Introduction
Acute liver injury (ALI) is a life-threatening disease, if not treated in time, it will eventually lead to acute liver failure when the extent of hepatocyte death exceeds the liver's regenerative capacity, and its pathogenesis involves direct damage and immune-mediated injury [1]. Oxidative stress plays a key role in hepatocyte injury because the liver is a major organ invaded by reactive oxygen species and reactive nitrogen species (RNS) [2–4]. Additionally, acute hepatic damage is closely related to inflammation, as inflammatory immune responses characterized by the expression of proinflammatory mediators such as TNF-α and extensive immune cell infiltration in the liver, eventually result in hepatic apoptosis [5]. Although the pathogenic mechanism and factors associated with ALI have been widely reported, the details of liver injury and the drugs effective for treating liver injury remain poorly understood.
Oxidative stress refers to the imbalance between ROS / RNS production from aerobic metabolism and the elimination of antioxidant defense [6]. The antioxidant defense system consists of glutathione (GSH) and its synthesis, phase II detoxification enzyme and active oxygen deactivation enzyme, which play a key role in protecting cells from oxidative damage [7]. Nrf2, the transcription factor nuclear factor-erythroid 2 related factor 2, plays a protective role in GSH synthesis, antioxidant stress system, conjugation, transport and excretion of the metabolites and serves as a pleiotropic target resistant to hepatic damage [8]. It has been reported that Nrf2 induced glutamic acid cysteine ligase gene expression contributes to GSH synthesis and meliorates NAPQI induced hepatotoxicity [9]. Furthermore, activation of Nrf2 decreases acetaminophen (AA) - sulfate formation and enhances elimination of AA- glucuronide due to increased expression of Mrp3 in Keap1-kd mice [10]. Another study finds that transforming growth factor β1 (TGFβ1) induces HO-1 protein expression and enhanced nuclear accumulation of Nrf2 in Human aortic smooth muscle cells (HAoSMC), which also demonstrates that Nrf2-ARE pathway represents a novel target for TGF-β1 in human vascular smooth muscle cells (SMC) [11]. However, the relationship between Nrf2 and TGFβ1 in the regulation of liver diseases is rarely reported.
TGFβ1, a member of the TGFβ family of growth and differentiation factors, controls cell differentiation and proliferation and plays key roles in skeletal diseases, fibrosis, and cancer [12]. TGFβ1 transduces its signal by directly binding TGFβ receptor 2 (TGFβr2) to form a constitutively active kinase and then recruiting TGFβ receptor 1 (TGFβr1) into a heterotetramer receptor complex, ultimately resulting in the phosphorylation of SMAD2 and SMAD3 [13]. Previous studies have shown that TGFβ1 increased in the liver and serum of mice or rats during liver failure [14, 15]. TGFβ1 was markedly elevated in both the liver tissue and the plasma in patients with acute liver failure [16, 17], indicating that TGFβ1 may play a vital role in the regulation of ALI. Upon this, candidate compounds with multi-effects on anti-inflammation, anti-oxidative stress and TGFβr1 and Nrf2 signaling pathway regulation seem to be a more suitable treatment strategy for ALI.
Lupeol, as a natural triterpenoid, is widely found in fruits such as strawberry, mango, grape and olive and vegetables such as white cabbage and green pepper [18]. As a pentacyclic triterpene, lupeol has been proven to have antioxidative, anti-inflammatory, and skin healing-promoting functions and to have inhibitory effects on breast cancer, prostate cancer and mouse melanoma [19, 20]. Besides, lupeol possesses many potential liver-protective effects. A previous study showed that lupeol has a protective effect on aflatoxin B1-induced peroxidative hepatic damage in rats and is as effective as silymarin [21]. Furthermore, other studies have found that lupeol is effective in combating oxidative stress-induced liver injury [22–24]. Thus, in view of the anti-oxidative and anti-inflammatory effects of lupeol, it may be an effective therapy for acute hepatic damage.
Therefore, we investigated the effects of lupeol on ALI in mice and zebrafish, as well as the related mechanisms. We found that lupeol relieved LPS/D-GalN-induced ALI by inhibiting hepatocyte inflammation, hepatic apoptosis and oxidative stress in the liver. Moreover, lupeol cured ALI by decreasing the expression of TGFβr1 and increasing the expression of Nrf2.
Results
Lupeol alleviates LPS-induced liver injury in zebrafish
First, we used the survival rate, body length, heart rate and morphological changes of zebrafish embryos to investigate the toxicology of lupeol. Zebrafish embryos (3 dpf) were treated with lupeol for 72 h. At 400 μM lupeol, the zebrafish larvae had abdominal swelling and a shorter body length (Figure 1A and Supplementary Figure 1A), indicating that 400 μM lupeol did have toxic effects on the developmental stages of zebrafish. Notably, 400 μM lupeol caused approximately 100% larval mortality after 72 h, and EC50=328.3μM (Figure 1B). In the body length test, compared with the control group, the 400 μM lupeol group showed decreased body length (Figure 1C). In the heart rate test, the heart rate was decreased in the 200 μM lupeol group (Figure 1D). Based on the results, we found that lupeol was toxic to zebrafish larvae at concentrations of more than 200 μM. Thus, we selected lupeol concentrations of 25, 50, and 100 μM for further zebrafish experimentation.
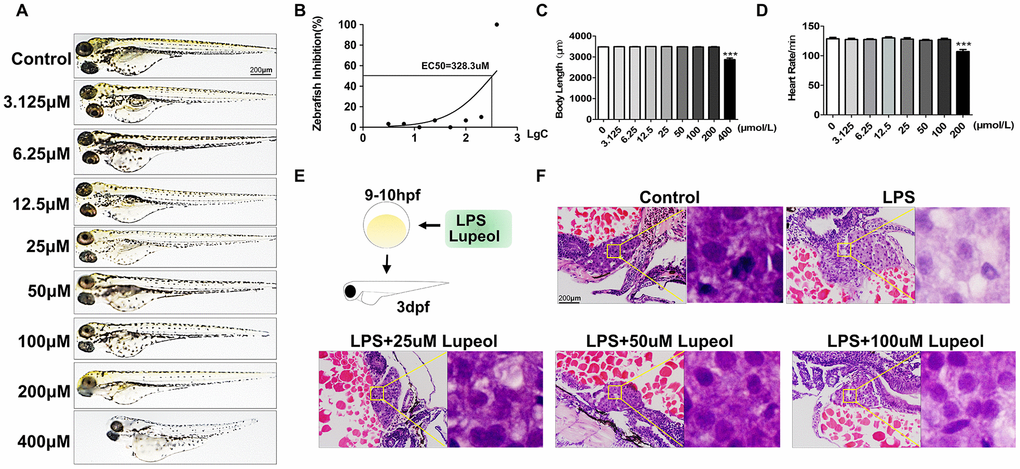
Figure 1. Lupeol attenuates liver injury induced by LPS in zebrafish. (A–D) Zebrafish larvae were treated with different concentrations of lupeol (0, 3.125, 6.25,12.5,25,50,100,200, and 400μmol) to observe changes in zebrafish morphology, survival rate, body length and heart rate. N=20, data are expressed as the mean ± SEM, *P<0.05, **P<0.01, ***P<0.001, control group vs administration group. (E) Schematic diagram of treatment on zebrafish. (F) H&E staining of the liver in zebrafish larva. Magnification, 400×, bar=200μm.
Previous studies have shown that zebrafish larvae grown in 10 μg/mL LPS until 3 dpf exhibit an obvious systemic inflammatory response and oxidative stress [25, 26]. In this study, we exposed 9-10 hpf zebrafish larvae to 10 μg/mL LPS until 3 dpf as a liver injury model (Figure 1E). Liver injury is the pathological basis of various hepatic diseases, including liver cell degeneration, necrosis and inflammatory cell infiltration. We examined the hepatoprotective effects of lupeol on LPS-induced ALI in zebrafish. H&E staining of liver sections revealed that LPS caused hepatocyte degeneration, and lupeol ameliorated the restoration of liver tissue structures in zebrafish larvae (Figure 1F).
These results suggested that lupeol antagonized liver injury by attenuating hepatic apoptosis. Moreover, the effective concentrations of lupeol were in the range of 25-100 μM, among which 100 μM was the most effective.
Lupeol inhibits liver damage caused induced by LPS/D-GalN in mice
LPS/D-GalN coinjection is a common method to establish an ALI model in mice, as D-GalN increases the sensitivity of rodents to LPS-induced hepatotoxicity [27]. To further verify the role of lupeol in ALI, an LPS/D-GalN-induced liver injury model was used (Figure 2A). As demonstrated by H&E staining, we found that the livers of the model groups exhibited diffuse necrosis, swollen hepatocytes and severe hemorrhage compared to those of the control group, while lupeol reduced liver necrosis (Figure 2D). In addition, serum ALT/AST levels were significantly increased in mice under LPS/D-Gal coinjection, while the levels were similar to those of the control group after lupeol treatment (Figure 2B, 2C).
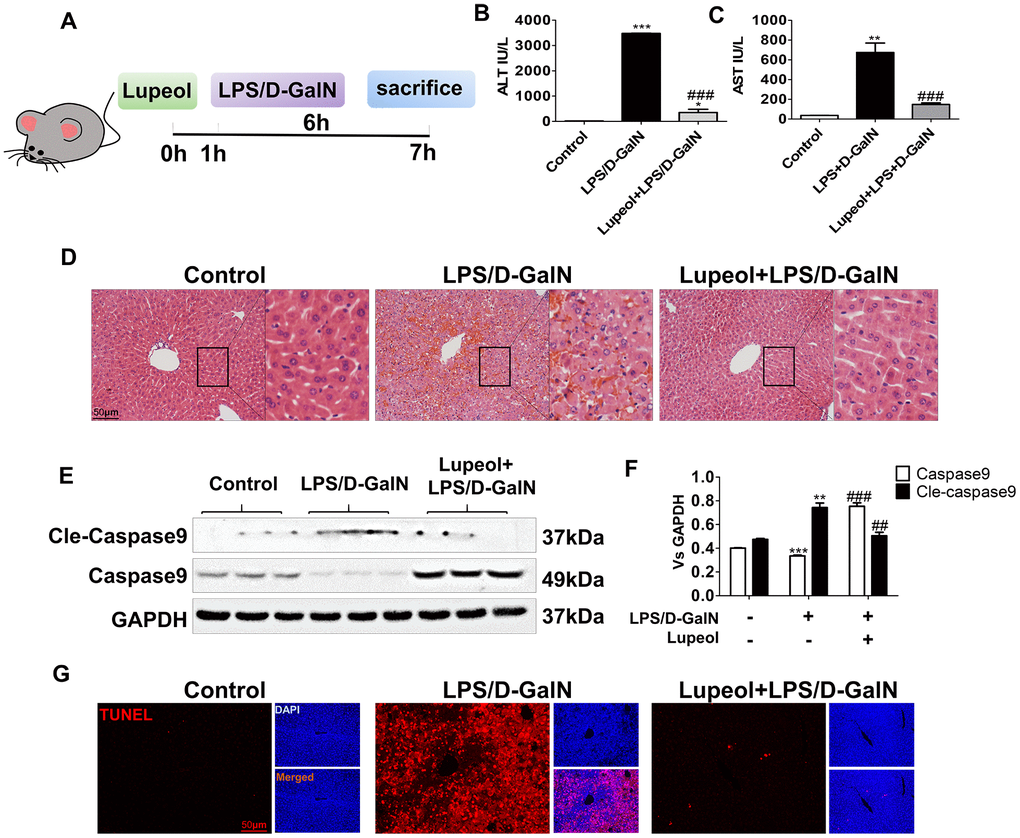
Figure 2. Lupeol alleviates LPS/GalN-induced liver injury in mice. (A) Diagrammatic sketch mice modeling and lupeol administration. (B, C) Changes in serum ALT and AST. (D) H&E staining was used to detect the liver histopathological changes. (E, F) Expression of caspase 9 and cleaved-caspase 9 protein in mice. (G) TUNEL staining of paraffin liver sections in control mice and mice treated with LPS/GalN or lupeol. Data are expressed as the mean ± SEM, n=3-6 per group, *P<0.05, **P<0.01, ***P<0.001, control group vs other groups. #P<0.05, ##P<0.01, ### P<0.001. Figures are magnified as 200x, bar=50μm.
Caspase 9, the initiator of apoptosis, activates the mitochondrial apoptosis pathway. Caspase 9 can combine with cytochrome c and the signal connector molecule Apaf-1 to form a complex after mitochondria release cytochrome c [28]. At the same time, it is cut into cleaved caspase 9. Cleaved caspase 9 further activates the downstream apoptosis executor caspase 3 to carry out a series of cascade reactions, which leads to apoptosis. In our study, the expression of caspase 9 was decreased, while cleaved caspase 9 was increased after LPS/D-GalN administration. After lupeol treatment, the expression of caspase-9 was increased, but the expression of cleaved caspase 9 was decreased (Figure 2E, 2F). Moreover, we used TUNEL staining to detect hepatocyte apoptosis in liver tissues and found that a large area of hepatocyte apoptosis occurred in the liver with LPS/D-GalN coinjection, while a significant reduction in hepatocyte apoptosis appeared with lupeol pretreatment (Figure 2GSupplementary Figure 1B).
These data showed that administration of LPS/D-GalN induced fatal ALI in mice. Lupeol inhibited aggravated liver injury by reducing hepatocyte apoptosis and decreasing the expression of cleaved caspase 9.
Lupeol restrains LPS/D-GalN-induced inflammatory responses in zebrafish and in mice
LPS/D-GalN induces liver injury mainly through inflammation. Generally, inflammation is caused by the release of TNF-α from natural killer cells, T lymphocytes and macrophages [29]. Vascular cell adhesion molecule-1 (VCAM-1), an adhesion molecule activated by TNF-α, recruits leukocytes to injury sites to initiate inflammatory responses [30, 31]. We found that lupeol treatment effectively reduced the expression of VCAM-1 in zebrafish (Figure 3A, 3B). Similarly, we also found that lupeol reduced VCAM-1 in mice (Figure 3C, 3D). Furthermore, we found that administration of lupeol significantly decreased the protein expression of TNF-α (Figure 3C–3F).
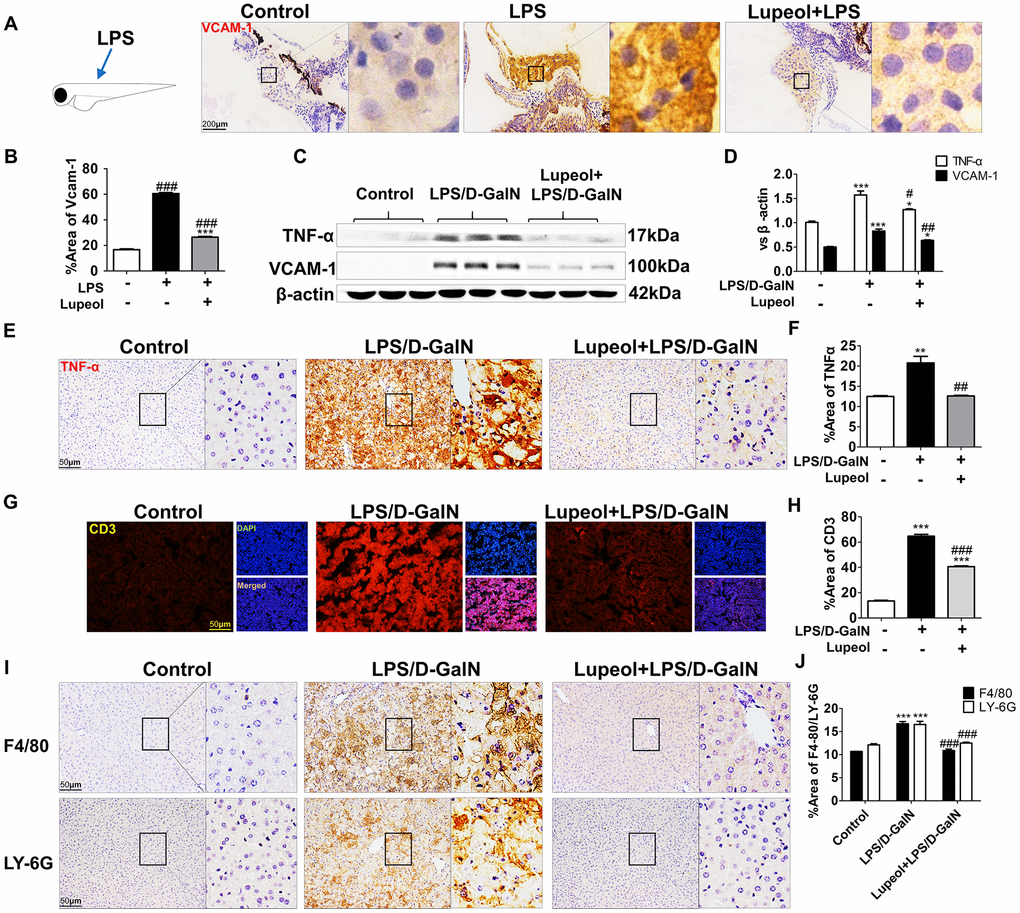
Figure 3. Lupeol restrains LPS/GalN-induced liver inflammation. (A) Immunohistochemistry staining of VCAM-1 in zebrafish, n=20, figures are magnified as 400x, bar=200μm. (B) Quantitative analysis of VCAM-1, data are represented as mean ± SEM. (C) Expression of TNF-α and VCAM-1 in mice. (D) Quantitative analysis of TNF-α and VCAM-1 protein. (E, F) Immunohistochemistry analysis for TNF-α location and expression in mice. (G, H) Immunofluorescence analysis for CD3 expression in mice. (I, J) Immunohistochemistry staining of F4/80 and LY-6G in mice and quantification of F4/80 and LY-6G expression. Data are shown as the mean ± SEM, n=3-4 group, *P<0.05, **P<0.01, ***P<0.001, control group vs other groups. #P<0.05, ##P<0.01, ### P<0.001. Figures are magnified as 200x, bar=50μm.
F4/80, a marker of macrophages, Ly-6G, a marker of neutrophils, and CD3, a marker of T cells, were detected by immunochemical staining in the livers of the mice. We found that compared with those in the control group, the numbers of cells with positive F4/80, Ly-6G and CD3 staining in the liver of the LPS/D-GalN-treated group were significantly increased, while pretreatment with lupeol inhibited the significant increase in immune cell infiltration (Figure 3G–3J). Collectively, these data indicated that lupeol reduced inflammatory responses in ALI.
Lupeol attenuates LPS/D-GalN-induced oxidative stress in zebrafish and in mice
Peroxynitrite (ONOO-) is a highly reactive oxygen species. Abnormal regulation of ONOO- in living systems is associated with diseases such as inflammatory conditions, auto-immune, and neurodegenerative diseases [32]. To detect the distribution and levels of reactive nitrogen species, we used liver-specific EGFP transgenic zebrafish and NP3, a fluorescent dye that can penetrate the cell membrane and blood-brain barrier, is suitable for detecting the level of ONOO- in living cells. After the larvae were incubated with the NP3 fluorescent probes for 10 min, we collected and photographed the larvae. When the level of ONOO- in the cell is higher, the blue fluorescence is stronger. We found that the level of RNS in zebrafish livers increased notably with LPS administration but decreased with treatment with 100 μM lupeol (Figure 4A, 4B).
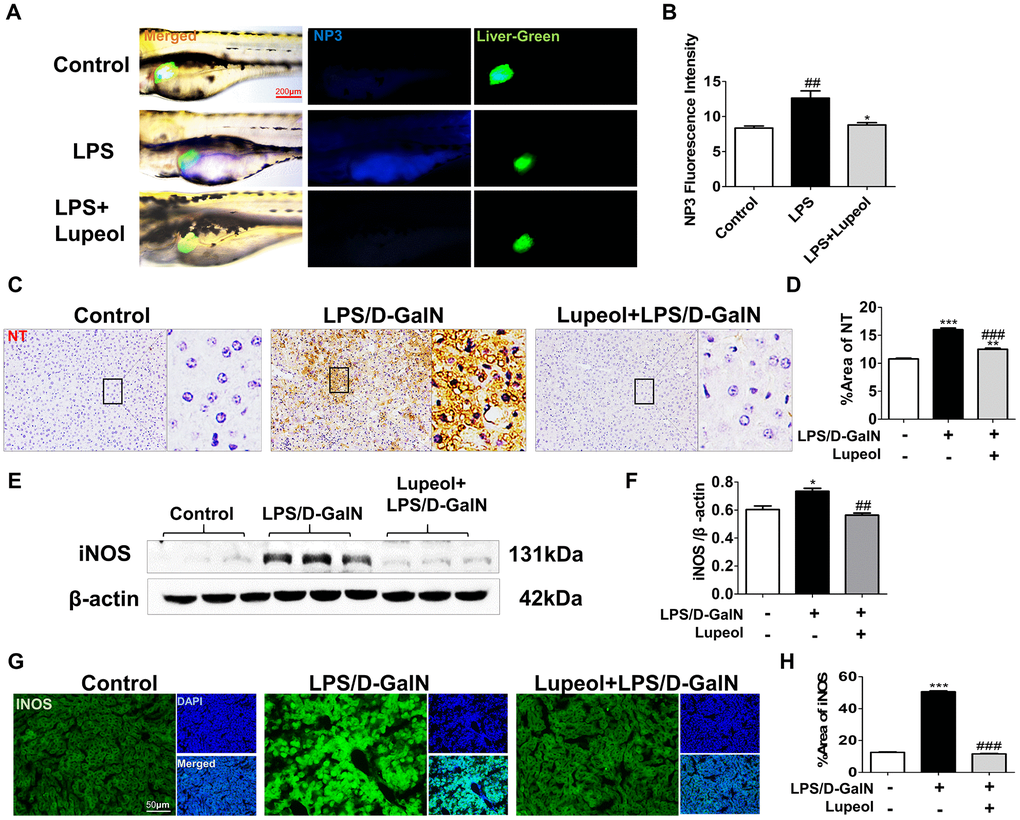
Figure 4. Lupeol reduces oxidative stress in zebrafish and in mice with ALI. (A) Fluorescence micrographs of ONOO- generation in the control zebrafish larvae and the zebrafish larvae treated with LPS or lupeol. N=3-5, figures are magnified as 400x, bar=200 μm. (B) Quantification of the amounts and distribution of ONOO-. Data are exhibited as mean ± SEM. (C, D) NT immunohistochemical staining of mice livers and its quantitative analysis. (E, F) Western blots and quantitative results for iNOS. (G, H) Immunofluorescence staining and quantification for iNOS. All data are shown as the mean ± SEM, n=3-4 group, *P<0.05 vs control group, #P<0.05 vs model group. Figures are magnified as 200x, bar=50μm.
Nitrotyrosine (NT) is an important tool for the detection of newly nitrosylated proteins, the determination of protein nitrosylation, and the measurement of nitrosylated protein levels in tissues and samples. Nitrated proteins were found to be significantly elevated in the livers of mice with LPS/D-GalN-induced injury. However, nitrated proteins decreased after lupeol treatment (Figure 4C, 4D). INOS levels increased in the livers of mice after LPS/D-GalN coinjection but decreased in the livers of mice pretreated with lupeol (Figure 4E–4H).
Generally, these results showed that lupeol could alleviate LPS/D-GalN-induced liver injury by inhibiting oxidative stress and partially by improving the antioxidant capacity in vivo.
Lupeol ameliorates LPS/D-GalN-induced liver injury through the TGFβ1 and Nrf2 pathway
Due to the pivotal role of the TGFβ1 pathway in the regulation of numerous cell processes, including extracellular matrix formation, cell proliferation, growth development, inhibition, and cell death [33], we detected whether lupeol treatment could affect the TGFβ1 pathway. Herein, lupeol efficiently repressed the protein expression of TGFβ1 in mice with LPS/D-GalN-induced ALI (Figure 5A, 5B).
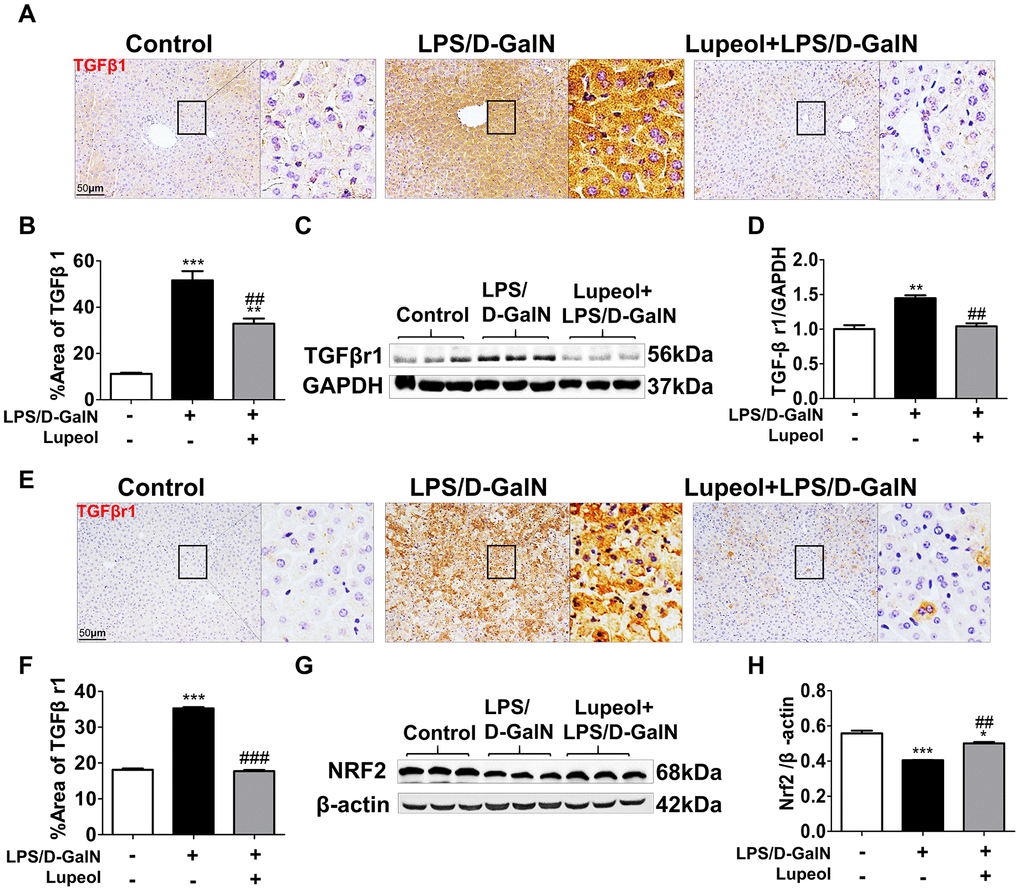
Figure 5. Lupeol downregulates TGFβr1 and upregulates Nrf2 pathway in mice with ALI. (A, B) Immunohistochemistry staining and quantitative analysis of TGFβ1. (C) Western blot analysis for the expressions of TGFβr1. (D) Quantitative analysis of TGFβr1 protein. (E, F) Immunohistochemistry analysis and quantification for detecting the expressions of TGFβr1. (G, H) Western blots and quantitative results for Nrf2. All data are shown as the mean ± SEM, n=3-4 group, *P<0.05 vs control group, #P<0.05 vs model group. Figures are magnified as 200x, bar=50μm.
Additionally, TGFβ1 transduces its signal by binding a heterotetramer receptor complex made up of TGF-β receptor 1 (TGFβr1) and TGF-β receptor 2 (TGFβr2). We found that lupeol could obviously decrease the protein expression of TGFβr1 in mice coinjected with LPS/D-GalN (Figure 5C–5F).
Moreover, the Nrf2 pathway plays a major role in regulating the expression of numerous antioxidant enzymes [34]. In our research, we found that lupeol promoted the expression of Nrf2, which was inhibited by LPS/D-GalN-induced liver injury (Figure 5G, 5H). In conclusion, lupeol could cure ALI by inhibiting TGFβ1 expression and promoting Nrf2 expression.
Discussion
ALI, a common disease that has high mortality, a poor prognosis, and no effective treatment, is associated with increased oxidative stress and inflammatory responses [5]. In this study, the principal findings obtained from histological and molecular studies are as follows. We found that lupeol could alleviate LPS/D-GalN-induced ALI by inhibiting inflammation caused by immune cell infiltration and oxidative stress resulting from RNS/ROS accumulation. Moreover, lupeol could protect the liver from LPS/D-GalN-induced injury by reducing the expression of TGFβr1 and increasing Nrf2, indicating that the TGFβr1-Nrf2 pathway was a possible target of lupeol.
Lupeol, which is present in diverse species of the plant kingdom, exhibits a spectrum of pharmacological activities against various diseases, such as cancer, microbial infections, arthritis, cardiovascular disease, diabetes, renal disease and liver disease [35–40]. A recent study demonstrated that lupeol ameliorated LPS/D-GalN-induced liver injury by inhibiting IRAK-mediated TLR inflammatory signaling [41]. In our study, we found that lupeol restored LPS/D-GalN -induced liver injury by repressing RNS/ROS accumulation and immune cell infiltration and that lupeol relieved ALI by regulating the TGFβ1 signaling pathway.
TGFβ1, which is involved in various stages of liver disease progression, plays a significant role in initial liver injury, liver inflammation, fibrosis and liver cancer [42, 43]. A previous study observed that Kupffer cells expressed high steady-state levels of TGFβ mRNA in CCl4-injured rat livers and that antisense S-oligodeoxynucleotides can restore CCL4-induced liver injury by downregulating TGFβ production [44]. As TGFβ plays a major role in liver regeneration, a recent study found that inhibition of TGFβR1 activity alleviated CCL4-induced intoxication by facilitating liver regeneration [45]. Moreover, a current study discovered that TGFβ signaling was activated in acute injury and that inhibition of TGFβR1 signaling reduced hepatocellular senescence by improving liver regeneration, function and outcome in acute liver injury [46]. In the present study, we further confirmed that TGFβ signaling was activated by LPS/GalN-induced liver injury. In addition, clinical trials of TGFβR1 or TGFβ inhibitors in human tumors are underway [46, 47]. Thus, our experiments provide a theoretical basis for the application of TGFβR1 or TGFβ inhibitors in acute liver damage and liver failure in humans.
Nrf2, considered a potential therapeutic target for preventing liver injury, plays an important role in the regulation of inflammation and oxidative stress [34, 48]. A recent study demonstrated that Nrf2 pathway was inhibited by a hepatotoxic drug matrine and accompanied with the activation of the ROS-mediated mitochondrial apoptosis pathway [49]. Moreover, another study showed that Licochalcone A has a hepatoprotective effect during LPS/GalN-induced liver injury by inducing the activation of Nrf2 and QSTM1(p62) signals and promoting autophagy via AMP-activated protein kinase (AMPK) signaling [50]. Furthermore, Nrf2 plays a major role in ameliorating various oxidative stress-associated diseases and exerts significant function in the antioxidant system, a recent study demonstrated that adropin reduced liver injury in nonalcoholic steatohepatitis by upregulating the expression of glutamate-cysteine ligase catalytic subunit(GCLC), glutamate-cysteine ligase regulatory subunit(GCLM) and glutathione peroxidase 1(Gpx1), dependent on Nrf2 transcriptional activity and increasing GSH levels [51]. Former study demonstrated that TGFβ1 induces Nrf2 mediated HO-1 expression and antioxidant response element activity in human aortic smooth muscle cells [11]. In this study, our results shown that lupeol administration improved hepatic antioxidant capacity to alleviate acute liver injury associated with Nrf2 up-regulation.
In summary, our results demonstrate that lupeol attenuates LPS/GalN-induced ALI by restraining hepatic inflammatory and oxidative stress and inhibits activation of TGFβ1 induced by LPS/GalN administration (Figure 6). However, our study does not address whether lupeol is effective for acute liver injury and failure in humans, and further human safety and efficacy studies are required. Furthermore, but there is no further experimental study on the effect of TGFβ1 on acute liver disease. Therefore, we will further clarify the signal transduction relationship between TGFβR1 and Nrf2 in our future research.
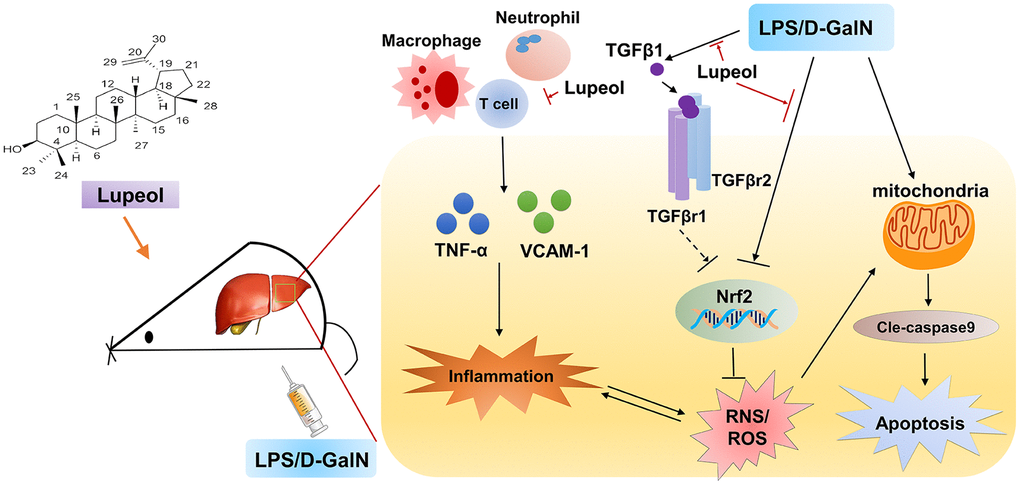
Figure 6. Graphical abstract: A schematic diagram describing the protective mechanisms of lupeol against LPS/GalN-induced acute liver injury.
Materials and Methods
Animals and ethics statement
Wild-type (WT) mice with a C57BL/6 background between 9 and 12 weeks of age were genotyped, housed in a temperature-controlled animal facility with alternating 12:12 h light-dark cycles and fasted overnight before surgery but allowed to drink water ad libitum. All the procedures used in the animal studies were approved by the National Institutional Animal Care and Ethical Committee of Southern Medical University. All the experimental methods performed in this study were in accordance with the approved guidelines.
ALI model and lupeol intervention
LPS (E. coli, L2630) and D-GalN (G0500) were purchased from Sigma (St. Louis, MO), and lupeol (B21602) was obtained from Shanghai Yuanye Bio-Technology. For the ALI model, male mice were intraperitoneally (i.p.) injected with LPS (20 μg/kg) 15 min before injection of D-GalN (700 mg/kg). Mice in drug treatment group was taken Lupeol (80 mg/kg each mouse) by intragastric administration 1 h before LPS and D-GalN treatment. Mice were sacrificed 6 h after LPS and D-GalN administration.
Zebrafish maintenance and treatment
WT and Tg(lfabp10α–EGFP) adults and larvae were maintained with a light: dark period of 14:10 h at 28° C. Embryos were gathered and developed in chorion water (0.5 mg/L methylene blue) for up to 5 days post fertilization (dpf) at 28.5° C. All zebrafish procedures were approved by the Institutional Animal Care and Use Committee of Southern Medical University.
Zebrafish larvae at 9-10 hpf were stochastically divided into six groups: the control group, the 0.1% DMSO group, the model group, and 3 therapy groups. In the control group, embryos were raised in fish water. In the 0.1% DMSO group, embryos were raised in 10 μg/mL LPS with 0.1% DMSO. In the model group, embryos were incubated in 10 μg/mL LPS until 3 dpf. In the therapy groups, lupeol was made soluble in dimethyl sulfoxide (DMSO) and diluted to 25 μM, 50 μM, and 100 μM with fish water. In addition, lupeol was added to the fish water 1 h before incubation in 10 μg/mL LPS. The larvae were raised in 6-well plates at a density of 30 larvae per well for approximately 61-62 h.
Histological analysis
Liver tissues or zebrafish larvae were fixed in 4% PFA overnight at 4° C, dehydrated, soaked in xylene, and embedded in paraffin in sequence and then sliced into 4 μm sections. Paraffin sections were dewaxed with xylene, dehydrated with different concentrations of ethanol, stained with hematoxylin and eosin, dehydrated, cleared, sealed, and finally imaged under a light microscope (Nikon Eclipse Ni-U; Nikon, Tokyo, Japan).
Serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) measurements
Serum was acquired by centrifugation of blood samples at 3000 g for 15 min, and AST and ALT activities were measured using an Alanine Aminotransferase Assay Kit (Nanjing Jiancheng Bioengineering Institute, C009-2-1) and an Aspartate Aminotransferase Assay Kit (Nanjing Jiancheng Bioengineering Institute, C010-2-1) according to the manufacturer’s instructions on a microplate reader at 510 nm.
Immunofluorescence and immunochemical staining
Liver tissues were collected and routinely embedded in OCT. Frozen liver samples were sliced into 14 μm sections. For immunofluorescence staining, after being washed with PBS 3 times, the sections were penetrated with methanol at -20° C for 10 min and sealed with 5% goat serum at room temperature for 1 h. Finally, frozen liver sections were stained with CD3 (Affinity, AF5405) and INOS (Abcam, ab178945) overnight at 4° C. After extensive washing, the frozen sections were incubated with the respective fluorescent secondary antibodies. Finally, the nucleus was stained with DAPI for 10 min.
For immunochemical staining, paraffin sections (4 μm) were first dewaxed in xylene I, II, and III and then rehydrated in 100%, 95%, 90%, 80%, and 70% ethanol. Then, the samples were boiled in 1X sodium citrate, maintained at a sub-boiling temperature for 10 min to repair antigen and cooled to room temperature. Afterwards, endogenous peroxidase enzyme was inactivated using 3% H2O2 in methanol for 10 min in the dark at room temperature. After blocking nonspecific binding with 5% goat serum at room temperature, the sections were stained with antibodies against F4/80 (Affinity, DF2789), NT (Cell Signaling Technology, 9691S), TNF-α (Abcam, ab1793), TGFβr1 (Abcam, ab31013), and TGFβ1 (HuaAn, H1113) overnight at 4° C. The next day, sections were incubated with the respective biotinylated secondary antibodies. Positive staining was visualized using DAB. The reaction was stopped in ice water. Then, the samples were counterstained with hematoxylin, dehydrated, paraffinized and finally mounted and sealed with neutral gum. The dyed sections were photographed with an optical microscope (Nikon Eclipse Ni-U; Nikon, Tokyo, Japan).
TUNEL assay
The TUNEL reaction was used to detect hepatocyte apoptosis in liver tissue by using frozen sections and the In Situ Cell Death Detection Kit (Roche). The cryosections were immersed in 0.01% Triton X-100 diluted in PBS for 10 min, washed with PBS, incubated with a 1:10 TUNEL working solution in a dark environment at 37° C for 1 h and washed 3 times with PBS. Then, DAPI was used to stain nuclei in the dark at room temperature for 5 min, and the samples were washed 3 times with PBS. The dyed cryosections were immediately photographed with an optical microscope (Nikon Eclipse Ni-U; Nikon, Tokyo, Japan).
Fluorescent probe detection
The fluorescent probe NP3 (FYRK-FP-01-001KY) was used to detect ONOO-. NP3 was used to measure RNS. At 3 dpf, live larvae were immediately transferred into 24-well plates and incubated with a 10 μM solution at 28° C in the dark for 10 min. Then, the fluorescence distribution of NP3 was visualized with a bright-field dissecting microscope (Nikon Eclipse Ni-U; Nikon, Tokyo, Japan).
Western blot analysis
Liver tissues were sonicated in ice-cold RIPA lysis buffer cell lysis buffer (Sigma) containing a phosphatase inhibitor cocktail (Sigma) and a protease inhibitor cocktail (Sigma). Protein concentrations were determined by a quantitative BCA assay. A total of 50 μg of protein was used for immunoblotting. Primary antibodies against INOS (1:1000, Abcam, ab178945), Nrf2 (1:1000, Proteintech, 16396-1AP), TGFβr1 (1:1000, Abcam, ab31013), caspase 9 (1:1000, Cell Signaling Technology, 9504P), cleaved caspase 9 (1:1000, Cell Signaling Technology, 9509P), TNF-α (1:1000, Abcam, ab1973), VCAM-1 (1:1000, HuaAn, HK0612), β-actin (1:2000, Affinity, T0022) and GAPDH (1:2000, Cell Signaling Technology, 2118S) were applied in the study.
Statistical analysis
The numerical results are shown as the mean ± standard deviation (SD). All statistical analyses were carried out with GraphPad Prism version 5.01 software and SPSS 20.0. One-way ANOVA or an unpaired t-test was used for statistical analysis, and Tukey’s multiple comparison test was used for the appropriate experiments. P-values less than 0.05 were considered statistically significant.
Supplementary Materials
Author Contributions
Lei Gao and Zhiping Lv: research concept and design; design of the study. Sha Huang: participated in the main experiments. Chan Mo, Ting Zeng, Yuqi Lai Chuying Zhou, Shuwen Xie, Limei Zhou and Yuhua Wang: organized the generation, collection, compilation and interpretation of data. Lei Gao and Zhiping Lv: participated in the drafting and revision of the manuscript. Shaohui Huang, Lei Gao and Zhiping Lv: obtained funding. Yuyao Chen: provided scientific suggestions, research concept and design.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This study was supported by the National Natural Science Foundation of China (81774170, 82074131, 81603501 and 81673774), the Natural Science Foundation of Guangdong Province (2018B030306012), the Scientific Research Initiative Program of Southern Medical University (CX2017N001), and Scientific research project in hygienism of Shenzhen Guangming District Science and technology innovation Bureau(2020R01126).
References
- 1. Rutherford A, Chung RT. Acute liver failure: mechanisms of hepatocyte injury and regeneration. Semin Liver Dis. 2008; 28:167–74. https://doi.org/10.1055/s-2008-1073116 [PubMed]
- 2. Radi R. Oxygen radicals, nitric oxide, and peroxynitrite: redox pathways in molecular medicine. Proc Natl Acad Sci USA. 2018; 115:5839–48. https://doi.org/10.1073/pnas.1804932115 [PubMed]
- 3. Radi R. Nitric oxide, oxidants, and protein tyrosine nitration. Proc Natl Acad Sci USA. 2004; 101:4003–08. https://doi.org/10.1073/pnas.0307446101 [PubMed]
- 4. Li S, Tan HY, Wang N, Zhang ZJ, Lao L, Wong CW, Feng Y. The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci. 2015; 16:26087–124. https://doi.org/10.3390/ijms161125942 [PubMed]
- 5. Zhang P, Yin Y, Wang T, Li W, Li C, Zeng X, Yang W, Zhang R, Tang Y, Shi L, Li R, Tao K. Maresin 1 mitigates concanavalin A-induced acute liver injury in mice by inhibiting ROS-mediated activation of NF-κB signaling. Free Radic Biol Med. 2020; 147:23–36. https://doi.org/10.1016/j.freeradbiomed.2019.11.033 [PubMed]
- 6. Zuo L, Zhou T, Pannell BK, Ziegler AC, Best TM. Biological and physiological role of reactive oxygen species—the good, the bad and the ugly. Acta Physiol (Oxf). 2015; 214:329–48. https://doi.org/10.1111/apha.12515 [PubMed]
- 7. Ramos-Tovar E, Muriel P. Free radicals, antioxidants, nuclear factor-E2-related factor-2 and liver damage. J Appl Toxicol. 2020; 40:151–168. https://doi.org/10.1002/jat.3880 [PubMed]
- 8. Gum SI, Cho MK. Recent updates on acetaminophen hepatotoxicity: the role of nrf2 in hepatoprotection. Toxicol Res. 2013; 29:165–72. https://doi.org/10.5487/TR.2013.29.3.165 [PubMed]
- 9. Gum SI, Cho MK. The amelioration of N-acetyl-p-benzoquinone imine toxicity by ginsenoside Rg3: the role of Nrf2-mediated detoxification and Mrp1/Mrp3 transports. Oxid Med Cell Longev. 2013; 2013:957947. https://doi.org/10.1155/2013/957947 [PubMed]
- 10. Reisman SA, Csanaky IL, Aleksunes LM, Klaassen CD. Altered disposition of acetaminophen in Nrf2-null and Keap1-knockdown mice. Toxicol Sci. 2009; 109:31–40. https://doi.org/10.1093/toxsci/kfp047 [PubMed]
- 11. Churchman AT, Anwar AA, Li FY, Sato H, Ishii T, Mann GE, Siow RC. Transforming growth factor-beta1 elicits Nrf2-mediated antioxidant responses in aortic smooth muscle cells. J Cell Mol Med. 2009; 13:2282–92. https://doi.org/10.1111/j.1582-4934.2009.00874.x [PubMed]
- 12. Morikawa M, Derynck R, Miyazono K. TGF-β and the TGF-β family: context-dependent roles in cell and tissue physiology. Cold Spring Harb Perspect Biol. 2016; 8:a021873. https://doi.org/10.1101/cshperspect.a021873 [PubMed]
- 13. Wrana JL, Attisano L, Wieser R, Ventura F, Massagué J. Mechanism of activation of the TGF-beta receptor. Nature. 1994; 370:341–47. https://doi.org/10.1038/370341a0 [PubMed]
- 14. McMillin M, Galindo C, Pae HY, Frampton G, Di Patre PL, Quinn M, Whittington E, DeMorrow S. Gli1 activation and protection against hepatic encephalopathy is suppressed by circulating transforming growth factor β1 in mice. J Hepatol. 2014; 61:1260–66. https://doi.org/10.1016/j.jhep.2014.07.015 [PubMed]
- 15. Eguchi S, Kamlot A, Ljubimova J, Hewitt WR, Lebow LT, Demetriou AA, Rozga J. Fulminant hepatic failure in rats: survival and effect on blood chemistry and liver regeneration. Hepatology. 1996; 24:1452–59. https://doi.org/10.1002/hep.510240626 [PubMed]
- 16. Yu X, Guo R, Ming D, Deng Y, Su M, Lin C, Li J, Lin Z, Su Z. The transforming growth factor β1/Interleukin-31 pathway is upregulated in patients with hepatitis B virus-related acute-on-chronic liver failure and is associated with disease severity and survival. Clin Vaccine Immunol. 2015; 22:484–92. https://doi.org/10.1128/CVI.00649-14 [PubMed]
- 17. Miwa Y, Harrison PM, Farzaneh F, Langley PG, Williams R, Hughes RD. Plasma levels and hepatic mRNA expression of transforming growth factor-beta1 in patients with fulminant hepatic failure. J Hepatol. 1997; 27:780–88. https://doi.org/10.1016/s0168-8278(97)80313-3 [PubMed]
- 18. Saleem M. Lupeol, a novel anti-inflammatory and anti-cancer dietary triterpene. Cancer Lett. 2009; 285:109–15. https://doi.org/10.1016/j.canlet.2009.04.033 [PubMed]
- 19. Beserra FP, Vieira AJ, Gushiken LFS, de Souza EO, Hussni MF, Hussni CA, Nóbrega RH, Martinez ERM, Jackson CJ, de Azevedo Maia GL, Rozza AL, Pellizzon CH. Lupeol, a Dietary Triterpene, Enhances Wound Healing in Streptozotocin-Induced Hyperglycemic Rats with Modulatory Effects on Inflammation, Oxidative Stress, and Angiogenesis. Oxid Med Cell Longev. 2019. https://doi.org/10.1155/2019/3182627 [PubMed]
- 20. Oliveira-Junior MS, Pereira EP, de Amorim VC, Reis LT, do Nascimento RP, da Silva VD, Costa SL. Lupeol inhibits LPS-induced neuroinflammation in cerebellar cultures and induces neuroprotection associated to the modulation of astrocyte response and expression of neurotrophic and inflammatory factors. Int Immunopharmacol. 2019; 70:302–12. https://doi.org/10.1016/j.intimp.2019.02.055 [PubMed]
- 21. Preetha SP, Kanniappan M, Selvakumar E, Nagaraj M, Varalakshmi P. Lupeol ameliorates aflatoxin B1-induced peroxidative hepatic damage in rats. Comp Biochem Physiol C Toxicol Pharmacol. 2006; 143:333–39. https://doi.org/10.1016/j.cbpc.2006.03.008 [PubMed]
- 22. Prasad S, Kalra N, Shukla Y. Hepatoprotective effects of lupeol and mango pulp extract of carcinogen induced alteration in Swiss albino mice. Mol Nutr Food Res. 2007; 51:352–9. https://doi.org/10.1002/mnfr.200600113 [PubMed]
- 23. Sunitha S, Nagaraj M, Varalakshmi P. Hepatoprotective effect of lupeol and lupeol linoleate on tissue antioxidant defence system in cadmium-induced hepatotoxicity in rats. Fitoterapia. 2001; 72:516–23. https://doi.org/10.1016/s0367-326x(01)00259-3 [PubMed]
- 24. Sudhahar V, Kumar SA, Varalakshmi P. Role of lupeol and lupeol linoleate on lipemic-oxidative stress in experimental hypercholesterolemia. Life Sci. 2006; 78:1329–35. https://doi.org/10.1016/j.lfs.2005.07.011 [PubMed]
- 25. Yang D, Zheng X, Chen S, Wang Z, Xu W, Tan J, Hu T, Hou M, Wang W, Gu Z, Wang Q, Zhang R, Zhang Y, Liu Q. Sensing of cytosolic LPS through caspy2 pyrin domain mediates noncanonical inflammasome activation in zebrafish. Nat Commun. 2018; 9:3052. https://doi.org/10.1038/s41467-018-04984-1 [PubMed]
- 26. Kim EA, Ding Y, Yang HW, Heo SJ, Lee SH. Soft coral Dendronephthya puetteri extract ameliorates inflammations by suppressing inflammatory mediators and oxidative stress in LPS-stimulated zebrafish. Int J Mol Sci. 2018; 19:2695. https://doi.org/10.3390/ijms19092695 [PubMed]
- 27. Arvelo MB, Cooper JT, Longo C, Daniel S, Grey ST, Mahiou J, Czismadia E, Abu-Jawdeh G, Ferran C. A20 protects mice from D-galactosamine/lipopolysaccharide acute toxic lethal hepatitis. Hepatology. 2002; 35:535–43. https://doi.org/10.1053/jhep.2002.31309 [PubMed]
- 28. Kim BI, Kim JH, Sim DY, Nam M, Jung JH, Shim B, Lee J, Kim SH. Inhibition of JAK2/STAT3 and activation of caspase-9/3 are involved in KYS05090S-induced apoptosis in ovarian cancer cells. Int J Oncol. 2019; 55:203–10. https://doi.org/10.3892/ijo.2019.4795 [PubMed]
- 29. Kong DH, Kim YK, Kim MR, Jang JH, Lee S. Emerging roles of vascular cell adhesion molecule-1 (VCAM-1) in immunological disorders and cancer. Int J Mol Sci. 2018; 19:1057. https://doi.org/10.3390/ijms19041057 [PubMed]
- 30. Schlesinger M, Bendas G. Vascular cell adhesion molecule-1 (VCAM-1)—an increasing insight into its role in tumorigenicity and metastasis. Int J Cancer. 2015; 136:2504–14. https://doi.org/10.1002/ijc.28927 [PubMed]
- 31. Cook-Mills JM, Marchese ME, Abdala-Valencia H. Vascular cell adhesion molecule-1 expression and signaling during disease: regulation by reactive oxygen species and antioxidants. Antioxid Redox Signal. 2011; 15:1607–38. https://doi.org/10.1089/ars.2010.3522 [PubMed]
- 32. Cao J, An W, Reeves AG, Lippert AR. A chemiluminescent probe for cellular peroxynitrite using a self-immolative oxidative decarbonylation reaction. Chem Sci. 2018; 9:2552–58. https://doi.org/10.1039/c7sc05087a [PubMed]
- 33. McMillin M, Grant S, Frampton G, Petrescu AD, Williams E, Jefferson B, DeMorrow S. The TGFβ1 receptor antagonist GW788388 reduces JNK activation and protects against acetaminophen hepatotoxicity in mice. Toxicol Sci. 2019; 170:549–61. https://doi.org/10.1093/toxsci/kfz122 [PubMed]
- 34. Xu D, Xu M, Jeong S, Qian Y, Wu H, Xia Q, Kong X. The role of Nrf2 in liver disease: novel molecular mechanisms and therapeutic approaches. Front Pharmacol. 2019; 9:1428. https://doi.org/10.3389/fphar.2018.01428 [PubMed]
- 35. Siddique HR, Saleem M. Beneficial health effects of lupeol triterpene: a review of preclinical studies. Life Sci. 2011; 88:285–93. https://doi.org/10.1016/j.lfs.2010.11.020 [PubMed]
- 36. Min TR, Park HJ, Ha KT, Chi GY, Choi YH, Park SH. Suppression of EGFR/STAT3 activity by lupeol contributes to the induction of the apoptosis of human non-small cell lung cancer cells. Int J Oncol. 2019; 55:320–30. https://doi.org/10.3892/ijo.2019.4799 [PubMed]
- 37. Shen X, Cui X, Cui H, Jin Y, Jin W, Sun H. Geraniol and lupeol inhibit growth and promote apoptosis in human hepatocarcinoma cells through the MAPK signaling pathway. J Cell Biochem. 2019; 120:5033–41. https://doi.org/10.1002/jcb.27779 [PubMed]
- 38. Hsu MJ, Peng SF, Chueh FS, Tsai CH, Tsai FJ, Huang CY, Tang CH, Yang JS, Hsu YM, Huang WW, Chung JG. Lupeol suppresses migration and invasion via p38/MAPK and PI3K/Akt signaling pathways in human osteosarcoma U-2 OS cells. Biosci Biotechnol Biochem. 2019; 83:1729–39. https://doi.org/10.1080/09168451.2019.1606693 [PubMed]
- 39. Prabhu B, Sivakumar A, Sundaresan S. Diindolylmethane and lupeol modulates apoptosis and cell proliferation in N-Butyl-N-(4-Hydroxybutyl) nitrosamine initiated and dimethylarsinic acid promoted rat bladder carcinogenesis. Pathol Oncol Res. 2016; 22:747–54. https://doi.org/10.1007/s12253-016-0054-9 [PubMed]
- 40. Machado VR, Sandjo LP, Pinheiro GL, Moraes MH, Steindel M, Pizzolatti MG, Biavatti MW. Synthesis of lupeol derivatives and their antileishmanial and antitrypanosomal activities. Nat Prod Res. 2018; 32:275–81. https://doi.org/10.1080/14786419.2017.1353982 [PubMed]
- 41. Kim SJ, Cho HI, Kim SJ, Kim JS, Kwak JH, Lee DU, Lee SK, Lee SM. Protective effects of lupeol against D-galactosamine and lipopolysaccharide-induced fulminant hepatic failure in mice. J Nat Prod. 2014; 77:2383–88. https://doi.org/10.1021/np500296b [PubMed]
- 42. Fabregat I, Moreno-Càceres J, Sánchez A, Dooley S, Dewidar B, Giannelli G, Ten Dijke P, and IT-LIVER Consortium. TGF-β signalling and liver disease. FEBS J. 2016; 283:2219–32. https://doi.org/10.1111/febs.13665 [PubMed]
- 43. Dooley S, ten Dijke P. TGF-β in progression of liver disease. Cell Tissue Res. 2012; 347:245–56. https://doi.org/10.1007/s00441-011-1246-y [PubMed]
- 44. Armendáriz-Borunda J, LeGros L
Jr , Campollo O, Panduro A, Rincón AR. Antisense S-oligodeoxynucleotides down-regulate TGFbeta-production by kupffer cells from CCl4-injured rat livers. Biochim Biophys Acta. 1997; 1353:241–52. https://doi.org/10.1016/s0167-4781(97)00066-3 [PubMed] - 45. Karkampouna S, Goumans MJ, Ten Dijke P, Dooley S, Kruithof-de Julio M. Inhibition of TGFβ type I receptor activity facilitates liver regeneration upon acute CCl4 intoxication in mice. Arch Toxicol. 2016; 90:347–57. https://doi.org/10.1007/s00204-014-1436-y [PubMed]
- 46. Bird TG, Müller M, Boulter L, Vincent DF, Ridgway RA, Lopez-Guadamillas E, Lu WY, Jamieson T, Govaere O, Campbell AD, Ferreira-Gonzalez S, Cole AM, Hay T, et al. TGFβ inhibition restores a regenerative response in acute liver injury by suppressing paracrine senescence. Sci Transl Med. 2018; 10:eaan1230. https://doi.org/10.1126/scitranslmed.aan1230 [PubMed]
- 47. Neuzillet C, Tijeras-Raballand A, Cohen R, Cros J, Faivre S, Raymond E, de Gramont A. Targeting the TGFβ pathway for cancer therapy. Pharmacol Ther. 2015; 147:22–31. https://doi.org/10.1016/j.pharmthera.2014.11.001 [PubMed]
- 48. Jadeja RN, Upadhyay KK, Devkar RV, Khurana S. Naturally occurring Nrf2 activators: potential in treatment of liver injury. Oxid Med Cell Longev. 2016; 2016:3453926. https://doi.org/10.1155/2016/3453926 [PubMed]
- 49. You L, Yang C, Du Y, Liu Y, Chen G, Sai N, Dong X, Yin X, Ni J. Matrine exerts hepatotoxic effects via the ROS-dependent mitochondrial apoptosis pathway and inhibition of Nrf2-mediated antioxidant response. Oxid Med Cell Longev. 2019; 2019:1045345. https://doi.org/10.1155/2019/1045345 [PubMed]
- 50. Lv H, Yang H, Wang Z, Feng H, Deng X, Cheng G, Ci X. Nrf2 signaling and autophagy are complementary in protecting lipopolysaccharide/d-galactosamine-induced acute liver injury by licochalcone A. Cell Death Dis. 2019; 10:313. https://doi.org/10.1038/s41419-019-1543-z [PubMed]
- 51. Chen X, Xue H, Fang W, Chen K, Chen S, Yang W, Shen T, Chen X, Zhang P, Ling W. Adropin protects against liver injury in nonalcoholic steatohepatitis via the Nrf2 mediated antioxidant capacity. Redox Biol. 2019; 21:101068. https://doi.org/10.1016/j.redox.2018.101068 [PubMed]



