Introduction
As the world's population structure is increasingly aging, the incidence of cancer is growing [1]. It is expected that by 2030, seventy percent of malignancies and 85 percent of tumor-related deaths will occur in elderly people (over 65 years) [2]. As a highly malignant tumor, pancreatic cancer (PC) is the fourth leading cause of tumor-related death and is expected to become the second leading cause by 2030 in the US [3, 4]. Due to the lack of clinical manifestations in the early stage, most PC patients are diagnosed at an advanced stage accompanied by metastasis [5, 6]. Although the overall incidence of PC among all age groups is only 11.7 percent, the incidence of PC is 66.4 percent in patients over 65 years old and 91.1 percent in patients over 80 years old [7]. Therefore, treating elderly patients with metastatic pancreatic cancer (mPC), an increasingly expanding group, is going to be a serious clinical challenge.
It has been widely recognized that elderly patients’ internal environment (chronic inflammation and immune system dysfunction) is more likely to induce cancer under the stimulation of carcinogens [8, 9]. Moreover, it has been reported that age is a significant negative prognostic factor for PC [10], and the immune system of elderly patients plays a key role in the development of PC [11]. Therefore, elderly patients may represent a distinct subgroup that needs more targeted clinical management plans [12]. At present, surgery is not recommended for pancreatic cancer patients with distant metastases, especially elderly patients, who are more likely to receive chemotherapy, radiotherapy and other nonsurgical treatments [7, 13]. The aim of this study was to explore the clinical characteristics and prognostic factors of elderly patients with mPC.
Results
Patient characteristics
We used X-tile software to divide the patients who diagnosed between 2004 and 2014 by age into three groups. The results indicated that the ages of 65 and 80 years old were appropriate cutoff values for age at diagnosis (Supplementary Figure 1). A total of 10,784 patients were enrolled in this study, including 3681 aged under 65 years, 4415 between 65 and 80 years, and 2688 over 80 years. The clinical characteristics of the mPC patients stratified by age are presented in Table 1. Approximately half of the patients were married (N=5329, 49.4%). Most patients were white (N=8560, 79.4%), and male patients accounted for 51.7% of all patients (N=5573). In total, 551 patients (5.1%) were treated surgically, and 3883 (36%) received chemotherapy. The collection of metastatic location data in the SEER database began in 2010; thus, detailed information about the metastatic sites of 5463 patients was included from 2010 to 2014.
Table 1. Comparison of characteristics of metastatic pancreatic cancer patients among different age groups.
| Age group | All patients | Age<65 | Age ≥65 and <80 | Age ≥80 | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Features of Patients (2004-2014) | 10784(100.0%) | 3681(100%) | 4415(100%) | 2688(100%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | Others | 5455(50.6%) | 1748(47.5%) | 2016(45.7%) | 1691(62.9%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 5329(49.4%) | 1933(52.5%) | 2399(54.3%) | 997(37.1%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race | Others | 2224(20.6%) | 902(24.5%) | 885(20%) | 437(16.3%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White | 8560(79.4%) | 2779(75.5%) | 3530(80%) | 2251(83.7%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | Male | 5573(51.7%) | 2191(59.5%) | 2288(51.8%) | 1094(40.7%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 5211(48.3%) | 1490(40.5%) | 2127(48.2%) | 1594(59.3%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | T0 | 515(4.8%) | 181(4.9%) | 218(4.9%) | 116(4.3%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 103(1.0%) | 32(0.9%) | 47(1.1%) | 24(0.9%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 766(7.1%) | 248(6.7%) | 316(7.2%) | 202(7.5%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3 | 1032(9.6%) | 390(10.6%) | 422(9.6%) | 220(8.2%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T4 | 1204(11.2%) | 472(12.8%) | 488(11.1%) | 244(9.1%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tx | 7164(66.4%) | 2358(64.1%) | 2924(66.2%) | 1882(70%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N stage | N0 | 3625(33.6%) | 1186(32.2%) | 1552(35.2%) | 887(33%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1 | 2125(19.7%) | 927(25.2%) | 823(18.6%) | 375(14%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nx | 5034(46.7%) | 1568(42.6%) | 2040(46.2%) | 1426(53.1%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Surgery | No | 10123(93.9%) | 3425(93%) | 4137(93.7%) | 2561(95.3%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 551(5.1%) | 220(6%) | 241(5.5%) | 90(3.3%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 110(1.0%) | 36(1%) | 37(0.8%) | 37(1.4%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | No | 10350(96.0%) | 3488(94.8%) | 4232(95.9% | 2630(97.8%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 434(4.0%) | 193(5.2%) | 183(4.1%) | 58(2.2%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemotherapy | No | 6901(64.0%) | 1896(51.5%) | 2722(61.7%) | 2283(84.9%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 3883(36.0%) | 1785(48.5%) | 1693(38.3%) | 405(15.1%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS | Live | 595(5.5%) | 305(8.3%) | 212(4.8%) | 78(2.9%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dead | 10189(94.5%) | 3376(91.7%) | 4203(95.2%) | 2610(97.1%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CSS | Others | 2104(19.5%) | 632(17.2%) | 849(19.2%) | 623(23.2%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dead of PC | 8680(80.5%) | 3049(82.8%) | 3566(80.8%) | 2065(76.8%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metastases of Patients (2010-2014) | 5463(100.0%) | 1769(100%) | 2238(100%) | 1456(100%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bone metastasis | No | 4318(79.0%) | 1416(80%) | 1782(79.6%) | 1120(76.9%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 480(8.8%) | 189(10.7%) | 202(9%) | 89(6.1%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 665(12.2%) | 164(9.3%) | 254(11.3%) | 247(17%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brain metastasis | No | 4702(86.1%) | 1563(88.4%) | 1945(86.9%) | 1194(82%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 67(1.2%) | 26(1.5%) | 29(1.3%) | 12(0.8%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 694(12.7%) | 180(10.2%) | 264(11.8%) | 250(17.2%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Liver metastasis | No | 1217(22.3%) | 386(21.8%) | 482(21.5%) | 349(24%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 3892(71.2%) | 1293(73.1%) | 1631(72.9%) | 968(66.5%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 354(6.5%) | 90(5.1%) | 125(5.6%) | 139(9.5%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lung metastasis | No | 3627(66.4%) | 1220(69%) | 1478(66%) | 929(63.8%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1165(21.3%) | 381(21.5%) | 495(22.1%) | 289(19.8%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 671(12.3%) | 168(9.5%) | 265(11.8%) | 238(16.3%) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS: overall survival; CSS: cancer-specific survival. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
On the one hand, compared with the number of mPC patients under 65 years old, more mPC patients between 65 and 80 years old were married, were white, were female, had T1 and T2 stage disease, and had N0 stage disease and were less likely to be treated with surgery, radiation, or chemotherapy (all P <0.001). Although the liver is the organ that most commonly develops metastasis, elderly patients have a greater chance of developing lung metastasis than younger patients and are less likely to develop metastasis in the liver, brain, or bone (all P <0.001). Detailed information is shown in Table 1. On the other hand, there was a large proportion of mPC patients older than 80 years who were unmarried (which contrasts with mPC patients between the ages of 65 and 80 years), were white, were female, had T1 and T2 stage disease, and had N0 stage disease and were less likely to be treated with surgery, radiation and chemotherapy (all P <0.001). Moreover, it may be more difficult to identify metastatic sites in elderly patients, including the liver, lung, brain, or bone (all of these sites have a lower diagnosis rate in elderly patients than in younger patients; all P <0.001, Table 1).
The analysis also indicated that elderly mPC patients had a higher mortality rate at the follow-up deadline but a lower tumor-specific mortality rate than younger patients (all P <0.001). Of the 10784 patients, mortality occurred in 10189 (94.5% of 10784) at the end of follow-up. Among them, 8680 (80.5% of 10784) patients died due to pancreatic cancer. Regarding CSS, the cancer-specific mortality rates were 82.8% in the under 65-year-old group, 80.8% in the 65- to 80-year-old group, and 76.8% in the over 80-year-old group. Regarding OS, the mortality rates were 91.7%, 95.2% and 97.1% in the three age groups, respectively.
Prognostic factors of mPC patients between 65 and 80 years old
Multivariate Cox regression analysis revealed that surgical resection was associated with better OS (HR=0.70, 95% CI=0.57-0.85) and CSS (HR=0.72, 95% CI=0.58-0.90) and that chemotherapy was also associated with better OS (HR=0.45, 95% CI=0.41-0.49) and CSS (HR=0.43, 95% CI=0.39-0.48) (Table 2). The correlations of chemotherapy or surgical resection with OS and CSS according to the log-rank test was also revealed through the survival curve (Figure 1). Moreover, the results demonstrated that factors associated with poor OS included being unmarried, T0 stage disease and lung metastasis. In addition, poor CSS tended to occur in patients with T0 stage disease or lung metastasis. The detailed patient characteristics are shown in Table 2.
Table 2. Multivariate analysis of overall survival and cancer-specific survival in metastatic pancreatic cancer with age above 65 and under 80 years old.
| Variables | OS | CSS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR | (95%CI) | P value | HR | (95%CI) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | Others | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 0.90 | (0.83-0.99) | 0.027 | 0.95 | (0.86-1.06) | 0.345 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race | Others | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White | 0.96 | (0.86-1.07) | 0.467 | 0.94 | (0.83-1.07) | 0.327 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | Male | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 0.96 | (0.88-1.05) | 0.338 | 1.05 | (0.95-1.17) | 0.348 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | T0 | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 0.60 | (0.40-0.89) | 0.012 | 0.57 | (0.35-0.93) | 0.025 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 0.74 | (0.59-0.95) | 0.015 | 0.76 | (0.57-1.00) | 0.053 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3 | 0.85 | (0.67-1.08) | 0.183 | 0.85 | (0.64-1.12) | 0.254 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T4 | 0.94 | (0.75-1.18) | 0.573 | 0.98 | (0.75-1.28) | 0.880 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tx | 0.81 | (0.66-0.99) | 0.036 | 0.81 | (0.64-1.03) | 0.084 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N stage | N0 | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1 | 0.96 | (0.85-1.08) | 0.496 | 0.96 | (0.83-1.11) | 0.580 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nx | 0.99 | (0.89-1.10) | 0.817 | 1.05 | (0.93-1.19) | 0.438 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Surgery | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.70 | (0.57-0.85) | <0.001 | 0.72 | (0.58-0.90) | 0.005 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 1.18 | (0.76-1.84) | 0.466 | 1.34 | (0.83-2.17) | 0.236 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.88 | (0.69-1.11) | 0.282 | 0.83 | (0.62-1.10) | 0.193 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemotherapy | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.45 | (0.41-0.49) | <0.001 | 0.43 | (0.39-0.48) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bone metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.05 | (0.88-1.24) | 0.592 | 1.02 | (0.83-1.25) | 0.830 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 1.12 | (0.77-1.62) | 0.560 | 1.03 | (0.67-1.58) | 0.894 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brain metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.27 | (0.85-1.88) | 0.241 | 1.26 | (0.79-2.01) | 0.331 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 0.96 | (0.66-1.41) | 0.848 | 0.99 | (0.64-1.52) | 0.947 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Liver metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.09 | (0.98-1.22) | 0.130 | 1.10 | (0.96-1.25) | 0.170 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 1.05 | (0.82-1.36) | 0.692 | 1.03 | (0.76-1.38) | 0.867 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lung metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.18 | (1.05-1.32) | 0.004 | 1.21 | (1.06-1.38) | 0.004 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 0.93 | (0.75-1.15) | 0.486 | 1.02 | (0.80-1.30) | 0.891 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS: overall survival; CSS: cancer-specific survival; HR: hazard ratio; CI: confidence interval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
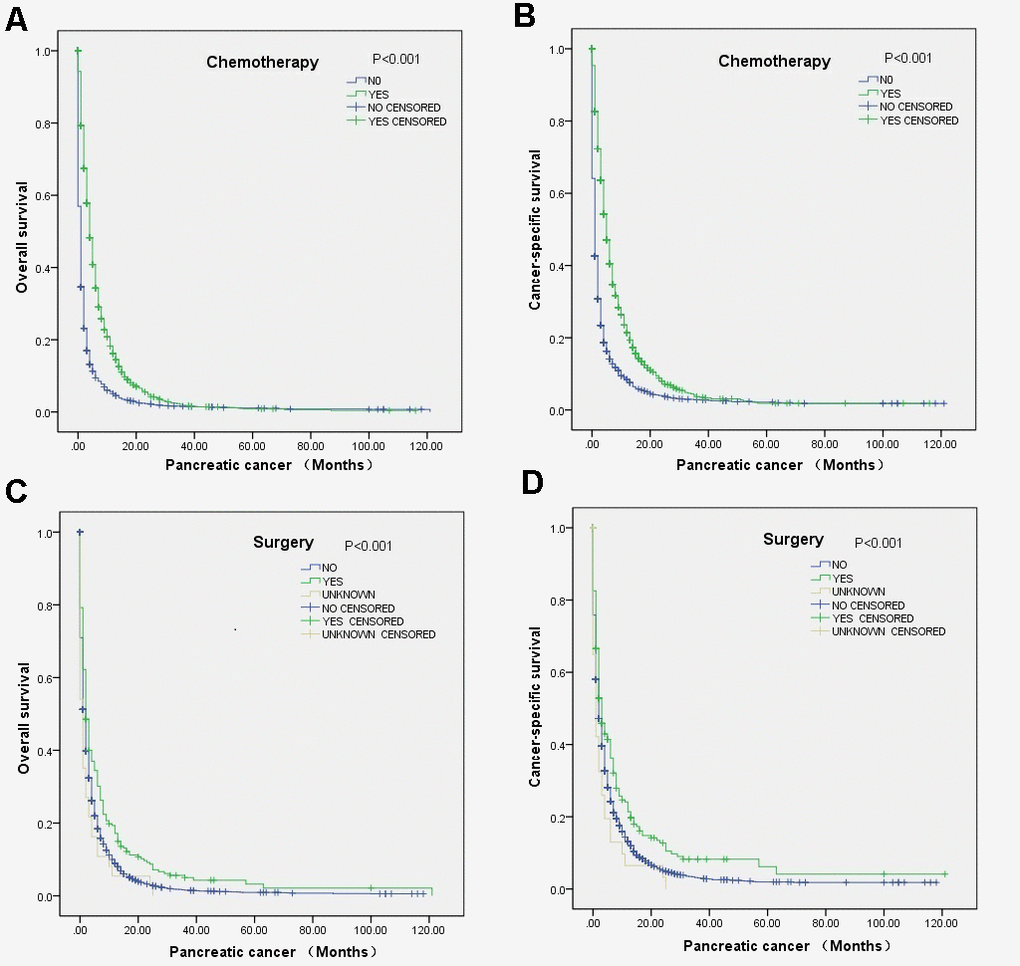
Figure 1. Survival curves of elderly mPC patients between 65 and 80 years old who received chemotherapy or surgery according to the log-rank test: (A) OS (P<0.001) and (B) CCS (P<0.001) for the chemotherapy, and (C) OS (P<0.001) and (D) CCS (P<0.001).for the surgery.
Prognostic factors of mPC patients aged above 80 years old
Multivariate Cox regression analysis indicated that chemotherapy was associated with improved OS (HR=0.45, 95% CI=0.41-0.49) and CSS (HR=0.43, 95% CI=0.39-0.48), and radiation showed a positive effect on OS (HR=0.64, 95% CI=0.45-0.92) but not CCS (Table 3). The relationships of chemotherapy and surgical resection with OS and CCS according to the log-rank test were also revealed through survival curves (Figure 2). Moreover, the analysis indicated that the factors associated with poor OS included male sex and liver metastasis (Table 3).
Table 3. Multivariate analysis of overall survival and cancer-specific survival in metastatic pancreatic cancer with age above 80 years old.
| Variables | OS | CSS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR | (95%CI) | P value | HR | (95%CI) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Marital status | Others | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Married | 0.95 | (0.84-1.07) | 0.364 | 1.02 | (0.88-1.18) | 0.830 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Race | Others | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| White | 1.08 | (0.93-1.25) | 0.304 | 1.01 | (0.85-1.21) | 0.904 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex | Male | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 0.88 | (0.79-0.99) | 0.037 | 1.05 | (0.91-1.22) | 0.476 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T stage | T0 | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1 | 0.75 | (0.40-1.38) | 0.354 | 0.73 | (0.33-1.63) | 0.440 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T2 | 1.08 | (0.81-1.45) | 0.593 | 1.18 | (0.81-1.72) | 0.382 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3 | 0.93 | (0.69-1.25) | 0.638 | 1.04 | (0.71-1.52) | 0.847 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T4 | 1.03 | (0.76-1.38) | 0.864 | 1.14 | (0.78-1.66) | 0.500 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tx | 1.06 | (0.82-1.36) | 0.674 | 1.18 | (0.85-1.63) | 0.333 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N stage | N0 | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1 | 1.06 | (0.9-1.240) | 0.47 | 1.2 | (0.99-1.46) | 0.063 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nx | 0.94 | (0.82-1.07) | 0.326 | 1.01 | (0.86-1.19) | 0.911 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Surgery | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.78 | (0.58-1.05) | 0.101 | 0.85 | (0.60-1.21) | 0.375 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 0.88 | (0.59-1.32) | 0.545 | 0.96 | (0.61-1.5) | 0.842 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.64 | (0.45-0.92) | 0.014 | 0.84 | (0.56-1.25) | 0.380 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chemotherapy | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.48 | (0.41-0.56) | <0.001 | 0.43 | (0.35-0.53) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bone metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 0.98 | (0.77-1.24) | 0.86 | 0.76 | (0.56-1.05) | 0.095 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 1.13 | (0.77-1.66) | 0.525 | 1.07 | (0.68-1.70) | 0.765 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Brain metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.42 | (0.79-2.58) | 0.243 | 1.48 | (0.72-3.04) | 0.289 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 1.11 | (0.76-1.62) | 0.581 | 1.28 | (0.82-2.00) | 0.284 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Liver metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.22 | (1.07-1.40) | 0.003 | 1.17 | (1.00-1.38) | 0.055 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 0.99 | (0.77-1.28) | 0.934 | 1.04 | (0.77-1.40) | 0.814 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lung metastasis | No | 1 (Referent) | 1 (Referent) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Yes | 1.00 | (0.87-1.16) | 0.992 | 1.00 | (0.84-1.19) | 0.996 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unknown | 0.84 | (0.64-1.10) | 0.201 | 0.79 | (0.58-1.10) | 0.160 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OS: overall survival; CSS: cancer-specific survival; HR: hazard ratio; CI: confidence interval. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
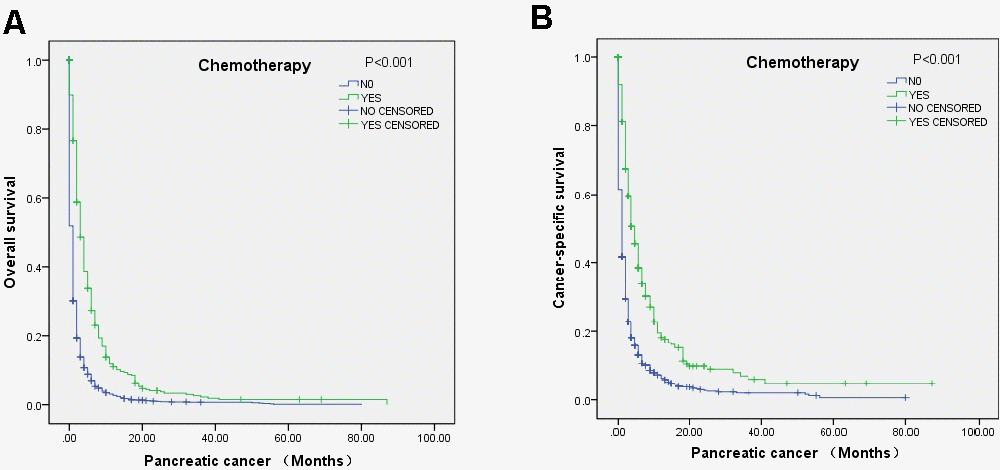
Figure 2. Survival curves of elderly mPC patients over 80 years old who received chemotherapy according to the log-rank test: (A) OS (p<0.001) and (B) CSS (P<0.001).
Validation set and performance
Patients who diagnosed at 2015 were set as the validation dataset, and detailed patients’ characteristics in different year of diagnosis was listed (Supplementary Table 1). For elderly group. we examined the performance of the prediction final model, both in the development dataset (2010-2014) and the validation dataset (2015) in terms of discrimination, which measured using values of 3-year time-dependent AUC. In the primary cohort, the model showed the discriminative ability in the validation cohort was agree with the development dataset (AUC: 0.699 vs 0.679, Supplementary Figure 2). Calibration was examined by plotting agreement between predicted and observed risks (Supplementary Figure 3). The imaginary line indicates a perfect calibration model in which the predicted probabilities are identical to the actual survival outcomes with the calibration slope of 1.0755 (0.9546 to 1.2117).
Discussion
According to the characteristics of previously reported age groups [7], the elderly patients in this study were divided into a 65- to 80-year-old group and an over 80-years-old group. The statistical results showed that elderly patients accounted for the majority of all mPC patients (65.9%), especially those between 65 and 80 years old (40.9%). A comparison of the clinical characteristics among patients of different ages revealed that white patients were more likely to have a longer life span, possibly because white patients tend to have more access to medical services [14, 15].
The results also showed that female sex was a favorable prognostic factor for mPC patients over 80 years old, which may be because females tend to pay more attention to their health and make greater use of health-care services than males [16, 17]. In addition, the proportion of patients between 65 and 80 years old who were married was the highest (2399/4415, 54.3%) among all age groups, while the proportion of married patients between 65 and 80 years old was sharply reduced (997/2688, 37.1%), which may be attributed to the higher death rate of their spouses during this age range. Further prognostic analysis indicated that marriage is a favorable prognostic factor for mPC patients between 65 and 80 years old, which agrees with many previous studies [18–21]. The result indicates that elderly people who lack care from a marriage partner have a poor prognosis. Therefore, elderly patients who do not have spouses should be given more attention.
Further study shows that there was an increase in the proportion of patients with T1 and T2 stage disease and a decrease in the proportion of patients with lymph node-negative disease (N0) among elderly patients. This result can be attributed to the frequent routine check-ups of elderly patients, which can allow for the diagnosis of disease at an early stage. Surprisingly, prognostic factor analysis suggested that patients with T0 stage disease between 65 and 80 years old had a better prognosis than those with Tn stage disease. Metastases that develop at an earlier T stage have a stronger ability to invade and metastasize; therefore, the tumor is more malignant, and the prognosis is correspondingly worse. This result is consistent with a previous study [20].
The older the cancer patient is, the more basic diseases he or she has and the more likely he or she will die from these diseases [7, 13], which was confirmed by our results. The present study showed that the overall mortality rate was higher among older patients; however, the tumor-specific mortality rate decreased gradually with increasing age. Such a result suggests that attention should be paid not only to the treatment of tumors in aging patients, especially in those over 80 years old, but also to the systemic conditions and basic diseases of these patients. Therefore, we tend to use the CSS as the prognostic indictor in the elderly group.
Moreover, the analysis of metastatic sites (collected from 2010 to 2014) suggested that the most common target organs for metastasis are the liver, lung, bone, and brain. A comparison among the age groups suggested that as age increases, except for the increase in lung metastasis between 65 and 80 years old, the metastasis rate in all sites showed a decreasing trend. This may be because tumors in elderly patients are often less aggressive than those in young patients. The liver and lungs are vital organs in the human body. Once metastasis occurs in these sites, patients may have a worse prognosis. Similarly, the present analysis also indicated that lung metastasis, which is related to both OS and CCS, is a poor prognostic factor for patients between 65 and 80 years old, while liver metastasis is related to OS in those over 80 years old.
Data on treatment showed that the older the patient is, the less likely he or she is to receive treatment, including surgery, radiation, and chemotherapy. Nonetheless, chemotherapy was the most common form of treatment, followed by surgery and radiation. Further analysis suggested that chemotherapy has the greatest therapeutic benefit for patients between 65 and 80 years old or older. Chemotherapy has been proven to be the primary treatment for mPC [22–25]. Gemcitabine alone or in combination with other chemotherapeutic drugs and the FOLFIRINOX regimen (leucovorin, fluorouracil, irinotecan, and oxaliplatin) are recommended according to the patient’s performance status as well as comorbidity profile [26]. Surgical treatment is not recommended for mPC, especially in elderly patients, according to clinical guidelines. However, surgery has been demonstrated to improve the prognosis of mPC patients [20]. The present analysis also indicated that patients between 65 and 80 years old may still benefit from surgery, but not those over 80 years old (which may be due to their physical condition and inability to tolerate surgery). This result is in agreement with those for other metastatic cancers, such as those originating from renal cells [27], the colorectum [28], and the prostate [29, 30], which have been proven to benefit from surgical treatment of the primary tumor. Although radiotherapy has a positive effect on OS among those between 65 and 80 years old, this treatment failed to show a correlation with CCS and had no association with death in patients over 80 years old. Such a result should be attributed to the lower sensitivity of pancreatic cancer to radiotherapy in elderly patients [23, 31]. During the validation process, its C-index value of prediction model was almost similar to the development dataset (AUC: 0.699 vs 0.679, Supplementary Figure 2). Moreover, the calibration ability detection indicates a perfect condition that the predicted probabilities are identical to the actual outcome with a calibration slope of 1.0755 (0.9546 to 1.2117).
The present study utilized a population-based cohort, which has the advantage of minimizing selection bias in comparisons. However, there are still some limitations, such as the relatively incomplete clinical information of many patients. For example, data about the sites of metastasis were collected only from 2010 to 2014, and patients enrolled before this period lacked relevant data. A large proportion of data regarding T stage, N stage, or metastatic sites were recorded as not otherwise specified or unknown. Information on the specific chemotherapy regimen used was not available. Because of these limitations, further evaluations of treatment in elderly patients were hindered, and we cannot rule out an alternative explanation for some of our findings. The remaining questions could be answered in future studies with more detailed information regarding clinical characteristics and treatment protocols. Our findings may be useful to establish a treatment policy for elderly mPC patients.
Conclusions
Elderly mPC patients differed from younger patients in many aspects. Chemotherapy, as the main treatment for elderly patients, can significantly improve their prognosis. Therefore, elderly patients with mPC are a special group of patients whose clinical characteristics and prognostic factors are different from those of young patients, and these patients require special treatment and attention. Chemotherapy is the most reasonable treatment and can improve the prognosis of elderly patients.
Materials and Methods
Patient cohort
The data examined in the present study were retrieved from the SEER-18 registry of the National Cancer Institute through SEER*Stat Software Version 8.3.5 software to query data from 18 SEER registries. As a publicly available database, the SEER database contains deidentified data; therefore, this study did not need approval from the institutional review board. Patients with a primary site of ‘pancreas’ between January 1, 2004, and December 31, 2014 were identified. The inclusion criteria were as follows: patients with International Classification of Diseases for Oncology, third edition (ICD-O-3) codes 8010, 8020, 8140, 8141 and 8144, and American Joint Committee on Cancer (AJCC) stage (6th edition) IV. Patients with unknown sites of cancer metastasis or unknown age and those who lacked survival data were excluded. The grouping of all patients by age was checked through X-tile software v3.6.1 (Yale University, New Haven, CT, USA), which was utilized to determine the optimal cutoff values [32]. Patients who diagnosed at 2015 were also collect at similar condition for validation.
Data collection
Information collected from each patient included age, race, sex, marital status, primary tumor site, T stage, N stage, M stage, surgical resection of the primary site, chemotherapy recode, cause-specific death classification, survival time, and vital status. Cancer-specific survival (CSS) and OS (overall survival) were defined as the time between diagnosis and death from mPC and between diagnosis and death from any cause, respectively. Detailed information on systematic treatment is not provided in the SEER database.
Statistical analysis
Clinical and demographic features were compared between different groups with the chi-square test. The Kaplan-Meier method with the log-rank test was used to examine CSS and OS. A Cox proportional hazards model was applied for multivariable survival analyses of CSS and OS. P<0.05 was considered statistically significant. IBM SPSS Statistics 22.0 (IBM, Armonk, NY, USA) was applied for all statistical analyses.
A prediction model for the elderly group was developed according to the results of multivariate Cox analysis and using the Empowerstats software (http://www.empowerstats.com/en/). The predictive accuracy of the model was assessed by ROC curve analysis. Besides, 3-year calibrations of the model were performed by comparing the predicted CSS to the observed CSS.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Author Contributions
All authors contributed in a manuscript as well as figure preparation are thoroughly familiar with its present version and are able to defend its content and conclusions.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This study was supported by a grant from the Doctoral Venture Capital fund of Henan Provincial People's Hospital (No. ZC20180077), the Special Project of Henan Provincial Key Research, Development and Promotion (Science and Technology) (No. 192102310119), and Joint Project of Medical Science and Technology Research Program of Henan Province (LHGJ20190577). Medical Science and Technology Research Plan of Henan Province, Project Co-built by Provincial Department (SB20190319). Chen Xiao-ping Foundation (CXPJJH1900001-2019203). These funds provided support for personnel and data collection.
References
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019; 69:7–34. https://doi.org/10.3322/caac.21551 [PubMed]
- 2. Yancik R, Ries LA. Cancer in older persons: an international issue in an aging world. Semin Oncol. 2004; 31:128–36. https://doi.org/10.1053/j.seminoncol.2003.12.024 [PubMed]
- 3. Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017; 67:7–30. https://doi.org/10.3322/caac.21387 [PubMed]
- 4. Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014; 74:2913–21. https://doi.org/10.1158/0008-5472.CAN-14-0155 [PubMed]
- 5. Luo G, Zhang Y, Guo P, Ji H, Xiao Y, Li K. Global patterns and trends in pancreatic cancer incidence: age, period, and birth cohort analysis. Pancreas. 2019; 48:199–208. https://doi.org/10.1097/MPA.0000000000001230 [PubMed]
- 6. Park HM, Park SJ, Han SS, Kim SH. Surgery for elderly patients with resectable pancreatic cancer, a comparison with non-surgical treatments: a retrospective study outcomes of resectable pancreatic cancer. BMC Cancer. 2019; 19:1090. https://doi.org/10.1186/s12885-019-6255-3 [PubMed]
- 7. Higuera O, Ghanem I, Nasimi R, Prieto I, Koren L, Feliu J. Management of pancreatic cancer in the elderly. World J Gastroenterol. 2016; 22:764–75. https://doi.org/10.3748/wjg.v22.i2.764 [PubMed]
- 8. Grimes A, Chandra SB. Significance of cellular senescence in aging and cancer. Cancer Res Treat. 2009; 41:187–95. https://doi.org/10.4143/crt.2009.41.4.187 [PubMed]
- 9. Lasry A, Ben-Neriah Y. Senescence-associated inflammatory responses: aging and cancer perspectives. Trends Immunol. 2015; 36:217–28. https://doi.org/10.1016/j.it.2015.02.009 [PubMed]
- 10. Wang H, Liu J, Xia G, Lei S, Huang X, Huang X. Survival of pancreatic cancer patients is negatively correlated with age at diagnosis: a population-based retrospective study. Sci Rep. 2020; 10:7048. https://doi.org/10.1038/s41598-020-64068-3 [PubMed]
- 11. Lianyuan T, Dianrong X, Chunhui Y, Zhaolai M, Bin J. The predictive value and role of stromal tumor-infiltrating lymphocytes in pancreatic ductal adenocarcinoma (PDAC). Cancer Biol Ther. 2018; 19:296–305. https://doi.org/10.1080/15384047.2017.1416932 [PubMed]
- 12. Li R, Vazquez J, Chau B, Novak J, Sedrak M, Warner S, Glaser S, Chen YJ, Dale W, Amini A. Corrigendum to 'Patterns of care and treatment outcomes in patients age 80 or older with non-metastatic pancreatic cancer. J Geriatr Oncol. 2020; 11:652. https://doi.org/10.1016/j.jgo.2020.11.005 [PubMed]
- 13. Gajda M, Kenig J. Treatment outcomes of pancreatic cancer in the elderly - literature review. Folia Med Cracov. 2018; 58:49–66. https://doi.org/10.24425/fmc.2018.125072 [PubMed]
- 14. Stepanikova I, Oates GR. Perceived discrimination and privilege in health care: the role of socioeconomic status and race. Am J Prev Med. 2017; 52:S86–94. https://doi.org/10.1016/j.amepre.2016.09.024 [PubMed]
- 15. Weissman J, Russell D, Jay M, Malaspina D. Racial, ethnic, and gender disparities in health care access and use among U.S. Adults with serious psychological distress. Psychiatr Serv. 2018; 69:517–22. https://doi.org/10.1176/appi.ps.201700221 [PubMed]
- 16. Bertakis KD, Azari R, Helms LJ, Callahan EJ, Robbins JA. Gender differences in the utilization of health care services. J Fam Pract. 2000; 49:147–52. [PubMed]
- 17. Redondo-Sendino A, Guallar-Castillón P, Banegas JR, Rodríguez-Artalejo F. Gender differences in the utilization of health-care services among the older adult population of Spain. BMC Public Health. 2006; 6:155. https://doi.org/10.1186/1471-2458-6-155 [PubMed]
- 18. Baine M, Sahak F, Lin C, Chakraborty S, Lyden E, Batra SK. Marital status and survival in pancreatic cancer patients: a SEER based analysis. PLoS One. 2011; 6:e21052. https://doi.org/10.1371/journal.pone.0021052 [PubMed]
- 19. Tao L, Xiu D, Sadula A, Ye C, Chen Q, Wang H, Zhang Z, Zhang L, Tao M, Yuan C. Surgical resection of primary tumor improves survival of pancreatic neuroendocrine tumor with liver metastases. Oncotarget. 2017; 8:79785–92. https://doi.org/10.18632/oncotarget.19523 [PubMed]
- 20. Tao L, Yuan C, Ma Z, Jiang B, Xiu D. Surgical resection of a primary tumor improves survival of metastatic pancreatic cancer: a population-based study. Cancer Manag Res. 2017; 9:471–79. https://doi.org/10.2147/CMAR.S145722 [PubMed]
- 21. Wang XD, Qian JJ, Bai DS, Li ZN, Jiang GQ, Yao J. Marital status independently predicts pancreatic cancer survival in patients treated with surgical resection: an analysis of the SEER database. Oncotarget. 2016; 7:24880–87. https://doi.org/10.18632/oncotarget.8467 [PubMed]
- 22. Kamisawa T, Wood LD, Itoi T, Takaori K. Pancreatic cancer. Lancet. 2016; 388:73–85. https://doi.org/10.1016/S0140-6736(16)00141-0 [PubMed]
- 23. Kuroda T, Kumagi T, Yokota T, Azemoto N, Hasebe A, Seike H, Nishiyama M, Inada N, Shibata N, Miyata H, Kawamura T, Imai Y, Ueno-Toshimori A, et al, and Ehime Pancreato-Cholangiology (EPOCH) Study Group. Efficacy of chemotherapy in elderly patients with unresectable pancreatic cancer: a multicenter review of 895 patients. BMC Gastroenterol. 2017; 17:66. https://doi.org/10.1186/s12876-017-0623-8 [PubMed]
- 24. Macchini M, Chiaravalli M, Zanon S, Peretti U, Mazza E, Gianni L, Reni M. Chemotherapy in elderly patients with pancreatic cancer: efficacy, feasibility and future perspectives. Cancer Treat Rev. 2019; 72:1–6. https://doi.org/10.1016/j.ctrv.2018.10.013 [PubMed]
- 25. Shin SH, Park Y, Hwang DW, Song KB, Lee JH, Kwon J, Yoo C, Alshammary S, Kim SC. Prognostic value of adjuvant chemotherapy following pancreaticoduodenectomy in elderly patients with pancreatic cancer. Anticancer Res. 2019; 39:1005–12. https://doi.org/10.21873/anticanres.13206 [PubMed]
- 26. Sohal DP, Mangu PB, Khorana AA, Shah MA, Philip PA, O'Reilly EM, Uronis HE, Ramanathan RK, Crane CH, Engebretson A, Ruggiero JT, Copur MS, Lau M, et al. Metastatic Pancreatic Cancer: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2016; 34:2784–96. https://doi.org/10.1200/JCO.2016.67.1412 [PubMed]
- 27. Flanigan RC, Salmon SE, Blumenstein BA, Bearman SI, Roy V, McGrath PC, Caton JR
Jr , Munshi N, Crawford ED. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med. 2001; 345:1655–59. https://doi.org/10.1056/NEJMoa003013 [PubMed] - 28. Temple LK, Hsieh L, Wong WD, Saltz L, Schrag D. Use of surgery among elderly patients with stage IV colorectal cancer. J Clin Oncol. 2004; 22:3475–84. https://doi.org/10.1200/JCO.2004.10.218 [PubMed]
- 29. Fossati N, Trinh QD, Sammon J, Sood A, Larcher A, Sun M, Karakiewicz P, Guazzoni G, Montorsi F, Briganti A, Menon M, Abdollah F. Identifying optimal candidates for local treatment of the primary tumor among patients diagnosed with metastatic prostate cancer: a SEER-based study. Eur Urol. 2015; 67:3–6. https://doi.org/10.1016/j.eururo.2014.08.056 [PubMed]
- 30. Culp SH, Schellhammer PF, Williams MB. Might men diagnosed with metastatic prostate cancer benefit from definitive treatment of the primary tumor? a SEER-based study. Eur Urol. 2014; 65:1058–66. https://doi.org/10.1016/j.eururo.2013.11.012 [PubMed]
- 31. Frakes J, Mellon EA, Springett GM, Hodul P, Malafa MP, Fulp WJ, Zhao X, Hoffe SE, Shridhar R, Meredith KL. Outcomes of adjuvant radiotherapy and lymph node resection in elderly patients with pancreatic cancer treated with surgery and chemotherapy. J Gastrointest Oncol. 2017; 8:758–65. https://doi.org/10.21037/jgo.2017.08.05 [PubMed]
- 32. Camp RL, Dolled-Filhart M, Rimm DL. X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res. 2004; 10:7252–59. https://doi.org/10.1158/1078-0432.CCR-04-0713 [PubMed]


