Introduction
Aging affects multiple cognitive domains in humans. While some individuals experience significant cognitive decline as they age, others maintain their cognitive abilities well into their later years [1–4]. This variability highlights the importance of understanding the neurobiological mechanisms underlying age-related cognitive impairment. Identifying these mechanisms will be crucial for developing effective interventions and treatments for age-related cognitive impairment and age-related diseases, such as Alzheimer’s disease.
The use of animal models, particularly non-human primates, is critical in aging research due to their close genetic, physiological, and behavioral similarities to humans [5]. For decades, macaques have been the primary non-human primate model for studying cognitive aging [6]. Macaques have the ability to perform complex cognitive tasks and substantial work has employed the macaque model to understand the underlying neural mechanisms that support cognitive functions, including memory, attention and executive function [6–10]. Further, their patterns of age-related cognitive decline closely mirror the impairments observed in humans, and they spontaneously develop age-related neuropathologies such as changes in neural excitability, including both prefrontal hypoexcitability and hippocampal hyperexcitability, and increased deposition of beta amyloid and phosphorylated tau [11–20]. However, macaques live up to 40 years and are considered aged around 20 years [21–23]. Thus, the long lifespan of the macaque poses logistical and practical challenges, particularly in conducting longitudinal studies that are essential for understanding the progression of cognitive impairment over time.
In this context, the common marmoset (Callithrix jacchus) has recently emerged as an alternative model for neuroscience research, particularly in the case of studies seeking to understand the biology of aging. Marmosets are a short-lived anthropoid primate typically living 10 to 12 years, although they can live up to 18 years. While there is currently no consensus on the exact age at which a marmoset is classified as aged, they are commonly considered aged between 7 and 8 years, as some age-related pathologies (e.g., reduced neurogenesis) emerge around this time [24–26]. Given their short lifespan, they offer a pragmatic solution for longitudinal studies, which are less feasible in the longer-lived macaque, and are critically important for understanding aging as a biological process that unfolds over an extended timeframe [24]. Indeed, marmosets also exhibit age-related cognitive impairment and undergo age-related changes in the prefrontal cortex and hippocampus [27–30]. Yet, the suitability of marmosets as a model for age-related cognitive impairment remains underexplored. Critically, it is not well established whether marmosets exhibit patterns of cognitive decline with age that are comparable to those observed in macaques and humans. While some studies have shown that marmosets have age-related impairments in the same domains as do macaques (e.g., cognitive flexibility, working memory) [28, 31, 32], another study reported age-related impairment in stimulus-reward association learning in marmosets that has not been seen in macaques [33].
Despite the growing popularity of the marmoset, comparative cognition studies with other non-human primate species are scarce [34–36]. Further, to date, no study has directly investigated age-related cognitive impairment in marmosets and macaques performing the identical cognitive task. This work is critically needed to evaluate the similarities and differences in age-related cognitive impairment across these species, and the extent to which marmosets and macaques are complementary animal models for aging investigations. Here, we utilized the Delayed Recognition Span Task (DRST), which is a complex working memory task that requires the prefrontal cortex and hippocampus, two areas that are affected early in the aging process, and in Alzheimer’s disease [37–40]. Previous work has demonstrated that older adults and macaques are impaired on this task compared to young controls, and people with Alzheimer’s disease are further impaired on this task compared to age-matched controls [16, 41–46]. Further, we previously showed that marmosets could perform this complex task and that older marmosets were impaired across multiple aspects of the DRST [28]. Utilizing this task, we conducted the first direct comparison of age-related cognitive impairment between marmosets and macaques, the two most commonly utilized primate species. This approach not only contributes to our understanding of cognitive aging in non-human primates, but also evaluates the potential of marmosets as a viable model for studying the neurobiology of age-related cognitive impairment. Comparative studies such as these are essential for advancing our understanding of cognitive decline mechanisms, ultimately guiding the development of targeted interventions and therapies for age-related cognitive disorders.
Results
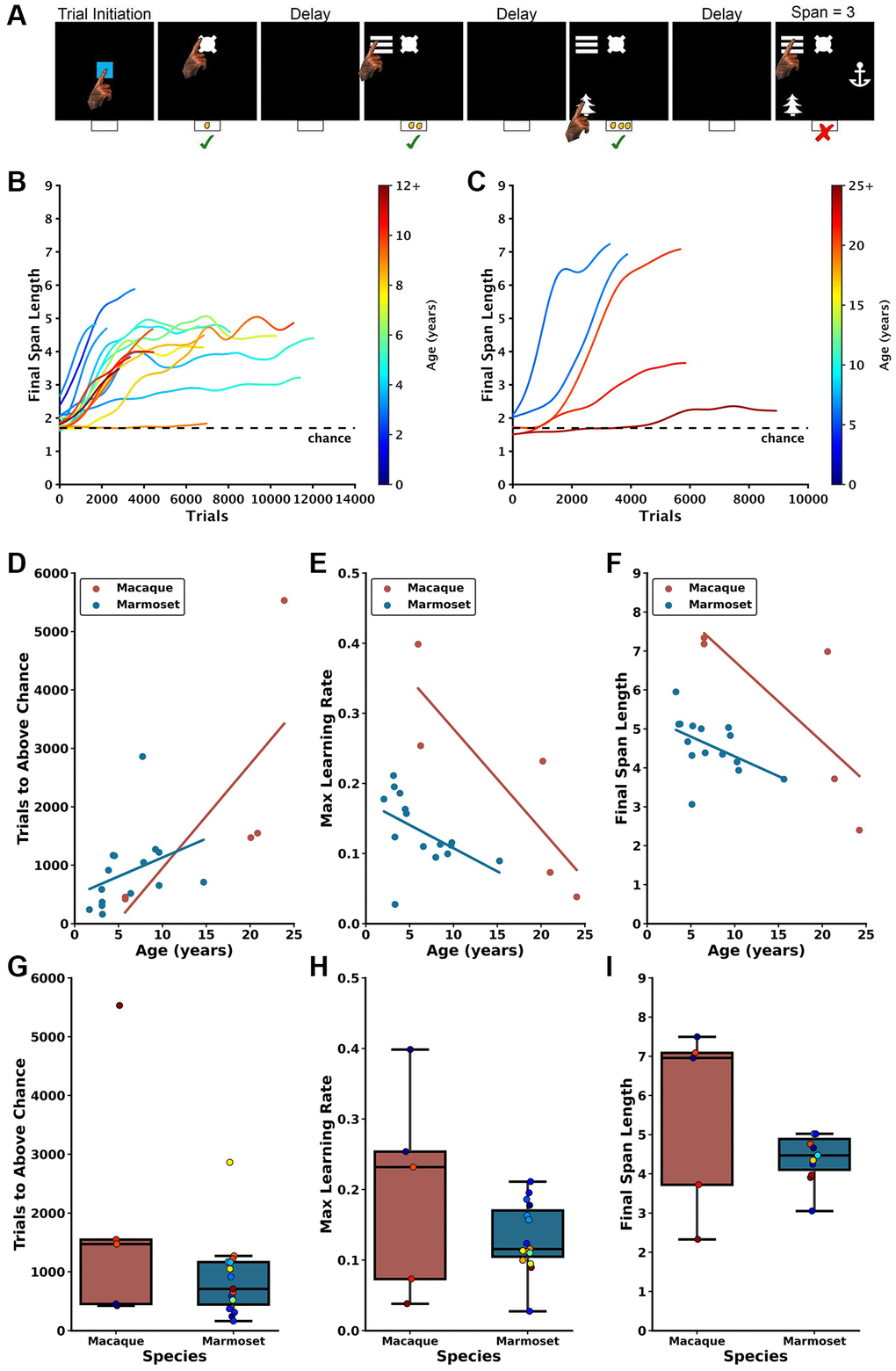
Some of the marmoset data presented here (Figures 1–3) were published previously [28].
Associations between age and delayed non-match-to-sample performance
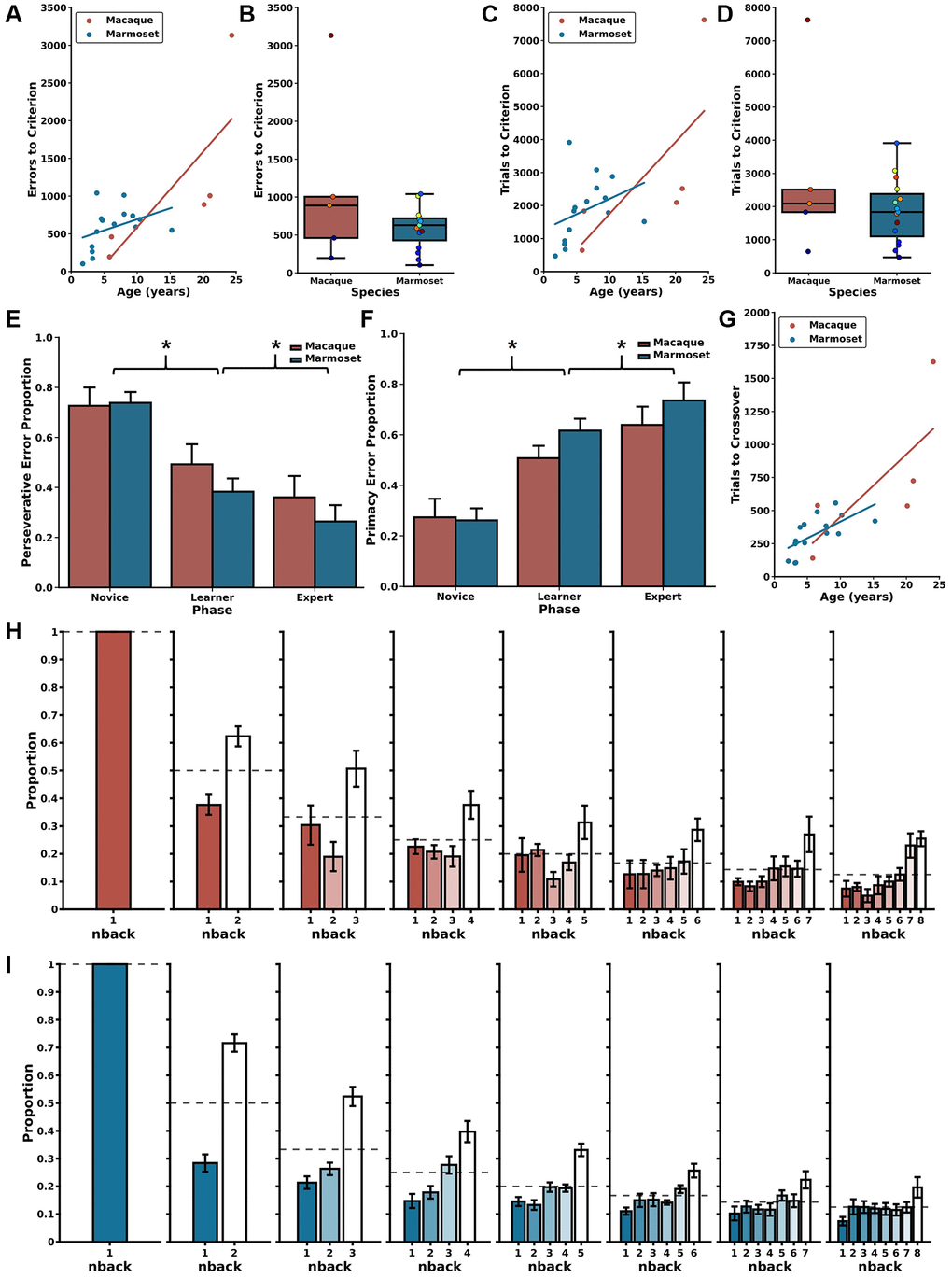
The first two parts of a DRST trial (Trial Difficulty Level (TDL)1 and TDL2, respectively) approximate a Delayed Non-Match-to-Sample (DNMS) paradigm. Specifically, the single stimulus presented in TDL1 is akin to a DNMS sample, and the two stimuli presented in TDL2 are akin to a DNMS choice. Therefore, by measuring performance of monkeys on DRST TDL2 trials, we can estimate DNMS task acquisition in the context of the DRST task. The two most frequently used dependent measures to assess DNMS performance are errors and trials to a learning criterion. We set the criterion a posteriori at 90% accuracy, achieved by responding correctly on at least 18 out of 20 consecutive trials. Spearman correlations revealed strong, significant associations between age and both errors to criterion (ETC) and trials to criterion (TTC) for both macaques and marmosets (Figure 2A, ETC; macaques: r(3) = 1.00, p = 1.40 × 10−24; marmosets: r(13) = 0.52, p = 4.78 × 10−2, Figure 2C, TTC; macaques: r(3) = 1.00, p = 1.40 × 10−24; marmosets: r(13) = 0.58, p = 2.37 × 10−2). Direct comparisons of macaque and marmoset DNMS performance revealed similar levels, whether measured by ETC or TTC, and regardless of age (Figure 2B, ETC: Mann Whitney U = 47.00, p = 0.44; Figure 2D, TTC: Mann Whitney U = 43.00, p = 0.67).
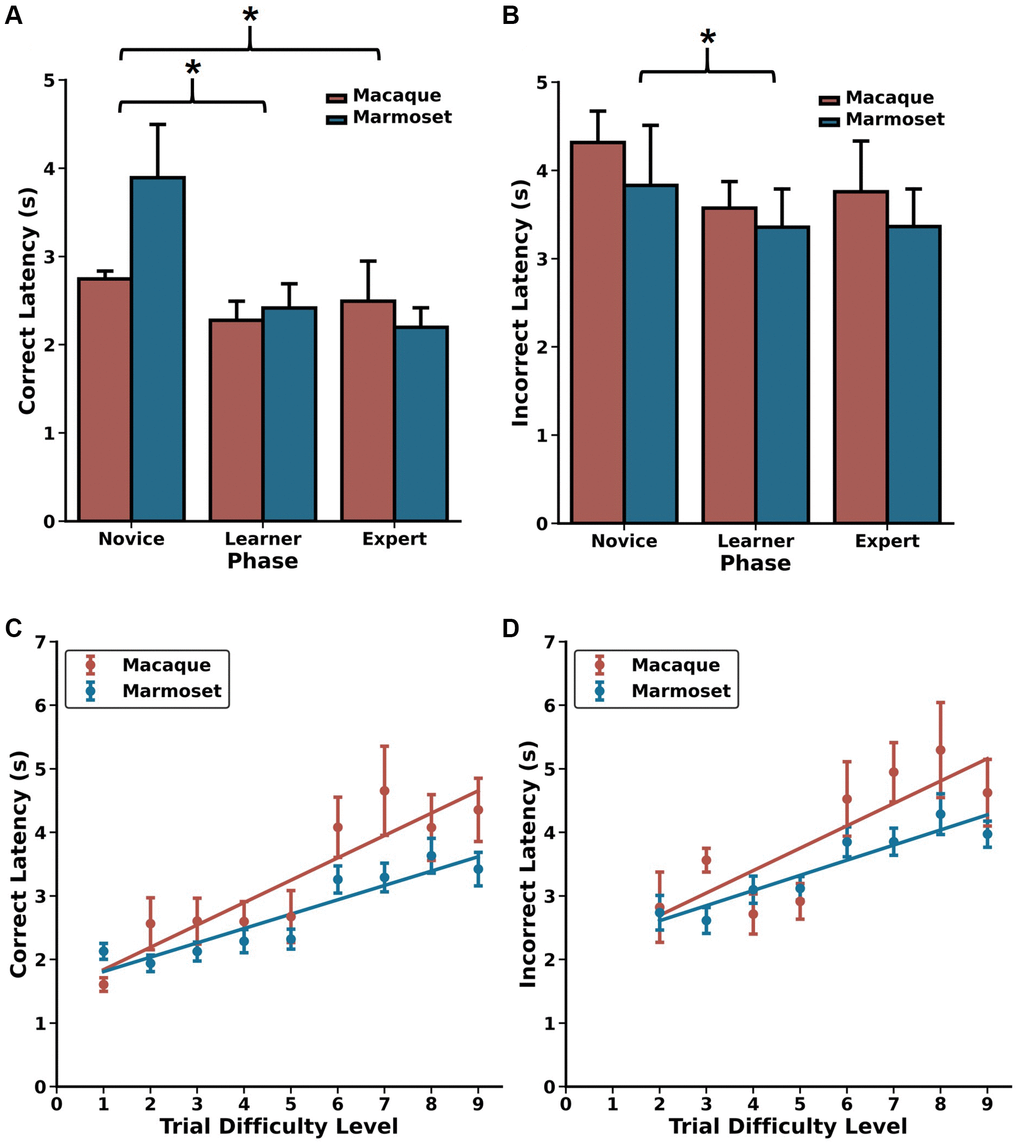
Similar choice latency patterns between macaques and marmosets reveal effects of cognitive load
One of the benefits of using infrared touch screens for evaluating cognitive performance is their capability to precisely and consistently measure choice response times. This metric is widely recognized as a reliable indicator of processing speed, and it shows associations with cognitive load and task complexity [49–51]. To investigate whether this trend persisted when monkeys were engaged in the DRST, we analyzed the response times for correct and incorrect choices made by each monkey in various Phases of the DRST, as well as for different levels of task difficulty in the Expert Phase. A Scheirer Ray Hare Test uncovered significant main effects related to response type (correct choice, incorrect choice) and DRST Phase (Novice, Learner, Expert), along with a significant interaction between these factors (Figure 3A, 3B; response type: H(1)=22.635, p = 1.959 × 10−6; phase: H(2) = 14.954, p = 5.66 × 10−4; interaction: H(2) = 44.733, p = 1.934 × 10−10). There was no main effect of species (H(1) = 0.159, p = 0.690).
When investigating how correct choice latency changed as a function of experience, we found that, across species, correct latencies were longer during the Novice Phase compared to the Learner and Expert Phases, with no significant difference between the Learner and Expert Phases (Wilcoxon signed-rank test; Novice vs. Learner: p = 3.0518 × 10−5, Novice vs. Expert: p =5.80 × 10−4, Learner vs. Expert: p = 0.117). We observed a similar pattern with incorrect latency, demonstrating that both macaques and marmosets make choices more rapidly as they gain experience and proficiency on the DRST (Wilcoxon signed-rank test; Novice vs. Learner: p = 0.029, Novice vs. Expert: p = 0.093, Learner vs. Expert: p = 1.00). To explore whether incorrect choices might be attributed to impulsiveness, we compared the response times of correct and incorrect choices within each of the Phases. During the Novice Phase, correct and incorrect choice response times were similar. However, during the Learner and Expert Phases, incorrect choice response times were significantly longer than those for correct choices (Figure 3A, 3B; Wilcoxon signed-rank tests; Novice: p = 0.216; Learner: p = 1.907 × 10−6; Expert: p = 3.052 × 10−5). This suggests that when monkeys made errors, it was unlikely due to impulsivity, as they took considerably more time to respond in such instances.
We next examined whether elevated cognitive load was reflected in the choice latency data from the Expert Phase. To do this, we analyzed correct and incorrect choice latency data separately for each of the TDLs. We found strong, positive associations between increasing TDL and increasing correct latency for both macaques and marmosets (Figure 3C, 3D; Spearman’s rank-order correlations; Macaque: r(7) = 0.900, p = 9.431 × 10−4; Marmoset: r(6) = 0.933, p = 2.359 × 10−4). Similar associations were also found between TDL and incorrect latency (Spearman’s rank-order correlations; Macaque: r(6) = 0.810, p = 1.490 × 10−2; Marmoset: r(6) = 0.952, p = 2.604 × 10−4). Together, these results demonstrate that, as TDLs increase, so does cognitive load, and this is reflected in increased processing time and longer choice latencies in both macaques and marmosets.
Species-specific effects of longer delays on DRST performance metrics
After reaching plateaued levels of performance on the DRST when trials included a 2 second delay between each stimulus presentation working memory was taxed further by the addition of longer delays. All macaques and a subset of the marmosets were tested with these longer delays which included 6, 10, and 14 seconds, and macaques were additionally tested with a 30 second delay.
First, we evaluated the effects of longer delays on DRST performance as measured by Final Span Length, and compared these effects across species. We found a significant main effect of species, no significant main effect of delay, and a significant species by delay interaction (Figure 4A; Scheirer Ray Hare: species H(1) = 19.25, p = 1.149 × 10−5; delay H(4) = 8.295, p = 0.0814, species x delay interaction H(4) = 70.189, p = 2.071 × 10−14). The significant main effect of species was driven by significant differences in performance between macaques and marmosets on all delays greater than 2 seconds (Mann-Whitney U-tests; 2 seconds U = 18.0, p = 0.440, 6 seconds U = 5.0, p = 0.0127, 10 seconds U = 2.0, p = 6.21 × 10−3, 14 seconds U = 1.0, p = 8.66 × 10−3). These differences emerged because marmosets exhibited a significant delay-dependent decrease in Final Span Length, whereas macaque performance trended towards a delay-dependent decrease in Final Span Length but did not reach statistical significance (Wilcoxon signed-rank test results in Table 2).
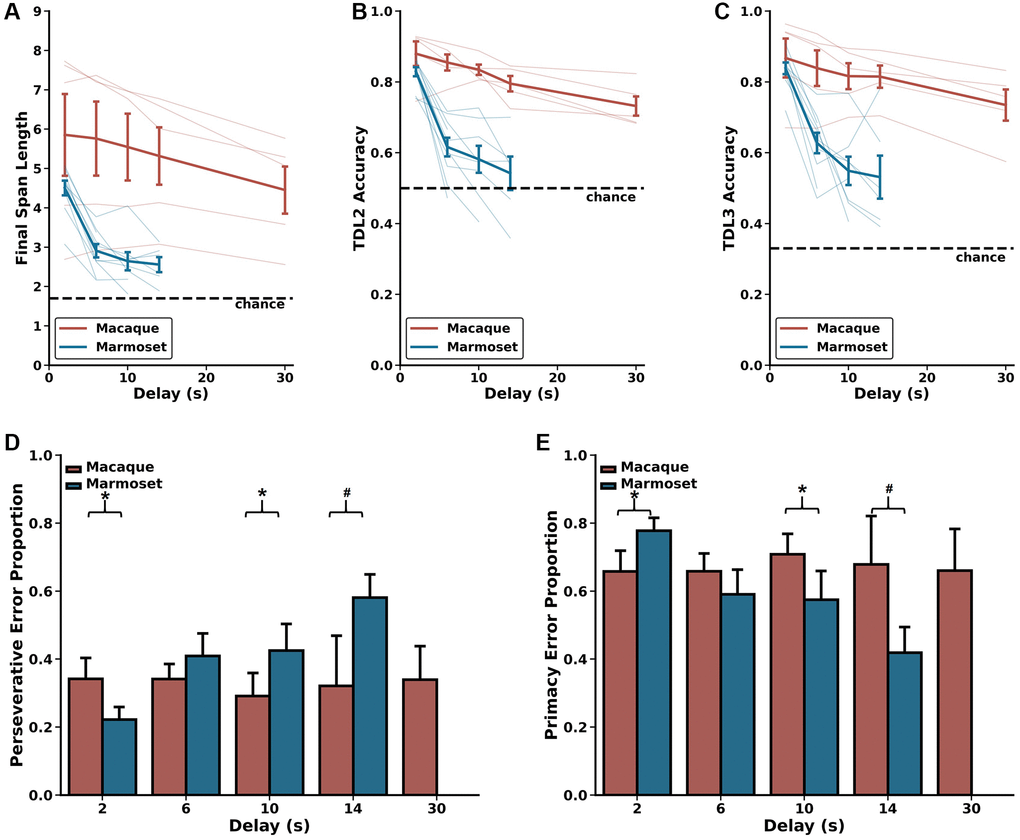
Figure 4. Delay-related effects on DRST performance. Marmosets (blue) show significant delay-dependent decreased DRST performance, whereas macaques (red) do not. Also, macaques have significantly higher performance than marmosets at delays longer than 2 seconds. These results are seen on several measures of performance including (A) average Final Span Length, (B) accuracy on the DNMS (TDL2) portion of the DRST, and (C) accuracy on TDL3 trials. (D) On TDL3 trials, marmosets’ perseverative errors increased in a delay-dependent manner, whereas macaque perseverative errors remained consistent across varying delays. (E) Marmosets’ primacy error rate showed a corresponding delay-dependent decrease, and macaque primacy errors remained consistent across the varying delays. Lightly shaded lines in (A–C) depict individual animal performance as a function of delay. Bold colored lines in (A–C) depict species average performance as a function of delay. mean ± SEM, *p < 0.05.
Table 2. P-values for Wilcoxon signed-rank tests to compare performance across delays by species for Final Span Length (FSL), accuracy on Trial Difficulty Level (TDL)2 and TDL3, and proportion of perseverative errors.
| Macaques | Marmosets | |||||||
| Delay (s) | FSL | TDL2 | TDL3 | Errors | FSL | TDL2 | TDL3 | Errors |
| 2 vs. 6 | 0.813 | 0.0625 | 0.0625 | 1 | 0.00195 | 0.00195 | 0.00195 | 0.00195 |
| 2 vs. 10 | 0.313 | 0.125 | 0.125 | 0.313 | 0.00781 | 0.00781 | 0.00781 | 0.0156 |
| 2 vs. 14 | 0.313 | 0.188 | 0.188 | 1 | 0.0313 | 0.0313 | 0.0313 | 0.0313 |
| 2 vs. 30 | 0.0625 | 0.0625 | 0.0625 | 1 | N/A | N/A | N/A | N/A |
| 6 vs. 10 | 0.188 | 0.313 | 0.313 | 0.273 | 0.0781 | 0.0547 | 0.0547 | 1 |
| 6 vs. 14 | 0.313 | 0.438 | 0.438 | 1 | 0.0625 | 0.0625 | 0.0625 | 0.156 |
| 6 vs. 30 | 0.0625 | 0.0625 | 0.0625 | 1 | N/A | N/A | N/A | N/A |
| 10 vs. 14 | 0.313 | 0.813 | 0.813 | 1 | 0.563 | 0.438 | 0.438 | 0.156 |
| 10 vs. 30 | 0.0625 | 0.0625 | 0.0625 | 0.625 | N/A | N/A | N/A | N/A |
| 14 vs. 30 | 0.0625 | 0.0625 | 0.0625 | 0.625 | N/A | N/A | N/A | N/A |
Next, we evaluated the effects of longer delays on TDL2 and TDL3 accuracy. We found significant main effects of species and delay and a significant species by delay interaction on both of these TDLs (TDL2: Figure 4B; Scheirer Ray Hare: species H(1) = 17.536, p = 2.819 × 10−5; delay H(4) = 17.449, p = 1.581 × 10−3; species x delay interaction H(4) = 110.182, p = 6.656 × 10−23; TDL3: Figure 4C; Scheirer Ray Hare: species H(1) = 14.239, p = 1.610 × 10−4; delay H(4) = 14.749, p = 5.250 × 10−3; species x delay interaction H(4) = 101.587, p = 4.518 × 10−21). The significant main effects of species were driven by significant differences in performance between macaques and marmosets on all delays greater than 2 seconds (Mann-Whitney U-tests; TDL2: 2 seconds U = 10.0, p = 0.0753, 6 seconds U = 0.0, p = 6.660 × 10−4, 10 seconds U = 0.0, p = 1.554 × 10−3, 14 seconds U = 0.0, p = 4.329 × 10−3; TDL3: 2 seconds U = 17.0, p = 0.371, 6 seconds U = 4.0, p = 7.992 × 10−3, 10 seconds U = 2.0, p = 6.216 × 10−3, 14 seconds U = 1.0, p = 8.658 × 10−3). These differences emerged because marmosets exhibited a delay-dependent decrease in accuracy on both TDL2 and TDL3, whereas macaques did not (Wilcoxon signed-rank test results in Table 2). Together, these results suggest that the effect of delay on performance varied as a function of species.
As described above, trials ending with a Final Span Length of two present a unique opportunity to investigate the prevalence with which monkeys committed perseverative and primacy errors. We assessed the proportion of these types of errors as a function of species and delay length and found no main effect of species, no main effect of delay, but a significant species by delay interaction (Figure 4D, 4E; Scheirer Ray Hare: species H(1) = 1.329, p = 0.249; delay H(4) = 16.204, p = 0.0940; species x delay interaction H(4) = 309.889, p = 1.260 × 10−60). This significant interaction is driven by the fact that marmosets exhibited a delay-dependent increase in perseverative errors and a corresponding delay-dependent decrease in primacy errors, whereas macaque perseverative and primacy errors were unchanged across varied delays (Wilcoxon signed-rank test results in Table 2). Thus, the proportion of error types changed as a function of delay only in marmosets.
Discussion
In this study, we conducted the first direct comparison of cognitive ability as a function of age in macaques and marmosets. We have previously demonstrated that marmosets exhibit robust and systematic impairments on the DRST [28]. By testing young and aged macaques on the identical working memory task, we found that they exhibit remarkably similar age-related learning and working memory impairments to marmosets. This work establishes that the patterns of age-related working memory deficits are largely conserved across the two most common non-human primate models used for cognitive aging research. Macaques demonstrate more robust performance than marmosets when working memory is taxed through increased delay durations.
Evaluation of macaque and marmoset performance in the context of prior work
In humans and non-human primates, cognitive functions that rely on the prefrontal cortex and hippocampus decline with age. As such, working memory deficits appear particularly early in the aging process [52, 53]. To measure working memory as a function of aging across macaques and marmosets, we used a touch screen version of the DRST. Using this task, we found strikingly similar associations between advancing age and working memory impairment in macaques and marmosets.
Specifically, we found that aged macaques have impaired ability to acquire the rules of the DRST, requiring more experience to perform above the levels expected by chance, and learning at a slower rate, than young macaques. This parallels our prior findings in the marmoset and therefore shows that both species exhibit age-related impairments in acquisition and learning of the DRST [28]. We also found age-related decreased working memory capacity in both macaques and marmosets. These findings align with previous work that has documented age-related impairments on the DRST in each of these species independently, albeit on similar, but non-identical task designs [28, 42, 54, 55]. It is important to note that prior work in macaques has employed versions of the DRST that require the subject to use either object identity or spatial location. In contrast, our approach allowed monkeys to use both object identity and spatial location, as our primary goal was to quantify working memory capacity independent of a particular domain. However, future work is needed to investigate if similar age-related changes would be seen if the task were selective for either visual or spatial information.
We capitalized on the fact that within the context of each DRST trial there existed an opportunity to directly compare performance between macaques and marmosets on the more commonly-used DNMS paradigm. We found that, in both species, aging was associated with impaired performance, measured by errors to a learning criterion. This aligns with numerous studies in macaques reporting similar findings [15, 56–59].
Although we find clear and compelling evidence for age-related working memory impairment, evaluation of individual animal learning curves revealed striking levels of between animal variability which was particularly evident in older individuals. Similar to previous reports in humans, similarly aged macaques and similarly aged marmosets demonstrated different working memory aptitudes. A subset of animals of each species performed at high levels, while others performed less optimally.
Given that a subset of the macaques approached ceiling levels of performance when 2-second delays were employed, we evaluated potential differences in DRST performance between macaques and marmosets under more challenging experimental conditions. Historically, working memory performance decreases as a function of longer delays that tax working memory [15, 37, 58, 60]. We found that increasing delays affected the performance of marmosets and macaques differently. Whereas macaques maintained stable levels of performance on delays up to 30 seconds, marmoset performance was severely impaired by increased delays. This clear species difference recapitulates prior work demonstrating that macaques are able to hold information in working memory for longer delays [61, 62]. Therefore, macaques, with more complex cortical structures, may have neural circuits that allow for better maintenance of information over extended delays. This invites comparative anatomy research to identify structural and functional differences in brain regions involved in working memory, such as the prefrontal cortex and hippocampus [34].
Limitations
The primary limitation of this study is the relatively small sample size of macaques. With macaques performing between 6,000 and 10,000 trials, we nevertheless, we were able to identify consistent patterns of age-related cognitive impairment. This extensive characterization additionally allowed us to differentiate learning from memory processes and conduct detailed error analyses. However, future research should aim to replicate and extend these findings with larger sample sizes to enhance generalizability. Another promising avenue for future research is investigating the impact of sex on age-related cognitive impairment. Prior studies suggest that age-related working memory impairment may manifest differently between males and females, with evidence in females linking these impairments to synaptic changes associated with hormonal shifts after menopause [33, 79–81]. Although the current study was not powered to examine sex differences, future research should incorporate analyses that consider sex as a critical variable, exploring whether marmosets and macaques exhibit similar sex-dependent cognitive aging trajectories.
Conclusion
Our study provides the first direct comparison of age-related cognitive impairments between macaques and marmosets, revealing that these species exhibit similar learning and working memory deficits with age. The observation that macaque working memory performance is more resilient to the effects of longer delays suggests a potentially larger working memory capacity compared to marmosets. Future work is needed to understand whether similar neural circuits underlie performance on this task across these species, and also to determine what age-related neuropathology gives rise to declining working memory.
Methods
Subjects
Marmosets
A total of 16 common marmosets (Callithrix jacchus) of both sexes participated in this study (8 female, 8 male) (Table 3). Data from these same individuals performing the DRST with a 2-sec delay have been published previously (Glavis-Bloom et al., 2022). For the present study, these marmosets performed additional DRST testing with longer delays. The marmosets ranged between 3.05 and 14.64 years of age at the onset of the study. Marmosets were housed singly or in pairs and were provided with species appropriate enrichment and diet. All procedures were carried out in accordance with the National Institutes of Health guidelines and were approved by the Salk Institute for Biological Studies Institutional Animal Care and Use Committee protocol number 17-00030.
Table 3. Description of subjects.
| Animal ID | Species | Sex | Age at start (years) | Age at end (years) |
| A19124 | Macaque | F | 5.73 | 6.98 |
| A19123 | Macaque | F | 5.74 | 6.55 |
| A19090 | Macaque | F | 19.92 | 20.65 |
| A19089 | Macaque | F | 20.7 | 21.44 |
| A18057 | Macaque | F | 23.66 | 24.55 |
| QK | Marmoset | F | 1.66 | 3.12 |
| AN | Marmoset | F | 3.05 | 3.31 |
| LO | Marmoset | M | 3.11 | 5.32 |
| JI | Marmoset | M | 3.12 | 5.3 |
| RJ | Marmoset | M | 3.14 | 3.47 |
| BZ | Marmoset | M | 3.81 | 5.12 |
| CC | Marmoset | F | 4.32 | 7.36 |
| VS | Marmoset | F | 4.48 | 5.84 |
| PA | Marmoset | F | 6.35 | 7.81 |
| EF | Marmoset | F | 7.6 | 9.81 |
| TR | Marmoset | M | 7.81 | 8.7 |
| FL | Marmoset | M | 8.68 | 9.15 |
| BL | Marmoset | F | 9.16 | 9.52 |
| CZ | Marmoset | M | 9.55 | 10.42 |
| BO | Marmoset | M | 9.56 | 10.44 |
| RZ | Marmoset | F | 14.64 | 15.46 |
Macaques
A total of five female rhesus macaques (Macaca mulatta) participated in this study (Table 3). Two of the monkeys were young (5.73 and 5.74 years of age), and three were aged (19.90, 20.70, and 23.66 years of age). All macaque monkeys were housed singly or in pairs in standard caging and were provided with species appropriate enrichment and diet. All procedures were carried out in accordance with the National Institutes of Health guidelines and were approved by the University of Washington Institutional Animal Care and Use Committee protocol number 4316-01.
Equipment
Cognitive testing for macaques and marmosets was administered via home cage mounted touch screen testing stations (Lafayette Instrument Company, Lafayette, IN, USA). These stations were self-contained and included an infrared touch screen (15 inches, 764 × 1028 pixels for macaques; 10.4 inches, 800 × 600 pixels for marmosets) and reward delivery system (pellet dispenser for macaques; peristaltic pump for liquid rewards for marmosets). Cognitive tasks were programmed using Animal Behavior Environment Test (ABET) Cognition software (Lafayette Instrument Company, Lafayette, IN, USA) that controlled all aspects of the task including the order of trials, timing, stimuli selection and display location, and delivery of rewards. The software also recorded detailed logs of task-related events (e.g., stimulus display, screen touches) with millisecond temporal resolution.
Statistical analyses
Raw data were extracted from the ABET-produced logs and analyzed using custom purposed Python scripts along with the statistics module within SciPy. Given the sample size and to control for outliers, non-parametric statistical tests were used throughout the study. Spearman’s rank-order correlations were used to assess the relationship between age and various dependent variable performance metrics, and between task parameters and performance. Scheirer Ray Hare Tests were used to assess two factor interactions across time with Wilcoxon’s signed-rank tests or Mann Whitney U post-hoc tests. Friedman’s Tests with Nemenyi post-hoc tests were used to identify within-factor differences. Performance was compared to chance using Chi-Square Goodness of Fit Tests, and Mann Whitney U-tests were used for species comparisons.
Cognitive testing
Cognitive testing methodologies are derived from Glavis-Bloom et al., 2022.
Touch training
All monkeys were trained to operate the touch screens via a positive reinforcement procedure. Briefly, monkeys learned, through trial and error, that interacting with the touch screen yielded rewards. For marmosets, to encourage initial physical engagement with the screen, Marshmallow Fluff™ was applied in each of the nine locations where a blue square stimulus was presented. Once the monkeys associated touching the screen with earning rewards, no additional Marshmallow Fluff™ was applied (Glavis-Bloom et al., 2022). The macaques had previously been trained to touch a physical target to earn rewards. Therefore, to encourage initial physical engagement with the screen, the physical target was placed near the screen. Over the course of several days of training, the number of stimuli on the screen was reduced so that by the end of the touch training procedure, all monkeys were touching a single stimulus displayed on the screen in any of the possible locations. Then, over an additional few days of training, the amount of reward earned per screen touch was also reduced. Marmosets were rewarded with sweetened liquid such as apple juice, and macaques were rewarded with fruit-flavored pellets (190 mg Dustless Precision Pellets, Bio-Serv, Flemington, NJ, USA).
Delayed recognition span task
The Delayed Recognition Span Task (DRST) measures working memory capacity (Figure 1A). Each trial of the DRST was initiated when a monkey touched a blue square in the center of the screen. Subsequently, a single black and white stimulus, chosen at random from a pool of 400 images, appeared on the screen in one of nine possible locations, also determined randomly (see Figure 1A for example stimuli). Upon touching this initial stimulus, the monkey received a small reward. After a delay, during which the screen remained blank, a two-alternative forced choice was presented. This choice included the original stimulus in its original location and a novel, visually distinct stimulus placed in a different pseudo-randomly selected location. If the monkey selected the novel stimulus, a correct response was recorded, a reward was dispensed, and another delay ensued. Subsequently, the first two stimuli reappeared in their original positions, and a third novel stimulus was introduced in a pseudo-randomly chosen location, with reward dispensed for selection of this new stimulus. This process continued with the introduction of novel stimuli after additional delays until the trial reached one of three possible conclusions: (1) the monkey successfully made nine consecutive correct selections; (2) the monkey failed to make a selection within a 12-second timeframe (i.e., omission); (3) the monkey made an incorrect response by selecting a non-novel stimulus. In cases of omission or incorrect responses, no reward was provided, and a five-second time-out period commenced before a new trial could be initiated. The “Final Span Length” for each trial was recorded as the number of correctly selected stimuli before the trial’s conclusion. The variations in the number of stimuli on the screen as trials progressed were referred to as trial difficulty levels (TDLs). Macaques and marmosets performed the DRST with a 2 second delay until performance levels plateaued. Subsequently, all macaques and a subset of the marmosets were tested on the DRST with delays greater than two seconds. Macaques and marmosets were tested with delays of 2, 6, 10, and 14 seconds, and macaques were additionally tested with a 30 second delay.
To maintain engagement and motivation, the quantity of reward increased in correspondence with the difficulty level of the trials. Specifically, marmosets received 0.05 mL of reward for accurate responses when one stimulus was on the screen, 0.1 mL for accurate responses when two, three, or four stimuli were on the screen, and 0.2 mL for accurate responses when five, six, seven, eight, or nine stimuli were on the screen. Likewise, macaques earned one reward pellet when responding to one stimulus on the screen, earned two reward pellets for correct responses when two, three, or four stimuli were on the screen, and three reward pellets for correctly responding when five or more stimuli were on the screen.
Each marmoset and macaque underwent testing sessions two to five days per week, and each session concluded after three hours or once the marmoset had earned 20 mL of reward, whichever event occurred first. Macaques underwent testing sessions three to five days a week and each session concluded after an hour or once the macaque earned 600 pellets, whichever event occurred first. Marmosets and macaques were not food or water restricted at any time throughout testing, and were naïve to touch screen cognitive testing prior to the beginning of the study.
The marmoset data used for the single delay analyses in Figures 1–3 are reproduced from Glavis-Bloom et al. 2022. The multi-delay data for both species are new and have not been published previously. All macaque data are new and have not been published previously.
Author Contributions
CRV, MLJ, EAB, and CGB conceived of the research. CRV, MLJ, PAA, SYZ, MNL, and CGB collected data and performed data analysis; CRV, PAA, and CGB wrote the manuscript; EAB and CGB revised the manuscript and provided supervision.
Acknowledgments
We thank Katie Williams for assistance in the care of the marmosets and technical support. We thank Kelly Morrisroe for assistance in training of the rhesus macaques and Paige Robertson for assistance with behavioral task programming.
Conflicts of Interest
None of the authors have financial or conflict of interests related to the manuscript.
Ethical Statement
All procedures were carried out in accordance with the National Institutes of Health guidelines. Macaque studies were approved by the University of Washington Institutional Animal Care and Use Committee protocol number 4316-01. Marmoset studies were approved by the Salk Institute for Biological Studies Institutional Animal Care and Use Committee protocol number 17-00030. All non-human primates were housed in proximity to conspecifics and provided species-appropriate enrichment and diet.
Funding
This research was supported by an AHA-Allen Initiative in Brain Health and Cognitive Impairment award made jointly through the American Heart Association and The Paul G. Allen Frontiers Group: 19PABH134610000AHA, National Institutes of Health grants 1R21AG068967-01 and P51OD010425, grants from the Larry L. Hillblom Foundation and the Don and Lorraine Freeberg Foundation, and the Fiona and Sanjay Jha Chair in Neuroscience.
References
- 1. Small SA, Perera GM, DeLaPaz R, Mayeux R, Stern Y. Differential regional dysfunction of the hippocampal formation among elderly with memory decline and Alzheimer's disease. Ann Neurol. 1999; 45:466–72. https://doi.org/10.1002/1531-8249(199904)45:4<466::aid-ana8>3.0.co;2-q [PubMed]
- 2. Cook Maher A, Makowski-Woidan B, Kuang A, Zhang H, Weintraub S, Mesulam MM, Rogalski E. Neuropsychological Profiles of Older Adults with Superior versus Average Episodic Memory: The Northwestern "SuperAger" Cohort. J Int Neuropsychol Soc. 2022; 28:563–73. https://doi.org/10.1017/S1355617721000837 [PubMed]
- 3. Stern Y, Albert M, Barnes CA, Cabeza R, Pascual-Leone A, Rapp PR. A framework for concepts of reserve and resilience in aging. Neurobiol Aging. 2023; 124:100–3. https://doi.org/10.1016/j.neurobiolaging.2022.10.015 [PubMed]
- 4. Vanderlip CR, Stark CEL, Initiative ADN. Digital cognitive assessments as low-burden markers for predicting future cognitive decline and tau accumulation across the Alzheimer’s spectrum. bioRxiv. 2024; 2024.05.23.595638. Available from: https://www.biorxiv.org/content/10.1101/2024.05.23.595638v1.
- 5. Izpisua Belmonte JC, Callaway EM, Caddick SJ, Churchland P, Feng G, Homanics GE, Lee KF, Leopold DA, Miller CT, Mitchell JF, Mitalipov S, Moutri AR, Movshon JA, et al. Brains, genes, and primates. Neuron. 2015; 86:617–31. https://doi.org/10.1016/j.neuron.2015.03.021 [PubMed]
- 6. Gray DT, Barnes CA. Experiments in macaque monkeys provide critical insights into age-associated changes in cognitive and sensory function. Proc Natl Acad Sci U S A. 2019; 116:26247–54. https://doi.org/10.1073/pnas.1902279116 [PubMed]
- 7. Zola SM, Squire LR, Teng E, Stefanacci L, Buffalo EA, Clark RE. Impaired recognition memory in monkeys after damage limited to the hippocampal region. J Neurosci. 2000; 20:451–63. https://doi.org/10.1523/JNEUROSCI.20-01-00451.2000 [PubMed]
- 8. Moore TL, Killiany RJ, Herndon JG, Rosene DL, Moss MB. Executive system dysfunction occurs as early as middle-age in the rhesus monkey. Neurobiol Aging. 2006; 27:1484–93. https://doi.org/10.1016/j.neurobiolaging.2005.08.004 [PubMed]
- 9. Gray DT, Smith AC, Burke SN, Gazzaley A, Barnes CA. Attentional updating and monitoring and affective shifting are impacted independently by aging in macaque monkeys. Behav Brain Res. 2017; 322:329–38. https://doi.org/10.1016/j.bbr.2016.06.056 [PubMed]
- 10. Zola-Morgan S, Squire LR, Amaral DG, Suzuki WA. Lesions of perirhinal and parahippocampal cortex that spare the amygdala and hippocampal formation produce severe memory impairment. J Neurosci. 1989; 9:4355–70. https://doi.org/10.1523/JNEUROSCI.09-12-04355.1989 [PubMed]
- 11. Yassa MA, Lacy JW, Stark SM, Albert MS, Gallagher M, Stark CE. Pattern separation deficits associated with increased hippocampal CA3 and dentate gyrus activity in nondemented older adults. Hippocampus. 2011; 21:968–79. https://doi.org/10.1002/hipo.20808 [PubMed]
- 12. Morrison JH, Baxter MG. The ageing cortical synapse: hallmarks and implications for cognitive decline. Nat Rev Neurosci. 2012; 13:240–50. https://doi.org/10.1038/nrn3200 [PubMed]
- 13. Thomé A, Gray DT, Erickson CA, Lipa P, Barnes CA. Memory impairment in aged primates is associated with region-specific network dysfunction. Mol Psychiatry. 2016; 21:1257–62. https://doi.org/10.1038/mp.2015.160 [PubMed]
- 14. Arnsten AFT, Datta D, Preuss TM. Studies of aging nonhuman primates illuminate the etiology of early-stage Alzheimer's-like neuropathology: An evolutionary perspective. Am J Primatol. 2021; 83:e23254. https://doi.org/10.1002/ajp.23254 [PubMed]
- 15. Baxter MG, Roberts MT, Roberts JA, Rapp PR. Neuropsychology of cognitive aging in rhesus monkeys. Neurobiol Aging. 2023; 130:40–9. https://doi.org/10.1016/j.neurobiolaging.2023.06.011 [PubMed]
- 16. Herndon JG, Moss MB, Rosene DL, Killiany RJ. Patterns of cognitive decline in aged rhesus monkeys. Behav Brain Res. 1997; 87:25–34. https://doi.org/10.1016/s0166-4328(96)02256-5 [PubMed]
- 17. Moss MB, Rosene DL, Peters A. Effects of aging on visual recognition memory in the rhesus monkey. Neurobiol Aging. 1988; 9:495–502. https://doi.org/10.1016/s0197-4580(88)80103-9 [PubMed]
- 18. Cabeza R, Anderson ND, Houle S, Mangels JA, Nyberg L. Age-related differences in neural activity during item and temporal-order memory retrieval: a positron emission tomography study. J Cogn Neurosci. 2000; 12:197–206. https://doi.org/10.1162/089892900561832 [PubMed]
- 19. Datta D, Leslie SN, Wang M, Morozov YM, Yang S, Mentone S, Zeiss C, Duque A, Rakic P, Horvath TL, van Dyck CH, Nairn AC, Arnsten AFT. Age-related calcium dysregulation linked with tau pathology and impaired cognition in non-human primates. Alzheimers Dement. 2021; 17:920–32. https://doi.org/10.1002/alz.12325 [PubMed]
- 20. Wang M, Gamo NJ, Yang Y, Jin LE, Wang XJ, Laubach M, Mazer JA, Lee D, Arnsten AF. Neuronal basis of age-related working memory decline. Nature. 2011; 476:210–3. https://doi.org/10.1038/nature10243 [PubMed]
- 21. Moore TL, Killiany RJ, Herndon JG, Rosene DL, Moss MB. Impairment in abstraction and set shifting in aged rhesus monkeys. Neurobiol Aging. 2003; 24:125–34. https://doi.org/10.1016/s0197-4580(02)00054-4 [PubMed]
- 22. Simmons HA. Age-Associated Pathology in Rhesus Macaques (Macaca mulatta). Vet Pathol. 2016; 53:399–416. https://doi.org/10.1177/0300985815620628 [PubMed]
- 23. Chiou KL, Montague MJ, Goldman EA, Watowich MM, Sams SN, Song J, Horvath JE, Sterner KN, Ruiz-Lambides AV, Martínez MI, Higham JP, Brent LJN, Platt ML, Snyder-Mackler N. Rhesus macaques as a tractable physiological model of human ageing. Philos Trans R Soc Lond B Biol Sci. 2020; 375:20190612. https://doi.org/10.1098/rstb.2019.0612 [PubMed]
- 24. Tardif SD, Mansfield KG, Ratnam R, Ross CN, Ziegler TE. The marmoset as a model of aging and age-related diseases. ILAR J. 2011; 52:54–65. https://doi.org/10.1093/ilar.52.1.54 [PubMed]
- 25. Freire-Cobo C, Edler MK, Varghese M, Munger E, Laffey J, Raia S, In SS, Wicinski B, Medalla M, Perez SE, Mufson EJ, Erwin JM, Guevara EE, et al. Comparative neuropathology in aging primates: A perspective. Am J Primatol. 2021; 83:e23299. https://doi.org/10.1002/ajp.23299 [PubMed]
- 26. Phillips KA, Lopez M, Bartling-John E, Meredith R, Buteau A, Alvarez A, Ross CN. Serum biomarkers associated with aging and neurodegeneration in common marmosets (Callithrix jacchus). Neurosci Lett. 2024; 819:137569. https://doi.org/10.1016/j.neulet.2023.137569 [PubMed]
- 27. Leuner B, Kozorovitskiy Y, Gross CG, Gould E. Diminished adult neurogenesis in the marmoset brain precedes old age. Proc Natl Acad Sci U S A. 2007; 104:17169–73. https://doi.org/10.1073/pnas.0708228104 [PubMed]
- 28. Glavis-Bloom C, Vanderlip CR, Reynolds JH. Age-Related Learning and Working Memory Impairment in the Common Marmoset. J Neurosci. 2022; 42:8870–80. https://doi.org/10.1523/JNEUROSCI.0985-22.2022 [PubMed]
- 29. Glavis-Bloom C, Vanderlip CR, Weiser Novak S, Kuwajima M, Kirk L, Harris KM, Manor U, Reynolds JH. Violation of the ultrastructural size principle in the dorsolateral prefrontal cortex underlies working memory impairment in the aged common marmoset (Callithrix jacchus). Front Aging Neurosci. 2023; 15:1146245. https://doi.org/10.3389/fnagi.2023.1146245 [PubMed]
- 30. Freire-Cobo C, Rothwell ES, Varghese M, Edwards M, Janssen WGM, Lacreuse A, Hof PR. Neuronal vulnerability to brain aging and neurodegeneration in cognitively impaired marmoset monkeys (Callithrix jacchus). Neurobiol Aging. 2023; 123:49–62. https://doi.org/10.1016/j.neurobiolaging.2022.12.001 [PubMed]
- 31. Vanderlip CR, Asch PA, Reynolds JH, Glavis-Bloom C. Domain-specific cognitive impairment reflects prefrontal dysfunction in aged common marmosets. eNeuro. Society for Neuroscience. 2023. Available from: https://www.eneuro.org/content/early/2023/08/07/ENEURO.0187-23.2023.
- 32. Vanderlip CR, Asch PA, Glavis-Bloom C. The Common Marmoset as a Translational Model for Longitudinal Studies of Cognitive Aging and Individual Vulnerability to Decline. bioRxiv. 2024; 2024.08.22.609213. Available from: https://www.biorxiv.org/content/10.1101/2024.08.22.609213v1.
- 33. Rothwell ES, Workman KP, Wang D, Lacreuse A. Sex differences in cognitive aging: a 4-year longitudinal study in marmosets. Neurobiol Aging. 2022; 109:88–99. https://doi.org/10.1016/j.neurobiolaging.2021.09.015 [PubMed]
- 34. Joyce MKP, Ivanov TG, Krienen FM, Mitchell JF, Ma S, Inoue W, Nandy AS, Datta D, Duque A, Arellano J, Gupta R, Gonzalez-Burgos G, Lewis DA, et al. Dopamine D1 receptor expression in dlPFC inhibitory parvalbumin neurons may contribute to higher visuospatial distractibility in marmosets versus macaques. bioRxiv. 2024; 2024.06.15.599163. Available from: https://www.biorxiv.org/content/10.1101/2024.06.15.599163v2.
- 35. Kell AJE, Bokor SL, Jeon Y-N, Toosi T, Issa EB. Marmoset core visual object recognition behavior is comparable to that of macaques and humans. iScience. Elsevier. 2023; 26. Available from: https://www.cell.com/iscience/abstract/S2589-0042(22)02061-2.
- 36. Nummela SU, Jutras MJ, Wixted JT, Buffalo EA, Miller CT. Recognition Memory in Marmoset and Macaque Monkeys: A Comparison of Active Vision. J Cogn Neurosci. 2019; 31:1318–28. https://doi.org/10.1162/jocn_a_01361 [PubMed]
- 37. Beason-Held LL, Rosene DL, Killiany RJ, Moss MB. Hippocampal formation lesions produce memory impairment in the rhesus monkey. Hippocampus. 1999; 9:562–74. https://doi.org/10.1002/(SICI)1098-1063(1999)9:5<562::AID-HIPO10>3.0.CO;2-X [PubMed]
- 38. Bor D, Duncan J, Lee AC, Parr A, Owen AM. Frontal lobe involvement in spatial span: converging studies of normal and impaired function. Neuropsychologia. 2006; 44:229–37. https://doi.org/10.1016/j.neuropsychologia.2005.05.010 [PubMed]
- 39. Jeneson A, Mauldin KN, Squire LR. Intact working memory for relational information after medial temporal lobe damage. J Neurosci. 2010; 30:13624–9. https://doi.org/10.1523/JNEUROSCI.2895-10.2010 [PubMed]
- 40. Small SA, Schobel SA, Buxton RB, Witter MP, Barnes CA. A pathophysiological framework of hippocampal dysfunction in ageing and disease. Nat Rev Neurosci. 2011; 12:585–601. https://doi.org/10.1038/nrn3085 [PubMed]
- 41. Salmon DP, Granholm E, McCullough D, Butters N, Grant I. Recognition memory span in mildly and moderately demented patients with Alzheimer's disease. J Clin Exp Neuropsychol. 1989; 11:429–43. https://doi.org/10.1080/01688638908400904 [PubMed]
- 42. Moss MB, Killiany RJ, Lai ZC, Rosene DL, Herndon JG. Recognition memory span in rhesus monkeys of advanced age. Neurobiol Aging. 1997; 18:13–9. https://doi.org/10.1016/s0197-4580(96)00211-4 [PubMed]
- 43. Maylor EA, Simpson EE, Secker DL, Meunier N, Andriollo-Sanchez M, Polito A, Stewart-Knox B, McConville C, O'Connor JM, Coudray C. Effects of zinc supplementation on cognitive function in healthy middle-aged and older adults: the ZENITH study. Br J Nutr. 2006; 96:752–60. [PubMed]
- 44. Belham FS, Satler C, Garcia A, Tomaz C, Gasbarri A, Rego A, Tavares MC. Age-related differences in cortical activity during a visuo-spatial working memory task with facial stimuli. PLoS One. 2013; 8:e75778. https://doi.org/10.1371/journal.pone.0075778 [PubMed]
- 45. Mazurek A, Bhoopathy RM, Read JC, Gallagher P, Smulders TV. Effects of age on a real-world What-Where-When memory task. Front Aging Neurosci. 2015; 7:74. https://doi.org/10.3389/fnagi.2015.00074 [PubMed]
- 46. Satler C, Belham FS, Garcia A, Tomaz C, Tavares MC. Computerized spatial delayed recognition span task: a specific tool to assess visuospatial working memory. Front Aging Neurosci. 2015; 7:53. https://doi.org/10.3389/fnagi.2015.00053 [PubMed]
- 47. Killiany RJ, Moss MB, Rosene DL, Herndon J. Recognition memory function in early senescent rhesus monkeys. Psychobiology. 2000; 28: 45–56. https://doi.org/10.3758/BF03330628
- 48. Moore TL, Bowley B, Shultz P, Calderazzo S, Shobin E, Killiany RJ, Rosene DL, Moss MB. Chronic curcumin treatment improves spatial working memory but not recognition memory in middle-aged rhesus monkeys. Geroscience. 2017; 39:571–84. https://doi.org/10.1007/s11357-017-9998-2 [PubMed]
- 49. Romberg C, Horner AE, Bussey TJ, Saksida LM. A touch screen-automated cognitive test battery reveals impaired attention, memory abnormalities, and increased response inhibition in the TgCRND8 mouse model of Alzheimer's disease. Neurobiol Aging. 2013; 34:731–44. https://doi.org/10.1016/j.neurobiolaging.2012.08.006 [PubMed]
- 50. Bopp KL, Verhaeghen P. Aging and n-Back Performance: A Meta-Analysis. J Gerontol Ser B. 2018. Available from: https://academic.oup.com/psychsocgerontology/advance-article/doi/10.1093/geronb/gby024/4944520.
- 51. De Boeck P, Jeon M. An Overview of Models for Response Times and Processes in Cognitive Tests. Front Psychol. 2019; 10:102. https://doi.org/10.3389/fpsyg.2019.00102 [PubMed]
- 52. Gazzaley A, Cooney JW, Rissman J, D'Esposito M. Top-down suppression deficit underlies working memory impairment in normal aging. Nat Neurosci. 2005; 8:1298–300. https://doi.org/10.1038/nn1543 [PubMed]
- 53. Upright NA, Baxter MG. Prefrontal cortex and cognitive aging in macaque monkeys. Am J Primatol. 2021; 83:e23250. https://doi.org/10.1002/ajp.23250 [PubMed]
- 54. Shobin E, Bowley MP, Estrada LI, Heyworth NC, Orczykowski ME, Eldridge SA, Calderazzo SM, Mortazavi F, Moore TL, Rosene DL. Microglia activation and phagocytosis: relationship with aging and cognitive impairment in the rhesus monkey. Geroscience. 2017; 39:199–220. https://doi.org/10.1007/s11357-017-9965-y [PubMed]
- 55. Moore TL, Medalla M, Ibañez S, Wimmer K, Mojica CA, Killiany RJ, Moss MB, Luebke JI, Rosene DL. Neuronal properties of pyramidal cells in lateral prefrontal cortex of the aging rhesus monkey brain are associated with performance deficits on spatial working memory but not executive function. Geroscience. 2023; 45:1317–42. https://doi.org/10.1007/s11357-023-00798-2 [PubMed]
- 56. Rapp PR, Amaral DG. Evidence for task-dependent memory dysfunction in the aged monkey. J Neurosci. 1989; 9:3568–76. https://doi.org/10.1523/JNEUROSCI.09-10-03568.1989 [PubMed]
- 57. Hara Y, Park CS, Janssen WG, Roberts MT, Morrison JH, Rapp PR. Synaptic correlates of memory and menopause in the hippocampal dentate gyrus in rhesus monkeys. Neurobiol Aging. 2012; 33:421.e17–28. https://doi.org/10.1016/j.neurobiolaging.2010.09.014 [PubMed]
- 58. Comrie AE, Gray DT, Smith AC, Barnes CA. Different macaque models of cognitive aging exhibit task-dependent behavioral disparities. Behav Brain Res. 2018; 344:110–9. https://doi.org/10.1016/j.bbr.2018.02.008 [PubMed]
- 59. Gray DT, Khattab S, Meltzer J, McDermott K, Schwyhart R, Sinakevitch I, Härtig W, Barnes CA. Retrosplenial cortex microglia and perineuronal net densities are associated with memory impairment in aged rhesus macaques. Cereb Cortex. 2023; 33:4626–44. https://doi.org/10.1093/cercor/bhac366 [PubMed]
- 60. Dumitriu D, Hao J, Hara Y, Kaufmann J, Janssen WG, Lou W, Rapp PR, Morrison JH. Selective changes in thin spine density and morphology in monkey prefrontal cortex correlate with aging-related cognitive impairment. J Neurosci. 2010; 30:7507–15. https://doi.org/10.1523/JNEUROSCI.6410-09.2010 [PubMed]
- 61. MILES RC. Delayed-response learning in the marmoset and the macaque. J Comp Physiol Psychol. 1957; 50:352–5. https://doi.org/10.1037/h0040257 [PubMed]
- 62. Collins P, Wilkinson LS, Everitt BJ, Robbins TW, Roberts AC. The effect of dopamine depletion from the caudate nucleus of the common marmoset (Callithrix jacchus) on tests of prefrontal cognitive function. Behav Neurosci. 2000; 114:3–17. https://doi.org/10.1037//0735-7044.114.1.3 [PubMed]
- 63. Kaplan E. The process approach to neuropsychological assessment. Aphasiology. 1988; 2: 309–11. https://doi.org/10.1080/02687038808248930
- 64. Thomas KR, Edmonds EC, Eppig J, Salmon DP, Bondi MW, and Alzheimer’s Disease Neuroimaging Initiative. Using Neuropsychological Process Scores to Identify Subtle Cognitive Decline and Predict Progression to Mild Cognitive Impairment. J Alzheimers Dis. 2018; 64:195–204. https://doi.org/10.3233/JAD-180229 [PubMed]
- 65. Edmonds EC, McDonald CR, Marshall A, Thomas KR, Eppig J, Weigand AJ, Delano-Wood L, Galasko DR, Salmon DP, Bondi MW, and Alzheimer's Disease Neuroimaging Initiative. Early versus late MCI: Improved MCI staging using a neuropsychological approach. Alzheimers Dement. 2019; 15:699–708. https://doi.org/10.1016/j.jalz.2018.12.009 [PubMed]
- 66. Sadoun A, Rosito M, Fonta C, Girard P. Key periods of cognitive decline in a nonhuman primate model of cognitive aging, the common marmoset (Callithrix jacchus). Neurobiol Aging. 2019; 74:1–14. https://doi.org/10.1016/j.neurobiolaging.2018.10.003 [PubMed]
- 67. Bussey TJ, Padain TL, Skillings EA, Winters BD, Morton AJ, Saksida LM. The touchscreen cognitive testing method for rodents: how to get the best out of your rat. Learn Mem. 2008; 15:516–23. https://doi.org/10.1101/lm.987808 [PubMed]
- 68. Smith SM, Garcia EL, Davidson CG, Thompson JJ, Lovett SD, Ferekides N, Federico Q, Bumanglag AV, Hernandez AR, Abisambra JF, Burke SN. Paired associates learning is disrupted after unilateral parietal lobe controlled cortical impact in rats: A trial-by-trial behavioral analysis. Behav Brain Res. 2023; 437:114106. https://doi.org/10.1016/j.bbr.2022.114106 [PubMed]
- 69. Peters A, Sethares C, Luebke JI. Synapses are lost during aging in the primate prefrontal cortex. Neuroscience. 2008; 152:970–81. https://doi.org/10.1016/j.neuroscience.2007.07.014 [PubMed]
- 70. Arnsten AF, Wang MJ, Paspalas CD. Neuromodulation of thought: flexibilities and vulnerabilities in prefrontal cortical network synapses. Neuron. 2012; 76:223–39. https://doi.org/10.1016/j.neuron.2012.08.038 [PubMed]
- 71. Dickstein DL, Weaver CM, Luebke JI, Hof PR. Dendritic spine changes associated with normal aging. Neuroscience. 2013; 251:21–32. https://doi.org/10.1016/j.neuroscience.2012.09.077 [PubMed]
- 72. Hara Y, Yuk F, Puri R, Janssen WG, Rapp PR, Morrison JH. Presynaptic mitochondrial morphology in monkey prefrontal cortex correlates with working memory and is improved with estrogen treatment. Proc Natl Acad Sci U S A. 2014; 111:486–91. https://doi.org/10.1073/pnas.1311310110 [PubMed]
- 73. Galvin VC, Yang S, Lowet AS, Datta D, Duque A, Arnsten AF, Wang M. M1 receptors interacting with NMDAR enhance delay-related neuronal firing and improve working memory performance. Curr Res Neurobiol. 2021; 2:100016. https://doi.org/10.1016/j.crneur.2021.100016 [PubMed]
- 74. Arnsten AFT, Datta D, Wang M. The genie in the bottle-magnified calcium signaling in dorsolateral prefrontal cortex. Mol Psychiatry. 2021; 26:3684–700. https://doi.org/10.1038/s41380-020-00973-3 [PubMed]
- 75. Galvin VC, Yang ST, Paspalas CD, Yang Y, Jin LE, Datta D, Morozov YM, Lightbourne TC, Lowet AS, Rakic P, Arnsten AFT, Wang M. Muscarinic M1 Receptors Modulate Working Memory Performance and Activity via KCNQ Potassium Channels in the Primate Prefrontal Cortex. Neuron. 2020; 106:649–61.e4. https://doi.org/10.1016/j.neuron.2020.02.030 [PubMed]
- 76. Hara Y, Park CS, Janssen WG, Punsoni M, Rapp PR, Morrison JH. Synaptic characteristics of dentate gyrus axonal boutons and their relationships with aging, menopause, and memory in female rhesus monkeys. J Neurosci. 2011; 31:7737–44. https://doi.org/10.1523/JNEUROSCI.0822-11.2011 [PubMed]
- 77. Rodriguez-Callejas JD, Fuchs E, Perez-Cruz C. Evidence of Tau Hyperphosphorylation and Dystrophic Microglia in the Common Marmoset. Front Aging Neurosci. 2016; 8:315. https://doi.org/10.3389/fnagi.2016.00315 [PubMed]
- 78. Perez-Cruz C, Rodriguez-Callejas JD. The common marmoset as a model of neurodegeneration. Trends Neurosci. 2023; 46:394–409. https://doi.org/10.1016/j.tins.2023.02.002 [PubMed]
- 79. Roberts JA, Gilardi KVK, Lasley B, Rapp PR. Reproductive senescence predicts cognitive decline in aged female monkeys. NeuroReport. 1997; 8: 2047–51. https://doi.org/10.1097/00001756-199705260-00048 [PubMed]
- 80. Rapp PR, Morrison JH, Roberts JA. Cyclic estrogen replacement improves cognitive function in aged ovariectomized rhesus monkeys. J Neurosci. 2003; 23:5708–14. https://doi.org/10.1523/JNEUROSCI.23-13-05708.2003 [PubMed]
- 81. Beckman D, Morrison JH. Towards developing a rhesus monkey model of early Alzheimer's disease focusing on women's health. Am J Primatol. 2021; 83:e23289. https://doi.org/10.1002/ajp.23289 [PubMed]