Introduction
Gastric cancer is the 4th most common type of cancer, with a global incidence of approximately 1 million annually [1]. Many risk factors are implicated in the etiology of gastric cancer, including Helicobacter pylori infection, lack of fiber food, irregular food intake, heredity, etc. [2]. Treatment options for gastric cancer are limited [3]. Many studies have explored the mechanisms involved in the development, progression, and metastases of gastric cancer. Nevertheless, the molecular mechanism of gastric cancer in tumor microenvironments remains unclear. Therefore, unraveling the pathogenesis of gastric cancer in the tumor microenvironment will facilitate the identification of diagnostic biomarkers and the development of novel treatment strategies [4].
Tumor microenvironments comprise heterogeneous populations, including gastric cancer cells and infiltrating immune cells, which are essential regulators of cancer development. A single-cell RNA sequencing of TME revealed the immune cell landscape at the single-cell level, which helps in identifying novel clusters of tumor-associated immune cells [5] and signature genes for different immune cells. In gastric cancer tissues, a down-regulated IRF8 transcription factor was reported in CD8+ tumor-infiltrating lymphocytes [6]. Pembrolizumab, an immune checkpoint inhibitor that targets PD-1 and its PD-1 interactions with PD-L1 and PD-L2, is a therapeutic approach for gastric cancer [7, 8]. Stratifying patients based on molecular and genomic signatures is essential to identify suitable immunotherapeutic methods for each subgroup.
Polycomb group (PcG) proteins are essential gene regulators that mediate the stable inheritance of cell states. Aberration of epigenetic regulation mediated by PcG proteins has been explored in several cancer types. The CBX protein family, critical canonical PcG components, regulate tumorigenesis and tumor progression by maintaining tumor suppressors and the undifferentiated state of cancer stem cells [9]. Eight members of CBX proteins have been identified in human genomes. These CBXs regulate heterochromatin, mediation of PRC1 binding to nucleosomes, recruitment as well as stabilization of PRC1 to distinct chromatin regions. These proteins have a conserved N-terminal chromodomain. Two groups of CBXs have been defined based on differences in molecular structures and functions. The heterochromatin protein 1β (HP1β) group contains CBX1/3/5, which are associated with the heterochromatin protein 1 (HP1) complex to interpret H3K9me3 marks mediated by H3K9 methyltransferases. The Pc group has a conserved C-terminal polycomb repressor box, comprising CBX2/4/6/7/8, deposited by polycomb repressive complex 2 to recognize H3K27me3 [10]. Previous studies have shown the aberrant expressions of CBX family proteins and their prognostic values in gastric cancer [11, 12]. For instance, CBX6 is up-regulated in hepatocellular carcinoma and associated with lower survival outcomes [13]. CBX7 positively regulates the phenotype of gastric cancer stem cells by downregulating p16 and upregulating microRNA-21 [14]. Nevertheless, the correlation between CBXs and immune cell infiltration in the gastric cancer microenvironment remains elusive.
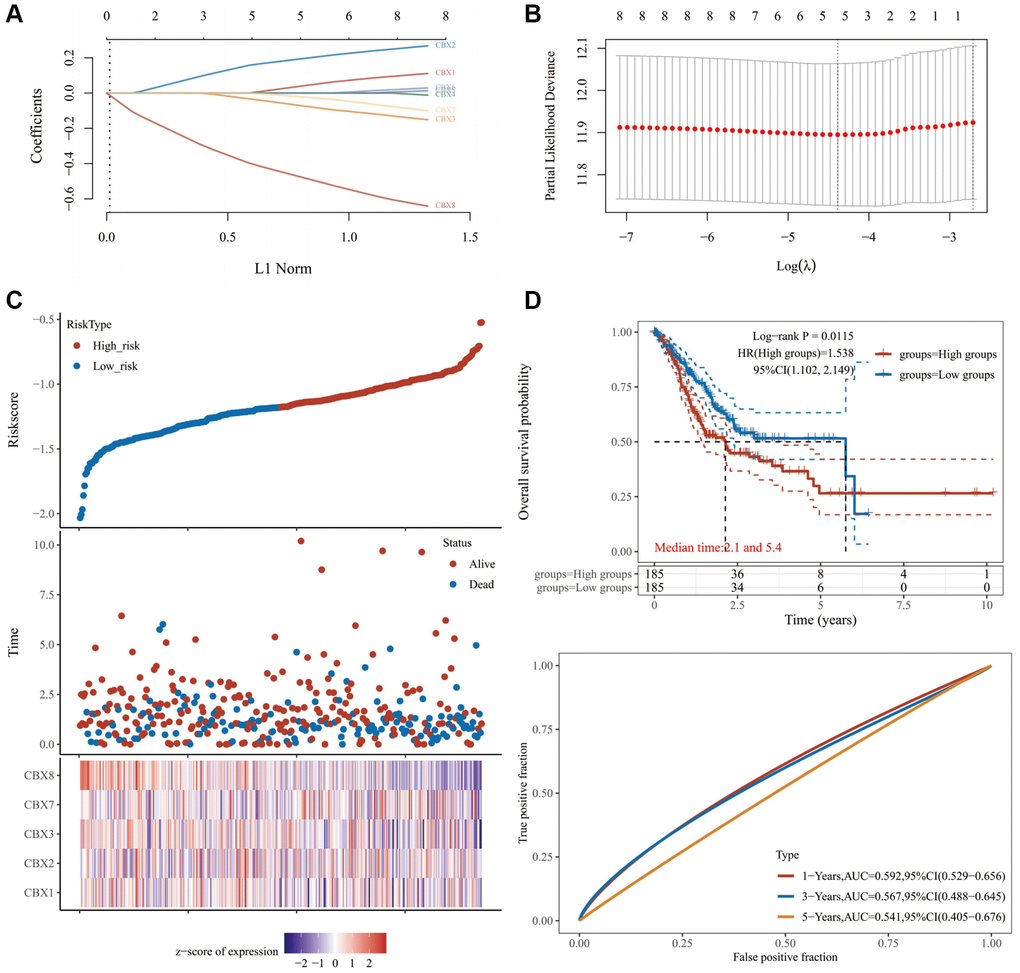
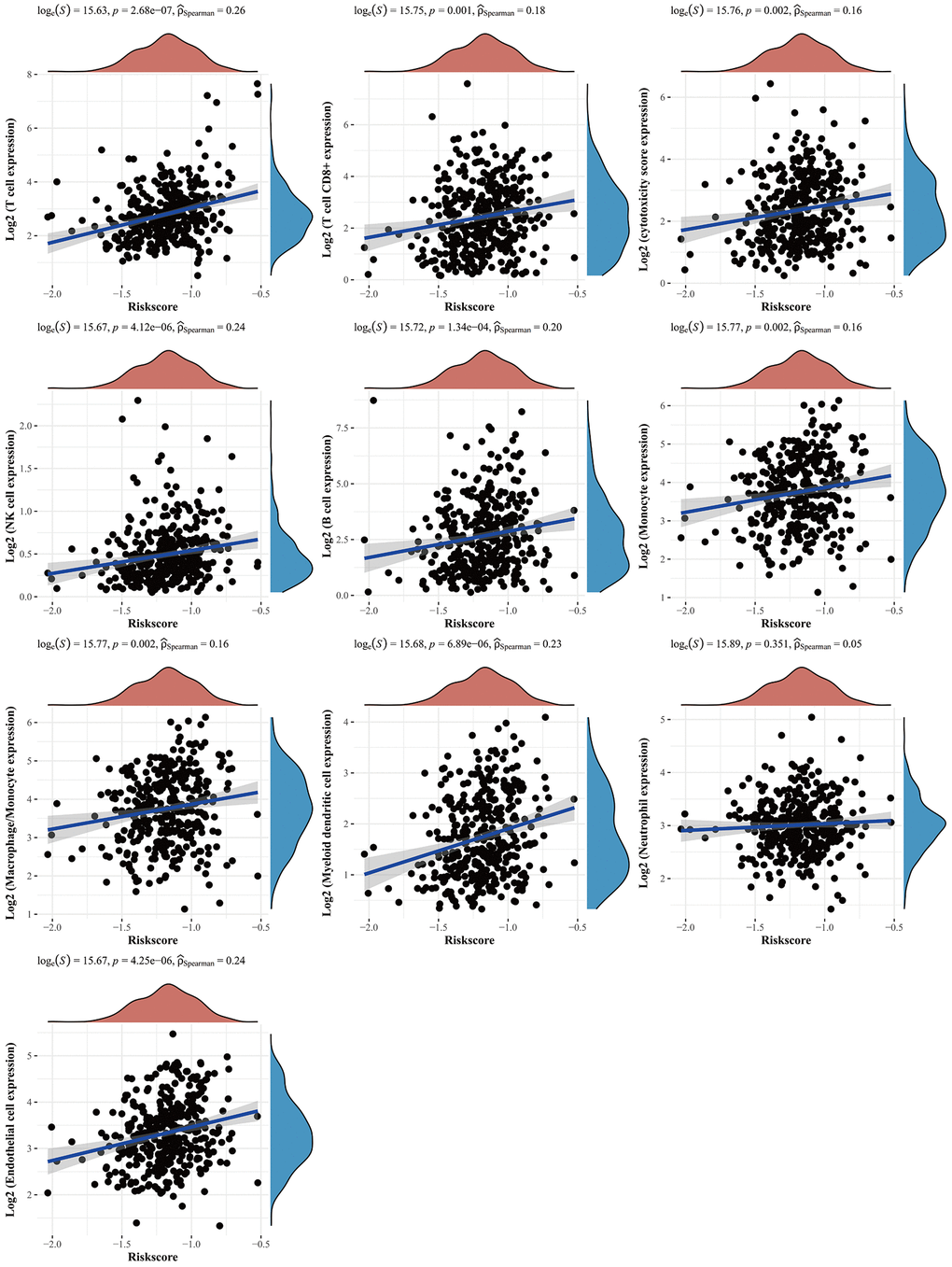
This study investigated the expression levels, clinical stages, mutations, risk factors, copy number variations (CNVs), and the immune microenvironment of gastric cancer. Consequently, we found that a prognostic CBXs model containing five CBX genes could predict overall survival for gastric cancer patients. Besides, a significant correlation was noted between the risk score of the CBXs-related prognostic gene model and immune-cell infiltration.
Results
Expression levels of different CBXs family members
First, we determined the expression levels of CBXs in different cancer types using the ONCOMINE database. Significantly upregulated mRNA expression of CBX1/2/3/4 was discovered in gastric cancer tissues compared to in normal control tissues (Supplementary Figure 1). Among the 8 CBXs family members, the expression levels of CBX1/3 were significantly upregulated, whereas CBX7 expression was significantly downregulated in other cancer types. Supplementary Table 1 summarizes the studies on gastric cancer. CBX1/2/3/4/6 were significantly up-regulated in different gastric adenocarcinoma types, whereas CBX7 was significantly down-regulated in diffuse gastric adenocarcinoma. These findings are in line with observation in different cancer types, which indicates the conserved function of the CBXs family among various tumor types.
RNA-seq data were downloaded from the TCGA, including 32 normal tissues and 375 gastric cancer tissues to verify the mRNA expression patterns of 8 CBXs in gastric cancer. Expression levels of CBX1/2/3/4/8 in gastric cancer samples were significantly upregulated, whereas mRNA expressions of CBX7 were significantly downregulated compared to the normal control in unpaired and paired analysis (Figure 1). The mRNA expression levels of 408 gastric cancer tissues were compared with 211 normal tissues using GEPIA online database. As shown in Supplementary Figure 2, the mRNA levels of CBX1/2/3/5/8 in gastric cancer tissues were significantly upregulated. CBX7 expression was significantly down-regulated in tumor samples, consistent with outcomes in other gastric cancer types (Supplementary Table 1).
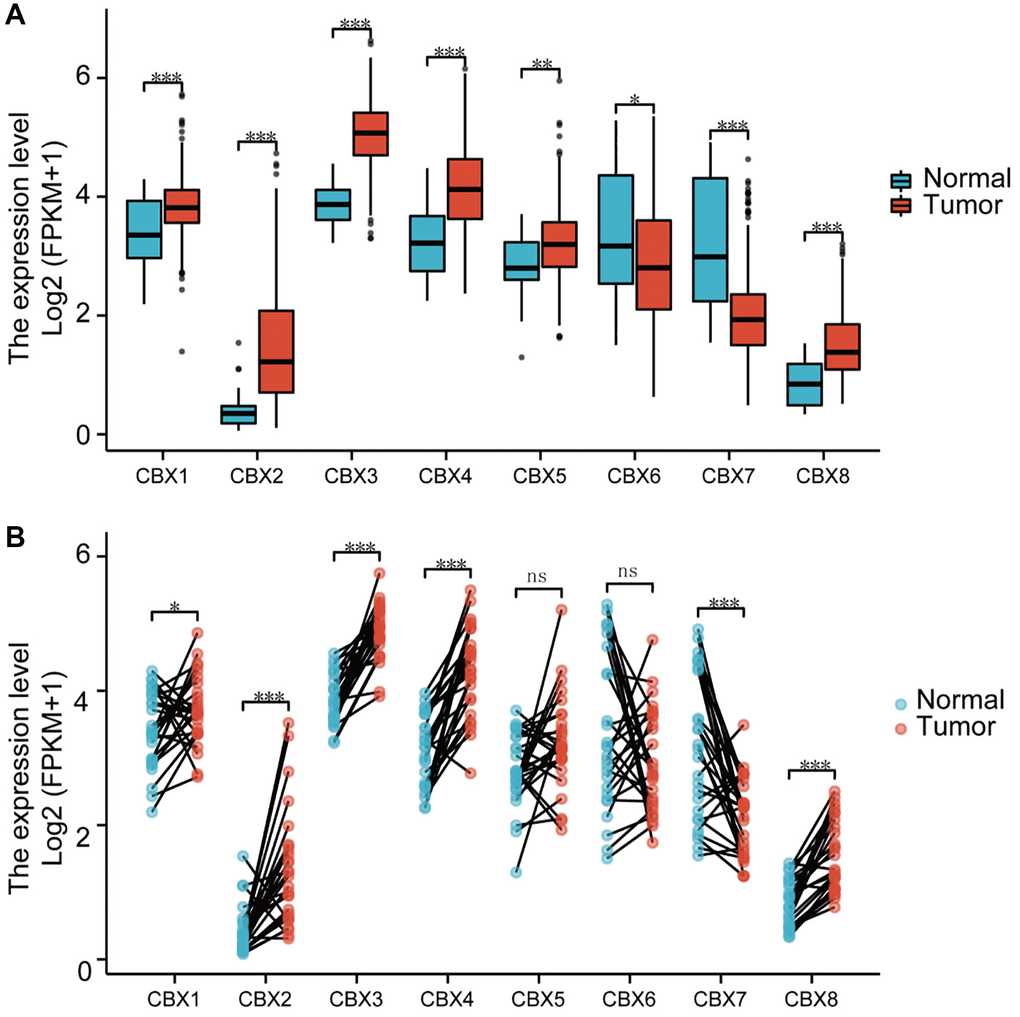
Figure 1. Analysis of CBXs mRNA expression levels. (A) unpaired samples containing 32 normal tissues and 375 gastric cancer tissues; (B) paired samples containing 32 normal tissues and corresponding gastric cancer tissues. Wilcoxon rank-sum test was used. *p < 0.05; **p < 0.01; ***p < 0.001; Abbreviation: ns: not significant.
Further, we evaluated protein expression patterns of CBXs in gastric cancer (Supplementary Figure 3). Protein levels of CBX2/3 increased in gastric cancer tissues. Suppressed protein expressions of CBX4/6/7 were observed in gastric cancer tissues. Additionally, similar protein expression levels of CBX5/8 were observed between normal tissues and gastric cancer tissues. Protein expression levels of CBX2/3/6/7 were in line with changes in mRNA expression levels.
Relationship between CBXs and clinicopathological features of patients with gastric cancer
We investigated the relationship between mRNA expression of CBXs and the clinical stage of gastric cancer patients. The mRNA expression levels of CBXs were not correlated with tumor stages in both databases (Supplementary Figures 4 and 5).
The relationship between CBXs mRNA expression levels and gastric cancer clinical grades was evaluated using the TISIDB database. The mRNA expression levels appeared high in patients with advanced cancer grades. Expression levels of CBX3/4/6/7/8 were significantly upregulated with clinical grades (Supplementary Figure 6). The highest mRNA expressions of CBX3/4/8 were found in grade 2, and the expression level dropped from grade 2 to 3 as the tumor grade increased. The highest mRNA expressions of CBX6/7 were found in grade 3. However, the expression levels of CBX1/2/5 did not significantly change with clinical grade. By integrating the results of mRNA and protein expression levels, CBX3/4/8 expression levels increased significantly from clinical grade 1 to 2 in gastric cancer.
Prognostic value of CBXs in gastric cancer patients
The correlation between CBXs and clinical outcomes in gastric cancer patients was examined using the microarray dataset and the RNA-seq data. The microarray dataset revealed that upregulated mRNA expressions of CBX3 (HR = 0.59, P = 1.4E-09) caused better overall survival outcomes among gastric cancer patients. In comparison, upregulated mRNA expression levels of CBX4 (HR = 1.25, p = 0.041), CBX5 (HR=2.08, p = 1.3E-13), CBX6 (HR = 1.5, p = 3.4E-06), CBX7 (HR = 1.52, p = 2e-06), and CBX8 (HR = 2.36, p = 3.1E-14) correlated with poor overall survival outcomes (Supplementary Figure 7). In addition, the RNA-seq data revealed that the mRNA expression levels of CBX1 (HR=1.61, p = 0.02) and CBX8 (HR = 0.62, p = 0.0048) significantly correlated with clinical outcomes in gastric cancer (Supplementary Figure 8). In general, mRNA expression levels of CBX1/3/4/5/6/7/8 significantly contributed to gastric cancer prognosis, confirming their potential application as biomarkers for the prediction of survival outcomes in gastric cancer patients.
Immune cell infiltration of CBXs in gastric cancer patients
A positive correlation was noted between CBX1 expression and CD4+ T cells as well as macrophage infiltration (Figure 2A). CBX2 and CBX8 expression levels inhibited CD8+ T cells, macrophages, neutrophils, and dendritic cells infiltration (Figure 2B, 2H). A negative correlation was noted between CBX3 expression and B cells, CD8+ T cells, CD4+ T cells, macrophages, neutrophils, and dendritic cell infiltration (Figure 2C). CBX4 expression levels promoted B cell infiltration but suppressed macrophage infiltration (Figure 2D). Moreover, CBX5 expression promoted CD4+ T cells and macrophage infiltration (Figure 2E). A negative correlation was noted between CBX6 expression and CD4+ T cells, macrophages, and dendritic cell infiltration (Figure 2F). CBX7 expression promoted all types of immune cell infiltration (Figure 2G).
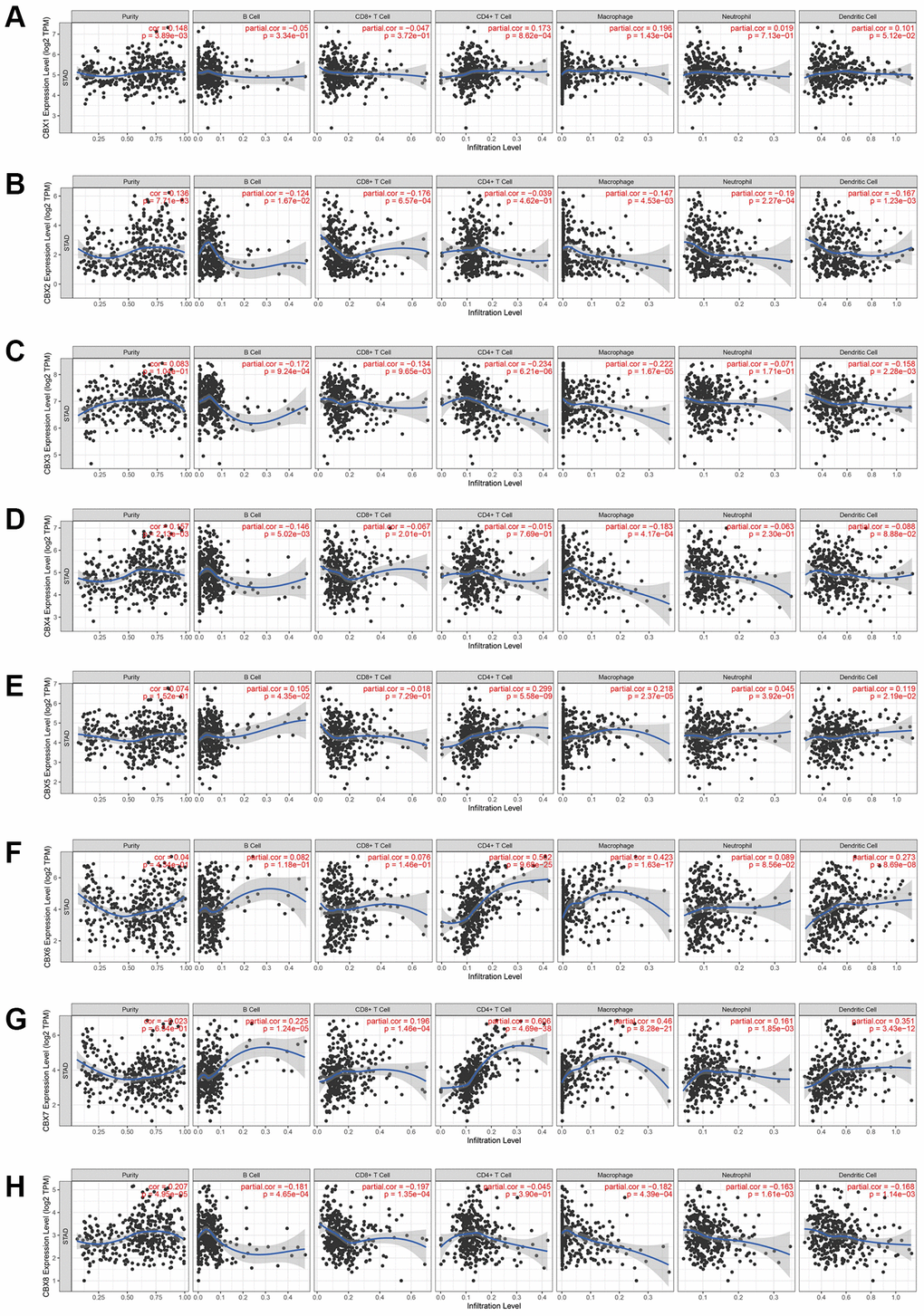
Figure 2. The correlation between CBXs and immune cell infiltration was analyzed by the TIMER database. (A) CBX1; (B) CBX2; (C) CBX3; (D) CBX4; (E) CBX5; (F) CBX6; (G) CBX7; (H) CBX8.
If a correlation coefficient >0.3 was defined as a strong correlation, then CBX6 promoted infiltration of CD4+ T cells (P = 9.28e-25, Cor = 0.502) and macrophages (P = 1.63e-17, Cor = 0.423), whereas CBX7 promoted the infiltration of CD4+ T cells (P = 4.69e-38, Cor = 0.606), macrophages (P = 8.28e-21, Cor = 0.46), and dendritic cells (P = 3.43e-12, Cor = 0.351).
Moreover, correlations between CBXs expression and tumor-infiltrating lymphocytes were discovered in various cancer types (Supplementary Figure 9).
The relationship between CNVs of CBXs and immune cell infiltration was evaluated. The CNVs of CBXs significantly correlated with immune cells, including B cells, CD8+ T cells, CD4+ T cells, macrophages, neutrophils, and dendritic cell infiltrations (Figure 3).
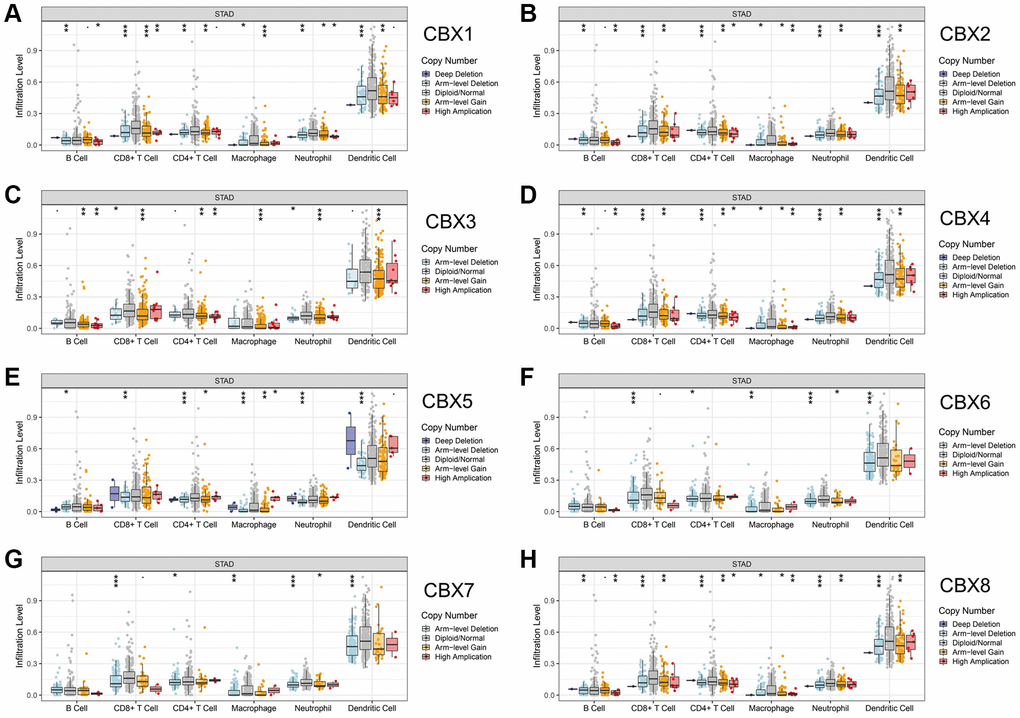
Figure 3. Correlation between CNV of CBXs and immune cell infiltration in gastric cancer analyzed by TIMER. (A) CBX1; (B) CBX2; (C) CBX3; (D) CBX4; (E) CBX5; (F) CBX6; (G) CBX7; (H) CBX8. *p < 0.05; **p < 0.01; ***p < 0.001.
Relationship between mRNA expressions of CBXs with immune subtypes and molecular subtypes in gastric cancer patients
CBXs significantly correlated with five immune subtypes analyzed in the TISIDB database (Figure 4). Expression levels of CBX1/2/3/4/8 in lymphocyte depleted subtype (C4) were significantly greater than those in other subtypes, and CBX6/7 had a significantly lower expression level in C4 and a higher expression level in inflammatory subtype (C3).
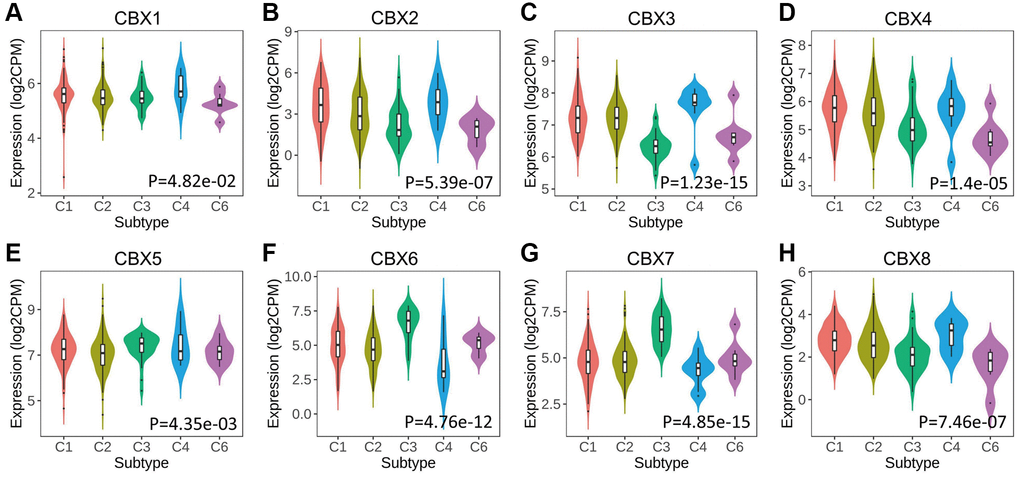
Figure 4. Relationship between CBXs and immune subtypes across in gastric cancer in TISIDB. (A) CBX1; (B) CBX2; (C) CBX3; (D) CBX4; (E) CBX5; (F) CBX6; (G) CBX7; (H) CBX8. C1: wound healing (n = 129); C2: IFN-gamma dominant (n = 210); C3: inflammatory (n = 36); C4: lymphocyte depleted (n = 9); C6: TGF-β dominant (n = 7).
The relationships between CBXs and the five molecular subtypes were investigated in the TISIDB database. Expression levels of CBXs except for CBX1 significantly correlated with five molecular subtypes. Expression levels of CBX2/3/4/8 in the genomically stable (GS) subtype were significantly low compared to the other subtypes, and CBX5/6/7 exhibited a higher expression level in the GS subtype (Figure 5).
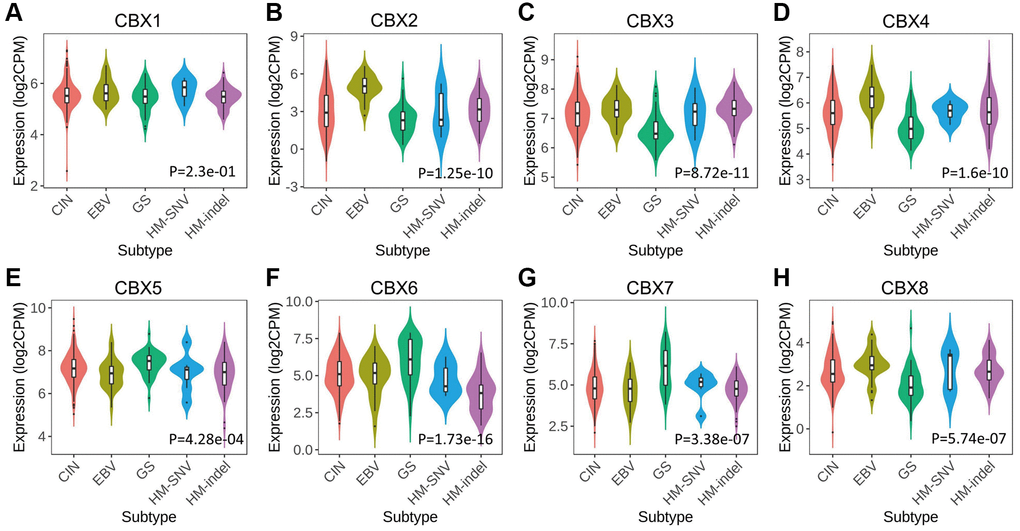
Figure 5. Relationship between CBXs and molecular subtypes in gastric cancer in TISIDB. (A) CBX1; (B) CBX2; (C) CBX3; (D) CBX4; (E) CBX5; (F) CBX6; (G) CBX7; (H) CBX8. Abbreviations: CIN: chromosomal instability (n = 223); EBV: Epstein–Barr virus positive (n = 30); GS: genomically stable (n = 50); HM-SNV: hypermutated-single-nucleotide variant predominant (n = 7) and HM-indel: hypermutated-insertion deletion mutation (n = 73).
Construction of a predictive nomogram
Univariate and multivariate regression analyses revealed that CBX3, CBX8, age, gender, pT stage, pN stage, and pM stage were independent factors for the prognosis of gastric cancer patients (Figure 9A, 9B). The predictive nomogram revealed that overall survival rates over 1, 2, and 3 years could be reasonably predicted (Figure 9C, 9D).
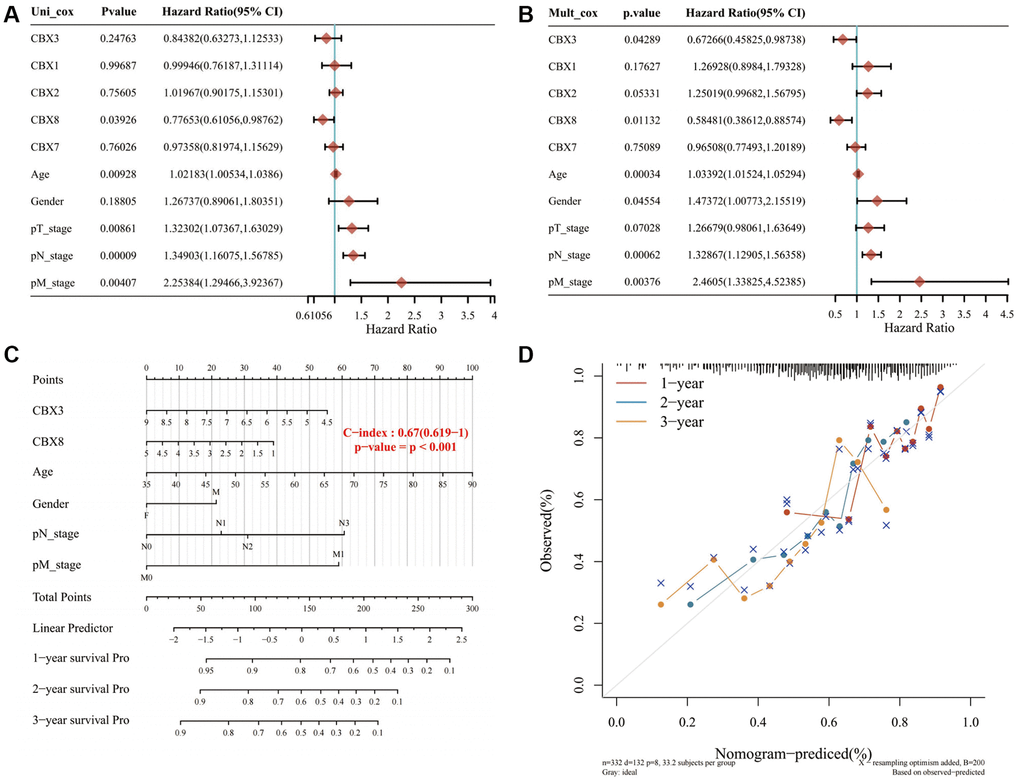
Figure 9. Constructing a prediction nomogram. (A, B) Univariate and multivariate regression analysis showed that CBX3, CBX8, age, gender, pT stage, pN stage, and pM stage were independent factors for the prognosis of gastric cancer patients; (C, D). The predictive nomogram suggested that overall survival rates over 1, 2 and 3 years could be reasonably predicted. A dashed diagonal line represents the ideal nomogram. Abbreviation: CBXs, chromobox proteins.
Risk factors associated with gastric cancer mortality
Further, the risk factors associated with mortality in 359 gastric cancer patients were assessed, among whom 140 died in the TIMER database. Table 1 presents a Cox proportional hazard model used to evaluate risk factors for mortality. Multivariate analysis revealed that six variables were risk factors for mortality in gastric cancer: stage II (HR = 2.125, p = 0.04); stage III (HR = 3.223, p = 0.001); stage IV (HR = 7.01, p < 0.001); age (HR = 1.042, p < 0.001); macrophages (HR = 475.661, p = 0.001), and CBX8 (HR = 0.595, p = 0.042) these variables were significantly associated with clinical outcomes of gastric cancer patients (Table 1). CBX6/7 significantly correlated with CD4+ T cells and macrophages, hence might be promising risk factors.
Table 1. The Cox regression model of clinical factors, tumor-infiltrating immune cells, and CBXs were analyzed by the TIMER database.
| coef | HR | 95% CI_low | 95% CI_up | P value | sig | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage II | 0.754 | 2.125 | 1.034 | 4.365 | 0.04 | * | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage III | 1.17 | 3.223 | 1.66 | 6.258 | 0.001 | ** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stage IV | 1.947 | 7.01 | 3.247 | 15.137 | 0 | *** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 0.041 | 1.042 | 1.023 | 1.061 | 0 | *** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender (male) | 0.193 | 1.213 | 0.832 | 1.768 | 0.315 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B cell | 3.082 | 21.802 | 0.244 | 1948.612 | 0.179 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CD8_Tcell | −1.731 | 0.177 | 0.008 | 3.94 | 0.274 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CD4_Tcell | −1.64 | 0.194 | 0.001 | 37.011 | 0.54 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Macrophage | 6.165 | 475.661 | 11.501 | 19672.08 | 0.001 | ** | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Neutrophil | −3.88 | 0.021 | 0 | 7.893 | 0.201 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dendritic | 1.25 | 3.491 | 0.267 | 45.573 | 0.34 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX1 | 0.25 | 1.284 | 0.9 | 1.833 | 0.168 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX2 | 0.137 | 1.146 | 0.904 | 1.453 | 0.258 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX3 | −0.166 | 0.847 | 0.57 | 1.257 | 0.41 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX4 | 0.126 | 1.134 | 0.759 | 1.695 | 0.538 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX5 | 0.06 | 1.062 | 0.791 | 1.426 | 0.69 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX6 | 0.068 | 1.07 | 0.863 | 1.328 | 0.537 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX7 | −0.246 | 0.782 | 0.591 | 1.035 | 0.086 | ns | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CBX8 | −0.52 | 0.595 | 0.36 | 0.982 | 0.042 | * | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: Coef: a regression coefficient; HR: hazard ratio; 95% CI: 95% confidential interval. *p < 0.05; **p < 0.01; ***p < 0.001; ns, not significant. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Figure 10 shows the receiver operating characteristic (ROC) curves for each CBXs gene. The area under the curve (AUC) for CBX3 was the highest at 0.959, indicating that CBX3-based prognostic indicators exert the best effect on patient stratification. Additionally, the AUC for CBX2/4/7/8 was more than 0.8, indicating the predictive efficacy of these genes.
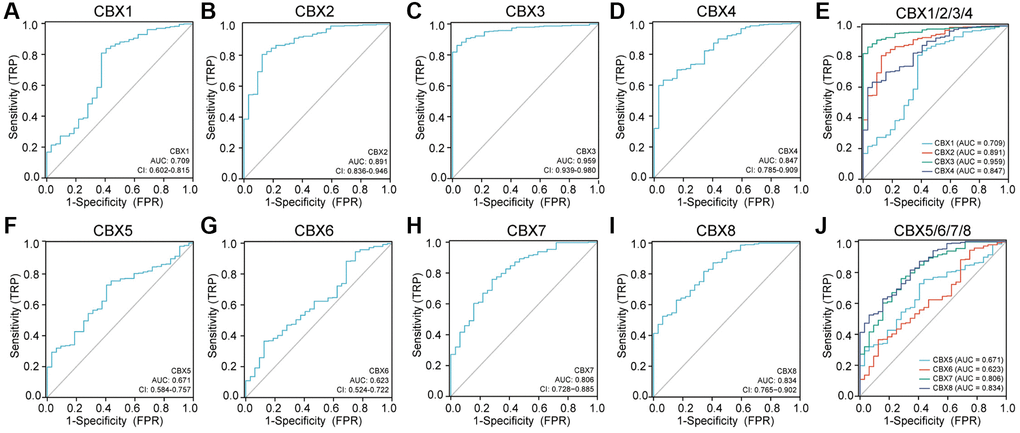
Figure 10. Receiver operating characteristic (ROC) curves for each CBXs gene in gastric cancer. (A) CBX1; (B) CBX2; (C) CBX3; (D) CBX4; (E) CBX5; (F) CBX6; (G) CBX7; (H) CBX8; (I) CBX1/2/3/4; (J) CBX5/6/7/8. Abbreviations: CI: confidence interval; AUC: area under curve; FPR: false positive rate; TPR: true positive rate.
Functional enrichment analysis of CBXs
A moderate to high correlation was observed in CBX2, CBX3, CBX4, CBX6, and CBX7, and a high correlation among CBX1, CBX5, and CBX8 (Figure 11A). Co-expression neighbor gene analysis of differentially expressed CBXs was performed using the GeneMANIA to explore potential interactions among them (Figure 11B). Metascape was used to analyze the functions of CBXs and their neighboring genes. As a result, GO term and pathways, including DNA duplex unwinding, covalent chromatin modification, regulation of PTEN gene transcription, chromatin-modifying enzymes, histone lysine methylation, and developmental processes involved in reproduction were linked to CBXs functions in gastric cancer (Figure 11C).
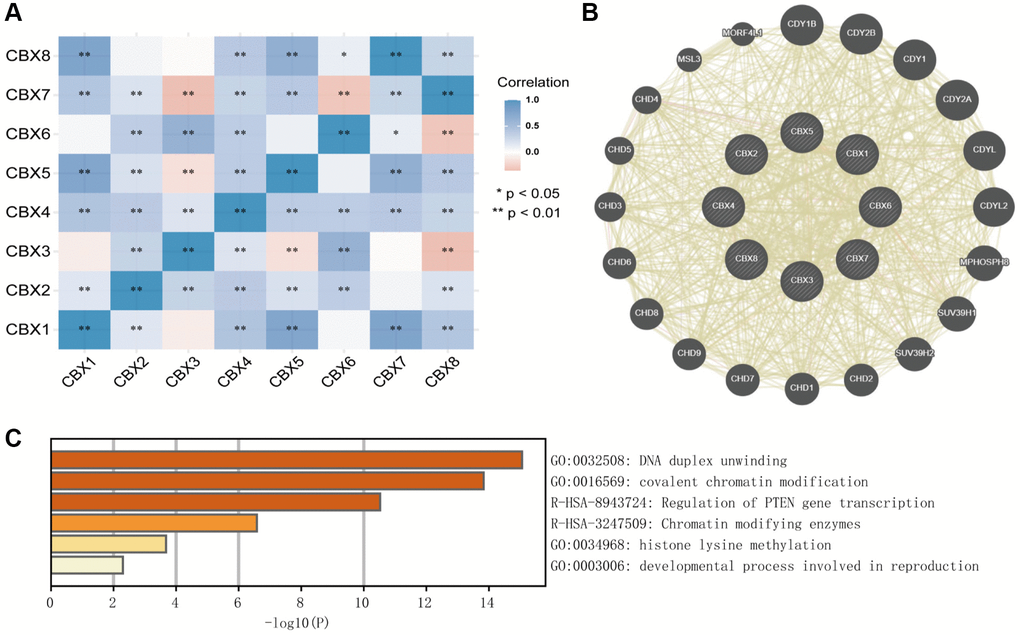
Figure 11. Predicted functions and pathways of CBXs and their coexpression neighbor genes in gastric cancer by Correlation heatmap (A), GeneMANIA (B), and Metascape (C).
Drug targets, miRNA targets, and transcription factor targets of CBXs
Drug targets, miRNA, and transcription factor targets of CBXs were investigated using the Enrichr databases. Three drugs, including Prednisolone, Phenacetin, and Pramoxine were identified for targeting CBXs in gastric cancer (Supplementary Table 2). The top three miRNA targets of CBXs included mmu-miR-493, his-miR-1296, and mmu-miR-5128 (Supplementary Table 3). Besides, the top three transcription factors (TEAD4, NRF1, and HINFP) were associated with the regulation of CBXs (Supplementary Table 4).
Discussion
Dysregulation of CBX family proteins has been analyzed in various cancer types [9, 19–21]. Evidence suggests that CBXs regulate tumorigenesis, tumor cell proliferation, invasion, and metastasis [10, 22]. Research has identified a correlation between CBX proteins and the tumor microenvironment [23]. Nonetheless, the tumorigenesis role of the CBXs family, specifically intercellular communication with immune cell infiltration remains understudied. Herein, we comprehensively analyzed 8 CBXs in gastric cancer as per their expression patterns, protein expression levels, clinicopathological parameters, prognostic values, biological functions, immune cell infiltration, copy number variation, and ROC curve of CBXs.
The mRNA and protein expression levels of CBX2/3 in gastric cancer tissues were significantly higher than in normal tissues, whereas CBX6/7 were down-regulated in gastric cancer. Protein expression levels of CBX1/2/5/8 were inconsistent with mRNA expression levels due to post-translational modification of CBX proteins. Studies have documented phosphorylation, SUMOylation/de-SUMOylation, and methylation/demethylation of CBXs. CBX4 is also a SUMO E3 ligase implicated in the regulation of SUMOylation and de-SUMOylation, and SUMOylation, mediating PRC1 recruitment of methylated histone 3 at K27 (H3K27me3), resulting in transcriptional repression of Gata4/6 transcription [24].
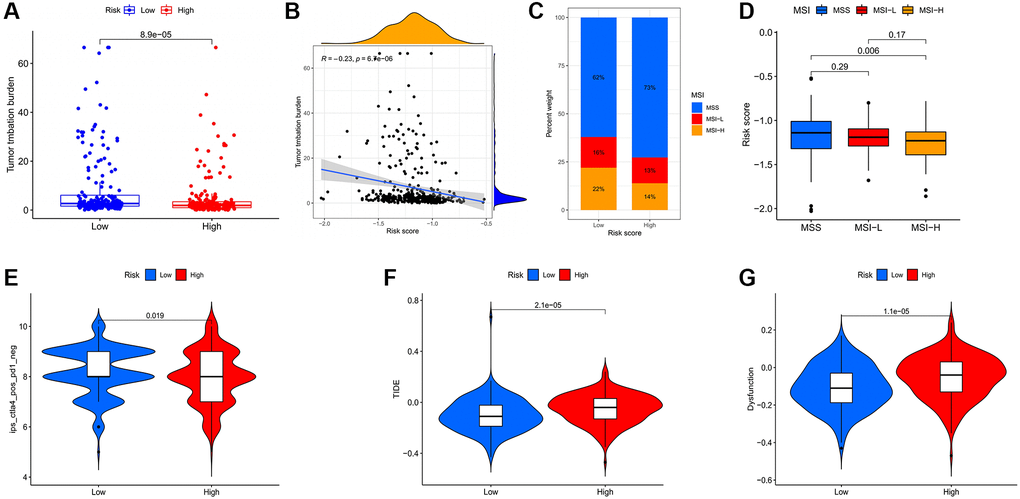
Furthermore, we noted that mRNA expression levels of CBX1/3/4/5/6/7/8 were significantly associated with gastric cancer prognosis. Notably, tumor stage and grade progression are influenced by protein expression levels, genetic mutations, tumor microenvironment, etc. We also observed dysregulated transcriptional expression of CBXs as tumors progressed. Studies indicate that CBX1 overexpression in gastric and breast cancers significantly correlates with shorter overall survival outcomes. Evidence shows that interactions between tumor and immune cells modulate tumor progression and recurrence, and consequently immunotherapeutic responses as well as clinical outcomes. CBX6/7 significantly correlates with immune cell infiltrations, particularly CD4+ T cells and macrophages, indicating that CBXs may also reveal immune status, hence regulating tumor status. We also revealed that the CNV of CBXs significantly correlates with immune cells. These results imply that CBXs could be critical regulators in gastric cancer progression. Previous studies have shown that CBX7-deficient upregulates FasL expression and consequently regulates CD4+ T cell apoptosis [25]. CBX2 promotes virus-infected macrophages by improving IFN-β transcription and promoting Jmjd3 recruitment to the Ifnb promoter [26]. The effects of CBX proteins on tumor states by regulating immune cell infiltrations warrant additional research. In addition, there were significant differences in immune checkpoints between low and high risk patients. The low risk patients could achieve a better response to immune checkpoint inhibitors.
Functional characterization of these genes revealed their relationship with chromatin modification and histone methylation. This was consistent with the roles of CBXs, a component of epigenetic regulation mediating proteins, PcG. We evaluated drug targets, miRNA targets, and transcription factor targets of the differentially expressed CBXs, and discovered that TEAD4, NRF1, and HINFP are critical transcription factors in the regulation of CBXs. Notably, TEAD4 is a downstream effector of the Hippo pathway. In coordination with YAP, TAZ, and VGLL, TEAD4 plays a critical role in cancer proliferation, including cell proliferation, metastasis, and cancer stem cell maintenance [27]. NRF1 mediates drug resistance in cancer via an oncometabolite, UDP-GlcNAc, which stimulates proteasome subunit genes in response to proteasome inhibitors, before maintaining proteasome activity and protecting cancer cells from proteotoxicity [28]. HINPF ablation inhibits histone H4 expression, disrupts the sub-nuclear organization of Histone Locus Bodies, and generates chromosomal fragility, hence sensitizing DNA to damage [29].
Conclusion
In conclusion, we comprehensively analyzed the potential effects of CBX protein family members in gastric cancer. Consequently, CBXs correlated with overall survival outcomes and could be vital prognostic markers in gastric cancer. Moreover, we found a prognostic CBXs model comprising five genes (CBX1, CBX2, CBX3, CBX7, and CBX8) for gastric cancer patients. Additional experiments and clinical cohort studies for CBXs are necessary to validate our results further.
Materials and Methods
ONCOMINE database
The ONCOMINE database (http://www.oncomine.org/) allows genome-wide expression analysis of integrated cancer microarray data [30]. Transcriptional expression of CBXs was investigated in gastric cancer tissues. Statistical differences in transcriptional expression levels between normal and cancer tissues were analyzed using the student’s t-test. Threshold settings were: P-value: 0.01; fold change: 1.5; gene rank: 10%; data type: mRNA.
The cancer genome atlas database
The cancer genome atlas (TCGA, https://www.cancer.gov/tcga) is a landmark cancer genome project, comprising sequencing and pathological data of 33 cancer types [31]. We downloaded HTSeq-FPKM formatted RNA-seq data, corresponding clinical data, and somatic mutation information of gastric cancer, including 375 tumor samples and 32 normal samples. Log2 transformation was performed for FPKM formatted RNA-seq data. The mRNA expression levels of unpaired samples and paired samples were visualized by the ggplot2 package. The ROC curve was drawn using the pROC package.
Correlation analysis of risk score and immunotherapy
The correlation of model risk scores with immune checkpoints and TMB was analyzed in the TCGA-STAD and GSE84437 datasets by the ggpubr package. The TIDE and T cell dysfunction scores of TCGA-STAD tumor samples were predicted from the TIDE database (http://tide.dfci.harvard.edu/login/) and the levels of these scores were compared between high and low risk groups. TCGA-STAD IPS and MIS scores data were downloaded from the TCIA database (https://tcia.at/home) to compare the levels of these scores for high and low risk groups.
TISIDB analysis
The TISIDB database (http://cis.hku.hk/TISIDB) merges 988 reported immune-related genes in the tumor microenvironment and provides a relationship between genes and immune cell infiltration by analyzing high-throughput screening data and genomics, transcriptomics, as well as clinical data [33]. Here, we established the relationships among expression levels of CBXs, clinical information, and subtype, and evaluated the correlations between CBXs expression and lymphocytes in gastric cancer.
GEPIA database
GEPIA (http://gepia.cancer-pku.cn/) is a web server with RNA sequencing expression data from the TCGA and GTEx projects [34]. Transcriptional expression differences of CBXs were compared between gastric cancer and normal gastric tissues.
Human protein atlas
The Human Protein Atlas (https://www.proteinatlas.org) is a website comprising immunohistochemistry-based expression cell lines and tissue data for most identified genes [35]. We directly compared protein expression levels of different CBXs family members by obtaining immunohistochemical images between human normal and gastric cancer tissues.
Kaplan-Meier plotter database
The predictive values of CBXs in gastric cancers were analyzed by the Kaplan-Meier plotter (http://kmplot.com/analysis/) [36]. Differences with P-values less than 0.05 (P < 0.05) were considered statistically significant.
GeneMANIA
GeneMANIA (http://www.genemania.org) is a flexible website that provides gene functions, protein interactions, relationships of genes and datasets, functionally similar genes, as well as similar genes with shared properties [37].
Metascape
Metascape (http://metascape.org) is a predictable and instinctive tool for gene annotation and gene enrichment analysis [38]. GO and KEGG in Metascape were used to analyze the functions of CBXs and CBXs co-expression genes.
TIMER
TIMER (https://cistrome.shinyapps.io/timer/) is a detailed resource for the systematic analysis of immune infiltrates [39, 40]. The correlation between the expression of CBXs and the abundance of immune cell infiltration was analyzed in the “Gene” module. Clinical relevance of infiltrated immune cells and CBXs expression in a multivariable Cox proportional hazards model were evaluated in the “Survival” module.
Enrichr
Enrichr (http://amp.pharm.mssm.edu/Enrichr/) is a comprehensive online resource for curated gene sets and gene function analysis [41]. Enrichr contains 184 annotated gene sets from 102 gene set libraries for analysis and download, including transcription, pathways, ontologies, diseases/drugs, cell types, etc.
Statistical analysis
The online databases were used to automatically perform statistical analyses, and the part of the code analysis was completed using the R package. For categorical variables, the chi-squared test was used, but for continuous variables, the Wilcoxon signed-rank test was applied. For comparisons, Spearman’s correlation analysis was utilized. P < 0.05 was considered statistically significant.
Data availability
The data supporting our results of this work are obtainable from TCGA (https://portal.gdc.cancer.gov/) and other data in the paper can be obtained from the corresponding author based on reasonable request.
Author Contributions
YinJiang Zhang: Data curation, Writing–original draft, Methodology. LinYi Zhao: Software. Xu He: Conceptualization. RongFei Yao: Visualization. Fan Lu: Data Curation. BiNan Lu: Supervision. ZongRan Pang: Validation, Writing - review and editing.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
References
- 1. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 2. Li WQ, Zhang JY, Ma JL, Li ZX, Zhang L, Zhang Y, Guo Y, Zhou T, Li JY, Shen L, Liu WD, Han ZX, Blot WJ, et al. Effects of Helicobacter pylori treatment and vitamin and garlic supplementation on gastric cancer incidence and mortality: follow-up of a randomized intervention trial. BMJ. 2019; 366:l5016. https://doi.org/10.1136/bmj.l5016 [PubMed]
- 3. Shitara K, Doi T, Dvorkin M, Mansoor W, Arkenau HT, Prokharau A, Alsina M, Ghidini M, Faustino C, Gorbunova V, Zhavrid E, Nishikawa K, Hosokawa A, et al. Trifluridine/tipiracil versus placebo in patients with heavily pretreated metastatic gastric cancer (TAGS): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2018; 19:1437–48. https://doi.org/10.1016/S1470-2045(18)30739-3 [PubMed]
- 4. Cristescu R, Lee J, Nebozhyn M, Kim KM, Ting JC, Wong SS, Liu J, Yue YG, Wang J, Yu K, Ye XS, Do IG, Liu S, et al. Molecular analysis of gastric cancer identifies subtypes associated with distinct clinical outcomes. Nat Med. 2015; 21:449–56. https://doi.org/10.1038/nm.3850 [PubMed]
- 5. Zhang M, Hu S, Min M, Ni Y, Lu Z, Sun X, Wu J, Liu B, Ying X, Liu Y. Dissecting transcriptional heterogeneity in primary gastric adenocarcinoma by single cell RNA sequencing. Gut. 2021; 70:464–75. https://doi.org/10.1136/gutjnl-2019-320368 [PubMed]
- 6. Fu K, Hui B, Wang Q, Lu C, Shi W, Zhang Z, Rong D, Zhang B, Tian Z, Tang W, Cao H, Wang X, Chen Z. Single-cell RNA sequencing of immune cells in gastric cancer patients. Aging (Albany NY). 2020; 12:2747–63. https://doi.org/10.18632/aging.102774 [PubMed]
- 7. Sasaki S, Nishikawa J, Sakai K, Iizasa H, Yoshiyama H, Yanagihara M, Shuto T, Shimokuri K, Kanda T, Suehiro Y, Yamasaki T, Sakaida I. EBV-associated gastric cancer evades T-cell immunity by PD-1/PD-L1 interactions. Gastric Cancer. 2019; 22:486–96. https://doi.org/10.1007/s10120-018-0880-4 [PubMed]
- 8. Kim ST, Cristescu R, Bass AJ, Kim KM, Odegaard JI, Kim K, Liu XQ, Sher X, Jung H, Lee M, Lee S, Park SH, Park JO, et al. Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer. Nat Med. 2018; 24:1449–58. https://doi.org/10.1038/s41591-018-0101-z [PubMed]
- 9. Jangal M, Lebeau B, Witcher M. Beyond EZH2: is the polycomb protein CBX2 an emerging target for anti-cancer therapy? Expert Opin Ther Targets. 2019; 23:565–78. https://doi.org/10.1080/14728222.2019.1627329 [PubMed]
- 10. van Wijnen AJ, Bagheri L, Badreldin AA, Larson AN, Dudakovic A, Thaler R, Paradise CR, Wu Z. Biological functions of chromobox (CBX) proteins in stem cell self-renewal, lineage-commitment, cancer and development. Bone. 2021; 143:115659. https://doi.org/10.1016/j.bone.2020.115659 [PubMed]
- 11. Ma T, Ma N, Chen JL, Tang FX, Zong Z, Yu ZM, Chen S, Zhou TC. Expression and prognostic value of Chromobox family members in gastric cancer. J Gastrointest Oncol. 2020; 11:983–98. https://doi.org/10.21037/jgo-20-223 [PubMed]
- 12. Lin K, Zhu J, Hu C, Bu F, Luo C, Zhu X, Zhu Z. Comprehensive analysis of the prognosis for chromobox family in gastric cancer. J Gastrointest Oncol. 2020; 11:932–51. https://doi.org/10.21037/jgo-20-208 [PubMed]
- 13. Zheng H, Jiang WH, Tian T, Tan HS, Chen Y, Qiao GL, Han J, Huang SY, Yang Y, Li S, Wang ZG, Gao R, Ren H, et al. CBX6 overexpression contributes to tumor progression and is predictive of a poor prognosis in hepatocellular carcinoma. Oncotarget. 2017; 8:18872–84. https://doi.org/10.18632/oncotarget.14770 [PubMed]
- 14. Ni SJ, Zhao LQ, Wang XF, Wu ZH, Hua RX, Wan CH, Zhang JY, Zhang XW, Huang MZ, Gan L, Sun HL, Dimri GP, Guo WJ. CBX7 regulates stem cell-like properties of gastric cancer cells via p16 and AKT-NF-κB-miR-21 pathways. J Hematol Oncol. 2018; 11:17. https://doi.org/10.1186/s13045-018-0562-z [PubMed]
- 15. Choucair K, Morand S, Stanbery L, Edelman G, Dworkin L, Nemunaitis J. TMB: a promising immune-response biomarker, and potential spearhead in advancing targeted therapy trials. Cancer Gene Ther. 2020; 27:841–53. https://doi.org/10.1038/s41417-020-0174-y [PubMed]
- 16. Rizzo A, Ricci AD, Brandi G. PD-L1, TMB, MSI, and Other Predictors of Response to Immune Checkpoint Inhibitors in Biliary Tract Cancer. Cancers (Basel). 2021; 13:558. https://doi.org/10.3390/cancers13030558 [PubMed]
- 17. Charoentong P, Finotello F, Angelova M, Mayer C, Efremova M, Rieder D, Hackl H, Trajanoski Z. Pan-cancer Immunogenomic Analyses Reveal Genotype-Immunophenotype Relationships and Predictors of Response to Checkpoint Blockade. Cell Rep. 2017; 18:248–62. https://doi.org/10.1016/j.celrep.2016.12.019 [PubMed]
- 18. Jiang P, Gu S, Pan D, Fu J, Sahu A, Hu X, Li Z, Traugh N, Bu X, Li B, Liu J, Freeman GJ, Brown MA, et al. Signatures of T cell dysfunction and exclusion predict cancer immunotherapy response. Nat Med. 2018; 24:1550–8. https://doi.org/10.1038/s41591-018-0136-1 [PubMed]
- 19. Liang YK, Lin HY, Chen CF, Zeng D. Prognostic values of distinct CBX family members in breast cancer. Oncotarget. 2017; 8:92375–87. https://doi.org/10.18632/oncotarget.21325 [PubMed]
- 20. Ning G, Huang YL, Zhen LM, Xu WX, Jiao Q, Yang FJ, Wu LN, Zheng YY, Song J, Wang YS, Xie C, Peng L. Transcriptional expressions of Chromobox 1/2/3/6/8 as independent indicators for survivals in hepatocellular carcinoma patients. Aging (Albany NY). 2018; 10:3450–73. https://doi.org/10.18632/aging.101658 [PubMed]
- 21. Xie X, Ning Y, Long J, Wang H, Chen X. Diverse CBX family members as potential prognostic biomarkers in non-small-cell lung cancer. FEBS Open Bio. 2020; 10:2206–15. https://doi.org/10.1002/2211-5463.12971 [PubMed]
- 22. Chang SC, Lai YC, Chen YC, Wang NK, Wang WS, Lai JI. CBX3/heterochromatin protein 1 gamma is significantly upregulated in patients with non-small cell lung cancer. Asia Pac J Clin Oncol. 2018; 14:e283–8. https://doi.org/10.1111/ajco.12820 [PubMed]
- 23. Connelly KE, Martin EC, Dykhuizen EC. CBX Chromodomain Inhibition Enhances Chemotherapy Response in Glioblastoma Multiforme. Yale J Biol Med. 2016; 89:431–40. [PubMed]
- 24. Kang X, Qi Y, Zuo Y, Wang Q, Zou Y, Schwartz RJ, Cheng J, Yeh ET. SUMO-specific protease 2 is essential for suppression of polycomb group protein-mediated gene silencing during embryonic development. Mol Cell. 2010; 38:191–201. https://doi.org/10.1016/j.molcel.2010.03.005 [PubMed]
- 25. Li J, Li Y, Cao Y, Yuan M, Gao Z, Guo X, Zhu F, Wang Y, Xu J. Polycomb chromobox (Cbx) 7 modulates activation-induced CD4+ T cell apoptosis. Arch Biochem Biophys. 2014; 564:184–8. https://doi.org/10.1016/j.abb.2014.10.004 [PubMed]
- 26. Sun D, Cao X, Wang C. Polycomb chromobox Cbx2 enhances antiviral innate immunity by promoting Jmjd3-mediated demethylation of H3K27 at the Ifnb promoter. Protein Cell. 2019; 10:285–94. https://doi.org/10.1007/s13238-018-0581-0 [PubMed]
- 27. Chen M, Huang B, Zhu L, Chen K, Liu M, Zhong C. Structural and Functional Overview of TEAD4 in Cancer Biology. Onco Targets Ther. 2020; 13:9865–74. https://doi.org/10.2147/OTT.S266649 [PubMed]
- 28. Sekine H, Motohashi H. Roles of CNC Transcription Factors NRF1 and NRF2 in Cancer. Cancers (Basel). 2021; 13:541. https://doi.org/10.3390/cancers13030541 [PubMed]
- 29. Ghule PN, Xie RL, Colby JL, Jones SN, Lian JB, Wijnen AJ, Stein JL, Stein GS. p53 checkpoint ablation exacerbates the phenotype of Hinfp dependent histone H4 deficiency. Cell Cycle. 2015; 14:2501–8. https://doi.org/10.1080/15384101.2015.1049783 [PubMed]
- 30. Rhodes DR, Kalyana-Sundaram S, Mahavisno V, Varambally R, Yu J, Briggs BB, Barrette TR, Anstet MJ, Kincead-Beal C, Kulkarni P, Varambally S, Ghosh D, Chinnaiyan AM. Oncomine 3.0: genes, pathways, and networks in a collection of 18,000 cancer gene expression profiles. Neoplasia. 2007; 9:166–80. https://doi.org/10.1593/neo.07112 [PubMed]
- 31. Wang Z, Jensen MA, Zenklusen JC. A Practical Guide to The Cancer Genome Atlas (TCGA). Methods Mol Biol. 2016; 1418:111–41. https://doi.org/10.1007/978-1-4939-3578-9_6 [PubMed]
- 32. Yoon SJ, Park J, Shin Y, Choi Y, Park SW, Kang SG, Son HY, Huh YM. Deconvolution of diffuse gastric cancer and the suppression of CD34 on the BALB/c nude mice model. BMC Cancer. 2020; 20:314. https://doi.org/10.1186/s12885-020-06814-4 [PubMed]
- 33. Ru B, Wong CN, Tong Y, Zhong JY, Zhong SSW, Wu WC, Chu KC, Wong CY, Lau CY, Chen I, Chan NW, Zhang J. TISIDB: an integrated repository portal for tumor-immune system interactions. Bioinformatics. 2019; 35:4200–2. https://doi.org/10.1093/bioinformatics/btz210 [PubMed]
- 34. Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017; 45:W98–102. https://doi.org/10.1093/nar/gkx247 [PubMed]
- 35. Asplund A, Edqvist PH, Schwenk JM, Pontén F. Antibodies for profiling the human proteome-The Human Protein Atlas as a resource for cancer research. Proteomics. 2012; 12:2067–77. https://doi.org/10.1002/pmic.201100504 [PubMed]
- 36. Szász AM, Lánczky A, Nagy Á, Förster S, Hark K, Green JE, Boussioutas A, Busuttil R, Szabó A, Győrffy B. Cross-validation of survival associated biomarkers in gastric cancer using transcriptomic data of 1,065 patients. Oncotarget. 2016; 7:49322–33. https://doi.org/10.18632/oncotarget.10337 [PubMed]
- 37. Warde-Farley D, Donaldson SL, Comes O, Zuberi K, Badrawi R, Chao P, Franz M, Grouios C, Kazi F, Lopes CT, Maitland A, Mostafavi S, Montojo J, et al. The GeneMANIA prediction server: biological network integration for gene prioritization and predicting gene function. Nucleic Acids Res. 2010; 38:W214–20. https://doi.org/10.1093/nar/gkq537 [PubMed]
- 38. Zhou Y, Zhou B, Pache L, Chang M, Khodabakhshi AH, Tanaseichuk O, Benner C, Chanda SK. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun. 2019; 10:1523. https://doi.org/10.1038/s41467-019-09234-6 [PubMed]
- 39. Li T, Fan J, Wang B, Traugh N, Chen Q, Liu JS, Li B, Liu XS. TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res. 2017; 77:e108–10. https://doi.org/10.1158/0008-5472.CAN-17-0307 [PubMed]
- 40. Li T, Fu J, Zeng Z, Cohen D, Li J, Chen Q, Li B, Liu XS. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020; 48:W509–14. https://doi.org/10.1093/nar/gkaa407 [PubMed]
- 41. Kuleshov MV, Jones MR, Rouillard AD, Fernandez NF, Duan Q, Wang Z, Koplev S, Jenkins SL, Jagodnik KM, Lachmann A, McDermott MG, Monteiro CD, Gundersen GW, Ma'ayan A. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016; 44:W90–7. https://doi.org/10.1093/nar/gkw377 [PubMed]