Introduction
Immune-checkpoint inhibitor (ICI) therapies have shown unprecedented durable responses in patients with advanced-stage cancers, including the success of anti-programmed cell death protein 1 (PD-1), anti-programmed death-ligand 1 (PD-L1) and anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), but the response rate is only 10%-40% [1, 2]. Therefore, it is important to identify the biomarkers that can accurately predict the ICI-therapy response.
More and more studies showed tumor mutation burden (TMB) is a clinical useful biomarker for identifying patients who benefited from ICI treatment [3, 4]. Recently, a pan-cancer study showed combining POLE and POLD1 mutation status into a simple model also can efficiently predict response to ICI therapy [5]. TP53 is one of the most frequently mutated gene in human cancers and has been formulated in a large number of studies for functions and mechanisms [6]. In brief, wild-type p53 plays a vital role in maintaining genomic stability and preventing oncogenesis by regulating many cellular processes, including promoting cell growth arrest, DNA repair, modulating autophagy and cancer metabolism [7], and TP53 is highly mutated in about 50% of human cancers. BRAF, is located on human chromosome 7 and encodes a RAS-regulated serine-threonine kinase that plays a part in ERK/MAPK signaling pathway. At the same time, the pathway is not only involved in regulating cellular biological functions, but is also related to tumor formation [8]. Up to this day, mutations in BRAF have been reported extensively in a variety of benign and malignant tumors [9, 10]. Comparing with POLE and POLD1, the mutation of TP53 and BRAF are more common in human cancer, and TP53 and BRAF had been shown to be linked to ICI therapies responses [11, 12].
In this study, we combined TP53 and BRAF mutation status into a biomarker model and found that the predict ability of TP53/BRAF mutation model is more powerful than POLE/POLD1 mutation model, and the combination of TP53/BRAF mutation model and TMB can more accurately predict the response to ICI therapy. Furthermore, we propose several possible molecular signaling pathways for the effect of TP53/BRAF mutations on the predictive value of ICI treatment response.
Materials and Methods
In this study, somatic mutation data for 46,697 patients were downloaded from cBioPortal (https://www.cbioportal.org) [13]. All nonsynonymous mutations were taken into account. The overall survival (OS) of 1,661 patients who received ICI therapy was defined from the date of the first ICI treatment to the time of death or most recent follow-up, and TMB was defined as the total number of somatic nonsynonymous mutations normalized to the total number of megabases sequenced [14].
The data of tumor-infiltrating immune cells levels for 11070 patients from TCGA by CIBERSORT14 was download from Tumor Immune Estimation Resource (TIMER) version 2.0 [15] (http://timer.cistrome.org/infiltration_estimation_for_tcga.csv.gz). The expression profiles of mRNAs and clinical survival data of 33 tumor types were obtained from the Pan-Cancer Atlas (https://gdc.cancer.gov/about-data/publications/pancanatlas).
The limma package V3.34.9 in R was used to identify differentially expressed mRNAs. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses were identified and visualized using R packages “clusterProfiler”. The cBioPortal online analysis tool was used for mutual exclusivity analysis between TP53 mutation and BRAF mutation. For survival analysis, Kaplan-Meier survival curves were generated and compared using the log-rank test, and the Cox regression model was used for multivariate survival analysis. Statistical tests were done with R software (version 4.0.2). Statistical significance was set at p values less than 0.05. Ethical approval was waived because we used only publicly available data and materials in this study.
Availability of data and materials
The datasets presented in this study can be found in online repositories. The names of the repositories and accession numbers can be found in the article material.
Results
TP53/BRAF mutation model has high frequency
The prevalence of TP53 and BRAF mutations in 46,697 patients with different cancer types is summarized in Figure 1. The mutation frequencies of TP53 and BRAF (33.51% and 5.30%) were significantly higher than that of POLE and POLD1 (2.74% and 1.45%). The relationships between TP53 mutation and BRAF mutation are mutually exclusive (Table 1).
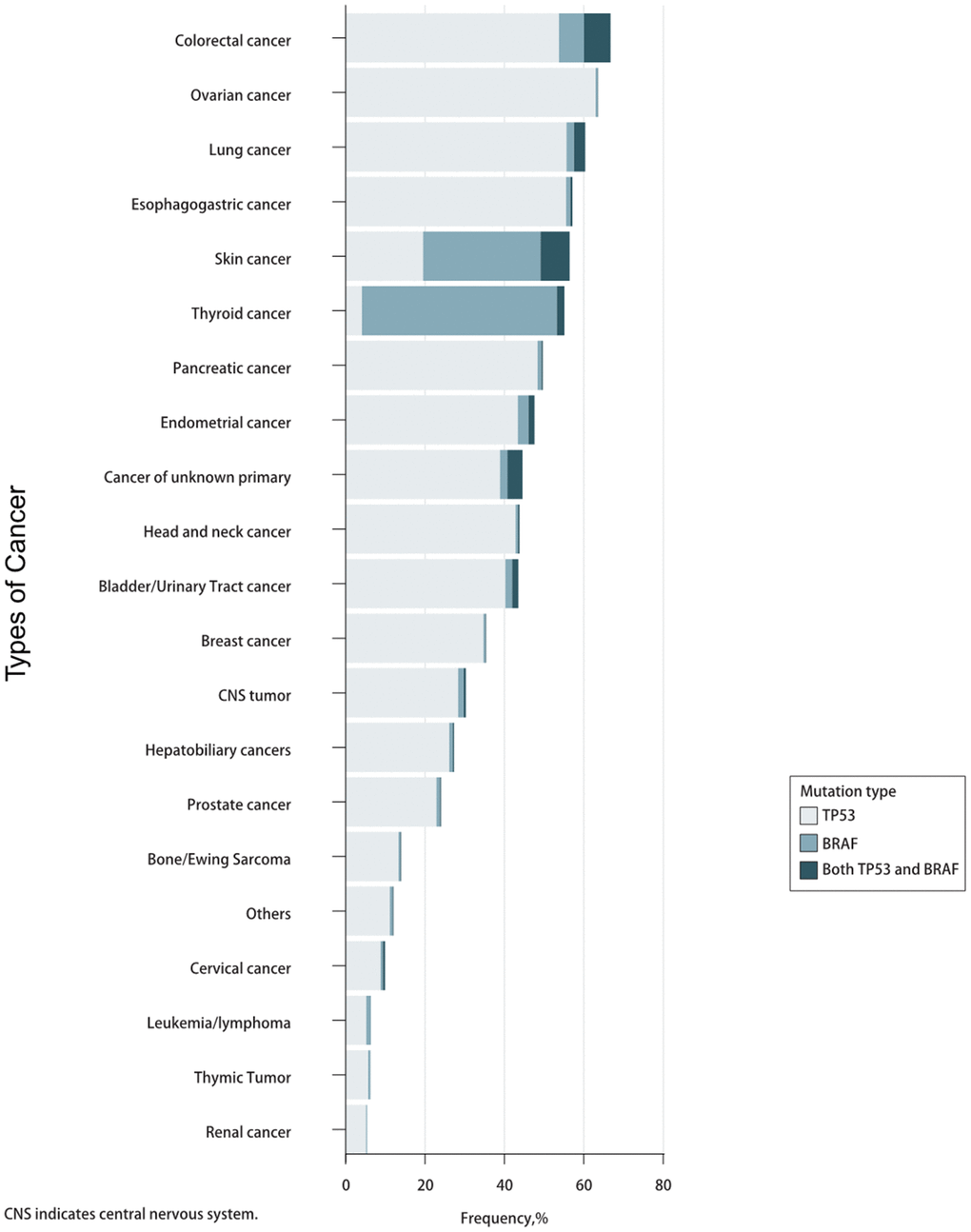
Figure 1. Prevalence of TP53/BRAF mutations in pan-cancer.
Table 1. Mutual exclusivity analysis between TP53 mutation and BRAF mutation in the whole cohort, TCGA subset, and MSKCC subset.
| Cohorts | TP53WT BRAFWT | TP53MUT BRAFWT | BRAFMUT TP53WT | TP53MUT BRAFMUT | Log2 odds ratio | p-value | q-value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Whole cohort | 27745 | 13519 | 1642 | 646 | -0.309 | <0.001 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TCGA subset | 5683 | 3661 | 636 | 209 | -0.971 | <0.001 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSKCC subset | 798 | 691 | 119 | 53 | -0.959 | <0.001 | <0.001 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TCGA, The Cancer Genome Atlas; MSKCC, Memorial Sloan Kettering Cancer Center. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
TP53/BRAF mutation model can predict immunotherapeutic effect and prognosis of patients
Based on the mutually exclusive relationship between TP53 mutation and BRAF mutation, we explored the immunotherapy response in patients with different combinations of TP53 mutation and BRAF mutation. Patients were divided into four genotypes, patients with BRAF mutation alone (BRAFMUTTP53WT) showed favorable survival (Median, 47 months), while those with TP53 mutation alone (TP53MUTBRAFWT) had the worst survival (Median, 13 months). Patients with both mutations or neither mutation (TP53MUTBRAFMUT or TP53WTBRAFWT) showed moderate survival (Median, 27 months and 20 months, respectively) (Figure 2A).
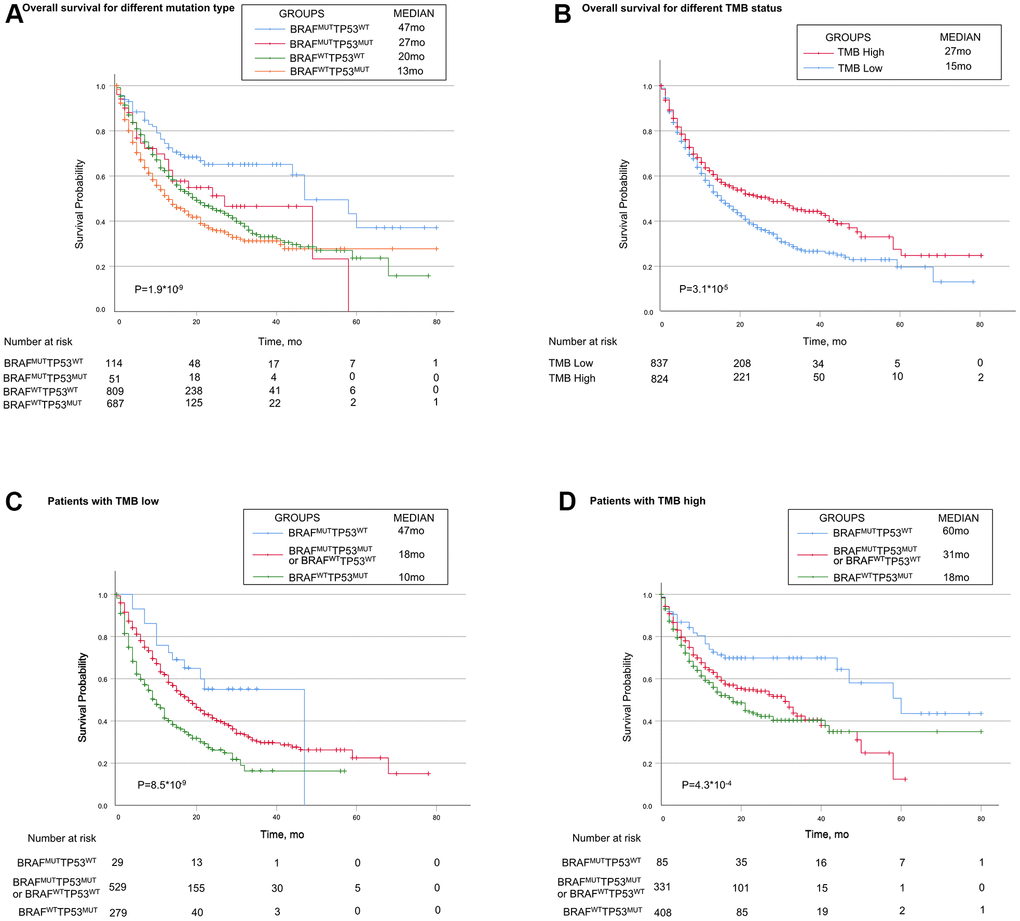
Figure 2. Associations of TP53 and BRAF mutation types with prognosis in patients treated with immune checkpoint inhibitors. (A) Patients with the BRAF mutation alone had the best prognosis, while patients with TP53 mutation alone had the worst prognosis. Patients with mutations in both or none had median survival. (B) Patients in high-TMB status group had longer OS than patients in low-TMB status group. (C, D) In both high-TMB/low-TMB status groups, TP53MUTBRAFWT indicated poorer OS, while BRAFMUTTP53WT did the opposite. BRAF indicates B-Raf Proto-Oncogene, Serine/Threonine Kinase gene; TP53 indicates tumor protein p53 gene; MUT indicates mutant genes; WT indicates wild type genes; TMB indicates tumor mutation burden; MSI indicates microsatellite instable.
In multivariable Cox regression analysis, TP53/BRAF mutation model and TMB were independent predictive factors for identifying patients who benefited from ICI treatment (both P<0.0001). However, POLE/POLD1 mutation model and MSI were not independent predictive factors (both P>0.05) (Table 2).
Table 2. Univariate and multivariable association of the TP53/BRAF mutation model with overall survival in 1,661 patients who received ICI therapy.
| Parameters | Univariate | Multivariable | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR (95%CI) | p value | HR (95%CI) | p value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender | 0.88 (0.77-1.01) | 0.078 | 0.89 (0.77-1.02) | 0.09 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 1.00 (0.99-1.00) | 0.071 | 1.00 (0.99-1.00) | 0.449 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| POLE/POLD1 mutation model | 0.62 (0.45-0.84) | 0.002 | 0.87 (0.63-1.21) | 0.399 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TMB | 0.98 (0.98-0.99) | <0.0001 | 0.98 (0.97-0.99) | <0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MSI | 0.98 (0.97-1.00) | 0.044 | 1.01 (0.99-1.03) | 0.235 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cancer type | 0.95 (0.93-0.98) | <0.0001 | 0.96 (0.94-0.98) | 0.0003 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TP53/BRAF mutation model | 1.41 (1.26-1.58) | <0.0001 | 1.42 (1.26-1.60) | <0.0001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TMB, tumor mutation burden; MSI, microsatellite instable. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Patients in high-TMB status group (the median TMB as cutoff) had longer OS than patients in low-TMB status group (median, 27 months vs 15 month; P =0.000031, Figure 2B). When stratified by TMB status, TP53/BRAF mutation model was still a statistically significant model for predicting ICI-therapy response. In both high-TMB status group and low-TMB status group, TP53/BRAF mutation model can successfully divide patients into three risk stratification: good response genotype (BRAFMUTTP53WT), intermediate response genotype (TP53MUTBRAFMUT or TP53WTBRAFWT), and poor response genotype (TP53MUTBRAFWT) (Figure 2C, 2D). In addition, the TP53/BRAF mutation model remained a statistically significant model when stratified according to patients' clinical information. Regardless of gender or age, TP53/BRAF mutation model can still classify patients into three risk stratification (Supplementary Figure 1A–1D).
We further performed MSI analysis and found that the low-MSI status group had a better prognosis (median, 19 months vs 15 month; P=0.0095, Supplementary Figure 1E). Exactly like the TMB model, MSI status can also stratify patients with mutated genetic risk (Supplementary Figure 1F, 1G).
Mechanism prediction of TP53/BRAF mutation model
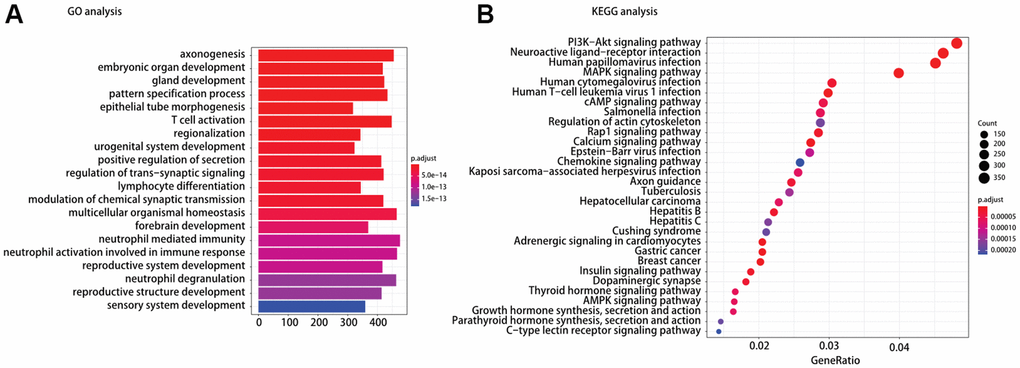
To understand the mechanism of oncogenesis underlying TP53/BRAF mutation correlates with response to ICI, functional enrichment characterization of different expression mRNAs between TP53MUTBRAFWT and BRAFMUTTP53WT was performed by GO and KEGG analysis. According to GO analysis, we found that the enriched GO terms were including T cell activation and lymphocyte differentiation. Moreover, KEGG pathway analysis indicated that most of different expression mRNAs were involved in PI3K−Akt signaling pathway, MAPK signaling pathway, Rap1 signaling pathway, chemokine signaling pathway, and AMPK signaling pathway in cancer (Figure 4).
Discussion
Up to this day, ICI therapies have shown powerful responses in cancer patients. However, the rate is not ideal enough, and the methods have the potential to play a greater role in the clinic. It’s critical to build more effective biomarker models and stratify the patients for predicting prognosis and applying better individualized treatments.
In the POLE/POLD1 mutation model, patients with either POLE or POLD1 mutations was associated with better ICI therapy response and longer OS than the wild-type population (34months vs 18months) [5]. However, the POLE/POLD1 mutation model was not a significant predictive factor for ICI therapy response after multivariable adjustment of TMB and TP53/BRAF mutation model. TP53/BRAF mutation model was a powerful and independent predictive factor for identifying patients who benefited from ICI treatment. In advanced tumors, TP53 and BRAF mutations are more common than POLE and POLD1 mutations, and TP53/BRAF mutation model is better than POLE/POLD1 mutation model in predicting ICI treatment response.
The biological implications of a mutually exclusive TP53 mutation and BRAF mutation are not understood at present. As mentioned above, TP53 is a tumor suppressor gene involved in the regulation of cell growth [19], BRAF is an oncogene involved in cellular responses to growth signals [20]. Missense mutations, insertions or deletions of TP53 lead to TP53 inactivation are very common. BRAF mutations, such as BRAF V600E mutations, cause the continuous activation of the downstream MEK-ERK signaling pathway [21]. In this study, concurrent TP53 mutation and BRAF mutation was seen in a small number of patients. Tumors carrying both TP53 mutations and BRAF mutations are less likely to response to ICI therapy than those showing only BRAF mutation. This could account for the TP53 inactivation and BRAF activation might be genetically redundant, and that alteration in both genes does not confer a further advantage.
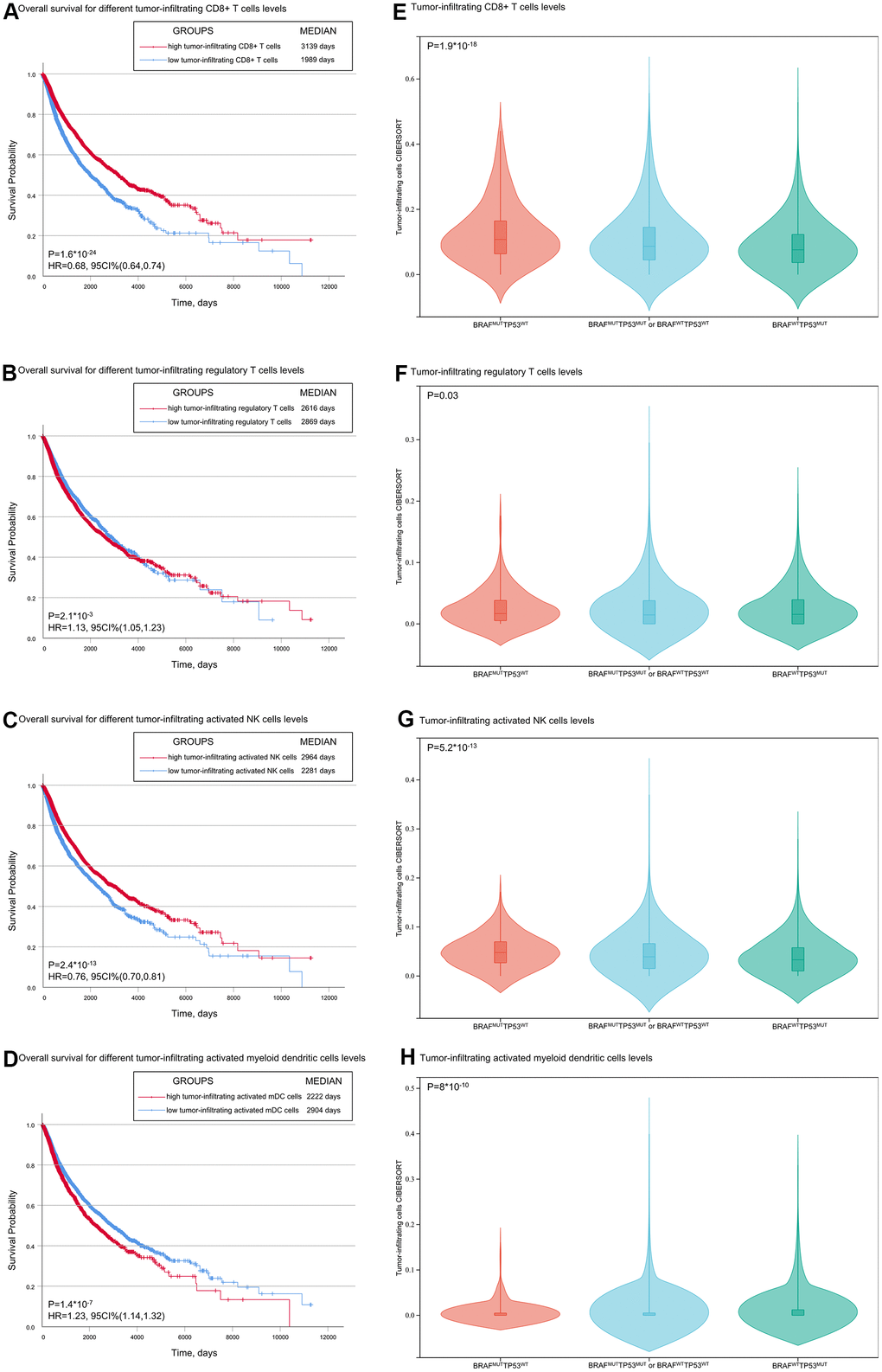
The molecular mechanisms explaining the effects of TP53/BRAF mutation on predictive value for ICI therapy response are presently unknown. Previously, we have shown statistically that the level of tumor-infiltrating CD8+ T cells is correlated with TP53/BRAF mutations, which may be one of the causes. According to KEGG analysis, we found five enriched signaling pathways closely related to tumor immunity. The PI3K−Akt signaling pathway plays a critical role in T and B cell development [22, 23]. The BRAF-MAPK signaling pathway correlates with the production of various immunosuppressive factors in regulating cancer-immune evasion [24]. The Rap1 signaling pathway activation leads to increased integrin affinity, leukocytes arrest rolling and actively lymphocyte migration and adhesion [25–27]. Chemokines signaling pathway are key molecules involved in the migration and homeostasis of immune cells [28]. The AMPK signaling pathway is involved in shaping the activity of lymphocytes [29, 30]. The above pathways may explain potential reasons why TP53/BRAF mutation of cancer patients contributes to the ICI therapy response. More detailed and specific studies are needed to elucidate the precise molecular mechanisms.
In this study, we show that a novel TP53/BRAF mutation model provides significant information about the stratification of response to ICI-therapy. Commonly, patients with high-TMB status have better response to ICI therapy than patients with low-TMB status. However, the genotype of TP53MUTBRAFWT in high-TMB status cohort have poorer response to ICI therapy than the genotype of BRAFMUTTP53WT in low-TMB status (Median, 18 months vs 47 month). Thus, TP53/BRAF mutation model can add predictive value to TMB in identifying patients who benefited from ICI treatment, which can enable more informed treatment decisions.
Conclusions
we combined TP53 and BRAF mutation status into a biomarker model which owns the ability to be more efficient than the POLE/POLD1 mutation model, and the combination of TP53/BRAF mutation model and TMB can more accurately predict the response to ICI therapy.
Supplementary Materials
Author Contributions
Dr Luo and Dr Wei had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Drs Yao, Liu, and Cao contributed equally to this work. Concept and design: Yao, Liu, Wei, Luo. Acquisition, analysis, or interpretation of data: All authors. Drafting of the manuscript: Yao, Liu, Cao, Tang, Li, Wei. Critical revision of the manuscript for important intellectual content: Wei, Luo. Statistical analysis: Wei, Feng, Luo. Obtained funding: Wei, Luo. Administrative, technical, or material support: Liu, Cao, Tang, Li, Feng. Supervision: Luo.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was supported by grants from National Natural Science Foundation of China (Award Number: 81725016, 81872094, 81602219), Natural Science Foundation of Guangdong Province (Award Number: 2017B020227004, 2017A030313538).
References
- 1. Sharma P, Hu-Lieskovan S, Wargo JA, Ribas A. Primary, Adaptive, and Acquired Resistance to Cancer Immunotherapy. Cell. 2017; 168:707–23. https://doi.org/10.1016/j.cell.2017.01.017 [PubMed]
- 2. Zou W, Wolchok JD, Chen L. PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci Transl Med. 2016; 8:328rv4. https://doi.org/10.1126/scitranslmed.aad7118 [PubMed]
- 3. Samstein RM, Lee CH, Shoushtari AN, Hellmann MD, Shen R, Janjigian YY, Barron DA, Zehir A, Jordan EJ, Omuro A, Kaley TJ, Kendall SM, Motzer RJ, et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat Genet. 2019; 51:202–6. https://doi.org/10.1038/s41588-018-0312-8 [PubMed]
- 4. Chan TA, Yarchoan M, Jaffee E, Swanton C, Quezada SA, Stenzinger A, Peters S. Development of tumor mutation burden as an immunotherapy biomarker: utility for the oncology clinic. Ann Oncol. 2019; 30:44–56. https://doi.org/10.1093/annonc/mdy495 [PubMed]
- 5. Wang F, Zhao Q, Wang YN, Jin Y, He MM, Liu ZX, Xu RH. Evaluation of POLE and POLD1 Mutations as Biomarkers for Immunotherapy Outcomes Across Multiple Cancer Types. JAMA Oncol. 2019; 5:1504–6. https://doi.org/10.1001/jamaoncol.2019.2963 [PubMed]
- 6. Levine AJ. p53: 800 million years of evolution and 40 years of discovery. Nat Rev Cancer. 2020; 20:471–80. https://doi.org/10.1038/s41568-020-0262-1 [PubMed]
- 7. Kastenhuber ER, Lowe SW. Putting p53 in Context. Cell. 2017; 170:1062–78. https://doi.org/10.1016/j.cell.2017.08.028 [PubMed]
- 8. Rubinfeld H, Seger R. The ERK cascade: a prototype of MAPK signaling. Mol Biotechnol. 2005; 31:151–74. https://doi.org/10.1385/MB:31:2:151 [PubMed]
- 9. Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S, Teague J, Woffendin H, Garnett MJ, Bottomley W, Davis N, Dicks E, Ewing R, et al. Mutations of the BRAF gene in human cancer. Nature. 2002; 417:949–54. https://doi.org/10.1038/nature00766 [PubMed]
- 10. Choueiri TK, Cheville J, Palescandolo E, Fay AP, Kantoff PW, Atkins MB, McKenney JK, Brown V, Lampron ME, Zhou M, Hirsch MS, Signoretti S. BRAF mutations in metanephric adenoma of the kidney. Eur Urol. 2012; 62:917–22. https://doi.org/10.1016/j.eururo.2012.05.051 [PubMed]
- 11. Long J, Wang A, Bai Y, Lin J, Yang X, Wang D, Yang X, Jiang Y, Zhao H. Development and validation of a TP53-associated immune prognostic model for hepatocellular carcinoma. EBioMedicine. 2019; 42:363–74. https://doi.org/10.1016/j.ebiom.2019.03.022 [PubMed]
- 12. Mazieres J, Drilon A, Lusque A, Mhanna L, Cortot AB, Mezquita L, Thai AA, Mascaux C, Couraud S, Veillon R, Van den Heuvel M, Neal J, Peled N, et al. Immune checkpoint inhibitors for patients with advanced lung cancer and oncogenic driver alterations: results from the IMMUNOTARGET registry. Ann Oncol. 2019; 30:1321–8. https://doi.org/10.1093/annonc/mdz167 [PubMed]
- 13. Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, Cerami E, Sander C, Schultz N. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. 2013; 6:pl1. https://doi.org/10.1126/scisignal.2004088 [PubMed]
- 14. Newman AM, Liu CL, Green MR, Gentles AJ, Feng W, Xu Y, Hoang CD, Diehn M, Alizadeh AA. Robust enumeration of cell subsets from tissue expression profiles. Nat Methods. 2015; 12:453–7. https://doi.org/10.1038/nmeth.3337 [PubMed]
- 15. Li T, Fu J, Zeng Z, Cohen D, Li J, Chen Q, Li B, Liu XS. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020; 48:W509–14. https://doi.org/10.1093/nar/gkaa407 [PubMed]
- 16. Snyder A, Makarov V, Merghoub T, Yuan J, Zaretsky JM, Desrichard A, Walsh LA, Postow MA, Wong P, Ho TS, Hollmann TJ, Bruggeman C, Kannan K, et al. Genetic basis for clinical response to CTLA-4 blockade in melanoma. N Engl J Med. 2014; 371:2189–99. https://doi.org/10.1056/NEJMoa1406498 [PubMed]
- 17. Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS, Miller ML, Rekhtman N, Moreira AL, et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science. 2015; 348:124–8. https://doi.org/10.1126/science.aaa1348 [PubMed]
- 18. McGranahan N, Furness AJ, Rosenthal R, Ramskov S, Lyngaa R, Saini SK, Jamal-Hanjani M, Wilson GA, Birkbak NJ, Hiley CT, Watkins TB, Shafi S, Murugaesu N, et al. Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science. 2016; 351:1463–9. https://doi.org/10.1126/science.aaf1490 [PubMed]
- 19. Aubrey BJ, Strasser A, Kelly GL. Tumor-Suppressor Functions of the TP53 Pathway. Cold Spring Harb Perspect Med. 2016; 6:a026062. https://doi.org/10.1101/cshperspect.a026062 [PubMed]
- 20. Pratilas CA, Solit DB. Therapeutic strategies for targeting BRAF in human cancer. Rev Recent Clin Trials. 2007; 2:121–34. https://doi.org/10.2174/157488707780599393 [PubMed]
- 21. Kotani H, Adachi Y, Kitai H, Tomida S, Bando H, Faber AC, Yoshino T, Voon DC, Yano S, Ebi H. Distinct dependencies on receptor tyrosine kinases in the regulation of MAPK signaling between BRAF V600E and non-V600E mutant lung cancers. Oncogene. 2018; 37:1775–87. https://doi.org/10.1038/s41388-017-0035-9 [PubMed]
- 22. Lucas CL, Chandra A, Nejentsev S, Condliffe AM, Okkenhaug K. PI3Kδ and primary immunodeficiencies. Nat Rev Immunol. 2016; 16:702–14. https://doi.org/10.1038/nri.2016.93 [PubMed]
- 23. Sobral-Leite M, Salomon I, Opdam M, Kruger DT, Beelen KJ, van der Noort V, van Vlierberghe RL, Blok EJ, Giardiello D, Sanders J, Van de Vijver K, Horlings HM, Kuppen PJ, et al. Cancer-immune interactions in ER-positive breast cancers: PI3K pathway alterations and tumor-infiltrating lymphocytes. Breast Cancer Res. 2019; 21:90. https://doi.org/10.1186/s13058-019-1176-2 [PubMed]
- 24. Sumimoto H, Imabayashi F, Iwata T, Kawakami Y. The BRAF-MAPK signaling pathway is essential for cancer-immune evasion in human melanoma cells. J Exp Med. 2006; 203:1651–6. https://doi.org/10.1084/jem.20051848 [PubMed]
- 25. Shimonaka M, Katagiri K, Nakayama T, Fujita N, Tsuruo T, Yoshie O, Kinashi T. Rap1 translates chemokine signals to integrin activation, cell polarization, and motility across vascular endothelium under flow. J Cell Biol. 2003; 161:417–27. https://doi.org/10.1083/jcb.200301133 [PubMed]
- 26. Katagiri K, Maeda A, Shimonaka M, Kinashi T. RAPL, a Rap1-binding molecule that mediates Rap1-induced adhesion through spatial regulation of LFA-1. Nat Immunol. 2003; 4:741–8. https://doi.org/10.1038/ni950 [PubMed]
- 27. Katagiri K, Imamura M, Kinashi T. Spatiotemporal regulation of the kinase Mst1 by binding protein RAPL is critical for lymphocyte polarity and adhesion. Nat Immunol. 2006; 7:919–28. https://doi.org/10.1038/ni1374 [PubMed]
- 28. Krieg C, Boyman O. The role of chemokines in cancer immune surveillance by the adaptive immune system. Semin Cancer Biol. 2009; 19:76–83. https://doi.org/10.1016/j.semcancer.2008.10.011 [PubMed]
- 29. Notarangelo LD. Combined immunodeficiencies with nonfunctional T lymphocytes. Adv Immunol. 2014; 121:121–90. https://doi.org/10.1016/B978-0-12-800100-4.00004-0 [PubMed]
- 30. Wang J, Li Z, Gao L, Qi Y, Zhu H, Qin X. The regulation effect of AMPK in immune related diseases. Sci China Life Sci. 2018; 61:523–33. https://doi.org/10.1007/s11427-017-9169-6 [PubMed]