Introduction
Subclinical thyroid dysfunction (STD) is common in the adult population and is characterized by the absence of symptoms, an abnormal thyroid-stimulating hormone (TSH) level, and normal free thyroxine and free triiodothyronine levels [1]. Reports of the prevalence of subclinical hypothyroidism (SH) have ranged from 4.0% to 20.0% and those of subclinical hyperthyroidism (SCH) from 0.7% to 9.0% [2]. Given the low rate of progression (<0.5%) of STD to overt disease within 5 years in older individuals [3] and the limited amount of evidence to suggest that early treatment alters the clinical course, there is controversy regarding the best treatment for STD. Investigation of the potential impact of STD on long-term adverse health outcomes would help to guide clinical management of individuals at high risk.
Previous studies have already addressed the long-term adverse health outcomes in patients with STD, including cardiovascular disease, stroke, all-cause mortality, chronic kidney disease, cognitive decline, decreased bone mineral density, and fracture. These studies found that STD was associated with an increased risk of long-term adverse health outcomes [4–9]. Sexual dimorphism exists in both thyroid disorders and in health outcomes. However, the sex-specific associations of STD with long-term adverse health outcomes remain unclear. In view of the small number of reports on sex-related differences in associations of subclinical SH and SCH with chronic kidney disease, cognitive decline, and decreased bone mineral density, we performed this systematic review and meta-analysis to assess differences in the association of STD and major adverse cardiovascular events (MACE) and fractures between men and women.
Methods
Data sources, search strategy, and selection criteria
The protocol for reporting a Meta-Analysis of Observational Studies in Epidemiology was used to perform and report this systematic review and meta-analysis [10]. Cohort studies that investigated the associations of SCH or SH with the risk of MACE and fractures according to sex were eligible for inclusion and no restrictions were placed on publication language or publication status. We systematically searched the PubMed, EmBase, and Cochrane Library databases for eligible studies from inception through to November 2021 using the following medical subject headings or text words as search terms: “hypothyroidism,” “subclinical hypothyroidism,” “hyperthyroidism,” “subclinical hyperthyroidism,” “thyroid diseases,” “thyroid function,” “thyroid status,” “cohort studies,” “prospective studies,” and “follow-up studies”. We also manually searched the reference lists of original articles to identify any additional studies that met the inclusion criteria.
The literature search and study selection were performed by two authors working independently. Any disagreement between the authors was resolved by discussion until consensus was reached. Studies were eligible for inclusion if they met the following criteria: (1) participants were a general population for which outcomes had not been previously investigated; (2) exposure and control, that is, SCH or SH and TSH were within the normal range; (3) outcomes included atrial fibrillation (AF), all-cause mortality, cardiac death, coronary heart disease (CHD), heart failure, MACE, stroke, any fracture (at various sites), hip fracture, non-vertebral fracture (including hip fracture and other fractures at non-vertebral sites), and vertebral fracture; (4) sex-based analysis specific for men and women or data stratified by sex; and (5) a cohort study design.
Data collection and quality assessment
Two authors independently extracted the following information: name of the study group, year of publication, country, design, sample size, patient age and sex, setting, definitions of SCH and SH, use of thyroid medication, outcomes reported, follow-up duration, and adjustment for confounding factors. The same two authors then used the Newcastle–Ottawa Scale to assess the quality of each study based on the three domains of selection, comparability, and outcome. The total scores in each study ranged from 0 to 9 stars, with a score of ≥7 stars indicating a high-quality study. Inconsistencies in data collection and quality assessment were resolved by a third author after consulting the original article.
Statistical analysis
The associations of SCH or SH with the risk of AF, all-cause mortality, cardiac death, CHD, heart failure, MACEs, stroke, any fracture, hip fracture, non-vertebral fracture, and vertebral fracture were evaluated to estimate the sex-specific effect with its 95% confidence interval (CI). Pooled relative risks (RRs) and 95% CIs were calculated for men and women using a random-effects model [11, 12]. Heterogeneity across the included studies was assessed using the I2 value and the Q statistic; significant heterogeneity was defined as an I2 value >50.0% or a P-value <0.10 [13]. Next, an indirect comparison of the pooled RRs were performed for men and women, and the RR ratio with the 95% CI was obtained. Sensitivity analysis was performed to evaluate the robustness of the pooled effect estimate of the sex difference after removing studies that reported data only for male or female subjects. Subgroup analyses were performed to detect sex differences based on duration of follow-up and level of adjustment. Reported outcomes that were adjusted for five or more factors were considered to be highly adjusted. Potential publication bias was assessed using a funnel plot with Egger’s test and Begg’s test [14, 15]. All pooled outcomes were two-sided, and a P-value <0.05 was considered statistically significant. The statistical analysis was performed using STATA software (version 10.0; StataCorp, College Station, TX, USA).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Results
Literature search
The initial electronic search produced 17,453 records, 9,871 of which were retained after removing duplicate records. A further 9,723 studies were removed because of irrelevant titles or abstracts. The remaining 148 studies were retrieved for full-text evaluation; 124 of these studies were removed because the subjects were not divided into male and female (n = 60), the same population was reported (n = 48), or a cohort design was not used (n = 16). Review of the reference lists of the relevant studies did not identify any additional eligible studies. Finally, 24 cohort studies [16–39] were selected for meta-analysis (Figure 1).
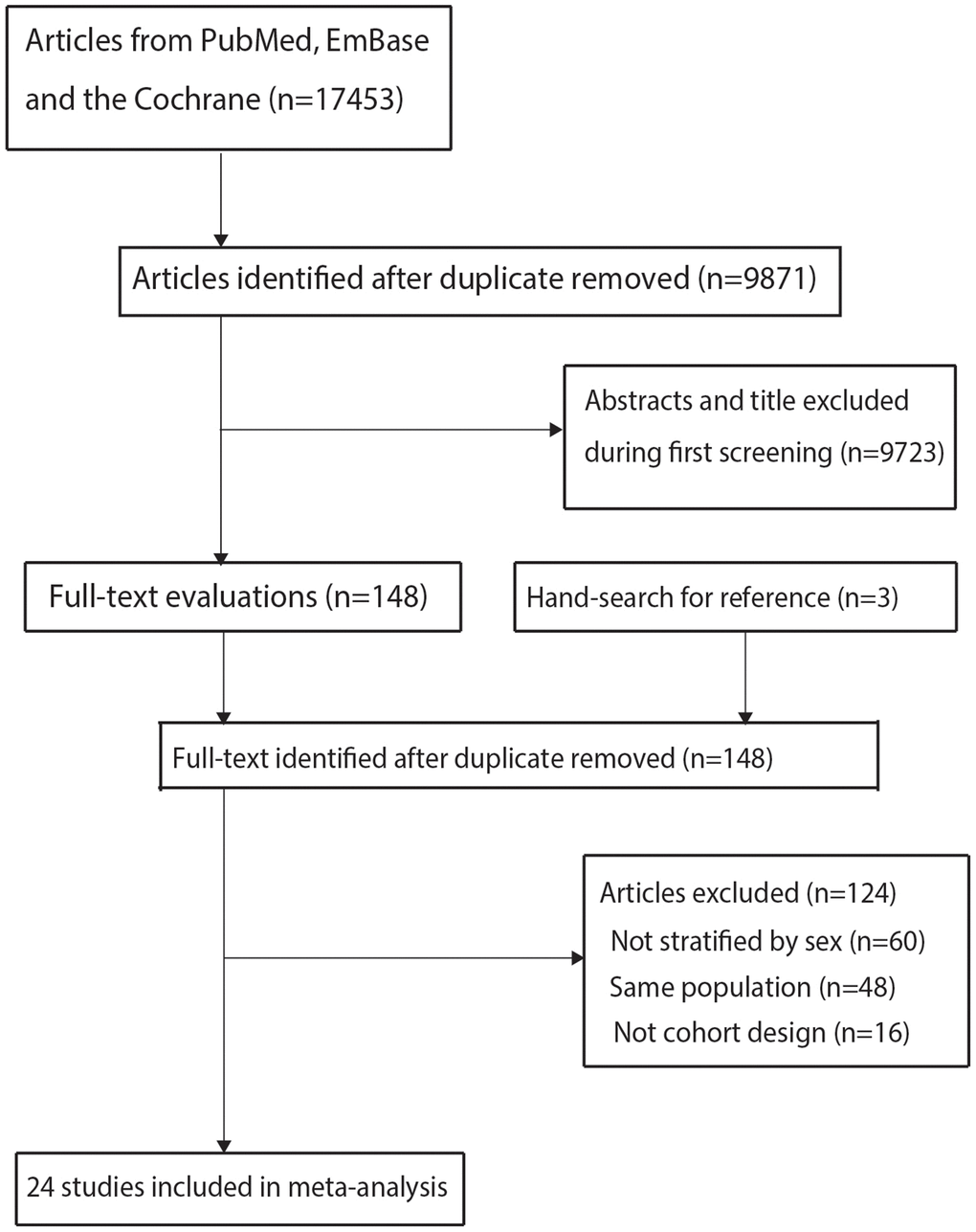
Figure 1. The details of literature search and study selection process.
Study characteristics
The background characteristics of the study participants are shown in Table 1. The 24 included studies had recruited a total of 3,480,682 individuals. Seventeen studies had a prospective cohort design and the remaining seven studies had a retrospective cohort design. Four studies were performed in Asia, 11 were performed in Europe, and the remaining nine were performed in the US or Australia. The follow-up duration ranged from 1.5 to 28.0 years and each study included 375–1,239,441 participants. The quality of each study is shown in Supplementary Table 1. All of the included studies were of high quality; three had 9 stars, sixteen had 8 stars, and the remaining five had 7 stars.
Table 1. The baseline characteristics of included studies.
| Study | Country | Design | Sample size | Age (years) | Gender | Setting | Hyperthyroidism | Hypothyroidism | TM users | Reported outcomes | Follow-up (years) | Adjusted factors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rotterdam 2000 [16] | Netherlands | Pro | 1,149 | >55.0 | Females | Population-based | NA | ≥4.0 | NA | MI | 4.6 | Age, BMI, cholesterol level, HDL-C, SBP, DBP, and smoking | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOF 2001 [17] | US | Pro | 9,704 | >65.0 | Females | Population-based | <0.50 | ≥5.5 | 12.2% | Fracture (hip, vertebral, non- vertebral), cardiac death, all-cause mortality | 3.7 | TM, previous hyperthyroidism, age, good or excellent self-rated health, current oral estrogen use | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Birmingham 2001 [18] | UK | Pro | 1,191 | >60.0 | Males and females | Population-based | <0.50 | ≥5.0 | NA | All-cause mortality, cardiac death | 8.2 | Age | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RERF 2004 [19] | Japan | Pro | 2,856 | 58.5 | Males and females | Population-based | NA | ≥5.0 | NA | MACE, CHD, stroke, all-cause mortality | 10.0 | Age, smoking | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sheffield 2008 [20] | UK | Pro | 375 | >50.0 | Females | Population-based | <0.45 | ≥4.5 | 5.0% | Fracture (hip, non-vertebral) | 10.0 | Age, smoking, BMI, history of DM, thyroid medication | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHS 2010 [21] | US | Pro | 3,576 | >65.0 | Males and females | Population-based | <0.45 | ≥4.5 | 8.3% | Fracture (hip) | 12.8 | Age, race, self-reported health status, frailty status, smoking, alcohol, height, weight, calcium intake, AOM | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OPUS 2010 [22] | Germany, France, England | Pro | 1,278 | >20.0 | Females | Population-based | <0.45 | ≥4.5 | 0.0% | Fracture (hip, non-vertebral) | 6.0 | Thyroid and thyroid-altering medication, AOM | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PROSPER 2012 [23] | Netherlands, Scotland, Ireland | Pro | 5,316 | >70.0 | Males and females | Population-based | <0.45 | NA | 2.5% | Heart failure | 3.2 | Age, sex, education, history of CVD, DM, BMI, smoking, SBP, LDL-C, creatinine, and β-blocker and antiarrhythmic use | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DNPR 2012 [24] | Denmark | Retro | 586,460 | >18.0 | Males and females | Population-based | <0.45 | ≥4.5 | NA | Atrial fibrillation, all-cause mortality, MACE, MI, heart failure, stroke | 5.5 | Sex, age, calendar year, Charlson comorbidity index, and socioeconomic status | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MrOS-US 2013 [25] | US | Pro | 1,588 | >65.0 | Males | Population-based | <0.55 | ≥4.78 | 7.6% | Fracture (hip, any, vertebral, non-vertebral), cardiac death, all-cause mortality | 11.1 | Age, clinic site, race, BMI, PA score, alcohol, smoking, corticosteroid use, and TM | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HUNT 2013 [26] | Norway | Pro | 25,205 | >40.0 | Males and females | Population-based | TSH <0.50 | >3.5 | 4.7% | Fracture (hip, any, non-vertebral), all-cause mortality, cardiac death, MI, heart failure | 12.5 | Age, BMI, and smoking | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WHI-OS 2013 [27] | US | Pro | 93,676 | >50.0 | Females | Population-based | NA | >4.68 | NA | MI, stroke | >5.0 | Age, ethnicity, gravidity, smoking, hormone therapy, and alcohol | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OPENTHYRO 2014 [28] | Denmark | Retro | 231,355 | >18.0 | Males and females | Population-based | <0.30 | >4.0 | NA | Fracture (hip, any) | 7.5 | Age, sex, Charlson index, prednisolone in last year, AOM, DM, dementia, CHF, malignancy, liver disease, rheumatic diseases, pulmonary disease, major osteoporotic fracture, and year | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ansung cohort study 2014 [29] | Korea | Pro | 2,968 | >40.0 | Males and females | Population-based | NA | >4.0 | NA | MACE | 10.0 | Reynolds risk score | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General Hospital Vienna 2015 [30] | Austria | Retro | 80,490 | >18.0 | Males and females | Population-based | NA | ≥4.5 | NA | All-cause mortality | 4.1 | Age | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDO 2015 [31] | US | Retro | 8,840 | >18.0 | Males and females | Population-based | NA | >5.0 | NA | All-cause mortality | 1.5 | Entry quarter, age, sex, race/ethnicity, cause of end-stage renal disease, vascular access, dialysis vintage, BMI, DM, CHF, cerebrovascular disease, MI, other cardiac disease, hypertension, and PAD | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| USRT 2017 [32] | US | Pro | 75,056 | >20.0 | Females | Population-based | <0.40 | >4.0 | NA | Cardiac death, stroke | 28.0 | Baseline year and age, race/ethnicity, BMI, family history of breast cancer, lifestyle and reproductive factors | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HIMS 2018 [33] | Australia | Pro | 4,248 | >70.0 | Males | Population-based | <0.40 | >4.0 | NA | Fracture (hip), all-cause mortality | 3.5 | Age, smoking, BMI, waist-hip ratio, alcohol, PA, hypertension, dyslipidaemia, DM, CVD, cancer, frailty, creatinine and vitamin D | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| US veterans 2018 [34] | US | Pro | 227,422 | 71.0 | Males and females | Population-based | <0.50 | >5.0 | 6.0% | All-cause mortality | 5.0 | Age, sex, race, ethnicity, DM, CHF, CVD, hypertension, hyperlipidemia, and Charlson comorbidity index | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Taiwan NHI 2018 [35] | China | Retro | 4,540 | >20.0 | Males and females | Population-based | NA | ≥4.5 | NA | All-cause mortality, stroke | 3.0 | Sex, age, DM, hyperlipidemia, hypertension, CVD, CHF, stroke, PAD, chronic obstructive pulmonary disease, asthma, and cancer | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| THIN 2019 [36] | UK | Retro | 863,072 | >18.0 | Males and females | Population-based | <0.40 | >4.0 | 81.7% | Fracture (any), CHD, heart failure, stroke, atrial fibrillation, all-cause mortality | 6.0 | Age, sex, BMI, smoking, fifth of townsend deprivation, prescription for lipid lowering drug, DM, hypertension, and prescription for levothyroxine | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NHANES 2020 [37] | US | Pro | 9,020 | >20.0 | Males and females | Population-based | NA | >5.6 | NA | All-cause mortality | 7.3 | Age, race/ethnicity, education status, smoking, cancer history, and estimated glomerular filtration rate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Korean NHI 2020 [38] | Korea | Retro | 1,239,441 | >20.0 | Males and females | Population-based | <0.45 | NA | NA | MI, stroke | 10.0 | Hypertension, fasting glucose, smoking, cholesterol, and obesity | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MrOS-Sweden 2021 [39] | Sweden | Pro | 1,856 | >69.0 | Males | Population-based | <0.45 | NA | 1.7% | Fracture (any, vertebral) | 8.9 | Age, MrOS site, levothyroxine treatment, BMI, appendicular lean mass, grip strength, walking speed, smoking, and total hip sBMD | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: AOM: antiosteoporosis medication; BMD: bone mineral density; BMI: body mass index; CHF: congestive heart failure; CVD: cardiovascular disease; DBP: diastolic blood pressure; DM: diabetes mellitus; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; PA: physical activity; PAD: peripheral arterial disease; Pro: prospective; Retro: retrospective; SBP: systolic blood pressure; TM: thyroid medication. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Atrial fibrillation
SCH and SH were not associated with an increased risk of AF irrespective of sex (Figure 2, Supplementary Figures 1 and 2). Moreover, we noted significant heterogeneity in the association of SCH or SH with the risk of AF in women. There were no significant sex-related differences in the association of SCH (RR ratio 1.10; 95% CI 0.74–1.66; P = 0.636) or SH (RR ratio 0.97; 95% CI 0.79–1.20; P = 0.780) with the risk of AF (Table 2). These findings persisted after removing indirect comparison studies (Table 3). The results of subgroup analyses were consistent with those of the overall analysis, and any sex-related differences in the association of SCH or SH with the risk of AF were not significant (Table 4). No significant publication bias with regard to the association of SCH or SH with the risk of AF was observed (Supplementary Figures 3 and 4).
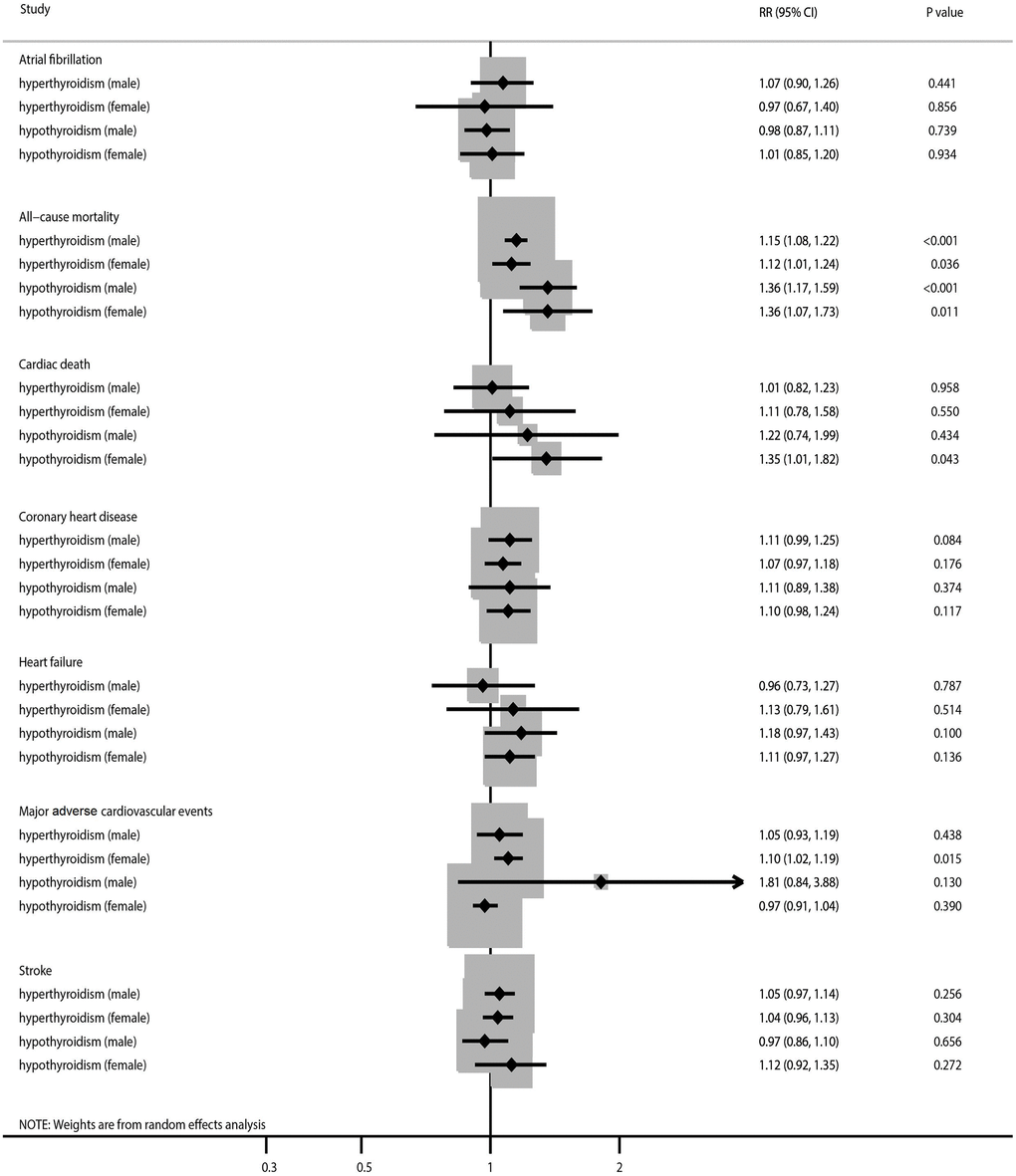
Figure 2. The summary results stratified by gender in the association of subclinical thyroid dysfunction with the risk of atrial fibrillation, all-cause mortality, cardiac death, coronary heart disease, heart failure, major adverse cardiovascular events, and stroke.
Table 2. Sex difference in long term health outcomes for patients with subclinical thyroid dysfunction.
| Outcomes | Subclinical thyroid dysfunction | Males | Females | RR ratio | P value |
| Atrial fibrillation | Hyperthyroidism | 1.07 (0.90–1.26) | 0.97 (0.66–1.40) | 1.10 (0.74–1.66) | 0.636 |
| Hypothyroidism | 0.98 (0.87–1.11) | 1.01 (0.85–1.20) | 0.97 (0.79–1.20) | 0.780 | |
| All-cause mortality | Hyperthyroidism | 1.15 (1.08–1.22) | 1.12 (1.01–1.24) | 1.03 (0.91–1.16) | 0.664 |
| Hypothyroidism | 1.36 (1.17–1.59) | 1.36 (1.07–1.73) | 1.00 (0.75–1.33) | 1.000 | |
| Cardiac death | Hyperthyroidism | 1.01 (0.82–1.23) | 1.11 (0.78–1.58) | 0.91 (0.61–1.37) | 0.649 |
| Hypothyroidism | 1.22 (0.74–1.99) | 1.35 (1.01–1.82) | 0.90 (0.51–1.61) | 0.730 | |
| Coronary heart disease | Hyperthyroidism | 1.11 (0.99–1.25) | 1.07 (0.97–1.18) | 1.04 (0.89–1.21) | 0.637 |
| Hypothyroidism | 1.11 (0.89–1.38) | 1.10 (0.98–1.24) | 1.01 (0.79–1.29) | 0.943 | |
| Heart failure | Hyperthyroidism | 0.96 (0.73–1.27) | 1.13 (0.79–1.61) | 0.85 (0.54–1.33) | 0.479 |
| Hypothyroidism | 1.18 (0.97–1.43) | 1.11 (0.97–1.27) | 1.06 (0.84–1.35) | 0.612 | |
| Major advance cardiovascular events | Hyperthyroidism | 1.05 (0.93–1.19) | 1.10 (1.02–1.19) | 0.96 (0.83–1.10) | 0.531 |
| Hypothyroidism | 1.81 (0.84–3.88) | 0.97 (0.91–1.04) | 1.87 (0.87–4.02) | 0.111 | |
| Stroke | Hyperthyroidism | 1.05 (0.97–1.14) | 1.04 (0.96–1.13) | 1.01 (0.90–1.13) | 0.870 |
| Hypothyroidism | 0.97 (0.86–1.10) | 1.12 (0.92–1.35) | 0.87 (0.69–1.09) | 0.216 | |
| Any fracture | Hyperthyroidism | 1.17 (1.00–1.36) | 1.02 (0.93–1.12) | 1.15 (0.96–1.37) | 0.134 |
| Hypothyroidism | 1.10 (0.96–1.26) | 0.99 (0.94–1.05) | 1.11 (0.96–1.29) | 0.160 | |
| Hip fracture | Hyperthyroidism | 1.47 (1.04–2.07) | 1.38 (1.01–1.89) | 1.07 (0.67–1.70) | 0.790 |
| Hypothyroidism | 0.97 (0.67–1.41) | 0.97 (0.89–1.06) | 1.00 (0.68–1.47) | 1.000 | |
| Non-vertebral fracture | Hyperthyroidism | 1.18 (0.45–3.11) | 1.24 (0.87–1.77) | 0.95 (0.34–2.66) | 0.925 |
| Hypothyroidism | 0.96 (0.63–1.46) | 1.31 (0.79–2.16) | 0.73 (0.38–1.41) | 0.353 | |
| Vertebral fracture | Hyperthyroidism | 3.07 (1.62–5.79) | 3.43 (1.52–7.71) | 0.90 (0.32–2.51) | 0.833 |
Table 3. Sensitivity analysis for direct comparisons of sex difference in long term health outcomes for patients with subclinical thyroid dysfunction.
| Outcomes | Subclinical thyroid dysfunction | Males | Females | RR ratio | P value |
| Atrial fibrillation | Hyperthyroidism | 1.07 (0.90–1.26) | 0.97 (0.66–1.40) | 1.03 (0.84–1.25) | 0.797 |
| Hypothyroidism | 0.98 (0.87–1.11) | 1.01 (0.85–1.20) | 0.99 (0.85–1.14) | 0.848 | |
| All-cause mortality | Hyperthyroidism | 1.15 (1.09–1.23) | 1.12 (1.01–1.25) | 1.01 (0.93–1.09) | 0.876 |
| Hypothyroidism | 1.39 (1.18–1.64) | 1.43 (1.09–1.86) | 0.97 (0.90–1.04) | 0.344 | |
| Cardiac death | Hyperthyroidism | 1.00 (0.80–1.20) | 0.90 (0.70–1.10) | 1.11 (0.82–1.51) | 0.496 |
| Hypothyroidism | 1.06 (0.65–1.72) | 1.76 (1.21–2.56) | 0.60 (0.33–1.11) | 0.106 | |
| Coronary heart disease | Hyperthyroidism | 1.11 (0.99–1.25) | 1.07 (0.97–1.18) | 1.01 (0.86–1.19) | 0.878 |
| Hypothyroidism | 1.11 (0.89–1.38) | 1.06 (0.98–1.16) | 0.99 (0.86–1.16) | 0.945 | |
| Heart failure | Hyperthyroidism | 0.96 (0.73–1.27) | 1.06 (0.71–1.57) | 0.96 (0.74–1.24) | 0.751 |
| Hypothyroidism | 1.18 (0.97–1.43) | 1.11 (0.97–1.27) | 1.08 (0.94–1.24) | 0.268 | |
| Major advance cardiovascular events | Hyperthyroidism | 1.05 (0.93–1.19) | 1.10 (1.02–1.19) | 0.95 (0.83–1.10) | 0.531 |
| Hypothyroidism | 1.81 (0.84–3.88) | 0.97 (0.91–1.04) | 1.59 (0.81–3.12) | 0.181 | |
| Stroke | Hyperthyroidism | 1.05 (0.97–1.14) | 1.04 (0.95–1.14) | 0.97 (0.88–1.08) | 0.620 |
| Hypothyroidism | 0.97 (0.86–1.10) | 1.10 (0.87–1.40) | 1.02 (0.87–1.18) | 0.841 | |
| Any fracture | Hyperthyroidism | 1.10 (0.94–1.30) | 1.02 (0.93–1.12) | 1.07 (0.90–1.28) | 0.423 |
| Hypothyroidism | 1.10 (0.94–1.29) | 0.99 (0.94–1.05) | 1.10 (0.92–1.33) | 0.302 | |
| Hip fracture | Hyperthyroidism | 1.34 (0.86–2.09) | 1.15 (0.98–1.34) | 1.13 (0.81–1.58) | 0.473 |
| Hypothyroidism | 0.88 (0.56–1.37) | 0.97 (0.88–1.07) | 0.87 (0.55–1.39) | 0.565 | |
| Non-vertebral fracture | Hyperthyroidism | 0.47 (0.07–3.41) | 1.08 (0.75–1.56) | 0.44 (0.06–3.14) | 0.410 |
| Hypothyroidism | 0.89 (0.40–1.98) | 1.11 (0.90–1.37) | 0.80 (0.35–1.83) | 0.601 |
Table 4. Subgroup analyses for investigated outcomes.
| Outcomes | Factors | Subgroups | Males | Females | RR ratio | P value |
| Atrial fibrillation (hyperthyroidism) | Follow-up (years) | ≥10.0 | − | − | − | − |
| <10.0 | 1.07 (0.90–1.26) | 0.97 (0.66–1.40) | 1.10 (0.73–1.67) | 0.641 | ||
| Adjusted level | High | 0.95 (0.77–1.18) | 0.81 (0.71–0.92) | 1.17 (0.91–1.51) | 0.211 | |
| Low | 1.20 (0.99–1.45) | 1.35 (1.20–1.51) | 0.89 (0.71–1.11) | 0.300 | ||
| Atrial fibrillation (hypothyroidism) | Follow-up (years) | ≥10.0 | − | − | − | − |
| <10.0 | 0.98 (0.87–1.11) | 1.01 (0.85–1.20) | 0.97 (0.79–1.20) | 0.780 | ||
| Adjusted level | High | 1.01 (0.86–1.18) | 1.09 (0.99–1.21) | 0.93 (0.77–1.12) | 0.425 | |
| Low | 0.94 (0.77–1.14) | 0.86 (0.77–0.97) | 1.09 (0.87–1.37) | 0.444 | ||
| All-cause mortality (hyperthyroidism) | Follow-up (years) | ≥10.0 | 0.78 (0.41–1.48) | − | − | − |
| <10.0 | 1.15 (1.09–1.23) | 1.12 (1.01–1.24) | 1.03 (0.91–1.16) | 0.663 | ||
| Adjusted level | High | 1.12 (1.00–1.25) | 1.14 (1.02–1.28) | 0.98 (0.84–1.15) | 0.827 | |
| Low | 1.17 (1.06–1.29) | 1.06 (0.79–1.42) | 1.10 (0.81–1.50) | 0.531 | ||
| All-cause mortality (hypothyroidism) | Follow-up (years) | ≥10.0 | 1.14 (0.83–1.58) | 1.10 (0.69–1.76) | 1.04 (0.59–1.83) | 0.902 |
| <10.0 | 1.34 (1.15–1.57) | 1.45 (1.11–1.89) | 0.92 (0.68–1.26) | 0.616 | ||
| Adjusted level | High | 1.31 (1.12–1.53) | 1.49 (1.16–1.91) | 0.88 (0.66–1.18) | 0.391 | |
| Low | 1.44 (0.75–2.74) | 1.25 (0.71–2.21) | 1.15 (0.49–2.73) | 0.747 | ||
| Cardiac death (hyperthyroidism) | Follow-up (years) | ≥10.0 | 1.12 (0.45–2.78) | 1.43 (0.62–2.31) | 0.78 (0.25–2.41) | 0.670 |
| <10.0 | 1.00 (0.82–1.22) | 0.90 (0.72–1.12) | 1.11 (0.83–1.50) | 0.487 | ||
| Adjusted level | High | 1.12 (0.45–2.78) | 0.97 (0.73–1.28) | 1.15 (0.45–2.99) | 0.767 | |
| Low | 1.00 (0.82–1.22) | 1.38 (0.55–3.43) | 0.72 (0.28–1.85) | 0.500 | ||
| Cardiac death (hypothyroidism) | Follow-up (years) | ≥10.0 | 1.22 (0.74–1.99) | 1.40 (0.98–2.01) | 0.87 (0.47–1.61) | 0.659 |
| <10.0 | − | 0.92 (0.25–3.36) | − | − | ||
| Adjusted level | High | 1.73 (0.49–6.09) | 1.21 (1.03–1.41) | 1.43 (0.40–5.09) | 0.581 | |
| Low | 1.06 (0.65–1.72) | 1.76 (1.21–2.56) | 0.60 (0.33–1.11) | 0.106 | ||
| Coronary heart disease (hyperthyroidism) | Follow-up (years) | ≥10.0 | 1.33 (0.84–2.09) | 1.20 (1.02–1.42) | 1.11 (0.68–1.80) | 0.678 |
| <10.0 | 1.05 (0.90–1.23) | 1.02 (0.91–1.14) | 1.03 (0.85–1.25) | 0.768 | ||
| Adjusted level | High | 1.09 (0.96–1.24) | 1.10 (0.96–1.27) | 0.99 (0.82–1.20) | 0.925 | |
| Low | 1.32 (0.79–2.18) | 0.99 (0.84–1.17) | 1.33 (0.78–2.27) | 0.291 | ||
| Coronary heart disease (hypothyroidism) | Follow-up (years) | ≥10.0 | 1.75 (0.46–6.58) | 1.17 (0.85–1.62) | 1.50 (0.38–5.88) | 0.564 |
| <10.0 | 1.07 (0.87–1.31) | 1.10 (0.95–1.26) | 0.97 (0.76–1.25) | 0.827 | ||
| Adjusted level | High | 1.05 (0.74–1.49) | 1.11 (0.90–1.37) | 0.95 (0.63–1.42) | 0.790 | |
| Low | 1.23 (0.81–1.89) | 1.13 (0.99–1.28) | 1.09 (0.70–1.69) | 0.707 | ||
| Heart failure (hyperthyroidism) | Follow-up (years) | ≥10.0 | − | 1.65 (0.89–3.07) | − | − |
| <10.0 | 0.96 (0.73–1.27) | 1.06 (0.71–1.57) | 0.91 (0.56–1.47) | 0.688 | ||
| Adjusted level | High | 0.88 (0.57–1.34) | 0.99 (0.61–1.60) | 0.89 (0.47–1.69) | 0.720 | |
| Low | 1.10 (0.93–1.30) | 1.25 (1.13–1.39) | 0.88 (0.72–1.07) | 0.203 | ||
| Heart failure (hypothyroidism) | Follow-up (years) | ≥10.0 | 0.62 (0.26–1.47) | 1.15 (0.86–1.54) | 0.54 (0.22–1.34) | 0.185 |
| <10.0 | 1.21 (1.01–1.45) | 1.11 (0.94–1.31) | 1.09 (0.85–1.39) | 0.491 | ||
| Adjusted level | High | 1.30 (0.96–1.75) | 1.20 (0.96–1.49) | 1.08 (0.75–1.57) | 0.673 | |
| Low | 0.98 (0.61–1.57) | 1.01 (0.91–1.11) | 0.97 (0.60–1.57) | 0.903 | ||
| Major advance cardiovascular events (hyperthyroidism) | Follow-up (years) | ≥10.0 | − | − | − | − |
| <10.0 | 1.05 (0.93–1.19) | 1.10 (1.02–1.19) | 0.95 (0.83–1.10) | 0.531 | ||
| Adjusted level | High | − | − | − | − | |
| Low | 1.05 (0.93–1.19) | 1.10 (1.02–1.19) | 0.95 (0.83–1.10) | 0.531 | ||
| Major advance cardiovascular events (hypothyroidism) | Follow-up (years) | ≥10.0 | 2.54 (1.42–4.52) | 1.04 (0.66–1.64) | 2.44 (1.17–5.10) | 0.017 |
| <10.0 | 1.03 (0.91–1.17) | 0.97 (0.91–1.04) | 1.06 (0.92–1.22) | 0.408 | ||
| Adjusted level | High | − | − | − | − | |
| Low | 1.81 (0.84–3.88) | 0.97 (0.91–1.04) | 1.87 (0.87–4.02) | 0.111 | ||
| Stroke (hyperthyroidism) | Follow-up (years) | ≥10.0 | 1.05 (0.95–1.16) | 1.13 (1.04–1.22) | 0.93 (0.82–1.06) | 0.260 |
| <10.0 | 1.05 (0.91–1.22) | 1.00 (0.90–1.11) | 1.05 (0.88–1.26) | 0.596 | ||
| Adjusted level | High | 1.07 (0.97–1.17) | 1.04 (0.93–1.16) | 1.03 (0.89–1.19) | 0.700 | |
| Low | 0.98 (0.80–1.20) | 1.03 (0.92–1.15) | 0.95 (0.75–1.20) | 0.673 | ||
| Stroke (hypothyroidism) | Follow-up (years) | ≥10.0 | 0.60 (0.11–3.17) | 1.28 (0.93–1.75) | 0.47 (0.08–2.59) | 0.385 |
| <10.0 | 0.97 (0.86–1.10) | 1.09 (0.88–1.35) | 0.89 (0.70–1.14) | 0.354 | ||
| Adjusted level | High | 0.99 (0.83–1.17) | 1.17 (0.89–1.55) | 0.85 (0.61–1.17) | 0.316 | |
| Low | 0.95 (0.79–1.15) | 0.93 (0.84–1.03) | 1.02 (0.83–1.26) | 0.845 | ||
| Any fracture (hyperthyroidism) | Follow-up (years) | ≥10.0 | 1.16 (0.62–2.18) | 1.14 (0.86–1.51) | 1.02 (0.51–2.03) | 0.961 |
| <10.0 | 1.17 (0.98–1.41) | 1.00 (0.91–1.11) | 1.17 (0.95–1.44) | 0.138 | ||
| Adjusted level | High | 1.18 (1.00–1.40) | 1.00 (0.91–1.11) | 1.18 (0.97–1.43) | 0.097 | |
| Low | 0.84 (0.37–1.90) | 1.14 (0.86–1.51) | 0.74 (0.31–1.75) | 0.489 | ||
| Any fracture (hypothyroidism) | Follow-up (years) | ≥10.0 | 0.85 (0.61–1.17) | 1.10 (0.93–1.30) | 0.77 (0.54–1.11) | 0.168 |
| <10.0 | 1.15 (1.02–1.30) | 0.98 (0.93–1.03) | 1.17 (1.03–1.34) | 0.017 | ||
| Adjusted level | High | 1.14 (1.01–1.28) | 0.98 (0.93–1.03) | 1.16 (1.02–1.32) | 0.022 | |
| Low | 0.72 (0.46–1.13) | 1.10 (0.93–1.30) | 0.65 (0.41–1.06) | 0.083 | ||
| Hip fracture (hyperthyroidism) | Follow-up (years) | ≥10.0 | 1.98 (0.87–4.50) | 1.59 (0.79–3.21) | 1.25 (0.42–3.67) | 0.691 |
| <10.0 | 1.30 (0.96–1.76) | 1.41 (0.85–2.32) | 0.92 (0.51–1.66) | 0.786 | ||
| Adjusted level | High | 1.64 (1.06–2.53) | 1.70 (0.97–2.97) | 0.96 (0.47–1.96) | 0.921 | |
| Low | 0.99 (0.40–2.44) | 1.28 (0.86–1.90) | 0.77 (0.29–2.08) | 0.610 | ||
| Hip fracture (hypothyroidism) | Follow-up (years) | ≥10.0 | 1.18 (0.53–2.65) | 1.11 (0.89–1.37) | 1.06 (0.46–2.45) | 0.886 |
| <10.0 | 0.79 (0.57–1.10) | 0.94 (0.85–1.04) | 0.84 (0.60–1.19) | 0.322 | ||
| Adjusted level | High | 1.06 (0.68–1.65) | 0.94 (0.85–1.03) | 1.13 (0.72–1.77) | 0.604 | |
| Low | 0.64 (0.37–1.10) | 1.20 (0.94–1.53) | 0.53 (0.29–0.97) | 0.039 | ||
| Non-vertebral fracture (hyperthyroidism) | Follow-up (years) | ≥10.0 | 1.18 (0.45–3.11) | 1.14 (0.80–1.61) | 1.04 (0.37–2.89) | 0.948 |
| <10.0 | − | 1.42 (0.71–2.83) | − | − | ||
| Adjusted level | High | 1.54 (0.63–3.76) | 2.05 (1.17–3.61) | 0.75 (0.26–2.16) | 0.595 | |
| Low | 0.47 (0.07–3.28) | 0.99 (0.74–1.34) | 0.47 (0.07–3.32) | 0.453 | ||
| Non-vertebral fracture (hypothyroidism) | Follow-up (years) | ≥10.0 | 0.96 (0.63–1.46) | 1.38 (0.78–2.45) | 0.70 (0.34–1.41) | 0.316 |
| <10.0 | − | 0.38 (0.02–6.69) | − | − | ||
| Adjusted level | High | 0.99 (0.60–1.62) | 2.05 (1.04–4.05) | 0.48 (0.21–1.12) | 0.090 | |
| Low | 0.89 (0.40–1.98) | 1.10 (0.90–1.36) | 0.81 (0.35–1.85) | 0.615 | ||
| Vertebral fracture (hyperthyroidism) | Follow-up (years) | ≥10.0 | 1.29 (0.18–9.32) | − | − | − |
| <10.0 | 3.39 (1.73–6.64) | 3.43 (1.52–7.71) | 0.99 (0.34–2.84) | 0.983 | ||
| Adjusted level | High | 3.07 (1.62–5.79) | 3.43 (1.52–7.71) | 0.90 (0.32–2.51) | 0.833 | |
| Low | − | − | − | − |
All-cause mortality
SCH and SH were associated with an increased risk of all-cause mortality in both men and women (Figure 2, Supplementary Figures 5 and 6). There was significant heterogeneity for all-cause mortality related to SCH in women and all-cause mortality related to SH in both sexes. No sex differences were observed in the association of SCH (RR ratio 1.03; 95% CI 0.91–1.16; P = 0.664) or SH (RR ratio 1.00; 95% CI 0.75–1.33; P = 1.000) with the risk of all-cause mortality (Table 2). The pooled conclusions for these associations were robust (Table 3). The results of the subgroup analyses were consistent with those of the overall analysis (Table 4). There was no significant publication bias with regard to studies of the association of SCH or SH with the risk of all-cause mortality (Supplementary Figures 7 and 8).
Cardiac death
An association was found between SH and an increased risk of cardiac death in women (Figure 2, Supplementary Figures 9 and 10). Moreover, there was significant heterogeneity in terms of cardiac death related to SCH. There were no significant sex-related differences between in the association of SCH (RR ratio 0.91; 95% CI 0.61–1.37; P = 0.649) or SH (RR ratio 0.90; 95% CI 0.51–1.61; P = 0.730) with the risk of cardiac death (Table 2). Sensitivity analysis indicated that the pooled conclusion for these associations was stable (Table 3). The results of the subgroup analyses were consistent with those of the overall analysis in all subgroups (Table 4). No significant publication bias was detected with regard to studies of the association of SCH or SH with the risk of cardiac death (Supplementary Figures 11 and 12).
Coronary heart disease
There was no significant association of SCH or SH with the risk of CHD, irrespective of sex (Figure 2, Supplementary Figures 13 and 14). We noted significant heterogeneity in the association of SH with the risk of CHD. No sex differences were observed in the association of SCH (RR ratio 1.04; 95% CI 0.89–1.21; P = 0.637) or SH (RR ratio 1.01; 95% CI 0.79–1.29; P = 0.943) with the risk of CHD (Table 2). The results of the sensitivity and subgroup analyses were consistent with those of the overall analysis, with no sex differences detected (Tables 3 and 4). However, there was potential publication bias in the association of SH with the risk of CHD (P-value for Egger’s test, 0.041; P-value for Begg’s test, 0.244; Supplementary Figures 15 and 16).
Heart failure
We did not find a significant association of SCH or SH with the risk of heart failure regardless of sex (Figure 2, Supplementary Figures 17 and 18). There was potentially significant heterogeneity for heart failure related to SCH or SH in both sexes. There were no significant sex-related differences in the association of SCH (RR ratio 0.85; 95% CI 0.54–1.33; P = 0.479) or SH (RR ratio 1.06; 95% CI 0.84–1.35; P = 0.612) with the risk of heart failure (Table 2). The results of the sensitivity and subgroup analyses were consistent with those of the overall analysis (Tables 3 and 4). There was no significant publication bias in terms of studies of the association of SCH or SH with the risk of heart failure (Supplementary Figures 19 and 20).
Major adverse cardiovascular events
SCH was associated with an increased risk of MACE in women and there was potentially significant heterogeneity for MACE related to SH in men (Figure 2, Supplementary Figures 21 and 22). There were no significant sex-related differences in the association of SCH (RR ratio 0.96; 95% CI 0.83–1.10; P = 0.531) or SH (RR ratio 1.87; 95% CI 0.87–4.02; P = 0.111) with the risk of MACE (Table 2). Sensitivity analysis indicated that the pooled conclusion regarding a sex-related difference was robust after removing the results of indirect comparisons (Table 3). Subgroup analysis found that men with SH had a higher risk of MACE when the follow-up duration was ≥10.0 years (RR ratio 2.44; 95% CI 1.17–5.10; P = 0.017; Table 4). No significant publication bias was detected with regard to the association of SH with the risk of MACE (Supplementary Figure 23).
Stroke
There was no significant association of SCH or SH with the risk of stroke regardless of sex (Figure 2, Supplementary Figures 24 and 25). No sex differences were observed in the association of SCH (RR ratio 1.01; 95% CI 0.90–1.13; P = 0.870) or SH (RR ratio 0.87; 95% CI 0.69–1.09; P = 0.216) with the risk of stroke (Table 2). The results of the sensitivity and subgroup analyses were consistent with those of the overall analysis (Tables 3 and 4). There was no significant publication bias in terms of the association of SCH or SH with the risk of stroke (Supplementary Figures 26 and 27).
Any fracture
We found an association of SCH with an increased risk of any fracture in men, with no significant heterogeneity observed (Figure 3, Supplementary Figures 28 and 29). Although the risk of any fracture with SCH (RR ratio 1.15; 95% CI 0.96–1.37; P = 0.134) or SH (RR ratio 1.11; 95% CI 0.96–1.29; P = 0.160) was higher in men, the sex differences were not statistically significant (Table 2). The results of the sensitivity analysis were consistent with those of the overall analysis (Table 3). Subgroup analysis indicated that the risk of any fracture was higher in men with SH than in women with SH when the follow-up duration was <10.0 years (RR ratio 1.17; 95% CI 1.03–1.34; P = 0.017) and in studies with a high level of adjustment (RR ratio 1.16; 95% CI 1.02–1.32; P = 0.022; Table 4). There was no significant publication bias with regard to the association of SCH or SH with the risk of any fracture (Supplementary Figures 30 and 31).
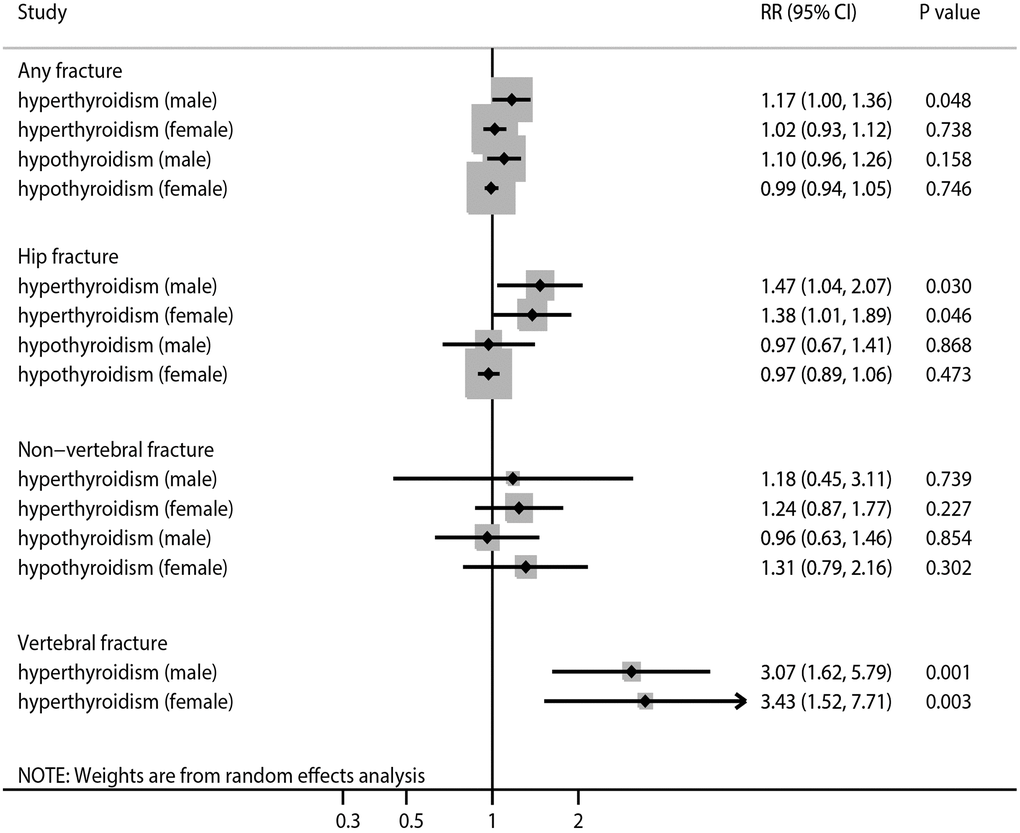
Figure 3. The summary results stratified by gender in the association of subclinical thyroid dysfunction with the risk of any fracture, hip fracture, non-vertebral fracture, and vertebral fracture.
Hip fracture
The results indicate that SCH was associated with an increased risk of hip fracture in both men and women, and there was potentially significant heterogeneity for hip fracture related to SH in men (Figure 3, Supplementary Figures 32 and 33). There were no significant differences in the association of SCH (RR ratio 1.07; 95% CI 0.67–1.70; P = 0.790) or SH (RR ratio 1.00; 95% CI 0.68–1.47; P = 1.000) with the risk of hip fracture between men and women (Table 2). The results of the sensitivity analysis were consistent with those of the overall analysis (Table 3). Subgroup analysis found that the risk of hip fracture was lower in men with SH than in women with SH on pooling of studies with a low level of adjustment (Table 4). There was significant publication bias in terms of the association of SCH with the risk of hip fracture (Supplementary Figures 34 and 35).
Non-vertebral fracture
There was no significant association of SCH or SH with the risk of non-vertebral fracture in men or women (Figure 3, Supplementary Figures 36 and 37). No sex differences were observed in the association of SCH (RR ratio 0.95; 95% CI 0.34–2.66; P = 0.925) or SH (RR ratio 0.73; 95% CI 0.38–1.41; P = 0.353) with the risk of non-vertebral fracture (Table 2). The results of sensitivity and subgroup analyses were consistent with those of the overall analysis (Tables 3 and 4). No significant publication bias was detected with regard to the association of SCH or SH with the risk of non-vertebral fracture (Supplementary Figures 38 and 39).
Vertebral fracture
The results indicate that SCH was associated with an increased risk of vertebral fracture in both men and women, with no evidence of heterogeneity across the included studies (Figure 3, Supplementary Figure 40). There was no significant difference in the association of SCH with the risk of vertebral fracture between men and women (RR ratio 0.90; 95% CI 0.32–2.51; P = 0.833; Table 2). The results of the sensitivity and subgroup analyses were consistent with those of the overall analysis (Tables 3 and 4). There was no significant publication bias in terms of the association between SCH and vertebral fracture risk (Supplementary Figure 41).
Discussion
This meta-analysis of 24 cohort studies reporting data on 3,480,682 individuals found no sex-related differences in the association of SCH or SH with the risk of AF, all-cause mortality, cardiac death, CHD, heart failure, MACE, stroke, any fracture, hip fracture, non-vertebral fracture, or vertebral fracture. Our conclusions were stable in sensitivity analyses. Subgroup analyses found that men with SH had an excess risk of MACE when compared with women if the duration of follow-up was ≥10.0 years. Moreover, the risk of any fracture was greater in men with SH than in women with SH when the follow-up duration was <10.0 years and in studies with a high level of adjustment. In studies with a low level of adjustment, the risk of hip fracture was greater in women with SH than in men with SH.
Although no systematic review and meta-analysis has investigated sex differences in the associations of STD with long-term health outcomes, several studies have assessed these associations with stratification according to sex. A meta-analysis by Collet et al. identified six such studies and found that the risk of CHD was higher in women with SH than in men with SH [40]. Baumgartner et al. analyzed data from 11 cohorts and found that women with SH had an excess risk of AF when compared with their male counterparts [41]. Blum et al. performed a meta-analysis of 13 studies and found that the risks of hip fracture, any fracture, non-vertebral fracture, and vertebral fracture were greater in men with SCH than in women with SCH [42]. However, the above studies did not address the heterogeneity across included studies and did not compare the effect estimates between men and women. Therefore, we performed this systematic review and meta-analysis to determine if there are sex differences in the association of STD with MACE and fractures.
There were no significant differences in the association of STD with the risk of AF, all-cause mortality, cardiac death, CHD, heart failure, MACE, or stroke. However, subgroup analysis found that the risk of MACE was greater in men with SH than in women with SH if the duration of follow-up was ≥10.0 years. A potential reason for this finding could be the high prevalence of metabolically unhealthy obese phenotypes in men with SH, regardless of age. However, metabolically unhealthy non-obese and obese phenotypes were found not to be associated with the risk of SH in women [43]. Moreover, the longer follow-up duration allowed us to obtain more cases of MACE, and the power was sufficient to detect a potential sex difference in the associations of SH with the risk of MACE.
Our results indicate no significant sex-related differences in the association of STD with the risk of any fracture, hip fracture, non-vertebral fracture, or vertebral fracture. However, we found an excess risk of any fracture in men with SH when compared with women if the follow-up duration was <10.0 years and in studies with a high level of adjustment. Sex hormone levels are important for skeletal health in younger and middle-aged men. The reduced gonadotropin and sex hormone levels in men with SH may contribute to their observed increase in fracture risk [44]. Interestingly, we found that women with SH had an excess risk of hip fracture in studies with a low level of adjustment, which could be explained by the significant reduction in bone mineral density at the femoral neck in women with SH, which has not been observed in men [45]. However, our stratified analyses based on the level of adjustment could have affect the pooled effect estimates of the sex difference because the characteristics of the study participants could have influenced the risk of fracture.
This study has several shortcomings that should be acknowledged. First, it included both prospective and retrospective cohort studies, and the pooled conclusions could be biased by uncontrolled confounding factors. Second, the TSH level varied during follow-up, which could affect the risk of major cardiovascular outcomes and fractures. Third, the background treatments for STD were not adjusted for in most of the studies, which could have affected the risks of major cardiovascular outcomes and fractures related to STD. Fourth, the results of our analysis are based on both indirect and direct comparisons, and the heterogeneity in characteristics between men and women could have contributed to these risks. Finally, there are some inherent limitations to a meta-analysis based on published articles, including inevitable publication bias and restriction of analyses.
This study found no sex-related differences in the associations of STD with MACE and fractures. However, subgroup analysis indicated potential sex differences in the risks of MACE, any fracture, and hip fracture related to SH, suggesting that individuals at high risk should be carefully monitored. Further large-scale prospective cohort studies should be performed to confirm if there are sex differences in the association of STD with MACE and fractures.
Author Contributions
W-WQ and F-HJ contributed to the conception and design of the study; F-HJ, Z-RS, and C-S collected the references, analyzed the data, and drafted the manuscript. F-HJ generated the figures. W-WQ modified and approved the manuscript. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This work was supported by grants from the Beijing Natural Science Foundation (7182049) and Basic-Clinical Research Cooperation Funding of Capital Medical University (17JL34) and National Key Research and Development Plan-Research (2020YFF0305101).
Editorial Note
This corresponding author has a verified history of publications using a personal email address for correspondence.
References
- 1. Beckett GJ, Toft AD. First-line thyroid function tests -- TSH alone is not enough. Clin Endocrinol (Oxf). 2003; 58:20–1. https://doi.org/10.1046/j.1365-2265.2003.01690.x [PubMed]
- 2. Yang G, Wang Y, Ma A, Wang T. Subclinical thyroid dysfunction is associated with adverse prognosis in heart failure patients with reduced ejection fraction. BMC Cardiovasc Disord. 2019; 19:83. https://doi.org/10.1186/s12872-019-1055-x [PubMed]
- 3. Roberts L, McCahon D, Johnson O, Haque MS, Parle J, Hobbs FR. Stability of thyroid function in older adults: the Birmingham Elderly Thyroid Study. Br J Gen Pract. 2018; 68:e718–26. https://doi.org/10.3399/bjgp18X698861 [PubMed]
- 4. Zhu H, Zhang J, Wang J, Zhao X, Gu M. Association of subclinical thyroid dysfunction with bone mineral density and fracture: a meta-analysis of prospective cohort studies. Endocrine. 2020; 67:685–98. https://doi.org/10.1007/s12020-019-02110-9 [PubMed]
- 5. Wang X, Zhao X, Huang X. Association of Subclinical Thyroid Dysfunction with Chronic Kidney Disease: A Systematic Review and Meta-analysis. Endocr Res. 2020; 45:41–9. https://doi.org/10.1080/07435800.2019.1645164 [PubMed]
- 6. Ning Y, Cheng YJ, Liu LJ, Sara JD, Cao ZY, Zheng WP, Zhang TS, Han HJ, Yang ZY, Zhang Y, Wang FL, Pan RY, Huang JL, et al. What is the association of hypothyroidism with risks of cardiovascular events and mortality? A meta-analysis of 55 cohort studies involving 1,898,314 participants. BMC Med. 2017; 15:21. https://doi.org/10.1186/s12916-017-0777-9 [PubMed]
- 7. Rieben C, Segna D, da Costa BR, Collet TH, Chaker L, Aubert CE, Baumgartner C, Almeida OP, Hogervorst E, Trompet S, Masaki K, Mooijaart SP, Gussekloo J, et al. Subclinical Thyroid Dysfunction and the Risk of Cognitive Decline: a Meta-Analysis of Prospective Cohort Studies. J Clin Endocrinol Metab. 2016; 101:4945–54. https://doi.org/10.1210/jc.2016-2129 [PubMed]
- 8. Chaker L, Baumgartner C, Ikram MA, Dehghan A, Medici M, Visser WE, Hofman A, Rodondi N, Peeters RP, Franco OH. Subclinical thyroid dysfunction and the risk of stroke: a systematic review and meta-analysis. Eur J Epidemiol. 2014; 29:791–800. https://doi.org/10.1007/s10654-014-9946-8 [PubMed]
- 9. Yang LB, Jiang DQ, Qi WB, Zhang T, Feng YL, Gao L, Zhao J. Subclinical hyperthyroidism and the risk of cardiovascular events and all-cause mortality: an updated meta-analysis of cohort studies. Eur J Endocrinol. 2012; 167:75–84. https://doi.org/10.1530/EJE-12-0015 [PubMed]
- 10. Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. 2000; 283:2008–12. https://doi.org/10.1001/jama.283.15.2008 [PubMed]
- 11. Ades AE, Lu G, Higgins JP. The interpretation of random-effects meta-analysis in decision models. Med Decis Making. 2005; 25:646–54. https://doi.org/10.1177/0272989X05282643 [PubMed]
- 12. DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986; 7:177–88. https://doi.org/10.1016/0197-2456(86)90046-2 [PubMed]
- 13. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–60. https://doi.org/10.1136/bmj.327.7414.557 [PubMed]
- 14. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997; 315:629–34. https://doi.org/10.1136/bmj.315.7109.629 [PubMed]
- 15. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50:1088–101. https://doi.org/10.2307/2533446 [PubMed]
- 16. Hak AE, Pols HA, Visser TJ, Drexhage HA, Hofman A, Witteman JC. Subclinical hypothyroidism is an independent risk factor for atherosclerosis and myocardial infarction in elderly women: the Rotterdam Study. Ann Intern Med. 2000; 132:270–8. https://doi.org/10.7326/0003-4819-132-4-200002150-00004 [PubMed]
- 17. Bauer DC, Ettinger B, Nevitt MC, Stone KL, and Study of Osteoporotic Fractures Research Group. Risk for fracture in women with low serum levels of thyroid-stimulating hormone. Ann Intern Med. 2001; 134:561–8. https://doi.org/10.7326/0003-4819-134-7-200104030-00009 [PubMed]
- 18. Parle JV, Maisonneuve P, Sheppard MC, Boyle P, Franklyn JA. Prediction of all-cause and cardiovascular mortality in elderly people from one low serum thyrotropin result: a 10-year cohort study. Lancet. 2001; 358:861–5. https://doi.org/10.1016/S0140-6736(01)06067-6 [PubMed]
- 19. Imaizumi M, Akahoshi M, Ichimaru S, Nakashima E, Hida A, Soda M, Usa T, Ashizawa K, Yokoyama N, Maeda R, Nagataki S, Eguchi K. Risk for ischemic heart disease and all-cause mortality in subclinical hypothyroidism. J Clin Endocrinol Metab. 2004; 89:3365–70. https://doi.org/10.1210/jc.2003-031089 [PubMed]
- 20. Finigan J, Greenfield DM, Blumsohn A, Hannon RA, Peel NF, Jiang G, Eastell R. Risk factors for vertebral and nonvertebral fracture over 10 years: a population-based study in women. J Bone Miner Res. 2008; 23:75–85. https://doi.org/10.1359/jbmr.070814 [PubMed]
- 21. Lee JS, Buzková P, Fink HA, Vu J, Carbone L, Chen Z, Cauley J, Bauer DC, Cappola AR, Robbins J. Subclinical thyroid dysfunction and incident hip fracture in older adults. Arch Intern Med. 2010; 170:1876–83. https://doi.org/10.1001/archinternmed.2010.424 [PubMed]
- 22. Murphy E, Glüer CC, Reid DM, Felsenberg D, Roux C, Eastell R, Williams GR. Thyroid function within the upper normal range is associated with reduced bone mineral density and an increased risk of nonvertebral fractures in healthy euthyroid postmenopausal women. J Clin Endocrinol Metab. 2010; 95:3173–81. https://doi.org/10.1210/jc.2009-2630 [PubMed]
- 23. Nanchen D, Gussekloo J, Westendorp RG, Stott DJ, Jukema JW, Trompet S, Ford I, Welsh P, Sattar N, Macfarlane PW, Mooijaart SP, Rodondi N, de Craen AJ, and PROSPER Group. Subclinical thyroid dysfunction and the risk of heart failure in older persons at high cardiovascular risk. J Clin Endocrinol Metab. 2012; 97:852–61. https://doi.org/10.1210/jc.2011-1978 [PubMed]
- 24. Selmer C, Olesen JB, Hansen ML, Lindhardsen J, Olsen AM, Madsen JC, Faber J, Hansen PR, Pedersen OD, Torp-Pedersen C, Gislason GH. The spectrum of thyroid disease and risk of new onset atrial fibrillation: a large population cohort study. BMJ. 2012; 345:e7895. https://doi.org/10.1136/bmj.e7895 [PubMed]
- 25. Waring AC, Harrison S, Fink HA, Samuels MH, Cawthon PM, Zmuda JM, Orwoll ES, Bauer DC, and Osteoporotic Fractures in Men (MrOS) Study. A prospective study of thyroid function, bone loss, and fractures in older men: The MrOS study. J Bone Miner Res. 2013; 28:472–9. https://doi.org/10.1002/jbmr.1774 [PubMed]
- 26. Svare A, Nilsen TI, Asvold BO, Forsmo S, Schei B, Bjøro T, Langhammer A. Does thyroid function influence fracture risk? Prospective data from the HUNT2 study, Norway. Eur J Endocrinol. 2013; 169:845–52. https://doi.org/10.1530/EJE-13-0546 [PubMed]
- 27. LeGrys VA, Funk MJ, Lorenz CE, Giri A, Jackson RD, Manson JE, Schectman R, Edwards TL, Heiss G, Hartmann KE. Subclinical hypothyroidism and risk for incident myocardial infarction among postmenopausal women. J Clin Endocrinol Metab. 2013; 98:2308–17. https://doi.org/10.1210/jc.2012-4065 [PubMed]
- 28. Abrahamsen B, Jørgensen HL, Laulund AS, Nybo M, Brix TH, Hegedüs L. Low serum thyrotropin level and duration of suppression as a predictor of major osteoporotic fractures-the OPENTHYRO register cohort. J Bone Miner Res. 2014; 29:2040–50. https://doi.org/10.1002/jbmr.2244 [PubMed]
- 29. Kim TH, Choi HS, Bae JC, Moon JH, Kim HK, Choi SH, Lim S, Park DJ, Park KS, Jang HC, Lee MK, Cho NH, Park YJ. Subclinical hypothyroidism in addition to common risk scores for prediction of cardiovascular disease: a 10-year community-based cohort study. Eur J Endocrinol. 2014; 171:649–57. https://doi.org/10.1530/EJE-14-0464 [PubMed]
- 30. Kovar FM, Fang IF, Perkmann T, Haslacher H, Slavka G, Födinger M, Endler G, Wagner OF. Subclinical hypothyroidism and mortality in a large Austrian cohort: a possible impact on treatment? Wien Klin Wochenschr. 2015; 127:924–30. https://doi.org/10.1007/s00508-015-0846-z [PubMed]
- 31. Rhee CM, Kim S, Gillen DL, Oztan T, Wang J, Mehrotra R, Kuttykrishnan S, Nguyen DV, Brunelli SM, Kovesdy CP, Brent GA, Kalantar-Zadeh K. Association of thyroid functional disease with mortality in a national cohort of incident hemodialysis patients. J Clin Endocrinol Metab. 2015; 100:1386–95. https://doi.org/10.1210/jc.2014-4311 [PubMed]
- 32. Journy NMY, Bernier MO, Doody MM, Alexander BH, Linet MS, Kitahara CM. Hyperthyroidism, Hypothyroidism, and Cause-Specific Mortality in a Large Cohort of Women. Thyroid. 2017; 27:1001–10. https://doi.org/10.1089/thy.2017.0063 [PubMed]
- 33. Siru R, Alfonso H, Chubb SAP, Golledge J, Flicker L, Yeap BB. Subclinical thyroid dysfunction and circulating thyroid hormones are not associated with bone turnover markers or incident hip fracture in older men. Clin Endocrinol (Oxf). 2018; 89:93–9. https://doi.org/10.1111/cen.13615 [PubMed]
- 34. Rhee CM, Kalantar-Zadeh K, Ravel V, Streja E, You AS, Brunelli SM, Nguyen DV, Brent GA, Kovesdy CP. Thyroid Status and Death Risk in US Veterans With Chronic Kidney Disease. Mayo Clin Proc. 2018; 93:573–85. https://doi.org/10.1016/j.mayocp.2018.01.024 [PubMed]
- 35. Lin HJ, Lin CC, Lin HM, Chen HJ, Lin CC, Chang CT, Chou CY, Huang CC. Hypothyroidism is associated with all-cause mortality in a national cohort of chronic haemodialysis patients. Nephrology (Carlton). 2018; 23:559–64. https://doi.org/10.1111/nep.13049 [PubMed]
- 36. Thayakaran R, Adderley NJ, Sainsbury C, Torlinska B, Boelaert K, Šumilo D, Price M, Thomas GN, Toulis KA, Nirantharakumar K. Thyroid replacement therapy, thyroid stimulating hormone concentrations, and long term health outcomes in patients with hypothyroidism: longitudinal study. BMJ. 2019; 366:l4892. https://doi.org/10.1136/bmj.l4892 [PubMed]
- 37. Inoue K, Ritz B, Brent GA, Ebrahimi R, Rhee CM, Leung AM. Association of Subclinical Hypothyroidism and Cardiovascular Disease With Mortality. JAMA Netw Open. 2020; 3:e1920745. https://doi.org/10.1001/jamanetworkopen.2019.20745 [PubMed]
- 38. Kim HJ, Kang T, Kang MJ, Ahn HS, Sohn SY. Incidence and Mortality of Myocardial Infarction and Stroke in Patients with Hyperthyroidism: A Nationwide Cohort Study in Korea. Thyroid. 2020; 30:955–65. https://doi.org/10.1089/thy.2019.0543 [PubMed]
- 39. Svensson J, Ohlsson C, Karlsson MK, Lorentzon M, Lewerin C, Mellström D. Subclinical hyperthyroidism is associated with increased risk of vertebral fractures in older men. Osteoporos Int. 2021; 32:2257–65. https://doi.org/10.1007/s00198-021-05964-w [PubMed]
- 40. Collet TH, Bauer DC, Cappola AR, Asvold BO, Weiler S, Vittinghoff E, Gussekloo J, Bremner A, den Elzen WP, Maciel RM, Vanderpump MP, Cornuz J, Dörr M, et al, and Thyroid Studies Collaboration. Thyroid antibody status, subclinical hypothyroidism, and the risk of coronary heart disease: an individual participant data analysis. J Clin Endocrinol Metab. 2014; 99:3353–62. https://doi.org/10.1210/jc.2014-1250 [PubMed]
- 41. Baumgartner C, da Costa BR, Collet TH, Feller M, Floriani C, Bauer DC, Cappola AR, Heckbert SR, Ceresini G, Gussekloo J, den Elzen WPJ, Peeters RP, Luben R, et al, and Thyroid Studies Collaboration. Thyroid Function Within the Normal Range, Subclinical Hypothyroidism, and the Risk of Atrial Fibrillation. Circulation. 2017; 136:2100–16. https://doi.org/10.1161/CIRCULATIONAHA.117.028753 [PubMed]
- 42. Blum MR, Bauer DC, Collet TH, Fink HA, Cappola AR, da Costa BR, Wirth CD, Peeters RP, Åsvold BO, den Elzen WP, Luben RN, Imaizumi M, Bremner AP, et al, and Thyroid Studies Collaboration. Subclinical thyroid dysfunction and fracture risk: a meta-analysis. JAMA. 2015; 313:2055–65. https://doi.org/10.1001/jama.2015.5161 [PubMed]
- 43. Wang Y, Lin H, Li Q, Guan L, Zhao M, Zhong F, Liu J, Yuan Z, Guo H, Song Y, Gao L, Zhao J. Association between different obesity phenotypes and hypothyroidism: a study based on a longitudinal health management cohort. Endocrine. 2021; 72:688–98. https://doi.org/10.1007/s12020-021-02677-2 [PubMed]
- 44. Abrahamsen B, Jørgensen HL, Laulund AS, Nybo M, Bauer DC, Brix TH, Hegedüs L. The excess risk of major osteoporotic fractures in hypothyroidism is driven by cumulative hyperthyroid as opposed to hypothyroid time: an observational register-based time-resolved cohort analysis. J Bone Miner Res. 2015; 30:898–905. https://doi.org/10.1002/jbmr.2416 [PubMed]
- 45. Xu N, Wang Y, Xu Y, Li L, Chen J, Mai X, Xu J, Zhang Z, Yang R, Sun J, Chen H, Chen R. Effect of subclinical hyperthyroidism on osteoporosis: A meta-analysis of cohort studies. Endocrine. 2020; 69:39–48. https://doi.org/10.1007/s12020-020-02259-8 [PubMed]



