Introduction
Acute pancreatitis (AP) is a prevalent gastrointestinal condition that often requires hospitalization due to its sudden onset, severe nature, and quick progression. Its recurrent occurrences can ultimately result in pancreatic failure and cerebral palsy [1]. While the majority of patients experience a self-limiting condition, approximately 20% of them progress to moderate or severe acute pancreatitis, with a fatality rate ranging from 20 to 40% [2]. Recognized risk factors for acute pancreatitis include smoking, hypertriglyceridemia, and autoimmune disorders [3]. Recent Mendelian randomization (MR) studies have found causal associations between acute pancreatitis and gallstones, type 2 diabetes mellitus (T2DM), smoking, body fat percentage, body mass fat index, trunk fat percentage, percent limb fat, and plasma triglycerides [4–6]. Researchers have thoroughly examined the causes and development of AP in recent years and have identified the significant involvement of the Interleukin (IL) family in AP [7]. Thus, identifying adjustable risk factors of the IL family for AP is crucial for the prompt treatment of the condition.
IL is a cytokine that is crucial for immune control and maintaining homeostasis, generated by multiple cells [8]. Increased IL-33 expression is associated with the seriousness of AP, and excessive IL-33 production worsens the inflammatory reaction and damage to the pancreas [9]. Overexpression of IL-6 leads to a sustained and exacerbated inflammatory response, which promotes the occurrence of pancreatic injury [10]. IL-17 not only exacerbates the damage to pancreatic acinar cells but also works together with other proinflammatory mediators [11]. Serum IL-18 levels are positively associated with polymorphonuclear neutrophil elastase (PMN-E), serving as an early and sensitive indicator for predicting the severity of AP [12]. There is a lack of research investigating the causal association between IL gene family and AP utilizing MR.
MR is essentially an instrumental variable method originated from econometrics. MR can be utilized to investigate the causation hypothesis, which is a genetic instrumental variable analysis, used to evaluate the possible connection between exposure and result [13]. Since MR employs instrumental variable analysis to mimic the randomization process of causal inference in randomized controlled trials (RCT), the design is less vulnerable to reverse causal bias [14]. In this study, the MR analysis was used to investigate the causal link between IL and AP based on the Open Genome-Wide Association Study (GWAS) database, offering a novel sight on the correlation between the IL gene family and AP.
Results
IL27 and IL1RN were identified as key genes
Based on the screening criteria of |log2-fold change (FC)| > 1 and P-value < 0.05, DEGs including 994 up-regulated and 183 down-regulated were identified in AP patients’ samples compared with health controls’ samples (Supplementary Table 1). A total of 6 DE-ILRGs were obtained, including IL1B, IL1RN, IL6, IL10, IL18 and IL27 (Figure 1A). Volcano plot and bidirectional hierarchical clustering heatmap were described to the DE-ILRGs (Figures 1B, 1C), which clearly indicated the samples tend to cluster in two distinct directions.
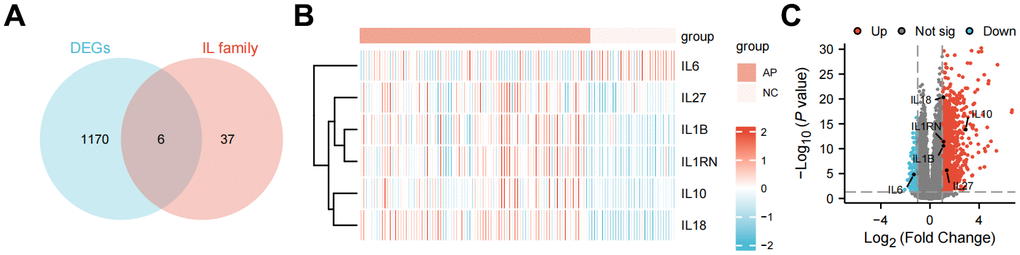
Figure 1. Differently expressed interleukin-related genes analysis for acute pancreatitis. (A) Venn diagram depicting genes associated with different expression genes only, IL family only, or both. (B) Gene expression heatmap of the differently expressed interleukin-related genes. Log2-transformed average expression in Illumina HiSeq 2500. (C) Volcano plot of different expression genes from GSE194331.
IL27 and IL1RN were causally associated with AP
Following screening, a total of 3 SNPs for IL27, 2 SNPs for IL1B, 3 SNPs for IL1RN, 1 SNP for IL10, 2 SNPs for IL18, and 4 SNPs for IL6 were identified as instrumental variables in the finn-b-K11_ACUTP dataset for UVMR analysis (Supplementary Table 2). The IVW approach results indicated that IL27 (P < 0.001, OR = 0.926) and IL1RN (P = 0.023, OR = 1.031) were significantly causally related to AP as shown in Figure 2A and Table 1.
Table 1. MR results.
| Exposure | Outcome | Method | nsnp | b | se | P-value |
| prot-b-14 | finn-b-K11_ACUTPANC | MR Egger | 3 | -0.01726 | 0.114862 | 0.9050455 |
| prot-b-14 | finn-b-K11_ACUTPANC | Weighted median | 3 | -0.07177 | 0.0575697 | 0.2125206 |
| prot-b-14 | finn-b-K11_ACUTPANC | Inverse variance weighted (multiplicative random effects) | 3 | -0.076834 | 0.0225784 | 0.0006665 |
| prot-b-14 | finn-b-K11_ACUTPANC | Simple median | 3 | -0.070402 | 0.0901814 | 0.4349939 |
| prot-a-1504 | finn-b-K11_ACUTPANC | MR Egger | 3 | -0.033993 | 0.3735759 | 0.9422313 |
| prot-a-1504 | finn-b-K11_ACUTPANC | Weighted median | 3 | 0.0323649 | 0.1146915 | 0.7777968 |
| prot-a-1504 | finn-b-K11_ACUTPANC | Inverse variance weighted (multiplicative random effects) | 3 | 0.0302148 | 0.0132439 | 0.0225236 |
| prot-a-1504 | finn-b-K11_ACUTPANC | Simple median | 3 | 0.031463 | 0.1200375 | 0.7932366 |
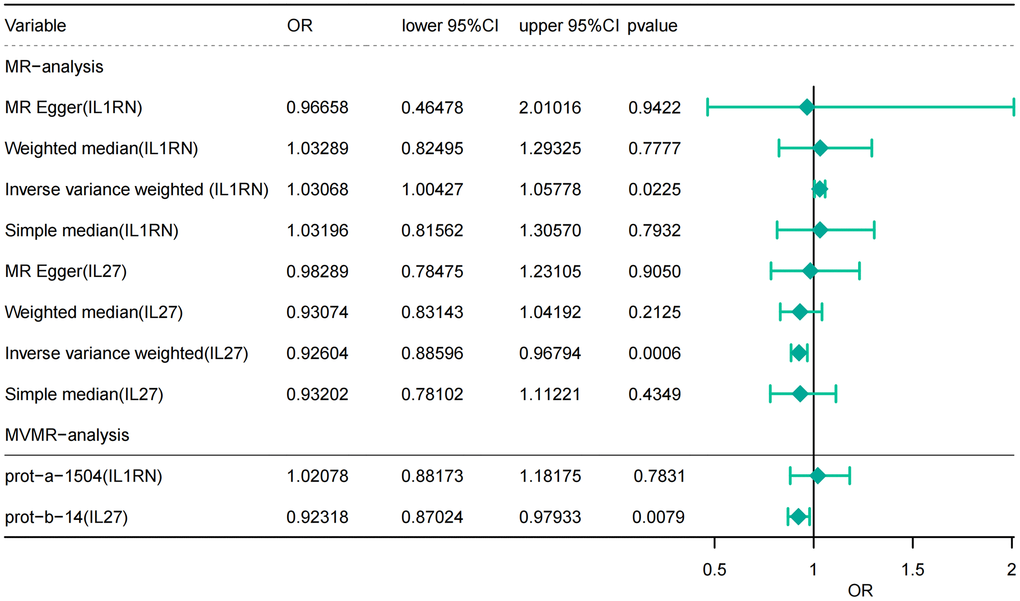
Figure 2. Causal estimates of genetically predicted IL-27 and IL-1RN using univariate and multivariate Mendelian randomization analysis. OR, odds ratios; CI, confidence interval.
IL27 as a protective factor and IL1RN as a risk factor
IL27 was identified as a protective factor and IL1RN was identified as a risk factor. The scatter plot showed a negative slope of the straight line for AP and IL27, suggesting that IL27 was a protective factor. Besides, a positive slope of the straight line for AP and IL1RN, suggesting that IL1RN was a risk factor (Figures 3A, 3B). The results of forest plots also confirmed the above results (Figures 4A, 4B). Meanwhile, the funnel plot showed that UVMR conformed to the second law of MR grouping (Figures 5A, 5B).
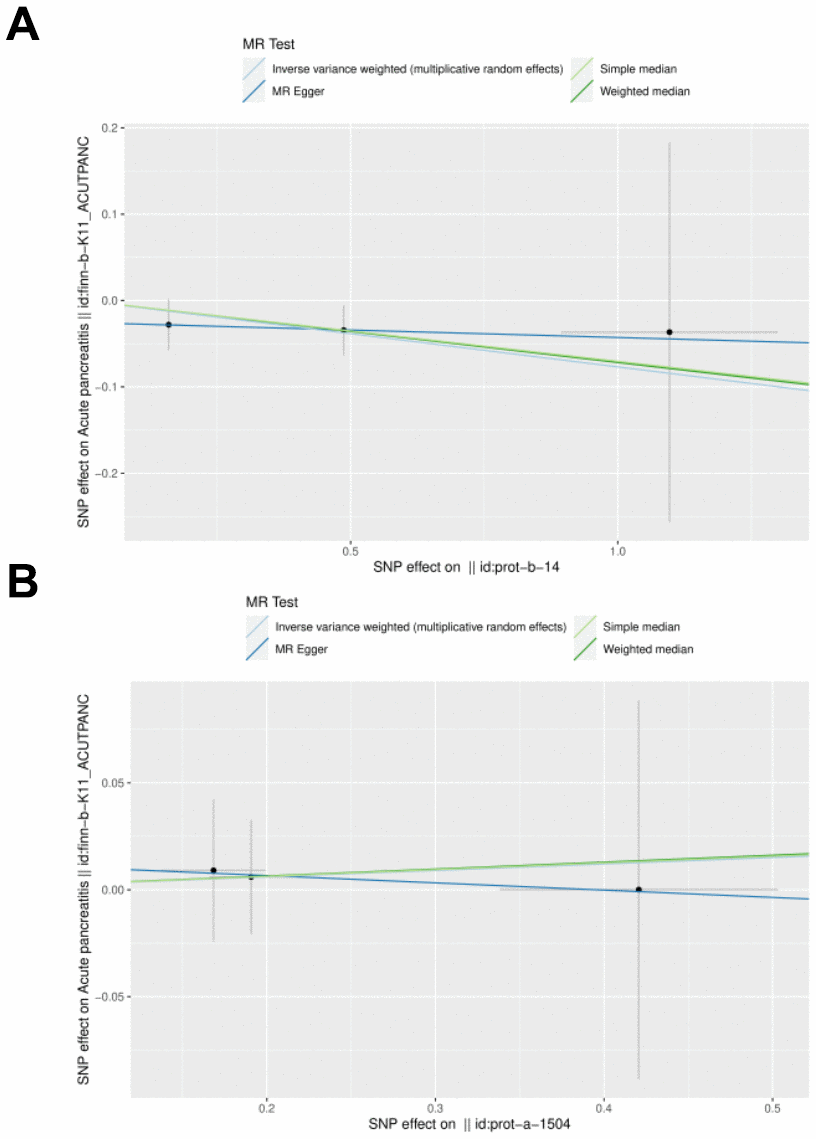
Figure 3. Protein-protein interaction network and drug prediction for differently expressed interleukin-related genes. (A) Gene related IL-27 and IL-1RN regulation network. (B) Drug regulatory network associated with IL-27 and IL-1RN.
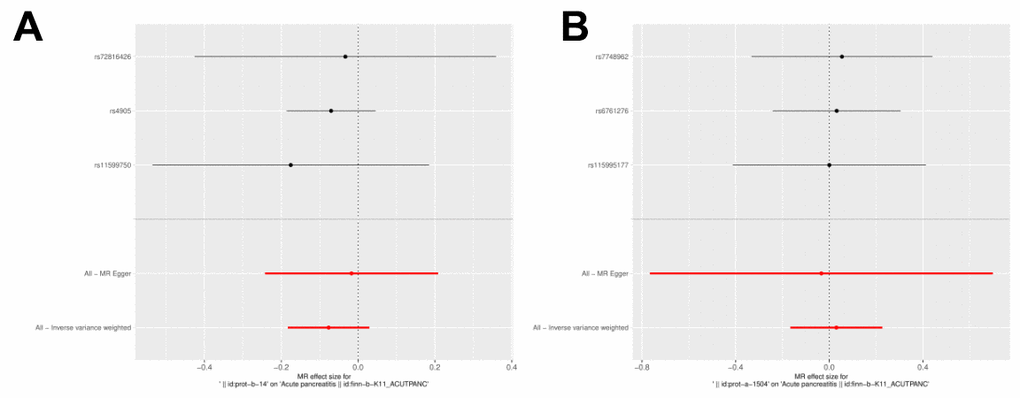
Figure 4. Scatter plot and effect estimates for four main estimators. (A) All 3 SNPs are plotted together with 95% confidence intervals representing their effect on both IL-27 (horizontal access) and on AP (vertical axis). (B) All 3 SNPs are plotted together with 95% confidence intervals representing their effect on both IL-1RN (horizontal access) and on AP (vertical axis).
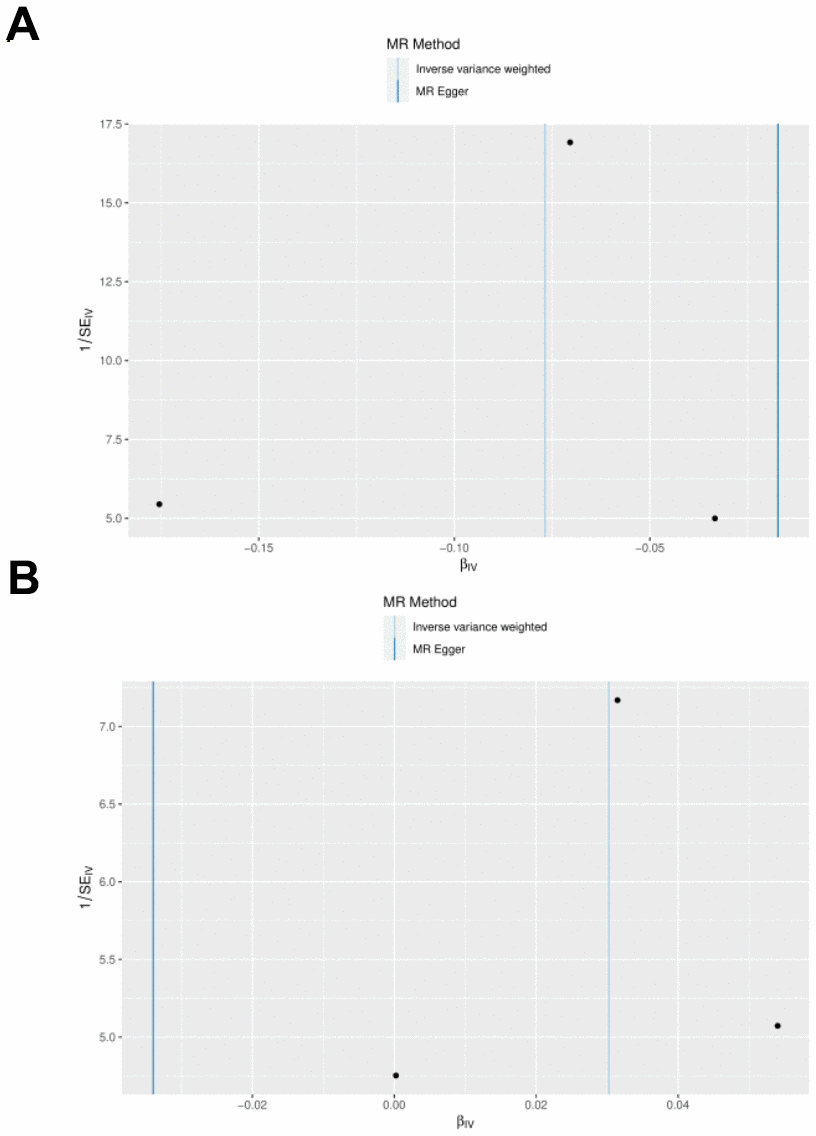
Figure 5. Forest plot of the causal effects of IL-1RN and IL-27 on acute pancreatitis. (A) MR estimate for elevated IL-27 on AP. (B) MR estimate for elevated IL-1RN on AP.
The reliability and sensitivity analysis
A sensitivity analysis was conducted to verify the reliability of the results. The Q-value for heterogeneity and the p-value for horizontal pleiotropy were both greater than 0.05, indicating that there was neither heterogeneity nor horizontal pleiotropy in the UVMR analysis (Table 2). Furthermore, there were no significant biases in the LOO analysis results, indicating that they were reliable (Figures 6A, 6B).
Table 2. MR sensitivity.
| Gene | P-value of heterogeneity | P-value of pleiotropy |
| IL1RN | 0.9826626 | 0.887608 |
| IL27 | 0.8405166 | 0.6615243 |
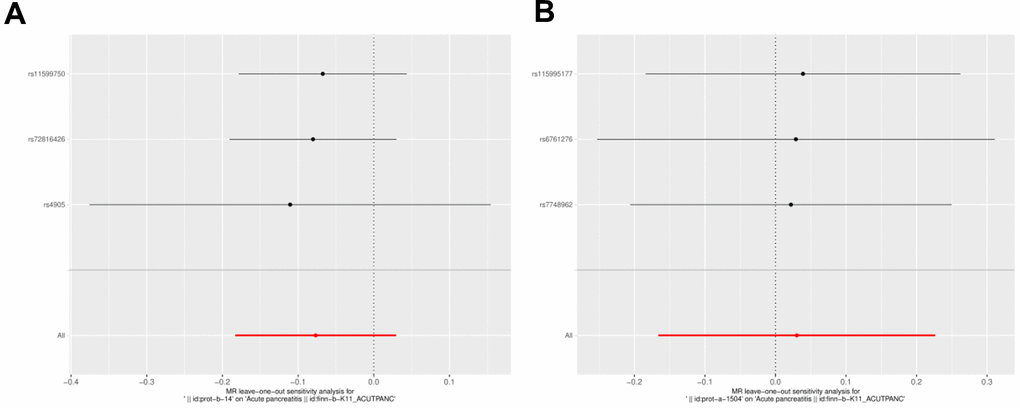
Figure 6. Funnel diagram of the random judgment of SNPs on IL-1RN and IL-27. (A) MR random judgment for elevated SNPs on IL-27. (B) MR random judgment for elevated SNPs on IL-1RN.
IL27 as a direct influence in MVMR
MVMR analysis results showed that IL1RN (P = 0.783, OR = 1.182) and IL27 (P = 0.008, OR = 0.980) attributes had not changed. However, IL1RN changed from a significant factor to a non-significant factor after correcting for the effect of IL27 (Figure 2B and Table 3).
Table 3. MVMR results.
| OR | 95%CI low | 95%CI high | P-value | |
| prot-a-1504(IL1RN) | 1.0207758 | 0.881725741 | 1.1817543 | 0.7831426 |
| prot-b-14(IL27) | 0.9231765 | 0.870239402 | 0.9793338 | 0.0079752 |
Construction of PPI network and drug prediction
The results of the PPI regulatory network showed that IL27 and IL1RN were enriched in pathways including cytokine receptors binding, cellular response to interleukin-1 and regulation of T cell activation, and interacted with IL36G and IL1R2 (Figure 7A and Supplementary Tables 3, 4). The drug prediction results showed that the gene-drug network includes 28 nodes and 26 edges. Among them, IL1RN gene predicted 12 drugs (TAE-684 and CT-GSK183, etc.) and IL27 gene predicted 14 drugs (ARQ-680 and EMS-690514, etc.) (Figure 7B).
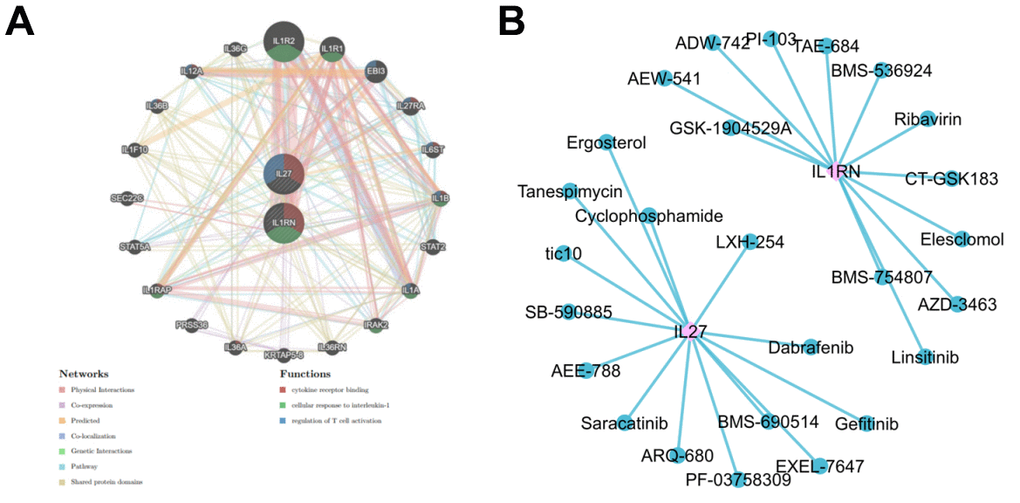
Figure 7. Forest plot of the sensitivity analysis of IL-1RN and IL-27 on acute pancreatitis. (A) MR sensitivity analysis for elevated IL-27 on AP. (B) MR sensitivity analysis for elevated IL-1RN on AP.
Discussion
This study found that IL-27 and IL-1RN were causally associated with AP. Moreover, IL-27 reduced the risk of disease, and IL-1RN was associated with increased risk of disease. AP is one of the most common acute diseases of the digestive system, which is characterized by local and systemic inflammation with a different clinical course [15]. Although the treatment of AP has evolved to be multidisciplinary, individualized and minimally invasive over the past decade, the mortality rate from severe acute pancreatitis (SAP) remains high [16]. The main reason for this is the release of a range of inflammatory response factors in the blood, leading to the occurrence of systemic inflammatory response syndrome and even multi-organ dysfunction syndrome [17]. Moreover, the reported risk factors for AP are almost based on the observational studies and case reports with small sample size [1]. This MR study provided convincing evidence, suggesting that a genetically predicted IL-1RN is causally associated with increased risk of AP whereas genetically predicted IL-27 level might exhibit protective effect on AP.
IL-27, a pleiotropic immunomodulatory cytokine, was first identified in 2002 [18]. IL-27 is expressed in multiple innate immune cells [19]. Dendritic cells located in the lamina propria of the intestine are also major producers of IL-27 after induction by Faecalis [20]. In addition to its multiple regulatory roles in innate immunity, IL-27 has been shown to have multiple functions in adaptive immunity, particularly its regulatory role in biology of T cells. With further study, the researchers found that IL-27 was mainly induced the differentiation of helper T cell subsets [21]. In previous experiments, IL-27 was highly expressed in AP, and the expression of IL-27 decreased significantly after the treatment of IFN-γ [22]. SNP of IL-27 was associated with genetic susceptibility to rheumatoid arthritis in the Chinese Han and Polish individuals, implying that IL-27 may be involved in the development of rheumatoid arthritis [23, 24]. A recent study in patients with Crohn’s disease reported higher levels of IL-27 in serum than in health controls [25]. Our study first reports that IL-27 plays a protective role in the development and progression of AP.
Earlier studies have found that IL-1 antagonism significantly attenuated pancreatic amylase release and tissue necrosis during pancreatitis [26]. IL-1 receptor antagonists encoded by IL-1RN, is member of the IL-1 gene cluster, which comes in two forms including a secreted form in macrophages and an intracellular form in epithelial cells [27]. Abnormal expression of IL-1RN has also been associated with sepsis, acute myeloid leukemia, and inflammatory bowel disease [28–30]. IL-1RN polymorphisms have been reported to be involved in the pathogenesis of type 2 diabetes mellitus [31]. Although IL-1RN expression is not necessary for the development of pancreatitis, it promotes the expansion of pancreatic damage and its associated inflammation [32]. IL-1RN appears to determine the severity of AP and the susceptibility to idiopathic AP [33]. We demonstrated for the first time from MR methods that IL-1RN can serve as a contributing factor for the development in AP.
To perform the MR analysis, three core assumptions need to be proved [34]. Evaluating the first presumption by DE-ILRGs linear regression of genetic variation and calculating the f-statistic, we found that all f-statistics for included variants were greater than 20, which suggests that the genetic variants used in our study were strongly in association with DE-ILRGs (P < 5×10-7, f-statistic > 20). Secondly, presumption requires that genetic instruments are independent of confounding factors in either DE-ILRGs or AP (Supplementary Table 2). To test this presumption, we utilized the PhenoScanner GWAS database to eliminate SNPs associated with available confounder (P < 1×10-7). Finally, we assume that genetic tools affect the AP only through DE-ILRG. The MR-Egger regression was performed to potentially test for pleiotropy and the result showed that there was no pleiotropy, suggesting that the third presumption may not be transgressed. The main advantage of this study is the MR design, which minimizes residual confounding and reduces reverse causation. Another advantage is MVMR, which can explain the relationship between IL-27 and IL-1RN. Our work may be constrained by gene pleiotropy, where a genetic variant has multiple direct correlations, thereby undermining conclusions drawn from a single route hypothesis. Our results were consistent across all MR approaches, and the Leave-One-Out (LOO) method was employed to identify potential pleiotropy.
Furthermore, we constructed a PPI network to select two genes in the AP group, which may play a key role in AP. Some studies have shown that through cellular response to IL-1, IL-1RN allelic polymorphism was strongly associated with an increased risk of T2DM, and the IL-1B allele polymorphism was associated with a decreased risk of T2DM [35]. IL-27 signaling in Tregs plays a crucial role for the proteinogenic properties of Tregs by upregulating CD39 [36]. Dabrafenib and Linsitinib have been tentatively applied to pancreatic ductal adenocarcinoma with poor prognosis after conventional cytotoxic chemotherapy [37, 38]. We found DE-ILRGs related drugs like Dabrafenib and Linsitinib from the CellMiner database, which can provide a new chance for treatment of acute pancreatitis.
Our Mendelian randomization analysis supports a possible causal link between IL and AP, addressing some information gaps. In a univariate analysis, there was a significant association between AP and IL-27 and IL1RN. IL-1RN was found to have a causal influence on AP, whereas IL-27 was found to have a preventive effect on AP. However, in multivariate analysis, after adjusting for the influence of IL 27, IL1RN transitioned from being a significant factor to an inconsequential factor. IL 27 directly contributes to AP when two exposure variables are present simultaneously. The validation of additional prospective experimental investigations and further clinical study involving a large number of samples is still required.
Materials and Methods
Data screening
We scanned the Gene Expression Omnibus database for high throughput screening data using the following parameters in order to screen the differentially expressed genes (DEGs) that were present between the health controls and the AP patients: a) the human species; b) the sample size is greater than one hundred; c) samples of blood taken from people who are experiencing AP [39]. In conclusion, high throughput screening results of GSE194331, which included 87 AP and 32 controls together with recurrence information, were collected by the use of the platform GPL16791 Illumina HiSeq 2500 [40].
Genetic instrument selection
The HUGO Gene nomenclature committee (HGNC) database was used in order to acquire interleukin related genes (ILRGs) [41]. Prot-b-14 of IL27, prot-a-1504 of IL1RN, prot-a-1495 of IL1B, prot-a-1539 of IL6, prot-a-1464 of IL10, prot-b-21 of IL18, and finn-b-K11_ACUTPANC of AP were among the datasets that were obtained from the GWAS database maintained by the Integrative Epidemiology Unit (IEU). [42]. Each of the 3,349 samples that were included in the prot-b-14 dataset contained a total of 5,270,646 single nucleotide polymorphisms (SNPs). The dataset known as prot-a-1504 contained 10,534,735 SNPs that were derived from 3,301 samples. A total of 16,380,428 SNPs were included in the finn-b-K11_ACUTPANC dataset. These SNPs were derived from 3,022 cases and 195,144 control samples.
Identification of DE-ILRGs and selection of IVs
Using the limma R package, we identified DEGs by analyzing the intersection of 87 AP samples and 32 healthy control samples. The criterion for identifying DEGs was established at a P-value of less than 0.05 and a log2-fold change (FC) greater than 1 [43]. Using the ComplexHeatmap R package (Version: 2.13.1), a hierarchical cluster heatmap that is based on Euclidean distance was constructed. This heatmap reflects the expression intensity and direction of DEGs of IL [44]. Through the process of overlapping ILRGs and the DEGs from GSE194331, differently expressed (DE)-ILRGs were produced. This allowed for the identification of IL genes that were strongly linked with disease. With the help of the TwoSampleM program (version 0.5.6), the SNPs that were found to be substantially linked with DE-ILRGs, were selected as instrumental variables (IVs) in this investigation. The criterion for identifying these SNPs was set at P < 5*10-7 [45]. Linkage disequilibrium analysis (LDA) was then used to eliminate single nucleotide polymorphisms (SNPs) (r2 = 0.001 and kb = 10000). The DE-ILRGs that had a P-value of less than 0.05 during the inverse variance weighted (IVW) analysis were determined to be the important genes. Furthermore, in order to do MR analysis, it was necessary to have three fundamental premises: (1) there was a strong and significant connection between intravenous (IV) and exposure; (2) there was no relationship between IVs and confounding factors; and (3) IVs could only influence outcomes through exposure and not through any other channels.
Multivariable (MV) MR analysis of exposure factors
MVMR analysis was carried out in order to make a discovery about the causal relationships that exist between important genes and AP at the multivariate level. The processing of data for the MVMR analysis was comparable to that of the UVMR study (p < 5*10-7, r2 = 0.001, and kb = 10000 are similar) [46]. All analyses were performed using the TwoSampleMR package.
Functional analysis of exposure factors
Through the use of the GeneMANIA website, a protein-protein interaction (PPI) network was established for key genes in order to determine whether or not there were connections between critical genes [47]. In addition, the gene-drug networks were constructed with the help of the CellMiner database [48].
Statistical analyses
In order to establish statistically significant causal associations between AP and DE-ILRGs (p < 0.05), the univariable Mendelian randomization (UVMR) study was conducted using MR-Egger, weighted median, IVW, and simple mode [49–51]. In the field of MR analysis, the IVW approach was the most significant. Based on the results of the odds ratio (OR) calculation, it was determined that the exposure factor was a risk factor if it was greater than one, while the exposure factor was a protection factor if it was greater than one. Scattered plots, forest plots, and funnel plots were utilized in the presentation of the acquired results. In addition, sensitivity analysis was carried out in order to assess the overall dependability of the UVMR data. There was no evidence of heterogeneity when the Q value was more than 0.05, as determined by the heterogeneity tests that were carried out. Horizontal pleiotropy tests were carried out by MR-Egger, and when the value of P was larger than 0.05, it indicated that there was no horizontal pleiotropy in the study. The leave-one-out (LOO) analysis was carried out by gradually removing each SNP. If there was no significant change in the influence of the remaining SNPs on the outcome variable, this might be interpreted as a hint that the results of the UVMR study were valid [52].
Supplementary Materials
Author Contributions
Conceptualization, methodology, and writing, review and editing were handled by Q.J., X.L., Z.L, and D.X; formal analysis was conducted by Q.J. and X.L.; data curation was performed by Q.J., Z.L., and D.X; writing-original draft preparation was done by Q.J. and D.X.; visualization was conducted by Q.J., X.L.; supervision was done by Q.J.; project administration was done by D.X. All authors have read and agreed to the published version of the manuscript.
Acknowledgments
We are glad to acknowledge the participants and investigators of GWAS and GEO study, and have been available at https://gwas.mrcieu.ac.uk and https://www.ncbi.nlm.nih.gov/geo/. The ILRGs catalogue has been contributed by the HUGO Gene nomenclature committee (HGNC) and has been available at https://ngdc.cncb.ac.cn/databasecommons/database/id/372. The PPI network and gene-drug networks were contributed by the GeneMANIA website and CellMiner database, and have been available at http://genemania.org/ and http://discover.nci.nih.gov/cellminer/.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Ethical Statement
No publicly available data sets of personal information were analyzed in this study. No additional ethical approval is required, as the original GWAS has already been approved by the appropriate ethical review committee.
Funding
This study was supported by grants from the National Natural Sciences Foundation of China (82170654).
References
- 1. Boxhoorn L, Voermans RP, Bouwense SA, Bruno MJ, Verdonk RC, Boermeester MA, van Santvoort HC, Besselink MG. Acute pancreatitis. Lancet. 2020; 396:726–34. https://doi.org/10.1016/S0140-6736(20)31310-6 [PubMed]
- 2. Bang JY, Wilcox CM, Arnoletti JP, Varadarajulu S. Superiority of endoscopic interventions over minimally invasive surgery for infected necrotizing pancreatitis: meta-analysis of randomized trials. Dig Endosc. 2020; 32:298–308. https://doi.org/10.1111/den.13470 [PubMed]
- 3. Mederos MA, Reber HA, Girgis MD. Acute Pancreatitis: A Review. JAMA. 2021; 325:382–90. https://doi.org/10.1001/jama.2020.20317 [PubMed]
- 4. Yuan S, Giovannucci EL, Larsson SC. Gallstone disease, diabetes, calcium, triglycerides, smoking and alcohol consumption and pancreatitis risk: Mendelian randomization study. NPJ Genom Med. 2021; 6:27. https://doi.org/10.1038/s41525-021-00189-6 [PubMed]
- 5. Tang QY, Yang Q, Yu XQ, Liu YX, Tong ZH, Li BQ, Chen YT, Yu EY, Li WQ. Association of demographic and clinical factors with risk of acute pancreatitis: An exposure-wide Mendelian randomization study. Mol Genet Genomic Med. 2023; 11:e2091. https://doi.org/10.1002/mgg3.2091 [PubMed]
- 6. Mi J, Liu Z, Jiang L, Li M, Wu X, Zhao N, Wan Z, Bai X, Feng Y. Mendelian randomization in blood metabolites identifies triglycerides and fatty acids saturation level as associated traits linked to pancreatitis risk. Front Nutr. 2022; 9:1021942. https://doi.org/10.3389/fnut.2022.1021942 [PubMed]
- 7. Yin YW, Sun QQ, Feng JQ, Hu AM, Liu HL, Wang Q. Influence of interleukin gene polymorphisms on development of acute pancreatitis: a systematic review and meta-analysis. Mol Biol Rep. 2013; 40:5931–41. https://doi.org/10.1007/s11033-013-2700-6 [PubMed]
- 8. Afonina IS, Müller C, Martin SJ, Beyaert R. Proteolytic Processing of Interleukin-1 Family Cytokines: Variations on a Common Theme. Immunity. 2015; 42:991–1004. https://doi.org/10.1016/j.immuni.2015.06.003 [PubMed]
- 9. Kotsiou OS, Gourgoulianis KI, Zarogiannis SG. IL-33/ST2 Axis in Organ Fibrosis. Front Immunol. 2018; 9:2432. https://doi.org/10.3389/fimmu.2018.02432 [PubMed]
- 10. Zheng M, Li H, Sun L, Brigstock DR, Gao R. Interleukin-6 participates in human pancreatic stellate cell activation and collagen I production via TGF-β1/Smad pathway. Cytokine. 2021; 143:155536. https://doi.org/10.1016/j.cyto.2021.155536 [PubMed]
- 11. Li G, Chen H, Liu L, Xiao P, Xie Y, Geng X, Zhang T, Zhang Y, Lu T, Tan H, Li L, Sun B. Role of Interleukin-17 in Acute Pancreatitis. Front Immunol. 2021; 12:674803. https://doi.org/10.3389/fimmu.2021.674803 [PubMed]
- 12. Wereszczynska-Siemiatkowska U, Mroczko B, Siemiatkowski A. Serum profiles of interleukin-18 in different severity forms of human acute pancreatitis. Scand J Gastroenterol. 2002; 37:1097–102. https://doi.org/10.1080/003655202320378310 [PubMed]
- 13. Burgess S, Thompson SG. Multivariable Mendelian randomization: the use of pleiotropic genetic variants to estimate causal effects. Am J Epidemiol. 2015; 181:251–60. https://doi.org/10.1093/aje/kwu283 [PubMed]
- 14. Smith GD, Ebrahim S. ‘Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol. 2003; 32:1–22. https://doi.org/10.1093/ije/dyg070 [PubMed]
- 15. Ahmed Ali U, Issa Y, Hagenaars JC, Bakker OJ, van Goor H, Nieuwenhuijs VB, Bollen TL, van Ramshorst B, Witteman BJ, Brink MA, Schaapherder AF, Dejong CH, Spanier BW, et al, and Dutch Pancreatitis Study Group. Risk of Recurrent Pancreatitis and Progression to Chronic Pancreatitis After a First Episode of Acute Pancreatitis. Clin Gastroenterol Hepatol. 2016; 14:738–46. https://doi.org/10.1016/j.cgh.2015.12.040 [PubMed]
- 16. Hajibandeh S, Jurdon R, Heaton E, Hajibandeh S, O’Reilly D. The risk of recurrent pancreatitis after first episode of acute pancreatitis in relation to etiology and severity of disease: A systematic review, meta-analysis and meta-regression analysis. J Gastroenterol Hepatol. 2023; 38:1718–33. https://doi.org/10.1111/jgh.16264 [PubMed]
- 17. Peery AF, Dellon ES, Lund J, Crockett SD, McGowan CE, Bulsiewicz WJ, Gangarosa LM, Thiny MT, Stizenberg K, Morgan DR, Ringel Y, Kim HP, DiBonaventura MD, et al. Burden of gastrointestinal disease in the United States: 2012 update. Gastroenterology. 2012; 143:1179–87.e3. https://doi.org/10.1053/j.gastro.2012.08.002 [PubMed]
- 18. Pflanz S, Timans JC, Cheung J, Rosales R, Kanzler H, Gilbert J, Hibbert L, Churakova T, Travis M, Vaisberg E, Blumenschein WM, Mattson JD, Wagner JL, et al. IL-27, a heterodimeric cytokine composed of EBI3 and p28 protein, induces proliferation of naive CD4+ T cells. Immunity. 2002; 16:779–90. https://doi.org/10.1016/s1074-7613(02)00324-2 [PubMed]
- 19. Povroznik JM, Robinson CM. IL-27 regulation of innate immunity and control of microbial growth. Future Sci OA. 2020; 6:FSO588. https://doi.org/10.2144/fsoa-2020-0032 [PubMed]
- 20. Alameddine J, Godefroy E, Papargyris L, Sarrabayrouse G, Tabiasco J, Bridonneau C, Yazdanbakhsh K, Sokol H, Altare F, Jotereau F. Faecalibacterium prausnitzii Skews Human DC to Prime IL10-Producing T Cells Through TLR2/6/JNK Signaling and IL-10, IL-27, CD39, and IDO-1 Induction. Front Immunol. 2019; 10:143. https://doi.org/10.3389/fimmu.2019.00143 [PubMed]
- 21. Yoshida H, Hamano S, Senaldi G, Covey T, Faggioni R, Mu S, Xia M, Wakeham AC, Nishina H, Potter J, Saris CJ, Mak TW. WSX-1 is required for the initiation of Th1 responses and resistance to L. major infection. Immunity. 2001; 15:569–78. https://doi.org/10.1016/s1074-7613(01)00206-0 [PubMed]
- 22. Meng H, Gong J, Fang L, Song Z, Wu F, Zhou B, Qian M. Effect of interferon-γ on NF-κB and cytokine IL-18 and IL-27 in acute pancreatitis. Bosn J Basic Med Sci. 2013; 13:114–8. https://doi.org/10.17305/bjbms.2013.2391 [PubMed]
- 23. Paradowska-Gorycka A, Raszkiewicz B, Jurkowska M, Felis-Giemza A, Romanowska-Prochnicka K, Mańczak M, Olesinska M. Association of single nucleotide polymorphisms in the IL27 gene with rheumatoid arthritis. Scand J Immunol. 2014; 80:298–305. https://doi.org/10.1111/sji.12209 [PubMed]
- 24. Yan J, Shang X, Rong X. Relationship between IL-27 gene polymorphism and susceptibility of rheumatoid arthritis in Chinese Han population. Int J Clin Exp Med. 2015; 8:6262–6. [PubMed]
- 25. Cui X, Jiao C, Wang D, Ye Z, Ma J, Tang N, Zhang H. Elevated Levels of IL-27 Are Associated with Disease Activity in Patients with Crohn’s Disease. Mediators Inflamm. 2021; 2021:5527627. https://doi.org/10.1155/2021/5527627 [PubMed]
- 26. Fink G, Yang J, Carter G, Norman J. Acute pancreatitis-induced enzyme release and necrosis are attenuated by IL-1 antagonism through an indirect mechanism. J Surg Res. 1997; 67:94–7. https://doi.org/10.1006/jsre.1996.4935 [PubMed]
- 27. Haskill S, Martin G, Van Le L, Morris J, Peace A, Bigler CF, Jaffe GJ, Hammerberg C, Sporn SA, Fong S. cDNA cloning of an intracellular form of the human interleukin 1 receptor antagonist associated with epithelium. Proc Natl Acad Sci USA. 1991; 88:3681–5. https://doi.org/10.1073/pnas.88.9.3681 [PubMed]
- 28. Granowitz EV, Santos AA, Poutsiaka DD, Cannon JG, Wilmore DW, Wolff SM, Dinarello CA. Production of interleukin-1-receptor antagonist during experimental endotoxaemia. Lancet. 1991; 338:1423–4. https://doi.org/10.1016/0140-6736(91)92725-h [PubMed]
- 29. Pasic I, Ren AH, Nampoothiri RV, Prassas I, Lipton JH, Mattsson J, Diamandis EP, Michelis FV. Multiplex proteomics using proximity extension assay for the identification of protein biomarkers predictive of acute graft-vs.-host disease in allogeneic hematopoietic cell transplantation. Clin Chem Lab Med. 2023; 61:1005–14. https://doi.org/10.1515/cclm-2022-0916 [PubMed]
- 30. De Ciucis CG, Fruscione F, De Paolis L, Mecocci S, Zinellu S, Guardone L, Franzoni G, Cappelli K, Razzuoli E. Toll-like Receptors and Cytokine Modulation by Goat Milk Extracellular Vesicles in a Model of Intestinal Inflammation. Int J Mol Sci. 2023; 24:11096. https://doi.org/10.3390/ijms241311096 [PubMed]
- 31. Masters SL, Dunne A, Subramanian SL, Hull RL, Tannahill GM, Sharp FA, Becker C, Franchi L, Yoshihara E, Chen Z, Mullooly N, Mielke LA, Harris J, et al. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nat Immunol. 2010; 11:897–904. https://doi.org/10.1038/ni.1935 [PubMed]
- 32. Norman JG, Fink G, Franz M, Guffey J, Carter G, Davison B, Sexton C, Glaccum M. Active interleukin-1 receptor required for maximal progression of acute pancreatitis. Ann Surg. 1996; 223:163–9. https://doi.org/10.1097/00000658-199602000-00008 [PubMed]
- 33. Smithies AM, Sargen K, Demaine AG, Kingsnorth AN. Investigation of the interleukin 1 gene cluster and its association with acute pancreatitis. Pancreas. 2000; 20:234–40. https://doi.org/10.1097/00006676-200004000-00003 [PubMed]
- 34. Rasooly D, Patel CJ. Conducting a Reproducible Mendelian Randomization Analysis Using the R Analytic Statistical Environment. Curr Protoc Hum Genet. 2019; 101:e82. https://doi.org/10.1002/cphg.82 [PubMed]
- 35. Jiao J, Wang Z, Guo Y, Liu J, Huang X, Ni X, Gao D, Sun L, Zhu X, Zhou Q, Yang Z, Yuan H. Association between IL-1B (-511)/IL-1RN (VNTR) polymorphisms and type 2 diabetes: a systematic review and meta-analysis. PeerJ. 2021; 9:e12384. https://doi.org/10.7717/peerj.12384 [PubMed]
- 36. Park YJ, Ryu H, Choi G, Kim BS, Hwang ES, Kim HS, Chung Y. IL-27 confers a protumorigenic activity of regulatory T cells via CD39. Proc Natl Acad Sci USA. 2019; 116:3106–11. https://doi.org/10.1073/pnas.1810254116 [PubMed]
- 37. Shin JE, An HJ, Park HS, Kim H, Shim BY. Efficacy of dabrafenib/trametinib in pancreatic ductal adenocarcinoma with BRAF NVTAP deletion: A case report. Front Oncol. 2022; 12:976450. https://doi.org/10.3389/fonc.2022.976450 [PubMed]
- 38. Jin Y, Gong S, Shang G, Hu L, Li G. Profiling of a novel circadian clock-related prognostic signature and its role in immune function and response to molecular targeted therapy in pancreatic cancer. Aging (Albany NY). 2023; 15:119–33. https://doi.org/10.18632/aging.204462 [PubMed]
- 39. Barrett T, Wilhite SE, Ledoux P, Evangelista C, Kim IF, Tomashevsky M, Marshall KA, Phillippy KH, Sherman PM, Holko M, Yefanov A, Lee H, Zhang N, et al. NCBI GEO: archive for functional genomics data sets--update. Nucleic Acids Res. 2013; 41:D991–5. https://doi.org/10.1093/nar/gks1193 [PubMed]
- 40. Nesvaderani M, Dhillon BK, Chew T, Tang B, Baghela A, Hancock RE, Eslick GD, Cox M. Gene Expression Profiling: Identification of Novel Pathways and Potential Biomarkers in Severe Acute Pancreatitis. J Am Coll Surg. 2022; 234:803–15. https://doi.org/10.1097/XCS.0000000000000115 [PubMed]
- 41. Seal RL, Braschi B, Gray K, Jones TEM, Tweedie S, Haim-Vilmovsky L, Bruford EA. Genenames.org: the HGNC resources in 2023. Nucleic Acids Res. 2023; 51:D1003–9. https://doi.org/10.1093/nar/gkac888 [PubMed]
- 42. Buniello A, MacArthur JAL, Cerezo M, Harris LW, Hayhurst J, Malangone C, McMahon A, Morales J, Mountjoy E, Sollis E, Suveges D, Vrousgou O, Whetzel PL, et al. The NHGRI-EBI GWAS Catalog of published genome-wide association studies, targeted arrays and summary statistics 2019. Nucleic Acids Res. 2019; 47:D1005–12. https://doi.org/10.1093/nar/gky1120 [PubMed]
- 43. Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015; 43:e47. https://doi.org/10.1093/nar/gkv007 [PubMed]
- 44. Gu Z, Eils R, Schlesner M. Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics. 2016; 32:2847–9. https://doi.org/10.1093/bioinformatics/btw313 [PubMed]
- 45. Hemani G, Zheng J, Elsworth B, Wade KH, Haberland V, Baird D, Laurin C, Burgess S, Bowden J, Langdon R, Tan VY, Yarmolinsky J, Shihab HA, et al. The MR-Base platform supports systematic causal inference across the human phenome. Elife. 2018; 7:e34408. https://doi.org/10.7554/eLife.34408 [PubMed]
- 46. Sanderson E. Multivariable Mendelian Randomization and Mediation. Cold Spring Harb Perspect Med. 2021; 11:a038984. https://doi.org/10.1101/cshperspect.a038984 [PubMed]
- 47. Franz M, Rodriguez H, Lopes C, Zuberi K, Montojo J, Bader GD, Morris Q. GeneMANIA update 2018. Nucleic Acids Res. 2018; 46:W60–4. https://doi.org/10.1093/nar/gky311 [PubMed]
- 48. Shankavaram UT, Varma S, Kane D, Sunshine M, Chary KK, Reinhold WC, Pommier Y, Weinstein JN. CellMiner: a relational database and query tool for the NCI-60 cancer cell lines. BMC Genomics. 2009; 10:277. https://doi.org/10.1186/1471-2164-10-277 [PubMed]
- 49. Bowden J, Davey Smith G, Burgess S. Mendelian randomization with invalid instruments: effect estimation and bias detection through Egger regression. Int J Epidemiol. 2015; 44:512–25. https://doi.org/10.1093/ije/dyv080 [PubMed]
- 50. Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent Estimation in Mendelian Randomization with Some Invalid Instruments Using a Weighted Median Estimator. Genet Epidemiol. 2016; 40:304–14. https://doi.org/10.1002/gepi.21965 [PubMed]
- 51. Burgess S, Scott RA, Timpson NJ, Davey Smith G, Thompson SG, and EPIC- InterAct Consortium. Using published data in Mendelian randomization: a blueprint for efficient identification of causal risk factors. Eur J Epidemiol. 2015; 30:543–52. https://doi.org/10.1007/s10654-015-0011-z [PubMed]
- 52. de Tommaso M, Marinazzo D, Stramaglia S. The measure of randomness by leave-one-out prediction error in the analysis of EEG after laser painful stimulation in healthy subjects and migraine patients. Clin Neurophysiol. 2005; 116:2775–82. https://doi.org/10.1016/j.clinph.2005.08.019 [PubMed]


