Introduction
Duchenne Muscular Dystrophy (DMD) is caused by mutations of the dystrophin gene, which leads to progressive muscle fiber damage and degeneration, resulting in cardiac or respiratory failure and ultimately premature death [1, 2]. DMD patients develop severe chronic inflammation in their muscles, which is characterized by the massive infiltration of immune cells into muscles and elevated activation of pro-inflammatory signaling in muscle cells [1, 2]. Our previous studies of mdx/Utro(−/−) mice (a severe murine model for DMD; dystrophin−/−; utrophin−/−) revealed the greatly increased number of SA-β-gal (Senescence-associated β-Galactosidase) + senescent cells in the dystrophic muscles, which is closely associated with severe dystrophic phenotypes [3, 4]. Our recent study further revealed that, macrophages in dystrophic muscles have aberrant activation of RhoA signaling, which is associated with upregulated expression of SASP (Senescence-associated secretary phenotypes) factors and senescence-like features of macrophages [3]. Therefore, it would be interesting to find out whether the repression or elimination of these senescence-like macrophages can be effective in recuing dystrophic muscle phenotypes.
One recent important progression in the studying of ageing mechanism has been the observation that the ageing immune system, or immunosenescence, has a causal role in driving systemic ageing and therefore represents a key therapeutic target to delay ageing [5]. It was verified that the senolytic ablation of senescent immune cells in the aged organism can effectively delay the systemic aging [5]. In normal conditions, senescent cells accumulated in body are specifically identified and removed by immune system [6, 7]; however, when the immune cell become senescent too, their ability to clear senescent cells would be greatly compromised, leading to excessive accumulation of senescent cells and aging-related disorders [5–7]. Therefore, potential clearance or repression of senescent immune cells would be greatly beneficial for body health.
Senescent cells accumulate with age and are considered to be greatly related to age-related disorders [5, 8]. Senescence is a cell fate elicited in response to external and internal cellular stress signals, which causes extensive changes in gene expression, histone modifications, organelle function, elevated protein production, and profound morphologic and metabolic shifts [9]. Senescent cells usually actively secret SASP factors, which act on the non-senescent normal cells and interfere with their normal function [10–12]. In recent years, elimination of senescent cells with senolytic drugs (senolytics) have arisen as a novel anti-aging strategy and therapeutic approach for treating many age-related diseases [13–20]. Senolytics are natural or synthesized compounds, including dasatinib and quercetin (D+Q), navitoclax (ABT263), 17-DMAG, and fisetin [13–16, 21]. Senolytics can selectively eliminate senescent cells by targeting anti-apoptotic signaling pathways that are overtly upregulated in senescent cells, such as BCL2 and BCL-XL [13–16, 21].
Therefore, in the current study, we investigated whether the treatment of utrophin−/−mdx mice with senolytic drug (fisetin) can be effective in eliminating the senescent immune cells and improve muscle phenotypes. We also performed in vitro treatment of cell co-culture system of immune cells and muscle stem cells to verify our observation in vivo. Our results will help verify the potential causal role of senescent immune cells in leading to severe muscle dystrophy and present a novel strategy for therapeutic treatment of DMD disease with senolytic drugs.
Results
Skeletal muscle of mdx/utro(−/−) mice develops higher level of muscle stem cell exhaustion, immune cell infiltration, and cellular senescence
Gastrocnemius muscles from hindlimbs were collected from 8-week old WT and mdx/utro(−/−) mice for histological analyses (Figure 1). Immunofluorescent staining of Pax7 and collagen type IV in muscle tissues revealed that, there is significantly reduced number of Pax7+ muscle stem cells in mdx/utro(−/−) muscles (Figure 1A); while in contrast, immunofluorescent staining of CD68 showed that, the number of CD68+ macrophages is greatly increased in mdx/utro(−/−) muscles (Figure 1B). Meanwhile, SA-β-Gal (senescence-associated β-Galactosidase, a marker for cellular senescence) staining further revealed that, there is obviously higher number of SA-β-Gal+ senescent cells in mdx/utro(−/−) muscles (Figure 1C).
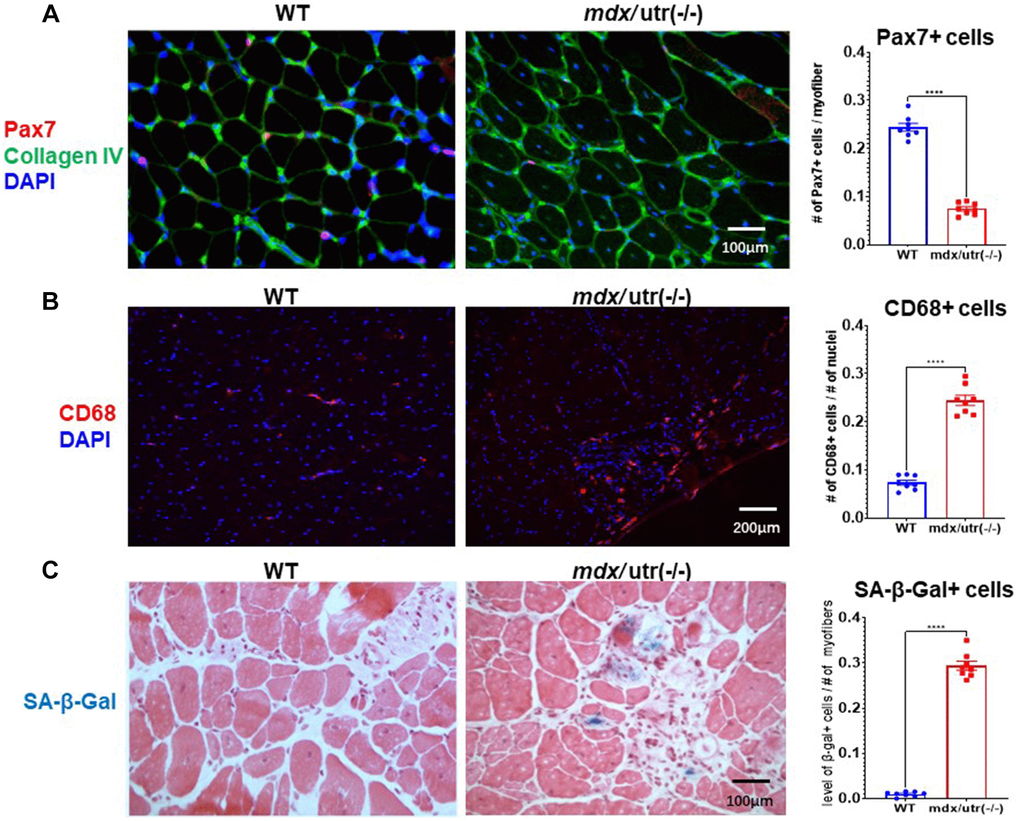
Figure 1. mdx/utro(−/−) muscle exhibits obvious stem cell exhaustion, immune cell infiltration and cellular senescence. (A) Immunofluorescent staining of Pax7 and Collagen type IV in muscle tissues of WT and mdx/utro(−/−) mice. (B) Immunofluorescent staining of CD68 in muscle tissues of WT and mdx/utro(−/−) mice. (C) Staining of SA-β-Gal in muscle tissues of WT and mdx/utro(−/−) mice. n = 8 mice/group for statistics, and data are presented as mean +/− SD.
CD68+ macrophages in mdx/Utro(−/−) muscle features higher level of mTORC1 activation
Our previous study of mdx/utro(−/−) mice revealed that, CD68+ macrophages in dystrophic muscle develop senescence-like phenotypes, including higher level of positive C12FDG (a fluorogenic substrate for β-galactosidase) signaling [3]. mTORC1 activation has been known as the central regulator of cellular senescence, which occurs in senescent human cells independent of growth factor signaling [22, 23]. In order to further verify whether CD68+ macrophages in mdx/utro(−/−) muscle may develop higher level of cellular senescence, co-immunostaining of CD68 and p-4E-BP1 (a substrate for mTORC1 signaling) in muscle tissues was performed. Results showed that, CD68+ macrophages in mdx/utro(−/−) muscle express higher amount of p-4E-BP1 protein (Figure 2A, 2C), indicating a high level of mTOR activation and senescence.
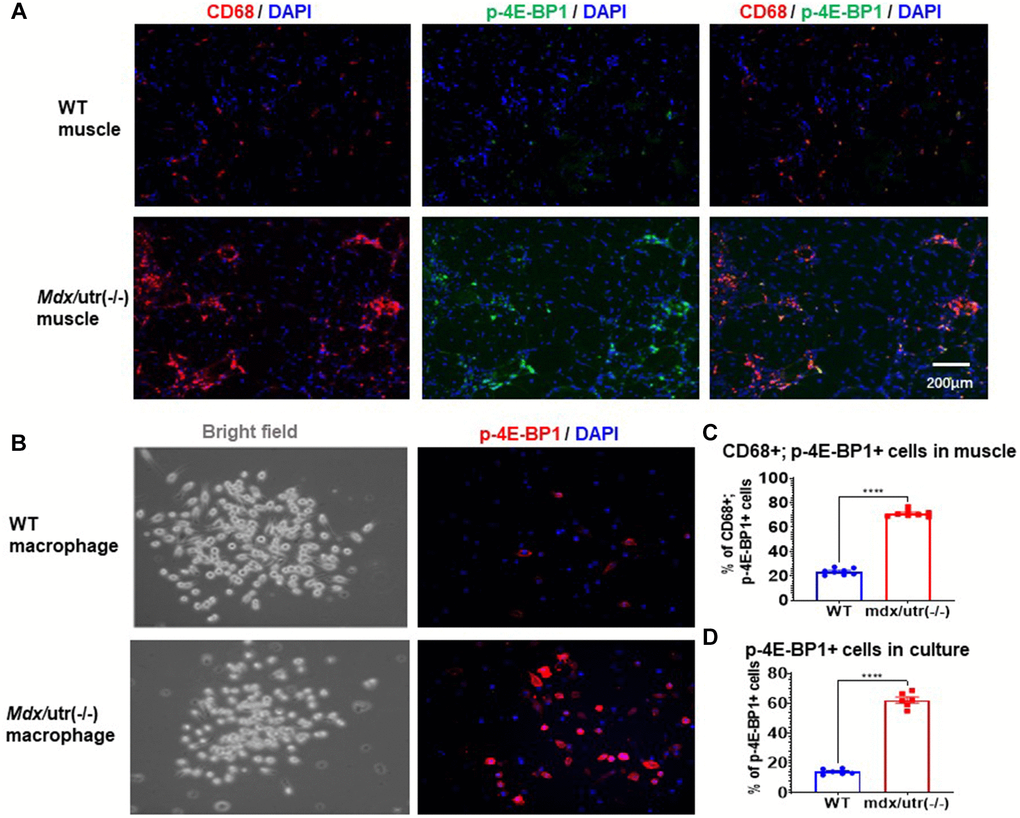
Figure 2. Macrophages in mdx/utro(−/−) muscle exhibits higher level of mTOR activation. (A) Immunofluorescent staining of CD68 and p-4E-BP1 in muscle tissues of WT and mdx/utro(−/−) mice. (B) Immunofluorescent staining of p-4E-BP1 in macrophages isolated from muscle tissues of WT and mdx/utro(−/−) mice. (C) Statistics of results in vivo. n = 8 mice/group. Data are presented as mean +/− SD. (D) Statistics of result in vitro, n = 6 (3 cell lineages x 2 replicates/group). Data are presented as mean +/− SD.
Also, CD68+ macrophages were isolated from WT and mdx/utro(−/−) muscles via flow cytometry based on CD68 expression, to compare their expression of p-4E-BP1 protein, and we observed significantly higher percentage of p-4E-BP1+ cells in CD68+ macrophages from mdx/utro(−/−) muscle (Figure 2B, 2D).
CD68+ macrophages from mdx/Utro(−/−) muscle features elevated expression of SASP factors and increased cellular senescence
The higher activation of mTOR signaling in mdx/utro(−/−) macrophage may suggest a potential development of cellular senescence. Thus, we then compared the expression of SASP factors in CD68+ macrophages isolated from WT and mdx/utro(−/−) muscles. RT-PCR result showed that, CD68+ macrophages from mdx/utro(−/−) muscle displayed up-regulated expression of SASP factors (i.e., TNF-α, IL-1α, IL-1β, IL-6, CXCL1, MCP1, and IFN-γ) and p16, and down-regulated expression of anti-inflammation factor IL10 (Figure 3A). Meanwhile, the fluorescent staining of SA-β-Gal revealed that there was higher percentage of SA-β-Gal+ cells in mdx/utro(−/−) macrophages, compared with WT macrophages (Figure 3B, 3C).
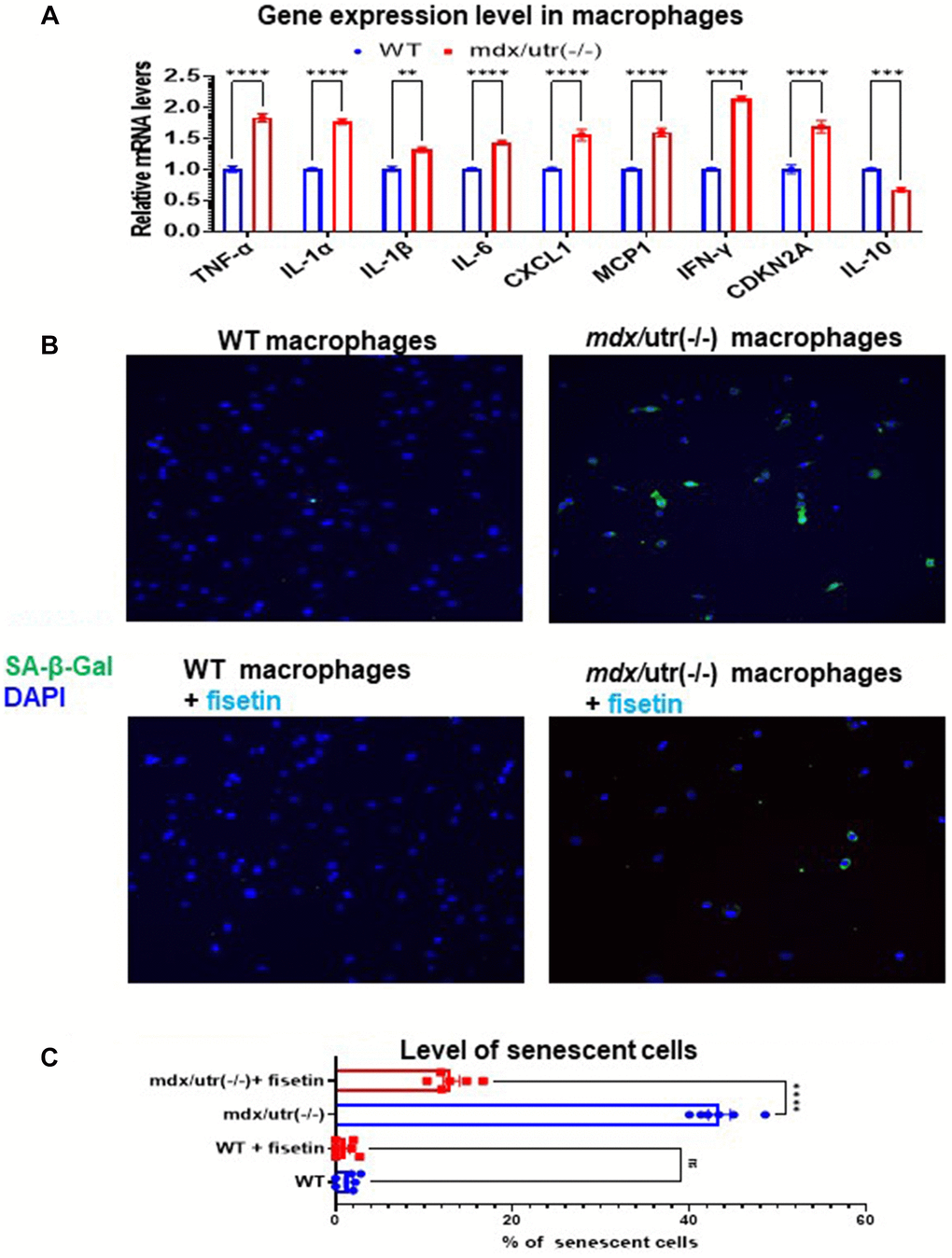
Figure 3. Fisetin treatment specifically remove senescent macrophages from mdx/utro(−/−) mouse. (A) Real time-PCR result of gene expression level of SASP factors, p16 and IL-10 in WT and mdx/utro(−/−) macrophages (n = 3 cell lineages). (B) Staining of SA-β-Gal in 4 groups of macrophages with fluorescent Senescence Assay Kit. Fisetin was applied to treat the WT and mdx/utro(−/−) macrophages for 48 hr at 20 μM of concentration. (C) Statistics of SA-β-Gal+ cells in 4 groups of cells. n = 6 (3 cell lineages x 2 replicates/group). Data are presented as mean +/− SD.
Further, we examined whether the senolytic drug fisetin may specifically remove SA-β-Gal+ senescent cells in mdx/utro(−/−) macrophages. Fisetin was applied to treat the WT and mdx/utro(−/−) macrophages at 20 μM of concentration for 48 hr. Result showed that, the percentage of SA-β-Gal+ senescent cells in mdx/utro(−/−) macrophages was obviously reduced after fisetin treatment (Figure 3B, 3C).
CD68+ macrophages isolated from mdx/utro(−/−) muscle negatively impact the function of muscle stem cell in a cell co-culture system
A cell coculture system was setup, to verify the potential direct impact of CD68+ macrophages in mdx/utro(−/−) muscle may exert on muscle progenitor/stem cells (MPCs). FAPs and MPCs were isolated from muscles of WT and mdx/utro(−/−) mice, and CD68+ macrophages from WT or mdx/utro(−/−) muscles and WT MPCs were co-cultured in a transwell system, with CD68+ macrophages from WT or mdx/utro(−/−) being at the upper chamber and WT MPCs being at lower chamber. Cell myogenesis and proliferation assays of WT MPCs showed that, the WT MPCs co-cultured with CD68+ macrophages from mdx/utro(−/−) developed impaired myogenesis and proliferation potentials (Figure 4).
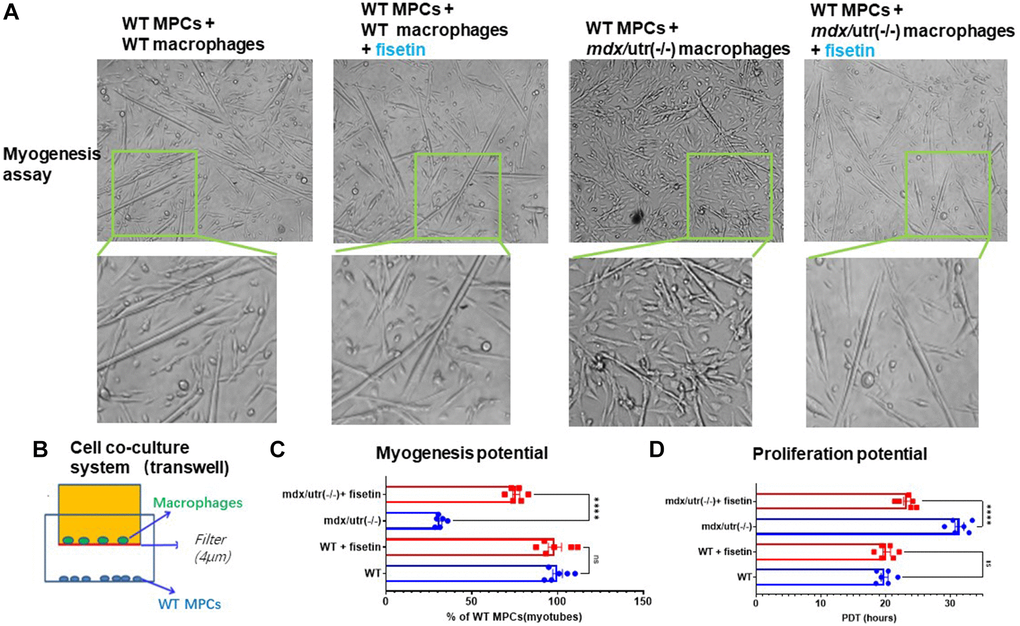
Figure 4. Macrophages from mdx/utro(−/−) muscle impair the function of co-cultured MPCs, which can be rescued with senolytic drug. (A) The progression of myotube formation was tracked in the myogenesis assay of WT MPCs co-cultured with WT or mdx/utro(−/−) macrophages, with or without fisetin treatment (20 μM, 96 h). (B) Schematic demonstration of the cell co-culture system of WT or mdx/utro(−/−) macrophages (upper chamber) and WT MPCs (lower chamber). (C) Statistics of result for myogenesis assay. (D) Statistics of result for proliferation assay. n = 6 (3 cell lineages x 2 replicates/group). Data are presented as mean +/− SD.
Treatment of co-culture system of mdx/utro(−/−) macrophages and WT MPCs with fisetin rescues the impaired function of WT MPCs
We then examined whether the removal of senescent cells in mdx/utro(−/−) macrophages with senolytic drug may rescue the function of muscle stem cells in the cell co-culture system. Fisetin was applied to treat the cell co-culture system of mdx/utro(−/−) macrophages and WT MPCs for 48 hr at 20 μM of concentration, and we observed improved myogenesis and proliferation potential of WT MPCs, compared to non-treated controls (Figure 4).
Treatment of mdx/utro(−/−) mice with fisetin results in increased number of muscle stem cells and improved muscle phenotypes
Finally, in order to investigate the efficacy of senolytics on muscle health in vivo, mdx/utro(−/−) mice were treated with fisetin via oral gavage (20 mg/kg for 5 consecutive days each week)) for 4 weeks. Immunostaining of muscle cryosections revealed that, compared to non-treated mice, the number of CD68+ macrophages and SA-β-Gal+ senescent cells was decreased and the number of Pax7+ MPCs was elevated in fisetin treated mice, while muscle phenotypes were also improved by observation of reduced fibrosis formation (collagen deposition) (Figure 5).
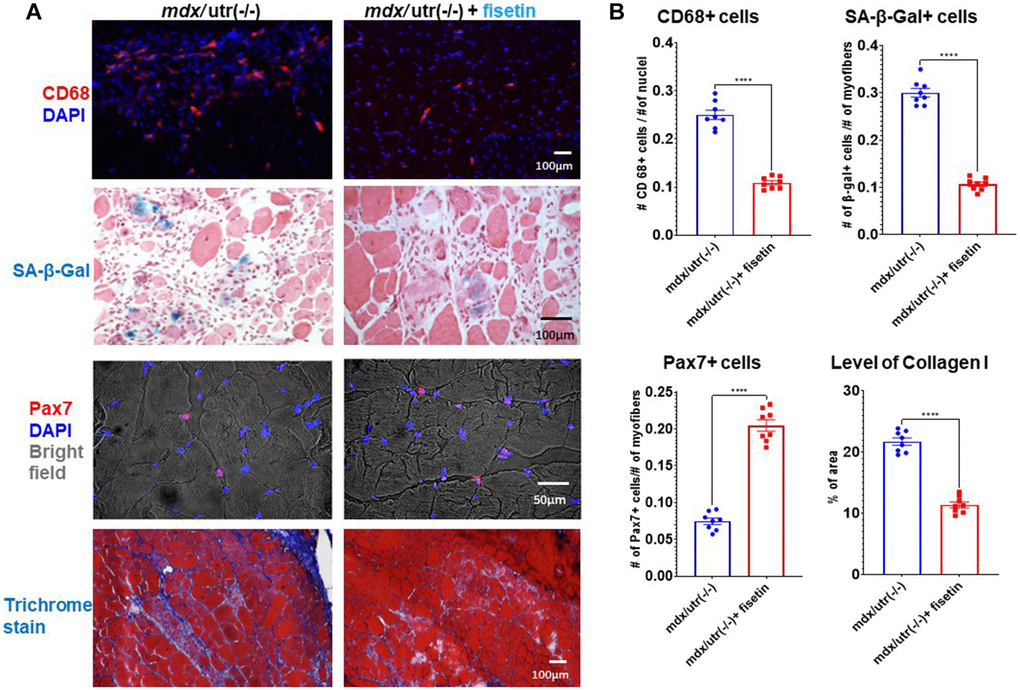
Figure 5. In vivo results of fisetin administration in mdx/utro(−/−) mice. (A) mdx/utro(−/−) mice were treated with fisetin via oral gavage (20 mg/kg for 5 consecutive days each week) for 4 weeks and muscle tissues were analyzed. Immunofluorescent staining of CD68 showed that the number of macrophages was decreased with fisetin administration; staining of SA-β-Gal showed that the number of senescent cells was decreased with fisetin administration; immunofluorescent staining of Pax7 showed that the number of MPCs was increased with fisetin administration; trichrome staining of collage deposition (fibrosis formation) showed that the amount of fibrosis formation was decreased with fisetin administration. (B) Statistics of in vivo results. n = 8 mice/group. Data are presented as mean +/− SD.
Discussion
Cellular senescence is a defined hallmark of aging, contributing to the aging process and age-related diseases and disorders. Senotherapeutics, including senolytics and senomorphics, has emerged as a promising anti-aging strategy, and was found to be able to prolong healthspan and lifespan of animals [12]. In recent years, senolytics have been widely studied in various types of tissues and disease models and have been proven for their beneficial effect in delaying aging or treating age-related diseases [11–14]. Interestingly, our recent study of mdx/utr(−/−) mice revealed that, macrophages are highly prevalent in severely dystrophic muscle of mdx/utr(−/−) mice and display senescence-like phenotypes, including the expression of SASP factors and positive staining for C12FDG [3]. However, the potential effect and mechanism of senolytics in regulating the function of MPCs in severely dystrophic muscles remains largely unclear. It was recently discovered that the ageing immune system, or immunosenescence, has a causal role in driving systemic ageing and therefore represents a key therapeutic target to delay ageing [5]. Therefore, here we investigated whether senolytic ablation of senescent immune cells in dystrophic muscle of mdx/utr(−/−) mice may promote the restoration of exhausted muscle stem cell pool and improve muscle phenotypes. Our current results show that, a substantial fraction of mdx/utr(−/−) macrophages develop senescence in the severely dystrophic muscles, which dominantly repress the proper function of MPCs by releasing SASPs; while the elimination of senescent macrophages with fisetin is able to rescue the function of MPCs.
Senescent cells are known as a source of age-related chronic inflammation via secreting multiple pro-inflammatory factors of SASPs [9, 10]. As senescent cells accumulate in the aged tissues, they can exert profound effects on the growth and function of normal cells by releasing SASP factors including exosomes [18, 20]. Senescent cells play a critical role in inducing or mediating various types of aging-related diseases [16–20]. A reduction in the senescent cell burden can be achieved with several different classes of senolytics including the combination of dasatinib and quercetin (D+Q), navitoclax (ABT263) and related Bcl-2 family member inhibitors, HSP90 inhibitors such as 17-DMAG and the flavonoid fisetin [16, 22–27]. Fisetin is a naturally occurring anti-oxidant that scavenges free radicals as well as upregulates synthesis of glutathione, and it also has been reported to inhibit apoptosis-related signaling BCL-XL [26], which may also be important for its senolytic activity. The positive effect of fisetin in removing senescent cell in aged tissues has been well-demonstrated, our current results have further demonstrated that fisetin is effective in removing senescent immune cells in dystrophic muscles.
Senescent macrophages are in fact also found to express senescence-related markers p16(Ink4a) and β-galactosidase (β-gal), and promote inflammation in diseased tissues [25, 26]. Our previous work has indicated increased cellular senescence in dystrophic muscles of mdx/utr(−/−) mice [3], however, whether or not macrophages in particular develop cellular senescence and promote senescence associated phenotypes was still unknown. To this end, here we further examined mdx/utr(−/−) mice and solved these puzzles.
Immune cells in the skeletal muscle are activated during muscle injury and promote the process of muscle regeneration by coordinating with muscle stem cells. However, studies with severely diseased muscles further demonstrate that immune cells can become dominantly activated and is inductive of increased fatty infiltration and fibrosis formation, while at the same time potently repress the proliferation and function of muscle stem cells [27]. Our current results in severely dystrophic muscle reveal a similar situation of interaction between macrophages and MPCs, showing that the function of MPCs is repressed by the senescent macrophages. As senescent cells accumulate in the aged or diseased tissues, it can exert profound effects on the growth and function of normal cells by releasing SASPs [9, 10]. Fisetin is a naturally-occurring flavone with low toxicity, a selective BCL-XL inhibitor [15], and our current results demonstrate the effective rescuing of muscle stem cell function and dystrophic muscle phenotype with fisetin administration. Because senescent cell usually takes weeks to reaccumulate, intermit administration of senolytics is efficient to achieve ideal results. Here in our study, it shows the weekly administration of fisetin in mice is effective in eliminating many of senescent cells and rescue the growth and function of MPCs.
Still, there is some limitation in our current study, such as the lack of results for other types of senolytic drugs than fisetin in potentially having better senolytic effect and improving severely dystrophic muscle, and results for other types of immune cells than senescent macrophage in potentially impacting the function of muscle stem cells, which can be further studied in our future researches. Also, here we have studied cells from a murine model of DMD, and the potential effect of fisetin in dystrophic muscle cells from human patients is to be further verified.
Therefore, our current results reveal that the senescence of macrophages play an important role in negatively regulating MPC function in severely dystrophic muscles of murine model of DMD, and the elimination of senescent macrophages with senolytics is effective in rescuing MPC function. Our results demonstrate that senescent macrophages may potentially serve as a new therapeutic target for treatment of muscular dystrophy or delaying the aging of skeletal muscle tissues.
Materials and Methods
Animal models
WT (C57BL/10J, male) mice were obtained from the Jackson Laboratory (Bar Harbor, ME, USA). Mdx/utr(−/−) mice (dystrophin/utrophin double knock out, dKO, or dystrophin−/−:utrophin−/−; male mice) were derived from our in-house colony. Mice were housed in groups of 4 on a 12:12-hour light-dark cycle at 20–23°C. At least 8 mice were used in each experimental sample group. All procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Pittsburgh (IACUC-1109718) and the University of Texas Health Science Center at Houston (AWC-18-0068). All mice were housed and maintained in the Center for Laboratory Animal Medicine and Care (CLAMC) at UTHealth (University of Texas Health Science Center at Houston) in accordance with established guidelines and protocols approved by the UTHealth Animal Welfare Committee.
Isolation of macrophages from skeletal muscles
Gastrocnemius (GM) muscle tissues were harvested from WT and mdx/utr(−/−) mice (8-week old, n = 3 for each group), and digested by serial 1-hr incubations at 37°C in 0.2% type XI collagenase (Sigma-Aldrich, Burlington, MA, USA), dispase (grade II, 240 units; Sigma-Aldrich), and 0.1% trypsin (Thermo Fisher Scientific, Waltham, MA, USA), as previously described [6]. Primary cells from muscle tissues were harvested after digestion, and macrophages were then isolated from these primary cells by Fluorescence activated cell sorting (FACS) of CD68+ cells with a cell sorter (BD FACSAria, San Jose, CA, USA). Macrophages were cultured and immunostaining with CD68 antibody was performed to validate the identity of the isolated cells to be macrophages; the primary muscle cells were also immunostained with CD68 antibody to serve as negative control.
SA-β-gal staining for senescent cells
The presence of senescent cells in skeletal muscle tissue [n = 8 for WT or mdx/utr(−/−) mice] and cells [n = 3 for WT or mdx/utr(−/−) cells, with 2 replicates for each cell population] cultured in vitro was individually measured using the regular senescence-associated β-Galactosidase (SA-β-gal) Staining Kit (Cell Signaling Technology, Danvers, MA, USA) and CellEvent Senescence Green Detection Kit (fluorescent) (Thermo Fisher Scientific), following the manufacturers’ protocol. The number of cells positive for β-gal activity at pH 6, a known characteristic of senescent cells, was determined.
Cell co-culture system of FAPs and MPCs
MPCs from WT muscle (n = 3, with 2 replicates for each cell population) were seeded in the lower chamber of a transwell (Thermo Fisher Scientific), with macrophage from WT or mdx/utr(−/−) mice (n = 3, with 2 replicates for each cell population) being seeded in the upper chamber. The pore size of the transwell membrane was 0.4 μm, and coated with a layer of Matrigel (20% of Matrigel in water, 0.2cm of thickness).
Myogenesis and proliferation assays
For myogenic differentiation assay, WT MPCs (n = 3, with 2 replicates for each cell population) were seeded as ~90% confluence in the lower chamber of transwell, with macrophages from WT and mdx/utr(−/−) mice (n = 3, with 2 replicates for each cell population) being co-cultured in the upper chamber (8000 cells/cm2); cells were co-cultured in the DMEM medium containing 2% HS (horse serum), and allowed for differentiation for 96 h with or without fisetin (Selleckchem, Houston, TX, USA), before being fixed and imaged to compare the number of myotubes formed by WT MPCs in different groups (WT MPCs + WT macrophages, WT MPCs + WT macrophages + fisetin, WT MPCs + mdx/utr(−/−) macrophages, and WT MPCs + mdx/utr(−/−) macrophages + fisetin). The concentration of fisetin used for cells was 20 μM, which was based upon previous studies that reported its senolytic effect [16, 24]. For proliferation assay, WT MPCs were seeded as 2000 cells/cm2 in the lower chamber of transwell, with macrophages being co-cultured in the upper chamber (5000 cells/cm2); cells were co-cultured in the DMEM medium containing 20% FBS and 1% CEE (Chicken Embryo Extracts) and allowed for proliferation for 72 h with or without fisetin (20 μM) before being observed to compare the number of WT MPCs in different groups and calculate cell population doubling time (PDT).
Fisetin treatment of cell co-culture system and mice
For in vitro cell treatment, fisetin (Selleckchem) was applied to treat the cell co-culture system of mdx/utr(−/−) macrophages and WT MPCs (n = 3, with 2 replicates for each cell population) at 20 μM of concentration, and the proliferation and myogenesis potential of WT MPCs was then compared to non-treated controls. For in vivo treatment of mdx/utr(−/−) mice [n = 8 for WT or mdx/utr(−/−) mice] , fisetin was dissolved in vehicle (10% ethanol, 30% polyethylene glycol, 60% phosal 50 pg). Mice were weighed and given fisetin (20 mg/kg for 5 consecutive days each week) or vehicle control alone by oral gavage for 4 weeks.
mRNA analysis via reverse transcriptase-PCR
Total RNA was obtained from MPCs or the skeletal muscles of mice (n = 8) using the RNeasy Mini Kit (Qiagen, Inc., Valencia, CA, USA) according to the manufacturer’s instructions. Reverse transcription was performed using an iScript cDNA Synthesis Kit (Bio-Rad Laboratories, Inc., Hercules, CA, USA). The sequences of primers are given in Table 1 for SASP genes (CXCL2, MCP1, IL-1α, IL-1β, IL-6 and IFN-γ), p16, IL-10, and GAPDH (glyceraldehyde 3-phosphate dehydrogenase). Real-Time PCR reactions were performed using an iCycler thermal cycler (Bio-Rad Laboratories, Inc.). The cycling parameters used for all primers were as follows: 95°C for 10 minutes; PCR, 40 cycles of 30 seconds at 95°C for denaturation, 1 minute at 54°C for annealing, and 30 seconds at 72°C for extension. All data were normalized to the expression of GAPDH.
Table 1. RT-PCR primer sequences.
| Gene | Primer sequence |
| GAPDH | Forward: TCCATGACAACTTTGGCATTG |
| Reverse: TCACGCCACAGCTTTCCA | |
| TNF-α | Forward: CCTGTAGCCCACGTCGTAG |
| Reverse: GGGAGTAGACAAGGTACAACCC | |
| IL-1α | Forward: TCTCAGATTCACAACTGTTCGTG |
| Reverse: AGAAAATGAGGTCGGTCTCACTA | |
| IL-1β | Forward: GCAACTGTTCCTGAACTCAACT |
| Reverse: ATCTTTTGGGGTCCGTCAACT | |
| CXCL1 | Forward: CTGGGATTCACCTCAAGAACATC |
| Reverse: CAGGGTCAAGGCAAGCCTC | |
| MCP1 | Forward: TAAAAACCTGGATCGGAACCAAA |
| Reverse: GCATTAGCTTCAGATTTACGGGT | |
| IFN-γ | Forward: CAGCAACAGCAAGGCGAAAAAGG |
| Reverse: TTTCCGCTTCCTGAGGCTGGAT | |
| CDKN2A | Forward: CGCAGGTTCTTGGTCACTGT |
| Reverse: TGTTCACGAAAGCCAGAGCG | |
| IL-10 | Forward: ATTTGAATTCCCTGGGTGAGAAG |
| Reverse: CACAGGGGAGAAATCGATGACA |
Trichrome staining
Fibrosis formation in muscle tissues of WT or mdx/utr(−/−) mice (n = 8) was visualized by Masson trichrome staining with the Trichrome Stain (Masson) Kit (Sigma-Aldrich). Sections were incubated in Weigert’s iron hematoxylin working solution for 10 min and rinsed under running water for 10 min. Slides were transferred to Biebrich scarlet-acid fuchsin solution for 15 min before incubation in aniline blue solution for another 5 min. Slides were then rinsed, dehydrated, and mounted as earlier. The ratio of the area of fibrotic collagen (blue) to the area of normal muscle (red) was quantified to measure fibrosis formation.
Immunofluorescent staining and imaging
Cultured cells (n = 3, with 2 replicates for each cell population) were fixed with 4% paraformaldehyde, and frozen muscle tissue sections were fixed with 10% formalin. The primary antibodies used – CD68 (Abcam, Boston, MA, USA), p-4E-BP1(Cell Signaling), Pax7 (DSHB, Iowa City, IA, USA), Collagen IV (Abcam), and fMHC (fast-type myosin heavy chain, R&D Systems, Minneapolis, MN, USA) – were all applied at a 1:100 to 1:300 dilution. Cell nucleus was stained with DNA binding reagent and 4′,6-diamidino-2-phenylindole (DAPI). Immunofluorescent images of myofibers were imaged and photographed with a Nikon high resolution microscope.
Measurements of results and statistical analysis
Image analysis was performed using ZEN 2.3 imaging software (ZEISS Microscopy, Germany) and Image J software (version 1.32j; National Institutes of Health, Bethesda, MD, USA). Data from at least six samples from each subject were pooled for statistical analysis. Results are given as the mean ± SEMx. The statistical significance of any difference was calculated using Student’s t-test or two-way ANOVA test. P values < 0.05 were considered statistically significant.
Author Contributions
PDR, JH, ZW and XM performed literature search, study design, and paper writing; LL, XY, ZS, WSH, QF, and YC performed experiments and data collection; LL, WSH, YC and XM performed data analysis.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
Procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Texas Health Science Center at Houston (AWC-18-0068). Mice were housed and maintained in the Center for Laboratory Animal Medicine and Care (CLAMC) in accordance with established guidelines and protocols approved by the UTHealth Animal Welfare Committee.
Funding
This study was supported by research funding from Shandong First Medical University and Shandong Academy of Medical Science to XM and ZW. This work was also supported by grant P01 AG062412 from the National Institutes of Health to PDR. We greatly appreciate the supports from the University of Texas Health Science Center at Houston.
References
- 1. Cruz-Guzmán Odel R, Rodríguez-Cruz M, Escobar Cedillo RE. Systemic Inflammation in Duchenne Muscular Dystrophy: Association with Muscle Function and Nutritional Status. Biomed Res Int. 2015; 2015:891972. https://doi.org/10.1155/2015/891972 [PubMed]
- 2. Rosenberg AS, Puig M, Nagaraju K, Hoffman EP, Villalta SA, Rao VA, Wakefield LM, Woodcock J. Immune-mediated pathology in Duchenne muscular dystrophy. Sci Transl Med. 2015; 7:299rv4. https://doi.org/10.1126/scitranslmed.aaa7322 [PubMed]
- 3. Mu X, Lin CY, Hambright WS, Tang Y, Ravuri S, Lu A, Matre P, Chen W, Gao X, Cui Y, Zhong L, Wang B, Huard J. Aberrant RhoA activation in macrophages increases senescence-associated secretory phenotypes and ectopic calcification in muscular dystrophic mice. Aging (Albany NY). 2020; 12:24853–71. https://doi.org/10.18632/aging.202413 [PubMed]
- 4. Mu X, Tang Y, Takayama K, Chen W, Lu A, Wang B, Weiss K, Huard J. RhoA/ROCK inhibition improves the beneficial effects of glucocorticoid treatment in dystrophic muscle: implications for stem cell depletion. Hum Mol Genet. 2017; 26:2813–24. https://doi.org/10.1093/hmg/ddx117 [PubMed]
- 5. Yousefzadeh MJ, Flores RR, Zhu Y, Schmiechen ZC, Brooks RW, Trussoni CE, Cui Y, Angelini L, Lee KA, McGowan SJ, Burrack AL, Wang D, Dong Q, et al. An aged immune system drives senescence and ageing of solid organs. Nature. 2021; 594:100–5. https://doi.org/10.1038/s41586-021-03547-7 [PubMed]
- 6. Prata LGP, Ovsyannikova IG, Tchkonia T, Kirkland JL. Senescent cell clearance by the immune system: Emerging therapeutic opportunities. Semin Immunol. 2018; 40:101275. https://doi.org/10.1016/j.smim.2019.04.003 [PubMed]
- 7. Song P, An J, Zou MH. Immune Clearance of Senescent Cells to Combat Ageing and Chronic Diseases. Cells. 2020; 9:671. https://doi.org/10.3390/cells9030671 [PubMed]
- 8. Childs BG, Durik M, Baker DJ, van Deursen JM. Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med. 2015; 21:1424–35. https://doi.org/10.1038/nm.4000 [PubMed]
- 9. Robbins PD, Jurk D, Khosla S, Kirkland JL, LeBrasseur NK, Miller JD, Passos JF, Pignolo RJ, Tchkonia T, Niedernhofer LJ. Senolytic Drugs: Reducing Senescent Cell Viability to Extend Health Span. Annu Rev Pharmacol Toxicol. 2021; 61:779–803. https://doi.org/10.1146/annurev-pharmtox-050120-105018 [PubMed]
- 10. Coppé JP, Desprez PY, Krtolica A, Campisi J. The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol. 2010; 5:99–118. https://doi.org/10.1146/annurev-pathol-121808-102144 [PubMed]
- 11. Victorelli S, Passos JF. Telomeres and Cell Senescence - Size Matters Not. EBioMedicine. 2017; 21:14–20. https://doi.org/10.1016/j.ebiom.2017.03.027 [PubMed]
- 12. Hickson LJ, Langhi Prata LGP, Bobart SA, Evans TK, Giorgadze N, Hashmi SK, Herrmann SM, Jensen MD, Jia Q, Jordan KL, Kellogg TA, Khosla S, Koerber DM, et al. Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine. 2019; 47:446–56. https://doi.org/10.1016/j.ebiom.2019.08.069 [PubMed]
- 13. Kirkland JL, Tchkonia T. Senolytic drugs: from discovery to translation. J Intern Med. 2020; 288:518–36. https://doi.org/10.1111/joim.13141 [PubMed]
- 14. Kirkland JL, Tchkonia T, Zhu Y, Niedernhofer LJ, Robbins PD. The Clinical Potential of Senolytic Drugs. J Am Geriatr Soc. 2017; 65:2297–301. https://doi.org/10.1111/jgs.14969 [PubMed]
- 15. Camell CD, Yousefzadeh MJ, Zhu Y, Prata LGP, Huggins MA, Pierson M, Zhang L, O'Kelly RD, Pirtskhalava T, Xun P, Ejima K, Xue A, Tripathi U, et al. Senolytics reduce coronavirus-related mortality in old mice. Science. 2021; 373:eabe4832. https://doi.org/10.1126/science.abe4832 [PubMed]
- 16. Yousefzadeh MJ, Zhu Y, McGowan SJ, Angelini L, Fuhrmann-Stroissnigg H, Xu M, Ling YY, Melos KI, Pirtskhalava T, Inman CL, McGuckian C, Wade EA, Kato JI, et al. Fisetin is a senotherapeutic that extends health and lifespan. EBioMedicine. 2018; 36:18–28. https://doi.org/10.1016/j.ebiom.2018.09.015 [PubMed]
- 17. Zhu Y, Tchkonia T, Pirtskhalava T, Gower AC, Ding H, Giorgadze N, Palmer AK, Ikeno Y, Hubbard GB, Lenburg M, O'Hara SP, LaRusso NF, Miller JD, et al. The Achilles' heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. 2015; 14:644–58. https://doi.org/10.1111/acel.12344 [PubMed]
- 18. Fuhrmann-Stroissnigg H, Ling YY, Zhao J, McGowan SJ, Zhu Y, Brooks RW, Grassi D, Gregg SQ, Stripay JL, Dorronsoro A, Corbo L, Tang P, Bukata C, et al. Identification of HSP90 inhibitors as a novel class of senolytics. Nat Commun. 2017; 8:422. https://doi.org/10.1038/s41467-017-00314-z [PubMed]
- 19. Zhu Y, Doornebal EJ, Pirtskhalava T, Giorgadze N, Wentworth M, Fuhrmann-Stroissnigg H, Niedernhofer LJ, Robbins PD, Tchkonia T, Kirkland JL. New agents that target senescent cells: the flavone, fisetin, and the BCL-XL inhibitors, A1331852 and A1155463. Aging (Albany NY). 2017; 9:955–63. https://doi.org/10.18632/aging.101202 [PubMed]
- 20. Xu M, Pirtskhalava T, Farr JN, Weigand BM, Palmer AK, Weivoda MM, Inman CL, Ogrodnik MB, Hachfeld CM, Fraser DG, Onken JL, Johnson KO, Verzosa GC, et al. Senolytics improve physical function and increase lifespan in old age. Nat Med. 2018; 24:1246–56. https://doi.org/10.1038/s41591-018-0092-9 [PubMed]
- 21. Kasselimi E, Pefani DE, Taraviras S, Lygerou Z. Ribosomal DNA and the nucleolus at the heart of aging. Trends Biochem Sci. 2022; 47:328–41. https://doi.org/10.1016/j.tibs.2021.12.007 [PubMed]
- 22. Papadopoli D, Boulay K, Kazak L, Pollak M, Mallette F, Topisirovic I, Hulea L. mTOR as a central regulator of lifespan and aging. F1000Res. 2019; 8:998. https://doi.org/10.12688/f1000research.17196.1 [PubMed]
- 23. Weichhart T. mTOR as Regulator of Lifespan, Aging, and Cellular Senescence: A Mini-Review. Gerontology. 2018; 64:127–34. https://doi.org/10.1159/000484629 [PubMed]
- 24. Saito Y, Miyajima M, Yamamoto S, Sato T, Miura N, Fujimiya M, Chikenji TS. Accumulation of Senescent Neural Cells in Murine Lupus With Depression-Like Behavior. Front Immunol. 2021; 12:692321. https://doi.org/10.3389/fimmu.2021.692321 [PubMed]
- 25. Abate M, Festa A, Falco M, Lombardi A, Luce A, Grimaldi A, Zappavigna S, Sperlongano P, Irace C, Caraglia M, Misso G. Mitochondria as playmakers of apoptosis, autophagy and senescence. Semin Cell Dev Biol. 2020; 98:139–53. https://doi.org/10.1016/j.semcdb.2019.05.022 [PubMed]
- 26. Hall BM, Balan V, Gleiberman AS, Strom E, Krasnov P, Virtuoso LP, Rydkina E, Vujcic S, Balan K, Gitlin I, Leonova K, Polinsky A, Chernova OB, Gudkov AV. Aging of mice is associated with p16(Ink4a)- and β-galactosidase-positive macrophage accumulation that can be induced in young mice by senescent cells. Aging (Albany NY). 2016; 8:1294–315. https://doi.org/10.18632/aging.100991 [PubMed]
- 27. Kim HK, Merrow AC, Shiraj S, Wong BL, Horn PS, Laor T. Analysis of fatty infiltration and inflammation of the pelvic and thigh muscles in boys with Duchenne muscular dystrophy (DMD): grading of disease involvement on MR imaging and correlation with clinical assessments. Pediatr Radiol. 2013; 43:1327–35. https://doi.org/10.1007/s00247-013-2696-z [PubMed]



