMed]
- 12. Nayak D, Roth TL, McGavern DB. Microglia development and function. Annu Rev Immunol. 2014; 32:367–402. https://doi.org/10.1146/annurev-immunol-032713-120240 [PubMed]
- 13. Hu X, Leak RK, Shi Y, Suenaga J, Gao Y, Zheng P, Chen J. Microglial and macrophage polarization—new prospects for brain repair. Nat Rev Neurol. 2015; 11:56–64. https://doi.org/10.1038/nrneurol.2014.207 [PubMed]
- 14. Liu Q, Zhang Y, Liu S, Liu Y, Yang X, Liu G, Shimizu T, Ikenaka K, Fan K, Ma J. Cathepsin C promotes microglia M1 polarization and aggravates neuroinflammation via activation of Ca2+-dependent PKC/p38MAPK/NF-κB pathway. J Neuroinflammation. 2019; 16:10. https://doi.org/10.1186/s12974-019-1398-3 [PubMed]
- 15. Zheng ZV, Lyu H, Lam SY, Lam PK, Poon WS, Wong GK. The dynamics of microglial polarization reveal the resident neuroinflammatory responses after subarachnoid hemorrhage. Transl Stroke Res. 2020; 11:433–49. https://doi.org/10.1007/s12975-019-00728-5 [PubMed]
- 16. Jackson L, Dong G, Althomali W, Sayed MA, Eldahshan W, Baban B, Johnson MH, Filosa J, Fagan SC, Ergul A. Delayed administration of angiotensin II type 2 receptor (AT2R) agonist compound 21 prevents the development of post-stroke cognitive impairment in diabetes through the modulation of microglia polarization. Transl Stroke Res. 2020; 11:762–75. https://doi.org/10.1007/s12975-019-00752-5 [PubMed]
- 17. Ma Y, Wang J, Wang Y, Yang GY. The biphasic function of microglia in ischemic stroke. Prog Neurobiol. 2017; 157:247–72. https://doi.org/10.1016/j.pneurobio.2016.01.005 [PubMed]
- 18. Nikolakopoulou AM, Dutta R, Chen Z, Miller RH, Trapp BD. Activated microglia enhance neurogenesis via trypsinogen secretion. Proc Natl Acad Sci USA. 2013; 110:8714–19. https://doi.org/10.1073/pnas.1218856110 [PubMed]
- 19. Ouyang Q, Li F, Xie Y, Han J, Zhang Z, Feng Z, Su D, Zou X, Cai Y, Zou Y, Tang Y, Jiang X. Meta-analysis of the safety and efficacy of stem cell therapies for ischemic stroke in preclinical and clinical studies. Stem Cells Dev. 2019; 28:497–514. https://doi.org/10.1089/scd.2018.0218 [PubMed]
- 20. Zhang R, Liu Y, Yan K, Chen L, Chen XR, Li P, Chen FF, Jiang XD. Anti-inflammatory and immunomodulatory mechanisms of mesenchymal stem cell transplantation in experimental traumatic brain injury. J Neuroinflammation. 2013; 10:106. https://doi.org/10.1186/1742-2094-10-106 [PubMed]
- 21. Han J, Feng Z, Xie Y, Li F, Lv B, Hua T, Zhang Z, Sun C, Su D, Ouyang Q, Cai Y, Zou Y, Tang Y, et al. Oncostatin m-induced upregulation of SDF-1 improves bone marrow stromal cell migration in a rat middle cerebral artery occlusion stroke model. Exp Neurol. 2019; 313:49–59. https://doi.org/10.1016/j.expneurol.2018.09.005 [PubMed]
- 22. Lv B, Li F, Han J, Fang J, Xu L, Sun C, Hua T, Zhang Z, Feng Z, Jiang X. Hif-1α overexpression improves transplanted bone mesenchymal stem cells survival in rat MCAO stroke model. Front Mol Neurosci. 2017; 10:80. https://doi.org/10.3389/fnmol.2017.00080 [PubMed]
- 23. Liu Y, Zhang R, Yan K, Chen F, Huang W, Lv B, Sun C, Xu L, Li F, Jiang X. Mesenchymal stem cells inhibit lipopolysaccharide-induced inflammatory responses of BV2 microglial cells through TSG-6. J Neuroinflammation. 2014; 11:135. https://doi.org/10.1186/1742-2094-11-135 [PubMed]
- 24. Troyer DL, Weiss ML. Wharton’s jelly-derived cells are a primitive stromal cell population. Stem Cells. 2008; 26:591–99. https://doi.org/10.1634/stemcells.2007-0439 [PubMed]
- 25. Friedman R, Betancur M, Boissel L, Tuncer H, Cetrulo C, Klingemann H. Umbilical cord mesenchymal stem cells: adjuvants for human cell transplantation. Biol Blood Marrow Transplant. 2007; 13:1477–86. https://doi.org/10.1016/j.bbmt.2007.08.048 [PubMed]
- 26. Chen MY, Lie PC, Li ZL, Wei X. Endothelial differentiation of wharton’s jelly-derived mesenchymal stem cells in comparison with bone marrow-derived mesenchymal stem cells. Exp Hematol. 2009; 37:629–40. https://doi.org/10.1016/j.exphem.2009.02.003 [PubMed]
- 27. Fischer UM, Harting MT, Jimenez F, Monzon-Posadas WO, Xue H, Savitz SI, Laine GA, Cox CS
Jr . Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev. 2009; 18:683–92. https://doi.org/10.1089/scd.2008.0253 [PubMed] - 28. Stonesifer C, Corey S, Ghanekar S, Diamandis Z, Acosta SA, Borlongan CV. Stem cell therapy for abrogating stroke-induced neuroinflammation and relevant secondary cell death mechanisms. Prog Neurobiol. 2017; 158:94–131. https://doi.org/10.1016/j.pneurobio.2017.07.004 [PubMed]
- 29. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004; 116:281–97. https://doi.org/10.1016/s0092-8674(04)00045-5 [PubMed]
- 30. Ameres SL, Martinez J, Schroeder R. Molecular basis for target RNA recognition and cleavage by human RISC. Cell. 2007; 130:101–12. https://doi.org/10.1016/j.cell.2007.04.037 [PubMed]
- 31. Mallory AC, Reinhart BJ, Jones-Rhoades MW, Tang G, Zamore PD, Barton MK, Bartel DP. MicroRNA control of PHABULOSA in leaf development: importance of pairing to the microRNA 5’ region. EMBO J. 2004; 23:3356–64. https://doi.org/10.1038/sj.emboj.7600340 [PubMed]
- 32. Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H, Xiang J, Zhang T, Theilen TM, et al. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat Cell Biol. 2015; 17:816–26. https://doi.org/10.1038/ncb3169 [PubMed]
- 33. Fong MY, Zhou W, Liu L, Alontaga AY, Chandra M, Ashby J, Chow A, O’Connor ST, Li S, Chin AR, Somlo G, Palomares M, Li Z, et al. Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat Cell Biol. 2015; 17:183–94. https://doi.org/10.1038/ncb3094 [PubMed]
- 34. Zhang L, Zhang S, Yao J, Lowery FJ, Zhang Q, Huang WC, Li P, Li M, Wang X, Zhang C, Wang H, Ellis K, Cheerathodi M, et al. Microenvironment-induced PTEN loss by exosomal microRNA primes brain metastasis outgrowth. Nature. 2015; 527:100–04. https://doi.org/10.1038/nature15376 [PubMed]
- 35. Song Y, Dou H, Li X, Zhao X, Li Y, Liu D, Ji J, Liu F, Ding L, Ni Y, Hou Y. Exosomal miR-146a contributes to the enhanced therapeutic efficacy of interleukin-1β-primed mesenchymal stem cells against sepsis. Stem Cells. 2017; 35:1208–21. https://doi.org/10.1002/stem.2564 [PubMed]
- 36. Théry C, Amigorena S, Raposo G, Clayton A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr Protoc Cell Biol. 2006; Chapter 3:Unit 3.22. https://doi.org/10.1002/0471143030.cb0322s30 [PubMed]
- 37. Liu W, Li R, Yin J, Guo S, Chen Y, Fan H, Li G, Li Z, Li X, Zhang X, He X, Duan C. Mesenchymal stem cells alleviate the early brain injury of subarachnoid hemorrhage partly by suppression of Notch1-dependent neuroinflammation: involvement of botch. J Neuroinflammation. 2019; 16:8. https://doi.org/10.1186/s12974-019-1396-5 [PubMed]
- 38. Xie Y, Peng J, Pang J, Guo K, Zhang L, Yin S, Zhou J, Gu L, Tu T, Mu Q, Liao Y, Zhang X, Chen L, Jiang Y. Biglycan regulates neuroinflammation by promoting M1 microglial activation in early brain injury after experimental subarachnoid hemorrhage. J Neurochem. 2020; 152:368–80. https://doi.org/10.1111/jnc.14926 [PubMed]
- 39. Ma Z, Zhang Z, Bai F, Jiang T, Yan C, Wang Q. Electroacupuncture pretreatment alleviates cerebral ischemic injury through α7 nicotinic acetylcholine receptor-mediated phenotypic conversion of microglia. Front Cell Neurosci. 2019; 13:537. https://doi.org/10.3389/fncel.2019.00537 [PubMed]
- 40. Fang SB, Zhang HY, Wang C, He BX, Liu XQ, Meng XC, Peng YQ, Xu ZB, Fan XL, Wu ZJ, Chen D, Zheng L, Zheng SG, Fu QL. Small extracellular vesicles derived from human mesenchymal stromal cells prevent group 2 innate lymphoid cell-dominant allergic airway inflammation through delivery of miR-146a-5p. J Extracell Vesicles. 2020; 9:1723260. https://doi.org/10.1080/20013078.2020.1723260 [PubMed]
- 41. Gong Z, Pan J, Shen Q, Li M, Peng Y. Mitochondrial dysfunction induces NLRP3 inflammasome activation during cerebral ischemia/reperfusion injury. J Neuroinflammation. 2018; 15:242. https://doi.org/10.1186/s12974-018-1282-6 [PubMed]
- 42. Treiber T, Treiber N, Meister G. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat Rev Mol Cell Biol. 2019; 20:5–20. https://doi.org/10.1038/s41580-018-0059-1 [PubMed]
- 43. Ying W, Riopel M, Bandyopadhyay G, Dong Y, Birmingham A, Seo JB, Ofrecio JM, Wollam J, Hernandez-Carretero A, Fu W, Li P, Olefsky JM. Adipose tissue macrophage-derived exosomal miRNAs can modulate in vivo and in vitro insulin sensitivity. Cell. 2017; 171:372–84.e12. https://doi.org/10.1016/j.cell.2017.08.035 [PubMed]
- 44. Zhao J, Li X, Hu J, Chen F, Qiao S, Sun X, Gao L, Xie J, Xu B. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. 2019; 115:1205–16. https://doi.org/10.1093/cvr/cvz040 [PubMed]
- 45. Mathiyalagan P, Liang Y, Kim D, Misener S, Thorne T, Kamide CE, Klyachko E, Losordo DW, Hajjar RJ, Sahoo S. Angiogenic mechanisms of human CD34+ stem cell exosomes in the repair of ischemic hindlimb. Circ Res. 2017; 120:1466–76. https://doi.org/10.1161/CIRCRESAHA.116.310557 [PubMed]
- 46. Song Y, Li Z, He T, Qu M, Jiang L, Li W, Shi X, Pan J, Zhang L, Wang Y, Zhang Z, Tang Y, Yang GY. M2 microglia-derived exosomes protect the mouse brain from ischemia-reperfusion injury via exosomal miR-124. Theranostics. 2019; 9:2910–23. https://doi.org/10.7150/thno.30879 [PubMed]
- 47. Simeoli R, Montague K, Jones HR, Castaldi L, Chambers D, Kelleher JH, Vacca V, Pitcher T, Grist J, Al-Ahdal H, Wong LF, Perretti M, Lai J, et al. Exosomal cargo including microRNA regulates sensory neuron to macrophage communication after nerve trauma. Nat Commun. 2017; 8:1778. https://doi.org/10.1038/s41467-017-01841-5 [PubMed]
- 48. Thomou T, Mori MA, Dreyfuss JM, Konishi M, Sakaguchi M, Wolfrum C, Rao TN, Winnay JN, Garcia-Martin R, Grinspoon SK, Gorden P, Kahn CR. Adipose-derived circulating miRNAs regulate gene expression in other tissues. Nature. 2017; 542:450–55. https://doi.org/10.1038/nature21365 [PubMed]
- 49. de Couto G, Gallet R, Cambier L, Jaghatspanyan E, Makkar N, Dawkins JF, Berman BP, Marbán E. Exosomal MicroRNA transfer into macrophages mediates cellular postconditioning. Circulation. 2017; 136:200–14. https://doi.org/10.1161/CIRCULATIONAHA.116.024590 [PubMed]
- 50. Otero-Ortega L, Gómez de Frutos MC, Laso-García F, Rodríguez-Frutos B, Medina-Gutiérrez E, López JA, Vázquez J, Díez-Tejedor E, Gutiérrez-Fernández M. Exosomes promote restoration after an experimental animal model of intracerebral hemorrhage. J Cereb Blood Flow Metab. 2018; 38:767–79. https://doi.org/10.1177/0271678X17708917 [PubMed]
- 51. Xin H, Li Y, Liu Z, Wang X, Shang X, Cui Y, Zhang ZG, Chopp M. MiR-133b promotes neural plasticity and functional recovery after treatment of stroke with multipotent mesenchymal stromal cells in rats via transfer of exosome-enriched extracellular particles. Stem Cells. 2013; 31:2737–46. https://doi.org/10.1002/stem.1409 [PubMed]
- 52. Xian P, Hei Y, Wang R, Wang T, Yang J, Li J, Di Z, Liu Z, Baskys A, Liu W, Wu S, Long Q. Mesenchymal stem cell-derived exosomes as a nanotherapeutic agent for amelioration of inflammation-induced astrocyte alterations in mice. Theranostics. 2019; 9:5956–75. https://doi.org/10.7150/thno.33872 [PubMed]
- 53. Ling X, Zhang G, Xia Y, Zhu Q, Zhang J, Li Q, Niu X, Hu G, Yang Y, Wang Y, Deng Z. Exosomes from human urine-derived stem cells enhanced neurogenesis via miR-26a/HDAC6 axis after ischaemic stroke. J Cell Mol Med. 2020; 24:640–54. https://doi.org/10.1111/jcmm.14774 [PubMed]
- 54. Moon GJ, Sung JH, Kim DH, Kim EH, Cho YH, Son JP, Cha JM, Bang OY. Application of mesenchymal stem cell-derived extracellular vesicles for stroke: biodistribution and MicroRNA study. Transl Stroke Res. 2019; 10:509–21. https://doi.org/10.1007/s12975-018-0668-1 [PubMed]
- 55. Lo WY, Peng CT, Wang HJ. MicroRNA-146a-5p mediates high glucose-induced endothelial inflammation via targeting interleukin-1 receptor-associated kinase 1 expression. Front Physiol. 2017; 8:551. https://doi.org/10.3389/fphys.2017.00551 [PubMed]
- 56. Osei ET, Florez-Sampedro L, Tasena H, Faiz A, Noordhoek JA, Timens W, Postma DS, Hackett TL, Heijink IH, Brandsma CA. miR-146a-5p plays an essential role in the aberrant epithelial-fibroblast cross-talk in COPD. Eur Respir J. 2017; 49:1602538. https://doi.org/10.1183/13993003.02538-2016 [PubMed]
- 57. Luo Q, Feng Y, Xie Y, Shao Y, Wu M, Deng X, Yuan WE, Chen Y, Shi X. Nanoparticle-microRNA-146a-5p polyplexes ameliorate diabetic peripheral neuropathy by modulating inflammation and apoptosis. Nanomedicine. 2019; 17:188–97. https://doi.org/10.1016/j.nano.2019.01.007 [PubMed]
- 58. Kawai T, Akira S. Pathogen recognition with toll-like receptors. Curr Opin Immunol. 2005; 17:338–44. https://doi.org/10.1016/j.coi.2005.02.007 [PubMed]
- 59. Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on toll-like receptors. Nat Immunol. 2010; 11:373–84. https://doi.org/10.1038/ni.1863 [PubMed]
- 60. Wang Z, Liu F, Wei M, Qiu Y, Ma C, Shen L, Huang Y. Chronic constriction injury-induced microRNA-146a-5p alleviates neuropathic pain through suppression of IRAK1/TRAF6 signaling pathway. J Neuroinflammation. 2018; 15:179. https://doi.org/10.1186/s12974-018-1215-4 [PubMed]
- 61. Ho BC, Yu IS, Lu LF, Rudensky A, Chen HY, Tsai CW, Chang YL, Wu CT, Chang LY, Shih SR, Lin SW, Lee CN, Yang PC, Yu SL. Inhibition of miR-146a prevents enterovirus-induced death by restoring the production of type I interferon. Nat Commun. 2014; 5:3344. https://doi.org/10.1038/ncomms4344 [PubMed]
- 62. Magilnick N, Reyes EY, Wang WL, Vonderfecht SL, Gohda J, Inoue JI, Boldin MP. miR-146a- Traf6 regulatory axis controls autoimmunity and myelopoiesis, but is dispensable for hematopoietic stem cell homeostasis and tumor suppression. Proc Natl Acad Sci USA. 2017; 114:E7140–49. https://doi.org/10.1073/pnas.1706833114 [PubMed]
- 63. Song S, Luo L, Sun B, Sun D. Roles of glial ion transporters in brain diseases. Glia. 2020; 68:472–94. https://doi.org/10.1002/glia.23699 [PubMed]
- 64. Song S, Wang S, Pigott VM, Jiang T, Foley LM, Mishra A, Nayak R, Zhu W, Begum G, Shi Y, Carney KE, Hitchens TK, Shull GE, Sun D. Selective role of Na+ /H+ exchanger in Cx3cr1+ microglial activation, white matter demyelination, and post-stroke function recovery. Glia. 2018; 66:2279–98. https://doi.org/10.1002/glia.23456 [PubMed]
- 65. Lyu C, Zhang Y, Gu M, Huang Y, Liu G, Wang C, Li M, Chen S, Pan S, Gu Y. IRAK-M deficiency exacerbates ischemic neurovascular injuries in experimental stroke mice. Front Cell Neurosci. 2018; 12:504. https://doi.org/10.3389/fncel.2018.00504 [PubMed]



