Introduction
Osteoporosis (OP) is a common metabolic bone disease in postmenopausal women and the elderly [1]. The pathological characteristics of osteoporosis include abnormal bone microarchitecture, decreased bone density, and increased bone fragility, which result in an increased risk of fractures [2]. An estimated 200 million people currently suffer from osteoporosis worldwide, and about 34 million patients in the USA have been diagnosed with osteoporosis or low bone mass [3]. Osteoporosis can cause pain, spinal deformities, and fragile fractures. Fragile fractures are usually caused by low-energy impacts, such as a fall from standing height, a slight collision, or other routine minor injuries. In a 50-year-old woman, the risk of developing osteoporotic fractures may be has high as 50% [4]. Vertebral and hip fractures are two common types of osteoporotic fractures. Vertebral fractures can cause long-term pain and severely affect the quality of life, while hip fractures can prevent patients from standing or walking and can even increase the risk of death [4]. Osteoporosis has thus become a major public health problem worldwide, placing heavy economic burdens on both patients and healthcare systems [3]. There is currently no gold standard treatment for osteoporosis.
Resveratrol (3,5,4′-trihydroxy-trans-stilbene) is a natural polyphenolic compound found in several plants, including grapes, berries, and peanuts [5, 6]. Growing evidence shows that resveratrol has various biological effects [7–9], including protective effects in osteoporosis. For example, bone mass density (BMD) reduction and microarchitectural deterioration were reversed by resveratrol in a rodent model [8]. Resveratrol may impact bone homeostasis by acting as both an anabolic and antiresorptive agent [10]. However, the detailed mechanisms underlying the effects of resveratrol on bone health remain unclear.
Bioinformatics, which uses computational techniques to organize and integrate experimental data, has been used to great effect in many studies [11, 12]. In this study, Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways associated with both resveratrol-targeted genes and osteoporosis progression were analyzed using bioinformatic tools. The p53 signaling pathway was identified as a key KEGG pathway involved in the protective effects of resveratrol on osteoporosis. We therefore performed an in vitro study to further examine whether resveratrol could counteract the negative effects of p53 on MDM2-mediated osteogenic differentiation.
Results
Resveratrol-targeted genes and the interaction network
In total, 30 resveratrol-targeted genes were identified in Search Tool for Interacting Chemicals (STITCH) using a three shell limit. Interaction networks among resveratrol-targeted genes were then constructed in STITCH (Figure 1A). TP53, SIRT1, PTGS1, SIRT3, ESR1, PPARG, NOS3, AKT1, SIRT5, and PTGS2 were identified as members of the first shell, indicating that resveratrol might directly affect these genes. The second shell consisted of ATM, BRCA1, FOXO1, MTOR, EP300, RICTOR, FOXO3, CDKN1A, KAT2B, and MDM2, and the third shell included HSP90AA1, HIPK2, NCOA3, CDKN2A, MAPK8, SRC, USP7, RCHY1, CREBBP, and SP1, indicating that resveratrol might have secondary effects on these genes. A network visualization constructed based on interaction weights indicated that TP53 had the highest weight of all of these genes (Figure 1B).
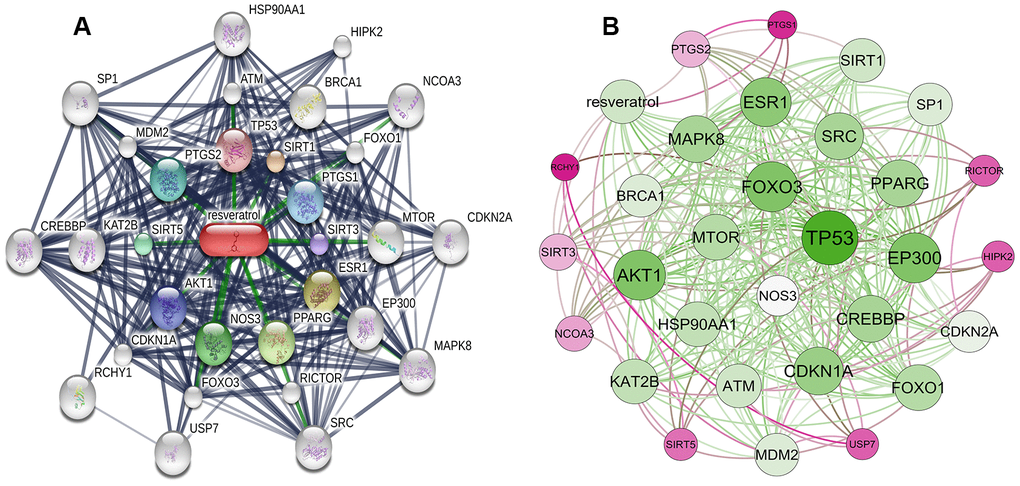
Figure 1. Construction of resveratrol-targeted genes interaction network. (A) Interaction network constructed using STITCH. First shell (chemical-protein): TP53, SIRT1, PTGS1, SIRT3, ESR1, PPARG, NOS3, AKT1, SIRT5, PTGS2. Second shell (protein-protein): ATM, BRCA1, FOXO1, MTOR, EP300, RICTOR, FOXO3, CDKN1A, KAT2B, MDM2. Third shell (protein-protein): HSP90AA1, HIPK2, NCOA3, CDKN2A, MAPK8, SRC, USP7, RCHY1, CREBBP, SP1. (B) Weighted interaction network indicating that TP53 had the highest weight.
Identification of KEGG pathways associated with osteoporosis and resveratrol-targeted genes
Enrichment analysis of the resveratrol-targeted genes using Database for Annotation, Visualization, and Integrated Discovery (DAVID) identified 33 KEGG pathways with p <0.05. 110 KEGG pathways involved in human osteoporosis were retrieved using miRWalk2.0. Twelve KEGG pathways associated with both osteoporosis and resveratrol-targeted genes were identified and visualized using a Venn Diagram online tool (Figure 2). Among them, the five KEGG pathways with smallest p values were prostate cancer pathway, pathway in cancer, glioma pathway, p53 signaling pathway, and cell cycle signaling pathway (Table 1).
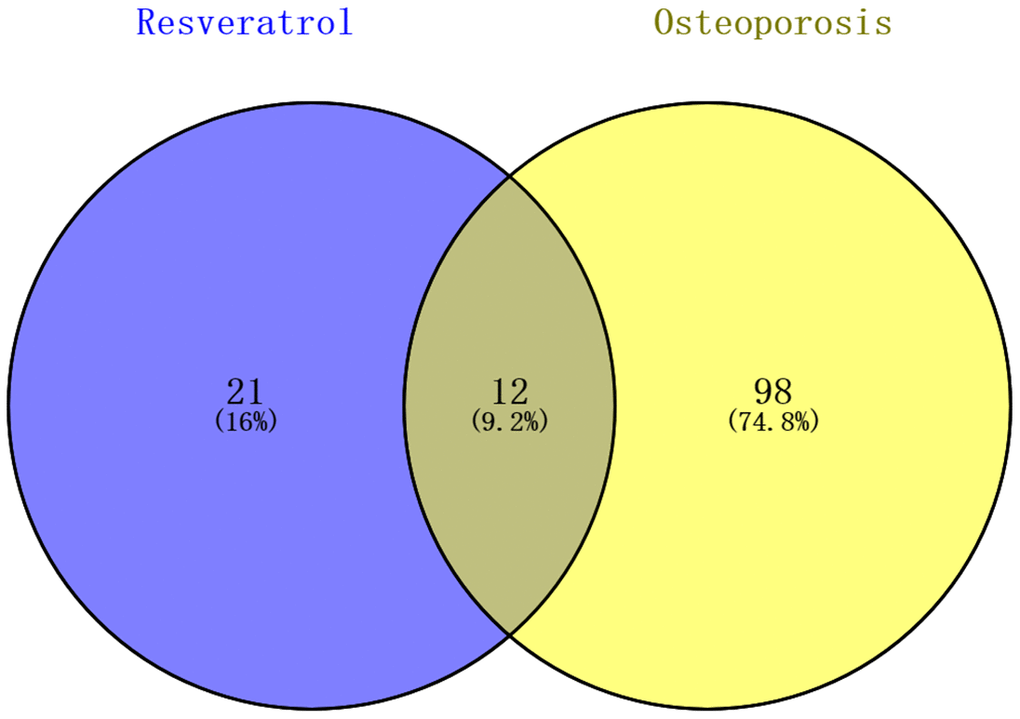
Figure 2. Identification KEGG pathways associated with both resveratrol-target genes and osteoporosis. 33 KEGG pathways associated with resveratrol-target genes and 110 associated with osteoporosis were identified; 12 (9.2%) KEGG pathways associated with both are shown in the Venn diagram.
Table 1. Top five KEGG pathways and associated genes.
| Term | KEGG pathway | Icariin-target genes | P-value |
| hsa05215 | Prostate cancer pathway | AKT1, CDKN1A, HSP90AA1, EP300, CREBBP, TP53, FOXO1, MDM2, MTOR | 9.5E-10 |
| hsa05200 | Pathways in cancer | HSP90AA1, PTGS2, PPARG, CREBBP, TP53, FOXO1, AKT1, CDKN1A, EP300, CDKN2A, MDM2, MAPK8, MTOR | 8.1E-9 |
| hsa05214 | Glioma pathway | AKT1, CDKN1A, CDKN2A, TP53, MDM2, MTOR | 4.4E-6 |
| hsa04115 | p53 signaling pathway | CDKN1A, CDKN2A, TP53, MDM2, RCHY1, ATM | 5.2E-6 |
| hsa04110 | Cell cycle signaling pathway | CDKN1A, CDKN2A, EP300, CREBBP, TP53, MDM2, ATM | 6.6E-6 |
Identification of hub genes
Among the 30 resveratrol-targeted genes, TP53, AKT1, EP300, CDKN1A, CREBBP, PPARG, MAPK8, FOXO1, MTOR, HSP90AA1, ATM, MDM2, CDKN2A, PTGS2, and RCHY1 were involved in the top five shared KEGG pathways (Figure 3). TP53, CDKN1A, and MDM2, which were involved in all top five KEGG pathways, were identified as hub genes. A circular visualization of chromosomal positions and connectivity of resveratrol-target genes is shown in Figure 4. Degree, betweenness, and closeness were highest for TP53.
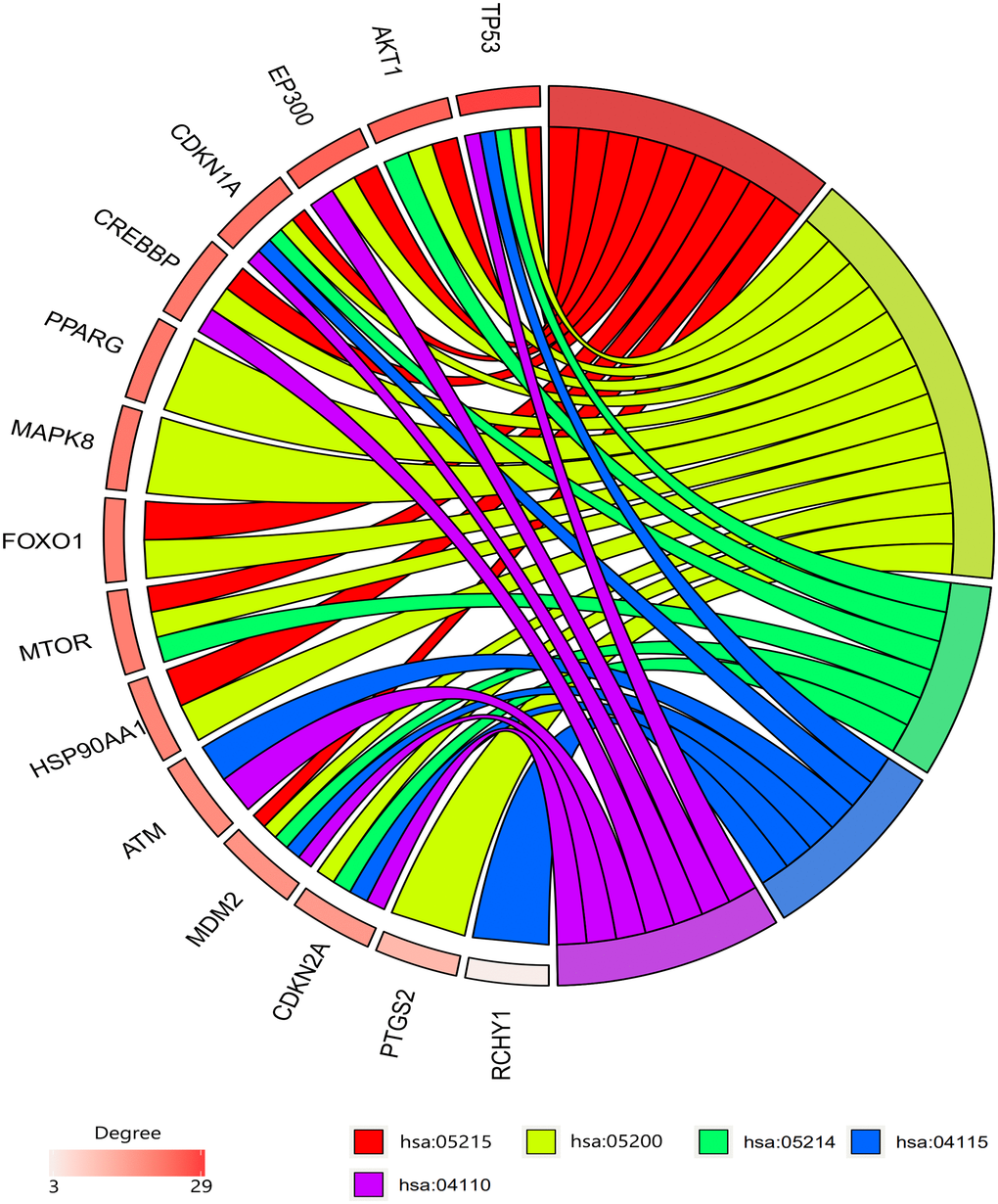
Figure 3. Gene enrichment analysis results. TP53, CDKN1A, and MDM2 were involved in all top five pathways. TP53, AKT1, and EP300 had the highest degrees. Hsa05215: prostate cancer pathway, hsa05200: pathway in cancer, hsa05214: glioma pathway, hsa04115; p53 signaling pathway, hsa04110: cell cycle signaling pathway.
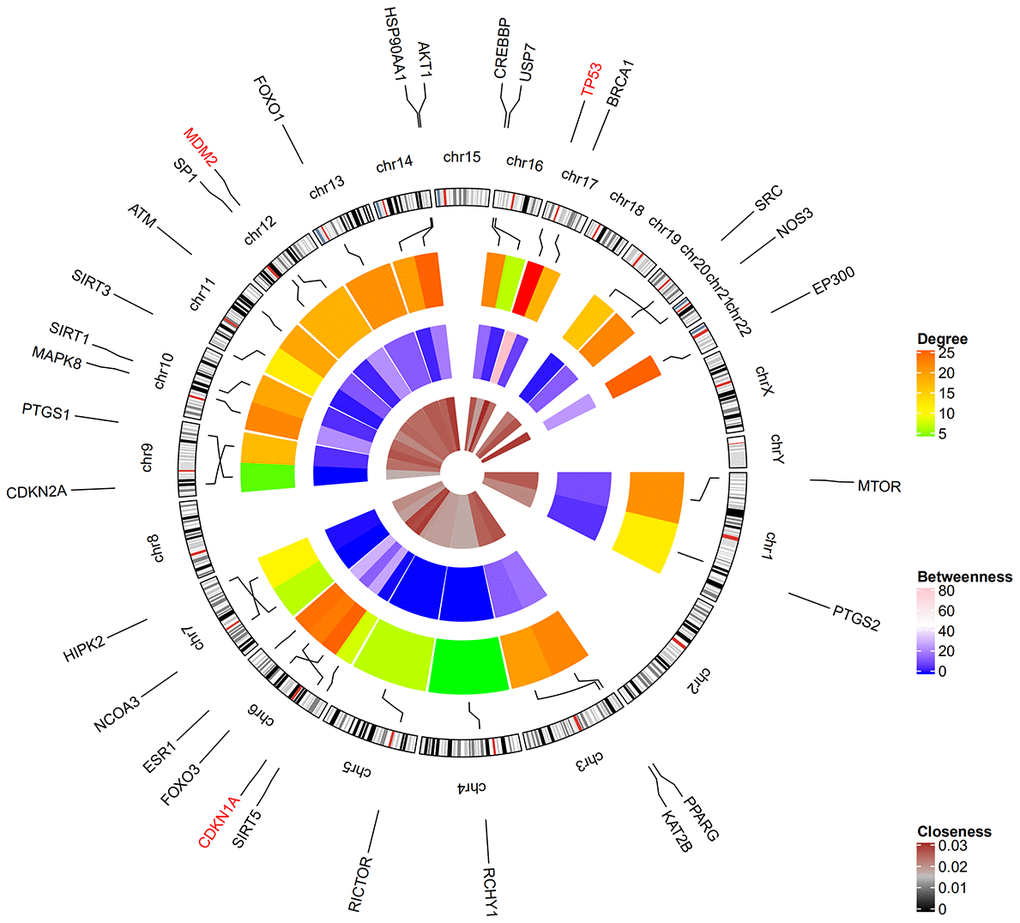
Figure 4. Circular visualization of chromosomal positions and connectivity of resveratrol-target genes. Gene names are shown in the outer circle. Different colors represent different degree, betweenness, and closeness values. The outer circle represents chromosomes; lines connect each gene to its chromosomal location. The three hub genes TP53, CDKN1A, and MDM2 highlighted in red are located in chr17, chr6, and chr12, respectively.
MDM2-mediated p53 degradation induces osteoblast differentiation in vitro
p53 gene and protein expression were measured in non-OP (n=10) and OP patients (n=10) using qRT-PCR and western blotting, respectively. p53 gene and protein expression were significantly higher in OP patients (p < 0.001, Figure 6A, 6B) than in non-OP patients. Transfection of human mesenchymal stem cells (hMSCs) with an MDM2 overexpression plasmid successfully increased MDM2 gene expression compared to untreated cells (Control) and empty vector transfection (Plasmid NC) (Figure 6C). As expected, p53 gene and protein expression decreased significantly in hMSCs transfected with MDM2 plasmid (p < 0.001 Figure 6D, 6E). In addition, expression of the osteogenic genes ALP OCN and Runx2 also increased significantly in MDM2 plasmid-treated hMSCs compared to untreated control and empty vector-treated cells (p < 0.001 Figure 6F). Alizarin red-mediated calcium staining also showed enhanced mineral deposition in the MDM2 plasmid group (p < 0.001 Figure 6G, 6H). These results indicated that MDM2 induced p53 degradation and promoted osteogenesis in hMSCs.
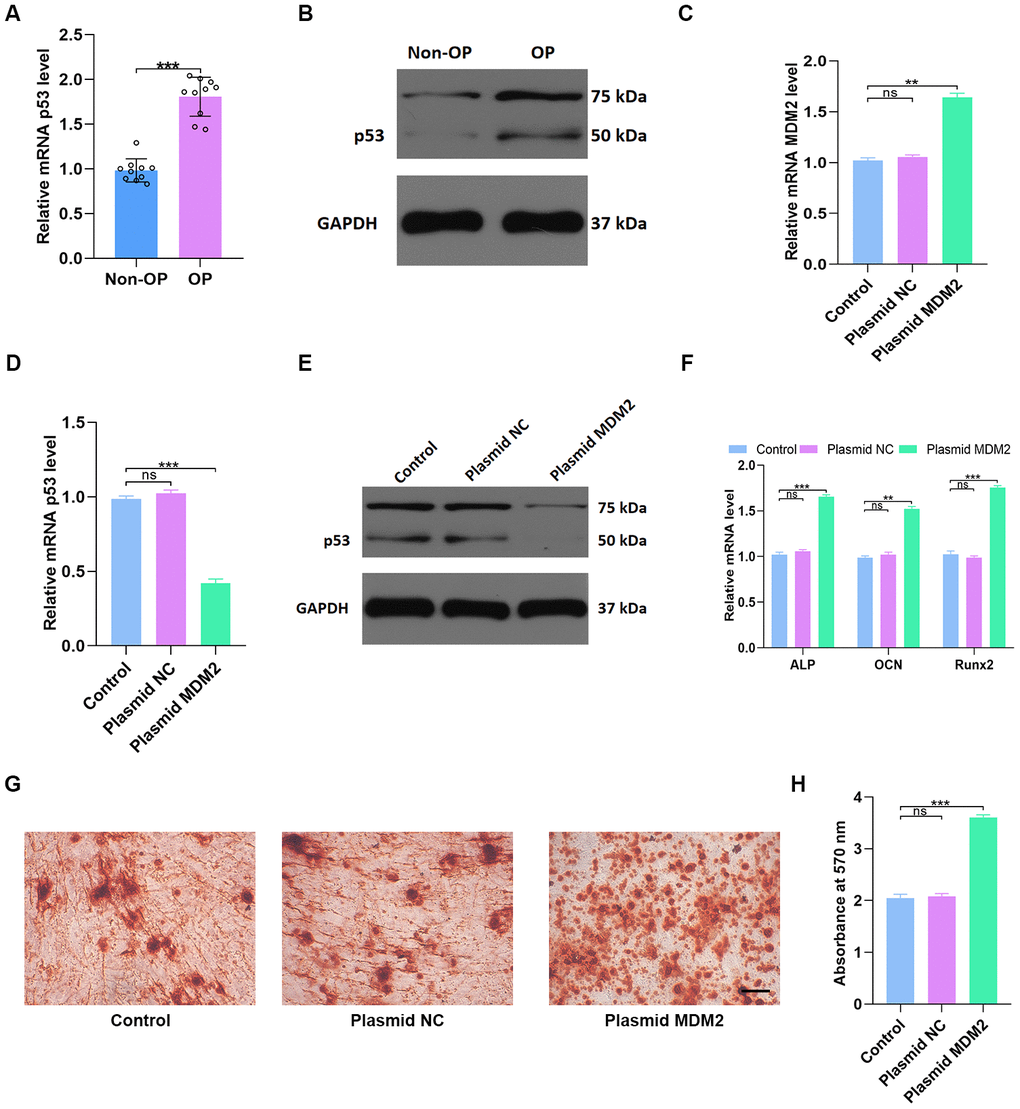
Figure 6. MDM2-mediated p53 degradation induces osteoblast differentiation in vitro. (A, B) p53 levels in non-OP patients and OP patients were measured by qRT-PCR and western blot; n=10 per group. (C) MDM2 expression in hMSCs was assessed by qRT-PCR analysis after different treatments. (D–E) p53 levels were measured by qRT-PCR and western blot in the three groups. (F) Osteogenic gene levels were measured by qRT-PCR. (G–H) Alizarin red-mediated calcium staining in hMSCs 21 days after transfection with different constructs. Scale bar = 10mm. Data are means ± SD. *p < 0.05, **p < 0.01, ***p < 0.001.
Resveratrol partially reverses p53-induced inhibition of osteogenic differentiation
The effects of different concentrations of resveratrol on MDM2 expression were measured by qRT-PCR. 10 μM resveratrol induced both the largest increase in MDM2 gene expression (Figure 7A) and the largest decrease in p53 expression (Figure 7B). As expected, ALP, OCN, and Runx2 expression were significantly increased by resveratrol treatment (Figure 7C–7D). Moreover, MDM2 gene expression was significantly higher (Figure 7E), while p53 expression was significantly lower (Figure 7F), in hMSCs treated with resveratrol than in the other groups. Furthermore, alizarin red-mediated calcium staining showed enhanced mineral deposition in the resveratrol group (p < 0.05 Figure 7G–7H), indicating that resveratrol might increase mineral bone mass. In summary, these results suggest that resveratrol promotes osteogenesis by inhibiting the p53 signaling pathway.
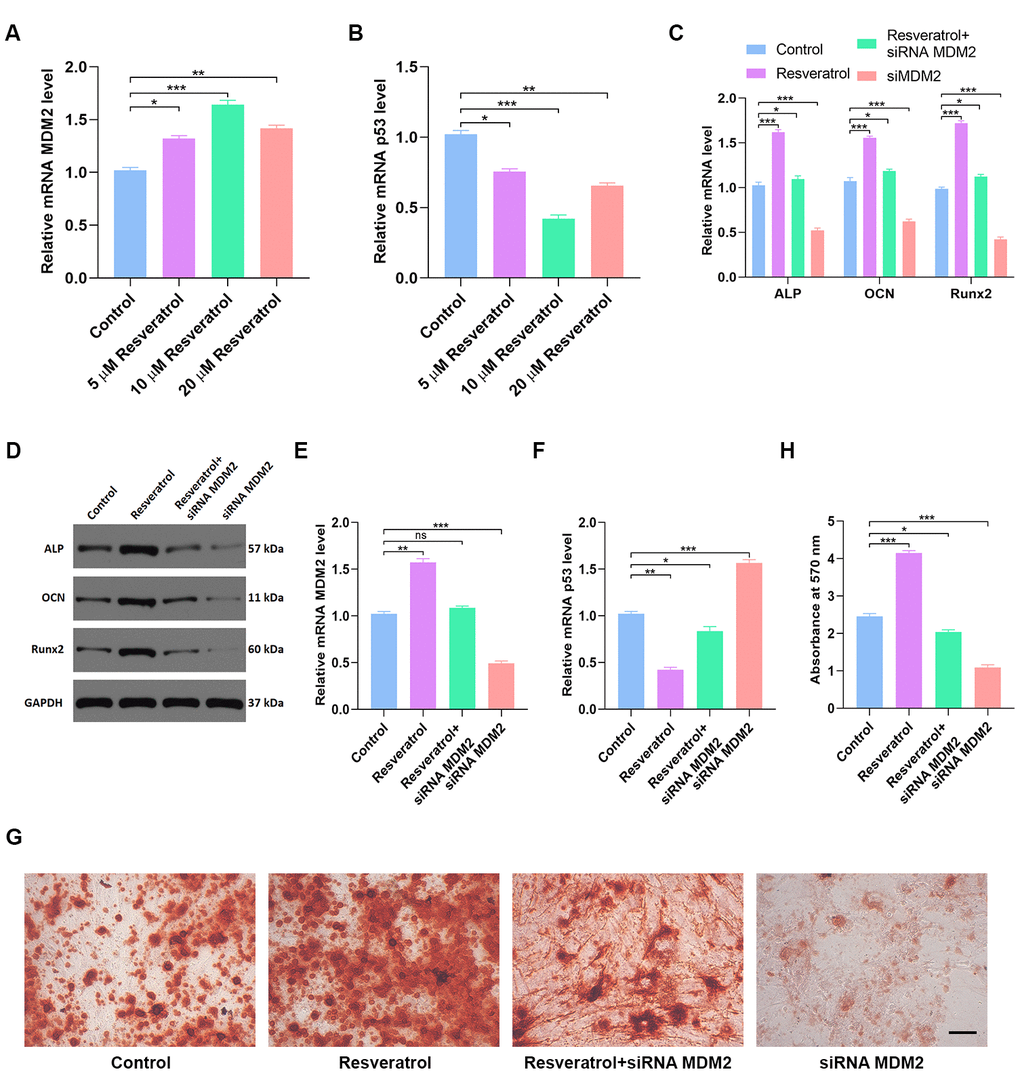
Figure 7. Resveratrol partially reverses p53-induced inhibition of osteogenic differentiation. (A) Effects of different concentrations of resveratrol on MDM2 expression were measured by qRT-PCR. (B) p53 expression in different groups was assessed by qRT-PCR. (C, D) Osteogenic genes expression in different groups was measured by qRT-PCR and western blot. (E) MDM2 expression in hMSCs after different treatments was assessed by qRT-PCR. (F) p53 expression in different groups was assessed by qRT-PCR. (G–H) Alizarin red-mediated calcium staining in hMSCs 21 days after transfection with different constructs. Scale bar = 10mm. Data are means ± SD. *p < 0.05, **p < 0.01, ***p < 0.001.
Discussion
Although osteoporosis is a widespread disease [13] that has become a major challenge for public health systems worldwide [3], there is currently no gold standard treatment. While resveratrol can reverse both osteoporosis-associated BMD reduction and microarchitectural deterioration [8], the mechanisms responsible for these effects remain unknown. In this study, bioinformatics analyses and in vitro studies were performed to investigate the molecular mechanisms of resveratrol’s effects on osteoporosis pathologies.
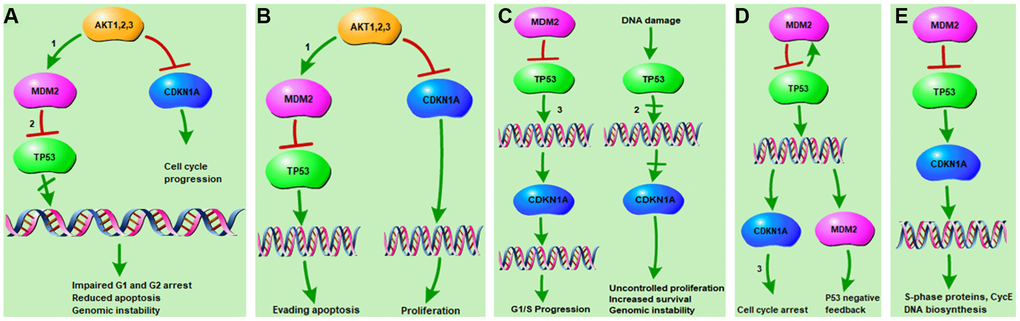
In this study, 12 KEGG pathways associated with both osteoporosis and resveratrol-targeted genes were identified using bioinformatics tools. Among these, the top five KEGG pathways with the smallest p values were prostate cancer pathway, pathway in cancer, glioma pathway, p53 pathway, and cell cycle pathway. Resveratrol-targeted genes were associated with G1/G2 arrest, apoptosis, and genomic instability in the prostate cancer pathway (Figure 5A), apoptosis and proliferation in pathway in cancer (Figure 5B), G1/S progression, proliferation, survival and genomic instability in the glioma pathway (Figure 5C), cell cycle arrest and p53 feedback in the p53 pathway (Figure 5D), and S-phase proteins and CycE DNA biosynthesis in the cell cycle pathway (Figure 5E). Thus, resveratrol-targeted genes exerted biological effects primarily through the p53 signaling pathway. p53 inhibits cancer development and progression via several mechanisms, including apoptosis, regulation of DNA replication, cell division, and inhibition of angiogenesis [14, 15]. The p53 protein is encoded by the TP53 gene, which was identified in this study as the hub gene with the highest degree of interaction in the network. TP53, CTNNB1, and SP1 modulate the expression of most of the differentially expressed genes that are upregulated and play important roles in primary osteoporosis [16]. Fu Jia et. al. demonstrated that pri-miR-34b/c rs4938723 and TP53 Arg72Pro polymorphisms may contribute to the risk of osteoporosis [17]. In this study, both qRT-PCR and western blots indicated that p53 was enriched in osteoporosis (Figure 6A, 6B). In addition, qRT-PCR and Alizarin-red staining showed that MDM2-mediated inhibition of p53 induced osteoblast differentiation in vitro (Figure 6C–6G), indicating that p53 promoted the pathological progression of osteoporosis.
Resveratrol (3,5,4′-trihydroxy-trans-stilbene) is a natural polyphenolic compound found in several plants [5, 6]. Accumulating evidence shows that resveratrol has anti-inflammatory, antioxidant, and other protective effects in osteoporosis and in aging-induced cognitive impairment [7–9]. Ali Mobasheri and Mehdi Shakibaei reported that resveratrol can modulate bone cell metabolism and bone turnover due to its osteogenic and osteoinductive properties [18, 19]. In this study, resveratrol partially reversed p53-induced inhibition of osteogenic differentiation in in vitro experiments (Figure 7). These results indicate that resveratrol may protect against osteoporosis by inhibiting the p53 signaling pathway.
Some limitations in this study should be considered when interpreting the results. Firstly, the effects of different durations of resveratrol treatment were not investigated. Furthermore, potential differences in p53 enrichment in different osteoporosis subtypes were not examined. Finally, additional studies are needed to compare the effects of resveratrol on osteoporosis development and progression with those of other drugs.
In conclusion, bioinformatics analysis revealed that the protective effects of resveratrol against osteoporosis were associated with its interaction with the prostate cancer pathway, pathway in cancer, glioma pathway, p53 signaling pathway, and cell cycle signaling pathway. Our in vitro experiment further indicated that resveratrol exerts anti-osteoporosis effects by inhibiting the p53 signaling pathway, and may thus serve as a promising osteoporosis treatment.
Materials and Methods
Construction of the resveratrol-targeted genes interaction network
The STITCH database integrates data on the effects of over 430000 chemicals on gene expression [20]. Resveratrol-targeted genes were identified using the default settings in STITCH, and the STITCH online tool was used to construct an interaction network for resveratrol and its targeted genes. Degree, betweenness, and closeness were analyzed for each gene in the network and visualized using Cytoscape 3.7.2. Degree represents the extent to which one node is associated with all the other nodes in the network, closeness represents how far one node is from other nodes in the network, and betweenness is the number of times a node acts as the shortest bridge between two other nodes. These measures for all resveratrol-targeted genes were then imported into Gephi software and a weighted interaction network was constructed.
Identification of hub genes
Enrichment information for the top five KEGG pathways was presented with GOplot, an R package that visually combines expression data with functional analysis [24]. Genes involved in all top five shared KEGG pathways were considered hub genes. Centrality in the network and chromosome position for all resveratrol-targeted genes were visualized using the circlize package for R [25].
Cell culture and transfection
BMSCs were kindly donated by the Huazhong University of Science and Technology, Wuhan, China. Cells were grown in a specific media designed for C57BL/6 mouse mesenchymal stem cells (#MUBMX-03011-440, Cyagen, Guangzhou, China) at 37°C in a 5% CO2 incubator. Cells were maintained for a maximum of 3 passages. Lipofectamine 3000 (#L3000001, ThermoFisher Scientific, USA) was used to transfect cells with siRNAs according to provided directions. MDM2 siRNA constructs (RIBOBIO, Guangzhou, China) were transfected at 50 nM. Plasmid NC and plasmid MDM2 were synthesized by GenePharma company (Shanghai, China). All in vitro experiments using cells were independently repeated three times.
qRT-PCR analysis
Total tissue/cell RNA was extracted using Trizol reagent (#15596018, Invitrogen, USA). RNA was reverse transcribed using the VersoTM cDNA Synthesis Kit (#AB-1054/A, ThermoFisher Scientific), and qRT-PCR reactions were performed using a Thermal Cycler C-1000 Touch system (#10021377, Bio-Rad CFX Manager, USA). GAPDH was used as a housekeeping gene. Target gene expression was quantified using the ΔΔCT method. The primers used in this study are listed in Table 2.
Table 2. Primers used in the experiments.
| Gene name | Primer sequence |
| hsa - p53 - Forward | CAGCACATGACGGAGGTTGT |
| hsa - p53 - Reverse | TCATCCAAATACTCCACACGC |
| hsa - MDM2 - Forward | GAATCATCGGACTCAGGTACATC |
| hsa - MDM2 - Reverse | TCTGTCTCACTAATTGCTCTCCT |
| hsa - ALP - Forward | ACCACCACGAGAGTGAACCA |
| hsa - ALP - Reverse | CGTTGTCTGAGTACCAGTCCC |
| hsa - OCN - Forward | CAAAGGTGCAGCCTTTGTGTC |
| hsa - OCN - Reverse | TCACAGTCCGGATTGAGCTCA |
| hsa - Runx2 - Forward | TGGTTACTGTCATGGCGGGTA |
| hsa - Runx2 - Reverse | TCTCAGATCGTTGAACCTTGCTA |
| hsa - GAPDH - Forward | CCGTTGAATTTGCCGTGA |
| hsa - GAPDH - Reverse | TGATGACCCTTTTGGCTCCC |
Western blotting
Lysis buffer (#AS1004, Aspen, South Africa) containing 1% protease inhibitor (#AS1008, Aspen) was used to lyse cells and tissue samples. Proteins were separated via SDS-PAGE, transferred to NC membranes (#IPVH00010, Millipore, USA), blocked with 5% nonfat milk, and stained overnight at 4°C with antibodies specific for ALP (1:1000, Sigma, USA,#ab95462), Osteocalcin (1:500, Sigma, USA, #ab93876), RunX2 (1:500, Sigma, USA, #ab23981), p53 (1:1,000, Sigma, USA, #SAB1302059), and GAPDH (1:10,000, Sigma, USA, #ab37168). Blots were then stained with appropriate horseradish peroxidase (HRP)-conjugated secondary antibodies (#AS1058, Aspen) and proteins were detected with a chemiluminescence detection system. Each experiment was independently repeated three times.
Alizarin red staining
BMSCs were grown in 6-well plates in media containing 100 nM dexamethasone, 50 mM ascorbic acid, and 10 mM b-glycerophosphate to promote osteogenesis (#HUXMA-90021, Cyagen, USA). Briefly, cells were washed twice with PBS and 10% formalin was added to fix the cells for 15 minutes. Subsequently, 1 mL 0.5% alizarin red staining solution was used to stain the cells at room temperature for 15 minutes. After rinsing with distilled water for 5 minutes, red mineralized nodules were analyzed via a charge-coupled device microscope. Absorbance was measured at 570 nm. Experiments were repeated in triplicate.
Statistical analysis
Data are shown as means ± SD. GraphPad Prism 8.0 (GraphPad Software, CA, USA) was used to perform statistical analyses. One-way analysis of variance (ANOVA) with Tukey’s post hoc test was used to compare three or more groups. Unpaired Student’s t-test was used to compare data between two groups. P < 0.05 was considered statistically significant. The study schema is shown in Figure 8.
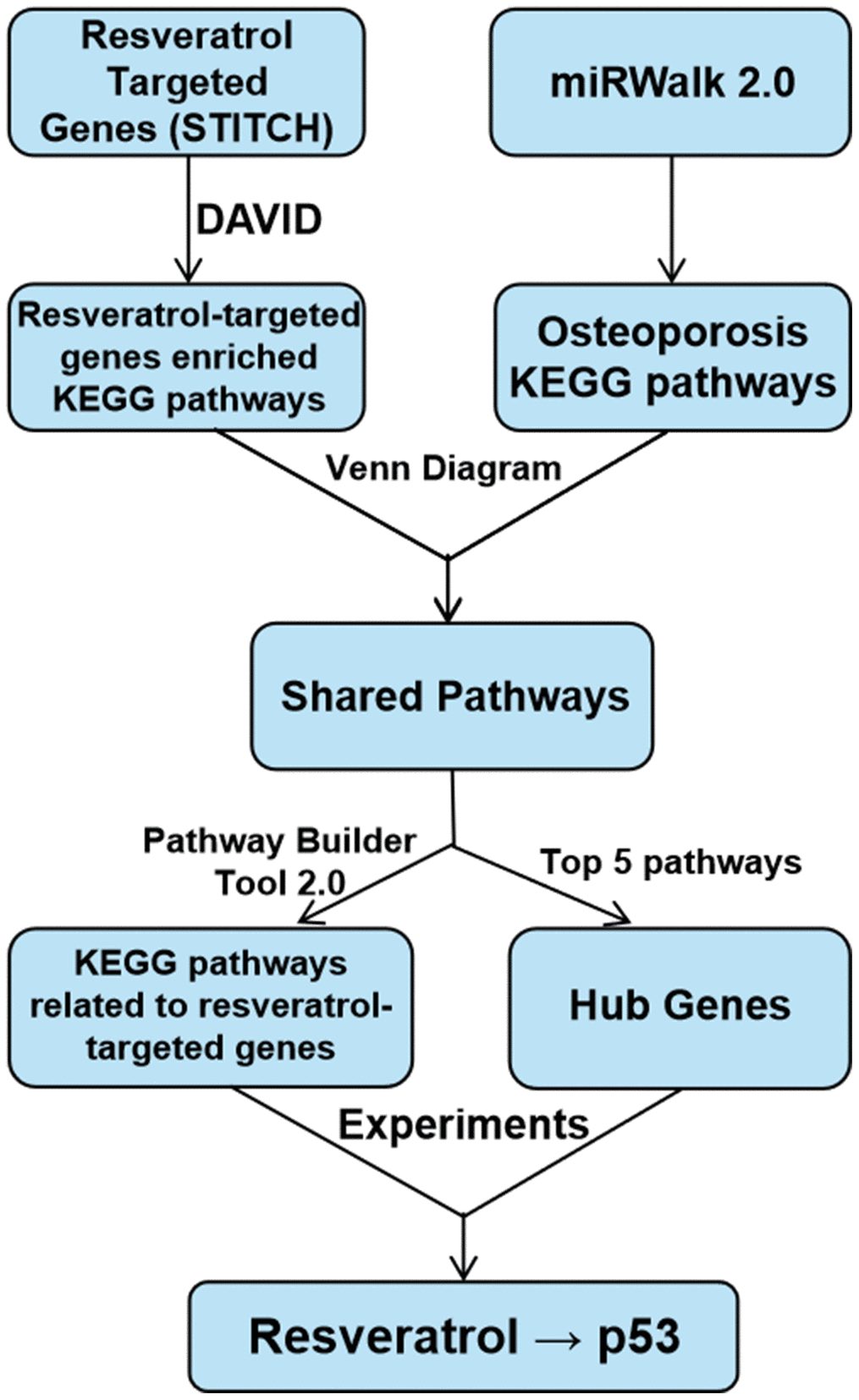
Figure 8. Study schema.
Author Contributions
YY and YX conceived of and designed the study; GY, HZ, and BL supervised the study; TY, ZW, and XY performed bioinformatics analysis and experiments; HZ and WH analyzed the data; JX and YZ provided advice and technical assistance; TY, ZW, and XY wrote the manuscript. All authors approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This study was supported by the National Science Foundation of China (No.31600754, NO.81472144).
References
- 1. Yan G, Huang Y, Cao H, Wu J, Jiang N, Cao X. Association of breastfeeding and postmenopausal osteoporosis in chinese women: a community-based retrospective study. BMC Womens Health. 2019; 19:110. https://doi.org/10.1186/s12905-019-0808-0 [PubMed]
- 2. Cai X, Yi X, Zhang Y, Zhang D, Zhi L, Liu H. Genetic susceptibility of postmenopausal osteoporosis on sulfide quinone reductase-like gene. Osteoporos Int. 2018; 29:2041–47. https://doi.org/10.1007/s00198-018-4575-9 [PubMed]
- 3. Rajasree Vijayakumar DB. Osteoporosis: An under-recognized public health problem. Journal of Local and Global Health Science. 2016; 2:1–13. https://doi.org/10.5339/jlghs.2016.2
- 4. Cummings SR, Melton LJ. Epidemiology and outcomes of osteoporotic fractures. Lancet. 2002; 359:1761–67. https://doi.org/10.1016/S0140-6736(02)08657-9 [PubMed]
- 5. Jang M, Cai L, Udeani GO, Slowing KV, Thomas CF, Beecher CW, Fong HH, Farnsworth NR, Kinghorn AD, Mehta RG, Moon RC, Pezzuto JM. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science. 1997; 275:218–20. https://doi.org/10.1126/science.275.5297.218 [PubMed]
- 6. Vinson JA. Flavonoids in foods as in vitro and in vivo antioxidants. Adv Exp Med Biol. 1998; 439:151–64. https://doi.org/10.1007/978-1-4615-5335-9_11 [PubMed]
- 7. Gocmez SS, Gacar N, Utkan T, Gacar G, Scarpace PJ, Tumer N. Protective effects of resveratrol on aging-induced cognitive impairment in rats. Neurobiol Learn Mem. 2016; 131:131–36. https://doi.org/10.1016/j.nlm.2016.03.022 [PubMed]
- 8. Feng J, Liu S, Ma S, Zhao J, Zhang W, Qi W, Cao P, Wang Z, Lei W. Protective effects of resveratrol on postmenopausal osteoporosis: regulation of SIRT1-NF-κB signaling pathway. Acta Biochim Biophys Sin (Shanghai). 2014; 46:1024–33. https://doi.org/10.1093/abbs/gmu103 [PubMed]
- 9. Csiszar A. Anti-inflammatory effects of resveratrol: possible role in prevention of age-related cardiovascular disease. Ann N Y Acad Sci. 2011; 1215:117–22. https://doi.org/10.1111/j.1749-6632.2010.05848.x [PubMed]
- 10. Tou JC. Evaluating resveratrol as a therapeutic bone agent: preclinical evidence from rat models of osteoporosis. Ann N Y Acad Sci. 2015; 1348:75–85. https://doi.org/10.1111/nyas.12840 [PubMed]
- 11. Song ZY, Chao F, Zhuo Z, Ma Z, Li W, Chen G. Identification of hub genes in prostate cancer using robust rank aggregation and weighted gene co-expression network analysis. Aging (Albany NY). 2019; 11:4736–56. https://doi.org/10.18632/aging.102087 [PubMed]
- 12. Schelker M, Feau S, Du J, Ranu N, Klipp E, MacBeath G, Schoeberl B, Raue A. Estimation of immune cell content in tumour tissue using single-cell RNA-seq data. Nat Commun. 2017; 8:2032. https://doi.org/10.1038/s41467-017-02289-3 [PubMed]
- 13. Hoffmann DB, Gruber J, Böker KO, Deppe D, Sehmisch S, Schilling AF, Lemus-Diaz N, Komrakova M, Schneider S. Effects of RANKL knockdown by virus-like particle-mediated RNAi in a rat model of osteoporosis. Mol Ther Nucleic Acids. 2018; 12:443–52. https://doi.org/10.1016/j.omtn.2018.06.001 [PubMed]
- 14. Chumakov PM. Versatile functions of p53 protein in multicellular organisms. Biochemistry (Mosc). 2007; 72:1399–421. https://doi.org/10.1134/s0006297907130019 [PubMed]
- 15. Jun HJ, Nguyen AH, Kim YH, Park KH, Kim D, Kim KK, Sim SJ. Distinct rayleigh scattering from hot spot mutant p53 proteins reveals cancer cells. Small. 2014; 10:2954–62. https://doi.org/10.1002/smll.201400004 [PubMed]
- 16. Xie W, Ji L, Zhao T, Gao P. Identification of transcriptional factors and key genes in primary osteoporosis by DNA microarray. Med Sci Monit. 2015; 21:1333–44. https://doi.org/10.12659/MSM.894111 [PubMed]
- 17. Jia F, Sun R, Li J, Li Q, Chen G, Fu W. Interactions of pri-miRNA-34b/c and TP53 polymorphisms on the risk of osteoporosis. Genet Test Mol Biomarkers. 2016; 20:398–401. https://doi.org/10.1089/gtmb.2015.0282 [PubMed]
- 18. Mobasheri A, Shakibaei M. Osteogenic effects of resveratrol in vitro: potential for the prevention and treatment of osteoporosis. Ann N Y Acad Sci. 2013; 1290:59–66. https://doi.org/10.1111/nyas.12145 [PubMed]
- 19. Rayalam S, Della-Fera MA, Baile CA. Synergism between resveratrol and other phytochemicals: implications for obesity and osteoporosis. Mol Nutr Food Res. 2011; 55:1177–85. https://doi.org/10.1002/mnfr.201000616 [PubMed]
- 20. Szklarczyk D, Santos A, von Mering C, Jensen LJ, Bork P, Kuhn M. STITCH 5: augmenting protein-chemical interaction networks with tissue and affinity data. Nucleic Acids Res. 2016; 44:D380–84. https://doi.org/10.1093/nar/gkv1277 [PubMed]
- 21. Dweep H, Gretz N. miRWalk2.0: a comprehensive atlas of microRNA-target interactions. Nat Methods. 2015; 12:697. https://doi.org/10.1038/nmeth.3485 [PubMed]
- 22. Huang da W, Sherman BT, Lempicki RA. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009; 37:1–13. https://doi.org/10.1093/nar/gkn923 [PubMed]
- 23. Huang da W, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009; 4:44–57. https://doi.org/10.1038/nprot.2008.211 [PubMed]
- 24. Walter W, Sánchez-Cabo F, Ricote M. GOplot: an R package for visually combining expression data with functional analysis. Bioinformatics. 2015; 31:2912–14. https://doi.org/10.1093/bioinformatics/btv300 [PubMed]
- 25. Gu Z, Gu L, Eils R, Schlesner M, Brors B. Circlize implements and enhances circular visualization in R. Bioinformatics. 2014; 30:2811–12. https://doi.org/10.1093/bioinformatics/btu393 [PubMed]