Introduction
Aging involves the progressive loss of physiological functions in all organs and tissues and therefore, increases vulnerability to death [1]. Age-related diseases, such as cancer, osteoporosis, and diabetes, are associated with an increase in cellular senescence [2, 3]. In fact, many age-related diseases occur in young individuals in response to genetic and environmental insults [3]. Thus, defining the genes and pathways that control aging is essential to improve the treatment of age-related diseases.
Target of rapamycin (TOR) signaling is strongly implicated in the aging processes of diverse organisms, including yeast, worms, flies, and mammals [4]. TOR is an evolutionally conserved serine/threonine kinase that forms the catalytic subunit in two multi-protein complexes, TORC1 and TORC2, which have distinct structures and functions in metabolism [5]. TORC1 is sensitive to rapamycin and plays a central role in cell growth. It controls the balance between anabolism and catabolism in response to multiple inputs, including amino-acid availability, growth factor levels, and intracellular energy status. When nutrients are sufficient, TORC1 is recruited to lysosomal membranes by the Rags GTPase, where it comes in contact with the Rheb GTPases, which promote TORC1 kinase activity [6, 7]. Activated TORC1 phosphorylates multiple downstream effectors, such as S6K and 4E-BP, to increase the synthesis of proteins, lipids, and nucleotides. In contrast, when nutrients are scarce, TORC1 is inactive and this triggers catabolic metabolism and autophagy to provide energy for cell survival. Thus, cells can rapidly fine-tune their metabolic state in response to the environment by modulating the activity of TORC1 [8].
In Drosophila, Rag GTPase is comprised of a heterodimer of RagA and RagC [6, 7]. When nutrients are sufficient, RagA binds GTP and RagC binds GDP to promote the recruitment of TORC1 to lysosomes, where the GTP-loaded form of Rheb GTPase binds and directly stimulates TORC1 kinase activity [9]. Two complexes, known as TSC and GATOR1, function as GAPs (GTPase-activating proteins) towards Rheb and RagA, respectively and thus, act as inhibitors of TORC1 activity [10, 11]. TSC comprises TSC1 and TSC2 and is repressed by growth factors and high cellular energy levels [12]. The GATOR1 complex, composed of Nprl2, Nprl3, and DEPDC5/Iml1, is inactivated by amino acids [13, 14]. In Drosophila and mammals, depleting any of the GATOR1 or TSC complex components results in a dramatic increase in TORC1 activity and cell size [13, 14]. Thus, TSC and GATOR1 cooperatively control TORC1 activity. Genetic manipulation or pharmacological treatment that reduces TORC1 activity promotes longevity in multiple model organisms, including yeast, Drosophila, and mice [15–17]. Although the relationship between decreased TORC1 activity and longevity is widely established, the mechanism remains unclear. Several models have been suggested as to how reduced TORC1 activity affects aging, including a reduction in global mRNA translation, increased autophagy, and the stimulation of stress-resistance pathways [3, 4].
One system that is affected by aging in humans is the digestive system or the gastrointestinal (GI) tract. The GI tract functions as a digestive system to absorb energy and nutrients from the environment and as a barrier epithelium to protect the animal from the exterior environment. Disruptions in this epithelial barrier negatively affect animal health and significantly decrease lifespan [18]. The human GI tract is divided into four distinct sections: the esophagus, stomach, small intestine, and large intestine (colon) [19]. They are separated from each other and control the movement of food or food residues from one section to another. During this process, the ingested food is digested, and the nutrients are absorbed. Finally, the remaining waste is expelled from the body as feces. Gastrointestinal motility is required to efficiently shuttle liquified food from the stomach into the intestine and thus, is essential to health [19, 20]. There are multiple changes associated with GI tract aging, including gastric motility, intestinal hyperplasia, and hormone secretion [21, 22]. Some clinical reports have shown increased morbidity associated with dysfunctional GI tract motility and gastric emptying in the aged population [20]. However, the underlying cellular and molecular mechanisms involved in the effect of aging on GI tract motility and gastric emptying are largely unknown.
The Drosophila digestive tract is divided into three discrete domains: foregut, midgut, and hindgut [23, 24]. The foregut, comprising the crop, esophagus, and cardia, serves as a storage site for ingested food [25, 26]. Functionally similar to the stomach, the crop is a structure consisting of a complex array of valves and sphincters that move food contents into the midgut. The midgut is a long tubular structure that is equivalent to the mammalian small intestine and is the main site for food digestion and nutrient absorption. The epithelial layer of the midgut mainly consists of two differentiated cell types - polyploid absorptive enterocytes (ECs) and hormone-secreting enteroendocrine (EE) cells, which originate from intestinal stem cells (ISCs) residing basally in the intestinal epithelium [27, 28]. Recent studies have established the Drosophila intestine as a model system to study intestinal tissue homeostasis, immune responses, and stress signaling [23, 24]. Damage to the intestinal epithelium can trigger ISC division and differentiation to replace lost cells and maintain tissue homeostasis [29]. The aging intestines exhibit increased proliferation of ISCs and reduced regeneration of epithelial cells, which leads to gut hyperplasia; metabolic disorders; barrier leakage; immune senescence; and ultimately, the death of the fly [30]. However, the mechanism of digestive dysfunction in aging flies remains elusive.
Here, we used mutations in the GATOR1 complex component, Nprl2, to explore how TORC1 activity promotes age-related physiological defects in the GI tract. Importantly, we found that crop emptying and GI tract motility defects in nprl2-mutant flies are phenocopied in aged wild-type flies. Moreover, we observed that the nprl2-mutant midgut displays age-related defects in gut morphology, ISC populations, metabolism, and immune responses. Our findings highlight the importance of TORC1 activity in aging and age-related GI tract disease and suggest that the Drosophila crop can be a useful model system to investigate the mechanisms of gastric emptying dysfunction in elderly individuals.
Results
The nprl2-mutant flies display shorter lifespan
Recently, the TOR pathway has emerged as an important modulator of aging [4]. Genetic and pharmacological inhibition of TORC1 activity extends lifespan and prevents multiple age-related diseases in several model organisms [17, 31–33]. Thus, we reasoned that mutations in nprl2, which have been previously shown to increase TORC1 activity, might affect Drosophila longevity [34]. To test this possibility, we compared the lifespan of nprl2 null mutants, nprl21, with the lifespan of the control stock, yw, under standard culture condition. As shown in Figure 1, the median lifespan of nprl21 flies (34 d) was markedly shorter than that of wild-type flies (64 d). Thus, mutations in nprl2 resulted in significantly decreased Drosophila lifespan.
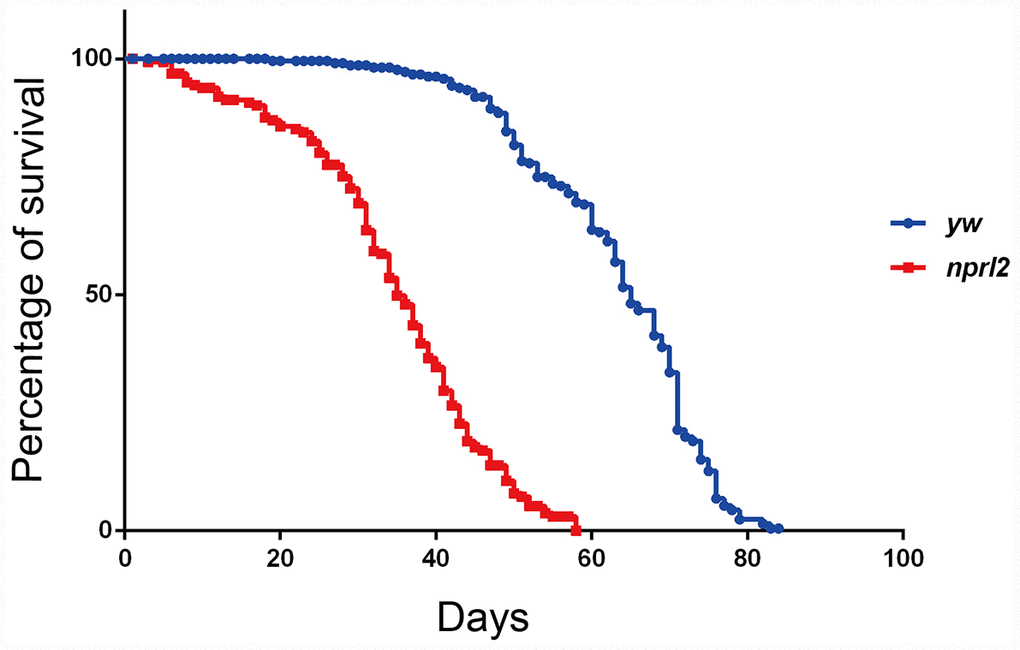
Figure 1. The nprl2 mutant had a decreased lifespan. Newly hatched male flies were cultured on standard food and counted each day. Survival curves of yw (n = 205) and nprl21 (n = 154) flies are shown. Pairwise comparisons by the Mantel-Cox log rank test showed P < 0.0001.
Nprl2 is required for GI tract digestive function
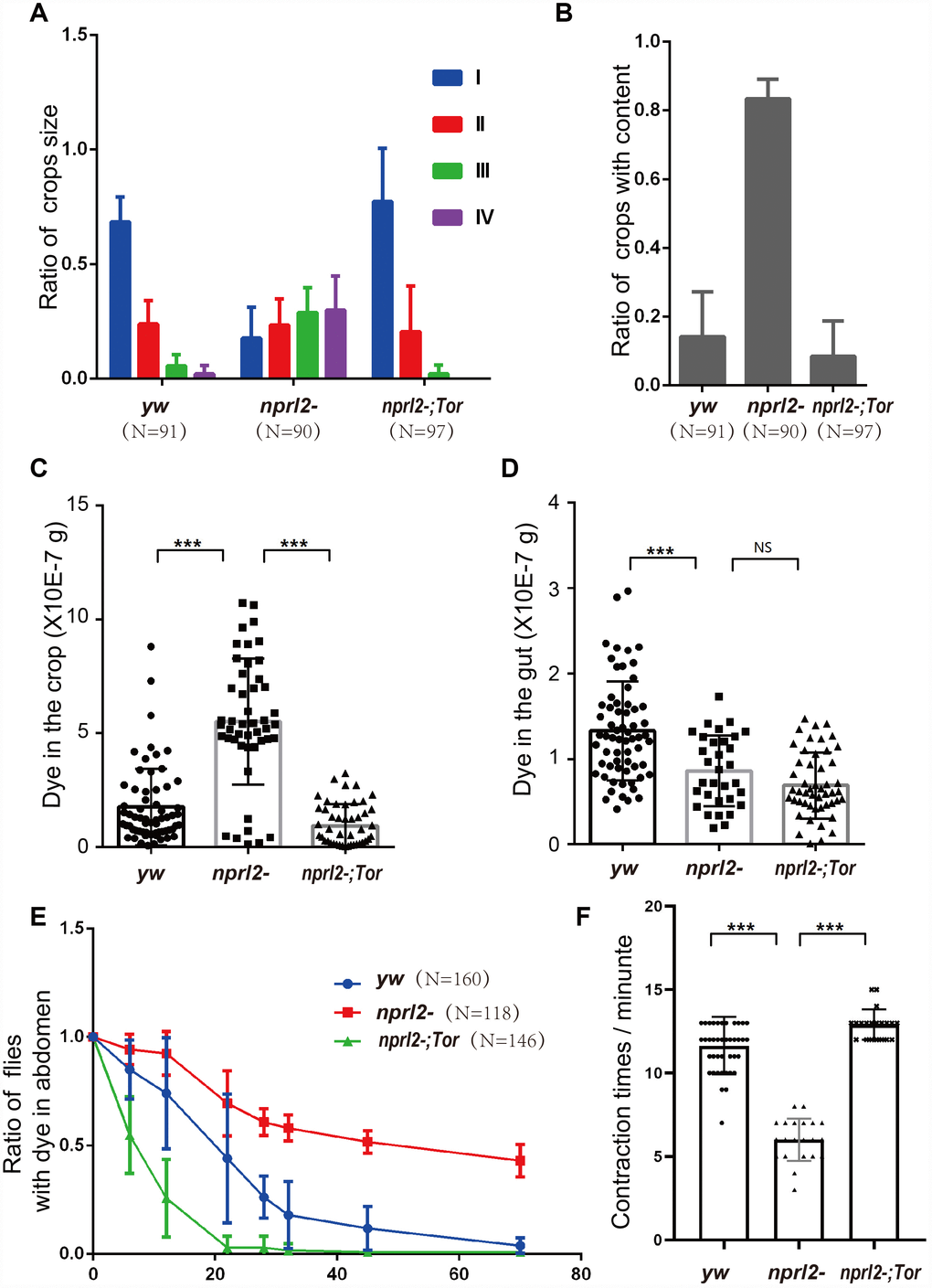
In humans, age contributes to decreased gastrointestinal tract motility and negatively affects GI tract digestive function, resulting in symptoms such as esophageal reflux, delayed gastric emptying, and constipation [19, 20]. Similarly, older monkeys have dramatically reduced intestinal motility [35]. We aimed to determine if Drosophilanprl2 mutants, which appear to have accelerated aging, exhibit age-related gastric dysfunction. The Drosophila crop, a foregut organ that is functionally comparable to the mammalian stomach, is primarily a reservoir for food and nutrients [23, 36]. Wild-type flies rarely exhibit large or extended crops when they have free access to food [37]. In contrast, we found that nplr2-mutant flies often developed a swollen crop as they increased in age. To better define the extent of the distended crop phenotype, we scored the crops for size and placed them in one of four categories, with 1 being the smallest crop and 4 being the largest (Figure 2A). Young wild-type adults had a small crop size, with less than 2% of flies showing crop sizes in class 3 or 4. As adult flies aged, the propensity for a large crop size increased, with approximately 10% of wild-type adults exhibiting a large crop size (class 3 or 4) at 15 d. This number rose to approximately 20% at 25 d, suggesting that age promoted crop distension in wild-type flies (Figure 2A). Consistent with the hypothesis that mutations in nprl2 accelerate the aging process, at 15 d, 50% of nprl2-mutant adults had a large crop size (class 3 or 4), which was nearly a five-fold increase relative to control adults. With the age increased, approximately 70% of nprl2-mutant adults displayed large crop at 25 d. As the crops of nprl2 mutants were small at 3 d (Figure 2A), we reasoned that the large crop size seen at 15 d resulted from increased food storage and/or retention. To test this hypothesis, we examined the percentage of nprl2-mutant and wild-type adults that clearly contained food in their crops. At 3 d, both control and nprl2-mutant flies had minimal food content in their crops. Consistent with the above findings that nprl2-mutant flies had larger crops at 15 and 25 days, the percentage of nprl2-mutant flies with crop food retention was significantly higher than the number of control flies with food retention (Figure 2B). To determine whether the distended crop was resulted from cell size increased in nprl2 mutant, we compared the crops that place in class I or class III in both control and nprl2 mutant flies (Supplementary Figure 1). Interestingly, the cell sizes between yw and nprl2 that place in the same class were similar. However, the cell sizes between small and large crops were obviously different, even the flies were same genotype. To confirm that the increased crop size and food accumulation in nprl2-mutant flies resulted from the loss of Nprl2, we overexpressed HA-tagged Nprl2, using the Ubi-GAL4 driver, in the nprl2-mutant background and found that the expression of Nprl2 fully rescued the crop size and food accumulation defects in 15-day-old nprl2 mutants (Supplementary Figure 2).
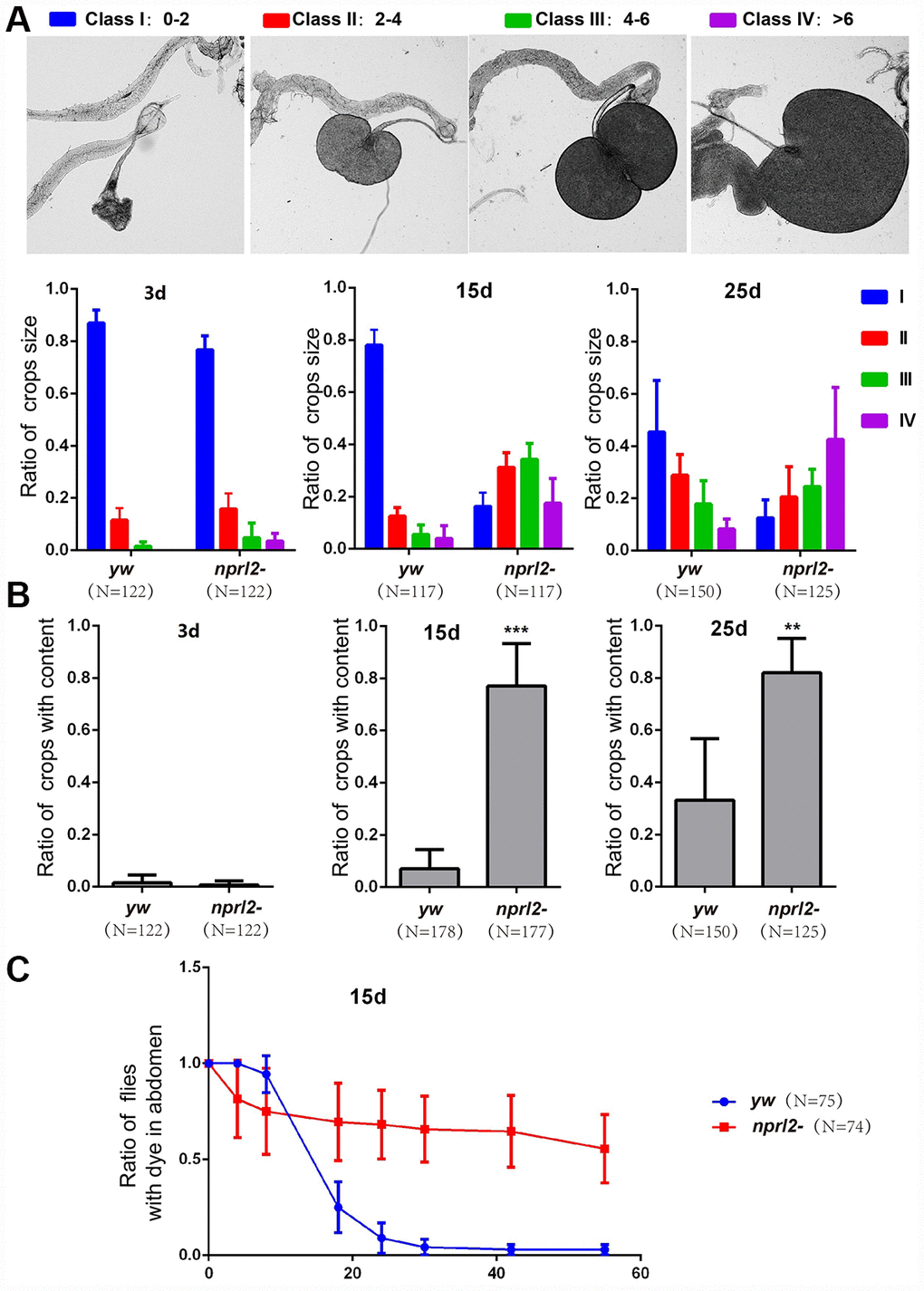
Figure 2. Nprl2 plays important roles in Drosophila digestive function. (A and B) Newly hatched yw and nprl21 flies were cultured on standard food for the indicated number of days and then dissected. (A) Crop sizes were grouped into four classes based on area (mm2). The ratio of different crop sizes was quantified. Error bars represent the SD from four independent experiments. (B) The ratio of flies with food content in their crops was quantified. Error bars represent the SD from four independent experiments. (C) 15 day-old flies were cultured on blue dyed food for 3 days and then transferred to standard food for the indicated time. The ratio of flies with blue dye in the digestive tube at each time point was calculated. Error bars represent the SD from three independent experiments. **P < 0.01, ***P < 0.001. N is the total number of flies used.
Although the Drosophila esophagus is directly connected to the proventriculus of the midgut, the ingested food is first diverted to the crop and is subsequently pumped into the midgut, rather than entering the midgut directly [36]. Thus, the crop plays a critical role in regulating food passaging. The abnormal size and amount of food accumulation in the crops of nprl2-mutant flies may be related to slow food passaging. To test this hypothesis, we fed flies with food that contains a non-absorbable blue dye (FD&C blue dye no. 1) and tracked the time taken to expel the dye. As shown in Figure 2C, less than 10% of control flies had dye in their digestive tracts after 24 hours. However, more than 50% of the nprl2-mutant flies still contained blue dye in their digestive tracts after 60 hours. Thus, crop (gastric) emptying was much slower in nprl2-mutant flies (Figure 2C). Taken together, our data indicated that Nprl2 is critical for the maintenance of digestive function in Drosophila.
The nprl2-mutant flies display GI tract motility defects
The phenotypes of distended crop, food storage, and prolonged food passage through the digestive tract found in nprl2-mutant flies may be caused by either excessive food ingestion or slow food digestion. To determine which of these two processes was involved, we first measured food ingestion by 15 day-old flies using the capillary feeder (CAFE) method [38]. As shown in Figure 3A, the amount of food intake was similar between control and nprl2-mutant flies. Next, we counted the number of feces to determine the differences in the rate of food digestion between nprl2-mutant and control flies. Interestingly, nprl2-mutant flies generated markedly less feces than control flies (Figure 3B). The reduction in defecation in nprl2-mutant flies may be due to slow GI motility. To test this hypothesis, we assessed food distribution in the GI tract by measuring the content of blue food dye in the crop and gut immediately after 30 min of feeding. As shown in Figure 3C, the crops of nprl2-mutant flies contained more blue dye than the crops of control flies. Consistent with the finding that the total amount of food intake was similar between nprl2-mutant and control flies, the guts of nprl2-mutant flies contained less food than the guts of control flies (Figure 3D). Thus, the movement of food was much slower from the crop to the gut in nprl2-mutant flies. Similar to the mammalian stomach, the crop is a structure consisting of a complex array of valves and sphincters that contracts dynamically to move the food into the midgut. We also found that the crop contraction rate was significantly reduced in nprl2-mutant flies (Figure 3E), which suggested that the muscle, neuron, or other factors that control crop contraction were affected by nprl2 mutation. These results suggested that GI tract dysmotility is related to slow food digestion in nprl2-mutant flies.
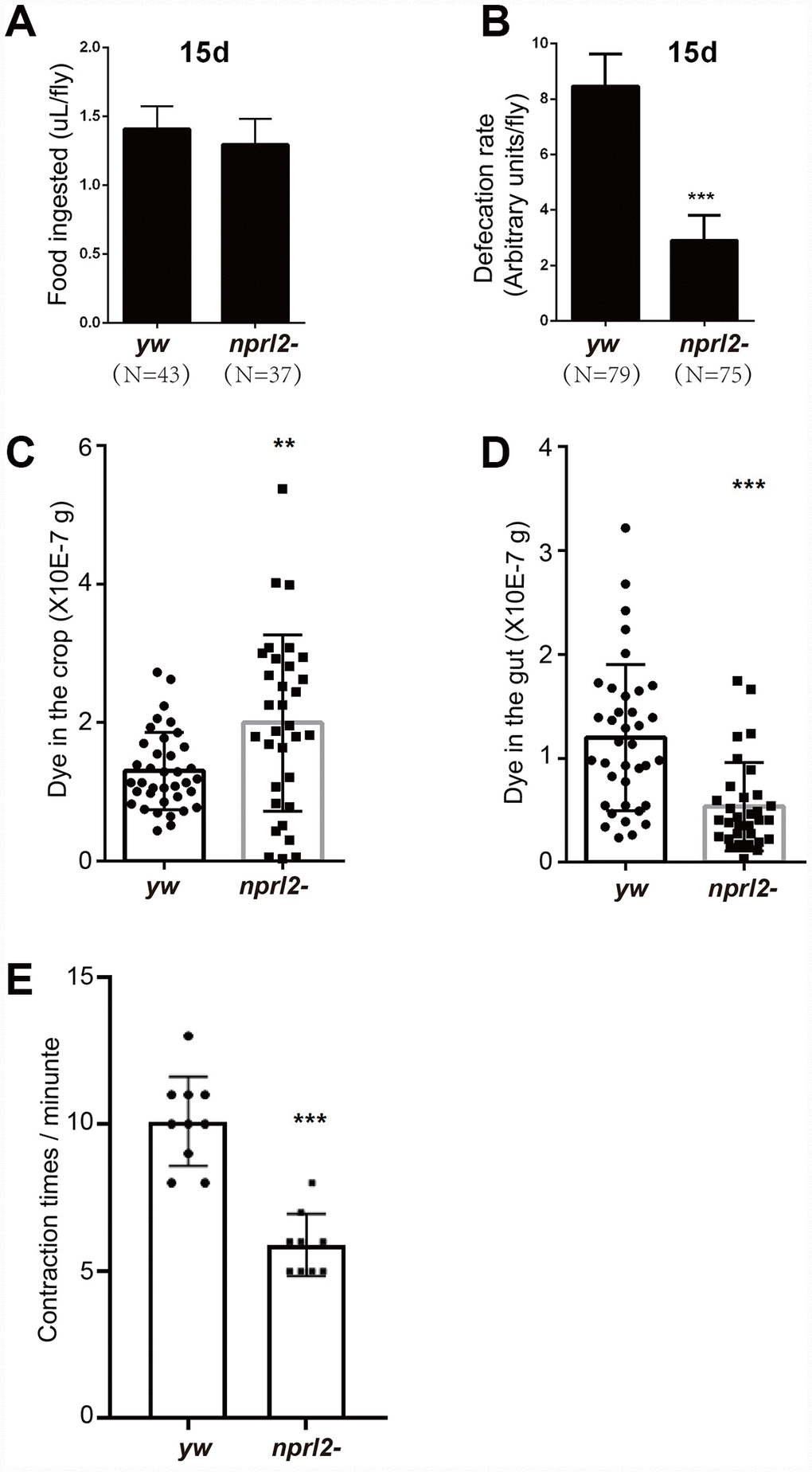
Figure 3. nprl2-mutant flies displayed GI tract motility defects. Fifteen-day-old yw and nprl21 males were used for the following analyses. (A) CAFE assay to quantify food ingestion. Error bars represent the SD from eight independent experiments. (B) Defecation rate assay to quantify food expulsion. Error bars represent the SD from five independent experiments. (C and D) Flies were starved in complete starvation medium (PBS with 1% agarose) for 10 hours and then fed dyed food for 30 min. The dyed food concentration in crops and guts was determined using a spectrophotometer. (E) The contraction rate of the crop was measured in dissected flies. Error bars represent the SD of the indicated number of data points. **P < 0.01, ***P < 0.001. N is the total number of flies used.
The nprl2-mutant flies display gut senescence
To efficiently acquire nutrients, food movement and digestion in the GI tract must be regulated and coordinated properly. Therefore, we further performed phenotypical analysis of the midgut, which is comparable to the mammalian intestine. Midgut epithelium homeostasis, maintained by ISCs, is essential for the preservation of intestinal integrity. During aging and stress, ISCs are induced to proliferate and generate daughter cells, which will further differentiate into ECs and EE cells to replace the damaged or lost cells and remodel the gut. Thus, gut length is dynamic and is influenced by aging or stress [39, 40]. Here we found that the guts of nprl2 mutants were markedly shorter and thinner than the guts of control flies at 15 d, which suggested that the nprl2 mutants failed to generate enough epithelial cells and that epithelial homeostasis was impaired (Figure 5). Furthermore, overexpressed HA-tagged Nprl2 in the nprl2-mutant background increased the gut length and width in 15-day-old nprl2 mutants (Supplementary Figure 5). Increased ISC proliferation and decreased terminal differentiation of ISC progeny is a hallmark of intestinal aging, which features the accumulation of polyploid cells expressing the ISC and enteroblasts (EB) marker, Esg. Previous studies have shown that loss of the tuberous sclerosis complex (TSC), another TORC1 inhibitor, results in ISC differentiation defects, with larger Esg+ cells and fewer EE cells, marked by Prospero expression [41, 42]. To test whether nprl2 mutants also have accelerated gut aging and decreased stem cell maintenance, we determined the cell composition at the posterior midgut. The nprl2-mutant flies had a greater number of ISCs marked by Esg expression and fewer EE cells marked by Prospero expression than control flies, which indicated the dysfunction of ISC maintenance and differentiation in nprl2 mutants (Figure 6). Interestingly, the gut length and cell composition phenotypes of nprl2 mutants were both rescued by the decrease in TORC1 activity through mutant one copy of Tor to TorA948V (Figures 5 and 6). These results suggested that the loss of epithelium maintenance is accelerated in nprl2-mutant flies through increased TORC1 activity.
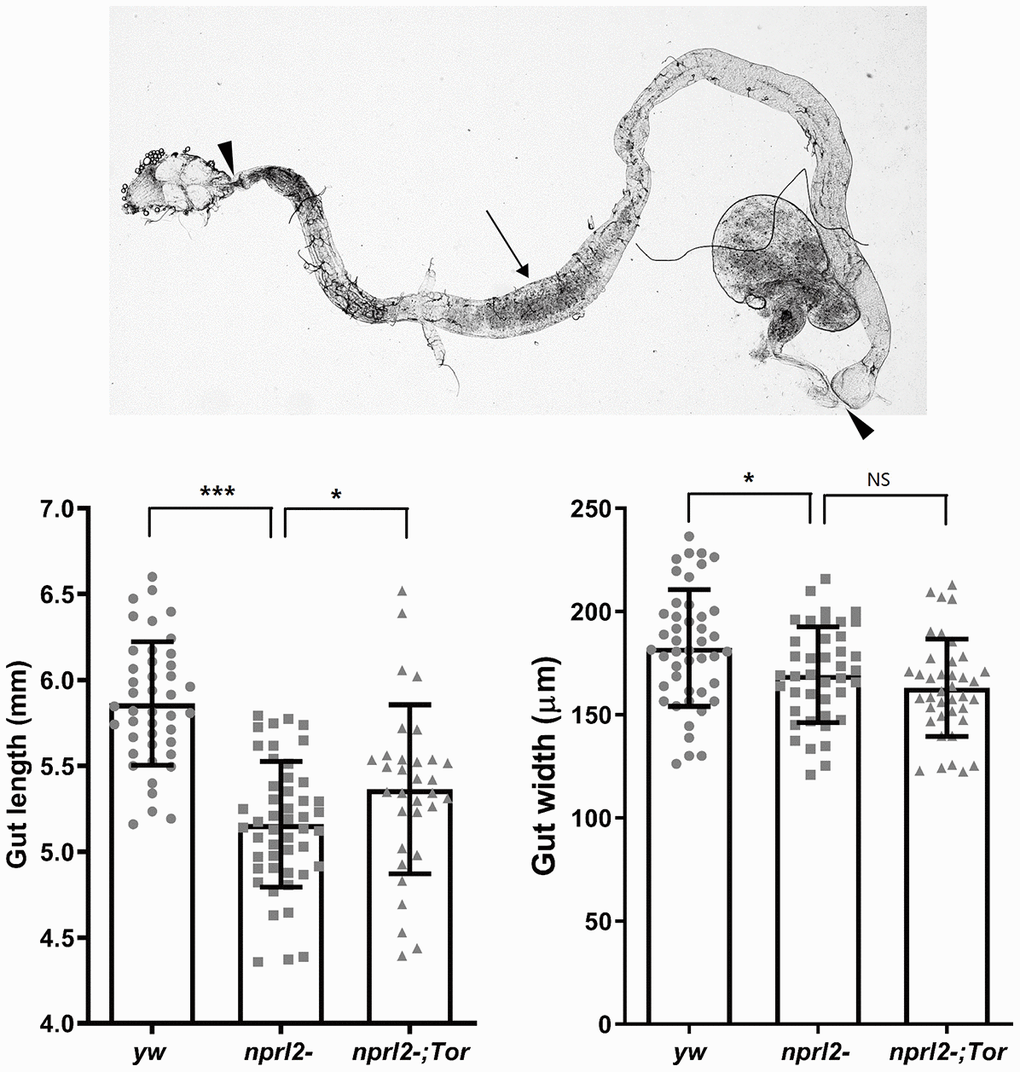
Figure 5. nprl2 mutation decreased gut length and width. Newly hatched male yw, nprl21, and nprl21; TorA594V/+ flies were cultured on standard food for 15 days and then dissected. Gut length (between the arrowhead) and gut width (arrow) were then measured. Error bars represent the SD of the indicated number of data points. *P < 0.05, ***P < 0.001. NS, not significant.
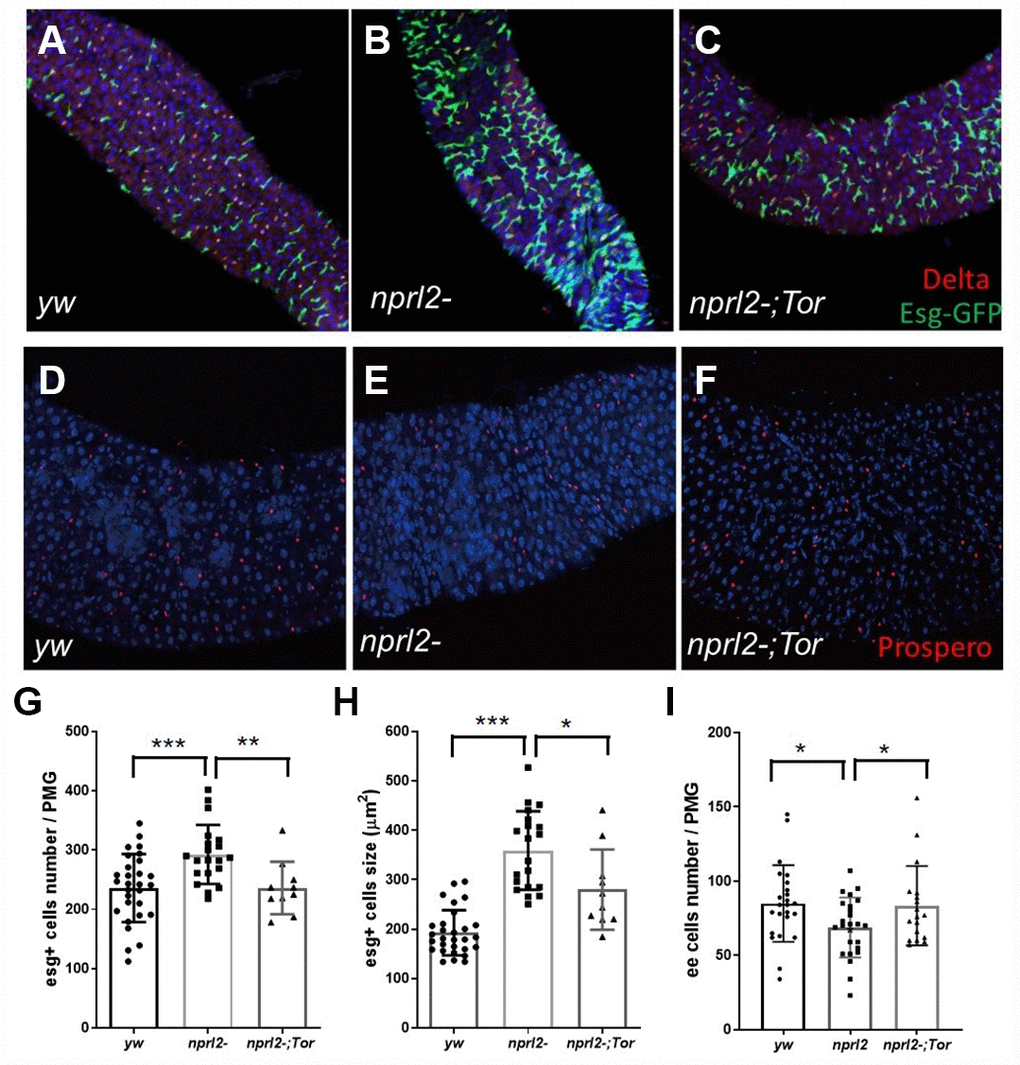
Figure 6. nprl2 mutation affected ISCs and EE cells in the midgut. (A–C) Newly hatched (A) yw; esg-Gal4, UAS-GFP, tub-Gal80ts, (B) nprl21; esg-Gal4, UAS-GFP, tub-Gal80ts and (C) nprl21; esg-Gal4, UAS-GFP, tub-Gal80ts / TorA594V flies were cultured on standard food for 15 days at 18°C, then transferred to 29°C and cultured for a further 3 days. Flies were dissected and stained with anti-Delta antibody. (D–F) Fifteen-day-old (D) yw, (E) nprl21 and (F) nprl21; TorA594V/+ flies were dissected and stained with an anti-Prospero antibody. (G) The number of Esg+ cells in the posterior midgut (PMG) was counted. (H) The size of Esg+ cells in the PMG was measured. (I) The number of Prospero+ cells in the PMG was counted. Error bars represent the SD of the indicated number of data points. *P < 0.05, **P < 0.01, ***P < 0.001.
Previous works have shown that the excessive proliferation and mis-differentiated ISCs are widely observed in the aged or damaged gut with a dysplastic phenotype [43, 44]. To monitor the proliferation of the ISCs, we used EdU method to detect the ratio of ISCs in S phase. Compared to only few ISCs labeled with EdU in control flies, large amount of ISCs in nprl2 mutant were labeled (Supplementary Figure 6). This result suggests that nprl2 mutant flies have more ISCs in proliferation state, which consistent with the hypothesis of nprl2 mutant promotes gut aging.
Next, we evaluated other common features of the aging gut, including metabolic disorders. A reduction in lipid storage in aged fly guts has been demonstrated through lipid-associated dye staining [45]. We used Nile red to stain fly guts and found that the nprl2-mutant flies had much less lipid storage than control flies in the anterior midgut at both 3 and l5 days (Figure 7). Furthermore, the amount of lipids was less at 15 days than at 3 days in control flies, which was consistent with previous reports that lipid storage decreases with age. These results suggested that nprl2 mutant promotes age-related metabolic disorder.
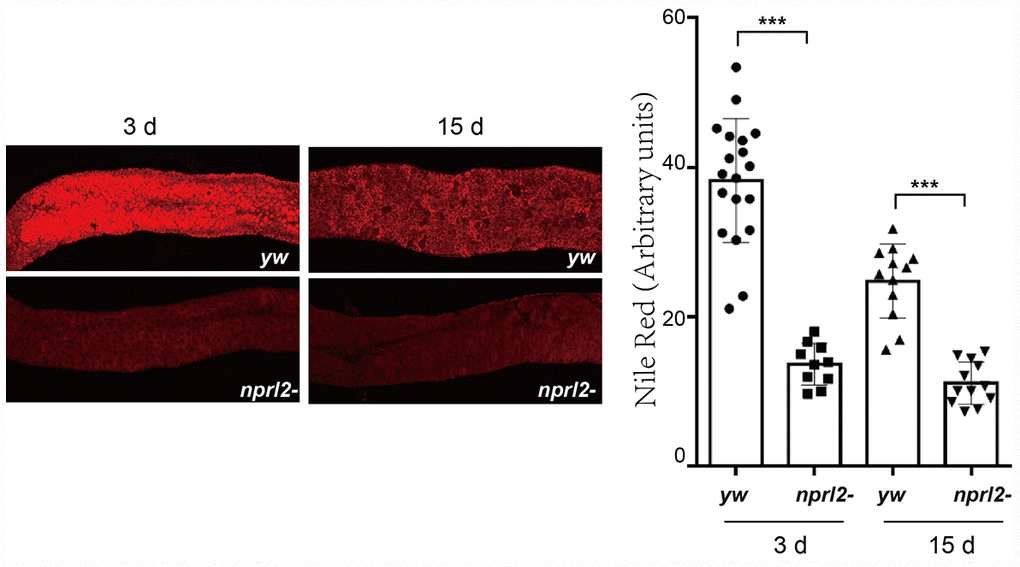
Figure 7. nprl2 mutation decreased lipid storage in the midgut. Newly hatched yw and nprl21 flies were cultured on standard food for 3 or 15 days and then dissected. The midgut was stained with Nile red. Error bars represent the SD of the indicated number of data points. ***P < 0.001.
The nprl2-mutant flies are sensitive to sodium dodecyl sulfate treatment
To assess the biological outcomes of the absence of Nprl2, we fed nprl2-mutant flies with the tissue-damaging agent, sodium dodecyl sulfate (SDS). SDS causes gut damage to flies and can be lethal [46]. When a low dose of 0.6% SDS in sucrose solution was given, the median lifespan of nprl21 flies (24 hours) was much shorter than the median lifespan of wild-type flies (90 hours, Figure 8). Increased vulnerability to a tissue-damaging reagent is consistent with the long staying of the food in the body. Interestingly, decreasing TORC1 activity through mutant one copy of Tor to TorA948V could partially rescued the effects of SDS treatment in nprl2 mutants, resulting in a median lifespan of 42 hours. Thus, these results suggested that Nprl2 protected the digestive tract from damage.
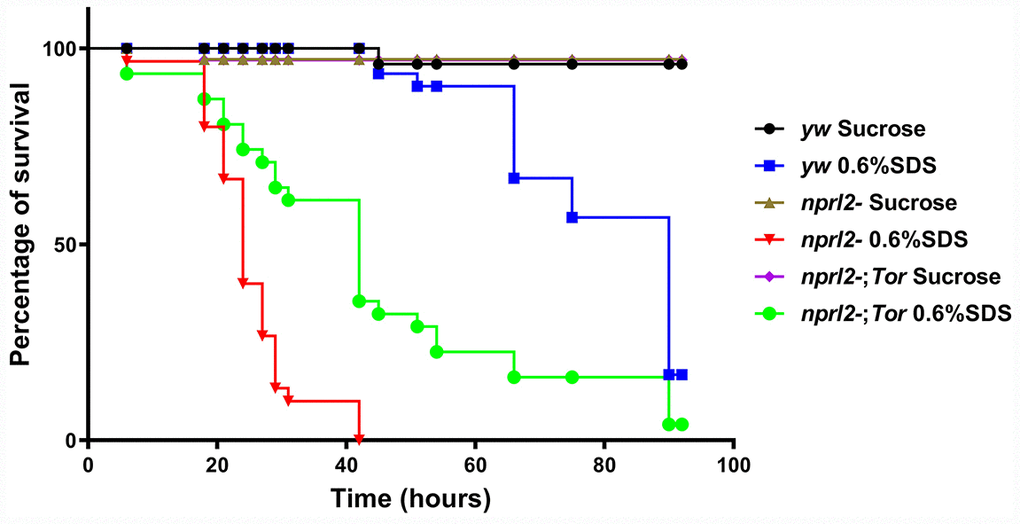
Figure 8. nprl2 mutants were sensitive to SDS treatment.yw,nprl21 and nprl21; TorA594V/+ flies were fed with 5% sucrose or with 0.6% SDS. Flies were counted at the indicated time points. Survival curves of the flies are shown. Pairwise comparisons by the Mantel-Cox log rank test showed that survival curves of the three genotypes of flies fed 0.6% SDS were different from each other: P < 0.001 (yw vs nprl21), P < 0.001 (nprl21 vs nprl21; TorA594V/+).
Discussion
Our study demonstrated the occurrence of abnormal crop size and digestive dysfunction with age. This age-related digestive disorder was reminiscent of GI motility and gastric emptying defects in the elderly, and thus underlines the relationship of aging and GI dysmotility. Mutation of nprl2 contributed to age-related crop distension and digestive dysfunction, and this could be rescued by decreasing TORC1 activity. The association between TORC1 activity and many age-related diseases remains elusive, but the work presented herein provides a possible mechanism for the promotion of digestive organ senescence by TORC1.
Aging affect crop size and digestive function in Drosophila
Base on the functional and structural similarity between Drosophila digestive tract and mammalian GI tract, the Drosophila intestine is widely used to study the mechanism of ISC proliferation and differentiation, barrier function, immune response and metabolic change during aging and stress [24, 30, 47]. Compared with the great progress had made on the midgut, little is known about the function of the Drosophila crop, which is analogous to the mammalian stomach. Some clinical studies have shown that a decreased rate of gastric emptying may be relevant to many symptoms that occur in the elderly [19, 20, 23]. Here, we found that the occurrence of a swollen crop and food accumulation in the crop were also correlated with age in Drosophila. Consistent with the gastrointestinal dysmotility seen in the elderly, crop contraction rate was also decreased in nprl2-mutant flies. These results suggested that aging may affect digestive function in the Drosophila crop, in a similar manner to mammals and this provide a good model for age-related GI dysmotility research. However, it should be noted that the swollen crop may have been due to defects in crop contraction and/or midgut digestion, which affect the passage of food.
Hyperactivated TORC1 promotes midgut aging
TORC1 activity plays important roles in ISC maintenance. Mutation of the TORC1 inhibitor, TSC2 results in defects in ISC division and differentiation, which impairs tissue homeostasis and increases the susceptible of flies to tissue damage [41]. On the contrary, inhibiting TORC1 activity through rapamycin treatment delays midgut aging [51]. Here, we found that the nprl2 mutant displayed an increase in ISC proliferation and a decrease in the number of differentiated EE cells, which was consistent with a previous report on mutations in the TORC1 inhibitor, TSC1 [42]. Furthermore, we found that lipid storage decreased in nprl2-mutant midguts, which can also be seen in aged flies [45]. Recently, it was reported that rapamycin treatment modulates tissue aging and protein aggregates, which suggests that the effects of TORC1 activity on aging may be mediated by autophagy [52]. Notably, autophagy is inhibited in GATOR1 mutants [34], which may be related to the aging and age-related digestive dysfunction seen in nprl2-mutant flies. Recently, a theory, proposed by Mikhail Blagosklonny, suggests that the hyperfunction of TORC1 and other nutrient-sensitive signaling is the cause of aging [53, 54], which was tested in C. elegans, a model system that also develop age-related pathology including digestive dysfunction and tumor formation [55, 56]. Here the age-related digestive dysfunction in nprl2 mutant flies might be contributed by the hyperfunction of TORC1.
In conclusion, hyperactivated TORC1 in nprl2-mutant flies decreased lifespan and promoted the occurrence of age-related crop enlargement, digestive dysfunction, and midgut senescence. Our findings suggested that Nprl2 is essential for the maintenance of GI function during aging, by suppressing TORC1 activity.
Materials and Methods
Fly stocks
The stocks yw, nprl21, and UASp-3×FLAG-3×HA-Nprl2, have been described previously [34]. y, w; TorA948V/CyO was kindly provided by Thomas P. Neufeld [57]. y w; esg-Gal4, UAS-GFP, tub-Gal80ts (TB00137) were obtained from Tsinghua Fly Center (Beijing, China). Flies were cultured at 60% humidity in a temperature-controlled incubator at 25 °C, with a 12 h on/off light cycle, unless otherwise noted. Only male flies were used in this study. The standard food consisted of 1% agar, 3% brewer’s yeast, 1.9% sucrose, 3.8% dextrose, and 9.1% cornmeal (all concentrations in wt/vol). The dyed medium was prepared using standard medium with FD&C Blue Dye No. 1 (SangoTech, Shanghai, China) added at a concentration of 0.5% wt/vol. For rapamycin treatment, rapamycin (LC Laboratories, Woburn, MA, US) was dissolved in methanol and mixed into the media when preparing food vials, to a final concentration at 200 μM. For SDS treatment, SDS was dissolved in 5% sucrose to a final concentration at 0.6%. For ISC experiments using esg-Gal4, UAS-GFP, tub-Gal80ts, fly crosses were performed at 18°C and newly enclosed flies of the appropriate genotypes were kept at 18°C and then transferred to 29°C for 3 days before dissection.
Western blots
Male Drosophila flies were homogenized in RIPA buffer containing complete protease inhibitors and phosphatase inhibitors (Roche, Basel, Switzerland). Western blotting was performed as described previously [13]. Antibodies were used at the following concentrations: Rabbit anti-p4E-BP antibody (Cell Signaling Technology, Danvers, MA, USA) was used at 1:500 and mouse anti-α-tubulin antibody was used at 1:10000 (Jackson Immuno-Research, West Grove, PA, USA). Band intensities were quantified using Image J software (NIH, Bethesda, MD, USA).
Crop size and gut length quantification
Enclosed male flies were collected and cultured on standard medium until the day of analysis. The Drosophila gut was dissected in phosphate-buffered saline (PBS) media, fixed for 20 min in 4% formaldehyde, and then washed twice and sealed with nail oil. Images were captured using a compound microscope. Gut length and crop area were quantified using Image J software.
Food retention assay
Flies were allowed to consume dyed food for 72 hours prior to quantification. After 72 hours, the dye was clearly observed in the digestive tract. The flies with dye in the gut were fed standard food and the ratio of flies with observed dye in the digestive tract was determined at the indicated time points.
Dyed food distribution assay
For immediate food distribution, flies were starved in complete starvation medium (PBS with 1% agarose) for 10 hours and then fed dyed food for 30 min prior to quantification. To determine stable-state food distribution, flies were fed dyed food for 72 hours prior to quantification. The fly gut was dissected, and the crop was carefully separated from the entire gut. Collected tissues were homogenized in 100 μL of PBS. The absorbance values at 630 nm were measured using a spectrophotometer. The dye concentration in crops and guts was calculated by comparison with a standard curve.
Crop motility assays
The crop motility assay was performed as previously described [48]. The male fly was pinned in a dissecting dish under PBS and the ventral cuticle was removed in order to view the crop. The contractions of a crop lobe were recorded from each fly for five repeats with each one is 30 second (with 30 second intervals between each recording period) and then the five counts were averaged.
Capillary Feeder (CAFE) assay
The CAFE assay was modified from previously described [38]. Briefly, 10 male flies were put in the inner chamber to get the liquid food (0.5% Blue dye in 5% sucrose media) for 24 hours and the amount of food loss was calculated. Each experiment included an identical CAFE chamber without flies to determine evaporative losses, which were subtracted from experimental readings.
Defecation rate
The defecation rate detection was modified from previously described [37]. Flies were allowed to consume dyed food for 3 days prior to quantification. The male flies were then transferred to empty cleared vials (10 flies per vial) for 1 hour and the number of blue fecal spots on the vial counted. The rate of defecation was calculated as the number of spots per fly per hour.
Nile red assay
Drosophila guts were dissected in PBS, fixed in 4% formaldehyde for 20 min, and then washed three times with PBS. The guts were incubated with Nile red for 30 min, washed twice, and mounted with ProLong ® Gold Antifade Reagent containing DAPI (Invitrogen, Carlsbad, CA, USA).
EdU labeling assay
The EdU labeling assay was modified from previously described with a Click-iT EdU Alexa Fluor 594 Imaging Kit (Thermo Fisher Scientific, C10339) [58]. Male flies were fed with 100 μM EdU and 5% sucrose for 24 hr. The EdU incorporation in gut was visualized by Click reaction according to the manufacturer’s instruction.
Immunofluorescence confocal microscopy
Immunofluorescence staining was performed as previously described [50]. The mouse anti-Prospero antibody (Developmental Studies Hybridoma Bank, University of Iowa) was used at 1:100. The mouse anti-discs large antibody (Developmental Studies Hybridoma Bank, University of Iowa) was used at 1:100. Alexa Fluor-conjugated anti-mouse secondary antibodies (Invitrogen) were used at a dilution of 1:1,000. Nuclei were visualized by staining with DAPI (Invitrogen). Images were acquired using a Zeiss 880 confocal microscope (Zeiss, Oberkochen, Germany) and fluorescence was quantified using Image J software.
Supplementary Materials
Acknowledgments
We thank members of the Animal Physiology Group in College of Bioscience and Biotechnology, Yangzhou University for comments on the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
Funding
This work was supported by National Natural Science Foundation of China (No. 31872287); Natural Science Foundation of Jiangsu Province (NO. BK20181456); Postdoctoral Science Foundation of China (No. 2019M650124); Natural Science Foundation of the Jiangsu Higher Education Institutions of China (No. 19KJB180008 and 18KJD180009); Six talent peaks project in Jiangsu Province (No. SWYY-146); Key Laboratory of Pollinating Insect Biology in China Ministry of Agriculture (No. 2017MFNZS07); ”Lvyang Jinfeng” Talents Attracting Plan (to Yuanyuan Fu); and Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) Intramural Research program (No. HD00163 16, to Mary A. Lilly).
References
- 1. Kennedy BK, Pennypacker JK. Drugs that modulate aging: the promising yet difficult path ahead. Transl Res. 2014; 163:456–65. https://doi.org/10.1016/j.trsl.2013.11.007 [PubMed]
- 2. Blagosklonny MV. Disease or not, aging is easily treatable. Aging (Albany NY). 2018; 10:3067–78. https://doi.org/10.18632/aging.101647 [PubMed]
- 3. Walters HE, Cox LS. mTORC Inhibitors as Broad-Spectrum Therapeutics for Age-Related Diseases. Int J Mol Sci. 2018; 19:19. https://doi.org/10.3390/ijms19082325 [PubMed]
- 4. Johnson SC, Rabinovitch PS, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature. 2013; 493:338–45. https://doi.org/10.1038/nature11861 [PubMed]
- 5. Saxton RA, Sabatini DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell. 2017; 169:361–71. https://doi.org/10.1016/j.cell.2017.03.035 [PubMed]
- 6. Kim E, Goraksha-Hicks P, Li L, Neufeld TP, Guan KL. Regulation of TORC1 by Rag GTPases in nutrient response. Nat Cell Biol. 2008; 10:935–45. https://doi.org/10.1038/ncb1753 [PubMed]
- 7. Sancak Y, Peterson TR, Shaul YD, Lindquist RA, Thoreen CC, Bar-Peled L, Sabatini DM. The Rag GTPases bind raptor and mediate amino acid signaling to mTORC1. Science. 2008; 320:1496–501. https://doi.org/10.1126/science.1157535 [PubMed]
- 8. Kim J, Guan KL. mTOR as a central hub of nutrient signalling and cell growth. Nat Cell Biol. 2019; 21:63–71. https://doi.org/10.1038/s41556-018-0205-1 [PubMed]
- 9. Lawrence RE, Zoncu R. The lysosome as a cellular centre for signalling, metabolism and quality control. Nat Cell Biol. 2019; 21:133–42. https://doi.org/10.1038/s41556-018-0244-7 [PubMed]
- 10. Jewell JL, Russell RC, Guan KL. Amino acid signalling upstream of mTOR. Nat Rev Mol Cell Biol. 2013; 14:133–39. https://doi.org/10.1038/nrm3522 [PubMed]
- 11. Inoki K, Li Y, Xu T, Guan KL. Rheb GTPase is a direct target of TSC2 GAP activity and regulates mTOR signaling. Genes Dev. 2003; 17:1829–34. https://doi.org/10.1101/gad.1110003 [PubMed]
- 12. Dibble CC, Elis W, Menon S, Qin W, Klekota J, Asara JM, Finan PM, Kwiatkowski DJ, Murphy LO, Manning BD. TBC1D7 is a third subunit of the TSC1-TSC2 complex upstream of mTORC1. Mol Cell. 2012; 47:535–46. https://doi.org/10.1016/j.molcel.2012.06.009 [PubMed]
- 13. Wei Y, Lilly MA. The TORC1 inhibitors Nprl2 and Nprl3 mediate an adaptive response to amino-acid starvation in Drosophila. Cell Death Differ. 2014; 21:1460–68. https://doi.org/10.1038/cdd.2014.63 [PubMed]
- 14. Bar-Peled L, Chantranupong L, Cherniack AD, Chen WW, Ottina KA, Grabiner BC, Spear ED, Carter SL, Meyerson M, Sabatini DM. A Tumor suppressor complex with GAP activity for the Rag GTPases that signal amino acid sufficiency to mTORC1. Science. 2013; 340:1100–06. https://doi.org/10.1126/science.1232044 [PubMed]
- 15. Kaeberlein M, Powers RW
3rd , Steffen KK, Westman EA, Hu D, Dang N, Kerr EO, Kirkland KT, Fields S, Kennedy BK. Regulation of yeast replicative life span by TOR and Sch9 in response to nutrients. Science. 2005; 310:1193–96. https://doi.org/10.1126/science.1115535 [PubMed] - 16. Kapahi P, Zid BM, Harper T, Koslover D, Sapin V, Benzer S. Regulation of lifespan in Drosophila by modulation of genes in the TOR signaling pathway. Curr Biol. 2004; 14:885–90. https://doi.org/10.1016/j.cub.2004.03.059 [PubMed]
- 17. Harrison DE, Strong R, Sharp ZD, Nelson JF, Astle CM, Flurkey K, Nadon NL, Wilkinson JE, Frenkel K, Carter CS, Pahor M, Javors MA, Fernandez E, Miller RA. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature. 2009; 460:392–95. https://doi.org/10.1038/nature08221 [PubMed]
- 18. Rera M, Clark RI, Walker DW. Intestinal barrier dysfunction links metabolic and inflammatory markers of aging to death in Drosophila. Proc Natl Acad Sci USA. 2012; 109:21528–33. https://doi.org/10.1073/pnas.1215849110 [PubMed]
- 19. Gidwaney NG, Bajpai M, Chokhavatia SS. Gastrointestinal Dysmotility in the Elderly. J Clin Gastroenterol. 2016; 50:819–27. https://doi.org/10.1097/MCG.0000000000000650 [PubMed]
- 20. Soenen S, Rayner CK, Horowitz M, Jones KL. Gastric Emptying in the Elderly. Clin Geriatr Med. 2015; 31:339–53. https://doi.org/10.1016/j.cger.2015.04.003 [PubMed]
- 21. Merchant HA, Liu F, Orlu Gul M, Basit AW. Age-mediated changes in the gastrointestinal tract. Int J Pharm. 2016; 512:382–95. https://doi.org/10.1016/j.ijpharm.2016.04.024 [PubMed]
- 22. Bhutto A, Morley JE. The clinical significance of gastrointestinal changes with aging. Curr Opin Clin Nutr Metab Care. 2008; 11:651–60. https://doi.org/10.1097/MCO.0b013e32830b5d37 [PubMed]
- 23. Apidianakis Y, Rahme LG. Drosophila melanogaster as a model for human intestinal infection and pathology. Dis Model Mech. 2011; 4:21–30. https://doi.org/10.1242/dmm.003970 [PubMed]
- 24. Zwick RK, Ohlstein B, Klein OD. Intestinal renewal across the animal kingdom: comparing stem cell activity in mouse and Drosophila. Am J Physiol Gastrointest Liver Physiol. 2019; 316:G313–22. https://doi.org/10.1152/ajpgi.00353.2018 [PubMed]
- 25. Stoffolano JG
Jr , Haselton AT. The adult Dipteran crop: a unique and overlooked organ. Annu Rev Entomol. 2013; 58:205–25. https://doi.org/10.1146/annurev-ento-120811-153653 [PubMed] - 26. Lemaitre B, Miguel-Aliaga I. The digestive tract of Drosophila melanogaster. Annu Rev Genet. 2013; 47:377–404. https://doi.org/10.1146/annurev-genet-111212-133343 [PubMed]
- 27. Ohlstein B, Spradling A. The adult Drosophila posterior midgut is maintained by pluripotent stem cells. Nature. 2006; 439:470–74. https://doi.org/10.1038/nature04333 [PubMed]
- 28. Micchelli CA, Perrimon N. Evidence that stem cells reside in the adult Drosophila midgut epithelium. Nature. 2006; 439:475–79. https://doi.org/10.1038/nature04371 [PubMed]
- 29. Guo Z, Lucchetta E, Rafel N, Ohlstein B. Maintenance of the adult Drosophila intestine: all roads lead to homeostasis. Curr Opin Genet Dev. 2016; 40:81–86. https://doi.org/10.1016/j.gde.2016.06.009 [PubMed]
- 30. Jasper H. Exploring the physiology and pathology of aging in the intestine of Drosophila melanogaster. Invertebr Reprod Dev. 2015; 59:51–58. https://doi.org/10.1080/07924259.2014.963713 [PubMed]
- 31. Tain LS, Mortiboys H, Tao RN, Ziviani E, Bandmann O, Whitworth AJ. Rapamycin activation of 4E-BP prevents parkinsonian dopaminergic neuron loss. Nat Neurosci. 2009; 12:1129–35. https://doi.org/10.1038/nn.2372 [PubMed]
- 32. Bjedov I, Toivonen JM, Kerr F, Slack C, Jacobson J, Foley A, Partridge L. Mechanisms of life span extension by rapamycin in the fruit fly Drosophila melanogaster. Cell Metab. 2010; 11:35–46. https://doi.org/10.1016/j.cmet.2009.11.010 [PubMed]
- 33. Bitto A, Ito TK, Pineda VV, LeTexier NJ, Huang HZ, Sutlief E, Tung H, Vizzini N, Chen B, Smith K, Meza D, Yajima M, Beyer RP, et al. Transient rapamycin treatment can increase lifespan and healthspan in middle-aged mice. eLife. 2016; 5:5. https://doi.org/10.7554/eLife.16351 [PubMed]
- 34. Wei Y, Reveal B, Cai W, Lilly MA. The GATOR1 Complex Regulates Metabolic Homeostasis and the Response to Nutrient Stress in Drosophila melanogaster. G3 (Bethesda). 2016; 6:3859–67. https://doi.org/10.1534/g3.116.035337 [PubMed]
- 35. Mitchell EL, Davis AT, Brass K, Dendinger M, Barner R, Gharaibeh R, Fodor AA, Kavanagh K. Reduced Intestinal Motility, Mucosal Barrier Function, and Inflammation in Aged Monkeys. J Nutr Health Aging. 2017; 21:354–61. https://doi.org/10.1007/s12603-016-0725-y [PubMed]
- 36. Ren J, Zhu H, Chi C, Mehrmohamadi M, Deng K, Wu X, Xu T. Beadex affects gastric emptying in Drosophila. Cell Res. 2014; 24:636–39. https://doi.org/10.1038/cr.2014.24 [PubMed]
- 37. Edgecomb RS, Harth CE, Schneiderman AM. Regulation of feeding behavior in adult Drosophila melanogaster varies with feeding regime and nutritional state. J Exp Biol. 1994; 197:215–35. [PubMed]
- 38. Ja WW, Carvalho GB, Mak EM, de la Rosa NN, Fang AY, Liong JC, Brummel T, Benzer S. Prandiology of Drosophila and the CAFE assay. Proc Natl Acad Sci USA. 2007; 104:8253–56. https://doi.org/10.1073/pnas.0702726104 [PubMed]
- 39. Buchon N, Broderick NA, Kuraishi T, Lemaitre B. Drosophila EGFR pathway coordinates stem cell proliferation and gut remodeling following infection. BMC Biol. 2010; 8:152. https://doi.org/10.1186/1741-7007-8-152 [PubMed]
- 40. Buchon N, Osman D, David FP, Fang HY, Boquete JP, Deplancke B, Lemaitre B. Morphological and molecular characterization of adult midgut compartmentalization in Drosophila. Cell Rep. 2013; 3:1725–38. https://doi.org/10.1016/j.celrep.2013.04.001 [PubMed]
- 41. Amcheslavsky A, Ito N, Jiang J, Ip YT. Tuberous sclerosis complex and Myc coordinate the growth and division of Drosophila intestinal stem cells. J Cell Biol. 2011; 193:695–710. https://doi.org/10.1083/jcb.201103018 [PubMed]
- 42. Quan Z, Sun P, Lin G, Xi R. TSC1/2 regulates intestinal stem cell maintenance and lineage differentiation through Rheb-TORC1-S6K but independently of nutritional status or Notch regulation. J Cell Sci. 2013; 126:3884–92. https://doi.org/10.1242/jcs.125294 [PubMed]
- 43. Choi NH, Kim JG, Yang DJ, Kim YS, Yoo MA. Age-related changes in Drosophila midgut are associated with PVF2, a PDGF/VEGF-like growth factor. Aging Cell. 2008; 7:318–34. https://doi.org/10.1111/j.1474-9726.2008.00380.x [PubMed]
- 44. Biteau B, Hochmuth CE, Jasper H. JNK activity in somatic stem cells causes loss of tissue homeostasis in the aging Drosophila gut. Cell Stem Cell. 2008; 3:442–55. https://doi.org/10.1016/j.stem.2008.07.024 [PubMed]
- 45. Karpac J, Biteau B, Jasper H. Misregulation of an adaptive metabolic response contributes to the age-related disruption of lipid homeostasis in Drosophila. Cell Rep. 2013; 4:1250–61. https://doi.org/10.1016/j.celrep.2013.08.004 [PubMed]
- 46. Zhou Y, Liu Z, Chen Y, Jin LH. Identification of the protective effects of traditional medicinal plants against SDS-induced Drosophila gut damage. Exp Ther Med. 2016; 12:2671–80. https://doi.org/10.3892/etm.2016.3641 [PubMed]
- 47. Miguel-Aliaga I, Jasper H, Lemaitre B. Anatomy and Physiology of the Digestive Tract of Drosophila melanogaster.. Genetics. 2018; 210:357–96. https://doi.org/10.1534/genetics.118.300224 [PubMed]
- 48. Peller CR, Bacon EM, Bucheger JA, Blumenthal EM. Defective gut function in drop-dead mutant Drosophila. J Insect Physiol. 2009; 55:834–39. https://doi.org/10.1016/j.jinsphys.2009.05.011 [PubMed]
- 49. Chtarbanova S, Lamiable O, Lee KZ, Galiana D, Troxler L, Meignin C, Hetru C, Hoffmann JA, Daeffler L, Imler JL. Drosophila C virus systemic infection leads to intestinal obstruction. J Virol. 2014; 88:14057–69. https://doi.org/10.1128/JVI.02320-14 [PubMed]
- 50. Wei Y, Reveal B, Reich J, Laursen WJ, Senger S, Akbar T, Iida-Jones T, Cai W, Jarnik M, Lilly MA. TORC1 regulators Iml1/GATOR1 and GATOR2 control meiotic entry and oocyte development in Drosophila. Proc Natl Acad Sci USA. 2014; 111:E5670–77. https://doi.org/10.1073/pnas.1419156112 [PubMed]
- 51. Fan X, Liang Q, Lian T, Wu Q, Gaur U, Li D, Yang D, Mao X, Jin Z, Li Y, Yang M. Rapamycin preserves gut homeostasis during Drosophila aging. Oncotarget. 2015; 6:35274–83. https://doi.org/10.18632/oncotarget.5895 [PubMed]
- 52. Schinaman JM, Rana A, Ja WW, Clark RI, Walker DW. Rapamycin modulates tissue aging and lifespan independently of the gut microbiota in Drosophila. Sci Rep. 2019; 9:7824. https://doi.org/10.1038/s41598-019-44106-5 [PubMed]
- 53. Blagosklonny MV, Hall MN. Growth and aging: a common molecular mechanism. Aging (Albany NY). 2009; 1:357–62. https://doi.org/10.18632/aging.100040 [PubMed]
- 54. Blagosklonny MV. Aging and immortality: quasi-programmed senescence and its pharmacologic inhibition. Cell Cycle. 2006; 5:2087–102. https://doi.org/10.4161/cc.5.18.3288 [PubMed]
- 55. Gems D, de la Guardia Y. Alternative Perspectives on Aging in Caenorhabditis elegans: Reactive Oxygen Species or Hyperfunction? Antioxid Redox Signal. 2013; 19:321–29. https://doi.org/10.1089/ars.2012.4840 [PubMed]
- 56. Wang H, Zhao Y, Ezcurra M, Benedetto A, Gilliat AF, Hellberg J, Ren Z, Galimov ER, Athigapanich T, Girstmair J, Telford MJ, Dolphin CT, Zhang Z, Gems D. A parthenogenetic quasi-program causes teratoma-like tumors during aging in wild-type C. elegans.. NPJ Aging Mech Dis. 2018; 4:6. https://doi.org/10.1038/s41514-018-0025-3 [PubMed]
- 57. Zhang Y, Billington CJ
Jr , Pan D, Neufeld TP. Drosophila target of rapamycin kinase functions as a multimer. Genetics. 2006; 172:355–62. https://doi.org/10.1534/genetics.105.051979 [PubMed] - 58. Obata F, Tsuda-Sakurai K, Yamazaki T, Nishio R, Nishimura K, Kimura M, Funakoshi M, Miura M. Nutritional Control of Stem Cell Division through S-Adenosylmethionine in Drosophila Intestine. Dev Cell. 2018; 44:741–751.e3. https://doi.org/10.1016/j.devcel.2018.02.017 [PubMed]