Introduction
The tumor microenvironment (TME) refers to the environment in which tumor cells grow, that includes the tumor cells, surrounding fibroblasts, immune and inflammatory cells, stromal cells, and other cells, as well as the extracellular matrix (ECM), microvessels and biomolecules in the nearby area [1, 2]. In contrast to the normal microenvironment, TME is characterized by low oxygen content, acid accumulation, and abnormal local immune status, which are conducive for proliferation, invasion, adhesion, angiogenesis, resistance to radiotherapy and chemotherapy, and emergence of malignant tumors. TME is a dynamic regulatory network involving multiple signals, in which a variety of cellular and molecular pathways can be potential therapeutic targets. Important anti-tumor therapeutic strategies involve blocking tumor-associated pathways in TME, inhibiting the activity of cells with tumor-enhancing effects, and promoting anti-tumor immunity, in combination with traditional treatments, such as surgery, chemotherapy, and radiotherapy. The molecular substances in TME are closely related to the pathogenesis and development of tumor and are potentially sensitive and specific tumor markers.
There are more than 100 chemical modifications of RNA, with methylation being the main form of modification in all types of RNA [3]. RNA methylation accounts for >60% of all RNA modifications. N6-methyladenosine (m6A) is the most abundant RNA modification in eukaryotes, and regulates the post-transcriptional expression of genes. m6A is mainly distributed in the protein coding sequence of mRNA, 3′ untranslated region (UTR), region around the stop codon, and long exon region [4, 5]. The role of m6A modification in the regulation of gene expression is closely related to various normal physiological processes, including cell differentiation, DNA damage response, biological clock, and sex determination, and the occurrence and development of diseases, such as tumors [5]. During tumor development, m6A modification can regulate the expression of oncogenes and tumor suppressor genes in tumor cells, thereby regulating tumor angiogenesis, extracellular matrix remodeling, epithelial-mesenchymal transition (EMT), and the immune microenvironment to promote tumorigenesis [6]. Recent research has found that m6A modification regulates the transcription level of mRNA encoding genes and also affects the transcription and generation of a variety of non-coding RNAs (ncRNAs) (such as microRNAs, lncRNAs, and circRNAs) [3]. Therefore, m6A modification participates in the regulation of various life processes in cells at multiple levels and plays a regulatory role in many diseases, including tumors [6].
This review aimed to summarize the biological characteristics of ncRNAs related to m6A modification and their roles in TME formation, explore the application prospects of ncRNAs related to m6A modification in clinical treatment, and provide new strategies for the accurate diagnosis and treatment of tumors.
Tumor microenvironment
Since Stephen Paget proposed the hypothesis of “seed and soil” theory of tumors in 1889, the relationship between tumors and TME has attracted extensive research attention. During the occurrence and development of tumor, TME can interact with tumor cells and play a key role in tumor proliferation, inflammation, immune evasion, metastasis and drug resistance. TME is a complex integrated system, and tumor and stromal cells (such as cancer-associated fibroblasts and tumor-related endothelial cells) and immune cells (such as myeloid-derived suppressor cells, tumor-associated macrophages, dendritic cells, and tumor-infiltrating lymphocytes) in TME can communicate in several ways to promote tumor growth, angiogenesis, immune escape, and metastasis (Figure 1). As an important epigenetic regulatory mechanism, ncRNAs can regulate gene expression at the genomic and chromosomal levels [7, 8], participate widely in signaling, and be transmitted through exosomes to influence recipient cells and promote TME formation.
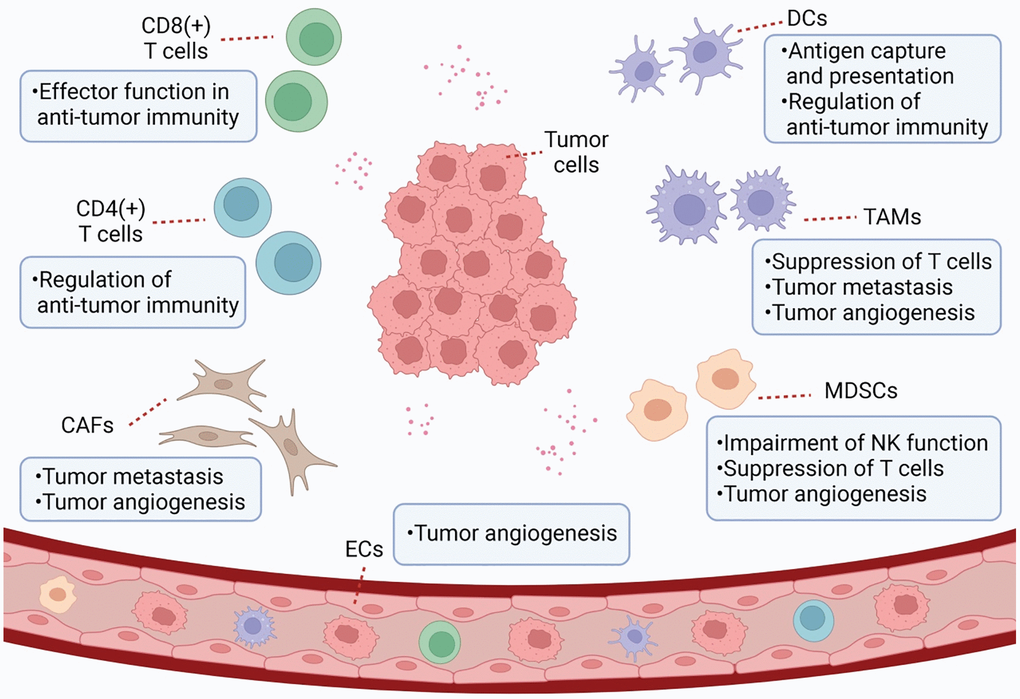
Figure 1. Schematic representation of TME. TME is a complex and comprehensive system. In addition to tumor cells, stromal cells (e.g., CAF and tumor-associated endothelial cells) and immune cells (e.g., MDSC, TAM, DC, and TILs) are also important components of TME.
A study by Tikhonova [3] profiled 17,374 single cells of the mouse bone marrow niche, identified previously unrecognized heterogeneity within the bone marrow microenvironment and demonstrate how the microenvironment responds to acute bone marrow stress at a single-cell level. We couple transcriptional profiling with fluorescent reporters and functional studies to demonstrate that vascular expression of the Notch receptor DLL4—identified by our single-cell studies—suppresses premature upregulation of the myeloid program in HSCs. They showed that single-cell transcriptomic profiling can translate to mechanistic insights into the regulation of the differentiation of stem and progenitor cells. They match haematopoietic factors to their cellular sources, and show that the loss of the vascular-endothelial-expressed Notch ligand DLL4 skews bone marrow haematopoiesis towards a significant transcriptional reprogramming and myeloid priming of HSPCs. Future studies are needed to investigate the functional consequences of niche heterogeneity on aberrant stem cell functions, such as immunodeficiency, haematopoietic malignancies and ageing. Targeting niche-associated factors could interfere with the initiation and progression of disease, as has recently been shown by targeting vascular CXCR4–CXCL12 interactions in acute lymphoblastic leukaemia [5].
A study by Zhou [6] conducted single cell RNA sequencing of human and mouse HCC tumors revealed heterogeneity of cancer-associated fibroblast (CAF). Cross-species analysis determined the prominent CD36+ CAFs exhibited high-level lipid metabolism and expression of macrophage migration inhibitory factor (MIF). Lineage-tracing assays showed CD36+CAFs were derived from hepatic stellate cells. Furthermore, CD36 mediated oxidized LDL uptake-dependent MIF expression via lipid peroxidation/p38/CEBPs axis in CD36+ CAFs, which recruited CD33+myeloid-derived suppressor cells (MDSCs) in MIF- and CD74-dependent manner. Co-implantation of CD36+ CAFs with HCC cells promotes HCC progression in vivo. Finally, CD36 inhibitor synergizes with anti-PD-1 immunotherapy by restoring antitumor T-cell responses in HCC. The work underscores the importance of elucidating the function of specific CAF subset in understanding the interplay between the tumor microenvironment and immune system.
Stromal cells in tumor microenvironment
Stromal cells in TME can be induced by signal factors secreted by tumor cells, and their gene expression and functional status are different from those of stromal cells in normal tissue. Stromal cells promote tumor progression by participating in the proliferation and migration of tumor cells, extracellular matrix remodeling, immune cell recruitment, and tumor angiogenesis.
There are numerous cancer-associated fibroblasts (CAFs) in TME. CAF can be derived from local normal fibroblasts, differentiated from bone marrow-derived mesenchymal cells, or generated by transdifferentiation of pericytes and other cell types [9, 10]. Transforming growth factor β (TGF-β), platelet-derived growth factors (PDGFs), and fibroblast growth factor (FGF) 2 are the main inducers of CAF activation [11]. Stromal derived factor 1 (SDF-1) secreted by CAF binds to CXCR4 on the surface of tumor cells and vascular endothelial cells (ECs), increasing tumor growth and malignancy as well as promoting angiogenesis [12]. Hepatocyte growth factor secreted by CAF enhances tumor invasion and metastasis by activating c-Met [13]. CAF can also induce the transcription of long non-coding RNA (lncRNA) HOTAIR by secreting TGF-β1, thereby promoting EMT and the metastasis of breast cancer cells [14]. In ovarian cancer, CAF upregulates lncRNA LINC00092 by secreting CXCL14 and promotes cancer metastasis by altering glycolysis [15].
Angiogenesis plays an important role in tumor growth and metastasis. Mesenchymal stem cells, tumor-associated macrophages and CAF secrete a variety of vascular growth factors into TME to promote angiogenesis and EC proliferation is the core process for neovascularization. CXCR7 expression is upregulated in tumor-associated EC, which promotes angiogenesis in TME through ERK1/2 phosphorylation [16]. Exosomes containing miR-23a secreted by tumor cells promote tumor microvasculogenesis by acting on SIRT1 in EC [17]. The vascular endothelial growth factor (VEGF)/VEGFR signaling pathway is important in tumor angiogenesis. VEGF upregulates Bcl-2 in vascular ECs and promotes tumor angiogenesis [18]. miR-134 inhibits angiogenesis in osteosarcoma by targeting the VEGF/VEGFR1 pathway [19]. In neuroblastoma, MALAT1 promotes EC migration and angiogenesis through the upregulation of FGF2 [20]. In addition, tumor cells and ECs directly interact to promote tumor angiogenesis through the mitogen-activated protein kinase (MAPK) and Notch pathways [21, 22]. Currently, anti-angiogenesis therapy is a conventional treatment strategy for tumor in clinical practice.
Immune cells in tumor microenvironment
During tumor development, tumor cells can take advantage of the negative regulation mechanism of the immune system to alter the functional status of various infiltrated immune cells, forming a microenvironment with low anti-tumor immunity, leading to tumor immune tolerance and immune escape. Therefore, inducing and enhancing the anti-tumor immune response is an important strategy to improve anti-tumor therapy efficacy.
TME comprises numerous immune cells, including myeloid-derived suppressor cells (MDSCs) that are a group of heterogeneous cells that originate from the myeloid system. MDSCs continue to differentiate into dendritic cells, macrophages, and granulocytes under normal circumstances, while under pathological conditions, MDSCs cannot differentiate but they develop into a cell population that can inhibit immune function. Several cytokines in TME can induce the proliferation of MDSCs, including COX2, interleukin6 (IL-6), granulocyte macrophage colony stimulating factor (GM-CSF), and VEGF [23]. Lnc-CHOP enhances the immunosuppressive function of MDSCs by promoting C/EBPβ activation and the expression of molecules associated with MDSC immunosuppressive activity [24]. However, lnc-C/EBPβ inhibits the activation of C/EBPβ and reduces MDSC function [25]. The immunosuppressive function of MDSCs mainly manifests as the suppression of the immune-killing effect of T cells and NK cells. MDSCs can inhibit T cells by promoting T cell apoptosis, consuming essential amino acids for T cell function, reducing T cell migration to lymph nodes, preventing T cell signaling, and inducing T cell differentiation imbalance [26, 27]. MDSCs inhibit NK cells by inhibiting activation receptor expression, downregulating perforin secretion, and limiting response to IL-2 by NK cells [28]. MDSCs are also involved in the regulation of tumor angiogenesis [29].
Macrophages are classified into M1 and M2 phenotypes [30]. M1 macrophages can secrete pro-inflammatory factors, such as IL-1, IL-12, and TNF-α, which are involved in defense against infections and tumoricidal activities [31]. In contrast, M2 macrophages highly express the immunosuppressive factor IL-10, which exerts anti-inflammatory and tumor-promoting effects [32]. Tumor-associated macrophages (TAMs) infiltrate the tumor area, showing a functional phenotype similar to that of M2 macrophages. A variety of chemokines, growth factors, and cytokines in TME recruit macrophages to infiltrate [33] and induce M2 phenotype transformation. TAM recruitment and polarization are regulated by ncRNAs. lncRNAs LNMAT1 [34] and lnc-BM [35], promote macrophage recruitment, while lncRNA-MM2P regulates the expression of M2-related genes in macrophages [36]. miR-21-3p, miR-125b-5p and miR-181d-5p can be delivered into TME by cancer-derived exosomes to promote M2 polarization of macrophages [37]. In TME, TAM can secrete matrix metalloproteinases (MMP) to promote basement membrane degradation, thus promoting metastasis. TAM can also secrete basic FGF, VEGF, CXCL8, and other factors that promote tumor angiogenesis [38]. TGF-β and IL-10 released by M2 macrophages can inhibit T cell immune responses and anti-tumor immunity.
Dendritic cells (DCs) have a strong ability for antigen uptake and processing, which can induce an anti-tumor immune response by presenting tumor antigens. DCs are divided into plasmacytoid dendritic cells (pDCs) and myeloid dendritic cells (mDCs). pDCs exert non-specific anti-infection and anti-tumor immunity by activating monocytes, macrophages, B cells, NK cells, and naive T cells [37]. mDCs mainly activate the transformation of initial CD4 cells to T helper (Th) 1 cells and CD8 cells to cytotoxic T lymphocytes (CTL) through antigen presentation and exert specific anti-infection and anti-tumor immunity [39]. Lnc-DC can bind to STAT3 to promote DC differentiation and improve the antigen presentation capacity [40]. However, in TME, various cytokines can induce DC differentiation disorders, resulting in abnormal immunomodulatory functions.
In TME, tumor-infiltrating lymphocytes (TILs) are influenced by different cytokines and activation mechanisms to produce different immune responses. CD8+T cells can recognize tumor antigens or secrete cytokines and play an effector-killing role in anti-tumor immunity. Lnc-Tim3 interacts with Tim-3 to release Bat3, resulting in CD8+ T-cell depletion and immune evasion in hepatocellular carcinoma (HCC) [41]. Activated CD8+T cells express cytotoxic T lymphocyte-associated antigen-4 (CTLA-4) and programmed death-1 (PD-1) [42]. Immunocheckpoint inhibitors (ICIs) targeting CTLA-4 and PD-1 have become important anti-tumor immunotherapies. The overexpression of miR-142-5p in tumor cells can block the PD-L1/PD-1 pathway and enhance anti-tumor immune function [43]. CD4+T cells assist and regulate immunity and can proliferate and differentiate into a variety of cell subsets: Th1, Th2, Th17, and regulatory T cells (Tregs) [44]. Interferon (IFN)-γ secreted by Th1 cells can activate cytotoxic CD8+T cells, DCs, and macrophages and play an anti-tumor role. Th2 cells secrete IL-4 to activate the tumor-promoting macrophages; moreover, Tregs can inhibit the proliferation and differentiation of T cells, hinder antigen presentation, mediate target cell death, and play an immunosuppressive part [45]. Several chemokines in TME and DC differentiation disorder can promote the recruitment, proliferation and activation of Tregs [46, 47]. The lncRNA SNHG1 regulates Treg cell differentiation through the miR-448/IDO pathway, affecting tumor immune escape [48], while specific Treg clearance can enhance the anti-tumor immune response [49, 50].
m6A modifications and noncoding RNAs
Molecular component of m6A RNA methylation
m6A methylation is a dynamic and reversible process [51], which is regulated by m6A methyltransferase complexes (m6A writers), m6A demethylases (m6A erasers), and m6A-binding proteins (m6A readers) (Figure 2A, 2B).
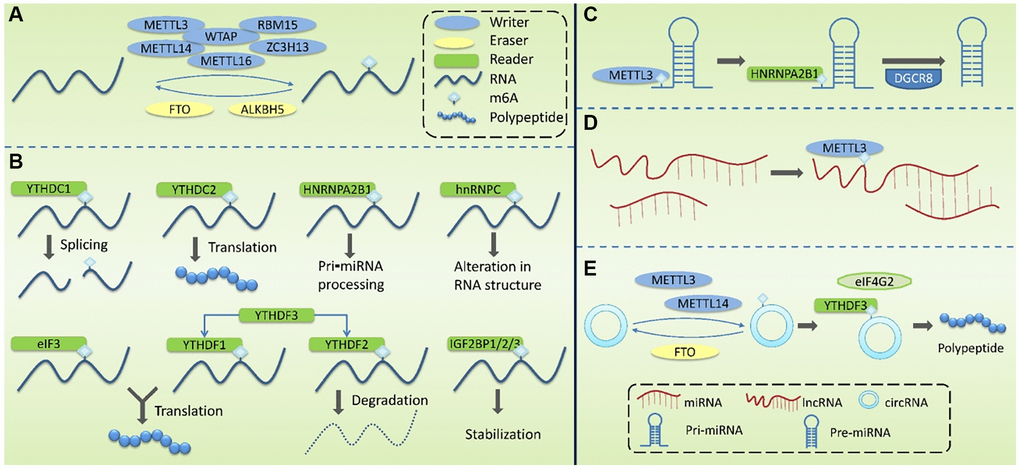
Figure 2. Molecular mechanism associated with the modification of m6A methylation. (A) m6A Writers are mainly composed of METTL3, METTL14, WTAP, METTL16, RBM15 and ZC3H13, mediating the modification of m6A methylation of RNA. Erasers mediate the process of m6A demethylation, mainly including FTO and ALKBH5. (B) m6A Readers recognize m6A methylation, including YTHDF1-3, YTHDC1-2, HNRNPC, HNRNPA2B1, eIF3, and IGF2BP1/2/3. (C) m6A modification plays a regulatory role in pri-miRNA to regulate the processing and maturation of miRNAs. (D) m6A modification plays a regulatory role in lncRNA to affect the RNA-RNA interaction function of lncRNA. (E) m6A modification plays a regulatory role in circRNA to promote its’ translation.
Writers take S-adenosylmethionine (SAM) as a methyl donor and mediate m6A methylation modification of RNA. These complexes are mainly composed of methyltransferase-like 3 (METTL3), methyltransferase-like 14 (METTL14), Wilms tumor1-associating protein (WTAP), methyltransferase-like 16 (METTL16), RNA binding motif protein 15 (RBM15), and zinc finger CCCH domain-containing protein 13 (ZC3H13). METTL3 contains a SAM-binding region [52] that identifies potential m6A modification sites and mediates the transfer of methyl from SAM to this site. METTL14 can form a heterodimer with METTL3 and specifically promote METTL3 recognizing RNA substrates. METTL3 and METTL14 are located on nuclear speckles and their localization is dependent on WTAP. By binding to the METTL3-METTL14 dimer, WTAP enables the methyltransferase complex to rapidly recognize potential m6A modification sites and activate the METTL3/METTL14 complex [53]. It has been reported that ZC3H13 can bind to WTAP and anchor the METTL3/METTL14 complex to the nucleus [54].
Erasers mediate the process of m6A demethylation, including the modulation of fat mass and obesity-associated gene (FTO) and AlkB homolog 5(ALKBH5). FTO oxidizes N-methyl at the m6A site to form hydroxymethyl groups [51, 55]. ALKBH5, co-locating with nuclear speaker in an RNaseA-sensitive manner, can directly catalyze the removal of methyl from 6-methylated adenosine and tends to demethylate specific m6A-modified single-strand RNA [56].
Readers recognize m6A methylation and act by specifically binding to the m6A-modified region or altering the RNA secondary structure to facilitate protein binding to RNA. Readers include YTH domain family (YTHDF) 1-3, YTH domain-containing proteins (YTHDC) 1-2, heterogeneous nuclear ribonucleoproteins (HNRNP), HNRNPC and HNRNPA2B1, eukaryotic translation initiation factor 3(eIF3), and insulin-like growth factor 2 mRNA-binding protein 1/2/3 (IGF2BP1/2/3). The role of YTHDF1-3 is mainly in the cytoplasm. YTHDF1 promotes the translation of m6A-labeled transcripts [57] and YTHDF2 promotes the degradation of m6A mRNA [58]. The combination of YTHDF3 with YTHDF1 enhanced its pro-translational ability, and the combination with YTHDF2 promoted its pro-degradation ability. The role of YTHDC1-2 is mainly in the nucleus. YTHDC1 may modulate pre-mRNA splicing factors to regulate RNA splicing [59], and YTHDC2 promotes mRNA translation [60]. The HNPNPC family regulates the selective splicing and structural alterations in mRNA [61]. HNRNPA2B1 interacts with the Di George Critical Region 8 (DGCR8) protein to promote pri-miRNA processing [62]. hnRNPC affects the local secondary structure of mRNA and lncRNA [61]. eIF3 binds to the m6A site in the 5′UTR of mRNA and promotes cap-independent mRNA translation [63]. IGF2BP1/2/3 increase the stability and translation efficiency of mRNA [64].
In recent years, m6A detection technology has developed rapidly, and a large number of m6A methylation sites have been identified by combining immunoprecipitation and high-throughput sequencing (meRIP-seq [65] and m6Aseq [66]). However, these two methods cannot identify m6A methylation sites that are very close to each other or accurately identify the m6A site. Photo-crosslinking co-immunoprecipitation techniques (such as PA-m6A-seq [67] and m6A-CLIP [68]) can achieve more accurate identification of m6A sites on the individual bases of RNA. In addition, the SCARLET method can detect single m6A sites with high accuracy [69]. However, it is expensive to detect m6A sites individually, and detection after bioinformatics prediction can greatly improve research efficiency. At present, multiple databases are available for researchers to use (Table 1), such as the WHISTLE database [70], SRAMP database (http://www.cuilab.cn/sramp/) [71], and HSM6AP database (http://lab.malab.cn/~lijing/HSM6AP.html) [72], which can be used to predict m6A sites. Verified m6A targets and potential m6A targets can be queried in the M6A2Target database (http://m6a2target.canceromics.org) [73], and WITMSG database (http://rnamd.com/intron/) can be used to predict m6A sites of introns [74]. m6A sites in specific cell lines or tissue types can be queried in the REPIC database (https://repicmod.uchicago.edu/repic) [75]. RMBase v2.0 database (http://rna.sysu.edu.cn/rmbase) [76] and iMRM (http://www.bioml.cn/XG_iRNA/home) [77] have also been used to predict methylation modification sites other than m6A. As m6A modification can be regulated by m6A regulatory proteins and affects a variety of physiological and pathological processes, databases integrating m6A functional annotations have emerged. RMBase v2.0 database (http://rna.sysu.edu.cn/rmbase) contains abundant information for exploring the relationship between RNA modification/miRNA binding and disease-associated single nucleotide polymorphisms (SNPs) [76]. Met-DB v2.0 database (http://compgenomics.utsa.edu/MeTDB/) integrates m6A “Writer,” “Eraser”, and “Reader” database, which facilitates the understanding of the function of m6A modification [78]. The development of technologies for m6A detection and prediction has greatly promoted in-depth research.
Table 1. m6A modification-associated databases.
| Database | Websites | Characteristic | Ref. |
| WHISTLE | Predicted m6A sites | [70] | |
| SRAMP | [71] | ||
| HSM6AP | [72] | ||
| M6A2Target | Searched verified m6A targets and potential m6A targets | [73] | |
| WITMSG | Predicted m6A sites of introns | [74] | |
| REPIC | Searched m6A sites in specific cell lines or tissue types | [75] | |
| RMBase v2.0 | a. Predicted methylation modification sites other than m6A b. Explored the relationship between RNA modification/miRNA binding and disease-associated SNPs | [76] | |
| iMRM | Predicted methylation modification sites other than m6A | [77] | |
| Met-DB v2.0 | Integrated m6A “Writer,” “Eraser”, and “Reader” database, which facilitates the understanding of the function of m6A modification | [78] |
Function and role of m6A RNA methylation
m6A modification occurs mainly in mRNA and it affects splicing, nuclear export, translation, and degradation of mRNA. Alterations in m6A modification can regulate mRNA processing (Table 2). FTO regulates exon splicing of the adipogenesis regulator RUNX1T1 by modulating m6A levels around the splicing site, thus regulating differentiation [79]. SR proteins are important regulators of alternative splicing. YTHDC1 promotes the RNA-binding ability of SRSF3 and inhibits that of SRSF10 to regulate mRNA splicing [59]. YTHDC1 also interacts with SRSF3 and RNA nuclear export factor 1 to regulate mRNA nuclear export [80]. The regulation of m6A modification in the degradation and translation of mature mRNA is another way to regulate gene expression. METTL3-mediated m6A modification of SOX2 mRNA increases transcription stability [81]. YTHDF1 [57] and IGF2BP1/2/3 are involved in regulating the translation of m6A-modified mRNA [64]. YTHDC2 enhances translation efficiency and reduces the stability of its target mRNA by interacting with translation and decay mechanisms [82]. It is reported that RNA m6A methyltransferase Mettl3 interacts with the 5′ external transcribed spacer (5′ETS) of the 47S rRNA precursor and modifies adenosine 196. Mettl3 knockdown results in the increase of pre-rRNA processing rates, while intracellular amounts of rRNA processing machinery components (U3, U8, U13, U14, and U17 small nucleolar RNA (snoRNA)and fibrillarin, nucleolin, Xrn2, and rrp9 proteins), rRNA degradation rates, and total amount of mature rRNA in the cell stay unchanged. Increased efficacy of pre-rRNA cleavage at A′ and A0 positions led to the decrease of 47S and 45S pre-rRNAs in the cell and increase of mature rRNA amount in the cytoplasm.
Table 2. Regulation of mRNAs by m6A modifications.
| Function | m6A regulatory proteins | Mechanism | Ref. |
| mRNA processing | FTO | Regulate exon splicing of the adipogenesis regulator RUNX1T1 by modulating m6A levels around the splicing site, thus regulating differentiation | [79] |
| YTHDC1 | Promote the RNA-binding ability of SRSF3 and inhibit that of SRSF10 to regulate mRNA splicing | [59] | |
| Interact with SRSF3 and RNA nuclear export factor 1 to regulate mRNA nuclear export | [80] | ||
| Degradation and translation of mature mRNA | METTL3 | Mediate m6A modification of SOX2 mRNA to increase transcription stability | [81] |
| YTHDF1 | Enhance translation efficiency of its target RNA | [57] | |
| IGF2BP1/2/3 | Regulate the translation of m6A-modified mRNA | [64] | |
| YTHDC2 | Enhance translation efficiency and reduce the stability of its target mRNA by interacting with translation and decay mechanisms | [82] |
m6A is widely present in various cells and affects cell differentiation, apoptosis, and other biological processes, as well as tumor development and other pathological processes (Table 3). Cell differentiation is a process by which cells acquire different structure and function during ontogeny, and selective gene expression occurs during this stage. The regulation of m6A in mRNA enables m6A to regulate gene expression, thus affecting cell differentiation and ontogeny. There are multiple binding sites of METTL3 on the transcripts of pluripotent genes in mouse embryonic stem cells. It has been reported that METTL3 knockout decreases the m6A level of transcripts of pluripotent genes, which could inhibit the differentiation ability of embryonic stem cells [83]. FTO can downregulate the expression of ASB2 and RARA by reducing m6A levels in UTRs of transcripts, resulting in the inhibition of ATRA-mediated acute myeloid leukemia (AML) cell differentiation [84].
Table 3. Biological functions and role of m6A regulatory proteins in human diseases.
| Functions | m6A regulatory proteins | Mechanisms | Ref. |
| Cell differentiation | METTL3 | Binded to transcripts of pluripotent gene and regulate differentiation ability of embryonic stem cells | [83] |
| FTO | Downregulated the expression of ASB2 and RARA by reducing m6A levels in UTRs of transcripts, resulting in the inhibition of ATRA-mediated AML cell differentiation | [84] | |
| Spermatogenesis | METTL3 | Altered splicing of spermatogenesis-related genes, thus regulating spermatogonial differentiation | [86] |
| METTL3 and METTL14 | Regulation of spermatogonial stem cells and spermatogenesis disorders | [85] | |
| ALKBH5 | Regulated nuclear export of RNA and sperm malformation in mice | [87] | |
| Central nervous system diseases | METTL3 | Regulated mice cerebellar development | [88] |
| METTL14 | Regulated mice cerebral cortex development | [89] | |
| FTO | Regulated mice memory | [91] | |
| Cardiovascular diseases | METTL3 | Contributed to cardiac hypertrophy | [94] |
| Promoted the maturation of miR-34a, which in turn inhibits SIRT1 and promotes the formation of abdominal aortic aneurysm | [96] | ||
| FTO | Participated in reduced cardiomyocyte contractile function during heart failure | [95] | |
| Tumors | METTL3 | Promoted the maturation of miR-25-3p, thus activating the Akt-P70S6K pathway and promoting the initiation and development of PDAC | [97] |
| FTO | Promoted glycolysis of breast cancer cells through the PI3K/AKT pathway | [98] | |
| Upregulated PKM2 expression through the demethylation of PKM2, thereby regulating glucose metabolism of HCC | [99] |
m6A is closely related to spermatogenesis as well as the development and function of the central nervous system. It has been reported that the knockout of METTL3 and METTL14 results in the loss of spermatogonial stem cells and impaired spermatogenesis [85]. Moreover, METTL3 knockout causes decreased m6A levels and altered splicing of spermatogenesis-related genes, leading to the downregulation of gene expression and the regulation of spermatogonial differentiation [86]. A previous study reported that ALKBH5 deficiency in mice increased m6A levels and nuclear export of RNA, ultimately causing sperm malformation in mice [87]. During the development of the central nervous system, METTL3 knockout can lead to decreased m6A in the cerebellum of mice and increased apoptosis of new small brain cells, leading to severe cerebellar hypoplasia [88]. METTL14 knockout resulted in impaired cerebral cortex development in mice [89]. m6A modification in the brain is also involved in cognitive functions such as learning and memory [90]. Artificial deficiency of FTO in the dorsal hippocampus enhanced memory in mice [91]. Notably, abnormal m6A modifications can contribute to the development of Alzheimer’s disease [92]. In addition, m6A modifications can regulate the circadian rhythm of metabolism [93].
Abnormal regulation of m6A gene expression may promote the occurrence of certain diseases [90]. Changes in the expression of m6A play a regulatory role in the occurrence and development of cardiovascular diseases, such as cardiac hypertrophy, heart failure, and aortic aneurysm [91]. Hypertrophic stimulation leads to increased METTL3-mediated m6A modification, which contributes to cardiac hypertrophy [94]. In heart failure, FTO expression is reduced, leading to increased m6A levels and reduced cardiomyocyte contractile function. Decreased cardiomyocyte contractile function induced by ischemia can be alleviated by increasing FTO expression [95]. m6A modification promotes the maturation of miR-34a, which in turn inhibits SIRT1 expression and promotes the formation of abdominal aortic aneurysms [96]. The regulation of m6A modifications in tumors involves several aspects. Cigarette smoke induces the overexpression of METTL3 and upregulation of m6A in pri-miR-25 to promote its processing and maturation. miR-25-3p inhibits PHLPP2 expression, which in turn activates the Akt-P70S6K pathway and promotes the initiation and development of pancreatic ductal adenocarcinoma (PDAC) [97]. The energy metabolism of tumor cells differs from that of normal cells. Aerobic glycolysis promotes adaptation and rapid proliferation of tumor cells in the hypoxic microenvironment. FTO overexpression in breast cancer cell lines promotes glycolysis through the PI3K/AKT pathway [98]. FTO upregulates the expression of PKM2 through the demethylation of PKM2 mRNA in HCC, thus regulating glucose metabolism [99]. Abnormal modification of m6A in TME can modulate tumor immune escape. A previous study analyzed the mRNA expression profiles of 1,938 gastric cancer (GC) samples and established an m6A scoring system based on the status of 21 m6A regulators [100]. An increased mutation load and immune activation were observed with low m6A scores. Effective immune infiltration was not observed with a high m6A score. In fact, the modification of m6A is involved in the entire process of tumorigenesis and development, including the regulation of tumor cell proliferation, angiogenesis, metastasis and invasion, immune escape, and other aspects by promoting the formation of TME.
Aging is a natural process of body decline, characterized by decreased function of tissues and organs and increased risk of age-related diseases [15]. Brain aging is a complex process that affects the structural and functional connectivity of the brain. Morphologically, brain aging is characterized by volume loss, cortical thinning, white matter degeneration, loss of brain rotation, and ventricular enlargement. Pathologically, brain aging is related to neuronal cell atrophy, dendritic degeneration, demyelination, small-vessel disease, metabolic slowdown, microglia activation, and white matter lesions [19]. The mechanisms underlying these changes remain unclear, leading to a lack of effective therapies [23]. Epigenetic changes are considered to be important markers of aging and cellular senescence. Given the abundance and age-related changes in RNA m6A methylation in the CNS, it must play an important role in aging and degenerative neurological diseases. Altered m6A methylation modifications and mutated RNA m6A methyltransferases are associated with a variety of neuropathological processes, providing a new dimension for the study of brain aging.
Regulation of ncRNAs by m6A modifications
m6A modification does not only occur in the ncRNA that can encode proteins, but also in miRNAs, and it plays an important regulatory role in the production and function of miRNAs (Figure 2C–2E; Table 4). ncRNA refers to functional RNA molecules that cannot be translated into proteins, including miRNAs, circRNAs, and lncRNAs.
Table 4. Regulation of ncRNAs by m6A modifications.
| ncRNA | m6A regulatory proteins | Mechanisms | Ref. |
| miRNAs | METTL3 | a. Marked pri-miRNA through m6A b. Increased Dicer splicing of pre-miRNAs | [103] |
| HNRNPA2B1 | Binded to m6A in pri-miRNA to recruit DGCR8, thereby promoting the maturation of miRNAs | [62] | |
| lncRNAs | HNRNPC and HNRNPG | Binded RNA sequences surrounding m6A in lncRNA MALAT1 | [61, 105] |
| — | Played a role in the lncRNA-miRNA interaction, thereby influencing miRNA level | [106] | |
| circRNAs | METTL3 and METTL14 | Enhanced m6A-driven circRNA translation | [107] |
| FTO | Inhibited m6A-driven circRNA translation | ||
| YTHDF3 and eIF4G2 | Played key roles in m6A-driven circRNA translation | ||
| YTHDF2 | Differentiated endogenous circRNA from exogenous circRNA and regulate innate immunity | [108, 109] |
miRNAs are non-coding single-stranded small RNAs (18–24 nucleotides in length), which regulate gene expression at the post-transcriptional level through the formation of an RNA-induced silencing complex (RISC) [101]. The generation and maturation of miRNAs are regulated by m6A. In the nucleus, DNA is first transcribed into primary miRNAs (pri-miRNAs), which are then processed into precursor miRNAs (pre-miRNAs) by a microprocessor complex containing DGCR8 and DROSHA. Pre-miRNAs are then cleaved using Dicer into mature single-stranded miRNAs in the cytoplasm [102]. In this process, METTL3 marks pri-miRNA through m6A, and HNRNPA2B1 binds to m6A in pri-miRNA to recruit DGCR8; this enables DGCR8 to recognize and bind to specific substrates, thereby promoting the maturation of miRNAs [62, 103]. METTL3 can also promote miRNA biosynthesis by increasing Dicer splicing of pre-miRNAs.
lncRNAs are non-coding RNAs with more than 200 nucleotides that play a regulatory role in chromosomal inactivation and modification, transcription, shearing, translation, and protein activity. lncRNAs can also adsorb miRNAs through sequence complementation and inhibit the targeted regulatory effect of miRNAs on mRNA [104]. m6A modification in lncRNAs may influence RNA-protein interactions. m6A in lncRNA may make the surrounding RNA sequences more likely to bind HNRNPC and HNRNPG [61, 105]. It also plays a role in the lncRNA-miRNA interaction, thereby influencing miRNA level [106].
circRNA is formed by the covalent binding of the 3′ end and 5′ end of precursor mRNA after reverse splicing and can participate in biological processes, such as the regulation of miRNA expression and gene transcription. m6A can promote circRNA translation, and m6A in circRNA can act as an internal ribosomal entry site (IRES) for cap-independent translation [107]. METTL3 and METTL14 enhance m6A-driven circRNA translation, whereas FTO-mediated m6A demethylation inhibits this translation process, with YTHDF3 and eIF4G2 playing key roles in this process. In addition, m6A also plays a role in circRNA immunity. Endogenous circRNA binding to YTHDF2 cannot activate RIG-I, whereas exogenous circRNAs lacking m6A modification can activate the RIG-I pathway, leading to interferon production and induction of innate immunity [108, 109]. This indicates that m6A can differentiate endogenous circRNAs from exogenous circRNAs and regulate innate immunity.
Regulation of m6A modifications by noncoding RNAs
m6A modification can regulate the generation and function of ncRNAs. Conversely, ncRNAs, as important functional regulatory molecules, can also influence m6A modification by interacting with or regulating m6A regulatory proteins and participating in the regulation of various physiological and pathophysiological processes (Figure 3). miR-493-3p and miR-145 can downregulate YTHDF2 mRNA, alter the intracellular m6A level, and ultimately inhibit the proliferation of prostate [110] and liver cancer cells [111]. However, miR-744-5p can silence HNRNPC, which influences miR-21 expression and Akt phosphorylation and ultimately promotes apoptosis [112]. miR-149-3p binds to the 3′UTR of FTO mRNA to downregulate its expression, inhibit adipogenic lineage differentiation, and enhance osteogenic lineage differentiation [113]. miR-141 regulates IGF2BP2 in PDAC, and IGF2BP2 activates the PI3K-Akt pathway in vivo and promotes pancreatic cancer growth [114]. miRNA let-7g inhibits breast cancer development by targeting the 3′-UTR of METTL3 mRNA [115]. miR-600 can also downregulate METTL3 and inhibit the proliferation and migration of lung cancer cells [116].
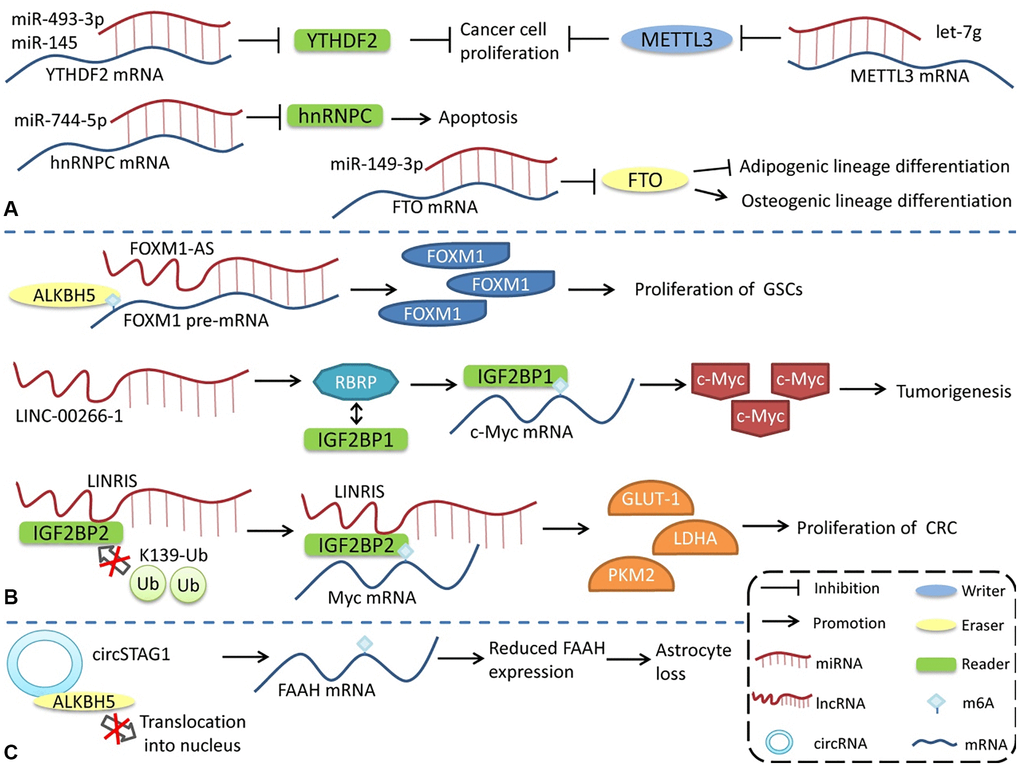
Figure 3. Regulation of m6A modifications by ncRNAs. ncRNAs can influence m6A modification by interacting with or regulating m6A regulatory proteins. (A) miRNA regulates the expression of m6A regulatory proteins. (B) lncRNA affects m6A modification. (C) circRNA interacts with m6A regulatory proteins to regulate m6A modification.
In contrast to the way miRNAs directly bind to m6A mRNA to regulate its expression, lncRNAs modulates m6A by interacting with m6A regulatory proteins. LncRNA LINC-00266-1-encoded RBRP interacts with IGF2BP1 to enhance the recognition of m6A on c-myc mRNA, enhance the stability and expression of c-myc mRNA, and promote tumorigenesis [117]. The long intergenic non-coding RNA LINRIS maintains the stability of IGF2BP2 by blocking K139 ubiquitination and promoting colorectal cancer (CRC) proliferation [118]. LINC01234 interacts with HNRNPA2B1, leading to the recruitment of DGCR8 and promoting the processing and maturation of miR-106b-5p, which inhibits CRY2 and promotes the growth of non-small cell lung cancer (NSCLC) cells [119]. FOXM1-AS is an antisense lncRNA of FOXM1 that promotes the interaction of ALKBH5 with newborn FOXM1 transcripts, regulates m6A modification and FOXM1 expression, and regulates the proliferation of glioblastoma stem cell-like cells (GSCs) [120]. LncRNA GAS5-AS, the antisense lncRNA of GAS5, can also interact with ALKBH5 to adjust the m6A modification of GAS5 to enhance its stability [121].
The regulation of circRNAs by m6A modification has also been reported. circSTAG1 can bind ALKBH5, inhibit its entry into the nucleus, upregulate the m6A modification level of FAAH mRNA, and affect its stability and expression, resulting in astrocyte dysfunction [122]. However, compared with miRNA and lncRNA, the regulation of m6A modification by circRNA has a broader research scope. Notably, some m6A regulatory proteins have multiple functions, and the effects of m6A regulatory proteins are not necessarily entirely attributable to the alterations in the level of m6A modification. However, more detailed studies are needed to identify the role of different pathways in various physiological and pathological processes.
In addition, m6A modification can not only regulate the function of exosomes [123], but exosomal ncRNAs can also enter target cells and influence diseases occurrence by regulating the expression of m6A-related proteins [124–126]. Yuan et al. demonstrated that human umbilical cord mesenchymal stem cell (hucMSC)-derived exosomal miR-26a-5p can enter the nucleus pulposus (NP) cells to inhibit pyroptotic NP cell death by targeting the METTL14/NLRP3 axis, thereby suppressing the progression of intervertebral disc degeneration (IVDD) [124].
Role of m6A modification and ncRNAs in TME
During tumorigenesis, tumor cells interact closely with stromal cells and immune cells in the microenvironment, jointly transforming the microenvironment into a site conducive to the growth, invasion, and metastasis of tumor. m6A-related ncRNA, as an important means of intracellular regulation of gene expression and functional status, as well as intercellular communication, can promote the formation and maturation of TME and play an important role in proliferation, angiogenesis, invasion, metastasis, immune escape, and other processes in tumor (Figure 4).
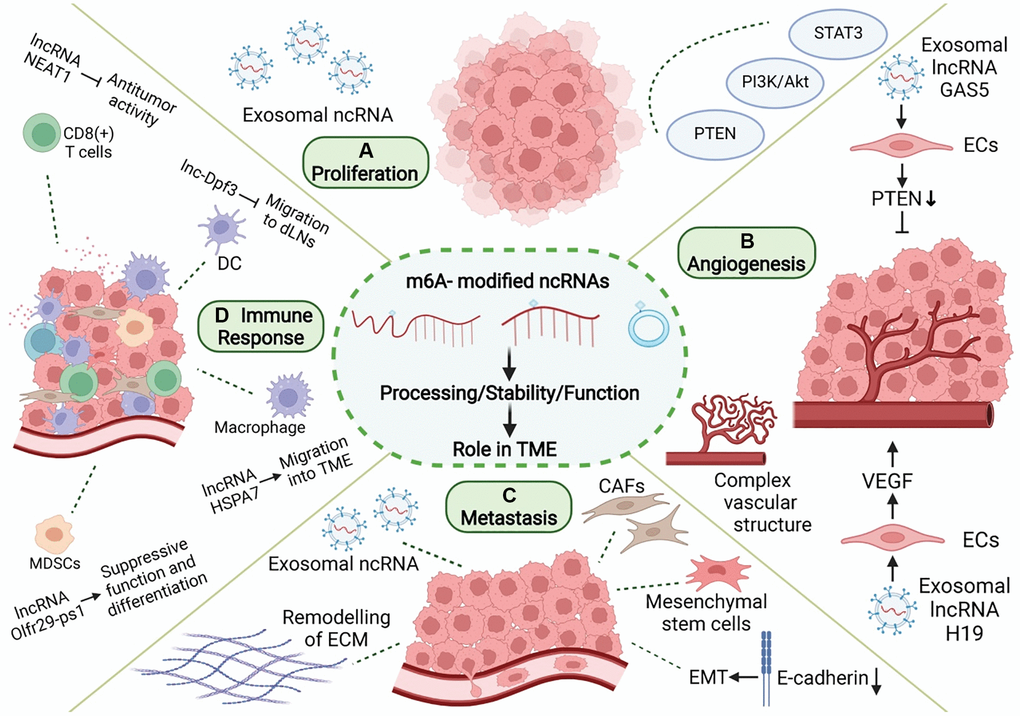
Figure 4. Role of m6A modification and ncRNAs in TME. In TME, m6A-related ncRNAs are involved in the regulation of proliferation, angiogenesis, invasion and metastasis, and immune escape in tumor. (A) m6A-related ncRNAs can affect tumor proliferation by regulating proliferation-related genes and signaling pathways, and can be transmitted in TME through exosomes. (B) m6A-related ncRNAs regulate tumor angiogenesis by regulating pro-angiogenic molecules, such as VEGF, and are involved in angiogenesis patterns of large vessels and complex vascular structures. (C) Tumor metastasis is related to EMT in tumor cells and the dissolution and remodeling of ECM. m6A-associated ncRNAs can also be delivered through exosomes, while regulating stromal cells in TME to promote metastasis. (D) m6A-related ncRNAs are involved in the recruitment, differentiation, and functional expression of immune cells in TME, and they promote the immune escape of tumor cells.
Mediation of tumor proliferation
Cell proliferation involves complex signal transduction. Uncontrolled proliferation of tumor cells is mostly related to the abnormality of proliferation-related genes and impaired signaling pathways involving cellular processes, such as oncogene activation, tumor suppressor gene inactivation, apoptosis resistance, and metabolic reprogramming [127, 128]. However, the malignant proliferation of tumors is related to tumor cells, and TME can also enhance this abnormal proliferation. In various malignant tumors, CAF in TME can promote tumor proliferation through the synthesis and secretion of TGF-β, fibroblast secretory protein 1 (FSP1), SDF-1, and other growth factors [129]. Mast cells can promote tumor proliferation through direct contact with tumor cells or the secretion of factors such as IL-17A [130].
m6A-related ncRNAs also play a regulatory role in tumor proliferation. m6A modification of ncRNAs can influence the expression of ncRNA by changing its stability or maturation process and regulating the expression of proliferation-related genes or signal pathways, thus promoting or inhibiting tumor proliferation. METTL3 promotes m6A-dependent miR-221/222 maturation and downregulates PTEN, thereby promoting the proliferation of bladder cancer cells [131]. Deoxycholic acid (DCA) levels are significantly decreased in gallbladder cancer (GBC) [132]. DCA can reduce the m6A level of pri-miR-92b, thereby reducing its expression and upregulating PTEN and consequently inhibiting tumor proliferation mediated by the PI3K/AKT pathway [132]. Moreover, m6A-related ncRNAs also play an important regulatory role in TME, which may serve as a bridge between tumor proliferation and TME formation. The signals of abnormal proliferation in a single tumor cell may also be transmitted to other tumor cells by m6A-associated ncRNAs through TME, thereby promoting the spread of malignant phenotypes. The links between these three factors have not been elucidated. ALKBH5 can mediate m6A demethylation of lncRNA PVT1 in osteosarcoma, thus upregulating PVT1 and promoting osteosarcoma cell proliferation [133]. PVT1 can also promote the transport and fusion of multivesicular bodies (MVB) to the plasma membrane, thus promoting exosome secretion by pancreatic cancer cells [134]. Exosomes are important intercellular communication molecules that can promote information transmission between tumor cells, and between tumor cells and the tumor matrix, thus promoting the formation of TME and tumor development. These results suggest that m6A-related ncRNAs may be involved in tumor development by regulating the formation of tumor cells and TME. IGF2BP2 promotes the stability and expression of ZFAS1 in an m6A-dependent manner [135]. In esophageal squamous cell carcinoma (ESCC), ZFAS1 can shuttle between tumor cells in the form of exosomes and act as ceRNA to downregulate miR-124, thereby upregulating STAT3 and ultimately inhibiting cellular apoptosis and promoting proliferation, migration, and invasion [136]. These results suggest that m6A-related ncRNA-mediated tumor proliferation may spread through exosomes in TME and increase tumor malignancy. In conclusion, tumor cells are closely related to other components of TME, and m6A-related ncRNAs can simultaneously regulate tumor proliferation and TME. Further studies are needed to confirm the potential crosstalk between the two factors.
Mediation of tumor angiogenesis
Tumor tissues require abundant blood flow to provide nutrients and excrete metabolites. In the process of tumor tissue growth and enlargement, the normal blood vessels of the original tissue are insufficient to support further growth. At this time, the components of TME can establish new blood circulation in the tumor tissue through complex signal communication. The rapid growth of tumor cells leads to a local anoxic microenvironment with metabolite accumulation, which promotes the formation of angiogenic mimicry and increases blood supply by promoting ECM remodeling and inducing the transformation of cancer stem cells (CSC) into endothelial phenotypes [137]. In addition, CAFs and TAMs in TME can also secrete angiogenic factors, such as VEGF, and cytokines, such as interleukin and free ncRNAs, into TME to activate the angiogenesis signaling pathway and promote tumor angiogenesis [138, 139].
m6A-related ncRNA is one of the components released by tumor cells into TME to promote angiogenesis and plays a regulatory role in the tumor angiogenesis microenvironment. MiR-155 can target FTO and upregulate m6A levels in clear cell renal cell carcinoma (RCC) [140]. However, melanoma-derived exosome miR-155 can be delivered to fibroblasts to downregulate SOCS1 and then activate the JAK2/STAT3 pathway, leading to upregulated VEGFa, FGF2, and MMP9 in the fibroblasts, enhanced reprogramming of fibroblasts into CAF, and improved angiogenesis promotion capacity [141]. m6A modification can regulate the expression of H19 and miRNA675 [142]. H19 is the precursor lncRNA of miRNA675, both of which regulate angiogenesis. MiRNA675-5p is involved in angiogenesis in anoxic microenvironments [143]. H19 is abundant in the exosomes secreted by CD90+ HCC cells. Through the adhesion of CD90 to human umbilical vein endothelial cells (HUVECs), exosomes can invade endothelial cells, deliver H19 to target cells, and stimulate angiogenesis through the synthesis and release of VEGF [144]. VIRMA is a regulatory component of m6A, and its knockout can reduce the stability and expression of lncRNA CCAT1 through decreased m6A level [145]. Pancreatic cancer (PC) cell-derived exosomes can transport CCAT1 to HUVEC. CCAT1 upregulates HMGA1 expression through competitive binding of miR-138-5p and enhances angiogenesis in HUVEC [146]. YTHDF3 promotes the degradation of lncRNA GAS5 in an m6A-dependent manner [147]. GAS5 is expressed at low levels in human lung cancer tissues, lung cancer cells, and cell culture supernatant exosomes. The overexpression of GAS5 in lung cancer cells can inhibit proliferation and tube formation in HUVECs in the form of exosomes, thereby affecting angiogenesis. The mechanism involves the regulation of PTEN expression, as well as PI3K and AKT phosphorylation through the “sponging” of miR-29-3p [148]. Therefore, tumor cells can increase the levels of pro-angiogenic m6A-related ncRNAs and decrease those of the anti-angiogenic m6A-related ncRNAs in secreted exosomes, thus regulating the angiogenesis process in TME. This provides a new research idea for anti-angiogenesis in tumor research. m6A modification of pri-miR-17-92 promotes its processing and maturation [149]. MiR-20a, a member of the miR-17-92 cluster, is associated with angiogenesis patterns in large vessels and complex vascular structures in breast cancer [150]. Tumor angiogenesis is an important event in TME, and changes in angiogenesis patterns may affect nutrient supply to various cells in TME, thus affecting their functional status. In addition, m6A modification can promote the splicing of the precursor miR-143-3p to promote its maturation. MiR-143-3p increases VEGFA expression in lung cancer cells by downregulating VASH1 and promoting angiogenesis [151]. miR-143-3p is highly expressed in the EVs of osteosarcoma cells and involved in the regulation of TME [152]. The specific role of miR-143-3p in TME is still unclear, and further research is needed to determine if miR-143-3p can also affect tumor angiogenesis by regulating TME. In conclusion, the multiple roles of m6A-related ncRNAs in TME may involve a regulatory network that modulates angiogenesis and tumor progression through multiple pathways.
Mediation of tumor metastasis
Tumor metastasis is closely related to EMT as well as the adhesion, invasion, and migration of tumor cells [153, 154]. The reduction in adhesion molecules on the surface of tumor cells allows cells to separate from each other, facilitating cell mobility. The ECM is a key regulator of tumor invasion and metastasis. Tumor or stromal cells can produce proteases (such as MMP) to dissolve ECM components and promote tumor cell migration. EMT is the transformation of tumor cells from an epithelial to a more aggressive mesenchymal phenotype, which promotes tumor metastasis. During EMT, E-cadherin and other cell adhesion proteins are gradually reduced, while N-cadherin, vimentin, and other mesenchymal marker proteins are enriched. Tumor metastasis is closely related to the enhanced invasion and metastatic ability of tumor cells as well as changes in TME. TME can promote tumor metastasis by promoting EMT and the invasion ability of tumor cells, promoting ECM remodeling and the formation of a premetastatic niche [155].
m6A-related ncRNAs play a significant regulatory role in many aspects of tumor metastasis. First, m6A-related ncRNAs can influence tumor invasion and metastasis by regulating the expression of metastasis-related genes. m6A modification can promote the splicing and maturation of miR-143-3p, while miR-143-3p can downregulate E-Cadherin and upregulate FN, vimetin, MMP2, and MMP-9 in lung cancer cells to promote the occurrence of EMT [151]. In nasopharyngeal carcinoma, m6A modification can enhance the stability of lncRNA FAM225A. As ceRNA, FAM225A inhibits the expression of miR-590-3p and miR-1275 and upregulates downstream ITGB3, promoting proliferation, migration, and invasion of cancer cells through the FAK/PI3K/Akt pathway [156]. In addition, future studies need to investigate if m6A-related ncRNAs regulate tumor metastasis by modulating TME. Small extracellular vesicles (sEV) derived from gastric cancer (GC) containing miR-151a-3p can be absorbed by hepatic Kupffer cells (KCs), and sEV-miR-151a-3p targets YTHDF3 and downregulates SUMO1 translation in an m6A-dependent manner, thereby inhibiting SP3, inducing TGF-β1 expression, and promoting the formation of a local liver metastasis microenvironment through the activation of the SMAD2/3 pathway [157]. METTL3 promotes the maturation of pri-miR-320b in an m6A-dependent manner, and miR-320b promotes EMT by downregulating PDCD4 and can be included in exosomes to promote lymphangiogenesis, thereby promoting ESCC metastasis [158]. LncRNA XIST is misexpressed in a variety of tumors and can regulate the malignant phenotype of tumors. YTHDF2 recognizes m6A modifications in XIST and mediates their degradation [159]. The deletion of XIST in breast cancer can promote the phenotypic transformation of microglia through the transport of miR-503 exosomes to microglia, leading to local immunosuppression and promoting brain metastasis in breast cancer [160]. ALKBH5 can upregulate NEAT1 by mediating m6A demethylation, thereby influencing the expression of EZH2 and inhibiting the invasion and metastasis of GC cells [161]. Bone metastases from prostate cancer are typically osteogenic. NEAT1 can be transferred to human bone marrow-derived mesenchymal stem cells (hBMSCs) through exosomes secreted by prostate cancer cells, thereby upregulating RUNX2, promoting osteogenic differentiation [162], and participating in the formation of the local bone metastasis microenvironment. METTL3 and YTHDF3 increase the stability of MALAT1 in an m6A-dependent manner, and MALAT1 acts as ceRNA to “sponge” miR-1914-3p, thereby upregulating YAP and promoting NSCLC invasion and metastasis [163]. In a GC study, MALAT1 increased the accumulation of SQSTM1 in tumor cells, which in turn activated NF-κB and increased the expression of IL-6. IL-6 promotes paracrine transformation of CAF [164]. CAF secrete various cytokines to promote tumor metastasis. This suggests that m6A-related ncRNAs can not only change the gene expression of tumor cells to enhance their ability to invade and metastasize but also promote tumor metastasis by inducing the formation of TME. Tumor cells and TME carry out complex information transmission to synergistically promote tumor invasion and metastasis, and m6A-related ncRNAs play an important mediating role in this process. However, further research is needed to gain clearer understanding of the regulatory network of TME in different tumor types.
Mediation of tumor immune response
Under normal circumstances, the immune surveillance function of the immune system can promptly detect and eliminate tumor cells caused by gene mutations; however, the formation of TME can protect tumor cells from the immune system. In TME, the recruitment, differentiation, and functional expression of immune cells are precisely regulated, forming a low anti-tumor immune response microenvironment to promote the immune escape of tumor cells. m6A modification plays an important role in maintaining the normal function of immune cells, and changes in m6A modification can affect the formation of an immunosuppressive microenvironment in tumors [165]. In DC, METTL3-mediated m6A modification promotes the expression of the costimulatory molecules, CD40 and CD80, and the proinflammatory cytokine IL-12, and promotes DC activation and maturation [166]. YTHDF1 recognizes the m6A modification of lysosomal protease transcription and promotes its expression, which in turn blocks cross-presentation of tumor antigens by DC and antigen-specific activation of CD8+ T cells [167]. METTL3 can also improve the stability of STAT1 mRNA by mediating m6A modifications, upregulating STAT1, and driving the polarization of M1 macrophages [168]. FTO knockout inhibits NF-κB signaling and decreases the stability of STAT1 and PPAR-γ mRNA, thus impeding macrophage activation [169]. In mouse models, METTL3 deficiency upregulated the mRNA expression of SOCS family by modulating mRNA m6A modification, thus inhibiting IL-7-mediated STAT5 activity and the homeostasis, proliferation, and differentiation of T cells [170]. METTL14/YTHDF1 knockdown in GC cells downregulated IFN-α, -β, and-γ transcription levels [171]. As m6A modification is ubiquitous in cells, its changes often involve alterations in multiple genes and signaling pathways, providing clues for subsequent in-depth studies.
m6A-related ncRNAs can regulate the migration, differentiation, and functional states of various immune cells, making the regulatory network of the tumor immune microenvironment more detailed and precise. The lncRNA Olfr29-ps1 can interact with miR-214-3p to inhibit its expression, thereby promoting the immunosuppressive function and differentiation of mononuclear MDSCs. In GM-CSF + IL6-induced MDSCs, Olfr29-ps1 was modified by m6A, which enhanced the regulation of Olfr29-ps1 on MDSCs [172]. The lncRNA HSPA7 is significantly overexpressed in glioblastoma (GBM) tissues, and m6A modification of HSPA7 promotes its expression. HSPA7 promotes macrophage migration to the GBM TME by activating the YAP1-LOX axis and can promote an immunosuppressive phenotype and inhibit the anti-tumor immune response [173]. In colorectal cancer, METTL3 upregulated the expression of miR-1246 by promoting the maturation of pri-miR-1246 through methylation [174]. However, exosomes shed by mutant p53 colon cancer can carry miR-1246 to the microenvironment containing macrophages, where it reprograms macrophages and recruits immunosuppressive T cells to promote tumor development [175]. DC migration to draining lymph nodes (DLNs) plays an important role in the initiation of anti-tumor T cells and adaptive immunity. CCR7 activation can prevent lnc-Dpf3 degradation by removing its m6A modification. Lnc-Dpf3 is upregulated after recognition by YTHDF2. Upregulated lnc-Dpf3 can directly bind to HIF-1α protein and inhibit the transcription of Ldha, an HIF-1α dependent glycolysis gene, thus inhibiting glycolysis metabolism in DCs and CC7-mediated DC migration to DLNs, which plays an immunosuppressive role in tumors [176]. In GC, ALKBH5 can upregulate lncRNA NEAT1 through demethylation [161]. In HCC, the downregulation of NEAT1 can enhance the anti-tumor function of CD8+ T cells [177]. m6A modification of circIGF2BP3 upregulates its expression by promoting its reverse splicing and cyclization. circIGF2BP3 upregulates PKP3 through competitive binding of miR-328-3p and miR-3173-5p, thereby inhibiting PD-L1 ubiquitination and promoting the immune escape of NSCLC cells [178]. As immunotherapy has become a widely used anti-tumor therapy, these studies undoubtedly provide a new research direction for precise tumor therapy.
Potential clinical application of m6A-modified ncRNAs in cancers
m6A-modified ncRNAs as therapeutic targets
The important role of m6A-related ncRNAs in TME makes them a new target for anti-tumor therapy. Because m6A methylation and demethylation are reversible processes, it is possible to change the expression of m6A-modified ncRNA by targeting m6A regulatory proteins, thus exerting an anti-tumor therapeutic effect. R-2HG can inhibit FTO by increasing m6A levels and suppressing leukemia cell proliferation. R-2HG combined with chemotherapy had a synergistic anticancer effect in a mouse leukemia model [194]. Meclofenamic acid is another FTO inhibitor [195], and its derivative, MA2, upregulates m6A levels in glioblastoma stem cells. In a glioblastoma mouse model, intratumoral injection of MA2 inhibited tumor growth and extended survival [196]. However, the unique advantages and disadvantages of ncRNA in animal and clinical trials can provide a reference for the development of m6A-related ncRNA-targeted drugs. The knockdown of lncRNA MALAT1 using an antisense oligonucleotide technique in a mouse breast cancer model resulted in decreased tumor growth and reduced metastasis [197]. MRX34 is an analog of tumor suppressor miR-34a and significantly inhibits tumor growth in mouse models [198]. In phase I clinical trials in patients with advanced solid tumors, partial responses (PR) or stable disease (SD) were achieved in some patients [199, 200], but further studies are needed to reduce immune-related toxicity of miRNA [200]. Intratumoral injection of circNRIP1 siRNA inhibited tumor growth in a GC mouse model [201]. In addition, a circRNA vaccine against SARS-COV-2 induced effective immune responses in both mouse and monkey models [202]. However, it is not clear if circRNA vaccines can be used to induce anti-tumor immunity.
The regulation of m6A-related ncRNAs in TME makes them a potential therapeutic target for tumor growth inhibition and enhanced sensitivity to chemotherapy. m6A modification can upregulate LINC00958 by enhancing its stability, and LINC00958 can promote the proliferation, migration, invasion, and adipogenesis of HCC through the miR-3619-5p/HDGF axis. PEGylated PLGA nanoparticles loaded with si-LINC00958 effectively inhibited tumor growth in a patient-derived xenograft (PDX) mouse model, significantly prolonged the overall survival of the mice, and showed no significant toxicity to the liver, kidney, lung, spleen, or heart [203] (Figure 5). The upregulation of m6A levels at specific sites of circRNA-SORE in HCC cells can enhance RNA stability, lead to upregulation of circRNA-SORE, and ultimately promote sorafenib resistance in cancer cells. In animal models, the local injection of circRNA-SORE interfering RNA significantly improved the efficacy of sorafenib [204]. High-risk human papillomavirus produces m6A-modified circRNA circE7, which translates into the E7 oncoprotein and promotes cell transformation. After circE7 knockdown by shRNA, tumor growth was inhibited both in vitro and in a xenograft, mouse model [205]. The practical application value of m6A-related ncRNAs as anti-tumor therapeutic targets remains to be further explored.
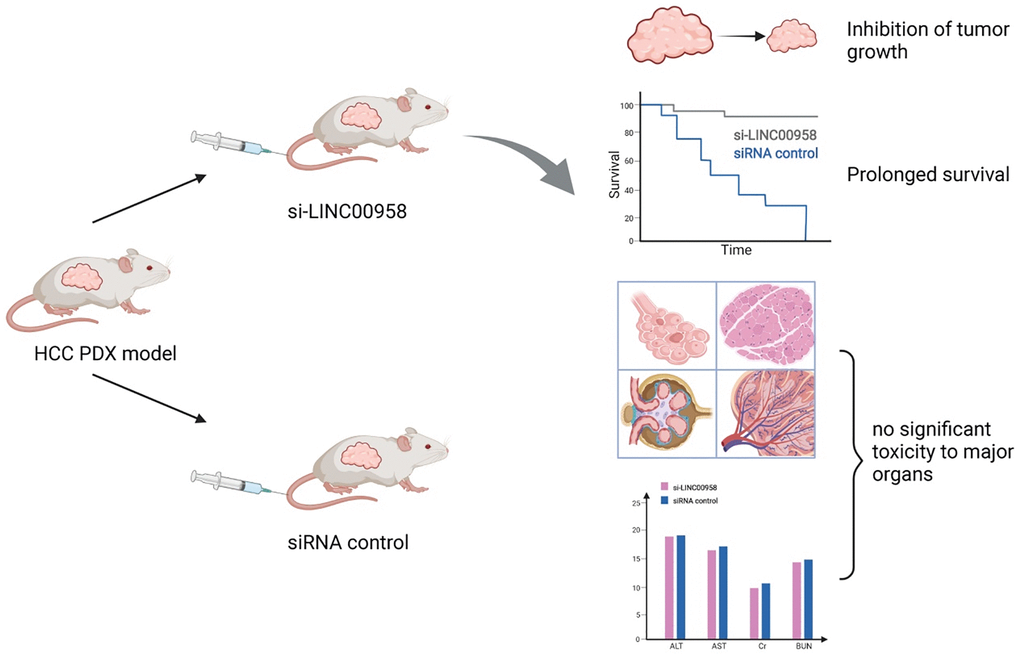
Figure 5. Schematic diagram of a patient-derived xenograft mouse model of liver cancer treated with si-LINC00958. A drug system loaded with si-LINC00958 effectively inhibited tumor growth in the PDX mouse model and significantly prolonged the overall survival of the mice. The systemic toxicity of the drug was evaluated using hematoxylin and eosin staining and blood indexes, and there was no evident toxicity to the liver, kidney, lung, spleen, and heart.
Hypoxia and m6A modification
Excessive distance between the vascular system and tumor cells can lead to diffuse hypoxia [93]. Under the condition of excessive tissue hypoxia, the homeostasis of the microenvironment is destroyed, resulting in hypoxia, hypoglycemia and acidic TME, which is conducive to tumor growth. Notably, hypoxia and tumor growth form a mutually positive feedback loop. Tumor cell proliferation leads to excessive oxygen depletion in Tmes and promotes anoxic environment in Tmes, thus providing suitable conditions for the occurrence and metastasis of tumors through proliferation, differentiation, drug resistance and other ways [116]. Cell hypoxia is mainly controlled by hypoxia-inducible factors (HIF), and heterodimer helix-loop-helix proteins composed of O2-unstable α and constitutive expressed β subunits (HIF1α, HIF2α and HIF-1β) participate in the coordination and regulation of many mechanisms to adapt tumor cells to the harsh environment [123]. HIF-1α and HIF-2α mainly recognize similar hypoxia response elements in target gene promoters and play a stable role in anoxic environment. HIF activates genes that control oxygen homeostasis in cells, including those involved in glucose metabolism and lactic acid metabolism. These molecular changes lead to glycolysis rather than oxidative metabolism to complete tumor adaptation [164].
Accurate m6A level is crucial for behavior and electrophysiological properties of mouse cortex in response to acute stress [135]. Moreover, m6A also plays a vital role in cellular response to external stimuli such as viral infection [136, 137], DNA damage [138] and heat shock response [139, 140]. For example, m6A modifications on transcripts rapidly recruit DNA polymerase to ultraviolet (UV) induced damage sites to facilitate DNA repair and cell survival [141]. m6A pathway may be important for hypoxic regulation by HIFs. Previous studies reported that hypoxic induction of ALKBH5 was dependent on HIFs and contributed to the breast cancer stem cell phenotype [142]. In the hypoxia/reoxygenation-treated cardiomyocytes, METTL3 is responsible for inhibiting autophagic flux and promoting apoptosis [143]. However, detailed regulatory mechanisms of cellular response to hypoxia by m6A pathway are still unclear.
Transcriptional activation by HIFs is the main pathway for hypoxic adaptation. Consistent with a prior report [144], a study by Wang et al. [145] observed up-regulation of ALKBH5 upon hypoxic stress. Moreover, the total m6A level in poly(A)+ RNAs was downregulated during this process. Down-regulation of total m6A levels of mRNA and protein levels of m6A readers under hypoxic condition might promote mRNA stability, reducing the need to produce new mRNAs. Upon hypoxic stress, HIF1A is stabilized, enters the nucleus, heterodimers with HIF1B to bind to the HRE elements in the promoters and activates target genes including ALKBH5. Considering that the repertoire of m6A regulator is far from complete, new m6A regulators are continually to be identified [146], we speculate that additional regulators are also involved in regulation of hypoxia. Reprogramming of m6A epitranscriptome further reshapes transcriptome and proteome to promote glycolysis and gluconeogenesis, and inhibit mitochondria oxidative respiratory chain, facilitating cells to response efficiently to hypoxia.
Moreover, many studies have reported that m6A genes participate in the formation of a hypoxic microenvironment. For example, FTO, an m6A regulator, promoted the progression of hypoxic TME formation via effects on glucose metabolism through FOXO1 mRNA expression. In turn, tumor hypoxia regulated the function of m6A reader YTHDF1 to drive the malignancy of hepatocellular carcinoma. Hypoxia has been reported to reprogram the chromatin by inducing changes in histone methylation to determine transcriptional activity, a process independent of HIFs [147, 148]. Meanwhile, m6A deposition was reported to occur cotranscriptionally guided by H3K36me3 (histone H3 trimethylation at lysine 36) [149]. Further studies of hypoxic stress are warranted to investigate whether m6A modification is involved in histone methylation-regulated transcripts, which may enhance our understandings of the molecular mechanism of hypoxia.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analysed during the current study.
Conclusions and perspectives
In recent years, with in-depth research on the mechanism of tumor development and anti-tumor therapy, m6A-related ncRNAs have received increasing attention because of their key roles in tumor growth, invasion, angiogenesis, metastasis, and immune tolerance, which is mediated by TME. The interaction between ncRNA and m6A has enriched our understanding of the complex regulatory network in TME, providing a new direction for studies on the role of post-transcriptional regulation in tumors. This interaction is a potential target for tumor diagnosis, prognosis evaluation, and treatment. Currently, the understanding of the role of m6A-related ncRNAs in TME is very limited, and more studies are needed to further reveal its regulatory mechanism and biological effects. In addition, the following limitations still exist: (1) Although m6A modification is widely present in all types of RNAs, current studies mainly focus on mRNA m6A modification, and many m6A-related ncRNAs have not been identified. (2) Because the role of m6A-related ncRNAs in tumors is still an emerging field, the genetic/epigenetic heterogeneity of different tumor cell lines and samples may lead to different results, which needs to be clarified by conducting follow-up studies. m6A-related ncRNAs show different functions in different tumors, which may be related to different downstream pathways, requiring further verification. (3) There are other kinds of RNA modifications besides m6A, such as m5C(5-methylcytidine), m6Am (N6-2′-O-dimethyladenosine), Ψ (pseudouridine), m1A (1-methyladenosine), m7G (7-methylguanosine), and hm5C (5-hydroxymethylcytidine). However, only a few of these have been studied; hence, it is necessary to consider the interaction between m6A and other modifications. Changes in specific phenotypes may not be entirely caused by alterations to a single type of RNA modification. (4) In terms of clinical applications, there is still a lack of specific methods for clinical diagnosis and treatment. As potential tumor markers, the diagnostic sensitivity and specificity of m6A-related ncRNAs need to be further validated, and their reference ranges in body fluids need to be determined. Sample handling and detection methods must be standardized. As a potential anti-tumor therapeutic target, more studies are needed to evaluate its efficacy and side effects and determine the appropriate administration method and dose. The role of m6A-related ncRNAs in TME is an exciting area of research. Clarifying the molecular mechanism of interaction between m6A-related ncRNA and the microenvironment, designing targeted drugs based on its complex and fine regulatory network, and combining it with traditional anti-tumor therapies to achieve improved efficacy and minimized side effects for individual precision medicine will be a major breakthrough in cancer research.
Author Contributions
Li Yin supervised the project, conceived and edited the manuscript. YanJun Zhang, Lijuan Zhan, Jing Li, Xue Jiang and Li Yin drafted the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This research was supported by the National Natural Science Foundation of China (81960658).
References
- 1. Nie H, Liao Z, Wang Y, Zhou J, He X, Ou C. Exosomal long non-coding RNAs: Emerging players in cancer metastasis and potential diagnostic biomarkers for personalized oncology. Genes Dis. 2020; 8:769–80. https://doi.org/10.1016/j.gendis.2020.12.004 [PubMed]
- 2. Zhang Q, Wang W, Zhou Q, Chen C, Yuan W, Liu J, Li X, Sun Z. Roles of circRNAs in the tumour microenvironment. Mol Cancer. 2020; 19:14. https://doi.org/10.1186/s12943-019-1125-9 [PubMed]
- 3. Cantara WA, Crain PF, Rozenski J, McCloskey JA, Harris KA, Zhang X, Vendeix FA, Fabris D, Agris PF. The RNA Modification Database, RNAMDB: 2011 update. Nucleic Acids Res. 2011; 39:D195–201. https://doi.org/10.1093/nar/gkq1028 [PubMed]
- 4. Yang Y, Li Q, Ling Y, Leng L, Ma Y, Xue L, Lu G, Ding Y, Li J, Tao S. m6A eraser FTO modulates autophagy by targeting SQSTM1/P62 in the prevention of canagliflozin against renal fibrosis. Front Immunol. 2023; 13:1094556. https://doi.org/10.3389/fimmu.2022.1094556 [PubMed]
- 5. Wu Y, Bao W, Ren J, Li C, Chen M, Zhang D, Zhu A. Integrated Profiles of Transcriptome and mRNA m6A Modification Reveal the Intestinal Cytotoxicity of Aflatoxin B1 on HCT116 Cells. Genes (Basel). 2022; 14:79. https://doi.org/10.3390/genes14010079 [PubMed]
- 6. Ma S, Chen C, Ji X, Liu J, Zhou Q, Wang G, Yuan W, Kan Q, Sun Z. The interplay between m6A RNA methylation and noncoding RNA in cancer. J Hematol Oncol. 2019; 12:121. https://doi.org/10.1186/s13045-019-0805-7 [PubMed]
- 7. Nie H, Wang Y, Liao Z, Zhou J, Ou C. The function and mechanism of circular RNAs in gastrointestinal tumours. Cell Prolif. 2020; 53:e12815. https://doi.org/10.1111/cpr.12815 [PubMed]
- 8. Wang Y, Nie H, He X, Liao Z, Zhou Y, Zhou J, Ou C. The emerging role of super enhancer-derived noncoding RNAs in human cancer. Theranostics. 2020; 10:11049–62. https://doi.org/10.7150/thno.49168 [PubMed]
- 9. Peng L, Wang D, Han Y, Huang T, He X, Wang J, Ou C. Emerging Role of Cancer-Associated Fibroblasts-Derived Exosomes in Tumorigenesis. Front Immunol. 2022; 12:795372. https://doi.org/10.3389/fimmu.2021.795372 [PubMed]
- 10. Zhou H, He X, He Y, Ou C, Cao P. Exosomal circRNAs: Emerging Players in Tumor Metastasis. Front Cell Dev Biol. 2021; 9:786224. https://doi.org/10.3389/fcell.2021.786224 [PubMed]
- 11. Mueller MM, Fusenig NE. Friends or foes - bipolar effects of the tumour stroma in cancer. Nat Rev Cancer. 2004; 4:839–49. https://doi.org/10.1038/nrc1477 [PubMed]
- 12. Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R, Carey VJ, Richardson AL, Weinberg RA. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell. 2005; 121:335–48. https://doi.org/10.1016/j.cell.2005.02.034 [PubMed]
- 13. Erez N, Glanz S, Raz Y, Avivi C, Barshack I. Cancer associated fibroblasts express pro-inflammatory factors in human breast and ovarian tumors. Biochem Biophys Res Commun. 2013; 437:397–402. https://doi.org/10.1016/j.bbrc.2013.06.089 [PubMed]
- 14. Ren Y, Jia HH, Xu YQ, Zhou X, Zhao XH, Wang YF, Song X, Zhu ZY, Sun T, Dou Y, Tian WP, Zhao XL, Kang CS, Mei M. Paracrine and epigenetic control of CAF-induced metastasis: the role of HOTAIR stimulated by TGF-ß1 secretion. Mol Cancer. 2018; 17:5. https://doi.org/10.1186/s12943-018-0758-4 [PubMed]
- 15. Zhao L, Ji G, Le X, Wang C, Xu L, Feng M, Zhang Y, Yang H, Xuan Y, Yang Y, Lei L, Yang Q, Lau WB, et al. Long Noncoding RNA LINC00092 Acts in Cancer-Associated Fibroblasts to Drive Glycolysis and Progression of Ovarian Cancer. Cancer Res. 2017; 77:1369–82. https://doi.org/10.1158/0008-5472.CAN-16-1615 [PubMed]
- 16. Yamada K, Maishi N, Akiyama K, Towfik Alam M, Ohga N, Kawamoto T, Shindoh M, Takahashi N, Kamiyama T, Hida Y, Taketomi A, Hida K. CXCL12-CXCR7 axis is important for tumor endothelial cell angiogenic property. Int J Cancer. 2015; 137:2825–36. https://doi.org/10.1002/ijc.29655 [PubMed]
- 17. Sruthi TV, Edatt L, Raji GR, Kunhiraman H, Shankar SS, Shankar V, Ramachandran V, Poyyakkara A, Kumar SVB. Horizontal transfer of miR-23a from hypoxic tumor cell colonies can induce angiogenesis. J Cell Physiol. 2018; 233:3498–514. https://doi.org/10.1002/jcp.26202 [PubMed]
- 18. Yadav A, Kumar B, Yu JG, Old M, Teknos TN, Kumar P. Tumor-Associated Endothelial Cells Promote Tumor Metastasis by Chaperoning Circulating Tumor Cells and Protecting Them from Anoikis. PLoS One. 2015; 10:e0141602. https://doi.org/10.1371/journal.pone.0141602 [PubMed]
- 19. Zhang L, Lv Z, Xu J, Chen C, Ge Q, Li P, Wei D, Wu Z, Sun X. MicroRNA-134 inhibits osteosarcoma angiogenesis and proliferation by targeting the VEGFA/VEGFR1 pathway. FEBS J. 2018; 285:1359–71. https://doi.org/10.1111/febs.14416 [PubMed]
- 20. Zhou L, Zhu Y, Sun D, Zhang Q. Emerging Roles of Long non-coding RNAs in The Tumor Microenvironment. Int J Biol Sci. 2020; 16:2094–103. https://doi.org/10.7150/ijbs.44420 [PubMed]
- 21. Pitulescu ME, Schmidt I, Giaimo BD, Antoine T, Berkenfeld F, Ferrante F, Park H, Ehling M, Biljes D, Rocha SF, Langen UH, Stehling M, Nagasawa T, et al. Dll4 and Notch signalling couples sprouting angiogenesis and artery formation. Nat Cell Biol. 2017; 19:915–27. https://doi.org/10.1038/ncb3555 [PubMed]
- 22. Noguera-Troise I, Daly C, Papadopoulos NJ, Coetzee S, Boland P, Gale NW, Lin HC, Yancopoulos GD, Thurston G. Blockade of Dll4 inhibits tumour growth by promoting non-productive angiogenesis. Nature. 2006; 444:1032–7. https://doi.org/10.1038/nature05355 [PubMed]
- 23. Talmadge JE, Gabrilovich DI. History of myeloid-derived suppressor cells. Nat Rev Cancer. 2013; 13:739–52. https://doi.org/10.1038/nrc3581 [PubMed]
- 24. Gao Y, Wang T, Li Y, Zhang Y, Yang R. Lnc-chop Promotes Immunosuppressive Function of Myeloid-Derived Suppressor Cells in Tumor and Inflammatory Environments. J Immunol. 2018; 200:2603–14. https://doi.org/10.4049/jimmunol.1701721 [PubMed]
- 25. Gao Y, Sun W, Shang W, Li Y, Zhang D, Wang T, Zhang X, Zhang S, Zhang Y, Yang R. Lnc-C/EBPβ Negatively Regulates the Suppressive Function of Myeloid-Derived Suppressor Cells. Cancer Immunol Res. 2018; 6:1352–63. https://doi.org/10.1158/2326-6066.CIR-18-0108 [PubMed]
- 26. Umansky V, Sevko A. Tumor microenvironment and myeloid-derived suppressor cells. Cancer Microenviron. 2013; 6:169–77. https://doi.org/10.1007/s12307-012-0126-7 [PubMed]
- 27. Hoechst B, Ormandy LA, Ballmaier M, Lehner F, Krüger C, Manns MP, Greten TF, Korangy F. A new population of myeloid-derived suppressor cells in hepatocellular carcinoma patients induces CD4(+)CD25(+)Foxp3(+) T cells. Gastroenterology. 2008; 135:234–43. https://doi.org/10.1053/j.gastro.2008.03.020 [PubMed]
- 28. Parker KH, Beury DW, Ostrand-Rosenberg S. Myeloid-Derived Suppressor Cells: Critical Cells Driving Immune Suppression in the Tumor Microenvironment. Adv Cancer Res. 2015; 128:95–139. https://doi.org/10.1016/bs.acr.2015.04.002 [PubMed]
- 29. Gabrilovich DI. Myeloid-Derived Suppressor Cells. Cancer Immunol Res. 2017; 5:3–8. https://doi.org/10.1158/2326-6066.CIR-16-0297 [PubMed]
- 30. Davies LC, Jenkins SJ, Allen JE, Taylor PR. Tissue-resident macrophages. Nat Immunol. 2013; 14:986–95. https://doi.org/10.1038/ni.2705 [PubMed]
- 31. Mosser DM, Edwards JP. Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 2008; 8:958–69. https://doi.org/10.1038/nri2448 [PubMed]
- 32. Qian BZ, Pollard JW. Macrophage diversity enhances tumor progression and metastasis. Cell. 2010; 141:39–51. https://doi.org/10.1016/j.cell.2010.03.014 [PubMed]
- 33. Dirkx AE, Oude Egbrink MG, Wagstaff J, Griffioen AW. Monocyte/macrophage infiltration in tumors: modulators of angiogenesis. J Leukoc Biol. 2006; 80:1183–96. https://doi.org/10.1189/jlb.0905495 [PubMed]
- 34. Chen C, He W, Huang J, Wang B, Li H, Cai Q, Su F, Bi J, Liu H, Zhang B, Jiang N, Zhong G, Zhao Y, et al. LNMAT1 promotes lymphatic metastasis of bladder cancer via CCL2 dependent macrophage recruitment. Nat Commun. 2018; 9:3826. https://doi.org/10.1038/s41467-018-06152-x [PubMed]
- 35. Wang S, Liang K, Hu Q, Li P, Song J, Yang Y, Yao J, Mangala LS, Li C, Yang W, Park PK, Hawke DH, Zhou J, et al. JAK2-binding long noncoding RNA promotes breast cancer brain metastasis. J Clin Invest. 2017; 127:4498–515. https://doi.org/10.1172/JCI91553 [PubMed]
- 36. Cao J, Dong R, Jiang L, Gong Y, Yuan M, You J, Meng W, Chen Z, Zhang N, Weng Q, Zhu H, He Q, Ying M, Yang B. LncRNA-MM2P Identified as a Modulator of Macrophage M2 Polarization. Cancer Immunol Res. 2019; 7:292–305. https://doi.org/10.1158/2326-6066.CIR-18-0145 [PubMed]
- 37. Chen X, Zhou J, Li X, Wang X, Lin Y, Wang X. Exosomes derived from hypoxic epithelial ovarian cancer cells deliver microRNAs to macrophages and elicit a tumor-promoted phenotype. Cancer Lett. 2018; 435:80–91. https://doi.org/10.1016/j.canlet.2018.08.001 [PubMed]
- 38. Hao NB, Lü MH, Fan YH, Cao YL, Zhang ZR, Yang SM. Macrophages in tumor microenvironments and the progression of tumors. Clin Dev Immunol. 2012; 2012:948098. https://doi.org/10.1155/2012/948098 [PubMed]
- 39. Dubsky P, Ueno H, Piqueras B, Connolly J, Banchereau J, Palucka AK. Human dendritic cell subsets for vaccination. J Clin Immunol. 2005; 25:551–72. https://doi.org/10.1007/s10875-005-8216-7 [PubMed]
- 40. Wang P, Xue Y, Han Y, Lin L, Wu C, Xu S, Jiang Z, Xu J, Liu Q, Cao X. The STAT3-binding long noncoding RNA lnc-DC controls human dendritic cell differentiation. Science. 2014; 344:310–3. https://doi.org/10.1126/science.1251456 [PubMed]
- 41. Ji J, Yin Y, Ju H, Xu X, Liu W, Fu Q, Hu J, Zhang X, Sun B. Long non-coding RNA Lnc-Tim3 exacerbates CD8 T cell exhaustion via binding to Tim-3 and inducing nuclear translocation of Bat3 in HCC. Cell Death Dis. 2018; 9:478. https://doi.org/10.1038/s41419-018-0528-7 [PubMed]
- 42. Declerck S, Vansteenkiste J. Immunotherapy for lung cancer: ongoing clinical trials. Future Oncol. 2014; 10:91–105. https://doi.org/10.2217/fon.13.166 [PubMed]
- 43. Jia L, Xi Q, Wang H, Zhang Z, Liu H, Cheng Y, Guo X, Zhang J, Zhang Q, Zhang L, Xue Z, Li Y, Da Y, et al. miR-142-5p regulates tumor cell PD-L1 expression and enhances anti-tumor immunity. Biochem Biophys Res Commun. 2017; 488:425–31. https://doi.org/10.1016/j.bbrc.2017.05.074 [PubMed]
- 44. Raphael I, Nalawade S, Eagar TN, Forsthuber TG. T cell subsets and their signature cytokines in autoimmune and inflammatory diseases. Cytokine. 2015; 74:5–17. https://doi.org/10.1016/j.cyto.2014.09.011 [PubMed]
- 45. Josefowicz SZ, Lu LF, Rudensky AY. Regulatory T cells: mechanisms of differentiation and function. Annu Rev Immunol. 2012; 30:531–64. https://doi.org/10.1146/annurev.immunol.25.022106.141623 [PubMed]
- 46. Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P, Evdemon-Hogan M, Conejo-Garcia JR, Zhang L, Burow M, Zhu Y, Wei S, Kryczek I, et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004; 10:942–9. https://doi.org/10.1038/nm1093 [PubMed]
- 47. Belkaid Y, Oldenhove G. Tuning microenvironments: induction of regulatory T cells by dendritic cells. Immunity. 2008; 29:362–71. https://doi.org/10.1016/j.immuni.2008.08.005 [PubMed]
- 48. Pei X, Wang X, Li H. LncRNA SNHG1 regulates the differentiation of Treg cells and affects the immune escape of breast cancer via regulating miR-448/IDO. Int J Biol Macromol. 2018; 118:24–30. https://doi.org/10.1016/j.ijbiomac.2018.06.033 [PubMed]
- 49. Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010; 363:711–23. https://doi.org/10.1056/NEJMoa1003466 [PubMed]
- 50. Telang S, Rasku MA, Clem AL, Carter K, Klarer AC, Badger WR, Milam RA, Rai SN, Pan J, Gragg H, Clem BF, McMasters KM, Miller DM, Chesney J. Phase II trial of the regulatory T cell-depleting agent, denileukin diftitox, in patients with unresectable stage IV melanoma. BMC Cancer. 2011; 11:515. https://doi.org/10.1186/1471-2407-11-515 [PubMed]
- 51. Jia G, Fu Y, Zhao X, Dai Q, Zheng G, Yang Y, Yi C, Lindahl T, Pan T, Yang YG, He C. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat Chem Biol. 2011; 7:885–7. https://doi.org/10.1038/nchembio.687 [PubMed]
- 52. Bokar JA, Shambaugh ME, Polayes D, Matera AG, Rottman FM. Purification and cDNA cloning of the AdoMet-binding subunit of the human mRNA (N6-adenosine)-methyltransferase. RNA. 1997; 3:1233–47. [PubMed]
- 53. Liu J, Yue Y, Han D, Wang X, Fu Y, Zhang L, Jia G, Yu M, Lu Z, Deng X, Dai Q, Chen W, He C. A METTL3-METTL14 complex mediates mammalian nuclear RNA N6-adenosine methylation. Nat Chem Biol. 2014; 10:93–5. https://doi.org/10.1038/nchembio.1432 [PubMed]
- 54. Wen J, Lv R, Ma H, Shen H, He C, Wang J, Jiao F, Liu H, Yang P, Tan L, Lan F, Shi YG, He C, et al. Zc3h13 Regulates Nuclear RNA m6A Methylation and Mouse Embryonic Stem Cell Self-Renewal. Mol Cell. 2018; 69:1028–38.e6. https://doi.org/10.1016/j.molcel.2018.02.015 [PubMed]
- 55. Fedeles BI, Singh V, Delaney JC, Li D, Essigmann JM. The AlkB Family of Fe(II)/α-Ketoglutarate-dependent Dioxygenases: Repairing Nucleic Acid Alkylation Damage and Beyond. J Biol Chem. 2015; 290:20734–42. https://doi.org/10.1074/jbc.R115.656462 [PubMed]
- 56. Xu C, Liu K, Tempel W, Demetriades M, Aik W, Schofield CJ, Min J. Structures of human ALKBH5 demethylase reveal a unique binding mode for specific single-stranded N6-methyladenosine RNA demethylation. J Biol Chem. 2014; 289:17299–311. https://doi.org/10.1074/jbc.M114.550350 [PubMed]
- 57. Wang X, Zhao BS, Roundtree IA, Lu Z, Han D, Ma H, Weng X, Chen K, Shi H, He C. N(6)-methyladenosine Modulates Messenger RNA Translation Efficiency. Cell. 2015; 161:1388–99. https://doi.org/10.1016/j.cell.2015.05.014 [PubMed]
- 58. Wang X, Lu Z, Gomez A, Hon GC, Yue Y, Han D, Fu Y, Parisien M, Dai Q, Jia G, Ren B, Pan T, He C. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature. 2014; 505:117–20. https://doi.org/10.1038/nature12730 [PubMed]
- 59. Xiao W, Adhikari S, Dahal U, Chen YS, Hao YJ, Sun BF, Sun HY, Li A, Ping XL, Lai WY, Wang X, Ma HL, Huang CM, et al. Nuclear m(6)A Reader YTHDC1 Regulates mRNA Splicing. Mol Cell. 2016; 61:507–19. https://doi.org/10.1016/j.molcel.2016.01.012 [PubMed]
- 60. Wojtas MN, Pandey RR, Mendel M, Homolka D, Sachidanandam R, Pillai RS. Regulation of m6A Transcripts by the 3'→5' RNA Helicase YTHDC2 Is Essential for a Successful Meiotic Program in the Mammalian Germline. Mol Cell. 2017; 68:374–87.e12. https://doi.org/10.1016/j.molcel.2017.09.021 [PubMed]
- 61. Liu N, Dai Q, Zheng G, He C, Parisien M, Pan T. N(6)-methyladenosine-dependent RNA structural switches regulate RNA-protein interactions. Nature. 2015; 518:560–4. https://doi.org/10.1038/nature14234 [PubMed]
- 62. Alarcón CR, Goodarzi H, Lee H, Liu X, Tavazoie S, Tavazoie SF. HNRNPA2B1 Is a Mediator of m(6)A-Dependent Nuclear RNA Processing Events. Cell. 2015; 162:1299–308. https://doi.org/10.1016/j.cell.2015.08.011 [PubMed]
- 63. Meyer KD, Patil DP, Zhou J, Zinoviev A, Skabkin MA, Elemento O, Pestova TV, Qian SB, Jaffrey SR. 5' UTR m(6)A Promotes Cap-Independent Translation. Cell. 2015; 163:999–1010. https://doi.org/10.1016/j.cell.2015.10.012 [PubMed]
- 64. Huang H, Weng H, Sun W, Qin X, Shi H, Wu H, Zhao BS, Mesquita A, Liu C, Yuan CL, Hu YC, Hüttelmaier S, Skibbe JR, et al. Recognition of RNA N6-methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat Cell Biol. 2018; 20:285–95. https://doi.org/10.1038/s41556-018-0045-z [PubMed]
- 65. Meyer KD, Saletore Y, Zumbo P, Elemento O, Mason CE, Jaffrey SR. Comprehensive analysis of mRNA methylation reveals enrichment in 3' UTRs and near stop codons. Cell. 2012; 149:1635–46. https://doi.org/10.1016/j.cell.2012.05.003 [PubMed]
- 66. Dominissini D, Moshitch-Moshkovitz S, Schwartz S, Salmon-Divon M, Ungar L, Osenberg S, Cesarkas K, Jacob-Hirsch J, Amariglio N, Kupiec M, Sorek R, Rechavi G. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature. 2012; 485:201–6. https://doi.org/10.1038/nature11112 [PubMed]
- 67. Chen K, Lu Z, Wang X, Fu Y, Luo GZ, Liu N, Han D, Dominissini D, Dai Q, Pan T, He C. High-resolution N(6) -methyladenosine (m(6) A) map using photo-crosslinking-assisted m(6) A sequencing. Angew Chem Int Ed Engl. 2015; 54:1587–90. https://doi.org/10.1002/anie.201410647 [PubMed]
- 68. Ke S, Alemu EA, Mertens C, Gantman EC, Fak JJ, Mele A, Haripal B, Zucker-Scharff I, Moore MJ, Park CY, Vågbø CB, Kusśnierczyk A, Klungland A, et al. A majority of m6A residues are in the last exons, allowing the potential for 3' UTR regulation. Genes Dev. 2015; 29:2037–53. https://doi.org/10.1101/gad.269415.115 [PubMed]
- 69. Liu N, Parisien M, Dai Q, Zheng G, He C, Pan T. Probing N6-methyladenosine RNA modification status at single nucleotide resolution in mRNA and long noncoding RNA. RNA. 2013; 19:1848–56. https://doi.org/10.1261/rna.041178.113 [PubMed]
- 70. Chen K, Wei Z, Zhang Q, Wu X, Rong R, Lu Z, Su J, de Magalhães JP, Rigden DJ, Meng J. WHISTLE: a high-accuracy map of the human N6-methyladenosine (m6A) epitranscriptome predicted using a machine learning approach. Nucleic Acids Res. 2019; 47:e41. https://doi.org/10.1093/nar/gkz074 [PubMed]
- 71. Zhou Y, Zeng P, Li YH, Zhang Z, Cui Q. SRAMP: prediction of mammalian N6-methyladenosine (m6A) sites based on sequence-derived features. Nucleic Acids Res. 2016; 44:e91. https://doi.org/10.1093/nar/gkw104 [PubMed]
- 72. Li J, He S, Guo F, Zou Q. HSM6AP: a high-precision predictor for the Homo sapiens N6-methyladenosine (m^6 A) based on multiple weights and feature stitching. RNA Biol. 2021; 18:1882–92. https://doi.org/10.1080/15476286.2021.1875180 [PubMed]
- 73. Deng S, Zhang H, Zhu K, Li X, Ye Y, Li R, Liu X, Lin D, Zuo Z, Zheng J. M6A2Target: a comprehensive database for targets of m6A writers, erasers and readers. Brief Bioinform. 2021; 22:bbaa055. https://doi.org/10.1093/bib/bbaa055 [PubMed]
- 74. Liu L, Lei X, Meng J, Wei Z. WITMSG: Large-scale Prediction of Human Intronic m6A RNA Methylation Sites from Sequence and Genomic Features. Curr Genomics. 2020; 21:67–76. https://doi.org/10.2174/1389202921666200211104140 [PubMed]
- 75. Liu S, Zhu A, He C, Chen M. REPIC: a database for exploring the N6-methyladenosine methylome. Genome Biol. 2020; 21:100. https://doi.org/10.1186/s13059-020-02012-4 [PubMed]
- 76. Xuan JJ, Sun WJ, Lin PH, Zhou KR, Liu S, Zheng LL, Qu LH, Yang JH. RMBase v2.0: deciphering the map of RNA modifications from epitranscriptome sequencing data. Nucleic Acids Res. 2018; 46:D327–34. https://doi.org/10.1093/nar/gkx934 [PubMed]
- 77. Liu K, Chen W. iMRM: a platform for simultaneously identifying multiple kinds of RNA modifications. Bioinformatics. 2020; 36:3336–42. https://doi.org/10.1093/bioinformatics/btaa155 [PubMed]
- 78. Liu H, Wang H, Wei Z, Zhang S, Hua G, Zhang SW, Zhang L, Gao SJ, Meng J, Chen X, Huang Y. MeT-DB V2.0: elucidating context-specific functions of N6-methyl-adenosine methyltranscriptome. Nucleic Acids Res. 2018; 46:D281–7. https://doi.org/10.1093/nar/gkx1080 [PubMed]
- 79. Zhao X, Yang Y, Sun BF, Shi Y, Yang X, Xiao W, Hao YJ, Ping XL, Chen YS, Wang WJ, Jin KX, Wang X, Huang CM, et al. FTO-dependent demethylation of N6-methyladenosine regulates mRNA splicing and is required for adipogenesis. Cell Res. 2014; 24:1403–19. https://doi.org/10.1038/cr.2014.151 [PubMed]
- 80. Roundtree IA, Luo GZ, Zhang Z, Wang X, Zhou T, Cui Y, Sha J, Huang X, Guerrero L, Xie P, He E, Shen B, He C. YTHDC1 mediates nuclear export of N6-methyladenosine methylated mRNAs. Elife. 2017; 6:e31311. https://doi.org/10.7554/eLife.31311 [PubMed]
- 81. Visvanathan A, Patil V, Arora A, Hegde AS, Arivazhagan A, Santosh V, Somasundaram K. Essential role of METTL3-mediated m6A modification in glioma stem-like cells maintenance and radioresistance. Oncogene. 2018; 37:522–33. https://doi.org/10.1038/onc.2017.351 [PubMed]
- 82. Hsu PJ, Zhu Y, Ma H, Guo Y, Shi X, Liu Y, Qi M, Lu Z, Shi H, Wang J, Cheng Y, Luo G, Dai Q, et al. Ythdc2 is an N6-methyladenosine binding protein that regulates mammalian spermatogenesis. Cell Res. 2017; 27:1115–27. https://doi.org/10.1038/cr.2017.99 [PubMed]
- 83. Geula S, Moshitch-Moshkovitz S, Dominissini D, Mansour AA, Kol N, Salmon-Divon M, Hershkovitz V, Peer E, Mor N, Manor YS, Ben-Haim MS, Eyal E, Yunger S, et al. Stem cells. m6A mRNA methylation facilitates resolution of naïve pluripotency toward differentiation. Science. 2015; 347:1002–6. https://doi.org/10.1126/science.1261417 [PubMed]
- 84. Li Z, Weng H, Su R, Weng X, Zuo Z, Li C, Huang H, Nachtergaele S, Dong L, Hu C, Qin X, Tang L, Wang Y, et al. FTO Plays an Oncogenic Role in Acute Myeloid Leukemia as a N6-Methyladenosine RNA Demethylase. Cancer Cell. 2017; 31:127–41. https://doi.org/10.1016/j.ccell.2016.11.017 [PubMed]
- 85. Lin Z, Hsu PJ, Xing X, Fang J, Lu Z, Zou Q, Zhang KJ, Zhang X, Zhou Y, Zhang T, Zhang Y, Song W, Jia G, et al. Mettl3-/Mettl14-mediated mRNA N6-methyladenosine modulates murine spermatogenesis. Cell Res. 2017; 27:1216–30. https://doi.org/10.1038/cr.2017.117 [PubMed]
- 86. Xu K, Yang Y, Feng GH, Sun BF, Chen JQ, Li YF, Chen YS, Zhang XX, Wang CX, Jiang LY, Liu C, Zhang ZY, Wang XJ, et al. Mettl3-mediated m6A regulates spermatogonial differentiation and meiosis initiation. Cell Res. 2017; 27:1100–14. https://doi.org/10.1038/cr.2017.100 [PubMed]
- 87. Zheng G, Dahl JA, Niu Y, Fedorcsak P, Huang CM, Li CJ, Vågbø CB, Shi Y, Wang WL, Song SH, Lu Z, Bosmans RP, Dai Q, et al. ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Mol Cell. 2013; 49:18–29. https://doi.org/10.1016/j.molcel.2012.10.015 [PubMed]
- 88. Wang CX, Cui GS, Liu X, Xu K, Wang M, Zhang XX, Jiang LY, Li A, Yang Y, Lai WY, Sun BF, Jiang GB, Wang HL, et al. METTL3-mediated m6A modification is required for cerebellar development. PLoS Biol. 2018; 16:e2004880. https://doi.org/10.1371/journal.pbio.2004880 [PubMed]
- 89. Yoon KJ, Ringeling FR, Vissers C, Jacob F, Pokrass M, Jimenez-Cyrus D, Su Y, Kim NS, Zhu Y, Zheng L, Kim S, Wang X, Doré LC, et al. Temporal Control of Mammalian Cortical Neurogenesis by m6A Methylation. Cell. 2017; 171:877–89.e17. https://doi.org/10.1016/j.cell.2017.09.003 [PubMed]
- 90. Nainar S, Marshall PR, Tyler CR, Spitale RC, Bredy TW. Evolving insights into RNA modifications and their functional diversity in the brain. Nat Neurosci. 2016; 19:1292–8. https://doi.org/10.1038/nn.4378 [PubMed]
- 91. Walters BJ, Mercaldo V, Gillon CJ, Yip M, Neve RL, Boyce FM, Frankland PW, Josselyn SA. The Role of The RNA Demethylase FTO (Fat Mass and Obesity-Associated) and mRNA Methylation in Hippocampal Memory Formation. Neuropsychopharmacology. 2017; 42:1502–10. https://doi.org/10.1038/npp.2017.31 [PubMed]
- 92. Han M, Liu Z, Xu Y, Liu X, Wang D, Li F, Wang Y, Bi J. Abnormality of m6A mRNA Methylation Is Involved in Alzheimer's Disease. Front Neurosci. 2020; 14:98. https://doi.org/10.3389/fnins.2020.00098 [PubMed]
- 93. Zhong X, Yu J, Frazier K, Weng X, Li Y, Cham CM, Dolan K, Zhu X, Hubert N, Tao Y, Lin F, Martinez-Guryn K, Huang Y, et al. Circadian Clock Regulation of Hepatic Lipid Metabolism by Modulation of m6A mRNA Methylation. Cell Rep. 2018; 25:1816–28.e4. https://doi.org/10.1016/j.celrep.2018.10.068 [PubMed]
- 94. Dorn LE, Lasman L, Chen J, Xu X, Hund TJ, Medvedovic M, Hanna JH, van Berlo JH, Accornero F. The N6-Methyladenosine mRNA Methylase METTL3 Controls Cardiac Homeostasis and Hypertrophy. Circulation. 2019; 139:533–45. https://doi.org/10.1161/CIRCULATIONAHA.118.036146 [PubMed]
- 95. Mathiyalagan P, Adamiak M, Mayourian J, Sassi Y, Liang Y, Agarwal N, Jha D, Zhang S, Kohlbrenner E, Chepurko E, Chen J, Trivieri MG, Singh R, et al. FTO-Dependent N6-Methyladenosine Regulates Cardiac Function During Remodeling and Repair. Circulation. 2019; 139:518–32. https://doi.org/10.1161/CIRCULATIONAHA.118.033794 [PubMed]
- 96. Zhong L, He X, Song H, Sun Y, Chen G, Si X, Sun J, Chen X, Liao W, Liao Y, Bin J. METTL3 Induces AAA Development and Progression by Modulating N6-Methyladenosine-Dependent Primary miR34a Processing. Mol Ther Nucleic Acids. 2020; 21:394–411. https://doi.org/10.1016/j.omtn.2020.06.005 [PubMed]
- 97. Zhang J, Bai R, Li M, Ye H, Wu C, Wang C, Li S, Tan L, Mai D, Li G, Pan L, Zheng Y, Su J, et al. Excessive miR-25-3p maturation via N6-methyladenosine stimulated by cigarette smoke promotes pancreatic cancer progression. Nat Commun. 2019; 10:1858. https://doi.org/10.1038/s41467-019-09712-x [PubMed]
- 98. Liu Y, Wang R, Zhang L, Li J, Lou K, Shi B. The lipid metabolism gene FTO influences breast cancer cell energy metabolism via the PI3K/AKT signaling pathway. Oncol Lett. 2017; 13:4685–90. https://doi.org/10.3892/ol.2017.6038 [PubMed]
- 99. Li J, Zhu L, Shi Y, Liu J, Lin L, Chen X. m6A demethylase FTO promotes hepatocellular carcinoma tumorigenesis via mediating PKM2 demethylation. Am J Transl Res. 2019; 11:6084–92. [PubMed]
- 100. Zhang B, Wu Q, Li B, Wang D, Wang L, Zhou YL. m6A regulator-mediated methylation modification patterns and tumor microenvironment infiltration characterization in gastric cancer. Mol Cancer. 2020; 19:53. https://doi.org/10.1186/s12943-020-01170-0 [PubMed]
- 101. Iwakawa HO, Tomari Y. The Functions of MicroRNAs: mRNA Decay and Translational Repression. Trends Cell Biol. 2015; 25:651–65. https://doi.org/10.1016/j.tcb.2015.07.011 [PubMed]
- 102. Treiber T, Treiber N, Meister G. Regulation of microRNA biogenesis and its crosstalk with other cellular pathways. Nat Rev Mol Cell Biol. 2019; 20:5–20. https://doi.org/10.1038/s41580-018-0059-1 [PubMed]
- 103. Alarcón CR, Lee H, Goodarzi H, Halberg N, Tavazoie SF. N6-methyladenosine marks primary microRNAs for processing. Nature. 2015; 519:482–5. https://doi.org/10.1038/nature14281 [PubMed]
- 104. Song J, Ye A, Jiang E, Yin X, Chen Z, Bai G, Zhou Y, Liu J. Reconstruction and analysis of the aberrant lncRNA-miRNA-mRNA network based on competitive endogenous RNA in CESC. J Cell Biochem. 2018; 119:6665–73. https://doi.org/10.1002/jcb.26850 [PubMed]
- 105. Liu N, Zhou KI, Parisien M, Dai Q, Diatchenko L, Pan T. N6-methyladenosine alters RNA structure to regulate binding of a low-complexity protein. Nucleic Acids Res. 2017; 45:6051–63. https://doi.org/10.1093/nar/gkx141 [PubMed]
- 106. Yang D, Qiao J, Wang G, Lan Y, Li G, Guo X, Xi J, Ye D, Zhu S, Chen W, Jia W, Leng Y, Wan X, Kang J. N6-Methyladenosine modification of lincRNA 1281 is critically required for mESC differentiation potential. Nucleic Acids Res. 2018; 46:3906–20. https://doi.org/10.1093/nar/gky130 [PubMed]
- 107. Yang Y, Fan X, Mao M, Song X, Wu P, Zhang Y, Jin Y, Yang Y, Chen LL, Wang Y, Wong CC, Xiao X, Wang Z. Extensive translation of circular RNAs driven by N6-methyladenosine. Cell Res. 2017; 27:626–41. https://doi.org/10.1038/cr.2017.31 [PubMed]
- 108. Paramasivam A, Vijayashree Priyadharsini J. Novel insights into m6A modification in circular RNA and implications for immunity. Cell Mol Immunol. 2020; 17:668–9. https://doi.org/10.1038/s41423-020-0387-x [PubMed]
- 109. Chen YG, Chen R, Ahmad S, Verma R, Kasturi SP, Amaya L, Broughton JP, Kim J, Cadena C, Pulendran B, Hur S, Chang HY. N6-Methyladenosine Modification Controls Circular RNA Immunity. Mol Cell. 2019; 76:96–109.e9. https://doi.org/10.1016/j.molcel.2019.07.016 [PubMed]
- 110. Li J, Meng S, Xu M, Wang S, He L, Xu X, Wang X, Xie L. Downregulation of N6-methyladenosine binding YTHDF2 protein mediated by miR-493-3p suppresses prostate cancer by elevating N6-methyladenosine levels. Oncotarget. 2017; 9:3752–64. https://doi.org/10.18632/oncotarget.23365 [PubMed]
- 111. Yang Z, Li J, Feng G, Gao S, Wang Y, Zhang S, Liu Y, Ye L, Li Y, Zhang X. MicroRNA-145 Modulates N6-Methyladenosine Levels by Targeting the 3'-Untranslated mRNA Region of the N6-Methyladenosine Binding YTH Domain Family 2 Protein. J Biol Chem. 2017; 292:3614–23. https://doi.org/10.1074/jbc.M116.749689 [PubMed]
- 112. Kleemann M, Schneider H, Unger K, Sander P, Schneider EM, Fischer-Posovszky P, Handrick R, Otte K. MiR-744-5p inducing cell death by directly targeting HNRNPC and NFIX in ovarian cancer cells. Sci Rep. 2018; 8:9020. https://doi.org/10.1038/s41598-018-27438-6 [PubMed]
- 113. Li Y, Yang F, Gao M, Gong R, Jin M, Liu T, Sun Y, Fu Y, Huang Q, Zhang W, Liu S, Yu M, Yan G, et al. miR-149-3p Regulates the Switch between Adipogenic and Osteogenic Differentiation of BMSCs by Targeting FTO. Mol Ther Nucleic Acids. 2019; 17:590–600. https://doi.org/10.1016/j.omtn.2019.06.023 [PubMed]
- 114. Xu X, Yu Y, Zong K, Lv P, Gu Y. Up-regulation of IGF2BP2 by multiple mechanisms in pancreatic cancer promotes cancer proliferation by activating the PI3K/Akt signaling pathway. J Exp Clin Cancer Res. 2019; 38:497. https://doi.org/10.1186/s13046-019-1470-y [PubMed]
- 115. Cai X, Wang X, Cao C, Gao Y, Zhang S, Yang Z, Liu Y, Zhang X, Zhang W, Ye L. HBXIP-elevated methyltransferase METTL3 promotes the progression of breast cancer via inhibiting tumor suppressor let-7g. Cancer Lett. 2018; 415:11–9. https://doi.org/10.1016/j.canlet.2017.11.018 [PubMed]
- 116. Wei W, Huo B, Shi X. miR-600 inhibits lung cancer via downregulating the expression of METTL3. Cancer Manag Res. 2019; 11:1177–87. https://doi.org/10.2147/CMAR.S181058 [PubMed]
- 117. Zhu S, Wang JZ, Chen D, He YT, Meng N, Chen M, Lu RX, Chen XH, Zhang XL, Yan GR. An oncopeptide regulates m6A recognition by the m6A reader IGF2BP1 and tumorigenesis. Nat Commun. 2020; 11:1685. https://doi.org/10.1038/s41467-020-15403-9 [PubMed]
- 118. Wang Y, Lu JH, Wu QN, Jin Y, Wang DS, Chen YX, Liu J, Luo XJ, Meng Q, Pu HY, Wang YN, Hu PS, Liu ZX, et al. LncRNA LINRIS stabilizes IGF2BP2 and promotes the aerobic glycolysis in colorectal cancer. Mol Cancer. 2019; 18:174. https://doi.org/10.1186/s12943-019-1105-0 [PubMed]
- 119. Chen Z, Chen X, Lei T, Gu Y, Gu J, Huang J, Lu B, Yuan L, Sun M, Wang Z. Integrative Analysis of NSCLC Identifies LINC01234 as an Oncogenic lncRNA that Interacts with HNRNPA2B1 and Regulates miR-106b Biogenesis. Mol Ther. 2020; 28:1479–93. https://doi.org/10.1016/j.ymthe.2020.03.010 [PubMed]
- 120. Zhang S, Zhao BS, Zhou A, Lin K, Zheng S, Lu Z, Chen Y, Sulman EP, Xie K, Bögler O, Majumder S, He C, Huang S. m6A Demethylase ALKBH5 Maintains Tumorigenicity of Glioblastoma Stem-like Cells by Sustaining FOXM1 Expression and Cell Proliferation Program. Cancer Cell. 2017; 31:591–606.e6. https://doi.org/10.1016/j.ccell.2017.02.013 [PubMed]
- 121. Wang X, Zhang J, Wang Y. Long noncoding RNA GAS5-AS1 suppresses growth and metastasis of cervical cancer by increasing GAS5 stability. Am J Transl Res. 2019; 11:4909–21. [PubMed]
- 122. Huang R, Zhang Y, Bai Y, Han B, Ju M, Chen B, Yang L, Wang Y, Zhang H, Zhang H, Xie C, Zhang Z, Yao H. N6-Methyladenosine Modification of Fatty Acid Amide Hydrolase Messenger RNA in Circular RNA STAG1-Regulated Astrocyte Dysfunction and Depressive-like Behaviors. Biol Psychiatry. 2020; 88:392–404. https://doi.org/10.1016/j.biopsych.2020.02.018 [PubMed]
- 123. Nair L, Zhang W, Laffleur B, Jha MK, Lim J, Lee H, Wu L, Alvarez NS, Liu ZP, Munteanu EL, Swayne T, Hanna JH, Ding L, et al. Mechanism of noncoding RNA-associated N6-methyladenosine recognition by an RNA processing complex during IgH DNA recombination. Mol Cell. 2021; 81:3949–64.e7. https://doi.org/10.1016/j.molcel.2021.07.037 [PubMed]
- 124. Yuan X, Li T, Shi L, Miao J, Guo Y, Chen Y. Human umbilical cord mesenchymal stem cells deliver exogenous miR-26a-5p via exosomes to inhibit nucleus pulposus cell pyroptosis through METTL14/NLRP3. Mol Med. 2021; 27:91. https://doi.org/10.1186/s10020-021-00355-7 [PubMed]
- 125. Song Z, Jia G, Ma P, Cang S. Exosomal miR-4443 promotes cisplatin resistance in non-small cell lung carcinoma by regulating FSP1 m6A modification-mediated ferroptosis. Life Sci. 2021; 276:119399. https://doi.org/10.1016/j.lfs.2021.119399 [PubMed]
- 126. Wang W, Qiao SC, Wu XB, Sun B, Yang JG, Li X, Zhang X, Qian SJ, Gu YX, Lai HC. Circ_0008542 in osteoblast exosomes promotes osteoclast-induced bone resorption through m6A methylation. Cell Death Dis. 2021; 12:628. https://doi.org/10.1038/s41419-021-03915-1 [PubMed]
- 127. Ou C, Sun Z, Li X, Li X, Ren W, Qin Z, Zhang X, Yuan W, Wang J, Yu W, Zhang S, Peng Q, Yan Q, et al. MiR-590-5p, a density-sensitive microRNA, inhibits tumorigenesis by targeting YAP1 in colorectal cancer. Cancer Lett. 2017; 399:53–63. https://doi.org/10.1016/j.canlet.2017.04.011 [PubMed]
- 128. He X, Yu B, Kuang G, Wu Y, Zhang M, Cao P, Ou C. Long noncoding RNA DLEU2 affects the proliferative and invasive ability of colorectal cancer cells. J Cancer. 2021; 12:428–37. https://doi.org/10.7150/jca.48423 [PubMed]
- 129. Xing F, Saidou J, Watabe K. Cancer associated fibroblasts (CAFs) in tumor microenvironment. Front Biosci (Landmark Ed). 2010; 15:166–79. https://doi.org/10.2741/3613 [PubMed]
- 130. Aponte-López A, Muñoz-Cruz S. Mast Cells in the Tumor Microenvironment. Adv Exp Med Biol. 2020; 1273:159–73. https://doi.org/10.1007/978-3-030-49270-0_9 [PubMed]
- 131. Han J, Wang JZ, Yang X, Yu H, Zhou R, Lu HC, Yuan WB, Lu JC, Zhou ZJ, Lu Q, Wei JF, Yang H. METTL3 promote tumor proliferation of bladder cancer by accelerating pri-miR221/222 maturation in m6A-dependent manner. Mol Cancer. 2019; 18:110. https://doi.org/10.1186/s12943-019-1036-9 [PubMed]
- 132. Lin R, Zhan M, Yang L, Wang H, Shen H, Huang S, Huang X, Xu S, Zhang Z, Li W, Liu Q, Shi Y, Chen W, et al. Deoxycholic acid modulates the progression of gallbladder cancer through N6-methyladenosine-dependent microRNA maturation. Oncogene. 2020; 39:4983–5000. https://doi.org/10.1038/s41388-020-1349-6 [PubMed]
- 133. Chen S, Zhou L, Wang Y. ALKBH5-mediated m6A demethylation of lncRNA PVT1 plays an oncogenic role in osteosarcoma. Cancer Cell Int. 2020; 20:34. https://doi.org/10.1186/s12935-020-1105-6 [PubMed]
- 134. Sun C, Wang P, Dong W, Liu H, Sun J, Zhao L. LncRNA PVT1 promotes exosome secretion through YKT6, RAB7, and VAMP3 in pancreatic cancer. Aging (Albany NY). 2020; 12:10427–40. https://doi.org/10.18632/aging.103268 [PubMed]
- 135. Lu S, Han L, Hu X, Sun T, Xu D, Li Y, Chen Q, Yao W, He M, Wang Z, Wu H, Wei M. N6-methyladenosine reader IMP2 stabilizes the ZFAS1/OLA1 axis and activates the Warburg effect: implication in colorectal cancer. J Hematol Oncol. 2021; 14:188. https://doi.org/10.1186/s13045-021-01204-0 [PubMed]
- 136. Li Z, Qin X, Bian W, Li Y, Shan B, Yao Z, Li S. Exosomal lncRNA ZFAS1 regulates esophageal squamous cell carcinoma cell proliferation, invasion, migration and apoptosis via microRNA-124/STAT3 axis. J Exp Clin Cancer Res. 2019; 38:477. https://doi.org/10.1186/s13046-019-1473-8 [PubMed]
- 137. Wei X, Chen Y, Jiang X, Peng M, Liu Y, Mo Y, Ren D, Hua Y, Yu B, Zhou Y, Liao Q, Wang H, Xiang B, et al. Mechanisms of vasculogenic mimicry in hypoxic tumor microenvironments. Mol Cancer. 2021; 20:7. https://doi.org/10.1186/s12943-020-01288-1 [PubMed]
- 138. Jiang X, Wang J, Deng X, Xiong F, Zhang S, Gong Z, Li X, Cao K, Deng H, He Y, Liao Q, Xiang B, Zhou M, et al. The role of microenvironment in tumor angiogenesis. J Exp Clin Cancer Res. 2020; 39:204. https://doi.org/10.1186/s13046-020-01709-5 [PubMed]
- 139. Fu LQ, Du WL, Cai MH, Yao JY, Zhao YY, Mou XZ. The roles of tumor-associated macrophages in tumor angiogenesis and metastasis. Cell Immunol. 2020; 353:104119. https://doi.org/10.1016/j.cellimm.2020.104119 [PubMed]
- 140. Yang W, Xie L, Wang P, Zhuang C. MiR-155 regulates m6A level and cell progression by targeting FTO in clear cell renal cell carcinoma. Cell Signal. 2022; 91:110217. https://doi.org/10.1016/j.cellsig.2021.110217 [PubMed]
- 141. Zhou X, Yan T, Huang C, Xu Z, Wang L, Jiang E, Wang H, Chen Y, Liu K, Shao Z, Shang Z. Melanoma cell-secreted exosomal miR-155-5p induce proangiogenic switch of cancer-associated fibroblasts via SOCS1/JAK2/STAT3 signaling pathway. J Exp Clin Cancer Res. 2018; 37:242. https://doi.org/10.1186/s13046-018-0911-3 [PubMed]
- 142. Hao J, Li C, Lin C, Hao Y, Yu X, Xia Y, Gao F, Jiang Z, Wang D. Targeted point mutations of the m6A modification in miR675 using RNA-guided base editing induce cell apoptosis. Biosci Rep. 2020; 40:BSR20192933. https://doi.org/10.1042/BSR20192933 [PubMed]
- 143. Lo Dico A, Costa V, Martelli C, Diceglie C, Rajata F, Rizzo A, Mancone C, Tripodi M, Ottobrini L, Alessandro R, Conigliaro A. MiR675-5p Acts on HIF-1α to Sustain Hypoxic Responses: A New Therapeutic Strategy for Glioma. Theranostics. 2016; 6:1105–18. https://doi.org/10.7150/thno.14700 [PubMed]
- 144. Conigliaro A, Costa V, Lo Dico A, Saieva L, Buccheri S, Dieli F, Manno M, Raccosta S, Mancone C, Tripodi M, De Leo G, Alessandro R. CD90+ liver cancer cells modulate endothelial cell phenotype through the release of exosomes containing H19 lncRNA. Mol Cancer. 2015; 14:155. https://doi.org/10.1186/s12943-015-0426-x [PubMed]
- 145. Barros-Silva D, Lobo J, Guimarães-Teixeira C, Carneiro I, Oliveira J, Martens-Uzunova ES, Henrique R, Jerónimo C. VIRMA-Dependent N6-Methyladenosine Modifications Regulate the Expression of Long Non-Coding RNAs CCAT1 and CCAT2 in Prostate Cancer. Cancers (Basel). 2020; 12:771. https://doi.org/10.3390/cancers12040771 [PubMed]
- 146. Han W, Sulidankazha Q, Nie X, Yilidan R, Len K. Retraction notice to “Pancreatic cancer cells-derived exosomal long non-coding RNA CCAT1/microRNA-138-5p/HMGA1 axis promotes tumor angiogenesis” [Life Sci. 278 (2021) 119495]. Life Sci. 2023; 319:121429. https://doi.org/10.1016/j.lfs.2023.121429 [PubMed]
- 147. Ni W, Yao S, Zhou Y, Liu Y, Huang P, Zhou A, Liu J, Che L, Li J. Long noncoding RNA GAS5 inhibits progression of colorectal cancer by interacting with and triggering YAP phosphorylation and degradation and is negatively regulated by the m6A reader YTHDF3. Mol Cancer. 2019; 18:143. https://doi.org/10.1186/s12943-019-1079-y [PubMed]
- 148. Cheng Y, Dai X, Yang T, Zhang N, Liu Z, Jiang Y. Low Long Noncoding RNA Growth Arrest-Specific Transcript 5 Expression in the Exosomes of Lung Cancer Cells Promotes Tumor Angiogenesis. J Oncol. 2019; 2019:2476175. https://doi.org/10.1155/2019/2476175 [PubMed]
- 149. Sun Y, Li S, Yu W, Zhao Z, Gao J, Chen C, Wei M, Liu T, Li L, Liu L. N6-methyladenosine-dependent pri-miR-17-92 maturation suppresses PTEN/TMEM127 and promotes sensitivity to everolimus in gastric cancer. Cell Death Dis. 2020; 11:836. https://doi.org/10.1038/s41419-020-03049-w [PubMed]
- 150. Luengo-Gil G, Gonzalez-Billalabeitia E, Perez-Henarejos SA, Navarro Manzano E, Chaves-Benito A, Garcia-Martinez E, Garcia-Garre E, Vicente V, Ayala de la Peña F. Angiogenic role of miR-20a in breast cancer. PLoS One. 2018; 13:e0194638. https://doi.org/10.1371/journal.pone.0194638 [PubMed]
- 151. Wang H, Deng Q, Lv Z, Ling Y, Hou X, Chen Z, Dinglin X, Ma S, Li D, Wu Y, Peng Y, Huang H, Chen L. N6-methyladenosine induced miR-143-3p promotes the brain metastasis of lung cancer via regulation of VASH1. Mol Cancer. 2019; 18:181. https://doi.org/10.1186/s12943-019-1108-x [PubMed]
- 152. Jerez S, Araya H, Hevia D, Irarrázaval CE, Thaler R, van Wijnen AJ, Galindo M. Extracellular vesicles from osteosarcoma cell lines contain miRNAs associated with cell adhesion and apoptosis. Gene. 2019; 710:246–57. https://doi.org/10.1016/j.gene.2019.06.005 [PubMed]
- 153. Ou C, Sun Z, He X, Li X, Fan S, Zheng X, Peng Q, Li G, Li X, Ma J. Targeting YAP1/LINC00152/FSCN1 Signaling Axis Prevents the Progression of Colorectal Cancer. Adv Sci (Weinh). 2019; 7:1901380. https://doi.org/10.1002/advs.201901380 [PubMed]
- 154. He X, Li S, Yu B, Kuang G, Wu Y, Zhang M, He Y, Ou C, Cao P. Up-regulation of LINC00467 promotes the tumourigenesis in colorectal cancer. J Cancer. 2019; 10:6405–13. https://doi.org/10.7150/jca.32216 [PubMed]
- 155. Ren B, Cui M, Yang G, Wang H, Feng M, You L, Zhao Y. Tumor microenvironment participates in metastasis of pancreatic cancer. Mol Cancer. 2018; 17:108. https://doi.org/10.1186/s12943-018-0858-1 [PubMed]
- 156. Zheng ZQ, Li ZX, Zhou GQ, Lin L, Zhang LL, Lv JW, Huang XD, Liu RQ, Chen F, He XJ, Kou J, Zhang J, Wen X, et al. Long Noncoding RNA FAM225A Promotes Nasopharyngeal Carcinoma Tumorigenesis and Metastasis by Acting as ceRNA to Sponge miR-590-3p/miR-1275 and Upregulate ITGB3. Cancer Res. 2019; 79:4612–26. https://doi.org/10.1158/0008-5472.CAN-19-0799 [PubMed]
- 157. Li B, Xia Y, Lv J, Wang W, Xuan Z, Chen C, Jiang T, Fang L, Wang L, Li Z, He Z, Li Q, Xie L, et al. miR-151a-3p-rich small extracellular vesicles derived from gastric cancer accelerate liver metastasis via initiating a hepatic stemness-enhancing niche. Oncogene. 2021; 40:6180–94. https://doi.org/10.1038/s41388-021-02011-0 [PubMed]
- 158. Liu T, Li P, Li J, Qi Q, Sun Z, Shi S, Xie Y, Liu S, Wang Y, Du L, Wang C. Exosomal and intracellular miR-320b promotes lymphatic metastasis in esophageal squamous cell carcinoma. Mol Ther Oncolytics. 2021; 23:163–80. https://doi.org/10.1016/j.omto.2021.09.003 [PubMed]
- 159. Yang X, Zhang S, He C, Xue P, Zhang L, He Z, Zang L, Feng B, Sun J, Zheng M. METTL14 suppresses proliferation and metastasis of colorectal cancer by down-regulating oncogenic long non-coding RNA XIST. Mol Cancer. 2020; 19:46. https://doi.org/10.1186/s12943-020-1146-4 [PubMed]
- 160. Xing F, Liu Y, Wu SY, Wu K, Sharma S, Mo YY, Feng J, Sanders S, Jin G, Singh R, Vidi PA, Tyagi A, Chan MD, et al. Loss of XIST in Breast Cancer Activates MSN-c-Met and Reprograms Microglia via Exosomal miRNA to Promote Brain Metastasis. Cancer Res. 2018; 78:4316–30. https://doi.org/10.1158/0008-5472.CAN-18-1102 [PubMed]
- 161. Zhang J, Guo S, Piao HY, Wang Y, Wu Y, Meng XY, Yang D, Zheng ZC, Zhao Y. ALKBH5 promotes invasion and metastasis of gastric cancer by decreasing methylation of the lncRNA NEAT1. J Physiol Biochem. 2019; 75:379–89. https://doi.org/10.1007/s13105-019-00690-8 [PubMed]
- 162. Mo C, Huang B, Zhuang J, Jiang S, Guo S, Mao X. LncRNA nuclear-enriched abundant transcript 1 shuttled by prostate cancer cells-secreted exosomes initiates osteoblastic phenotypes in the bone metastatic microenvironment via miR-205-5p/runt-related transcription factor 2/splicing factor proline- and glutamine-rich/polypyrimidine tract-binding protein 2 axis. Clin Transl Med. 2021; 11:e493. https://doi.org/10.1002/ctm2.493 [PubMed]
- 163. Jin D, Guo J, Wu Y, Du J, Yang L, Wang X, Di W, Hu B, An J, Kong L, Pan L, Su G. m6A mRNA methylation initiated by METTL3 directly promotes YAP translation and increases YAP activity by regulating the MALAT1-miR-1914-3p-YAP axis to induce NSCLC drug resistance and metastasis. J Hematol Oncol. 2019; 12:135. https://doi.org/10.1186/s13045-019-0830-6 [PubMed]
- 164. Wang Z, Wang X, Zhang T, Su L, Liu B, Zhu Z, Li C. LncRNA MALAT1 promotes gastric cancer progression via inhibiting autophagic flux and inducing fibroblast activation. Cell Death Dis. 2021; 12:368. https://doi.org/10.1038/s41419-021-03645-4 [PubMed]
- 165. Shulman Z, Stern-Ginossar N. The RNA modification N6-methyladenosine as a novel regulator of the immune system. Nat Immunol. 2020; 21:501–12. https://doi.org/10.1038/s41590-020-0650-4 [PubMed]
- 166. Wang H, Hu X, Huang M, Liu J, Gu Y, Ma L, Zhou Q, Cao X. Mettl3-mediated mRNA m6A methylation promotes dendritic cell activation. Nat Commun. 2019; 10:1898. https://doi.org/10.1038/s41467-019-09903-6 [PubMed]
- 167. Han D, Liu J, Chen C, Dong L, Liu Y, Chang R, Huang X, Liu Y, Wang J, Dougherty U, Bissonnette MB, Shen B, Weichselbaum RR, et al. Anti-tumour immunity controlled through mRNA m6A methylation and YTHDF1 in dendritic cells. Nature. 2019; 566:270–4. https://doi.org/10.1038/s41586-019-0916-x [PubMed]
- 168. Liu Y, Liu Z, Tang H, Shen Y, Gong Z, Xie N, Zhang X, Wang W, Kong W, Zhou Y, Fu Y. The N6-methyladenosine (m6A)-forming enzyme METTL3 facilitates M1 macrophage polarization through the methylation of STAT1 mRNA. Am J Physiol Cell Physiol. 2019; 317:C762–75. https://doi.org/10.1152/ajpcell.00212.2019 [PubMed]
- 169. Gu X, Zhang Y, Li D, Cai H, Cai L, Xu Q. N6-methyladenosine demethylase FTO promotes M1 and M2 macrophage activation. Cell Signal. 2020; 69:109553. https://doi.org/10.1016/j.cellsig.2020.109553 [PubMed]
- 170. Li HB, Tong J, Zhu S, Batista PJ, Duffy EE, Zhao J, Bailis W, Cao G, Kroehling L, Chen Y, Wang G, Broughton JP, Chen YG, et al. m6A mRNA methylation controls T cell homeostasis by targeting the IL-7/STAT5/SOCS pathways. Nature. 2017; 548:338–42. https://doi.org/10.1038/nature23450 [PubMed]
- 171. Zhang C, Zhang M, Ge S, Huang W, Lin X, Gao J, Gong J, Shen L. Reduced m6A modification predicts malignant phenotypes and augmented Wnt/PI3K-Akt signaling in gastric cancer. Cancer Med. 2019; 8:4766–81. https://doi.org/10.1002/cam4.2360 [PubMed]
- 172. Shang W, Gao Y, Tang Z, Zhang Y, Yang R. The Pseudogene Olfr29-ps1 Promotes the Suppressive Function and Differentiation of Monocytic MDSCs. Cancer Immunol Res. 2019; 7:813–27. https://doi.org/10.1158/2326-6066.CIR-18-0443 [PubMed]
- 173. Zhao R, Li B, Zhang S, He Z, Pan Z, Guo Q, Qiu W, Qi Y, Zhao S, Wang S, Chen Z, Zhang P, Guo X, et al. The N6-Methyladenosine-Modified Pseudogene HSPA7 Correlates With the Tumor Microenvironment and Predicts the Response to Immune Checkpoint Therapy in Glioblastoma. Front Immunol. 2021; 12:653711. https://doi.org/10.3389/fimmu.2021.653711 [PubMed]
- 174. Peng W, Li J, Chen R, Gu Q, Yang P, Qian W, Ji D, Wang Q, Zhang Z, Tang J, Sun Y. Upregulated METTL3 promotes metastasis of colorectal Cancer via miR-1246/SPRED2/MAPK signaling pathway. J Exp Clin Cancer Res. 2019; 38:393. https://doi.org/10.1186/s13046-019-1408-4 [PubMed]
- 175. Cooks T, Pateras IS, Jenkins LM, Patel KM, Robles AI, Morris J, Forshew T, Appella E, Gorgoulis VG, Harris CC. Mutant p53 cancers reprogram macrophages to tumor supporting macrophages via exosomal miR-1246. Nat Commun. 2018; 9:771. https://doi.org/10.1038/s41467-018-03224-w [PubMed]
- 176. Liu J, Zhang X, Chen K, Cheng Y, Liu S, Xia M, Chen Y, Zhu H, Li Z, Cao X. CCR7 Chemokine Receptor-Inducible lnc-Dpf3 Restrains Dendritic Cell Migration by Inhibiting HIF-1α-Mediated Glycolysis. Immunity. 2019; 50:600–15.e15. https://doi.org/10.1016/j.immuni.2019.01.021 [PubMed]
- 177. Yan K, Fu Y, Zhu N, Wang Z, Hong JL, Li Y, Li WJ, Zhang HB, Song JH. Repression of lncRNA NEAT1 enhances the antitumor activity of CD8(+)T cells against hepatocellular carcinoma via regulating miR-155/Tim-3. Int J Biochem Cell Biol. 2019; 110:1–8. https://doi.org/10.1016/j.biocel.2019.01.019 [PubMed]
- 178. Liu Z, Wang T, She Y, Wu K, Gu S, Li L, Dong C, Chen C, Zhou Y. N6-methyladenosine-modified circIGF2BP3 inhibits CD8+ T-cell responses to facilitate tumor immune evasion by promoting the deubiquitination of PD-L1 in non-small cell lung cancer. Mol Cancer. 2021; 20:105. https://doi.org/10.1186/s12943-021-01398-4 [PubMed]
- 179. Ge L, Zhang N, Chen Z, Song J, Wu Y, Li Z, Chen F, Wu J, Li D, Li J, Wang C, Wang H, Wang J. Level of N6-Methyladenosine in Peripheral Blood RNA: A Novel Predictive Biomarker for Gastric Cancer. Clin Chem. 2020; 66:342–51. https://doi.org/10.1093/clinchem/hvz004 [PubMed]
- 180. Lobo J, Costa AL, Cantante M, Guimarães R, Lopes P, Antunes L, Braga I, Oliveira J, Pelizzola M, Henrique R, Jerónimo C. m6A RNA modification and its writer/reader VIRMA/YTHDF3 in testicular germ cell tumors: a role in seminoma phenotype maintenance. J Transl Med. 2019; 17:79. https://doi.org/10.1186/s12967-019-1837-z [PubMed]
- 181. Chen RX, Chen X, Xia LP, Zhang JX, Pan ZZ, Ma XD, Han K, Chen JW, Judde JG, Deas O, Wang F, Ma NF, Guan X, et al. N6-methyladenosine modification of circNSUN2 facilitates cytoplasmic export and stabilizes HMGA2 to promote colorectal liver metastasis. Nat Commun. 2019; 10:4695. https://doi.org/10.1038/s41467-019-12651-2 [PubMed]
- 182. Wen S, Wei Y, Zen C, Xiong W, Niu Y, Zhao Y. Long non-coding RNA NEAT1 promotes bone metastasis of prostate cancer through N6-methyladenosine. Mol Cancer. 2020; 19:171. https://doi.org/10.1186/s12943-020-01293-4 [PubMed]
- 183. Ban Y, Tan P, Cai J, Li J, Hu M, Zhou Y, Mei Y, Tan Y, Li X, Zeng Z, Xiong W, Li G, Li X, et al. LNCAROD is stabilized by m6A methylation and promotes cancer progression via forming a ternary complex with HSPA1A and YBX1 in head and neck squamous cell carcinoma. Mol Oncol. 2020; 14:1282–96. https://doi.org/10.1002/1878-0261.12676 [PubMed]
- 184. Wang J, Ding W, Xu Y, Tao E, Mo M, Xu W, Cai X, Chen X, Yuan J, Wu X. Long non-coding RNA RHPN1-AS1 promotes tumorigenesis and metastasis of ovarian cancer by acting as a ceRNA against miR-596 and upregulating LETM1. Aging (Albany NY). 2020; 12:4558–72. https://doi.org/10.18632/aging.102911 [PubMed]
- 185. Lin G, Wang H, Wu Y, Wang K, Li G. Hub Long Noncoding RNAs with m6A Modification for Signatures and Prognostic Values in Kidney Renal Clear Cell Carcinoma. Front Mol Biosci. 2021; 8:682471. https://doi.org/10.3389/fmolb.2021.682471 [PubMed]
- 186. Huang S, Lyu S, Gao Z, Zha W, Wang P, Shan Y, He J, Li Y. m6A-Related lncRNAs Are Potential Biomarkers for the Prognosis of Metastatic Skin Cutaneous Melanoma. Front Mol Biosci. 2021; 8:687760. https://doi.org/10.3389/fmolb.2021.687760 [PubMed]
- 187. Zhang X, Shen L, Cai R, Yu X, Yang J, Wu X, Zhu Y, Liu X. Comprehensive Analysis of the Immune-Oncology Targets and Immune Infiltrates of N6-Methyladenosine-Related Long Noncoding RNA Regulators in Breast Cancer. Front Cell Dev Biol. 2021; 9:686675. https://doi.org/10.3389/fcell.2021.686675 [PubMed]
- 188. Konno M, Koseki J, Asai A, Yamagata A, Shimamura T, Motooka D, Okuzaki D, Kawamoto K, Mizushima T, Eguchi H, Takiguchi S, Satoh T, Mimori K, et al. Distinct methylation levels of mature microRNAs in gastrointestinal cancers. Nat Commun. 2019; 10:3888. https://doi.org/10.1038/s41467-019-11826-1 [PubMed]
- 189. Geng X, Zhang Y, Li Q, Xi W, Yu W, Shi L, Lin X, Sun S, Wang H. Screening and functional prediction of differentially expressed circular RNAs in human glioma of different grades. Aging (Albany NY). 2020; 13:1989–2014. https://doi.org/10.18632/aging.202192 [PubMed]
- 190. Zhang B, Chen Z, Tao B, Yi C, Lin Z, Li Y, Shao W, Lin J, Chen J. m6A target microRNAs in serum for cancer detection. Mol Cancer. 2021; 20:170. https://doi.org/10.1186/s12943-021-01477-6 [PubMed]
- 191. Zhang P, Liu G, Lu L. N6-Methylandenosine-Related lncRNA Signature Is a Novel Biomarkers of Prognosis and Immune Response in Colon Adenocarcinoma Patients. Front Cell Dev Biol. 2021; 9:703629. https://doi.org/10.3389/fcell.2021.703629 [PubMed]
- 192. Xu F, Huang X, Li Y, Chen Y, Lin L. m6A-related lncRNAs are potential biomarkers for predicting prognoses and immune responses in patients with LUAD. Mol Ther Nucleic Acids. 2021; 24:780–91. https://doi.org/10.1016/j.omtn.2021.04.003 [PubMed]
- 193. Qiu Y, Wang X, Fan Z, Zhan S, Jiang X, Huang J. Integrated analysis on the N6-methyladenosine-related long noncoding RNAs prognostic signature, immune checkpoints, and immune cell infiltration in clear cell renal cell carcinoma. Immun Inflamm Dis. 2021; 9:1596–612. https://doi.org/10.1002/iid3.513 [PubMed]
- 194. Su R, Dong L, Li C, Nachtergaele S, Wunderlich M, Qing Y, Deng X, Wang Y, Weng X, Hu C, Yu M, Skibbe J, Dai Q, et al. R-2HG Exhibits Anti-tumor Activity by Targeting FTO/m6A/MYC/CEBPA Signaling. Cell. 2018; 172:90–105.e23. https://doi.org/10.1016/j.cell.2017.11.031 [PubMed]
- 195. Huang Y, Yan J, Li Q, Li J, Gong S, Zhou H, Gan J, Jiang H, Jia GF, Luo C, Yang CG. Meclofenamic acid selectively inhibits FTO demethylation of m6A over ALKBH5. Nucleic Acids Res. 2015; 43:373–84. https://doi.org/10.1093/nar/gku1276 [PubMed]
- 196. Cui Q, Shi H, Ye P, Li L, Qu Q, Sun G, Sun G, Lu Z, Huang Y, Yang CG, Riggs AD, He C, Shi Y. m6A RNA Methylation Regulates the Self-Renewal and Tumorigenesis of Glioblastoma Stem Cells. Cell Rep. 2017; 18:2622–34. https://doi.org/10.1016/j.celrep.2017.02.059 [PubMed]
- 197. Arun G, Diermeier S, Akerman M, Chang KC, Wilkinson JE, Hearn S, Kim Y, MacLeod AR, Krainer AR, Norton L, Brogi E, Egeblad M, Spector DL. Differentiation of mammary tumors and reduction in metastasis upon Malat1 lncRNA loss. Genes Dev. 2016; 30:34–51. https://doi.org/10.1101/gad.270959.115 [PubMed]
- 198. Daige CL, Wiggins JF, Priddy L, Nelligan-Davis T, Zhao J, Brown D. Systemic delivery of a miR34a mimic as a potential therapeutic for liver cancer. Mol Cancer Ther. 2014; 13:2352–60. https://doi.org/10.1158/1535-7163.MCT-14-0209 [PubMed]
- 199. Beg MS, Brenner AJ, Sachdev J, Borad M, Kang YK, Stoudemire J, Smith S, Bader AG, Kim S, Hong DS. Phase I study of MRX34, a liposomal miR-34a mimic, administered twice weekly in patients with advanced solid tumors. Invest New Drugs. 2017; 35:180–8. https://doi.org/10.1007/s10637-016-0407-y [PubMed]
- 200. Hong DS, Kang YK, Borad M, Sachdev J, Ejadi S, Lim HY, Brenner AJ, Park K, Lee JL, Kim TY, Shin S, Becerra CR, Falchook G, et al. Phase 1 study of MRX34, a liposomal miR-34a mimic, in patients with advanced solid tumours. Br J Cancer. 2020; 122:1630–7. https://doi.org/10.1038/s41416-020-0802-1 [PubMed]
- 201. Zhang X, Wang S, Wang H, Cao J, Huang X, Chen Z, Xu P, Sun G, Xu J, Lv J, Xu Z. Circular RNA circNRIP1 acts as a microRNA-149-5p sponge to promote gastric cancer progression via the AKT1/mTOR pathway. Mol Cancer. 2019; 18:20. https://doi.org/10.1186/s12943-018-0935-5 [PubMed]
- 202. Qu L, Yi Z, Shen Y, Lin L, Chen F, Xu Y, Wu Z, Tang H, Zhang X, Tian F, Wang C, Xiao X, Dong X, et al. Circular RNA vaccines against SARS-CoV-2 and emerging variants. Cell. 2022; 185:1728–44.e16. https://doi.org/10.1016/j.cell.2022.03.044 [PubMed]
- 203. Zuo X, Chen Z, Gao W, Zhang Y, Wang J, Wang J, Cao M, Cai J, Wu J, Wang X. M6A-mediated upregulation of LINC00958 increases lipogenesis and acts as a nanotherapeutic target in hepatocellular carcinoma. J Hematol Oncol. 2020; 13:5. https://doi.org/10.1186/s13045-019-0839-x [PubMed]
- 204. Xu J, Wan Z, Tang M, Lin Z, Jiang S, Ji L, Gorshkov K, Mao Q, Xia S, Cen D, Zheng J, Liang X, Cai X. N6-methyladenosine-modified CircRNA-SORE sustains sorafenib resistance in hepatocellular carcinoma by regulating β-catenin signaling. Mol Cancer. 2020; 19:163. https://doi.org/10.1186/s12943-020-01281-8 [PubMed]
- 205. Zhao J, Lee EE, Kim J, Yang R, Chamseddin B, Ni C, Gusho E, Xie Y, Chiang CM, Buszczak M, Zhan X, Laimins L, Wang RC. Transforming activity of an oncoprotein-encoding circular RNA from human papillomavirus. Nat Commun. 2019; 10:2300. https://doi.org/10.1038/s41467-019-10246-5 [PubMed]



