Introduction
Extranodal NK/T-cell lymphoma, nasal type (NKTCL) is a subtype of mature NK or T cell lymphoma characterized by extranodal involvement and association with Epstein-Barr virus [1–3]. It has a geographical predilection for Asian and Latin American populations [4, 5]. Majority (70–80%) of cases present localized disease predominately occurring in the upper aerodigestive tract (UADT), especially nasal cavity and Waldeyer’s ring [1, 6]. Tumor cells exhibit a cytotoxic phenotype, primarily characterized by the expression of perforin and granzyme B [1].
NKTCL is unique among common aggressive lymphomas in terms of treatment principles. It is highly sensitive to radiation (RT) but resistant to chemotherapy (CT), especially anthracycline (ANT)-based regimens [7–9]. RT is the backbone for the treatment of localized NKTCL and is the most effective modality to make rapid locoregional disease control (LRC) [8, 10–12]. RT alone achieved excellent complete remission (CR) rate of 83–100% and satisfactory 5-year overall survival (OS) rate of 83.3–88.8% for stage I diseases [8, 13, 14]. Besides, the addition of CT to RT brings further survival benefits, especially among high-risk cases [7, 14, 15]. Currently, the combined chemoradiation (CRT), arranged sequentially or concurrently, is regarded as a standard treatment modality for early stage NKTCL.
In clinical practice, first-line sequential induction CT followed by RT is widely used in treating early stage NKTCL. On one hand, the sequential modality achieves similar responses and survivals as concurrent CRT [16–24]. On the other, it is much easier to arrange without waiting for RT planning, and is also more tolerable since it avoids intensifying mucositis and overlapping of other toxicities [25]. In the modality of sequential CT and RT, though non-ANT-based regimens yielded satisfactory ORR of 81–92% after induction CT, the remission was not deep with only half or less cases reaching CR [17, 26–29]. Fortunately, RT still produced excellent LRC among those non-CRs, and dramatically improved the CR rate to 70–100% at the completion of RT [26, 29–31]. Therefore, in the setting of effective RT, is it still necessary to achieve early deep remission from induction CT? Up to now, however, the association between induction CT response and survival of localized NKTCL has not been fully explored.
Optimization of risk stratification is vital for clinical decision guidance in localized NKTCL since 10–37% of cases still have refractory disease or progression after first-line CRT [32]. Li and his colleagues reviewed 214 patients with early stage NKTCL, and results revealed that systemic dissemination was the main failure pattern [32]. The 5-year cumulative incidences of systemic failure (SF) were similar between patients receiving RT alone and CRT (28.5% vs. 24.1%, P = 0.758) [32]. Therefore, it is inferred that more effective systemic therapy is one of the key elements for further cure of this disease. At present, an early recognition and selection of vulnerable patients for intensive therapy are warranted. Apart from well-established pretreatment prognostic factors [33–37], treatment-related variables reflecting tumor reaction to therapy, such as post-treatment EBV-DNA positivity, positron emission tomography (PET) scan after primary treatment, and inadequate dose of RT were reported to predict early relapse and poor prognosis for localized NKTCL [12, 38, 39]. Here, we suppose that the induction CT response, which reflects tumor’s sensitivity to systemic therapy, might also be a potential prognostic factor for long-term survivals and might be used in individualized treatment for localized NKTCL.
Therefore, among stage I(E)/II(E) NKTCL patients who received first-line sequential CT and RT, we conducted this retrospective study to assess the association between induction CT response and long-term survivals; we also analyzed the failure patterns and further explored its potential role in guiding treatment strategy for NKTCL.
Results
Patient population description
Patients who progressed rapidly during or after induction CT with no chance for subsequent RT were excluded. Finally, a total of 203 consecutive patients who were newly-diagnosed localized NKTCL and underwent first-line CT and RT from 2010 to 2020 from Peking University Cancer Hospital were retrospectively reviewed as the primary cohort; another independent group of 67 patients from Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College were analyzed as the validation cohort. Baseline patient characteristics were summarized in Table 1. For patients in the primary cohort, the median age was 42 (range, 15–83) years, and the male-to-female ratio was 2.12:1. All patients had good performance status score of 0–1. Stage I disease accounted for 45.8%, and stage II accounted for 54.2% of patients. Elevated lactate dehydrogenase (LDH) was presented in 24.1% of cases, PTI in 53.7% of cases, and B symptoms in 38.9% of cases. Patients in the primary cohort presented a high-risk population with intermediate- and high-risk patients (NRI > 1) accounted for 76.4% according to NRI stratification. Baseline characteristics were well balanced between the validation and primary cohort except for the serum LDH, which was higher among the validation cohort of patients (40.3% vs. 24.1%, P = 0.018).
Table 1. Patient baseline characteristics.
| Characteristics | Primary cohort | Validation cohort | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender | 1.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 138 (68.0) | 46 (68.7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 65 (32.0) | 21 (31.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 0.646 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤60 | 183 (90.1) | 59 (88.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| >60 | 20 (9.9) | 8 (11.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ECOG score | 0.248 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0–1 | 203 (100.0) | 66 (98.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≥2 | 0 (0) | 1 (1.5) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Primary site | 0.617 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nasal cavity | 158 (77.8) | 50 (74.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Extra-nasal | 45 (22.2) | 17 (25.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage | 1.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I (E) | 93 (45.8) | 31 (46.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| II (E) | 110 (54.2) | 36 (53.7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B symptom | 0.069 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Absent | 130 (59.1) | 30 (44.8) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Present | 90 (40.9) | 37 (55.2) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Primary tumor invasion | 0.779 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No | 94 (46.3) | 33 (49.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 109 (53.7) | 34 (50.7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Serum LDH | 0.018 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Normal | 154 (75.9) | 40 (59.7) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Elevated | 49 (24.1) | 27 (40.3) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IPI score | 1.000 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low risk | 187 (92.1) | 60 (89.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intermediate-low risk | 14 (6.9) | 7 (10.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intermediate-high risk | 1 (0.5) | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High risk | 1 (0.5) | 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PINK | 0.254 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | 184 (90.6) | 57 (85.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1 | 19 (9.4) | 10 (14.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NRI | 0.154 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low risk | 48 (23.6) | 8 (11.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intermediate-low risk | 52 (25.6) | 17 (25.4) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Intermediate-high risk | 69 (34.0) | 30 (44.8) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| High risk | 34 (16.7) | 12 (17.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: ECOG: eastern cooperative oncology group performance status; LDH: lactate dehydrogenase; IPI: International Prognostic Index; PINK: Prognostic index of natural killer lymphoma; NRI: nomogram-revised risk index. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Treatment responses
Treatment response was shown in Figure 1. A total of 134 patients (66.0%) used PET scanning for induction response assessment. The initial CR rate after induction CT was 53.7%. All but one patient (99.1%) with initial CR maintained final CR after RT. Subsequent RT further deepened the remission: 69 out of 81 (85.2%) cases with initial partial remission (PR), and 5 out of 13 (38.5%) initial non-responders achieved final CR after RT. In conclusion, compared with initial non-CRs, patients who were highly sensitive to induction CT had higher final CR rate after subsequent RT (99.1% vs. 78.7%, P < 0.001). That was the same in the validation cohort: the final CR rate was 96.3% for patients with initial CR versus 77.8% for patients with initial non-CR (P < 0.001).
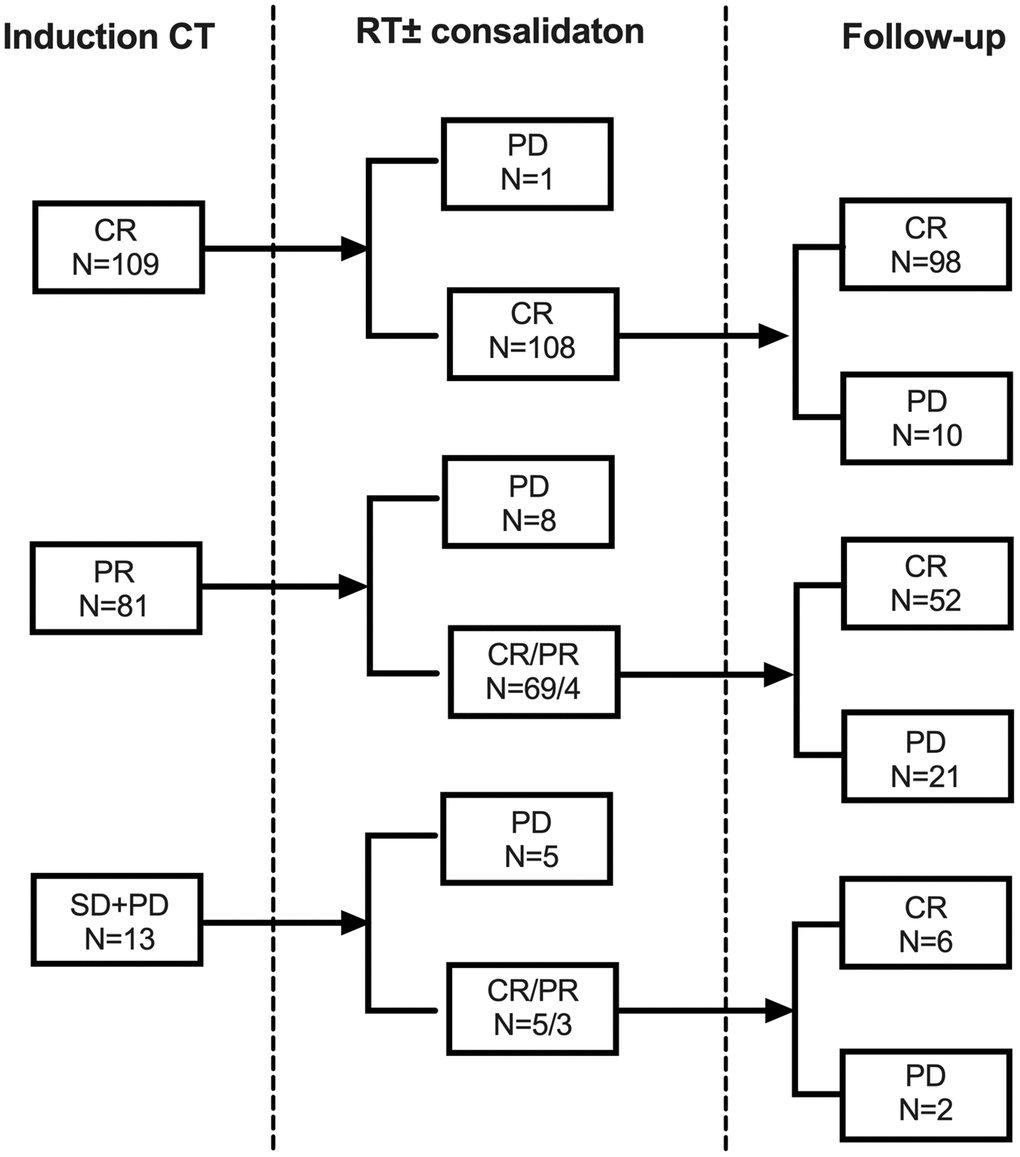
Figure 1. Responses from induction chemotherapy and subsequent radiotherapy for early stage NKTCL in the primary cohort. Abbreviations: CT: chemotherapy; RT: radiotherapy; CR: complete remission; PR: partial remission; SD: stable disease; PD: progression of disease.
Induction CT responses predicted survivals in localized NKTCL receiving CRT
Till the last visit in February 2022, the median follow-up period was 59.2 months for surviving patients in the primary cohort and 62.7 months in the validation cohort. The 5-year progression-free survival (PFS) and OS were 74.6% and 81.5%, respectively for the primary cohort, and 70.2% and 78.2%, respectively for the validation cohort (Figure 2). Survival outcomes were comparable for patients receiving different regimens in the primary cohort and the validation cohort, respectively.
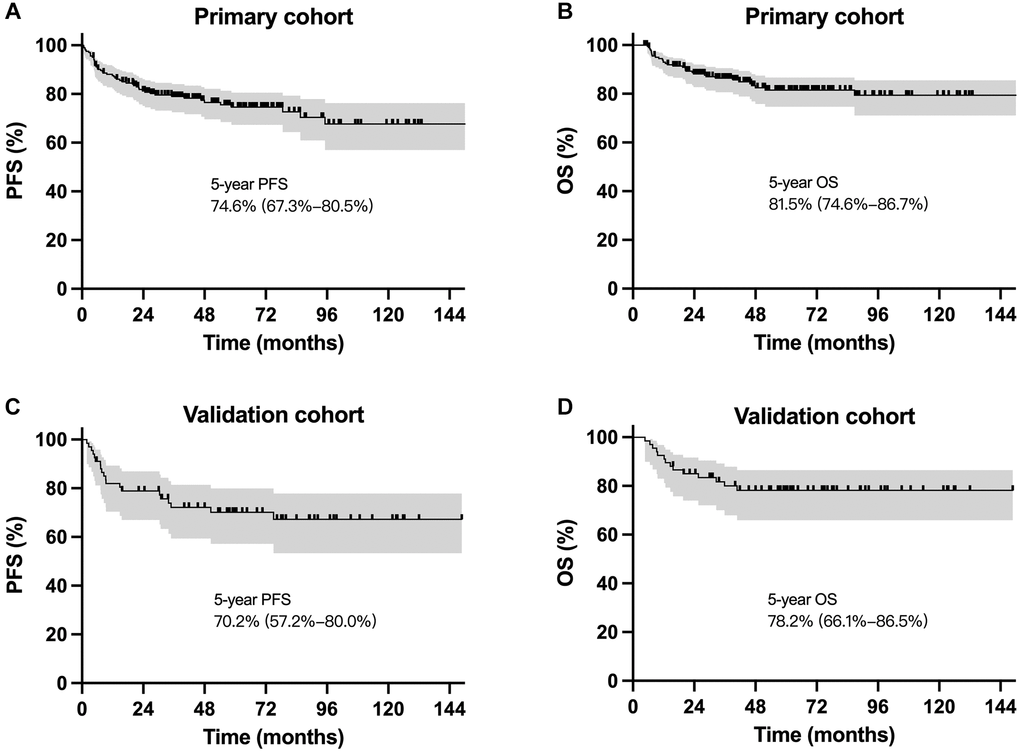
Figure 2. Survival curves for patients with localized NKTCL receiving sequential chemoradiotherapy. Progression-free survival and overall survival cures for the primary cohort (A and B) and the validation cohort (C and D).
For patients with initial CR after induction CT in the primary cohort, the 5-year PFS and OS rates were 90.0% and 92.2%, respectively, much superior to those of initial PRs (61.4% and 70.7%, P < 0.001; Figure 3A and 3B) or non-responders (30.8% and 60.6%, P < 0.001; Figure 3A and 3B). Furthermore, among patients who had final CR after subsequent RT, those with initial CR still had superior 5-year PFS compared with initial non-CRs (90.9% vs. 69.7%, P = 0.002; Figure 3E); though the difference did not reach statistical significance, initial CR did present a tendency of prolonged OS over initial non-CRs (92.1% vs. 81.9%, P = 0.063; Figure 3F). Similar results were observed among patients in the validation cohort (Figure 3C, 3D, 3G and 3H).
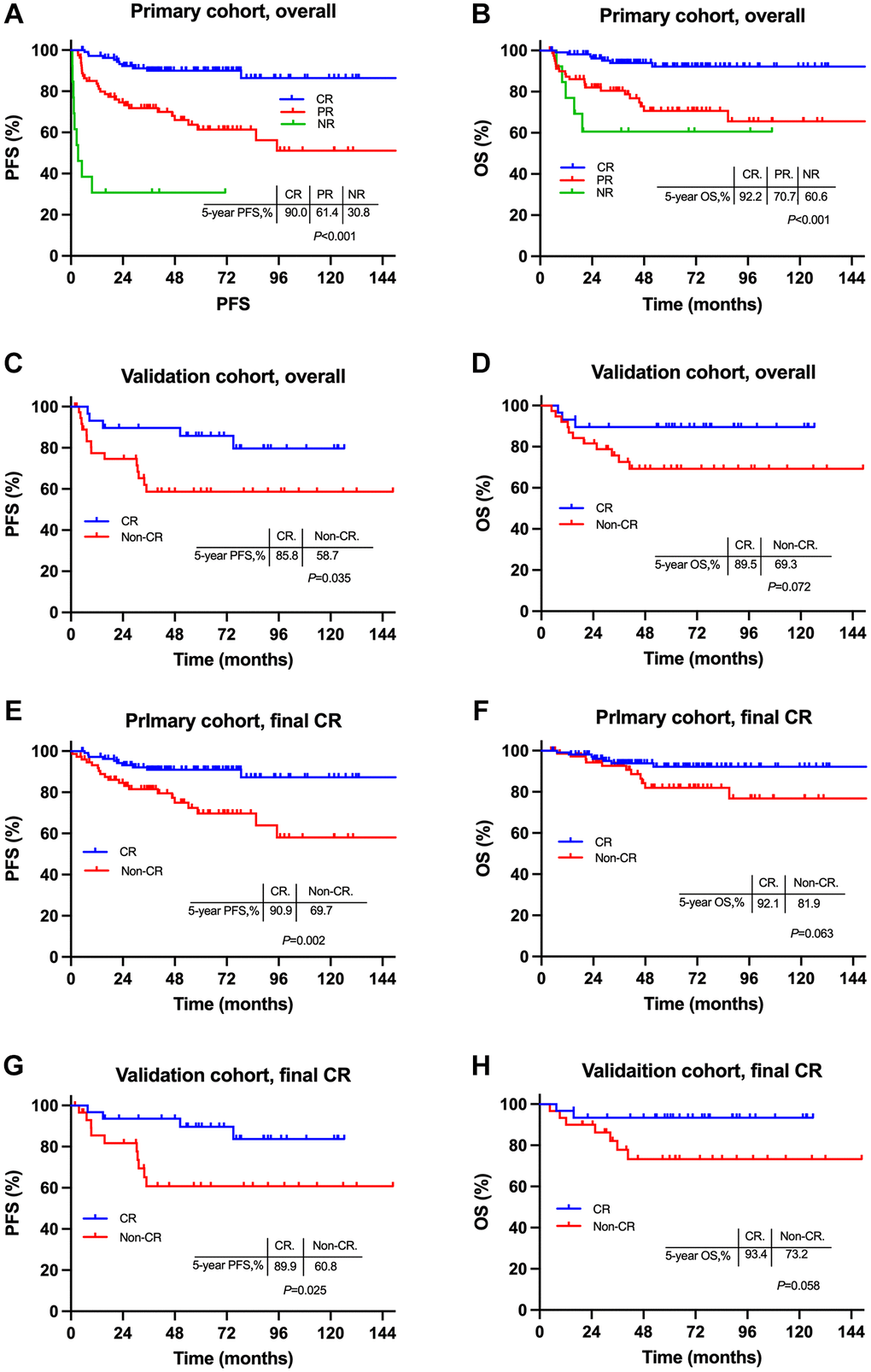
Figure 3. Comparisons of survivals between early stage NKTCL patients with different induction chemotherapy responses. Survival cures for entire patients in the primary cohort (A and B) and the validation cohort (C and D); survival cures for patients with final CR in the primary cohort (E and F) and the validation cohort (G and H). Abbreviations: PFS: progression-free survival; OS: overall survival; CR: complete remission; PR: partial remission; NR: non-response.
Multivariate analysis demonstrated induction CT response as an independent predictor for PFS and OS (Table 2). It was no doubt that patients with initial non-responders (vs. initial CR) to induction CT had higher risk of developing relapse (HR = 19.90, 95% CI, 7.338–54.46; P < 0.001) and death (HR = 4.256, 95% CI, 1.239–14.62; P = 0.021). Noteworthy, those with initial PR also had significantly higher relapse hazard (HR = 3.934, 95% CI, 1.928–8.025; P < 0.001) and death hazard (HR = 4.801, 95% CI, 1.960–11.76, P = 0.001) in comparison to initial CRs.
Table 2. Multivariate analysis for survivals for patients in the primary cohort.
| Characteristics | PFS | OS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR (95% CI) | P-value | HR (95% CI) | P-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, >60 y vs. ≤60 y | 2.069 (0.847–5.057) | 0.111 | 3.750 (1.449–9.705) | 0.006 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Primary site, extra-nasal vs. nasal | 1.171 (0.601–2.282) | 0.643 | 1.172 (0.603–2.279) | 0.639 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage, II (E) vs. I (E) | 3.141 (1.512–6.528) | 0.002 | 3.053(1.472–6.335) | 0.003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B symptom, yes vs. no | 1.035 (0.554–1.933) | 0.913 | 1.182 (0.648–2.154) | 0.586 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDH, elevated vs. normal | 1.703 (0.909–3.191) | 0.096 | 1.186 (0.550–2.555) | 0.664 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PTI, yes vs. no | 1.228 (0.648–2.326) | 0.529 | 2.214 (0.977–5.017) | 0.057 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Induction response | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PR vs. CR | 3.934 (1.928–8.025) | <0.001 | 4.801 (1.960–11.76) | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-response vs. CR | 19.90 (7.338–54.46) | <0.001 | 4.256 (1.239–14.62) | 0.021 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: CR: complete remission; PR: partial remission; LDH: lactate dehydrogenase; PTI: primary tumor invasion; PFS: progression-free survival; OS: overall survival; HR: hazard ratio; 95% CI: 95% confidence interval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
SF was the main failure pattern for localized NKTCL
Till the last follow up, a total of 49 (24.1%) patients failed the treatment in the primary cohort including 24 (11.8%) cases with locoregional failure (LRF) and 33 (16.3%) cases with SF. Failure patterns were summarized in Figure 4A. Most failures occurred within the first two years after diagnosis. The 5-year cumulative incidences of overall failure (OF), LRF and SF were 25.4%, 10.3% and 17.9%, respectively (Figure 4B). Patients with initial CR had much lower OF, LRF and SF rates of 10.0%, 3.1% and 7.0%, respectively (Figure 4C). And the corresponding rates were 43.3%, 19.4%, and 30.9%, respectively, for patients with initial non-CR (Figure 4D). SF was the main failure pattern for localized NKTCL regardless of induction CT response, indicating the necessity of more intensive and effective systemic therapy.
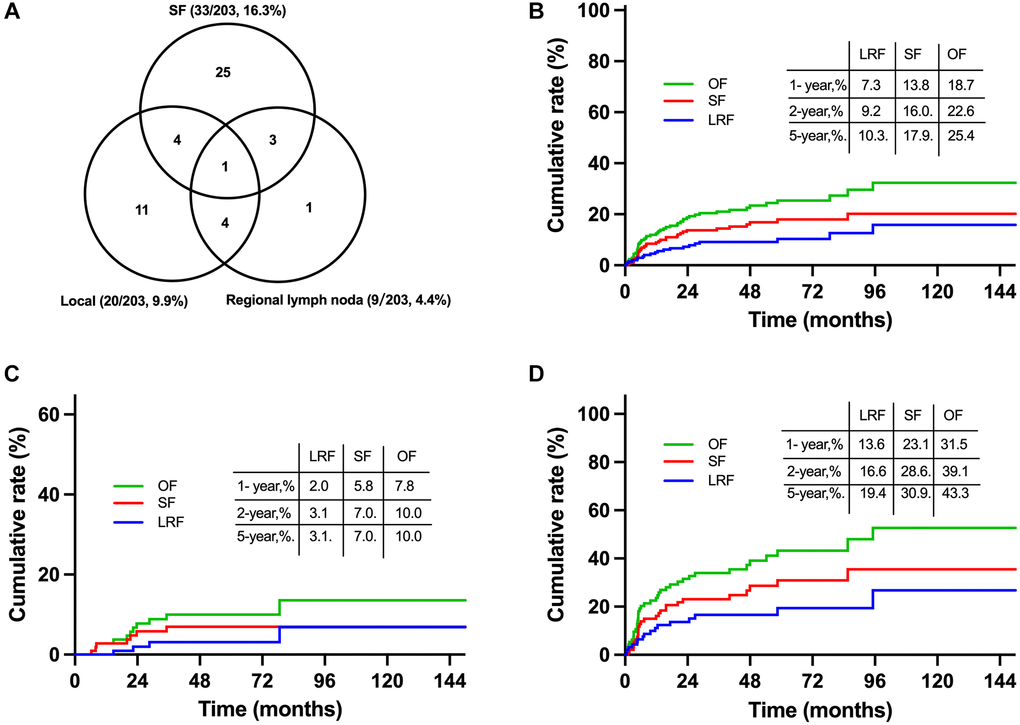
Figure 4. Failure patterns and cumulative failure incidences for early stage NKTCL receiving combined chemoradiation in the primary cohort. Failure patterns for entire patients in the primary cohort (A), cumulative failure rates for entire patients (B), patients with initial CR (C) and non-CR (D) from induction chemotherapy. Abbreviations: OF: overall failure; SF: systemic failure; LRF: locoregional failure; CR: complete remission.
Induction CT response-adapted individualized therapy for localized NKTCL
We subsequently explored the influence of intensive systemic therapy on survivals among patients with different induction responses. For patients with initial PR after induction CT, those who had ≥ 6 total cycles of CT had significantly superior survivals to those who had < 6 total cycles of CT (5-year PFS, 62.8% vs. 43.3%, P = 0.006; 5-year OS, 76.7% vs. 54.7%, P = 0.026; Figure 5A and 5B). However, for those with initial CR after induction CT, survivals were comparable between a total ≥6 and <6 cycles of CT (5-year PFS, 92.0% vs. 84.3%, P = 0.174; 5-year OS, 93.5% vs. 88.0%, P = 0.340; Figure 5C and 5D). Similar results were observed in the validation cohort (Figure 5E–5H). These results revealed that the more intensive systemic therapy improved survivals of patients with initial non-CR whose main failure pattern was high rate of SF. Based on these results, we suggested an individualized treatment strategy based on induction CT response for localized NKTCL: more intensive therapy of ≥6 total cycles of CT be recommended to those who did not reached CR from induction CT, rather than those who had initial CR. This induction response-adapted strategy needs to be further validated by large-sample prospective studies since death event was few in some certain groups in our analysis.
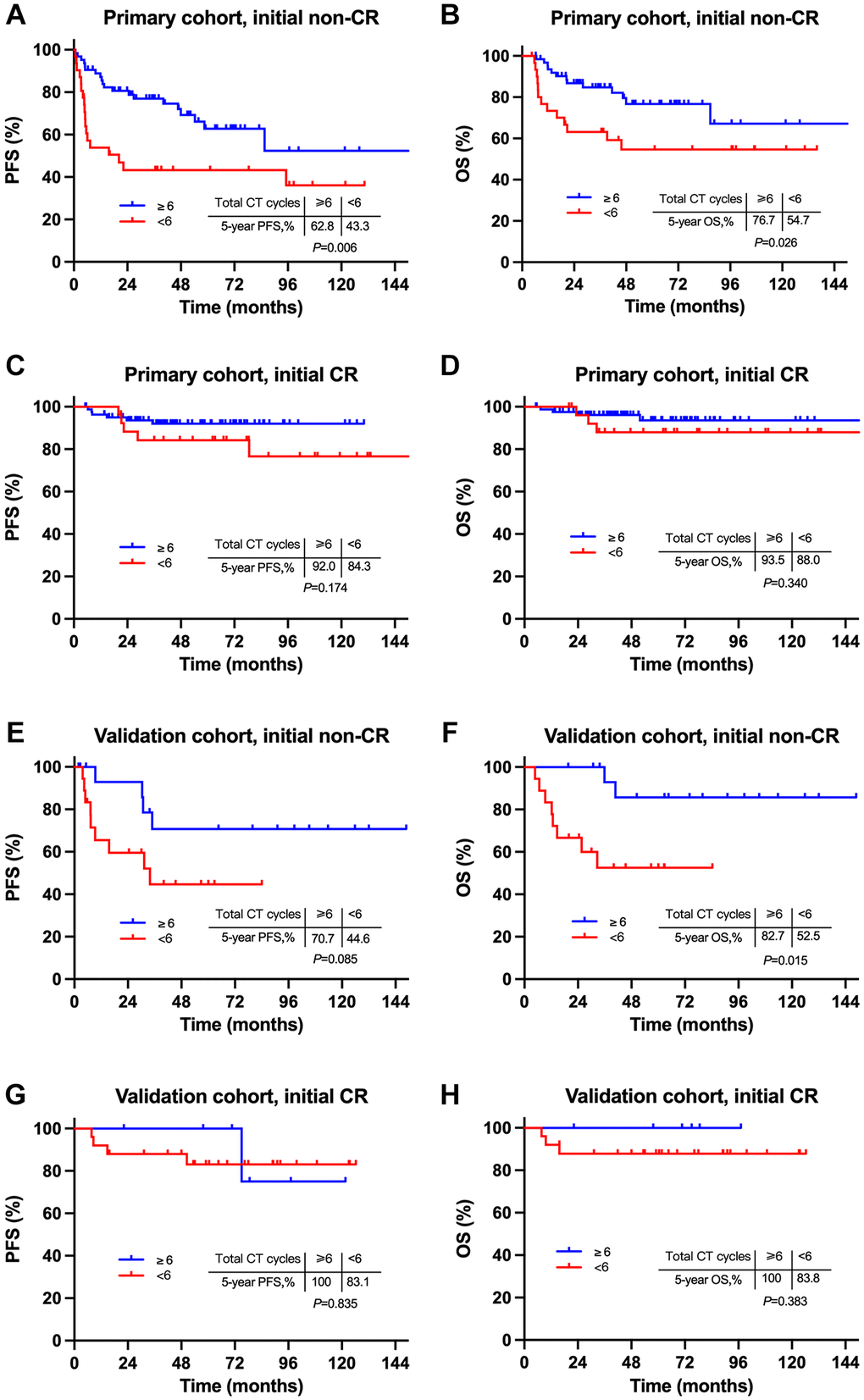
Figure 5. Comparison of survivals between intensive and non-intensive chemotherapy for patients with different induction response. Comparison of survivals between ≥6 cycles and <6 cycles of CT for patients with initial non-CR (A and B) and initial CR (C and D) in the primary cohort, initial non-CR (E and F) and initial CR (G and H) in the validation cohort. Abbreviations: PFS: progression-free survival; OS: overall survival; CR: complete remission.
Discussion
This is a comparably large-scale, real-world study which focuses on the association between first-line induction CT response and long-term survivals among early stage UADT-NKTCL. Firstly, our results revealed that early deep remission from induction CT was associated with prolonged PFS and OS in the presence of combined RT for localized NKTCL. Though most patients with initial non-CR got final CR after subsequent RT, they still had significantly higher relapse and death hazard than those with initial CR. Secondly, we demonstrated SF as the main failure pattern for patients receiving CRT, and those with initial non-CR had significantly higher cumulative incidence of SF. Lastly and importantly, we suggested an induction CT response-adapted individualized therapy for localized NKTCL that more intensive therapy of ≥6 total cycles of CT might be recommended for those with initial non-CR rather than initial CR from induction CT.
It is well established that RT is the most effective treatment modality for rapid LRC in localized NKTCL [8, 14, 29]. The CR rate of RT alone reached 70–100% for localized diseases, significantly higher than that of CT alone (25–50%) [8, 13, 40]. Improved LRC was revealed to translate into survival benefits: an absolute gain of 10% LRC provided a 9.54% improvement in PFS and 8.79% improvement in OS [10]. Meanwhile, the survival benefit of adding CT to RT has also been proved by several comparison studies [14, 15, 41]. In a retrospective analysis of 1360 early stage NKTCL patients from 20 institutions in China, combined CT+RT (vs. RT alone) significantly improved PFS (63.5% vs. 54.2%, P < 0.001) and OS (73.2% vs. 60.9%, P < 0.001) for intermediate-/high-risk NKTCL [15]. Moreover, the addition of new-regimens to RT achieved more survival benefit compared with ANT-based regimens (5-year OS, 59.5% vs. 44.5%) [41]. However, the association between tumor’s sensitivity to CT and survivals has not been fully discussed in localized NKTCL.
Our results proved induction CT response as an independent prognostic factor in NKTCL. In line with published studies, the CR rate was 53.7% after induction CT and improved to 89.7% after RT in our study [16, 17, 20–23, 26]. Patients who were highly sensitive to induction CT had higher final CR rate at the completion of RT (99.1% vs. 85.2%, P < 0.001), which might explain the survival advantage of patients with initial CR over initial non-CR from induction CT (5-year OS, 92.2% for initial CR vs. 70.7% for initial PR vs. 60.6% for initial non-response, P < 0.001). Furthermore, among patients who had final CR after RT, those with initial CR showed a tendency of prolonged OS, though the difference was not statistically significant. This indicated that RT was vital in improving LRC and long-term survivals of localized NKTCL, and meanwhile, deep remission from induction CT also contributed to the survival benefit. Conclusively, we demonstrated that initial CR from induction CT was associated with improved survivals in the modality of CRT for localized NKTCL. These results were consistent with Jing Yang’s study [42]. Yang reported in a study of 193 stage I-II NKTCL patients who received sequential or sandwich chemoradiation. The CR rate of induction GELOX and EPOCHL (etoposide, vincristine, doxorubicin, prednisone, cyclophosphamide, L-asparaginase/pegaspargase) were 75% and 56%, respectively; patients who achieved CR from induction chemotherapy had superior PFS than those with non-CRs (P = 0.033) [42].
Actually, early deep remission from upfront or early RT has been reported essential in improving survivals for localized NKTCL and no more than 3 cycles of induction CT prior to RT were recommended [15, 29, 40, 43]. In a multicenter comprehensive study of 1273 localized NKTCL, early RT+CT yielded superior 5-year OS to initial CT +RT (72.2% vs. 58.3%, P = 0.017) [14]. In our prior comparison analysis of 75 patients with localized NKTCL receiving first-line IMRT and GDP, upfront RT achieved a higher initial CR rate (91.1% vs. 40.0%; P < 0.001), 5-year LRC (90.8% vs. 66.9%; P = 0.020) and PFS (81.6% vs. 56.0%, P = 0.017), as compared to initial CT followed by RT [29]. In the present study, majority (87.2%) of patients received 2–3 cycles of induction CT prior to RT. In this setting of early RT in our study, initial CR from induction predicted superior survivals over initial non-CRs, indicating that early deep remission from induction CT might also be a positive prognostic factor, and the delay of deep remission might negatively impact survivals for localized NKTCL.
Our results revealed that most treatment failure occurred within the first two years of treatment. In line with published studies, the 5-year cumulative failure rate was 25.4% for localized NKTCL in this study [18, 19, 21, 32, 37]. Furthermore, we demonstrated SF (17.9%) as the main failure pattern for early stage NKTCL receiving first-line CRT. In the old therapy era of CHOP or CHOP-like regimens, the cumulative incidences of SF at 5 years was 24.1% for patients who received CRT compared with 28.5% for those who received RT alone (P = 0.758) [32]. Though it was hard to make a direct comparison, the SF rate in this study was slightly lower than that in Li’s study, partly due to the administration of more effective new regimens. However, our results showed SF was still the main failure pattern, especially for those who did not obtain initial CR from induction (30.9%). It was therefore inferred that more intensive systemic therapy was in urgent need to prevent systemic dissemination, which was the key and difficult point for treatment of localized NKTCL in the modern era.
Therefore, we proposed a response-adapted individualized therapy for patients with localized NKTCL in this study. For patients who did not reach CR from induction CT, more intensive therapy of ≥6 total cycles of CT might be recommended (5-year OS, 76.7% vs. 54.7%, P = 0.026). However, more intensive therapy did not bring any further survival benefit for patients with initial CR. Conclusively, the induction response might be used for better risk stratification and the induction response-adapted individualized strategy might be of potential value in decision-making in NKTCL. Actually, apart from increased drug dosage which have been discussed above, combination of CRT with new anticancer agents including immune checkpoint inhibitors, epigenetic drugs, adaptive immune therapy and other agents might also be of potential value to decrease SF and prolong survivals for localized NKTCL, some of which are under investigation currently [9, 44–47].
This study has some limitations. Firstly, multiple CT regimens, some of which were not widely used in NKTCL were included in this study. However, all regimens were L-asparaginase/pegaspargase-based in the primary cohort. Besides, though COEPL is modified from CHOP, it is not an ANT-based regimen but incorporates L-asparaginase/pegaspargase. Survival analysis in the present study demonstrated it as an effective regimen for early stage NKTCL. Secondly, though the response assessment techniques were not identical, more than half of patients used PET scanning for induction response assessment. Lastly, this study was limited by its retrospective nature, and the sample size of the validation cohort was comparably small. Therefore, large-sample strictly-designed prospective researches are warrant to validate our conclusion.
Methods
Eligibility and study population
Consecutive patients who were newly-diagnosed localized NKTCL and underwent first-line CT and RT from 2010 to 2020 from Peking University Cancer Hospital (the primary cohort) and Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College (the validation cohort) were retrospectively reviewed. Inclusion criteria were: (1) newly diagnosed NKTCL with typical morphology and immunophenotype that included CD20/CD79ɑ, CD3ɛ, CD56, TIA-1, Gram-B, perforin, and EBV-encoded RNA in situ hybridization, according to WHO classification; (2) tumors primarily occurring in the UADT; (3) Ann Arbor stages I(E)/II(E); (4) patients received first-line induction CT followed by involved-field RT with or without consolidation therapies; (5) at least one measurable lesion; Exclusion criteria were: (1) extra-UADT tumors; (2) advanced (stage III/IV) disease; (3) patients who were treated with initial RT or CT alone; (4) incomplete clinicopathologic and follow-up information.
We observed in clinical practice that a small group of localized NKTCL developed rapidly systemically progression (such as hemophagocytic syndrome) before or in the induction phase, leaving nearly no possibilities for local treatment. It was no doubt that prognosis of these patients was fairly poor. Therefore, to better explore the influence of induction CT response on survivals in the presence of combined RT, we excluded these patients from analyze. Finally, 203 eligible patients who received first-line induction CT and RT were included in the primary cohort and 67 cases in the validation cohort.
Disease evaluation
Initial clinical evaluation included history and physical examination, PET of the whole body, nasopharyngeal endoscopy of the nasal and oral cavities, magnetic resonance of head and neck, computed tomography of chest, abdomen and pelvis, and bone marrow aspiration.
Treatment procedure and response assessment
A median 2 (range, 2–6) cycles of induction CT was given to each patient prior to involved-field RT. The CT regimens changed along with time and center. All patients in the primary cohort received L-asparaginase/pegaspargase-based regimens, including COEPL (72.9%), CHOPL/CHOPEL (24.1%), and GDPL/GELOX (3.0%). Patients in the validation cohort received L-asparaginase/pegaspargase-based regimens (42/67, 62.7%) or gemcitabine-based regimens (25/67, 37.3%). Drugs and dosages were listed in Supplementary Table 1. Extended involved-field RT was delivered using intensity modulated radiation (IMRT) or volumetric modulated Arc therapy (VMAT) techniques. The median RT dose was 54 Gy (range, 45-56; dose per fraction, 1.8–2.0 Gy) in the primary cohort and 50 Gy (range, 50–56; dose per fraction, 1.8–2.0 Gy) in the validation cohort. At the completion of RT, additional CT was given to fulfill a total 4–8 cycles of therapy.
Treatment responses were assessed after induction CT (initial response), at the completion of RT with/without consolidation therapy (final response). Examination by PET or computed tomography scans of the head, neck, chest, abdomen, and pelvis; magnetic resonance imaging of the head and neck; were repeated to assess treatment response. Response assessment was followed by the Lugano classification response criteria for NHL [48].
Statistics
Initial non-response was defined as stable disease (SD) or progression of disease (PD) from induction CT. The ORR was defined as the proportion of patients classified as CR and PR. PFS was defined as the period from the date of treatment until the date of disease progression, relapse, or death from any reason. OS was defined as the interval from the date of first treatment until the date of death from any reason or last follow-up. Survival was analyzed using the Kaplan-Meier method and compared using a log-rank test. Multivariable Cox proportional hazards regression was also performed. All statistical analyses were used SPSS 24.0 software. A two-tailed P value < 0.05 was considered statistically significant.
Supplementary Materials
Author Contributions
J.Z., YQ.S. and M.D. designed the study; Y.S., Y.X., M.W., Y.C., B.C., SN.Q., WP.L., YX.L., and NJ.L. contributed to the acquisition of data; F.Q., WY.Z. and N.D. analyzed and interpreted the data; F.Q. and WY.Z. contributed to the writing of the manuscript; and all authors revised the manuscript and gave final approval.
Acknowledgments
The authors thank all physicians and nurses who participated in this study as well as all enrolled patients at the Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, and Peking University Cancer Hospital.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement and Consent
This was a retrospective non-invasive study with ethical exemptions. Each patient provided written Informed Consent Form before treatment.
Funding
This study was supported by National Natural Science Foundation of China (grant number, 82070205 and 81870154), and Capital Health Research and Development of Special (grant number, 2022-1-2152).
References
- 1. Wang H, Fu BB, Gale RP, Liang Y. NK-/T-cell lymphomas. Leukemia. 2021; 35:2460–8. https://doi.org/10.1038/s41375-021-01313-2 [PubMed]
- 2. Tamaru JI. 2016 revision of the WHO classification of lymphoid neoplasms. Rinsho Ketsueki. 2017; 58:2188–93. https://doi.org/10.11406/rinketsu.58.2188 [PubMed]
- 3. Mundy-Bosse B, Weinstock DM. Breaking Down the Barriers to Define and Treat NK/T Cell Lymphoma. Cancer Cell. 2020; 37:263–5. https://doi.org/10.1016/j.ccell.2020.02.009 [PubMed]
- 4. Haverkos BM, Pan Z, Gru AA, Freud AG, Rabinovitch R, Xu-Welliver M, Otto B, Barrionuevo C, Baiocchi RA, Rochford R, Porcu P. Extranodal NK/T Cell Lymphoma, Nasal Type (ENKTL-NT): An Update on Epidemiology, Clinical Presentation, and Natural History in North American and European Cases. Curr Hematol Malig Rep. 2016; 11:514–27. https://doi.org/10.1007/s11899-016-0355-9 [PubMed]
- 5. Sun J, Yang Q, Lu Z, He M, Gao L, Zhu M, Sun L, Wei L, Li M, Liu C, Zheng J, Liu W, Li G, Chen J. Distribution of lymphoid neoplasms in China: analysis of 4,638 cases according to the World Health Organization classification. Am J Clin Pathol. 2012; 138:429–34. https://doi.org/10.1309/AJCP7YLTQPUSDQ5C [PubMed]
- 6. Jo JC, Yoon DH, Kim S, Lee BJ, Jang YJ, Park CS, Huh J, Lee SW, Ryu JS, Suh C. Clinical features and prognostic model for extranasal NK/T-cell lymphoma. Eur J Haematol. 2012; 89:103–10. https://doi.org/10.1111/j.1600-0609.2012.01796.x [PubMed]
- 7. Kim SJ, Yoon SE, Kim WS. Treatment of localized extranodal NK/T cell lymphoma, nasal type: a systematic review. J Hematol Oncol. 2018; 11:140. https://doi.org/10.1186/s13045-018-0687-0 [PubMed]
- 8. Li YX, Yao B, Jin J, Wang WH, Liu YP, Song YW, Wang SL, Liu XF, Zhou LQ, He XH, Lu N, Yu ZH. Radiotherapy as primary treatment for stage IE and IIE nasal natural killer/T-cell lymphoma. J Clin Oncol. 2006; 24:181–9. https://doi.org/10.1200/JCO.2005.03.2573 [PubMed]
- 9. Yamaguchi M, Suzuki R, Oguchi M. Advances in the treatment of extranodal NK/T-cell lymphoma, nasal type. Blood. 2018; 131:2528–40. https://doi.org/10.1182/blood-2017-12-791418 [PubMed]
- 10. Yang Y, Cao JZ, Lan SM, Wu JX, Wu T, Zhu SY, Qian LT, Hou XR, Zhang FQ, Zhang YJ, Zhu Y, Xu LM, Yuan ZY, et al. Association of Improved Locoregional Control With Prolonged Survival in Early-Stage Extranodal Nasal-Type Natural Killer/T-Cell Lymphoma. JAMA Oncol. 2017; 3:83–91. https://doi.org/10.1001/jamaoncol.2016.5094 [PubMed]
- 11. Wang H, Li YX, Wang WH, Jin J, Dai JR, Wang SL, Liu YP, Song YW, Wang ZY, Liu QF, Fang H, Qi SN, Liu XF, Yu ZH. Mild toxicity and favorable prognosis of high-dose and extended involved-field intensity-modulated radiotherapy for patients with early-stage nasal NK/T-cell lymphoma. Int J Radiat Oncol Biol Phys. 2012; 82:1115–21. https://doi.org/10.1016/j.ijrobp.2011.02.039 [PubMed]
- 12. Vargo JA, Patel A, Glaser SM, Balasubramani GK, Farah RJ, Marks SM, Beriwal S. The impact of the omission or inadequate dosing of radiotherapy in extranodal natural killer T-cell lymphoma, nasal type, in the United States. Cancer. 2017; 123:3176–85. https://doi.org/10.1002/cncr.30697 [PubMed]
- 13. Li YX, Wang H, Jin J, Wang WH, Liu QF, Song YW, Wang ZY, Qi SN, Wang SL, Liu YP, Liu XF, Yu ZH. Radiotherapy alone with curative intent in patients with stage I extranodal nasal-type NK/T-cell lymphoma. Int J Radiat Oncol Biol Phys. 2012; 82:1809–15. https://doi.org/10.1016/j.ijrobp.2010.10.040 [PubMed]
- 14. Yang Y, Zhu Y, Cao JZ, Zhang YJ, Xu LM, Yuan ZY, Wu JX, Wang W, Wu T, Lu B, Zhu SY, Qian LT, Zhang FQ, et al. Risk-adapted therapy for early-stage extranodal nasal-type NK/T-cell lymphoma: analysis from a multicenter study. Blood. 2015; 126:1424–32. https://doi.org/10.1182/blood-2015-04-639336 [PubMed]
- 15. Qi SN, Yang Y, Zhang YJ, Huang HQ, Wang Y, He X, Zhang LL, Wu G, Qu BL, Qian LT, Hou XR, Zhang FQ, Qiao XY, et al. Risk-based, response-adapted therapy for early-stage extranodal nasal-type NK/T-cell lymphoma in the modern chemotherapy era: A China Lymphoma Collaborative Group study. Am J Hematol. 2020; 95:1047–56. https://doi.org/10.1002/ajh.25878 [PubMed]
- 16. Michot JM, Mazeron R, Danu A, Lazarovici J, Ghez D, Antosikova A, Willekens C, Chamseddine AN, Minard V, Dartigues P, Bosq J, Carde P, Koscielny S, et al. Concurrent Etoposide, Steroid, High-dose Ara-C and Platinum chemotherapy with radiation therapy in localised extranodal natural killer (NK)/T-cell lymphoma, nasal type. Eur J Cancer. 2015; 51:2386–95. https://doi.org/10.1016/j.ejca.2015.07.009 [PubMed]
- 17. Wang L, Wang ZH, Chen XQ, Wang KF, Huang HQ, Xia ZJ. First-line combination of GELOX followed by radiation therapy for patients with stage IE/IIE ENKTL: An updated analysis with long-term follow-up. Oncol Lett. 2015; 10:1036–40. https://doi.org/10.3892/ol.2015.3327 [PubMed]
- 18. Zhu F, Liu T, Pan H, Xiao Y, Li Q, Liu X, Chen W, Wu G, Zhang L. Long-term outcomes of upfront concurrent chemoradiotherapy followed by P-GDP regimen in newly diagnosed early stage extranodal nasal-type NK/T cell lymphoma: A prospective single-center phase II study. Medicine (Baltimore). 2020; 99:e21705. https://doi.org/10.1097/MD.0000000000021705 [PubMed]
- 19. Qi F, Wang WH, He XH, Chen B, Gui L, Fang H, Liu P, Wang SL, Yang JL, Song YW, Yang S, Qi SN, Zhou SY, et al. Phase 2 Study of First-line Intensity Modulated Radiation Therapy Followed by Gemcitabine, Dexamethasone, and Cisplatin for High-Risk, Early Stage Extranodal Nasal-Type NK/T-Cell Lymphoma: The GREEN Study. Int J Radiat Oncol Biol Phys. 2018; 102:61–70. https://doi.org/10.1016/j.ijrobp.2018.05.046 [PubMed]
- 20. Yamaguchi M, Tobinai K, Oguchi M, Ishizuka N, Kobayashi Y, Isobe Y, Ishizawa K, Maseki N, Itoh K, Usui N, Wasada I, Kinoshita T, Ohshima K, et al. Phase I/II study of concurrent chemoradiotherapy for localized nasal natural killer/T-cell lymphoma: Japan Clinical Oncology Group Study JCOG0211. J Clin Oncol. 2009; 27:5594–600. https://doi.org/10.1200/JCO.2009.23.8295 [PubMed]
- 21. Xu PP, Xiong J, Cheng S, Zhao X, Wang CF, Cai G, Zhong HJ, Huang HY, Chen JY, Zhao WL. A Phase II Study of Methotrexate, Etoposide, Dexamethasone and Pegaspargase Sandwiched with Radiotherapy in the Treatment of Newly Diagnosed, Stage IE to IIE Extranodal Natural-Killer/T-Cell Lymphoma, Nasal-Type. EBioMedicine. 2017; 25:41–9. https://doi.org/10.1016/j.ebiom.2017.10.011 [PubMed]
- 22. Yoon DH, Kim SJ, Jeong SH, Shin DY, Bae SH, Hong J, Park SK, Yhim HY, Yang DH, Lee H, Kang HJ, Lee MH, Eom HS, et al. Phase II trial of concurrent chemoradiotherapy with L-asparaginase and MIDLE chemotherapy for newly diagnosed stage I/II extranodal NK/T-cell lymphoma, nasal type (CISL-1008). Oncotarget. 2016; 7:85584–91. https://doi.org/10.18632/oncotarget.11319 [PubMed]
- 23. Kim SJ, Kim K, Kim BS, Kim CY, Suh C, Huh J, Lee SW, Kim JS, Cho J, Lee GW, Kang KM, Eom HS, Pyo HR, et al. Phase II trial of concurrent radiation and weekly cisplatin followed by VIPD chemotherapy in newly diagnosed, stage IE to IIE, nasal, extranodal NK/T-Cell Lymphoma: Consortium for Improving Survival of Lymphoma study. J Clin Oncol. 2009; 27:6027–32. https://doi.org/10.1200/JCO.2009.23.8592 [PubMed]
- 24. Li J, Li Y, Zhong M, Liu W, Liu X, Li J, Li K, Yi P. A Multicenter Retrospective Comparison of Sequential versus Sandwich Chemoradiotherapy for Stage IE-IIE Extranodal Natural Killer/T-Cell Lymphoma, Nasal Type. J Cancer. 2018; 9:1598–606. https://doi.org/10.7150/jca.24310 [PubMed]
- 25. Pokrovsky VS, Vinnikov D. Defining the toxicity of current regimens for extranodal NK/T cell lymphoma: a systematic review and metaproportion. Expert Rev Anticancer Ther. 2019; 19:93–104. https://doi.org/10.1080/14737140.2019.1549992 [PubMed]
- 26. Jiang M, Zhang H, Jiang Y, Yang Q, Xie L, Liu W, Zhang W, Ji X, Li P, Chen N, Zhao S, Wang F, Zou L. Phase 2 trial of "sandwich" L-asparaginase, vincristine, and prednisone chemotherapy with radiotherapy in newly diagnosed, stage IE to IIE, nasal type, extranodal natural killer/T-cell lymphoma. Cancer. 2012; 118:3294–301. https://doi.org/10.1002/cncr.26629 [PubMed]
- 27. Ma X, Guo Y, Pang Z, Wang B, Lu H, Gu YJ, Guo X. A randomized phase II study of CEOP with or without semustine as induction chemotherapy in patients with stage IE/IIE extranodal NK/T-cell lymphoma, nasal type in the upper aerodigestive tract. Radiother Oncol. 2009; 93:492–7. https://doi.org/10.1016/j.radonc.2009.08.045 [PubMed]
- 28. Ghione P, Qi S, Imber BS, Seshan V, Moskowitz A, Galasso N, Lunning M, Straus D, Sauter C, Dahi P, Dogan A, Yahalom J, Horwitz S. Modified SMILE (mSMILE) and intensity-modulated radiotherapy (IMRT) for extranodal NK-T lymphoma nasal type in a single-center population. Leuk Lymphoma. 2020; 61:3331–41. https://doi.org/10.1080/10428194.2020.1811864 [PubMed]
- 29. Qi F, Chen B, Wang J, Lin X, Qi S, Yang J, Zhou S, Wang S, Gui L, Fang H, Liu P, Song Y, Yang S, et al. Upfront radiation is essential for high-risk early-stage extranodal NK/T-cell lymphoma, nasal type: comparison of two sequential treatment modalities combining radiotherapy and GDP (gemcitabine, dexamethasone, and cisplatin) in the modern era. Leuk Lymphoma. 2019; 60:2679–88. https://doi.org/10.1080/10428194.2019.1599111 [PubMed]
- 30. Pokrovsky VS, Vinnikov D. L-Asparaginase for newly diagnosed extra-nodal NK/T-cell lymphoma: systematic review and meta-analysis. Expert Rev Anticancer Ther. 2017; 17:759–68. https://doi.org/10.1080/14737140.2017.1344100 [PubMed]
- 31. Kwong YL, Kim WS, Lim ST, Kim SJ, Tang T, Tse E, Leung AY, Chim CS. SMILE for natural killer/T-cell lymphoma: analysis of safety and efficacy from the Asia Lymphoma Study Group. Blood. 2012; 120:2973–80. https://doi.org/10.1182/blood-2012-05-431460 [PubMed]
- 32. Li YX, Liu QF, Wang WH, Jin J, Song YW, Wang SL, Liu YP, Liu XF, Zhou LQ, Yu ZH. Failure patterns and clinical implications in early stage nasal natural killer/T-cell lymphoma treated with primary radiotherapy. Cancer. 2011; 117:5203–11. https://doi.org/10.1002/cncr.26167 [PubMed]
- 33. Kim TM, Park YH, Lee SY, Kim JH, Kim DW, Im SA, Kim TY, Kim CW, Heo DS, Bang YJ, Chang KH, Kim NK. Local tumor invasiveness is more predictive of survival than International Prognostic Index in stage I(E)/II(E) extranodal NK/T-cell lymphoma, nasal type. Blood. 2005; 106:3785–90. https://doi.org/10.1182/blood-2005-05-2056 [PubMed]
- 34. Lee J, Park YH, Kim WS, Lee SS, Ryoo BY, Yang SH, Park KW, Kang JH, Park JO, Lee SH, Kim K, Jung CW, Park YS, et al. Extranodal nasal type NK/T-cell lymphoma: elucidating clinical prognostic factors for risk-based stratification of therapy. Eur J Cancer. 2005; 41:1402–8. https://doi.org/10.1016/j.ejca.2005.03.010 [PubMed]
- 35. Cheung MM, Chan JK, Lau WH, Ngan RK, Foo WW. Early stage nasal NK/T-cell lymphoma: clinical outcome, prognostic factors, and the effect of treatment modality. Int J Radiat Oncol Biol Phys. 2002; 54:182–90. https://doi.org/10.1016/s0360-3016(02)02916-4 [PubMed]
- 36. Wang ZY, Liu QF, Wang H, Jin J, Wang WH, Wang SL, Song YW, Liu YP, Fang H, Ren H, Wu RY, Chen B, Zhang XM, et al. Clinical implications of plasma Epstein-Barr virus DNA in early-stage extranodal nasal-type NK/T-cell lymphoma patients receiving primary radiotherapy. Blood. 2012; 120:2003–10. https://doi.org/10.1182/blood-2012-06-435024 [PubMed]
- 37. Liu ZL, Bi XW, Zhang XW, Lei DX, Liu PP, Yang H, Gao Y, Jiang YX, Jiang WQ, Xia Y. Characteristics, Prognostic Factors, and Survival of Patients with NK/T-Cell Lymphoma of Non-upper Aerodigestive Tract: A 17-Year Single-Center Experience. Cancer Res Treat. 2019; 51:1557–67. [PubMed]
- 38. Wang L, Wang H, Wang JH, Xia ZJ, Lu Y, Huang HQ, Jiang WQ, Zhang YJ. Post-treatment plasma EBV-DNA positivity predicts early relapse and poor prognosis for patients with extranodal NK/T cell lymphoma in the era of asparaginase. Oncotarget. 2015; 6:30317–26. https://doi.org/10.18632/oncotarget.4505 [PubMed]
- 39. Kim SJ, Choi JY, Hyun SH, Ki CS, Oh D, Ahn YC, Ko YH, Choi S, Jung SH, Khong PL, Tang T, Yan X, Lim ST, et al, and Asia Lymphoma Study Group. Risk stratification on the basis of Deauville score on PET-CT and the presence of Epstein-Barr virus DNA after completion of primary treatment for extranodal natural killer/T-cell lymphoma, nasal type: a multicentre, retrospective analysis. Lancet Haematol. 2015; 2:e66–74. https://doi.org/10.1016/S2352-3026(15)00002-2 [PubMed]
- 40. Huang MJ, Jiang Y, Liu WP, Li ZP, Li M, Zhou L, Xu Y, Yu CH, Li Q, Peng F, Liu JY, Luo F, Lu Y. Early or up-front radiotherapy improved survival of localized extranodal NK/T-cell lymphoma, nasal-type in the upper aerodigestive tract. Int J Radiat Oncol Biol Phys. 2008; 70:166–74. https://doi.org/10.1016/j.ijrobp.2007.05.073 [PubMed]
- 41. Qi SN, Yang Y, Song YQ, Wang Y, He X, Hu C, Zhang LL, Wu G, Qu BL, Qian LT, Hou XR, Zhang FQ, Qiao XY, et al. First-line non-anthracycline-based chemotherapy for extranodal nasal-type NK/T-cell lymphoma: a retrospective analysis from the CLCG. Blood Adv. 2020; 4:3141–53. https://doi.org/10.1182/bloodadvances.2020001852 [PubMed]
- 42. Yang J, Fu R, Liu X, Wei L, Wang L. Optimal induction treatment regimens for extranodal NK/T-cell lymphoma: lessons learned, challenges, and proposals. Aging Pathobiol Ther. 2020; 2:226–9. https://doi.org/10.31491/APT.2020.12.045
- 43. Zang J, Li C, Luo SQ, Wang JH, Xu M, Zhao LN, Li WW, Yang H, Xiao F, Hitchcock YJ, Shi M. Early radiotherapy has an essential role for improving survival in patients with stage I-II nasal-type of NK/T cell lymphoma treated with L-asparaginase-containing chemotherapy--a single institution experience. Ann Hematol. 2015; 94:583–91. https://doi.org/10.1007/s00277-014-2244-4 [PubMed]
- 44. Lv K, Li X, Yu H, Chen X, Zhang M, Wu X. Selection of new immunotherapy targets for NK/T cell lymphoma. Am J Transl Res. 2020; 12:7034–47. [PubMed]
- 45. Xu J, Xu X, Chen J, Wang J, Jiang C, Lv C, Chen B. Sustained remission of multi-line relapsed extranodal NK/T-cell lymphoma, nasal type, following sintilimab and chidamide: A case report. Medicine (Baltimore). 2021; 100:e24824. https://doi.org/10.1097/MD.0000000000024824 [PubMed]
- 46. Xue W, Zhang M. Updating targets for natural killer/T-cell lymphoma immunotherapy. Cancer Biol Med. 2021; 18:52–62. https://doi.org/10.20892/j.issn.2095-3941.2020.0400 [PubMed]
- 47. Cho J, Kim SJ, Park WY, Kim J, Woo J, Kim G, Yoon SE, Ko YH, Kim WS. Immune subtyping of extranodal NK/T-cell lymphoma: a new biomarker and an immune shift during disease progression. Mod Pathol. 2020; 33:603–15. https://doi.org/10.1038/s41379-019-0392-8 [PubMed]
- 48. Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, Lister TA, and Alliance, Australasian Leukaemia and Lymphoma Group, and Eastern Cooperative Oncology Group, and European Mantle Cell Lymphoma Consortium, and Italian Lymphoma Foundation, and European Organisation for Research, and Treatment of Cancer/Dutch Hemato-Oncology Group, and Grupo Español de Médula Ósea, and German High-Grade Lymphoma Study Group, and German Hodgkin's Study Group, and Japanese Lymphorra Study Group, and Lymphoma Study Association, and NCIC Clinical Trials Group, and Nordic Lymphoma Study Group, and Southwest Oncology Group, and United Kingdom National Cancer Research Institute. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014; 32:3059–68. https://doi.org/10.1200/JCO.2013.54.8800 [PubMed]



