Introduction
Age-related macular degeneration (AMD), a progressive chronic disease of the central retina, is a major cause of blindness worldwide [1]. The prevalence of AMD is likely to increase as a consequence of the exponential increase in the aging population, and the projected number of people with AMD in 2040 is around 288 million [2]. Therefore, AMD will be a major medical and socioeconomic challenge worldwide in the coming years.
The precise pathogenesis is still poorly understood. It is generally accepted that AMD is the result of a complex interaction between genetic and environmental factors [3].
Several risk factors involved in the pathogenesis of AMD have been described, including genetic predisposition, age, and other modifiable factors such as smoking, light and ultraviolet (UV) exposure, dietary factors, and hypertension, which may influence the molecular mechanisms or cellular processes involved in development of the disease.
It is well established that the prevalence and incidence of many diseases are related to the month or season of birth [4–8]. There is increasing evidence that environmental factors in prenatal and early postnatal life can have significant effect on the development of various diseases later in adulthood. According to the “fetal origins hypothesis” of the developmental origins of the diseases, the susceptibility to many chronic diseases is determined in utero and has a lasting effect on the disease process [9].
No study has investigated whether there is an association between the season or month of birth and the risk of AMD. The aim of this study was to examine whether the month or season of birth is related to the risk of neovascular AMD (n-AMD) in Italy.
Results
Overall, in Italy, 60,818 patients received intravitreal injections of anti- vascular endothelial growth factor (VEGF) for n-AMD from January 1, 2013 to July 29, 2015. Consecutive years having at least 1000 individuals treated were found during the span 1925 to 1944 (included). A total of 46,826 patients born in these years were treated with anti-VEGF; after excluding 981 patients born outside Italy, a final sample of 45,845 patients (19,207 men and 26,638 women) who were born in Italy and treated with intravitreal anti-VEGF for n-AMD was obtained. In the same years, 20,140,426 people were born in Italy (10,334,262 males and 9,806,164 females) and were included in the analysis.
Figure 1 shows the observed cases and the expected cases of n-AMD by month. In the analysis of the season-specific risk of n-AMD, we compared the number of people with n-AMD born in each season with that in the general population. The risk of n-AMD was 2.5% higher for people born in summer (OR = 1.03; [95% CI, 1.01–1.06]; χ2 = 3.057, P = 0.002; Bonferroni corrected P = 0.008) and 3% lower for those born in winter (OR = 0.96 ([95% CI, 0.94–0.98]; χ2 4.03, P = 0.0001; Bonferroni corrected P = 0.0004) (Table 1 and Fig. 2).
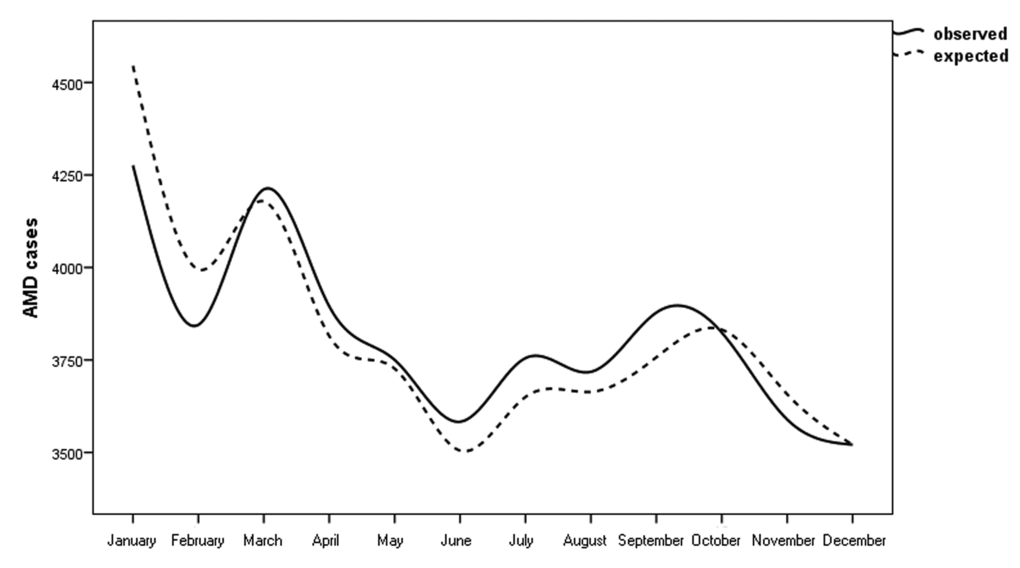
Figure 1. Pooled analysis of observed/expected births in people with neovascular AMD in Italy between 1925 and 1944 (n = 45845) with 95% confidence intervals.
Table 1. All borns, observed number of people with neovascular AMD compared with the expected number, odds ratio with 95% CI, according to season.
| All borns | Observed n-AMD cases | Expected n-AMD cases | OR, 95 CI, p | |||||||||
| male | female | all | male | female | all | male | female | all | male | female | all | |
| Winter | 2889994 | 2697229 | 5587223 | 5245 | 7087 | 12332 | 5371,3 | 7326,9 | 12718,0 | 0.967; 0.94-0.99; p=0.042 1 | 0.955; 0.93-0.98; p=0.001 3 | 0.958; 0.94-0.98; p=0.0001 5 |
| Spring | 2490013 | 2362684 | 4852697 | 4756 | 6472 | 11228 | 4627,9 | 6418,1 | 11046,0 | 1.037; 1.01-1.07; p=0.030 2 | 1.011; 0.98-1.04; p=0.439 | 1.022; 1.01-1.04; p=0.047 6 |
| Summer | 2498596 | 2365135 | 4863731 | 4739 | 6612 | 11351 | 4643,8 | 6424,8 | 11071,2 | 1.027; 0.99-1.06; 0.108 | 1.039; 1.011-1.069; p=0.006 4 | 1.034; 1.01-1.06; p=0.002 7 |
| Autumn | 2455659 | 2381116 | 4836775 | 4467 | 6467 | 10934 | 4564,0 | 6468,2 | 11009,8 | 0.972; 0.94-1.01; p=0.099 | 1.0; 0.97-1.03; p=0.986 | 0.990; 0.97-1.01; p=0.407 |
| Total | 10334262 | 9806164 | 20140426 | 19207 | 26638 | 45845 | 19207 | 26638 | 45845 | |||
| p (Bonferroni correction) | 1 p=0.168 | 3 p=0.004 | 5 p=0.0004 | |||||||||
| 2 p=0.120 | 4 p=0.024 | 6 p=0.188 | ||||||||||
| 7 p=0.008 | ||||||||||||
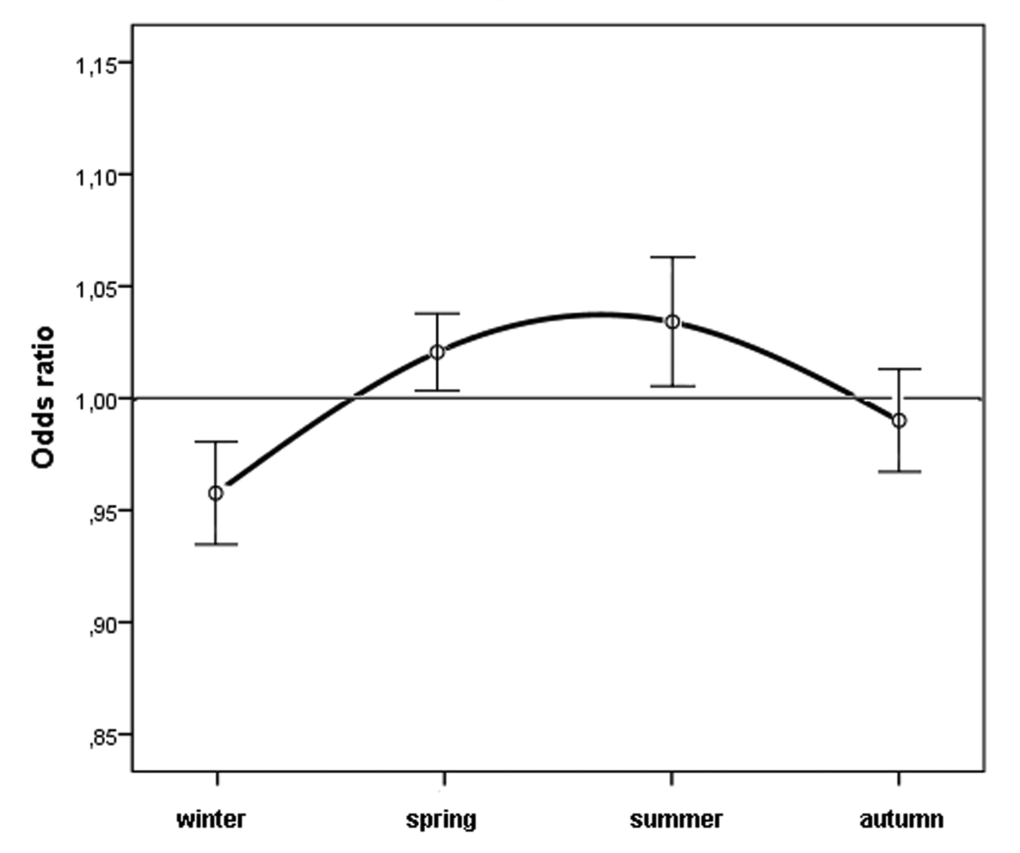
Figure 2. Odds ratios for people with neovascular AMD being born in different seasons in Italy between 1925 and 1944 (n = 45845) with 95% confidence intervals.
When we considered the month of birth, the risk of n-AMD was 5.9% lower for people born in January (OR = 0.93 [95% CI, 0.91–0.96); χ2 = 4.20, P = 0.0001, Bonferroni corrected P = 0.0012) (Table 2 and Fig. 3). The results of the subgroup analysis are shown in Tables 1 and 2, and Figures 4 and 5. In women, the risk of n-AMD was 2.9% higher for those born in summer (OR = 1.04 [95% CI, 1.01–1.07); χ2 = 2.68, P = 0.006, Bonferroni corrected P = 0.024) and 3.2% lower for those born in winter (OR = 0.95 [95% CI, 0.93–0.98); χ2 = 3.29, P = 0.001, Bonferroni corrected P = 0.004). In women, no significant differences were found between different months after Bonferroni correction. In men, no significant differences were found between seasons or months.
Table 2. All borns, observed number of people with neovascular AMD compared with the expected number, odds ratio with 95% CI, according to month.
| All borns | Observed n-AMD cases | Expected n-AMD cases | OR, 95 CI, p | |||||||||
| male | female | all | male | female | all | male | female | all | male | female | all | |
| January | 1052187 | 944371 | 1996558 | 1838 | 2438 | 4276 | 1955,6 | 2565,3 | 4544,7 | 0.933; 0.89-0.98; p=0.005 1 | 0.945; 0.91-0.98; p=0.008 3 | 0.935; 0.91-0.96; p=0.0001 5 |
| February | 897659 | 857274 | 1754933 | 1598 | 2247 | 3845 | 1668,4 | 2328,7 | 3994,7 | 0.954; 0.90-1.01; p=0.070 | 0.961; 0.92-1.01; p=0.075 | 0.959; 0.93-0.99; p=0.013 6 |
| March | 940148 | 895584 | 1835732 | 1809 | 2402 | 4211 | 1747,3 | 2432,8 | 4178,6 | 1.041; 0.99-1.09; P=0.121 | 0.986; 0.95-1.03; p=0.512 | 1.008; 0.97-1.04; p=0.599 |
| April | 858981 | 816864 | 1675845 | 1634 | 2261 | 3895 | 1596,5 | 2219,0 | 3814,7 | 1.026; 0.98-1.08; p=0.326 | 1.020; 0.98-1.07; p=0.351 | 1.023; 0.99-1.06; p=0.174 |
| May | 839404 | 797651 | 1637055 | 1590 | 2160 | 3750 | 1560,1 | 2166,8 | 3726,4 | 1.021; 0.97-1.07; p=0.429 | 0.997; 0.95-1.04; p=0.879 | 1.006; 0.97-1.04; p=0.686 |
| june | 791628 | 748169 | 1539797 | 1532 | 2051 | 3583 | 1471,3 | 2032,4 | 3505,0 | 1.045; 0.99-1.10; p=0.099 | 1.010; 0.97-1.06; p=0.667 | 1.024; 0.99-1.06; p=0.169 |
| July | 823977 | 779541 | 1603518 | 1593 | 2162 | 3755 | 1531,4 | 2117,6 | 3650,0 | 1.044; 0.99-1.010; p=0.101 | 1.023; 0.98-1.07; p=0.314 | 1.031; 0.99-1.07; p=0.069 |
| August | 826973 | 782608 | 1609581 | 1561 | 2157 | 3718 | 1537,0 | 2125,9 | 3663,8 | 1.017; 0.97-1.07; p=0.523 | 1.016; 0.97-1.06; p=0.482 | 1.016; 0.98-1.05; p=0.350 |
| September | 847646 | 802986 | 1650632 | 1585 | 2293 | 3878 | 1575,4 | 2181,3 | 3757,3 | 1.006; 0.96-1.06; p=0.800 | 1.056; 1.01-1.10; p=0.012 4 | 1.035; 1.01-1.07; p=0.039 7 |
| October | 863423 | 819891 | 1683314 | 1575 | 2249 | 3824 | 1604,7 | 2227,2 | 3831,7 | 0.979; 0.93-1.03; p=0.438 | 1.011; 0.97-1.06; p=0.629 | 0.998; 0.96-1.03; p=0.897 |
| November | 821012 | 785681 | 1606693 | 1451 | 2138 | 3589 | 1525,9 | 2134,3 | 3657,3 | 0.946; 0.89-0.99; p=0.045 2 | 1.002; 0.96-1.05; p=0.933 | 0.980; 0.95-1.01; p=0.239 |
| December | 771224 | 775544 | 1546768 | 1441 | 2080 | 3521 | 1433,4 | 2106,7 | 3520,9 | 1.005; 0.95-1.06; p=0.834 | 0.986; 0.94-1.03; p=0.543 | 1.0; 0.97-1.04; p=0.998 |
| total | 10334262 | 9806164 | 20140426 | 19207 | 26638 | 45845 | 19207 | 26638 | 45845 | |||
| p (Bonferroni correction) | 1 p=0.06 | 3 p=0.096 | 5 p=0.0012 | |||||||||
| 2 p=0.54 | 4 p=0.144 | 6 p=0.156 | ||||||||||
| 7 p=0.468 | ||||||||||||
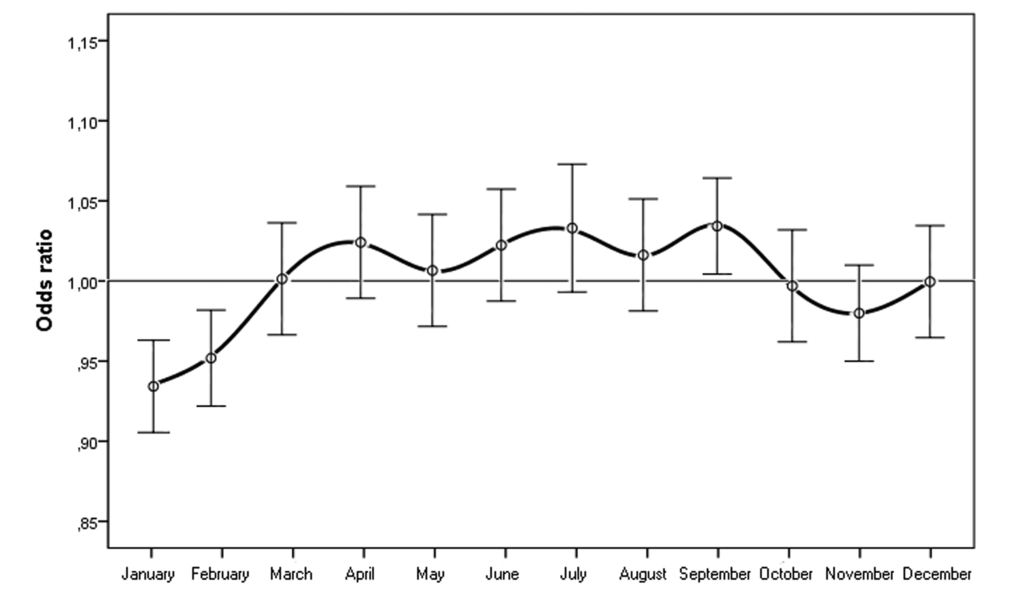
Figure 3. Odds ratios for people with neovascular AMD being born in different months in Italy between 1925 and 1944 (n = 45845) with 95% confidence intervals.
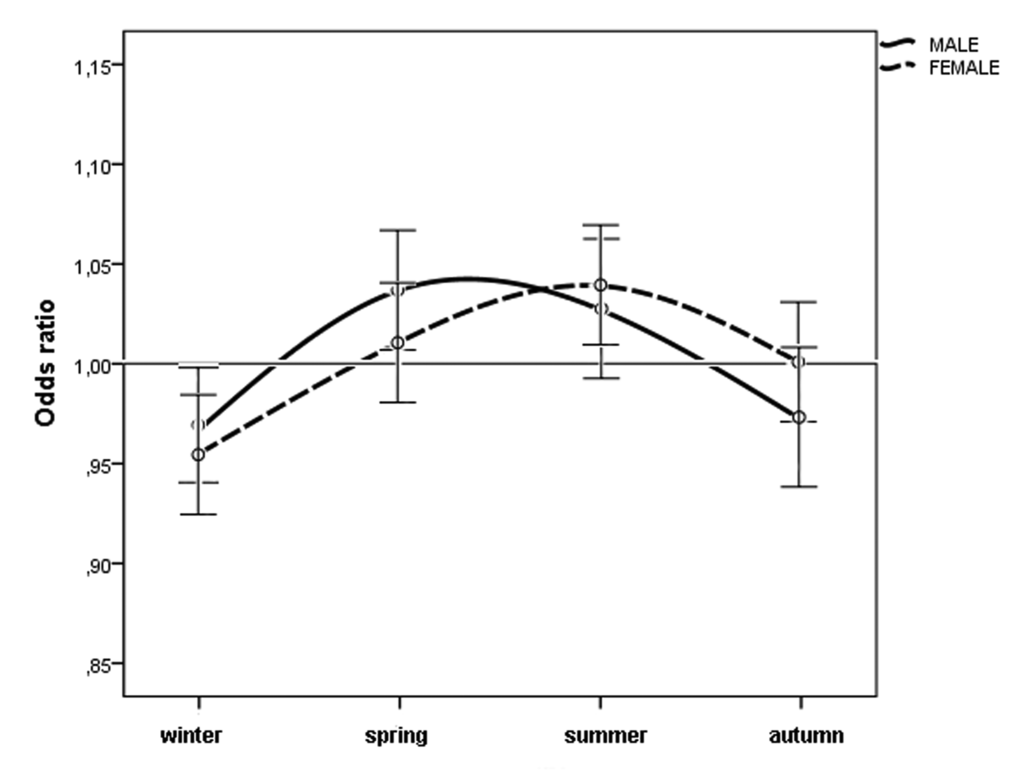
Figure 4. Odds ratios for men and women with neovascular AMD being born in different seasons in Italy between 1925 and 1944 (n = 45845) with 95% confidence intervals.
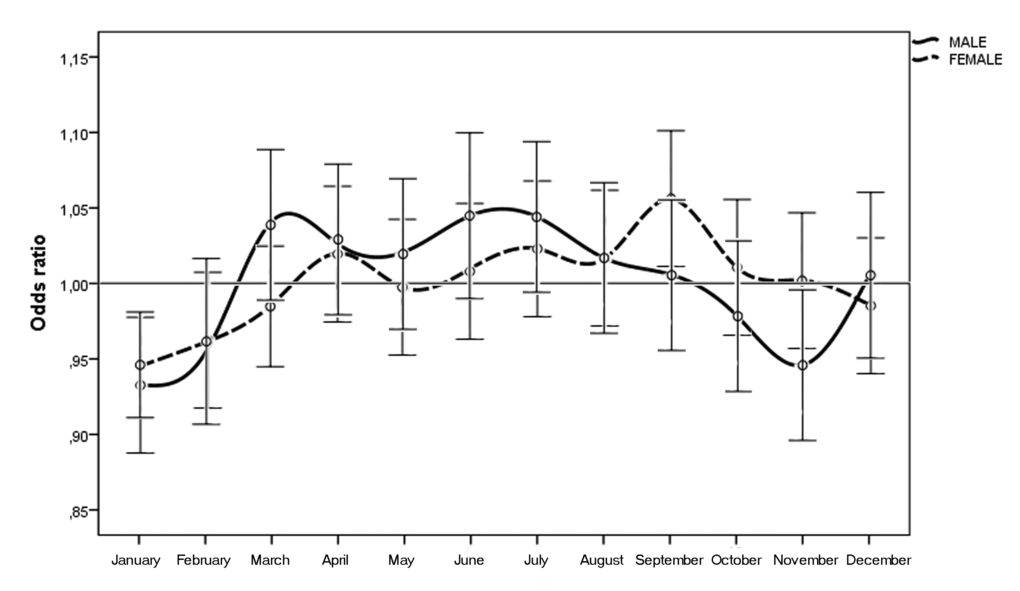
Figure 5. Odds ratios for men and women with neovascular AMD being born in different months in Italy between 1925 and 1944 (n = 45845) with 95% confidence intervals.
Discussion
In this study, we analyzed the month of birth in the years 1925 to 1944 of patients with n- AMD who were treated with intravitreal anti-VEGF injections in Italy. We found a seasonal variation: the number of people who developed n-AMD was higher than expected in summer, and lower in winter.
Several diseases exhibit an effect of birth month or birth season on the incidence [4–8]. A meta-analysis of studies on multiple sclerosis showed a higher risk in patients born in April and May and lower risk in those born in October and November, as well as a relationship between risk and latitude [6]. Similar trends have been reported for other immune-mediated diseases. A large study of people with type 1 diabetes in the USA found that the relative percentage of observed to expected births was lower in November–February and higher in April–July. This effect was seen in both men and women but was evident only in regions in the northern latitudes, which suggested a role of factors related to geographic latitude such as solar irradiance [5].
A national study in the UK showed that the distribution of births of patients who developed an immune-mediated disease (e.g., multiple sclerosis, rheumatoid arthritis, ulcerative colitis, or systemic lupus erythematosus) differed significantly from that of the general population. The peak of births for those who developed one of these diseases occurred in April and a trough in October, and the risk correlated inversely with predicted second-trimester UVB exposure and third-trimester vitamin D status [7].
Several seasonal factors acting during pregnancy and the perinatal period have been proposed to explain these data, including infections, dietary factors (e.g., low intake of antioxidants in winter), and vitamin D deficiency through lack of sun exposure during pregnancy [8]. This latter plays a significant role, since increased serum vitamin D concentration is associated with a decreased risk of multiple sclerosis [10,11], and vitamin D supplementation during the first year of life is associated with a significantly decreased risk of type 1 diabetes [12].
The biologically active form of vitamin D is generated in the skin during exposure to UV radiation from sunlight, and its levels follow a seasonal distribution. In the Unites States, a study of more than 3 million people reported a peak of 25-hydroxyvitamin D (25(OH)D) level in August and lowest level in February [13]. In young women in Italy, the serum concentration in summer was reported to be twice that in winter (120 nmol/l in July vs 60 nmol/l in February) [14]. During pregnancy, when a higher amount of vitamin D is required, many pregnant women can develop a vitamin D deficiency. In Ireland, a higher prevalence of vitamin D deficiency (25(OH)D <27.5 nmol/l) was observed in pregnant than in nonpregnant women, both in spring (44% vs. 18%) and in winter (35% vs 29%) [15]. Newborns can have lower vitamin D levels than their mothers. In the USA, in winter and spring, a vitamin D deficiency (25(OH)D <37 nmol/l) was found in 5% of new mothers and in 9.7% of their newborns [16]. In Italy, low vitamin D levels (25(OH)D<25 nmol/l) were found in 18% of new mothers and in 38% of their newborns [17].
Gestational vitamin D concentration is considered important for the development of the immune system. The vitamin D acts as an immunomodulator by inhibiting the proliferation of the T cells, shifting the T-cell population toward noninflammatory T helper 2 (Th2) and T-regulatory cells, and suppressing the transcription of genes encoding key Th1 proinflammatory cytokines [18]. Genes associated with many autoimmune diseases are enriched for vitamin D receptor-binding sites [19–21], suggesting that the combined effect of vitamin D deficiency, genetic variation, and exposure to other environmental agents may concur in the pathogenesis. Seasonal changes in immune function are also mediated by the pineal hormone melatonin, which has a proinflammatory action [31], by increasing the release of Th1 cytokines [22].
In animal models, gestational vitamin D deficiency affects brain development [23], and the myelination process [24], and it could have an effect on retinal cells. Vitamin D is involved in the pathogenesis of neovascular AMD, and a significant correlation has been found between reduced plasma vitamin D level and the prevalence of AMD [25–28]. A recent meta-analysis reported a correlation between latitude, insolation, and the incidence of severe AMD; higher incidence was found in locations with insolation <3 kWh/m2/day compared with those with insolation >3 kWh/m2/day (P < 0.001) [29].
An effect of the month of birth has also been reported for ocular conditions such as myopia. In Israel, a study found increased rates of moderate and severe myopia in people born in summer and lower rates in those born in winter (9% and 8.5%, and 2.2% and 2.7%, respectively). These differences correlated with the number of daylight hours, which is thought to relate to the melatonin level, or an imbalance in the melatonin–dopamine relationship [30]. Similarly, a study in the UK reported a 4.1% increased rate of severe myopia in summer–autumn and 3.6% lower rate in winter–spring. However, the lack of relationship between severe myopia and the number of daylight hours, suggested the influence of other season-related factors, such as the birth weight [31].
Studies of the effect of birth month or birth season have observed significant variability in birth month in the general population. This factor, combined with the inevitable heterogeneity in region of origin and year of birth, can lead to false-positive associations [32]. In this study, we included people born throughout Italy over a 20-year period (1925–1944), thus avoiding the possible effect of a single year.
This study has several limitations. Although season of birth is a well-defined variable, it is only one possible factor in the pathogenesis of the n-AMD. It is associated with various other environmental factors, such as meteorological factors, daily sunlight exposure, and alterations in air pollution and food supply, as well as behavioral variables, including dietary habits and physical activity levels. Personal factors, such as smoking, urban or rural residency, alimentary habits, and migration (inside or outside Italy) could have caused different levels of exposure to risk factors and had an effect on the development of n-AMD.
In conclusion, this study shows that there is seasonal variation in the risk of developing neovascular AMD in Italy. The factors causing such changes should be determined.
Methods
In this study, all consecutive patients recorded in the anti-VEGF monitoring registry of the Italian Medicines Agency (AIFA) who received intravitreal injections of anti-VEGF for n-AMD between January 1, 2013 and July 29, 2015 were included. The system monitors the registration process throughout the country and uploading of clinical data, and allows access to data about reimbursement by the National Health Service [33,34].
The inclusion criteria were patients born in Italy and treated with intravitreal anti-VEGF injection for n-AMD. Patients born outside Italy were excluded. The data obtained from the AIFA database for each patient were year and month of birth, region of birth, and sex. Other parameters allowing personal identification (such as city or date of birth, or initials of patients) were not provided. The drugs used for intravitreal injections of anti-VEGF recorded in the AIFA registry included in this study were pegaptanib sodium (Macugen; EyetechInc, Palm Beach Gardens, FL), bevacizumab (Avastin; Genentech Inc., South San Francisco, CA), ranibizumab (Lucentis; Genentech Inc.), and aflibercept (EYLEA; Regeneron Pharmaceuticals, Inc, Tarrytown, NY).
From the initial cohort of patients, to ensure a sufficient sample size across all 12 months, as previously described [4], we included in the analysis only those consecutive years having at least 1000 individuals treated. For the same years, the numbers of all births in Italy were extracted from the Italian National Institute of Statistics along with information about the month of birth, sex, and region of birth. These data were used to calculate the expected number of births of people who would later develop AMD under the assumption that the birth distributions for patients and the whole population are equal.
Institutional review board approval was obtained.
Statistical analysis
The n-AMD incidence rate was calculated per 100,000 residents per year. The distribution of season of birth was compared between the study population (with n-AMD) and the corresponding Italian population. We compared the observed number of n- AMD cases with the expected number of n-AMD cases in each season, and we analyzed the season-specific risk of n-AMD compared with the other 3 seasons using the odds ratios (ORs) and 95% confidence intervals (CIs) and a 2 × 2 chi-square test. Significant (P ≤ 0.05) values for a single season were corrected for the 4 comparisons using the Bonferroni correction.
We also compared the observed number of n-AMD cases with the expected number of n-AMD cases in each month, and we analyzed the month-specific risk of n-AMD compared with the other 11 months using ORs and 95% CIs and a 2 × 2 chi-square test. Significant (P ≤ 0.05) values for single months were corrected for the 12 comparisons using the Bonferroni correction.
Subgroup analyses were performed for women and men. We calculated 95% CIs based on the Poisson distribution for describing the variation in the number of events during a period of time.
Data were analyzed using IBM SPSS Software 22 version (IBM Corp., Armonk, NY, USA). All P-values were 2-sided, and P ≤ 0.05 was considered to be significant.
Conflicts of Interest
The authors have no conflict of interests to declare.
AIFA Disclosure.
The views expressed in this work are personal and may not be understood or quoted as being made on behalf of, or reflecting the position of AIFA, EMA, or of one of their committees or working parties.
References
- 1. Lim LS, Mitchell P, Seddon JM, Holz FG, Wong TY. Age-related macular degeneration. Lancet. 2012; 379:1728–38. https://doi.org/10.1016/S0140-6736(12)60282-7 [PubMed]
- 2. Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, Wong TY. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014; 2:e106–16. https://doi.org/10.1016/S2214-109X(13)70145-1 [PubMed]
- 3. Chakravarthy U, Wong TY, Fletcher A, Piault E, Evans C, Zlateva G, Buggage R, Pleil A, Mitchell P. Clinical risk factors for age-related macular degeneration: a systematic review and meta-analysis. BMC Ophthalmol. 2010; 10:31. https://doi.org/10.1186/1471-2415-10-31 [PubMed]
- 4. Boland MR, Shahn Z, Madigan D, Hripcsak G, Tatonetti NP. Birth month affects lifetime disease risk: a phenome-wide method. J Am Med Inform Assoc. 2015; 22:1042–53. https://doi.org/10.1093/jamia/ocv046 [PubMed]
- 5. Kahn HS. 1, Morgan TM, Case LD, Dabelea D, Mayer-Davis EJ, Lawrence JM, Marcovina SM, Imperatore G; SEARCH for Diabetes in Youth Study Group. Association of Type 1 Diabetes With Month of Birth Among U.S. Youth. Diabetes Care. 2009; 32:2010–15. https://doi.org/10.2337/dc09-0891 [PubMed]
- 6. Dobson R, Giovannoni G, Ramagopalan S. The month of birth effect in multiple sclerosis: systematic review, meta-analysis and effect of latitude. J Neurol Neurosurg Psychiatry. 2013; 84:427–32. https://doi.org/10.1136/jnnp-2012-303934 [PubMed]
- 7. Disanto G, Chaplin G, Morahan JM, Giovannoni G, Hyppönen E, Ebers GC, Ramagopalan SV. Month of birth, vitamin D and risk of immune-mediated disease: a case control study. BMC Med. 2012; 10:69. https://doi.org/10.1186/1741-7015-10-69 [PubMed]
- 8. Salzer J, Svenningsson A, Sundström P. Season of birth and multiple sclerosis in Sweden. Acta Neurol Scand. 2010; 121:20–23. https://doi.org/10.1111/j.1600-0404.2009.01181.x [PubMed]
- 9. Rasmussen KM. The “fetal origins” hypothesis: challenges and opportunities for maternal and child nutrition. Annu Rev Nutr. 2001; 21:73–95. https://doi.org/10.1146/annurev.nutr.21.1.73 [PubMed]
- 10. Simpson S
Jr , Taylor B, Blizzard L, Ponsonby AL, Pittas F, Tremlett H, Dwyer T, Gies P, van der Mei I. Higher 25-hydroxyvitamin D is associated with lower relapse risk in multiple sclerosis. Ann Neurol. 2010; 68:193–203. [PubMed] - 11. Soilu-Hänninen M, Laaksonen M, Laitinen I, Erälinna JP, Lilius EM, Mononen I. A longitudinal study of serum 25-hydroxyvitamin D and intact parathyroid hormone levels indicate the importance of vitamin D and calcium homeostasis regulation in multiple sclerosis. J Neurol Neurosurg Psychiatry. 2008; 79:152–57. https://doi.org/10.1136/jnnp.2006.105320 [PubMed]
- 12. Hyppönen E, Läärä E, Reunanen A, Järvelin MR, Virtanen SM. Intake of vitamin D and risk of type 1 diabetes: a birth-cohort study. Lancet. 2001; 358:1500–03. https://doi.org/10.1016/S0140-6736(01)06580-1 [PubMed]
- 13. Kasahara AK, Singh RJ, Noymer A. Vitamin D (25OHD) Serum Seasonality in the United States. PLoS One. 2013; 8:e65785. https://doi.org/10.1371/journal.pone.0065785 [PubMed]
- 14. Romagnoli E, Caravella P, Scarnecchia L, Martinez P, Minisola S. Hypovitaminosis D in an Italian population of healthy subjects and hospitalized patients. Br J Nutr. 1999; 81:133–37. [PubMed]
- 15. Holmes VA, Barnes MS, Alexander HD, McFaul P, Wallace JM. Vitamin D deficiency and insufficiency in pregnant women: a longitudinal study. Br J Nutr. 2009; 102:876–81. https://doi.org/10.1017/S0007114509297236 [PubMed]
- 16. Bodnar LM, Catov JM, Roberts JM, Simhan HN. Prepregnancy obesity predicts poor vitamin D status in mothers and their neonates. J Nutr. 2007; 137:2437–42. [PubMed]
- 17. Cadario F, Savastio S, Magnani C, Cena T, Pagliardini V, Bellomo G, Bagnati M, Vidali M, Pozzi E, Pamparana S, Zaffaroni M, Genoni G, Bona G. High Prevalence of Vitamin D Deficiency in Native versus Migrant Mothers and Newborns in the North of Italy: A Call to Act with a Stronger Prevention Program. PLoS One. 2015; 10:e0129586. https://doi.org/10.1371/journal.pone.0129586 [PubMed]
- 18. Hewison M. An update on vitamin D and human immunity. Clin Endocrinol (Oxf). 2012; 76:315–25. https://doi.org/10.1111/j.1365-2265.2011.04261.x [PubMed]
- 19. Harvey L, Burne TH, McGrath JJ, Eyles DW. Developmental vitamin D3 deficiency induces alterations in immune organ morphology and function in adult offspring. J Steroid Biochem Mol Biol. 2010; 121:239–42. https://doi.org/10.1016/j.jsbmb.2010.03.050 [PubMed]
- 20. Yu S, Cantorna MT. Epigenetic reduction in invariant NKT cells following in utero vitamin D deficiency in mice. J Immunol. 2011; 186:1384–90. https://doi.org/10.4049/jimmunol.1002545 [PubMed]
- 21. Ramagopalan SV, Heger A, Berlanga AJ, Maugeri NJ, Lincoln MR, Burrell A, Handunnetthi L, Handel AE, Disanto G, Orton SM, Watson CT, Morahan JM, Giovannoni G, et al. A ChIP-seq defined genome-wide map of vitamin D receptor binding: associations with disease and evolution. Genome Res. 2010; 20:1352–60. https://doi.org/10.1101/gr.107920.110 [PubMed]
- 22. Carrillo-Vico A, Lardone PJ, Alvarez-Sánchez N, Rodríguez-Rodríguez A, Guerrero JM. Melatonin: buffering the immune system. Int J Mol Sci. 2013; 14:8638–83. https://doi.org/10.3390/ijms14048638 [PubMed]
- 23. Cui X, McGrath JJ, Burne TH, Mackay-Sim A, Eyles DW. Maternal vitamin D depletion alters neurogenesis in the developing rat brain. Int J Dev Neurosci. 2007; 25:227–32. https://doi.org/10.1016/j.ijdevneu.2007.03.006 [PubMed]
- 24. Chabas JF, Stephan D, Marqueste T, Garcia S, Lavaut MN, Nguyen C, Legre R, Khrestchatisky M, Decherchi P, Feron F. Cholecalciferol (vitamin D3) improves myelination and recovery after nerve injury. PLoS One. 2013; 8:e65034. https://doi.org/10.1371/journal.pone.0065034 [PubMed]
- 25. Graffe A, Milea D, Annweiler C, Beauchet O, Mauget-Faÿsse M, Beauchet O, Kodjikian L, Milea D. Association between hypovitaminosis D and late stages of age-related macular degeneration: a case-control study. J Am Geriatr Soc. 2012; 60:1367–69. https://doi.org/10.1111/j.1532-5415.2012.04015.x [PubMed]
- 26. Itty S, Day S, Lyles KW, Stinnett SS, Vajzovic LM, Mruthyunjaya P. Vitamin D deficiency in neovascular versus nonneovascular age-related macular degeneration. Retina. 2014; 34:1779–86. https://doi.org/10.1097/IAE.0000000000000178 [PubMed]
- 27. Kim EC, Han K, Jee D. Inverse relationship between high blood 25-hydroxyvitamin D and late stage of age-related macular degeneration in a representative Korean population. Invest Ophthalmol Vis Sci. 2014; 55:4823–31. https://doi.org/10.1167/iovs.14-14763 [PubMed]
- 28. Seddon JM, Reynolds R, Shah HR, Rosner B. Smoking, dietary betaine, methionine, and vitamin D in monozygotic twins with discordant macular degeneration: epigenetic implications. Ophthalmology. 2011; 118:1386–94. [PubMed]
- 29. Reibaldi M, Longo A, Pulvirenti A, Avitabile T, Russo A, Cillino S, Mariotti C, Casuccio A. Geo-Epidemiology of Age-Related Macular Degeneration: New Clues Into the Pathogenesis. Am J Ophthalmol. 2016; 161:78–93.e1, 2. https://doi.org/10.1016/j.ajo.2015.09.031 [PubMed]
- 30. Mandel Y, Grotto I, El-Yaniv R, Belkin M, Israeli E, Polat U, Bartov E. Season of birth, natural light, and myopia. Ophthalmology. 2008; 115:686–92. https://doi.org/10.1016/j.ophtha.2007.05.040 [PubMed]
- 31. McMahon G, Zayats T, Chen YP, Prashar A, Williams C, Guggenheim JA. Season of birth, daylight hours at birth, and high myopia. Ophthalmology. 2009; 116:468–73. https://doi.org/10.1016/j.ophtha.2008.10.004 [PubMed]
- 32. Fiddes B, Wason J, Sawcer S. Confounding in association studies: month of birth and multiple sclerosis. J Neurol. 2014; 261:1851–56. https://doi.org/10.1007/s00415-014-7241-y [PubMed]
- 33. Xoxi E, De Nigro L, Tomino C, Pani L. The Italian post-marketing registries. Pharmaceutical Programming. 2012; 5:57–60.
- 34. Montilla S, Xoxi E, Russo P, Cicchetti A, Pani L. Monitoring registries at Italian Medicines Agency: fostering access, guaranteening sustainability. Int J Technol Assess Health Care. 2015; 31:210–13. [PubMed]



