Introduction
Liver cancer is a prevalent malignancy and the principal reason for tumor mortality globally, in which hepatocellular carcinoma (HCC) depicts 70–85% of the entire liver carcinoma weight [1]. Although there have been recent improvements, most HCC cases are identified at an advanced stage, resulting in bad outcomes and a considerable recurrence frequency [2]. Though previous investigations have explored the unusual expression of various proteins in HCC, the mechanism of HCC development is still mostly undiscovered [3]. Autophagy is a lysosome-related program and represents an intricate function during tumorigenesis [4]. When denied growth factors, nutrients, and oxygen, cancer cells sustain their durability by autophagy-associated degeneration of damaged organelles and misfolded proteins [5]. As the previous studies, autophagy is a crucial process during liver cancer development and a potential therapeutic target for liver cancer therapy (Allaire, 2019 #35; Huang, 2018 #34; Tang, 2019 #33), but the mechanisms are poorly understood.
MicroRNAs (miRNAs) are extensively expressed in many species, including viruses, plants, and animals [6, 7]. MiRNAs serve the non-coding, endogenous, and small regulators and negatively modulate the targeted genes' mRNA 3′-untranslated region (3′-UTR) by causing mRNAs degeneration suppressing protein translation [8–10]. Multiple miRNAs are reported to participate in the regulation of liver cancer [11, 12]. Meanwhile, miR-513b-5p has been reported to inhibit progression of embryonal carcinoma and lung cancer [13, 14]. Moreover, it has been uncovered that miR-511 reduces migration, invasion, and proliferation of HCC cells by targeting phosphoinositide-3-kinase regulatory subunit 3 [15]. MiR-132 represses migration, invasion, and proliferation of HCC cells through down-regulating PIK3R3 [16]. PIK3R3 acts as an oncogene of various cancers, containing glioma, lung cancer, and gastric cancer [17–19]. However, the impact of miR-513b-5p on PIK3R3 in HCC is still unreported. As several miRNAs are involved in the modulation of autophagy in HCC and based on the crucial role of miR-513b-5p in cancer development, we selected miR-513b-5p as an example to evaluate its function in autophagy during liver cancer progression.
In the present study, we were interested in the miR-513b-5p function in the modulation of autophagy during liver cancer progression. We demonstrated that miR-513b-5p attenuated autophagy during the malignant progression of liver cancer by targeting PIK3R3.
Results
MiR-513b-5p inhibits autophagy in liver cancer cells
Firstly, we analyzed the expression of miR-513b-5p in the normal liver LO2 cells and liver cancer cells, including H7402, HCCLM3, HepG2 and Huh-7 cells. We observed that we miR-513b-5p was decreased in the liver cancer cells compared with the normal liver cells (Figure 1A). We then were interested in the function of miR-513b-5p in the regulation of autophagy in HCC, we analyzed the autophagy related markers, such as LC3, beclin1, and p62 in HCC cells. For this purpose, the liver cancer cell lines, including HepG2 and Huh-7 cells, were treated with miR-513b-5p and the expression of miR-513b-5p was enhanced in the cells (Figure 1B). Functionally, the treatment of miR-513b-5p mimic repressed the LC3-II and beclin1 expression but induced p62 expression in the HepG2, Huh-7, and HCCLM3 cells (Figure 1C–1F, Supplementary Figure 1). Taken together, these results indicate that miR-513b-5p inhibits autophagy in liver cancer cells.
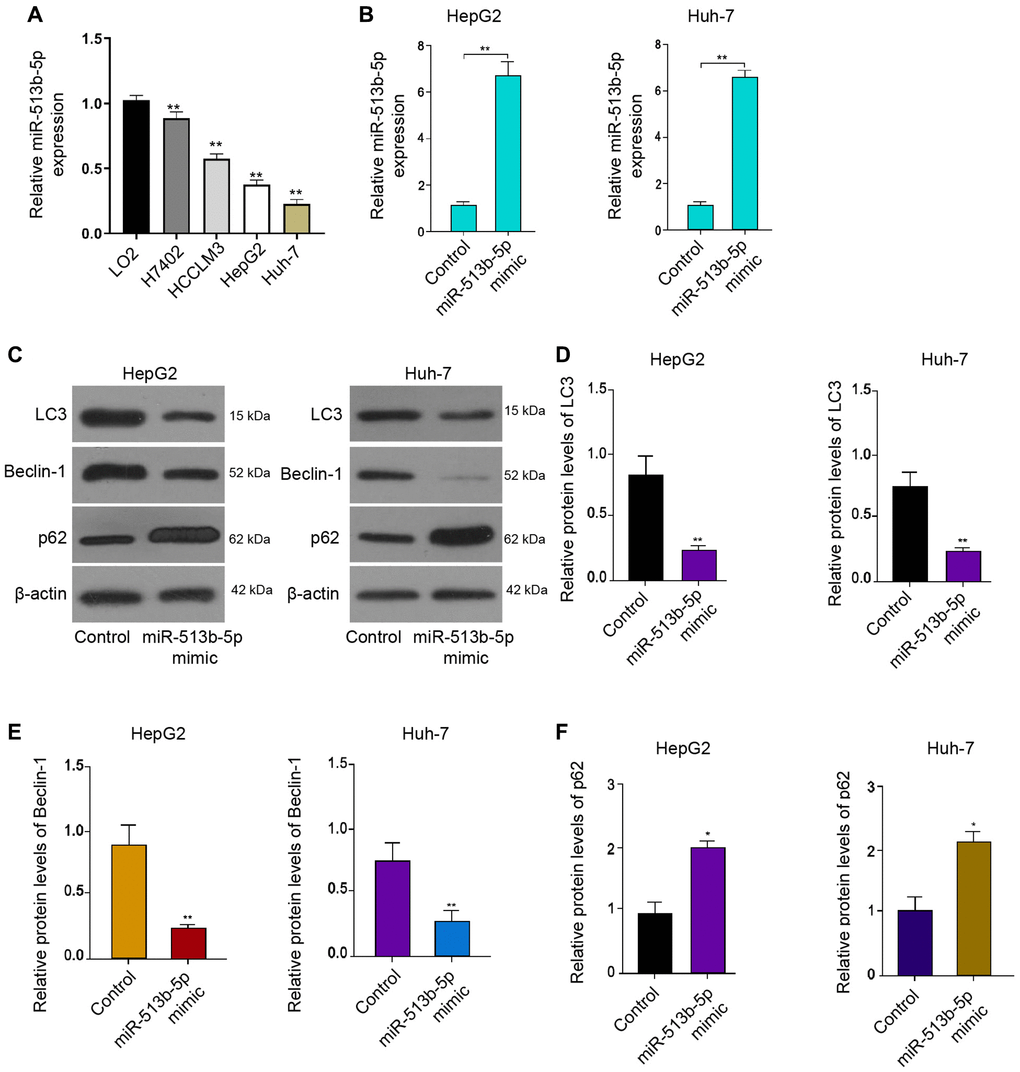
Figure 1. MiR-513b-5p inhibits autophagy in liver cancer cells. (A) The measurement of miR-513b-5p expression using qPCR. (B–F) The HepG2 and Huh-7 cells were treated with miR-513b-5p mimic. (B) The measurement of miR-513b-5p expression using qPCR. (C–F) The detection of LC3, beclin1, and p62 expression using Western blot analysis.
MiR-513b-5p represses liver cancer cell proliferation in vitro
We then further explored the effect of miR-513b-5p on HCC cell proliferation in vitro, to this end, we performed MTT assays and colony formation assays in the HepG2, Huh-7, and HCCLM3 cells. The treatment of miR-513b-5p suppressed the cell viability in the HepG2, Huh-7, and HCCLM3 cells (Figure 2A and 2B, Supplementary Figure 2A). Similarly, the colony formation ability of HepG2, Huh-7, and HCCLM3 cells was attenuated by miR-513b-5p (Figure 2C and 2D, Supplementary Figure 2B). Collectively, it suggests that miR-513b-5p represses liver cancer cell proliferation in vitro.
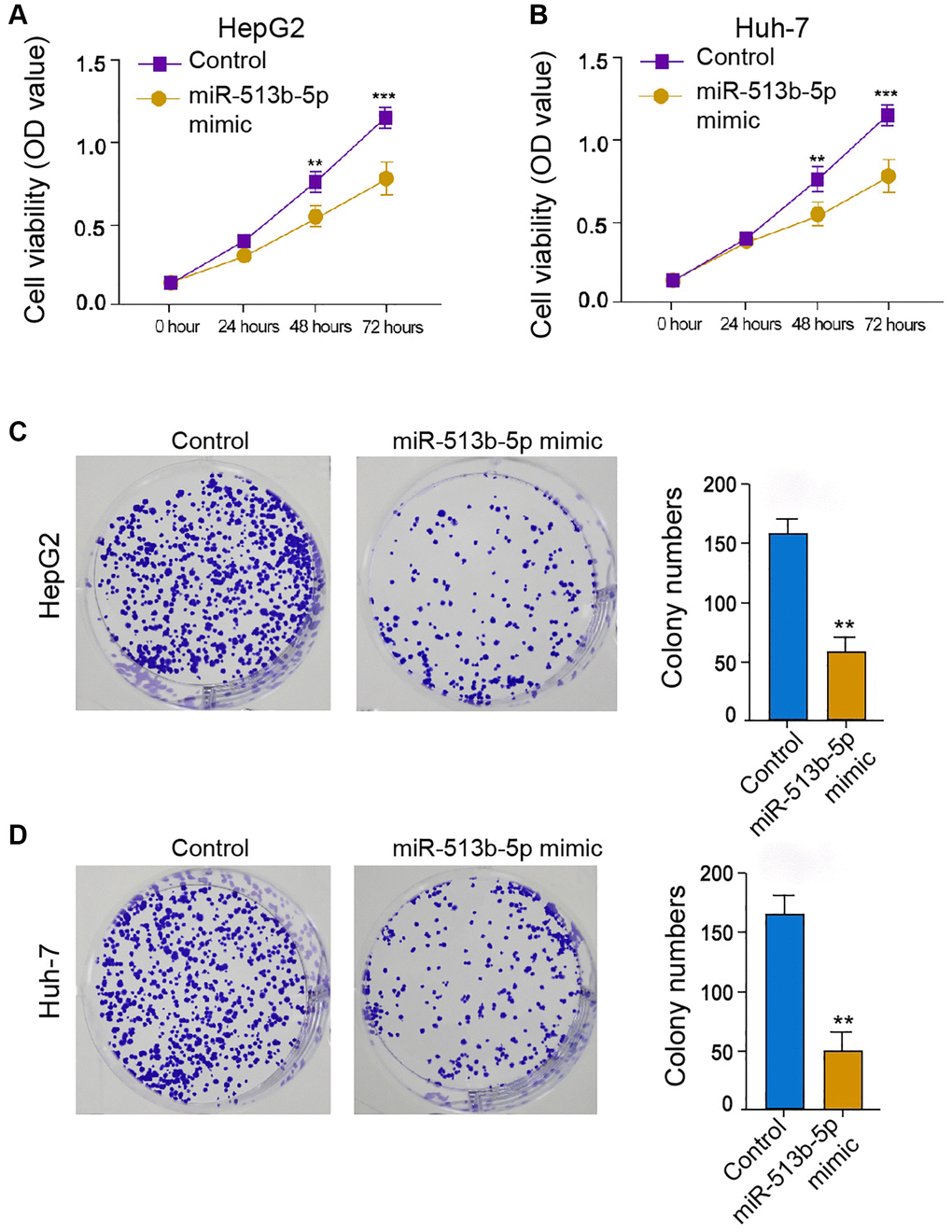
Figure 2. MiR-513b-5p represses liver cancer cell proliferation in vitro. (A–D) The HepG2 and Huh-7 cells were treated with miR-513b-5p mimic. (A and B) The analysis of cell proliferation using MTT assays. (C and D) The analysis of cell proliferation using colony formation assays.
MiR-513b-5p reduces tumor growth of liver cancer cells in vivo
We then further assessed the role of miR-513b-5p during tumor growth of HepG2 cells in vivo. Interestingly, the treatment of miR-513b-5p mimic remarkably alleviated the tumor growth phenotypes, including tumor size, tumor weight, and tumor volume (Figure 3A–3C). These data indicate that miR-513b-5p reduces tumor growth of liver cancer cells in vivo.
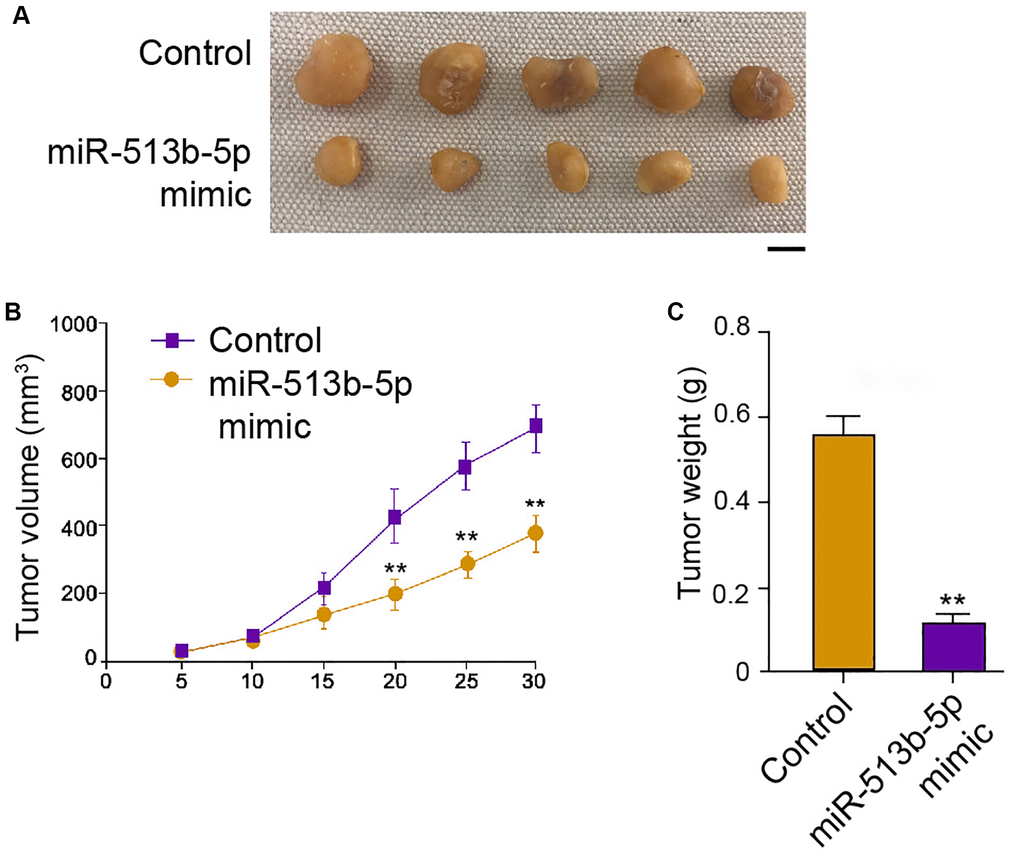
Figure 3. MiR-513b-5p reduces tumor growth of liver cancer cells in vivo. (A–C) The analysis of tumor growth of HepG2 cells treated with miR-513b-5p mimic using tumorigenicity assays in the nude mice. The tumor size (Scale bar = 10 mm), tumor weight, and tumor volume were shown.
MiR-513b-5p suppresses migration/invasion and enhances apoptosis of liver cancer cells in vitro
Given that the migration, invasion, and apoptosis are the crucial phenotype of cancer progression, we then concerned about the impact of miR-513b-5p on liver cancer cells migration/invasion and apoptosis in vitro. We found that miR-513b-5p mimic significantly restrained the migration/invasion capability of HepG2, Huh-7, and HCCLM3 cells (Figure 4A and 4B, Supplementary Figure 3A). Moreover, HepG2, Huh-7, and HCCLM3 cells apoptosis was induced in the miR-513b-5p mimic-treated cells (Figure 4C and 4D, Supplementary Figure 3B). Taken together, these data indicate that miR-513b-5p suppresses migration/invasion and enhances apoptosis of liver cancer cells in vitro.
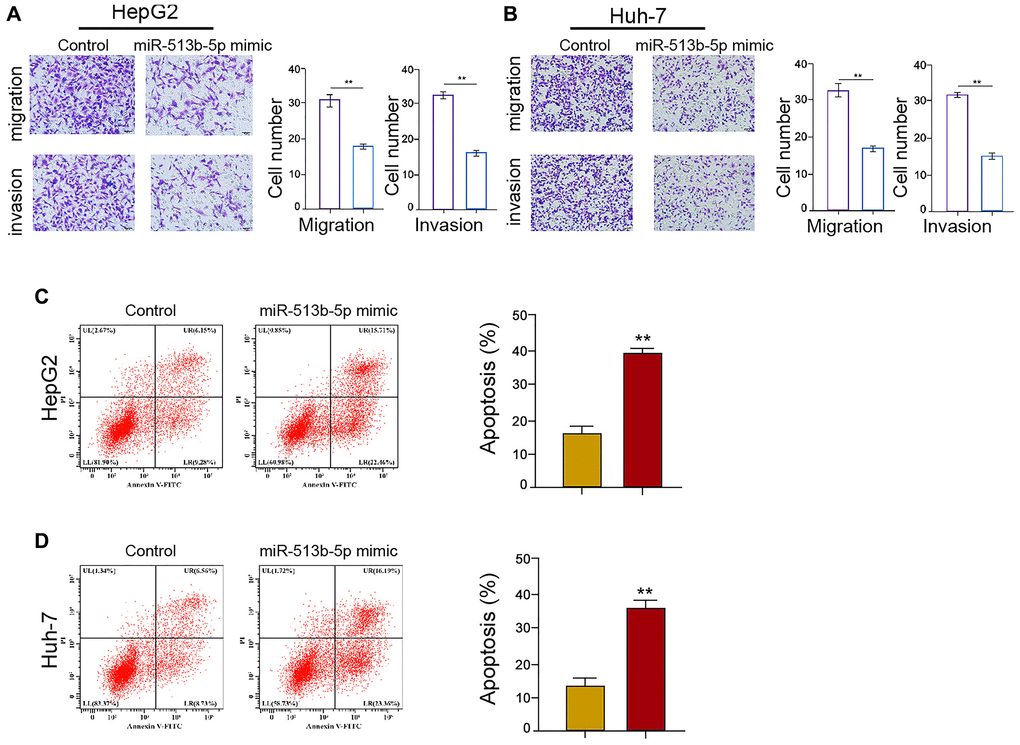
Figure 4. MiR-513b-5p suppresses migration/invasion and enhances apoptosis of liver cancer cells in vitro. (A–D) The HepG2 and Huh-7 cells were treated with miR-513b-5p mimic. (A and B) The analysis of cell migration/invasion using transwell assays. (C and D) The analysis of cell apoptosis using flow cytometry.
PIK3R3 is targeted by miR-513b-5p in liver cancer cells
Next, we tried to explore the potential mechanism underlying miR-513b-5p-mediated HCC progression. The predicted analysis demonstrated the potential binding of miR-513b-5p with PIK3R3 mRNA 3′UTR (Figure 5A). MiR-513b-5p mimic remarkably repressed the luciferase activities of PIK3R3 mRNA 3′UTR in the HepG2, Huh-7, and HCCLM3 cells (Figure 5B and 5C, Supplementary Figure 4). Both of the mRNA and protein levels of PIK3R3 were down-regulated by miR-513b-5p mimic in the HepG2, Huh-7, and HCCLM3 cells (Figure 5D–5F, Supplementary Figure 3B). Collectively, it indicates that PIK3R3 is targeted by miR-513b-5p in liver cancer cells.
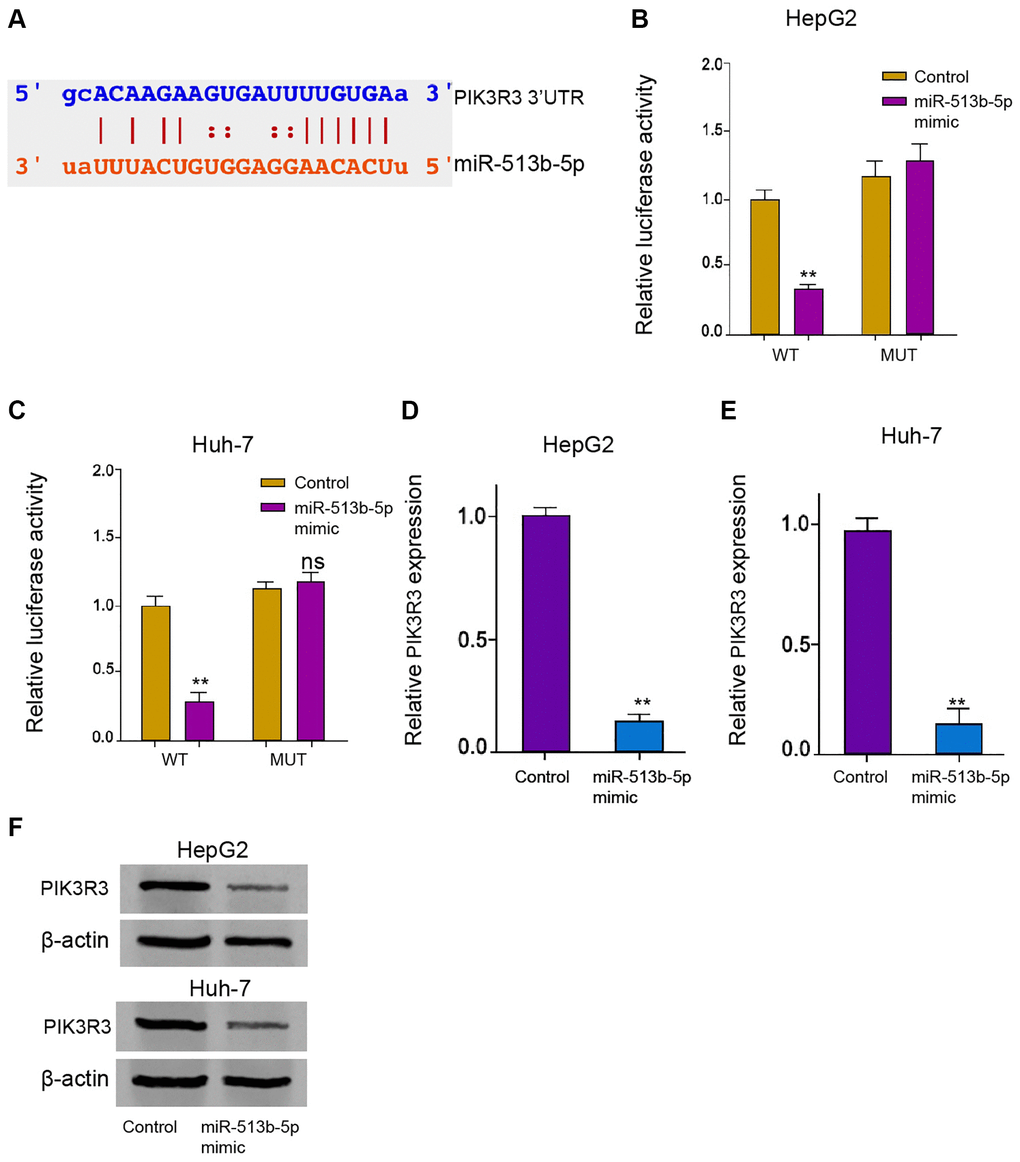
Figure 5. PIK3R3 is targeted by miR-513b-5p in liver cancer cells. (A) The interaction prediction analysis of miR-513b-5p with PIK3R3 mRNA 3′UTR using ENCORI online database. (B–E) The HepG2 and Huh-7 cells were treated with miR-513b-5p mimic. (B and C) The analysis of luciferase activities using luciferase reporter gene assays. (D and E) The analysis of PIK3R3 mRNA expression using qPCR. (F) The detection of PIK3R3 expression using Western blot analysis.
PIK3R3 is involved in miR-513b-5p-inhibited autophagy liver cancer cells
Furthermore, the treatment of miR-513b-5p mimic repressed the LC3-II expression but induced p62 expression in the HepG2 and Huh-7 cells, in which the PIK3R3 overexpression partly reversed this effect in the cells (Figure 6A and 6B). Meanwhile, PIK3R3 overexpression partly rescued the cell proliferation and blocked the cell apoptosis, which were mediated by miR-513b-5p mimic in the HepG2, Huh-7, and HCCLM3 cells (Figure 6C–6F, Supplementary Figure 5), implying that PIK3R3 is involved in miR-513b-5p-inhibited autophagy liver cancer cells.
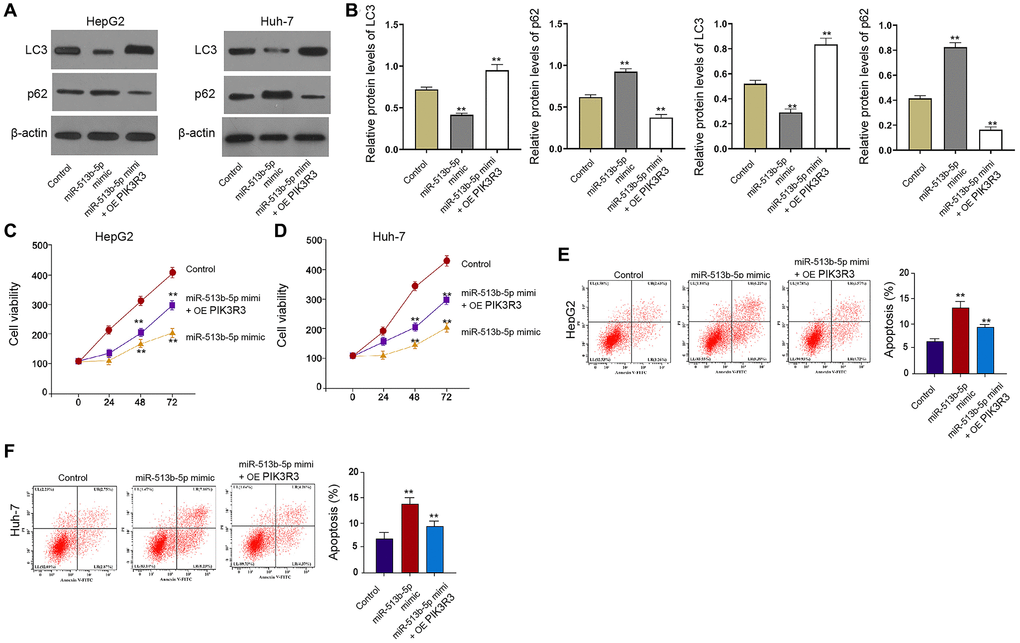
Figure 6. PIK3R3 is involved in miR-513b-5p-inhibited autophagy liver cancer cells. (A–F) The HepG2 and Huh-7 cells were treated with miR-513b-5p mimic and pcDNA3.1- PIK3R3. (A and B) The detection of LC3, beclin1, and p62 expression using Western blot analysis. (C and D) The analysis of cell proliferation using MTT assays. (E and F) The analysis of cell apoptosis using flow cytometry.
Moreover, autophagy inhibitor 3-MA enhanced miR-513b-5p mimic-promoted cell proliferation and miR-513b-5p mimic-inhibited cell apoptosis in the HepG2, Huh-7, and HCCLM3 cells (Figure 7, Supplementary Figure 6).
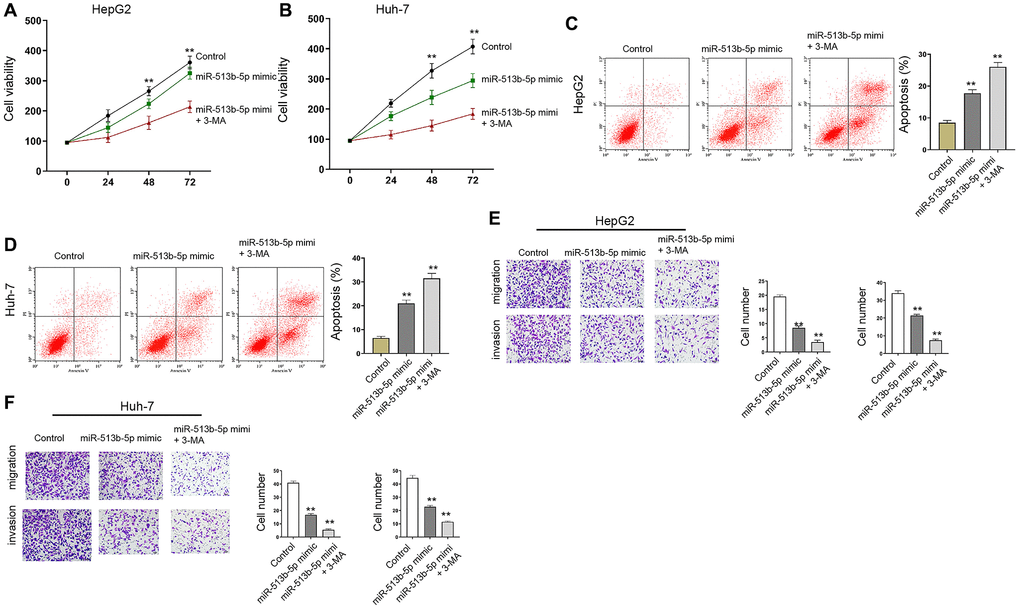
Figure 7. Autophagy inhibitor 3-MA reverses miR-513b-5p-mediated liver cancer progression in vitro. (A–F) The HepG2 and Huh-7 cells were treated with miR-513b-5p mimic and 3-MA (5mM). (A and B) The analysis of cell proliferation using MTT assays. (C and D) The analysis of cell apoptosis using flow cytometry. (E and F) The analysis of cell migration/invasion using transwell assays.
Discussion
It has been well-identified that autophagy is the critical cellular process and the regulators of autophagy are potential therapeutic targets for the liver cancer. The HGF/MET signaling controls autophagy and metabolism to regulate chemoresistance of liver cancer [20]. Autophagy regulates glycolytic metabolism by hexokinase 2 degradation in liver cancer [21]. Long noncoding RNA HULC enhances liver cancer progression via repressing PTEN through autophagy-mediated miR-15a [22]. Moreover, several miRNAs have been reported to regulate autophagy in liver cancer cells. MiR-638 promotes cell autophagy and apoptosis through repressing EZH2 in liver cancer [23]. MiR-223 enhancement attenuates doxorubicin-stimulated autophagy and relieves chemoresistance by targeting FOXO3 sin HCC cells [24]. In addition, it has been reported that miR-513b-5p enhances P53 expression by repressing IRF2 to decreases proliferation of testicular embryonal carcinoma cells [14]. MiR-513b-5p targets DUSP11 and is involved in AZIN1-AS1-promoted lung cancer progression [13]. Our data showed that miR-513b-5p decreased autophagy-related phenotypes in liver cancer cells. miR-513b-5p repressed proliferation and migration/invasion, and enhanced apoptosis of liver cancer cell in vitro and reduced tumor growth of liver cancer cells in vivo. These data indicate a novel function of miR-513b-5p in the autophagy modulation and liver cancer progression, providing important evidence of the role of miR-513b-5p in malignancies. In this study, we just evaluated the function of miR-513b-5p as a example in the modulation of autophagy in HCC, Whether or not have other miRNAs as well as miR-513b-5p to suppress autophagy in HCC progression and then synergy with miR-513b-5p should be explored in future investigations. Multiple factors may affect the outcome of miR-513b-5p-mediated autophagy and cancer progression. According to the previous study, several upstream factors, including long non-coding RNA FTX [25], long non-coding RNA UCA1 [26], long non-coding RNA LINC00861 [27], long non-coding RNA AZIN1-AS1 [13], circular RNA G004213 [28], and circular RNA circ_LARP4 [29], in the modulation of cancer progression, such as pancreatic cancer, osteosarcoma, cervical cancer, non-small cell lung cancer, and ovarian cancer. Whether these factors modulated miR-513b-5p-mediated autophagy in liver cancer should be validated in future investigations. Moreover, the miR-513b-5p mimic-enhanced p62 is associated with aggresome formation and maintain the survival of HCC. Thus, the miR-513b-5p is not sufficient to suppress the HCC relapse. More evidence should be constructed in future investigations to validate this issue.
Previous investigations have demonstrated the function of PIK3R3. It has been reported that miR-601 is a potential cancer inhibitor by repressing PIK3R3 in HCC cells [30]. MiR-1287 represses the migration, invasion, and proliferation of HCC cells by targeting PIK3R3 [31]. In our study, we identified that PIK3R3 was targeted by miR-513b-5p in liver cancer cells and involved in miR-513b-5p-inhibited autophagy liver cancer cells. Our finding provides new knowledge of the mechanism involving PIK3R3 of miR-513b-5p-mediated cancer progression. Meanwhile, PIK3R3 may be just one of the downstream targets of PIK3R3 in the modulation of cancer development, more potential factors response to miR-513b-5p-regulated cancer progression need to be explored in other investigations. In addition, some reported factors, including E2F5 [26], PTEN/AKT/mTOR signaling [27], DUSP11 [13], PRPF39 [28], and LARP4 [29], are involved in miR-513b-5p regulated cancer progression. The correlation of miR-513b-5p with these factors in the regulation of tumorigenesis and autophagy in liver cancer should be confirmed in future studies. There are still some limitations of this study. For example, we just used the miR-513b-5p mimic but not miR-513b-5p inhibitor to investigate the function of miR-513b-5p in HCC cells, the effect of miR-513b-5p inhibitor on autophagy in HCC cells needs to validate in further experiments. Meanwhile, we just evaluated the effect of miR-513b-5p on HCC cell growth in vivo, the function of miR-513b-5p/ PIK3R3 needs to be confirmed in the model. Importantly, the clinical significance of miR-513b-5p in HCC should be assessed in clinical HCC samples. This study provides the new knowledge of the function of miR-513b-5p in regulation autophagy during HCC and the correlation of miR-513b-5p with PIK3R3 in this process. The therapeutic agents or strategies of targeting miR-513b-5p/ PIK3R3 axis should be developed and designed and may benefit the routine clinical practice in HCC. Meanwhile, it could promote the application of miR-513b-5p to be a potential drug in HCC.
We concluded that miR-513b-5p repressed autophagy during the malignant progression of HCC by targeting PIK3R3. MiR-513b-5p may be applied as a therapeutic target for HCC.
Materials and Methods
Cell culture
The LO2, H7402, HCCLM3, HepG2, and Huh-7 cells were obtained in American Type Tissue Culture Collection. The cells were cultured in the DMEM (BI, USA) containing 0.1 mg/mL streptomycin (BI, USA), 100 units/mL penicillin (BI, USA), and 10% fetal bovine serum (BI, USA), at a condition of 37°C with 5% CO2. The lentiviral plasmids carrying miR-196a-5p mimic/inhibitor, and pcDNA3.1- PIK3R3 were synthesized and obtained (Genscript, China). The autophagy inhibitor 3-Methyladenine (3-MA) were purchased from Selleck (USA).
Quantitative reverse transcription-PCR (qRT-PCR)
The RNA was extracted from BC cells by using TriZol reagent (Thermo) after treatment, reverse transcribed to cDNA by using Super Script III kit (Invitrogen). Subsequently, the relative level was quantified by SYBR Premix kit (Takara, Japan), and normalized to GAPDH and 18 s. The results were calculated with 2-△△Ct method. All primers were obtained from RiboBio (China).
MTT assays
The cell viability was measured by MTT assays in the HepG2 and Huh-7 cells. Briefly, after the indicated treatment, about 2 × 104 cells were put into 96 wells and cultured for 12 hours. After indicated treatment, the cells were added with the MTT solution (10 μL, 5 mg/mL) and cultured for an extra 4 hours. Discarded medium, and 150 μL DMSO was used to treat the wells. An ELISA browser was applied to analyze the absorbance at 570nm (Bio-Tek EL 800, USA).
Western blot analysis
Total proteins were obtained from the mice tissues or cells with RIPA buffer (CST, USA). Protein concentrations were analyzed by applying the BCA Protein Quantification Kit (Abbkine, USA). Same concentration of protein was divided by SDS-PAGE (12% polyacrylamide gels), transferred to PVDF membranes (Millipore, USA) in the subsequent step. The membranes were hindered with 5% milk and hatched overnight at 4°C with the primary antibodies for PIK3R3 (Abcam, USA), LC3 (Abcam, USA), beclin1 (Abcam, USA), p62 (Abcam, USA), and β-actin (Abcam, USA). Then, the corresponding second antibodies (Abcam, USA) were used for hatching the membranes 1 hour at room temperature, followed by the visualization by using an Odyssey CLx Infrared Imaging System.
Colony formation assays
The HCC cells were transfected as the indication, digested and suspended as single cells, seeded into 6-well plates with 1000 cells in each well. The cells were placed in incubator for two weeks, and the medium was changed every three days till the visible clones formed. The formed clones were stained by 0.5% crystal violate resolved in methanol, captured and counted by a microscope (Leica, Germany).
Tumorigenicity
The tumor growth of liver cancer cells in vivo was analyzed in nude mice of Balb/c (male, 4-week-old) (n = 5). About 1 × 107 cells HepG2 cells treated with miR-513b-5p mimic A. After 5 days of injection, we measured tumor growth every 5 days. We sacrificed the mice after 30 days of injection, and tumors were scaled. The width and length of tumor, and the body weight of mice were measured at indicated time. Tumor volume was calculated by the formula: width (mm)2 × length (mm)/2. The mice were anesthetized to death when tumor size reached 1000 mm3, and the tumors were collected. Animal care and method procedures were authorized by the Animal Ethics Committee of Affiliated Wuming Hospital, Guangxi Medical University.
Transwell assays
The migration and invasion ability of HCC cells were determined via using a transwell chamber (Corning, USA). To detect migration, HCC cells (1 × 105 cells/well) transfected as the indication were seeded into the upper chambers with FBS-free medium, while the lower chambers were filled with complete DMEM medium. After 24 hours incubation, the membranes of upper chambers were fixed by 4% paraformaldehyde for 15 min, and stained by 0.5% crystal violet for 30 minutes. The migrated cells were photographed and counted. For cell invasion, the process was similar with that of migration experiment, only that the upper chambers were coated with Matrigel (BD Bioscience, USA).
Analysis of cell apoptosis
For cell apoptosis detection, the apoptotic cells were stained with an FITC-Annexin V/PI detection kit (CST, USA). In brief, about 2 × 105 HepG2 and Huh-7 cells were plated on 6-well dishes. And the cells were harvested, washed with PBS, then stained with FITC-Annexin V (5 μL) and PI (5 μL) for 10 minutes, respectively. The samples were then detected in a flow cytometry (BD Biosciences, USA).
Luciferase reporter gene assay
The potential binding sites of miR-513b-5p and the 3′UTR region of PIK3R3 were predicted by ENCORI website. The wild type sequences of the 3′UTR region of PIK3R3 were cloned into the pmirGLO vectors (Promega, USA) to obtain the PIK3R3-WT. The site-specific mutated sequences of the 3′UTR of PIK3R3 were inserted into pmirGLO vectors to obtain the PIK3R3-Mut. The cells were transfected with PIK3R3-WT and Mut along with miR-513b-5p mimic. After 24 hours incubation, the cells were lysed and the luciferase intensity was detected by a Dual Luciferase assay kit (Promega, USA). As control, the luciferase activities of Renilla were measured.
Statistical analysis
Data were expressed as mean ± SD, and the statistical analysis was presented: *P < 0.05, **P < 0.01, ***P < 0.001, in which P < 0.05 were considered as statistically significant. The unpaired Student’s t-test and one-way ANOVA was used to compare the difference.
Supplementary Materials
Author Contributions
Wei Jin and Yilei Liang designed and performed experiments, analysed data and wrote the paper; Shuyou Li and Guoxiang Lin designed and performed experiments; Haiying Liang, Zhenni Zhang, Weiming Zhang, and Rongjun Nie designed experiments, analysed data and wrote the paper.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- 1. Conde de la Rosa L, Garcia-Ruiz C, Vallejo C, Baulies A, Nuñez S, Monte MJ, Marin JJG, Baila-Rueda L, Cenarro A, Civeira F, Fuster J, Garcia-Valdecasas JC, Ferrer J, et al. STARD1 promotes NASH-driven HCC by sustaining the generation of bile acids through the alternative mitochondrial pathway. J Hepatol. 2021; 74:1429–41. https://doi.org/10.1016/j.jhep.2021.01.028 [PubMed]
- 2. Dai X, Guo Y, Hu Y, Bao X, Zhu X, Fu Q, Zhang H, Tong Z, Liu L, Zheng Y, Zhao P, Fang W. Immunotherapy for targeting cancer stem cells in hepatocellular carcinoma. Theranostics. 2021; 11:3489–501. https://doi.org/10.7150/thno.54648 [PubMed]
- 3. Kong FE, Li GM, Tang YQ, Xi SY, Loong JHC, Li MM, Li HL, Cheng W, Zhu WJ, Mo JQ, Gong YF, Tang H, Zhao Y, et al. Targeting tumor lineage plasticity in hepatocellular carcinoma using an anti-CLDN6 antibody-drug conjugate. Sci Transl Med. 2021; 13:eabb6282. https://doi.org/10.1126/scitranslmed.abb6282 [PubMed]
- 4. Llovet JM, De Baere T, Kulik L, Haber PK, Greten TF, Meyer T, Lencioni R. Locoregional therapies in the era of molecular and immune treatments for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2021; 18:293–313. https://doi.org/10.1038/s41575-020-00395-0 [PubMed]
- 5. Qian Y, Liu Q, Li P, Han Y, Zhang J, Xu J, Sun J, Wu A, Song S, Lu W. Highly Tumor-Specific and Long-Acting Iodine-131 Microbeads for Enhanced Treatment of Hepatocellular Carcinoma with Low-Dose Radio-Chemoembolization. ACS Nano. 2021; 15:2933–46. https://doi.org/10.1021/acsnano.0c09122 [PubMed]
- 6. Alles J, Fehlmann T, Fischer U, Backes C, Galata V, Minet M, Hart M, Abu-Halima M, Grässer FA, Lenhof HP, Keller A, Meese E. An estimate of the total number of true human miRNAs. Nucleic Acids Res. 2019; 47:3353–64. https://doi.org/10.1093/nar/gkz097 [PubMed]
- 7. Kabekkodu SP, Shukla V, Varghese VK, D'Souza J, Chakrabarty S, Satyamoorthy K. Clustered miRNAs and their role in biological functions and diseases. Biol Rev Camb Philos Soc. 2018; 93:1955–86. https://doi.org/10.1111/brv.12428 [PubMed]
- 8. Lu Q, Wu R, Zhao M, Garcia-Gomez A, Ballestar E. miRNAs as Therapeutic Targets in Inflammatory Disease. Trends Pharmacol Sci. 2019; 40:853–65. https://doi.org/10.1016/j.tips.2019.09.007 [PubMed]
- 9. Roux J, Gonzàlez-Porta M, Robinson-Rechavi M. Comparative analysis of human and mouse expression data illuminates tissue-specific evolutionary patterns of miRNAs. Nucleic Acids Res. 2012; 40:5890–900. https://doi.org/10.1093/nar/gks279 [PubMed]
- 10. Saliminejad K, Khorram Khorshid HR, Soleymani Fard S, Ghaffari SH. An overview of microRNAs: Biology, functions, therapeutics, and analysis methods. J Cell Physiol. 2019; 234:5451–65. https://doi.org/10.1002/jcp.27486 [PubMed]
- 11. Klingenberg M, Matsuda A, Diederichs S, Patel T. Non-coding RNA in hepatocellular carcinoma: Mechanisms, biomarkers and therapeutic targets. J Hepatol. 2017; 67:603–18. https://doi.org/10.1016/j.jhep.2017.04.009 [PubMed]
- 12. Wei L, Wang X, Lv L, Liu J, Xing H, Song Y, Xie M, Lei T, Zhang N, Yang M. The emerging role of microRNAs and long noncoding RNAs in drug resistance of hepatocellular carcinoma. Mol Cancer. 2019; 18:147. https://doi.org/10.1186/s12943-019-1086-z [PubMed]
- 13. Cai Y, Wu Q, Liu Y, Wang J. AZIN1-AS1, A Novel Oncogenic LncRNA, Promotes the Progression of Non-Small Cell Lung Cancer by Regulating MiR-513b-5p and DUSP11. Onco Targets Ther. 2020; 13:9667–78. https://doi.org/10.2147/OTT.S261497 [PubMed]
- 14. Wang X, Zhang X, Wang G, Wang L, Lin Y, Sun F. Hsa-miR-513b-5p suppresses cell proliferation and promotes P53 expression by targeting IRF2 in testicular embryonal carcinoma cells. Gene. 2017; 626:344–53. https://doi.org/10.1016/j.gene.2017.05.033 [PubMed]
- 15. Cao G, Dong W, Meng X, Liu H, Liao H, Liu S. MiR-511 inhibits growth and metastasis of human hepatocellular carcinoma cells by targeting PIK3R3. Tumour Biol. 2015; 36:4453–59. https://doi.org/10.1007/s13277-015-3085-z [PubMed]
- 16. Zhang W, Liu Y, Fu Y, Han W, Xu H, Wen L, Deng Y, Liu K. Long non-coding RNA LINC00160 functions as a decoy of microRNA-132 to mediate autophagy and drug resistance in hepatocellular carcinoma via inhibition of PIK3R3. Cancer Lett. 2020; 478:22–33. https://doi.org/10.1016/j.canlet.2020.02.014 [PubMed]
- 17. Sun H, Feng X. MicroRNA-367 directly targets PIK3R3 to inhibit proliferation and invasion of oral carcinoma cells. Biosci Rep. 2020; 40:BSR20193867. https://doi.org/10.1042/BSR20193867 [PubMed]
- 18. Sun Q, Yang Z, Li P, Wang X, Sun L, Wang S, Liu M, Tang H. A novel miRNA identified in GRSF1 complex drives the metastasis via the PIK3R3/AKT/NF-κB and TIMP3/MMP9 pathways in cervical cancer cells. Cell Death Dis. 2019; 10:636. https://doi.org/10.1038/s41419-019-1841-5 [PubMed]
- 19. Zhu Y, Zhao H, Rao M, Xu S. MicroRNA-365 inhibits proliferation, migration and invasion of glioma by targeting PIK3R3. Oncol Rep. 2017; 37:2185–92. https://doi.org/10.3892/or.2017.5458 [PubMed]
- 20. Huang X, Gan G, Wang X, Xu T, Xie W. The HGF-MET axis coordinates liver cancer metabolism and autophagy for chemotherapeutic resistance. Autophagy. 2019; 15:1258–79. https://doi.org/10.1080/15548627.2019.1580105 [PubMed]
- 21. Jiao L, Zhang HL, Li DD, Yang KL, Tang J, Li X, Ji J, Yu Y, Wu RY, Ravichandran S, Liu JJ, Feng GK, Chen MS, et al. Regulation of glycolytic metabolism by autophagy in liver cancer involves selective autophagic degradation of HK2 (hexokinase 2). Autophagy. 2018; 14:671–84. https://doi.org/10.1080/15548627.2017.1381804 [PubMed]
- 22. Xin X, Wu M, Meng Q, Wang C, Lu Y, Yang Y, Li X, Zheng Q, Pu H, Gui X, Li T, Li J, Jia S, Lu D. Long noncoding RNA HULC accelerates liver cancer by inhibiting PTEN via autophagy cooperation to miR15a. Mol Cancer. 2018; 17:94. https://doi.org/10.1186/s12943-018-0843-8 [PubMed]
- 23. Zhang H, Liang H, Wu S, Zhang Y, Yu Z. MicroRNA-638 induces apoptosis and autophagy in human liver cancer cells by targeting enhancer of zeste homolog 2 (EZH2). Environ Toxicol Pharmacol. 2021; 82:103559. https://doi.org/10.1016/j.etap.2020.103559 [PubMed]
- 24. Zhou Y, Chen E, Tang Y, Mao J, Shen J, Zheng X, Xie S, Zhang S, Wu Y, Liu H, Zhi X, Ma T, Ni H, et al. miR-223 overexpression inhibits doxorubicin-induced autophagy by targeting FOXO3a and reverses chemoresistance in hepatocellular carcinoma cells. Cell Death Dis. 2019; 10:843. https://doi.org/10.1038/s41419-019-2053-8 [PubMed]
- 25. Li S, Zhang Q, Liu W, Zhao C. Silencing of FTX suppresses pancreatic cancer cell proliferation and invasion by upregulating miR-513b-5p. BMC Cancer. 2021; 21:290. https://doi.org/10.1186/s12885-021-07975-6 [PubMed]
- 26. Zhang Z, Wu X, Han Q, Huang Z. Downregulation of long non-coding RNA UCA1 represses tumorigenesis and metastasis of osteosarcoma via miR-513b-5p/E2F5 axis. Anticancer Drugs. 2021; 32:602–13. https://doi.org/10.1097/CAD.0000000000001034 [PubMed]
- 27. Liu H, Zhang L, Ding X, Sui X. LINC00861 inhibits the progression of cervical cancer cells by functioning as a ceRNA for miR-513b-5p and regulating the PTEN/AKT/mTOR signaling pathway. Mol Med Rep. 2021; 23:24. https://doi.org/10.3892/mmr.2020.11662 [PubMed]
- 28. Qin L, Zhan Z, Wei C, Li X, Zhang T, Li J. Has-circRNA-G004213 promotes cisplatin sensitivity by regulating miR-513b-5p/PRPF39 in liver cancer. Mol Med Rep. 2021; 23:421. https://doi.org/10.3892/mmr.2021.12060 [PubMed]
- 29. Lin W, Ye H, You K, Chen L. Up-regulation of circ_LARP4 suppresses cell proliferation and migration in ovarian cancer by regulating miR-513b-5p/LARP4 axis. Cancer Cell Int. 2020; 20:5. https://doi.org/10.1186/s12935-019-1071-z [PubMed]
- 30. Song Y, He S, Zhuang J, Wang G, Ni J, Zhang S, Ye Y, Xia W. MicroRNA-601 serves as a potential tumor suppressor in hepatocellular carcinoma by directly targeting PIK3R3. Mol Med Rep. 2019; 19:2431–39. https://doi.org/10.3892/mmr.2019.9857 [PubMed]
- 31. Lu J, Tang L, Xu Y, Ge K, Huang J, Gu M, Zhong J, Huang Q. Mir-1287 suppresses the proliferation, invasion, and migration in hepatocellular carcinoma by targeting PIK3R3. J Cell Biochem. 2018; 119:9229–38. https://doi.org/10.1002/jcb.27190 [PubMed]



