Introduction
Dialysis patients exhibit an increased all-cause and cardiovascular (CV) mortality when compared to the general aging population [1]. In particular, with the increased aging of the population, calcification of vessels and cardiac valves is highly prevalent in maintenance hemodialysis (mHD) patients and has been associated with an increased CV risk as well as with all-cause mortality [2]. In patients with end stage renal disease (ESRD), besides traditional risk factors such as increased age, hypertension (HT), diabetes (DM) and dyslipidemia, non-traditional CV risk factors, such as mineral metabolism abnormalities, extreme PTH serum levels, excessive administration of calcium salts as phosphate binders, chronic inflammation, malnutrition and oxidative stress play an important role in development of CV diseases [3].
In previous studies, male gender, dialysis vintage, smoking, calcium-phosphate product, high serum high-sensitivity C reactive protein, low Kt/Vurea, Diabetes Mellitus and ethnicity were independent risk factors for CV calcifications [4,5]. Type 2 diabetic patients show an increased risk of CV events that is similar to the risk of non-diabetic patients with coronary artery disease [6], regardless of other concomitant CV risk factors [7]. Moreover, both type 1 and type 2 DM are frequently complicated by renal disease [8] and kidney disease in diabetic patients dramatically increases the incidence of CV events [9-12]. Therefore, renal disease must be considered an independent risk factor for CV disease at least as strong as DM. Indeed, regardless of DM, gender and ethnicity, incident HD patients show a CV morbidity/mortality increased by ten to one hundred times compared to the age-matched general population: CV disease-related mortality rate of a 25 to 35 year-old remic patient may be similar to that of people 85 or more years old [13].
The primary aim of this study was to evaluate the long term predictive value for CV events of coronary artery calcifications (CACs) detected by multi-layer spiral computed tomography (MSCT) in non-diabetic Caucasian mHD patients.
The secondary aim of the study was then to evaluate whether conventional “atherogenic indices” were an independent risk factor for CACs. The following “atherogenic indices” were investigated: total cholesterol (TC)/high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol/HDL cholesterol and Triglyceride Logarithm/HDL cholesterol.
Results
Demographic, clinical features and laboratory findings of the study population divided on AS values <400 vs. >400 are summarized in Table 1. We selected 400 AS units as cut-off value according to previous literature [14].
Table 1. Demographic, clinical and laboratory characteristics of study population by agatstone score
| AS ≤ 400 | AS >400 | p (Fisher Test) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Demographics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Patients | 72/205 (35,1%)*** | 133/205 (64,9%)*** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 52,50±12,65* | 63,98±10,82* | 0,126 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 34/72 | 41/133 | 0,119 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 38/72 | 92/133 | 0,236 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Clinical Characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BM | 23,98±4,80* | 24,81±3,77* | 0,039 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 22/72 (30,5%)*** | 55/133 (41,4%)*** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HD vintage (months) | 37 (6-276)** | 48 (6-252)** | 0,579 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Kt/Vurea | 1,31 (1-1,75)** | 1,28 (0,85-1,84)** | 0,957 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Laboratory data | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CPR (mg/l) | 1,12±1,09 | 1,08±1,19 | 0,885 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hemoglobin (g/dl) | 11,47±1,09* | 11,61±1,2* | 0,472 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Calcium (mmol/l) | 9,43±0,77* | 9,47±1,01* | 0,120 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phosphorus (mmol/l) | 6,00±1,19* | 5,62±1,34* | 0,258 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ca × P | 55,41±12,50* | 53,19±14,52* | 0,166 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| iPTH (pg/l) | 383 (61-1498)** | 451 (2,43-2500)** | 0,015 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total cholesterol (mmol/l) | 164,83±45,95* | 157,12±38,42* | 0,080 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL cholesterol (mmol/l) | 48,2±14,53* | 42,90±12,70* | 0,188 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL cholesterol (mmol/l) | 79,50 (38-311,4)** | 93 (40-254,2)** | 0,032 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Triglycerides (mmol/l) | 143 (50-747)** | 171 (53-716)** | 0,796 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Serum Albumin (g/l) | 4,23±0,30* | 4,12±0,39* | 0,043 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: AS, Agatstone Score; HD, hemodialysis; Ca, calcium; P, phosphorus; iPTH, Intact Parathyroid Hormone; LDL, Low-Density Lipoprotein; HDL, High-Density Lipoprotein. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Normally distributed data are expressed as mean ± standard deviation | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non normally distributes data are expressed as median with ranges | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data are expressed as percentage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Among the 205 mHD patients enrolled into the study, 72 (35.1%) showed an AS ≤ 400 units and 133 (64.9%) an AS >400 units. Age, HD-vintage, Kt/Vurea, C-reactive protein (CRP), serum haemoglobin (Hb), glucose, calcium, phosphorus and calcium-phosphorus product, fibrinogen, total cholesterol, HDL cholesterol and serum triglycerides did not differ significantly in patients with AS above or below 400. Statistically significantly higher values were found in patients with AS >400 units for BMI (p=0.039) (values less than 25 in both groups), serum intact parathyroid hormone (iPTH) (p=0.015) and serum LDL cholesterol (p=0.032) (values less than 100mg/dl in both groups), whereas serum albumin was statistically lower (p=0.043) in patients with AS >400 units but still above 4mg/dl.
Drug therapy for dyslipidemia and for the management of calcium/phosphorus metabolism of study population did not differ in patients with AS above or below 400 (NS) (Table 2).
Table 2. Drug therapy of patients
| AS ≤ 400 (n=72) | AS >400 (n=133) | p value | |
|---|---|---|---|
| 1.Phosphate binders: | |||
| a-calcium carbonate | 19,44% (n=14) | 20,30% (n=27) | 0,495 |
| b-sevelamer | 65,28% (n=47) | 67,67% (n=90) | 0,225 |
| 2.Vitamin D analogues: | 80,55% (n=58) | 81,95% (n=109) | 0,531 |
| 3.Statins: | 58,33% (n=42) | 56,31% (n=75) | 0,589 |
The “atherogenic indices” or lipoprotein ratio stratified by AS values, in male and female patients, are described in Tables 3 A and B, respectively. In male patients, no statistically significant differences were observed for TC/HDL-cholesterol ratio, LDL/HDL cholesterol ratio and Logarithmic transformation of the triglyceride/HDL cholesterol molar concentration ratio in patients with AS above or below 400. Moreover, all parameters were in the normal range. In female patients, TC/HDL-cholesterol ratio did not differ in patients with AS above or below 400. Although a statistical difference was observed for both LDL/HDL cholesterol ratio and Logarithmic transformation of the triglyceride/HDL cholesterol molar concentration ratio in patients with AS above or below 400, these values were in the normal range.
Table 3B. Atherogenic indices values by Agatstone score in female
| AS≤400 | AS>400 | p value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TC/HDL-C | 3,29±1,09a | 3,79±1,33a | 0,09 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL-C/HDL-C | 1,83±0,79a | 2,52±1,27a | 0,0074 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Log Trigl/HDL col | 0,04±0,01a | 0,05±0,16a | 0,009* | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data are expressed as mean ± standard deviation. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| P<0,05 is considered statistical significant. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| We applied Mann/Whitney Test | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviation: HDL-C, High-Density Lipoprotein Cholesterol; LDL-C, Low-Density Lipoprotein Cholesterol; TC, Total Cholesterol; Log Trigl, Triglyceride Logarithm. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No correlation was found between serum iPTH levels and AS value (r =-0.046, p=0.64). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Survival analysis
One-hundred two patients (49.7%) had a CV event during the seven year follow-up period. Kaplan-Meier survival analysis is shown in Figure 1. Seven-year actuarial survival exceeded 90% among patients of groups 1 (AS=0) and 2 (AS≤400), but fell to about 50% in patients of group 3 (400
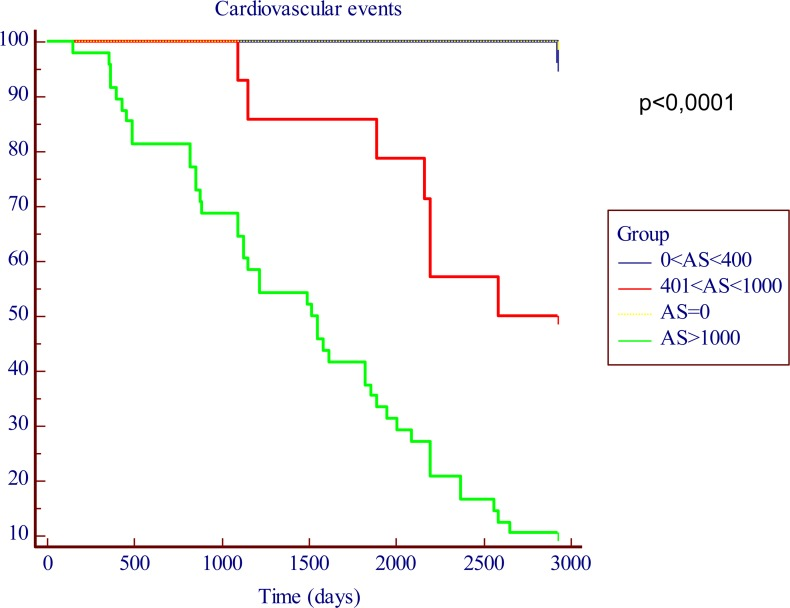
Figure 1. Kaplan-Meier in function of the AS cut-off levels. The time is expressed as days.
Seven-year actuarial survival exceeded 90% among patients of groups 1 (AS=0) and 2 (AS≤400), but fell to about 50% in patients of group 3 (400
We found a predictive role, both for an higher AS and an higher CV mortality, linked to the serum iPTH levels; in fact, as shown in Table 1, the iPTH values were significantly higher in pts with an AS > 400 (451 pg/L, [range 2.43 to 2500 pg/L] vs 383 pg/L [range 61 to 1498 pg/L], p < 0.05); furthermore the Kaplan-Meier analysis showed a significantly decreased seven-year actuarial survival (p < 0.05) in patients with iPTH levels > 300 pg/L compared to patients with iPTH levels < 300 pg/L (Figure 2).
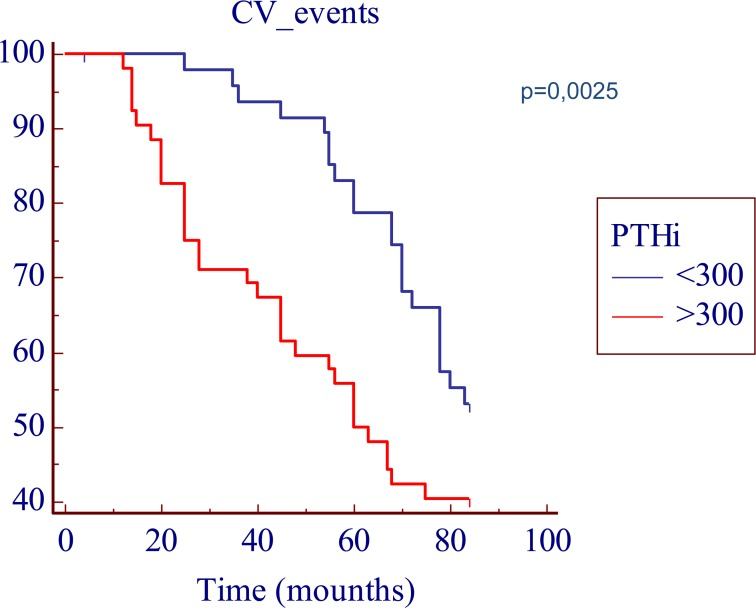
Figure 2. Kaplan-Meier survival plot in function of the iPTH cut-off levels. The time is expressed as months.
Nine out of the 205 patients enrolled into the study were censored: four died for non-CV causes and 5 underwent renal transplantation.
Discussion
With the changing medical treatments and with the changes in the living environment, the age of the population is increasing in all countries, posing novel challenges to the medical and social areas [15-27]. While the changes related to increased age implies significant problems for the society, relevant modifications needs to be clarified for the physical, biological and mental health. This has fostered important research in distinct medical aspects, from the major role played by the telomers [28,29] to the study in different animal models, from bacteria, C. elegans to drosophila or mice where the cell autonomy and the underlying molecular pathways have been deeply investigated [30-39]. Recently it has become apparent that the complex genetic and epigenetic gene regulation in aging is also affected by a large array of microRNA [40-48], that contribute to the cellular signalling [49-58], which in turn regulate several cellular function, such as for example metabolism [59-64], cell division [65-66], or DNA damage [67-70]. A significant expansion of the latter is the involvement of p53 [71-76] and its family members p63 [77-79] and p73 [80-83] with their metabolic regulation. All these biochemical alterations, profoundly affect different organs, such as for example the neural [84-95] and the cardiovascular [96,97] systems. Accordingly, in the present manuscript we investigated a specific aspect of vascular biology.
Calcification of vessels and cardiac valves is highly prevalent in maintenance hemodialysis (mHD) patients and has been associated with an increased CV risk as well as with all-cause mortality [2].
Vascular calcifications can affect the media or intima. Medial artery calcifications are frequently found in DM and uremia [98]. Vascular calcifications are associated with adverse clinical outcomes [99]. Hemodynamic disturbances related to vascular calcifications include loss of arterial elasticity, an increased arterial stiffness and pulse wave velocity, with subsequent development of myocardial ischemia and heart failure [100].
Pathogenesis of vascular calcifications is complex. It does not only consist in a simple precipitation of calcium and phosphate, but is instead an active process in which vascular smooth muscle cells (VSMCs), undergo apoptosis and vesicles formation, are transformed into osteoblast-like cells, inducing both matrix formation and attracting local factors involved in the mineralization process [100].
Jono et al. suggested that elevated intracellular phosphate may directly stimulate VSMCs to transform into calcifying cells by activating genes associated with osteoblastic function [101]. Hyperfosfatemia is responsible for the enhanced number and activity of osteoclasts, being a major contributor to increased bone resorption in chronic kidney disease [102].
Cardiac calcifications predict CV events and all-cause mortality in non diabetic and add incremental predictive value to conventional CV risk factors [103].
In the present prospective, cohort and long-term study we assessed CACS by using a semi-quantitative calcification score. In patients with CACS less than 400 AS units seven-year actuarial survival was very high (more than 90%) compared to patients with AS> 400. A striking increase in long term CV morbidity/mortality associated with the increase of baseline CACS was observed. Seven-year actuarial survival dropped to less than 10% in patients with extensive calcifications (AS>1000).
We found a significant predictive role, both for an higher AS and for the serum iPTH levels; in fact, the seven year actuarial survival was lower both in patients with an AS > 400 and in patients with iPTH levels > 300.
Other studies have addressed this issue using less invasive methodology to assess CV calcifications in both diabetic and non-diabetic mHD population [104]; in contrast with the results of previous studies, age, male gender, dialysis vintage, calcium-phosphate product, CRP serum levels, serum albumin, Kt/Vurea, were not independent risk factors for CACS in our non-diabetic study population, suggesting that the more accurate, semi-quantitative method used for measurement, the anatomic location and the exclusion of diabetic patients probably avoided confounding factors. Interestingly, “atherogenic indices” were not independent risk factors for CACS and LDL cholesterol was at target levels regardless to the extent of cardiac calcifications suggesting other factors to be important for CACS formation.
Our findings corroborate the well-known poor impact of traditional cardiovascular risk factor in non diabetic hemodialysis patients, and support that usual risk factors, as well as age, blood pressure and lipid levels are working with other factors, in particular iPTH levels, to contribute to the development and progression of vascular calcifications. It is worthy to note that higher serum iPTH levels are strongly associated with the presence of cardiac calcification, because a deregulated calcium-phosphorus is as important long term CV risk factor in non diabetic mHD patients and stress the importance of optimal calcium-phosphorus metabolism control in order to prevent or reduce the occurrence of CV events [105].
Although accurate MSCT is more invasive and the ionizing radiation dose is higher compared to roentgenography, it is likely that patients with higher iPTH and phosphorus levels could be suitable for screening and widespread clinical use in asymptomatic patients, may although provide additional information in situations in which a more accurate assessment of CV risk would be necessary.
Conclusions
Following our previous interest on aging [106-108], here we show that extensive coronary artery calcifications strongly predict long term CV morbidity and mortality in non diabetic Caucasian mHD patients independently of atherogenic indices. A full cost benefit analysis would be needed before more widespread screening of MSCT could be advocated in clinical practice, employing a CV risk stratification plan that includes iPTH with other usual stronger atherogenic indices, linked to cholesterol and triglycerides ratios.
Materials and Methods
Study design and patients
This prospective and cohort study was designed specifically to evaluate CACS as a possible predictor of CV events in mHD patients with no evidence of cardiovascular disease. Patients recruited from five dialysis facilities, had coronary artery calcifications measured by multi-layer spiral computed tomography (MSCT). All of them were performed at our university centre. Clinical data, features of dialytic treatment and information on outcome were provided for each patient at regular six-month interval. Recruitment started in January 2003. Participants' clinical status was annually reviewed annually up to January 2011.
The study protocol complied with the declaration of Helsinki and was appointed by our local ethical committee and by ethical committee of each of the participating institutions. A written fully informed consent was provided by all patients before entering the study.
Men and women aged 18 years or more, on mHD since 6 months at least were considered eligible for the study.
Exclusion criteria were: diabetes, pregnancy, known coronary artery disease, congestive heart failure, uncontrolled HT, cerebro-vascular ischemic events in the last six months, cardiac arrhythmias (that would impede assessment of CACS with computer tomography), neoplastic disease, inability to provide informed consent or other medical conditions likely to limit life expectancy or requiring extensive medical treatment.
Two-hundred five (105 male and 100 female) Caucasian mHD patients were enrolled into the study. At enrolment time each patients performed the coronary MSCT. Mean age was 59.85±12.77 years, HD vintage was 62.30±55 months.
All patients received standard bicarbonate dialysis with1.5–2.0m2 hollow-fiber low-flux polysulphone membranes (Lo-PS Diacap Polysulphone, B. Braun gmbh, Melsungen, Germany), four hours three times weekly, through a well-functioning native A-V fistula or a cuffed internal jugular indwelling venous catheter. The vascular access performance was satisfactory with a blood flow of at least 300ml/min and a Kt/V urea ratio>1.3 [109].
Body mass index (BMI) was calculated upon the post-dialysis body weight (“dry weight”) at enrollment by dividing weight in kilograms by the square of height in meters for every patient.
Laboratory measurements
After an overnight fast blood samples were taken for baseline measurements of serum glucose, serum Hb, total and HDL cholesterol, (LDL cholesterol was calculated by Friedewald method), triglycerides, serum Calcium and Phosphorus, iPTH, fibrinogen and CRP. Blood samples were drawn in a midweek non-dialysis day.
Atherogenic indices were calculated as follows: total cholesterol (mg/dl) /HDL cholesterol (mg/dl) (normal value <5 for male and <4.5 for female), LDL cholesterol (mg/dl)/HDL cholesterol (mg/dl) (normal value<3.5 for male and <3 for female). An atherogenic plasma index [log (triglycerides mg/dl/HDL cholesterol mg/dl)], >0.5 has been proposed as cut-off point indicating atherogenic risk. Risk categories and target levels for total cholesterol/HDL-cholesterol, LDL-cholesterol/HDL-cholesterol in primary and secondary prevention, stratified by gender, were considered [110].
Cardiac calcifications
Quantification of CACS was obtained with a coronary MSCT. The measurement of CACS was expressed in Agatston score (AS) units [111].
MSCT was performed with a 64-channel multidetector scanner (LightSpeed VCT, General Electric Medical Systems, EU) and retrospective synchronization technique. Images were acquired without enhancement with the patient in the supine position. All scans were performed with the following parameters: detector collimation 4 × 2.5mm, reconstruction interval 10mm, gantry rotation time 0.5 sec, tube voltage 120Kv, intensity 300mA, field of view 25cm, cranio-caudal scan direction.
Calcium was scored according to the Agatston method to quantify the amount of calcification in the coronary arteries.
Coronary calcification was defined as the presence of four or more contiguous pixels with more than 130 Hounsfield units (HU). The investigator scored each of the 20 slices individually, using GE Medical Systems-Advantage Workstation software. Thereby, each plaque score was generated as the product of the area and density. This method has previously been described in detail by Agatston et al. [112,113]. According to the AS patients were then divided in four groups: group 1(AS=0), group 2 (0
Clinical endpoints
Primary clinical endpoint was a fatal major CV event: death due to myocardial infarction (MI), heart failure or other CV causes (aortic aneurysm rupture, stroke, i.e.) during the 7 year follow-up period following baseline evaluation. Clinical outcome was assessed by direct interview contact to the patients and inspection of medical or other records.
Statistical analysis
Results related to biochemical and radiological findings are expressed as the mean ± standard deviation (SD) of three independent determinations. Two-tailed independent-sample T test and two sided Fisher's exact test were employed for analysis of results; p-values <0.05 were considered statistically significant.
Seven-year actuarial survival was calculated in the four groups separately by Kaplan-Meier method. The “log rank” test was employed to compare survival curves. Patients who died for causes other than CV diseases and transplanted patients were censored. To study the linear relationship between i-PTH levels and AS values, non parametric correlation (Spearman p) was used. Data were elaborated through the MedCalc Statistical Software (MedCalc Software, 9030 Mariakerke, Belgium).
Acknowledgments
We wish to thank the study group: Nephrology and dialysis Unit, Aurelia Hospital, Rome: Vega Agostino, Paolo Barone; Nephrology and dialysis Unit, G.B.Grassi Hospital, Ostia: Massimo Morosetti; Nephrology and dialysis Unit, San Giovanni dell'Addolorata Hospital, Roma: Alessandro Balducci, Micaela Manni; Nephrology and Hypertension Unit, Ospedale Israelitico, Roma: Giorgio Coen; Lazio Regional Agency for Transplantations and Related Pathologies.
Conflicts of Interest
The authors have no conflict of interests to declare.
References
- 1. Schlieper G, Brandenburg V, Djuric Z, Damjanovic T, Markovic N, Schurgers L, Kruger T, Westenfeld R, Ackermann D, Haselhuhn A, Dimkovic S, Ketteler M, Floege J, et al. Risk factors for cardiovascular calcifications in non diabetic Caucasian haemo-dialysis patients. Kidney Blood Press Res. 2009; 32: 161 -168. [PubMed] .
- 2. Bellasi A, Ferramosca E, Muntner P, Ratti C, Wildman RP, Block GA, Raggi P. Correlation of simple imaging tests and coronary artery calcium measured by computed tomography in hemodialysis patients. Kidney Intern. 2006; 70: 1623 -1628. .
- 3. Moe SM and Chen NX. Mechanisms of vascular calcification in chronic kidney disease. J Am Soc Nephrol. 2008; 19: 213 -216. [PubMed] .
- 4. Kendrik J and Chonchol M. The role of phosphorus in the development and progression of vascular calcifications. Am, J. Kidney Dis. 2011; 58: 826 -834. [PubMed] .
- 5. Bild DE, Detrano R, Peterson D, Guerci A, Liu K, Shahar E, Ouyang P, Jackson S, Saad MF. Ethnic differences in coronary calcification: the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation. 2005; 111: 1313 -1320. [PubMed] .
- 6. Nathan DM, Meigs J, Singer DE. The epidemiology of cardiovascular disease in type 2 diabetes mellitus: how sweet it is … or is it? Lancet. 1997; 350: 4 -9. [PubMed] .
- 7. Juutilainen A, Lehto S, Rönnemaa T, Pyörälä K, Laakso M. Type 2 diabetes as a “coronary heart disease equivalent”: an 18-year prospective population-based study in Finnish subjects. Diabetes Care. 2005; 28: 2901 -2907. [PubMed] .
- 8. Molitch ME, DeFronzo RA, Franz MJ, Keane WF, Mogensen CE, Parving HH, Steffes MW. American Diabetes Association. Nephropathy in diabetes. Diabetes Care. 2004; 27: S79 -S83. [PubMed] .
- 9. Soedamah-Muthu SS, Chaturvedi N, Toeller M Ferriss B, Reboldi P, Michel G, Manes C, Fuller JH, EURODIAB Prospective Complications Study Group. Risk factors for coronary heart disease in type 1 diabetic patients in Europe: the EURODIAB Prospective Complications Study. Diabetes Care. 2004; 27: 530 -537. [PubMed] .
- 10. Sowers JR and Lester MA. Diabetes and cardiovascular disease. Diabetes Care. 1999; 22: 14 -20. .
- 11. Dorman JS, LaPorte RE, Kuller LH, Cruickshanks KJ, Orchard TJ, Wagener DK, Becker DJ, Cavender DE, Drash AL. The Pittsburgh insulin-dependent diabetes mellitus (IDDM) morbidity and mortality study: mortality results. Diabetes. 1984; 33: 271 -276. [PubMed] .
- 12. Orchard TJ, Costacou T, Kretowski A, Nesto RW. Type 1 diabetes and coronary artery disease. Diabetes Care. 2006; 29: 2528 -2538. [PubMed] .
- 13. Foley RN, Parfrey PS, Sarnak MJ. Clinical epidemiology of cardiovascular disease in chronic renal disease. Am J Kidney Dis. 1998; 32: S112 -S119. [PubMed] .
- 14. Elkeles R. Computer tomography imaging, coronary calcium and atherosclerosis. Expert Rev Cardiovascular Ther. 2008; 8: 1083 -1093. .
- 15. Blagosklonny MV. Koschei the immortal and anti-aging drugs. Cell Death Dis. 2014; 4: e1552 [PubMed] .
- 16. Blagosklonny MV. Geroconversion: irreversible step to cellular senescence. Cell Cycle. 2014; 13: 3628 -3635. [PubMed] .
- 17. Harper S. Economic and social implications of aging societies. Science. 2014; 346: 587 -591. [PubMed] .
- 18. Fontana L, Kennedy BK, Longo VD, Seals D, Melov S. Medical research: treat ageing. Nature. 2014; 511: 405 -407. [PubMed] .
- 19. Stewart L. Gerontology: Will you still need me, will you still feed me? Nature. 2014; 514: S14 -S155. [PubMed] .
- 20. Scudellari M. Ageing research: Blood to blood. Nature. 2015; 517: 426 -429. [PubMed] .
- 21. Guarente L. Aging research-where do we stand and where are we going? Cell. 2014; 159: 15 -19. [PubMed] .
- 22. Berman AE, Leontieva OV, Natarajan V, McCubrey JA, Demidenko ZN, Nikiforov MA. Recent progress in genetics of aging, senescence and longevity: focusing on cancer-related genes. Oncotarget. 2012; 3: 1522 -1532. [PubMed] .
- 23. de Souto Barreto P. Ageing: Research needs social science. Nature. 2014; 512: 253 [PubMed] .
- 24. Purcell M, Kruger A, Tainsky MA. Gene expression profiling of replicative and induced senescence. Cell Cycle. 2014; 13: 3927 -3937. [PubMed] .
- 25. Moskalev AA, Aliper AM, Smit-McBride Z, Buzdin A, Zhavoronkov A. Genetics and epigenetics of aging and longevity. Cell Cycle. 2014; 13: 1063 -1077. [PubMed] .
- 26. van Deursen JM. The role of senescent cells in ageing. Nature. 2014; 509: 439 -446. [PubMed] .
- 27. Gutchess A. Plasticity of the aging brain: new directions in cognitive neuroscience. Science. 2014; 346: 579 -582. [PubMed] .
- 28. Gao B, Li K, Wei YY, Zhang J, Li J, Zhang L, Gao JP, Li YY, Huang LG, Lin P, Wei YQ. Zinc finger protein 637 protects cells against oxidative stress-induced premature senescence by mTERT-mediated telomerase activity and telomere maintenance. Cell Death Dis. 2014; 5: e1334 [PubMed] .
- 29. Freund A, Zhong FL, Venteicher AS, Meng Z, Veenstra TD, Frydman J, Artandi SE. Proteostatic control of telomerase function through TRiC-mediated folding of TCAB1. Cell. 2014; 159: 1389 -1403. [PubMed] .
- 30. Merino MM, Rhiner C, Lopez-Gay JM, Buechel D, Hauert B, Moreno E. Elimination of unfit cells maintains tissue health and prolongs lifespan. Cell. 2015; 160: 461 -476. [PubMed] .
- 31. Gatta V, D'Aurora M, Granzotto A, Stuppia L, Sensi SL. Early and sustained altered expression of aging-related genes in young 3xTg-AD mice. Cell Death Dis. 2014; 5: e1054 [PubMed] .
- 32. Saint-Léger A, Koelblen M, Civitelli L, Bah A, Djerbi N, Giraud-Panis MJ, Londoño-Vallejo A, Ascenzioni F, Gilson E. The basic N-terminal domain of TRF2 limits recombination endonuclease action at human telomeres. Cell Cycle. 2014; 13: 2469 -2474. [PubMed] .
- 33. Heintz C and Mair W. You are what you host: microbiome modulation of the aging process. Cell. 2014; 156: 408 -11. [PubMed] .
- 34. Finch CE and Tower J. Sex-specific aging in flies, worms, and missing great-granddads. Cell. 2014; 156: 398 -399. [PubMed] .
- 35. Bansal A, Zhu LJ, Yen K, Tissenbaum HA. Uncoupling lifespan and healthspan in Caenorhabditis elegans longevity mutants. Proc Natl Acad Sci U S A. 2015; 112: E277 -286. [PubMed] .
- 36. Rozhok AI, Salstrom JL, DeGregori J. Stochastic modeling indicates that aging and somatic evolution in the hematopoetic system are driven by non-cell-autonomous processes. Aging (Albany NY). 2014; 6: 1033 -1048. [PubMed] .
- 37. Arlia-Ciommo A, Piano A, Leonov A, Svistkova V, Titorenko VI. Quasi-programmed aging of budding yeast: a trade-off between programmed processes of cell proliferation, differentiation, stress response, survival and death defines yeast lifespan. Cell Cycle. 2014; 13: 3336 -3349. [PubMed] .
- 38. Geißler S, Textor M, Schmidt-Bleek K, Klein O, Thiele M, Ellinghaus A, Jacobi D, Ode A, Perka C, Dienelt A, Klose J, Kasper G, Duda GN, Strube P. In serum veritas-in serum sanitas? Cell non-autonomous aging compromises differentiation and survival of mesenchymal stromal cells via the oxidative stress pathway. Cell Death Dis. 2013; 4: e970 [PubMed] .
- 39. Fulle S, Sancilio S, Mancinelli R, Gatta V, Di Pietro R. Dual role of the caspase enzymes in satellite cells from aged and young subjects. Cell Death Dis. 2013; 4: e955 [PubMed] .
- 40. Bonafè M and Olivieri F. Circulating microRNAs in aging. Oncotarget. 2015; 6: 1340 -1341. [PubMed] .
- 41. Grammatikakis I, Panda AC, Abdelmohsen K, Gorospe M. Long noncoding RNAs(lncRNAs) and the molecular hallmarks of aging. Aging (Albany NY). 2014; 6: 992 -1009. [PubMed] .
- 42. Rivetti di Val Cervo P, Lena AM, Nicoloso M, Rossi S, Mancini M, Zhou H, Saintigny G, Dellambra E, Odorisio T, Mahé C, Calin GA, Candi E, Melino G. p63-microRNA feedback in keratinocyte senescence. Proc Natl Acad Sci U S A. 2012; 109: 1133 -1138. [PubMed] .
- 43. Zhou Y, Hu Y, Yang M, Jat P, Li K, Lombardo Y, Xiong D, Coombes RC, Raguz S, Yagüe E. The miR-106b~25 cluster promotes bypass of doxorubicin-induced senescence and increase in motility and invasion by targeting the E-cadherin transcriptional activator EP300. Cell Death Differ. 2014; 21: 462 -474. [PubMed] .
- 44. Mogilyansky E and Rigoutsos I. The miR-17/92 cluster: a comprehensive update on its genomics, genetics, functions and increasingly important and numerous roles in health and disease. Cell Death Differ. 2013; 20: 1603 -1614. [PubMed] .
- 45. Grigoriev A and Bonini NM. Age-dependent patterns of microRNA RISC loading. Aging (Albany NY). 2014; 6: 705 -706. [PubMed] .
- 46. Lena AM, Mancini M, Rivetti di Val Cervo P, Saintigny G, Mahé C, Melino G, Candi E. MicroRNA-191 triggers keratinocytes senescence by SATB1 and CDK6 downregulation. Biochem Biophys Res Commun. 2012; 423: 509 -514. [PubMed] .
- 47. Vasa-Nicotera M, Chen H, Tucci P, Yang AL, Saintigny G, Menghini R, Mahè C, Agostini M, Knight RA, Melino G, Federici M. miR-146a is modulated in human endothelial cell with aging. Atherosclerosis. 2011; 217: 326 -330. [PubMed] .
- 48. Menghini R, Casagrande V, Cardellini M, Martelli E, Terrinoni A, Amati F, Vasa-Nicotera M, Ippoliti A, Novelli G, Melino G, Lauro R, Federici M. MicroRNA 217 modulates endothelial cell senescence via silent information regulator 1. Circulation. 2009; 120: 1524 -1532. [PubMed] .
- 49. Ramachandran I, Ganapathy V, Gillies E, Fonseca I, Sureban SM, Houchen CW, Reis A, Queimado L. Wnt inhibitory factor 1 suppresses cancer stemness and induces cellular senescence. Cell Death Dis. 2014; 5: e1246 [PubMed] .
- 50. Macià A, Vaquero M, Gou-Fàbregas M, Castelblanco E, Valdivielso JM, Anerillas C, Mauricio D, Matias-Guiu X, Ribera J, Encinas M. Sprouty1 induces a senescence-associated secretory phenotype by regulating NFκB activity: implications for tumorigenesis. Cell Death Differ. 2014; 21: 333 -343. [PubMed] .
- 51. Coll NS, Smidler A, Puigvert M, Popa C, Valls M, Dangl JL. The plant metacaspase AtMC1 in pathogen-triggered programmed cell death and aging: functional linkage with autophagy. Cell Death Differ. 2014; 21: 1399 -1408. [PubMed] .
- 52. Burnley P, Rahman M, Wang H, Zhang Z, Sun X, Zhuge Q, Su DM. Role of the p63-FoxN1 regulatory axis in thymic epithelial cell homeostasis during aging. Cell Death Dis. 2013; 4: e932 [PubMed] .
- 53. He YH, Lu X, Yang LQ, Xu LY, Kong QP. Association of the insulin-like growth factor binding protein 3 (IGFBP-3) polymorphism with longevity in Chinese nonagenarians and centenarians. Aging (Albany NY). 2014; 6: 944 -956. [PubMed] .
- 54. Bernal GM, Wahlstrom JS, Crawley CD, Cahill KE, Pytel P, Liang H, Kang S, Weichselbaum RR, Yamini B. Loss of Nfkb1 leads to early onset aging. Aging (Albany NY). 2014; 6: 931 -943. [PubMed] .
- 55. Mohamed JS, Wilson JC, Myers MJ, Sisson KJ, Alway SE. Dysregulation of SIRT-1 in aging mice increases skeletal muscle fatigue by a PARP-1-dependent mechanism. Aging (Albany NY). 2014; 6: 820 -834. [PubMed] .
- 56. Stepanova M, Rodriguez E, Birerdinc A, Baranova A. Age-independent rise of inflammatory scores may contribute to accelerated aging in multi-morbidity. Oncotarget. 2015; 6: 1414 -1421. [PubMed] .
- 57. Ohanna M, Bonet C, Bille K, Allegra M, Davidson I, Bahadoran P, Lacour JP, Ballotti R, Bertolotto C. SIRT1 promotes proliferation and inhibits the senescence-like phenotype in human melanoma cells. Oncotarget. 2014; 5: 2085 -2095. [PubMed] .
- 58. Ruan L, Zhang Z, Mu L, Burnley P, Wang L, Coder B, Zhuge Q, Su DM. Biological significance of FoxN1 gain-of-function mutations during T and B lymphopoiesis in juvenile mice. Cell Death Dis. 2014; 5: e1457 [PubMed] .
- 59. Chin RM, Fu X, Pai MY, Vergnes L, Hwang H, Deng G, Diep S, Lomenick B, Meli VS, Monsalve GC, Hu E, Whelan SA, Wang JX, Jung G, Solis GM, et al. The metabolite α-ketoglutarate extends lifespan by inhibiting ATP synthase and TOR. Nature. 2014; 510: 397 -401. [PubMed] .
- 60. Rufini A, Niklison-Chirou MV, Inoue S, Tomasini R, Harris IS, Marino A, Federici M, Dinsdale D, Knight RA, Melino G, Mak TW. TAp73 depletion accelerates aging through metabolic dysregulation. Genes Dev. 2012; 26: 2009 -2014. [PubMed] .
- 61. Rotblat B, Southwell AL, Ehrnhoefer DE, Skotte NH, Metzler M, Franciosi S, Leprivier G, Somasekharan SP, Barokas A, Deng Y, Tang T, Mathers J, Cetinbas N, Daugaard M, Kwok B, et al. HACE1 reduces oxidative stress and mutant Huntingtin toxicity by promoting the NRF2 response. Proc Natl Acad Sci U S A. 2014; 111: 3032 -3037. [PubMed] .
- 62. Gitenay D, Wiel C, Lallet-Daher H, Vindrieux D, Aubert S, Payen L, Simonnet H, Bernard D. Glucose metabolism and hexosamine pathway regulate oncogene-induced senescence. Cell Death Dis. 2014; 5: e1089 [PubMed] .
- 63. Klang IM, Schilling B, Sorensen DJ, Sahu AK, Kapahi P, Andersen JK, Swoboda P, Killilea DW, Gibson BW, Lithgow GJ. Iron promotes protein insolubility and aging in C. elegans. Aging (Albany NY). 2014; 6: 975 -991. [PubMed] .
- 64. Anisimov VN, Popovich IG, Zabezhinski MA, Egormin PA, Yurova MN, Semenchenko AV, Tyndyk ML, Panchenko AV, Trashkov AP, Vasiliev AG, Khaitsev NV. Sex differences in aging, life span and spontaneous tumorigenesis in 129/Sv mice neonatally exposed to metformin. Cell Cycle. 2015; 14: 46 -55. [PubMed] .
- 65. Yun Y, Holt JE, Lane SI, McLaughlin EA, Merriman JA, Jones KT. Reduced ability to recover from spindle disruption and loss of kinetochore spindle assembly checkpoint proteins in oocytes from aged mice. Cell Cycle. 2014; 13: 1938 -1947. [PubMed] .
- 66. Shomper M, Lappa C, FitzHarris G. Kinetochore microtubule establishment is defective in oocytes from aged mice. Cell Cycle. 2014; 13: 1171 -1179. [PubMed] .
- 67. Lans H, Lindvall JM, Thijssen K, Karambelas AE, Cupac D, Fensgård O, Jansen G, Hoeijmakers JH, Nilsen H, Vermeulen W. DNA damage leads to progressive replicative decline but extends the life span of long-lived mutant animals. Cell Death Differ. 2013; 20: 1709 -1718. [PubMed] .
- 68. Fausti F, Di Agostino S, Cioce M, Bielli P, Sette C, Pandolfi PP, Oren M, Sudol M, Strano S, Blandino G. ATM kinase enables the functional axis of YAP, PML and p53 to ameliorate loss of Werner protein-mediated oncogenic senescence. Cell Death Differ. 2013; 20: 1498 -1509. [PubMed] .
- 69. Danilov A, Shaposhnikov M, Plyusnina E, Kogan V, Fedichev P, Moskalev A. Selective anticancer agents suppress aging in Drosophila. Oncotarget. 2013; 4: 1507 -1526. [PubMed] .
- 70. Oh JH, Gertych A, Tajbakhsh J. Nuclear DNA methylation and chromatin condensation phenotypes are distinct between normally proliferating/aging, rapidly growing/immortal, and senescent cells. Oncotarget. 2013; 4: 474 -493. [PubMed] .
- 71. Müller M, Schleithoff ES, Stremmel W, Melino G, Krammer PH, Schilling T. One, two, three--p53, p63, p73 and chemosensitivity. Drug Resist Updat. 2006; 9: 288 -306. [PubMed] .
- 72. Rufini A, Tucci P, Celardo I, Melino G. Senescence and aging: the critical roles of p53. Oncogene. 2013; 32: 5129 -5143. [PubMed] .
- 73. Levine AJ, Tomasini R, McKeon FD, Mak TW, Melino G. The p53 family: guardians of maternal reproduction. Nat Rev Mol Cell Biol. 2011; 12: 259 -265. [PubMed] .
- 74. Kim J, Nakasaki M, Todorova D, Lake B, Yuan CY, Jamora C, Xu Y. p53 induces skin aging by depleting Blimp1+ sebaceous gland cells. Cell Death Dis. 2014; 5: e1141 [PubMed] .
- 75. Tomasini R, Mak TW, Melino G. The impact of p53 and p73 on aneuploidy and cancer. Trends Cell Biol. 2008; 18: 244 -252. [PubMed] .
- 76. Zhang J, Xu E, Ren C, Yan W, Zhang M, Chen M, Cardiff RD, Imai DM, Wisner E, Chen X. Mice deficient in Rbm38, a target of the p53 family, are susceptible to accelerated aging and spontaneous tumors. Proc Natl Acad Sci U S A. 2014; 111: 18637 -18642. [PubMed] .
- 77. Candi E, Rufini A, Terrinoni A, Giamboi-Miraglia A, Lena AM, Mantovani R, Knight R, Melino G. DeltaNp63 regulates thymic development through enhanced expression of FgfR2 and Jag2. Proc Natl Acad Sci U S A. 2007; 104: 11999 -2004. [PubMed] .
- 78. Tucci P, Agostini M, Grespi F, Markert EK, Terrinoni A, Vousden KH, Muller PA, Dötsch V, Kehrloesser S, Sayan BS, Giaccone G, Lowe SW, Takahashi N, Vandenabeele P, Knight RA, Levine AJ, Melino G. Loss of p63 and its microRNA-205 target results in enhanced cell migration and metastasis in prostate cancer. Proc Natl Acad Sci U S A. 2012; 109: 15312 -15317. [PubMed] .
- 79. Melino G, Gallagher E, Aqeilan RI, Knight R, Peschiaroli A, Rossi M, Scialpi F, Malatesta M, Zocchi L, Browne G, Ciechanover A, Bernassola F. Itch: a HECT-type E3 ligase regulating immunity, skin and cancer. Cell Death Differ. 2008; 15: 1103 -1112. [PubMed] .
- 80. Agostini M, Niklison-Chirou MV, Catani MV, Knight RA, Melino G, Rufini A. TAp73 promotes anti-senescence-anabolism not proliferation. Aging (Albany NY). 2014; 6: 921 -930. [PubMed] .
- 81. He Z, Liu H, Agostini M, Yousefi S, Perren A, Tschan MP, Mak TW, Melino G, Simon HU. p73 regulates autophagy and hepatocellular lipid metabolism through a transcriptional activation of the ATG5 gene. Cell Death Differ. 2013; 20: 1415 -1424. [PubMed] .
- 82. Amelio I, Inoue S, Markert EK, Levine AJ, Knight RA, Mak TW, Melino G. TAp73 opposes tumor angiogenesis by promoting hypoxia-inducible factor 1α degradation. Proc Natl Acad Sci U S A. 2015; 112: 226 -231. [PubMed] .
- 83. Inoue S, Tomasini R, Rufini A, Elia AJ, Agostini M, Amelio I, Cescon D, Dinsdale D, Zhou L, Harris IS, Lac S, Silvester J, Li WY, Sasaki M, Haight J, Brüstle A, Wakeham A, McKerlie C, Jurisicova A, Melino G, Mak TW. TAp73 is required for spermatogenesis and the maintenance of male fertility. Proc Natl Acad Sci U S A. 2014; 111: 1843 -1848. [PubMed] .
- 84. Baruch K, Deczkowska A, David E, Castellano JM, Miller O, Kertser A, Berkutzki T, Barnett-Itzhaki Z, Bezalel D, Wyss-Coray T, Amit I, Schwartz M. Aging-induced type I interferon response at the choroid plexus negatively affects brain function. Science. 2014; 346: 89 -93. [PubMed] .
- 85. Katsimpardi L, Litterman NK, Schein PA, Miller CM, Loffredo FS, Wojtkiewicz GR, Chen JW, Lee RT, Wagers AJ, Rubin LL. Vascular and neurogenic rejuvenation of the aging mouse brain by young systemic factors. Science. 2014; 344: 630 -634. [PubMed] .
- 86. Dong CM, Wang XL, Wang GM, Zhang WJ, Zhu L, Gao S, Yang DJ, Qin Y, Liang QJ, Chen YL, Deng HT, Ning K, Liang AB, Gao ZL, Xu J. A stress-induced cellular aging model with postnatal neural stem cells. Cell Death Dis. 2014; 5: e1116 [PubMed] .
- 87. Kook SY, Jeong H, Kang MJ, Park R, Shin HJ, Han SH, Son SM, Song H, Baik SH, Moon M, Yi EC, Hwang D, Mook-Jung I. Crucial role of calbindin-D28k in the pathogenesis of Alzheimer's disease mouse model. Cell Death Differ. 2014; 21: 1575 -1587. [PubMed] .
- 88. Banday AR, Baumgartner M, Al Seesi S, Karunakaran DK, Venkatesh A, Congdon S, Lemoine C, Kilcollins AM, Mandoiu I, Punzo C, Kanadia RN. Replication-dependent histone genes are actively transcribed in differentiating and aging retinal neurons. Cell Cycle. 2014; 13: 2526 -2541. [PubMed] .
- 89. Niklison-Chirou MV, Steinert JR, Agostini M, Knight RA, Dinsdale D, Cattaneo A, Mak TW, Melino G. TAp73 knockout mice show morphological and functional nervous system defects associated with loss of p75 neurotrophin receptor. Proc Natl Acad Sci U S A. 2013; 110: 18952 -18957. [PubMed] .
- 90. Milosch N, Tanriöver G, Kundu A, Rami A, François JC, Baumkötter F, Weyer SW, Samanta A, Jäschke A, Brod F, Buchholz CJ, Kins S, Behl C, Müller UC, Kögel D. Holo-APP and G-protein-mediated signaling are required for sAPPα-induced activation of the Akt survival pathway. Cell Death Dis. 2014; 5: e1391 [PubMed] .
- 91. Tan MS, Tan L, Jiang T, Zhu XC, Wang HF, Jia CD, Yu JT. Amyloid-β induces NLRP1-dependent neuronal pyroptosis in models of Alzheimer's disease. Cell Death Dis. 2014; 5: e1382 [PubMed] .
- 92. Li S, Deng J, Hou H, Tian J, Giunta B, Wang Y, Sawmiller D, Smith A, Sanberg PR, Obregon D, Mori T, Tan J. Specific antibody binding to the APP672-699 region shifts APP processing from α- to β-cleavage. Cell Death Dis. 2014; 5: e1374 [PubMed] .
- 93. LeBlanc AC, Ramcharitar J, Afonso V, Hamel E, Bennett DA, Pakavathkumar P, Albrecht S. Caspase-6 activity in the CA1 region of the hippocampus induces age-dependent memory impairment. Cell Death Differ. 2014; 21: 696 -706. [PubMed] .
- 94. Tang Y, Li T, Li J, Yang J, Liu H, Zhang XJ, Le W. Jmjd3 is essential for the epigenetic modulation of microglia phenotypes in the immune pathogenesis of Parkinson's disease. Cell Death Differ. 2014; 21: 369 -380. [PubMed] .
- 95. Zhu X, Prasad S, Gaedicke S, Hettich M, Firat E, Niedermann G. Patient-derived glioblastoma stem cells are killed by CD133-specific CAR T cells but induce the T cell aging marker CD57. Oncotarget. 2015; 6: 171 -184. [PubMed] .
- 96. Larson ED, St Clair JR, Sumner WA, Bannister RA, Proenza C. Depressed pacemaker activity of sinoatrial node myocytes contributes to the age-dependent decline in maximum heart rate. Proc Natl Acad Sci U S A. 2013; 110: 18011 -18016. [PubMed] .
- 97. Liu F, Li N, Long B, Fan YY, Liu CY, Zhou QY, Murtaza I, Wang K, Li PF. Cardiac hypertrophy is negatively regulated by miR-541. Cell Death Dis. 2014; 5: e1171 [PubMed] .
- 98. Coen G, Pierantozzi A, Spizzichino D, Sardella D, Mantella D, Manni M, Pellegrino L, Romagnoli A, Pacifici R, Zuccaro P, Digiulio S. Risk factors of one year increment of coronary calcifications and survival in hemodialysis patients. BMC Nephrology. 2010; 11: 10 -19. [PubMed] .
- 99. Moe SM and Chen NX. Mechanisms of vascular calcification in chronic kidney disease. J Am Soc Nephrol. 2008; 19: 213 -216. [PubMed] .
- 100. Covic A, Kanbay M, Voroneanu L, Turgut F, Serban DN, Serban IL, Goldsmith DJ. Vascular calcification in chronic kidney disease. Clinical Science. 2010; 119: 111 -121. [PubMed] .
- 101. Jono S, McKee MD, Murry CE, Shioi A, Nishizawa Y, Mori K, Morii H, Giachelli CM. Phosphate regulation of vascular smooth muscle cell calcification. Circ Res. 2000; 87: e10 -e17. [PubMed] .
- 102. KDIGO Clinical Practice Guideline for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. 2009; 76: S1 -S130. .
- 103. Ishimura E, Okuno S, Kitatani K, Kim M, Shoji T, Nakatani T, Inaba M, Nishizawa Y. Different risk factors for peripheral vascular calcification between diabetic and non-diabetic haemodialysis patients-importance of glycaemic control. Diabetologia. 2002; 45: 1446 -1448. [PubMed] .
- 104. Stompor T. Coronary artery calcification in chronic kidney disease: An update. World J Cardio.l. 2014; 6: 115 -129. .
- 105. NKF-K/DOQI. Clinical Practice Guidelines for Hemodialysis Adequacy. Am J Kidney Dis. 1997; 30: S15 -S66. .
- 106. Mancini M, Lena AM, Saintigny G, Mahé C, Di Daniele N, Melino G, Candi E. MicroRNAs in human skin ageing. Ageing Res Rev. 2014; 17: 9 -15. [PubMed] .
- 107. Galluzzi L, Bravo-San Pedro JM, Vitale I, Aaronson SA, Abrams JM, Adam D, Alnemri ES, Altucci L, Andrews D, Annicchiarico-Petruzzelli M, Baehrecke EH, Bazan NG, Bertrand MJ, et al. Essential versus accessory aspects of cell death: recommendations of the NCCD. Cell Death Differ. 2015; 22: 58 -73. [PubMed] .
- 108. Noce A, Ferrannini M, Fabrini R, Bocedi A, Dessì M, Galli F, Federici G, Palumbo R, Di Daniele N, Ricci G. Erythrocyte glutathione transferase: a new biomarker for hemodialysis adequacy, overcoming the Kt/V(urea) dogma? Cell Death Dis. 2012; 3: e377 [PubMed] .
- 109. Millán J, Pintó X, Muñoz A, Zúñiga M, Rubiés-Prat J, Pallardo LF, Masana L, Mangas A, Hernández-Mijares A, González-Santos P, Ascaso JF. Pedro-Botet Lipoprotein ratios:physiological significance and clinical usefulness in cardiovascular prevention. Vasc Health Risk Manag. 2009; 5: 757 -765. [PubMed] .
- 110. Coen G, Manni M, Agnoli A, Balducci A, Dessi M, De Angelis S, Jankovic L, Mantella D, Morosetti M, Naticchia A, Nofroni I, Romagnoli A, Gallucci MT, et al. Fetuin-A and other risk factors in hemodialysis patients. ASAIO J. 2006; 52: 150 -156. [PubMed] .
- 111. Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M Jr, Detrano R. Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol. 1990; 15: 827 -832. [PubMed] .
- 112. Taniwaki H, Ishimura E, Tabata T, Tsujimoto Y, Shioi A, Shoji T, Inaba M, Inoue T, Nishizawa Y. Aortic calcification in haemodialysis patients with diabetes mellitus. Nephrol Dial Transplant. 2005; 20: 2472 -2247. [PubMed] .
- 113. Hoffmann U, Brady TJ, Muller J. Use of new imaging techniques to screen for coronary artery disease. Circulation. 2003; 108: e50 -53. [PubMed] .



