Introduction
Rpd3, a Drosophila histone deacetylase (HDAC), modulates chromatin structures and affects signaling pathways by interacting with several chromatin remodeling complexes [1-3]. The Rpd3 protein is known to mediate epigenetic effects like long-term memory and lifespan. The adult brain-specific changes of Rpd3 expression results in impaired long-term courtship memory [4]. However, systemic downregulation of Rpd3 extends lifespan [5,6], which is also observed in yeast with reduced Rpd3 expression [7]. Recently, it has been reported that Rpd3 interacts partially with the insulin signaling longevity pathway [8]. As several long-lived mutant flies display increased resistance to numerous stressors including oxidation, starvation, and heat compared to wild-type flies [9-12], Rpd3 downregulation also enhances stress resistance with extended lifespan [6]. Inhibition of mammalian HDACs is shown to be a proven anticancer therapeutic and potential treatment of many other diseases including HIV infection, Alzheimer's disease, and cardiac remodeling [13,14]. Consistently, it was recently reported that decreased Rpd3 expression in Drosophila heart tissue enhances cardiac function (decreased heart failure and accelerated heart recovery) and resistance against stressors [6].
Loco, a fly regulator of G-protein signaling (RGS) protein, has been identified as an activator or repressor with GTPase-activating protein (GAP) activity in the G-protein signaling pathway [15,16]. The Loco protein functions in neuroblast (NB) asymmetric division [17] and blood-brain barrier formation [18], which are mediated by interactions with several GPCRs and the inhibitory G proteins (Gαi and Gαo). Beyond these specific cellular functions, it is reported that Loco globally regulates stress resistance and lifespan [19]. Reduced Loco expression results in longer lifespan for flies with stronger resistance to the stressors, higher manganese-containing superoxide dismutase (MnSOD) activity, and higher fat content [19]. In contrast, upregulation of the loco gene shortens lifespan significantly with lower stress resistance and reduced fat content [19], also showing that its RGS domain containing GAP activity is related to the regulation of longevity [19]. Notably, the expressional changes of yeast RGS2 and rat RGS14, homologues of the fly Loco, also affect oxidative stress resistance and lifespan in the respective species [19], indicating that the Loco/RGS14 signaling pathway is evolutionarily conserved in various organisms for the regulation of longevity.
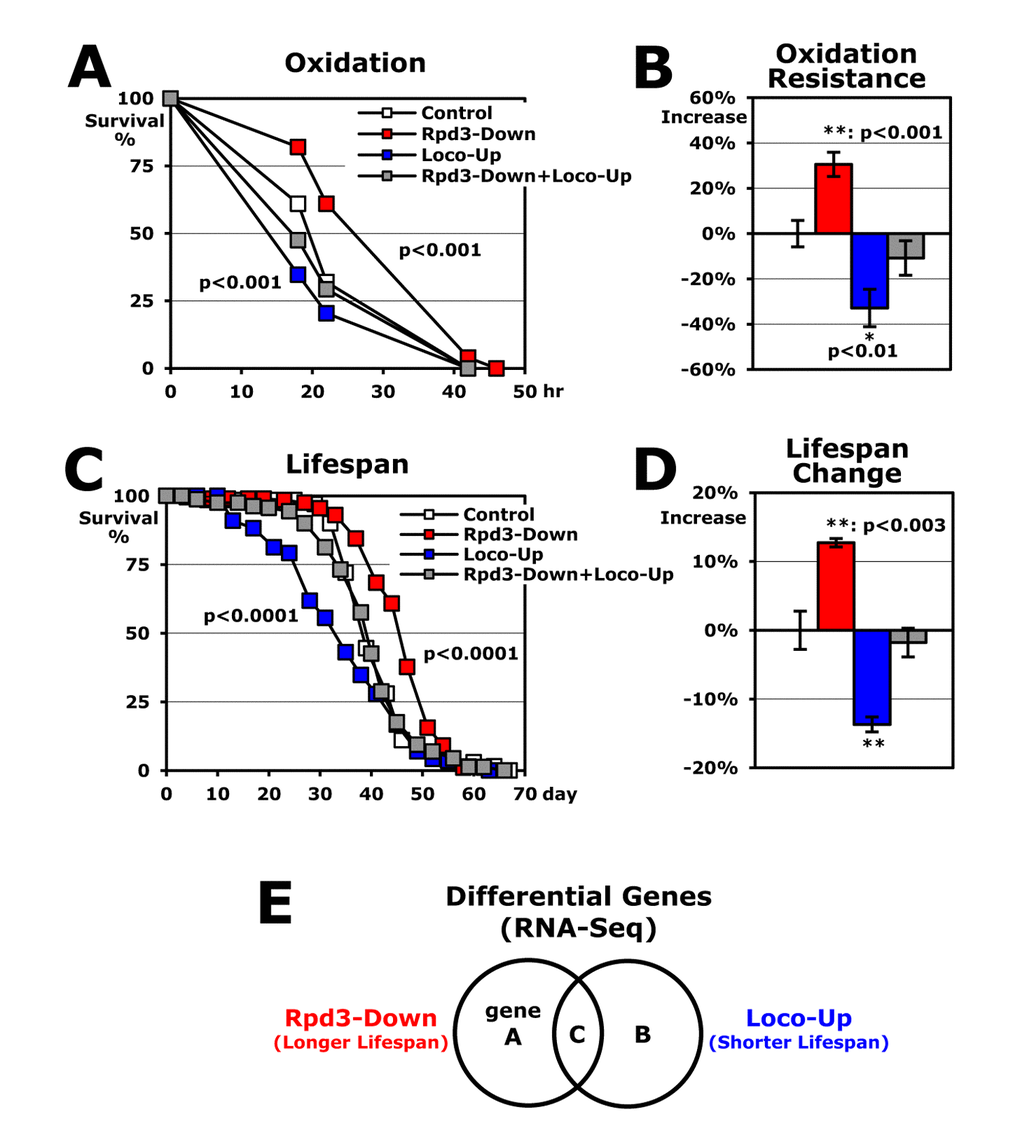
To investigate whether the nuclear protein Rpd3 interacts with the cytoplasmic membrane-associated protein Loco for the longevity regulation, here, we measured stress resistance and lifespan with a combination of the rpd3 downregulation to extend lifespan [5,6] and the loco upregulation to shorten lifespan [19]. Then, we examined gene expression profiles between those flies in order to find the common target genes modulated by Rpd3 and Loco for the longevity.
Results
Longer lifespan enhanced by changes in Rpd3 and Loco exhibits the reduced catabolism
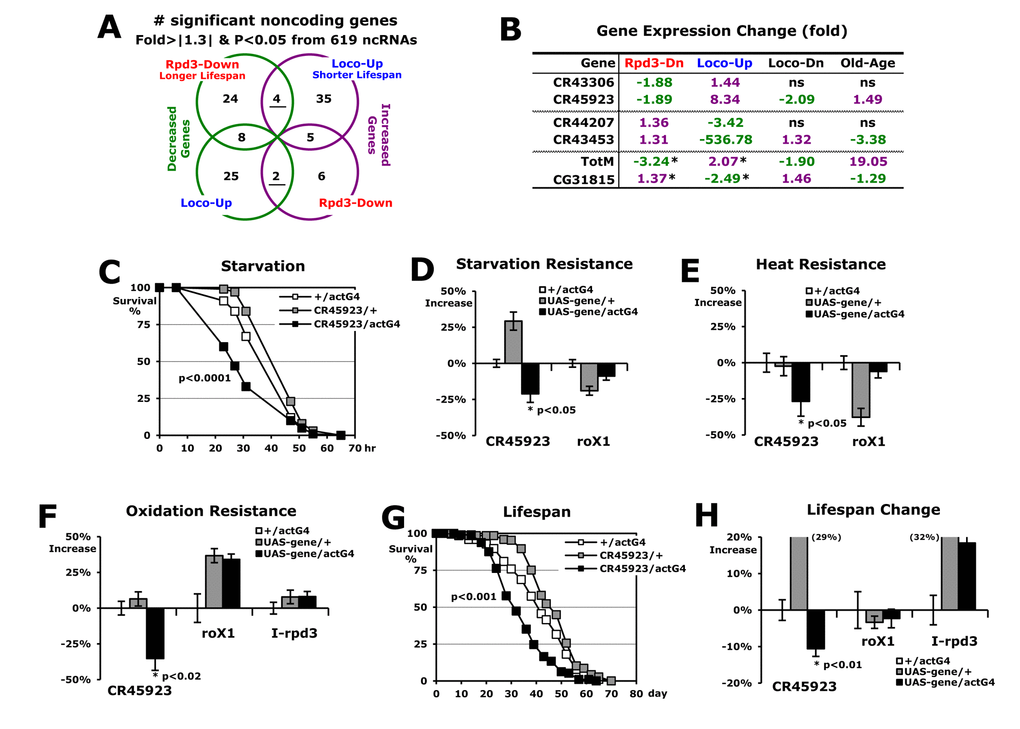
With a total of 49,162,435 reads of RNA-seq experiments, RNA expression levels of the 13,244 genes were compared between 2-day-old male flies of experimental and control groups (Rpd3-Down/ or Loco-Up/common control). First, we statistically filtered out the genes that are non-specifically changed in the expression with a p-value < 0.05 (Fisher’s exact test). Then, with a cut-off value of ± 1.3-fold changes, we selected the candidates as significantly changed genes (Figure 2A). In the rpd3-downregulated flies (Rpd3-Down), 647 genes showed decreased expressions and 633 gene expressions were increased with the loco upregulation (Loco-Up) (Figure 2A). From these two gene groups, only 29 genes were overlapped reflecting the opposite expression between the longer lifespan (Rpd3-Down) and shorter lifespan (Loco-Up) (Figure 2A). This indicates that the 29 expressions decrease in the long-lived flies with Rpd3 downregulation and conversely increase in the short lifespan of Loco-upregulated flies. Reverse analysis (Figure 2A) revealed that 10 genes show an expression pattern of an increase in the longer lifespan and a decrease in the shorter lifespan. Collectively, these 39 total genes (~ 0.3% from 13,244 genes initially screened) were considered as candidate target genes (Figure 2A) which both Loco and Rpd3 modulate for stress resistance and lifespan (Figure 1).

Figure 2. Longer lifespan induced by the change of Rpd3 and Loco results from the reduced catabolism. (A) The gene expression analyses (Rpd3-Down or Loco-Up/control). From RNA-seq experiments using 2-day-old male flies, the genes changed more than 1.3-fold were selected with p-value <0.05 (Fisher’s exact test). The underlined genes represent that their expressions decrease (green) and increase (purple) in a different group, respectively. (B) With the genes oppositely changed in the Rpd3-Down and Loco-Up (*: total 39 in A), the gene ontology was analyzed using a DAVID web tool (http://david.abcc.ncifcrf.gov/home.jsp). %: involved genes/total 39 genes; p-value: a modified Fisher Exact. (C) Expressional changes of selected genes in RNA-seq and real-time PCR analyses. The fold changes were averaged from 4 ~ 13 independent experiments using four different RNA batches. Bold change: p-value < 0.05 ~ 0.0001. (D) Functional analyses (http://flybase.org) of genes which were selected with more than 1.5-fold changes and p-value <0.05 in both Rpd3-Down (longer lifespan) and Loco-Up (shorter lifespan) groups. *: genes tested as a representative of each functional subgroup with RT-PCR analysis in (C).
When the gene ontology of these 39 genes was analyzed with a DAVID web tool (Figure 2B), 69% of them were shown to be related to the signaling pathways. Interestingly, many of them are metabolic enzymes, suggesting that Loco and Rpd3 change the metabolism for the longevity. Through RT-PCR analysis (Figure 2C), we confirmed expression changes of candidate genes obtained from the RNA-seq experiments (Figure 2A). As representatives of each functional subgroup in Figure 2D, they showed opposite expressions in the longer lifespan (Rpd3-Down) and shorter lifespan (Loco-Up), respectively (Figure 2C). However, the Rpd3 and Loco did not affect expressions of each other. Among the 39 candidate genes (Figure 2A), we next examined more significantly changed genes with more than 1.5-fold changes [21-23] in both Rpd3-Down and Loco-Up groups (Figure 2D). Interestingly, the activities of catabolic enzymes such as mannosidase, lipase, endopeptidase, and phosphodiesterase [24] were reduced in the long-lived flies with Rpd3 downregulation (Figure 2D). Uptake and storage proteins (ninaD and Lsp2) of diets to be catabolized [25,26] were also decreased in the flies with longer lifespan (Figure 2D). Inversely, those catabolic activities that break down carbohydrates, lipids, and peptides were high in the short lifespan of Loco-upregulated flies (Figure 2D). Other notable changes were an apparent decrease in the detoxifying molecule and antibacterial peptides (AMP) in the longer lifespan of Rpd3 downregulation (Figure 2D). As defending mechanisms, these proteins are essential to protect flies against toxins and bacterial infections [27-29], and the AMP expressions increase during the aging process [22], which may prepare pathogen defenses promptly in the old-aged flies that have weak immune responses. However, it has been reported that lesser expressions of AMP genes are found in longer-lived flies [22] and the AMP downregulation enhances stress resistance, lifespan, and fat content in adult flies [30]. Lifespan extension through hormesis (the beneficial effects of low-level toxins and stressors) also occurs with reduced immune function [31]. These data imply that less activation of AMP synthesis has a beneficial effect for the longevity process under the normal condition without bacterial infection. Consistent with this hypothesis, higher expressions of AMP genes were detected in the short lifespan of Loco-upregulated flies (Figure 2D).
On the contrary, expressions of Octα2R, nimC1, and CG9825 genes were increased in the long-lived flies under Rpd3 downregulation (Figure 2D). Octα2R (α2-adrenergic-like octopamine receptor) is activated by octopamine and epinephrine, resulting in the inhibition of cAMP production in a dose-dependent manner [32]. The cAMP reduction has been observed in several long-lived animals such as AC5 (type 5 adenylyl cyclase) KO mice and Loco (Regulator of G-protein signaling protein) mutant flies [19,33]. It was reported that reduction of nimC1 (EGF-like receptor) makes flies live for a shorter time [34] and the downregulation of the CG9825 gene (organic anion transporter) is defective in the pain (heat) response [35]. Consistently, the decrease of nimC1 and CG9825 expressions were shown in the short-lived flies with Loco upregulation (Figure 2D).
Discussion
Rpd3 or Loco downregulation extends lifespan with higher stress resistance [5,6,19]. Here, we found that the nuclear protein Rpd3 genetically interacts with the cytoplasmic membrane-associated protein Loco in the longevity mechanism (Figure 1A-D). Given that the changes in Rpd3 and Loco did not affect the expression of each other (Figure 2C), they may interact post-translationally through a signaling pathway. As a regulator of G-protein signaling protein, Loco increases adenylate cyclase (AC) activity by inactivating the inhibitory Gαi•GTP protein [17,18,33], and the mammalian homologue, RGS14, interacts with activated H-Ras and Raf-1 kinases, which subsequently inhibit ERK phosphorylation [37-39]. Consistently, we previously showed that downregulation of Loco significantly diminishes cAMP amounts [19] and increases p-ERK levels with higher resistance to the oxidative stress [12]. In mammals, p-ERK is shown to interact with PP1 (protein phosphatase 1) that dephosphorylates p-HDAC1 [40-42]. Considering mammalian HDAC1 phosphorylation promotes complex formation and enhances the enzyme activity of HDAC1 [42-44], one possibility is that Loco downregulation may decrease Rpd3 activity through the reduction of phospho-Rpd3 levels, subsequently resulting in the longer lifespan (Figure 1).
A second possibility of interaction between Rpd3 and Loco is that they regulate the metabolic pathways that converge to impact longevity (Figure 2). When we examined the target genes showing opposite expression between the rpd3-downregulated long-lived flies and the loco-upregulated short-lived flies (Figure 2A-C), we found that the expressions of catabolism process genes were significantly reduced in the long-lived flies (Figure 2D). Given that these gene expressions were analyzed in young flies (2 days of age in Figure 2A), the reduced catabolism is considered to be necessary from a young age for the longer lifespan of the Rpd3- and Loco-downregulated old flies (Figure 2D). This reduced catabolism is related to the dietary restriction (DR) that extends lifespan in several species [39]. Also, it was recently reported that Rpd3 interacts partially with the insulin signaling pathway of the DR longevity mechanism [8,45].
The noncoding RNA, dntRL (CR45923), was found to be a putative target gene that is modulated by Rpd3 and Loco for the longevity (Figure 3B). Surprisingly, this RNA itself could affect stress resistance and lifespan (Figure 3C-H). The fly database (http://flybase.org) indicates that the dntRL gene is expressed in fat body tissue, which functions in metabolic homeostasis, stress tolerance, growth, and longevity in Drosophila [46,47]. The antimicrobial peptides (AMPs) genes are also dominantly expressed in the fat body [27] as other target genes modulated by Rpd3 and Loco for the longevity (Figure 3B). Together with a report that the dntRL gene was screened during a mitochondrial disease-related study [48], it is considered that the dntRL gene may be involved in the metabolic mechanism as a long noncoding RNA (1,364 nt).
Materials and Methods
Fly genotypes and constructs
The y1 w1 (lab stock from Bloomington) flies were used as a wild-type background. The rpd3- (P{PZ}HDAC104556: Bloomington), UAS-loco (P{XP}locod06164: Exelixis at Harvard Medical School), and act-Gal4 (Bloomington) flies were obtained from the Drosophila stock center. The loco- (locoP283: W. Chia [17],) and UAS-roX1 (RL. Kelley [49],) were kindly provided. These flies were six times isogenized with y1 w1 before the stress resistance and aging tests. Virgin flies were collected from a bottle in which larval density was controlled in a standard cornmeal medium, and were used for all fly experiments including stress response, aging, and gene expression studies [19]. To construct UAS-CR45923 and UAS-I-rpd3 transgenes (Figure 3), the CR45923 (1,364 bp) and rpd3 (1,563 bp) cDNAs were cloned into the pCRII-TOPO vector (Invitrogen). After the sequences were confirmed, the plasmids were subcloned into an EcoRI/XhoI or XhoI/XbaI digested pUASTattB vector.
Stress response and aging assays
To measure stress responses, 100 newly enclosed flies (20 flies per vial) were kept in a standard cornmeal medium at 25°C for 2 days [9,19,50]. For the heat test, these 2-day-old adult flies were transferred to new vials containing standard cornmeal medium and maintained at 37°C with 30% humidity. For the starvation test, the 2-day-old adult flies were transferred to new vials (2.5 × 9.3 cm) containing two filter circles (2.4-cm diameter, Fisher Scientific) soaked in 300 μl of distilled water, and were maintained at 25°C under moist conditions with 100 μl of water added every 12 hrs. For the oxidative stress test, the 2-day-old adult flies were starved for 6 hrs at 25°C as described above. Then, the flies were transferred to new vials containing two filter circles wetted with 300 μl of 20 mM methyl viologen hydrate (Paraquat, Fisher Scientific) in a 5% sucrose solution and maintained at 25°C. The median survival times of flies under each stress (heat, starvation, or oxidation) were calculated from the survival curves of 3 ~ 6 independent experiments. For the aging test, 200 virgin flies (20 flies per vial) were counted and transferred to fresh standard cornmeal vials every 3 - 4 days [9]. Mean lifespan was calculated from the lifespan curves and averaged with standard error of mean (SEM) from several repeat experiments.
RNA-seq and RT-PCR analyses
Total RNAs were extracted with the TRIzol Reagent (Invitrogen) from the whole body of male adult flies and then ribosomal RNAs were depleted from the total RNAs using the RiboMinus Eukaryote Kit (Invitrogen). With these coding and noncoding RNAs, the Illumina compatible strand-specific RNA-Seq protocol was used to make the cDNA library, which was then sequenced by Illumina NextSeq500 sequencer in the Genomics facility (http://research.njms.rutgers.edu/genomics/). The significance of the expression changes was evaluated by the Fisher’s exact test (p-value). For the RT-PCR experiment to check transcriptional expression of the genes, 5 μg of the total RNA purified from adult flies were treated with DNase I (RNase-free, Roche) and used to produce oligo dT-primed cDNAs (SuperScript II RT, Invitrogen). Then, the cDNAs were used as templates for quantitative real-time PCR that was performed with power SYBR green PCR mix (Applied Biosystems) [51]. The rp49 gene was used as an internal reference for normalizing the quality of total RNA purified from each fly. Expressional fold of the various genes was determined by the comparative CT method (ABI Prism 7700 Sequence Detection System User Bulletin #2, Applied Biosystems).
Acknowledgements
We thank Catherine Uhle for the CR45923 work, Hardik Parikh for research assistance, Dr. Jiyeon Park for RNA-seq analysis, Christopher Brady for critical reading, and Dr. Hyangyee Oh for research support.
Conflicts of Interest
The authors have no conflict of interests to declare.
Funding
This work was supported by the New Jersey Health Foundation (NJHF) and National Institute on Aging (NIA) grant AG042021.
References
- 1. Burgio G, Cipressa F, Ingrassia AM, Cenci G, Corona DF. The histone deacetylase Rpd3 regulates the heterochromatin structure of Drosophila telomeres. J Cell Sci. 2011; 124:2041–48. https://doi.org/10.1242/jcs.078261 [PubMed]
- 2. Zhang Z, Feng J, Pan C, Lv X, Wu W, Zhou Z, Liu F, Zhang L, Zhao Y. Atrophin-Rpd3 complex represses Hedgehog signaling by acting as a corepressor of CiR. J Cell Biol. 2013; 203:575–83. https://doi.org/10.1083/jcb.201306012 [PubMed]
- 3. Meier K, Brehm A. Chromatin regulation: how complex does it get? Epigenetics. 2014; 9:1485–95. https://doi.org/10.4161/15592294.2014.971580 [PubMed]
- 4. Fitzsimons HL, Scott MJ. Genetic modulation of Rpd3 expression impairs long-term courtship memory in Drosophila. PLoS One. 2011; 6:e29171. https://doi.org/10.1371/journal.pone.0029171 [PubMed]
- 5. Rogina B, Helfand SL, Frankel S. Longevity regulation by Drosophila Rpd3 deacetylase and caloric restriction. Science. 2002; 298:1745. https://doi.org/10.1126/science.1078986 [PubMed]
- 6. Kopp ZA, Hsieh JL, Li A, Wang W, Bhatt DT, Lee A, Kim SY, Fan D, Shah V, Siddiqui E, Ragam R, Park K, Ardeshna D, et al. Heart-specific Rpd3 downregulation enhances cardiac function and longevity. Aging (Albany NY). 2015; 7:648–63. https://doi.org/10.18632/aging.100806 [PubMed]
- 7. Kim S, Benguria A, Lai CY, Jazwinski SM. Modulation of life-span by histone deacetylase genes in Saccharomyces cerevisiae. Mol Biol Cell. 1999; 10:3125–36. https://doi.org/10.1091/mbc.10.10.3125 [PubMed]
- 8. Woods JK, Ziafazeli T, Rogina B. Rpd3 interacts with insulin signaling in Drosophila longevity extension. Aging (Albany NY). 2016; 8:3028–44. https://doi.org/10.18632/aging.101110 [PubMed]
- 9. Lin YJ, Seroude L, Benzer S. Extended life-span and stress resistance in the Drosophila mutant methuselah. Science. 1998; 282:943–46. https://doi.org/10.1126/science.282.5390.943 [PubMed]
- 10. Clancy DJ, Gems D, Harshman LG, Oldham S, Stocker H, Hafen E, Leevers SJ, Partridge L. Extension of life-span by loss of CHICO, a Drosophila insulin receptor substrate protein. Science. 2001; 292:104–06. https://doi.org/10.1126/science.1057991 [PubMed]
- 11. Vermeulen CJ, Loeschcke V. Longevity and the stress response in Drosophila. Exp Gerontol. 2007; 42:153–59. https://doi.org/10.1016/j.exger.2006.09.014 [PubMed]
- 12. Lin YR, Parikh H, Park Y. Loco signaling pathway in longevity. Small GTPases. 2011; 2:158–61. https://doi.org/10.4161/sgtp.2.3.16390 [PubMed]
- 13. Eom GH, Kook H. Posttranslational modifications of histone deacetylases: implications for cardiovascular diseases. Pharmacol Ther. 2014; 143:168–80. https://doi.org/10.1016/j.pharmthera.2014.02.012 [PubMed]
- 14. Millard CJ, Watson PJ, Fairall L, Schwabe JW. Targeting Class I Histone Deacetylases in a “Complex” Environment. Trends Pharmacol Sci. 2017; 38:363–77. https://doi.org/10.1016/j.tips.2016.12.006 [PubMed]
- 15. Hunt TW, Fields TA, Casey PJ, Peralta EG. RGS10 is a selective activator of G α i GTPase activity. Nature. 1996; 383:175–77. https://doi.org/10.1038/383175a0 [PubMed]
- 16. Neubig RR, Siderovski DP. Regulators of G-protein signalling as new central nervous system drug targets. Nat Rev Drug Discov. 2002; 1:187–97. https://doi.org/10.1038/nrd747 [PubMed]
- 17. Yu F, Wang H, Qian H, Kaushik R, Bownes M, Yang X, Chia W. Locomotion defects, together with Pins, regulates heterotrimeric G-protein signaling during Drosophila neuroblast asymmetric divisions. Genes Dev. 2005; 19:1341–53. https://doi.org/10.1101/gad.1295505 [PubMed]
- 18. Schwabe T, Bainton RJ, Fetter RD, Heberlein U, Gaul U. GPCR signaling is required for blood-brain barrier formation in drosophila. Cell. 2005; 123:133–44. https://doi.org/10.1016/j.cell.2005.08.037 [PubMed]
- 19. Lin YR, Kim K, Yang Y, Ivessa A, Sadoshima J, Park Y. Regulation of longevity by regulator of G-protein signaling protein, Loco. Aging Cell. 2011; 10:438–47. https://doi.org/10.1111/j.1474-9726.2011.00678.x [PubMed]
- 20. Brand AH, Perrimon N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development. 1993; 118:401–15. [PubMed]
- 21. Hamada FN, Park PJ, Gordadze PR, Kuroda MI. Global regulation of X chromosomal genes by the MSL complex in Drosophila melanogaster. Genes Dev. 2005; 19:2289–94. https://doi.org/10.1101/gad.1343705 [PubMed]
- 22. Landis GN, Abdueva D, Skvortsov D, Yang J, Rabin BE, Carrick J, Tavaré S, Tower J. Similar gene expression patterns characterize aging and oxidative stress in Drosophila melanogaster. Proc Natl Acad Sci USA. 2004; 101:7663–68. https://doi.org/10.1073/pnas.0307605101 [PubMed]
- 23. Chamilos G, Lewis RE, Hu J, Xiao L, Zal T, Gilliet M, Halder G, Kontoyiannis DP. Drosophila melanogaster as a model host to dissect the immunopathogenesis of zygomycosis. Proc Natl Acad Sci USA. 2008; 105:9367–72. https://doi.org/10.1073/pnas.0709578105 [PubMed]
- 24. Nemčovičová I, Šesták S, Rendić D, Plšková M, Mucha J, Wilson IB. Characterisation of class I and II α-mannosidases from Drosophila melanogaster. Glycoconj J. 2013; 30:899–909. https://doi.org/10.1007/s10719-013-9495-5 [PubMed]
- 25. Benes H, Spivey DW, Miles J, Neal K, Edmondson RG. Fat-body-specific expression of the Drosophila Lsp-2 gene. SAAS Bull Biochem Biotechnol. 1990; 3:129–33. [PubMed]
- 26. Yang J, O’Tousa JE. Cellular sites of Drosophila NinaB and NinaD activity in vitamin A metabolism. Mol Cell Neurosci. 2007; 35:49–56. https://doi.org/10.1016/j.mcn.2007.02.001 [PubMed]
- 27. Hoffmann JA, Reichhart JM. Drosophila innate immunity: an evolutionary perspective. Nat Immunol. 2002; 3:121–26. https://doi.org/10.1038/ni0202-121 [PubMed]
- 28. Kim T, Kim YJ. Overview of innate immunity in Drosophila. J Biochem Mol Biol. 2005; 38:121–27. [PubMed]
- 29. Aggarwal K, Silverman N. Positive and negative regulation of the Drosophila immune response. BMB Rep. 2008; 41:267–77. https://doi.org/10.5483/BMBRep.2008.41.4.267 [PubMed]
- 30. Lin YR, Parikh H, Park Y. Stress resistance and lifespan enhanced by downregulation of antimicrobial peptide genes in the Imd pathway. Aging (Albany NY). 2018; 10:622–31. https://doi.org/10.18632/aging.101417 [PubMed]
- 31. McClure CD, Zhong W, Hunt VL, Chapman FM, Hill FV, Priest NK. Hormesis results in trade-offs with immunity. Evolution. 2014; 68:2225–33. https://doi.org/10.1111/evo.12453 [PubMed]
- 32. Qi YX, Xu G, Gu GX, Mao F, Ye GY, Liu W, Huang J. A new Drosophila octopamine receptor responds to serotonin. Insect Biochem Mol Biol. 2017; 90:61–70. https://doi.org/10.1016/j.ibmb.2017.09.010 [PubMed]
- 33. Yan L, Vatner DE, O’Connor JP, Ivessa A, Ge H, Chen W, Hirotani S, Ishikawa Y, Sadoshima J, Vatner SF. Type 5 adenylyl cyclase disruption increases longevity and protects against stress. Cell. 2007; 130:247–58. https://doi.org/10.1016/j.cell.2007.05.038 [PubMed]
- 34. Neely GG, Kuba K, Cammarato A, Isobe K, Amann S, Zhang L, Murata M, Elmén L, Gupta V, Arora S, Sarangi R, Dan D, Fujisawa S, et al. A global in vivo Drosophila RNAi screen identifies NOT3 as a conserved regulator of heart function. Cell. 2010; 141:142–53. https://doi.org/10.1016/j.cell.2010.02.023 [PubMed]
- 35. Neely GG, Hess A, Costigan M, Keene AC, Goulas S, Langeslag M, Griffin RS, Belfer I, Dai F, Smith SB, Diatchenko L, Gupta V, Xia CP, et al. A genome-wide Drosophila screen for heat nociception identifies α2δ3 as an evolutionarily conserved pain gene. Cell. 2010; 143:628–38. https://doi.org/10.1016/j.cell.2010.09.047 [PubMed]
- 36. Park Y, Kelley RL, Oh H, Kuroda MI, Meller VH. Extent of chromatin spreading determined by roX RNA recruitment of MSL proteins. Science. 2002; 298:1620–23. https://doi.org/10.1126/science.1076686 [PubMed]
- 37. Willard FS, Willard MD, Kimple AJ, Soundararajan M, Oestreich EA, Li X, Sowa NA, Kimple RJ, Doyle DA, Der CJ, Zylka MJ, Snider WD, Siderovski DP. Regulator of G-protein signaling 14 (RGS14) is a selective H-Ras effector. PLoS One. 2009; 4:e4884. https://doi.org/10.1371/journal.pone.0004884 [PubMed]
- 38. Shu FJ, Ramineni S, Hepler JR. RGS14 is a multifunctional scaffold that integrates G protein and Ras/Raf MAPkinase signalling pathways. Cell Signal. 2010; 22:366–76. https://doi.org/10.1016/j.cellsig.2009.10.005 [PubMed]
- 39. Fontana L, Partridge L, Longo VD. Extending healthy life span--from yeast to humans. Science. 2010; 328:321–26. https://doi.org/10.1126/science.1172539 [PubMed]
- 40. Quevedo C, Salinas M, Alcázar A. Initiation factor 2B activity is regulated by protein phosphatase 1, which is activated by the mitogen-activated protein kinase-dependent pathway in insulin-like growth factor 1-stimulated neuronal cells. J Biol Chem. 2003; 278:16579–86. https://doi.org/10.1074/jbc.M212936200 [PubMed]
- 41. Canettieri G, Morantte I, Guzmán E, Asahara H, Herzig S, Anderson SD, Yates JR
3rd , Montminy M. Attenuation of a phosphorylation-dependent activator by an HDAC-PP1 complex. Nat Struct Biol. 2003; 10:175–81. https://doi.org/10.1038/nsb895 [PubMed] - 42. Galasinski SC, Resing KA, Goodrich JA, Ahn NG. Phosphatase inhibition leads to histone deacetylases 1 and 2 phosphorylation and disruption of corepressor interactions. J Biol Chem. 2002; 277:19618–26. https://doi.org/10.1074/jbc.M201174200 [PubMed]
- 43. Pflum MK, Tong JK, Lane WS, Schreiber SL. Histone deacetylase 1 phosphorylation promotes enzymatic activity and complex formation. J Biol Chem. 2001; 276:47733–41. https://doi.org/10.1074/jbc.M105590200 [PubMed]
- 44. Pluemsampant S, Safronova OS, Nakahama K, Morita I. Protein kinase CK2 is a key activator of histone deacetylase in hypoxia-associated tumors. Int J Cancer. 2008; 122:333–41. https://doi.org/10.1002/ijc.23094 [PubMed]
- 45. Frankel S, Woods J, Ziafazeli T, Rogina B. RPD3 histone deacetylase and nutrition have distinct but interacting effects on Drosophila longevity. Aging (Albany NY). 2015; 7:1112–29. https://doi.org/10.18632/aging.100856 [PubMed]
- 46. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006; 444:860–67. https://doi.org/10.1038/nature05485 [PubMed]
- 47. Hull-Thompson J, Muffat J, Sanchez D, Walker DW, Benzer S, Ganfornina MD, Jasper H. Control of metabolic homeostasis by stress signaling is mediated by the lipocalin NLaz. PLoS Genet. 2009; 5:e1000460. https://doi.org/10.1371/journal.pgen.1000460 [PubMed]
- 48. Fernández-Ayala DJ, Chen S, Kemppainen E, O’Dell KM, Jacobs HT. Gene expression in a Drosophila model of mitochondrial disease. PLoS One. 2010; 5:e8549. https://doi.org/10.1371/journal.pone.0008549 [PubMed]
- 49. Kelley RL, Lee OK, Shim YK. Transcription rate of noncoding roX1 RNA controls local spreading of the Drosophila MSL chromatin remodeling complex. Mech Dev. 2008; 125:1009–19. https://doi.org/10.1016/j.mod.2008.08.003 [PubMed]
- 50. Kim K, Lin YR, Park Y. Enhancement of stress resistances and downregulation of Imd pathway by lower developmental temperature in Drosophila melanogaster. Exp Gerontol. 2010; 45:984–87. https://doi.org/10.1016/j.exger.2010.08.033 [PubMed]
- 51. Park SW, Oh H, Lin YR, Park Y. MSL cis-spreading from roX gene up-regulates the neighboring genes. Biochem Biophys Res Commun. 2010; 399:227–31. https://doi.org/10.1016/j.bbrc.2010.07.059 [PubMed]