Introduction
Musculoskeletal soft tissue injuries (MSTIs), including ligament, tendon, and muscle injuries, commonly occur during recreational or occupational activities, affecting both athletes and non-athletes. It is estimated that 47% of children and adolescents have experienced at least one physical activity-related injury [1]. However, determining the exact incidence of MSTIs in the general population remains challenging. During the 2016 Olympic Games, 8% of athletes sustained sports-related injuries [2], highlighting the significant impact of MSTIs on individuals’ well-being, work capacity, and participation in physical activities, along with the substantial economic burden associated with managing these conditions [3].
The etiology of MSTIs remains largely unclear, with both intrinsic (such as genetic factors) and extrinsic (such as physical activity and chronic overuse) factors being proposed as contributing factors [4, 5]. Inherited genetic factors may predispose individuals to an increased or decreased risk of MSTIs. In recent years, numerous studies have focused on decoding the genetic basis of MSTIs to understand the underlying molecular mechanisms [6]. To date, a multitude of genes have been implicated in MSTIs, including collagen-encoding, transforming growth factor-β (TGF-β), matrix metalloproteinase (MMP), and growth/differentiation factor genes [7].
Collagens, the predominant proteins in mammals, play a crucial role as the major structural proteins in ligaments and tendons. Type I collagen (COL1) accounts for nearly 90% of the total content in ligaments and tendons, consisting of two α1 chains and one α2 chain, providing structural and mechanical stability to biological tissues [8].
Khoschnau et al. [9] initially reported the rs1800012 polymorphism in the COL1A1 gene, which was significantly associated with a decreased risk of shoulder dislocation and cruciate ligament rupture. This study sparked further investigation into this area. Several studies have explored the role of the COL1A1 polymorphisms in MSTI susceptibility [10–19]. However, the findings have been mixed and inconclusive due to variations in ethnic backgrounds, clinical heterogeneity, and gender differences. This study was designed to synthesize the existing evidence to assess the conflicting results and elucidate the correlations between COL1A1 polymorphisms and susceptibility to MSTIs.
Materials and Methods
Literature search
A comprehensive literature search was conducted across multiple databases, including Web of Science, PubMed, EMBASE, Cochrane Library, and Wanfang. The keywords for literature search string were: (Achilles tendon OR Tendon injury OR Tendinopathy OR Achilles tendon rupture OR ACL injury OR Anterior cruciate ligament injury OR Ligament injury OR Anterior cruciate ligament tear OR ACL tear OR Tennis elbow OR Lateral epicondylitis OR Rotator cuff tear OR Musculoskeletal injury OR Muscle injury OR Soft tissue injuries OR Tendon-ligament injuries) AND (Collagen Type I Alpha I OR Collagen Type I Alpha1 OR Collagen Type1 Alpha 1 OR Collagen Type I α1 OR Collagen Type 1 α1 OR Type 1 Collagen α1 OR Type I Collagen α1 OR COL1A1) AND (Mutation OR Variant OR Variation OR Polymorphism). Literature search was conducted without any restriction on language. The reference lists of eligible studies were screened for additional relevant articles. Two authors (RG and SG) independently performed the literature search, with any discrepancies resolved by a third author.
Inclusion and exclusion criteria
Inclusion criteria comprised (i) studies examining the associations between COL1A1 polymorphisms and MSTIs; (ii) studies in which MSTI diagnosis was established through clinical evaluation, imaging, or surgery; (iii) studies in which healthy individuals without MSTIs served as controls; and (iv) studies in which detailed genotype data were available to calculate the odds ratios (ORs) and 95% confidence intervals (95% CIs). Exclusion criteria included (i) duplicate publications; (ii) reviews, conference abstracts, commentary articles, or case reports; and (iii) animal studies. In cases of overlapping data, only the most comprehensive study was included.
Evaluation of study quality
Two authors (RG and SG) independently assessed study quality using the Newcastle-Ottawa Scale (NOS), considering “selection,” “comparability,” and “outcome” criteria, with discrepancies resolved by consulting a third investigator. Studies scoring >5 points were deemed high quality.
Data extraction
Two review authors (RG and SG) independently extracted relevant information, including author details, publication year, country of origin, ethnicity, gender, study type, diagnostic methods, genotyping techniques, genotype counts, and Hardy-Weinberg equilibrium (HWE) test results [20]. Discrepancies were resolved by a third investigator.
Statistical analysis
Associations were assessed using ORs and 95% CIs. The pooled effect size was calculated under the allele (T vs. G), homozygote (TT vs. GG), heterozygote (TG vs. GG), dominant (TT + TG vs. GG), and recessive (TT vs. TG + GG) models. Heterogeneity was evaluated using Q-statistics and I2-statistics, with data pooled using fixed-effect or random-effect models based on heterogeneity levels (P > 0.10, I2 ≤ 50%). HWE in the control group was assessed using the chi-squared test. Subgroup analyses were conducted based on ethnicity and injury type. Statistical analyses were performed using RevMan 5.3 software.
Sensitivity analysis and publication bias
To assess the reliability and robustness of the pooled outcomes, we conducted a sensitivity analysis by sequentially excluding each study and recalculating the ORs and 95% CIs. Additionally, we utilized funnel plots and Egger’s and Begg’s linear regression tests to examine potential publication bias.
Trial sequential analysis
We performed trial sequential analysis (TSA) to estimate the required information size (RIS) based on a 20% relative risk reduction, a 5% overall type I error, and an 80% statistical test power [21]. TSA was conducted using TSA 0.9.5.10 Beta software.
Availability of data and materials
All data generated during this study are included in this published article.
Results
Literature identification
A total of 358 items were initially identified. After removing duplicates and reviewing titles and abstracts, 229 irrelevant articles were excluded, leaving 31 articles for full-text review. Ultimately, 16 articles [10–19, 22–27] were deemed eligible for final data analysis (Figure 1).
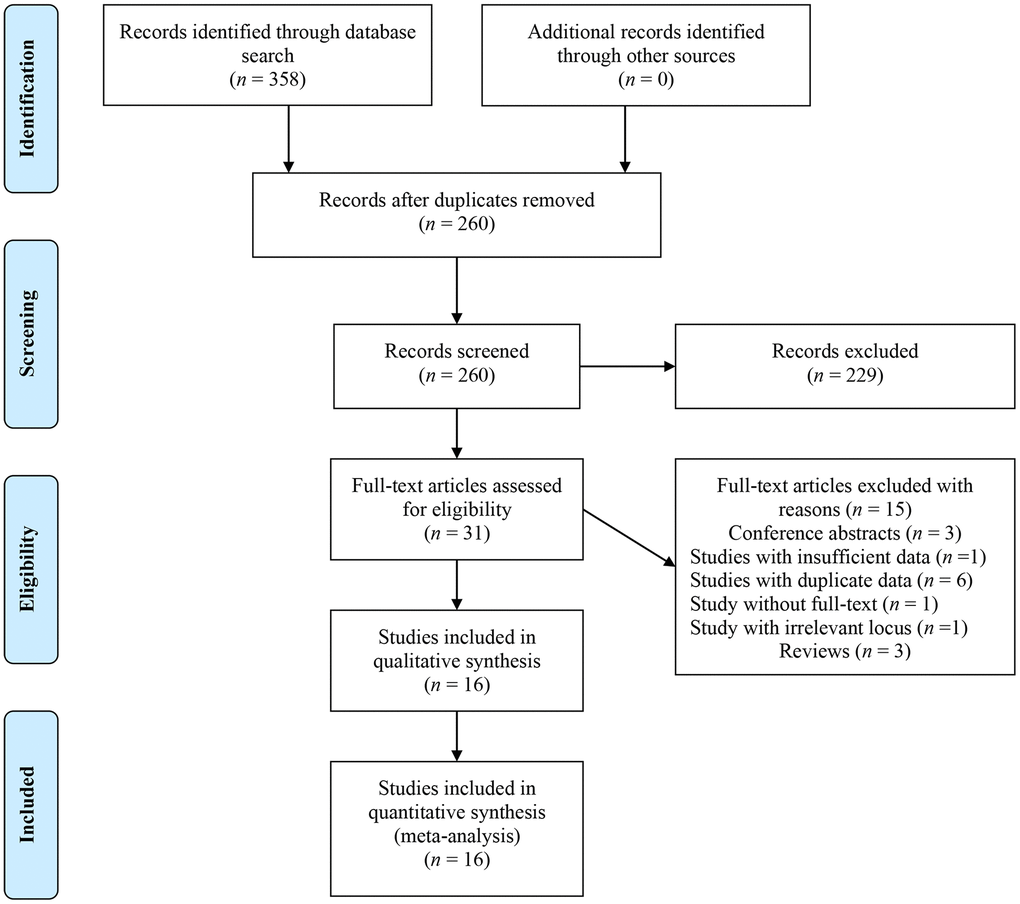
Figure 1. Flow chart of literature identification.
Main characteristics
Table 1 displays the basic characteristics of the included studies. These 16 articles [10–19, 22–27] published from 2008 to 2023 consisted of one cross-sectional study [15], three cohort studies [10, 12, 17], and remaining case-control studies [11, 13, 14, 16, 18, 19, 22–27]. Geographically, five studies [11, 14, 15, 17, 18] were conducted in Asian populations, two [19, 26] in mixed populations, and the rest in European populations. The injury types studied included anterior cruciate ligament injury (ACLI), shoulder dislocation, musculoskeletal injury, tennis elbow, Achilles tendinopathy (TEN), Achilles tendon rupture, tendinopathy, and patellar tendinopathy. Notably, two studies [15, 16] on the rs1800012 polymorphism and one [18] on the rs1107946 polymorphism deviated from HWE. All studies scored ≥6 points in quality assessment, indicating high quality (Table 2).
Table 1. Main characteristics of included studies.
| Study ID | Year | Country | Ethnicity | Gender | Study design | Genotyping methods | Diagnosis | Case | Control | HWE | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT | TG | GG | TT | TG | GG | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rs1800012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Erduran M | 2014 | Turkey | Caucasian | Both | Case-control | PCR-RFLP | TE | 2 | 32 | 69 | 7 | 35 | 61 | 0.53 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ficek K | 2013 | Poland | Caucasian | Male | Case-control | PCR | ACLI | 0 | 26 | 65 | 6 | 41 | 96 | 0.55 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gibbon A | 2020 | South Africa, UK | Caucasian | Both | Case-control | PCR | TEN, RUP, ACLI | 7 | 119 | 299 | 15 | 125 | 326 | 0.49 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haug KBF | 2018 | Norway | Caucasian | Both | Cohort | TaqMan assay | PTEN | 0 | 14 | 19 | 6 | 23 | 64 | 0.07 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Khoschnau S | 2008 | Sweden | Caucasian | Both | Case-control | PCR | CLR, SD | 2 | 99 | 257 | 12 | 83 | 230 | 0.20 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Le´znicka K | 2021 | Poland | Caucasian | Both | Case-control | TaqMan assay | MI | 8 | 13 | 32 | 9 | 9 | 43 | < 0.01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Massidda M | 2023 | Italy | Caucasian | Both | Case-control | PCR-RFLP | ACLI | 6 | 41 | 38 | 4 | 33 | 44 | 0.48 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prabhakar S | 2019 | India | Asian | Both | Case-control | PCR | ACLI | 39 | 7 | 1 | 43 | 7 | 0 | 0.60 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shukla M | 2020 | India | Asian | Both | Case-control | PCR | ACLI | 0 | 14 | 76 | 0 | 12 | 64 | 0.46 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sivertsen EA | 2019 | Norway, Finland | Caucasian | Female | Cohort | TaqMan assay | ACLI | 2 | 38 | 79 | 15 | 205 | 512 | 0.29 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stępień-Słodkowska M | 2017 | Poland | Caucasian | Male | Case-control | TaqMan assay | ACLI | 2 | 46 | 90 | 5 | 39 | 139 | 0.28 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Zhao D | 2020 | China | Asian | Both | Cross-sectional | PCR-RFLP | ACLI | 1 | 2 | 98 | 1 | 2 | 107 | < 0.01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rs1107946 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ficek K | 2013 | Poland | Caucasian | Male | Case-control | PCR | ACLI | 1 | 30 | 60 | 3 | 33 | 107 | 0.81 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gibbon A | 2020 | South Africa, UK | Caucasian | Both | Case-control | PCR | TEN, RUP, ACLI | 5 | 97 | 313 | 12 | 112 | 349 | 0.41 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lopes LR | 2023 | Brazil | Mixed | Both | Case-control | TaqMan assay | TEND | 7 | 18 | 29 | 14 | 61 | 110 | 0.18 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mirghaderi SP | 2022 | Iran | Asian | Male | Case-control | TaqMan assay | ACLI | 33 | 43 | 124 | 29 | 57 | 114 | < 0.01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Miyamoto-Mikami E | 2021 | Japan | Asian | Both | Cohort | TaqMan assay | MI | 37 | 87 | 67 | 223 | 652 | 498 | 0.70 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Perini JA | 2022 | Brazil | Mixed | Both | Case-control | TaqMan assay | ACLI | 10 | 43 | 92 | 15 | 65 | 110 | 0.23 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prabhakar S | 2019 | India | Asian | Both | Case-control | PCR | ACLI | 23 | 17 | 7 | 23 | 19 | 8 | 0.25 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sivertsen EA | 2019 | Norway, Finland | Caucasian | Female | Cohort | TaqMan assay | ACLI | 3 | 34 | 82 | 15 | 168 | 549 | 0.62 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stępień-Słodkowska M | 2017 | Poland | Caucasian | Male | Case-control | TaqMan assay | ACLI | 1 | 48 | 89 | 4 | 46 | 133 | 0.99 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: PCR: Polymerase chain reaction; PCR-RFLP: Polymerase chain reaction–restriction fragment length polymorphism; CLR: cruciate ligament rupture; SD: shoulder dislocation; ACLI: anterior cruciate ligament injury; MI: musculoskeletal injury; TE: tennis elbow; TEN: Achilles tendinopathy; TEND: tendinopathy; RUP: Achilles tendon rupture; PTEN: patellar tendinopathy; HWE: Hardy-Weinberg Equilibrium. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Quality assessment of included studies.
| Study ID | Selection | Control for important factor | Exposure | ||||||
| Adequate definition of cases | Representativeness of cases | Selection of control subjects | Definition of control subjects | Exposure assessment | Same method of ascertainment for all subjects | Non-response rate | Total | ||
| Erduran M, 2014 | ★ | ★ | ☆ | ★ | ★★ | ★ | ★ | ★ | 8 |
| Ficek K, 2013 | ★ | ☆ | ☆ | ★ | ★★ | ★ | ★ | ★ | 7 |
| Gibbon A, 2020 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Haug KBF, 2018 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Khoschnau S, 2008 | ★ | ☆ | ★ | ★ | ★☆ | ★ | ★ | ★ | 7 |
| Le´znicka K, 2021 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Lopes LR, 2023 | ★ | ☆ | ☆ | ★ | ★★ | ★ | ★ | ★ | 7 |
| Massidda M, 2023 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Mirghaderi SP, 2022 | ★ | ☆ | ☆ | ★ | ★★ | ★ | ★ | ★ | 7 |
| Miyamoto-Mikami E, 2021 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Perini JA, 2022 | ★ | ☆ | ☆ | ★ | ★★ | ★ | ★ | ★ | 7 |
| Prabhakar S, 2019 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Shukla M, 2020 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Sivertsen EA, 2019 | ★ | ☆ | ★ | ★ | ★☆ | ★ | ★ | ★ | 7 |
| Stępień-Słodkowska M, 2017 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 6 |
| Zhao D, 2020 | ★ | ☆ | ☆ | ★ | ★☆ | ★ | ★ | ★ | 7 |
Meta-analyses and subgroup analyses
Association of the rs1800012 polymorphism and MSTIs
Twelve studies [10–16, 22–25, 27] analyzed the correlation between the rs1800012 polymorphism and MSTIs, involving 1643 cases and 2423 controls. Low heterogeneity was observed, and the fixed-effects model was applied. The combined data showed that TT genotype carriers had a significantly decreased risk of MSTIs (TT vs. GG, OR = 0.53, 95% CI 0.35–0.82, P = 0.004; TT vs. TG + GG, OR = 0.54, 95% CI 0.36–0.80, P = 0.002; Figure 2).
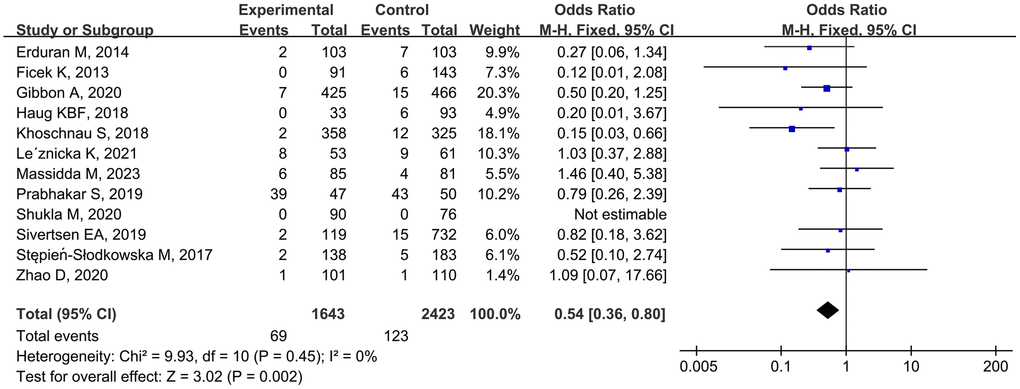
Figure 2. Forest plot of rs1800012 polymorphism and musculoskeletal soft tissue injuries (TT vs. TG+GG).
Stratified analyses by injury type revealed that TG-genotype carriers tended to have an increased risk of ACLI (TG vs. GG, OR = 1.25, 95% CI 1.02–1.55, P = 0.03). Ethnicity-based analysis indicated that TT genotype individuals had a reduced risk of MSTIs among Caucasians (TT vs. GG, OR = 0.53, 95% CI 0.34–0.83, P = 0.005; TT vs. TG + GG, OR = 0.50, 95% CI 0.32–0.78, P = 0.002; Table 3) but not among Asians.
Table 3. Association of COL1A1 gene polymorphisms and musculoskeletal soft tissue injuries.
| Polymorphism/ genetic models | Association | No. of cohorts | Heterogeneity | Statistical model | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR | 95% CI | P | I2 (%) | P | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rs1800012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Overall | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 0.98 | 0.86–1.12 | 0.79 | 12 | 2 | 0.43 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 0.53 | 0.35–0.82 | 0.004 | 12 | 9 | 0.36 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 1.16 | 0.99–1.35 | 0.07 | 12 | 0 | 0.54 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 1.05 | 0.90–1.22 | 0.53 | 12 | 0 | 0.56 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 0.54 | 0.36–0.80 | 0.002 | 12 | 0 | 0.45 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ACLI | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 1.09 | 0.91–1.30 | 0.34 | 8 | 0 | 0.61 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 0.66 | 0.34–1.26 | 0.20 | 8 | 0 | 0.63 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 1.25 | 1.02–1.55 | 0.03 | 8 | 0 | 0.72 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 1.19 | 0.97–1.46 | 0.10 | 8 | 0 | 0.65 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 0.88 | 0.73–1.06 | 0.18 | 8 | 0 | 0.64 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Caucasian | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 0.99 | 0.87–1.13 | 0.84 | 9 | 26 | 0.21 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 0.53 | 0.34–0.83 | 0.005 | 9 | 25 | 0.22 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 1.17 | 1.00–1.36 | 0.05 | 9 | 13 | 0.33 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 1.05 | 0.91–1.23 | 0.50 | 9 | 12 | 0.33 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 0.50 | 0.32–0.78 | 0.002 | 9 | 15 | 0.31 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asian | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 0.90 | 0.51–1.61 | 0.73 | 3 | 0 | 0.85 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 0.60 | 0.08–4.58 | 0.63 | 3 | 0 | 0.55 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 0.94 | 0.44–1.98 | 0.86 | 3 | 0 | 0.82 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 0.93 | 0.45–1.92 | 0.85 | 3 | 0 | 0.78 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 0.83 | 0.30–2.31 | 0.72 | 3 | 0 | 0.84 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rs1107946 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Overall | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 1.05 | 0.94–1.18 | 0.40 | 9 | 0 | 0.45 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 1.05 | 0.80–1.37 | 0.73 | 9 | 0 | 0.67 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 1.05 | 0.90–1.22 | 0.57 | 9 | 26 | 0.22 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 1.05 | 0.91–1.21 | 0.51 | 9 | 13 | 0.33 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 1.09 | 0.85–1.40 | 0.50 | 9 | 0 | 0.65 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ACLI | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 1.04 | 0.89–1.21 | 0.61 | 7 | 0 | 0.45 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 0.92 | 0.63–1.37 | 0.69 | 7 | 0 | 0.88 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 1.07 | 0.89–1.30 | 0.47 | 7 | 42 | 0.11 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 1.06 | 0.88–1.27 | 0.53 | 7 | 28 | 0.21 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 0.99 | 0.69–1.42 | 0.96 | 7 | 0 | 0.83 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Caucasian | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 1.09 | 0.91–1.31 | 0.36 | 4 | 31 | 0.22 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 0.63 | 0.30–1.30 | 0.21 | 4 | 0 | 0.59 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 1.22 | 0.99–1.50 | 0.07 | 4 | 30 | 0.23 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 1.17 | 0.95–1.43 | 0.14 | 4 | 35 | 0.20 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 0.60 | 0.29–1.24 | 0.17 | 4 | 0 | 0.62 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Asian | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T vs. G | 1.04 | 0.88–1.23 | 0.65 | 3 | 0 | 0.69 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. GG | 1.16 | 0.83–1.61 | 0.38 | 3 | 0 | 0.90 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TG vs. GG | 0.88 | 0.68–1.15 | 0.37 | 3 | 0 | 0.47 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT+TG vs. GG | 0.96 | 0.75–1.22 | 0.74 | 3 | 0 | 0.59 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TT vs. TG+GG | 1.20 | 0.89–1.61 | 0.22 | 3 | 0 | 0.97 | F | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: OR: odds ratios; CI: confidence interval; F: fixed-effects model; R: random-effects model; ACLI: anterior cruciate ligament injury. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Association of the rs1107946 polymorphism and MSTIs
Nine studies [11–13, 17–19, 23, 25, 26] examined the association of the rs1107946 polymorphism with MSTIs, involving 1400 cases and 3529 controls. As minimal between-study heterogeneity was detected, the fixed-effects model was used. The pooled data did not show any significant association between the rs1107946 polymorphism and MSTIs (TT vs. TG + GG, OR = 1.09, 95% CI 0.85–1.40, P = 0.50; Figure 3). Subgroup analyses by injury type and ethnicity also revealed no significant association (Table 3).
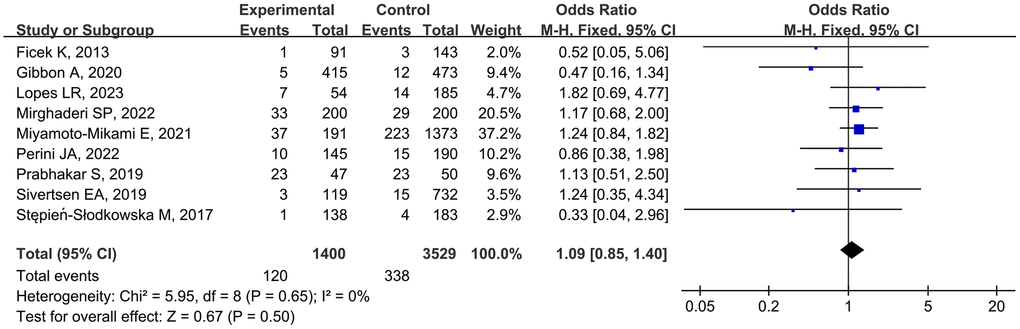
Figure 3. Forest plot of rs1107946 polymorphism and musculoskeletal soft tissue injuries (TT vs. TG+GG).
Sensitivity analysis and publication bias
Sequential removal of each study did not result in significant fluctuations in the re-pooled ORs and 95% CIs, indicating the stability and robustness of the results. Funnel plots displayed symmetrical patterns, suggesting no significant publication bias (Figure 4). Egger’s and Begg’s linear regression tests did not find significant publication bias (Supplementary Table 1).
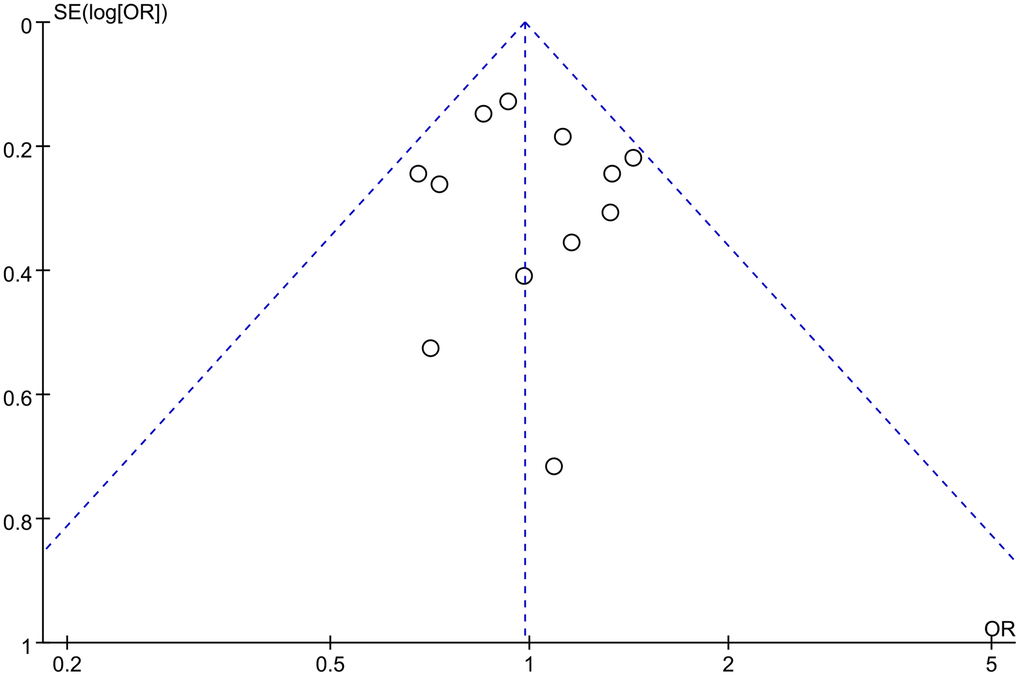
Figure 4. Funnel plot of rs1800012 polymorphism and musculoskeletal soft tissue injuries (TT vs. TG+GG).
Trial sequential analysis
For the rs1800012 polymorphism, the cumulative Z-curves did not surpass the TSA monitoring boundaries or the RIS line under five genetic models, indicating insufficient evidence and the need for further studies (Figure 5). In contrast, for the rs1107946 polymorphism, the cumulative Z-curves exceeded the RIS line but did not surpass the TSA monitoring boundaries under the allele, heterozygote, and dominant models, suggesting sufficient evidence. However, the cumulative Z-curves did not cross the TSA monitoring boundaries or the RIS line under the homozygote and recessive models, indicating the need for additional studies to obtain definitive outcomes.
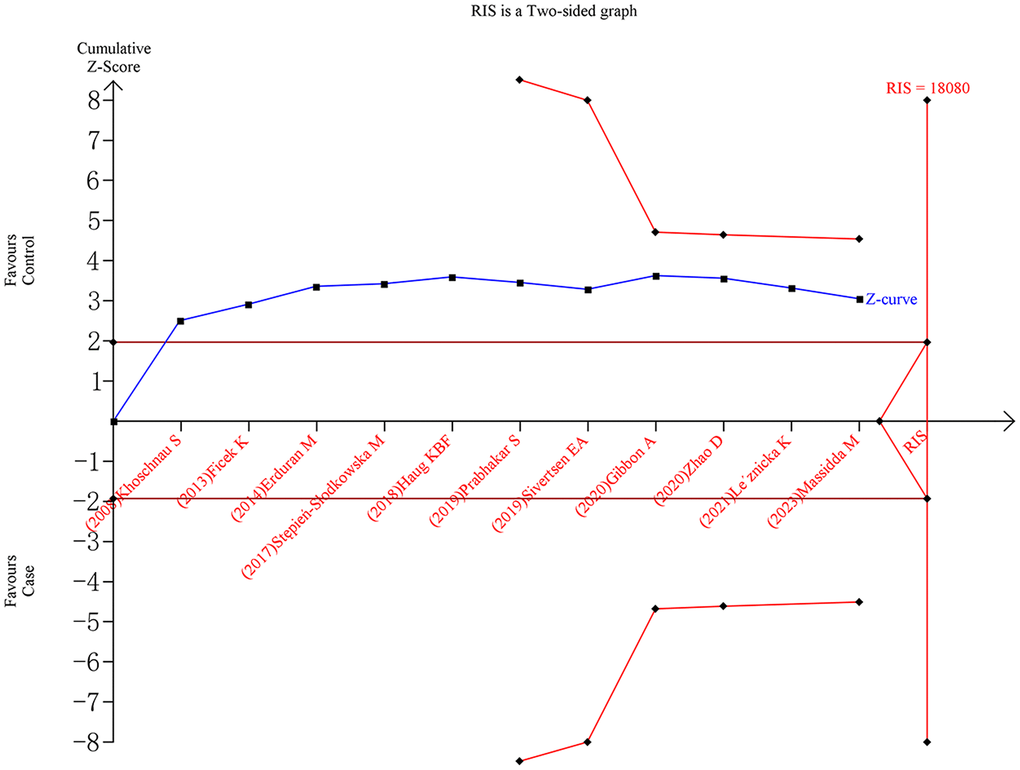
Figure 5. Trial sequential analysis of rs1800012 polymorphism and musculoskeletal soft tissue injuries (TT vs. TG+GG).
Discussion
Understanding the etiology of MSTIs is crucial for risk reduction and improved prognosis. However, the causative mechanisms of MSTIs are complex and not fully understood. Genetic factors play a significant role in MSTIs, as indicated by mounting evidence and the postulation of familial predisposition by researchers [28, 29]. Over 80 loci have been associated with MSTIs [30]. The association of COL1A1 polymorphisms with MSTIs has been extensively studied but with inconsistent findings. Therefore, this meta-analysis was conducted to synthesize existing evidence and clarify this association. Our findings support a protective role of the TT genotype of the rs1800012 polymorphism in MSTIs, particularly among Caucasians. However, the rs1107946 polymorphism showed no association with MSTIs across different ethnicities and injury types.
Tendons and ligaments are vital components of the musculoskeletal system, with COL1 playing a crucial role. COL1, also known as fibril-forming collagen, is abundant and widely used in tissue engineering due to its structural properties. Although COL1 fibers have high tensile strength and are resistant to most proteases, abnormal accumulation can lead to fibrotic diseases. COL1 is encoded by the COL1A1 and COL1A2 genes, which are respectively translated into α1 and α2 polypeptide chains in a coordinated pattern. Two α1 chains interplay with one α2 to form a triple-helical structure. The genetic aspects of the synthesis, function, and degradation of COL1 fibers and its association with diseases are under active studies [31]. In human genome, the COL1A1 gene is mapped to chromosome 17q21.33, which is 18 kb in length and consists of 52 exons.
COL1A1 gene variants are associated with various musculoskeletal disorders, including intervertebral disc degeneration [32], osteoporosis [33], osteoarthritis [34], and osteogenesis imperfecta [35]. The rs1800012 polymorphism is a guanine (G) to thymine (T) transversion within an Sp1 binding element in intron 1 (position +1245) of the COL1A1 gene [36]. Mann et al. [37] found that the T allele of rs1800012 is associated with stronger transcriptional activity and higher α1 to α2 chain ratio, which was reflected by the increased mRNA ratio of COL1A1 to COL1A2. Ireland et al. [38] observed elevated levels of COL1 and COL3 mRNAs in samples from individuals with TEN.
Rs1107946 is a frequently studied variant located in the proximal promoter of the COL1A1 gene, specifically at position-1997 in intron 1. In contrast to the rs1800012 polymorphism, the G allele of rs1107946 exhibits higher transcriptional activity than the T allele. Individuals with the TT genotype for the rs1107946 polymorphism have a significantly lower proportion of α1 chain homotrimers, potentially leading to increased resistance against injury [17]. Furthermore, rs1107946 is strongly linked with the rs1800012 polymorphism [39]. Ficek et al. [23] reported that while the rs1800012 and rs1107946 polymorphisms were not directly associated with ACLI, the COL1A1 G-T haplotype demonstrated a protective effect against ACLI among professional soccer players [23]. Additionally, Jin et al. [40] observed that COLIA1 variants regulate transcription through DNA-protein interactions. They suggested that the G-del-T haplotype (rs1107946-rs2412298-rs1800012) leads to higher transcriptional activity of COL1A1, resulting in increased production of the α1 chain, disruption of the α1 and α2 chain ratio, and generation of mature COL1 with an altered structure. Perini et al. [19] found that a separate polymorphism in the COL1A1 and COL1A2 genes was not directly associated with ACLI, but the cumulative effects of COL1A1 and COL1A2 variants might contribute to the risk of ACLI among athletes.
Wang et al. [41] conducted a meta-analysis involving 933 cases and 1381 controls to investigate the relationship between rs1800012 and tendon-ligament injuries. Compared with Wang et al.’s study, the present study has several advantages. First, new evidence was incorporated into this study, leading to an increase in the sample size and enhancement of statistical power. Moreover, while Wang et al.’s study focused solely on Caucasian participants, our study included subjects from Asian and Brazilian populations. Ethnicity stratification was performed to assess potential differences among variant ethnicities. Furthermore, the rs1107946 polymorphism, a crucial locus within the COL1A1 gene, was examined in this meta-analysis. Additionally, TSA was conducted to evaluate the adequacy of the current data to achieve positive outcomes.
Several limitations should be acknowledged in this study. First, most of the included studies had small sample sizes, which may have limited statistical power, as also indicated by TSA results. Second, the majority of research focused on Caucasians and Asians, necessitating the replication of findings in other ancestral groups to ensure generalizability. Third, all studies were observational, indicating a lower level of evidence. Fourth, due to limited data availability, adjustments for confounding factors such as gender, age, body mass index, occupation, and exercise intensity were not feasible. Fifth, the participant group in this study was heterogeneous, encompassing various injuries and occupations. Lastly, the meta-analysis only analyzed two loci within the COL1A1 gene, whereas MSTIs are complex and likely influenced by multiple genes interacting synergistically. The interaction network of COL1A1 and its related genes, including ADAMTS2, COL1A2, COL5A2, and RUNX2, is illustrated in Figure 6, suggesting a potential role of COL1A1 in the pathogenesis of MSTIs.
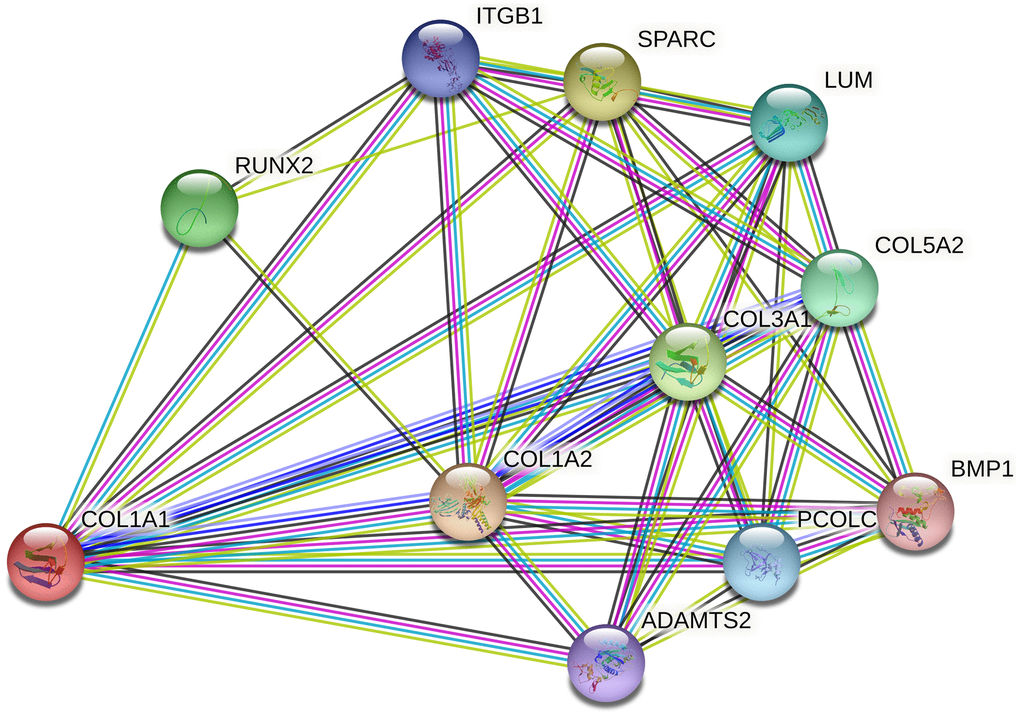
Figure 6. Network of COL1A1 with its potentially functional partners.
In conclusion, this study supports the protective effect of the TT genotype of the rs1800012 polymorphism against MSTIs, particularly among Caucasians. However, the rs1107946 polymorphism does not show an association with MSTIs. Given the limitations outlined, larger-scale prospective studies across diverse ethnic backgrounds are warranted to validate these findings and provide a more comprehensive understanding of genetic influences on MSTIs.
Supplementary Materials
Author Contributions
ZW designed this study. RG, and SG performed the literature search and selection, data extraction, and data analysis. NS, AA, HW, XF drafted the manuscript. All authors reviewed and approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This study was supported by the Graduate's Innovation Foundation of Xinjiang Medical University (XJ2022G159).
References
- 1. Räisänen AM, Kokko S, Pasanen K, Leppänen M, Rimpelä A, Villberg J, Parkkari J. Prevalence of adolescent physical activity-related injuries in sports, leisure time, and school: the National Physical Activity Behaviour Study for children and Adolescents. BMC Musculoskelet Disord. 2018; 19:58. https://doi.org/10.1186/s12891-018-1969-y [PubMed]
- 2. Soligard T, Steffen K, Palmer D, Alonso JM, Bahr R, Lopes AD, Dvorak J, Grant ME, Meeuwisse W, Mountjoy M, Pena Costa LO, Salmina N, Budgett R, Engebretsen L. Sports injury and illness incidence in the Rio de Janeiro 2016 Olympic Summer Games: A prospective study of 11274 athletes from 207 countries. Br J Sports Med. 2017; 51:1265–71. https://doi.org/10.1136/bjsports-2017-097956 [PubMed]
- 3. Herzog MM, Marshall SW, Lund JL, Pate V, Spang JT. Cost of Outpatient Arthroscopic Anterior Cruciate Ligament Reconstruction Among Commercially Insured Patients in the United States, 2005-2013. Orthop J Sports Med. 2017; 5:2325967116684776. https://doi.org/10.1177/2325967116684776 [PubMed]
- 4. Kozlovskaia M, Vlahovich N, Ashton KJ, Hughes DC. Biomedical Risk Factors of Achilles Tendinopathy in Physically Active People: a Systematic Review. Sports Med Open. 2017; 3:20. https://doi.org/10.1186/s40798-017-0087-y [PubMed]
- 5. van der Vlist AC, Breda SJ, Oei EHG, Verhaar JAN, de Vos RJ. Clinical risk factors for Achilles tendinopathy: a systematic review. Br J Sports Med. 2019; 53:1352–61. https://doi.org/10.1136/bjsports-2018-099991 [PubMed]
- 6. Rahim M, Collins M, September A. Genes and Musculoskeletal Soft-Tissue Injuries. Med Sport Sci. 2016; 61:68–91. https://doi.org/10.1159/000445243 [PubMed]
- 7. Ribbans WJ, September AV, Collins M. Tendon and Ligament Genetics: How Do They Contribute to Disease and Injury? A Narrative Review. Life (Basel). 2022; 12:663. https://doi.org/10.3390/life12050663 [PubMed]
- 8. Naomi R, Ridzuan PM, Bahari H. Current Insights into Collagen Type I. Polymers (Basel). 2021; 13:2642. https://doi.org/10.3390/polym13162642 [PubMed]
- 9. Mokone GG, Schwellnus MP, Noakes TD, Collins M. The COL5A1 gene and Achilles tendon pathology. Scand J Med Sci Sports. 2006; 16:19–26. https://doi.org/10.1111/j.1600-0838.2005.00439.x [PubMed]
- 10. Haug KBF, Visnes H, Sivertsen EA, Bahr R. Genetic variation in candidate genes and patellar tendinopathy: Prospective cohort study of 126 elite volleyball players. Transl Sports Med. 2018; 1:73–8. https://doi.org/10.1002/tsm2.13
- 11. Prabhakar S, John R, Dhillon MS, Anand A, Bammidi S. Are COL1A1 gene polymorphisms associated with anterior cruciate ligament tear in the Indian population? Results of a preliminary case-control study. Muscles Ligaments Tendons J. 2019; 8:15–22. https://doi.org/10.11138/mltj/2018.8.1.015
- 12. Sivertsen EA, Haug KBF, Kristianslund EK, Trøseid AS, Parkkari J, Lehtimäki T, Mononen N, Pasanen K, Bahr R. No Association Between Risk of Anterior Cruciate Ligament Rupture and Selected Candidate Collagen Gene Variants in Female Elite Athletes From High-Risk Team Sports. Am J Sports Med. 2019; 47:52–8. https://doi.org/10.1177/0363546518808467 [PubMed]
- 13. Gibbon A, Raleigh SM, Ribbans WJ, Posthumus M, Collins M, September AV. Functional COL1A1 variants are associated with the risk of acute musculoskeletal soft tissue injuries. J Orthop Res. 2020; 38:2290–8. https://doi.org/10.1002/jor.24621 [PubMed]
- 14. Shukla M, Gupta R, Pandey V, Tiwari PK, Amrathlal RS. COLIA1 + 1245 G > T Sp1 Binding Site Polymorphism is Not Associated with ACL Injury Risks Among Indian Athletes. Indian J Orthop. 2020; 54:647–54. https://doi.org/10.1007/s43465-020-00119-1 [PubMed]
- 15. Zhao D, Zhang Q, Lu Q, Hong C, Luo T, Duan Q, Shu S, Lv J, Zhao W. Correlations Between the Genetic Variations in the COL1A1, COL5A1, COL12A1, and β-fibrinogen Genes and Anterior Cruciate Ligament Injury in Chinese Patientsa. J Athl Train. 2020; 55:515–21. https://doi.org/10.4085/1062-6050-335-18 [PubMed]
- 16. Leźnicka K, Żyżniewska-Banaszak E, Gębska M, Machoy-Mokrzyńska A, Krajewska-Pędzik A, Maciejewska-Skrendo A, Leońska-Duniec A. Interactions between Gene Variants within the COL1A1 and COL5A1 Genes and Musculoskeletal Injuries in Physically Active Caucasian. Genes (Basel). 2021; 12:1056. https://doi.org/10.3390/genes12071056 [PubMed]
- 17. Miyamoto-Mikami E, Kumagai H, Tanisawa K, Taga Y, Hirata K, Kikuchi N, Kamiya N, Kawakami R, Midorikawa T, Kawamura T, Kakigi R, Natsume T, Zempo H, et al. Female Athletes Genetically Susceptible to Fatigue Fracture Are Resistant to Muscle Injury: Potential Role of COL1A1 Variant. Med Sci Sports Exerc. 2021; 53:1855–64. https://doi.org/10.1249/MSS.0000000000002658 [PubMed]
- 18. Mirghaderi SP, Salimi M, Kheirollahi M, Mortazavi SMJ, Akbari-Aghdam H. Anterior cruciate ligament injury and its postoperative outcomes are not associated with polymorphism in COL1A1 rs1107946 (G/T): a case-control study in the Middle East elite athletes. J Orthop Surg Res. 2022; 17:462. https://doi.org/10.1186/s13018-022-03341-9 [PubMed]
- 19. Perini JA, Lopes LR, Guimarães JAM, Goes RA, Pereira LFA, Pereira CG, Mandarino M, Villardi AM, de Sousa EB, Cossich VRA. Influence of type I collagen polymorphisms and risk of anterior cruciate ligament rupture in athletes: a case-control study. BMC Musculoskelet Disord. 2022; 23:154. https://doi.org/10.1186/s12891-022-05105-2 [PubMed]
- 20. Salanti G, Amountza G, Ntzani EE, Ioannidis JP. Hardy-Weinberg equilibrium in genetic association studies: an empirical evaluation of reporting, deviations, and power. Eur J Hum Genet. 2005; 13:840–8. https://doi.org/10.1038/sj.ejhg.5201410 [PubMed]
- 21. Gao S, Xu T, Mao C, Cheng J, Xun C, Liang W, Sheng W. Lack of Associations between Endoplasmic Reticulum Aminopeptidase 2 Gene Polymorphisms and Ankylosing Spondylitis: A Meta-analysis with Trial Sequential Analysis. Immunol Invest. 2022; 51:715–26. https://doi.org/10.1080/08820139.2020.1869253 [PubMed]
- 22. Erduran M, Altinisik J, Meric G, Ates O, Ulusal AE, Akseki D. Is Sp1 binding site polymorphism within COL1A1 gene associated with tennis elbow? Gene. 2014; 537:308–11. https://doi.org/10.1016/j.gene.2013.12.014 [PubMed]
- 23. Ficek K, Cieszczyk P, Kaczmarczyk M, Maciejewska-Karłowska A, Sawczuk M, Cholewinski J, Leonska-Duniec A, Stepien-Slodkowska M, Zarebska A, Stepto NK, Bishop DJ, Eynon N. Gene variants within the COL1A1 gene are associated with reduced anterior cruciate ligament injury in professional soccer players. J Sci Med Sport. 2013; 16:396–400. https://doi.org/10.1016/j.jsams.2012.10.004 [PubMed]
- 24. Khoschnau S, Melhus H, Jacobson A, Rahme H, Bengtsson H, Ribom E, Grundberg E, Mallmin H, Michaëlsson K. Type I collagen alpha1 Sp1 polymorphism and the risk of cruciate ligament ruptures or shoulder dislocations. Am J Sports Med. 2008; 36:2432–6. https://doi.org/10.1177/0363546508320805 [PubMed]
- 25. Stepien-Slodkowska M, Ficek K, Zietek P, Kaczmarczyk M, Lubkowska W, Szark-Eckardt M, Cieszczyk P. Is the Combination of COL1A1 Gene Polymorphisms a Marker of Injury Risk? J Sport Rehabil. 2017; 26:234–8. https://doi.org/10.1123/jsr.2015-0151 [PubMed]
- 26. Lopes LR, Guimarães JAM, Amaral MVG, Pereira CG, Wainchtock VS, Goes RA, Miranda VAR, Perini JA. Genetic Polymorphisms in COL1A2 gene and the Risk of Tendinopathy: Case-Control Study. Rev Bras Ortop (Sao Paulo). 2023; 58:478–86. https://doi.org/10.1055/s-0042-1757959 [PubMed]
- 27. Massidda M, Flore L, Scorcu M, Monteleone G, Tiloca A, Salvi M, Tocco F, Calò CM. Collagen Gene Variants and Anterior Cruciate Ligament Rupture in Italian Athletes: A Preliminary Report. Genes (Basel). 2023; 14:1418. https://doi.org/10.3390/genes14071418 [PubMed]
- 28. Flynn RK, Pedersen CL, Birmingham TB, Kirkley A, Jackowski D, Fowler PJ. The familial predisposition toward tearing the anterior cruciate ligament: a case control study. Am J Sports Med. 2005; 33:23–8. https://doi.org/10.1177/0363546504265678 [PubMed]
- 29. Ly AJ, Reddy YC, Jain NB, Du L, Atem F, Khazzam M. The role of familial predisposition in imaging-confirmed atraumatic rotator cuff tears. J Shoulder Elbow Surg. 2022; 31:819–23. https://doi.org/10.1016/j.jse.2021.10.008 [PubMed]
- 30. Rahim M, Gibbon A, Collins M, September AV. Chapter Fifteen - Genetics of musculoskeletal soft tissue injuries: Current status, challenges, and future directions. Sports, Exercise, and Nutritional Genomics. 2019; 317–39. https://doi.org/10.1016/B978-0-12-816193-7.00015-4
- 31. Trefilova VV, Shnayder NA, Petrova MM, Kaskaeva DS, Tutynina OV, Petrov KV, Popova TE, Balberova OV, Medvedev GV, Nasyrova RF. The Role of Polymorphisms in Collagen-Encoding Genes in Intervertebral Disc Degeneration. Biomolecules. 2021; 11:1279. https://doi.org/10.3390/biom11091279 [PubMed]
- 32. Xie G, Liang C, Yu H, Zhang Q. Association between polymorphisms of collagen genes and susceptibility to intervertebral disc degeneration: a meta-analysis. J Orthop Surg Res. 2021; 16:616. https://doi.org/10.1186/s13018-021-02724-8 [PubMed]
- 33. Wu J, Yu M, Zhou Y. Association of collagen type I alpha 1 +1245G/T polymorphism and osteoporosis risk in post-menopausal women: a meta-analysis. Int J Rheum Dis. 2017; 20:903–10. https://doi.org/10.1111/1756-185X.13052 [PubMed]
- 34. Lian K, Zmuda JM, Nevitt MC, Lui L, Hochberg MC, Greene D, Li J, Wang J, Lane NE. Type I collagen alpha1 Sp1 transcription factor binding site polymorphism is associated with reduced risk of hip osteoarthritis defined by severe joint space narrowing in elderly women. Arthritis Rheum. 2005; 52:1431–6. https://doi.org/10.1002/art.21011 [PubMed]
- 35. Ward LM, Lalic L, Roughley PJ, Glorieux FH. Thirty-three novel COL1A1 and COL1A2 mutations in patients with osteogenesis imperfecta types I-IV. Hum Mutat. 2001; 17:434. https://doi.org/10.1002/humu.1124 [PubMed]
- 36. Birk DE, Fitch JM, Babiarz JP, Doane KJ, Linsenmayer TF. Collagen fibrillogenesis in vitro: interaction of types I and V collagen regulates fibril diameter. J Cell Sci. 1990; 95:649–57. https://doi.org/10.1242/jcs.95.4.649 [PubMed]
- 37. Mann V, Hobson EE, Li B, Stewart TL, Grant SF, Robins SP, Aspden RM, Ralston SH. A COL1A1 Sp1 binding site polymorphism predisposes to osteoporotic fracture by affecting bone density and quality. J Clin Invest. 2001; 107:899–907. https://doi.org/10.1172/JCI10347 [PubMed]
- 38. Ireland D, Harrall R, Curry V, Holloway G, Hackney R, Hazleman B, Riley G. Multiple changes in gene expression in chronic human Achilles tendinopathy. Matrix Biol. 2001; 20:159–69. https://doi.org/10.1016/s0945-053x(01)00128-7 [PubMed]
- 39. García-Giralt N, Enjuanes A, Bustamante M, Mellibovsky L, Nogués X, Carreras R, Díez-Pérez A, Grinberg D, Balcells S. In vitro functional assay of alleles and haplotypes of two COL1A1-promoter SNPs. Bone. 2005; 36:902–8. https://doi.org/10.1016/j.bone.2004.12.012 [PubMed]
- 40. Jin H, van't Hof RJ, Albagha OM, Ralston SH. Promoter and intron 1 polymorphisms of COL1A1 interact to regulate transcription and susceptibility to osteoporosis. Hum Mol Genet. 2009; 18:2729–38. https://doi.org/10.1093/hmg/ddp205 [PubMed]
- 41. Wang C, Li H, Chen K, Wu B, Liu H. Association of polymorphisms rs1800012 in COL1A1 with sports-related tendon and ligament injuries: a meta-analysis. Oncotarget. 2017; 8:27627–34. https://doi.org/10.18632/oncotarget.15271 [PubMed]



