Introduction
Since the turn of the century, the incidence and mortality of cancer have increased significantly compared to that in previous years [1]. Although various treatment options, such as surgery, radiation, and chemotherapy, are available, the overall survival rates of patients with cancer have not improved significantly [2]. Therefore, identification of new tumor biomarkers and therapeutic targets can better guide clinical tumor treatment.
Non-SMC condensin I complex subunit G (NCAPG) is a subunit of the condensin complex, which condenses and stabilizes chromosomes during mitosis and meiosis [3]. Knockdown of NCAPG significantly reduced the viability of hepatocellular carcinoma cells by regulating Bax, cleaved caspase-3, E-cadherin, N-cadherin, cyclin A1, CDK2, and Bcl-2, and the expression of HOXB9 induces apoptosis and cell cycle arrest in the DNA synthesis phase [4]. In addition, upregulation of NCAPG can activate multiple signaling pathways to promote cell proliferation and anti-apoptotic activity and regulate DNA replication and mismatch repair in different cancer types [5–7].
Studies have shown that NCAPG is overexpressed in several tumors and associated with clinical features of cancer, such as tumor proliferation, metastasis, invasion, and patient survival [5, 8–10]. However, its role in various types of tumors remains controversial. For example, NCAPG is overexpressed in hepatocellular carcinoma [11] and glioma [12] but underexpressed in out-of-niche primary tumor cells of multiple myeloma and acute myeloid leukemia [13, 14]. Therefore, we performed a meta-analysis to explore the relationship between NCAPG upregulation and the clinical characteristics of cancer, analyze the prognostic value of NCAPG for cancer patients, and validate its role by bioinformatics methods.
Materials and Methods
Literature search
Two authors independently searched four databases, namely, Pubmed, Embase, Web of Science, and the Cochrane Library for studies published before April 30, 2022. The following search terms were used: (“Neoplasms” OR “Carcinoma” OR “Prognosis” OR “Diagnosis” OR “Survival”) AND (“non-SMC condensin I complex subunit G” OR “NCAPG”).
Inclusion criteria
The following inclusion criteria were considered when screening the databases: (1) the original literature was in English; (2) cancers with abnormal NCAPG expression were investigated; (3) high and low NCAPG expression was delineated; and (4) HR and 95% CIs of the OS can be obtained or calculated from the survival curve.
Exclusion criteria
The exclusion criteria were as follows: (1) reviews, publication letters, retracted literature, and case reports; (2) insufficient data; (3) bioinformatics analysis; and (4) studies not relevant to NCAPG.
Data extraction
Data extraction was performed by two investigators for all included studies and submitted to a third researcher to resolve disagreements. The following data were extracted according to the inclusion criteria: first author, publication date, country of origin, cancer type, number of cases, follow-up time, measurement method of NCAPG expression, outcome measures, HR and 95% CIs for OS.
Quality assessment
The quality of the literature was evaluated using the Newcastle–Ottawa Scale. The evaluation was conducted independently by two investigators, and when disagreements arose, a third investigator participated in the discussion. The total score was 9 points, and a score of ≥6 points indicated high-quality research [15].
Validation of the bioinformatics database
The GEPIA2.0 database (http://gepia.cancer-pku.cn/index.html) is a platform for sequencing and expression data, that includes most tumors and normal tissue samples [16]. Moreover, this study used the “Expression DIY” module to explore the differences between NCAPG transcripts from cancer tissue samples and normal tissue samples. In addition, we downloaded tumor transcription samples from the TCGA database (https://portal.gdc.cancer.gov/) [17] and used R (survival and timeROC packages) to perform a cox regression and ROC analysis of the survival rate. Next, we used the Kaplan-Meier Plotter database (https://kmplot.com/analysis/) to analyze the effect of the NCAPG gene on the survival rate in different cancers for additional data supplementation [18]. To validate the prognostic tumor status of NCAPG, we utilized the PrognoScan database to verify the survival information of this gene in multiple cancer datasets. We used the UALCAN database (http://ualcan.path.uab.edu/index.html) [19] to validate clinical information on NCAPG expression in tumors and explore the methylation and phosphorylation of NCAPG in these tumors.
Molecular role and functional enrichment analysis of NCAPG
GeneMANIA (http://www.genemania.org) is an online tool for protein-protein interactions that helped in identifying genes with similar functions relative to that of the NCAPG gene in this study [20, 21]. We used the STRING database (https://string-db.org/) for GO and KEGG pathway analyses of NCAPG [22]. Finally, we used the multiMiR package (version 4.12) to identify competitive endogenous RNA of NCAPG and constructed a NCAPG network of competitive endogenous RNA interactions of target genes through Cytoscape software (version 3.8.2, https://cytoscape.org/) [23].
Data processing and statistical analysis
The K-M curves of the included studies were processed by Enguage Digitizer 11.3 software to obtain HR values and 95% CIs. Further data analysis was performed using Review Manager 5.3 software. Survival outcomes were calculated by logarithmic HR values and their standard errors. In addition, the correlation between NCAPG upregulation and clinicopathological parameters of cancers (age, gender, degree of differentiation, TNM stage, metastasis, vascular invasion) was assessed by calculating the ORs and 95% CIs. Cochran’s Q test and I2 test assessed the heterogeneity to determine the effect model. A fixed model was used if the included studies had no significant heterogeneity (I2 < 50%, P > 0.1), while a random model was used otherwise. A sensitivity analysis of the included studies was performed using STATA 12.0 software to assess the stability of the results. Publication bias was evaluated using Begg’s rank correlation and Egger’s linear regression. At P < 0.05, publication bias was observed. If publication bias was present, then the trim-and-fill method was used to further assess the stability of the pooled results.
Data availability statement
All relevant data is contained within the article: The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding authors.
Results
Literature selection
A flow chart depicting the literature screening process is shown in Figure 1. A total of 340 articles were screened, and 180 duplicate articles were excluded by searching PubMed, EMBASE, the Cochrane Library, and Web of Science. After scrutinizing the titles and abstracts, 133 more studies were excluded. As eight studies did not record the hazard ratios (HRs) and/or Kaplan-Meier (K-M) curves for overall survival (OS), 11 articles were excluded from bioinformatics analysis. Finally, we included eight studies in the meta-analysis [5, 6, 8, 9, 12, 24–26], all of which were cohort studies, and all outcome measures were OS.
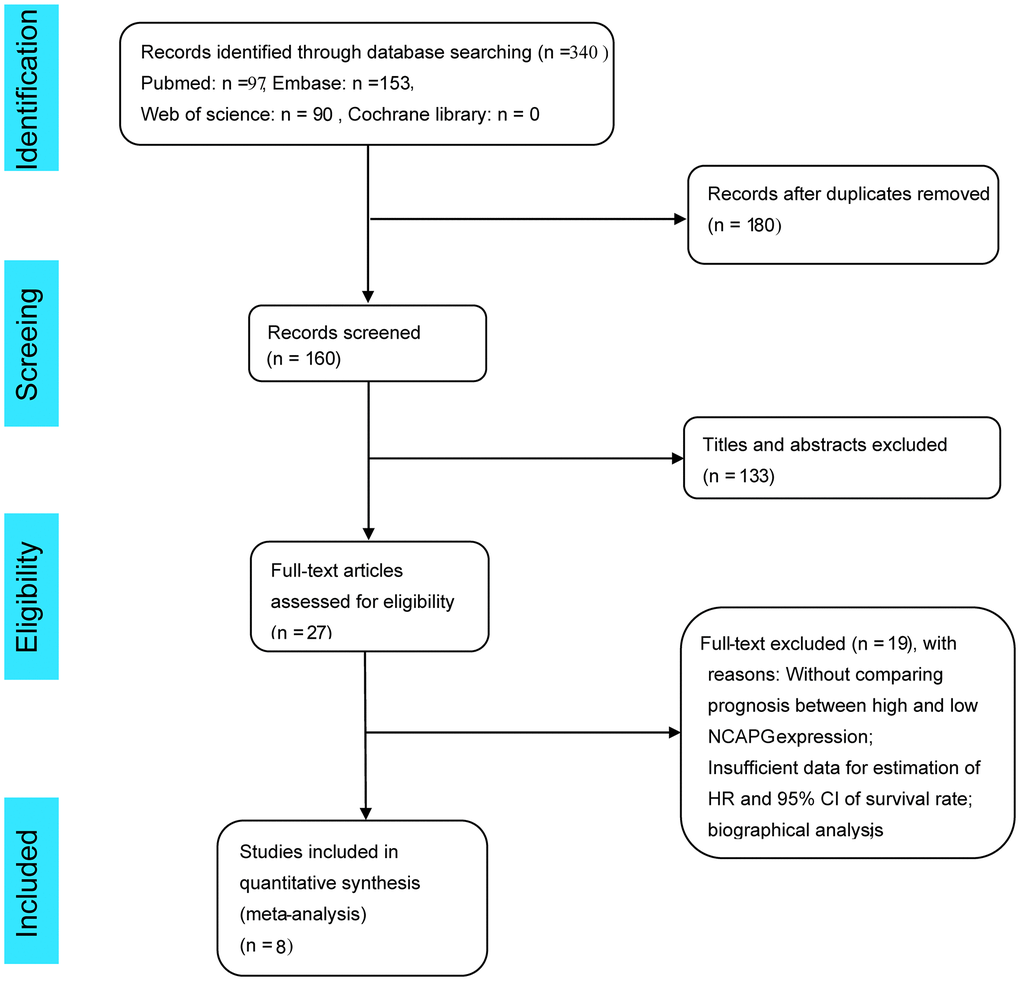
Figure 1. Flow chart of literature screening for this meta-analysis.
Study characteristics and quality assessment
The characteristics of the included studies are presented in Table 1. All the studies were conducted in China, and 1096 patients were recruited. The types of cancer included non-small cell lung cancer (NSCLC), glioma, breast cancer, gastric cancer, and hepatocellular carcinoma. All included studies assessed the association between OS and NCAPG expression, with follow-ups ranging from 80 to 140 months. All but one study reported clinicopathological parameters. All studies measured NCAPG expression via immunohistochemical staining, except for one study that performed RNA sequencing instead. Newcastle–Ottawa Scale scores were ≥6, indicating that the included studies were of moderate to high quality.
Table 1. Characteristics of included studies.
| Name | Year | Region | Ethnic | Type | Sample size (high/low) | FollowUp (months) | Method | Outcome | HR estimation method | HR (95% CI) | NOS |
| Jiang | 2020 | China | Asian | BC | 103 (35/68) | 120 | IHC | OS | K-M | 7.57 (3.13, 18.29) | 7 |
| Sun | 2022 | China | Asian | NSCLC | 156 (84/72) | 140 | IHC | OS, CP | REP | 2.35 (1.3, 4.27) | 7 |
| Sun | 2020 | China | Asian | GC | 135 (71/64) | 120 | IHC | OS, CP | REP | 2.03 (1.23, 3.35) | 7 |
| Wang | 2022 | China | Asian | NSCLC | 60 (28/32) | 80 | IHC | OS, CP | K-M | 3.45 (1.26, 9.49) | 6 |
| Wang | 2019 | China | Asian | HCC | 70 (35/35) | 120 | RNA-Seq | OS, CP | K-M | 2.34 (1.08, 5.07) | 7 |
| Wu | 2021 | China | Asian | NSCLC | 292 (164/128) | 120 | IHC | OS, CP | REP | 2.05 (1.35, 3.11) | 7 |
| Zheng | 2022 | China | Asian | Glioma | 140 (47/93) | 120 | IHC | OS, CP | K-M | 9.34 (3.75, 23.26) | 7 |
| Zhou | 2022 | China | Asian | NSCLC | 140 (70/70) | 100 | IHC | OS, CP | REP | 2.32 (1.41, 3.81) | 6 |
Correlation of NCAPG expression with OS and cancer type
Eight studies reported that NCAPG upregulation was associated with tumors. Therefore, a pooled analysis of the eight studies was performed. As shown in Figure 2, a random model was used because of the absence of obvious heterogeneity (I2 = 56%, P = 0.02). The pooled results suggested that high NCAPG expression rates were correlated with worse OS in patients with different cancers (HR = 2.90, 95% confidence interval (CI) = 2.06–4.10, P < 0.00001).
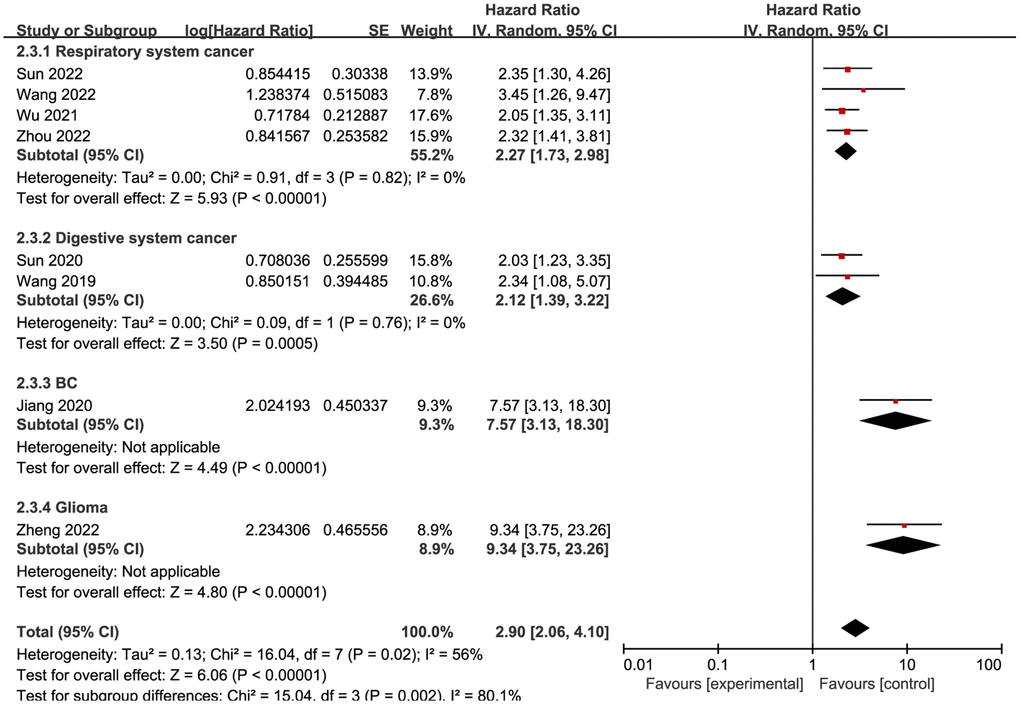
Figure 2. Forest plot of the pooled OS for subgroup analysis.
Due to the existence of heterogeneity, a subgroup analysis was performed to further explore the impact of high NCAPG expression on survival in different types of cancer (Figure 2). Four studies [5, 9, 25, 26] investigated cancers of the respiratory system, two studies [8, 24] investigated cancers of the digestive system, and the remaining two studies investigated other types of cancers, namely, glioma [12] and breast cancer [6]. According to the forest plot, upregulation of NCAPG expression in cancer tissues was related to worse OS regardless of the group (respiratory cancer, HR = 2.27, 95% CI = 1.73–2.98, P < 0.00001; digestive cancer, HR = 2.12, 95% CI = 1.39–3.22, P = 0.0005; breast cancer, HR = 7.57, 95% CI = 3.13–18.30, P < 0.00001; glioma, HR = 9.34, 95% CI = 3.75–23.26, P < 0.00001).
Correlation of NCAPG expression with clinicopathological parameters
As all studies included in this meta-analysis reported clinicopathological parameters, we analyzed the high expression of NCAPG and these parameters. As shown in Figure 3, the upregulation of NCAPG was not significantly correlated with gender (male vs. female, odds ratio (OR) = 1.20, 95% CI = 0.92–1.56, P = 0.19, fixed model; Figure 3A), age (young vs. old, OR = 0.68, 95% CI = 0.44–1.06, P = 0.09, random model; Figure 3B), vascular invasion (yes vs. no, OR = 2.23, 95% CI = 0.81–6.11, P = 0.12, random model; Figure 3D), differentiation (well differentiated vs. poorly differentiated, OR = 1.20, 95% CI = 0.50–2.90, P = 0.68, fixed model; Figure 3E), TNM stage (III–IV vs. I–II, OR = 1.64, 95% CI = 0.92–2.92, P = 0.09, random model; Figure 3G), or T classification (T3+T4 vs. T1+T2, OR = 1.68, 95% CI = 0.83–3.40, P = 0.15, random model; Figure 3J), although it was correlated with distant metastasis (yes vs. no, OR = 5.65, 95% CI 2.60–12.26, P < 0.0001, fixed model; Figure 3C), lymph node metastasis (yes vs. no, OR = 2.08, 95% CI = 1.54–2.80, P < 0.00001, fixed model; Figure 3F), relapse (yes vs. no, OR = 2.62, 95% CI = 1.02–6.70, P = 0.04, random model; Figure 3H), and clinical stage (III–IV vs. I–II, OR = 2.23, 95% CI = 1.44–3.44, P = 0.0003, fixed model; Figure 3I). The results of database validation of the clinicopathological characteristics and NCAPG expression data (Supplementary Figure 1) are consistent with most of our previous meta-analysis results.
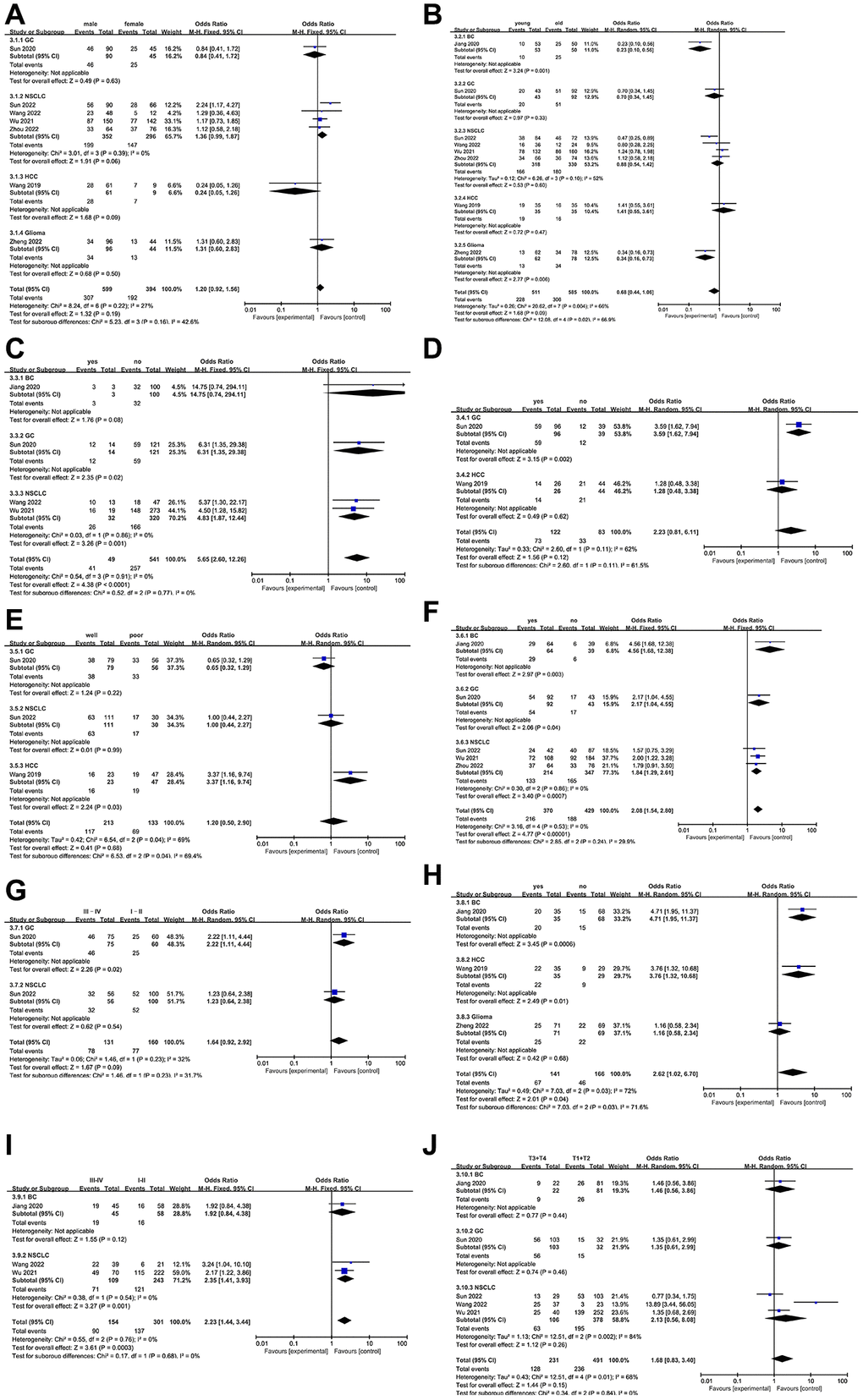
Figure 3. Forest plot of the relationship between high NCAPG expression and clinicopathological parameters. (A) gender, (B) age, (C) distant metastasis, (D) vascular invasion, (E) differentiation, (F) lymph node metastasis, (G) TNM stage, (H) relapse, (I) clinical stage, (J) T classification.
Sensitivity analysis and publication bias
To verify the robustness of the results, we performed a sensitivity analysis (Figure 4). Removal of one or more articles did not significantly affect the results, indicating that the results were relatively stable. Begg’s and Egger’s tests indicated publication bias for OS and distant metastasis (Table 2, Supplementary Figure 2). We analyzed the stability of the study further using the trim-and-fill method. For OS, the results indicated that the estimated number of missing studies was 0 and the adjusted HR was 2.90 (95% CI = 2.06–4.10, P < 0.001), which indicated that upregulation of NCAPG was associated with poorer OS, suggesting that our result is reliable. In addition, for distant metastases, two studies were estimated to be missing and the adjusted OR was 4.85 (95% CI = 2.48–9.58, P < 0.001), which indicated a reliable result.
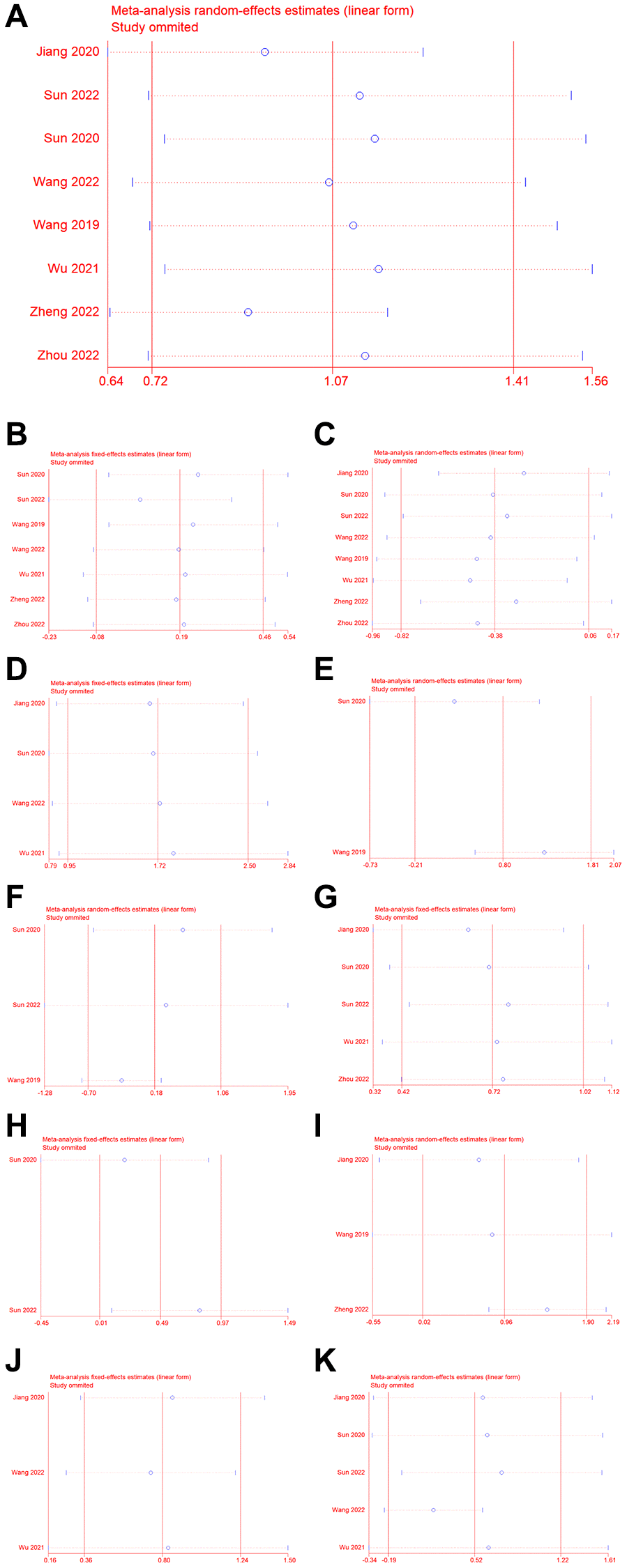
Figure 4. Sensitivity analysis. (A) OS, (B) gender, (C) age, (D) distant metastasis, (E) vascular invasion, (F) differentiation, (G) lymph node metastasis, (H) TNM stage, (I) relapse, (J) clinical stage, (K) T classification.
Table 2. Results of Begg’s and Egger’s tests for publication bias.
| Analysis type | Begg’s test | Egger’s test | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Z | P | t | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS | 2.60 | 0.009 | 3.13 | 0.020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (male vs. female) | 0.90 | 0.368 | −1.08 | 0.328 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (young vs. old) | 0.62 | 0.536 | −1.16 | 0.291 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distant metastasis (yes vs. no) | 1.70 | 0.089 | 6.99 | 0.020 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vascular invasion (yes vs. no) | 0.00 | 1.000 | NA | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Differentiation (well vs. poor) | 1.04 | 0.296 | 12.42 | 0.051 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lymph node metastasis (yes vs. no) | 0.24 | 0.806 | 1.15 | 0.332 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM stage (III−IV vs. I−II) | 0.00 | 1.000 | NA | NA | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Relapse (yes vs. no) | 0.00 | 1.000 | 1.59 | 0.357 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Clinical stage (III−IV vs. I−II) | 0.00 | 1.000 | 0.79 | 0.573 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T classification (T3+T4 vs. T1+T2) | 1.22 | 0.221 | 2.27 | 0.108 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| If P < 0.05, the results are in bold. Abbreviation: NA: Not available. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Validation of NCAPG expression against public databases
We evaluated the NCAPG expression levels and performed a survival analysis in various cancers using the GEPIA2.0 database to validate our results. The findings showed that, compared with normal tissues, the expression of NCAPG was significantly upregulated in tumors, including BRCA, GBM, LIHC, LUAD, LUSC, and STAD (Figure 5A). We next performed a univariate Cox survival analysis on the above tumors, and it showed that the OS of LIHC and LUAD was related to NCAPG (Figure 5B). The progression-free survival (PFS) of BRCA, LIHC, LUAD, and STAD was related to NCAPG (Figure 5C). The relapse-free survival (RFS) of LIHC, LUSC, and STAD was related to NCAPG (Figure 5D), and the disease-specific survival (DSS) of LIHC and LUAD was related to NCAPG (Figure 5E). We then performed receiver operating characteristic (ROC) survival analysis of the data on the above tumors (Supplementary Figure 3), and the area under the curve of GBM, LIHC, and STAD was greater than 0.7. Therefore, we concluded that NCAPG may be a good prognostic indicator of various cancers. In addition, we analyzed the survival prognosis of the above tumors using the Kaplan-Meier plotter database based on the median cutoff value of NCAPG expression (including probes 218663_at and 218663_s_at) in cancer. The results showed that NCAPG expression can be used to predict OS in breast cancer (P < 0.01; Figure 6A), RFS (P < 0.01; Figure 6B), PPS (P < 0.01; Figure 6C), DMFS (P < 0.01; Figure 6D), liver cancer, PFS, PFS, and DSS (P < 0.01; Figure 6E), lung cancer (P < 0.01; Figure 6F), FP (P < 0.01; Figure 6G), and PPS (P < 0.01; Figure 6H), gastric cancer (P < 0.01; Figure 6I), FP (P < 0.01; Figure 6J,), and PPS (P < 0.01; Figure 6K). Moreover, the findings were validated against multiple cancer datasets using the PrognoScan database. We collected 29 datasets on breast and lung cancers. As shown in Table 3, NCAPG expression in these tumors significantly affected prognosis-related indicators, such as OS, DSS, RFS, and PFS (P < 0.05).
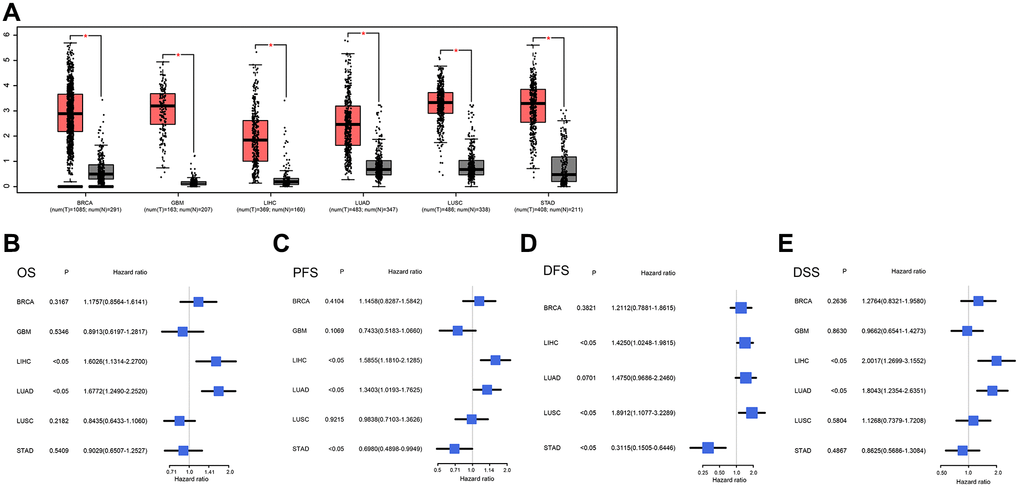
Figure 5. (A) Expression levels of NCAPG in cancer tissues and normal tissues in GEPIA2. From left to right are gastric cancer (STAD), lung cancer (LUAD), liver cancer (LIHC), glioma (GBM) and breast cancer (BRCA). The red box represents the expression level of NCAPG in cancer tissues; the gray box represents the expression level of NCAPG in normal tissues, the screening criteria were log2FC|>1 and P < 0.01, (B) OS of BRCA, GBM, LIHC, LUAD, LUSC and STAD, (C) PFS of BRCA, GBM, LIHC, LUAD, LUSC and STAD, (D) DFS of BRCA, LIHC, LUAD, LUSC and STAD, (E) BRCA, GBM, LIHC, LUAD, LUSC and STAD's DSS.
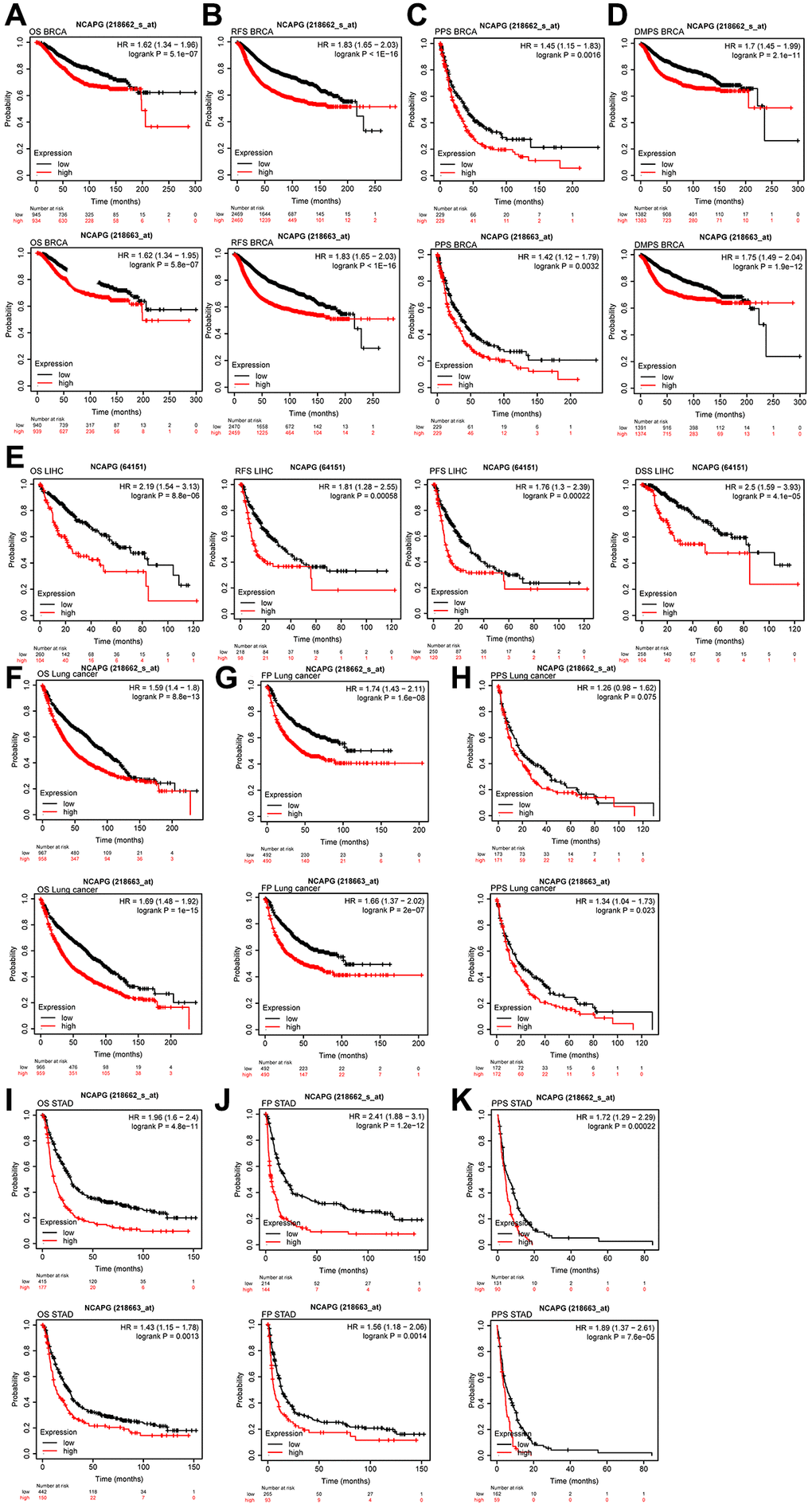
Figure 6. (A) OS of NCAPG (218663-s-at) in BRCA (n = 1879), OS of NCAPG (218663-at) in BRCA (n = 1879), (B) RFS of NCAPG (218663-s-at) in BRCA (n = 4929), RFS of NCAPG (218663-at) in BRCA (n = 4929), (C) PPS of NCAPG (218663-s-at) in BRCA (n = 458), PPS of NCAPG (218663-at) in BRCA (n = 458)(D) DMPS of NCAPG (218663-s-at) in BRCA (n = 2765), DMPS of NCAPG (218663-at) in BRCA (n = 2765), (E) OS (n = 364), PFS (n = 316), PFS (n = 370)and DSS (n = 362)of NCAPG in LIHC, (F) OS of NCAPG (218663-s-at) in lung cancer (n = 1925), OS of NCAPG (218663-at) in lung cancer (n = 1925), (G) FP of NCAPG (218663-s-at) in lung cancer (n = 982), FP of NCAPG (218663-at) in lung cancer (n = 982), (H) PPS of NCAPG (218663-s-at) in lung cancer (n = 344), PPS of NCAPG (218663-at) in lung cancer (n = 344), (I) OS of NCAPG (218663-s-at) in STAD (n = 592), OS of NCAPG (218663-at) in STAD (n = 592), (J) FP of NCAPG (218663-s-at) in STAD (n = 358), FP of NCAPG (218663-at) in STAD (n = 358), (K) PPS of NCAPG (218663-s-at) in STAD (n = 221), PPS of NCAPG (218663-at) in STAD (n = 221).
Table 3. NCAPG-related cancer GEO database classifications.
| Dataset | Cancer type | Prognostic factor | Cox P-value | ln (HR) | HR (95% CI) |
| GSE5287 | Bladder cancer | Overall Survival | 0.043472 | 0.555305 | 1.74 (1.02–2.99) |
| GSE5287 | Bladder cancer | Overall Survival | 0.328603 | 0.223676 | 1.25 (0.80–1.96) |
| GSE13507 | Bladder cancer | Overall Survival | 0.000399636 | 0.380311 | 1.46 (1.19–1.81) |
| GSE13507 | Bladder cancer | Disease Specific Survival | 7.89E-05 | 0.698904 | 2.01 (1.42–2.85) |
| GSE12417-GPL96 | Blood cancer | Overall Survival | 0.294458 | −0.153815 | 0.86 (0.64–1.14) |
| GSE12417-GPL96 | Blood cancer | Overall Survival | 0.391317 | −0.121301 | 0.89 (0.67–1.17) |
| GSE12417-GPL570 | Blood cancer | Overall Survival | 0.924132 | −0.0155483 | 0.98 (0.71–1.36) |
| GSE12417-GPL570 | Blood cancer | Overall Survival | 0.895552 | 0.0205993 | 1.02 (0.75–1.39) |
| GSE5122 | Blood cancer | Overall Survival | 0.852157 | −0.025255 | 0.98 (0.75–1.27) |
| GSE5122 | Blood cancer | Overall Survival | 0.980633 | −0.00489207 | 1.00 (0.67–1.48) |
| GSE8970 | Blood cancer | Overall Survival | 0.116843 | −0.317877 | 0.73 (0.49–1.08) |
| GSE8970 | Blood cancer | Overall Survival | 0.14972 | −0.274213 | 0.76 (0.52–1.10) |
| GSE4475 | Blood cancer | Overall Survival | 0.0095096 | −0.464977 | 0.63 (0.44–0.89) |
| E-TABM-346 | Blood cancer | Overall Survival | 0.865305 | −0.0630235 | 0.94 (0.45–1.94) |
| E-TABM-346 | Blood cancer | Overall Survival | 0.951871 | 0.0174584 | 1.02 (0.58–1.79) |
| E-TABM-346 | Blood cancer | Event Free Survival | 0.707167 | −0.126166 | 0.88 (0.46–1.70) |
| E-TABM-346 | Blood cancer | Event Free Survival | 0.998653 | 0.000438628 | 1.00 (0.60–1.66) |
| GSE16131-GPL96 | Blood cancer | Overall Survival | 0.86364 | 0.0296222 | 1.03 (0.73–1.44) |
| GSE16131-GPL96 | Blood cancer | Overall Survival | 0.774936 | −0.0475906 | 0.95 (0.69–1.32) |
| GSE2658 | Blood cancer | Disease Specific Survival | 0.0835894 | 0.410555 | 1.51 (0.95–2.40) |
| GSE2658 | Blood cancer | Disease Specific Survival | 0.00039476 | 0.382343 | 1.47 (1.19–1.81) |
| GSE4271 | Brain cancer | Overall Survival | 0.00174277 | 0.58775 | 1.80 (1.25–2.60) |
| GSE7696 | Brain cancer | Overall Survival | 0.993869 | 0.00119241 | 1.00 (0.74–1.36) |
| GSE7696 | Brain cancer | Overall Survival | 0.567686 | 0.0919879 | 1.10 (0.80–1.50) |
| GSE16581 | Brain cancer | Overall Survival | 0.844517 | 0.141127 | 1.15 (0.28–4.72) |
| GSE16581 | Brain cancer | Overall Survival | 0.542849 | 0.658295 | 1.93 (0.23–16.10) |
| GSE19615 | Breast cancer | Distant Metastasis Free Survival | 0.375869 | 0.346581 | 1.41 (0.66–3.05) |
| GSE19615 | Breast cancer | Distant Metastasis Free Survival | 0.53393 | 0.279045 | 1.32 (0.55–3.18) |
| GSE12276 | Breast cancer | Relapse Free Survival | 0.000105043 | 0.427798 | 1.53 (1.24–1.90) |
| GSE6532-GPL570 | Breast cancer | Distant Metastasis Free Survival | 0.115457 | 0.314844 | 1.37 (0.93–2.03) |
| GSE6532 | Breast cancer | Distant Metastasis Free Survival | 0.0361386 | 0.384826 | 1.47 (1.03–2.11) |
| GSE6532-GPL570 | Breast cancer | Relapse Free Survival | 0.115457 | 0.314844 | 1.37 (0.93–2.03) |
| GSE6532 | Breast cancer | Relapse Free Survival | 0.0361386 | 0.384826 | 1.47 (1.03–2.11) |
| GSE9195 | Breast cancer | Distant Metastasis Free Survival | 0.434869 | 0.261618 | 1.30 (0.67–2.50) |
| GSE9195 | Breast cancer | Distant Metastasis Free Survival | 0.165806 | 0.510274 | 1.67 (0.81–3.43) |
| GSE9195 | Breast cancer | Relapse Free Survival | 0.224745 | 0.397302 | 1.49 (0.78–2.83) |
| GSE9195 | Breast cancer | Relapse Free Survival | 0.499771 | 0.202634 | 1.22 (0.68–2.21) |
| GSE12093 | Breast cancer | Distant Metastasis Free Survival | 0.0122697 | 0.923643 | 2.52 (1.22–5.19) |
| GSE11121 | Breast cancer | Distant Metastasis Free Survival | 0.00994604 | 0.574941 | 1.78 (1.15–2.75) |
| GSE1378 | Breast cancer | Relapse Free Survival | 0.755055 | 0.0639828 | 1.07 (0.71–1.59) |
| GSE1379 | Breast cancer | Relapse Free Survival | 0.728436 | 0.0804905 | 1.08 (0.69–1.71) |
| GSE2034 | Breast cancer | Distant Metastasis Free Survival | 0.00249838 | 0.515644 | 1.67 (1.20–2.34) |
| GSE1456 | Breast cancer | Overall Survival | 0.000786964 | 1.06803 | 2.91 (1.56–5.43) |
| GSE1456 | Breast cancer | Disease Specific Survival | 0.000648569 | 0.983901 | 2.67 (1.52–4.71) |
| GSE1456 | Breast cancer | Relapse Free Survival | 0.000279683 | 1.15084 | 3.16 (1.70–5.88) |
| GSE7378 | Breast cancer | Disease Free Survival | 0.0335011 | 0.654287 | 1.92 (1.05–3.52) |
| GSE7378 | Breast cancer | Disease Free Survival | 0.0709317 | 0.554641 | 1.74 (0.95–3.18) |
| E-TABM-158 | Breast cancer | Distant Metastasis Free Survival | 0.681173 | 0.0873489 | 1.09 (0.72–1.66) |
| E-TABM-158 | Breast cancer | Overall Survival | 0.203745 | −0.227607 | 0.80 (0.56–1.13) |
| E-TABM-158 | Breast cancer | Relapse Free Survival | 0.361575 | −0.17199 | 0.84 (0.58–1.22) |
| E-TABM-158 | Breast cancer | Disease Specific Survival | 0.0899647 | −0.399162 | 0.67 (0.42–1.06) |
| E-TABM-158 | Breast cancer | Overall Survival | 0.361575 | −0.17199 | 0.84 (0.58–1.22) |
| E-TABM-158 | Breast cancer | Distant Metastasis Free Survival | 0.757249 | 0.0666703 | 1.07 (0.70–1.63) |
| E-TABM-158 | Breast cancer | Relapse Free Survival | 0.203745 | −0.227607 | 0.80 (0.56–1.13) |
| E-TABM-158 | Breast cancer | Disease Specific Survival | 0.0351901 | −0.454509 | 0.63 (0.42–0.97) |
| GSE3494 | Breast cancer | Disease Specific Survival | 0.00163843 | 0.590531 | 1.80 (1.25–2.61) |
| GSE4922 | Breast cancer | Disease Free Survival | 4.93E-05 | 0.808544 | 2.24 (1.52–3.32) |
| GSE2990 | Breast cancer | Distant Metastasis Free Survival | 0.0759889 | 0.351849 | 1.42 (0.96–2.10) |
| GSE2990 | Breast cancer | Relapse Free Survival | 0.105799 | 0.500621 | 1.65 (0.90–3.03) |
| GSE2990 | Breast cancer | Relapse Free Survival | 0.123393 | 0.239449 | 1.27 (0.94–1.72) |
| GSE2990 | Breast cancer | Distant Metastasis Free Survival | 0.00239721 | 0.895966 | 2.45 (1.37–4.37) |
| GSE2990 | Breast cancer | Distant Metastasis Free Survival | 0.0796503 | 0.412016 | 1.51 (0.95–2.39) |
| GSE2990 | Breast cancer | Relapse Free Survival | 0.1058 | 0.313035 | 1.37 (0.94–2.00) |
| GSE2990 | Breast cancer | Distant Metastasis Free Survival | 0.142013 | 0.556377 | 1.74 (0.83–3.67) |
| GSE2990 | Breast cancer | Relapse Free Survival | 0.00369961 | 0.72696 | 2.07 (1.27–3.38) |
| GSE7390 | Breast cancer | Distant Metastasis Free Survival | 0.17505 | 0.143945 | 1.15 (0.94–1.42) |
| GSE7390 | Breast cancer | Overall Survival | 0.0654623 | 0.211501 | 1.24 (0.99–1.55) |
| GSE7390 | Breast cancer | Relapse Free Survival | 0.217378 | 0.140286 | 1.15 (0.92–1.44) |
| GSE7390 | Breast cancer | Distant Metastasis Free Survival | 0.279784 | 0.152457 | 1.16 (0.88–1.54) |
| GSE7390 | Breast cancer | Relapse Free Survival | 0.133969 | 0.127109 | 1.14 (0.96–1.34) |
| GSE7390 | Breast cancer | Overall Survival | 0.203224 | 0.191366 | 1.21 (0.90–1.63) |
| GSE12945 | Colorectal cancer | Disease Free Survival | 0.390324 | 0.626282 | 1.87 (0.45–7.81) |
| GSE12945 | Colorectal cancer | Overall Survival | 0.115402 | 0.789413 | 2.20 (0.82–5.88) |
| GSE12945 | Colorectal cancer | Disease Free Survival | 0.672657 | −0.773745 | 0.46 (0.01–16.70) |
| GSE12945 | Colorectal cancer | Overall Survival | 0.16887 | 1.33458 | 3.80 (0.57–25.43) |
| GSE17536 | Colorectal cancer | Disease Specific Survival | 0.589054 | −0.130516 | 0.88 (0.55–1.41) |
| GSE17536 | Colorectal cancer | Disease Specific Survival | 0.828392 | 0.0428782 | 1.04 (0.71–1.54) |
| GSE17536 | Colorectal cancer | Overall Survival | 0.856783 | 0.0390747 | 1.04 (0.68–1.59) |
| GSE17536 | Colorectal cancer | Overall Survival | 0.262151 | 0.198586 | 1.22 (0.86–1.73) |
| GSE17536 | Colorectal cancer | Disease Free Survival | 0.109121 | −0.506703 | 0.60 (0.32–1.12) |
| GSE17536 | Colorectal cancer | Disease Free Survival | 0.268629 | −0.263513 | 0.77 (0.48–1.23) |
| GSE14333 | Colorectal cancer | Disease Free Survival | 0.0827771 | −0.427442 | 0.65 (0.40–1.06) |
| GSE14333 | Colorectal cancer | Disease Free Survival | 0.021572 | −0.444143 | 0.64 (0.44–0.94) |
| GSE17537 | Colorectal cancer | Overall Survival | 0.908953 | 0.0357876 | 1.04 (0.56–1.91) |
| GSE17537 | Colorectal cancer | Overall Survival | 0.616503 | 0.136126 | 1.15 (0.67–1.95) |
| GSE17537 | Colorectal cancer | Disease Free Survival | 0.713261 | 0.125822 | 1.13 (0.58–2.22) |
| GSE17537 | Colorectal cancer | Disease Free Survival | 0.33555 | 0.294475 | 1.34 (0.74–2.44) |
| GSE17537 | Colorectal cancer | Disease Specific Survival | 0.142937 | 0.741175 | 2.10 (0.78–5.66) |
| GSE17537 | Colorectal cancer | Disease Specific Survival | 0.0989951 | 0.789785 | 2.20 (0.86–5.63) |
| GSE22138 | Eye cancer | Distant Metastasis Free Survival | 0.440403 | 0.174746 | 1.19 (0.76–1.86) |
| GSE22138 | Eye cancer | Distant Metastasis Free Survival | 0.0942372 | 0.746158 | 2.11 (0.88–5.05) |
| GSE2837 | Head and neck cancer | Relapse Free Survival | 0.160158 | −0.644551 | 0.52 (0.21–1.29) |
| jacob-00182-CANDF | Lung cancer | Overall Survival | 0.426986 | 0.197876 | 1.22 (0.75–1.99) |
| jacob-00182-CANDF | Lung cancer | Overall Survival | 0.184801 | 0.237298 | 1.27 (0.89–1.80) |
| jacob-00182-HLM | Lung cancer | Overall Survival | 0.565996 | 0.0919948 | 1.10 (0.80–1.50) |
| jacob-00182-HLM | Lung cancer | Overall Survival | 0.572148 | 0.0813385 | 1.08 (0.82–1.44) |
| jacob-00182-MSK | Lung cancer | Overall Survival | 0.0865412 | 0.267983 | 1.31 (0.96–1.78) |
| jacob-00182-MSK | Lung cancer | Overall Survival | 0.0414568 | 0.367399 | 1.44 (1.01–2.06) |
| GSE13213 | Lung cancer | Overall Survival | 0.00524186 | 0.3675 | 1.44 (1.12–1.87) |
| GSE31210 | Lung cancer | Relapse Free Survival | 3.00E-05 | 0.623234 | 1.86 (1.39–2.50) |
| GSE31210 | Lung cancer | Overall Survival | 0.00404387 | 0.597241 | 1.82 (1.21–2.73) |
| jacob-00182-UM | Lung cancer | Overall Survival | 0.158649 | 0.165838 | 1.18 (0.94–1.49) |
| jacob-00182-UM | Lung cancer | Overall Survival | 0.313668 | 0.0915618 | 1.10 (0.92–1.31) |
| GSE3141 | Lung cancer | Overall Survival | 0.251246 | 0.222022 | 1.25 (0.85–1.82) |
| GSE3141 | Lung cancer | Overall Survival | 0.439787 | 0.186023 | 1.20 (0.75–1.93) |
| GSE14814 | Lung cancer | Overall Survival | 0.347644 | 0.275749 | 1.32 (0.74–2.34) |
| GSE14814 | Lung cancer | Disease Specific Survival | 0.213061 | 0.40874 | 1.50 (0.79–2.86) |
| GSE14814 | Lung cancer | Disease Specific Survival | 0.163201 | 0.63031 | 1.88 (0.77–4.56) |
| GSE14814 | Lung cancer | Overall Survival | 0.339429 | 0.394737 | 1.48 (0.66–3.34) |
| GSE8894 | Lung cancer | Relapse Free Survival | 0.194985 | 0.138056 | 1.15 (0.93–1.41) |
| GSE8894 | Lung cancer | Relapse Free Survival | 0.0887164 | 0.16656 | 1.18 (0.98–1.43) |
| GSE4573 | Lung cancer | Overall Survival | 0.102369 | 0.458103 | 1.58 (0.91–2.74) |
| GSE4573 | Lung cancer | Overall Survival | 0.238414 | 0.356487 | 1.43 (0.79–2.58) |
| GSE17710 | Lung cancer | Relapse Free Survival | 0.257306 | 0.342858 | 1.41 (0.78–2.55) |
| GSE17710 | Lung cancer | Relapse Free Survival | 0.203291 | 0.389413 | 1.48 (0.81–2.69) |
| GSE17710 | Lung cancer | Overall Survival | 0.168822 | 0.435278 | 1.55 (0.83–2.87) |
| GSE17710 | Lung cancer | Overall Survival | 0.127973 | 0.489001 | 1.63 (0.87–3.06) |
| GSE9891 | Ovarian cancer | Overall Survival | 0.0458145 | 0.171656 | 1.19 (1.00–1.41) |
| DUKE-OC | Ovarian cancer | Overall Survival | 0.190803 | −0.132614 | 0.88 (0.72–1.07) |
| DUKE-OC | Ovarian cancer | Overall Survival | 0.774339 | −0.0393488 | 0.96 (0.73–1.26) |
| GSE26712 | Ovarian cancer | Overall Survival | 0.104731 | −0.203939 | 0.82 (0.64–1.04) |
| GSE26712 | Ovarian cancer | Overall Survival | 0.493253 | −0.135951 | 0.87 (0.59–1.29) |
| GSE26712 | Ovarian cancer | Disease Free Survival | 0.817429 | −0.0412868 | 0.96 (0.68–1.36) |
| GSE26712 | Ovarian cancer | Disease Free Survival | 0.112295 | −0.181627 | 0.83 (0.67–1.04) |
| GSE17260 | Ovarian cancer | Overall Survival | 0.374023 | 0.125722 | 1.13 (0.86–1.50) |
| GSE17260 | Ovarian cancer | Progression Free Survival | 0.249324 | 0.123087 | 1.13 (0.92–1.39) |
| GSE14764 | Ovarian cancer | Overall Survival | 0.20335 | 0.297358 | 1.35 (0.85–2.13) |
| GSE14764 | Ovarian cancer | Overall Survival | 0.330025 | 0.268734 | 1.31 (0.76–2.25) |
| GSE19234 | Skin cancer | Overall Survival | 0.00485285 | 1.28566 | 3.62 (1.48–8.85) |
| GSE30929 | Soft tissue cancer | Distant Recurrence Free Survival | 0.000134506 | 0.466765 | 1.59 (1.26–2.03) |
Molecular role and functional enrichment analysis results of NCAPG
We used the GeneMANIA database for the protein-molecular interaction analysis of NCAPG and its related molecules, such as NCAPG2, NCAPH, and SMC4 (Figure 7A). Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) functional and pathway enrichment analyses of NCAPG were performed using the STRING database. The most abundant GO terms were nuclear division, cell division, and cell cycle process (Table 4). In addition, the KEGG pathway analysis confirmed that these co-expressed genes were significantly involved in the p53 signaling pathway, cell cycle, and cellular senescence (Table 4). These results indicate that NCAPG is involved in the biological pathways of cancer. Additionally, we used the multiMiR package to identify NCAPG-related miRNAs and lncRNAs (the screening criterion for miRNAs was a predicted cutoff of 500,000, and the screening criteria for lncRNAs were lnc_mi$pancancerNum>10 and lnc_mi$clipExpNum>4) that may interact with NCAPG (Figure 7B). We identified 20 miRNAs and 13 lncRNAs, which could provide direction for future experimental designs. DNA methylation directly affects the occurrence and progression of cancers. We used the UALCAN database to investigate the DNA methylation of NCAPG. Our results showed that NCAPG methylation levels were significantly reduced in BRCA, GBM, LIHC, LUAD, and LUSC tissues compared to normal tissues (Figure 8A–8F), which may explain the difference in NCAPG expression between BRCA, GBM, LIHC, LUAD, and LUSC tissues and normal tissues. Post-translational modification is a key molecular mechanism underlying NCAPG activation. Therefore, we examined the changes in NCAPG phosphorylation levels between tumor tissues and normal tissues. The Clinical Proteomic Tumor Analysis Consortium database includes four cancers: BRCA, GBM, LIHC, and LUAD. Compared with the normal samples, the phosphorylation levels at 674/973/975/984 of NCAPG were higher in BRCA, GBM, LIHC, and LUAD, respectively (Figure 8G). The specific results are shown in Figure 8H–8M. Since the p53 signaling pathway is highly enriched, we also explored the phosphorylation of NCAPG in this pathway, and the results are shown in Figure 8N–8R.
Table 4. Functional enrichment analysis and pathway enrichment analysis of NCAPG genes.
| GO ID | Term description | Ontology | Count | False discovery rate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000280 | Nuclear division | BP | 11 | 1.26E-16 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0140014 | Mitotic nuclear division | BP | 10 | 1.26E-16 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0030261 | Chromosome condensation | BP | 8 | 5.29E-16 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0051301 | Cell division | BP | 11 | 7.60E-15 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000070 | Mitotic sister chromatid segregation | BP | 8 | 4.14E-13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0007076 | Mitotic chromosome condensation | BP | 6 | 8.07E-13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0022402 | Cell cycle process | BP | 11 | 7.24E-12 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0010032 | Meiotic chromosome condensation | BP | 5 | 1.78E-11 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:1903046 | Meiotic cell cycle process | BP | 6 | 1.29E-07 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0051276 | Chromosome organization | BP | 9 | 1.48E-07 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0007077 | Mitotic nuclear envelope disassembly | BP | 3 | 3.34E-05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0022414 | Reproductive process | BP | 8 | 5.10E-05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0051304 | Chromosome separation | BP | 3 | 0.00033 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0140013 | Meiotic nuclear division | BP | 4 | 0.00049 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:1905448 | Positive regulation of mitochondrial atp synthesis coupled electron transport | BP | 2 | 0.0023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0051987 | Positive regulation of attachment of spindle microtubules to kinetochore | BP | 2 | 0.0029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0031145 | Anaphase-promoting complex-dependent catabolic process | BP | 3 | 0.0046 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0045132 | Meiotic chromosome segregation | BP | 3 | 0.0051 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0035404 | Histone-serine phosphorylation | BP | 2 | 0.0082 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0055015 | Ventricular cardiac muscle cell development | BP | 2 | 0.0082 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0034501 | Protein localization to kinetochore | BP | 2 | 0.01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0051782 | Negative regulation of cell division | BP | 2 | 0.0139 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000086 | G2/M transition of mitotic cell cycle | BP | 3 | 0.0145 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0007292 | Female gamete generation | BP | 3 | 0.0145 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0051383 | Kinetochore organization | BP | 2 | 0.0145 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0007051 | Spindle organization | BP | 3 | 0.0162 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0007093 | Mitotic cell cycle checkpoint | BP | 3 | 0.0197 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0045931 | Positive regulation of mitotic cell cycle | BP | 3 | 0.0197 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0060045 | Positive regulation of cardiac muscle cell proliferation | BP | 2 | 0.0247 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000226 | Microtubule cytoskeleton organization | BP | 4 | 0.0261 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0010971 | Positive regulation of g2/m transition of mitotic cell cycle | BP | 2 | 0.0261 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0018105 | Peptidyl-serine phosphorylation | BP | 3 | 0.0261 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0010389 | Regulation of g2/m transition of mitotic cell cycle | BP | 3 | 0.0338 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:1901991 | Negative regulation of mitotic cell cycle phase transition | BP | 3 | 0.0412 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000796 | Condensin complex | CC | 6 | 3.01E-14 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000793 | Condensed chromosome | CC | 8 | 3.53E-11 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000799 | Nuclear condensin complex | CC | 4 | 2.14E-09 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000794 | Condensed nuclear chromosome | CC | 6 | 4.97E-09 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000797 | Condensin core heterodimer | CC | 3 | 7.47E-07 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000779 | Condensed chromosome, centromeric region | CC | 5 | 1.06E-06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0098687 | Chromosomal region | CC | 6 | 1.80E-06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0005694 | Chromosome | CC | 9 | 2.71E-06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000228 | Nuclear chromosome | CC | 8 | 6.95E-06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0043232 | Intracellular non-membrane-bounded organelle | CC | 11 | 3.32E-05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000307 | Cyclin-dependent protein kinase holoenzyme complex | CC | 3 | 0.00021 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0097125 | Cyclin b1-cdk1 complex | CC | 2 | 0.00021 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0005813 | Centrosome | CC | 5 | 0.00087 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0032991 | Protein-containing complex | CC | 10 | 0.0011 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0005634 | Nucleus | CC | 11 | 0.0019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0005819 | Spindle | CC | 4 | 0.0026 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0072686 | Mitotic spindle | CC | 3 | 0.0029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000922 | Spindle pole | CC | 3 | 0.0064 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0030496 | Midbody | CC | 3 | 0.0083 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0000780 | Condensed nuclear chromosome, centromeric region | CC | 2 | 0.0092 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0005829 | Cytosol | CC | 9 | 0.0132 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0005654 | Nucleoplasm | CC | 8 | 0.0161 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0005876 | Spindle microtubule | CC | 2 | 0.018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GO:0035173 | Histone kinase activity | MF | 3 | 0.00041 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KEGG ID | Term description | Count | False discovery rate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsa04115 | p53 signaling pathway | 3 | 0.0029 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsa04914 | Progesterone-mediated oocyte maturation | 3 | 0.0032 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsa04110 | Cell cycle | 3 | 0.0043 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsa04114 | Oocyte meiosis | 3 | 0.0043 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsa04218 | Cellular senescence | 3 | 0.005 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| hsa05170 | Human immunodeficiency virus 1 infection | 3 | 0.0101 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: BP: Biological Process; CC: Cellular Component; ME: Molecular Function; KEGG: Kyoto Encyclopedia of Genes and Genomes. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
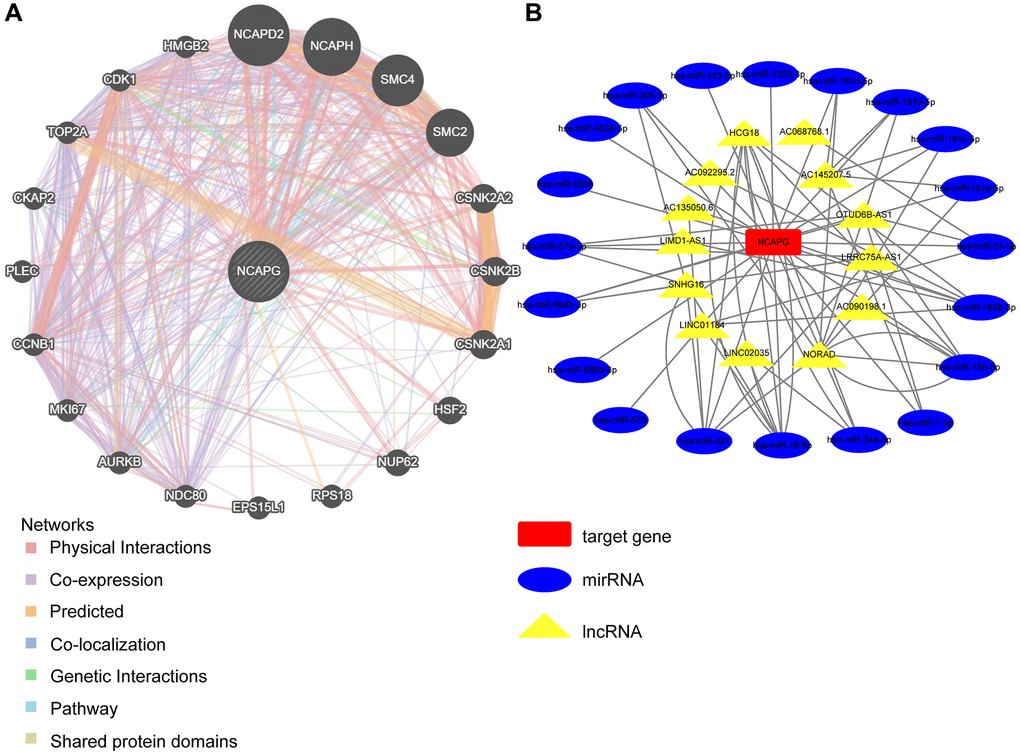
Figure 7. Network analysis between NCAPG and target genes (A) PPI network for KIF23 was constructed in Gene MANIA, Different colors of the network edge indicate the bioinformatics methods applied: physical interaction, co-expression, predicted, co-localization, pathway, genetic interaction and shared protein domains. Abbreviation: PPI: protein–protein interaction. (B) The relationship between NCAPG and non-coding RNA, the red square represents the target gene NCAPG, the blue oval represents miRNA, and the yellow triangle represents lncRNA.
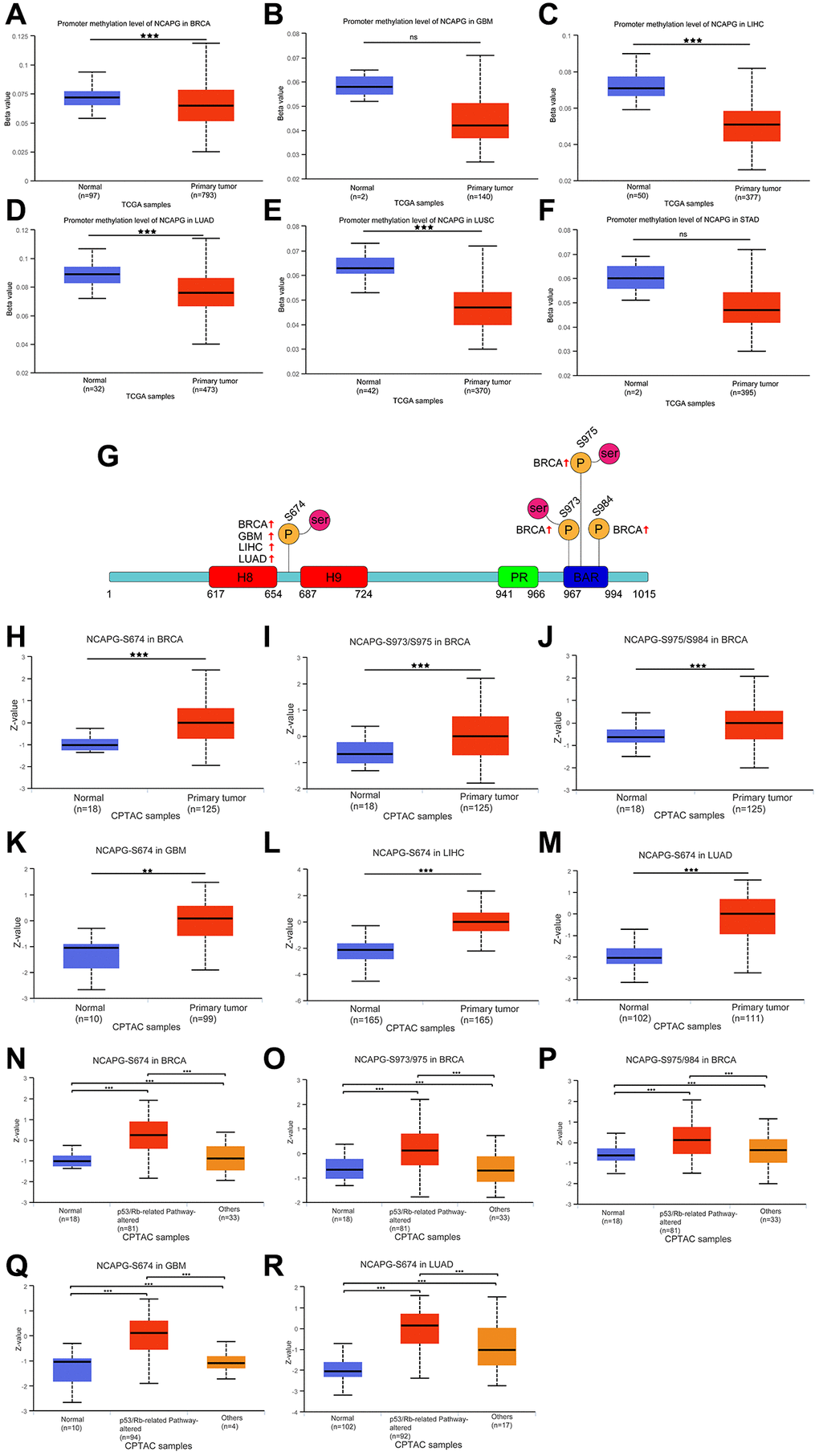
Figure 8. DNA methylation features of NCAPG in BRCA (A), GBM (B), LIHC (C), LUAD (D), LUSC (E) and STAD (F). Phosphorylation of NCAPG in several selected cancers according to the CPTAC database. (G) The schematic diagram and phosphorylation sites of the NCAPG protein are shown. The phosphorylation of NCAPG at S674, S973, S975 and 984 in BRCA (H–J), S674 in GBM (K), S674 in LIHC (L), S674 in LUAD (M). The P53 pathway phosphorylation of NCAPG at S674, S973, S975 and 984 in BRCA (N–P), S674 in GBM (Q), S674 in LUAD (R), from the UALCAN database. *p < 0:05, **p < 0:01, and ***p < 0:001, Abbreviation: ns: No statistical significance.
Discussion
NCAPG expression was initially found to be correlated with the prognosis of liver cancer. Later on, the expression of NCAPG was often closely associated with the survival outcome and clinical pathology of patients with diseases such as NSCLC, renal clear cell carcinoma, breast cancer, and gastric cancer [27]. To better verify and summarize the value of this gene and avoid the errors caused by small samples or small queues, we used meta-methods and bioinformatics jointly.
First, in this study, the results of the meta-analysis showed that high expression of NCAPG is associated with poor prognosis, suggesting its role as a proto-oncogene in cancer. Second, eight studies were included in this meta-analysis. Our results suggest that cancer patients with upregulated NCAPG expression have a 2.90-fold worse OS than those with low expression. We also performed a subgroup analysis according to the different systems. The results showed that NCAPG might be a potential prognostic marker for cancers of the respiratory, digestive, and other systems. Third, from the perspective of bioinformatics, univariate cox regression analysis showed that NCAPG was a bad prognostic factor for LIHC, LUAD, and STAD. We also verified this inference using the Kaplan-Meier plotter database and found that the high expression of NCAPG was related to the poor prognosis of BRCA, LIHC, LUAD, and STAD. We calculated the relationship between the expression of NCAPG and the annual survival rate of the cancer (Supplementary Figure 3). The results showed that NCAPG could predict the survival and prognosis of GBM, LIHC, and STAD.
Additionally, we assessed the association between NCAPG expression and clinicopathological parameters. The pooled results showed that the upregulation of NCAPG was not associated with age, sex, vascular invasion, differentiation, TNM stage, and T classification but was associated with distant metastasis, lymph node metastasis, relapse, and clinical stage. The results of the sensitivity and publication bias analyses demonstrated the reliability of the results. To our knowledge, this is the first meta-analysis to demonstrate the prognostic value of NCAPG in cancer.
To further explore the relationship between NCAPG expression and the clinicopathological features of different cancers, a subgroup analysis was performed. We found that NCAPG overexpression was significantly correlated with positive lymph node metastasis in gastric cancer and NSCLC, TNM stage in hepatocellular carcinoma, age in glioma, differentiation in hepatocellular carcinoma and glioma, and vascular invasion in gastric cancer. Next, we verified the clinicopathological features of cancers with NCAPG expression using the UALCAN database (Supplementary Figure 1). The results showed that the expression of NCAPG was related to the age, lymph node metastasis, and stage of tumor patients.
How NCAPG can precisely regulate oncogenes remains unknown, but some studies have proved that it may be related to the following mechanisms. NCAPG induces epigenetic changes of tumors through a variety of signal pathways and molecules. In lung cancer, NCAPG expression activates TGF-β signaling pathway [5]. In breast cancer, it is related to the SRC/STAT3 signaling pathway [6] and p53 signaling pathway [28]. In colorectal cancer, it is associated with Wnt/β-catenin signaling pathway [29]. In hepatocellular carcinoma, it is related to activation of PI3K/AKT signaling pathway [7]. In cardiac adenocarcinoma, Wnt/β-catenin signaling pathway [30] and PI3K/AKT signaling pathway are involved [31]. In endometrial carcinoma, it is related to Wnt/β-catenin signaling pathway [32]. In oral squamous cell carcinoma, it is related to miR-378a-3p-mediated GSK-3β/β-catenin signaling [33]. In prostate cancer, it interacts with miR-99a-3p [34]. In bladder cancer, it is related to NF-κB signaling pathway [35]. In ovarian cancer, it is related to p38 MAPK signaling pathway [36].
We also analyzed the GO and KEGG pathways of NCAPG. The richest GO terms were nuclear division, cell division, and cell cycle processes, all of which are related to cancer cell proliferation. In addition, KEGG pathway analysis confirmed that NCAPG-related genes were involved in the p53 signaling pathway, cell senescence, and cell cycle, which are involved in the mechanism underlying carcinogenesis. We also explored the changes in phosphorylation and methylation of NCAPG in these tumors. Finally, we identified the miRNAs and lncRNAs related to NCAPG.
This study has a few limitations. First, a total of eight studies were included in this study, which is a relatively small sample size and may affect the accuracy of the results; therefore, additional research is needed to confirm the findings. Second, it is necessary to fully verify and clarify the role and mechanism of NCAPG in cancer through cell models in vitro and in vivo. Furthermore, some studies used the K-M curve to extract the HR of OS, which may have had an impact on the results and led to publication bias. Finally, due to the different methods used to evaluate NCAPG expression and different cutoff value standards, statistical errors may have been introduced.
Conclusion
In conclusion, our study is the first to systematically address the prognostic and clinical significance of the NCAPG gene in cancer. We provide meta-analytical and bioinformatic evidence that NCAPG acts as an oncogenic mRNA with great potential as a biological prognostic marker for cancer. However, this study had certain limitations, and more basic experiments are needed to verify these conclusions.
Supplementary Materials
Author Contributions
Yingjun Xie (XYJ): Conceptualization and Methodology, Administrative support: Jie Lin (LJ): Provide learning materials or patients, Writing-Original draft preparation. Gangyi Li (LGY): Data Collection and Analysis, Writing-Original draft preparation. Yanping Bai (BYP): Data analysis and interpretation.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
This study was registered in PROSPERO (registration number CRD42022333964).
Funding
Special Medical and Health Personnel Program of Jilin Province Finance Department (2019SCZT015), Health Technology Innovative Program of Jilin Province (2018J053).
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 2. Zugazagoitia J, Guedes C, Ponce S, Ferrer I, Molina-Pinelo S, Paz-Ares L. Current Challenges in Cancer Treatment. Clin Ther. 2016; 38:1551–66. https://doi.org/10.1016/j.clinthera.2016.03.026 [PubMed]
- 3. Murphy LA, Sarge KD. Phosphorylation of CAP-G is required for its chromosomal DNA localization during mitosis. Biochem Biophys Res Commun. 2008; 377:1007–11. https://doi.org/10.1016/j.bbrc.2008.10.114 [PubMed]
- 4. Gu C, Wang Z, Zhou N, Li G, Kou Y, Luo Y, Wang Y, Yang J, Tian F. Mettl14 inhibits bladder TIC self-renewal and bladder tumorigenesis through N6-methyladenosine of Notch1. Mol Cancer. 2019; 18:168. https://doi.org/10.1186/s12943-019-1084-1 [PubMed]
- 5. Wu Y, Lin Y, Pan J, Tu X, Xu Y, Li H, Chen Y. NCAPG promotes the progression of lung adenocarcinoma via the TGF-β signaling pathway. Cancer Cell Int. 2021; 21:443. https://doi.org/10.1186/s12935-021-02138-w [PubMed]
- 6. Jiang L, Ren L, Chen H, Pan J, Zhang Z, Kuang X, Chen X, Bao W, Lin C, Zhou Z, Huang D, Yang J, Huang H, et al. NCAPG confers trastuzumab resistance via activating SRC/STAT3 signaling pathway in HER2-positive breast cancer. Cell Death Dis. 2020; 11:547. https://doi.org/10.1038/s41419-020-02753-x [PubMed]
- 7. Gong C, Ai J, Fan Y, Gao J, Liu W, Feng Q, Liao W, Wu L. NCAPG Promotes The Proliferation Of Hepatocellular Carcinoma Through PI3K/AKT Signaling. Onco Targets Ther. 2019; 12:8537–52. https://doi.org/10.2147/OTT.S217916 [PubMed]
- 8. Sun DP, Lin CC, Hung ST, Kuang YY, Hseu YC, Fang CL, Lin KY. Aberrant Expression of NCAPG is Associated with Prognosis and Progression of Gastric Cancer. Cancer Manag Res. 2020; 12:7837–46. https://doi.org/10.2147/CMAR.S248318 [PubMed]
- 9. Zhou Y, Fan Y, Mao Y, Lou M, Liu X, Yuan K, Tong J. NCAPG is a prognostic biomarker of immune infiltration in non-small-cell lung cancer. Biomark Med. 2022; 16:523–35. https://doi.org/10.2217/bmm-2021-1090 [PubMed]
- 10. Sang L, Wu X, Yan T, Naren D, Liu X, Zheng X, Zhang N, Wang H, Li Y, Gong Y. The m6A RNA methyltransferase METTL3/METTL14 promotes leukemogenesis through the mdm2/p53 pathway in acute myeloid leukemia. J Cancer. 2022; 13:1019–30. https://doi.org/10.7150/jca.60381 [PubMed]
- 11. Liu W, Liang B, Liu H, Huang Y, Yin X, Zhou F, Yu X, Feng Q, Li E, Zou Z, Wu L. Overexpression of non-SMC condensin I complex subunit G serves as a promising prognostic marker and therapeutic target for hepatocellular carcinoma. Int J Mol Med. 2017; 40:731–8. https://doi.org/10.3892/ijmm.2017.3079 [PubMed]
- 12. Zheng G, Han T, Hu X, Yang Z, Wang J, Wen Z, Li H, Wang H. NCAPG Promotes Tumor Progression and Modulates Immune Cell Infiltration in Glioma. Front Oncol. 2022; 12:770628. https://doi.org/10.3389/fonc.2022.770628 [PubMed]
- 13. Liang ML, Hsieh TH, Ng KH, Tsai YN, Tsai CF, Chao ME, Liu DJ, Chu SS, Chen W, Liu YR, Liu RS, Lin SC, Ho DM, et al. Downregulation of miR-137 and miR-6500-3p promotes cell proliferation in pediatric high-grade gliomas. Oncotarget. 2016; 7:19723–37. https://doi.org/10.18632/oncotarget.7736 [PubMed]
- 14. Cohen Y, Gutwein O, Garach-Jehoshua O, Bar-Haim A, Kornberg A. The proliferation arrest of primary tumor cells out-of-niche is associated with widespread downregulation of mitotic and transcriptional genes. Hematology. 2014; 19:286–92. https://doi.org/10.1179/1607845413Y.0000000125 [PubMed]
- 15. Hu SP, Ge MX, Gao L, Jiang M, Hu KW. LncRNA HCP5 as a potential therapeutic target and prognostic biomarker for various cancers: a meta-analysis and bioinformatics analysis. Cancer Cell Int. 2021; 21:686. https://doi.org/10.1186/s12935-021-02404-x [PubMed]
- 16. Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017; 45:W98–102. https://doi.org/10.1093/nar/gkx247 [PubMed]
- 17. Colaprico A, Silva TC, Olsen C, Garofano L, Cava C, Garolini D, Sabedot TS, Malta TM, Pagnotta SM, Castiglioni I, Ceccarelli M, Bontempi G, Noushmehr H. TCGAbiolinks: an R/Bioconductor package for integrative analysis of TCGA data. Nucleic Acids Res. 2016; 44:e71. https://doi.org/10.1093/nar/gkv1507 [PubMed]
- 18. Győrffy B. Survival analysis across the entire transcriptome identifies biomarkers with the highest prognostic power in breast cancer. Comput Struct Biotechnol J. 2021; 19:4101–9. https://doi.org/10.1016/j.csbj.2021.07.014 [PubMed]
- 19. Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi BVS, Varambally S. UALCAN: A Portal for Facilitating Tumor Subgroup Gene Expression and Survival Analyses. Neoplasia. 2017; 19:649–58. https://doi.org/10.1016/j.neo.2017.05.002 [PubMed]
- 20. Franz M, Rodriguez H, Lopes C, Zuberi K, Montojo J, Bader GD, Morris Q. GeneMANIA update 2018. Nucleic Acids Res. 2018; 46:W60–4. https://doi.org/10.1093/nar/gky311 [PubMed]
- 21. Warde-Farley D, Donaldson SL, Comes O, Zuberi K, Badrawi R, Chao P, Franz M, Grouios C, Kazi F, Lopes CT, Maitland A, Mostafavi S, Montojo J, et al. The GeneMANIA prediction server: biological network integration for gene prioritization and predicting gene function. Nucleic Acids Res. 2010; 38:W214–20. https://doi.org/10.1093/nar/gkq537 [PubMed]
- 22. Szklarczyk D, Gable AL, Nastou KC, Lyon D, Kirsch R, Pyysalo S, Doncheva NT, Legeay M, Fang T, Bork P, Jensen LJ, von Mering C. The STRING database in 2021: customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021; 49:D605–12. https://doi.org/10.1093/nar/gkaa1074 [PubMed]
- 23. Szklarczyk D, Morris JH, Cook H, Kuhn M, Wyder S, Simonovic M, Santos A, Doncheva NT, Roth A, Bork P, Jensen LJ, von Mering C. The STRING database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 2017; 45:D362–8. https://doi.org/10.1093/nar/gkw937 [PubMed]
- 24. Wang Y, Gao B, Tan PY, Handoko YA, Sekar K, Deivasigamani A, Seshachalam VP, OuYang HY, Shi M, Xie C, Goh BKP, Ooi LL, Man Hui K. Genome-wide CRISPR knockout screens identify NCAPG as an essential oncogene for hepatocellular carcinoma tumor growth. FASEB J. 2019; 33:8759–70. https://doi.org/10.1096/fj.201802213RR [PubMed]
- 25. Wang X, Tian X, Sui X, Li X, Zhao X, Han K, Sun L, Dong Y. Increased expression of NCAPG (Non-SMC condensing I complex subunit G) is associated with progression and poor prognosis of lung adenocarcinoma. Bioengineered. 2022; 13:6113–25. https://doi.org/10.1080/21655979.2022.2035124 [PubMed]
- 26. Sun H, Zhang H, Yan Y, Li Y, Che G, Zhou C, Nicot C, Ma H. NCAPG promotes the oncogenesis and progression of non-small cell lung cancer cells through upregulating LGALS1 expression. Mol Cancer. 2022; 21:55. https://doi.org/10.1186/s12943-022-01533-9 [PubMed]
- 27. Cai X, Gao J, Shi C, Guo WZ, Guo D, Zhang S. The role of NCAPG in various of tumors. Biomed Pharmacother. 2022; 155:113635. https://doi.org/10.1016/j.biopha.2022.113635 [PubMed]
- 28. Dong M, Xu T, Cui X, Li H, Li X, Xia W. NCAPG upregulation mediated by four microRNAs combined with activation of the p53 signaling pathway is a predictor of poor prognosis in patients with breast cancer. Oncol Lett. 2021; 21:323. https://doi.org/10.3892/ol.2021.12585 [PubMed]
- 29. Shi Y, Ge C, Fang D, Wei W, Li L, Wei Q, Yu H. NCAPG facilitates colorectal cancer cell proliferation, migration, invasion and epithelial-mesenchymal transition by activating the Wnt/β-catenin signaling pathway. Cancer Cell Int. 2022; 22:119. https://doi.org/10.1186/s12935-022-02538-6 [PubMed]
- 30. Zhang X, Zhu M, Wang H, Song Z, Zhan D, Cao W, Han Y, Jia J. Overexpression of NCAPG inhibits cardia adenocarcinoma apoptosis and promotes epithelial-mesenchymal transition through the Wnt/β-catenin signaling pathway. Gene. 2021; 766:145163. https://doi.org/10.1016/j.gene.2020.145163 [PubMed]
- 31. Zhang X, Wang H, Han Y, Zhu M, Song Z, Zhan D, Jia J. NCAPG Induces Cell Proliferation in Cardia Adenocarcinoma via PI3K/AKT Signaling Pathway. Onco Targets Ther. 2020; 13:11315–26. https://doi.org/10.2147/OTT.S276868 [PubMed]
- 32. Liu C, Yan Y, Di F, Li W, Yin X, Dong L. Inhibition of NCAPG expression inactivates the Wnt/β-catenin signal to suppresses endometrial cancer cell growth in vitro. Environ Toxicol. 2021; 36:2512–20. https://doi.org/10.1002/tox.23364 [PubMed]
- 33. Li J, Sun S, Li J, Zhao X, Li Z, Sha T, Cui Z. NCAPG, mediated by miR-378a-3p, regulates cell proliferation, cell cycle progression, and apoptosis of oral squamous cell carcinoma through the GSK-3β/β-catenin signaling. Neoplasma. 2021; 68:1201–11. https://doi.org/10.4149/neo_2021_210421N544 [PubMed]
- 34. Arai T, Okato A, Yamada Y, Sugawara S, Kurozumi A, Kojima S, Yamazaki K, Naya Y, Ichikawa T, Seki N. Regulation of NCAPG by miR-99a-3p (passenger strand) inhibits cancer cell aggressiveness and is involved in CRPC. Cancer Med. 2018; 7:1988–2002. https://doi.org/10.1002/cam4.1455 [PubMed]
- 35. Tang F, Yu H, Wang X, Shi J, Chen Z, Wang H, Wan Z, Fu Q, Hu X, Zuhaer Y, Liu T, Yang Z, Peng J. NCAPG promotes tumorigenesis of bladder cancer through NF-κB signaling pathway. Biochem Biophys Res Commun. 2022; 622:101–7. https://doi.org/10.1016/j.bbrc.2022.07.007 [PubMed]
- 36. Yu H, Zou D, Ni N, Zhang S, Zhang Q, Yang L. Overexpression of NCAPG in ovarian cancer is associated with ovarian cancer proliferation and apoptosis via p38 MAPK signaling pathway. J Ovarian Res. 2022; 15:98. https://doi.org/10.1186/s13048-022-01030-z [PubMed]



