Introduction
The liver is a multifunctional internal organ closely related to human health and performs several vital physiological functions, such as decomposition of blood toxins, metabolic storage, lipid/glucose metabolism, secretion of serum proteins [1]. Hepatocytes, as the primary cell type within the liver, are responsible for these crucial functions. Previous single-cell transcriptome studies have carefully drawn the molecular atlas of adult liver cells [2], however, effective systematic integration of single-cell transcriptome studies on hepatocytes are urgently needed during liver regeneration [3]. Taken together, In-depth understanding of the specific mechanisms and state changes of hepatocytes during regeneration deepen our understanding of liver disease, opening up novel strategies for patients across the spectrum of liver diseases.
The liver is an exceptional organ with an innate ability to regenerate following injury. Even after the removal of up to 70% of its mass, the remaining tissue can regenerate an original-sized liver within a few months [4]. Hepatocytes represent the major cell type in the liver, and over 98% of them contribute to damage repair. During post-hepatectomy acute liver injury, newborn hepatocytes arise from the self-replication of residual hepatocytes, highlighting the proliferation and division of mature hepatocytes as the fastest and most effective method of liver regeneration [1]. Notably, Single-cell RNA sequencing (scRNA-seq) is a powerful approach to explore cell structure and characterize cell identity [5], especially identifying different cell populations and explaining the heterogeneity or correlation between cells. In conclusion, liver regeneration is a critical topic of interest in clinical scenarios and cutting-edge cellular therapies for liver disease. Advancing our understanding of the molecular basis and exploring the source of the proliferation process at the single-cell level will provide valuable insights into therapeutic targets for patients of liver disease.
The present study aims to investigate the key signatures and cell signaling pathways involved in liver regeneration. Our research team aims to identify molecules that could potentially be used to treat patients experiencing liver failure. Furthermore, the identified genes may serve as potential biomarkers to predict the likelihood of spontaneous liver regeneration in patients with damaged liver tissue, bridging the gap between bench-side research and bedside clinical application.
Materials and Methods
Data collection
The data collected in this study were mainly from the GEO database (https://www.ncbi.nlm.nih.gov/geo/), and all the sequencing data were related to PHx. Details of these data are provided in Table 1 [6–11]. The collection of partitioned genes was mainly from the references.
Table 1. Overview of the information of analyzed datasets.
| Dataset | Year | Species | Platform | Data type | Team | |
| GSE151309 | 2021 | Mus musculus | GPL24247 | Illumina NovaSeq 6000 | scRNA-seq | Chembazhi UV, et al. |
| GSE158866 | 2020 | Mus musculus | GPL24247 | Illumina NovaSeq 6000 | scRNA-seq | Chen T, et al. |
| GSE133213 | 2022 | Mus musculus | GPL18480 GPL21103 | Illumina HiSeq 1500 Illumina HiSeq 4000 | Bulk RNA-seq | Belenguer, et al. |
| GSE215423 | 2022 | Mus musculus | GPL24247 | Illumina NovaSeq 6000 | Bulk RNA-seq | Guo, et al. |
| GSE83598 | 2017 | Mus musculus | GPL7202 | Agilent-014868 Whole Mouse Genome Microarray 4x44K G4122F | Microarray | |
| GSE4528 | 2007 | Mus musculus | GPL339 | [MOE430A] Affymetrix Mouse Expression 430A Array | Microarray | |
| GSE167034 | 2022 | Mus musculus | GPL1261 | [Mouse430_2] Affymetrix Mouse Genome 430 2.0 Array | Microarray | Holland C, et al. |
| GSE158864 | 2020 | Mus musculus | GPL17021 | Illumina HiSeq 2500 | Bulk RNA-seq | Chen T, et al. |
| GSE212692 | 2023 | Mus musculus | GPL21103 GPL24247 | Illumina HiSeq 4000 (Mus musculus) Illumina NovaSeq 6000 (Mus musculus) | scRNA-seq | Li L, et al. |
High dimensional WGCNA (hdWGCNA)
HdWGCNA can be used to perform weighted gene co-expression network analysis (WGCNA) in high dimensional data such as single cell RNA-seq [12]. HdWGCNA could be utilized to construct co-expression networks in a cell type-specific manner, identifies robust modules of related genes, and provides the biological context for these modules. In this article, we used hdWGCNA to explore the function of hepatocytes after hepatectomy. And finally screening characteristic genes of a hepatocyte regeneration subgroup. The R package version of hdWGCNA used in this study is 0.1.1.9010.
Metacell algorithm
Single-cell transcriptome data have the characteristics of high background noise, low gene detection rate and sparse expression matrix. At present, genes with known functions and acquaintances can often be gathered together. Herein, obtaining metacell using the metacell algorithm [13, 14] can often better reflect the real expression of the cell. We performed the metacell analysis on the single cell sequencing data of hepatocytes to provide reliable cell data for further training and testing model for regenerative subpopulations.
Construction of machine learning model
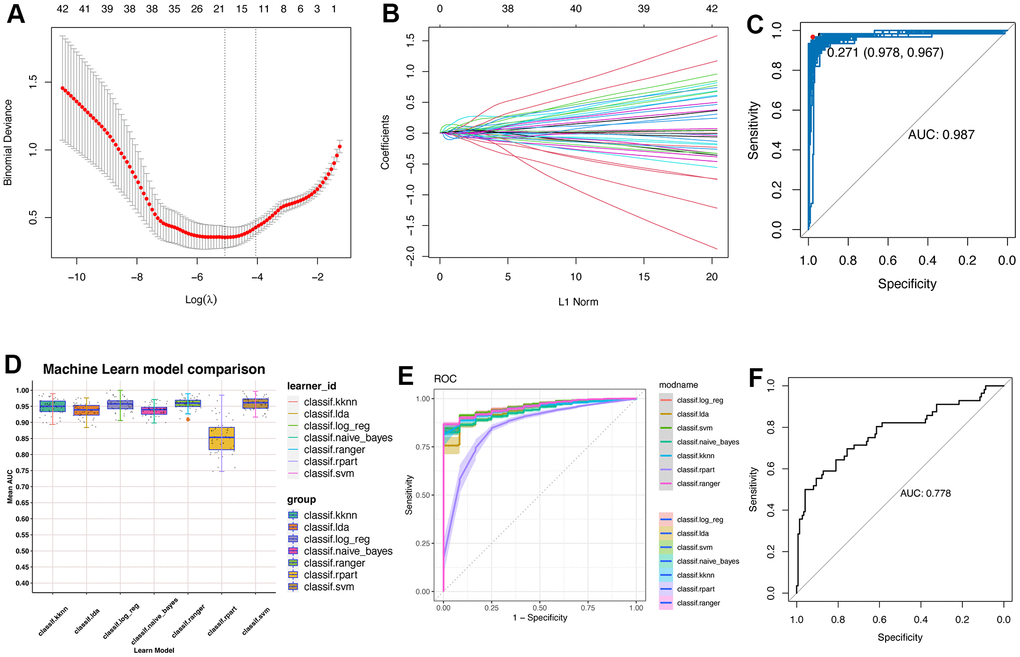
The use of single cell sequencing and key regulatory genes identified by hdWGCNA through machine learning may also help to improve understanding of liver regeneration. We use the Least Absolute Shrinkage and Selection Operator (Lasso) analysis and learn from seven machine learning algorithms to screen, get the appropriate algorithm and gene, so as to better characterize the state of hepatocytes. The seven machine learning algorithms were performed using the mlr3verse (version 0.2.7) package in R (https://CRAN.R-project.org/package=mlr3verse).
Single sample gene set enrichment analysis (ssGSEA) and gene set enrichment analysis (GSEA)
To better understand the activation of the interested gene sets, ssGSEA algorithm in gene set variation analysis (GSVA, version 1.42.0) package [15] and the GSEA pipeline, which could uncover the detailed pathways of interest [16], were utilized. And clusterProfiler package (version 1.42.0) [17] and fgsea package (version 1.42.0) [18] were adopted to better visualize the interested and enriched pathways, including Gene Ontology (GO) and KEGG signalings. We used GSEA and fgsea analyses to understand the potential role of Birc5 in liver regeneration.
Pseudo-time analysis
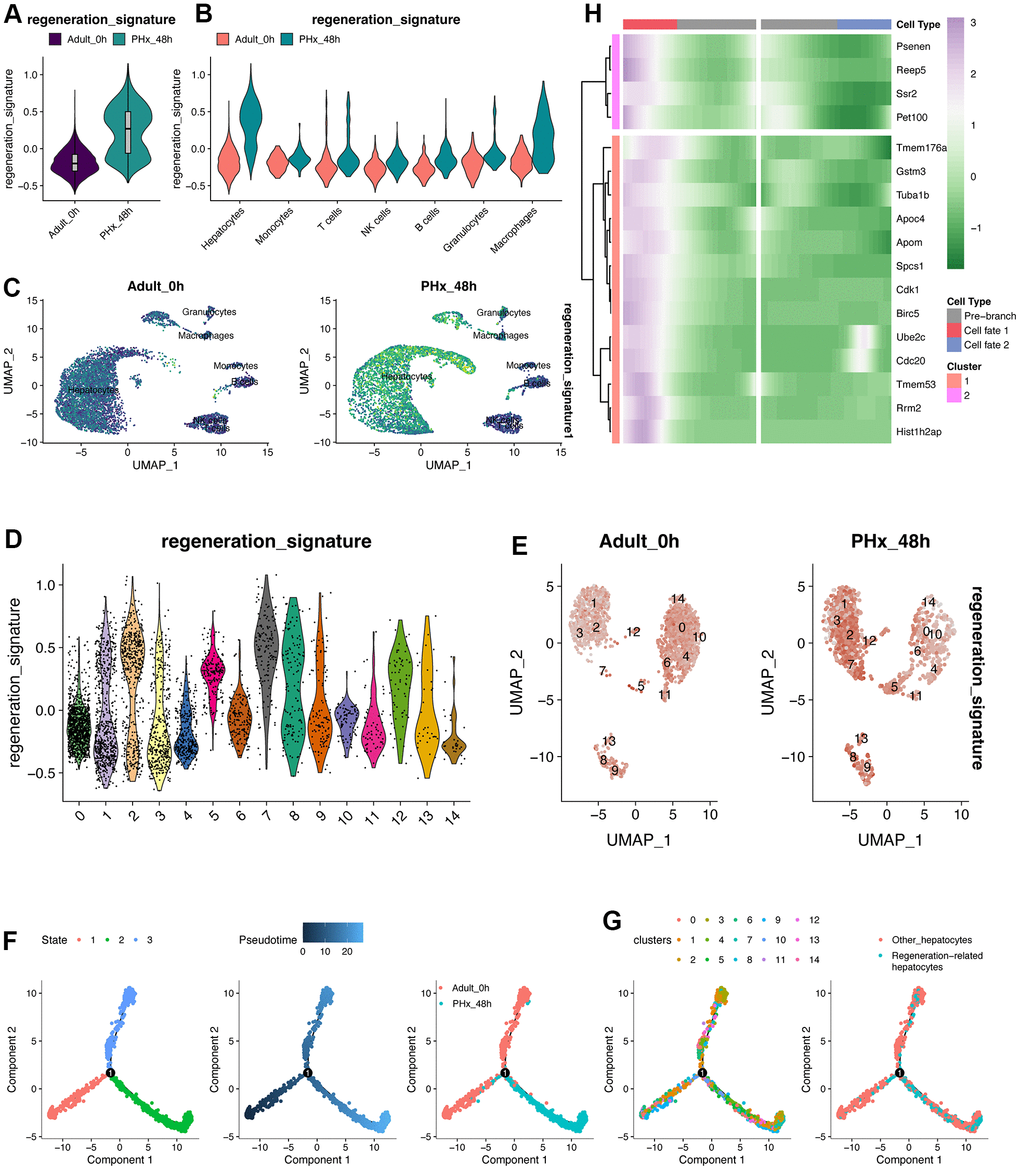
Monocle2 algorithm (version 2.22.0) [19] can clearly see the changing trend of up-regulation and down-regulation and even find hidden change patterns: We used trajectory analysis to in-depth explore the process of complete release of regeneration ability from mature hepatocytes to hepatectomy after 48h. And the changing expression of 17 key genes with time series was depicted.
Cellular communication analysis
Cellular communication plays important roles in diseases. CellChat package (version 1.0.0) makes more detailed modifications to the signaling mechanism to recognize different levels of signal changes [20]. Using cell communication analysis, we explored the interaction of regeneration-related subsets with other cell types and compared them with other hepatocyte populations, as well as the differences and connections between hepatocytes and other non-essential cells during the current regeneration process. We stochastically selected 4000 hepatocytes for running the pipeline. Other parameters were set as default.
Analysis of transcriptional factor regulatory network
Single-cell regulatory network interference and clustering (SCENIC, version 1.2.4) [21] is a gene regulatory networks (GRNs) algorithm specially developed for single cell data. SCENIC introduced the gene co-expression network deduced by transcription factor (TF) motifs, thereafter identifying high-reliability GRNs dominated by TFs. By using the SCENIC algorithm to explore the specific transcription factors of regeneration-related subpopulations compared with other subpopulations, we further revealed the possible relationship between Hmgb1 and liver regeneration.
Partial hepatectomy liver model (PHx)
6~8weeks adult mice were housed in 12h light-dark cycle with standard diet. 2/3rd partial hepatectomy (PHx) procedure was implemented according to previously protocol [22]. Briefly, an 8~10mm incision was made on the skin below mediastinum with anesthetized (2% tribromoethanol, intraperitoneal injection), left lateral lobe and the median lobe were ligated by a 4-0 silk thread, and the lobes were resected just above the knots. Following this, peritoneum and skin were then closed with 6-0 sutures, and then mice were fed with normal saline solution and allowed to recover on a pre-warmed pad. Sham mice that underwent a similar surgical intervention except for the actual resection of the liver sections.
Liver function test
Retro-orbital punctures were implemented to collect peripheral blood from mice into vacuum blood collection tubes. The upper layer of serum was isolated and centrifugation at 12000g for 8 min and stored at −80° C for further measurement. Serum levels of ALT and AST were performed at third-party inspection structure (Yuxiu Biotechnology, Shanghai, China).
Tissue embedding and immunofluorescence staining
Tissue was collected in PBS and washed for 3 times, and then split into 10~15 mm size, fixed in 10% neutral buffered formalin for 48~72 h and embedded in paraffin. 3~4 μm thick sections were cut, deparaffinized in xylene, rehydrated and antigen retrieved in Tris-EDTA buffer (pH=9.0). The sections were washed with PBST (PBS+1%Tween20) and blocked for 1h at room temperature in blocking buffer (10% normal donkey serum+3% BSA in PBS), followed by primary antibody incubation overnight at 4° C. The next day, tissues were washed with PBST for 3times and incubated with fluorescence-conjugated secondary antibodies (Abcam) for 30~45 minutes at 37° C and washed with PBST again, mounted with mounting medium containing DAPI (Abcam). The used antibody included: rabbit anti-GS (Abcam, 49873, 1:1000 dilution), mouse anti-CDH1(Biosciences, 610181, 1:50 dilution), rabbit anti-Survivin (Abconal, A1551, 1:50 dilution), rabbit anti-Ki67(Abbomax, 500-1874, 1:200 dilution), mouse anti-β-catenin (Biosciences, 610153, 1:400 dilution). For continuous labelling of DNA synthesis post-PHx, EDU solution (Yeasen, 40284ES50, 0.5~1mg/ml dilution) was intraperitoneal injection every 12h before sacrifice within the first 3 days post-PHx. Images were acquired using Leica confocal microscope (Leica+STED+3X). Images were analyzed by the Image J software.
Quantitative rt-PCR
After grinding liver tissue, Total RNA was extracted using Trizol reagent (Thermofisher, MA, USA) and reverse-transcribed into cDNA with 5×PrimeScriptTM RT Master Mix ((Takara Bio Inc., Shiga, Japan). 2×SYBR Green qRT-PCR Master Mix (Selleck, Selleck) was used to implement qRT-PCR. Target gene expression was uniformized according to the level of GAPDH expression in different samples and calculated using the 2−ΔΔCt. The primer sequences were shown in Table 2.
Table 2. Primers for all 17-genes identified by machine learning algorithms.
| Gene | Forward primer (5′-3′) | Reversed primer (5′-3′) |
| Mus-Birc5 | GAGGCTGGCTTCATCCACTG | CTTTTTGCTTGTTGTTGGTCTCC |
| Mus-Ssr2 | TGTTAGCCGTCAGTCAAGCAG | GGAGCCGACGTTGTAGATGTT |
| Mus-Gstm3 | CCCCAACTTTGACCGAAGC | GGTGTCCATAACTTGGTTCTCCA |
| Mus-Tmem53 | TGGTCCTGGCGATAGCAAC | TACCATACGTTCCACATCCCT |
| Mus-Ube2c | CTCCGCCTTCCCTGAGTCA | GGTGCGTTGTAAGGGTAGCC |
| Mus-Cdk1 | AGAAGGTACTTACGGTGTGGT | GAGAGATTTCCCGAATTGCAGT |
| Mus-Psenen | GTGTTTCGGATGTCCCTCTACC | CAGCTCCCTCTTACGCTGG |
| Mus-Spcs1 | GCCCACCCAGATGGATTACAA | GCCACGTACCCGTAGATAAACC |
| Mus-Apom | TAACTCCATGAATCAGTGCCCT | CCCGCAATAAAGTACCACAGG |
| Mus-Reep5 | GGTTCCTGCACGAGAAGAACT | GAGAGAGGCTCCATAACCGAA |
| Mus-Hmgb1 | GGCGAGCATCCTGGCTTATC | GGCTGCTTGTCATCTGCTG |
| Mus-Cdc20 | TTCGTGTTCGAGAGCGATTTG | ACCTTGGAACTAGATTTGCCAG |
| Mus-Tmem176a | GCCGGATGCTCATTGCTAAG | ATGGCCTATGTAGAGGGTTCC |
| Mud-GAPDH | AGGTCGGTGTGAACGGATTTG | TGTAGACCATGTAGTTGAGGTCA |
Statistical analysis
Statistically significant difference is estimated by using Student’s 2-tailed t test. P values of less than 0.05 were considered statistically significant. The correlation analysis was performed using the cor function in R (R version 4.1.0).
Results
Single cell RNA-sequencing analysis of liver regeneration at 48h after partial hepatectomy (PHx)
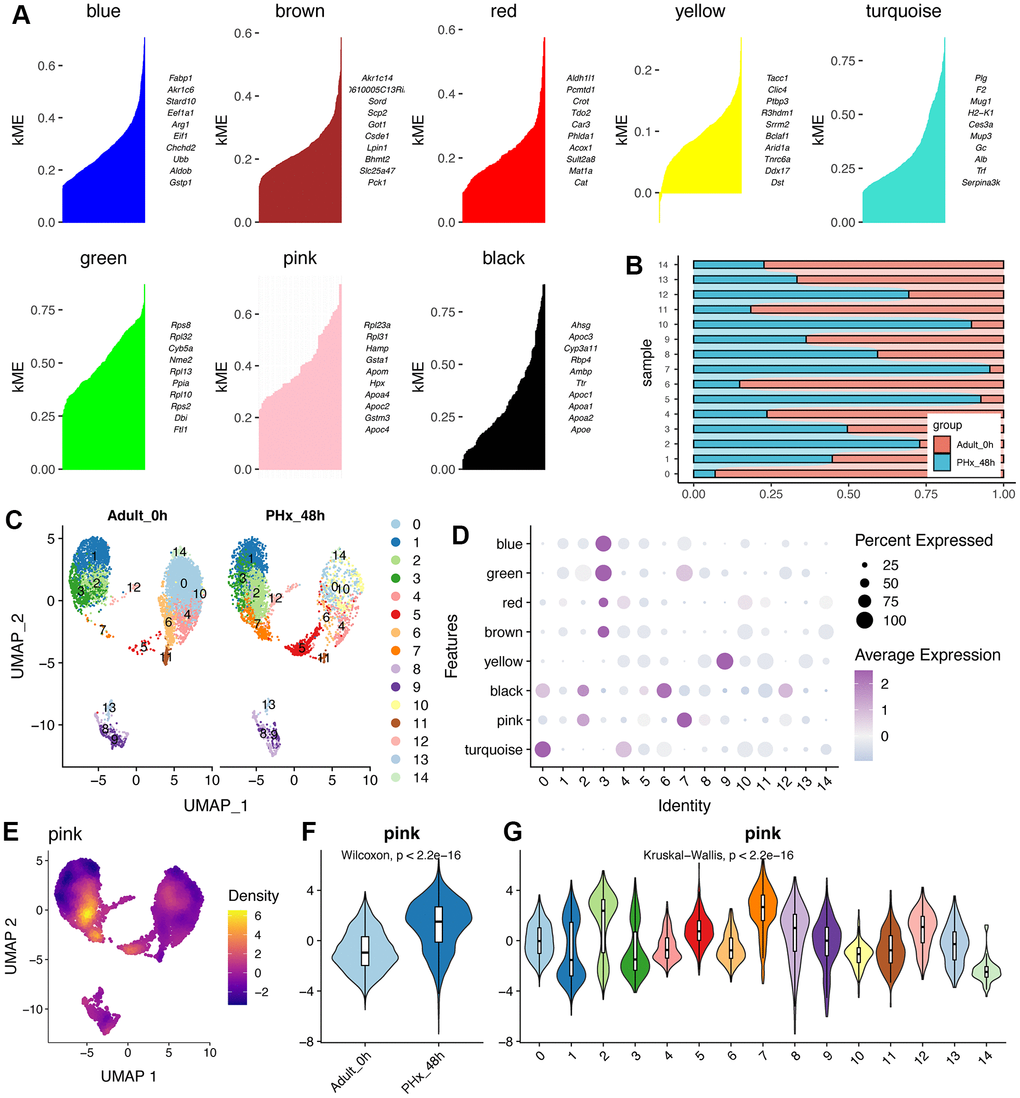
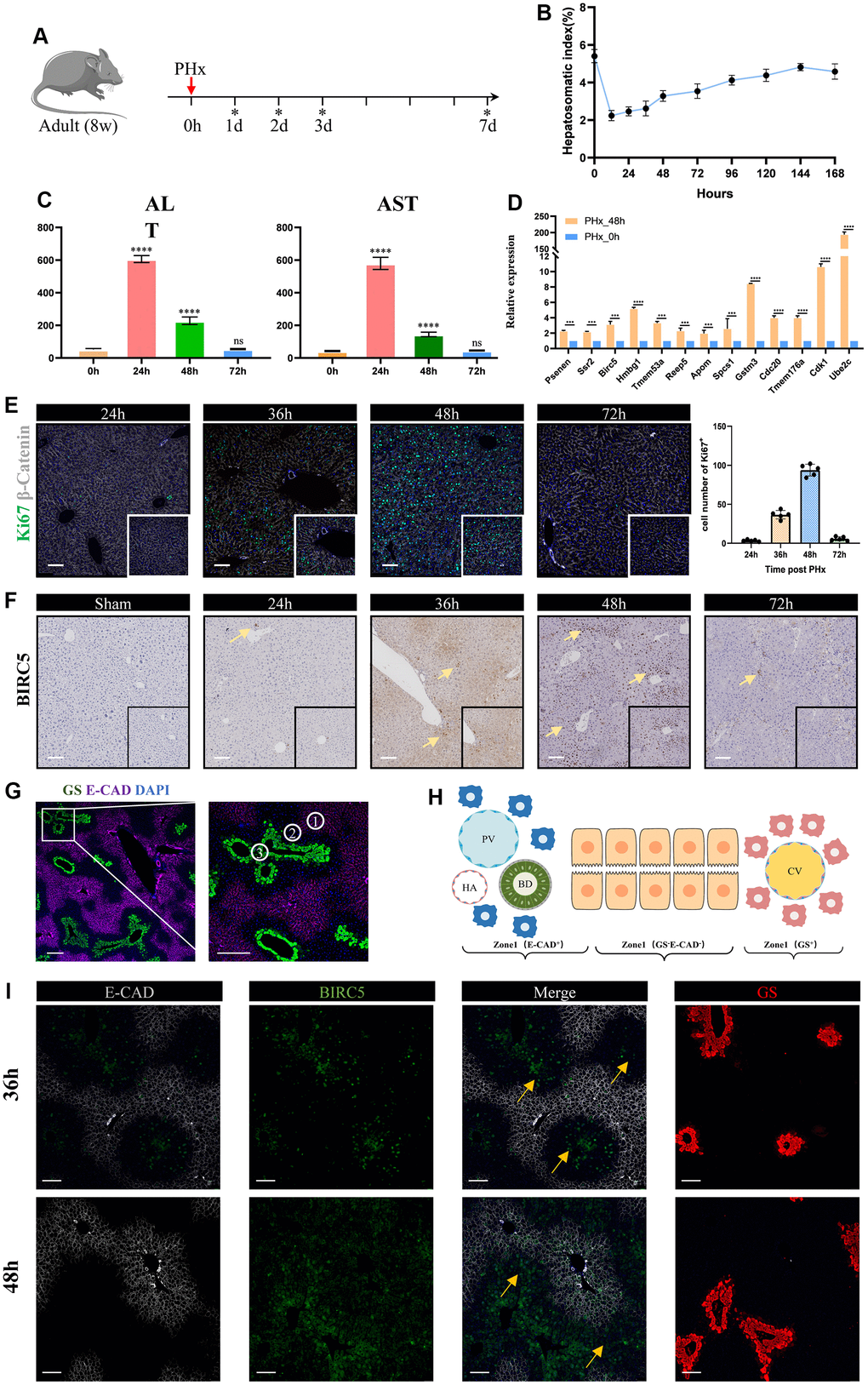
Liver has great power of regeneration function and the proliferative ability will reach peak at 48h after partial hepatectomy (PHx) [6, 7]. Therefore, we tended to explore changes between the proliferative state and quiescent state of liver organ. Here, we collected two scRNA-seq data containing mouse liver cells from adult_0H and PHx_48h groups (Figure 1A). After removing batch effect and with stringent criteria, we had narrowed and filtered 15065 liver cells, and finally 13032 liver parenchymal and nonparenchymal cells for subsequent analyses (Supplementary Figures 1, 2). Clearly, we could find that different cell types were distributed and clustered separately using reduction analysis and UMAP plot visualization (Figure 1A). Then, the proportion of different cell types were showed in Figure 1B–1E. Interestingly, the obtained non-parenchymal liver cells will increase at 48h after PHx both in the two 48h scRNA-seq samples (Figure 1E), indicating the involvement of cell-cell communication during regeneration.
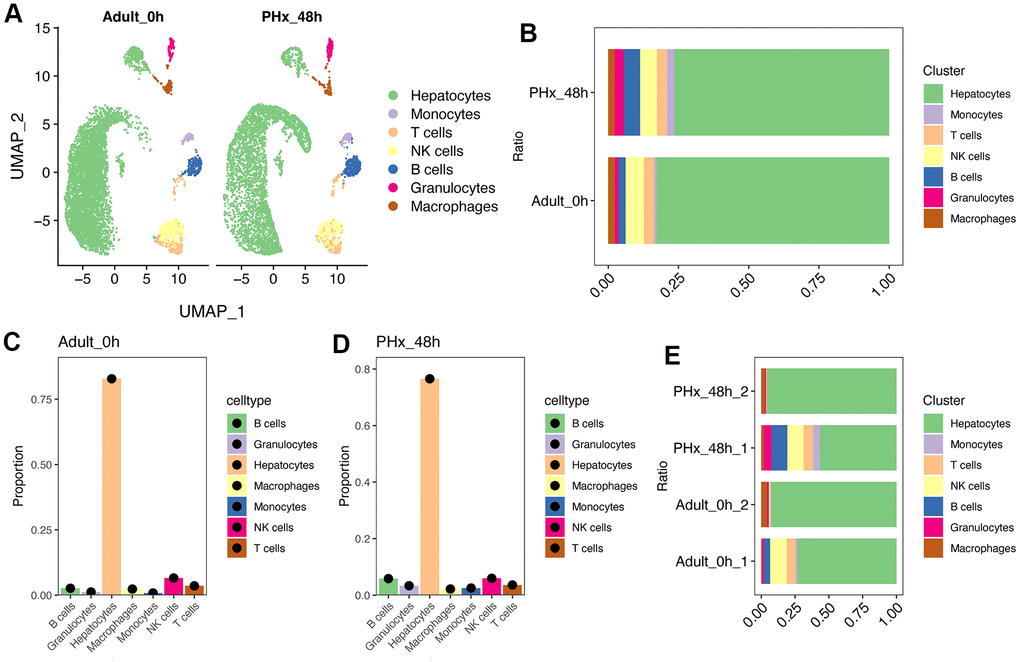
Figure 1. Single cell analysis of cell proportion changes of liver regeneration after PHx. (A) UMAP plot shows the distribution of different cell types of PHx livers. (B) Cell proportion of seven cell types in PHx livers. (C) Bar plot indicates the cell proportion of all seven cell types in liver at 0h after PHx. (D) Bar plot indicates the cell proportion of all seven cell types in liver at 48h after PHx. (E) Cell proportion of seven cell types in PHx livers. The cell proportion in four samples of liver was showed.
Transcriptional factor network and pseudotime analysis revealed the potential regulatory regulons and the communication differences during regeneration
The good performance of the constructed regeneration model indicates the gene expression varied largely during PHx. Therefore, we adopted SCENIC pipeline [21] for further uncovering the differences of transcriptional factors. As a result, several TFs were found out (Figure 5A). According to the calculated Regulon Specificity Score (RSS), The top TFs were different from the two groups, including Hmgb1, Rora, Pole4, Pole3, Rxra and Nr2f6 (Figure 5B). According to the inferred TFs, we further performed the reduction analysis and result implied the different distribution of regeneration-related hepatocytes and other hepatocytes from PHx_0h and PHx_48h groups (Figure 5C). And the mRNA expression level and inferred activity score of Hmgb1 were both higher in regeneration-related hepatocytes rather than other hepatocytes (Figure 5C). Therefore, we inferred our that Hmgb1 might be vital in the regeneration-related hepatocytes of PHx_48h group. Then, we performed the correlation analysis of the inferred TFs and 17 regeneration-related genes. Results showed that Hmgb1 and Pole3 had significant correlated with various gens (Figure 5D).
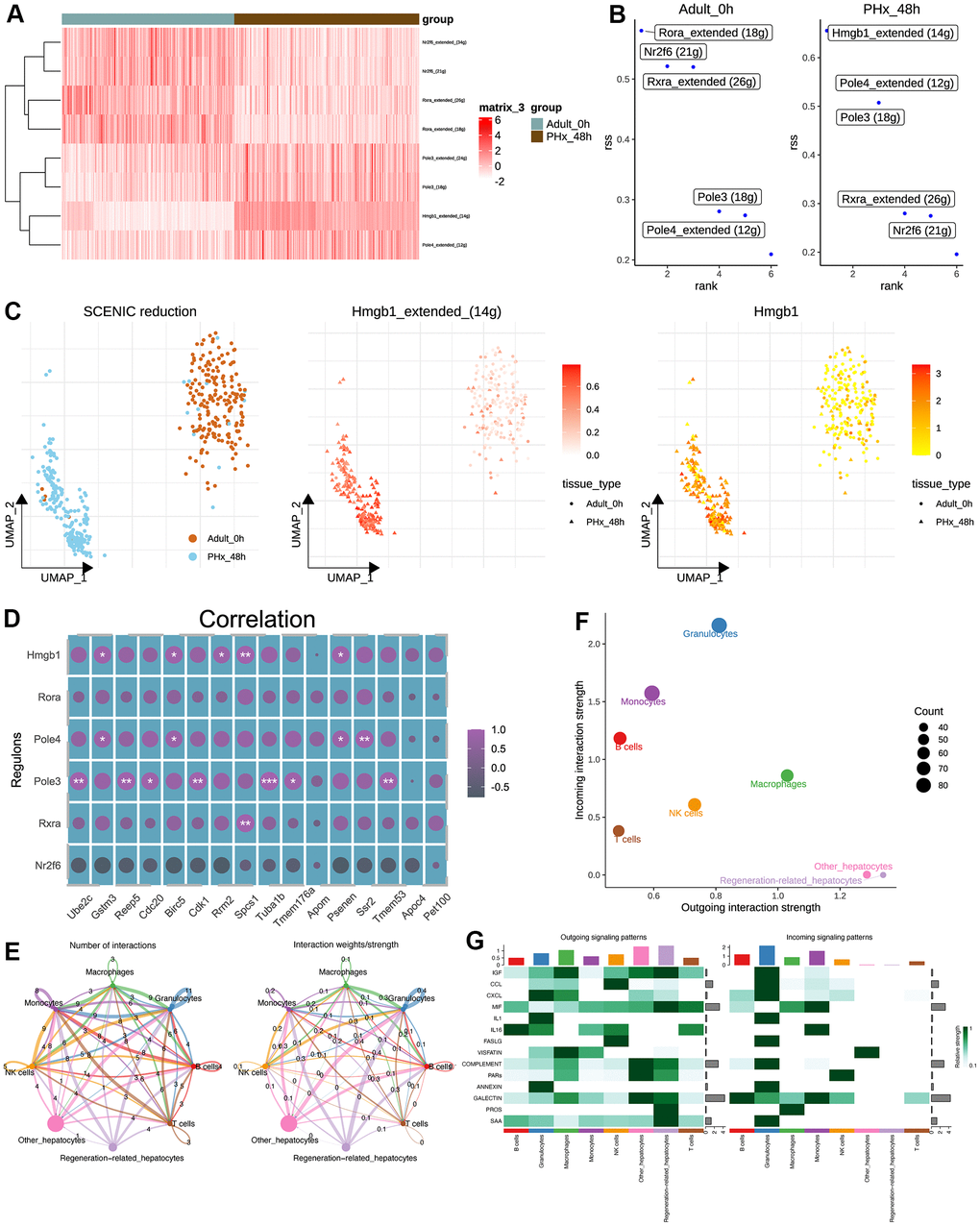
Figure 5. Transcriptional regulatory network and cell-cell communication analyses of hepatocytes. (A) Heatmap shows the differences of TFs in hepatocytes between the PHx 0h and PHx 48h groups. Only 400 hepatocytes were randomly chosen for SCENIC analysis. (B) Top activities of TFs between different groups of hepatocytes. RSS indicates Regulon Specificity Score. (C) Activity distribution of Hmgb1 and expression distribution of Hmgb1 in hepatocytes. (D) Correlation analysis of SCENIC-identified regulons and 17 machine-learning-identified feature genes in bulk RNA-seq of PHx samples. (E) Cellchat analysis of all cell types. Both interaction numbers and interaction strengths were showed. (F) Scatter plot indicates the differences of incoming and outgoing interaction strengths among all cell types. (G) Top cell cytokines were showed in heatmap across all cell types in PHx.
Parallelly, the cell-cell communication analysis was also performed using cellchat pipeline [20]. Collectively, the overall communication ability with other non-parenchymal cell types was still strong during the regeneration process (Figure 5E). Of interest, we found that the outgoing interaction strength, indicating the secretory ability, of regeneration-related hepatocytes was stronger than other hepatocytes (Figure 5F), including IGF, MIF and SAA signaling (Figure 5G).
Functional analysis indicated the involvement of Birc5 in cell cycle and liver regeneration
Subsequently, we conducted the GO and KEGG analyses of the 17 genes (Figure 7A, 7B). Notably, the proliferation-related pathways, including “p53 signaling pathway”, “Cell cycle” and “Regulation of nuclear division” were highly enriched. Then, we explored the average expression level of the 17 genes from single cell data (Figure 7C), especially Birc5 (Figure 7D). Then, GSE167034 dataset was used to run the GSEA analysis of Birc5, which could be used to better understand the potential function of Birc5. Interestingly, result showed that Birc5 had been highly correlated with the “Reactome_cell_cycle” pathway and stem cell-related pathways, consistent with the enrichment analysis of 17 genes (Figure 7E, 7F). Finally, we performed the correlation analysis between Birc5 and Transcriptional factor Hmgb1, which also highlighted the expression correlation between them (Figure 7G).
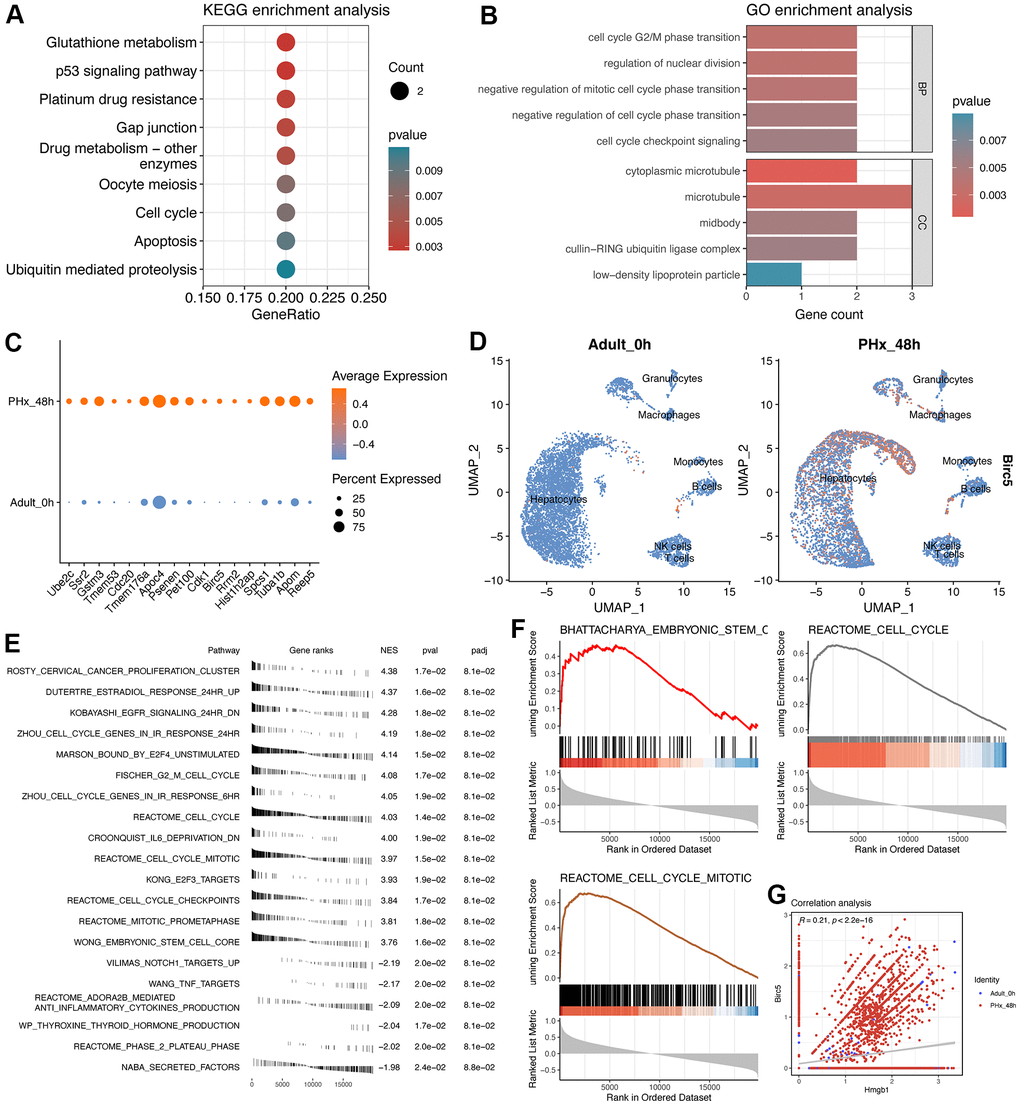
Figure 7. Enrichment analyses of 17-signature-related genes and GSEA analysis of Birc5. (A) KEGG enrichment analysis of 17 regeneration-signature-related genes. Only significant terms were showed and the red color represents lower P value. (B) GO enrichment analysis of 17 regeneration-signature-related genes, showing in bar plot. BP indicates biological process while CC indicates cellular component. (C) Average expression of the 17 genes between PHx 0h and PHx 48 groups, which was displayed using dot plot. (D) Distribution of Birc5 expression in all cell types between PHx 0h and PHx 48 groups. (E) fGSEA analysis of Birc5 and only top significant terms were listed. (F) GSEA analysis of Birc5 gene. The method for ranking detected genes was according to the correlation coefficients with Birc5 gene, which could make us to be capable of enriching Birc5-related terms. (G) correlation analysis between birc5 and Hmgb1.
Loss of spatial characteristics in hepatocytes during regeneration process
Spatial characteristics of hepatocytes indicates the maturation and biological function of hepatocytes. Herein, we collected the genes/biomarkers related to Portal Vein area (PV) and Central Vein area (CV), including Cyp2e1, Cyp2f2, Glul, Ass1 and so on (Figure 8A). Result indicated that the expression of PV- or CV-related genes will be down-regulated in almost all cell types (Figure 8A), especially hepatocytes (Figure 8B). Then, we utilized the “addmodule” function in Seurat packages to further calculate the overall signatures of PV and CV in all 15 hepatocyte clusters (Figure 8C–8F). Obviously, the PV and CV signature score will decrease when the hepatocytes tried to change its status from quiescent state into proliferative state (Figure 8E, 8F). The landmark genes, including Cyp2e1, Cyp2f2, Glul and Ass1, also supported the results that almost all clusters of hepatocytes lose the spatial characteristics to some extent, no matter PV or CV (Figure 8G). Notably, we also found that the identified regeneration signature and Birc5 were both negatively correlated with both PV and CV signature, respectively (Figure 8H).
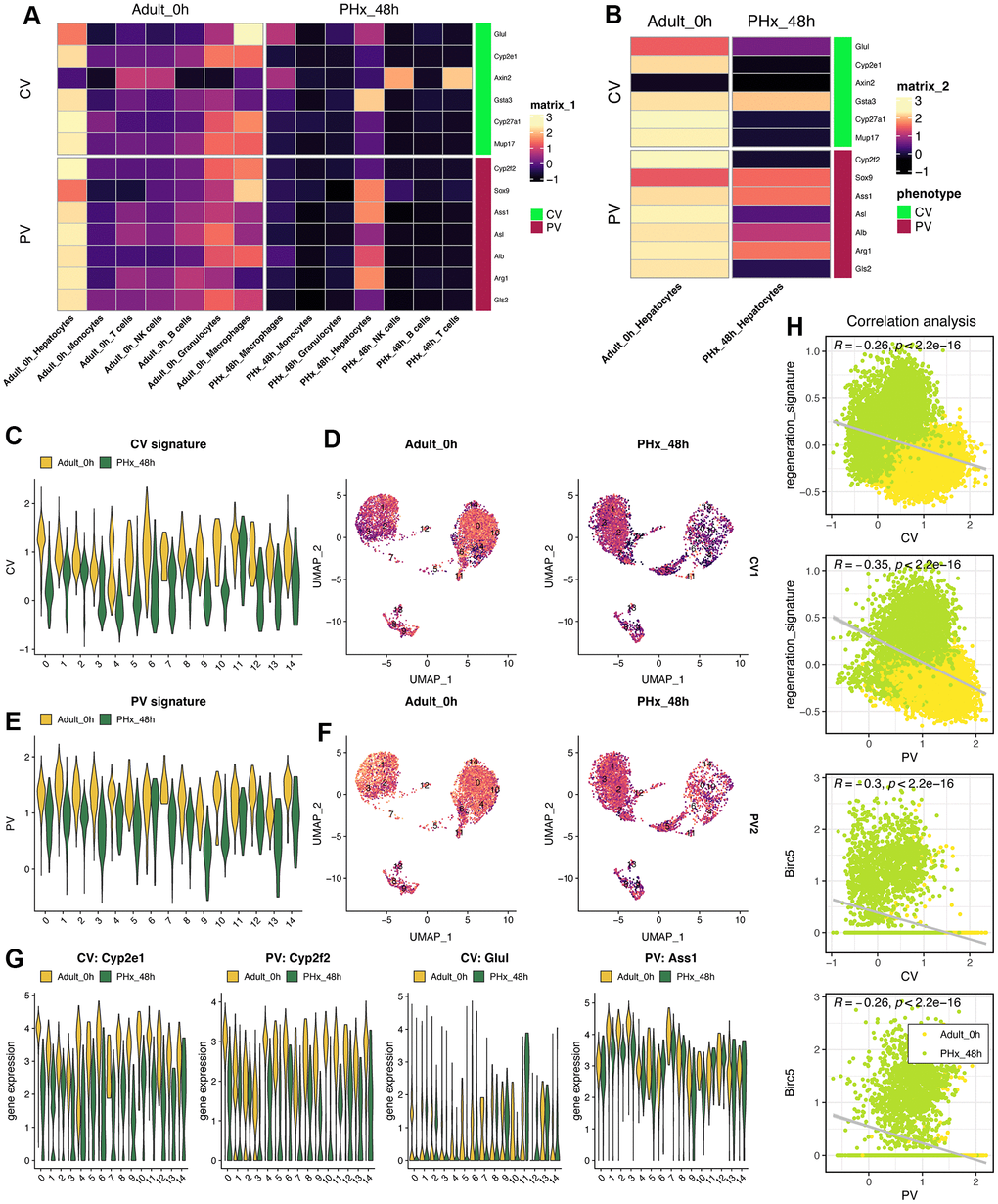
Figure 8. Characteristics of liver zonation in mice after PHx. (A) Expression levels of liver zonation genes across all cell types at PHx 0h and PHx 48h groups. PV: periportal vein area; CV: pericentral vein area. (B) Expression levels of liver zonation genes in hepatocytes with the comparison between the PHx 0h group and the PHx 48h group. (C) Violin plot shows the CV signature score in all 15 hepatocytes clusters. (D) Distribution of the CV signature score in all 15 hepatocytes clusters. The CV signature score was calculated using “addmodule” function in seurat package. (E) Violin plot shows the PV signature score in all 15 hepatocytes clusters. (F) Distribution of the PV signature score in all 15 hepatocytes clusters. The PV signature score was calculated using “addmodule” function in seurat package. (G) Expression levels of Cyp2e1, Cyp2f2, Glul and Ass1, which were typical biomarkers of PV or CV, in all hepatocyte clusters. Yellow indicates the PHx 0h group while Green indicates the PHx 48h group. (H) Correlation analyses among regeneration signature, CV signature, PV signature and Birc5 gene in all single hepatocyte cells from both PHx 0h and 48h groups.
Discussion
Liver transplantation is the only effective treatment for patients with end-stage liver diseases, however, the current supply of liver organ was not enough to cover patient need [23]. Therefore, understanding the mechanism of hepatic proliferation and finding ways to promote hepatocyte growth in vivo after transplantation are urgently needed. Cells related to the hepatic lineage have great potential in the treatment of end-stage liver diseases. Recent studies have demonstrated that cell plasticity and epigenic remodeling that occur in hepatocytes during liver damage repair. Through chromatin remodeling, a proportion of quiescent hepatocytes ultimately transformed into proliferative state, generating transcriptomic alterations which allows cells to re-enter the cell cycle [24]. Utilizing scRNA-seq techniques, we generated an unbiased and comprehensive cell atlas related to liver regeneration. We collected liver tissue samples from normal mice and mice 48 hours following PHx surgery to annotate different cell subsets and reveal changes in the transcriptome of hepatocytes, as well as illustrate their interactions during liver regeneration. This study provides a critical foundation for further understanding the reprogramming effects and molecular mechanisms of mature hepatocytes during liver regeneration. regeneration. Numerous studies and several pioneer factors, such as EGFR, IL-6, TGFβ revealed that liver regeneration and tumor development are inseparable process [11, 25, 26]. Of note, a signature of a hepatocyte subset related to liver regeneration was demonstrated to be useful in determining the regenerative capacity of hepatocytes, particularly Birc5, indicating that it may be a promising key regulatory factor. Moreover, The Birc5 have also been confirmed during rat PHx liver injury models and increased during 5-7days after human transplantation [27], partly indicating the reliability of our identified signature and the crucial of Birc5 for hepatocyte proliferation and regeneration in PHx.
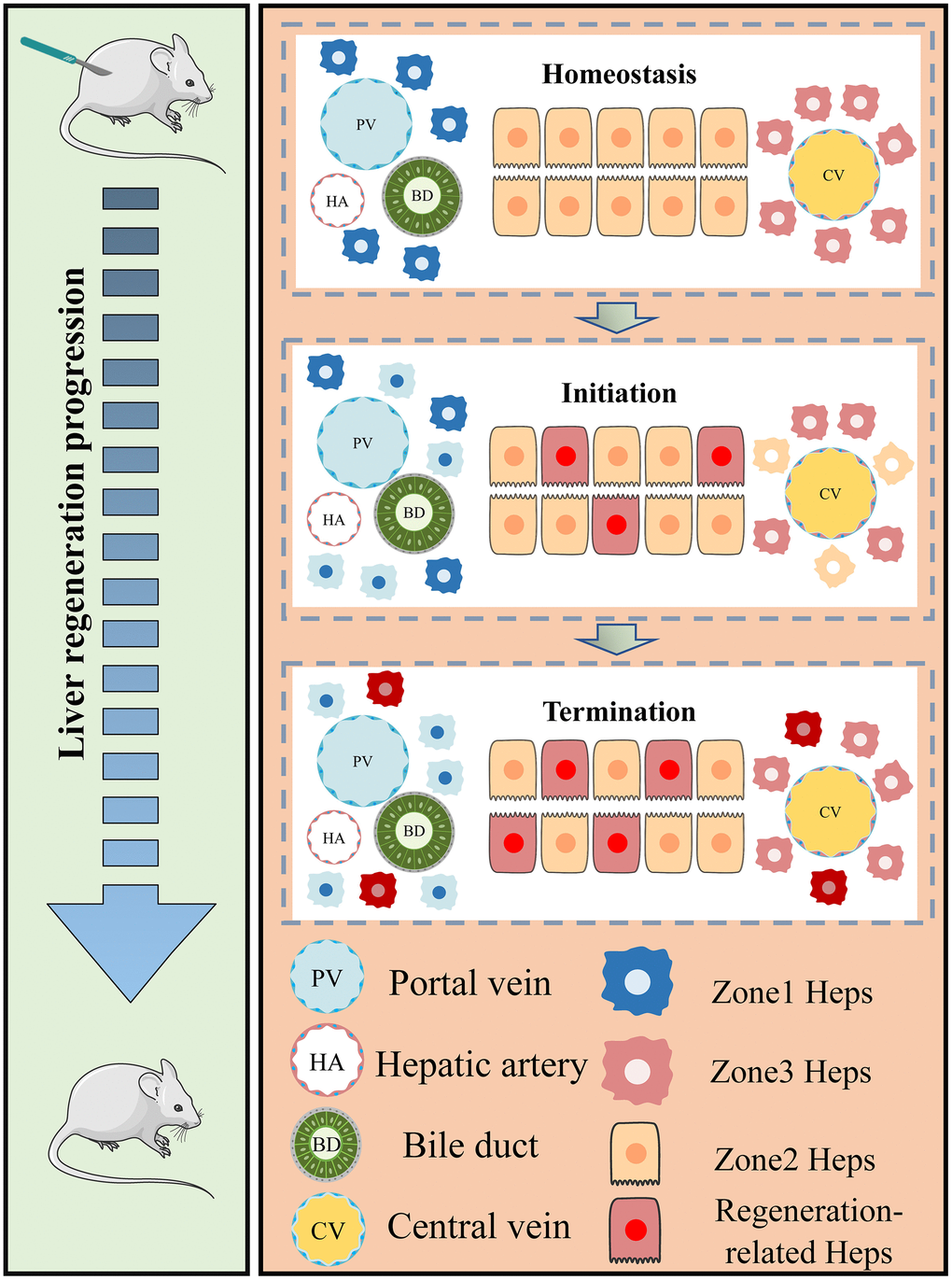
Under normal circumstances, the liver is a relatively stationary organ, and mature hepatocytes are largely quiescent. Random DNA-labeling revealed that only 1.9% hepatocytes divided more than once in 8 to 10 months [28]. However, the liver possesses an exceptional ability to regenerate rapidly following partial hepatectomy, enabling surgical resection of a portion of the liver as well as living donor and segmented liver transplantation [29]. Recent evidence suggests that hepatocytes proliferate randomly in all hepatic regions to maintain homeostasis [30]. In the process from partial hepatectomy (PHx) to regeneration, the hepatocyte lineage tracing studies showed that approximately 98% of newly produced hepatocytes were derived from pre-existing hepatocytes, with the remaining fraction likely to be derived from pre-existing hepatic progenitor cells [31]. Particularly, the obtained non-parenchymal liver cells will increase at 48h after PHx both in the two 48h scRNA-seq samples, indicating the involvement of cell-cell communication during regeneration. Advancement in the field of liver regeneration is potential role of macrophage during inflammation and reprogramming process [11, 32]. Recently, Li et al. has reported that hepatocytes could be triggered and induced into proliferative and regenerative state by Kupffer-cell-derived IL-6 after suffering injury [11]. Thus, understanding the mechanism of hepatocyte regeneration, mastering the process of hepatocyte proliferation from static to dynamic, and identifying the regeneration state of hepatocytes are conducive to better culturing hepatocytes in vitro and determining the state of hepatocytes after transplantation into damaged livers.
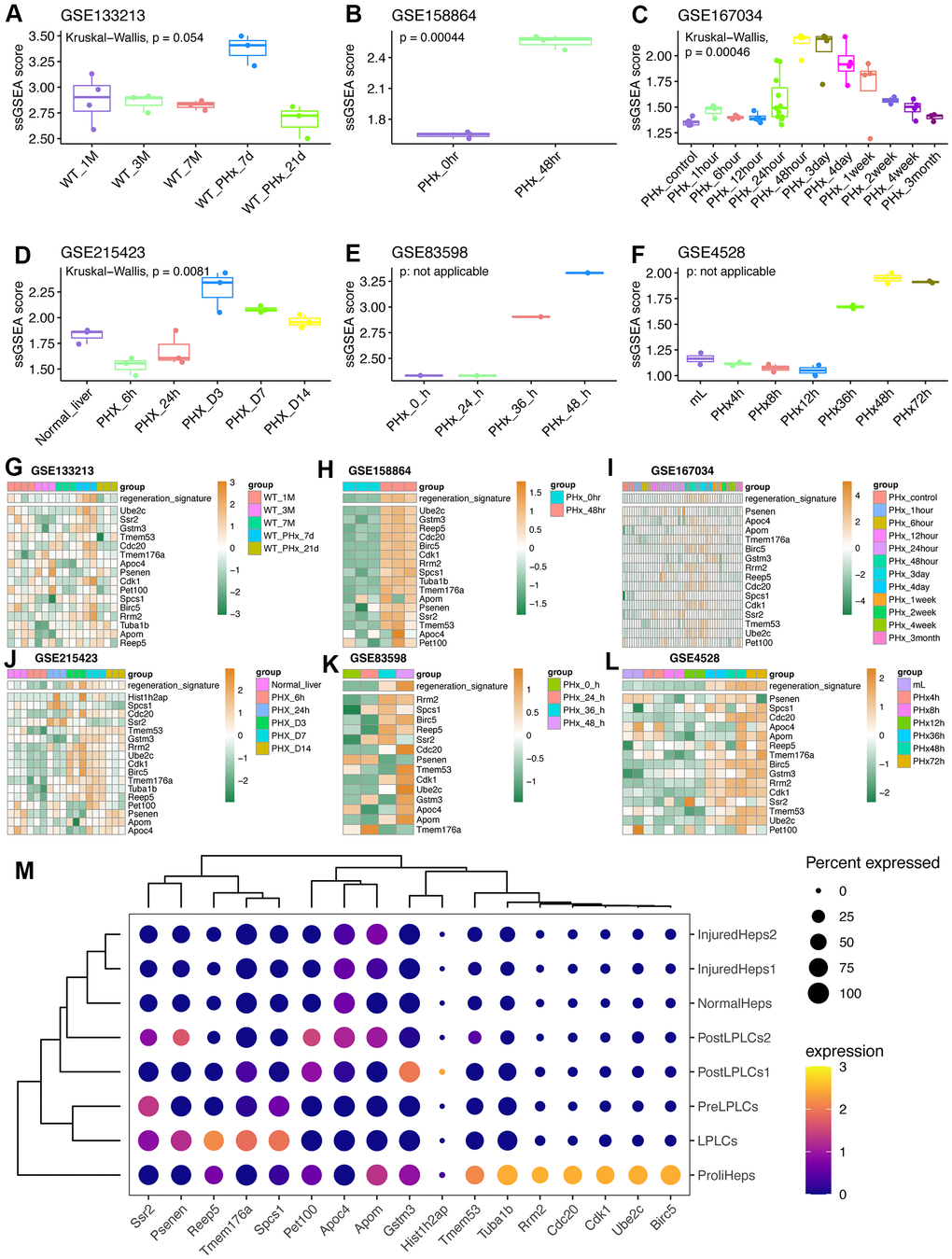
In this work, we screened and obtained a 17-gene model using hdWGCNA, univariate analysis, multivariate analysis, LASSO analysis, and seven other machine learning analyses, including genes of Ube2c, Ssr2, Gstm3, Tmem53, Cdc20, Tmem176a, Apoc4, Psenen, Pet100, Cdk1, Birc5, Rrm2, Hist1h2ap, Spcs1, Tuba1b, Apom and Reep5. The process of hepatocyte proliferation from static state to dynamic state often needs to enter proliferation cycle. Herein, it can be seen that some cycle-related genes, such as Cdk1 and Cdc20, are included in this model. Using enrichment analysis, we also found that these 17 genes were closely related to the cycle. In particular, GSEA and fGSEA analyses of the Birc5 gene revealed that Birc5 was strongly associated with cell cycle. More interestingly, in several external validation datasets, the score levels of Birc5 and 17 genes were highly consistent, better reflecting the proliferative status of regeneration-related hepatocytes, and seemed to be a relatively reliable biomarker/indicator. Another fascinating part in the field of liver regeneration is liver zonation, which remain several unsolved problems and several unanswered questions remain regarding how liver zonation can drive hepatic pathophysiology. Our single-cell analysis revealed that zonal differences of hepatocytes gene expression decrease, which is consistent with previous results about liver regeneration post PHx [7]. Previous studies with different lineage tracing strategies came up with widely varying conclusion [33, 34]. Our work revealed that midlobular hepatocytes were primary involved in acute liver injury and further studies are needed to explore how liver zonation affect liver regeneration-related hepatocytes and its role in spatial compartmentalization.
Survivin, also known as Birc5 and as part of the chromosomal complex during mitosis, directs the aggregation and segregation of chromosomes as well as cytoplasmic division [35]. When it occurs at the interphase, it is mainly located in the cytoplasm and participates in the anti-apoptotic function of cells [35]. Another finding suggests that survivin may be regulated by metabolic stress, which is consistent with the significant metabolic changes observed following PHx [36]. This work emphasized that Birc5 can act as a potential marker for estimating hepatocyte entering the peak of proliferation, with a significant negative correlation with metabolic zonation of liver, suggesting that it plays an important role in the process of liver regeneration and disruption of liver zonation.
As transcription factors are an important regulatory factor for cell fate determination [37], we tended to explore regulatory factors for triggering the static hepatocytes into proliferative and regenerative hepatocytes. Therefore, SCENIC pipeline was adopted and we found that Hmgb1 might be a potential target for hepatocyte-based regenerative medicine. The high mobility group box 1 gene (Hmgb1) encodes a very abundant non-histone nuclear protein. Hmgb1 is not only a nuclear factor that enhances transcription but also an important cytokine that mediates the pathological effects of cancers, sepsis and other diseases [38]. Hmgb1 was typical and known to be participated in immune response and inflammation [38]. Recently, a study shows that IL6, another known proinflammatory factor, can be secreted by Kupffer cells and get involved in the reprogramming and regeneration of hepatocytes in liver injury via STAT3 pathway [11]. Using the sequencing data from this study, we also found that the identified genes were mainly expressed in proliferative hepatocytes and liver progenitor-like cells (LPLC), partly validating the reliability of the signature.
In this study, we utilized scRNA-seq data to identify key cell subsets and their associated specific signatures during liver regeneration following PHx. Furthermore, we sought to determine whether these signatures could be applicable to other forms of liver injury. After analyzing published scRNA-seq data regarding the expression of Birc5 and Hmgb1, we found that hepatocyte reprogramming was likely closely linked to these genes. It is noteworthy to consider whether Birc5 and Hmgb1 may also play important roles in other types of liver injury.
Conclusions
In conclusion, our study has identified a regeneration-related signature that can be used to estimate liver regeneration. These findings will contribute to a better understanding and interpretation of the biological and clinical aspects of liver-regenerating hepatocytes, providing a fundamental basis for the development of effective diagnosis and therapy. The genes identified in this study, especially Birc5, may be used to evaluate the liver state of patients with liver failure. Alternatively, clinicians could use these molecules as biomarkers to predict the likelihood of spontaneous liver regeneration in patients.
Supplementary Materials
Author Contributions
Overall analysis of the databases, study design, and manuscript preparation and revision were carried out by YD in collaboration with SQJ and XCW. YD and XCW jointly developed the bioinformatic analysis methods. CY and YD conducted the PHx model. HQ and KF performed the verification assays. JFL and JS provided valuable input in the research conception and design, and contributed to the manuscript revision and project funding with YHY. All authors have thoroughly reviewed and approved the final versions of the manuscript.
Acknowledgments
We thank JS and JFL for assistance with constructive discussion.
Conflicts of Interest
The authors declare no conflicts of interest.
Ethical Statement
This animal experiments were approved by the ethics committee of the First Affiliated Hospital of Nanchang University.
Funding
No funding was provided to this study.
Editorial Note
This corresponding author has a verified history of publications using a personal email address for correspondence.
References
- 1. Michalopoulos GK, Bhushan B. Liver regeneration: biological and pathological mechanisms and implications. Nat Rev Gastroenterol Hepatol. 2021; 18:40–55. https://doi.org/10.1038/s41575-020-0342-4 [PubMed]
- 2. Wesley BT, Ross ADB, Muraro D, Miao Z, Saxton S, Tomaz RA, Morell CM, Ridley K, Zacharis ED, Petrus-Reurer S, Kraiczy J, Mahbubani KT, Brown S, et al. Single-cell atlas of human liver development reveals pathways directing hepatic cell fates. Nat Cell Biol. 2022; 24:1487–98. https://doi.org/10.1038/s41556-022-00989-7 [PubMed]
- 3. Ramachandran P, Matchett KP, Dobie R, Wilson-Kanamori JR, Henderson NC. Single-cell technologies in hepatology: new insights into liver biology and disease pathogenesis. Nat Rev Gastroenterol Hepatol. 2020; 17:457–72. https://doi.org/10.1038/s41575-020-0304-x [PubMed]
- 4. Clemens MM, McGill MR, Apte U. Mechanisms and biomarkers of liver regeneration after drug-induced liver injury. Adv Pharmacol. 2019; 85:241–62. https://doi.org/10.1016/bs.apha.2019.03.001 [PubMed]
- 5. Huang H, Wu L, Lu L, Zhang Z, Qiu B, Mo J, Luo Y, Xi Z, Feng M, Wan P, Zhu J, Yu D, Wu W, et al. Single-cell transcriptomics uncovers cellular architecture and developmental trajectories in hepatoblastoma. Hepatology. 2023; 77:1911–28. https://doi.org/10.1002/hep.32775 [PubMed]
- 6. Chembazhi UV, Bangru S, Hernaez M, Kalsotra A. Cellular plasticity balances the metabolic and proliferation dynamics of a regenerating liver. Genome Res. 2021; 31:576–91. https://doi.org/10.1101/gr.267013.120 [PubMed]
- 7. Chen T, Oh S, Gregory S, Shen X, Diehl AM. Single-cell omics analysis reveals functional diversification of hepatocytes during liver regeneration. JCI Insight. 2020; 5:e141024. https://doi.org/10.1172/jci.insight.141024 [PubMed]
- 8. Belenguer G, Mastrogiovanni G, Pacini C, Hall Z, Dowbaj AM, Arnes-Benito R, Sljukic A, Prior N, Kakava S, Bradshaw CR, Davies S, Vacca M, Saeb-Parsy K, et al. RNF43/ZNRF3 loss predisposes to hepatocellular-carcinoma by impairing liver regeneration and altering the liver lipid metabolic ground-state. Nat Commun. 2022; 13:334. https://doi.org/10.1038/s41467-021-27923-z [PubMed]
- 9. Guo R, Jiang M, Wang G, Li B, Jia X, Ai Y, Chen S, Tang P, Liu A, Yuan Q, Xie X. IL6 supports long-term expansion of hepatocytes in vitro. Nat Commun. 2022; 13:7345. https://doi.org/10.1038/s41467-022-35167-8 [PubMed]
- 10. Holland CH, Ramirez Flores RO, Myllys M, Hassan R, Edlund K, Hofmann U, Marchan R, Cadenas C, Reinders J, Hoehme S, Seddek AL, Dooley S, Keitel V, et al. Transcriptomic Cross-Species Analysis of Chronic Liver Disease Reveals Consistent Regulation Between Humans and Mice. Hepatol Commun. 2022; 6:161–77. https://doi.org/10.1002/hep4.1797 [PubMed]
- 11. Li L, Cui L, Lin P, Liu Z, Bao S, Ma X, Nan H, Zhu W, Cen J, Mao Y, Ma X, Jiang L, Nie Y, et al. Kupffer-cell-derived IL-6 is repurposed for hepatocyte dedifferentiation via activating progenitor genes from injury-specific enhancers. Cell Stem Cell. 2023; 30:283–99.e9. https://doi.org/10.1016/j.stem.2023.01.009 [PubMed]
- 12. Morabito S, Miyoshi E, Michael N, Shahin S, Martini AC, Head E, Silva J, Leavy K, Perez-Rosendahl M, Swarup V. Single-nucleus chromatin accessibility and transcriptomic characterization of Alzheimer’s disease. Nat Genet. 2021; 53:1143–55. https://doi.org/10.1038/s41588-021-00894-z [PubMed]
- 13. Li H, van der Leun AM, Yofe I, Lubling Y, Gelbard-Solodkin D, van Akkooi ACJ, van den Braber M, Rozeman EA, Haanen JB, Blank CU, Horlings HM, David E, Baran Y, et al. Dysfunctional CD8 T Cells Form a Proliferative, Dynamically Regulated Compartment within Human Melanoma. Cell. 2019; 176:775–89.e18. https://doi.org/10.1016/j.cell.2018.11.043 [PubMed]
- 14. Baran Y, Bercovich A, Sebe-Pedros A, Lubling Y, Giladi A, Chomsky E, Meir Z, Hoichman M, Lifshitz A, Tanay A. MetaCell: analysis of single-cell RNA-seq data using K-nn graph partitions. Genome Biol. 2019; 20:206. https://doi.org/10.1186/s13059-019-1812-2 [PubMed]
- 15. Hänzelmann S, Castelo R, Guinney J. GSVA: gene set variation analysis for microarray and RNA-seq data. BMC Bioinformatics. 2013; 14:7. https://doi.org/10.1186/1471-2105-14-7 [PubMed]
- 16. Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA. 2005; 102:15545–50. https://doi.org/10.1073/pnas.0506580102 [PubMed]
- 17. Yu G, Wang LG, Han Y, He QY. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012; 16:284–7. https://doi.org/10.1089/omi.2011.0118 [PubMed]
- 18. Korotkevich G, Sukhov V, Budin N, Shpak B, Artyomov MN, Sergushichev A. Fast gene set enrichment analysis. bioRxiv. 2021:060012.
- 19. Trapnell C, Cacchiarelli D, Grimsby J, Pokharel P, Li S, Morse M, Lennon NJ, Livak KJ, Mikkelsen TS, Rinn JL. The dynamics and regulators of cell fate decisions are revealed by pseudotemporal ordering of single cells. Nat Biotechnol. 2014; 32:381–6. https://doi.org/10.1038/nbt.2859 [PubMed]
- 20. Jin S, Guerrero-Juarez CF, Zhang L, Chang I, Ramos R, Kuan CH, Myung P, Plikus MV, Nie Q. Inference and analysis of cell-cell communication using CellChat. Nat Commun. 2021; 12:1088. https://doi.org/10.1038/s41467-021-21246-9 [PubMed]
- 21. Aibar S, González-Blas CB, Moerman T, Huynh-Thu VA, Imrichova H, Hulselmans G, Rambow F, Marine JC, Geurts P, Aerts J, van den Oord J, Atak ZK, Wouters J, Aerts S. SCENIC: single-cell regulatory network inference and clustering. Nat Methods. 2017; 14:1083–6. https://doi.org/10.1038/nmeth.4463 [PubMed]
- 22. Mitchell C, Willenbring H. A reproducible and well-tolerated method for 2/3 partial hepatectomy in mice. Nat Protoc. 2008; 3:1167–70. https://doi.org/10.1038/nprot.2008.80 [PubMed]
- 23. Yang LS, Shan LL, Saxena A, Morris DL. Liver transplantation: a systematic review of long-term quality of life. Liver Int. 2014; 34:1298–313. https://doi.org/10.1111/liv.12553 [PubMed]
- 24. Wang X, He Q, Zhou C, Xu Y, Liu D, Fujiwara N, Kubota N, Click A, Henderson P, Vancil J, Marquez CA, Gunasekaran G, Schwartz ME, et al. Prolonged hypernutrition impairs TREM2-dependent efferocytosis to license chronic liver inflammation and NASH development. Immunity. 2023; 56:58–77.e11. https://doi.org/10.1016/j.immuni.2022.11.013 [PubMed]
- 25. Oh SH, Swiderska-Syn M, Jewell ML, Premont RT, Diehl AM. Liver regeneration requires Yap1-TGFβ-dependent epithelial-mesenchymal transition in hepatocytes. J Hepatol. 2018; 69:359–67. https://doi.org/10.1016/j.jhep.2018.05.008 [PubMed]
- 26. Huang M, Long J, Yao Z, Zhao Y, Zhao Y, Liao J, Lei K, Xiao H, Dai Z, Peng S, Lin S, Xu L, Kuang M. METTL1-Mediated m7G tRNA Modification Promotes Lenvatinib Resistance in Hepatocellular Carcinoma. Cancer Res. 2023; 83:89–102. https://doi.org/10.1158/0008-5472.CAN-22-0963 [PubMed]
- 27. Baba HA, Wohlschlaeger J, Schmitz KJ, Nadalin S, Lang H, Benesch A, Gu Y, Biglarnia AR, Sotiropoulos GC, Takeda A, Takeda N, von Wnuck Lipinski K, Levkau B. Survivin is upregulated during liver regeneration in rats and humans and is associated with hepatocyte proliferation. Liver Int. 2009; 29:585–92. https://doi.org/10.1111/j.1478-3231.2008.01911.x [PubMed]
- 28. Miyajima A, Tanaka M, Itoh T. Stem/progenitor cells in liver development, homeostasis, regeneration, and reprogramming. Cell Stem Cell. 2014; 14:561–74. https://doi.org/10.1016/j.stem.2014.04.010 [PubMed]
- 29. Hu S, Liu S, Bian Y, Poddar M, Singh S, Cao C, McGaughey J, Bell A, Blazer LL, Adams JJ, Sidhu SS, Angers S, Monga SP. Single-cell spatial transcriptomics reveals a dynamic control of metabolic zonation and liver regeneration by endothelial cell Wnt2 and Wnt9b. Cell Rep Med. 2022; 3:100754. https://doi.org/10.1016/j.xcrm.2022.100754 [PubMed]
- 30. Chen F, Jimenez RJ, Sharma K, Luu HY, Hsu BY, Ravindranathan A, Stohr BA, Willenbring H. Broad Distribution of Hepatocyte Proliferation in Liver Homeostasis and Regeneration. Cell Stem Cell. 2020; 26:27–33.e4. https://doi.org/10.1016/j.stem.2019.11.001 [PubMed]
- 31. Malato Y, Naqvi S, Schürmann N, Ng R, Wang B, Zape J, Kay MA, Grimm D, Willenbring H. Fate tracing of mature hepatocytes in mouse liver homeostasis and regeneration. J Clin Invest. 2011; 121:4850–60. https://doi.org/10.1172/JCI59261 [PubMed]
- 32. Goikoetxea-Usandizaga N, Serrano-Maciá M, Delgado TC, Simón J, Fernández Ramos D, Barriales D, Cornide ME, Jiménez M, Pérez-Redondo M, Lachiondo-Ortega S, Rodríguez-Agudo R, Bizkarguenaga M, Zalamea JD, et al. Mitochondrial bioenergetics boost macrophage activation, promoting liver regeneration in metabolically compromised animals. Hepatology. 2022; 75:550–66. https://doi.org/10.1002/hep.32149 [PubMed]
- 33. Wei Y, Wang YG, Jia Y, Li L, Yoon J, Zhang S, Wang Z, Zhang Y, Zhu M, Sharma T, Lin YH, Hsieh MH, Albrecht JH, et al. Liver homeostasis is maintained by midlobular zone 2 hepatocytes. Science. 2021; 371:eabb1625. https://doi.org/10.1126/science.abb1625 [PubMed]
- 34. He L, Pu W, Liu X, Zhang Z, Han M, Li Y, Huang X, Han X, Li Y, Liu K, Shi M, Lai L, Sun R, et al. Proliferation tracing reveals regional hepatocyte generation in liver homeostasis and repair. Science. 2021; 371:eabc4346. https://doi.org/10.1126/science.abc4346 [PubMed]
- 35. Wheatley SP, Altieri DC. Survivin at a glance. J Cell Sci. 2019; 132:jcs223826. https://doi.org/10.1242/jcs.223826 [PubMed]
- 36. Alimujiang M, Sun J, Chen S, Bai N, Chen S, Hu F, Ma J, Xu Y, Xu J, Ma X, Yang Y. Survivin is essential for thermogenic program and metabolic homeostasis in mice. Mol Metab. 2022; 58:101446. https://doi.org/10.1016/j.molmet.2022.101446 [PubMed]
- 37. Stadhouders R, Filion GJ, Graf T. Transcription factors and 3D genome conformation in cell-fate decisions. Nature. 2019; 569:345–54. https://doi.org/10.1038/s41586-019-1182-7 [PubMed]
- 38. Chen R, Kang R, Tang D. The mechanism of HMGB1 secretion and release. Exp Mol Med. 2022; 54:91–102. https://doi.org/10.1038/s12276-022-00736-w [PubMed]