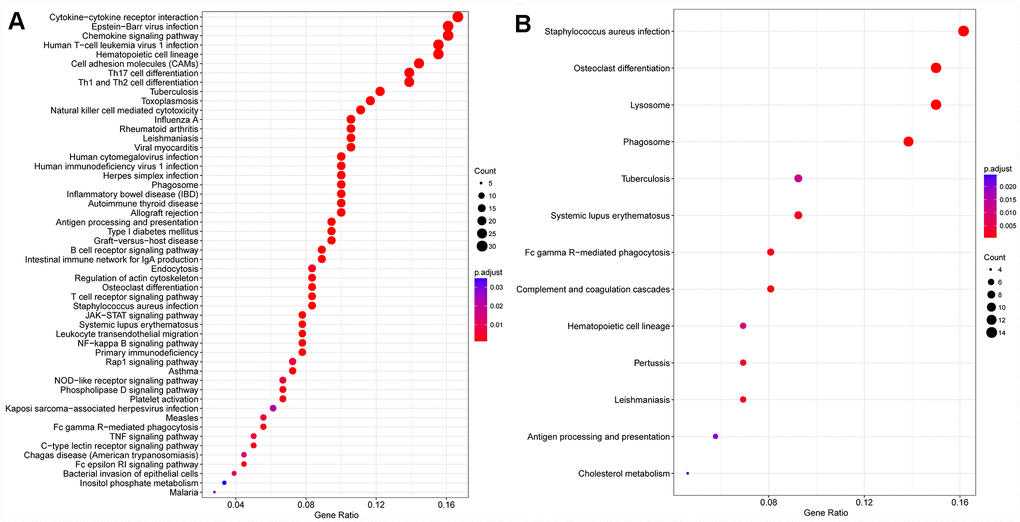
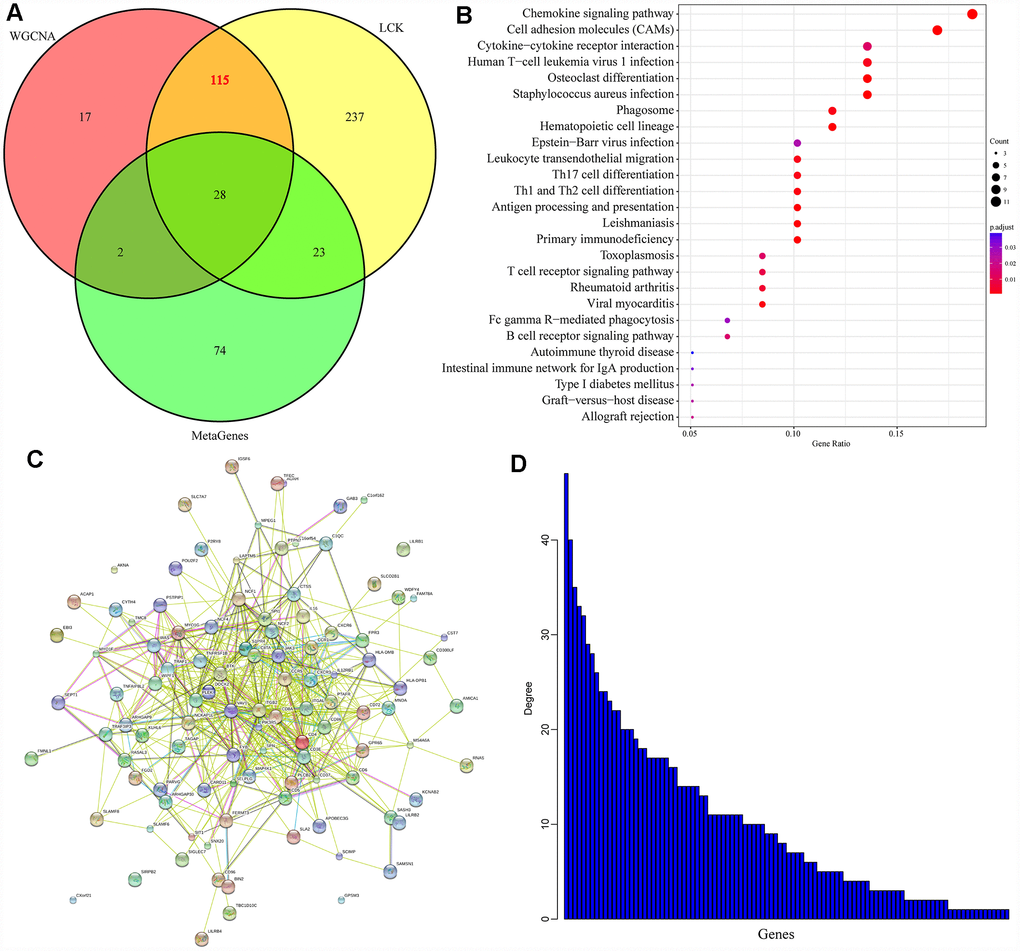
Introduction
In recent years, with the increasing understanding of the immune microenvironment of breast cancer tissues, immune escape has come to be considered an important marker of breast cancer development [1–4]. After tumor occurrence, tumor cells continuously interact with the immune microenvironment and gradually acquire the capacity for immune escape [5]. Both innate immunity (facilitated by macrophages and neutrophils) and adaptive immunity (facilitated by T cells and B cells) are impaired in patients with breast cancer. These impairments alter the immune microenvironment and promote the occurrence and development of tumors by (1) stimulating tumor angiogenesis, (2) altering the biological characteristics of tumors, (3) screening for tumor cells that are more suitable for survival in the host microenvironment and (4) regulating the activity of tumor stem cells. Therefore, targeted tumor immune microenvironment therapy for breast cancer has become a research hotspot [6]. However, such research has mostly been limited to preclinical experiments or clinical data-mining studies with small sample sizes [7].
Based on the expression of immune-related genes in The Cancer Genome Atlas (TCGA) database, researchers have developed a variety of immune scoring methods to investigate the interactions between tumor cells and immune cells in tumor tissues [8]. For instance, ‘Estimation of STromal and Immune cells in MAlignant Tumours using Expression data’ (ESTIMATE) is a tool that uses gene expression data to predict the purity of tumors and the presence of infiltrating stromal/immune cells in tumor tissues [9]. Previous ESTIMATE analyses have revealed that stromal/immune cell infiltration is associated with an improved prognosis in patients with various types of tumors, including prostate cancer and colorectal cancer [10, 11]. However, similar research has not been conducted in breast cancer.
Therefore, in this study, we used a series of bioinformatic tools to identify suitable immune scoring methods for different clinical subtypes of breast cancer, in order to discover diagnostic and prognostic markers of breast cancer.
Results
Selection of the lymphocyte-specific kinase (LCK) metagene as a representative gene in the breast cancer immune microenvironment
We obtained gene expression data from patients with different breast cancer subtypes from TCGA, and used Spearman correlation coefficients to calculate the correlations between different immune-related scores in these patients (Figure 1A–1D). With the exception of the neoantigen score, all the immune scores exhibited strong positive correlations with one another, especially in patients with the Her-2-like subtype. In all four subtypes of breast cancer, the LCK metagene score exhibited the highest mean correlation score with the other types of immune-related scores (luminal A: 77.7, luminal B: 69.4, Her-2-like: 87.7, triple-negative breast cancer [TNBC]: 76.7). Thus, we selected the LCK metagene as a representative gene in the breast cancer immune microenvironment.
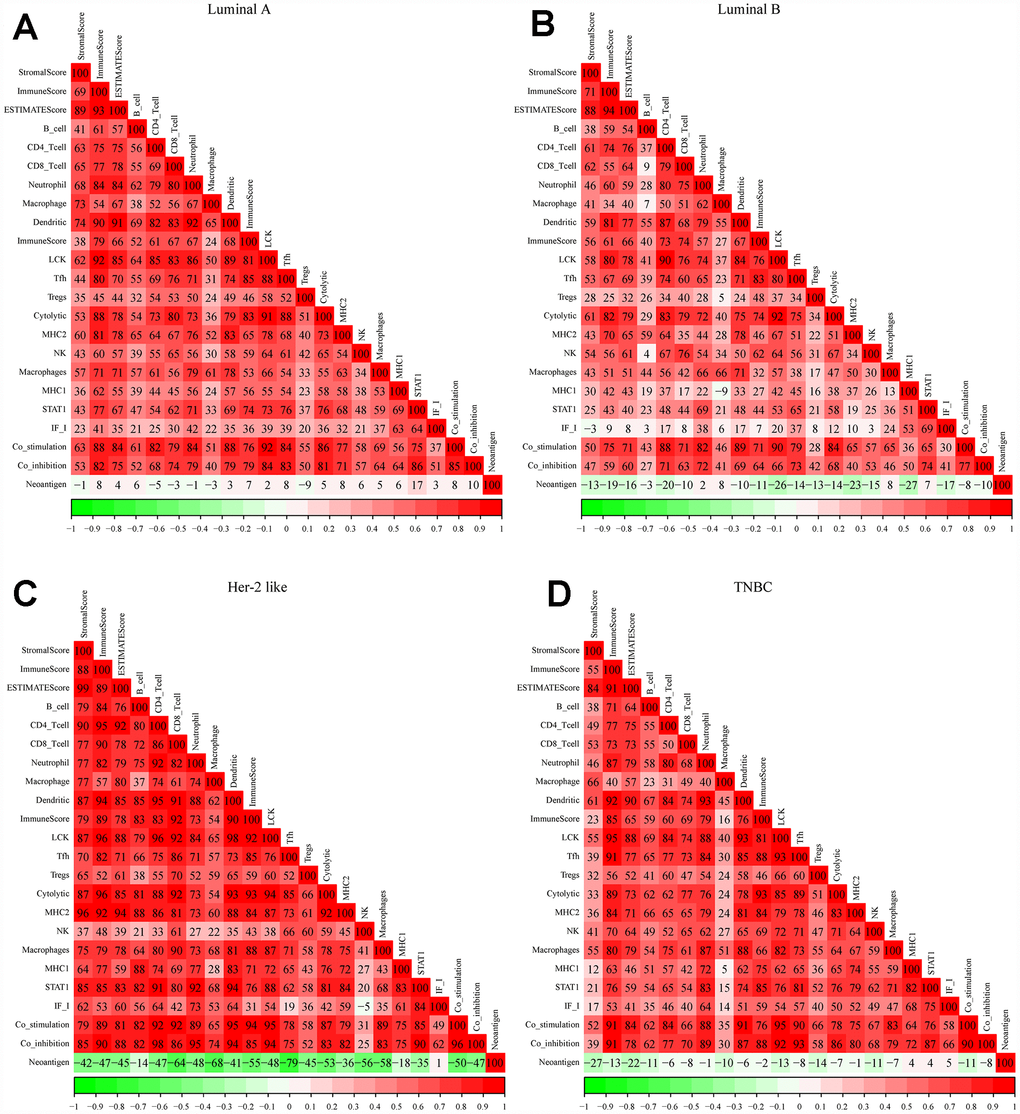
Figure 1. Correlations between different immune scores in patients with different breast cancer subtypes. (A) Luminal A subtype, (B) Luminal B subtype, (C) Her-2-like subtype, (D) TNBC subtype. Spearman correlation coefficients are color-coded to indicate positive (red) or negative (green) associations.
Next, we analyzed the distribution of LCK metagene levels in patients of the four breast cancer subtypes at different clinical stages (Figure 2A–2D). LCK metagene expression was significantly upregulated in stage I in TNBC, suggesting that high LCK metagene expression may be a positive prognostic factor in TNBC.
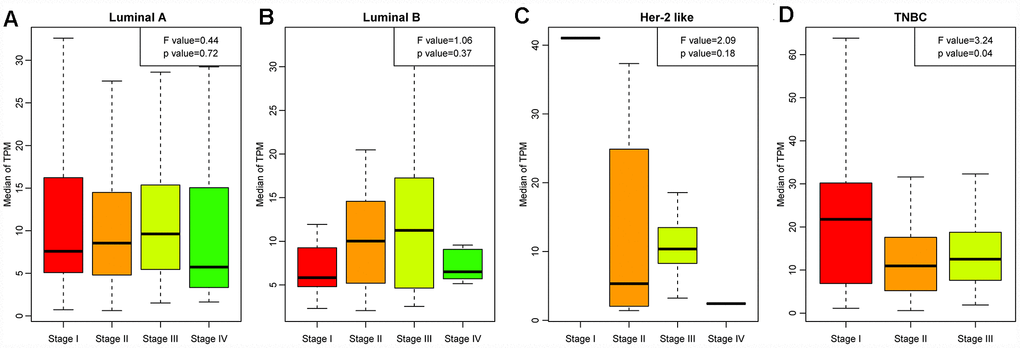
Figure 2. LCK metagene scores of patients at different clinical stages. (A) Luminal A subtype, (B) Luminal B subtype, (C) Her-2-like subtype, (D) TNBC subtype. Data are presented as the mean ± standard error of the mean (SEM).
We then divided the patients of each breast cancer subtype into two groups based on the median LCK metagene level, and assessed the prognostic differences between patients with high and low LCK metagene levels (Figure 3A–3D). The prognosis of the high expression group was better than that of the low expression group in all four subtypes. We also observed significant differences in LCK metagene expression among the four subtypes (Figure 3E). The median LCK metagene score was significantly higher in the TNBC group than in the other groups, suggesting that LCK metagene expression can be used as a prognostic marker in breast cancer.
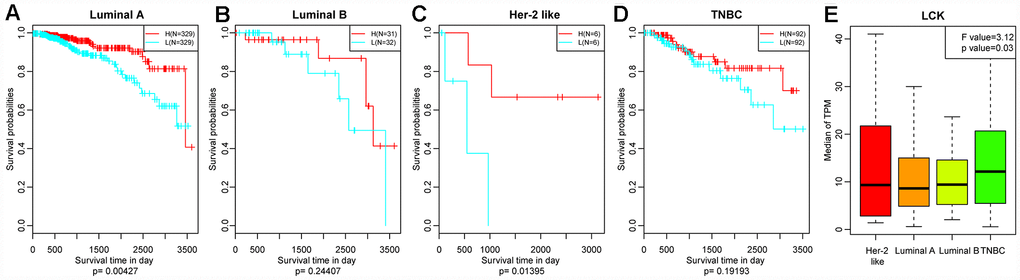
Figure 3. Relationship between the LCK metagene score and prognosis. (A) Luminal A subtype, (B) Luminal B subtype, (C) Her-2-like subtype, (D) TNBC subtype. Data were analyzed in KM plotter. H: high LCK metagene score; L: low LCK metagene score. The log-rank p values are shown. (E) LCK metagene scores of patients with different breast cancer subtypes. Data are presented as the mean ± SEM.
Next, we downloaded single-nucleotide polymorphism data on BRCA1, BRCA2, TP53 and PTEN [12], and divided patients into mutant and wild-type groups. We then determined the LCK metagene expression in each of these groups (Figure 4A–4D). LCK metagene expression was significantly greater in the BRCA2, TP53 and PTEN mutant groups than in their wild-type counterparts, but did not differ significantly between the BRCA1 mutant and wild-type groups.
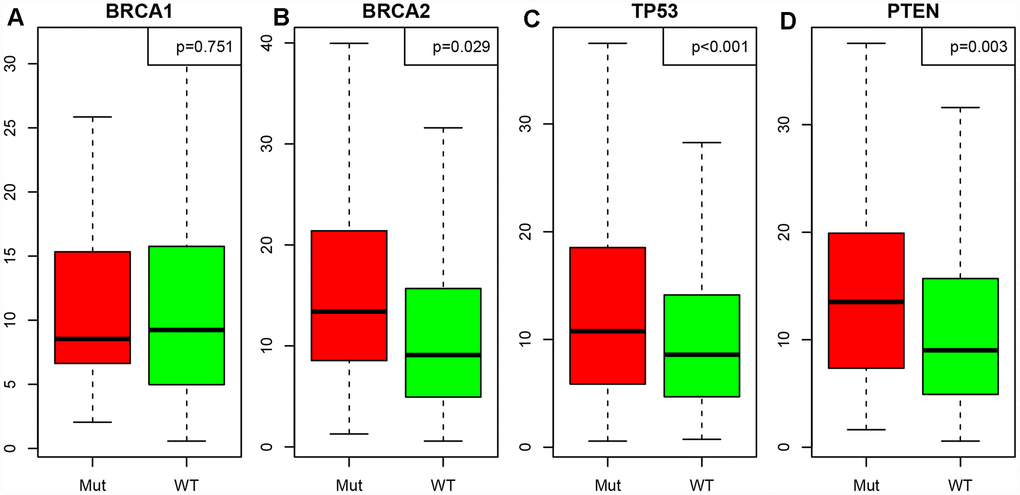
Figure 4. Correlation between the LCK metagene score and gene mutations. (A) BRCA1, (B) BRCA2, (C) TP53, (D) PTEN. Mut: mutant; WT: wild-type. Data are presented as the mean ± SEM.
Thus, the LCK metagene can be regarded as a representative gene in the immune microenvironment of breast cancer, and can be explored as a marker for the development of drugs to treat breast cancer.
Identification of differentially expressed genes (DEGs) in the high and low LCK metagene expression groups
Next, we used the DESeq2 function in the R software package [13] to analyze the genetic differences between the high and low LCK metagene expression groups, and obtained 403 DEGs. The volcano plot is shown in Figure 6. There were significantly more upregulated genes than downregulated genes in the high LCK metagene expression group.
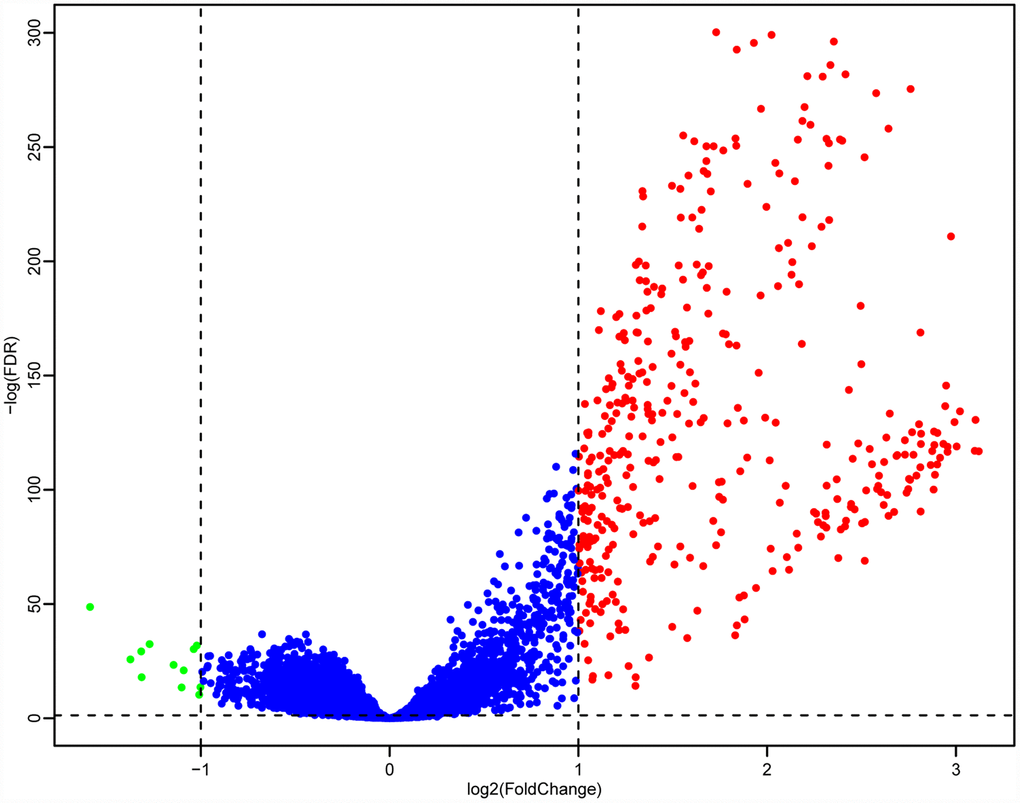
Figure 6. Volcano maps of DEGs. Red represents genes that were upregulated in patients with high LCK metagene scores, while green represents genes that were upregulated in patients with low LCK metagene scores.
External data validation
To verify the prognostic value of these 22 genes, we used the breast cancer dataset from the online tool Kaplan-Meier (KM) plotter [15] (http://kmplot.com) to analyze the relationship between the expression of these genes and the overall survival of breast cancer patients. We assessed the 18 genes with available data on the KM plotter platform, and divided the samples into high and low expression groups according to the median mRNA level of each gene. The KM curves of the 18 genes are shown in Figure 8. High expression of these 18 genes was associated with a good prognosis, consistent with our earlier analysis results.
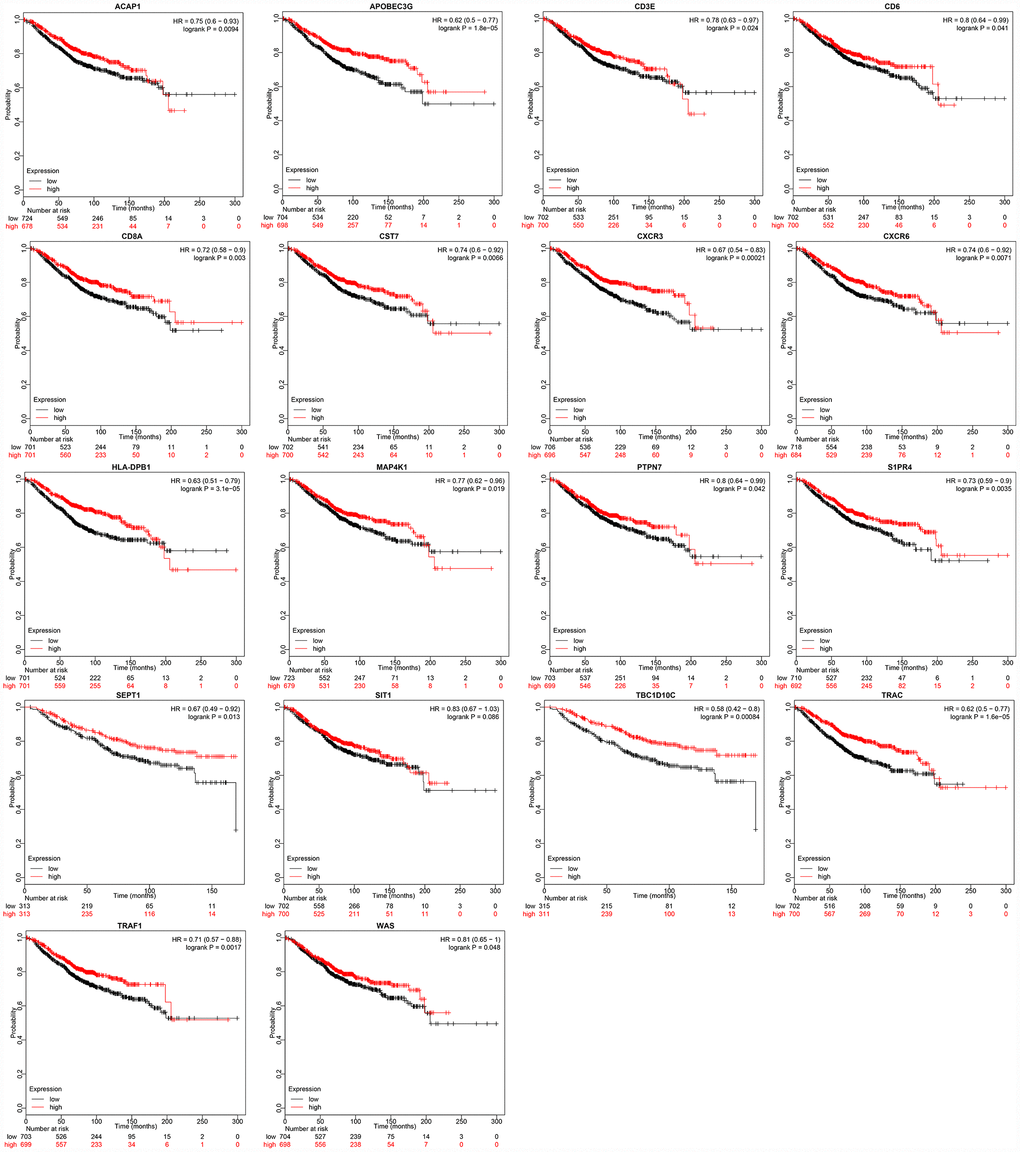
Figure 8. Relationship between immune microenvironment-related genes and breast cancer patient prognosis. Data were analyzed with KM plotter. Probabilities indicate overall survival; HR: hazard ratio.
Specimen verification results
We then analyzed the expression of the 22 prognosis-related genes in tumor and non-tumor tissues from five breast cancer patients. The fold-changes in these 22 genes between tumor and non-tumor tissues (T/N) are shown in Supplementary Figure 6. In most cases, the fold-change (T/N) was less than 0.05, indicating that the gene was downregulated in breast cancer tumors (all data are available upon request).
Discussion
In recent years, breast cancer treatment has included surgery, chemotherapy, endocrine therapy, targeted therapy and radiotherapy. However, traditional therapies have failed to save some refractory patients [16–18]. In this context, increasing attention has been paid to the tumor microenvironment and tumor immunity [19]. Stimulating the immune system and enhancing the anti-tumor function of the tumor microenvironment may be a novel way to kill tumor cells [20]. Therefore, screening genes related to the immune environment of breast cancer is an important step towards predicting the prognosis of patients and identifying new therapeutic targets. In this study, we used the database of TCGA to search for immune microenvironmental markers associated with the overall survival of breast cancer patients. We found that the expression of 22 genes correlated significantly with overall survival, and verified these results in KM plotter.
We first demonstrated that the LCK metagene score correlated highly with various other immune-related scores, along with the tumor clinical stage, the prognosis and the mutation status of multiple tumor suppressor genes (BRCA2, TP53 and PTEN) in patients with the four subtypes of breast cancer (luminal A, luminal B, Her-2-like and TNBC). BRCA1 and BRCA2 have been described as “breast cancer susceptibility genes,” so failure to properly repair mutations in these genes increases the risk for breast cancer [21]. Our data indicated that BRCA2 mutations, but not BRCA1 mutations, correlated highly with LCK metagene expression. It is worth noting that BRCA1 and BRCA2 are unrelated proteins that were discovered separately [22]. Until now, little has been reported about the relationship between the LCK metagene and BRCA1/2. Based on the current evidence, BRCA1 and LCK metagene expression may be independent markers of breast cancer.
Secondly, taking the gene members of the LCK metagene as the research object, we used a WGCNA to detect representative genes from the relevant gene modules of the LCK metagene. We also analyzed the DEGs between samples with high and low LCK metagene scores to identify co-expressed genes that correlated significantly with members of the LCK metagene in terms of their mRNA levels. Then, we assessed the overlap between these gene sets, and performed a survival analysis to determine which of these co-expressed genes were significantly associated with the prognosis of breast cancer patients. We explored the functions of these genes through an enrichment analysis, and further verified our findings with external data sets. We ultimately found 22 potential immune-related diagnostic and prognostic markers: WAS, ACAP1, SIT1, SEPT1, MAP4K1, CXCR3, TRAC, CD8A, CXCR6, TRBC2, TRAF1, PTPN7, CD3E, TBC1D10C, APOBEC3G, JAML, TRBV28, TRBV7-9, HLA-DPB1, S1PR4, CD6 and CST7.
The CD8 antigen is a cell-surface glycoprotein found on most cytotoxic T lymphocytes, and facilitates efficient cell-cell interactions within the immune system. CD8A encodes the CD8 alpha chain, and was one of the 22 immune-related genes identified in this study. Cytotoxic T lymphocytes can recognize and eliminate infected cells and tumor cells. CD8A homodimers on the surface of natural killer cells enable these cells to conjugate with and lyse multiple target cells, thus promoting survival [23]. Higher CD8A expression has been associated with a better prognosis in breast cancer patients [24]. Thus, therapeutically inducing CD8A could enhance the function of cytotoxic T lymphocytes, enabling them to kill more cancer cells.
CXCR3, another one of the 22 genes we identified, encodes a G protein-coupled receptor that is selective for three chemokines: CXCL9, CXCL10 and CXCL11. Binding of chemokines to CXCR3 induces cellular responses that are involved in leukocyte trafficking, most notably integrin activation, cytoskeletal changes and chemotactic migration [25]. In mice, CXCR3 deficiency was reported to promote the development of breast cancer by stimulating the M2 polarization of macrophages [26]. Higher CXCR3 expression was found to predict favorable outcomes in breast cancer patients treated with tamoxifen [27].
Another immune-related prognostic gene detected in this study was CXCR6. This gene is expressed by activated natural killer cells. In a previous study, irradiation was reported to induce CXCL16 chemokine expression in cancer cells and to enhance the migration of CXCR6+ natural killer cells to breast cancer cells for their destruction [28].
Among the 22 genes we identified, 15 (ACAP1, MAP4K1, CXCR3, TRAC, CD8A, CXCR6, TRBC2, TRAF1, CD3E, APOBEC3G, TRBV28, TRBV7-9, HLA-DPB1, CD6 and CST7) have previously been reported to be involved in the occurrence, development, malignant transformation and pathology of breast cancer, and to be associated with the survival and prognosis of patients [29–33]. This emphasizes the reliability and accuracy of our biological information mining results based on TCGA database screening and KM plotter database verification. The same gene expression trends were detected in matched tumor and non-tumor tissues, further validating our results. However, the remaining seven genes (S1PR4, SIT1, AML, PTPN7, WAS, TBC1D10C and SEPT1) have not been reported to be associated with breast cancer in experimental or clinical studies. Among these genes, SIT1 and JAML are of the greatest interest to us. SIT1 regulates the proliferation, activation and survival of memory T cells, thus affecting the generation of regulatory T cells, the immune escape of tumors and the resistance of tumors to immunotherapy [34]. JAML is involved in the proliferation and survival of T cells, as well as the production and release of cytokines and growth factors; thus, JAML regulates the sensitivity of tumor cells to relevant vaccines [35].
This study had several limitations. Firstly, our prognostic analyses of genes were based on overall survival, but information on relapse-free survival was lacking. Secondly, although most of the genes we identified could be verified in external databases and patient samples, the KM plotter platform could only verify 18 genes. Thirdly, the data from TCGA came from tissues; thus, although we identified 22 genes that could be therapeutic targets in breast cancer, we could not determine which cells highly expressed these genes. However, using an online tool (http://biogps.org), we were able to determine which types of cells typically express certain genes. For instance, CD19+ B cells highly express HLA-DBP1, and CD8+ T cells highly express CD8A. Single-cell sequencing can now be used to explore which cells highly express specific genes [36]; thus, our data could provide a reference for future single-cell sequencing analyses.
Although many studies have described the correlation between gene expression and survival in breast cancer patients, the results of most of these studies have been verified in animal tumor models, in vitro cell models or small numbers of human samples. The infiltration of immune cells (regulatory T cells, M2 tumor-related macrophages and CD20+ B cells) in the microenvironment of breast cancer is an important predictor of prognosis [37–39]. Retrospective studies have indicated that higher expression of immune checkpoint molecules (PD-L1, PD-1, CTLA-4 and LAG3) is associated with a higher survival rate in TNBC patients [40]. However, the complexity of the breast cancer microenvironment requires more comprehensive analyses in larger study populations. Fortunately, the rapid development of whole-genome sequencing and the development of high-throughput tumor databases such as TCGA have made it possible to analyze ‘big data’ from large-scale breast cancer populations. Furthermore, the ESTIMATE method can be applied to detect the infiltration of stromal and immune cells into tumor samples based on gene expression data [10]. In this study, we focused on immune microenvironment-related genes that were involved in the occurrence, development and malignant transformation of breast cancer and the overall survival of patients. Our results have helped to decode the complex microenvironment of breast cancer, and can be used as a source of potential immune-related diagnostic/prognostic markers or therapeutic targets for breast cancer.
Materials and Methods
Data sources and pre-processing
Counts data (see TCGA-BRCA counts.txt; all materials are available upon request), single-nucleotide polymorphism data (see TCGA.mutect.somatic.maf) and clinical follow-up information (see Merge clinilcal.txt) were downloaded from the database of TCGA. RNA-Seq data (reads per kilobase million) were downloaded from TCGA and converted into transcripts per kilobase million (TPM) expression profiles (see Merge TCGA-BRCA TPM.txt). Thirteen metagenes (see ImmuneScore.genes.ids.txt) corresponded to various immune cell types and reflected the corresponding immune functions [41]. The median mRNA levels of these immune metagenes were used for scoring (see meta.score.txt). The scores of immune cells in samples (six categories) were calculated through the Tumor Immune Estimation Resource (https://cistrome. shinyapps.io/timer/) (immu.score.txt) [42]. The immune and stromal scores of samples were calculated with the ESTIMATE function of the R software package (est.score.txt). The immune neoantigen score was calculated by a previously reported method [43] (Neoantigen.txt).
Screening representative genes in the breast cancer immune microenvironment
We used Spearman correlation coefficients to calculate the correlations between different immune-related scores in different breast cancer subtypes. Based on the results, the LCK metagene was selected as a representative gene in the breast cancer immune microenvironment. Next, we analyzed the relationship between LCK metagene expression and clinical stage. We also classified samples into high and low expression groups according to the median mRNA level of the LCK metagene, and performed KM analysis to determine the prognostic differences between the groups. In addition, we analyzed the relationship between LCK metagene expression and BRCA1, BRCA2, TP53 and PTEN mutations.
Specimen verification
We collected five paired tumor and non-tumor samples from breast cancer patients. Then, we used a PrimeScript™ RT reagent Kit (Cat#RR037A, Takara, Japan) to extract RNA from these samples and to reverse-transcribe the RNA to cDNA. Gene expression was detected by quantitative polymerase chain reaction experiments (TB Green™ Premix Ex Taq™ II, Cat#RR820A, Takara, Japan). Transcription products were quantified relative to beta-actin. The tumor and non-tumor tissues were collected with informed consent. This study was approved by the Ethics Committee of the Obstetrics and Gynecology Hospital of Fudan University.
Ethical statement
Samples from patients in this study were used with approval from the Ethics Committee of the Obstetrics and Gynecology Hospital of Fudan University and with consent from all the patients.
Acknowledgments
The authors thank members of their laboratory and their collaborators for their research work.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
Science and Technology Commission of Shanghai Municipality (No. 17411961100).
References
- 1. O’Meara T, Safonov A, Casadevall D, Qing T, Silber A, Killelea B, Hatzis C, Pusztai L. Immune microenvironment of triple-negative breast cancer in African-American and Caucasian women. Breast Cancer Res Treat. 2019; 175:247–59. https://doi.org/10.1007/s10549-019-05156-5 [PubMed]
- 2. Takada K, Kashiwagi S, Goto W, Asano Y, Takahashi K, Hatano T, Takashima T, Tomita S, Motomura H, Ohsawa M, Hirakawa K, Ohira M. Significance of re-biopsy for recurrent breast cancer in the immune tumour microenvironment. Br J Cancer. 2018; 119:572–79. https://doi.org/10.1038/s41416-018-0197-4 [PubMed]
- 3. Romaniuk A, Lyndin M. Immune microenvironment as a factor of breast cancer progression. Diagn Pathol. 2015; 10:79. https://doi.org/10.1186/s13000-015-0316-y [PubMed]
- 4. Xiao Y, Ma D, Zhao S, Suo C, Shi J, Xue MZ, Ruan M, Wang H, Zhao J, Li Q, Wang P, Shi L, Yang WT, et al; AME Breast Cancer Collaborative Group. Multi-Omics Profiling Reveals Distinct Microenvironment Characterization and Suggests Immune Escape Mechanisms of Triple-Negative Breast Cancer. Clin Cancer Res. 2019; 25:5002–5014. https://doi.org/10.1158/1078-0432.CCR-18-3524 [PubMed]
- 5. Burugu S, Asleh-Aburaya K, Nielsen TO. Immune infiltrates in the breast cancer microenvironment: detection, characterization and clinical implication. Breast Cancer. 2017; 24:3–15. https://doi.org/10.1007/s12282-016-0698-z [PubMed]
- 6. Steven A, Seliger B. The Role of Immune Escape and Immune Cell Infiltration in Breast Cancer. Breast Care (Basel). 2018; 13:16–21. https://doi.org/10.1159/000486585 [PubMed]
- 7. Zhao J, Wang Y, Lao Z, Liang S, Hou J, Yu Y, Yao H, You N, Chen K. Prognostic immune-related gene models for breast cancer: a pooled analysis. Onco Targets Ther. 2017; 10:4423–33. https://doi.org/10.2147/OTT.S144015 [PubMed]
- 8. Hernández-Prieto S, Romera A, Ferrer M, Subiza JL, López-Asenjo JA, Jarabo JR, Gómez AM, Molina EM, Puente J, González-Larriba JL, Hernando F, Pérez-Villamil B, Díaz-Rubio E, Sanz-Ortega J. A 50-gene signature is a novel scoring system for tumor-infiltrating immune cells with strong correlation with clinical outcome of stage I/II non-small cell lung cancer. Clin Transl Oncol. 2015; 17:330–8. https://doi.org/10.1007/s12094-014-1235-1 [PubMed]
- 9. Yoshihara K, Shahmoradgoli M, Martínez E, Vegesna R, Kim H, Torres-Garcia W, Treviño V, Shen H, Laird PW, Levine DA, Carter SL, Getz G, Stemke-Hale K, et al. Inferring tumour purity and stromal and immune cell admixture from expression data. Nat Commun. 2013; 4:2612. https://doi.org/10.1038/ncomms3612 [PubMed]
- 10. Donovan MJ, Fernandez G, Scott R, Khan FM, Zeineh J, Koll G, Gladoun N, Charytonowicz E, Tewari A, Cordon-Cardo C. Development and validation of a novel automated Gleason grade and molecular profile that define a highly predictive prostate cancer progression algorithm-based test. Prostate Cancer Prostatic Dis. 2018; 21:594–603. https://doi.org/10.1038/s41391-018-0067-4 [PubMed]
- 11. Tian Y, Li J, Zhou T, Tong D, Chi S, Kong X, Ding K, Li J. Spatially varying effects of predictors for the survival prediction of nonmetastatic colorectal Cancer. BMC Cancer. 2018; 18:1084. https://doi.org/10.1186/s12885-018-4985-2 [PubMed]
- 12. do Valle IF, Giampieri E, Simonetti G, Padella A, Manfrini M, Ferrari A, Papayannidis C, Zironi I, Garonzi M, Bernardi S, Delledonne M, Martinelli G, Remondini D, Castellani G. Optimized pipeline of MuTect and GATK tools to improve the detection of somatic single nucleotide polymorphisms in whole-exome sequencing data. BMC Bioinformatics. 2016 (Suppl 12); 17:341. https://doi.org/10.1186/s12859-016-1190-7 [PubMed]
- 13. Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014; 15:550. https://doi.org/10.1186/s13059-014-0550-8 [PubMed]
- 14. Reimand J, Isserlin R, Voisin V, Kucera M, Tannus-Lopes C, Rostamianfar A, Wadi L, Meyer M, Wong J, Xu C, Merico D, Bader GD. Pathway enrichment analysis and visualization of omics data using g:Profiler, GSEA, Cytoscape and EnrichmentMap. Nat Protoc. 2019; 14:482–517. https://doi.org/10.1038/s41596-018-0103-9 [PubMed]
- 15. Hsu HM, Chu CM, Chang YJ, Yu JC, Chen CT, Jian CE, Lee CY, Chiang YT, Chang CW, Chang YT. Six novel immunoglobulin genes as biomarkers for better prognosis in triple-negative breast cancer by gene co-expression network analysis. Sci Rep. 2019; 9:4484. https://doi.org/10.1038/s41598-019-40826-w [PubMed]
- 16. Pommier SJ, Morgan RE, Limbach KE, Jackson CM, Naik AM, Peckham JL, Muller PJ, Condron ME, Jameson NE, Pommier RF. Surgical resection of breast cancers: molecular analysis of cancer stem cells in residual disease. Surgery. 2019; 165:1008–13. https://doi.org/10.1016/j.surg.2019.01.001 [PubMed]
- 17. Goetz MP, Gradishar WJ, Anderson BO, Abraham J, Aft R, Allison KH, Blair SL, Burstein HJ, Dang C, Elias AD, Farrar WB, Giordano SH, Goldstein LJ, et al. NCCN Guidelines Insights: Breast Cancer, Version 3.2018. J Natl Compr Canc Netw. 2019; 17:118–26. https://doi.org/10.6004/jnccn.2019.0009 [PubMed]
- 18. Tong CW, Wu M, Cho WC, To KK. Recent Advances in the Treatment of Breast Cancer. Front Oncol. 2018; 8:227. https://doi.org/10.3389/fonc.2018.00227 [PubMed]
- 19. Nakasone ES, Hurvitz SA, McCann KE. Harnessing the immune system in the battle against breast cancer. Drugs Context. 2018; 7:212520. https://doi.org/10.7573/dic.212520 [PubMed]
- 20. Esteva FJ, Hubbard-Lucey VM, Tang J, Pusztai L. Immunotherapy and targeted therapy combinations in metastatic breast cancer. Lancet Oncol. 2019; 20:e175–86. https://doi.org/10.1016/S1470-2045(19)30026-9 [PubMed]
- 21. Friedenson B. The BRCA1/2 pathway prevents hematologic cancers in addition to breast and ovarian cancers. BMC Cancer. 2007; 7:152. https://doi.org/10.1186/1471-2407-7-152 [PubMed]
- 22. Irminger-Finger I, Ratajska M, Pilyugin M. New concepts on BARD1: regulator of BRCA pathways and beyond. Int J Biochem Cell Biol. 2016; 72:1–17. https://doi.org/10.1016/j.biocel.2015.12.008 [PubMed]
- 23. Zhu J, Peng T, Johnston C, Phasouk K, Kask AS, Klock A, Jin L, Diem K, Koelle DM, Wald A, Robins H, Corey L. Immune surveillance by CD8αα+ skin-resident T cells in human herpes virus infection. Nature. 2013; 497:494–97. https://doi.org/10.1038/nature12110 [PubMed]
- 24. Araujo JM, Gomez AC, Aguilar A, Salgado R, Balko JM, Bravo L, Doimi F, Bretel D, Morante Z, Flores C, Gomez HL, Pinto JA. Effect of CCL5 expression in the recruitment of immune cells in triple negative breast cancer. Sci Rep. 2018; 8:4899. https://doi.org/10.1038/s41598-018-23099-7 [PubMed]
- 25. Lasagni L, Francalanci M, Annunziato F, Lazzeri E, Giannini S, Cosmi L, Sagrinati C, Mazzinghi B, Orlando C, Maggi E, Marra F, Romagnani S, Serio M, Romagnani P. An alternatively spliced variant of CXCR3 mediates the inhibition of endothelial cell growth induced by IP-10, Mig, and I-TAC, and acts as functional receptor for platelet factor 4. J Exp Med. 2003; 197:1537–49. https://doi.org/10.1084/jem.20021897 [PubMed]
- 26. Oghumu S, Varikuti S, Terrazas C, Kotov D, Nasser MW, Powell CA, Ganju RK, Satoskar AR. CXCR3 deficiency enhances tumor progression by promoting macrophage M2 polarization in a murine breast cancer model. Immunology. 2014; 143:109–19. https://doi.org/10.1111/imm.12293 [PubMed]
- 27. Hilborn E, Sivik T, Fornander T, Stål O, Nordenskjöld B, Jansson A. C-X-C ligand 10 and C-X-C receptor 3 status can predict tamoxifen treatment response in breast cancer patients. Breast Cancer Res Treat. 2014; 145:73–82. https://doi.org/10.1007/s10549-014-2933-7 [PubMed]
- 28. Yoon MS, Pham CT, Phan MT, Shin DJ, Jang YY, Park MH, Kim SK, Kim S, Cho D. Irradiation of breast cancer cells enhances CXCL16 ligand expression and induces the migration of natural killer cells expressing the CXCR6 receptor. Cytotherapy. 2016; 18:1532–42. https://doi.org/10.1016/j.jcyt.2016.08.006 [PubMed]
- 29. Hoffman JD, Graff RE, Emami NC, Tai CG, Passarelli MN, Hu D, Huntsman S, Hadley D, Leong L, Majumdar A, Zaitlen N, Ziv E, Witte JS. Cis-eQTL-based trans-ethnic meta-analysis reveals novel genes associated with breast cancer risk. PLoS Genet. 2017; 13:e1006690. https://doi.org/10.1371/journal.pgen.1006690 [PubMed]
- 30. Frazao A, Messaoudene M, Nunez N, Dulphy N, Roussin F, Sedlik C, Zitvogel L, Piaggio E, Toubert A, Caignard A. CD16(+)NKG2A(high) Natural Killer Cells Infiltrate Breast Cancer-Draining Lymph Nodes. Cancer Immunol Res. 2019; 7:208–218. https://doi.org/10.1158/2326-6066.CIR-18-0085 [PubMed]
- 31. Sakaguchi N, Maeda K. Germinal Center B-Cell-Associated Nuclear Protein (GANP) Involved in RNA Metabolism for B Cell Maturation. Adv Immunol. 2016; 131:135–86. https://doi.org/10.1016/bs.ai.2016.02.003 [PubMed]
- 32. Chaudhuri S, Cariappa A, Tang M, Bell D, Haber DA, Isselbacher KJ, Finkelstein D, Forcione D, Pillai S. Genetic susceptibility to breast cancer: HLA DQB*03032 and HLA DRB1*11 may represent protective alleles. Proc Natl Acad Sci USA. 2000; 97:11451–54. https://doi.org/10.1073/pnas.97.21.11451 [PubMed]
- 33. Tong WL, Callahan BM, Tu YN, Zaman S, Chobrutskiy BI, Blanck G. Immune receptor recombinations from breast cancer exome files, independently and in combination with specific HLA alleles, correlate with better survival rates. Breast Cancer Res Treat. 2019; 173:167–177. https://doi.org/10.1007/s10549-018-4961-1 [PubMed]
- 34. Cerkovnik P, Novaković BJ, Stegel V, Novaković S. Changes in expression of genes involved in antitumor immunity in mice vaccinated with tumor vaccine composed of irradiated syngeneic tumor cells and CpG oligodeoxynucleotides. Mol Immunol. 2016; 79:1–13. https://doi.org/10.1016/j.molimm.2016.09.016 [PubMed]
- 35. Roh SE, Jeong Y, Kang MH, Bae YS. Junctional adhesion molecules mediate transendothelial migration of dendritic cell vaccine in cancer immunotherapy. Cancer Lett. 2018; 434:196–205. https://doi.org/10.1016/j.canlet.2018.07.029 [PubMed]
- 36. Wagner J, Rapsomaniki MA, Chevrier S, Anzeneder T, Langwieder C, Dykgers A, Rees M, Ramaswamy A, Muenst S, Soysal SD, Jacobs A, Windhager J, Silina K, et al. A Single-Cell Atlas of the Tumor and Immune Ecosystem of Human Breast Cancer. Cell. 2019; 177:1330–1345.e18. https://doi.org/10.1016/j.cell.2019.03.005 [PubMed]
- 37. Dziobek K, Biedka M, Nowikiewicz T, Szymankiewicz M, Łukaszewska E, Dutsch-Wicherek M. Analysis of Treg cell population in patients with breast cancer with respect to progesterone receptor status. Contemp Oncol (Pozn). 2018; 22:236–39. https://doi.org/10.5114/wo.2018.80038 [PubMed]
- 38. Lao L, Fan S, Song E. Tumor Associated Macrophages as Therapeutic Targets for Breast Cancer. Adv Exp Med Biol. 2017; 1026:331–70. https://doi.org/10.1007/978-981-10-6020-5_16 [PubMed]
- 39. Arias-Pulido H, Cimino-Mathews A, Chaher N, Qualls C, Joste N, Colpaert C, Marotti JD, Foisey M, Prossnitz ER, Emens LA, Fiering S. The combined presence of CD20 + B cells and PD-L1 + tumor-infiltrating lymphocytes in inflammatory breast cancer is prognostic of improved patient outcome. Breast Cancer Res Treat. 2018; 171:273–82. https://doi.org/10.1007/s10549-018-4834-7 [PubMed]
- 40. Wang K, Li HL, Xiong YF, Shi Y, Li ZY, Li J, Zhang X, Li HY. Development and validation of nomograms integrating immune-related genomic signatures with clinicopathologic features to improve prognosis and predictive value of triple-negative breast cancer: A gene expression-based retrospective study. Cancer Med. 2019; 8:686–700. https://doi.org/10.1002/cam4.1880 [PubMed]
- 41. Safonov A, Jiang T, Bianchini G, Győrffy B, Karn T, Hatzis C, Pusztai L. Immune Gene Expression Is Associated with Genomic Aberrations in Breast Cancer. Cancer Res. 2017; 77:3317–24. https://doi.org/10.1158/0008-5472.CAN-16-3478 [PubMed]
- 42. Li T, Fan J, Wang B, Traugh N, Chen Q, Liu JS, Li B, Liu XS. TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res. 2017; 77:e108–10. https://doi.org/10.1158/0008-5472.CAN-17-0307 [PubMed]
- 43. Rooney MS, Shukla SA, Wu CJ, Getz G, Hacohen N. Molecular and genetic properties of tumors associated with local immune cytolytic activity. Cell. 2015; 160:48–61. https://doi.org/10.1016/j.cell.2014.12.033 [PubMed]
- 44. Langfelder P, Horvath S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics. 2008; 9:559. https://doi.org/10.1186/1471-2105-9-559 [PubMed]