Introduction
After the onset of spinal cord injury (SCI), the reduction of control over sympathetic nerve cells renders the autonomic reflex circuits in the nervous system dysfunctional, triggering pathology such as dysreflexia as well as SCI [1–3]. Such disorders induce the imbalance in the autonomic signals in the digestive tract, leading to dysfunctions in mucosal secretion, colonic mobility, as well as vascular tone [4, 5]. Amongst the problems faced by people with SCI, neurogenic bowel dysfunction (NBD) has actually been a significant issue that may seriously impact the life of SCI patients. Two major indications of NBD, i.e., constipation as well as fecal incontinence, occur in nearly half of SCI patients [6, 7].
Moxibustion has been a treatment used in traditional Chinese medicine for many years to induce pain relief at the points of acupuncture called acupoints [8]. Moxibustion trigger self-healing by impacting the functions of the neuroendocrine immune system [9]. While the use of thermal therapy to replace moxibustion has been controversial, both treatments involve similar mechanisms in pain relief as well as self-healing [9, 10]. Past research actually presented that moxibustion modifies the expression levels of genes such as cytokines to play a beneficial role in the treatments of inflammatory as well as autoimmune conditions [11]. Moreover, it has been found that the combined treatment of moxibustion with acupuncture could recover the motor function and preserve the neuron cells in SCI rats [12].
It was discovered that moxibustion therapy elevated the levels of Firmicutes while reducing the number of Bacteroidetes as well as Proteobacteria. Regarding the efficacy of moxibustion therapy, it was proposed that short term moxibustion therapy might dramatically impact the microbiome in the digestive tract. In general, in ulcer colitis mice, the quantity of beneficial bacteria was reduced, while the number of numerous opportunistic pathogens was increased [13].
Discrepancies of microbiota composition in the gut are called “dysbiosis” and are actually triggered by a lot of factors, such as genes in the host, lifestyle, as well as direct exposure to microbes or even different clinical procedures [14]. Dysbiosis has been linked to both inflammation in the intestinal tract irritation as well as many health conditions outside the digestive tract, like obesity, atopic eczema, allergic reaction, as well as diabetic issues [3, 15, 16].
As reported by previous studies, the severe neurological and psychological complications induced by SCI could induce gut dysbiosis. For example, SCI patients could be subjected to sudden and dramatic life changes, causing acute and chronic psychological stress. Neurogenic damages could develop in bladder and bowel due to spinal autonomic circuitry after SCI. And dysautonomia induced by SCI could also damage the immune system, increasing the patients’ possibility to use antibiotics against infections. Therefore, it was presented that SCI induces gut dysbiosis, which in turn hinders the healing as well as worsening the pathology of SCI [17]. In addition, microbiota in the digestive tract can impact serotonin formation, neurotransmission, as well as metabolic process in a sex-specific way [18, 19].
Macrophage polarization appears to induce different macrophage reactions depending on the perceived stimulations. Macrophage polarization has actually been characterized into 2 types, i.e., M1 as well as M2 polarization, with each type of polarization pertaining to particular immune system feedbacks, amongst which both the advancement as well as relief of inflammation play crucial roles [20].
It has been shown that gut dysbiosis could exacerbate neurological impairment and spinal cord pathology during post-SCI recovery, and hence was considered as a disease-modifying factor after traumatic spinal cord injury [17, 21]. In this study, we aimed to study the effect of MOX on microbiota dysbiosis and macrophage polarization to unveil the mechanism underlying the therapeutic effect of MOX on SCI treatment. Accordingly, we hypothesized that MOX could alleviate spinal cord injury by promoting microbiota dysbiosis and inhibiting the polarization of macrophages.
Materials and Methods
Animal model
The experimental processes of animal studies have actually been reviewed and approved by our Animal Ethics Committee. Female C57BL/6 adult mice weighing 20 to 22 g were purchased from the Facility of Experimental Animals at our institution and housed in an air-conditioned animal facility subjected to a 12 h: 12 h light:dark cycle. The temperature in the animal facility was maintained at 22 ± 1°C while the humidity level in the animal facility was kept to 50 ± 10%. All mice had ad libitum access to food as well as water during the study. Subsequently, the mice were divided into 4 groups, i.e., 1. SHAM group (N = 8, mice treated with a sham surgical procedure); 2. SHAM +MOX group (N = 8, mice treated with a sham surgical procedure and then given MOX); 3. SCI group (N = 8, mice treated with a surgical procedure to induce SCI); and 4. SCI + MOX group (N = 8, mice treated with a surgical procedure to induce SCI and then given MOX). To induce SCI, the mice in SCI groups were anesthetized by using 2% isoflurane. Then, laminectomy was conducted at the level of T10 and the mice got a contusion injury induced by dropping an impactor (Infinite Horizons, Precision Systems and Instrumentation, Lexington, KY, USA). After that, the laceration was sutured and the mice were returned to a warming enclosure till they were fully awake. Post-operatively, the animals were given 0.5 ml of Ringer's solution via S.C. injection once a day for 5 times. The mice in the SHAM groups were given laminectomy at level T10 without receiving the injury induced by the impactor. And for the MOX treatment, moxibustion was delivered by using moxa cones that were 0.6 centimeter tall, 0.5 centimeter in diameter and 1.8 gram in weight (Nanyang Hanyi Moxa, Nanyang, China). The moxa cones were positioned on bilateral acupoints ST25 (acupoints as illustrated in Supplementary Figure 1) to deliver 10 minutes of moxibustion once daily for 28 days. The skin temperature at the site of acupuncture was kept at 43°C during the entire treatment procedure. By the completion of this study, all mice were sacrificed by a lethal intravenous dose of sodium pentobarbital at 100 mg/kg.
RNA isolation and real time PCR
A Trizol reagent (Invitrogen, Carlsbad, CA, USA) was utilized in accordance with the recommended protocol provided by the manufacturer to isolate total RNA content from cell as well as tissue samples. The concentration as well as quality of isolate total RNA samples was identified by utilizing a Nano Drop spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) in accordance with the recommended protocol provided by the manufacturer. Then, reverse transcription of isolate total RNA was done by using a Hairpin-it real time PCR Quantitation assay kit (GenePharma, Shanghai, China) along with a PrimeScript RT Master Mix (TaKaRa, Tokyo, Japan) in accordance with the recommended protocol provided by the assay kit manufacturers. In the next step, real time PCR was executed by using a SYBR Green PCR master mix (Applied Biosystems, Foster City, CA, USA) on an 7900HT real time PCR machine (Applied Biosystems, Foster City, CA, USA) in accordance with the recommended protocol provided by the manufacturers. Finally, the relative expression of M2 markers of colonic macrophages, i.e., CD206 (Forward primer sequence: 5′-AGCCAACACCAGCTCCTCAAGA-3′; Reverse primer sequence: 5′-CAAAACGCTCGCGCATTGTCCA-3′) and Arg-1 (Forward primer sequence: 5′-TCATCTGGGTGGATGCTCACAC-3′; Reverse primer sequence: 5′-GAGAATCCTGGCACATCGGGAA-3′), as well as M1 markers of colonic macrophages, i.e., CXCL-9 (Forward primer sequence: 5′-CTGTTCCTGCATCAGCACCAAC-3′; Reverse primer sequence: 5′-TGAACTCCATTCTTCAGTGTAGCA-3′), iNOS (Forward primer sequence: 5′-GCTCTACACCTCCAATGTGACC-3′; Reverse primer sequence: 5′-CTGCCGAGATTTGAGCCTCATG-3′) and caspase-3 (Forward primer sequence: 5′-GGAAGCGAATCAATGGACTCTGG-3′; Reverse primer sequence: 5′-GCATCGACATCTGTACCAGACC-3′), was calculated by normalization to the expression of internal control gene GAPDH (Forward primer sequence: 5′-GTCTCCTCTGACTTCAACAGCG-3′; Reverse primer sequence: 5′-ACCACCCTGTTGCTGTAGCCAA-3′) via making use of the 2−ΔΔCT procedure.
Western blot analysis
Total protein was separated from tissue samples and assessed by utilizing a BCA protein assay kit (Tiangen Biotech, Beijing, China) in accordance with the recommended protocol provided by the manufacturer. Then, 10 μg of proteins in every sample were resolved by using 10% SDS-PAGE before they were electro-transferred onto polyvinylidene fluoride (PVDF) membranes (Millipore, Billerica, MA, USA) for immunoblotting. Then, the membranes were blocked with 10 FBS and incubated with anti-caspase-3 primary antibodies (dilution of 1:5000; ab32351, Abcam, Cambridge, MA, USA) as well as HRP-labeled secondary antibodies (dilution of 1:1000; ab6747, Abcam, Cambridge, MA, USA) in accordance with the recommended protocol provided by the antibody manufacturer. Finally, the grey levels of protein bands were quantified by utilizing Image J software to determine the protein expression of caspase-3 in each sample.
ELISA
After the behavior examinations, all mice were killed through cervical dislocation and their blood samples were collected. In the next step, the expression of IL-17, IFN-γ, MCP-1, and IL-1β in each sample was determined by utilizing commercial ELISA kits (Catalog # BMS6001 for IL-17; Catalog # KMC4021 for IFN-γ; Catalog # EMCCL12 for MCP-1; Catalog # BMS6002 for IL-1β; Thermo Fisher Scientific, Waltham, MA, USA) in accordance with the recommended protocol provided by the kit manufacturer.
TUNEL assay
The status of apoptosis in collected tissue samples was evaluated by using a TUNEL assay (Roche, Basel, Switzerland) in accordance with the recommended protocol provided by the assay kit manufacturer.
BMS score
The Basso Mouse Scale (BMS) score was assessed based on hindlimb movement after the moxibustion treatment. In brief, the mice were analyzed in an open area for 4 minutes pre-operatively as well as on D3, D7, D14, D21 and D28 postoperatively. The left and right hindlimb performance was ranked independently before the scores averaged to produce the total score and sub-scores of BMS. Locomotion parameters were evaluated by utilizing DigiGait Image Analysis software. In each behavioral test, the mice were trained at a rate of 15 cm/s for 7 days prior to the induction of SCI before they were tested at a rate of 9 cm/s and their images were recorded with a high-speed camera.
QIIME test
To evaluate the Ace index of operational taxonomic unit (OTU) level, Shannon index of OTU level as well as the relative abundances of Lactobacillales, Clostridiales and Bifidobacteriales, the mice in each group were subjected to QIIME tests. In brief, paired end readings were combined by utilizing a FLASH software program. Then, quality filtering of raw tags was carried out to get high quality tags depending on the requirements in the QIIME software. In the next step, the tag information was compared to the reference information by making use of a UCHIME algorithm to identify chimera sequences, which were then eliminated to acquire effective tags. For OTU clustering as well as species annotation, all effective tags were subjected to sequence analysis by utilizing the Uparse program.
Test of body weight, food intake, energy expenditure (EE) and respiratory quotient (RQ) in each mouse group
Total EE, which was shown as kcal/day, was actually assessed prior to as well as throughout the entire experiment by using Weir’s formula, i.e., VCO2 (EEVCO2) = 3.941 × VCO2 (L/min)/RQ + 1.11 × VCO2 (L/min) × 1440.
Statistical analysis
All results were the average of three experiments. All data were shown as the average ± standard deviation. The correlation between BMS score and the levels of Clostridiales/Lactobacillales was analyzed by using linear regression. One-way ANOVA was utilized for comparing different groups. Statistical evaluations were done by making use of SPSS 22.0 software (SPSS, Chicago, IL). P < 0.05 was actually taken into consideration as statistically significant.
Results
MOX treatment mitigated dysregulated gut bacterial composition in SCI mice
QIIME was used to measure the level and diversity of bacterial composition based on the OTU level in each group. Moreover, relative abundance of Lactobacillales, Clostridiales and Bifidobacteriales was also compared between different groups. As indicated by the results, the SCI group showed a significant increase in the Ace index (Figure 1A) and Shannon index (Figure 1B). Accordingly, MOX treatment significantly reduced the richness and diversity of the intestinal microbiota in SCI mice while exhibiting no effect in the SHAM group. As shown in Figure 1C and compared with the SHAM group, the induction of SCI markedly reduced the levels of Lactobacillales and Bifidobacteriales while elevating the level of Clostridiales. Accordingly, MOX treatment alleviated SCI-induced up-regulation of Clostridiales and down-regulation of Lactobacillales but exhibited no obvious effect on the level of Bifidobacteriales.
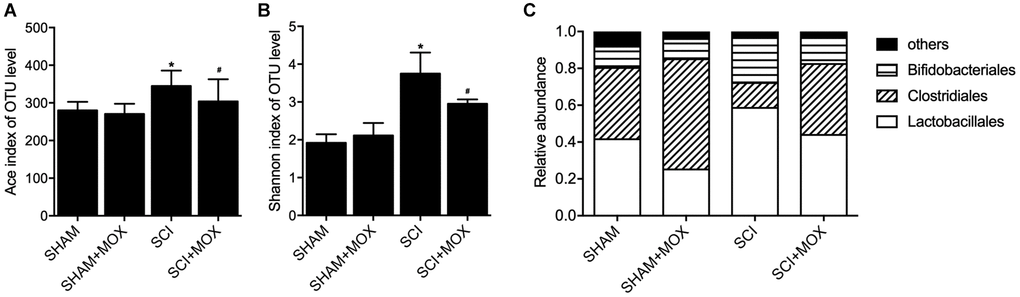
Figure 1. MOX treatment mitigated the dysregulation of gut bacterial composition in SCI mice (*P value < 0.05 vs. SHAM group; #P value < 0.05 vs. SCI group). (A) Ace index in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (B) Shannon index in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (C) Relative abundance of Lactobacillales, Clostridiales and Bifidobacteriales in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group.
MOX treatment mitigated the dysregulation of weight gain and metabolic profiling in SCI mice
After comparing the body weight in all mouse groups from Day 0 to Day 28 after model establishment (Figure 2A), we found that the SHAM and SHAM+MOX groups showed stable and comparable values of body weight index, but the mice in the SCI group lost body weight after surgery, and MOX treatment gradually aimed weight gain in SCI mice. In addition, metabolic parameters including food intake, EE and RQ were assessed in each group. As shown by the results, the food intake (Figure 2B), EE (Figure 2C) and RQ (Figure 2D) of SCI mice were all evidently decreased, while MOX treatment significantly mitigated the reduction in these metabolic parameters in SCI mice. Noteworthy, MOX treatment upon sham-operated mice showed no effect on the indexes, indicating that MOX treatment was only effective in SCI mice.
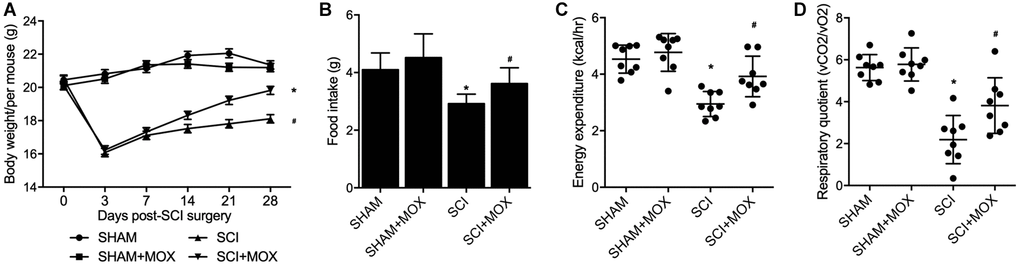
Figure 2. MOX treatment mitigated the dysregulation of weight gain and metabolic profiling in SCI mice (*P value < 0.05 vs. SHAM group; #P value < 0.05 vs. SCI group). (A) Body weight during 0-28 days post-SCI surgery in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (B) Food intake in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (C) Energy expenditure in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (D) Respiratory quotient in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group.
MOX treatment improved locomotor recovery in SCI mice
BMS was performed to evaluate the changes and subsequent recovery in locomotion of each group. As shown in Figure 3A, the BMS score remained unchanged in the SHAM and SHAM+MOX groups but was evidently reduced in the SCI group, and the subsequent post-SCI recovery was accompanied with a stepwise increase in the BMS score. Also, mice treated with MOX exhibited a greater improvement in locomotion activity. We also analyzed the correlation between Clostridiales/Lactobacillales and BMS score. As indicated by the results, the BMS score was negatively correlated with the level of Clostridiales (Figure 3B) and positively correlated with the level of Lactobacillales (Figure 3C), indicating the possibility of using certain bacteria to predict the functional recovery after SCI.
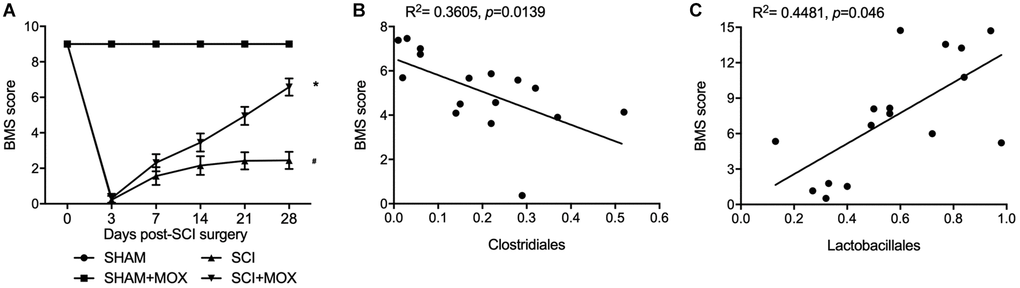
Figure 3. MOX treatment improved locomotor recovery in SCI mice. (A) BMS score during 0–28 days post-SCI surgery in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group (*P value < 0.05 vs. SHAM group; #P value < 0.05 vs. SCI group); (B) Correlation analysis of BMS score and Clostridiales level in the SCI group and SCI+MOX group; (C) Correlation analysis of BMS score and Lactobacillales level in the SCI group and SCI+MOX group.
MOX treatment reduced SCI-induced up-regulation of IL-17, IFN-γ, MCP-1 and IL-1β expression in SCI mice
ELISA was performed to measure the levels of IL-17, IFN-γ, MCP-1 and IL-1β in each mouse group. Compared with those in the SHAM group, the levels of IL-17 (Figure 4A), IFN-γ (Figure 4B), MCP-1 (Figure 4C) and IL-1β (Figure 4D) were increased in the SCI group, and MOX treatment reduced the expression of IL-17, IFN-γ, MCP-1 and IL-1β in the SCI+MOX group. Moreover, no evident difference was observed between the SHAM+MOX and SHAM groups.
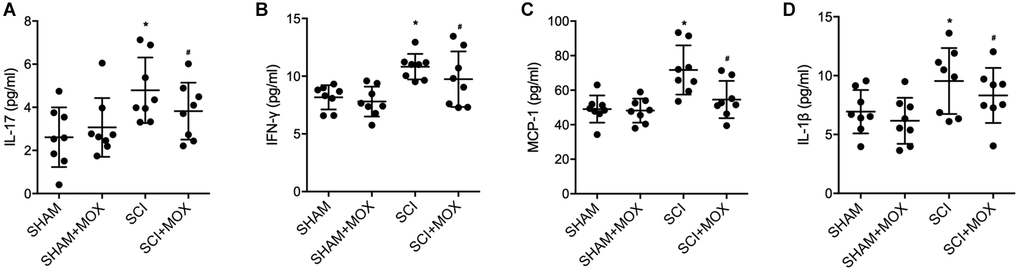
Figure 4. MOX treatment reduced SCI-induced up-regulation of IL-17, IFN-γ, MCP-1 and IL-1β expression. (A) Level of IL-17 in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (B) Level of IFN-γ in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (C) Level of MCP-1 in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group. (D) Level of IL-1β in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group.
MOX treatment preserved colonic macrophage phenotypes in SCI mice
To measure macrophage polarization in each group, the expression of M1 markers including CXCL-9 and iNOS, as well as the expression of M2 markers including CD206 and Arginase-1, were observed in colonic macrophages. As shown in Figure 5, the level of M2 markers CD206 mRNA (Figure 5A) and Arg-1 mRNA (Figure 5B) were decreased in SCI mice, while the levels of M1 markers CXCL-9 mRNA (Figure 5C) and iNOS mRNA (Figure 5D) were increased in SCI mice. Furthermore, the dysregulation of these markers in SCI mice was recovered by the treatment with MOX.
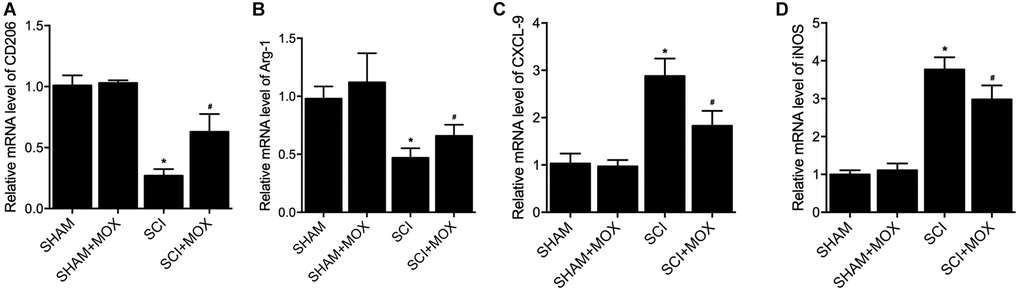
Figure 5. MOX treatment preserved colonic macrophage phenotypes in SCI mice. (A) Level of CO206 mRNA in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (B) Level of Arg-1 mRNA in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (C) Level of CXCL-9 mRNA in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (D) Level of iNOS mRNA in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group.
MOX treatment suppressed cell apoptosis in SCI mice
TUNEL assay was utilized to measure the apoptosis of cells in spinal cord tissues. As shown in Figure 6, the apoptotic index was most evidently increased in the SCI group, while the SCI+MOX group showed an apoptotic index lower than that in the SCI group. Moreover, compared with that in the SHAM group, the expression of caspase-3 mRNA (Figure 7A) and protein (Figure 7B) was also increased after the induction of SCI, and was reduced by MOX treatment in the SCI+MOX group.
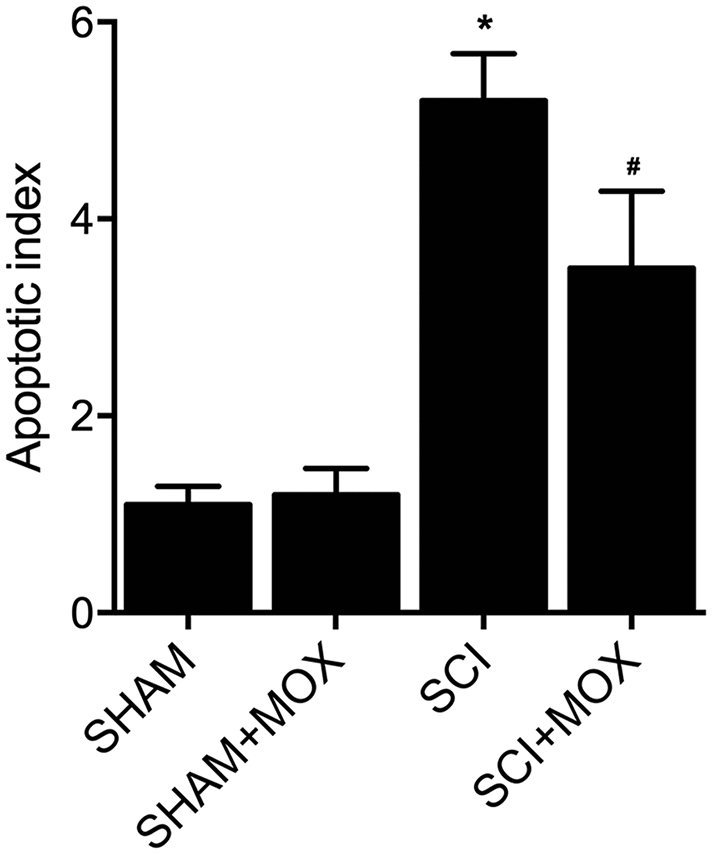
Figure 6. TUNEL assay validated that MOX treatment suppressed cell apoptosis in SCI mice (*P value < 0.05 vs. SHAM group; #P value < 0.05 vs. SCI group).
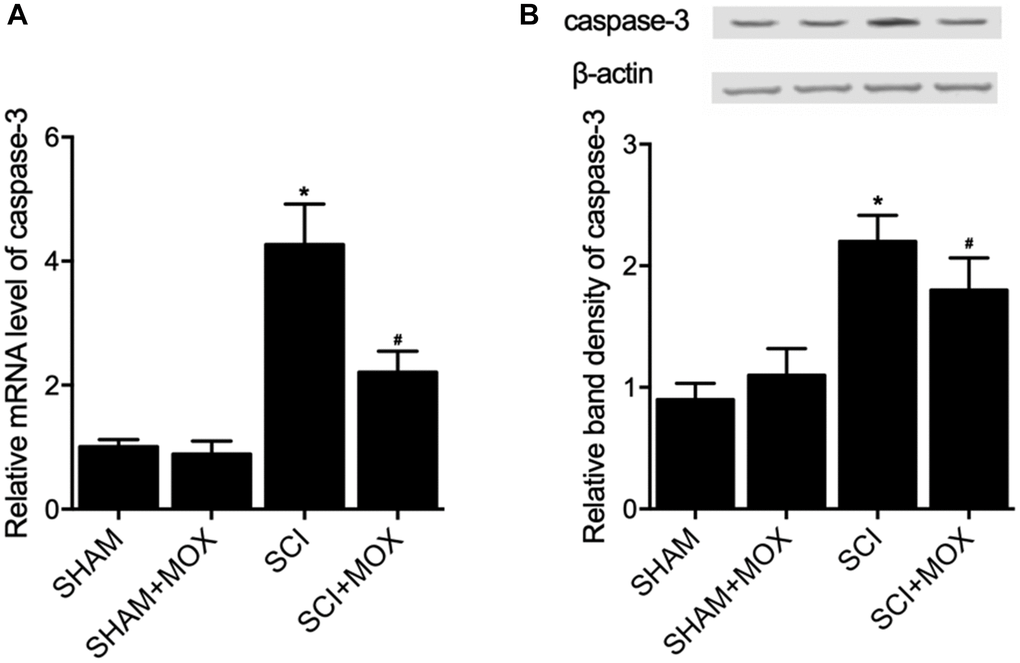
Figure 7. MOX treatment suppressed SCI-induced up-regulation of caspase-3 expression in SCI mice (*P value < 0.05 vs. SHAM group; #P value < 0.05 vs. SCI group). (A) Relative mRNA level of caspase-3 in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group; (B) Relative band density of caspase-3 protein in the SHAM group, SHAM+MOX group, SCI group and SCI+MOX group.
Discussion
Moxibustion was analyzed in many IBD animal models and appeared to alleviate inflammation in rodents by lowering the expression of TNF receptors [22, 23]. Moxibustion can likewise upregulate the expression of tight junction protein to reduce the apoptosis of epithelial cells [24, 25]. Furthermore, rats with DSS induced colitis showed enhanced expression of inflammatory cytokines, minimized expression of anti-inflammatory cytokines, as well as gut dysbiosis inflamed, and the effects of DSS could be alleviated by moxibustion therapy, which exerted its curative effects through regulating the microbiome as well as the immunity in intestinal tract mucus [13]. In this study, we found that the Ace index and Shannon index in the SCI group were significantly increased, while the MOX treatment significantly reduced the values of above indexes. The relative abundance of Lactobacillales and Bifidobacteriales was reduced while the relative level of Clostridiales was elevated in the SCI group, while the MOX treatment mitigated the above issues. Meanwhile, we also found that the BMS score remained unchanged between the SHAM and SHAM+MOX groups but was evidently reduced in the SCI group, while the MOX treatment greatly improved locomotion activity. Accordingly, the BMS score was negatively correlated with the level of Clostridiales and positively correlated with the level of Lactobacillales.
The impact of moxibustion might travel a long distance as a result of neural reflexes [26]. Past researches have actually uncovered that moxibustion can activate distinct cytokines as well as signaling pathways while enhancing the autophagic activity of macrophages [9, 11, 25, 27, 28]. Additionally, dysbiosis might promote the pathogenesis of post-SCI intraspinal disorders, and recent information revealed that dysbiosis after SCI triggers the reduction of butyrate-producing bacteria in the gut [29–31]. That is why regular use of probiotics containing lactic acid bacteria such as Lactobacillus as well as Bifidobacterium can actually promote post-SCI healing in mice [17, 32, 33]. At the same time, it was shown that gut Lactobacillus as well as Bifidobacterium levels were increased after SCI, suggesting the therapeutic outcome of probiotics in the SCI treatment.
Nonetheless, numerous factors can influence the curative outcome of regulating gut dysbiosis. The immune responses are important post-SCI physiological processes, and the activation of macrophages has been shown to be a necessary step in triggering such responses [34, 35]. The differing levels of the M1 as well as M2 macrophages can determine the profile of post-SCI immune responses [36]. The M1 as well as M2 macrophages may be individually modulated by cytokines released from Th2 and Th1 helper cells, respectively [37]. Microbial stimulations, including lipopolysaccharides as well as cytokines related to Th1 helper cells like interferon-γ can promote M1 polarization of macrophages, which exert pro-inflammatory, tumor resistance as well as microbicidal effects due to their ability of antigen presentation and higher yield of IL-6, IL-23 as well as IL-12, along with high level of secretion of reactive oxygen intermediates and NO [38–41]. On the other hand, Th2 cytokines like IL-4 as well as IL-13 can promote M2 polarization of macrophages, which exert anti-inflammatory, tumor promoting as well as parasite clearance effects [42]. In this study, we found that the levels of IL-17, IFN-γ, MCP-1 and IL-1β were increased in the SCI group, and MOX treatment reduced the levels of IL-17, IFN-γ, MCP-1 and IL-1β in the SCI+MOX group. Similarly, the levels of M2 markers CD206 mRNA and Arg-1 mRNA were decreased but the levels of M1 markers CXCL-9 mRNA and iNOS mRNA were increased in SCI mice, while the dysregulation of these markers was alleviated by the MOX treatment. The phenotype change of macrophages can be impacted by the changes in post-SCI neuro-inflammatory environment [43]. It was proposed that both macrophages and microglia might be polarized in reaction to various stimulations, including the presence of various cytokine clusters [44–46]. Furthermore, both IL-13 and IL-4 are anti-inflammatory and are cytokines frequently utilized for inducing alternative polarization in Raw264.7 macrophages. In addition, IL-13 and IL-4, along with their receptors IL-13R and IL-4R, are important signals required to induce above polarization [47].
Conclusion
By investigating the effect of MOX on SCI mice, it can be concluded that the treatment with MOX promoted microbiota dysbiosis and macrophage polarization, while alleviating spinal cord injury via the down-regulation of inflammatory cytokines.
Supplementary Materials
Author Contributions
Zhuang Zhang and Dongjian Xia designed this study, Zhuang Zhang and Rubo Sui collected the literatures, Zhuang Zhang, Rubo Sui, Lili Ge and Dongjian Xia performed the experiments, Rubo Sui and Lili Ge analyzed the data, Dongjian Xia visualized the data, Zhuang Zhang and Rubo Sui composed the manuscript, Dongjian Xia reviewed and improved this study, all authors approved the submission of this final manuscript.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This study was funded by 2018 Liaoning Province Natural Science Foundation Project (Grant No. 20180550818) and 2017 Liaoning Provincial Education Department Youth Foundation Project (Grant No. JYTQN201736).
References
- 1. Brommer B, Engel O, Kopp MA, Watzlawick R, Müller S, Prüss H, Chen Y, DeVivo MJ, Finkenstaedt FW, Dirnagl U, Liebscher T, Meisel A, Schwab JM. Spinal cord injury-induced immune deficiency syndrome enhances infection susceptibility dependent on lesion level. Brain. 2016; 139:692–707. https://doi.org/10.1093/brain/awv375 [PubMed]
- 2. Meisel C, Schwab JM, Prass K, Meisel A, Dirnagl U. Central nervous system injury-induced immune deficiency syndrome. Nat Rev Neurosci. 2005; 6:775–86. https://doi.org/10.1038/nrn1765 [PubMed]
- 3. Arumugam M, Raes J, Pelletier E, Le Paslier D, Yamada T, Mende DR, Fernandes GR, Tap J, Bruls T, Batto JM, Bertalan M, Borruel N, Casellas F, et al, and MetaHIT Consortium. Enterotypes of the human gut microbiome. Nature. 2011; 473:174–80. https://doi.org/10.1038/nature09944 [PubMed]
- 4. Cervi AL, Lukewich MK, Lomax AE. Neural regulation of gastrointestinal inflammation: role of the sympathetic nervous system. Auton Neurosci. 2014; 182:83–8. https://doi.org/10.1016/j.autneu.2013.12.003 [PubMed]
- 5. Elenkov IJ, Wilder RL, Chrousos GP, Vizi ES. The sympathetic nerve--an integrative interface between two supersystems: the brain and the immune system. Pharmacol Rev. 2000; 52:595–638. [PubMed]
- 6. Koo BI, Bang TS, Kim SY, Ko SH, Kim W, Ko HY. Anorectal Manometric and Urodynamic Parameters According to the Spinal Cord Injury Lesion. Ann Rehabil Med. 2016; 40:528–33. https://doi.org/10.5535/arm.2016.40.3.528 [PubMed]
- 7. Tate DG, Forchheimer M, Rodriguez G, Chiodo A, Cameron AP, Meade M, Krassioukov A. Risk Factors Associated With Neurogenic Bowel Complications and Dysfunction in Spinal Cord Injury. Arch Phys Med Rehabil. 2016; 97:1679–86. https://doi.org/10.1016/j.apmr.2016.03.019 [PubMed]
- 8. Yi SH. Thermal properties of direct and indirect moxibustion. J Acupunct Meridian Stud. 2009; 2:273–9. https://doi.org/10.1016/S2005-2901(09)60068-6 [PubMed]
- 9. Cakmak YO. A review of the potential effect of electroacupuncture and moxibustion on cell repair and survival: the role of heat shock proteins. Acupunct Med. 2009; 27:183–6. https://doi.org/10.1136/aim.2009.001420 [PubMed]
- 10. Deng H, Shen X. The mechanism of moxibustion: ancient theory and modern research. Evid Based Complement Alternat Med. 2013; 2013:379291. https://doi.org/10.1155/2013/379291 [PubMed]
- 11. Bao CH, Wu LY, Wu HG, Shi Y, Liu HR, Zhang R, Yu LQ, Wang JH. Moxibustion inhibits apoptosis and tumor necrosis factor-alpha/tumor necrosis factor receptor 1 in the colonic epithelium of Crohn's disease model rats. Dig Dis Sci. 2012; 57:2286–95. https://doi.org/10.1007/s10620-012-2161-0 [PubMed]
- 12. Ding LL, Hu SF, He XW, Zhang P, Zhao FF, Liu TP, Zhang Q, He F, Yu Y, Xiong P, Wang CK. Acupuncture combined with moxibustion promote the recovery of spinal cord injury in correlation with Shh/Gli-1 signaling pathway. J Spinal Cord Med. 2022; 45:106–16. https://doi.org/10.1080/10790268.2020.1766900 [PubMed]
- 13. Qi Q, Liu YN, Jin XM, Zhang LS, Wang C, Bao CH, Liu HR, Wu HG, Wang XM. Moxibustion treatment modulates the gut microbiota and immune function in a dextran sulphate sodium-induced colitis rat model. World J Gastroenterol. 2018; 24:3130–44. https://doi.org/10.3748/wjg.v24.i28.3130 [PubMed]
- 14. Round JL, Mazmanian SK. The gut microbiota shapes intestinal immune responses during health and disease. Nat Rev Immunol. 2009; 9:313–23. https://doi.org/10.1038/nri2515 [PubMed]
- 15. Elinav E, Strowig T, Kau AL, Henao-Mejia J, Thaiss CA, Booth CJ, Peaper DR, Bertin J, Eisenbarth SC, Gordon JI, Flavell RA. NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell. 2011; 145:745–57. https://doi.org/10.1016/j.cell.2011.04.022 [PubMed]
- 16. Henao-Mejia J, Elinav E, Jin C, Hao L, Mehal WZ, Strowig T, Thaiss CA, Kau AL, Eisenbarth SC, Jurczak MJ, Camporez JP, Shulman GI, Gordon JI, et al. Inflammasome-mediated dysbiosis regulates progression of NAFLD and obesity. Nature. 2012; 482:179–85. https://doi.org/10.1038/nature10809 [PubMed]
- 17. Kigerl KA, Hall JC, Wang L, Mo X, Yu Z, Popovich PG. Gut dysbiosis impairs recovery after spinal cord injury. J Exp Med. 2016; 213:2603–20. https://doi.org/10.1084/jem.20151345 [PubMed]
- 18. Clarke G, Grenham S, Scully P, Fitzgerald P, Moloney RD, Shanahan F, Dinan TG, Cryan JF. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol Psychiatry. 2013; 18:666–73. https://doi.org/10.1038/mp.2012.77 [PubMed]
- 19. Bolnick DI, Snowberg LK, Hirsch PE, Lauber CL, Org E, Parks B, Lusis AJ, Knight R, Caporaso JG, Svanbäck R. Individual diet has sex-dependent effects on vertebrate gut microbiota. Nat Commun. 2014; 5:4500. https://doi.org/10.1038/ncomms5500 [PubMed]
- 20. Atri C, Guerfali FZ, Laouini D. Role of Human Macrophage Polarization in Inflammation during Infectious Diseases. Int J Mol Sci. 2018; 19:1801. https://doi.org/10.3390/ijms19061801 [PubMed]
- 21. Kigerl KA, Mostacada K, Popovich PG. Gut Microbiota Are Disease-Modifying Factors After Traumatic Spinal Cord Injury. Neurotherapeutics. 2018; 15:60–7. https://doi.org/10.1007/s13311-017-0583-2 [PubMed]
- 22. Wu HG, Zhou LB, Pan YY, Huang C, Chen HP, Shi Z, Hua XG. Study of the mechanisms of acupuncture and moxibustion treatment for ulcerative colitis rats in view of the gene expression of cytokines. World J Gastroenterol. 1999; 5:515–7. https://doi.org/10.3748/wjg.v5.i6.515 [PubMed]
- 23. Shi Y, Zhou EH, Wu HG, Zhou CL, Wang QY, Qi L. Moxibustion treatment restoring the intestinal epithelium barrier in rats with Crohn's disease by down-regulating tumor necrosis factor alpha, tumor necrosis factor receptor 1, and tumor necrosis factor receptor 2. Chin J Integr Med. 2011; 17:212–7. https://doi.org/10.1007/s11655-011-0669-3 [PubMed]
- 24. Shi Y, Qi L, Wang J, Xu MS, Zhang D, Wu LY, Wu HG. Moxibustion activates mast cell degranulation at the ST25 in rats with colitis. World J Gastroenterol. 2011; 17:3733–8. https://doi.org/10.3748/wjg.v17.i32.3733 [PubMed]
- 25. Wang XM, Lu Y, Wu LY, Yu SG, Zhao BX, Hu HY, Wu HG, Bao CH, Liu HR, Wang JH, Yao Y, Hua XG, Guo HY, Shen LR. Moxibustion inhibits interleukin-12 and tumor necrosis factor alpha and modulates intestinal flora in rat with ulcerative colitis. World J Gastroenterol. 2012; 18:6819–28. https://doi.org/10.3748/wjg.v18.i46.6819 [PubMed]
- 26. Noguchi E, Ohsawa H, Takagi K. Neural mechanism of localized changes in skeletal muscle blood flow caused by moxibustion-like thermal stimulation of anesthetized rats. J Physiol Sci. 2009; 59:421–7. https://doi.org/10.1007/s12576-009-0052-8 [PubMed]
- 27. Li X, Guo G, Shen F, Kong L, Liang F, Sun G. Moxibustion Activates Macrophage Autophagy and Protects Experimental Mice against Bacterial Infection. Evid Based Complement Alternat Med. 2014; 2014:450623. https://doi.org/10.1155/2014/450623 [PubMed]
- 28. Kogure M, Mimura N, Ikemoto H, Ishikawa S, Nakanishi-Ueda T, Sunagawa M, Hisamitsu T. Moxibustion at mingmen reduces inflammation and decreases IL-6 in a collagen-induced arthritis mouse model. J Acupunct Meridian Stud. 2012; 5:29–33. https://doi.org/10.1016/j.jams.2011.11.004 [PubMed]
- 29. Ferrante RJ, Kubilus JK, Lee J, Ryu H, Beesen A, Zucker B, Smith K, Kowall NW, Ratan RR, Luthi-Carter R, Hersch SM. Histone deacetylase inhibition by sodium butyrate chemotherapy ameliorates the neurodegenerative phenotype in Huntington's disease mice. J Neurosci. 2003; 23:9418–27. [PubMed]
- 30. Butchbach MER, Lumpkin CJ, Harris AW, Saieva L, Edwards JD, Workman E, Simard LR, Pellizzoni L, Burghes AHM. Protective effects of butyrate-based compounds on a mouse model for spinal muscular atrophy. Exp Neurol. 2016; 279:13–26. https://doi.org/10.1016/j.expneurol.2016.02.009 [PubMed]
- 31. Gungor B, Adiguzel E, Gursel I, Yilmaz B, Gursel M. Intestinal Microbiota in Patients with Spinal Cord Injury. PLoS One. 2016; 11:e0145878. https://doi.org/10.1371/journal.pone.0145878 [PubMed]
- 32. Furusawa Y, Obata Y, Fukuda S, Endo TA, Nakato G, Takahashi D, Nakanishi Y, Uetake C, Kato K, Kato T, Takahashi M, Fukuda NN, Murakami S, et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature. 2013; 504:446–50. https://doi.org/10.1038/nature12721 [PubMed]
- 33. El Aidy S, Dinan TG, Cryan JF. Gut Microbiota: The Conductor in the Orchestra of Immune-Neuroendocrine Communication. Clin Ther. 2015; 37:954–67. https://doi.org/10.1016/j.clinthera.2015.03.002 [PubMed]
- 34. Wynn TA, Chawla A, Pollard JW. Macrophage biology in development, homeostasis and disease. Nature. 2013; 496:445–55. https://doi.org/10.1038/nature12034 [PubMed]
- 35. Ginhoux F, Jung S. Monocytes and macrophages: developmental pathways and tissue homeostasis. Nat Rev Immunol. 2014; 14:392–404. https://doi.org/10.1038/nri3671 [PubMed]
- 36. Zhang SJ, Song XY, He M, Yu SB. Effect of TGF-β1/SDF-1/CXCR4 signal on BM-MSCs homing in rat heart of ischemia/perfusion injury. Eur Rev Med Pharmacol Sci. 2016; 20:899–905. [PubMed]
- 37. Mills CD, Kincaid K, Alt JM, Heilman MJ, Hill AM. M-1/M-2 macrophages and the Th1/Th2 paradigm. J Immunol. 2000; 164:6166–73. https://doi.org/10.4049/jimmunol.164.12.6166 [PubMed]
- 38. Montes VN, Turner MS, Subramanian S, Ding Y, Hayden-Ledbetter M, Slater S, Goodspeed L, Wang S, Omer M, Den Hartigh LJ, Averill MM, O'Brien KD, Ledbetter J, Chait A. T cell activation inhibitors reduce CD8+ T cell and pro-inflammatory macrophage accumulation in adipose tissue of obese mice. PLoS One. 2013; 8:e67709. https://doi.org/10.1371/journal.pone.0067709 [PubMed]
- 39. Hirayama D, Iida T, Nakase H. The Phagocytic Function of Macrophage-Enforcing Innate Immunity and Tissue Homeostasis. Int J Mol Sci. 2017; 19:92. https://doi.org/10.3390/ijms19010092 [PubMed]
- 40. Verreck FA, de Boer T, Langenberg DM, Hoeve MA, Kramer M, Vaisberg E, Kastelein R, Kolk A, de Waal-Malefyt R, Ottenhoff TH. Human IL-23-producing type 1 macrophages promote but IL-10-producing type 2 macrophages subvert immunity to (myco)bacteria. Proc Natl Acad Sci U S A. 2004; 101:4560–5. https://doi.org/10.1073/pnas.0400983101 [PubMed]
- 41. Houghton AM, Hartzell WO, Robbins CS, Gomis-Rüth FX, Shapiro SD. Macrophage elastase kills bacteria within murine macrophages. Nature. 2009; 460:637–41. https://doi.org/10.1038/nature08181 [PubMed]
- 42. Mantovani A, Sozzani S, Locati M, Allavena P, Sica A. Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 2002; 23:549–55. https://doi.org/10.1016/s1471-4906(02)02302-5 [PubMed]
- 43. Plemel JR, Wee Yong V, Stirling DP. Immune modulatory therapies for spinal cord injury--past, present and future. Exp Neurol. 2014; 258:91–104. https://doi.org/10.1016/j.expneurol.2014.01.025 [PubMed]
- 44. Boehler RM, Kuo R, Shin S, Goodman AG, Pilecki MA, Gower RM, Leonard JN, Shea LD. Lentivirus delivery of IL-10 to promote and sustain macrophage polarization towards an anti-inflammatory phenotype. Biotechnol Bioeng. 2014; 111:1210–21. https://doi.org/10.1002/bit.25175 [PubMed]
- 45. Lee SI, Jeong SR, Kang YM, Han DH, Jin BK, Namgung U, Kim BG. Endogenous expression of interleukin-4 regulates macrophage activation and confines cavity formation after traumatic spinal cord injury. J Neurosci Res. 2010; 88:2409–19. https://doi.org/10.1002/jnr.22411 [PubMed]
- 46. Sato A, Ohtaki H, Tsumuraya T, Song D, Ohara K, Asano M, Iwakura Y, Atsumi T, Shioda S. Interleukin-1 participates in the classical and alternative activation of microglia/macrophages after spinal cord injury. J Neuroinflammation. 2012; 9:65. https://doi.org/10.1186/1742-2094-9-65 [PubMed]
- 47. Gordon S, Martinez FO. Alternative activation of macrophages: mechanism and functions. Immunity. 2010; 32:593–604. https://doi.org/10.1016/j.immuni.2010.05.007 [PubMed]



